6c0e389ff12e4cf6ed5804dab70af787.ppt
- Количество слайдов: 40

Outcome Measurements as Clinical Biomarkers Sindhu Ramchandren, M. D. , M. S Assistant Professor of Neurology Wayne State University-Detroit Medical Center sramchan@med. wayne. edu

Impact of Charcot Marie Tooth Disease (CMT) • Prevalence: 1 in 2500 people • Childhood/adolescent onset; higharched feet, distal atrophy, areflexic • Financial burden unclear; but, for example, cost of Diabetic PN alone: 4 to 10 billion dollars annually • Adverse Impact on Quality of Life (QOL): pain, disability, depression Despite promising animal studies, no therapy that is curative or disease modifying exists in CMT
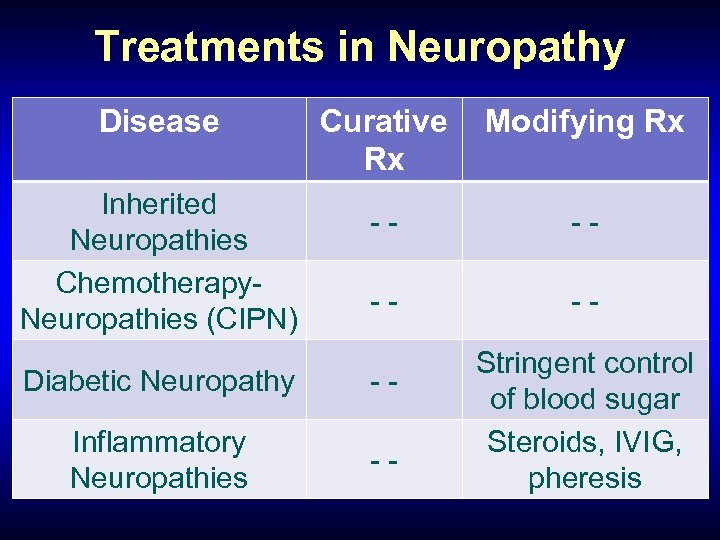
Treatments in Neuropathy Disease Inherited Neuropathies Chemotherapy. Neuropathies (CIPN) Curative Rx Modifying Rx -- -- Diabetic Neuropathy -- Inflammatory Neuropathies -- Stringent control of blood sugar Steroids, IVIG, pheresis
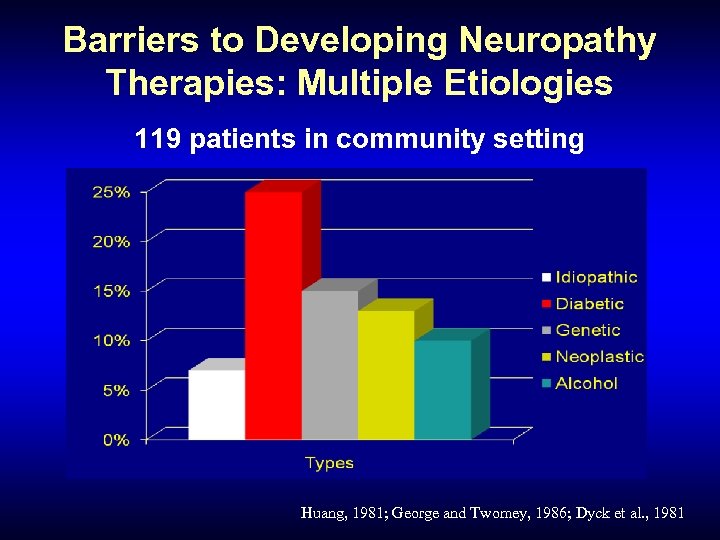
Barriers to Developing Neuropathy Therapies: Multiple Etiologies 119 patients in community setting Huang, 1981; George and Twomey, 1986; Dyck et al. , 1981
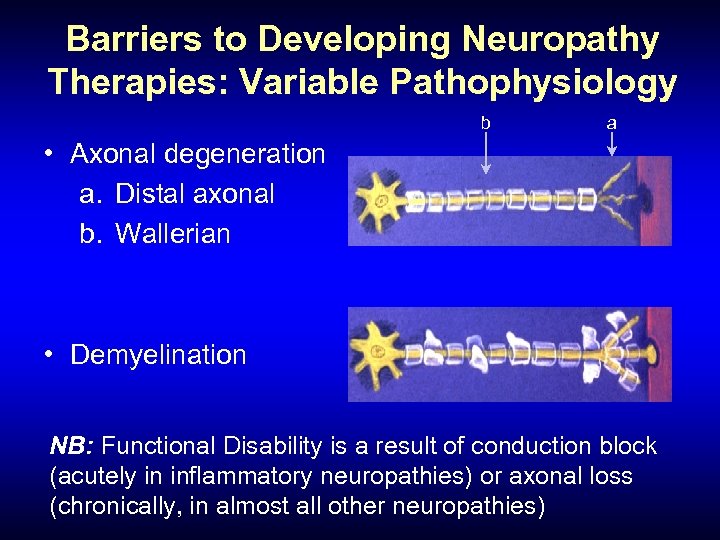
Barriers to Developing Neuropathy Therapies: Variable Pathophysiology b a • Axonal degeneration a. Distal axonal b. Wallerian • Demyelination NB: Functional Disability is a result of conduction block (acutely in inflammatory neuropathies) or axonal loss (chronically, in almost all other neuropathies)
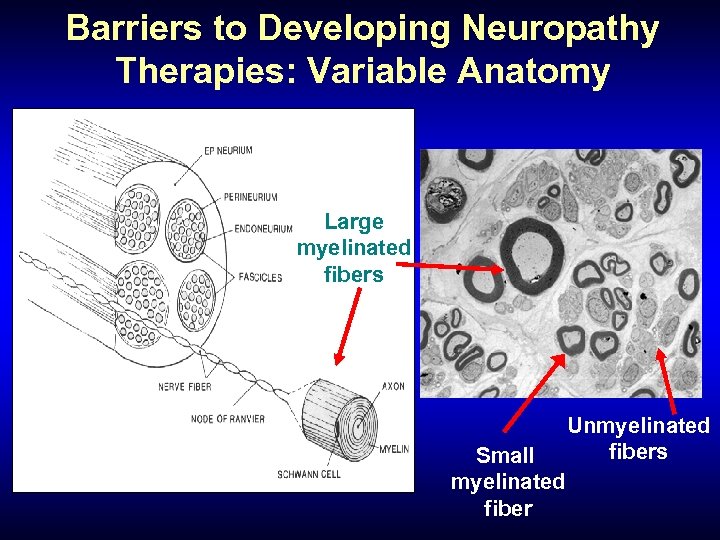
Barriers to Developing Neuropathy Therapies: Variable Anatomy Large myelinated fibers Small myelinated fiber Unmyelinated fibers

Clinical-Pathophysiologic Correlates • Large fibers: “Negative symptoms” – Weakness, wasting, poor balance, sensory loss, areflexia • Small fibers: “Positive symptoms” – Allodynia, hyperesthesia, cramps, tingling, pins and needles, burning, etc.

Identifying Barriers to Developing Neuropathy Therapies • Not one disease; multiple etiologies • Variable pathophysiology: – Axonal vs. demyelinating processes – Small vs. Large fiber involvement • Given the above, what outcome measure should we choose for a neuropathy trial? – Need to better understand natural history of the disease, in order to select an outcome measure that can improve in a 1 -2 year trial

Outcome Measures and the FDA • FDA requirement for Rx approval: improvement in a Clinically Meaningful Outcome Measure • In the absence of clinically meaningful OMs (or if trial cost of gathering such OM data is prohibitive), a Surrogate Measure can be used. – A Biomarker, which is a potential surrogate measure, is a clinical or laboratory measure of disease activity • Selecting the best outcome measure for your clinical trial requires careful study of the disease itself.

“Why should I care what outcome measure is chosen for a clinical trial? If the drug works, the patient will be cured, wouldn’t it be obvious? ”

Chronic Inflammatory Demyelinating Polyradiculoneuropathy (CIDP) • Prevalence of 0. 8 - 1. 9 per 100, 000 adults • Aggressive: progression or recurrent relapses > 8 weeks from symptom onset • Large fiber (vibration, proprioception) involved more than small (pain, temperature) • NCS: Patchy demyelinating neuropathy with conduction block and temporal dispersion early on; later: secondary axonal loss • Unfortunately- not much natural history data collected on effect on outcome measures in CIDP

IVIG in CIDP Clinical Trials TRIAL OUTCOME MEASURE Cross -over placebo trial of high-dose IVIG in CIDP (1 -2 courses 8 days apart; 7 patients) 1 point Δ in Rankin score RCT of High-dose IVIG vs. placebo in CIDP (single treatment over 5 days) 1 point Δ in Rankin score (score of at least 3 for inclusion - van Doorn et al. , Neurology 1990 - Vermeulen et al. , JNNP 1993 Cross-over placebo trial of high-dose 20 point Δ in Neurological disability score IVIG in Rx-naïve CIDP (2 courses over 8 weeks) - Hahn et al. , Brain 1996 RCT of IVIG vs. Prednisolone in CIDP 11 point Δ in ODS - Hughes et al. , Ann Neurol 2001 RCT of IVIG vs. placebo Δ in Average Muscle Score (modified MRC) IVIG for CIDP (ICE trial) Difference betwn-groups in proportion of INCAT responders vs. non-responders (1 point Δ in INCAT = response) - Mendell et al. , Neurology 2001 - Hughes et al. , Lancet Neurol 2008
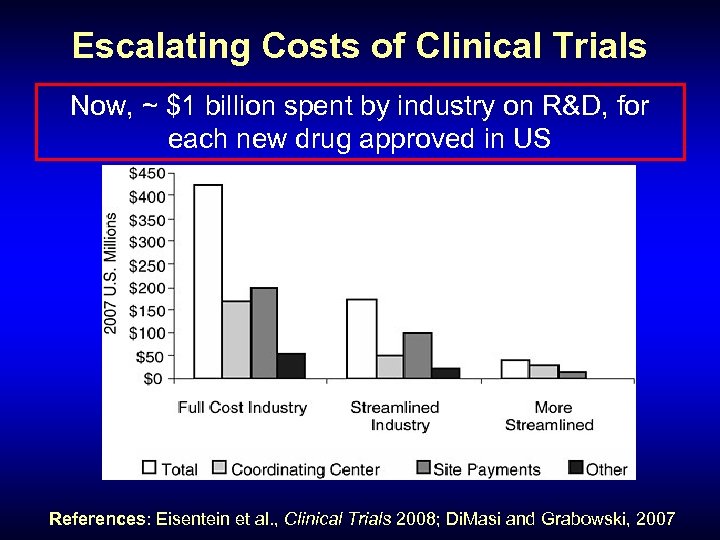
Escalating Costs of Clinical Trials Now, ~ $1 billion spent by industry on R&D, for each new drug approved in US References: Eisentein et al. , Clinical Trials 2008; Di. Masi and Grabowski, 2007
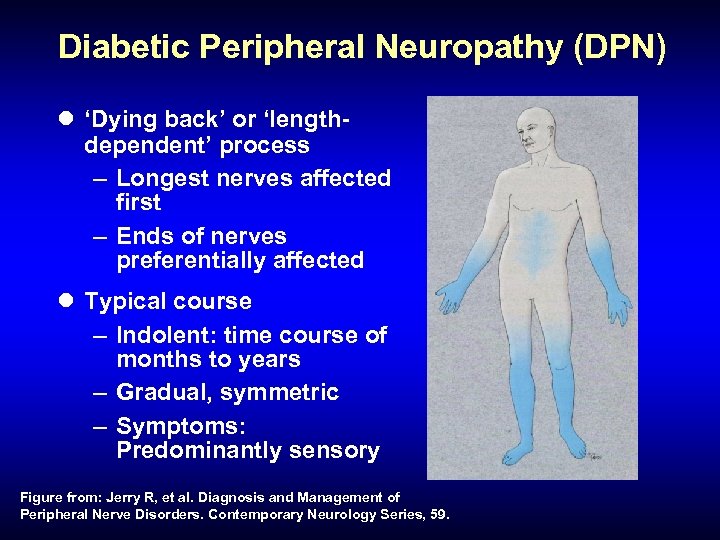
Diabetic Peripheral Neuropathy (DPN) l ‘Dying back’ or ‘lengthdependent’ process – Longest nerves affected first – Ends of nerves preferentially affected l Typical course – Indolent: time course of months to years – Gradual, symmetric – Symptoms: Predominantly sensory Figure from: Jerry R, et al. Diagnosis and Management of Peripheral Nerve Disorders. Contemporary Neurology Series, 59.
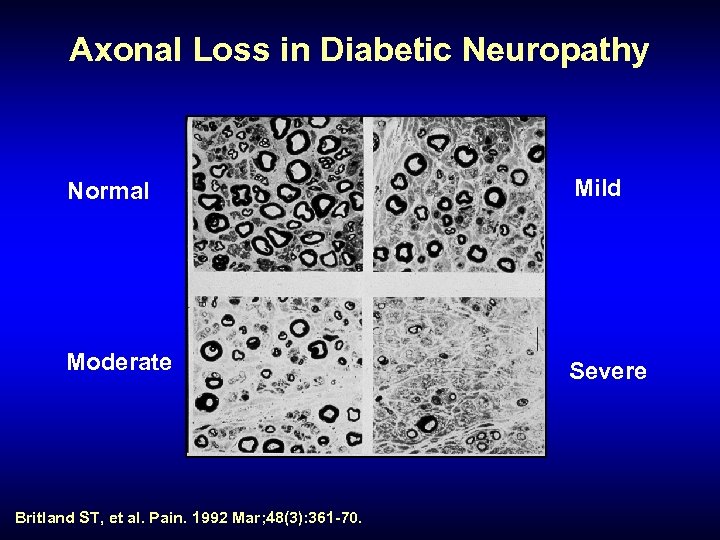
Axonal Loss in Diabetic Neuropathy Normal Mild Moderate Severe Britland ST, et al. Pain. 1992 Mar; 48(3): 361 -70.
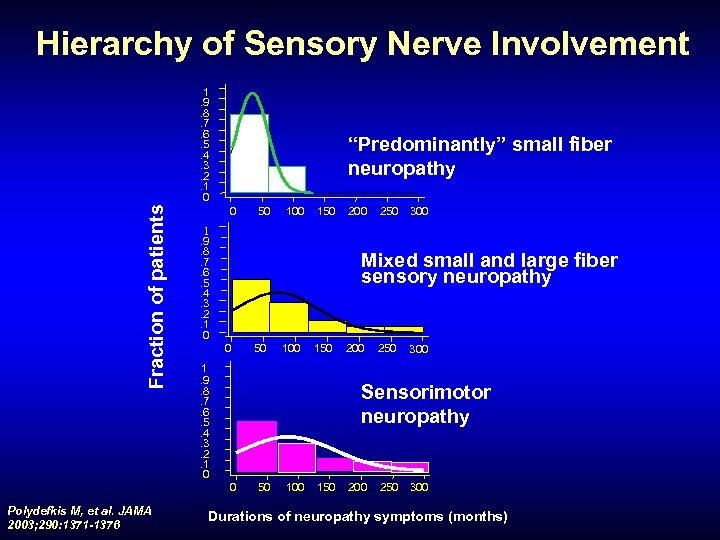
Hierarchy of Sensory Nerve Involvement Fraction of patients 1. 9. 8. 7. 6. 5. 4. 3. 2. 1 0 “Predominantly” small fiber neuropathy 0 100 150 200 250 300 1 . 9. 8. 7. 6. 5. 4. 3. 2. 1 0 Mixed small and large fiber sensory neuropathy 0 50 100 150 1. 9. 8. 7. 6. 5. 4. 3. 2. 1 0 200 250 300 Sensorimotor neuropathy 0 Polydefkis M, et al. JAMA 2003; 290: 1371 -1376 50 50 100 150 200 250 300 Durations of neuropathy symptoms (months)

Past Clinical Trials. Diabetic Peripheral Neuropathy • Natural history of DPN- vey slow progression • DPN Trials until the mid-90 s may have been inefficiently designed*: – Enrolled patients with mild and severe PN – Short trial duration – Looked for reversal of endpoints such as NCS abnormalities that (a) did not have time to change and (b) were more likely to slow in progression than reverse Need clinically meaningful end-points that can change within the allotted study time frame - *Dyck PJ, et al. Diabetes Care 2007; 30: 2619– 2625 - *Ziegler D, et al. Int Rev Neurobiol 2002; 50: 431– 463 - *Carrington AL, et al. Diabetes Care 2001; 25: 2010– 2015

Diabetic Neuropathy Trials: Future • Validated Pain Scales have taken over as the patient reported outcome measure of choice • Need to identify Clinical Outcome Measures – no consensus: measures used include NIS-LL, NCS… – Intraepidermal nerve fiber density is problematic: • 20 -50% of small fiber neuropathies are idiopathic • Clinical Exam not very sensitive or specific • Processes other than neuropathy cause small fiber loss • IENF density has not been shown to improve clinical management or patient health outcome – New research: early changes in axon excitability due to ion channel dysfunction may precede longterm axonal loss (Sung et al; Diabetes 2012: 1592 -8)

Charcot Marie Tooth Disease (CMT) • Natural history: variable, but for CMT 1 A, slow progression • NCS in CMT 1 A: Symmetric demyelinating neuropathy, typically without conduction block or temporal dispersion • Despite demyelination, disability in CMT 1 A correlates to secondary axonal loss (i. e. chronic denervation)
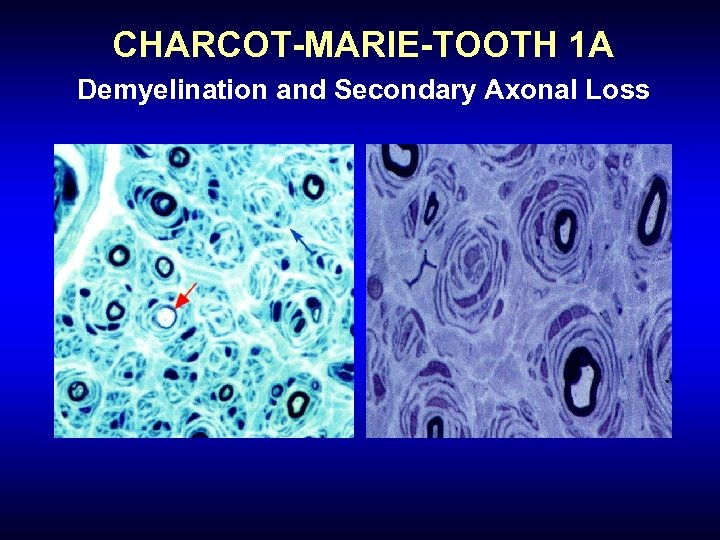
CHARCOT-MARIE-TOOTH 1 A Demyelination and Secondary Axonal Loss

Past Failures in CMT trials: Ineffective Drugs Vs. Trial Design Errors HISTORICAL RATE OF CHANGE/TIME FOR OUTCOME MEASURE No change in 8 years Nerve Conduction Velocities (Sancho 1999; Sahenk 1999; Krajewski 2000; Shy 2008) Charcot-Marie-Tooth Neuropathy Score (CMTNS) 1. 4 points/2 years (CMT 1 A); 2. 89 points/10 years (CMT 1 X) (Shy, 2008) OUTCOME MEASURES USED IN CMT TRIALS Myelinated fiber regeneration Sahenk et al. , Neurology 2005 Myosin isoform changes Smith et al. , Muscle Nerve 2006 2. 5 m/sec/yr improvement in NCV Burns et al. , Lancet Neurol 2009 10 m/sec improvement in NCV Verhamme et al. , BMC Med 2009 5 -point improvement in CMTNS Micallef et al. , Lancet Neurol 2009 1. 5 betwn-group in CMTNS in 2 yrs Pareyson et al. , Lancet Neurol 2011

Need to Develop and Validate new Clinical Outcome Measures • Inherited Neuropathy Consortium (NINDSNS 065712; PI Dr. Shy): identify reliable, sensitive and valid outcome measures for clinical trials in pediatric CMT. • Clinical and related disease-quantifying measures are in development (ex. CMTPed. S) • There is currently no Patient-Reported Outcome (PRO) measure being developed for future trials.

Patient-Reported Outcome (PRO) Measures • PROs provide Direct measure of patient’s perspective on a given domain (depression, pain, function (ADLs), without clinician interpretation • FDA defines treatment benefit as a Direct measure of improvement Direct measures include: - Survival - How patient feels, or - How patient functions Surrogate measures of improvement include: - Blood pressure, Cholesterol, Hb. A 1 C, etc. • Health-Related Quality of Life (QOL) is a specific PRO measure that assesses the patient’s perspective on the impact of chronic illnesses on their life’s quality or function.

Cancer Treat Rep. 1985 Oct; 69(10): 1155 -9. Food and Drug Administration requirements for approval of new anticancer drugs. Johnson JR, Temple R. Abstract For approval of new drugs, the law requires a minimum of two independent well-controlled studies for each indication. The preferred study design is prospective, randomized, and comparative. The use of historical controls, while sometimes acceptable, must be justified. A favorable effect on survival and/or quality of life is generally required for approval.
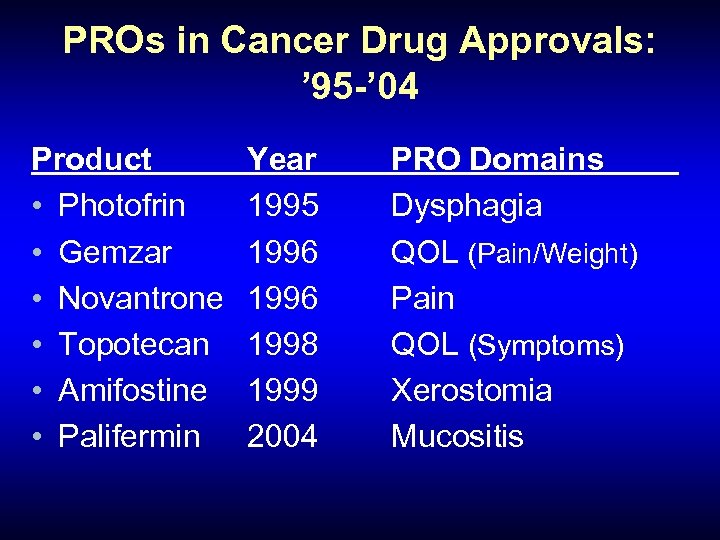
PROs in Cancer Drug Approvals: ’ 95 -’ 04 Product • Photofrin • Gemzar • Novantrone • Topotecan • Amifostine • Palifermin Year 1995 1996 1998 1999 2004 PRO Domains Dysphagia QOL (Pain/Weight) Pain QOL (Symptoms) Xerostomia Mucositis

QOL as a PRO in Clinical Trials • QOL instruments have prognostic value in many chronic illnesses, including cancer (Mapes 2003, Singh 2005; Cunningham 2005, Fang 2004; Gotay 2008; Movsas 2009; Quinten 2009; Sprangers 2010) • In neuromuscular disorders, QOL may identify biological processes that affect long-term function earlier than traditional trial endpoints, because it is a more comprehensive assessment of one’s health status • In addition, PROs provide invaluable data on toxicity and tolerability of a therapy in a clinical trial • However, the decision on whether to use PROs as primary or secondary endpoints in clinical trial must be guided by their added value (ie, will the PRO data make a difference to study conclusions at the end of a trial? )

Utilization of QOL Instruments in Neuropathy Trials* • Systematic literature review to assess the utilization of QOL in randomized controlled neuropathy trials • Only 21% of randomized neuropathy trials utilized QOL as a primary or secondary outcome measure. • Anonymous survey of 169 neuromuscular specialists in the United States to assess their awareness of validated QOL measures in neuropathy. • Among the 25. 4% survey respondents, 97. 7% acknowledged the importance of assessing QOL, but less than a third could identify a validated QOL measure. *-Ramchandren S and De. Sousa C
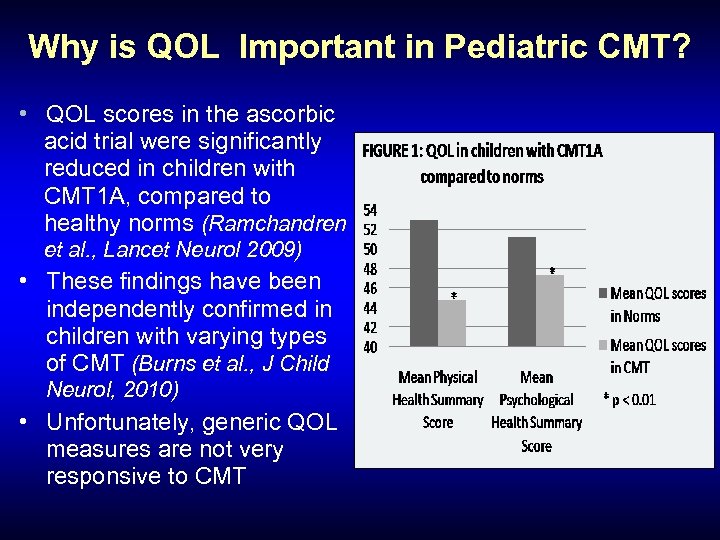
Why is QOL Important in Pediatric CMT? • QOL scores in the ascorbic acid trial were significantly reduced in children with CMT 1 A, compared to healthy norms (Ramchandren et al. , Lancet Neurol 2009) • These findings have been independently confirmed in children with varying types of CMT (Burns et al. , J Child Neurol, 2010) • Unfortunately, generic QOL measures are not very responsive to CMT

Study Goals and Methods* 1. Collaborate with the Inherited Neuropathy Consortium to develop and validate a disease-specific pediatric CMT quality of life instrument (p. CMTQOL) through an iterative process 2. Identify new clinical outcome measures that accurately reflects CMT disease progression in children, among composite neuropathy scores, electrophysiology and QOL data * -Ramchandren, K 23 NS 072279

Systematic Review of Existing QOL Measures Analysis of generic pediatric QOL measures (PEDSQL, TACQOL, CHQ) Analysis of adult generic QOL measure (SF 36): Summary of domains from CMT literature: Physical physical function, physical Physical function, role physical disability, toe and complaints, daily living, physical, bodily pain, heel walk, independent bodily pain, general health ambulation, forearm/hand intrinsic muscle strength, lower limb weakness, leg cramps, pain Social cognitive, positive moods and negative moods (psychological function), physical role-social, family activities, family cohesion Emotional role-social, mental health, selfesteem, parental emotion and time impact Other vitality, social function, family, leisure items, community involvement/ productivity role emotional, mental health, selfesteem financial status, living situation, religion Interaction in our CMT clinic: Fatigue Psychosocial stressors (Arnold, 2005), depression Inadequate sleep

Cross-sectional studies correlating generic QOL domains to functional assessments* • 70 children (40 boys, 30 girls) aged 5 -16 years (mean age 9. 1, SD 3. 0 yrs) • Independent determinants of reduced QOL in children with CMT 1 A were leg cramps, hand tremor, and reduced long jump distance • Many standardized clinical and electrophysiologic measures currently used as end-points in clinical trials of CMT correlated poorly with QOL measured by the CHQ. QOL Domain Predictor variable β Multiple weight r 2 model Physical Health Summary Long jump. 456** Leg cramps . 359** Psychological Health Summary Leg cramps . 365** . 322 . 386 . 329** Hand tremor *Burns J, Ramchandren S, Ryan MM, Shy M, Ouvrier R. Neurology 2010

Developing questions pertinent to the domains: collaboration with Neuro-QOL • • Neuro-QOL: an offshoot of the NIH Roadmap initiative Patient-Reported Outcomes Measurement Information System (PROMIS) Benefits: – – – Large database of items pertinent to specific domains (depression, anger, physical function), normative data, Rasch and Item response theory (IRT) analyses, CAT capability These item banks will be accessible to clinical researchers eventually through web-based computer adaptive testing systems PROMIS core group is interested in collaborating with investigators interested in developing disease-specific instruments using questions from their item bank.

Conceptual Scheme for the Pediatric CMT QOL Instrument. Ratified by Expert Consensus* • Physical symptoms - Fatigue/weakness, pain, sleep, cramps • Physical function - ADL’s, Upper extremity and lower extremity functions, balance • Social role-physical - Physical activities with peers, adults • Mental symptoms - Stigma, anxiety/fear, depression, stress • Mental function - Perceived cognitive function • Social role-emotional - Self esteem, emotional bonding with peers, adults Physical Domain Mental Domain *ENMC meeting, Naarden, September 2009
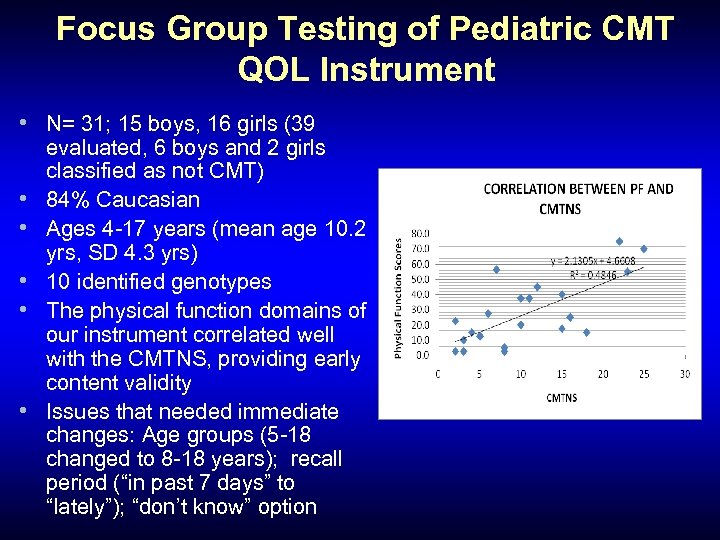
Focus Group Testing of Pediatric CMT QOL Instrument • N= 31; 15 boys, 16 girls (39 • • • evaluated, 6 boys and 2 girls classified as not CMT) 84% Caucasian Ages 4 -17 years (mean age 10. 2 yrs, SD 4. 3 yrs) 10 identified genotypes The physical function domains of our instrument correlated well with the CMTNS, providing early content validity Issues that needed immediate changes: Age groups (5 -18 changed to 8 -18 years); recall period (“in past 7 days” to “lately”); “don’t know” option

Final Version of p. CMT-QOL Three versions: Six Domains 1. A child version (for children ages 8 -18) PHYSICAL 2. A parent-proxy version (for children ages 8 -18) Function (10 questions) 3. A parent-proxy version (for children ages 7 and younger) Symptoms (10 questions) Social Activities (10 questions) EMOTIONAL Feelings (10 questions) Cognition (10 questions) Social Skills (10 questions)


Patient Enrollment in p. CMT-QOL Table 1: Summary of enrollment and demographic data Sample size: (N; enrolled to date September 5 th, 2012) = 131 Site breakdown: Children’s hospital of Philadelphia= 13 National Hospital of Neurology /Neurosurgery, UK = 29 University of Rochester = 5 Wayne State University = 36 Children’s Hospital at Westmead, Sydney, Australia = 43 University of Iowa = 5 Gender: 65 males , 63 females 3 missing data Ages 8 -18: 99; Ages 7 and under: 30; 2 missing data

Other Measures being Investigated • Disability Severity Index: An anchor for CMT – Categorization of disability severity based on walking/movement aid used; both patient and physician perspectives – This will serve as an Anchor in future, i. e. will help to determine the minimum change in our QOL measure that is functionally important to patient – Status: 200 patients, 94 physicians (3 of whom have CMT) enrolled; data analysis in progress

Conclusions • There is a critical need for new therapies and new clinical outcome measures in Peripheral Neuropathy • QOL is a useful outcome measure since it is severely reduced in pediatric inherited neuropathy and therefore can be targeted for improvement in a therapeutic trial • We have developed a disease-specific pediatric CMT QOL outcome measure and completed preliminary testing for content validity • Future steps include psychometric testing of the final instrument through Dr. Shy’s RDCRC- Inherited Neuropathy Consortium study, as well as identification of other clinical outcome measures.

Acknowledgements • Mentors - Dr. Michael Shy (U Iowa), Dr. Lawrence Lum (KCI), Dr. Jeff Loeb (WSU), Dr. Deborah Ellis (WSU), Dr. Penner (WSU) • INC Collaborators - Dr. Richard Finkel, Dr. Joshua Burns, Dr. Rosemary Shy, Dr. Mary Reilly, Dr. Muntoni, Dr. Pareyson, Dr. Hermann • Coordinators - Carly Siskind, Lindsey Miller, Shawna Feely, Stephanie Neal • Biostatisticians - Dr. Templin (WSU), Dr. Mc. Dermott (U of Rochester), Dr. Ingemar Merkies (Spaarne Hospital, The Netherlands) • Grant support for Dr. Ramchandren: - NIH (NINDS): 1 K 23 NS 072279
6c0e389ff12e4cf6ed5804dab70af787.ppt