bc9e5ae24754ed9283be143bf33a8eb5.ppt
- Количество слайдов: 38
 Overview of Medical Devices Regulations Velocity Science Sarah Chandler A/Manager, Device Licensing Services Division Medical Devices Bureau Health Canada sarah. chandler@canada. ca
Overview of Medical Devices Regulations Velocity Science Sarah Chandler A/Manager, Device Licensing Services Division Medical Devices Bureau Health Canada sarah. chandler@canada. ca
 Medical Device Stakeholders 2
Medical Device Stakeholders 2
 Brief History of the Canadian Medical Device Regulatory Framework 1975 Medical Devices Regulations (MDR) come into force on September 2. Medical Devices Review Committee (MDRC) created to assess regulation. 2015 1997 1991 Greater than 35000 Medical Device Licences renewed. The Medical Devices Bureau is unified under one Directorate. 1977 1992 MDR amended to include premarket review for some devices. MDRC publish their report which includes several recommendations. 1998 The new MDR comes into effect, making licensing a requirement. 3
Brief History of the Canadian Medical Device Regulatory Framework 1975 Medical Devices Regulations (MDR) come into force on September 2. Medical Devices Review Committee (MDRC) created to assess regulation. 2015 1997 1991 Greater than 35000 Medical Device Licences renewed. The Medical Devices Bureau is unified under one Directorate. 1977 1992 MDR amended to include premarket review for some devices. MDRC publish their report which includes several recommendations. 1998 The new MDR comes into effect, making licensing a requirement. 3
 Regulatory Framework Canada has incorporated successful elements from other regulatory systems into our own regulations: Ø Risk Based Classification System Ø Quality System Requirement Ø Pre-Market Licensing Ø Post-Market Regulation 4
Regulatory Framework Canada has incorporated successful elements from other regulatory systems into our own regulations: Ø Risk Based Classification System Ø Quality System Requirement Ø Pre-Market Licensing Ø Post-Market Regulation 4
 Regulatory Tools 5
Regulatory Tools 5
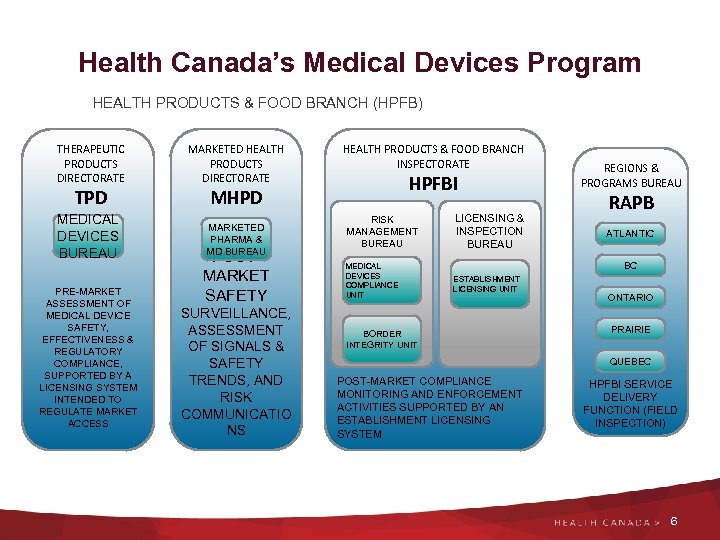 Health Canada’s Medical Devices Program HEALTH PRODUCTS & FOOD BRANCH (HPFB) THERAPEUTIC PRODUCTS DIRECTORATE TPD MEDICAL DEVICES BUREAU PRE-MARKET ASSESSMENT OF MEDICAL DEVICE SAFETY, EFFECTIVENESS & REGULATORY COMPLIANCE, SUPPORTED BY A LICENSING SYSTEM INTENDED TO REGULATE MARKET ACCESS MARKETED HEALTH PRODUCTS DIRECTORATE HEALTH PRODUCTS & FOOD BRANCH INSPECTORATE HPFBI MHPD MARKETED PHARMA & MD BUREAU POSTMARKET SAFETY SURVEILLANCE, ASSESSMENT OF SIGNALS & SAFETY TRENDS, AND RISK COMMUNICATIO NS RISK MANAGEMENT BUREAU LICENSING & INSPECTION BUREAU MEDICAL DEVICES COMPLIANCE UNIT ESTABLISHMENT LICENSING UNIT REGIONS & PROGRAMS BUREAU RAPB ATLANTIC BC BORDER INTEGRITY UNIT ONTARIO PRAIRIE QUEBEC POST-MARKET COMPLIANCE MONITORING AND ENFORCEMENT ACTIVITIES SUPPORTED BY AN ESTABLISHMENT LICENSING SYSTEM HPFBI SERVICE DELIVERY FUNCTION (FIELD INSPECTION) 6
Health Canada’s Medical Devices Program HEALTH PRODUCTS & FOOD BRANCH (HPFB) THERAPEUTIC PRODUCTS DIRECTORATE TPD MEDICAL DEVICES BUREAU PRE-MARKET ASSESSMENT OF MEDICAL DEVICE SAFETY, EFFECTIVENESS & REGULATORY COMPLIANCE, SUPPORTED BY A LICENSING SYSTEM INTENDED TO REGULATE MARKET ACCESS MARKETED HEALTH PRODUCTS DIRECTORATE HEALTH PRODUCTS & FOOD BRANCH INSPECTORATE HPFBI MHPD MARKETED PHARMA & MD BUREAU POSTMARKET SAFETY SURVEILLANCE, ASSESSMENT OF SIGNALS & SAFETY TRENDS, AND RISK COMMUNICATIO NS RISK MANAGEMENT BUREAU LICENSING & INSPECTION BUREAU MEDICAL DEVICES COMPLIANCE UNIT ESTABLISHMENT LICENSING UNIT REGIONS & PROGRAMS BUREAU RAPB ATLANTIC BC BORDER INTEGRITY UNIT ONTARIO PRAIRIE QUEBEC POST-MARKET COMPLIANCE MONITORING AND ENFORCEMENT ACTIVITIES SUPPORTED BY AN ESTABLISHMENT LICENSING SYSTEM HPFBI SERVICE DELIVERY FUNCTION (FIELD INSPECTION) 6
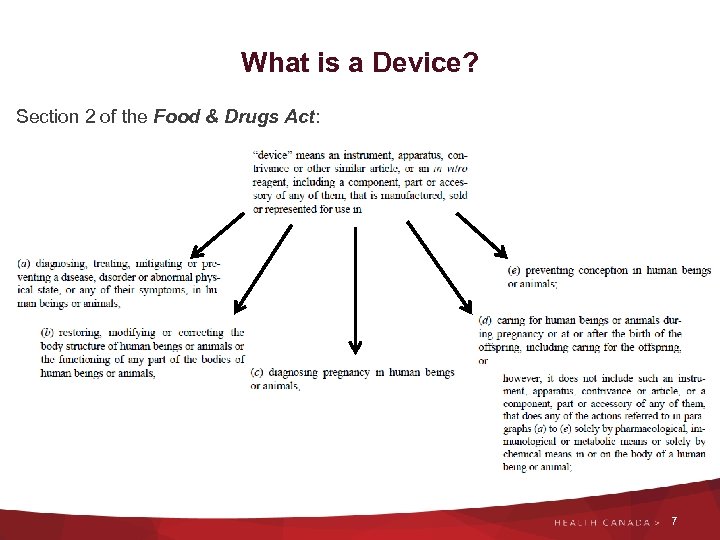 What is a Device? Section 2 of the Food & Drugs Act: 7
What is a Device? Section 2 of the Food & Drugs Act: 7
 What is a Medical Device? • Section 1 of the Medical Devices Regulations: 8
What is a Medical Device? • Section 1 of the Medical Devices Regulations: 8
 Regulatory Provisions • All devices offered for sale in Canada must comply with the Food and Drugs Act: – Cannot advertise or represent by label a treatment for a Schedule A disease or disorder (Section 3) – Cannot sell or advertise a device that may cause harm – Cannot sell or advertise a device in a misleading or deceptive way • All medical devices (those used on human beings) must also comply with the Medical Devices Regulations 9
Regulatory Provisions • All devices offered for sale in Canada must comply with the Food and Drugs Act: – Cannot advertise or represent by label a treatment for a Schedule A disease or disorder (Section 3) – Cannot sell or advertise a device that may cause harm – Cannot sell or advertise a device in a misleading or deceptive way • All medical devices (those used on human beings) must also comply with the Medical Devices Regulations 9
 Regulatory Provisions • The Regulations apply to: – The sale and advertising for sale of a medical device – The importation of a medical device for sale or for use on individuals, other than importation for personal use • Manufacturers of medical devices apply to Health Canada to receive either a Licence or an Authorization to sell their devices. 10
Regulatory Provisions • The Regulations apply to: – The sale and advertising for sale of a medical device – The importation of a medical device for sale or for use on individuals, other than importation for personal use • Manufacturers of medical devices apply to Health Canada to receive either a Licence or an Authorization to sell their devices. 10
 Regulatory Provisions A manufacturer: – Sells a medical device under their own name, trademark, design, trade name or other name owned or controlled by the person – Is responsible for designing, manufacturing, assembling, processing, labelling, packaging, refurbishing or modifying the device, assigning it a purpose – Performs or has someone perform on their behalf Manufacturer = Name on the label 11
Regulatory Provisions A manufacturer: – Sells a medical device under their own name, trademark, design, trade name or other name owned or controlled by the person – Is responsible for designing, manufacturing, assembling, processing, labelling, packaging, refurbishing or modifying the device, assigning it a purpose – Performs or has someone perform on their behalf Manufacturer = Name on the label 11
 Classification of Medical Devices • MDR sets out a system for classifying medical devices into one of four classes. Class I Low Risk Class III Moderate Risk Class IV High Risk Increasing Regulatory Oversight 12
Classification of Medical Devices • MDR sets out a system for classifying medical devices into one of four classes. Class I Low Risk Class III Moderate Risk Class IV High Risk Increasing Regulatory Oversight 12
 Risk-Based Regulatory System Class IV Premarket regulatory oversight based on licensing system. Market authorization is granted to manufacturers who demonstrate conformity to requirements set out in MDR. Post-market surveillance intended to enforce and promote compliance with MDR Premarket oversight with establishment licensing Post-market surveillance to promote compliance with MDR 4 3 - Certification that device is designed & manufactured according to ISO 13485 - Evidence of safety, effectiveness & labelling reviewed to validate compliance with MDR. More stringent review than Class III - Certification that device is designed & manufactured according to ISO 13485 - Evidence of safety, effectiveness & labelling reviewed to validate compliance with MDR. Class II 2 - Certification that device is manufactured according to international Quality Management System standard (ISO 13485). - Manufacturer’s attestation that device satisfies the safety, effectiveness requirements in MDR. Label reviewed. Class I 1 - Premarket regulatory oversight based on establishment licensing - Post-market oversight based on compliance to applicable sections of the MDR 13
Risk-Based Regulatory System Class IV Premarket regulatory oversight based on licensing system. Market authorization is granted to manufacturers who demonstrate conformity to requirements set out in MDR. Post-market surveillance intended to enforce and promote compliance with MDR Premarket oversight with establishment licensing Post-market surveillance to promote compliance with MDR 4 3 - Certification that device is designed & manufactured according to ISO 13485 - Evidence of safety, effectiveness & labelling reviewed to validate compliance with MDR. More stringent review than Class III - Certification that device is designed & manufactured according to ISO 13485 - Evidence of safety, effectiveness & labelling reviewed to validate compliance with MDR. Class II 2 - Certification that device is manufactured according to international Quality Management System standard (ISO 13485). - Manufacturer’s attestation that device satisfies the safety, effectiveness requirements in MDR. Label reviewed. Class I 1 - Premarket regulatory oversight based on establishment licensing - Post-market oversight based on compliance to applicable sections of the MDR 13
 Quality Management System • • ISO 13485: 2003 Quality Management System Certificate Recognized Registrars: BSI (US) Intertek (CN) LRQA (US) SGS (UK) DQS (GER) TUV-N/S (US) UL (USA) Dekra (Netherlands) G-MED (France) NSAI (Ireland) SAI (Australia) LGA (GER) TGA (Australia) TUV (US) 14
Quality Management System • • ISO 13485: 2003 Quality Management System Certificate Recognized Registrars: BSI (US) Intertek (CN) LRQA (US) SGS (UK) DQS (GER) TUV-N/S (US) UL (USA) Dekra (Netherlands) G-MED (France) NSAI (Ireland) SAI (Australia) LGA (GER) TGA (Australia) TUV (US) 14
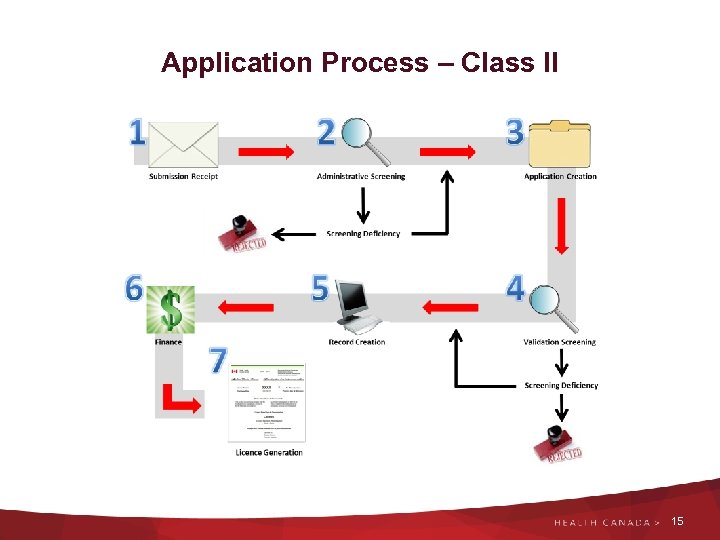 Application Process – Class II 15
Application Process – Class II 15
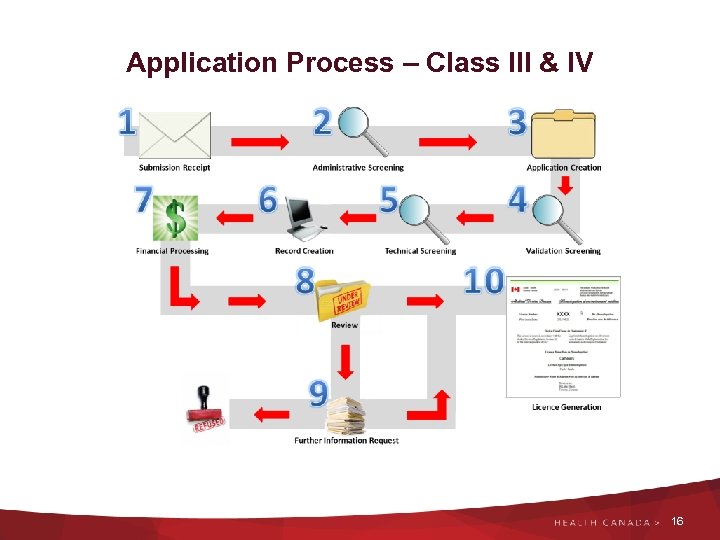 Application Process – Class III & IV 16
Application Process – Class III & IV 16
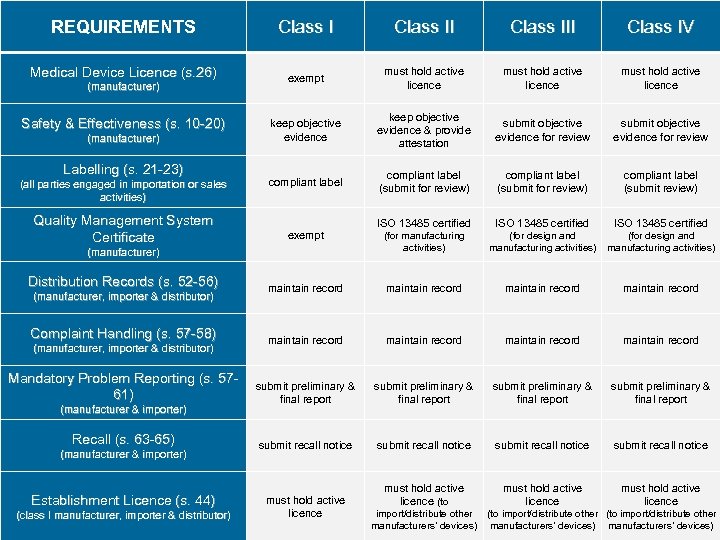 REQUIREMENTS Class III Class IV Medical Device Licence (s. 26) exempt must hold active licence keep objective evidence & provide attestation submit objective evidence for review compliant label (submit for review) compliant label (submit review) ISO 13485 certified (for manufacturing activities) (for design and manufacturing activities) maintain record maintain record submit preliminary & final report submit recall notice must hold active licence (to must hold active licence (manufacturer) Safety & Effectiveness (s. 10 -20) (manufacturer) Labelling (s. 21 -23) (all parties engaged in importation or sales activities) Quality Management System Certificate exempt (manufacturer) Distribution Records (s. 52 -56) (manufacturer, importer & distributor) Complaint Handling (s. 57 -58) (manufacturer, importer & distributor) Mandatory Problem Reporting (s. 5761) (manufacturer & importer) Recall (s. 63 -65) (manufacturer & importer) Establishment Licence (s. 44) (class I manufacturer, importer & distributor) must hold active licence import/distribute other manufacturers’ devices) (to import/distribute other manufacturers’ devices)
REQUIREMENTS Class III Class IV Medical Device Licence (s. 26) exempt must hold active licence keep objective evidence & provide attestation submit objective evidence for review compliant label (submit for review) compliant label (submit review) ISO 13485 certified (for manufacturing activities) (for design and manufacturing activities) maintain record maintain record submit preliminary & final report submit recall notice must hold active licence (to must hold active licence (manufacturer) Safety & Effectiveness (s. 10 -20) (manufacturer) Labelling (s. 21 -23) (all parties engaged in importation or sales activities) Quality Management System Certificate exempt (manufacturer) Distribution Records (s. 52 -56) (manufacturer, importer & distributor) Complaint Handling (s. 57 -58) (manufacturer, importer & distributor) Mandatory Problem Reporting (s. 5761) (manufacturer & importer) Recall (s. 63 -65) (manufacturer & importer) Establishment Licence (s. 44) (class I manufacturer, importer & distributor) must hold active licence import/distribute other manufacturers’ devices) (to import/distribute other manufacturers’ devices)
 Premarket Review Document Requirements 18
Premarket Review Document Requirements 18
 Classification of Medical Devices • To determine the classification of a device, you must apply all of the rules in Schedule 1 of the Medical Devices Regulations. • You must consider the labelled indications for use, or claims made for the device. 19
Classification of Medical Devices • To determine the classification of a device, you must apply all of the rules in Schedule 1 of the Medical Devices Regulations. • You must consider the labelled indications for use, or claims made for the device. 19
 Classification of Medical Devices • Non In vitro Diagnostic Devices – Large number of variables – Rapid technological advances – Classification criteria based on: • The human body • Inherent device related potential hazards • Customary device use (O. R. , I. C. U. , N. I. C. U. ) 20
Classification of Medical Devices • Non In vitro Diagnostic Devices – Large number of variables – Rapid technological advances – Classification criteria based on: • The human body • Inherent device related potential hazards • Customary device use (O. R. , I. C. U. , N. I. C. U. ) 20
 Definitions • “Invasive device” means a medical device that is intended to come into contact with the surface of the eye or penetrate the body, either through a body orifice or through the body surface. • “Surgically invasive device” means an invasive device that is intended to enter the body through an artificially created opening that provides access to body structures and fluids. 21
Definitions • “Invasive device” means a medical device that is intended to come into contact with the surface of the eye or penetrate the body, either through a body orifice or through the body surface. • “Surgically invasive device” means an invasive device that is intended to enter the body through an artificially created opening that provides access to body structures and fluids. 21
 Definitions • “Central cardiovascular system” means the heart, pericardium, pulmonary veins, pulmonary arteries, cardiac veins, coronary arteries, common carotid arteries, cerebral arteries, brachiocephalic artery, aorta, inferior and superior vena cava, renal arteries, iliac arteries and femoral arteries. • “Surgical or dental instrument” means a reusable medical device that is intended for surgical or dental use, including cutting, drilling, sawing, scraping, clamping, hammering, puncturing, dilating, retracting or clipping, without connection to an active device. 22
Definitions • “Central cardiovascular system” means the heart, pericardium, pulmonary veins, pulmonary arteries, cardiac veins, coronary arteries, common carotid arteries, cerebral arteries, brachiocephalic artery, aorta, inferior and superior vena cava, renal arteries, iliac arteries and femoral arteries. • “Surgical or dental instrument” means a reusable medical device that is intended for surgical or dental use, including cutting, drilling, sawing, scraping, clamping, hammering, puncturing, dilating, retracting or clipping, without connection to an active device. 22
 Definitions • “Active device” means a medical device that depends for its operation on a source of energy other than energy generated by the human body or gravity. A medical device that transmits or withdraws energy or a substance to or from a patient without substantially altering the energy or the substance is not an active device. 23
Definitions • “Active device” means a medical device that depends for its operation on a source of energy other than energy generated by the human body or gravity. A medical device that transmits or withdraws energy or a substance to or from a patient without substantially altering the energy or the substance is not an active device. 23
 Definitions • “Active Diagnostic Device” means an active device that, whether used alone or in combination with another medical device, is intended to supply information for the purpose of detecting, monitoring or treating a physiological condition, state of health, illness or congenital deformity. • “Active Therapeutic Device” means an active device that, whether used alone or in combination with another medical device, is intended to support, modify, replace or restore a biological function or structure for the purpose of treating or mitigating an illness or injury or a symptom of an illness or injury. 24
Definitions • “Active Diagnostic Device” means an active device that, whether used alone or in combination with another medical device, is intended to supply information for the purpose of detecting, monitoring or treating a physiological condition, state of health, illness or congenital deformity. • “Active Therapeutic Device” means an active device that, whether used alone or in combination with another medical device, is intended to support, modify, replace or restore a biological function or structure for the purpose of treating or mitigating an illness or injury or a symptom of an illness or injury. 24
 Classification of Medical Devices • In vitro Diagnostic Devices (IVDDs) – IVDD means a medical device that is intended to be used in vitro for the examination of specimens taken from the body. – The greater the risk associated with a false positive/false negative, the higher the Class. 25
Classification of Medical Devices • In vitro Diagnostic Devices (IVDDs) – IVDD means a medical device that is intended to be used in vitro for the examination of specimens taken from the body. – The greater the risk associated with a false positive/false negative, the higher the Class. 25
 Definitions • “Near patient IVDD” means an in vitro diagnostic device that is intended for use outside a laboratory, for testing at home or at the point of care, such as a pharmacy, a health care professional’s office or the bedside. 26
Definitions • “Near patient IVDD” means an in vitro diagnostic device that is intended for use outside a laboratory, for testing at home or at the point of care, such as a pharmacy, a health care professional’s office or the bedside. 26
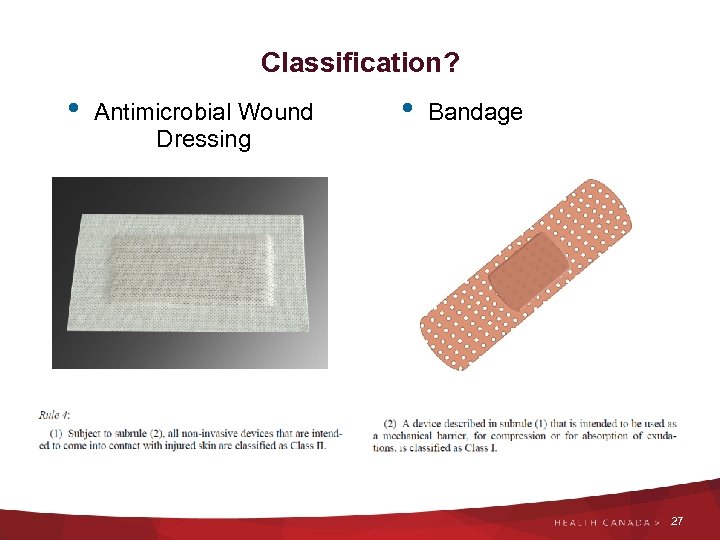 Classification? • Antimicrobial Wound Dressing • Bandage 27
Classification? • Antimicrobial Wound Dressing • Bandage 27
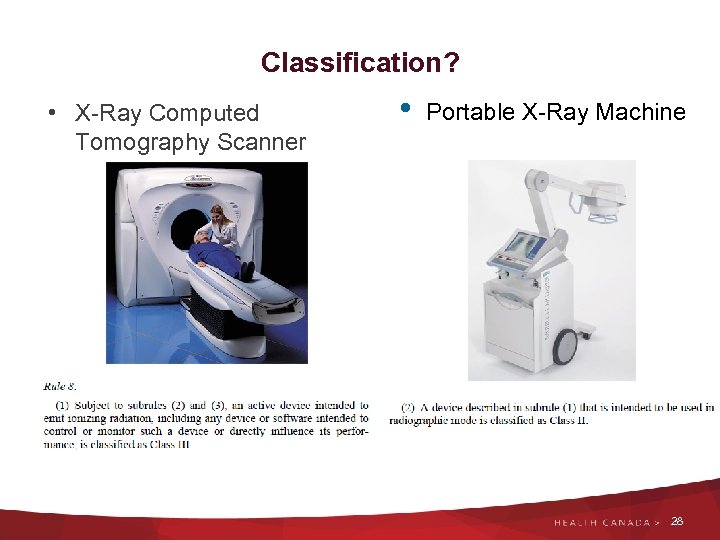 Classification? • X-Ray Computed Tomography Scanner • Portable X-Ray Machine 28
Classification? • X-Ray Computed Tomography Scanner • Portable X-Ray Machine 28
 Classification? • Natural Absorbable Suture X 29
Classification? • Natural Absorbable Suture X 29
 Classification? • Oral Thermometer X 30
Classification? • Oral Thermometer X 30
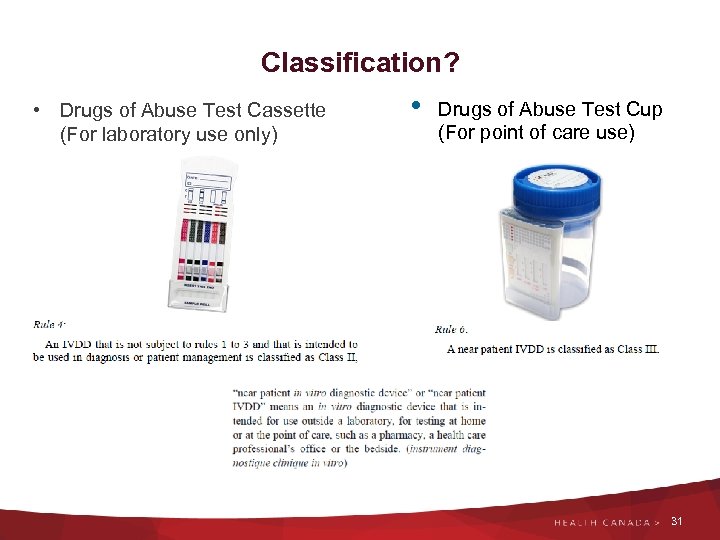 Classification? • Drugs of Abuse Test Cassette (For laboratory use only) • Drugs of Abuse Test Cup (For point of care use) 31
Classification? • Drugs of Abuse Test Cassette (For laboratory use only) • Drugs of Abuse Test Cup (For point of care use) 31
 Classification? • Pregnancy Test 32
Classification? • Pregnancy Test 32
 Overview • • Class I (low risk) – Class IV (high risk) Pre-market resources apply primarily to Class III & IV Product licensing applies to Class II, III & IV Quality Management System requirements apply to Class II, III & IV • Post-market requirements apply to all classes 33
Overview • • Class I (low risk) – Class IV (high risk) Pre-market resources apply primarily to Class III & IV Product licensing applies to Class II, III & IV Quality Management System requirements apply to Class II, III & IV • Post-market requirements apply to all classes 33
 Other Regulatory Provisions • Part 2: Special Access Programme – Access to custom-made devices (other than massproduced device) – Access to devices for emergency use or if conventional therapies have failed, are unavailable or are unsuitable – Applications in prescribed format must be received from a health care professional – Authorization for sale granted to manufacturer of the device 34
Other Regulatory Provisions • Part 2: Special Access Programme – Access to custom-made devices (other than massproduced device) – Access to devices for emergency use or if conventional therapies have failed, are unavailable or are unsuitable – Applications in prescribed format must be received from a health care professional – Authorization for sale granted to manufacturer of the device 34
 Other Regulatory Provisions • Part 3: Investigational Testing – Grants authorization to conduct clinical trials involving human subjects – Applications are received from the manufacturer, including a risk assessment and detailed protocol – Allows manufacturers to acquire evidence of safety and effectiveness as required by Sections 10 -20 of the Regulations 35
Other Regulatory Provisions • Part 3: Investigational Testing – Grants authorization to conduct clinical trials involving human subjects – Applications are received from the manufacturer, including a risk assessment and detailed protocol – Allows manufacturers to acquire evidence of safety and effectiveness as required by Sections 10 -20 of the Regulations 35
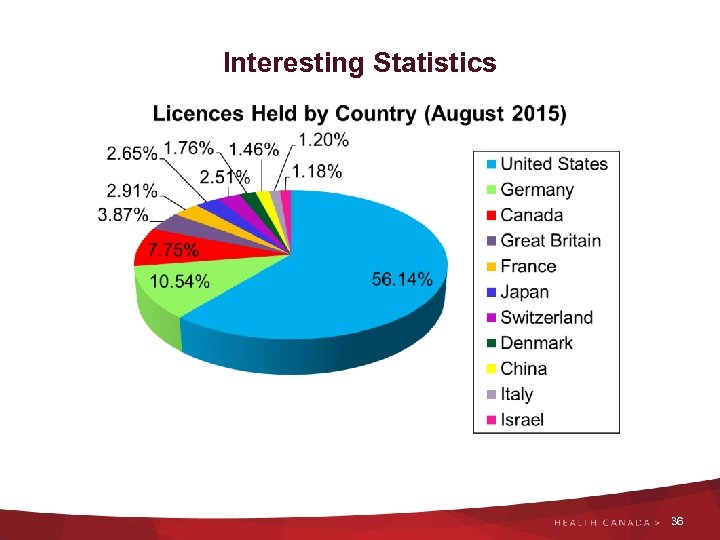 Interesting Statistics 36
Interesting Statistics 36
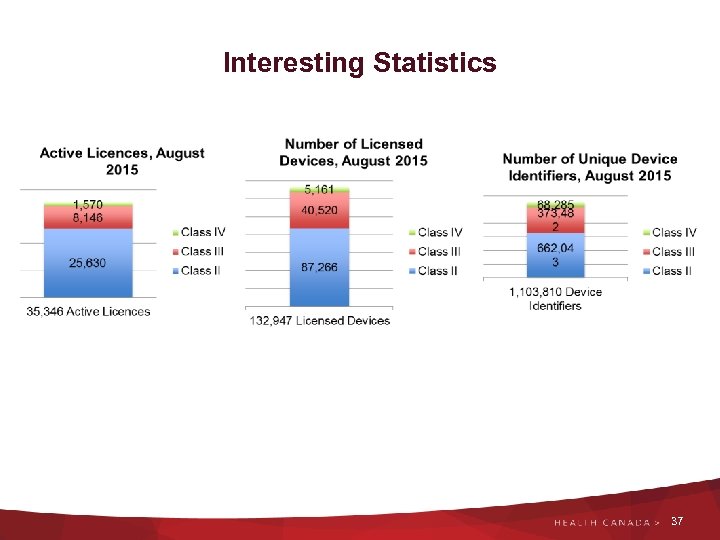 Interesting Statistics 37
Interesting Statistics 37
 Thank You! 38
Thank You! 38


