e5cb64dbe73fdac00dee2a8a43ca5e81.ppt
- Количество слайдов: 77
 Opioid Pharmacology and Management Principles Part I Steven Stanos, DO © 2005 Rehabilitation Institute of Chicago
Opioid Pharmacology and Management Principles Part I Steven Stanos, DO © 2005 Rehabilitation Institute of Chicago
 © 2005 Rehabilitation Institute of Chicago
© 2005 Rehabilitation Institute of Chicago
 “Opiate” “Opioid” “Narcotic” © 2005 Rehabilitation Institute of Chicago
“Opiate” “Opioid” “Narcotic” © 2005 Rehabilitation Institute of Chicago
 © 2005 Rehabilitation Institute of Chicago
© 2005 Rehabilitation Institute of Chicago
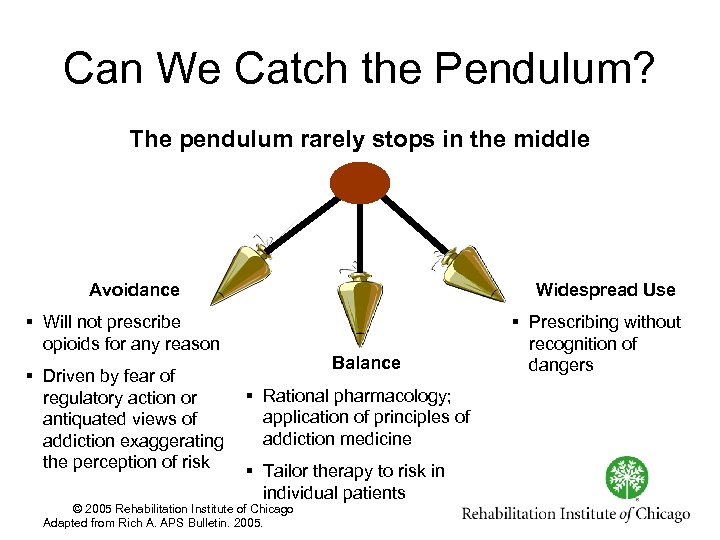 Can We Catch the Pendulum? The pendulum rarely stops in the middle Avoidance Widespread Use § Will not prescribe opioids for any reason § Driven by fear of regulatory action or antiquated views of addiction exaggerating the perception of risk Balance § Rational pharmacology; application of principles of addiction medicine § Tailor therapy to risk in individual patients © 2005 Rehabilitation Institute of Chicago Adapted from Rich A. APS Bulletin. 2005. § Prescribing without recognition of dangers
Can We Catch the Pendulum? The pendulum rarely stops in the middle Avoidance Widespread Use § Will not prescribe opioids for any reason § Driven by fear of regulatory action or antiquated views of addiction exaggerating the perception of risk Balance § Rational pharmacology; application of principles of addiction medicine § Tailor therapy to risk in individual patients © 2005 Rehabilitation Institute of Chicago Adapted from Rich A. APS Bulletin. 2005. § Prescribing without recognition of dangers
 Holy Grail ? • Potent analgesic • More less potent agents • Drugs for reduced renal function • Better formulations • Less divertible © 2005 Rehabilitation Institute of Chicago
Holy Grail ? • Potent analgesic • More less potent agents • Drugs for reduced renal function • Better formulations • Less divertible © 2005 Rehabilitation Institute of Chicago
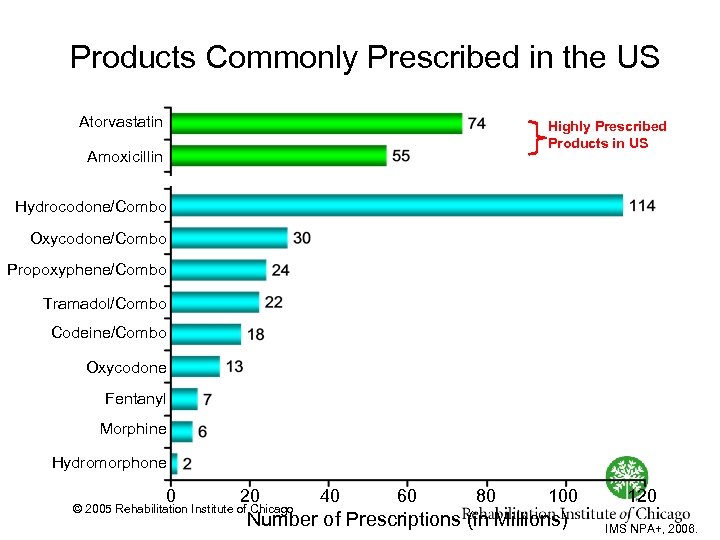 Products Commonly Prescribed in the US Atorvastatin Highly Prescribed Products in US Amoxicillin Hydrocodone/Combo Oxycodone/Combo Propoxyphene/Combo Tramadol/Combo Codeine/Combo Oxycodone Fentanyl Morphine Hydromorphone 0 20 © 2005 Rehabilitation Institute of Chicago 40 60 80 100 Number of Prescriptions (in Millions) 120 IMS NPA+, 2006.
Products Commonly Prescribed in the US Atorvastatin Highly Prescribed Products in US Amoxicillin Hydrocodone/Combo Oxycodone/Combo Propoxyphene/Combo Tramadol/Combo Codeine/Combo Oxycodone Fentanyl Morphine Hydromorphone 0 20 © 2005 Rehabilitation Institute of Chicago 40 60 80 100 Number of Prescriptions (in Millions) 120 IMS NPA+, 2006.
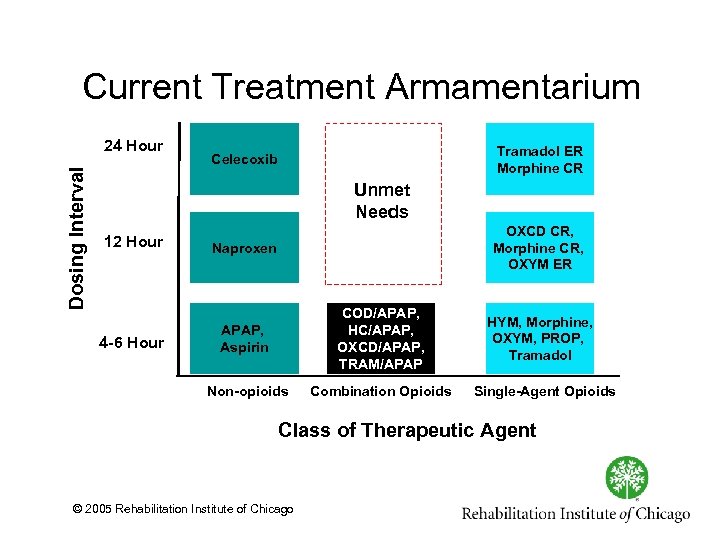 Current Treatment Armamentarium Dosing Interval 24 Hour Tramadol ER Morphine CR Celecoxib Unmet Needs 12 Hour OXCD CR, Morphine CR, OXYM ER Naproxen Non-opioids 4 -6 Hour APAP, Aspirin COD/APAP, HC/APAP, OXCD/APAP, TRAM/APAP Combination Opioids HYM, Morphine, OXYM, PROP, Tramadol Single-Agent Opioids Class of Therapeutic Agent © 2005 Rehabilitation Institute of Chicago
Current Treatment Armamentarium Dosing Interval 24 Hour Tramadol ER Morphine CR Celecoxib Unmet Needs 12 Hour OXCD CR, Morphine CR, OXYM ER Naproxen Non-opioids 4 -6 Hour APAP, Aspirin COD/APAP, HC/APAP, OXCD/APAP, TRAM/APAP Combination Opioids HYM, Morphine, OXYM, PROP, Tramadol Single-Agent Opioids Class of Therapeutic Agent © 2005 Rehabilitation Institute of Chicago
 Opioids—the Science Mechanisms 1 • Biogenesis of endogenous peptides • Anatomic distribution • Endocrine changes • Endogenous opioids and stress 2 • Receptor characteristics 1. Bailey CP, et al. Curr Opin Pharmacol. 2005; 5: 60– 68 [Evidence Level C] 2. Bruehl S, et al. Clin J Pain. 2004; 20: 283– 292. [Evidence Level A] © 2005 Rehabilitation Institute of Chicago
Opioids—the Science Mechanisms 1 • Biogenesis of endogenous peptides • Anatomic distribution • Endocrine changes • Endogenous opioids and stress 2 • Receptor characteristics 1. Bailey CP, et al. Curr Opin Pharmacol. 2005; 5: 60– 68 [Evidence Level C] 2. Bruehl S, et al. Clin J Pain. 2004; 20: 283– 292. [Evidence Level A] © 2005 Rehabilitation Institute of Chicago
 Genetic Polymorphisms and Response to Opioids • Subtype variants of mu opioid receptor 1, 2 – Alter binding affinities – Change in receptor densities 3 • Interindividual variability in pain perception and sensitivity to analgesics 1, 2 – – Efficacy Side effects Tolerance profiles Risk of drug abuse 1. Landau R. Anesthesiology. 2006; 105; 235– 237 [Evidence Level C]; 2. Pasternak GW. J Pain Symptom Manage. © 2005 Rehabilitation Institute of Chicago 2005; 29(suppl): S 2–S 9 [Evidence Level C]; 3. Ross JR, et al. Pharmacogenomics J. 2005; 5: 324– 336. [Evidence Level B]
Genetic Polymorphisms and Response to Opioids • Subtype variants of mu opioid receptor 1, 2 – Alter binding affinities – Change in receptor densities 3 • Interindividual variability in pain perception and sensitivity to analgesics 1, 2 – – Efficacy Side effects Tolerance profiles Risk of drug abuse 1. Landau R. Anesthesiology. 2006; 105; 235– 237 [Evidence Level C]; 2. Pasternak GW. J Pain Symptom Manage. © 2005 Rehabilitation Institute of Chicago 2005; 29(suppl): S 2–S 9 [Evidence Level C]; 3. Ross JR, et al. Pharmacogenomics J. 2005; 5: 324– 336. [Evidence Level B]
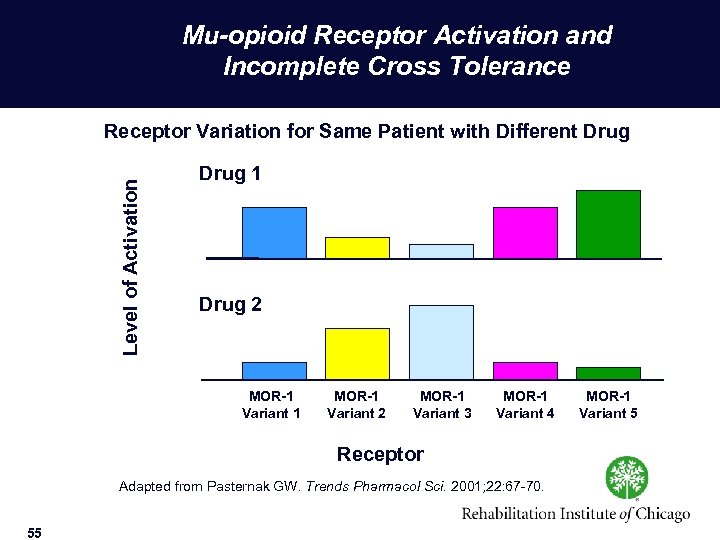 Mu-opioid Receptor Activation and Incomplete Cross Tolerance Level of Activation Receptor Variation for Same Patient with Different Drug 1 Drug 2 MOR-1 Variant 1 MOR-1 Variant 2 MOR-1 Variant 3 MOR-1 Variant 4 Receptor Adapted from Pasternak GW. Trends Pharmacol Sci. 2001; 22: 67 70. © 2004 Rehabilitation Institute of Chicago 55 MOR-1 Variant 5
Mu-opioid Receptor Activation and Incomplete Cross Tolerance Level of Activation Receptor Variation for Same Patient with Different Drug 1 Drug 2 MOR-1 Variant 1 MOR-1 Variant 2 MOR-1 Variant 3 MOR-1 Variant 4 Receptor Adapted from Pasternak GW. Trends Pharmacol Sci. 2001; 22: 67 70. © 2004 Rehabilitation Institute of Chicago 55 MOR-1 Variant 5
 Opioids I. Syntheric phenylpiperidines Meperidine, Fentanyl, II. Synthetic Pseudopiperidines Methadone, Propoxyphene III. Naturally Occurring Alkaloids A. Natural: Heroin, Morphine, Codeine B. Semi-synthetic: Hydromorphone, Oxycodone, Oxymorphone C. Tramadol © 2005 Rehabilitation Institute of Chicago
Opioids I. Syntheric phenylpiperidines Meperidine, Fentanyl, II. Synthetic Pseudopiperidines Methadone, Propoxyphene III. Naturally Occurring Alkaloids A. Natural: Heroin, Morphine, Codeine B. Semi-synthetic: Hydromorphone, Oxycodone, Oxymorphone C. Tramadol © 2005 Rehabilitation Institute of Chicago
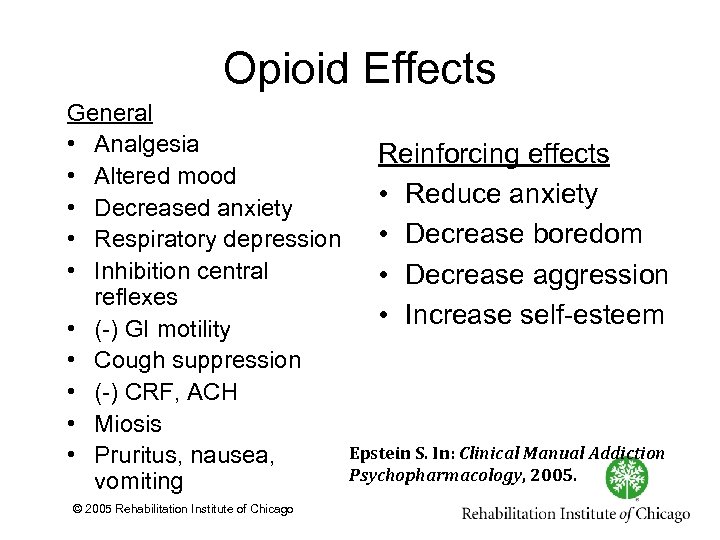 Opioid Effects General • Analgesia • Altered mood • Decreased anxiety • Respiratory depression • Inhibition central reflexes • ( ) GI motility • Cough suppression • ( ) CRF, ACH • Miosis • Pruritus, nausea, vomiting © 2005 Rehabilitation Institute of Chicago Reinforcing effects • Reduce anxiety • Decrease boredom • Decrease aggression • Increase self esteem Epstein S. In: Clinical Manual Addiction Psychopharmacology, 2005.
Opioid Effects General • Analgesia • Altered mood • Decreased anxiety • Respiratory depression • Inhibition central reflexes • ( ) GI motility • Cough suppression • ( ) CRF, ACH • Miosis • Pruritus, nausea, vomiting © 2005 Rehabilitation Institute of Chicago Reinforcing effects • Reduce anxiety • Decrease boredom • Decrease aggression • Increase self esteem Epstein S. In: Clinical Manual Addiction Psychopharmacology, 2005.
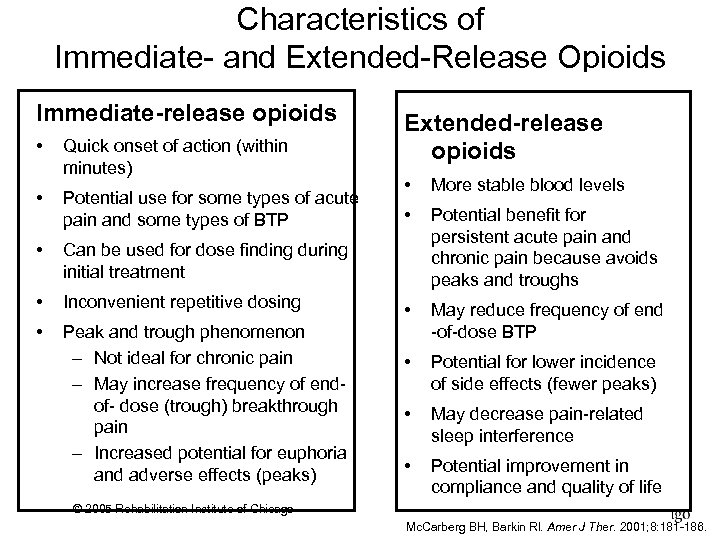 Characteristics of Immediate and Extended Release Opioids Immediate-release opioids • Quick onset of action (within minutes) • Potential use for some types of acute pain and some types of BTP • Inconvenient repetitive dosing • Peak and trough phenomenon – Not ideal for chronic pain – May increase frequency of end of dose (trough) breakthrough pain – Increased potential for euphoria and adverse effects (peaks) • More stable blood levels • Potential benefit for persistent acute pain and chronic pain because avoids peaks and troughs • May reduce frequency of end of dose BTP • Potential for lower incidence of side effects (fewer peaks) • May decrease pain related sleep interference • Potential improvement in compliance and quality of life Can be used for dose finding during initial treatment • Extended-release opioids © 2005 Rehabilitation Institute of Chicago Mc. Carberg BH, Barkin Rl. Amer J Ther. 2001; 8: 181 186.
Characteristics of Immediate and Extended Release Opioids Immediate-release opioids • Quick onset of action (within minutes) • Potential use for some types of acute pain and some types of BTP • Inconvenient repetitive dosing • Peak and trough phenomenon – Not ideal for chronic pain – May increase frequency of end of dose (trough) breakthrough pain – Increased potential for euphoria and adverse effects (peaks) • More stable blood levels • Potential benefit for persistent acute pain and chronic pain because avoids peaks and troughs • May reduce frequency of end of dose BTP • Potential for lower incidence of side effects (fewer peaks) • May decrease pain related sleep interference • Potential improvement in compliance and quality of life Can be used for dose finding during initial treatment • Extended-release opioids © 2005 Rehabilitation Institute of Chicago Mc. Carberg BH, Barkin Rl. Amer J Ther. 2001; 8: 181 186.
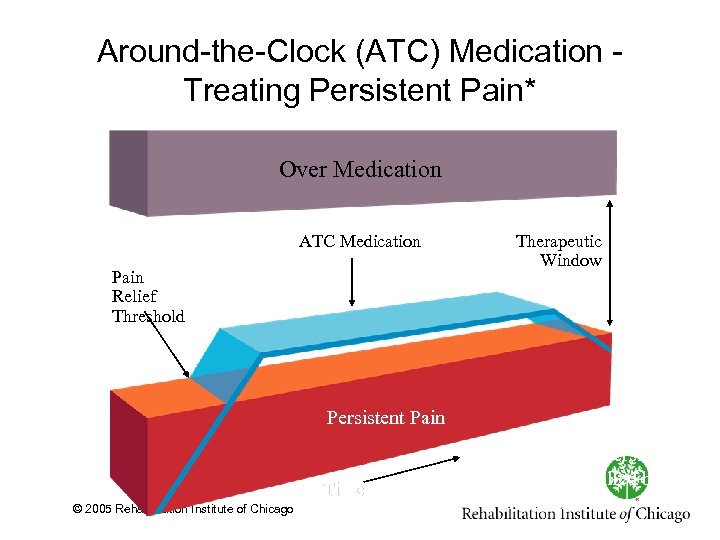 Around the Clock (ATC) Medication Treating Persistent Pain* Over Medication ATC Medication Pain Relief Threshold Therapeutic Window Persistent Pain Time © 2005 Rehabilitation Institute of Chicago *Baseline or persistent pain is controlled by ATC medication.
Around the Clock (ATC) Medication Treating Persistent Pain* Over Medication ATC Medication Pain Relief Threshold Therapeutic Window Persistent Pain Time © 2005 Rehabilitation Institute of Chicago *Baseline or persistent pain is controlled by ATC medication.
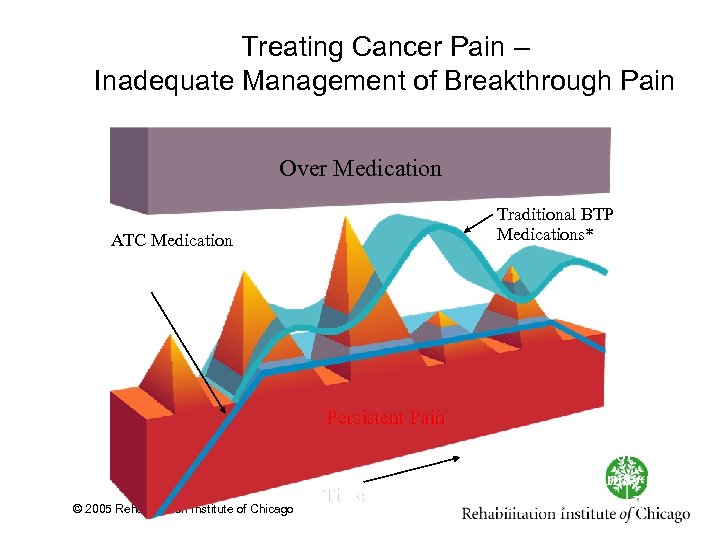 Treating Cancer Pain – Inadequate Management of Breakthrough Pain Over Medication Traditional BTP Medications* ATC Medication Persistent Pain © 2005 Rehabilitation Institute of Chicago Time *Delayed onset of action, type and dose may not adequately treat a BTP episode.
Treating Cancer Pain – Inadequate Management of Breakthrough Pain Over Medication Traditional BTP Medications* ATC Medication Persistent Pain © 2005 Rehabilitation Institute of Chicago Time *Delayed onset of action, type and dose may not adequately treat a BTP episode.
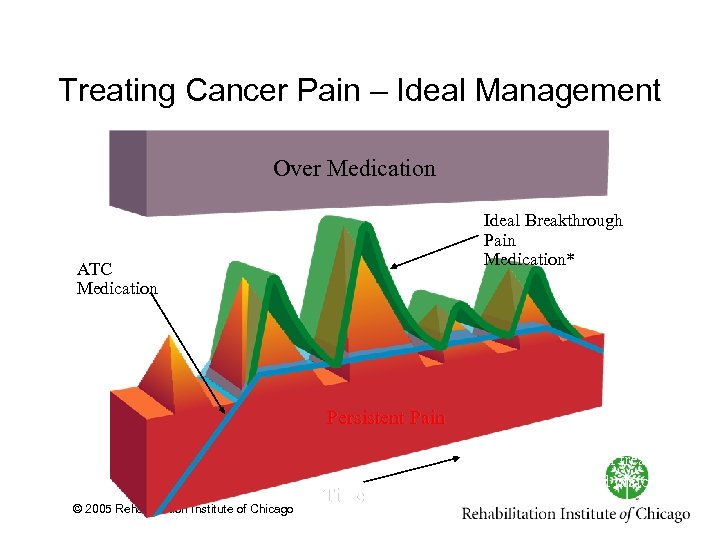 Treating Cancer Pain – Ideal Management Over Medication Ideal Breakthrough Pain Medication* ATC Medication Persistent Pain © 2005 Rehabilitation Institute of Chicago Time *Breakthrough pain treatment has rapid onset and matches the intensity and duration of BTP episode.
Treating Cancer Pain – Ideal Management Over Medication Ideal Breakthrough Pain Medication* ATC Medication Persistent Pain © 2005 Rehabilitation Institute of Chicago Time *Breakthrough pain treatment has rapid onset and matches the intensity and duration of BTP episode.
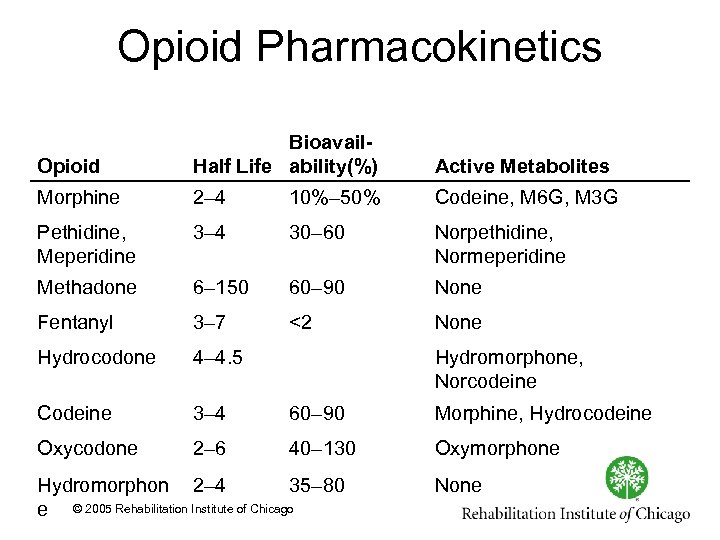 Opioid Pharmacokinetics Opioid Bioavail. Half Life ability(%) Active Metabolites Morphine 2– 4 10%– 50% Codeine, M 6 G, M 3 G Pethidine, Meperidine 3– 4 30– 60 Norpethidine, Normeperidine Methadone 6– 150 60– 90 None Fentanyl 3– 7 <2 None Hydrocodone 4– 4. 5 Codeine 3– 4 60– 90 Morphine, Hydrocodeine Oxycodone 2– 6 40– 130 Oxymorphone Hydromorphone, Norcodeine Hydromorphon 2– 4 35– 80 e © 2005 Rehabilitation Institute of Chicago None
Opioid Pharmacokinetics Opioid Bioavail. Half Life ability(%) Active Metabolites Morphine 2– 4 10%– 50% Codeine, M 6 G, M 3 G Pethidine, Meperidine 3– 4 30– 60 Norpethidine, Normeperidine Methadone 6– 150 60– 90 None Fentanyl 3– 7 <2 None Hydrocodone 4– 4. 5 Codeine 3– 4 60– 90 Morphine, Hydrocodeine Oxycodone 2– 6 40– 130 Oxymorphone Hydromorphone, Norcodeine Hydromorphon 2– 4 35– 80 e © 2005 Rehabilitation Institute of Chicago None
 Propoxyphene (Darvocet) • • With acetaminophen (Darvocet N 100) Structurally related to methadone Peak plasma concentration 2 h Liver metabolism to norproxyphene © 2005 Rehabilitation Institute of Chicago
Propoxyphene (Darvocet) • • With acetaminophen (Darvocet N 100) Structurally related to methadone Peak plasma concentration 2 h Liver metabolism to norproxyphene © 2005 Rehabilitation Institute of Chicago
 FDA ruling on Propoxyphene • • • UK: phased removal in 2005 Pubic Citizen petitioned for removal ASHSP recommends FDA withdrawal (Jan ‘ 09) No more effective than APAP alone, similar to tramadol Supported by Beers Criteria as inappropriate for the elderly • FDA Advisory Committee (Jan. ’ 09): 14 12 in support of removing from market • July 7, 2009: FDA: black box warning, remain on market © 2005 Rehabilitation Institute of Chicago
FDA ruling on Propoxyphene • • • UK: phased removal in 2005 Pubic Citizen petitioned for removal ASHSP recommends FDA withdrawal (Jan ‘ 09) No more effective than APAP alone, similar to tramadol Supported by Beers Criteria as inappropriate for the elderly • FDA Advisory Committee (Jan. ’ 09): 14 12 in support of removing from market • July 7, 2009: FDA: black box warning, remain on market © 2005 Rehabilitation Institute of Chicago
 Morphine (Gold Standard) Hydrophilic • Slower entry into CNS • Active metabolites (M 3 G, M 6 G) Morphine 3 glucuronide • inactive? , CNS excitation Morphine 6 glucuroide • Active analgesic, 2 100 x more potent (centrally), Induces liver enzymes © 2005 Rehabilitation Institute of Chicago MS IR®: 15, 30 mg MS Contin®: 15, 30, 60, 100, 200 mg Kadian®: 20, 30, 50, 60 mg Avinza®: 30, 60, 90, 120 mg
Morphine (Gold Standard) Hydrophilic • Slower entry into CNS • Active metabolites (M 3 G, M 6 G) Morphine 3 glucuronide • inactive? , CNS excitation Morphine 6 glucuroide • Active analgesic, 2 100 x more potent (centrally), Induces liver enzymes © 2005 Rehabilitation Institute of Chicago MS IR®: 15, 30 mg MS Contin®: 15, 30, 60, 100, 200 mg Kadian®: 20, 30, 50, 60 mg Avinza®: 30, 60, 90, 120 mg
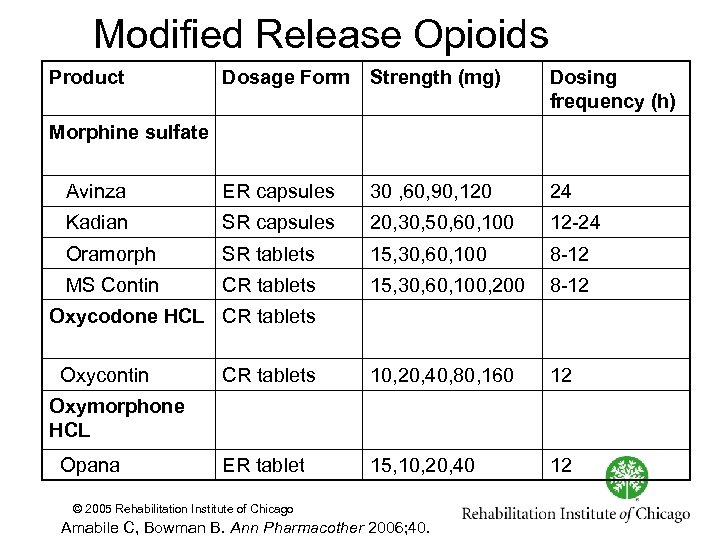 Modified Release Opioids Product Dosage Form Strength (mg) Dosing frequency (h) Avinza ER capsules 30 , 60, 90, 120 24 Kadian SR capsules 20, 30, 50, 60, 100 12 24 Oramorph SR tablets 15, 30, 60, 100 8 12 MS Contin CR tablets 15, 30, 60, 100, 200 8 12 CR tablets 10, 20, 40, 80, 160 12 ER tablet 15, 10, 20, 40 12 Morphine sulfate Oxycodone HCL CR tablets Oxycontin Oxymorphone HCL Opana © 2005 Rehabilitation Institute of Chicago Amabile C, Bowman B. Ann Pharmacother 2006; 40.
Modified Release Opioids Product Dosage Form Strength (mg) Dosing frequency (h) Avinza ER capsules 30 , 60, 90, 120 24 Kadian SR capsules 20, 30, 50, 60, 100 12 24 Oramorph SR tablets 15, 30, 60, 100 8 12 MS Contin CR tablets 15, 30, 60, 100, 200 8 12 CR tablets 10, 20, 40, 80, 160 12 ER tablet 15, 10, 20, 40 12 Morphine sulfate Oxycodone HCL CR tablets Oxycontin Oxymorphone HCL Opana © 2005 Rehabilitation Institute of Chicago Amabile C, Bowman B. Ann Pharmacother 2006; 40.
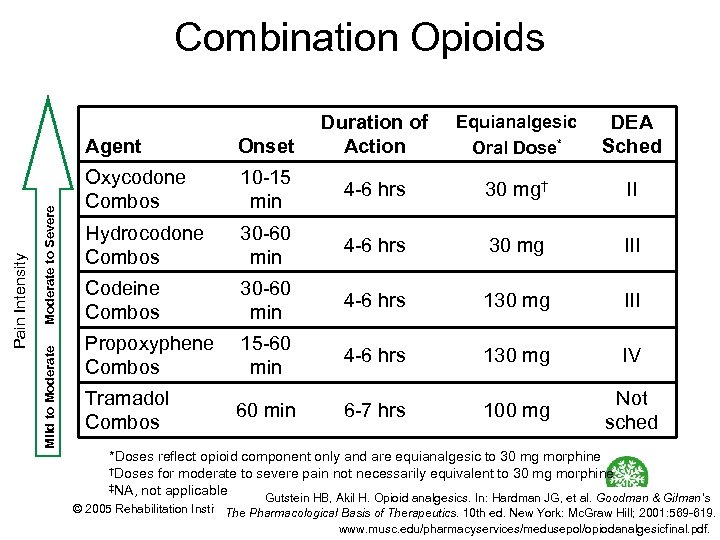 Combination Opioids Duration of Action Equianalgesic Oral Dose* DEA Sched Moderate to Severe Mild to Moderate Pain Intensity Agent Onset Oxycodone Combos 10 15 min 4 6 hrs 30 mg† II Hydrocodone Combos 30 60 min 4 6 hrs 30 mg III Codeine Combos 30 60 min 4 6 hrs 130 mg III Propoxyphene Combos 15 60 min 4 6 hrs 130 mg IV Tramadol Combos 60 min 6 7 hrs 100 mg Not sched *Doses reflect opioid component only and are equianalgesic to 30 mg morphine †Doses for moderate to severe pain not necessarily equivalent to 30 mg morphine ‡NA, not applicable Gutstein HB, Akil H. Opioid analgesics. In: Hardman JG, et al. Goodman & Gilman’s © 2005 Rehabilitation Institute of Pharmacological Basis of Therapeutics. 10 th ed. New York: Mc. Graw Hill; 2001: 569 619. Chicago The www. musc. edu/pharmacyservices/medusepol/opiodanalgesicfinal. pdf.
Combination Opioids Duration of Action Equianalgesic Oral Dose* DEA Sched Moderate to Severe Mild to Moderate Pain Intensity Agent Onset Oxycodone Combos 10 15 min 4 6 hrs 30 mg† II Hydrocodone Combos 30 60 min 4 6 hrs 30 mg III Codeine Combos 30 60 min 4 6 hrs 130 mg III Propoxyphene Combos 15 60 min 4 6 hrs 130 mg IV Tramadol Combos 60 min 6 7 hrs 100 mg Not sched *Doses reflect opioid component only and are equianalgesic to 30 mg morphine †Doses for moderate to severe pain not necessarily equivalent to 30 mg morphine ‡NA, not applicable Gutstein HB, Akil H. Opioid analgesics. In: Hardman JG, et al. Goodman & Gilman’s © 2005 Rehabilitation Institute of Pharmacological Basis of Therapeutics. 10 th ed. New York: Mc. Graw Hill; 2001: 569 619. Chicago The www. musc. edu/pharmacyservices/medusepol/opiodanalgesicfinal. pdf.
 Hydrocodone/ Oxycodone – Prodrug (nonactive in form delivered) – Dependent on P 450 2 D 6 enzyme system – Concurrent medication therapy may affect prodrug metabolism if drug(s) inhibit P 450 2 D 6 – Tylenol combination: Norco, Lortab, Percocet, Roxicet © 2005 Rehabilitation Institute of Chicago
Hydrocodone/ Oxycodone – Prodrug (nonactive in form delivered) – Dependent on P 450 2 D 6 enzyme system – Concurrent medication therapy may affect prodrug metabolism if drug(s) inhibit P 450 2 D 6 – Tylenol combination: Norco, Lortab, Percocet, Roxicet © 2005 Rehabilitation Institute of Chicago
 © 2005 Rehabilitation Institute of Chicago
© 2005 Rehabilitation Institute of Chicago
 Acetaminophen (Tylenol) © 2005 Rehabilitation Institute of Chicago
Acetaminophen (Tylenol) © 2005 Rehabilitation Institute of Chicago
 FDA Decisions: June 30, 2009 • 20 17: ban combination products • 21 16: lower maximum daily APAP • 24 13: limit maximum single dose APAP • 26 11: 1, 000 mg dose APAP prescription • Final decision pending © 2005 Rehabilitation Institute of Chicago
FDA Decisions: June 30, 2009 • 20 17: ban combination products • 21 16: lower maximum daily APAP • 24 13: limit maximum single dose APAP • 26 11: 1, 000 mg dose APAP prescription • Final decision pending © 2005 Rehabilitation Institute of Chicago
 Acetaminophen (APAP) • A centrally acting analgesic that increases the pain threshold • Mechanism of action is not fully known – – May selectively inhibit a distinct form of COX (COX 3) Most likely has no affinity for the active site of COX, but blocks activity by reducing the active oxidized form of COX to an inactive form • Indicated to reduce fever and for the temporary relief of minor aches and pains • Fewer GI side effects than NSAIDs/COX 2 inhibitors • Adverse effects associated with chronic use Roberts LJ II, et al. In: Hardman JG, et al, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 2001: 703 705. Lucas R, et al. FASEB J. 2005; 19: 635 637. © 2005 Rehabilitation Institute of Chicago
Acetaminophen (APAP) • A centrally acting analgesic that increases the pain threshold • Mechanism of action is not fully known – – May selectively inhibit a distinct form of COX (COX 3) Most likely has no affinity for the active site of COX, but blocks activity by reducing the active oxidized form of COX to an inactive form • Indicated to reduce fever and for the temporary relief of minor aches and pains • Fewer GI side effects than NSAIDs/COX 2 inhibitors • Adverse effects associated with chronic use Roberts LJ II, et al. In: Hardman JG, et al, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 2001: 703 705. Lucas R, et al. FASEB J. 2005; 19: 635 637. © 2005 Rehabilitation Institute of Chicago
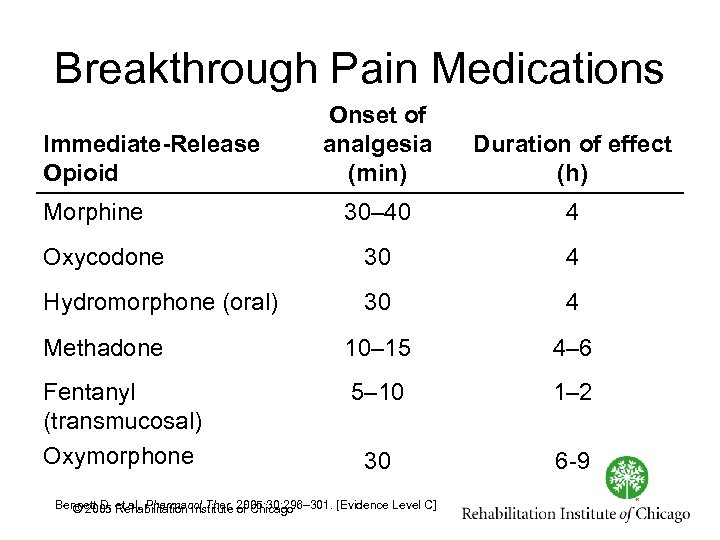 Breakthrough Pain Medications Onset of analgesia (min) Duration of effect (h) 30– 40 4 Oxycodone 30 4 Hydromorphone (oral) 30 4 Methadone 10– 15 4– 6 Fentanyl (transmucosal) Oxymorphone 5– 10 1– 2 30 6 9 Immediate-Release Opioid Morphine Bennett D, et al. Pharmacol Ther. 2005; 30: 296– 301. [Evidence Level C] © 2005 Rehabilitation Institute of Chicago
Breakthrough Pain Medications Onset of analgesia (min) Duration of effect (h) 30– 40 4 Oxycodone 30 4 Hydromorphone (oral) 30 4 Methadone 10– 15 4– 6 Fentanyl (transmucosal) Oxymorphone 5– 10 1– 2 30 6 9 Immediate-Release Opioid Morphine Bennett D, et al. Pharmacol Ther. 2005; 30: 296– 301. [Evidence Level C] © 2005 Rehabilitation Institute of Chicago
 New Short Acting Opioids for Breakthrough Pain (BTP) • • Transmucosal fentanyl (Actiq®) Fentanyl buccal tablet (Fentora™) Oxymorphone (Opana®) Tapentadol (Nucynta®) © 2005 Rehabilitation Institute of Chicago
New Short Acting Opioids for Breakthrough Pain (BTP) • • Transmucosal fentanyl (Actiq®) Fentanyl buccal tablet (Fentora™) Oxymorphone (Opana®) Tapentadol (Nucynta®) © 2005 Rehabilitation Institute of Chicago
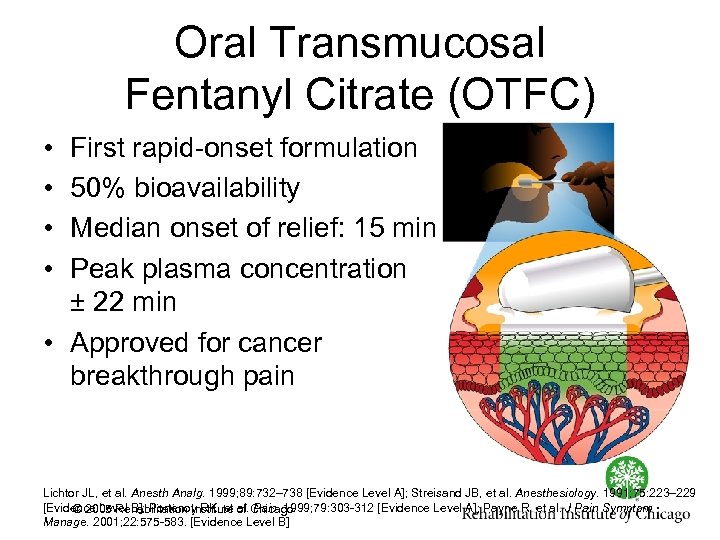 Oral Transmucosal Fentanyl Citrate (OTFC) • • First rapid onset formulation 50% bioavailability Median onset of relief: 15 min Peak plasma concentration ± 22 min • Approved for cancer breakthrough pain Lichtor JL, et al. Anesth Analg. 1999; 89: 732– 738 [Evidence Level A]; Streisand JB, et al. Anesthesiology. 1991; 75: 223– 229 [Evidence Level B]; Portenoy RK, et of Chicago © 2005 Rehabilitation Institute al. Pain. 1999; 79: 303 312 [Evidence Level A]; Payne R, et al. J Pain Symptom Manage. 2001; 22: 575 583. [Evidence Level B]
Oral Transmucosal Fentanyl Citrate (OTFC) • • First rapid onset formulation 50% bioavailability Median onset of relief: 15 min Peak plasma concentration ± 22 min • Approved for cancer breakthrough pain Lichtor JL, et al. Anesth Analg. 1999; 89: 732– 738 [Evidence Level A]; Streisand JB, et al. Anesthesiology. 1991; 75: 223– 229 [Evidence Level B]; Portenoy RK, et of Chicago © 2005 Rehabilitation Institute al. Pain. 1999; 79: 303 312 [Evidence Level A]; Payne R, et al. J Pain Symptom Manage. 2001; 22: 575 583. [Evidence Level B]
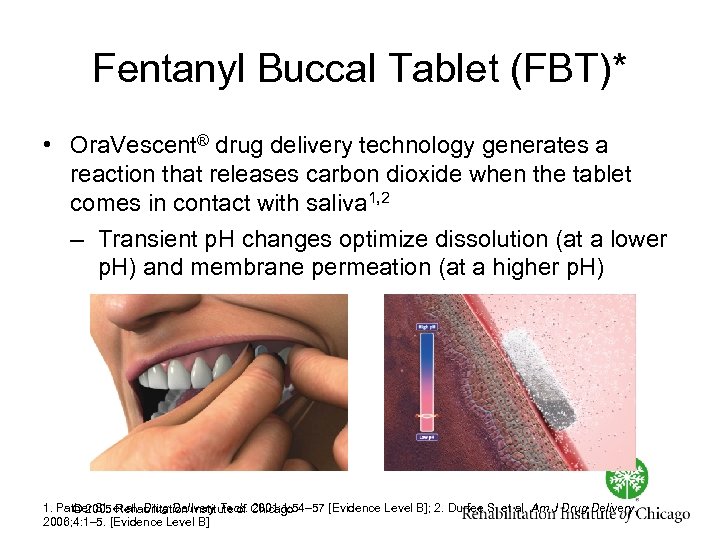 Fentanyl Buccal Tablet (FBT)* • Ora. Vescent® drug delivery technology generates a reaction that releases carbon dioxide when the tablet comes in contact with saliva 1, 2 – Transient p. H changes optimize dissolution (at a lower p. H) and membrane permeation (at a higher p. H) 1. Pather SI, et al. Drug Delivery Tech. Chicago © 2005 Rehabilitation Institute of 2001; 1: 54– 57 [Evidence Level B]; 2. Durfee S, et al. Am J Drug Delivery. 2006; 4: 1– 5. [Evidence Level B]
Fentanyl Buccal Tablet (FBT)* • Ora. Vescent® drug delivery technology generates a reaction that releases carbon dioxide when the tablet comes in contact with saliva 1, 2 – Transient p. H changes optimize dissolution (at a lower p. H) and membrane permeation (at a higher p. H) 1. Pather SI, et al. Drug Delivery Tech. Chicago © 2005 Rehabilitation Institute of 2001; 1: 54– 57 [Evidence Level B]; 2. Durfee S, et al. Am J Drug Delivery. 2006; 4: 1– 5. [Evidence Level B]
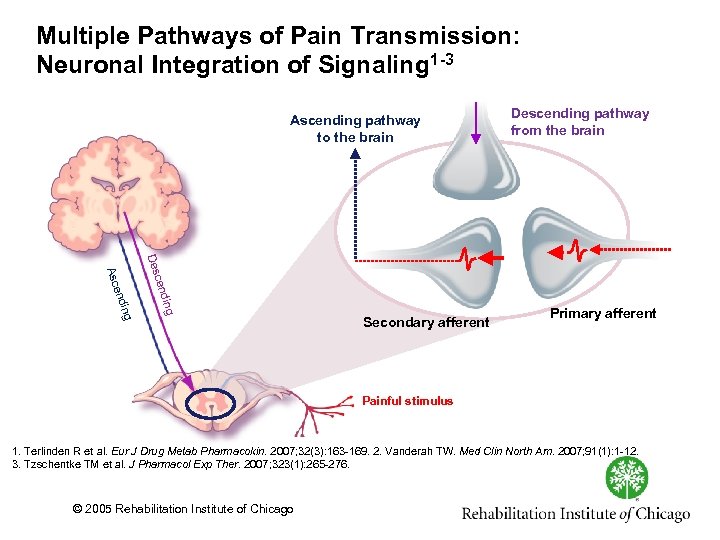 Multiple Pathways of Pain Transmission: Neuronal Integration of Signaling 1 -3 Ascending pathway to the brain Descending pathway from the brain ding cen Des ing end Asc Secondary afferent Primary afferent Painful stimulus 1. Terlinden R et al. Eur J Drug Metab Pharmacokin. 2007; 32(3): 163 169. 2. Vanderah TW. Med Clin North Am. 2007; 91(1): 1 12. 3. Tzschentke TM et al. J Pharmacol Exp Ther. 2007; 323(1): 265 276. © 2005 Rehabilitation Institute of Chicago
Multiple Pathways of Pain Transmission: Neuronal Integration of Signaling 1 -3 Ascending pathway to the brain Descending pathway from the brain ding cen Des ing end Asc Secondary afferent Primary afferent Painful stimulus 1. Terlinden R et al. Eur J Drug Metab Pharmacokin. 2007; 32(3): 163 169. 2. Vanderah TW. Med Clin North Am. 2007; 91(1): 1 12. 3. Tzschentke TM et al. J Pharmacol Exp Ther. 2007; 323(1): 265 276. © 2005 Rehabilitation Institute of Chicago
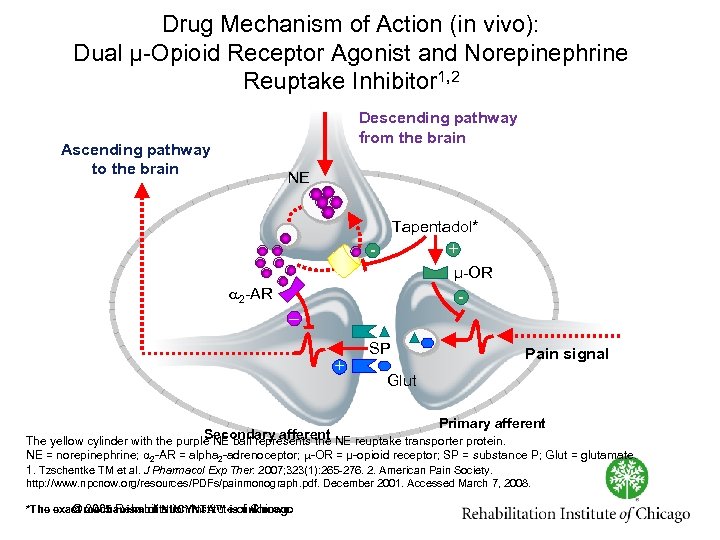 Drug Mechanism of Action (in vivo): Dual μ Opioid Receptor Agonist and Norepinephrine Reuptake Inhibitor 1, 2 Descending pathway from the brain Ascending pathway to the brain NE Tapentadol* + μ OR 2 AR – + SP Pain signal Glut Primary afferent Secondary afferent NE reuptake transporter protein. The yellow cylinder with the purple NE ball represents the NE = norepinephrine; 2 AR = alpha 2 adrenoceptor; OR = μ opioid receptor; SP = substance P; Glut = glutamate. 1. Tzschentke TM et al. J Pharmacol Exp Ther. 2007; 323(1): 265 276. 2. American Pain Society. http: //www. npcnow. org/resources/PDFs/painmonograph. pdf. December 2001. Accessed March 7, 2008. © 2005 Rehabilitation Institute unknown. *The exact mechanism of NUCYNTA™ isof Chicago
Drug Mechanism of Action (in vivo): Dual μ Opioid Receptor Agonist and Norepinephrine Reuptake Inhibitor 1, 2 Descending pathway from the brain Ascending pathway to the brain NE Tapentadol* + μ OR 2 AR – + SP Pain signal Glut Primary afferent Secondary afferent NE reuptake transporter protein. The yellow cylinder with the purple NE ball represents the NE = norepinephrine; 2 AR = alpha 2 adrenoceptor; OR = μ opioid receptor; SP = substance P; Glut = glutamate. 1. Tzschentke TM et al. J Pharmacol Exp Ther. 2007; 323(1): 265 276. 2. American Pain Society. http: //www. npcnow. org/resources/PDFs/painmonograph. pdf. December 2001. Accessed March 7, 2008. © 2005 Rehabilitation Institute unknown. *The exact mechanism of NUCYNTA™ isof Chicago
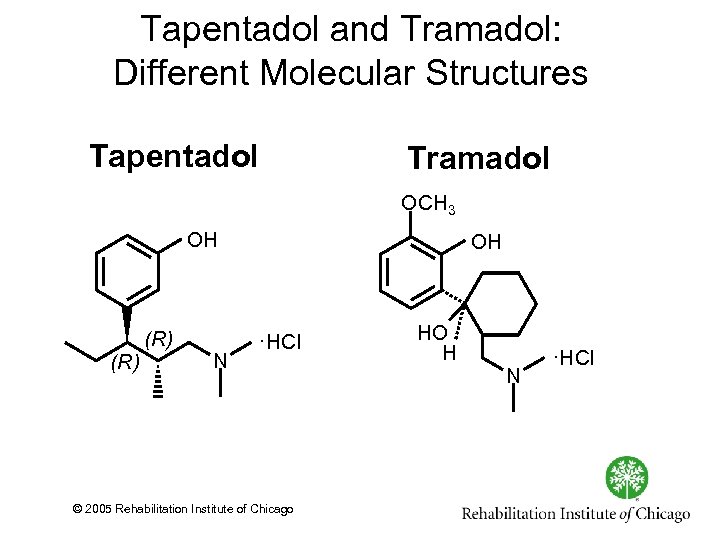 Tapentadol and Tramadol: Different Molecular Structures Tapentadol Tramadol OCH 3 OH (R) N OH ·HCl © 2005 Rehabilitation Institute of Chicago HO H N ·HCl
Tapentadol and Tramadol: Different Molecular Structures Tapentadol Tramadol OCH 3 OH (R) N OH ·HCl © 2005 Rehabilitation Institute of Chicago HO H N ·HCl
 Tapentadol and Tramadol: Prescribing Information Differences Feature Tapentadol Tramadol Pain severity Moderate to severe Moderate to moderately severe Mechanism of action • Binds to μ receptors • Inhibits reuptake of norepinephrine • Inhibits reuptake of serotonin Activity of metabolites Metabolites are not active M 1 metabolite with 6 and 200 fold greater potency than parent drug for analgesia and μ OR affinity, respectively Metabolism Primarily Phase 2 conjugation to glucuronide and sulfate O demethylation produces M 1 (CYP 2 D 6); N demethylation (CYP 3 A 4 and CYP 2 B 6); sulfation/glucuronidation Federal scheduling Schedule II Unscheduled OR = opioid receptor. © 2005 Rehabilitation Institute of Chicago
Tapentadol and Tramadol: Prescribing Information Differences Feature Tapentadol Tramadol Pain severity Moderate to severe Moderate to moderately severe Mechanism of action • Binds to μ receptors • Inhibits reuptake of norepinephrine • Inhibits reuptake of serotonin Activity of metabolites Metabolites are not active M 1 metabolite with 6 and 200 fold greater potency than parent drug for analgesia and μ OR affinity, respectively Metabolism Primarily Phase 2 conjugation to glucuronide and sulfate O demethylation produces M 1 (CYP 2 D 6); N demethylation (CYP 3 A 4 and CYP 2 B 6); sulfation/glucuronidation Federal scheduling Schedule II Unscheduled OR = opioid receptor. © 2005 Rehabilitation Institute of Chicago
 Tapentadol: Indication and Dosing • • • Tapentadol is indicated for the relief of moderate to severe acute pain in patients 18 years of age or older The dose is 50 mg, 75 mg, or 100 mg every 4 to 6 hours depending upon pain intensity – Good clinical practice dictates that the lowest starting dose be used and, as always, the dose should be individualized for the patient – 50 mg is the lowest dose available – Daily doses on first day of therapy above 700 mg and on subsequent days above 600 mg have not been studied in controlled clinical trials and are not recommended Clinical considerations should include: – Patient's medical history and concomitant medication use – Previous experience prescribing similar drugs – Severity of the pain being treated – Ability to monitor the patient © 2005 Rehabilitation Institute of Chicago
Tapentadol: Indication and Dosing • • • Tapentadol is indicated for the relief of moderate to severe acute pain in patients 18 years of age or older The dose is 50 mg, 75 mg, or 100 mg every 4 to 6 hours depending upon pain intensity – Good clinical practice dictates that the lowest starting dose be used and, as always, the dose should be individualized for the patient – 50 mg is the lowest dose available – Daily doses on first day of therapy above 700 mg and on subsequent days above 600 mg have not been studied in controlled clinical trials and are not recommended Clinical considerations should include: – Patient's medical history and concomitant medication use – Previous experience prescribing similar drugs – Severity of the pain being treated – Ability to monitor the patient © 2005 Rehabilitation Institute of Chicago
 Tapentadol: Clinical Pharmacokinetics • Bioavailability is 32% after single dose administration in fasted state • Low plasma protein binding (~20%) • 97% metabolized – Occurs mainly via Phase 2 pathways (55% O glucuronide, 15% sulfate of tapentadol) – Other metabolism (15%) via P 450 enzymes (13% by CYP 2 C 9 and CYP 2 C 19, 2% by CYP 2 D 6) • 99% of tapentadol and its metabolites eliminated via renal clearance • Metabolites lack analgesic activity • Time to maximum serum concentration (Tmax): 1. 25 h • Half life (t 1/2): 4 h © 2005 Rehabilitation Institute of Chicago
Tapentadol: Clinical Pharmacokinetics • Bioavailability is 32% after single dose administration in fasted state • Low plasma protein binding (~20%) • 97% metabolized – Occurs mainly via Phase 2 pathways (55% O glucuronide, 15% sulfate of tapentadol) – Other metabolism (15%) via P 450 enzymes (13% by CYP 2 C 9 and CYP 2 C 19, 2% by CYP 2 D 6) • 99% of tapentadol and its metabolites eliminated via renal clearance • Metabolites lack analgesic activity • Time to maximum serum concentration (Tmax): 1. 25 h • Half life (t 1/2): 4 h © 2005 Rehabilitation Institute of Chicago
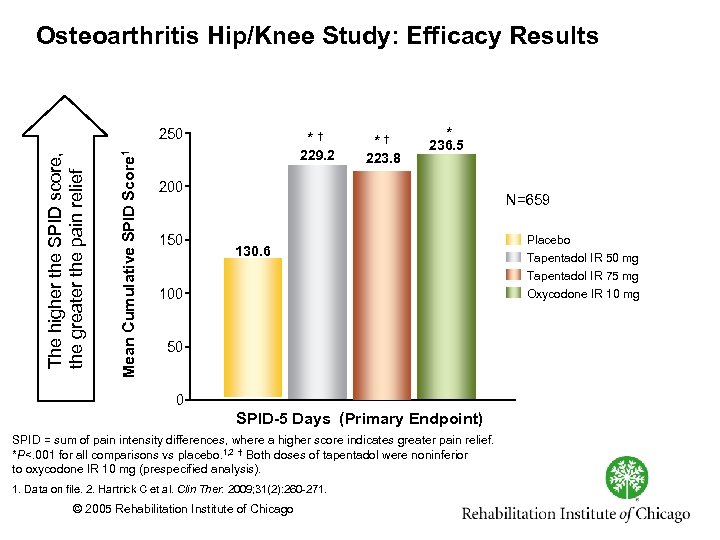 Osteoarthritis Hip/Knee Study: Efficacy Results Mean Cumulative SPID Score 1 The higher the SPID score, the greater the pain relief 250 *† 229. 2 *† 223. 8 * 236. 5 200 150 N=659 130. 6 100 50 0 SPID-5 Days (Primary Endpoint) SPID = sum of pain intensity differences, where a higher score indicates greater pain relief. *P<. 001 for all comparisons vs placebo. 1, 2 † Both doses of tapentadol were noninferior to oxycodone IR 10 mg (prespecified analysis). 1. Data on file. 2. Hartrick C et al. Clin Ther. 2009; 31(2): 260 271. © 2005 Rehabilitation Institute of Chicago Placebo Tapentadol IR 50 mg Tapentadol IR 75 mg Oxycodone IR 10 mg
Osteoarthritis Hip/Knee Study: Efficacy Results Mean Cumulative SPID Score 1 The higher the SPID score, the greater the pain relief 250 *† 229. 2 *† 223. 8 * 236. 5 200 150 N=659 130. 6 100 50 0 SPID-5 Days (Primary Endpoint) SPID = sum of pain intensity differences, where a higher score indicates greater pain relief. *P<. 001 for all comparisons vs placebo. 1, 2 † Both doses of tapentadol were noninferior to oxycodone IR 10 mg (prespecified analysis). 1. Data on file. 2. Hartrick C et al. Clin Ther. 2009; 31(2): 260 271. © 2005 Rehabilitation Institute of Chicago Placebo Tapentadol IR 50 mg Tapentadol IR 75 mg Oxycodone IR 10 mg
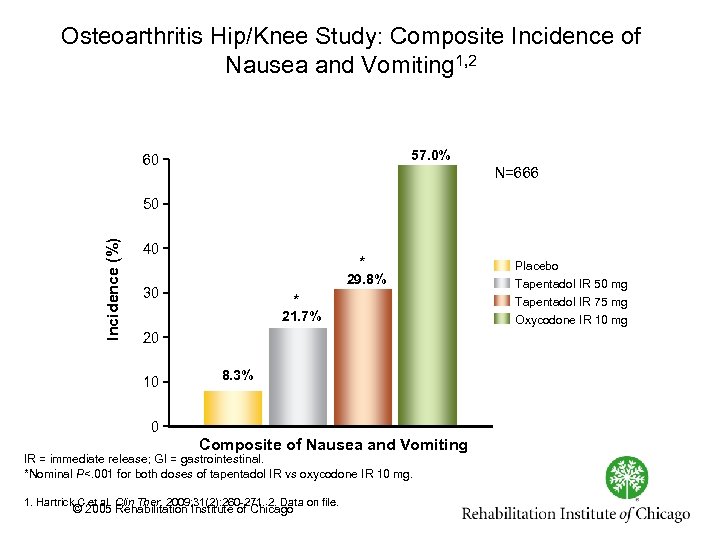 Osteoarthritis Hip/Knee Study: Composite Incidence of Nausea and Vomiting 1, 2 57. 0% 60 N=666 Incidence (%) 50 40 * 29. 8% 30 * 21. 7% 20 10 8. 3% 0 Composite of Nausea and Vomiting IR = immediate release; GI = gastrointestinal. *Nominal P<. 001 for both doses of tapentadol IR vs oxycodone IR 10 mg. 1. Hartrick C et al. Clin Ther. 2009; 31(2): 260 271. 2. Data on file. © 2005 Rehabilitation Institute of Chicago Placebo Tapentadol IR 50 mg Tapentadol IR 75 mg Oxycodone IR 10 mg
Osteoarthritis Hip/Knee Study: Composite Incidence of Nausea and Vomiting 1, 2 57. 0% 60 N=666 Incidence (%) 50 40 * 29. 8% 30 * 21. 7% 20 10 8. 3% 0 Composite of Nausea and Vomiting IR = immediate release; GI = gastrointestinal. *Nominal P<. 001 for both doses of tapentadol IR vs oxycodone IR 10 mg. 1. Hartrick C et al. Clin Ther. 2009; 31(2): 260 271. 2. Data on file. © 2005 Rehabilitation Institute of Chicago Placebo Tapentadol IR 50 mg Tapentadol IR 75 mg Oxycodone IR 10 mg
 New Long Acting Opioids • Tramadol ER (Ultram® ER) oral tablet – Q 24 h ER technology 1 • Oxymorphone ER (Opana ER®) oral tablet 2– 4 – Q 12 h ER technology • Hydrophilic gel matrix with slowly eroding core • Tramadol HCL ER (Ryzolt®) 1. Gana TJ, et al. Curr Med Res Opin. 2006; 22: 1391– 1401 [Evidence Level A]; 2. Prommer E. Support Care Cancer. 2006; 14: 109– 115 [Evidence Level C]; 3. Durfee S. Am J Drug Deliv. 2006; 4: 1– 5 [Evidence Level B]; 4. Mc. IIwain H, et al. Am J Ther. 2005; 12: 106– 112. [Evidence Level B] © 2005 Rehabilitation Institute of Chicago
New Long Acting Opioids • Tramadol ER (Ultram® ER) oral tablet – Q 24 h ER technology 1 • Oxymorphone ER (Opana ER®) oral tablet 2– 4 – Q 12 h ER technology • Hydrophilic gel matrix with slowly eroding core • Tramadol HCL ER (Ryzolt®) 1. Gana TJ, et al. Curr Med Res Opin. 2006; 22: 1391– 1401 [Evidence Level A]; 2. Prommer E. Support Care Cancer. 2006; 14: 109– 115 [Evidence Level C]; 3. Durfee S. Am J Drug Deliv. 2006; 4: 1– 5 [Evidence Level B]; 4. Mc. IIwain H, et al. Am J Ther. 2005; 12: 106– 112. [Evidence Level B] © 2005 Rehabilitation Institute of Chicago
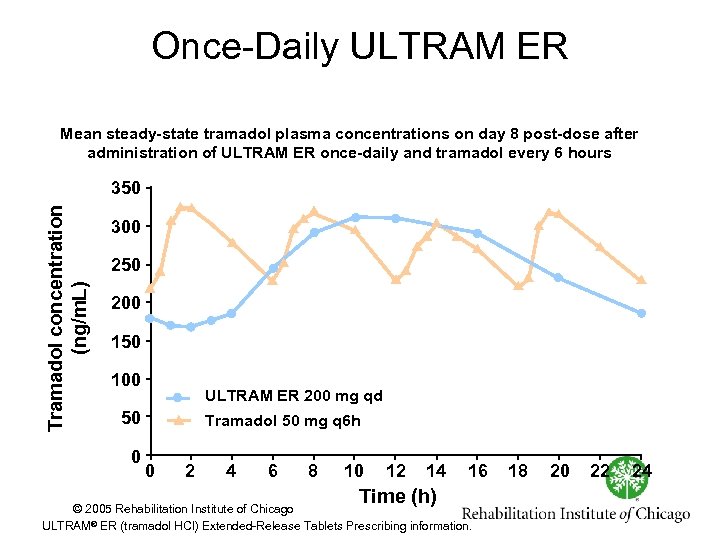 Once Daily ULTRAM ER Mean steady-state tramadol plasma concentrations on day 8 post-dose after administration of ULTRAM ER once-daily and tramadol every 6 hours Tramadol concentration (ng/m. L) 350 300 250 200 150 100 ULTRAM ER 200 mg qd 50 0 Tramadol 50 mg q 6 h 0 2 4 6 8 10 12 14 Time (h) 16 © 2005 Rehabilitation Institute of Chicago ULTRAM® ER (tramadol HCl) Extended Release Tablets Prescribing information. 18 20 22 24
Once Daily ULTRAM ER Mean steady-state tramadol plasma concentrations on day 8 post-dose after administration of ULTRAM ER once-daily and tramadol every 6 hours Tramadol concentration (ng/m. L) 350 300 250 200 150 100 ULTRAM ER 200 mg qd 50 0 Tramadol 50 mg q 6 h 0 2 4 6 8 10 12 14 Time (h) 16 © 2005 Rehabilitation Institute of Chicago ULTRAM® ER (tramadol HCl) Extended Release Tablets Prescribing information. 18 20 22 24
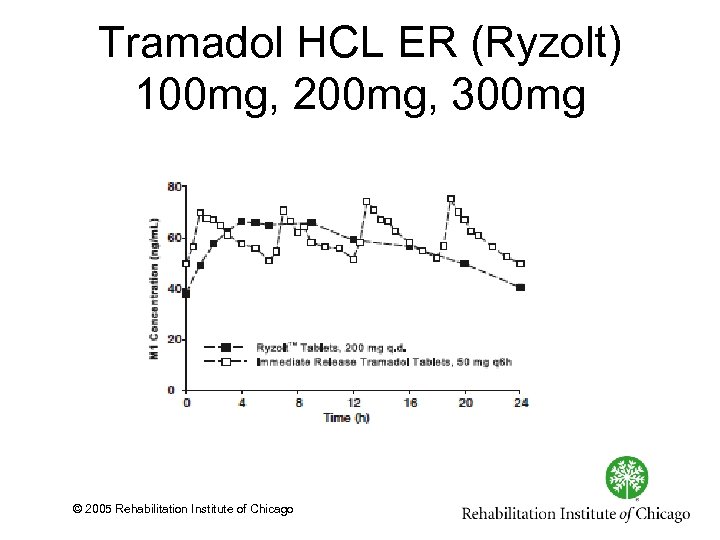 Tramadol HCL ER (Ryzolt) 100 mg, 200 mg, 300 mg © 2005 Rehabilitation Institute of Chicago
Tramadol HCL ER (Ryzolt) 100 mg, 200 mg, 300 mg © 2005 Rehabilitation Institute of Chicago
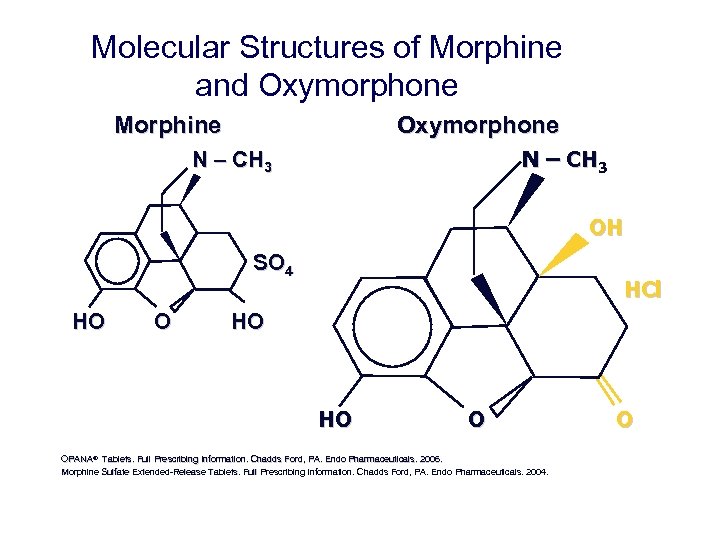 Molecular Structures of Morphine and Oxymorphone Morphine Oxymorphone N – CH 3 OH SO 4 HO O HCl HO HO O OPANA® Tablets. Full Prescribing Information. Chadds Ford, PA. Endo Pharmaceuticals. 2006. Morphine Sulfate Extended Release Tablets. Full Prescribing Information. Chadds Ford, PA. Endo Pharmaceuticals. 2004. O
Molecular Structures of Morphine and Oxymorphone Morphine Oxymorphone N – CH 3 OH SO 4 HO O HCl HO HO O OPANA® Tablets. Full Prescribing Information. Chadds Ford, PA. Endo Pharmaceuticals. 2006. Morphine Sulfate Extended Release Tablets. Full Prescribing Information. Chadds Ford, PA. Endo Pharmaceuticals. 2004. O
 Methadone (Dolophine) • Synthetic, μ agonists, NMDA antagonist • L/D enantiomers (50: 50) • Oral bioavailability varies (40%– 99%) • Biphasic plasma level decline • No active metabolites • Metabolized by liver (CYP 3 A 4) © 2005 Rehabilitation Institute of Chicago L/D = light/darkness ratio; NMDA = N methyl D aspartate.
Methadone (Dolophine) • Synthetic, μ agonists, NMDA antagonist • L/D enantiomers (50: 50) • Oral bioavailability varies (40%– 99%) • Biphasic plasma level decline • No active metabolites • Metabolized by liver (CYP 3 A 4) © 2005 Rehabilitation Institute of Chicago L/D = light/darkness ratio; NMDA = N methyl D aspartate.
 Methadone Dosing: Gradual Conversion Mor-E (mg/d) Calculate Meth (mg/d) <200 15 mg Initial Meth Dose Increment Example 5 mg q 8 h 5– 7 d Mor 90 mg/d Meth 5 mg q 8 h 200– 500 ~7% Morph dose Calculated dose given q 8 h 5– 7 d 300 mg Mor = 21 mg/d Met 7. 5 mg q 8 h >500 1/3 calculated Add 1/3 dose q 8 h dose q 5 d; ↓ previous opioid q 5 d ~7% Morph dose Complete conversion in 15 d Goodman F, et al. Methadone Dosing Recommendations for Treatment of Chronic Pain. Available at: http: //www. pbm. va. gov/monitoring/Methadone%20 Dosing%20 Final%20(Rev%20081103). pdf. Accessed November 28, 2006. [Evidence Level C] © 2005 Rehabilitation Institute of Chicago
Methadone Dosing: Gradual Conversion Mor-E (mg/d) Calculate Meth (mg/d) <200 15 mg Initial Meth Dose Increment Example 5 mg q 8 h 5– 7 d Mor 90 mg/d Meth 5 mg q 8 h 200– 500 ~7% Morph dose Calculated dose given q 8 h 5– 7 d 300 mg Mor = 21 mg/d Met 7. 5 mg q 8 h >500 1/3 calculated Add 1/3 dose q 8 h dose q 5 d; ↓ previous opioid q 5 d ~7% Morph dose Complete conversion in 15 d Goodman F, et al. Methadone Dosing Recommendations for Treatment of Chronic Pain. Available at: http: //www. pbm. va. gov/monitoring/Methadone%20 Dosing%20 Final%20(Rev%20081103). pdf. Accessed November 28, 2006. [Evidence Level C] © 2005 Rehabilitation Institute of Chicago
 Methadone: prolonged QT and Torsade de Pointes (Td. P) § Risk Factors: heart disease, hypokalemia, hypomagnesemia, concomitant CYP 3 A 4 inhibitors § Med. Watch System (N= 5503) — <1% with Td. P or PQ prolongation — Average dose 410 /+ 349/day — 30% within recommended range (60 100 mg/day) — 75% with risk factors Pearson E. Pharmacoepidem Drug Saf 2005: 14: 747 53.
Methadone: prolonged QT and Torsade de Pointes (Td. P) § Risk Factors: heart disease, hypokalemia, hypomagnesemia, concomitant CYP 3 A 4 inhibitors § Med. Watch System (N= 5503) — <1% with Td. P or PQ prolongation — Average dose 410 /+ 349/day — 30% within recommended range (60 100 mg/day) — 75% with risk factors Pearson E. Pharmacoepidem Drug Saf 2005: 14: 747 53.
 “Adverse Effects Cases Risk Stratification American Greetings.
“Adverse Effects Cases Risk Stratification American Greetings.
 Formulation Strategies Intended to Reduce Abuse Physical Approach 1, 2 Pharmacologic Approach 1, 2 Examples include • Advanced dispensing formulations • Tamper resistant formulations Examples include • Addition of antagonist • Prodrug formulation Aversive Approach 1, 2 Examples include • Addition of non treatment related ingredients such as niacin or capsaicin 1. Gershell L et al. Nat Rev Drug Discov. 2006; 5(11): 889 890. 2. Katz NP et al. Clin J Pain. 2007; 23(8): 648 660.
Formulation Strategies Intended to Reduce Abuse Physical Approach 1, 2 Pharmacologic Approach 1, 2 Examples include • Advanced dispensing formulations • Tamper resistant formulations Examples include • Addition of antagonist • Prodrug formulation Aversive Approach 1, 2 Examples include • Addition of non treatment related ingredients such as niacin or capsaicin 1. Gershell L et al. Nat Rev Drug Discov. 2006; 5(11): 889 890. 2. Katz NP et al. Clin J Pain. 2007; 23(8): 648 660.
 EMBEDA™ - 01 How Does the EMBEDA™ Pharmacologic Abuse-deterrent Technology Work? AL O MORPHINE SULFATE NALTREXONE CORE Chewing or Crushing Force Naltrexone Core Releases and Mixes Naltrexone Morphine 1. If taken as directed, EMBEDA™ is intended to provide effective pain relief with long-acting morphine pellets; the naltrexone passes out of the body without effect 2. If EMBEDA™ is crushed, chewed or extracted: A. the naltrexone (opioid antagonist) releases and mixes with morphine B. the morphine-naltrexone mixture is Opioid Receptor intended to prevent euphoria from morphine by competing for the same opioid receptor in the brain…thus deterring abuse of the product
EMBEDA™ - 01 How Does the EMBEDA™ Pharmacologic Abuse-deterrent Technology Work? AL O MORPHINE SULFATE NALTREXONE CORE Chewing or Crushing Force Naltrexone Core Releases and Mixes Naltrexone Morphine 1. If taken as directed, EMBEDA™ is intended to provide effective pain relief with long-acting morphine pellets; the naltrexone passes out of the body without effect 2. If EMBEDA™ is crushed, chewed or extracted: A. the naltrexone (opioid antagonist) releases and mixes with morphine B. the morphine-naltrexone mixture is Opioid Receptor intended to prevent euphoria from morphine by competing for the same opioid receptor in the brain…thus deterring abuse of the product
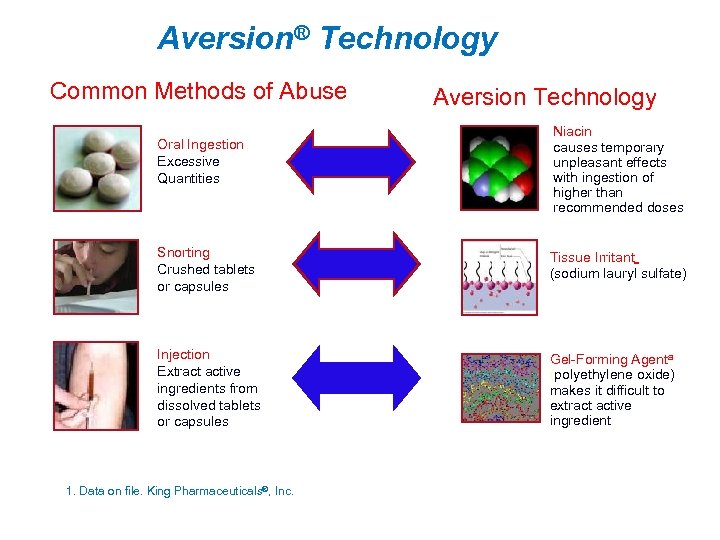 Aversion® Technology Common Methods of Abuse Oral Ingestion Excessive Quantities Aversion Technology Niacin causes temporary unpleasant effects with ingestion of higher than recommended doses Snorting Crushed tablets or capsules Injection Extract active ingredients from dissolved tablets or capsules a Two Tissue Irritant (sodium lauryl sulfate) Gel Forming Agenta (polyethylene oxide) makes it difficult to extract active ingredient additional excipients synergize polyethylene oxide 1. Data on file. King Pharmaceuticals®, Inc.
Aversion® Technology Common Methods of Abuse Oral Ingestion Excessive Quantities Aversion Technology Niacin causes temporary unpleasant effects with ingestion of higher than recommended doses Snorting Crushed tablets or capsules Injection Extract active ingredients from dissolved tablets or capsules a Two Tissue Irritant (sodium lauryl sulfate) Gel Forming Agenta (polyethylene oxide) makes it difficult to extract active ingredient additional excipients synergize polyethylene oxide 1. Data on file. King Pharmaceuticals®, Inc.
 Adverse Effects: Nausea and Vomiting § Chemoreceptor trigger zone (CTZ) § Cortex § Peripheral pathways § Vestibular sensitivity
Adverse Effects: Nausea and Vomiting § Chemoreceptor trigger zone (CTZ) § Cortex § Peripheral pathways § Vestibular sensitivity
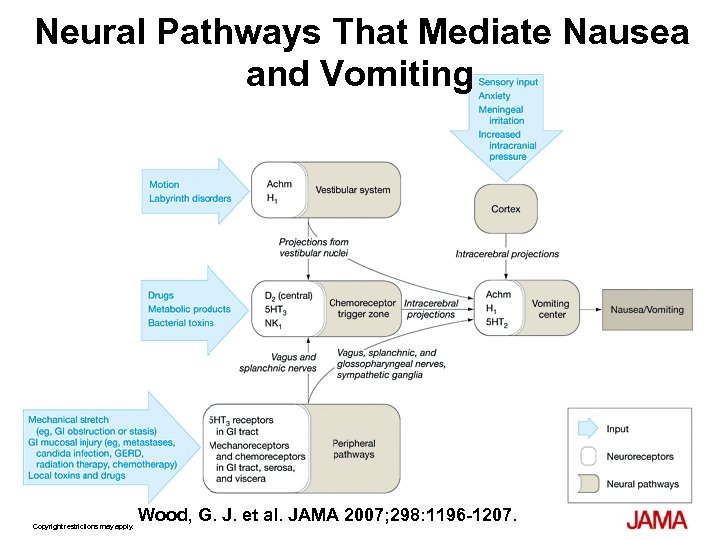 Neural Pathways That Mediate Nausea and Vomiting Copyright restrictions may apply. Wood, G. J. et al. JAMA 2007; 298: 1196 -1207.
Neural Pathways That Mediate Nausea and Vomiting Copyright restrictions may apply. Wood, G. J. et al. JAMA 2007; 298: 1196 -1207.
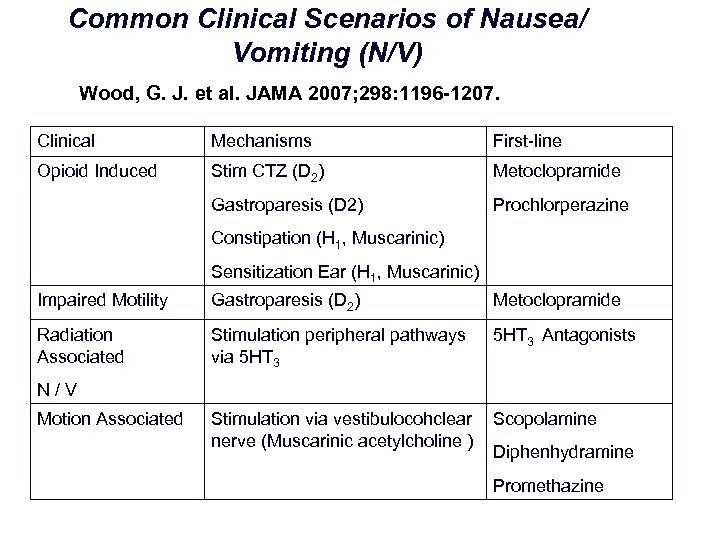 Common Clinical Scenarios of Nausea/ Vomiting (N/V) Wood, G. J. et al. JAMA 2007; 298: 1196 -1207. Clinical Mechanisms First line Opioid Induced Stim CTZ (D 2) Metoclopramide Gastroparesis (D 2) Prochlorperazine Constipation (H 1, Muscarinic) Sensitization Ear (H 1, Muscarinic) Impaired Motility Gastroparesis (D 2) Metoclopramide Radiation Associated Stimulation peripheral pathways via 5 HT 3 Antagonists Stimulation via vestibulocohclear nerve (Muscarinic acetylcholine ) Scopolamine N/V Motion Associated Diphenhydramine Promethazine
Common Clinical Scenarios of Nausea/ Vomiting (N/V) Wood, G. J. et al. JAMA 2007; 298: 1196 -1207. Clinical Mechanisms First line Opioid Induced Stim CTZ (D 2) Metoclopramide Gastroparesis (D 2) Prochlorperazine Constipation (H 1, Muscarinic) Sensitization Ear (H 1, Muscarinic) Impaired Motility Gastroparesis (D 2) Metoclopramide Radiation Associated Stimulation peripheral pathways via 5 HT 3 Antagonists Stimulation via vestibulocohclear nerve (Muscarinic acetylcholine ) Scopolamine N/V Motion Associated Diphenhydramine Promethazine
 Nausea: Treatment § Antihistamines (H 1) — Diphenhydramin (Benadryl): 25 50 mg PO — Scopolamine: 1. 5 mg TD patch Q 72 hrs § Antipsychotics (D 2/CTZ) — Haloperidol (Haldol): 0. 5 – 4 mg PO — Prochlorperazine (Compazine): 5 10 mg PO; 25 mg PR — Promethazine (H 1) (Phenergan): 12. 5 25 mg PO; 25 mg PR § Prokinetic agents (D 2) — Metoclopramide (Reglan): 5 20 mg PO/IV § Serotonin Antagonist (5 HT 3) — Ondansetron (Zofran): 4 8 mg PO or IV Swegle JM, Logeman CL. Amer Fam Phys 2006; 74: 1347 54
Nausea: Treatment § Antihistamines (H 1) — Diphenhydramin (Benadryl): 25 50 mg PO — Scopolamine: 1. 5 mg TD patch Q 72 hrs § Antipsychotics (D 2/CTZ) — Haloperidol (Haldol): 0. 5 – 4 mg PO — Prochlorperazine (Compazine): 5 10 mg PO; 25 mg PR — Promethazine (H 1) (Phenergan): 12. 5 25 mg PO; 25 mg PR § Prokinetic agents (D 2) — Metoclopramide (Reglan): 5 20 mg PO/IV § Serotonin Antagonist (5 HT 3) — Ondansetron (Zofran): 4 8 mg PO or IV Swegle JM, Logeman CL. Amer Fam Phys 2006; 74: 1347 54
 Opoid Induced Sedation § Methylphenidate 1, 2 (Sch II): 5 10 mg TID PRN § Dextroamphetamine (Sch II): 2. 5 – 7. 5 mg BID, PRN § Caffeine § Donepezil (Aricept)3, 4: 2. 5 – 10 mg/day § Modafinil (Provigil) (Sch IV): 100 – 600 mg/ day § Differential Diagnosis 5: depression, hypothyroidism, opioid dose, opioid adverse effect, endocrine, sleep disorder 1. Bruera E, et al. J Pain Symptom Manage 1989; 4: 3 6. 2. Bruera e, et al. Pain 1992; 50: 75 7. 3. Slatkin et al. J Pain Symptom Manage 2001; 21: 425 38. 4. Bruera E, et al. J Pain Symptom Manage 2003; 26: 1049 54. 5. Reissig JE, Rybarczyk A. Ann Pharmacother 2005; 39: 727 31.
Opoid Induced Sedation § Methylphenidate 1, 2 (Sch II): 5 10 mg TID PRN § Dextroamphetamine (Sch II): 2. 5 – 7. 5 mg BID, PRN § Caffeine § Donepezil (Aricept)3, 4: 2. 5 – 10 mg/day § Modafinil (Provigil) (Sch IV): 100 – 600 mg/ day § Differential Diagnosis 5: depression, hypothyroidism, opioid dose, opioid adverse effect, endocrine, sleep disorder 1. Bruera E, et al. J Pain Symptom Manage 1989; 4: 3 6. 2. Bruera e, et al. Pain 1992; 50: 75 7. 3. Slatkin et al. J Pain Symptom Manage 2001; 21: 425 38. 4. Bruera E, et al. J Pain Symptom Manage 2003; 26: 1049 54. 5. Reissig JE, Rybarczyk A. Ann Pharmacother 2005; 39: 727 31.
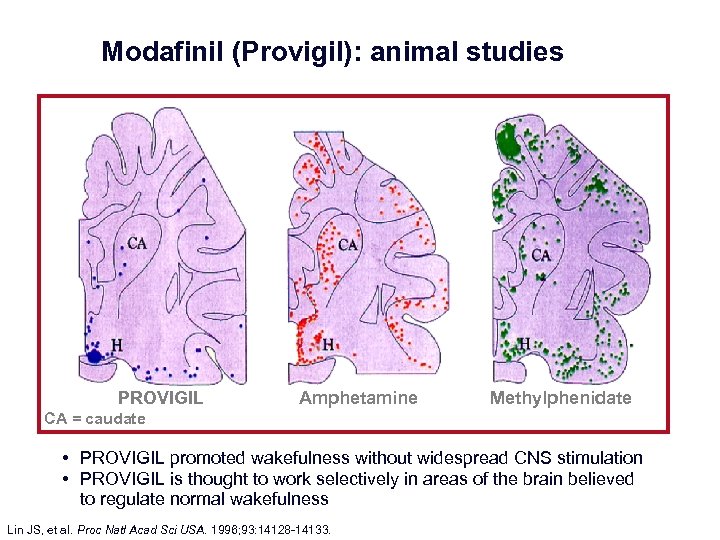 Modafinil (Provigil): animal studies PROVIGILhypothalamus Amphetamine H= Methylphenidate CA = caudate • PROVIGIL promoted wakefulness without widespread CNS stimulation • PROVIGIL is thought to work selectively in areas of the brain believed to regulate normal wakefulness Lin JS, et al. Proc Natl Acad Sci USA. 1996; 93: 14128 14133.
Modafinil (Provigil): animal studies PROVIGILhypothalamus Amphetamine H= Methylphenidate CA = caudate • PROVIGIL promoted wakefulness without widespread CNS stimulation • PROVIGIL is thought to work selectively in areas of the brain believed to regulate normal wakefulness Lin JS, et al. Proc Natl Acad Sci USA. 1996; 93: 14128 14133.
 Modafinil (Provigil) § Mechanisms: decrease GABA release in cortex, weak dopamine release, amygdala? § Scheduled IV: 100 mg, 200 mg § FDA: narcolepsy, shift work sleep disorder § Webster L et al, Pain Medicine 2003; 4: 135 40 — Retrospective, N = 11, Epworth Sleepiness Scale (ESS) — Dose range: 264 mg – 427 mg/ day — Opioid dose: 536 mg – 810 mg/ day
Modafinil (Provigil) § Mechanisms: decrease GABA release in cortex, weak dopamine release, amygdala? § Scheduled IV: 100 mg, 200 mg § FDA: narcolepsy, shift work sleep disorder § Webster L et al, Pain Medicine 2003; 4: 135 40 — Retrospective, N = 11, Epworth Sleepiness Scale (ESS) — Dose range: 264 mg – 427 mg/ day — Opioid dose: 536 mg – 810 mg/ day
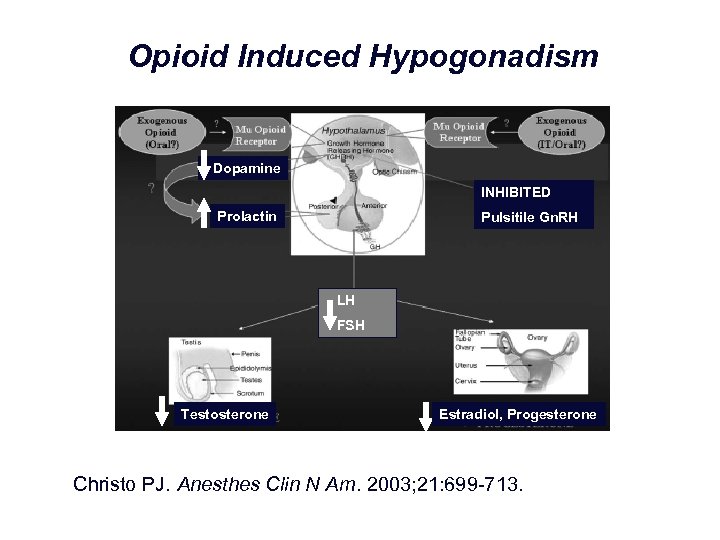 Opioid Induced Hypogonadism Dopamine INHIBITED Prolactin Pulsitile Gn. RH LH FSH Testosterone Estradiol, Progesterone Christo PJ. Anesthes Clin N Am. 2003; 21: 699 713.
Opioid Induced Hypogonadism Dopamine INHIBITED Prolactin Pulsitile Gn. RH LH FSH Testosterone Estradiol, Progesterone Christo PJ. Anesthes Clin N Am. 2003; 21: 699 713.
 Hypogonadism: (OPIAD) § Sexual dysfunction, infertility Lab § Depression § Free testosterone (FT) § Decreased energy level § Total testosterone (TT) § Muscle wasting and osteoporosis § Luetinizing hormone (LH) § Lowered pain threshold § Estradiol (E 2) § Impaired wound healing § Prostate specific antigen (PSA) § “Opioid Induced Androgen Deficiency” (OPIAD) § Follicle stimulating (FSH)
Hypogonadism: (OPIAD) § Sexual dysfunction, infertility Lab § Depression § Free testosterone (FT) § Decreased energy level § Total testosterone (TT) § Muscle wasting and osteoporosis § Luetinizing hormone (LH) § Lowered pain threshold § Estradiol (E 2) § Impaired wound healing § Prostate specific antigen (PSA) § “Opioid Induced Androgen Deficiency” (OPIAD) § Follicle stimulating (FSH)
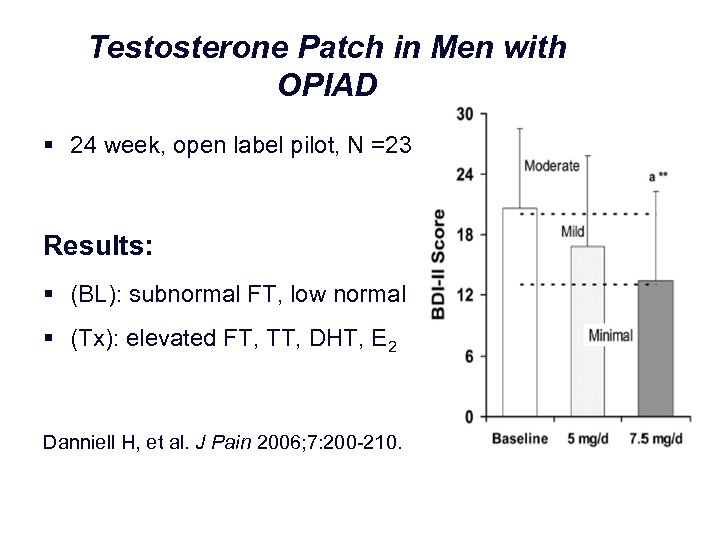 Testosterone Patch in Men with OPIAD § 24 week, open label pilot, N =23 Results: § (BL): subnormal FT, low normal LH § (Tx): elevated FT, TT, DHT, E 2 Danniell H, et al. J Pain 2006; 7: 200 210.
Testosterone Patch in Men with OPIAD § 24 week, open label pilot, N =23 Results: § (BL): subnormal FT, low normal LH § (Tx): elevated FT, TT, DHT, E 2 Danniell H, et al. J Pain 2006; 7: 200 210.
 Opioid Induced Sensorineural Hearing Loss § Hydrocodone/ APAP abuse 1 15 60 tablets/ day cochlear toxicity • Hydrocodone/APAP 2 50 300 mg/ day association with Hep C Severe impairment 1. Oh AK. et al. Neurology 2000; 54: 2345. § Propoxyphene abuse 3 2. Ho et al. Pain Physician 2007; 10: 467 72. 3. Harell M. et al. Laryngoscope 1978; 88: 1518 21.
Opioid Induced Sensorineural Hearing Loss § Hydrocodone/ APAP abuse 1 15 60 tablets/ day cochlear toxicity • Hydrocodone/APAP 2 50 300 mg/ day association with Hep C Severe impairment 1. Oh AK. et al. Neurology 2000; 54: 2345. § Propoxyphene abuse 3 2. Ho et al. Pain Physician 2007; 10: 467 72. 3. Harell M. et al. Laryngoscope 1978; 88: 1518 21.
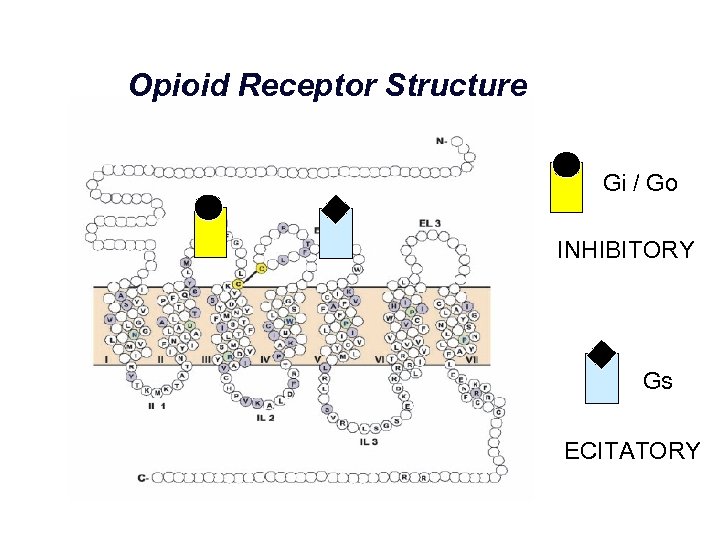 Opioid Receptor Structure Gi / Go INHIBITORY Gs ECITATORY Waldhoer M, et al. Annu Rev Biochem. 2004; 73: 953 990.
Opioid Receptor Structure Gi / Go INHIBITORY Gs ECITATORY Waldhoer M, et al. Annu Rev Biochem. 2004; 73: 953 990.
 Opioid Induced Abnormal Pain Sensitivity • Desensitization process leading to opioid tolerance • Sensitization (pronociceptive) process leading to opioid induced pain sensitivity Increased spinal dynorphin Descending facilitation Glutamatergic system © 2005 Rehabilitation Institute of Chicago
Opioid Induced Abnormal Pain Sensitivity • Desensitization process leading to opioid tolerance • Sensitization (pronociceptive) process leading to opioid induced pain sensitivity Increased spinal dynorphin Descending facilitation Glutamatergic system © 2005 Rehabilitation Institute of Chicago
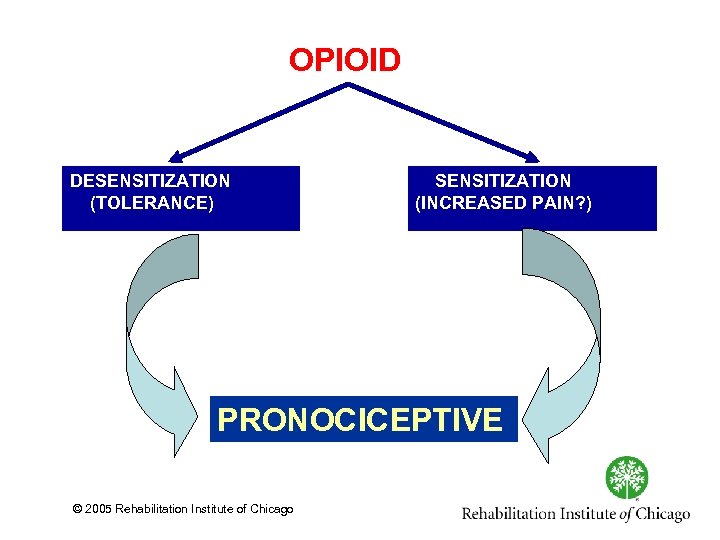 OPIOID DESENSITIZATION (TOLERANCE) SENSITIZATION (INCREASED PAIN? ) PRONOCICEPTIVE © 2005 Rehabilitation Institute of Chicago
OPIOID DESENSITIZATION (TOLERANCE) SENSITIZATION (INCREASED PAIN? ) PRONOCICEPTIVE © 2005 Rehabilitation Institute of Chicago
 Opioid Induced Hyperalgesia (OIH) • Mao, J (2002) • Post Surgical Studies: Increased pain and analgesic use (Guignard B, 2006) • Problems: opioid naïve patients, post surgical, methadone maintenance • (Cohen S, et al 2008) – 355 patients, interventional procedures – Results: enhanced pain perception in opioid patients with local lidocaine prep – Correlated with dose and duration of Tx © 2005 Rehabilitation Institute of Chicago
Opioid Induced Hyperalgesia (OIH) • Mao, J (2002) • Post Surgical Studies: Increased pain and analgesic use (Guignard B, 2006) • Problems: opioid naïve patients, post surgical, methadone maintenance • (Cohen S, et al 2008) – 355 patients, interventional procedures – Results: enhanced pain perception in opioid patients with local lidocaine prep – Correlated with dose and duration of Tx © 2005 Rehabilitation Institute of Chicago
 “Men in general are quick to believe that with which they wish to be true. ” Julius Caesar © 2005 Rehabilitation Institute of Chicago
“Men in general are quick to believe that with which they wish to be true. ” Julius Caesar © 2005 Rehabilitation Institute of Chicago
 TOLERANCE and DEPENDENCE © 2005 Rehabilitation Institute of Chicago X = ADDICTION
TOLERANCE and DEPENDENCE © 2005 Rehabilitation Institute of Chicago X = ADDICTION
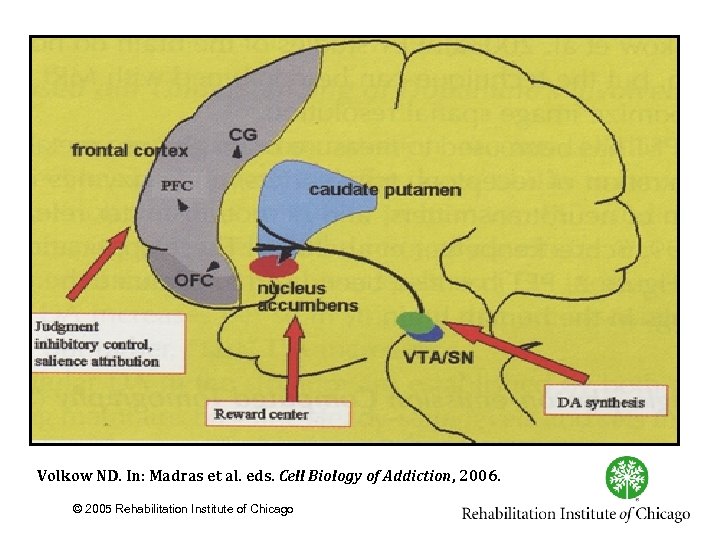 Volkow ND. In: Madras et al. eds. Cell Biology of Addiction, 2006. © 2005 Rehabilitation Institute of Chicago
Volkow ND. In: Madras et al. eds. Cell Biology of Addiction, 2006. © 2005 Rehabilitation Institute of Chicago
 “Pseudoaddiction” Inadequate Pain Management FRUSTRATION ANGER PATIENT TEAM ISOLATION AVOIDANCE CRISIS © 2005 Rehabilitation Institute of Chicago Weissman, Haddox, Pain (1989)
“Pseudoaddiction” Inadequate Pain Management FRUSTRATION ANGER PATIENT TEAM ISOLATION AVOIDANCE CRISIS © 2005 Rehabilitation Institute of Chicago Weissman, Haddox, Pain (1989)
 Predicting aberrant behaviors • High risk – Family history of substance abuse – Legal problems – Drug or alcohol abuse • Other – Cigarette use, higher opioid dose, less opioid reported side effects, MVA – Mental health disorders Michna E, et al. J Pain Sym Management 2004; 28: 250 -8. © 2005 Rehabilitation Institute of Chicago
Predicting aberrant behaviors • High risk – Family history of substance abuse – Legal problems – Drug or alcohol abuse • Other – Cigarette use, higher opioid dose, less opioid reported side effects, MVA – Mental health disorders Michna E, et al. J Pain Sym Management 2004; 28: 250 -8. © 2005 Rehabilitation Institute of Chicago
 Aberrant Drug Related Behavior: Implications • Differential diagnosis of aberrant drug related behavior – Addiction – Pseudoaddiction – Other psychiatric disorders • Axis I and Axis II disorders • Mild encephalopathy • Family disturbances – Criminal intent: diversion Portenoy RK, et al. In Lowinson JH, et al. (eds): Comprehensive Textbook of Substance Abuse, Fourth Edition. Baltimore: Williams and Wilkins, 2005, pp. 863 903. © 2005 Rehabilitation Institute of Chicago
Aberrant Drug Related Behavior: Implications • Differential diagnosis of aberrant drug related behavior – Addiction – Pseudoaddiction – Other psychiatric disorders • Axis I and Axis II disorders • Mild encephalopathy • Family disturbances – Criminal intent: diversion Portenoy RK, et al. In Lowinson JH, et al. (eds): Comprehensive Textbook of Substance Abuse, Fourth Edition. Baltimore: Williams and Wilkins, 2005, pp. 863 903. © 2005 Rehabilitation Institute of Chicago
 Predicting opioid misuse at 1 year Misuse (32%) • Predictors cocaine abuse DUI conviction male cannabinoids “Opioid misuse” • • • Ives TJ, et al. BMC Health Ser Res 2006; 6: 46. © 2005 Rehabilitation Institute of Chicago ( ) tox screen (+) for other Multiple providers Diversion Forgery Stimulant use
Predicting opioid misuse at 1 year Misuse (32%) • Predictors cocaine abuse DUI conviction male cannabinoids “Opioid misuse” • • • Ives TJ, et al. BMC Health Ser Res 2006; 6: 46. © 2005 Rehabilitation Institute of Chicago ( ) tox screen (+) for other Multiple providers Diversion Forgery Stimulant use
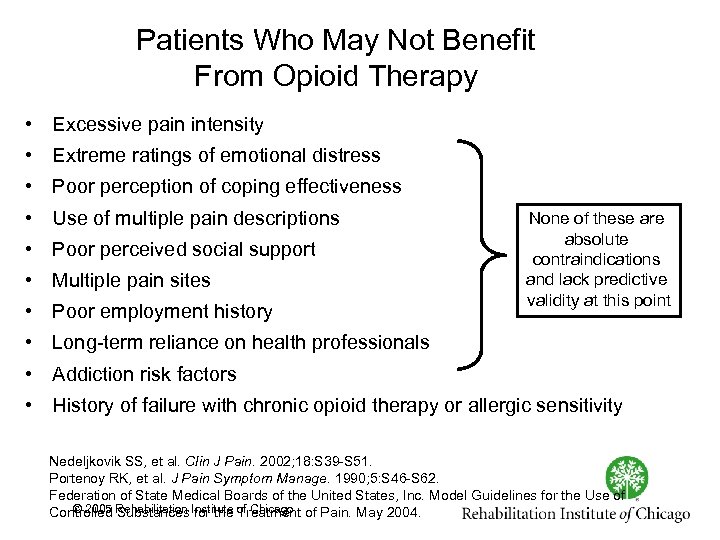 Patients Who May Not Benefit From Opioid Therapy • Excessive pain intensity • Extreme ratings of emotional distress • Poor perception of coping effectiveness • Use of multiple pain descriptions • Poor perceived social support • Multiple pain sites • Poor employment history None of these are absolute contraindications and lack predictive validity at this point • Long term reliance on health professionals • Addiction risk factors • History of failure with chronic opioid therapy or allergic sensitivity Nedeljkovik SS, et al. Clin J Pain. 2002; 18: S 39 S 51. Portenoy RK, et al. J Pain Symptom Manage. 1990; 5: S 46 S 62. Federation of State Medical Boards of the United States, Inc. Model Guidelines for the Use of © 2005 Substances for the Treatment Controlled Rehabilitation Institute of Chicago of Pain. May 2004.
Patients Who May Not Benefit From Opioid Therapy • Excessive pain intensity • Extreme ratings of emotional distress • Poor perception of coping effectiveness • Use of multiple pain descriptions • Poor perceived social support • Multiple pain sites • Poor employment history None of these are absolute contraindications and lack predictive validity at this point • Long term reliance on health professionals • Addiction risk factors • History of failure with chronic opioid therapy or allergic sensitivity Nedeljkovik SS, et al. Clin J Pain. 2002; 18: S 39 S 51. Portenoy RK, et al. J Pain Symptom Manage. 1990; 5: S 46 S 62. Federation of State Medical Boards of the United States, Inc. Model Guidelines for the Use of © 2005 Substances for the Treatment Controlled Rehabilitation Institute of Chicago of Pain. May 2004.
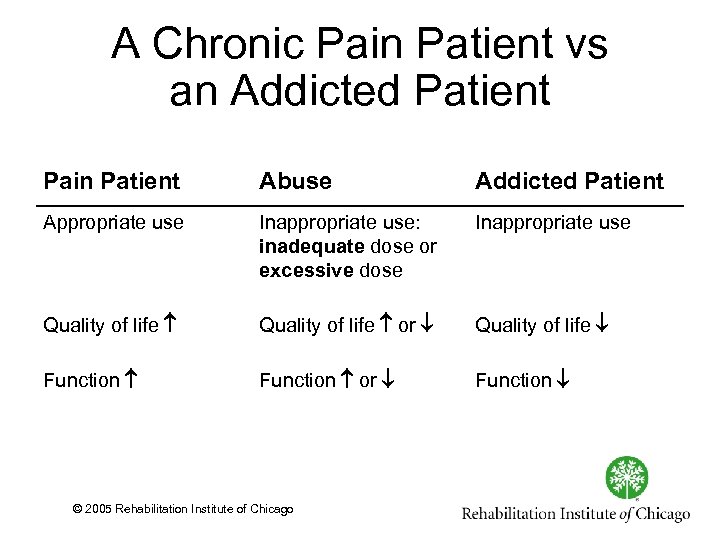 A Chronic Pain Patient vs an Addicted Patient Pain Patient Abuse Addicted Patient Appropriate use Inappropriate use: inadequate dose or excessive dose Inappropriate use Quality of life or Quality of life Function or Function © 2005 Rehabilitation Institute of Chicago
A Chronic Pain Patient vs an Addicted Patient Pain Patient Abuse Addicted Patient Appropriate use Inappropriate use: inadequate dose or excessive dose Inappropriate use Quality of life or Quality of life Function or Function © 2005 Rehabilitation Institute of Chicago
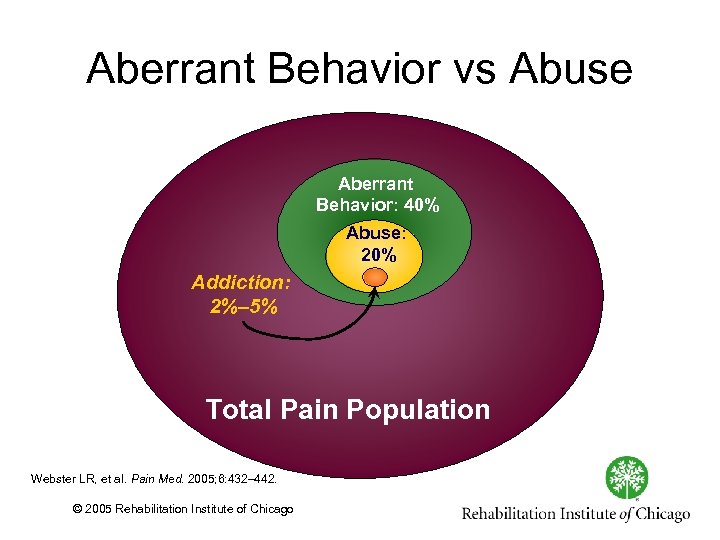 Aberrant Behavior vs Abuse Aberrant Behavior: 40% Abuse: 20% Addiction: 2%– 5% Total Pain Population Webster LR, et al. Pain Med. 2005; 6: 432– 442. © 2005 Rehabilitation Institute of Chicago Webster LR, Webster RM. Pain Med. 2005; 6: 432– 442;
Aberrant Behavior vs Abuse Aberrant Behavior: 40% Abuse: 20% Addiction: 2%– 5% Total Pain Population Webster LR, et al. Pain Med. 2005; 6: 432– 442. © 2005 Rehabilitation Institute of Chicago Webster LR, Webster RM. Pain Med. 2005; 6: 432– 442;
 Summary • Remember opioid pharmacokinetics • Side effect management important to improving chances of success • Opioid hyperalgesia • Endocrine effects • Risk stratify patients © 2005 Rehabilitation Institute of Chicago
Summary • Remember opioid pharmacokinetics • Side effect management important to improving chances of success • Opioid hyperalgesia • Endocrine effects • Risk stratify patients © 2005 Rehabilitation Institute of Chicago


