f427a6d79c4c45ca9758dd043035517d.ppt
- Количество слайдов: 24
 Overview of the ETCTN May 30, 2015 Percy Ivy, MD ETCTN and ETCTN-Phase 1 Program Director Associate Chief, Investigational Drug Branch Cancer Therapy Evaluation Program
Overview of the ETCTN May 30, 2015 Percy Ivy, MD ETCTN and ETCTN-Phase 1 Program Director Associate Chief, Investigational Drug Branch Cancer Therapy Evaluation Program
 Goals and Objectives of the Experimental Therapeutics Clinical Trials Network (ETCTN) Research and Development for New Treatments • • Dose and schedule in early treatment trials Novel combination therapies Tumor Characterization in Biomarker-driven studies • • • Molecular characterization: expression, sequence and epigenetics Validated biomarker assays in qualified labs Functional imaging Enhanced understanding of cancer biology • Bedside to bench and back Education and Training for young investigators
Goals and Objectives of the Experimental Therapeutics Clinical Trials Network (ETCTN) Research and Development for New Treatments • • Dose and schedule in early treatment trials Novel combination therapies Tumor Characterization in Biomarker-driven studies • • • Molecular characterization: expression, sequence and epigenetics Validated biomarker assays in qualified labs Functional imaging Enhanced understanding of cancer biology • Bedside to bench and back Education and Training for young investigators
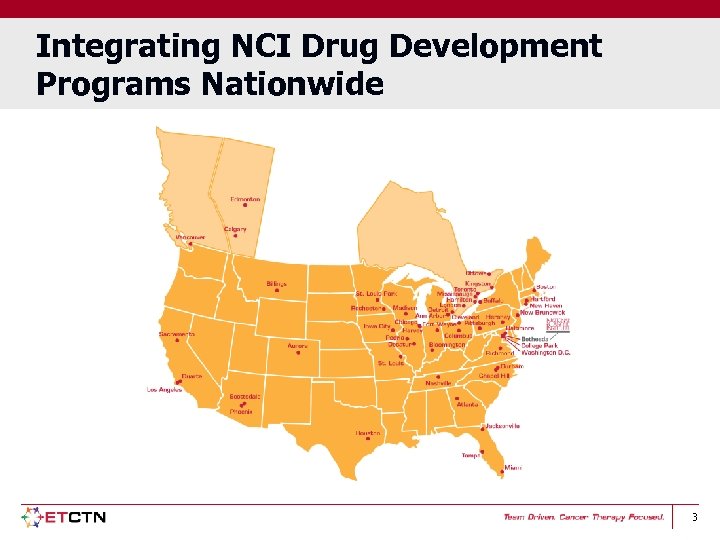 Integrating NCI Drug Development Programs Nationwide 3
Integrating NCI Drug Development Programs Nationwide 3
 ETCTN Phase 1 Principal Investigators Institutions PIs City of Hope/Beckman Res. Inst. Newman, Edward Lenz, Heinz-Joseph Ohio State U. Grever, Michael Dana-Farber/Harvard Cancer Center Kufe, Donald Flaherty, Keith Shapiro, Geoffrey Duke U. North Carolina Wash. U. Hurwitz, Herbert Dees, Elizabeth Lockhart, Albert Johns Hopkins Carducci, Michael Gocke, Christopher Gojo, Ivana Rudek, Michelle Mayo - Rochester NCI-DTC Erlichman, Charles Huluska, Paul Sausville, Ed Kummar, Shivaani Rutgers-Cancer Inst. NJ Di. Paola, Robert U. Wisconsin Liu, Glenn U. Chicago Ratain, Mark Maitland, Michael U. Health Network Siu, Lillian Sullivan, Dan U. Pittsburgh Chu, Edward Beumer, Jan U. Texas – MDACC U. Colorado – Denver Yao, James Eckhardt, Gail Meric-Bernstam, Funda Yale University Lorusso, Patricia Eder, Paul Berlin, Jordan 4
ETCTN Phase 1 Principal Investigators Institutions PIs City of Hope/Beckman Res. Inst. Newman, Edward Lenz, Heinz-Joseph Ohio State U. Grever, Michael Dana-Farber/Harvard Cancer Center Kufe, Donald Flaherty, Keith Shapiro, Geoffrey Duke U. North Carolina Wash. U. Hurwitz, Herbert Dees, Elizabeth Lockhart, Albert Johns Hopkins Carducci, Michael Gocke, Christopher Gojo, Ivana Rudek, Michelle Mayo - Rochester NCI-DTC Erlichman, Charles Huluska, Paul Sausville, Ed Kummar, Shivaani Rutgers-Cancer Inst. NJ Di. Paola, Robert U. Wisconsin Liu, Glenn U. Chicago Ratain, Mark Maitland, Michael U. Health Network Siu, Lillian Sullivan, Dan U. Pittsburgh Chu, Edward Beumer, Jan U. Texas – MDACC U. Colorado – Denver Yao, James Eckhardt, Gail Meric-Bernstam, Funda Yale University Lorusso, Patricia Eder, Paul Berlin, Jordan 4
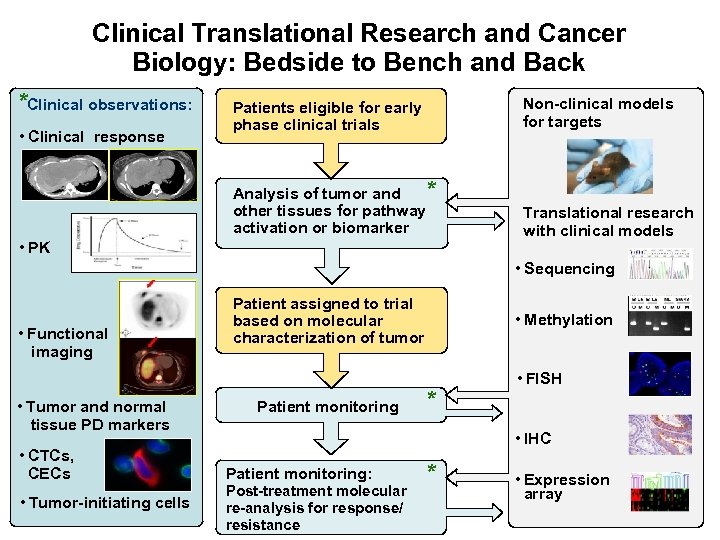 Clinical Translational Research and Cancer Biology: Bedside to Bench and Back *Clinical observations: Non-clinical models for targets * Analysis of tumor and other tissues for pathway activation or biomarker • Clinical response Patients eligible for early phase clinical trials Translational research with clinical models • PK • Sequencing • Functional imaging • Tumor and normal tissue PD markers • CTCs, CECs • Tumor-initiating cells Patient assigned to trial based on molecular characterization of tumor Patient monitoring • Methylation * • FISH • IHC Patient monitoring: Post-treatment molecular re-analysis for response/ resistance * • Expression array
Clinical Translational Research and Cancer Biology: Bedside to Bench and Back *Clinical observations: Non-clinical models for targets * Analysis of tumor and other tissues for pathway activation or biomarker • Clinical response Patients eligible for early phase clinical trials Translational research with clinical models • PK • Sequencing • Functional imaging • Tumor and normal tissue PD markers • CTCs, CECs • Tumor-initiating cells Patient assigned to trial based on molecular characterization of tumor Patient monitoring • Methylation * • FISH • IHC Patient monitoring: Post-treatment molecular re-analysis for response/ resistance * • Expression array
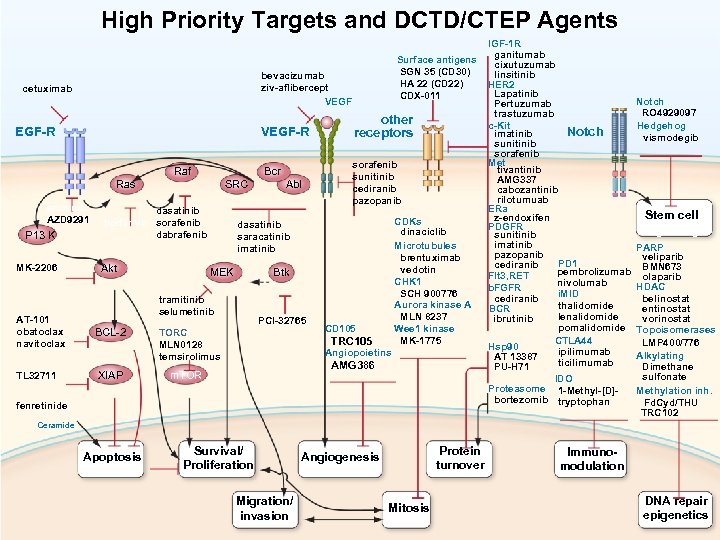 High Priority Targets and DCTD/CTEP Agents bevacizumab ziv-aflibercept VEGF cetuximab EGF-R VEGF-R Ras erlotinib AZD 9291 P 13 K MK-2206 Surface antigens SGN 35 (CD 30) HA 22 (CD 22) CDX-011 Raf SRC dasatinib tipifarnib sorafenib dabrafenib Akt BCL-2 TL 32711 XIAP Abl sorafenib sunitinib cediranib pazopanib CDKs dinaciclib Microtubules brentuximab vedotin CHK 1 SCH 900776 Aurora kinase A MLN 8237 Wee 1 kinase MK-1775 dasatinib saracatinib imatinib MEK Btk tramitinib selumetinib AT-101 obatoclax navitoclax Bcr other receptors PCI-32765 TORC MLN 0128 temsirolimus CD 105 TRC 105 Angiopoietins AMG 386 m. TOR IGF-1 R ganitumab cixutuzumab linsitinib HER 2 Lapatinib Pertuzumab trastuzumab c-Kit Notch imatinib sunitinib sorafenib Met tivantinib AMG 337 cabozantinib rilotumuab ERa z-endoxifen PDGFR sunitinib imatinib pazopanib PD 1 cediranib pembrolizumab Flt 3, RET nivolumab b. FGFR i. MID cediranib thalidomide BCR lenalidomide ibrutinib pomalidomide CTLA 44 Hsp 90 ipilimumab AT 13387 ticilimumab PU-H 71 IDO Proteasome 1 -Methyl-[D]bortezomib tryptophan fenretinide Notch RO 4929097 Hedgehog vismodegib Stem cell signaling PARP veliparib BMN 673 olaparib HDAC belinostat entinostat vorinostat Topoisomerases LMP 400/776 Alkylating Dimethane sulfonate Methylation inh. Fd. Cyd/THU TRC 102 Ceramide Apoptosis Survival/ Proliferation Migration/ invasion Protein turnover Angiogenesis Mitosis Immunomodulation DNA repair epigenetics 6
High Priority Targets and DCTD/CTEP Agents bevacizumab ziv-aflibercept VEGF cetuximab EGF-R VEGF-R Ras erlotinib AZD 9291 P 13 K MK-2206 Surface antigens SGN 35 (CD 30) HA 22 (CD 22) CDX-011 Raf SRC dasatinib tipifarnib sorafenib dabrafenib Akt BCL-2 TL 32711 XIAP Abl sorafenib sunitinib cediranib pazopanib CDKs dinaciclib Microtubules brentuximab vedotin CHK 1 SCH 900776 Aurora kinase A MLN 8237 Wee 1 kinase MK-1775 dasatinib saracatinib imatinib MEK Btk tramitinib selumetinib AT-101 obatoclax navitoclax Bcr other receptors PCI-32765 TORC MLN 0128 temsirolimus CD 105 TRC 105 Angiopoietins AMG 386 m. TOR IGF-1 R ganitumab cixutuzumab linsitinib HER 2 Lapatinib Pertuzumab trastuzumab c-Kit Notch imatinib sunitinib sorafenib Met tivantinib AMG 337 cabozantinib rilotumuab ERa z-endoxifen PDGFR sunitinib imatinib pazopanib PD 1 cediranib pembrolizumab Flt 3, RET nivolumab b. FGFR i. MID cediranib thalidomide BCR lenalidomide ibrutinib pomalidomide CTLA 44 Hsp 90 ipilimumab AT 13387 ticilimumab PU-H 71 IDO Proteasome 1 -Methyl-[D]bortezomib tryptophan fenretinide Notch RO 4929097 Hedgehog vismodegib Stem cell signaling PARP veliparib BMN 673 olaparib HDAC belinostat entinostat vorinostat Topoisomerases LMP 400/776 Alkylating Dimethane sulfonate Methylation inh. Fd. Cyd/THU TRC 102 Ceramide Apoptosis Survival/ Proliferation Migration/ invasion Protein turnover Angiogenesis Mitosis Immunomodulation DNA repair epigenetics 6
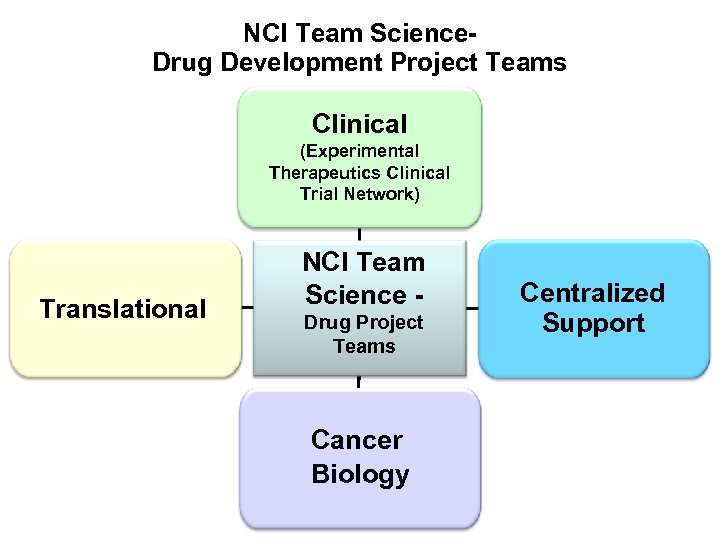 NCI Team Science. Drug Development Project Teams Clinical (Experimental Therapeutics Clinical Trial Network) Translational NCI Team Science Drug Project Teams Cancer Biology Centralized Support
NCI Team Science. Drug Development Project Teams Clinical (Experimental Therapeutics Clinical Trial Network) Translational NCI Team Science Drug Project Teams Cancer Biology Centralized Support
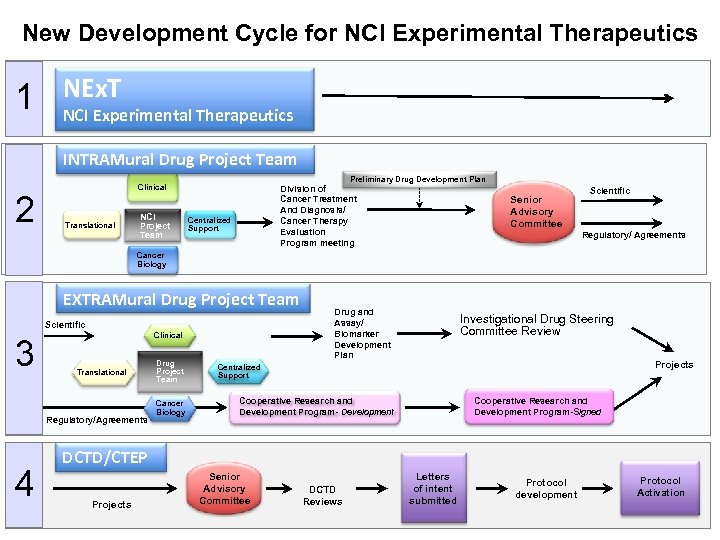 New Development Cycle for NCI Experimental Therapeutics 1 NEx. T NCI Experimental Therapeutics INTRAMural Drug Project Team 2 Preliminary Drug Development Plan Clinical Translational NCI Project Team Division of Cancer Treatment And Diagnosis/ Cancer Therapy Evaluation Program meeting Centralized Support Senior Advisory Committee Scientific Regulatory/ Agreements Cancer Biology EXTRAMural Drug Project Team Scientific 3 Clinical Translational Regulatory/Agreements 4 Drug Project Team Cancer Biology Drug and Assay/ Biomarker Development Plan Investigational Drug Steering Committee Review Projects Centralized Support Cooperative Research and Development Program- Development Cooperative Research and Development Program-Signed DCTD/CTEP Projects Senior Advisory Committee DCTD Reviews Letters of intent submitted Protocol development Protocol Activation
New Development Cycle for NCI Experimental Therapeutics 1 NEx. T NCI Experimental Therapeutics INTRAMural Drug Project Team 2 Preliminary Drug Development Plan Clinical Translational NCI Project Team Division of Cancer Treatment And Diagnosis/ Cancer Therapy Evaluation Program meeting Centralized Support Senior Advisory Committee Scientific Regulatory/ Agreements Cancer Biology EXTRAMural Drug Project Team Scientific 3 Clinical Translational Regulatory/Agreements 4 Drug Project Team Cancer Biology Drug and Assay/ Biomarker Development Plan Investigational Drug Steering Committee Review Projects Centralized Support Cooperative Research and Development Program- Development Cooperative Research and Development Program-Signed DCTD/CTEP Projects Senior Advisory Committee DCTD Reviews Letters of intent submitted Protocol development Protocol Activation
 Challenges for the Early Phase Therapeutics Network Accrual • • Smaller patient populations due to molecularly-defined diseases A scalable/flexible program that can rapidly adapt to accrual needs Biomarkers • • • Often requires biopsies Fit for purpose, validated assays Functional imaging More Facile Mechanisms for Translation • • To and From Bench to Bedside Collaborations More predictive animal models to evaluate tumor heterogeneity
Challenges for the Early Phase Therapeutics Network Accrual • • Smaller patient populations due to molecularly-defined diseases A scalable/flexible program that can rapidly adapt to accrual needs Biomarkers • • • Often requires biopsies Fit for purpose, validated assays Functional imaging More Facile Mechanisms for Translation • • To and From Bench to Bedside Collaborations More predictive animal models to evaluate tumor heterogeneity
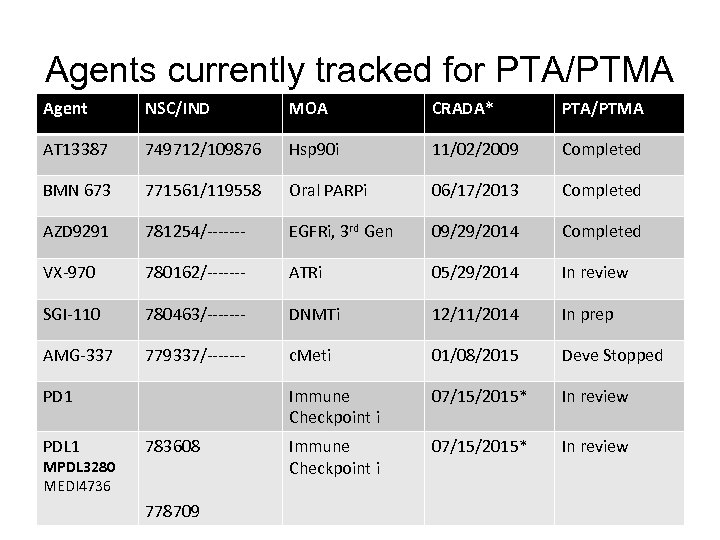 Agents currently tracked for PTA/PTMA Agent NSC/IND MOA CRADA* PTA/PTMA AT 13387 749712/109876 Hsp 90 i 11/02/2009 Completed BMN 673 771561/119558 Oral PARPi 06/17/2013 Completed AZD 9291 781254/------- EGFRi, 3 rd Gen 09/29/2014 Completed VX-970 780162/------- ATRi 05/29/2014 In review SGI-110 780463/------- DNMTi 12/11/2014 In prep AMG-337 779337/------- c. Meti 01/08/2015 Deve Stopped Immune Checkpoint i 07/15/2015* In review PD 1 PDL 1 MPDL 3280 MEDI 4736 783608 778709
Agents currently tracked for PTA/PTMA Agent NSC/IND MOA CRADA* PTA/PTMA AT 13387 749712/109876 Hsp 90 i 11/02/2009 Completed BMN 673 771561/119558 Oral PARPi 06/17/2013 Completed AZD 9291 781254/------- EGFRi, 3 rd Gen 09/29/2014 Completed VX-970 780162/------- ATRi 05/29/2014 In review SGI-110 780463/------- DNMTi 12/11/2014 In prep AMG-337 779337/------- c. Meti 01/08/2015 Deve Stopped Immune Checkpoint i 07/15/2015* In review PD 1 PDL 1 MPDL 3280 MEDI 4736 783608 778709
 NCI-Sponsored Infrastructure
NCI-Sponsored Infrastructure
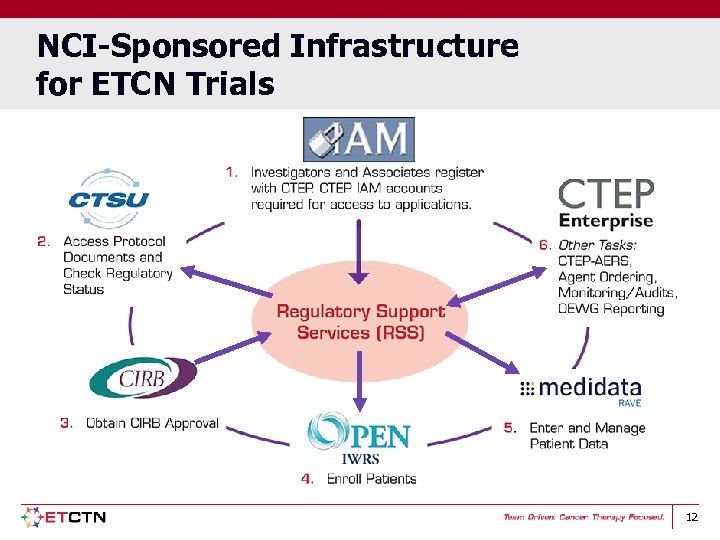 NCI-Sponsored Infrastructure for ETCN Trials 12
NCI-Sponsored Infrastructure for ETCN Trials 12
![Medidata Rave [Medidata Rave-focused webinar scheduled for April 28, 2014] Medidata Rave [Medidata Rave-focused webinar scheduled for April 28, 2014]](https://present5.com/presentation/f427a6d79c4c45ca9758dd043035517d/image-13.jpg) Medidata Rave [Medidata Rave-focused webinar scheduled for April 28, 2014]
Medidata Rave [Medidata Rave-focused webinar scheduled for April 28, 2014]
 Medidata Rave § Web based EDC (electronic data collection) to be used in all future NCI studies § CTEP-IAM account (user name and password) required to access Rave; must also have one of the Rave-specific roles. § Users must also be on the ETCTN site roster of a participating organization and have approval for a given trial at their site to manage data in Rave. § All ETCTN studies will use the same standard Theradex e. CRFs resulting in expedited study set-up following approval § Built-in, real-time edit checks will minimize queries and after-thefact data cleaning § Queries entered by CTMS monitors and auditors can be resolved within Rave by the site staff 14
Medidata Rave § Web based EDC (electronic data collection) to be used in all future NCI studies § CTEP-IAM account (user name and password) required to access Rave; must also have one of the Rave-specific roles. § Users must also be on the ETCTN site roster of a participating organization and have approval for a given trial at their site to manage data in Rave. § All ETCTN studies will use the same standard Theradex e. CRFs resulting in expedited study set-up following approval § Built-in, real-time edit checks will minimize queries and after-thefact data cleaning § Queries entered by CTMS monitors and auditors can be resolved within Rave by the site staff 14
![Web Reporting [Will be covered in a future focused webinar; TBD] Web Reporting [Will be covered in a future focused webinar; TBD]](https://present5.com/presentation/f427a6d79c4c45ca9758dd043035517d/image-15.jpg) Web Reporting [Will be covered in a future focused webinar; TBD]
Web Reporting [Will be covered in a future focused webinar; TBD]
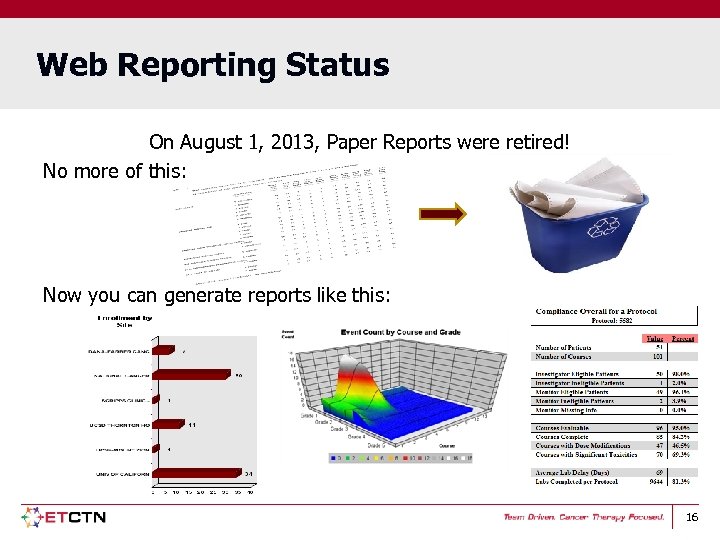 Web Reporting Status On August 1, 2013, Paper Reports were retired! No more of this: Now you can generate reports like this: 16
Web Reporting Status On August 1, 2013, Paper Reports were retired! No more of this: Now you can generate reports like this: 16
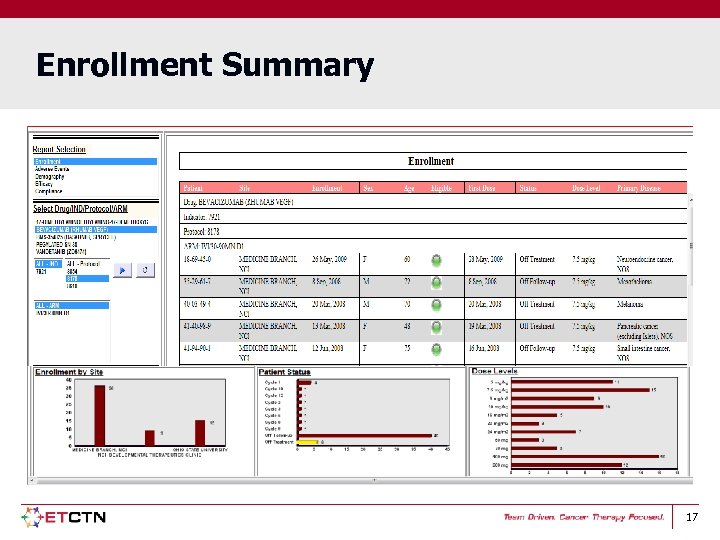 Enrollment Summary 17
Enrollment Summary 17
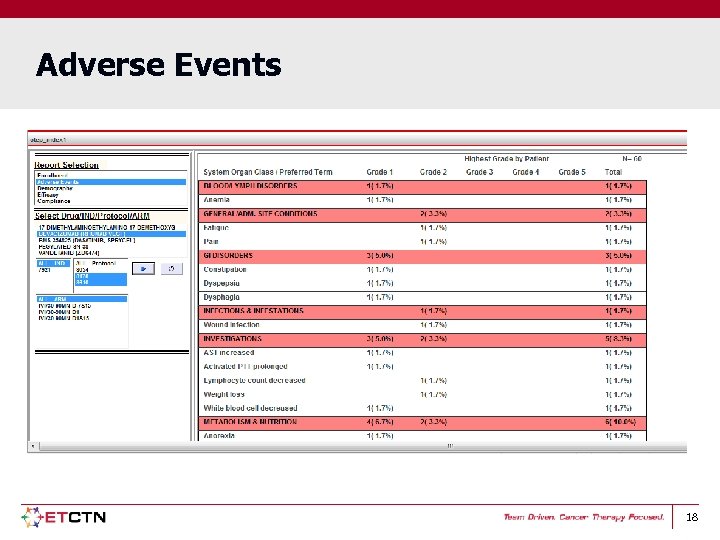 Adverse Events 18
Adverse Events 18
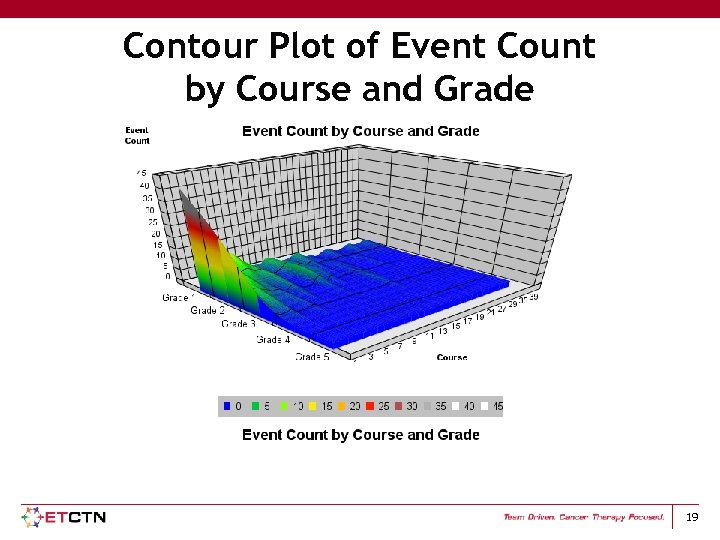 Contour Plot of Event Count by Course and Grade 19
Contour Plot of Event Count by Course and Grade 19
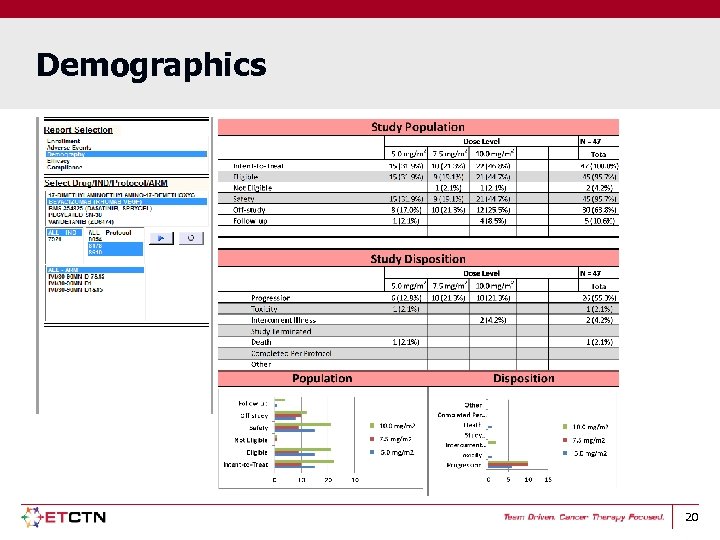 Demographics 20
Demographics 20
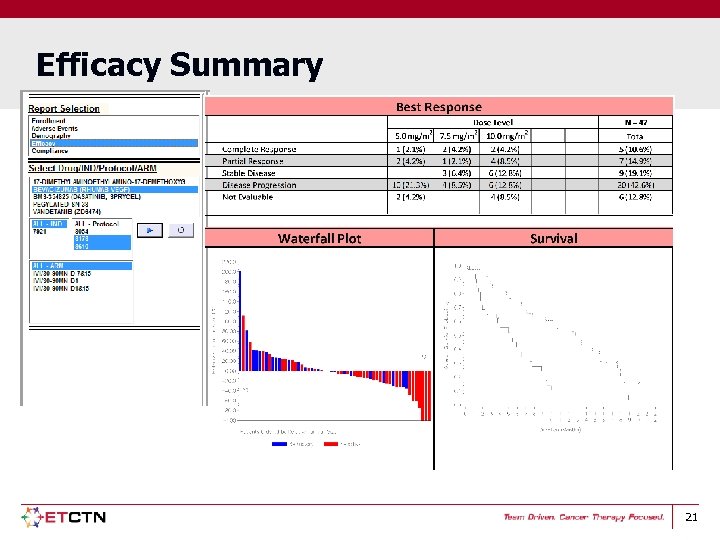 Efficacy Summary 21
Efficacy Summary 21
 National Cancer Institute ETCTN Education and Training U. S. DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health Since program launch, we have held a number of educational webinars for ETCTN members: § For Leadership: § For Site Staff: § Kick-off and Overview § Introduction to the ETCTN, § Rosters and Roles Centralized Services, and § Patient Enrollment the CTSU Website § NCI CIRB § Patient Enrollment § PIO Updates § Regulatory Processes § Data Management § Biomarkers § Implementing Drug Project Teams § Web Reporting
National Cancer Institute ETCTN Education and Training U. S. DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health Since program launch, we have held a number of educational webinars for ETCTN members: § For Leadership: § For Site Staff: § Kick-off and Overview § Introduction to the ETCTN, § Rosters and Roles Centralized Services, and § Patient Enrollment the CTSU Website § NCI CIRB § Patient Enrollment § PIO Updates § Regulatory Processes § Data Management § Biomarkers § Implementing Drug Project Teams § Web Reporting
 National Cancer Institute Educational Materials U. S. DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health • Educational Materials on the ETCTN-CTSU website includes: links to the webinar recordings, checklists, and information sheets on 14 different topics : § § § § Protocol Development Protocol Amendments Person Registration & CTEP-IAM Rosters & Roles The CTSU Protocol Access & Communications Regulatory Processing § § § § The NCI CIRB Patient Enrollment Agent Ordering Data Management SAE Reporting CDUS Reporting Auditing and Monitoring • All documents will be posted to the ETCTN pages on the CTEP website once development is complete
National Cancer Institute Educational Materials U. S. DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health • Educational Materials on the ETCTN-CTSU website includes: links to the webinar recordings, checklists, and information sheets on 14 different topics : § § § § Protocol Development Protocol Amendments Person Registration & CTEP-IAM Rosters & Roles The CTSU Protocol Access & Communications Regulatory Processing § § § § The NCI CIRB Patient Enrollment Agent Ordering Data Management SAE Reporting CDUS Reporting Auditing and Monitoring • All documents will be posted to the ETCTN pages on the CTEP website once development is complete
 National Cancer Institute U. S. DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health
National Cancer Institute U. S. DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health


