ff5d80739585a3d7b283ce72cb52da0e.ppt
- Количество слайдов: 21
 Organic Chemistry
Organic Chemistry
 Organic Chemistry Chemical compounds fall into one of two categories: Organic compounds Inorganic compounds Organic compounds can be found naturally or they can be synthesized, but they all contain C and a few other elements. There are far more organic compounds than inorganic compounds.
Organic Chemistry Chemical compounds fall into one of two categories: Organic compounds Inorganic compounds Organic compounds can be found naturally or they can be synthesized, but they all contain C and a few other elements. There are far more organic compounds than inorganic compounds.
 Organic Compounds fuels carbohydrates, fats, proteins vitamins penicillin, sulfa drugs plastics cotton, silk, wool rubber DNA ibuprofen, aspirin, morphine Organic chemistry is part of our everyday lives!
Organic Compounds fuels carbohydrates, fats, proteins vitamins penicillin, sulfa drugs plastics cotton, silk, wool rubber DNA ibuprofen, aspirin, morphine Organic chemistry is part of our everyday lives!
 Properties of Organic Compounds usually molecular (contain non-metals bonded with covalent bonds) low melting points nonpolar (insoluble, or only slightly soluble, in water) very stable (not reactive)
Properties of Organic Compounds usually molecular (contain non-metals bonded with covalent bonds) low melting points nonpolar (insoluble, or only slightly soluble, in water) very stable (not reactive)
 Carbon forms 4 covalent bonds, i. e. , it will share electrons with 4 other elements. Prefixes are used in the name to indicate how many carbons are in the compound.
Carbon forms 4 covalent bonds, i. e. , it will share electrons with 4 other elements. Prefixes are used in the name to indicate how many carbons are in the compound.
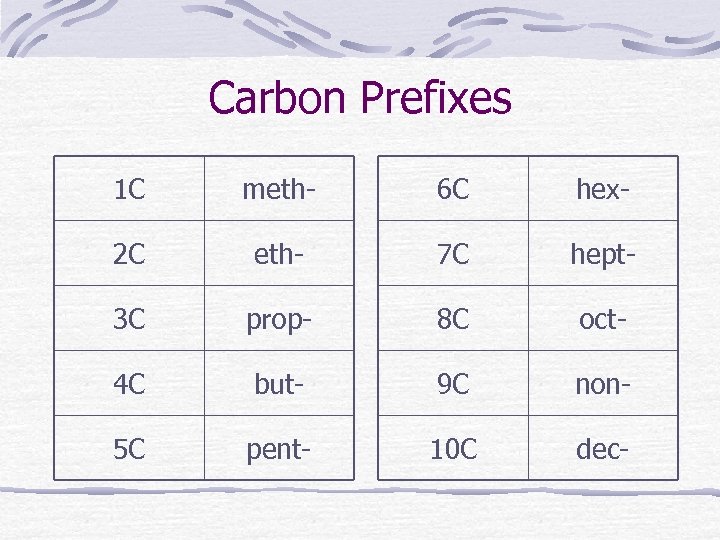 Carbon Prefixes 1 C meth- 6 C hex- 2 C eth- 7 C hept- 3 C prop- 8 C oct- 4 C but- 9 C non- 5 C pent- 10 C dec-
Carbon Prefixes 1 C meth- 6 C hex- 2 C eth- 7 C hept- 3 C prop- 8 C oct- 4 C but- 9 C non- 5 C pent- 10 C dec-
 Memory Aid Many Equestrian People Buy Pretty Handsome Horses Of Noble Descent.
Memory Aid Many Equestrian People Buy Pretty Handsome Horses Of Noble Descent.
 Naming Alkanes
Naming Alkanes
 Alkanes are hydrocarbons (consisting of only C and H) that only contain single bonds. Start with the Prefix that matches the number of carbons Alkanes have the ending –ane in their name.
Alkanes are hydrocarbons (consisting of only C and H) that only contain single bonds. Start with the Prefix that matches the number of carbons Alkanes have the ending –ane in their name.
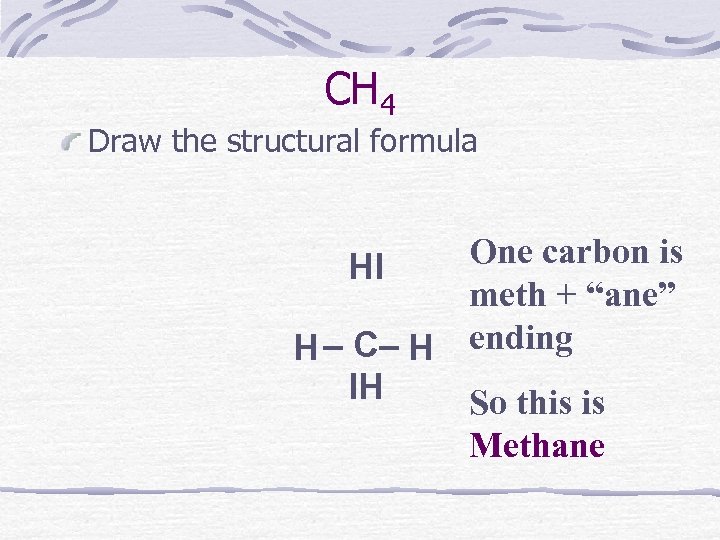 CH 4 Draw the structural formula One carbon is HI meth + “ane” H – C– H ending IH So this is Methane
CH 4 Draw the structural formula One carbon is HI meth + “ane” H – C– H ending IH So this is Methane
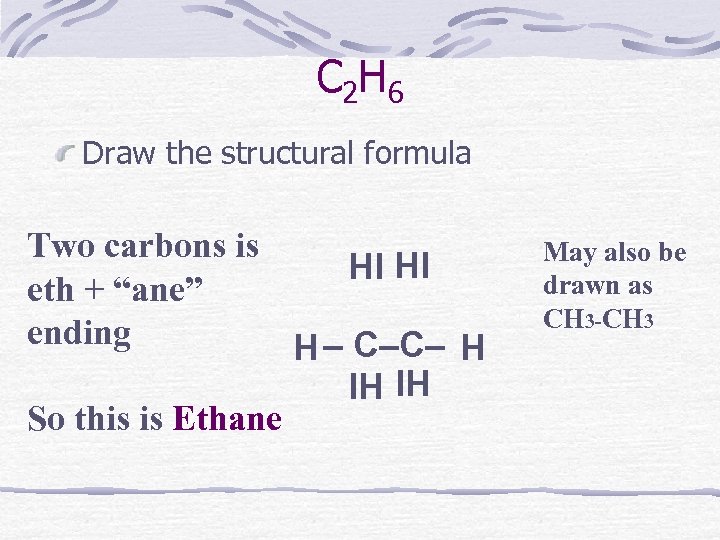 C 2 H 6 Draw the structural formula Two carbons is HI HI eth + “ane” ending H – C–C– H IH IH So this is Ethane May also be drawn as CH 3 -CH 3
C 2 H 6 Draw the structural formula Two carbons is HI HI eth + “ane” ending H – C–C– H IH IH So this is Ethane May also be drawn as CH 3 -CH 3
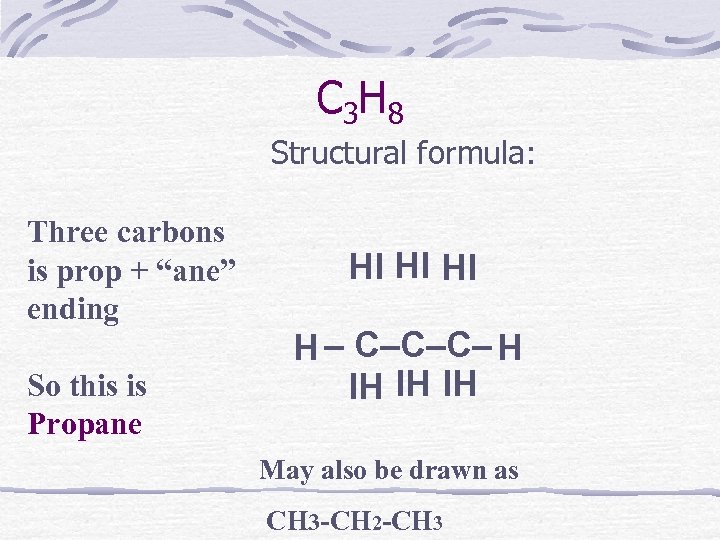 C 3 H 8 Structural formula: Three carbons is prop + “ane” ending So this is Propane HI HI HI H – C–C–C– H IH IH IH May also be drawn as CH 3 -CH 2 -CH 3
C 3 H 8 Structural formula: Three carbons is prop + “ane” ending So this is Propane HI HI HI H – C–C–C– H IH IH IH May also be drawn as CH 3 -CH 2 -CH 3
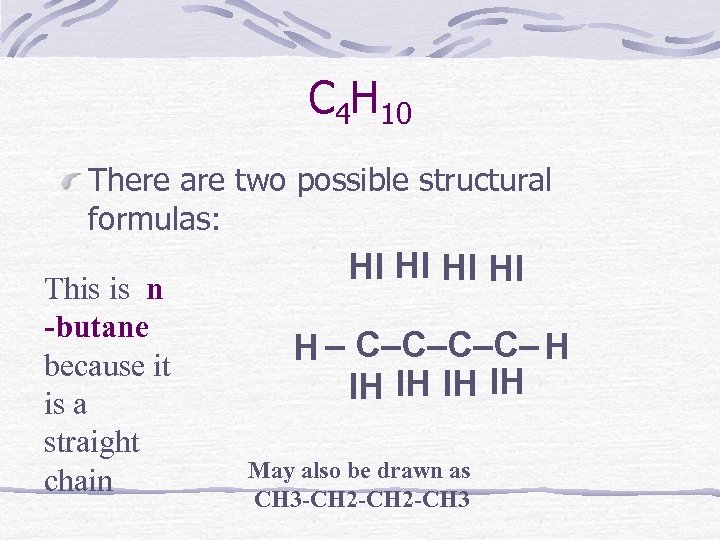 C 4 H 10 There are two possible structural formulas: This is n -butane because it is a straight chain HI HI H – C–C– H IH IH May also be drawn as CH 3 -CH 2 -CH 3
C 4 H 10 There are two possible structural formulas: This is n -butane because it is a straight chain HI HI H – C–C– H IH IH May also be drawn as CH 3 -CH 2 -CH 3
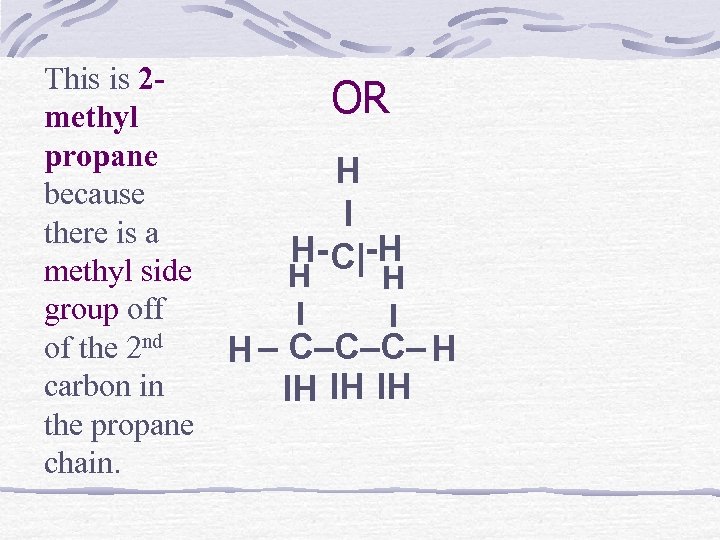 This is 2 methyl propane because there is a methyl side group off of the 2 nd carbon in the propane chain. OR H I H- C|-H H I H – C–C–C– H IH IH IH
This is 2 methyl propane because there is a methyl side group off of the 2 nd carbon in the propane chain. OR H I H- C|-H H I H – C–C–C– H IH IH IH
 Isomers are compounds that have the same molecular formula, but different structural formulas. Isomers usually have different chemical and physical properties as well. We must have some way of specifying which form of butane we are talking about.
Isomers are compounds that have the same molecular formula, but different structural formulas. Isomers usually have different chemical and physical properties as well. We must have some way of specifying which form of butane we are talking about.
 Normal Alkanes that have a continuous chain of carbons are called normal alkanes. The first isomer of butane is a normal alkane and is sometimes referred to as n-butane.
Normal Alkanes that have a continuous chain of carbons are called normal alkanes. The first isomer of butane is a normal alkane and is sometimes referred to as n-butane.
 Branched Alkanes The second isomer has a branch off the second carbon. These branches are referred to as alkyl groups. The name of the alkyl group is determined by the prefix according to the number of carbons and the ending “-yl” The branch in this case would be called methyl
Branched Alkanes The second isomer has a branch off the second carbon. These branches are referred to as alkyl groups. The name of the alkyl group is determined by the prefix according to the number of carbons and the ending “-yl” The branch in this case would be called methyl
 Rules for Naming Alkanes 1. Count the carbons of the longest continuous chain. This will be the prefix. Add –ane to it. 2. Name the alkyl groups attached to the main chain. If there are two or more of the same name, use the counting prefixes (di, tri, etc. )
Rules for Naming Alkanes 1. Count the carbons of the longest continuous chain. This will be the prefix. Add –ane to it. 2. Name the alkyl groups attached to the main chain. If there are two or more of the same name, use the counting prefixes (di, tri, etc. )
 Rules for Naming Alkanes, cont’d 3. Number the carbons, starting with the carbon closest to the branch. 4. List the alkyl groups first, in alphabetical order, using a number to indicate which carbon it branches from.
Rules for Naming Alkanes, cont’d 3. Number the carbons, starting with the carbon closest to the branch. 4. List the alkyl groups first, in alphabetical order, using a number to indicate which carbon it branches from.
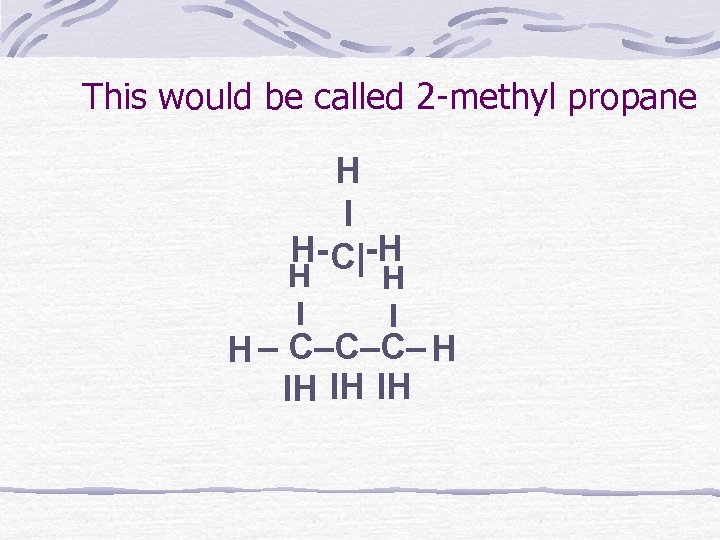 This would be called 2 -methyl propane H I H- C|-H H I H – C–C–C– H IH IH IH
This would be called 2 -methyl propane H I H- C|-H H I H – C–C–C– H IH IH IH
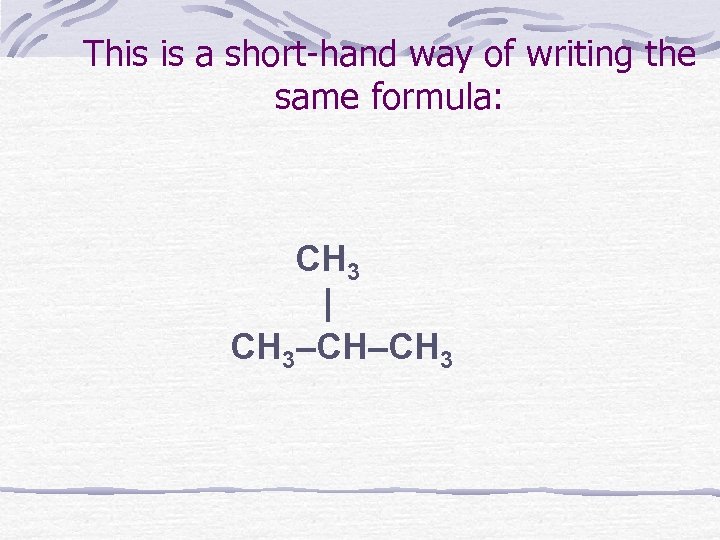 This is a short-hand way of writing the same formula: CH 3 | CH 3–CH–CH 3
This is a short-hand way of writing the same formula: CH 3 | CH 3–CH–CH 3


