e1b01b37212062e8d3b6b56900d012f4.ppt
- Количество слайдов: 59
 Ordering phenomena on the perovskite and spinel lattices: synchrotron and neutron studies Paolo G. Radaelli ISIS Facility, Rutherford Appleton Laboratory and Dept of Physics & Astronomy, University College London
Ordering phenomena on the perovskite and spinel lattices: synchrotron and neutron studies Paolo G. Radaelli ISIS Facility, Rutherford Appleton Laboratory and Dept of Physics & Astronomy, University College London
![Powder diffraction in the 21 st century? Na. V 2 O 5 [1] • Powder diffraction in the 21 st century? Na. V 2 O 5 [1] •](https://present5.com/presentation/e1b01b37212062e8d3b6b56900d012f4/image-2.jpg) Powder diffraction in the 21 st century? Na. V 2 O 5 [1] • In many systems single crystal diffraction is the tool of choice to study ordering phenomena. There are many classic and recent examples, esp. in low-dimensional systems. • Yet, powder diffraction still gives a significant contribution, esp. in systems where the high-temperature phase is highly symmetric, (perovskites, spinels, pyrochlores, etc. ) [1] J. L. de Boer, A. Meetsma, J. Baas, and T. T. M. Palstra, PRL 84 (2000) 3962. Paolo G. Radaelli, Oxford, Sept 2003
Powder diffraction in the 21 st century? Na. V 2 O 5 [1] • In many systems single crystal diffraction is the tool of choice to study ordering phenomena. There are many classic and recent examples, esp. in low-dimensional systems. • Yet, powder diffraction still gives a significant contribution, esp. in systems where the high-temperature phase is highly symmetric, (perovskites, spinels, pyrochlores, etc. ) [1] J. L. de Boer, A. Meetsma, J. Baas, and T. T. M. Palstra, PRL 84 (2000) 3962. Paolo G. Radaelli, Oxford, Sept 2003
 Outline • Charge ordering in manganites: methodology. • Frustrated systems. Ø Fe 3 O 4 revisited. • Dimer formation on the pyrochlore lattice: Ø ½-filled system: Cu. Ir 2 S 4 Ø 1 -filled system: Mg. Ti 2 O 4 • Data quality issues. • Summary. Paolo G. Radaelli, Oxford, Sept 2003
Outline • Charge ordering in manganites: methodology. • Frustrated systems. Ø Fe 3 O 4 revisited. • Dimer formation on the pyrochlore lattice: Ø ½-filled system: Cu. Ir 2 S 4 Ø 1 -filled system: Mg. Ti 2 O 4 • Data quality issues. • Summary. Paolo G. Radaelli, Oxford, Sept 2003
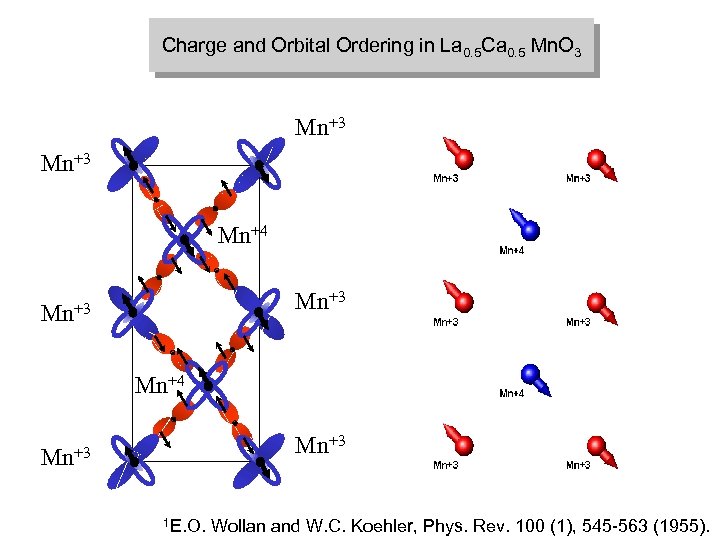 Charge and Orbital Ordering in La 0. 5 Ca 0. 5 Mn. O 3 Mn+3 Mn+4 Mn+3 1 E. O. Wollan and W. C. Koehler, Phys. Rev. 100 (1), 545 -563 (1955).
Charge and Orbital Ordering in La 0. 5 Ca 0. 5 Mn. O 3 Mn+3 Mn+4 Mn+3 1 E. O. Wollan and W. C. Koehler, Phys. Rev. 100 (1), 545 -563 (1955).
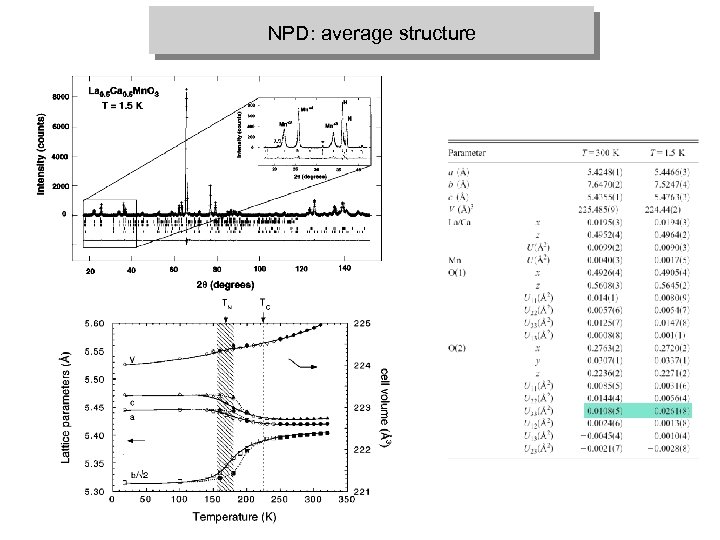 NPD: average structure
NPD: average structure
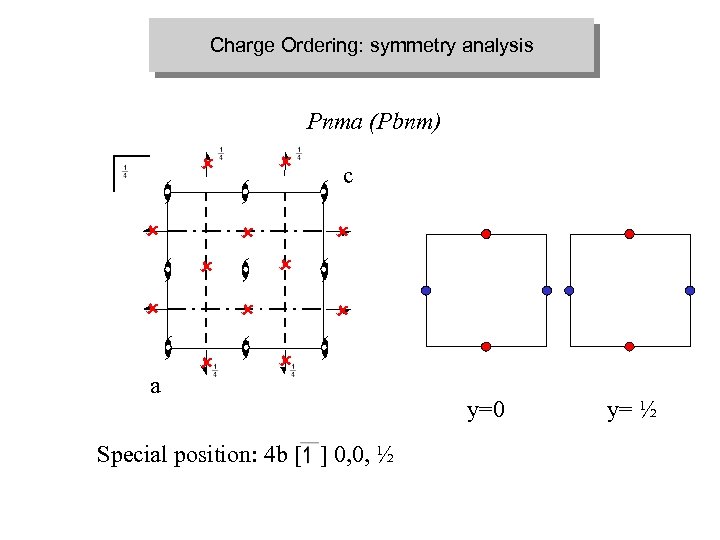 Charge Ordering: symmetry analysis Pnma (Pbnm) û û û a û û û c û û Special position: 4 b [ ] 0, 0, ½ y=0 y= ½
Charge Ordering: symmetry analysis Pnma (Pbnm) û û û a û û û c û û Special position: 4 b [ ] 0, 0, ½ y=0 y= ½
![Charge Ordering: symmetry analysis P 21/m c a Special position: 4 b [ ] Charge Ordering: symmetry analysis P 21/m c a Special position: 4 b [ ]](https://present5.com/presentation/e1b01b37212062e8d3b6b56900d012f4/image-7.jpg) Charge Ordering: symmetry analysis P 21/m c a Special position: 4 b [ ] 0, 0, ½ y=0 y= ½
Charge Ordering: symmetry analysis P 21/m c a Special position: 4 b [ ] 0, 0, ½ y=0 y= ½
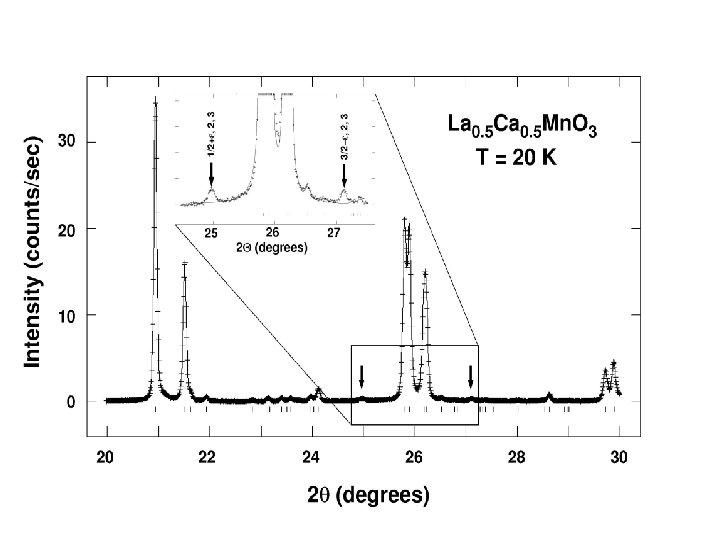
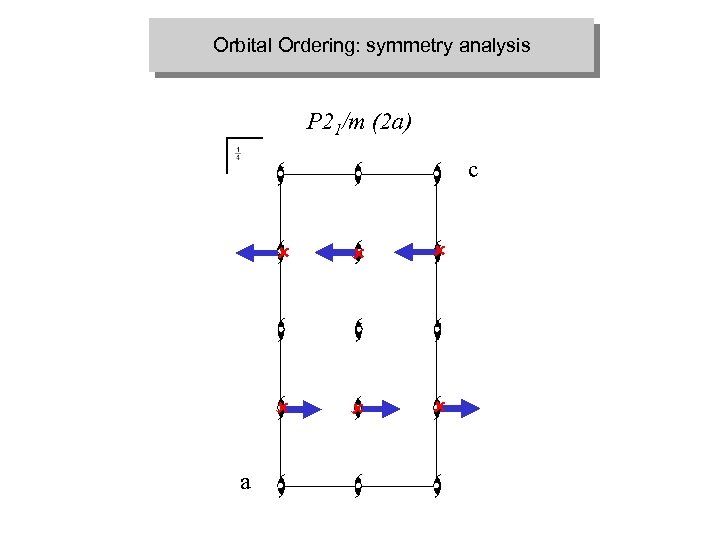 Orbital Ordering: symmetry analysis P 21/m (2 a) c û û û a û û û
Orbital Ordering: symmetry analysis P 21/m (2 a) c û û û a û û û
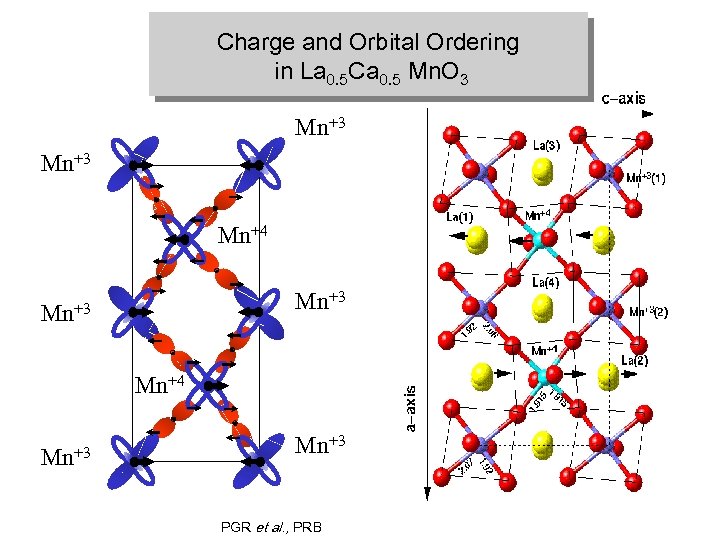 Charge and Orbital Ordering in La 0. 5 Ca 0. 5 Mn. O 3 Mn+3 Mn+4 Mn+3 PGR et al. , PRB
Charge and Orbital Ordering in La 0. 5 Ca 0. 5 Mn. O 3 Mn+3 Mn+4 Mn+3 PGR et al. , PRB
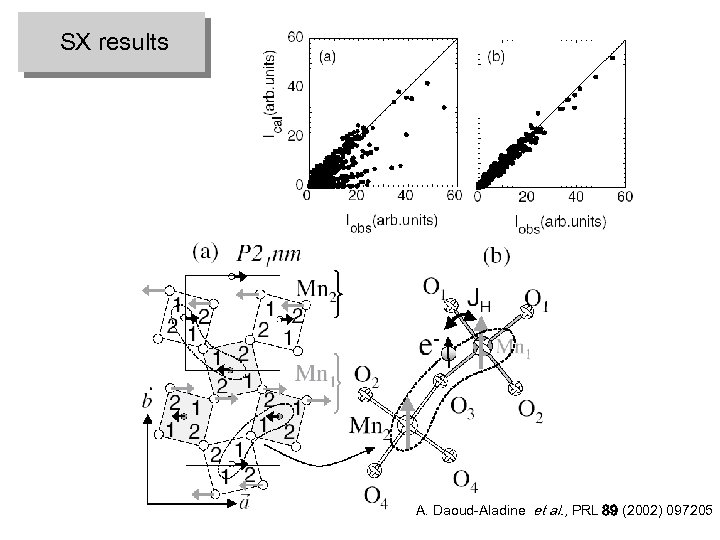 SX results A. Daoud-Aladine et al. , PRL 89 (2002) 097205
SX results A. Daoud-Aladine et al. , PRL 89 (2002) 097205
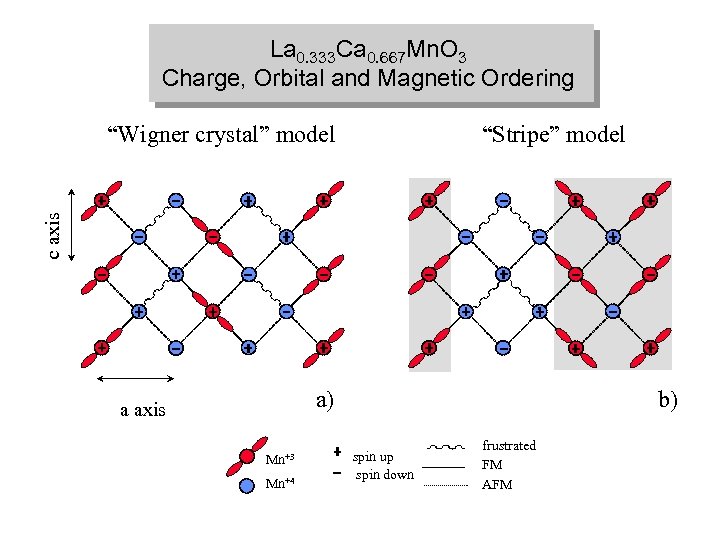 La 0. 333 Ca 0. 667 Mn. O 3 Charge, Orbital and Magnetic Ordering “Stripe” model c axis “Wigner crystal” model a) a axis Mn+3 Mn+4 b) spin up spin down frustrated FM AFM
La 0. 333 Ca 0. 667 Mn. O 3 Charge, Orbital and Magnetic Ordering “Stripe” model c axis “Wigner crystal” model a) a axis Mn+3 Mn+4 b) spin up spin down frustrated FM AFM
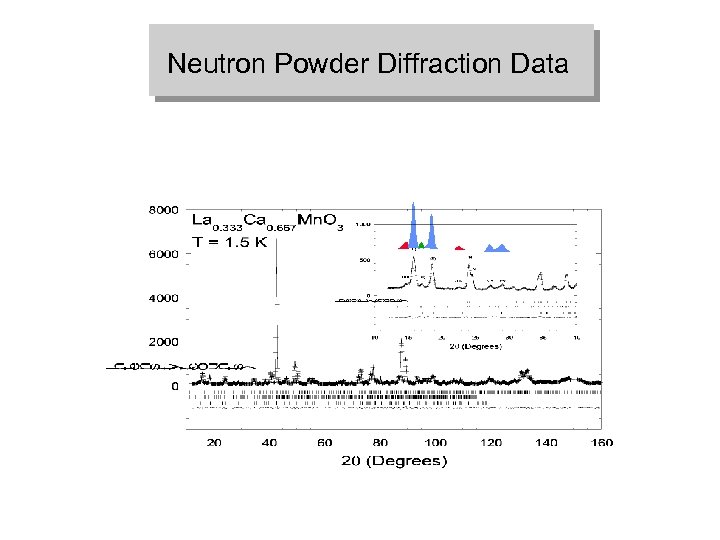 Neutron Powder Diffraction Data
Neutron Powder Diffraction Data
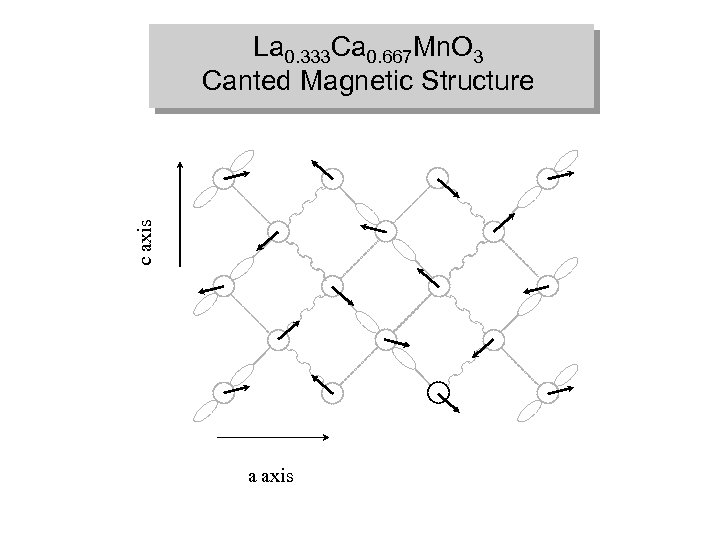 c axis La 0. 333 Ca 0. 667 Mn. O 3 Canted Magnetic Structure a axis
c axis La 0. 333 Ca 0. 667 Mn. O 3 Canted Magnetic Structure a axis
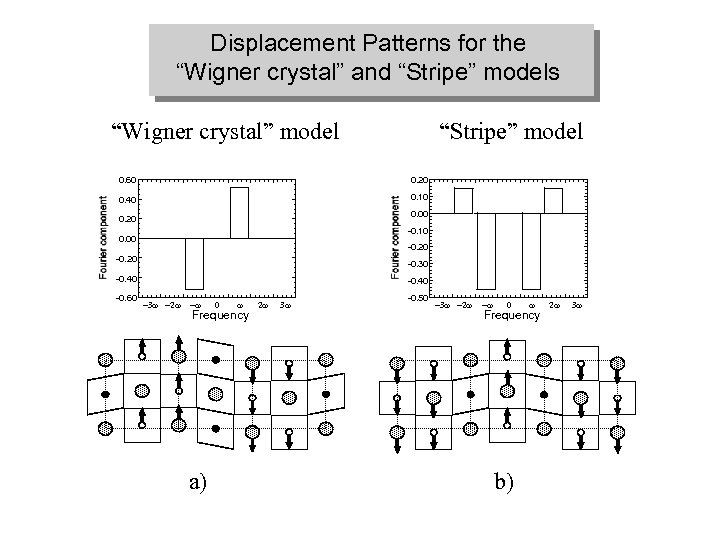 Displacement Patterns for the “Wigner crystal” and “Stripe” models “Wigner crystal” model “Stripe” model 0. 60 0. 20 0. 40 0. 10 0. 00 0. 20 -0. 10 0. 00 -0. 20 -0. 30 -0. 40 -0. 60 -0. 40 -3 w -2 w -w 0 w Frequency a) 2 w 3 w -0. 50 -3 w -2 w -w 0 w Frequency b) 2 w 3 w
Displacement Patterns for the “Wigner crystal” and “Stripe” models “Wigner crystal” model “Stripe” model 0. 60 0. 20 0. 40 0. 10 0. 00 0. 20 -0. 10 0. 00 -0. 20 -0. 30 -0. 40 -0. 60 -0. 40 -3 w -2 w -w 0 w Frequency a) 2 w 3 w -0. 50 -3 w -2 w -w 0 w Frequency b) 2 w 3 w
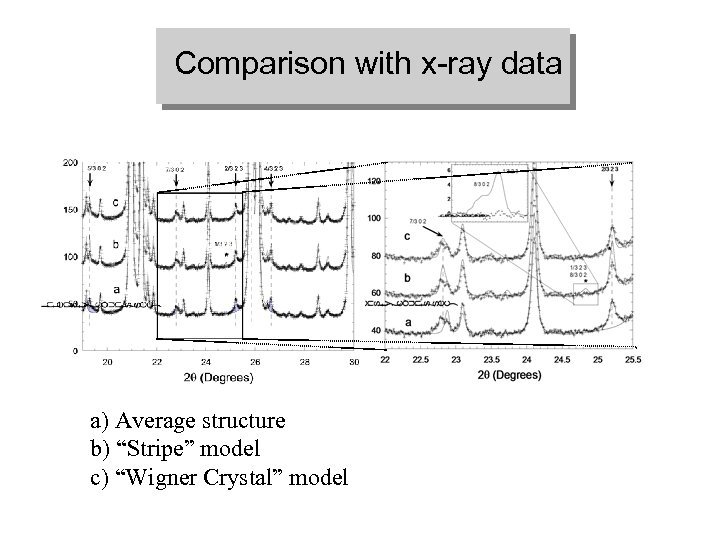 Comparison with x-ray data a) Average structure b) “Stripe” model c) “Wigner Crystal” model
Comparison with x-ray data a) Average structure b) “Stripe” model c) “Wigner Crystal” model
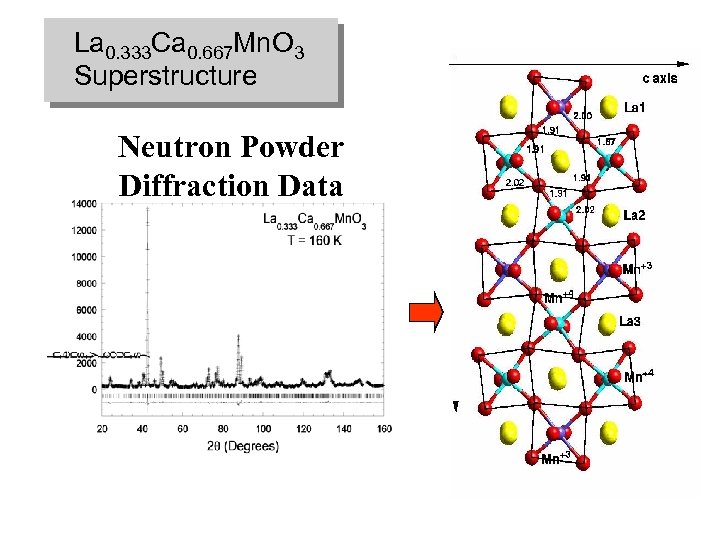 La 0. 333 Ca 0. 667 Mn. O 3 Superstructure Neutron Powder Diffraction Data
La 0. 333 Ca 0. 667 Mn. O 3 Superstructure Neutron Powder Diffraction Data
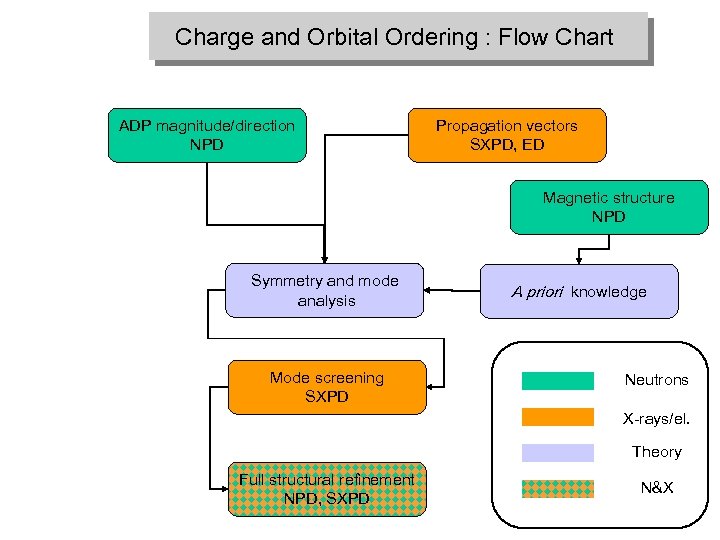 Charge and Orbital Ordering : Flow Chart ADP magnitude/direction NPD Propagation vectors SXPD, ED Magnetic structure NPD Symmetry and mode analysis Mode screening SXPD A priori knowledge Neutrons X-rays/el. Theory Full structural refinement NPD, SXPD N&X
Charge and Orbital Ordering : Flow Chart ADP magnitude/direction NPD Propagation vectors SXPD, ED Magnetic structure NPD Symmetry and mode analysis Mode screening SXPD A priori knowledge Neutrons X-rays/el. Theory Full structural refinement NPD, SXPD N&X
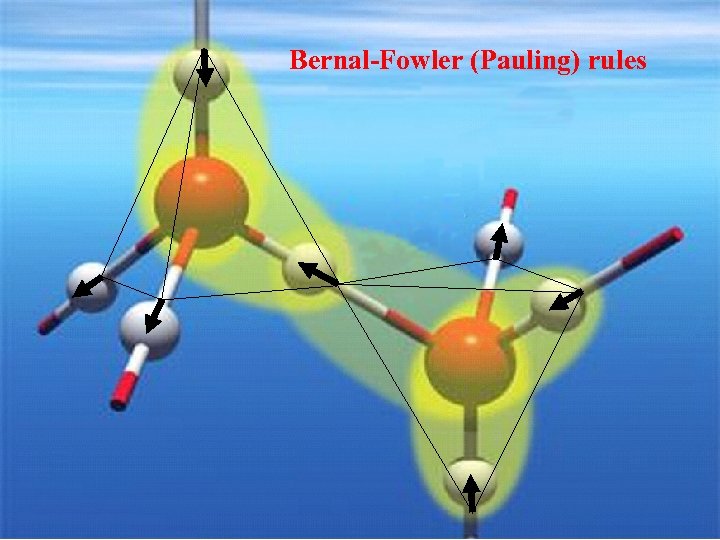 Bernal-Fowler (Pauling) rules
Bernal-Fowler (Pauling) rules
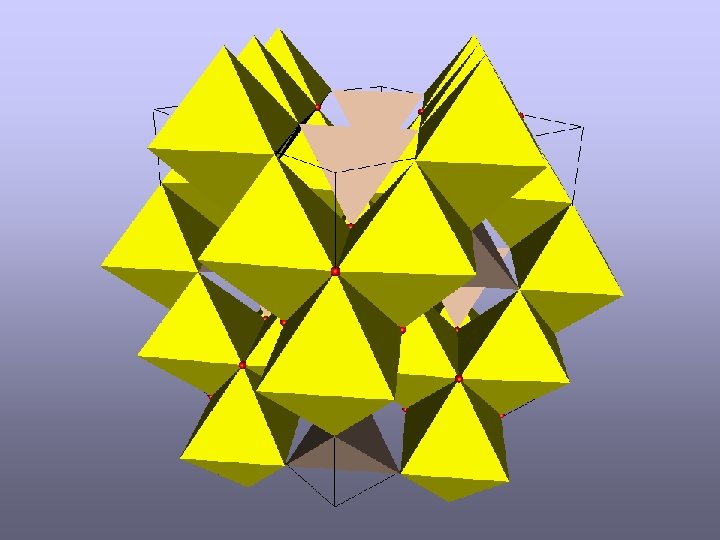
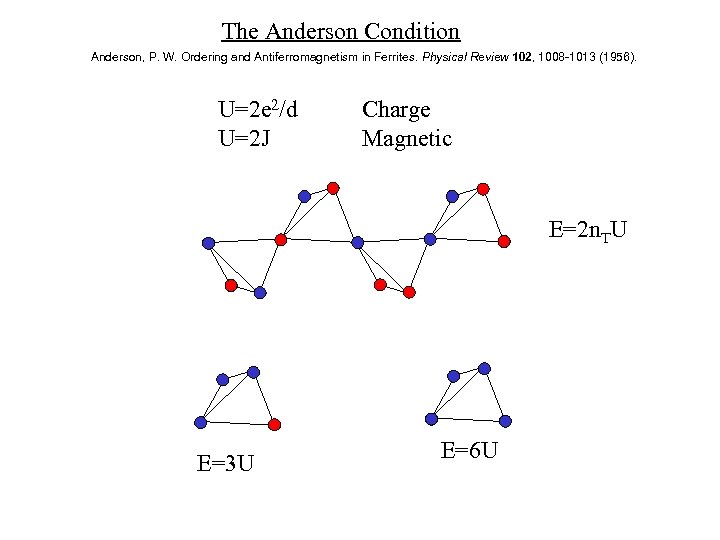 The Anderson Condition Anderson, P. W. Ordering and Antiferromagnetism in Ferrites. Physical Review 102, 1008 -1013 (1956). U=2 e 2/d U=2 J Charge Magnetic E=2 n. TU E=3 U E=6 U
The Anderson Condition Anderson, P. W. Ordering and Antiferromagnetism in Ferrites. Physical Review 102, 1008 -1013 (1956). U=2 e 2/d U=2 J Charge Magnetic E=2 n. TU E=3 U E=6 U
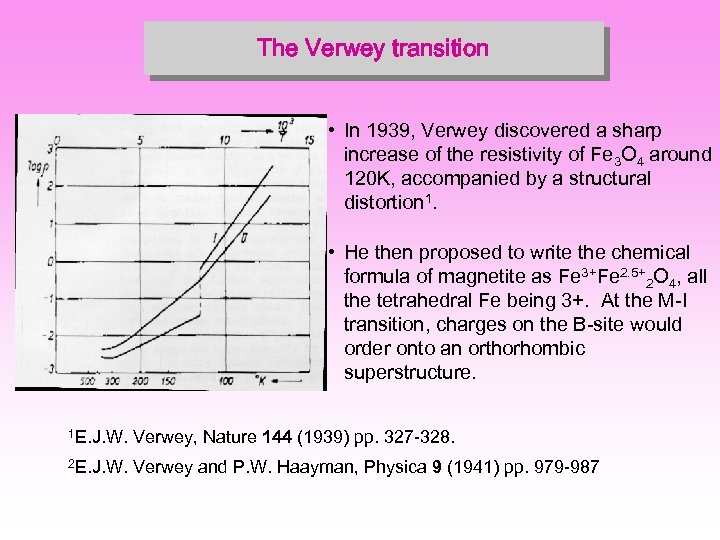 The Verwey transition • In 1939, Verwey discovered a sharp increase of the resistivity of Fe 3 O 4 around 120 K, accompanied by a structural distortion 1. • He then proposed to write the chemical formula of magnetite as Fe 3+Fe 2. 5+2 O 4, all the tetrahedral Fe being 3+. At the M-I transition, charges on the B-site would order onto an orthorhombic superstructure. 1 E. J. W. Verwey, Nature 144 (1939) pp. 327 -328. 2 E. J. W. Verwey and P. W. Haayman, Physica 9 (1941) pp. 979 -987
The Verwey transition • In 1939, Verwey discovered a sharp increase of the resistivity of Fe 3 O 4 around 120 K, accompanied by a structural distortion 1. • He then proposed to write the chemical formula of magnetite as Fe 3+Fe 2. 5+2 O 4, all the tetrahedral Fe being 3+. At the M-I transition, charges on the B-site would order onto an orthorhombic superstructure. 1 E. J. W. Verwey, Nature 144 (1939) pp. 327 -328. 2 E. J. W. Verwey and P. W. Haayman, Physica 9 (1941) pp. 979 -987
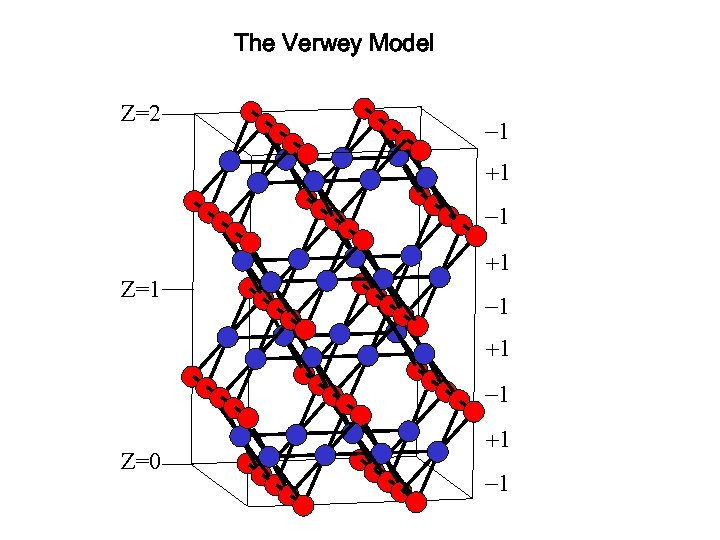 The Verwey Model Z=2 -1 +1 Z=1 -1 +1 -1 Z=0 +1 -1
The Verwey Model Z=2 -1 +1 Z=1 -1 +1 -1 Z=0 +1 -1
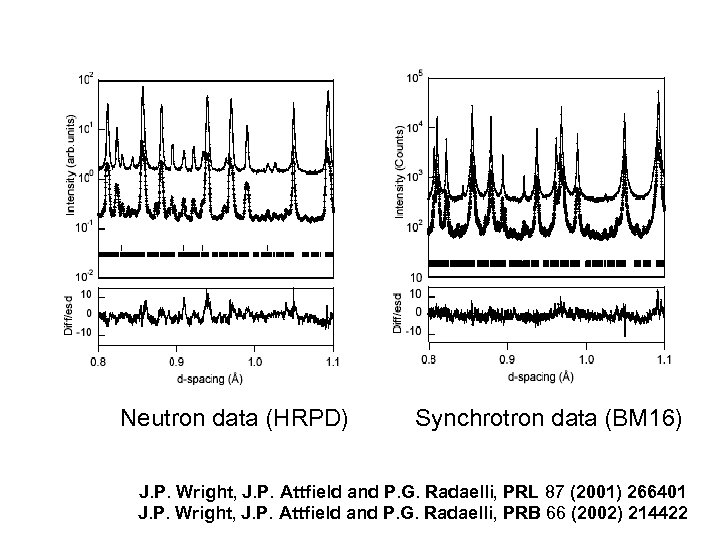 Neutron data (HRPD) Synchrotron data (BM 16) J. P. Wright, J. P. Attfield and P. G. Radaelli, PRL 87 (2001) 266401 J. P. Wright, J. P. Attfield and P. G. Radaelli, PRB 66 (2002) 214422
Neutron data (HRPD) Synchrotron data (BM 16) J. P. Wright, J. P. Attfield and P. G. Radaelli, PRL 87 (2001) 266401 J. P. Wright, J. P. Attfield and P. G. Radaelli, PRB 66 (2002) 214422
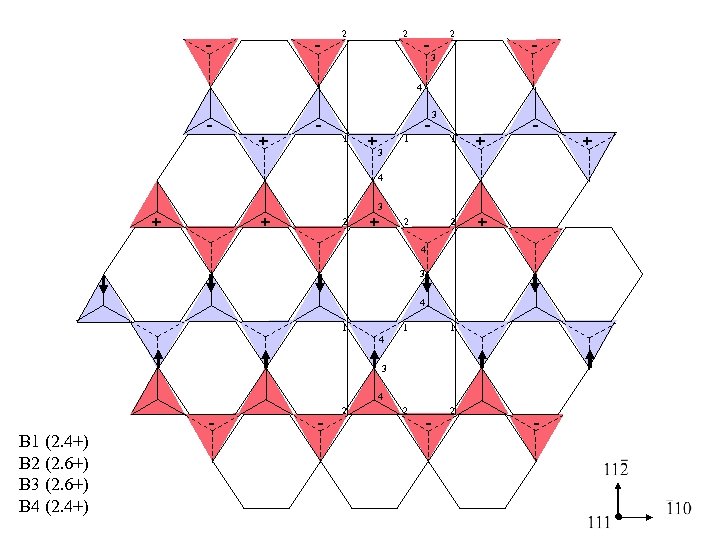 - - 2 2 -3 2 - 4 - + - 1 +3 - 3 1 + 2 1 - + 4 + 3 + 2 4 3 4 1 1 1 2 2 4 3 4 B 1 (2. 4+) B 2 (2. 6+) B 3 (2. 6+) B 4 (2. 4+) - - 2 - - +
- - 2 2 -3 2 - 4 - + - 1 +3 - 3 1 + 2 1 - + 4 + 3 + 2 4 3 4 1 1 1 2 2 4 3 4 B 1 (2. 4+) B 2 (2. 6+) B 3 (2. 6+) B 4 (2. 4+) - - 2 - - +
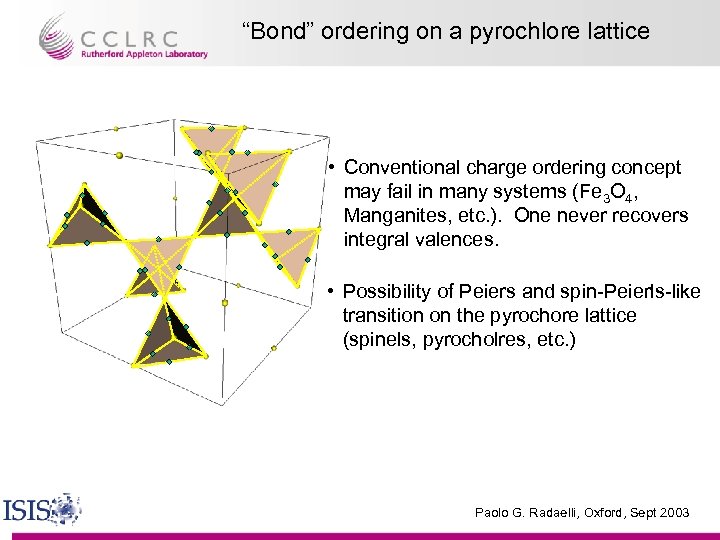 “Bond” ordering on a pyrochlore lattice • Conventional charge ordering concept may fail in many systems (Fe 3 O 4, Manganites, etc. ). One never recovers integral valences. • Possibility of Peiers and spin-Peierls-like transition on the pyrochore lattice (spinels, pyrocholres, etc. ) Paolo G. Radaelli, Oxford, Sept 2003
“Bond” ordering on a pyrochlore lattice • Conventional charge ordering concept may fail in many systems (Fe 3 O 4, Manganites, etc. ). One never recovers integral valences. • Possibility of Peiers and spin-Peierls-like transition on the pyrochore lattice (spinels, pyrocholres, etc. ) Paolo G. Radaelli, Oxford, Sept 2003
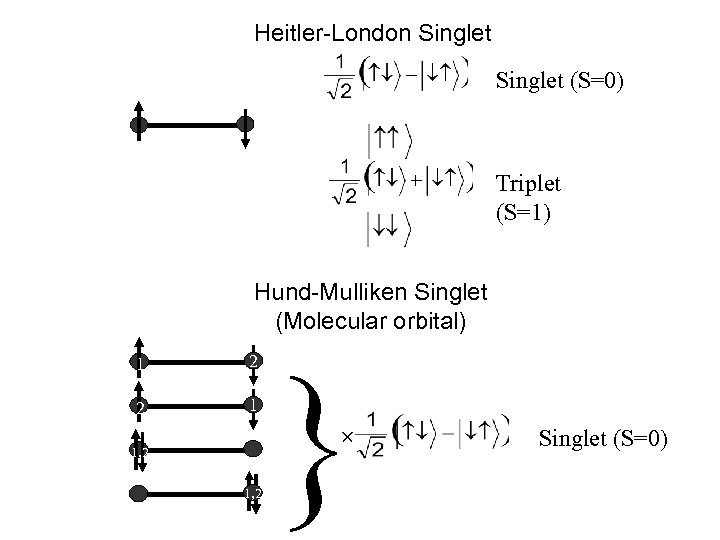 Heitler-London Singlet (S=0) Triplet (S=1) Hund-Mulliken Singlet (Molecular orbital) 1 2 2 1 } 1, 2 Singlet (S=0)
Heitler-London Singlet (S=0) Triplet (S=1) Hund-Mulliken Singlet (Molecular orbital) 1 2 2 1 } 1, 2 Singlet (S=0)
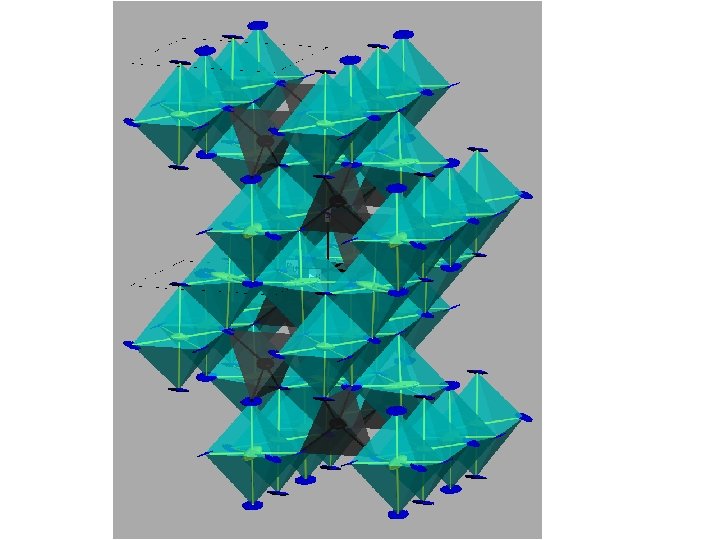
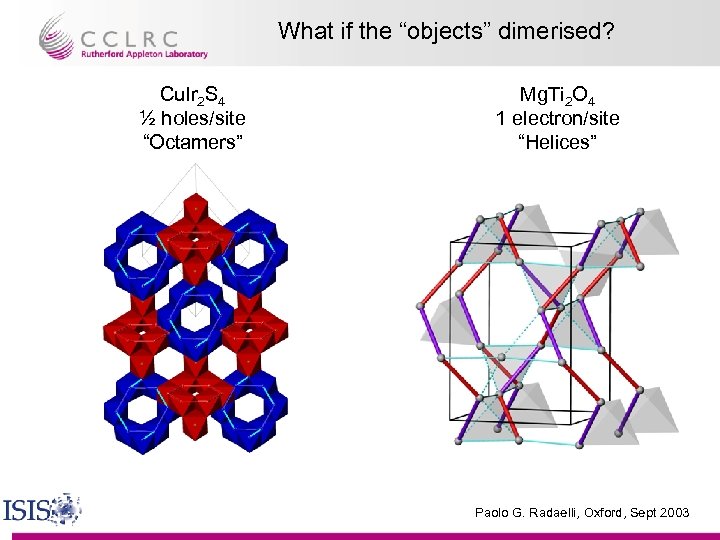 What if the “objects” dimerised? Cu. Ir 2 S 4 ½ holes/site “Octamers” Mg. Ti 2 O 4 1 electron/site “Helices” Paolo G. Radaelli, Oxford, Sept 2003
What if the “objects” dimerised? Cu. Ir 2 S 4 ½ holes/site “Octamers” Mg. Ti 2 O 4 1 electron/site “Helices” Paolo G. Radaelli, Oxford, Sept 2003
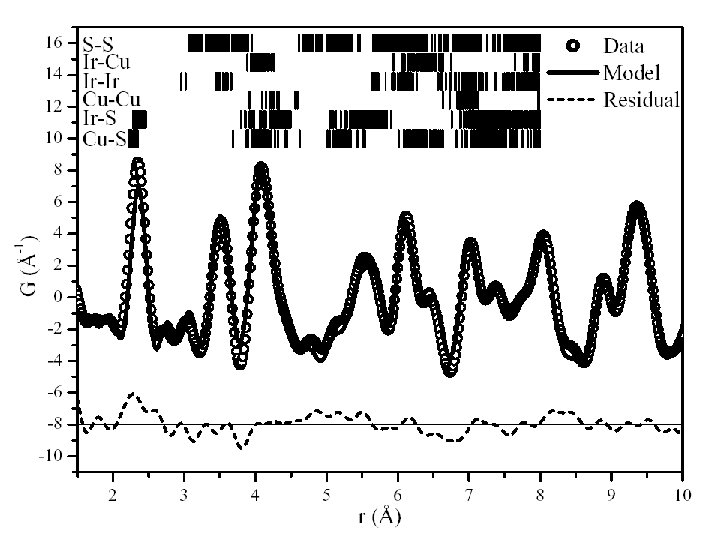
 Cu. Ir 2 S 4 • Y. Koyama ‡ • Yew-San Hor§ • Valery Kiryukhin§ • Hiroki Ishibashi§|| • Sang-Wook Cheong§ Sample synthesis, transport meas. synchrotron data • Y. Horibe†‡§ • C. H. Chen† Electron Microscopy • Matthias J. Gutmann* • Richard M. Ibberson* Neutron diffraction PDF analysis * ISIS Facility, Rutherford Appleton Laboratory † Bell Laboratories, Lucent Technologies, Murray Hill, New Jersey 07974, USA ‡ Department of Materials Science and Engineering, Waseda University, Japan § Department of Physics and Astronomy, Rutgers University New Jersey, USA. || Department of Materials Science, Osaka Prefecture University, Osaka, Japan. Paolo G. Radaelli, Oxford, Sept 2003
Cu. Ir 2 S 4 • Y. Koyama ‡ • Yew-San Hor§ • Valery Kiryukhin§ • Hiroki Ishibashi§|| • Sang-Wook Cheong§ Sample synthesis, transport meas. synchrotron data • Y. Horibe†‡§ • C. H. Chen† Electron Microscopy • Matthias J. Gutmann* • Richard M. Ibberson* Neutron diffraction PDF analysis * ISIS Facility, Rutherford Appleton Laboratory † Bell Laboratories, Lucent Technologies, Murray Hill, New Jersey 07974, USA ‡ Department of Materials Science and Engineering, Waseda University, Japan § Department of Physics and Astronomy, Rutgers University New Jersey, USA. || Department of Materials Science, Osaka Prefecture University, Osaka, Japan. Paolo G. Radaelli, Oxford, Sept 2003
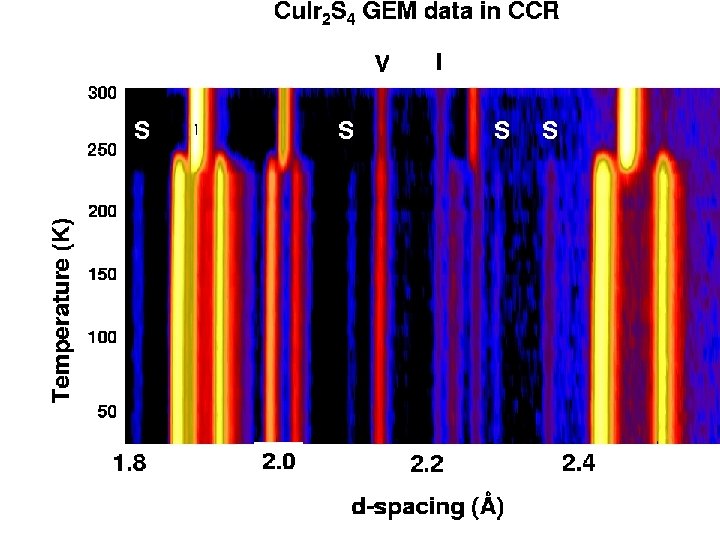
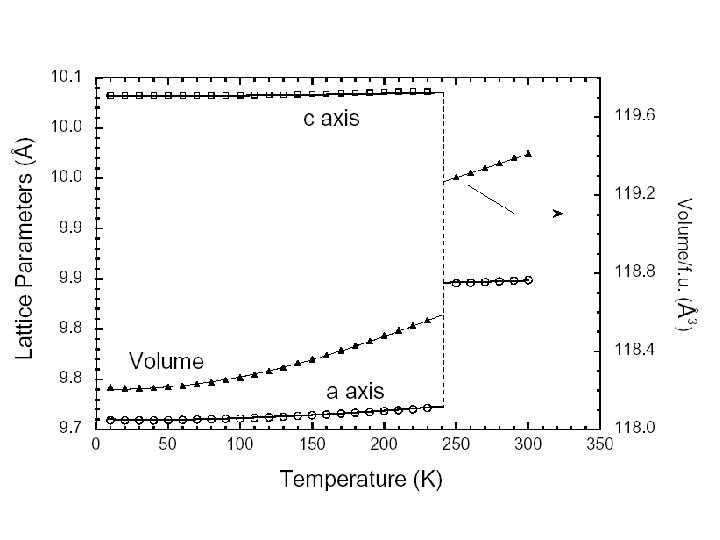
 Cu. Ir 2 S 4: Charge Ordering and spin pairing. • The copper iridium thiospinel Cu. Ir 2 S 4 is a very young relative of magnetite, since it was synthesized for the first time in 19941. • It is reminiscent of Fe 3 O 4 in that it is halffilled and has a coupled M-I and structural transition (at 230 K), but with one striking difference: it undergoes a Pauli paramagnet-to-diamagnet phase transition at the same time 2. 1 S. 2 Nagata et al. , Physica B 194 -196 (1994) P. 1077. T. Furubayashi et al. , J. Phys. Soc. J 63 (1994) p. 3333. Paolo G. Radaelli, Oxford, Sept 2003
Cu. Ir 2 S 4: Charge Ordering and spin pairing. • The copper iridium thiospinel Cu. Ir 2 S 4 is a very young relative of magnetite, since it was synthesized for the first time in 19941. • It is reminiscent of Fe 3 O 4 in that it is halffilled and has a coupled M-I and structural transition (at 230 K), but with one striking difference: it undergoes a Pauli paramagnet-to-diamagnet phase transition at the same time 2. 1 S. 2 Nagata et al. , Physica B 194 -196 (1994) P. 1077. T. Furubayashi et al. , J. Phys. Soc. J 63 (1994) p. 3333. Paolo G. Radaelli, Oxford, Sept 2003
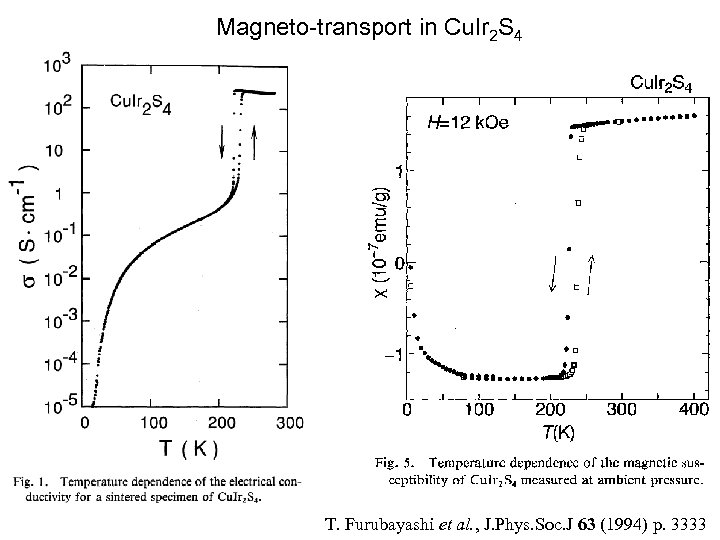 Magneto-transport in Cu. Ir 2 S 4 T. Furubayashi et al. , J. Phys. Soc. J 63 (1994) p. 3333
Magneto-transport in Cu. Ir 2 S 4 T. Furubayashi et al. , J. Phys. Soc. J 63 (1994) p. 3333
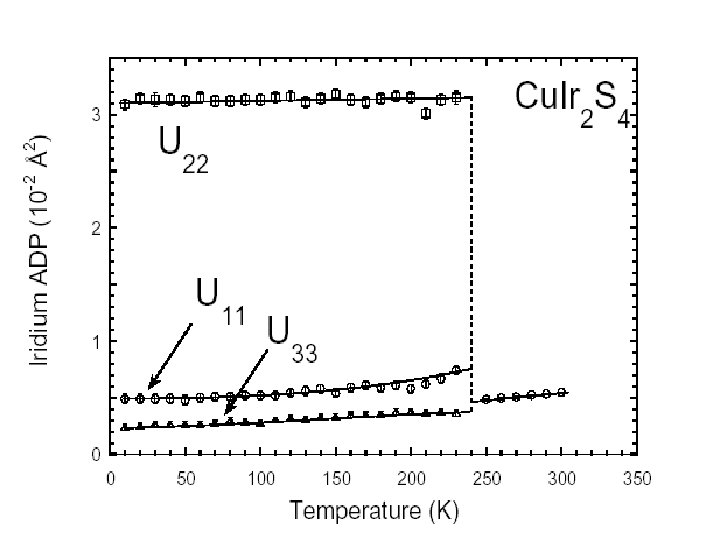
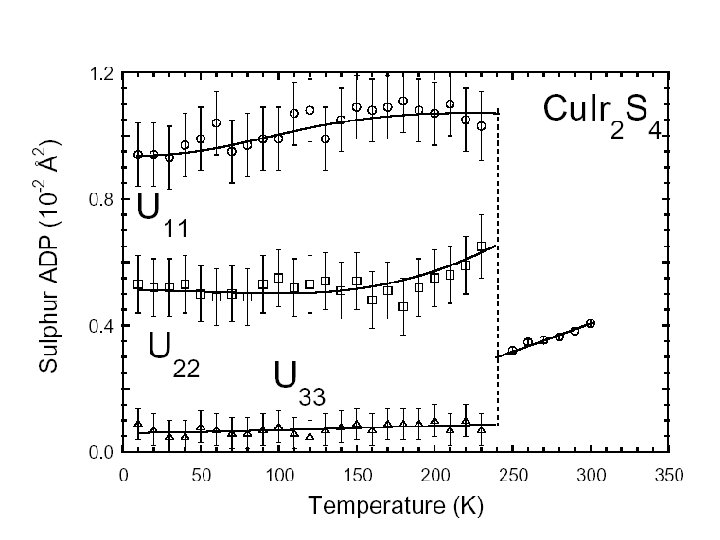
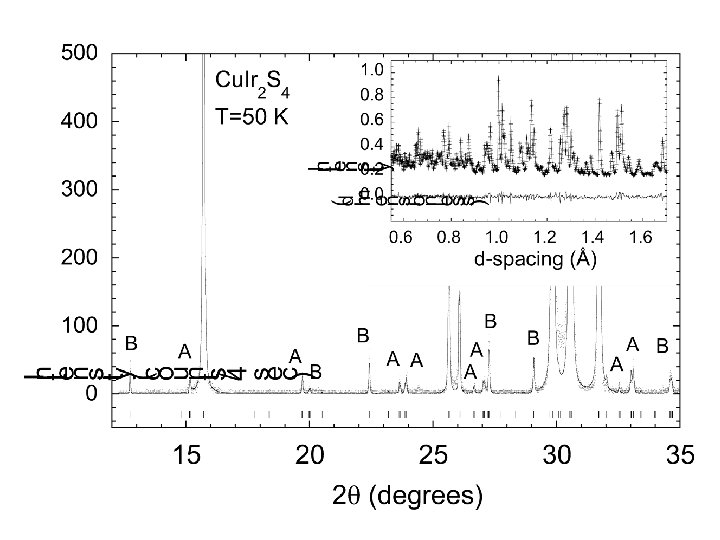
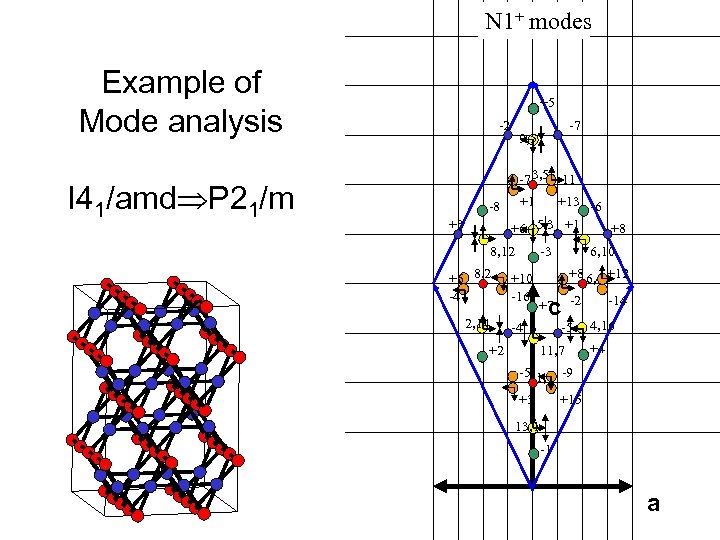 N 1+ modes Example of Mode analysis I 41/amd P 21/m +5 -2 -7 9, 5 -7 3, 5 -11 -8 +3 +1 +13 -6 +6 15, 3 +1 +8 8, 12 +6 8, 2 -4 2, 14 -3 +10 -16 6, 10 +8 6, 4 +12 c +7 -4 +2 -2 -5 11, 7 -14 4, 16 +4 -5 1, 7 -9 +3 +15 13, 1 -1 a
N 1+ modes Example of Mode analysis I 41/amd P 21/m +5 -2 -7 9, 5 -7 3, 5 -11 -8 +3 +1 +13 -6 +6 15, 3 +1 +8 8, 12 +6 8, 2 -4 2, 14 -3 +10 -16 6, 10 +8 6, 4 +12 c +7 -4 +2 -2 -5 11, 7 -14 4, 16 +4 -5 1, 7 -9 +3 +15 13, 1 -1 a
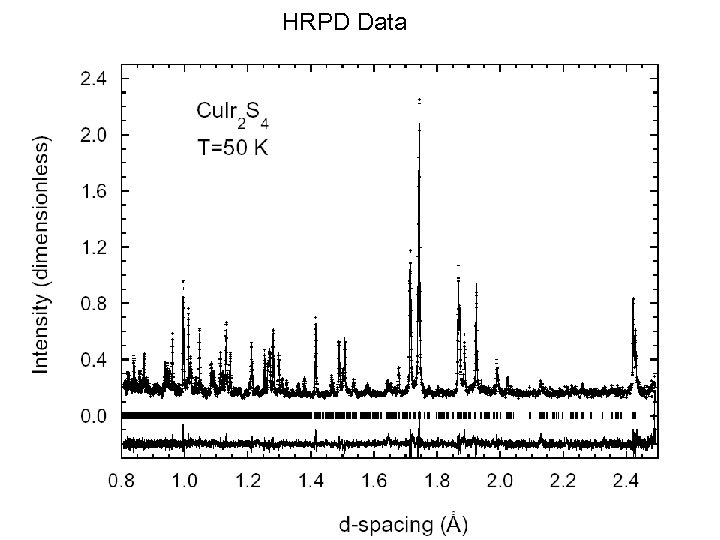 HRPD Data
HRPD Data
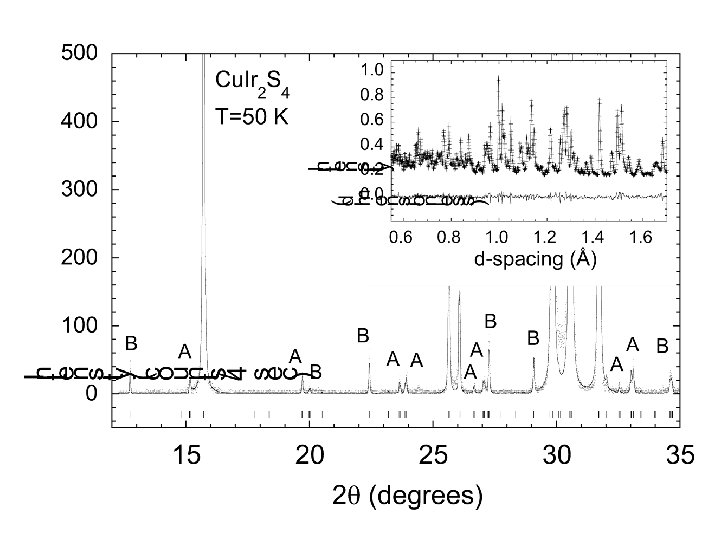
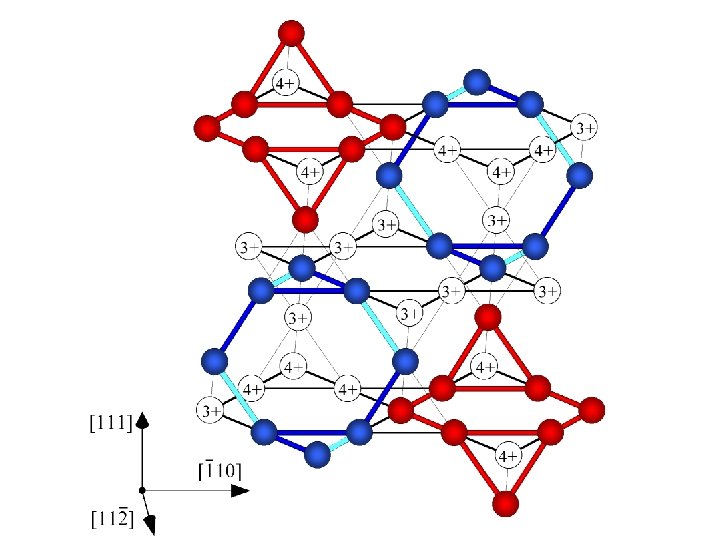
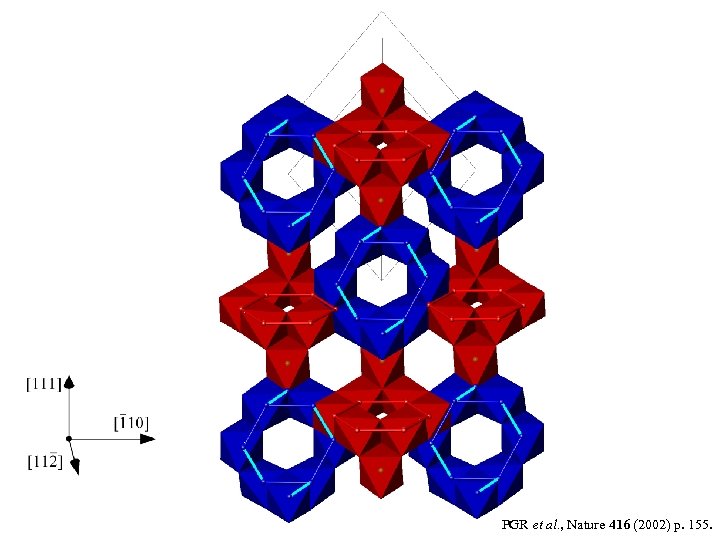 PGR et al. , Nature 416 (2002) p. 155.
PGR et al. , Nature 416 (2002) p. 155.
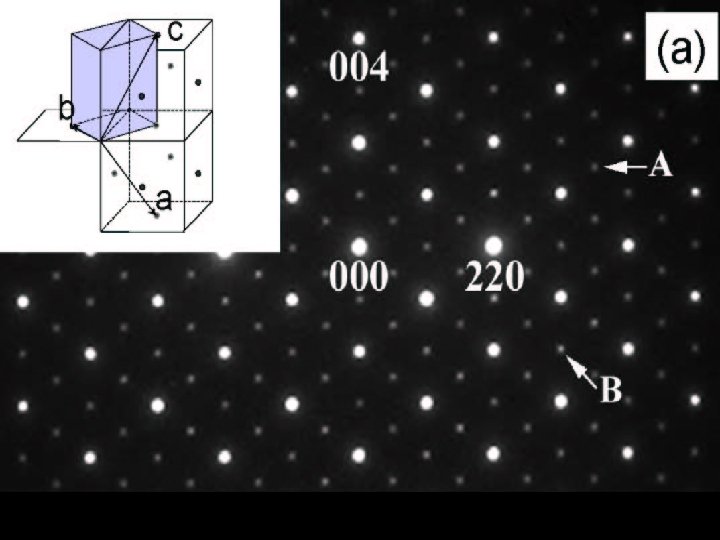
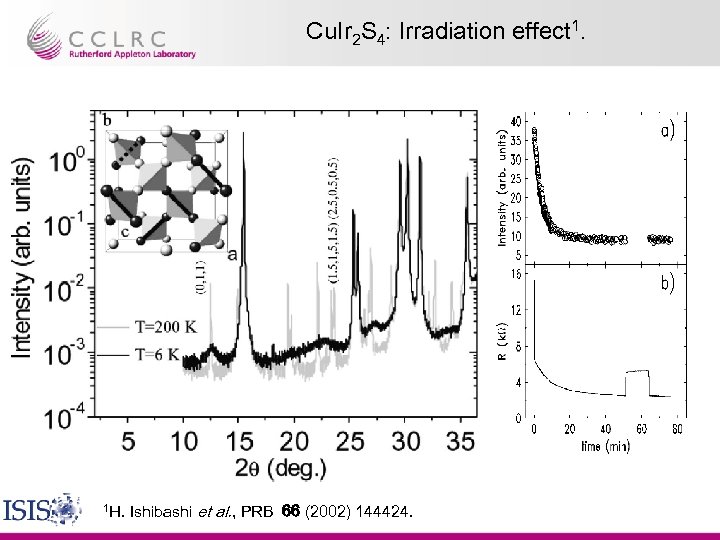 Cu. Ir 2 S 4: Irradiation effect 1. 1 H. Ishibashi et al. , PRB 66 (2002) 144424.
Cu. Ir 2 S 4: Irradiation effect 1. 1 H. Ishibashi et al. , PRB 66 (2002) 144424.
 Cu. Ir 2 S 4: Conclusions. • The crystal structure of Cu. Ir 2 S 4 at low temperatures accounts well for the observed transport and magnetic properties. Band structure calculations indicate HM rather than HL singlets (a Peierls rather than a spin-Peierls transition). • The ‘Octamer-based’ description of the spinel crystal structure is simple and elegant. Yet, the underlying CO structure represents a step up in complexity with respect to all previously known arrangements. • X-ray irradiation destroys the octamers but preserves the dimers. Paolo G. Radaelli, Oxford, Sept 2003
Cu. Ir 2 S 4: Conclusions. • The crystal structure of Cu. Ir 2 S 4 at low temperatures accounts well for the observed transport and magnetic properties. Band structure calculations indicate HM rather than HL singlets (a Peierls rather than a spin-Peierls transition). • The ‘Octamer-based’ description of the spinel crystal structure is simple and elegant. Yet, the underlying CO structure represents a step up in complexity with respect to all previously known arrangements. • X-ray irradiation destroys the octamers but preserves the dimers. Paolo G. Radaelli, Oxford, Sept 2003
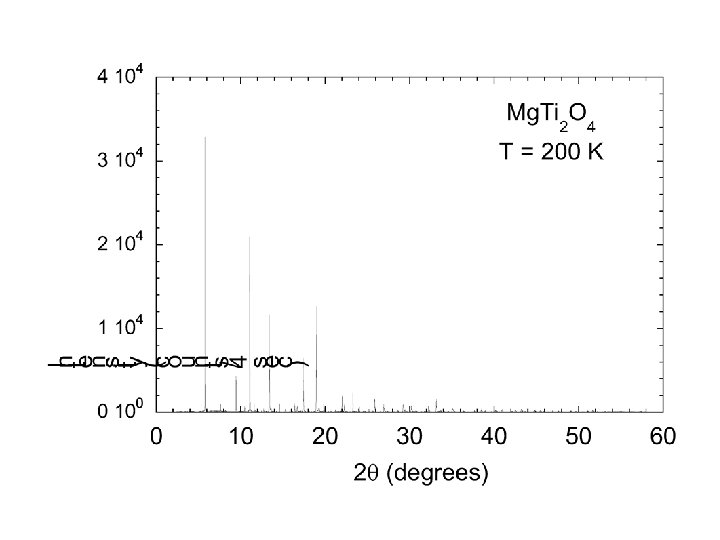
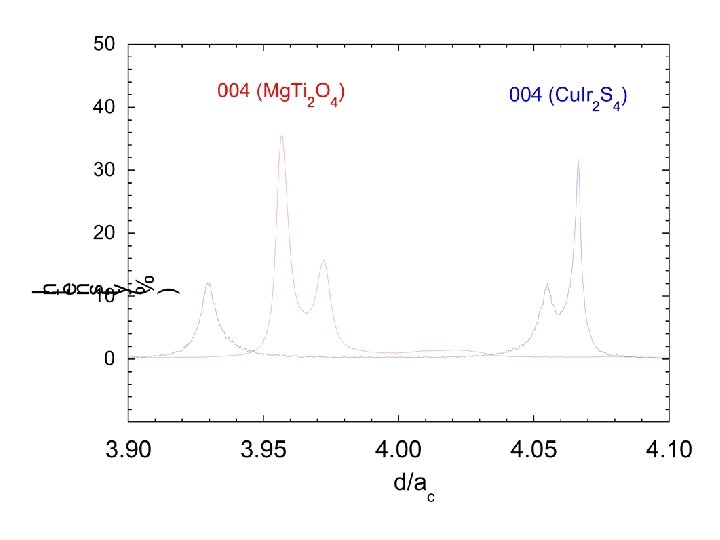
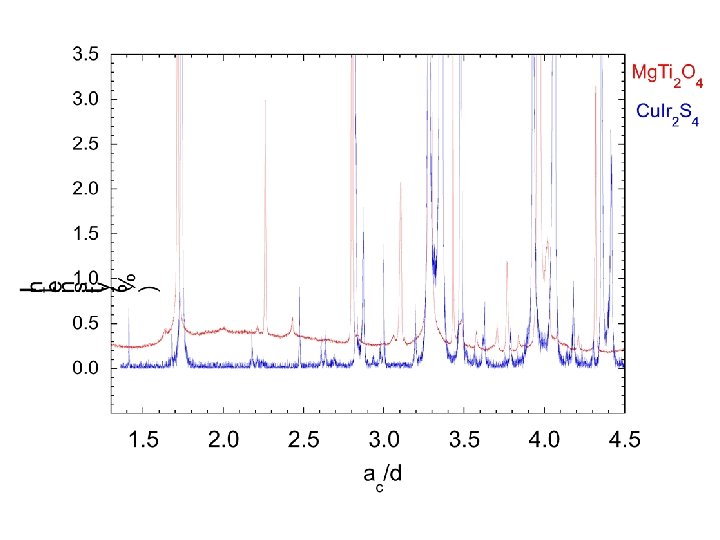
 Mg. Ti 2 O 4 • William Ratcliff II § • Sang-Wook Cheong§ Sample synthesis, transport meas. • Marek Schmidt¶* Synchrotron/Neutron diffraction • Keith Refson* • Nic Harrison ‡|| Band structure calculations * ISIS Facility, Rutherford Appleton Laboratory ¶Dept. of physics and Astronomy, Michigan State University † Bell Laboratories, Lucent Technologies, Murray Hill, New Jersey 07974, USA § Department of Physics and Astronomy, Rutgers University New Jersey, USA. ‡ Department of Chemistry, Imperial College London || CCLRC, Daresbury Laboratory. Paolo G. Radaelli, Oxford, Sept 2003
Mg. Ti 2 O 4 • William Ratcliff II § • Sang-Wook Cheong§ Sample synthesis, transport meas. • Marek Schmidt¶* Synchrotron/Neutron diffraction • Keith Refson* • Nic Harrison ‡|| Band structure calculations * ISIS Facility, Rutherford Appleton Laboratory ¶Dept. of physics and Astronomy, Michigan State University † Bell Laboratories, Lucent Technologies, Murray Hill, New Jersey 07974, USA § Department of Physics and Astronomy, Rutgers University New Jersey, USA. ‡ Department of Chemistry, Imperial College London || CCLRC, Daresbury Laboratory. Paolo G. Radaelli, Oxford, Sept 2003
 Mg. Ti 2 O 4: Dimerisation with a “twist”. • Mg. Ti 2 O 4 is another spinel with related phenomenology. Ti is in the singlevalence state 3+ (3 d 1, S=1/2) 1. • Similar to Cu. Ir 2 S 4, Mg. Ti 2 O 4 has a coupled M-I and structural transition (at 260 K), which is also accompanied by a strong reduction of the magnetic susceptibility and a structural transition 1. 1 M. Isobe and Y. Ueda, J. Phys. Soc. Japan 71 (2002) p. 1848. Paolo G. Radaelli, Oxford, Sept 2003
Mg. Ti 2 O 4: Dimerisation with a “twist”. • Mg. Ti 2 O 4 is another spinel with related phenomenology. Ti is in the singlevalence state 3+ (3 d 1, S=1/2) 1. • Similar to Cu. Ir 2 S 4, Mg. Ti 2 O 4 has a coupled M-I and structural transition (at 260 K), which is also accompanied by a strong reduction of the magnetic susceptibility and a structural transition 1. 1 M. Isobe and Y. Ueda, J. Phys. Soc. Japan 71 (2002) p. 1848. Paolo G. Radaelli, Oxford, Sept 2003
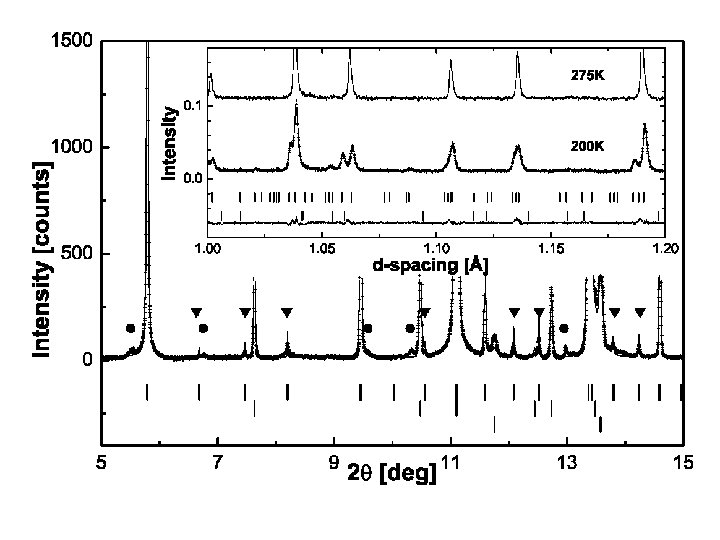
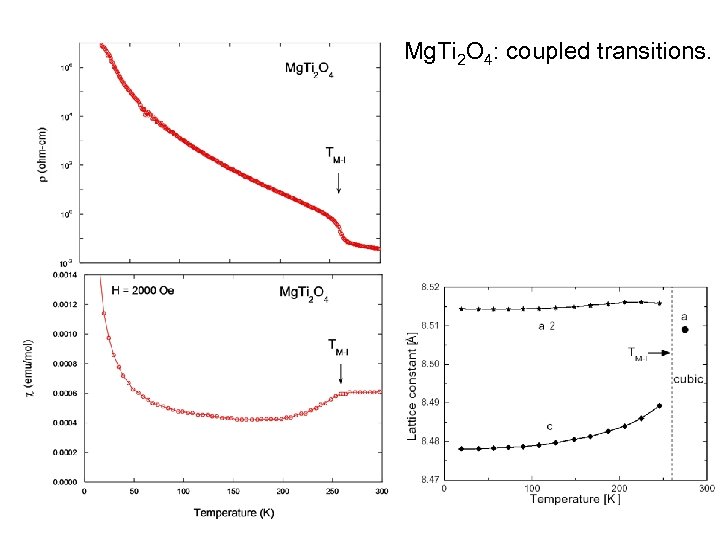 Mg. Ti 2 O 4: coupled transitions.
Mg. Ti 2 O 4: coupled transitions.
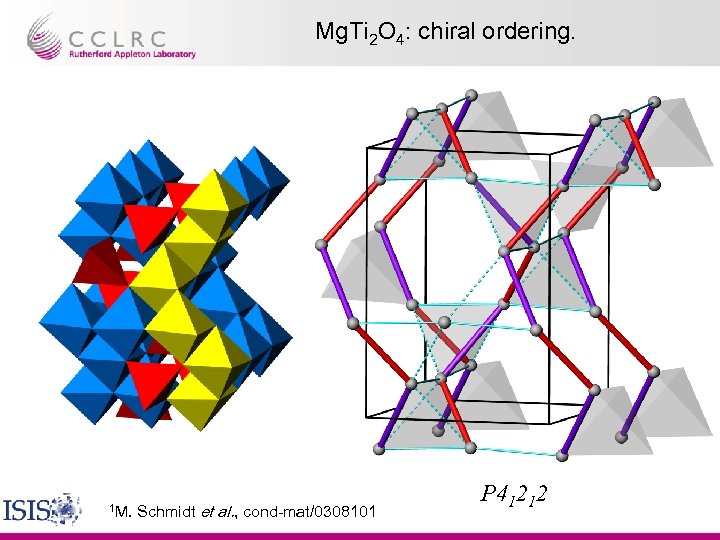 Mg. Ti 2 O 4: chiral ordering. 1 M. Schmidt et al. , cond-mat/0308101 P 41212
Mg. Ti 2 O 4: chiral ordering. 1 M. Schmidt et al. , cond-mat/0308101 P 41212
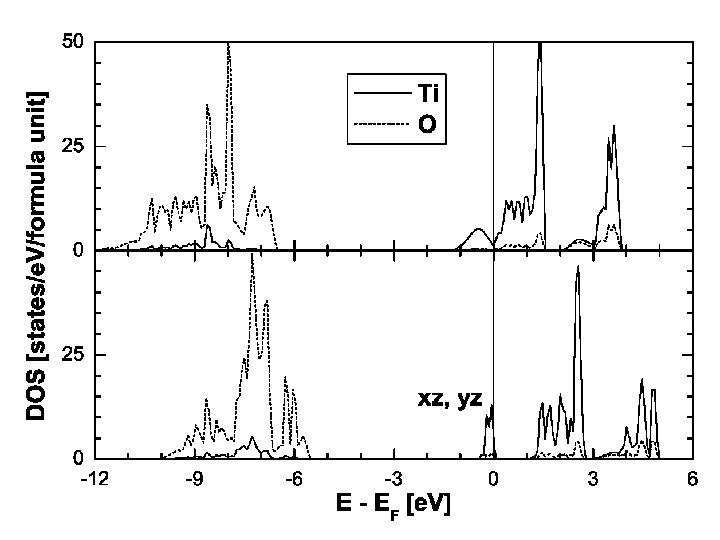
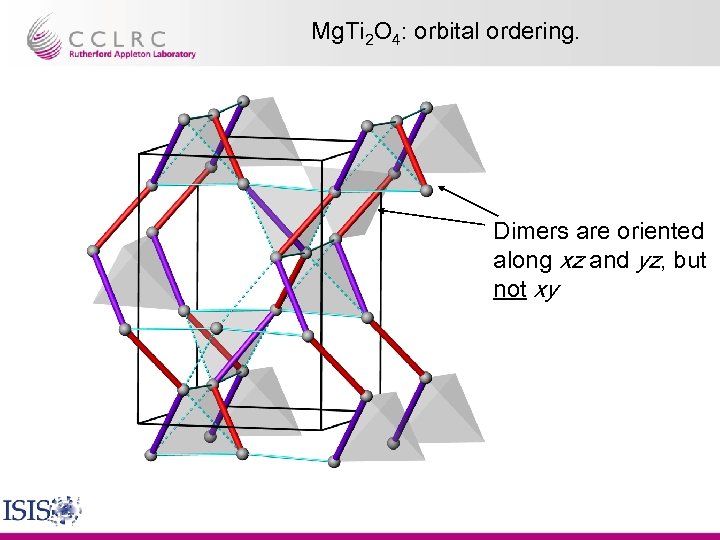 Mg. Ti 2 O 4: orbital ordering. Dimers are oriented along xz and yz, but not xy
Mg. Ti 2 O 4: orbital ordering. Dimers are oriented along xz and yz, but not xy
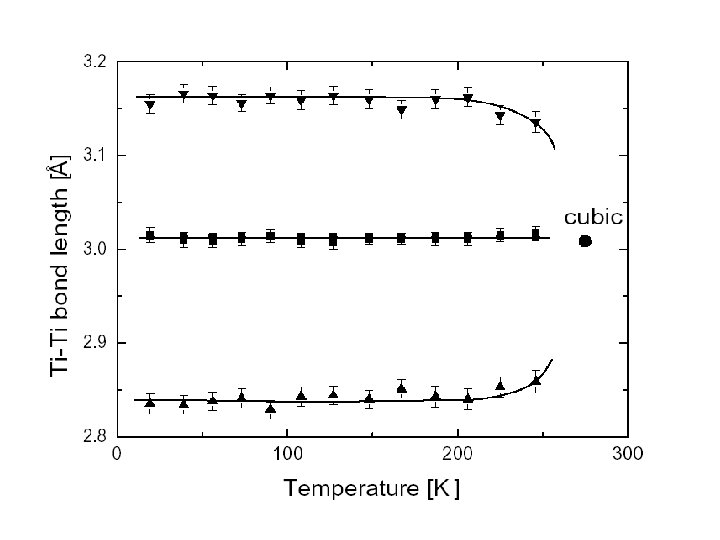
 Mg. Ti 2 O 4: Conclusions. • At low temperatures, the Ti atoms dimerise and Mg. Ti 2 O 4 distorts to a chiral space group. The chiral structural elements are “helices” of …-long-shortlong…. Ti-Ti bonds. • This results in xz/yz orbital ordering, the formation of HM singlets and the opening of a gap in the electronic DOS, accounting well for the observed properties. • This can be thought of as ordering of the dimers on a network of isolated octahedra at 1/6 filling, leading to a different type of degeneracy with respect to the original frustrated lattice. Paolo G. Radaelli, Oxford, Sept 2003
Mg. Ti 2 O 4: Conclusions. • At low temperatures, the Ti atoms dimerise and Mg. Ti 2 O 4 distorts to a chiral space group. The chiral structural elements are “helices” of …-long-shortlong…. Ti-Ti bonds. • This results in xz/yz orbital ordering, the formation of HM singlets and the opening of a gap in the electronic DOS, accounting well for the observed properties. • This can be thought of as ordering of the dimers on a network of isolated octahedra at 1/6 filling, leading to a different type of degeneracy with respect to the original frustrated lattice. Paolo G. Radaelli, Oxford, Sept 2003


