Lecture_6_Octahedral.ppt
- Количество слайдов: 57
![Octahedral rhenium(III) cluster complexes [{Re 6 Q 8}L 6] Q=S, Se, Te, Cl, Br, Octahedral rhenium(III) cluster complexes [{Re 6 Q 8}L 6] Q=S, Se, Te, Cl, Br,](https://present5.com/presentation/5652994_182163104/image-1.jpg) Octahedral rhenium(III) cluster complexes [{Re 6 Q 8}L 6] Q=S, Se, Te, Cl, Br, O L=Cl, Br, I, OH, CN, SCN… N-, P-, O-, S- donor organic ligands Cluster cores: {Re 6 Se 4 Br 4}6+ {Re 5 Os. Se 8}3+ { Re 6 Se 8}2+ / 3+ L VEC=24 / 23 e per Re 6 {Re 5 Mo. Se 8}+ Cluster complexes: [Re 6 S 8(H 2 O)6]2+ cationic complex [Re 6 S 8(H 2 O)4(OH)2]0 neutral complex [Re 6 S 8 Br 6]4 - anionic complex [Re 6 S 8(SO 3)6]10 - anionic complex
Octahedral rhenium(III) cluster complexes [{Re 6 Q 8}L 6] Q=S, Se, Te, Cl, Br, O L=Cl, Br, I, OH, CN, SCN… N-, P-, O-, S- donor organic ligands Cluster cores: {Re 6 Se 4 Br 4}6+ {Re 5 Os. Se 8}3+ { Re 6 Se 8}2+ / 3+ L VEC=24 / 23 e per Re 6 {Re 5 Mo. Se 8}+ Cluster complexes: [Re 6 S 8(H 2 O)6]2+ cationic complex [Re 6 S 8(H 2 O)4(OH)2]0 neutral complex [Re 6 S 8 Br 6]4 - anionic complex [Re 6 S 8(SO 3)6]10 - anionic complex
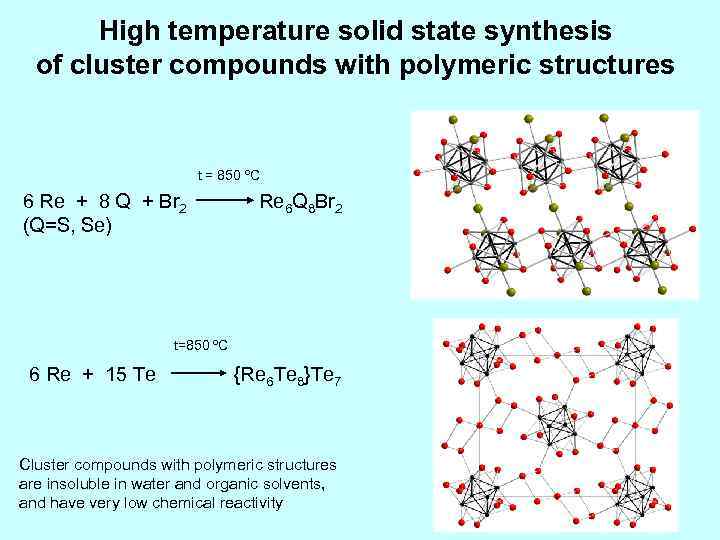 High temperature solid state synthesis of cluster compounds with polymeric structures t = 850 ºC 6 Re + 8 Q + Br 2 Re 6 Q 8 Br 2 (Q=S, Se) t=850 ºC 6 Re + 15 Te {Re 6 Te 8}Te 7 Cluster compounds with polymeric structures are insoluble in water and organic solvents, and have very low chemical reactivity
High temperature solid state synthesis of cluster compounds with polymeric structures t = 850 ºC 6 Re + 8 Q + Br 2 Re 6 Q 8 Br 2 (Q=S, Se) t=850 ºC 6 Re + 15 Te {Re 6 Te 8}Te 7 Cluster compounds with polymeric structures are insoluble in water and organic solvents, and have very low chemical reactivity
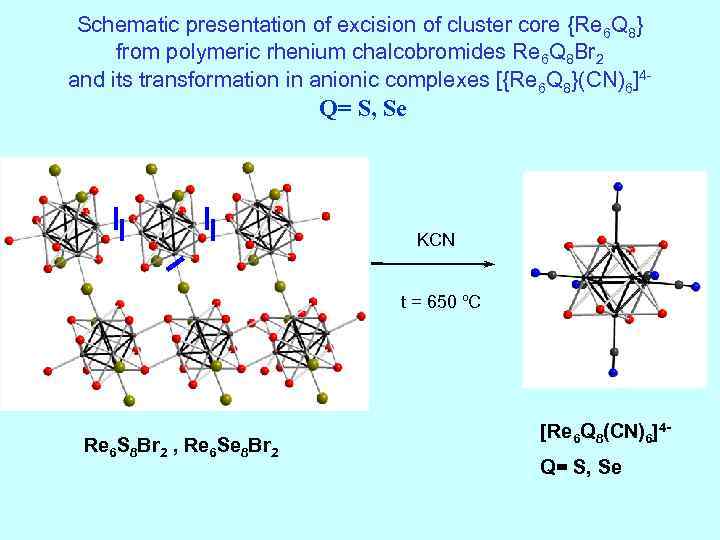 Schematic presentation of excision of cluster core {Re 6 Q 8} from polymeric rhenium chalcobromides Re 6 Q 8 Br 2 and its transformation in anionic complexes [{Re 6 Q 8}(CN)6]4 - Q= S, Se KCN t = 650 ºC Re 6 S 8 Br 2 , Re 6 Se 8 Br 2 [Re 6 Q 8(CN)6]4 - Q= S, Se
Schematic presentation of excision of cluster core {Re 6 Q 8} from polymeric rhenium chalcobromides Re 6 Q 8 Br 2 and its transformation in anionic complexes [{Re 6 Q 8}(CN)6]4 - Q= S, Se KCN t = 650 ºC Re 6 S 8 Br 2 , Re 6 Se 8 Br 2 [Re 6 Q 8(CN)6]4 - Q= S, Se
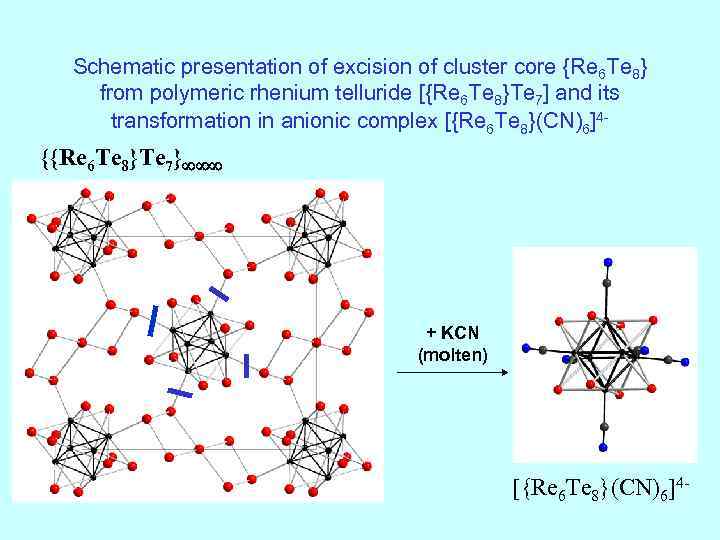 Schematic presentation of excision of cluster core {Re 6 Te 8} from polymeric rhenium telluride [{Re 6 Te 8}Te 7] and its transformation in anionic complex [{Re 6 Te 8}(CN)6]4 - {{Re 6 Te 8}Te 7} + KCN (molten) [{Re 6 Te 8}(CN)6]4 -
Schematic presentation of excision of cluster core {Re 6 Te 8} from polymeric rhenium telluride [{Re 6 Te 8}Te 7] and its transformation in anionic complex [{Re 6 Te 8}(CN)6]4 - {{Re 6 Te 8}Te 7} + KCN (molten) [{Re 6 Te 8}(CN)6]4 -
![Discovery of octahedral rhenium cluster chalcocyanide complexes [Re 6 Q 8(CN)6]4 - (Q=S, Se, Discovery of octahedral rhenium cluster chalcocyanide complexes [Re 6 Q 8(CN)6]4 - (Q=S, Se,](https://present5.com/presentation/5652994_182163104/image-5.jpg) Discovery of octahedral rhenium cluster chalcocyanide complexes [Re 6 Q 8(CN)6]4 - (Q=S, Se, Te) Excision of {Re 6 Q 8} cluster core from polymeric compounds: 650 o. C [{Re 6 Te 8}(CN)6]4 - {Re 6 Te 8}Te 7 + KCN (molten) A. Slougui, Yu. V. Mironov, A. Perrin, V. E. Fedorov, Croatica Chemica Acta, 1995, v. 68, pp. 885 -890. Yu. V. Mironov, A. V. Virovets, V. E. Fedorov, N. V. Podberezskaya, O. V. Shishkin, Yu. T. Struchkov, Polyhedron, 1995, v. 14, pp. 3171 -3173. 650 o. C {Re 6 Q 8}Br 2 + KCN (molten) Q=S, Se N. G. Naumov, A. V. Virovets, N. V. Podberezskaya, V. E. Fedorov, Russ. J. Struct. Chem. , 1997, v. 38, pp. 857 -862. H. Imoto, N. G. Naumov, A. V. Virovets, T. Saito, V. E. Fedorov, Russ. J. Struct. Chem. , 1998, v. 39, pp. 720 -727. [{Re 6 Q 8}(CN)6]4 -
Discovery of octahedral rhenium cluster chalcocyanide complexes [Re 6 Q 8(CN)6]4 - (Q=S, Se, Te) Excision of {Re 6 Q 8} cluster core from polymeric compounds: 650 o. C [{Re 6 Te 8}(CN)6]4 - {Re 6 Te 8}Te 7 + KCN (molten) A. Slougui, Yu. V. Mironov, A. Perrin, V. E. Fedorov, Croatica Chemica Acta, 1995, v. 68, pp. 885 -890. Yu. V. Mironov, A. V. Virovets, V. E. Fedorov, N. V. Podberezskaya, O. V. Shishkin, Yu. T. Struchkov, Polyhedron, 1995, v. 14, pp. 3171 -3173. 650 o. C {Re 6 Q 8}Br 2 + KCN (molten) Q=S, Se N. G. Naumov, A. V. Virovets, N. V. Podberezskaya, V. E. Fedorov, Russ. J. Struct. Chem. , 1997, v. 38, pp. 857 -862. H. Imoto, N. G. Naumov, A. V. Virovets, T. Saito, V. E. Fedorov, Russ. J. Struct. Chem. , 1998, v. 39, pp. 720 -727. [{Re 6 Q 8}(CN)6]4 -
![Chalcocyanide octahedral cluster complexes [Re 6 Q 8(CN)6]n- (Q=S, Se, Te; n=3, 4) Chalcocyanides Chalcocyanide octahedral cluster complexes [Re 6 Q 8(CN)6]n- (Q=S, Se, Te; n=3, 4) Chalcocyanides](https://present5.com/presentation/5652994_182163104/image-6.jpg) Chalcocyanide octahedral cluster complexes [Re 6 Q 8(CN)6]n- (Q=S, Se, Te; n=3, 4) Chalcocyanides with homometallic cluster core: [Re 6 Q 8(CN)6]4 - (Q=S, Se, Te) VEC=24, diamagnetic [Re 6 Q 8(CN)6]3 - (Q=S, Se, Te) VEC=23, paramagnetic Chalcocyanides with heterometallic cluster cores: [Re 5 Mo. S 8(CN)6]5 - VEC=24, diamagnetic [Re 4 Mo 2 S 8(CN)6]5 - VEC=23, paramagnetic [Re 5 Os. Se 8(CN)6]3 - VEC=24, diamagnetic [Re 4 Os 2(CN)6]2 - VEC=24, diamagnetic Mixed-ligands cluster complexes: trans-[Re 6 S 8(CN)2 S 4]4 - VEC=24, diamagnetic trans-[Re 6 Se 8(CN)4 Br 2]4 - VEC=24, diamagnetic [Re 6(Q 8 -x. Q’x)(CN)6]4 - (Q, Q’=S, Se, Te) VEC=24, diamagnetic etc.
Chalcocyanide octahedral cluster complexes [Re 6 Q 8(CN)6]n- (Q=S, Se, Te; n=3, 4) Chalcocyanides with homometallic cluster core: [Re 6 Q 8(CN)6]4 - (Q=S, Se, Te) VEC=24, diamagnetic [Re 6 Q 8(CN)6]3 - (Q=S, Se, Te) VEC=23, paramagnetic Chalcocyanides with heterometallic cluster cores: [Re 5 Mo. S 8(CN)6]5 - VEC=24, diamagnetic [Re 4 Mo 2 S 8(CN)6]5 - VEC=23, paramagnetic [Re 5 Os. Se 8(CN)6]3 - VEC=24, diamagnetic [Re 4 Os 2(CN)6]2 - VEC=24, diamagnetic Mixed-ligands cluster complexes: trans-[Re 6 S 8(CN)2 S 4]4 - VEC=24, diamagnetic trans-[Re 6 Se 8(CN)4 Br 2]4 - VEC=24, diamagnetic [Re 6(Q 8 -x. Q’x)(CN)6]4 - (Q, Q’=S, Se, Te) VEC=24, diamagnetic etc.
![Some chalcocyanide cluster salts of type M 4[Re 6 Q 8(CN)6] Q= S Se Some chalcocyanide cluster salts of type M 4[Re 6 Q 8(CN)6] Q= S Se](https://present5.com/presentation/5652994_182163104/image-7.jpg) Some chalcocyanide cluster salts of type M 4[Re 6 Q 8(CN)6] Q= S Se Te
Some chalcocyanide cluster salts of type M 4[Re 6 Q 8(CN)6] Q= S Se Te
![Properties of [Re 6 Q 8(CN)6]n- (Q= S, Se, Te) cluster anions Electrochemical behavior Properties of [Re 6 Q 8(CN)6]n- (Q= S, Se, Te) cluster anions Electrochemical behavior](https://present5.com/presentation/5652994_182163104/image-8.jpg) Properties of [Re 6 Q 8(CN)6]n- (Q= S, Se, Te) cluster anions Electrochemical behavior in CH 3 CN: Chemical oxidation [Re 6 Q 8(CN)6]4 - - ē Re 6 Q 8(CN)6]3 - CH 3 CN [Re 6 Q 8(CN)6]4 - + ½ Br 2 Re 6 Q 8(CN)6]3 - + Br- or H O 2 S VEC=24 VEC=23 E = 0. 66 V 1/2 Effective magnetic moments for compounds Se containing oxidized anions [Re 6 Q 8(CN)6]3 - E = 0. 36 V 1/2 VEC=23 Te Compound E = 0. 08 V (PPh 4)3[Re 6 S 8(CN)6] 1. 9 (PPh 4)2(H)[Re 6 Se 8(CN)6] 4 H 2 O 1. 8 (NBu 4)2(H)[Re 6 Te 8(CN)6] 1. 9 1/2 1, 0 0, 8 0, 6 0, 4 0, 2 E vs NHE, V 0, 0 -0, 2 -0, 4 eff, BM (T=298 K)
Properties of [Re 6 Q 8(CN)6]n- (Q= S, Se, Te) cluster anions Electrochemical behavior in CH 3 CN: Chemical oxidation [Re 6 Q 8(CN)6]4 - - ē Re 6 Q 8(CN)6]3 - CH 3 CN [Re 6 Q 8(CN)6]4 - + ½ Br 2 Re 6 Q 8(CN)6]3 - + Br- or H O 2 S VEC=24 VEC=23 E = 0. 66 V 1/2 Effective magnetic moments for compounds Se containing oxidized anions [Re 6 Q 8(CN)6]3 - E = 0. 36 V 1/2 VEC=23 Te Compound E = 0. 08 V (PPh 4)3[Re 6 S 8(CN)6] 1. 9 (PPh 4)2(H)[Re 6 Se 8(CN)6] 4 H 2 O 1. 8 (NBu 4)2(H)[Re 6 Te 8(CN)6] 1. 9 1/2 1, 0 0, 8 0, 6 0, 4 0, 2 E vs NHE, V 0, 0 -0, 2 -0, 4 eff, BM (T=298 K)
![Change in color of cluster anion [Re 6 Se 8(CN)6]4 during its oxidation in Change in color of cluster anion [Re 6 Se 8(CN)6]4 during its oxidation in](https://present5.com/presentation/5652994_182163104/image-9.jpg) Change in color of cluster anion [Re 6 Se 8(CN)6]4 during its oxidation in bromine water K 4[Re 6 Se 8(CN)6] + ½ Br 2 K 3[Re 6 Se 8(CN)6] + Br yellow-orange green
Change in color of cluster anion [Re 6 Se 8(CN)6]4 during its oxidation in bromine water K 4[Re 6 Se 8(CN)6] + ½ Br 2 K 3[Re 6 Se 8(CN)6] + Br yellow-orange green
![Properties of [Re 6 Q 8(CN)6]n- cluster anions (Q= S, Se, Te) Comparison of Properties of [Re 6 Q 8(CN)6]n- cluster anions (Q= S, Se, Te) Comparison of](https://present5.com/presentation/5652994_182163104/image-10.jpg) Properties of [Re 6 Q 8(CN)6]n- cluster anions (Q= S, Se, Te) Comparison of interatomic distances (Å ) in anions [Re 6 Q 8(CN)6]4 - and [Re 6 Q 8(CN)6]3 - (Q=S, Se, Te) Q COMPOUNDS Re-Re Re-Q S Cs 2 Co[Re 6 S 8(CN)6] 2 H 2 O 2. 599 2. 411 (PPh 4)3[Re 6 S 8(CN)6] 2. 599 2. 401 Se K 4[Re 6 Se 8(CN)6] 3. 5 H 2 O 2. 526 2. 633 (PPh 4)2(H)[Re 6 Se 8(CN)6] 4 H 2 O 2. 635 Te 2. 519 Cs 4[Re 6 Te 8(CN)6] 2 H 2 O 2. 684 2. 698 (NBu 4)2(H)[Re 6 Te 8(CN)6] 2. 689 2. 686
Properties of [Re 6 Q 8(CN)6]n- cluster anions (Q= S, Se, Te) Comparison of interatomic distances (Å ) in anions [Re 6 Q 8(CN)6]4 - and [Re 6 Q 8(CN)6]3 - (Q=S, Se, Te) Q COMPOUNDS Re-Re Re-Q S Cs 2 Co[Re 6 S 8(CN)6] 2 H 2 O 2. 599 2. 411 (PPh 4)3[Re 6 S 8(CN)6] 2. 599 2. 401 Se K 4[Re 6 Se 8(CN)6] 3. 5 H 2 O 2. 526 2. 633 (PPh 4)2(H)[Re 6 Se 8(CN)6] 4 H 2 O 2. 635 Te 2. 519 Cs 4[Re 6 Te 8(CN)6] 2 H 2 O 2. 684 2. 698 (NBu 4)2(H)[Re 6 Te 8(CN)6] 2. 689 2. 686
![Properties of [Re 6 Q 8(CN)6]n- (Q= S, Se, Te) cluster anions Vibrational frequencies Properties of [Re 6 Q 8(CN)6]n- (Q= S, Se, Te) cluster anions Vibrational frequencies](https://present5.com/presentation/5652994_182163104/image-11.jpg) Properties of [Re 6 Q 8(CN)6]n- (Q= S, Se, Te) cluster anions Vibrational frequencies of CN-ligands ( CN) in [Re 6 Q 8(CN)6] 4 -/3 - anions CN , cm-1 Q [Re 6 Q 8(CN)6]4 - [Re 6 Q 8(CN)6]3 - CN S Se Te 2117 2111 2078 2137 2130 2106 20 19 28
Properties of [Re 6 Q 8(CN)6]n- (Q= S, Se, Te) cluster anions Vibrational frequencies of CN-ligands ( CN) in [Re 6 Q 8(CN)6] 4 -/3 - anions CN , cm-1 Q [Re 6 Q 8(CN)6]4 - [Re 6 Q 8(CN)6]3 - CN S Se Te 2117 2111 2078 2137 2130 2106 20 19 28
![Luminescent properties of cluster anions [Re 6 Q 8(CN)6]4 - (Q= S, Se, Te) Luminescent properties of cluster anions [Re 6 Q 8(CN)6]4 - (Q= S, Se, Te)](https://present5.com/presentation/5652994_182163104/image-12.jpg) Luminescent properties of cluster anions [Re 6 Q 8(CN)6]4 - (Q= S, Se, Te) 12 h Characteristics of (Ph 4 P)4[Re 6 Q 8(CN)6], Q = S, Se, Te Q lmax nm t, s fem. S 720 11. 2 0. 056 Se 720 17. 1 0. 140 Te 750 0. 57 0. 004 For comparison: (Bu 4 N)2 Mo 6 Cl 14 fem. = 0. 19 Te
Luminescent properties of cluster anions [Re 6 Q 8(CN)6]4 - (Q= S, Se, Te) 12 h Characteristics of (Ph 4 P)4[Re 6 Q 8(CN)6], Q = S, Se, Te Q lmax nm t, s fem. S 720 11. 2 0. 056 Se 720 17. 1 0. 140 Te 750 0. 57 0. 004 For comparison: (Bu 4 N)2 Mo 6 Cl 14 fem. = 0. 19 Te
![In rhenium chalcocyanide complexes [Re 6( 3 -Q)8(CN)6]n– six metal atoms form an octahedron In rhenium chalcocyanide complexes [Re 6( 3 -Q)8(CN)6]n– six metal atoms form an octahedron](https://present5.com/presentation/5652994_182163104/image-13.jpg) In rhenium chalcocyanide complexes [Re 6( 3 -Q)8(CN)6]n– six metal atoms form an octahedron cluster Re 6. Eight 3 chalcogene ligands (S, Se, or Te) form a cube around the octahedron Re 6. Six terminal CN-ligands are coordinated to each rhenium atom via carbon. CN ligands are ambidentate and able to coordinate to other metals via N atoms. The anions [Re 6( 3 -Q)8(CN)6]n– can be considered as geometrical analogs of mononuclear cyanide complex [Fe(CN)6]4– but possessing larger linear and voluminous sizes due to cluster core {Re 6 Q 8}2+. These cluster anions can be used as building blocks in design of coordination polymers. 10. 3 Å [Fe(CN)6]4–
In rhenium chalcocyanide complexes [Re 6( 3 -Q)8(CN)6]n– six metal atoms form an octahedron cluster Re 6. Eight 3 chalcogene ligands (S, Se, or Te) form a cube around the octahedron Re 6. Six terminal CN-ligands are coordinated to each rhenium atom via carbon. CN ligands are ambidentate and able to coordinate to other metals via N atoms. The anions [Re 6( 3 -Q)8(CN)6]n– can be considered as geometrical analogs of mononuclear cyanide complex [Fe(CN)6]4– but possessing larger linear and voluminous sizes due to cluster core {Re 6 Q 8}2+. These cluster anions can be used as building blocks in design of coordination polymers. 10. 3 Å [Fe(CN)6]4–
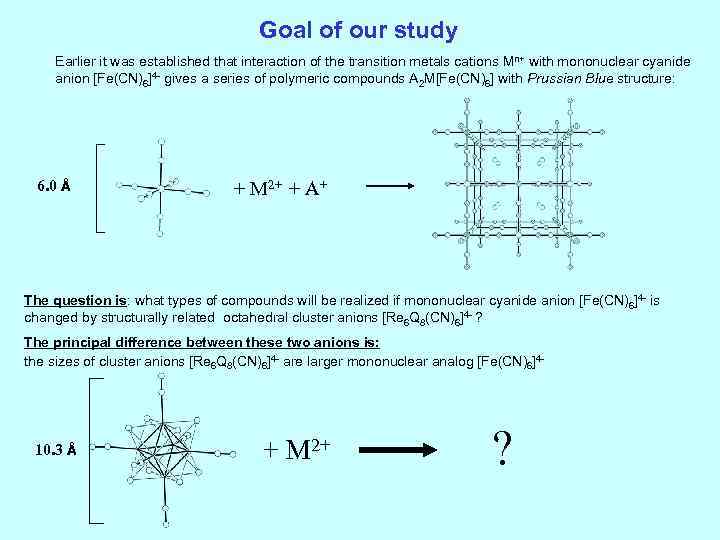 Goal of our study Earlier it was established that interaction of the transition metals cations Mn+ with mononuclear cyanide anion [Fe(CN)6]4 - gives a series of polymeric compounds A 2 M[Fe(CN)6] with Prussian Blue structure: 6. 0 Å + M 2+ + A+ The question is: what types of compounds will be realized if mononuclear cyanide anion [Fe(CN)6]4 - is changed by structurally related octahedral cluster anions [Re 6 Q 8(CN)6]4 - ? The principal difference between these two anions is: the sizes of cluster anions [Re 6 Q 8(CN)6]4 - are larger mononuclear analog [Fe(CN)6]4 - 10. 3 Å + M 2+ ?
Goal of our study Earlier it was established that interaction of the transition metals cations Mn+ with mononuclear cyanide anion [Fe(CN)6]4 - gives a series of polymeric compounds A 2 M[Fe(CN)6] with Prussian Blue structure: 6. 0 Å + M 2+ + A+ The question is: what types of compounds will be realized if mononuclear cyanide anion [Fe(CN)6]4 - is changed by structurally related octahedral cluster anions [Re 6 Q 8(CN)6]4 - ? The principal difference between these two anions is: the sizes of cluster anions [Re 6 Q 8(CN)6]4 - are larger mononuclear analog [Fe(CN)6]4 - 10. 3 Å + M 2+ ?
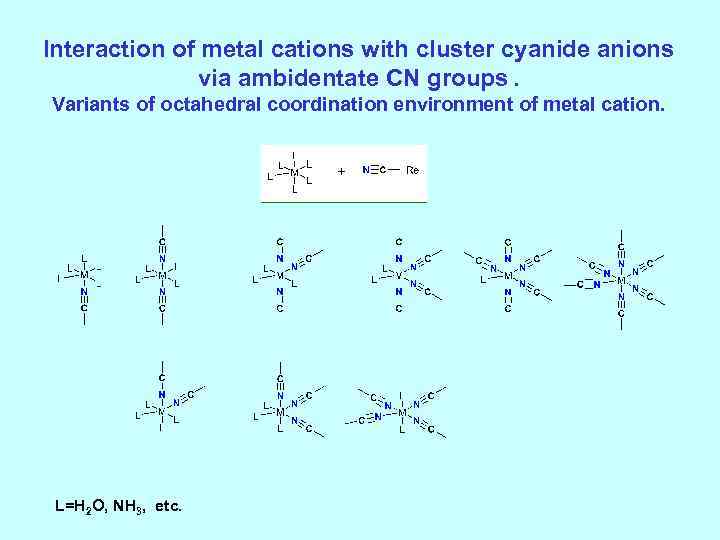 Interaction of metal cations with cluster cyanide anions via ambidentate CN groups. Variants of octahedral coordination environment of metal cation. L=H 2 O, NH 3, etc.
Interaction of metal cations with cluster cyanide anions via ambidentate CN groups. Variants of octahedral coordination environment of metal cation. L=H 2 O, NH 3, etc.
![Design of solids based on octahedral chalcocyanide cluster complexes [Re 6 Q 8(CN)6]4 -/3 Design of solids based on octahedral chalcocyanide cluster complexes [Re 6 Q 8(CN)6]4 -/3](https://present5.com/presentation/5652994_182163104/image-16.jpg) Design of solids based on octahedral chalcocyanide cluster complexes [Re 6 Q 8(CN)6]4 -/3 General approach: [M(H 2 O)6]2+ + [Re 6 Q 8(CN)6]4 -/3 - + Ni [{Ni(NH 3)5}2 Re 6 Te 8(CN)6]· 2 H 2 O (n-Bu 4 N)2[{Ni(H 2 O)5}{Re 6 Se 8(CN)6}]· 2 H 2 O
Design of solids based on octahedral chalcocyanide cluster complexes [Re 6 Q 8(CN)6]4 -/3 General approach: [M(H 2 O)6]2+ + [Re 6 Q 8(CN)6]4 -/3 - + Ni [{Ni(NH 3)5}2 Re 6 Te 8(CN)6]· 2 H 2 O (n-Bu 4 N)2[{Ni(H 2 O)5}{Re 6 Se 8(CN)6}]· 2 H 2 O
![Design of solids based on octahedral cyanoclusters Isolated anionic fragment [Cu(en)2{Re 6 Te 8(CN)6}2]6 Design of solids based on octahedral cyanoclusters Isolated anionic fragment [Cu(en)2{Re 6 Te 8(CN)6}2]6](https://present5.com/presentation/5652994_182163104/image-17.jpg) Design of solids based on octahedral cyanoclusters Isolated anionic fragment [Cu(en)2{Re 6 Te 8(CN)6}2]6 in the structure of (Et 4 N)2[Cu. NH 3(en)2]2 [Cu(en)2{Re 6 Te 8(CN)6}2] 2 H 2 O Coordination of cations in the structure: {Cu(NC)2(en)2}2+ [Cu. NH 3(en)2]2+
Design of solids based on octahedral cyanoclusters Isolated anionic fragment [Cu(en)2{Re 6 Te 8(CN)6}2]6 in the structure of (Et 4 N)2[Cu. NH 3(en)2]2 [Cu(en)2{Re 6 Te 8(CN)6}2] 2 H 2 O Coordination of cations in the structure: {Cu(NC)2(en)2}2+ [Cu. NH 3(en)2]2+
![Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов Строение (Et 4 N)2[Cu(NH 3)(en)2]2[{Cu(en)2}{Re Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов Строение (Et 4 N)2[Cu(NH 3)(en)2]2[{Cu(en)2}{Re](https://present5.com/presentation/5652994_182163104/image-18.jpg) Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов Строение (Et 4 N)2[Cu(NH 3)(en)2]2[{Cu(en)2}{Re 6 Te 8(CN)6}2]· 2 H 2 O
Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов Строение (Et 4 N)2[Cu(NH 3)(en)2]2[{Cu(en)2}{Re 6 Te 8(CN)6}2]· 2 H 2 O
![The cluster compounds with infinite linear chains [Ni(NH 3)2(en)2]2[{Ni(NH 3)4}Re 6 Q 8(CN)6]Cl 2 The cluster compounds with infinite linear chains [Ni(NH 3)2(en)2]2[{Ni(NH 3)4}Re 6 Q 8(CN)6]Cl 2](https://present5.com/presentation/5652994_182163104/image-19.jpg) The cluster compounds with infinite linear chains [Ni(NH 3)2(en)2]2[{Ni(NH 3)4}Re 6 Q 8(CN)6]Cl 2 n. H 2 O [Co(NH 3)2(en)2]2[{Co(en)2}Re 6 Te 8(CN)6]Cl 2 H 2 O (Q = Se, Te) H, Se, Te, Cl atoms and water molecules are omitted for clarity
The cluster compounds with infinite linear chains [Ni(NH 3)2(en)2]2[{Ni(NH 3)4}Re 6 Q 8(CN)6]Cl 2 n. H 2 O [Co(NH 3)2(en)2]2[{Co(en)2}Re 6 Te 8(CN)6]Cl 2 H 2 O (Q = Se, Te) H, Se, Te, Cl atoms and water molecules are omitted for clarity
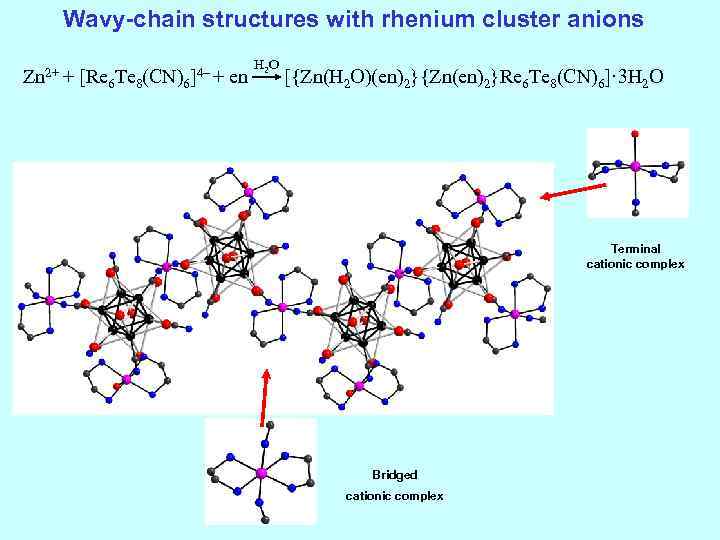 Wavy-chain structures with rhenium cluster anions H 2 O Zn 2+ + [Re 6 Te 8(CN)6]4– + en [{Zn(H 2 O)(en)2}{Zn(en)2}Re 6 Te 8(CN)6]· 3 H 2 O Terminal cationic complex Bridged cationic complex
Wavy-chain structures with rhenium cluster anions H 2 O Zn 2+ + [Re 6 Te 8(CN)6]4– + en [{Zn(H 2 O)(en)2}{Zn(en)2}Re 6 Te 8(CN)6]· 3 H 2 O Terminal cationic complex Bridged cationic complex
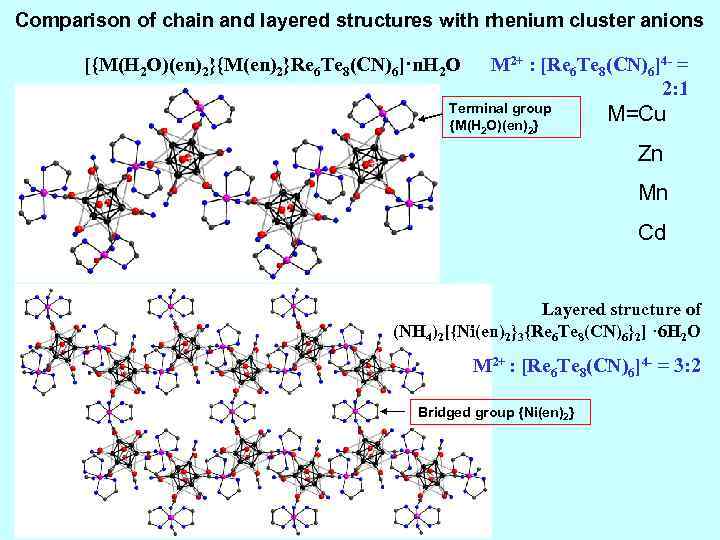 Comparison of chain and layered structures with rhenium cluster anions [{M(H 2 O)(en)2}{M(en)2}Re 6 Te 8(CN)6]·n. H 2 O M 2+ : [Re 6 Te 8(CN)6]4 - = 2: 1 Terminal group {M(H 2 O)(en)2} M=Cu Zn Mn Cd Layered structure of (NH 4)2[{Ni(en)2}3{Re 6 Te 8(CN)6}2] · 6 H 2 O M 2+ : [Re 6 Te 8(CN)6]4 - = 3: 2 Bridged group {Ni(en)2}
Comparison of chain and layered structures with rhenium cluster anions [{M(H 2 O)(en)2}{M(en)2}Re 6 Te 8(CN)6]·n. H 2 O M 2+ : [Re 6 Te 8(CN)6]4 - = 2: 1 Terminal group {M(H 2 O)(en)2} M=Cu Zn Mn Cd Layered structure of (NH 4)2[{Ni(en)2}3{Re 6 Te 8(CN)6}2] · 6 H 2 O M 2+ : [Re 6 Te 8(CN)6]4 - = 3: 2 Bridged group {Ni(en)2}
![Layered structure of compounds Cs 2[{M(H 2 O)2}{Re 6 S 8(CN)6}] (M=Mn 2+, Fe Layered structure of compounds Cs 2[{M(H 2 O)2}{Re 6 S 8(CN)6}] (M=Mn 2+, Fe](https://present5.com/presentation/5652994_182163104/image-22.jpg) Layered structure of compounds Cs 2[{M(H 2 O)2}{Re 6 S 8(CN)6}] (M=Mn 2+, Fe 2+, Co 2+, Cd 2+) Orthorhombic, Space group Imma H 2 O Co N C Coordination environment of Co 2+ cation Co-N(NC) 2. 16 Å Co-O(OH 2 ) 2. 05; 2. 09 Å O-Co-O 180 o Short S-S contacts: 3. 32 Å within layer; 3. 67 Å between adjacent layers Similar structure is not realized for Se N. G. Naumov, A. V. Virovets, Y. I. Mironov, S. B. Artemkina, V. E. Fedorov, Ukraine Chem. J. , 1999, v. 65, pp. 21 -27.
Layered structure of compounds Cs 2[{M(H 2 O)2}{Re 6 S 8(CN)6}] (M=Mn 2+, Fe 2+, Co 2+, Cd 2+) Orthorhombic, Space group Imma H 2 O Co N C Coordination environment of Co 2+ cation Co-N(NC) 2. 16 Å Co-O(OH 2 ) 2. 05; 2. 09 Å O-Co-O 180 o Short S-S contacts: 3. 32 Å within layer; 3. 67 Å between adjacent layers Similar structure is not realized for Se N. G. Naumov, A. V. Virovets, Y. I. Mironov, S. B. Artemkina, V. E. Fedorov, Ukraine Chem. J. , 1999, v. 65, pp. 21 -27.
![View of polymeric layer in the compound [{Mn(H 2 O)3}2{Re 6 Se 8(CN)6}]∙ 3. View of polymeric layer in the compound [{Mn(H 2 O)3}2{Re 6 Se 8(CN)6}]∙ 3.](https://present5.com/presentation/5652994_182163104/image-23.jpg) View of polymeric layer in the compound [{Mn(H 2 O)3}2{Re 6 Se 8(CN)6}]∙ 3. 3 H 2 O A layer of composition [{Mn(H 2 O)3}4{Re 6 Se 8(CN)6}3]4– H 2 O Mn N C Mn 2 Mn 1
View of polymeric layer in the compound [{Mn(H 2 O)3}2{Re 6 Se 8(CN)6}]∙ 3. 3 H 2 O A layer of composition [{Mn(H 2 O)3}4{Re 6 Se 8(CN)6}3]4– H 2 O Mn N C Mn 2 Mn 1
![Structure of (H 3 O)[{Nd(H 2 O)3(DMF)3}{Re 6 Se 8(CN)6}]·n. H 2 O Space Structure of (H 3 O)[{Nd(H 2 O)3(DMF)3}{Re 6 Se 8(CN)6}]·n. H 2 O Space](https://present5.com/presentation/5652994_182163104/image-24.jpg) Structure of (H 3 O)[{Nd(H 2 O)3(DMF)3}{Re 6 Se 8(CN)6}]·n. H 2 O Space group R 3 c, a=32. 514, c=25. 255 Coordination framework of the structure The same open structure is found for Ln = Pr, Nd, Sm, Ho and Q=S, Se, Te. [{La(H 2 O)2(DMF)4}{Re 6 Se 8(CN)6}]4 H 2 O with oxidized [Re 6 Se 8(CN)6]3 - anion has similar framework. Coordination environment of Nd 3+ cation (CN=9) N 2 N 4 N 5 Nd O 2 O 1 O 6 O 3 O 5 O 4 S. B. Artemkina, N. G. Naumov, A. V. Virovets, S. A. Gromilov, D. Fenske, V. E. Fedorov, Inorg. Chem. Comm. , 2001, v. 4, pp. 423 -426.
Structure of (H 3 O)[{Nd(H 2 O)3(DMF)3}{Re 6 Se 8(CN)6}]·n. H 2 O Space group R 3 c, a=32. 514, c=25. 255 Coordination framework of the structure The same open structure is found for Ln = Pr, Nd, Sm, Ho and Q=S, Se, Te. [{La(H 2 O)2(DMF)4}{Re 6 Se 8(CN)6}]4 H 2 O with oxidized [Re 6 Se 8(CN)6]3 - anion has similar framework. Coordination environment of Nd 3+ cation (CN=9) N 2 N 4 N 5 Nd O 2 O 1 O 6 O 3 O 5 O 4 S. B. Artemkina, N. G. Naumov, A. V. Virovets, S. A. Gromilov, D. Fenske, V. E. Fedorov, Inorg. Chem. Comm. , 2001, v. 4, pp. 423 -426.
![Structure of (NH 4)2[{Ni(en)2}3{Re 6 Te 8(CN)6}2]· 6 H 2 O with one-dimensional channels Structure of (NH 4)2[{Ni(en)2}3{Re 6 Te 8(CN)6}2]· 6 H 2 O with one-dimensional channels](https://present5.com/presentation/5652994_182163104/image-25.jpg) Structure of (NH 4)2[{Ni(en)2}3{Re 6 Te 8(CN)6}2]· 6 H 2 O with one-dimensional channels Ball-and-stick presentation Space-filling presentation
Structure of (NH 4)2[{Ni(en)2}3{Re 6 Te 8(CN)6}2]· 6 H 2 O with one-dimensional channels Ball-and-stick presentation Space-filling presentation
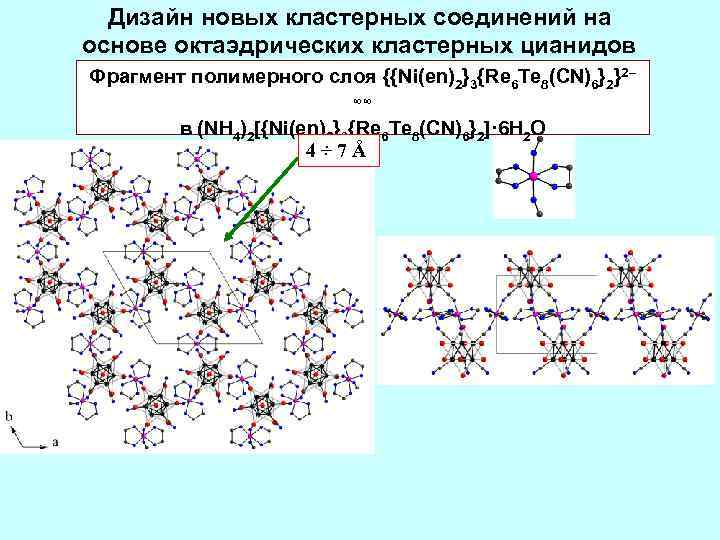 Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов Фрагмент полимерного слоя {{Ni(en)2}3{Re 6 Te 8(CN)6}2}2– ∞∞ в (NH 4)2[{Ni(en)2}3{Re 6 Te 8(CN)6}2]· 6 H 2 O 4÷ 7Å
Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов Фрагмент полимерного слоя {{Ni(en)2}3{Re 6 Te 8(CN)6}2}2– ∞∞ в (NH 4)2[{Ni(en)2}3{Re 6 Te 8(CN)6}2]· 6 H 2 O 4÷ 7Å
![Reactions of cluster anions [Re 6 Q 8(CN)6]4 - with Cu 2+ ions and Reactions of cluster anions [Re 6 Q 8(CN)6]4 - with Cu 2+ ions and](https://present5.com/presentation/5652994_182163104/image-27.jpg) Reactions of cluster anions [Re 6 Q 8(CN)6]4 - with Cu 2+ ions and chiral 1, 2 S, 3 S, 4, - tetraaminobutane (threo-tab) Y. Mironov, N. Naumov, K. Brylev, O. Efremova, V. Fedorov, K. Hegetschweiler, Angew. Chem. , Int. Ed. , 2004, v. 43, N 10, pp. 1297 -1300. K 4 Re 6 Te 8(CN)6 + Cu. Cl 2 + tab + NH 4 OH [{Cu(tab)}2 Re 6 Te 8(CN)6] K 4 Re 6 Q 8(CN)6 + Cu. Cl 2 + tab + NH 4 OH [{Cu 2(tab)}3(NH 3)}Re 6 Q 8(CN)6] Q=S, Se
Reactions of cluster anions [Re 6 Q 8(CN)6]4 - with Cu 2+ ions and chiral 1, 2 S, 3 S, 4, - tetraaminobutane (threo-tab) Y. Mironov, N. Naumov, K. Brylev, O. Efremova, V. Fedorov, K. Hegetschweiler, Angew. Chem. , Int. Ed. , 2004, v. 43, N 10, pp. 1297 -1300. K 4 Re 6 Te 8(CN)6 + Cu. Cl 2 + tab + NH 4 OH [{Cu(tab)}2 Re 6 Te 8(CN)6] K 4 Re 6 Q 8(CN)6 + Cu. Cl 2 + tab + NH 4 OH [{Cu 2(tab)}3(NH 3)}Re 6 Q 8(CN)6] Q=S, Se
![The structure of [{Cu(tab)}2 Re 6 Te 8(CN)6] 13. 5 H 2 O Orthorhombic, The structure of [{Cu(tab)}2 Re 6 Te 8(CN)6] 13. 5 H 2 O Orthorhombic,](https://present5.com/presentation/5652994_182163104/image-28.jpg) The structure of [{Cu(tab)}2 Re 6 Te 8(CN)6] 13. 5 H 2 O Orthorhombic, Space group P 212121, a = 13. 767, b = 19. 692, c = 21. 780 Å a). View of one layer b). Packing of wavy layers b b c a Y. Mironov, N. Naumov, K. Brylev, O. Efremova, V. Fedorov, K. Hegetschweiler, Angew. Chem. , Int. Ed. , 2004, v. 43, N 10, pp. 1297 -1300.
The structure of [{Cu(tab)}2 Re 6 Te 8(CN)6] 13. 5 H 2 O Orthorhombic, Space group P 212121, a = 13. 767, b = 19. 692, c = 21. 780 Å a). View of one layer b). Packing of wavy layers b b c a Y. Mironov, N. Naumov, K. Brylev, O. Efremova, V. Fedorov, K. Hegetschweiler, Angew. Chem. , Int. Ed. , 2004, v. 43, N 10, pp. 1297 -1300.
![The plane layer in the structure of [{Cu 2(tab)3(NH 3)}Re 6 Se 8(CN)6]· 2. The plane layer in the structure of [{Cu 2(tab)3(NH 3)}Re 6 Se 8(CN)6]· 2.](https://present5.com/presentation/5652994_182163104/image-29.jpg) The plane layer in the structure of [{Cu 2(tab)3(NH 3)}Re 6 Se 8(CN)6]· 2. 5 H 2 O
The plane layer in the structure of [{Cu 2(tab)3(NH 3)}Re 6 Se 8(CN)6]· 2. 5 H 2 O
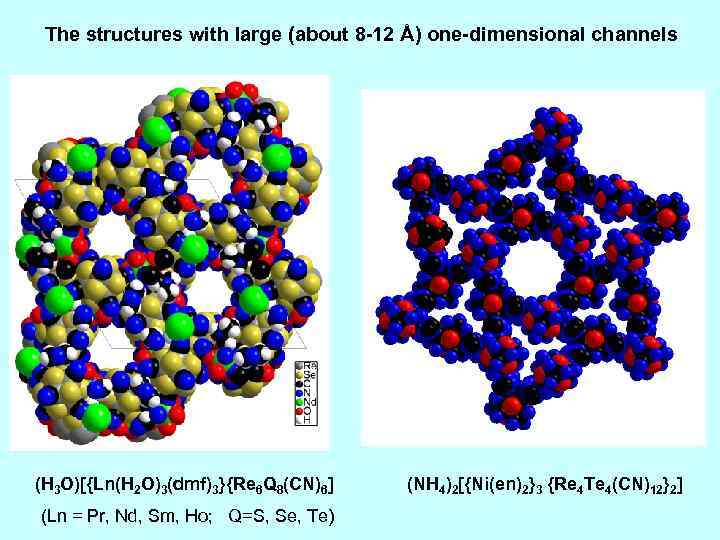 The structures with large (about 8 -12 Å) one-dimensional channels (H 3 O)[{Ln(H 2 O)3(dmf)3}{Re 6 Q 8(CN)6] (Ln = Pr, Nd, Sm, Ho; Q=S, Se, Te) (NH 4)2[{Ni(en)2}3 {Re 4 Te 4(CN)12}2]
The structures with large (about 8 -12 Å) one-dimensional channels (H 3 O)[{Ln(H 2 O)3(dmf)3}{Re 6 Q 8(CN)6] (Ln = Pr, Nd, Sm, Ho; Q=S, Se, Te) (NH 4)2[{Ni(en)2}3 {Re 4 Te 4(CN)12}2]
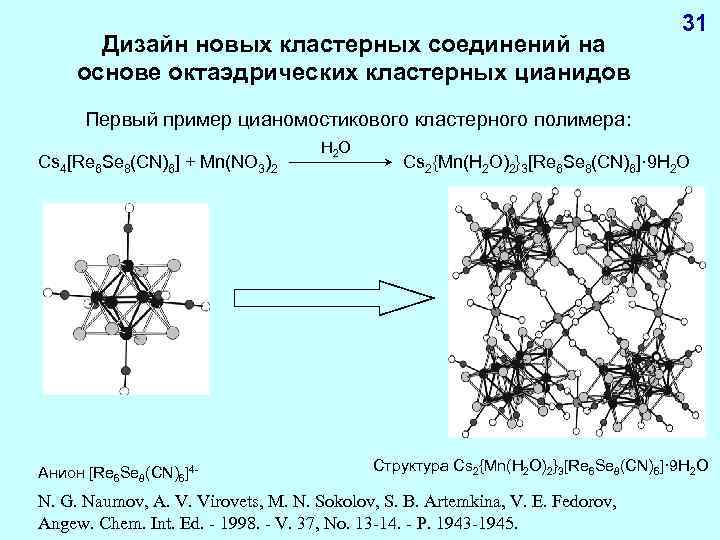 Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов 31 Первый пример цианомостикового кластерного полимера: Cs 4[Re 6 Se 8(CN)6] + Mn(NO 3)2 Анион [Re 6 Se 8(CN)6]4 - H 2 O Cs 2{Mn(H 2 O)2}3[Re 6 Se 8(CN)6]· 9 H 2 O Структура Cs 2{Mn(H 2 O)2}3[Re 6 Se 8(CN)6]· 9 H 2 O N. G. Naumov, A. V. Virovets, M. N. Sokolov, S. B. Artemkina, V. E. Fedorov, Angew. Chem. Int. Ed. - 1998. - V. 37, No. 13 -14. - P. 1943 -1945.
Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов 31 Первый пример цианомостикового кластерного полимера: Cs 4[Re 6 Se 8(CN)6] + Mn(NO 3)2 Анион [Re 6 Se 8(CN)6]4 - H 2 O Cs 2{Mn(H 2 O)2}3[Re 6 Se 8(CN)6]· 9 H 2 O Структура Cs 2{Mn(H 2 O)2}3[Re 6 Se 8(CN)6]· 9 H 2 O N. G. Naumov, A. V. Virovets, M. N. Sokolov, S. B. Artemkina, V. E. Fedorov, Angew. Chem. Int. Ed. - 1998. - V. 37, No. 13 -14. - P. 1943 -1945.
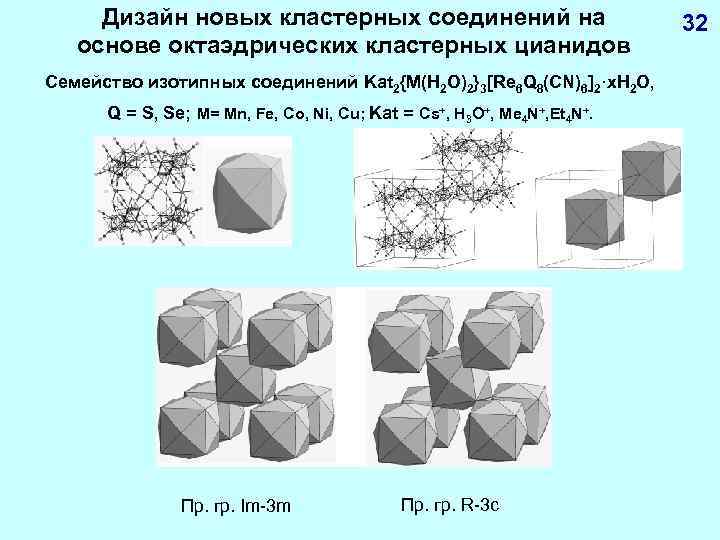 Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов Семейство изотипных соединений Kat 2{M(H 2 O)2}3[Re 6 Q 8(CN)6]2·x. H 2 O, Q = S, Se; M= Mn, Fe, Co, Ni, Cu; Kat = Cs+, H 3 O+, Me 4 N+, Et 4 N+. Пр. гр. Im-3 m Пр. гр. R-3 c 32
Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов Семейство изотипных соединений Kat 2{M(H 2 O)2}3[Re 6 Q 8(CN)6]2·x. H 2 O, Q = S, Se; M= Mn, Fe, Co, Ni, Cu; Kat = Cs+, H 3 O+, Me 4 N+, Et 4 N+. Пр. гр. Im-3 m Пр. гр. R-3 c 32
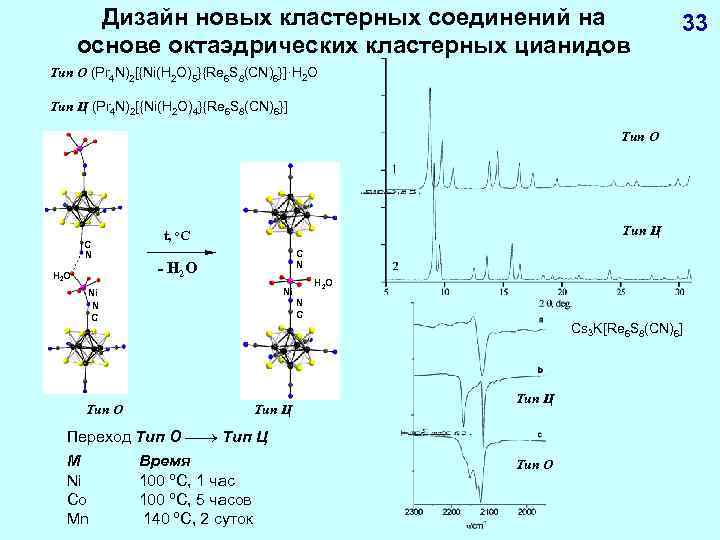 Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов 33 Тип О (Pr 4 N)2[{Ni(H 2 O)5}{Re 6 S 8(CN)6}]·H 2 O Тип Ц (Pr 4 N)2[{Ni(H 2 O)4}{Re 6 S 8(CN)6}] Тип О C N H 2 O Тип Ц t, o. C C N - H 2 O Тип О H 2 O Ni Ni N C Cs 3 K[Re 6 S 8(CN)6] Тип Ц Переход Тип О Тип Ц M Ni Co Mn Время 100 ºC, 1 час 100 ºC, 5 часов 140 ºC, 2 суток Тип О
Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов 33 Тип О (Pr 4 N)2[{Ni(H 2 O)5}{Re 6 S 8(CN)6}]·H 2 O Тип Ц (Pr 4 N)2[{Ni(H 2 O)4}{Re 6 S 8(CN)6}] Тип О C N H 2 O Тип Ц t, o. C C N - H 2 O Тип О H 2 O Ni Ni N C Cs 3 K[Re 6 S 8(CN)6] Тип Ц Переход Тип О Тип Ц M Ni Co Mn Время 100 ºC, 1 час 100 ºC, 5 часов 140 ºC, 2 суток Тип О
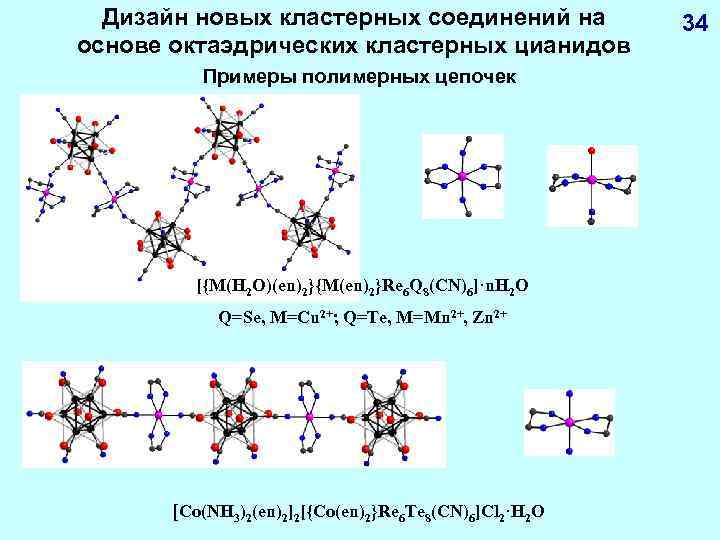 Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов Примеры полимерных цепочек [{M(H 2 O)(en)2}{M(en)2}Re 6 Q 8(CN)6]·n. H 2 O Q=Se, M=Cu 2+; Q=Te, M=Mn 2+, Zn 2+ [Co(NH 3)2(en)2]2[{Co(en)2}Re 6 Te 8(CN)6]Cl 2·H 2 O 34
Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов Примеры полимерных цепочек [{M(H 2 O)(en)2}{M(en)2}Re 6 Q 8(CN)6]·n. H 2 O Q=Se, M=Cu 2+; Q=Te, M=Mn 2+, Zn 2+ [Co(NH 3)2(en)2]2[{Co(en)2}Re 6 Te 8(CN)6]Cl 2·H 2 O 34
![Соединения с этилендиамином M 2+ + en + [Re 6 Q 8(CN)6]4– Ионные комплексы Соединения с этилендиамином M 2+ + en + [Re 6 Q 8(CN)6]4– Ионные комплексы](https://present5.com/presentation/5652994_182163104/image-35.jpg) Соединения с этилендиамином M 2+ + en + [Re 6 Q 8(CN)6]4– Ионные комплексы Молекулярные комплексы Цепочечные полимеры Слоистые полимеры 35
Соединения с этилендиамином M 2+ + en + [Re 6 Q 8(CN)6]4– Ионные комплексы Молекулярные комплексы Цепочечные полимеры Слоистые полимеры 35
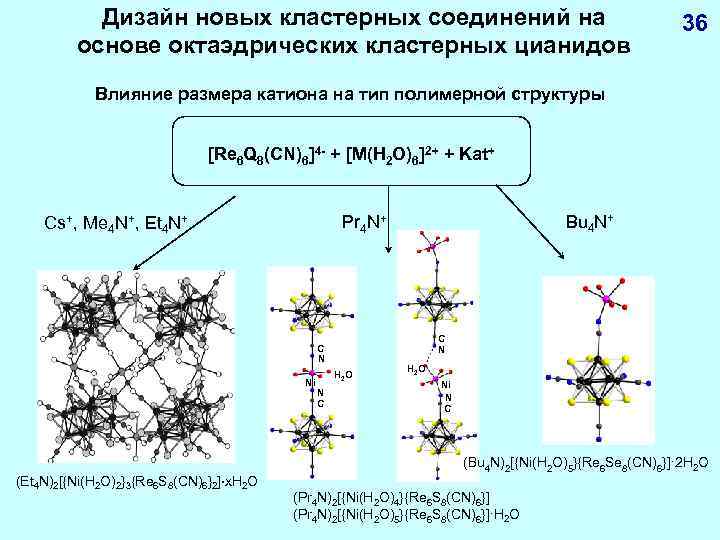 Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов 36 Влияние размера катиона на тип полимерной структуры [Re 6 Q 8(CN)6]4 - + [M(H 2 O)6]2+ + Kat+ C N Ni (Et 4 N)2[{Ni(H 2 O)2}3{Re 6 S 8(CN)6}2] x. H 2 O Bu 4 N+ Pr 4 N+ Cs+, Me 4 N+, Et 4 N+ H 2 O N C H 2 O Ni N C (Bu 4 N)2[{Ni(H 2 O)5}{Re 6 Se 8(CN)6}]· 2 H 2 O (Pr 4 N)2[{Ni(H 2 O)4}{Re 6 S 8(CN)6}] (Pr 4 N)2[{Ni(H 2 O)5}{Re 6 S 8(CN)6}]·H 2 O
Дизайн новых кластерных соединений на основе октаэдрических кластерных цианидов 36 Влияние размера катиона на тип полимерной структуры [Re 6 Q 8(CN)6]4 - + [M(H 2 O)6]2+ + Kat+ C N Ni (Et 4 N)2[{Ni(H 2 O)2}3{Re 6 S 8(CN)6}2] x. H 2 O Bu 4 N+ Pr 4 N+ Cs+, Me 4 N+, Et 4 N+ H 2 O N C H 2 O Ni N C (Bu 4 N)2[{Ni(H 2 O)5}{Re 6 Se 8(CN)6}]· 2 H 2 O (Pr 4 N)2[{Ni(H 2 O)4}{Re 6 S 8(CN)6}] (Pr 4 N)2[{Ni(H 2 O)5}{Re 6 S 8(CN)6}]·H 2 O
![Структурные аналогии с моноядерными цианометаллатами 37 [Mo 6 Q 8(CN)6]7–/6– + M 2+ K Структурные аналогии с моноядерными цианометаллатами 37 [Mo 6 Q 8(CN)6]7–/6– + M 2+ K](https://present5.com/presentation/5652994_182163104/image-37.jpg) Структурные аналогии с моноядерными цианометаллатами 37 [Mo 6 Q 8(CN)6]7–/6– + M 2+ K 7[Mo 6 Se 8(CN)6](вода) + [Mn(salen)]Ac(метанол) ? (аморфный осадок) K 5[Mn. Mo 6 Se 8(CN)6]· 9 H 2 O salen – бис-(салицилиден)этилендиамин Фрагмент каркаса {Mn. Mo 6 Se 8(CN)6}5– Пр. гр. Fm 3 m a = 15, 226(6) Å V = 3530(2) Å3 Z = 4 Лигандное окружение марганца
Структурные аналогии с моноядерными цианометаллатами 37 [Mo 6 Q 8(CN)6]7–/6– + M 2+ K 7[Mo 6 Se 8(CN)6](вода) + [Mn(salen)]Ac(метанол) ? (аморфный осадок) K 5[Mn. Mo 6 Se 8(CN)6]· 9 H 2 O salen – бис-(салицилиден)этилендиамин Фрагмент каркаса {Mn. Mo 6 Se 8(CN)6}5– Пр. гр. Fm 3 m a = 15, 226(6) Å V = 3530(2) Å3 Z = 4 Лигандное окружение марганца
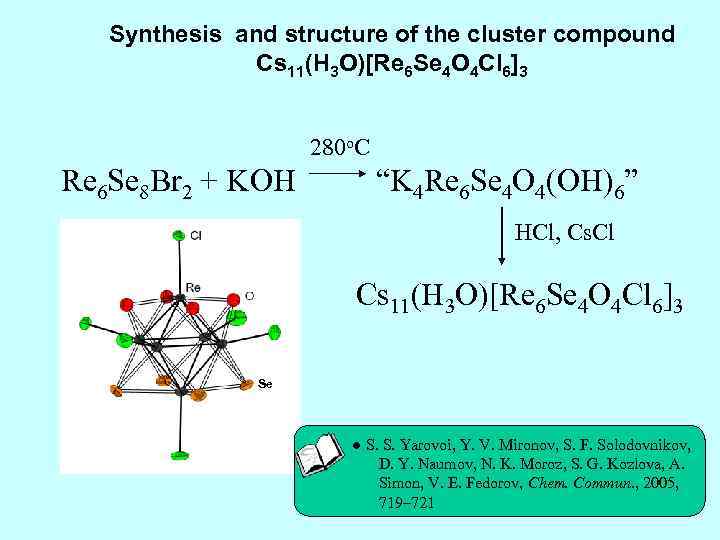 Synthesis and structure of the cluster compound Cs 11(H 3 O)[Re 6 Se 4 O 4 Cl 6]3 280 o. C Re 6 Se 8 Br 2 + KOH “K 4 Re 6 Se 4 O 4(OH)6” HCl, Cs. Cl Cs 11(H 3 O)[Re 6 Se 4 O 4 Cl 6]3 Se ● S. S. Yarovoi, Y. V. Mironov, S. F. Solodovnikov, D. Y. Naumov, N. K. Moroz, S. G. Kozlova, A. Simon, V. E. Fedorov, Chem. Commun. , 2005, 719– 721
Synthesis and structure of the cluster compound Cs 11(H 3 O)[Re 6 Se 4 O 4 Cl 6]3 280 o. C Re 6 Se 8 Br 2 + KOH “K 4 Re 6 Se 4 O 4(OH)6” HCl, Cs. Cl Cs 11(H 3 O)[Re 6 Se 4 O 4 Cl 6]3 Se ● S. S. Yarovoi, Y. V. Mironov, S. F. Solodovnikov, D. Y. Naumov, N. K. Moroz, S. G. Kozlova, A. Simon, V. E. Fedorov, Chem. Commun. , 2005, 719– 721
![Structure of [{Mg(H 2 O)5}2 Re 6 Q 8(OH)6]. 6 H 2 O (Q=S, Structure of [{Mg(H 2 O)5}2 Re 6 Q 8(OH)6]. 6 H 2 O (Q=S,](https://present5.com/presentation/5652994_182163104/image-39.jpg) Structure of [{Mg(H 2 O)5}2 Re 6 Q 8(OH)6]. 6 H 2 O (Q=S, Se) [{Mg(H 2 O)5}2 Re 6 S 8(OH)6]. 6 H 2 O Sp. gr. P 1 a = 8. 4986(18) Å b = 8. 6593(19) Å c = 10. 920(2) Å a= 78. 179(4)o β = 85. 273(3)o g= 76. 809(3)o V = 765. 3(3) Å3
Structure of [{Mg(H 2 O)5}2 Re 6 Q 8(OH)6]. 6 H 2 O (Q=S, Se) [{Mg(H 2 O)5}2 Re 6 S 8(OH)6]. 6 H 2 O Sp. gr. P 1 a = 8. 4986(18) Å b = 8. 6593(19) Å c = 10. 920(2) Å a= 78. 179(4)o β = 85. 273(3)o g= 76. 809(3)o V = 765. 3(3) Å3
![Structure of the compounds[{Ca(H 2 O)3}2 Re 6 Q 8(OH)6] Sp. gr. P 3, Structure of the compounds[{Ca(H 2 O)3}2 Re 6 Q 8(OH)6] Sp. gr. P 3,](https://present5.com/presentation/5652994_182163104/image-40.jpg) Structure of the compounds[{Ca(H 2 O)3}2 Re 6 Q 8(OH)6] Sp. gr. P 3, a = 8. 3359(8) Å, c = 9. 3100(18) Å, V = 560. 25(13) Å3, (Q=S) Sp. gr. P 3, a = 8. 4729(6) Å, c = 9. 4353(14) Å, V = 586. 61(11) Å3, (Q=Se)
Structure of the compounds[{Ca(H 2 O)3}2 Re 6 Q 8(OH)6] Sp. gr. P 3, a = 8. 3359(8) Å, c = 9. 3100(18) Å, V = 560. 25(13) Å3, (Q=S) Sp. gr. P 3, a = 8. 4729(6) Å, c = 9. 4353(14) Å, V = 586. 61(11) Å3, (Q=Se)
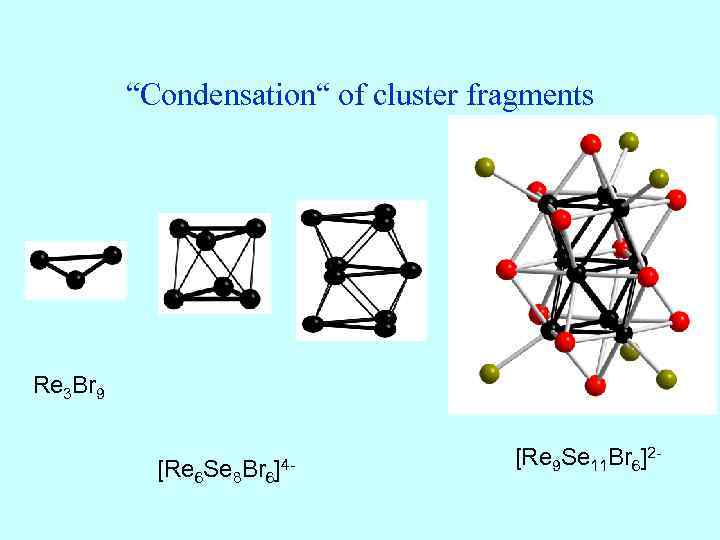 “Condensation“ of cluster fragments Re 3 Br 9 [Re 6 Se 8 Br 6 ]4 - [Re 9 Se 11 Br 6]2 -
“Condensation“ of cluster fragments Re 3 Br 9 [Re 6 Se 8 Br 6 ]4 - [Re 9 Se 11 Br 6]2 -
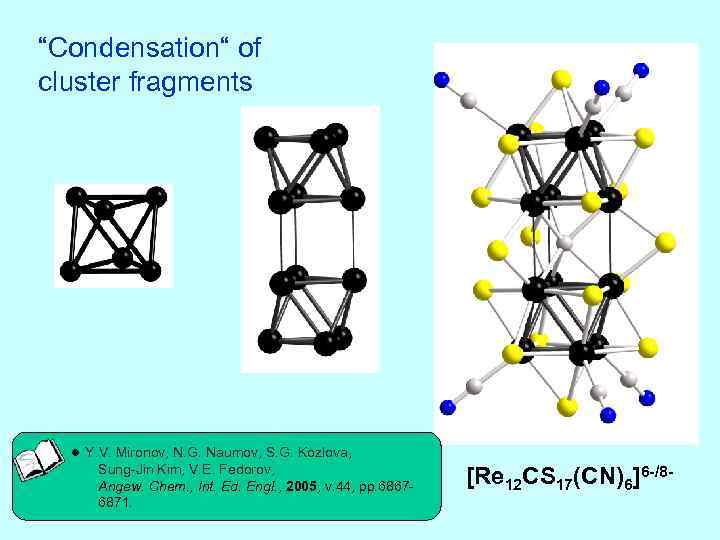 “Condensation“ of cluster fragments ● Y. V. Mironov, N. G. Naumov, S. G. Kozlova, Sung-Jin Kim, V. E. Fedorov, Angew. Chem. , Int. Ed. Engl. , 2005, v. 44, pp. 68676871. [Re 12 CS 17(CN)6]6 -/8 -
“Condensation“ of cluster fragments ● Y. V. Mironov, N. G. Naumov, S. G. Kozlova, Sung-Jin Kim, V. E. Fedorov, Angew. Chem. , Int. Ed. Engl. , 2005, v. 44, pp. 68676871. [Re 12 CS 17(CN)6]6 -/8 -
![The comparison of rhenium sulfide cyanide anions: [Re 4 S 4(CN)12]4–, [Re 6 S The comparison of rhenium sulfide cyanide anions: [Re 4 S 4(CN)12]4–, [Re 6 S](https://present5.com/presentation/5652994_182163104/image-43.jpg) The comparison of rhenium sulfide cyanide anions: [Re 4 S 4(CN)12]4–, [Re 6 S 8(CN)6]4–/3– and [Re 12 CS 17(CN)6]8–/6–
The comparison of rhenium sulfide cyanide anions: [Re 4 S 4(CN)12]4–, [Re 6 S 8(CN)6]4–/3– and [Re 12 CS 17(CN)6]8–/6–
![[{Ni(NH 3)5}2{Ni(NH 3)4}Re 12 CS 17(CN)6] [{Ni(NH 3)4}3 {Re 12 CS 17(CN)6}] [{Ni(NH 3)5}2{Ni(NH 3)4}Re 12 CS 17(CN)6] [{Ni(NH 3)4}3 {Re 12 CS 17(CN)6}]](https://present5.com/presentation/5652994_182163104/image-44.jpg) [{Ni(NH 3)5}2{Ni(NH 3)4}Re 12 CS 17(CN)6] [{Ni(NH 3)4}3 {Re 12 CS 17(CN)6}]
[{Ni(NH 3)5}2{Ni(NH 3)4}Re 12 CS 17(CN)6] [{Ni(NH 3)4}3 {Re 12 CS 17(CN)6}]
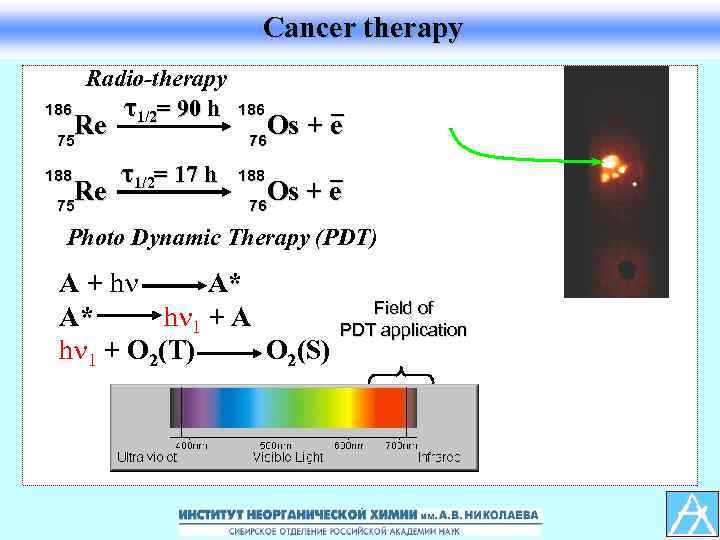 Cancer therapy 186 Radio-therapy τ1/2= 90 h Re 75 188 Re 75 186 _ Os + e 76 τ1/2= 17 h 188 _ Os + e 76 Photo Dynamic Therapy (PDT) A + h A* A* h 1 + A h 1 + O 2(T) O 2(S) Field of PDT application
Cancer therapy 186 Radio-therapy τ1/2= 90 h Re 75 188 Re 75 186 _ Os + e 76 τ1/2= 17 h 188 _ Os + e 76 Photo Dynamic Therapy (PDT) A + h A* A* h 1 + A h 1 + O 2(T) O 2(S) Field of PDT application
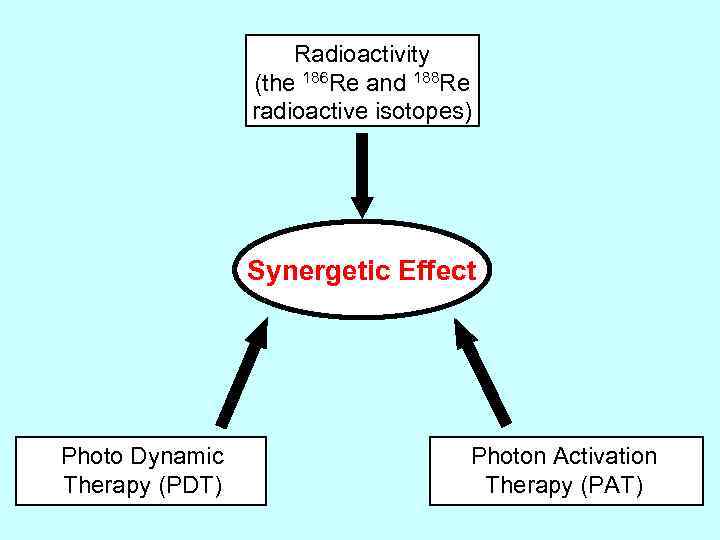 Radioactivity (the 186 Re and 188 Re radioactive isotopes) Synergetic Effect Photo Dynamic Therapy (PDT) Photon Activation Therapy (PAT)
Radioactivity (the 186 Re and 188 Re radioactive isotopes) Synergetic Effect Photo Dynamic Therapy (PDT) Photon Activation Therapy (PAT)
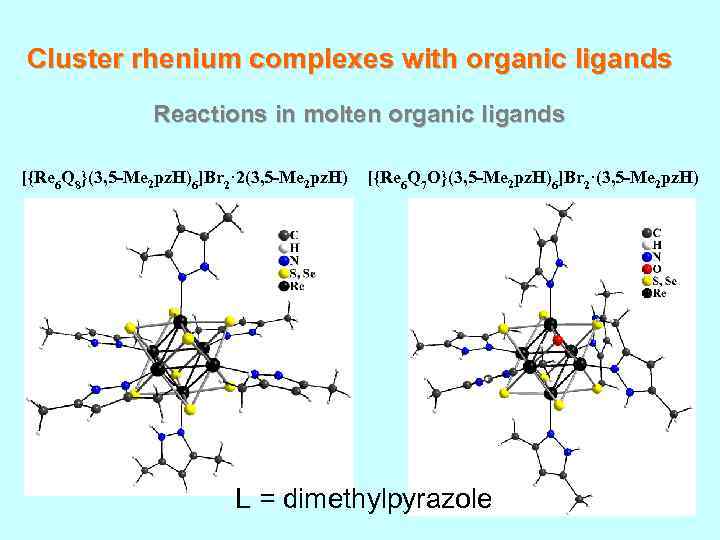 Cluster rhenium complexes with organic ligands Reactions in molten organic ligands [{Re 6 Q 8}(3, 5 -Me 2 pz. H)6]Br 2· 2(3, 5 -Me 2 pz. H) [{Re 6 Q 7 O}(3, 5 -Me 2 pz. H)6]Br 2·(3, 5 -Me 2 pz. H) L = dimethylpyrazole
Cluster rhenium complexes with organic ligands Reactions in molten organic ligands [{Re 6 Q 8}(3, 5 -Me 2 pz. H)6]Br 2· 2(3, 5 -Me 2 pz. H) [{Re 6 Q 7 O}(3, 5 -Me 2 pz. H)6]Br 2·(3, 5 -Me 2 pz. H) L = dimethylpyrazole
![Reactions in molten organic ligands Pyrazine (pyz) Cs 4[{Re 6 S 8}Br 6] Reactions in molten organic ligands Pyrazine (pyz) Cs 4[{Re 6 S 8}Br 6]](https://present5.com/presentation/5652994_182163104/image-48.jpg) Reactions in molten organic ligands Pyrazine (pyz) Cs 4[{Re 6 S 8}Br 6] Cs 3[{Re 6 Q 7 Br}Br 6] (Q=S, Se) 160°C cis-[{Re 6 S 8}(pyz)4 Br 2]·pyz fac-[{Re 6 Q 7 Br}(pyz)3 Br 3]
Reactions in molten organic ligands Pyrazine (pyz) Cs 4[{Re 6 S 8}Br 6] Cs 3[{Re 6 Q 7 Br}Br 6] (Q=S, Se) 160°C cis-[{Re 6 S 8}(pyz)4 Br 2]·pyz fac-[{Re 6 Q 7 Br}(pyz)3 Br 3]
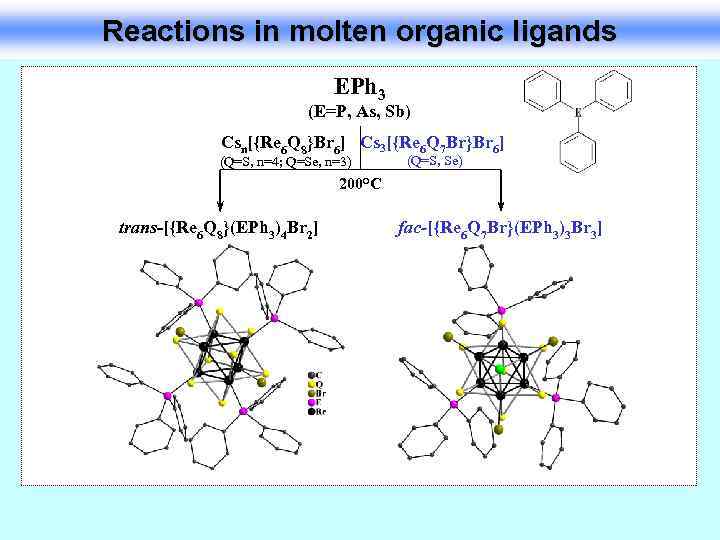 Reactions in molten organic ligands EPh 3 (E=P, As, Sb) Csn[{Re 6 Q 8}Br 6] Cs 3[{Re 6 Q 7 Br}Br 6] (Q=S, n=4; Q=Se, n=3) (Q=S, Se) 200°C trans-[{Re 6 Q 8}(EPh 3)4 Br 2] fac-[{Re 6 Q 7 Br}(EPh 3)3 Br 3]
Reactions in molten organic ligands EPh 3 (E=P, As, Sb) Csn[{Re 6 Q 8}Br 6] Cs 3[{Re 6 Q 7 Br}Br 6] (Q=S, n=4; Q=Se, n=3) (Q=S, Se) 200°C trans-[{Re 6 Q 8}(EPh 3)4 Br 2] fac-[{Re 6 Q 7 Br}(EPh 3)3 Br 3]
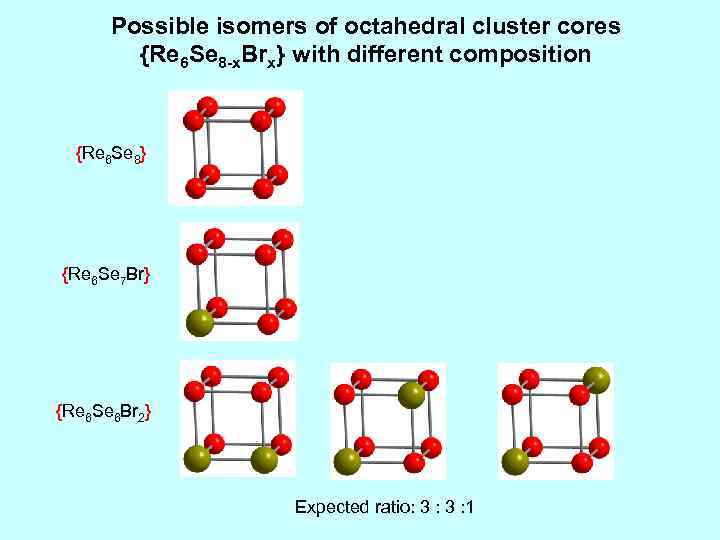 Possible isomers of octahedral cluster cores {Re 6 Se 8 -x. Brx} with different composition {Re 6 Se 8} {Re 6 Se 7 Br} {Re 6 Se 6 Br 2} Expected ratio: 3 : 1
Possible isomers of octahedral cluster cores {Re 6 Se 8 -x. Brx} with different composition {Re 6 Se 8} {Re 6 Se 7 Br} {Re 6 Se 6 Br 2} Expected ratio: 3 : 1
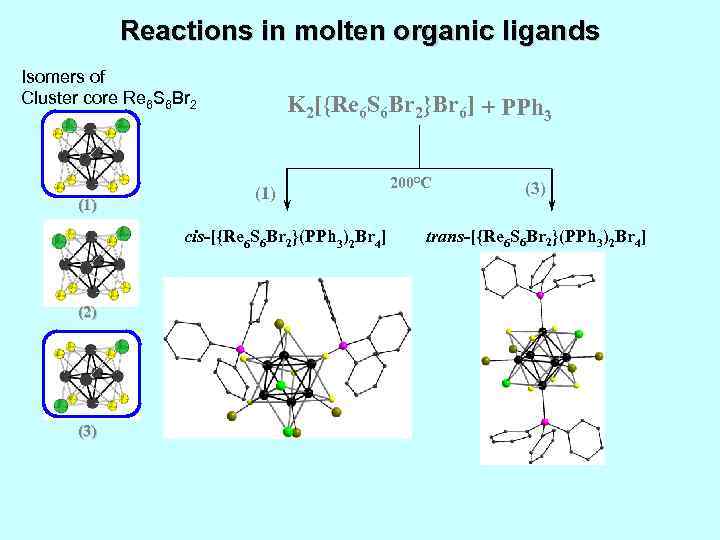 Reactions in molten organic ligands Isomers of Cluster core Re 6 S 6 Br 2 (1) K 2[{Re 6 S 6 Br 2}Br 6] + PPh 3 (1) cis-[{Re 6 S 6 Br 2}(PPh 3)2 Br 4] (2) (3) 200°C (3) trans-[{Re 6 S 6 Br 2}(PPh 3)2 Br 4]
Reactions in molten organic ligands Isomers of Cluster core Re 6 S 6 Br 2 (1) K 2[{Re 6 S 6 Br 2}Br 6] + PPh 3 (1) cis-[{Re 6 S 6 Br 2}(PPh 3)2 Br 4] (2) (3) 200°C (3) trans-[{Re 6 S 6 Br 2}(PPh 3)2 Br 4]
![Octahedral rhenium cluster complexes with new ligands [Re 6 S 8(CH 3 NH 2)2(OH)4]2 Octahedral rhenium cluster complexes with new ligands [Re 6 S 8(CH 3 NH 2)2(OH)4]2](https://present5.com/presentation/5652994_182163104/image-52.jpg) Octahedral rhenium cluster complexes with new ligands [Re 6 S 8(CH 3 NH 2)2(OH)4]2 - [Re 6 S 8(CH 3 COO)6]4 -
Octahedral rhenium cluster complexes with new ligands [Re 6 S 8(CH 3 NH 2)2(OH)4]2 - [Re 6 S 8(CH 3 COO)6]4 -
![Octahedral rhenium cluster complexes with new ligands [Re 6 S 8(SO 3)6]10 Octahedral rhenium cluster complexes with new ligands [Re 6 S 8(SO 3)6]10](https://present5.com/presentation/5652994_182163104/image-53.jpg) Octahedral rhenium cluster complexes with new ligands [Re 6 S 8(SO 3)6]10
Octahedral rhenium cluster complexes with new ligands [Re 6 S 8(SO 3)6]10
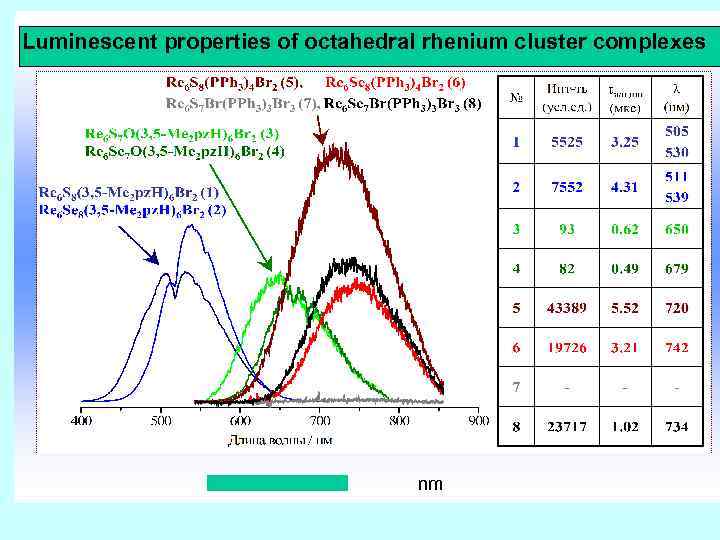 Luminescent properties of octahedral rhenium cluster complexes nm
Luminescent properties of octahedral rhenium cluster complexes nm
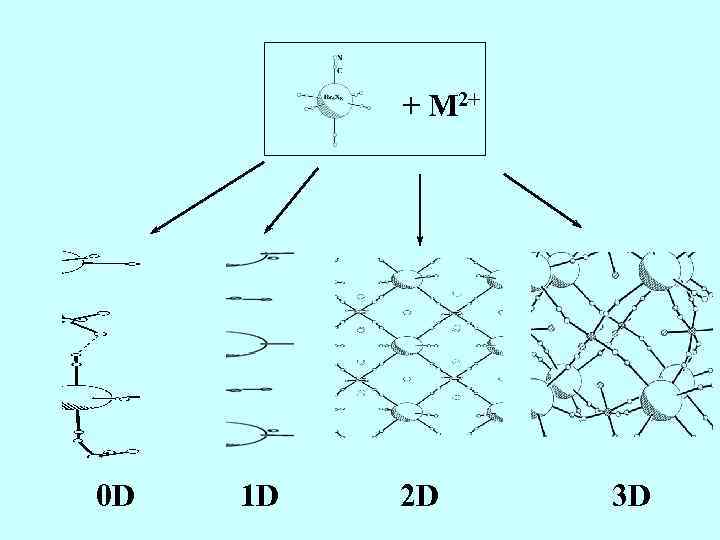 + M 2+ 0 D 1 D 2 D 3 D
+ M 2+ 0 D 1 D 2 D 3 D
![Samples of isomorphic substitution of [M(CN)6] [Re 6 Q 8(CN)6] for some compounds Fe Samples of isomorphic substitution of [M(CN)6] [Re 6 Q 8(CN)6] for some compounds Fe](https://present5.com/presentation/5652994_182163104/image-56.jpg) Samples of isomorphic substitution of [M(CN)6] [Re 6 Q 8(CN)6] for some compounds Fe 4[Fe(CN)6]3 14 H 2 O Fm 3 m a = 10. 166(3) V = 1050. 6 Å3 Fe 4[Re 6 Se 8(CN)6]3 36 H 2 O Fm 3 m a = 14. 2455(10) V = 2890. 9(4) Å3 J. R. Long et al. Na 2 Zn 3[Fe(CN)6]2 9 H 2 O (Me 4 N)[Mn(H 2 O)4][Fe(CN)6] 4 H 2 O Na 2 Cu[Fe(CN)6] n. H 2 O La(H 2 O)3[Cr(CN)6] 2 H 2 O R 3 c a = 12. 469(2) c = 32. 92(2) V = 4432. 6 Å3 Na 2 Zn 3[Re 6 Se 8(CN)6]2 24 H 2 O P 4/n a = 10. 67 b = 10. 568 V = 1203. 2 Å3 (Pr 4 N)2[Mn(H 2 O)4][Re 6 S 8(CN)6] P 12/m 1 a = 6. 998 b = 7. 055 c = 9. 889 = 97. 56 V = 484. 0 Å3 Cs 2[Co(H 2 O)2][Re 6 S 8(CN)6] P 63/m a = 7. 7053(4) c = 14. 8155(9) V = 761. 77(7) Å3 J. R. Long et al. Our data Nd(H 2 O)3[Re 6 Te 8(CN)6] 2 H 2 O Our data R 3 c a = 17. 087(1) c = 49. 643(6) V = 12552(2) Å3 I 4/m a = 13. 255(2) c = 14. 432(3) Å V = 2535. 6(8) Å3 Imma a = 18. 4651(6) b = 10. 6368(3) c = 13. 3162(4) V = 2586. 8(1) Å3 P 63/m a = 9. 303(1) c = 20. 222(4) V = 1515. 7(4) Å3
Samples of isomorphic substitution of [M(CN)6] [Re 6 Q 8(CN)6] for some compounds Fe 4[Fe(CN)6]3 14 H 2 O Fm 3 m a = 10. 166(3) V = 1050. 6 Å3 Fe 4[Re 6 Se 8(CN)6]3 36 H 2 O Fm 3 m a = 14. 2455(10) V = 2890. 9(4) Å3 J. R. Long et al. Na 2 Zn 3[Fe(CN)6]2 9 H 2 O (Me 4 N)[Mn(H 2 O)4][Fe(CN)6] 4 H 2 O Na 2 Cu[Fe(CN)6] n. H 2 O La(H 2 O)3[Cr(CN)6] 2 H 2 O R 3 c a = 12. 469(2) c = 32. 92(2) V = 4432. 6 Å3 Na 2 Zn 3[Re 6 Se 8(CN)6]2 24 H 2 O P 4/n a = 10. 67 b = 10. 568 V = 1203. 2 Å3 (Pr 4 N)2[Mn(H 2 O)4][Re 6 S 8(CN)6] P 12/m 1 a = 6. 998 b = 7. 055 c = 9. 889 = 97. 56 V = 484. 0 Å3 Cs 2[Co(H 2 O)2][Re 6 S 8(CN)6] P 63/m a = 7. 7053(4) c = 14. 8155(9) V = 761. 77(7) Å3 J. R. Long et al. Our data Nd(H 2 O)3[Re 6 Te 8(CN)6] 2 H 2 O Our data R 3 c a = 17. 087(1) c = 49. 643(6) V = 12552(2) Å3 I 4/m a = 13. 255(2) c = 14. 432(3) Å V = 2535. 6(8) Å3 Imma a = 18. 4651(6) b = 10. 6368(3) c = 13. 3162(4) V = 2586. 8(1) Å3 P 63/m a = 9. 303(1) c = 20. 222(4) V = 1515. 7(4) Å3
 57 Способы выращивания монокристаллов для РСт. А 1) диффузия в геле в U-образной трубке; 2) диффузия в трубках; 3) медленное удаление летучего компонента; 4) диффузия через газовую фазу; 5) гидротермальный синтез и многие другие
57 Способы выращивания монокристаллов для РСт. А 1) диффузия в геле в U-образной трубке; 2) диффузия в трубках; 3) медленное удаление летучего компонента; 4) диффузия через газовую фазу; 5) гидротермальный синтез и многие другие


