99060643d63e19e3fa52f8ef66777849.ppt
- Количество слайдов: 55
 NSCLC Adiuvante Dott. Domenico Galetta
NSCLC Adiuvante Dott. Domenico Galetta
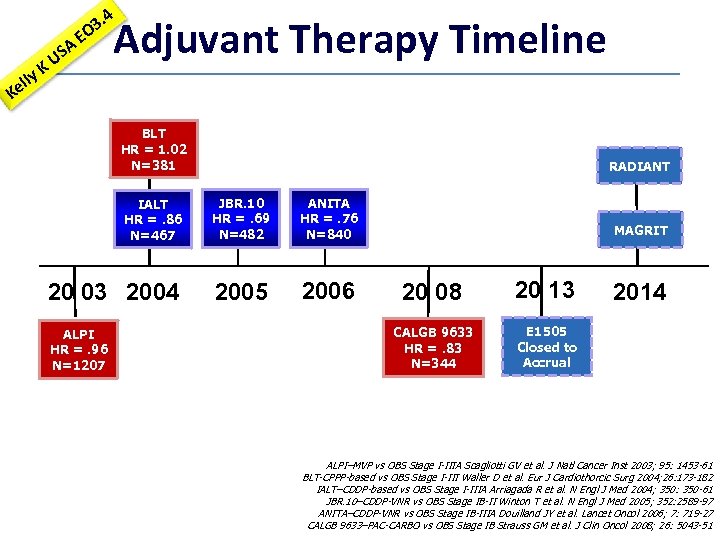 y ell K A US E 3. 4 O Adjuvant Therapy Timeline K BLT HR = 1. 02 N=381 RADIANT IALT HR =. 86 N=467 JBR. 10 HR =. 69 ~ N=482 ANITA HR =. 76 N=840 20 03 2004 2005 2006 ALPI HR =. 96 N=1207 6 MAGRIT 20 08 20 13 CALGB 9633 HR =. 83 N=344 E 1505 Closed to Accrual 2014 ALPI–MVP vs OBS Stage I-IIIA Scagliotti GV et al. J Natl Cancer Inst 2003; 95: 1453 -61 BLT-CPPP-based vs OBS Stage I-III Waller D et al. Eur J Cardiothorcic Surg 2004; 26: 173 -182 IALT–CDDP-based vs OBS Stage I-IIIA Arriagada R et al. N Engl J Med 2004; 350: 350 -61 JBR. 10–CDDP-VNR vs OBS Stage IB-II Winton T et al. N Engl J Med 2005; 352: 2589 -97 ANITA–CDDP-VNR vs OBS Stage IB-IIIA Douilland JY et al. Lancet Oncol 2006; 7: 719 -27 CALGB 9633–PAC-CARBO vs OBS Stage IB Strauss GM et al. J Clin Oncol 2008; 26: 5043 -51
y ell K A US E 3. 4 O Adjuvant Therapy Timeline K BLT HR = 1. 02 N=381 RADIANT IALT HR =. 86 N=467 JBR. 10 HR =. 69 ~ N=482 ANITA HR =. 76 N=840 20 03 2004 2005 2006 ALPI HR =. 96 N=1207 6 MAGRIT 20 08 20 13 CALGB 9633 HR =. 83 N=344 E 1505 Closed to Accrual 2014 ALPI–MVP vs OBS Stage I-IIIA Scagliotti GV et al. J Natl Cancer Inst 2003; 95: 1453 -61 BLT-CPPP-based vs OBS Stage I-III Waller D et al. Eur J Cardiothorcic Surg 2004; 26: 173 -182 IALT–CDDP-based vs OBS Stage I-IIIA Arriagada R et al. N Engl J Med 2004; 350: 350 -61 JBR. 10–CDDP-VNR vs OBS Stage IB-II Winton T et al. N Engl J Med 2005; 352: 2589 -97 ANITA–CDDP-VNR vs OBS Stage IB-IIIA Douilland JY et al. Lancet Oncol 2006; 7: 719 -27 CALGB 9633–PAC-CARBO vs OBS Stage IB Strauss GM et al. J Clin Oncol 2008; 26: 5043 -51
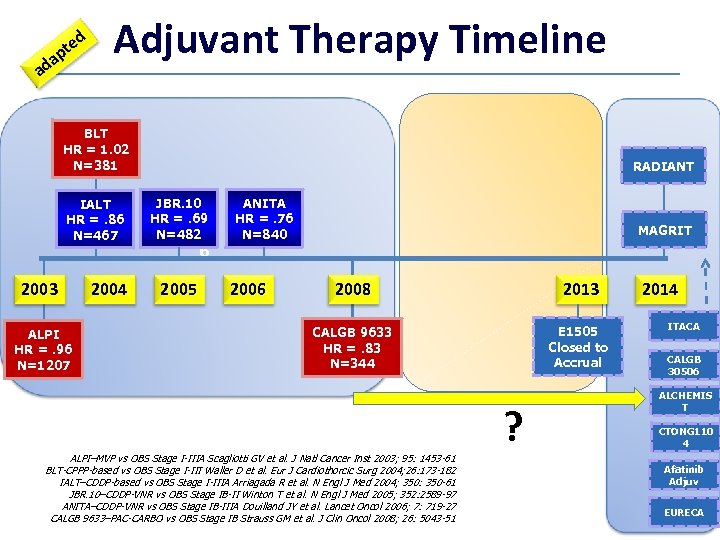 d e pt a ad Adjuvant Therapy Timeline BLT HR = 1. 02 N=381 IALT HR =. 86 N=467 2003 ALPI HR =. 96 N=1207 2004 RADIANT JBR. 10 HR =. 69 N=482~ 6 2005 ANITA HR =. 76 N=840 2006 MAGRIT 2008 2013 CALGB 9633 HR =. 83 N=344 E 1505 Closed to Accrual ALPI–MVP vs OBS Stage I-IIIA Scagliotti GV et al. J Natl Cancer Inst 2003; 95: 1453 -61 BLT-CPPP-based vs OBS Stage I-III Waller D et al. Eur J Cardiothorcic Surg 2004; 26: 173 -182 IALT–CDDP-based vs OBS Stage I-IIIA Arriagada R et al. N Engl J Med 2004; 350: 350 -61 JBR. 10–CDDP-VNR vs OBS Stage IB-II Winton T et al. N Engl J Med 2005; 352: 2589 -97 ANITA–CDDP-VNR vs OBS Stage IB-IIIA Douilland JY et al. Lancet Oncol 2006; 7: 719 -27 CALGB 9633–PAC-CARBO vs OBS Stage IB Strauss GM et al. J Clin Oncol 2008; 26: 5043 -51 ? 2014 ITACA CALGB 30506 ALCHEMIS T CTONG 110 4 Afatinib Adjuv EURECA
d e pt a ad Adjuvant Therapy Timeline BLT HR = 1. 02 N=381 IALT HR =. 86 N=467 2003 ALPI HR =. 96 N=1207 2004 RADIANT JBR. 10 HR =. 69 N=482~ 6 2005 ANITA HR =. 76 N=840 2006 MAGRIT 2008 2013 CALGB 9633 HR =. 83 N=344 E 1505 Closed to Accrual ALPI–MVP vs OBS Stage I-IIIA Scagliotti GV et al. J Natl Cancer Inst 2003; 95: 1453 -61 BLT-CPPP-based vs OBS Stage I-III Waller D et al. Eur J Cardiothorcic Surg 2004; 26: 173 -182 IALT–CDDP-based vs OBS Stage I-IIIA Arriagada R et al. N Engl J Med 2004; 350: 350 -61 JBR. 10–CDDP-VNR vs OBS Stage IB-II Winton T et al. N Engl J Med 2005; 352: 2589 -97 ANITA–CDDP-VNR vs OBS Stage IB-IIIA Douilland JY et al. Lancet Oncol 2006; 7: 719 -27 CALGB 9633–PAC-CARBO vs OBS Stage IB Strauss GM et al. J Clin Oncol 2008; 26: 5043 -51 ? 2014 ITACA CALGB 30506 ALCHEMIS T CTONG 110 4 Afatinib Adjuv EURECA
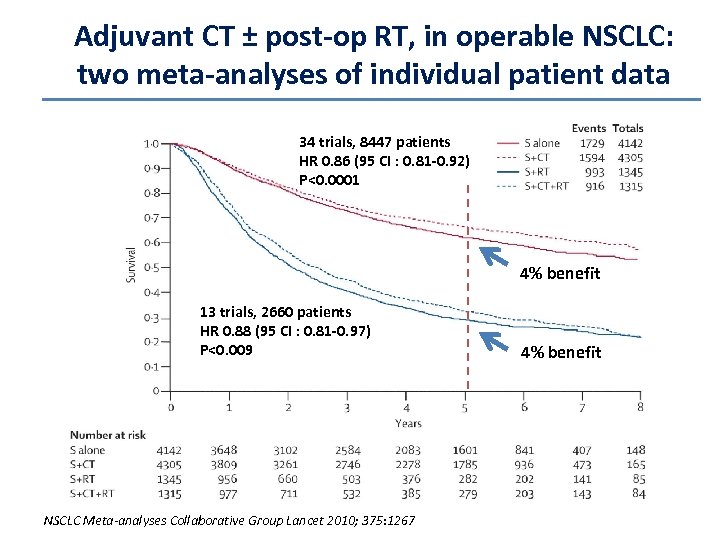 Adjuvant CT ± post-op RT, in operable NSCLC: two meta-analyses of individual patient data 34 trials, 8447 patients HR 0. 86 (95 CI : 0. 81 -0. 92) P<0. 0001 4% benefit 13 trials, 2660 patients HR 0. 88 (95 CI : 0. 81 -0. 97) P<0. 009 NSCLC Meta-analyses Collaborative Group Lancet 2010; 375: 1267 4% benefit
Adjuvant CT ± post-op RT, in operable NSCLC: two meta-analyses of individual patient data 34 trials, 8447 patients HR 0. 86 (95 CI : 0. 81 -0. 92) P<0. 0001 4% benefit 13 trials, 2660 patients HR 0. 88 (95 CI : 0. 81 -0. 97) P<0. 009 NSCLC Meta-analyses Collaborative Group Lancet 2010; 375: 1267 4% benefit
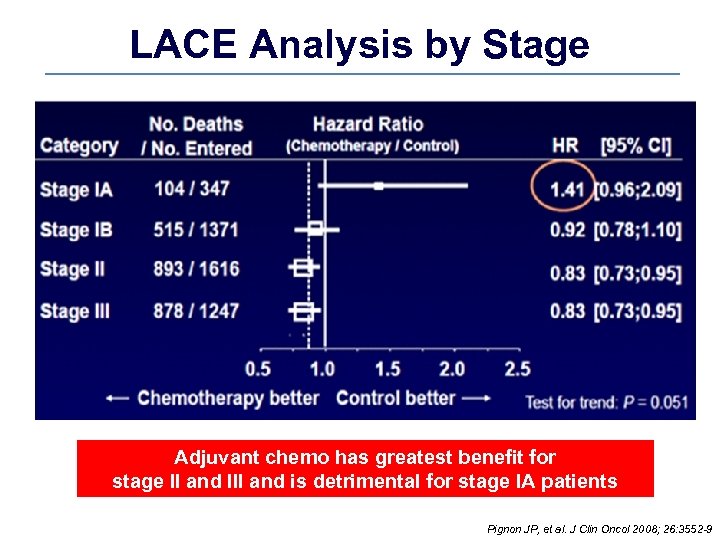 LACE Analysis by Stage Adjuvant chemo has greatest benefit for stage II and III and is detrimental for stage IA patients Pignon JP, et al. J Clin Oncol 2008; 26: 3552 -9
LACE Analysis by Stage Adjuvant chemo has greatest benefit for stage II and III and is detrimental for stage IA patients Pignon JP, et al. J Clin Oncol 2008; 26: 3552 -9
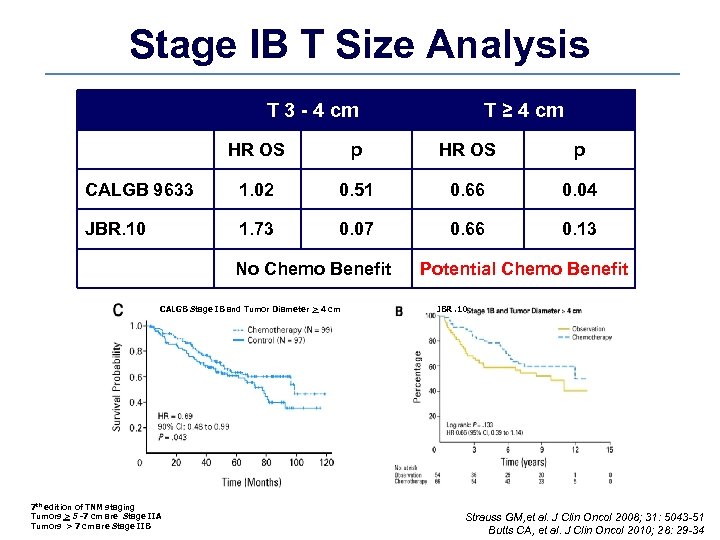 Stage IB T Size Analysis T 3 - 4 cm T ≥ 4 cm HR OS p CALGB 9633 1. 02 0. 51 0. 66 0. 04 JBR. 10 1. 73 0. 07 0. 66 0. 13 No Chemo Benefit CALGB Stage IB and Tumor Diameter > 4 cm 7 th edition of TNM staging Tumors > 5 -7 cm are Stage IIA Tumors > 7 cm are Stage IIB Potential Chemo Benefit JBR. 10 Strauss GM, et al. J Clin Oncol 2008; 31: 5043 -51 Butts CA, et al. J Clin Oncol 2010; 28: 29 -34
Stage IB T Size Analysis T 3 - 4 cm T ≥ 4 cm HR OS p CALGB 9633 1. 02 0. 51 0. 66 0. 04 JBR. 10 1. 73 0. 07 0. 66 0. 13 No Chemo Benefit CALGB Stage IB and Tumor Diameter > 4 cm 7 th edition of TNM staging Tumors > 5 -7 cm are Stage IIA Tumors > 7 cm are Stage IIB Potential Chemo Benefit JBR. 10 Strauss GM, et al. J Clin Oncol 2008; 31: 5043 -51 Butts CA, et al. J Clin Oncol 2010; 28: 29 -34
 y ell K A US E 3. 4 O What Have We Learned? K 1. CDDP based adjuvant chemotherapy improves the cure rate for patients with Stage II-IIIA NSCLC with a PS of 0 -1. 3. No role for adjuvant chemotherapy in patients with a tumor size < 3 cm (Stage IA in both 6 th and 7 th Staging classification). 2. Controversial role for adjuvant chemotherapy in patients with a tumor size of > 4 cm with subset analyses suggesting a benefit. 4. Non-cancer mortality may be increased in patients receiving chemotherapy.
y ell K A US E 3. 4 O What Have We Learned? K 1. CDDP based adjuvant chemotherapy improves the cure rate for patients with Stage II-IIIA NSCLC with a PS of 0 -1. 3. No role for adjuvant chemotherapy in patients with a tumor size < 3 cm (Stage IA in both 6 th and 7 th Staging classification). 2. Controversial role for adjuvant chemotherapy in patients with a tumor size of > 4 cm with subset analyses suggesting a benefit. 4. Non-cancer mortality may be increased in patients receiving chemotherapy.
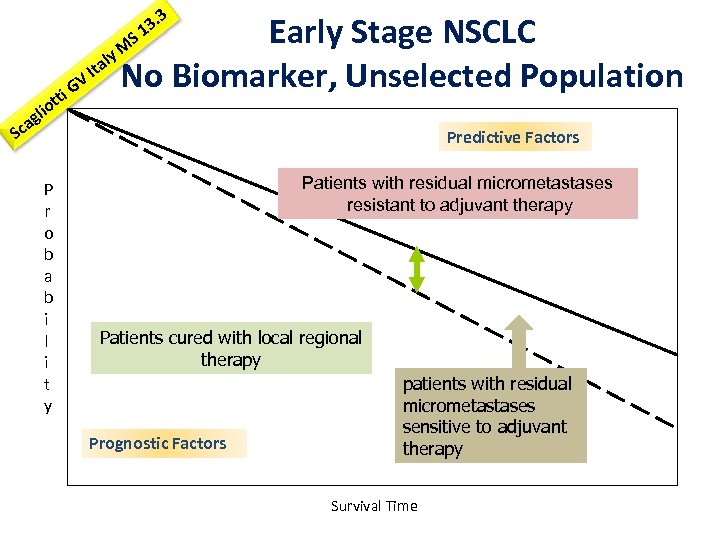 3. 3 1 Early Stage NSCLC l Ita No Biomarker, Unselected Population V i. G t S y. M t o gli a Sc P r o b a b i l i t y Predictive Factors Patients with residual micrometastases resistant to adjuvant therapy Patients cured with local regional therapy Prognostic Factors patients with residual micrometastases sensitive to adjuvant therapy Survival Time
3. 3 1 Early Stage NSCLC l Ita No Biomarker, Unselected Population V i. G t S y. M t o gli a Sc P r o b a b i l i t y Predictive Factors Patients with residual micrometastases resistant to adjuvant therapy Patients cured with local regional therapy Prognostic Factors patients with residual micrometastases sensitive to adjuvant therapy Survival Time
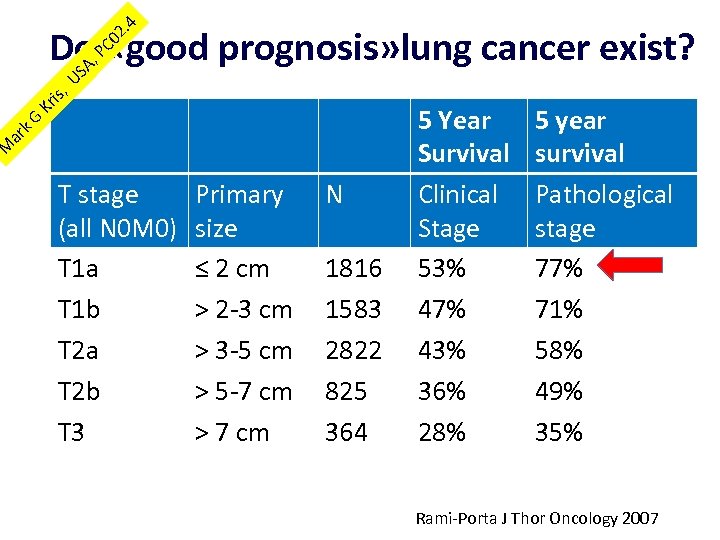 M ar k. G Kr is, US A, PC 02 . 4 Do «good prognosis» lung cancer exist? T stage (all N 0 M 0) T 1 a T 1 b T 2 a T 2 b T 3 Primary size ≤ 2 cm > 2 -3 cm > 3 -5 cm > 5 -7 cm > 7 cm N 1816 1583 2822 825 364 5 Year Survival Clinical Stage 53% 47% 43% 36% 28% 5 year survival Pathological stage 77% 71% 58% 49% 35% Rami-Porta J Thor Oncology 2007
M ar k. G Kr is, US A, PC 02 . 4 Do «good prognosis» lung cancer exist? T stage (all N 0 M 0) T 1 a T 1 b T 2 a T 2 b T 3 Primary size ≤ 2 cm > 2 -3 cm > 3 -5 cm > 5 -7 cm > 7 cm N 1816 1583 2822 825 364 5 Year Survival Clinical Stage 53% 47% 43% 36% 28% 5 year survival Pathological stage 77% 71% 58% 49% 35% Rami-Porta J Thor Oncology 2007
 is Kr M A US PC 2. 4 0 Prognostic factors in lung cancer There are no “good prognosis lung cancers” 23% of patients with tumor less than 2 cm (stage p. T 1 a. N 0 M 0) are dead at 5 years. All patients with breast cancer with this degree of risk are reccomended additional therapy with primary treatment 3. 3 ly ta I M S 1 V • Phase II “proof of concept” studies less applicable to adjuvant setting. i. G tt o. In adjuvant studies overall response rate is NOT an endpoint. • gli a Sc • Survival is much longer and potentially impacted by additional lines of therapy at relapse. • Quality of life issues and adverse events. • Early stage NSCLC are less frequently reported than in other types of tumors (e. g. breast).
is Kr M A US PC 2. 4 0 Prognostic factors in lung cancer There are no “good prognosis lung cancers” 23% of patients with tumor less than 2 cm (stage p. T 1 a. N 0 M 0) are dead at 5 years. All patients with breast cancer with this degree of risk are reccomended additional therapy with primary treatment 3. 3 ly ta I M S 1 V • Phase II “proof of concept” studies less applicable to adjuvant setting. i. G tt o. In adjuvant studies overall response rate is NOT an endpoint. • gli a Sc • Survival is much longer and potentially impacted by additional lines of therapy at relapse. • Quality of life issues and adverse events. • Early stage NSCLC are less frequently reported than in other types of tumors (e. g. breast).
 Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
 Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
 y. M V Use of microarrays in NSCLC o gli a Sc G tti l Ita S 3. 3 1 • • • Need for complicated methods. Large number of genes used in gene profilings. In most of the studies need of fresh tissue. Lack of both reproducibility and independent validation of the results. Genes varied considerably and only few genes have been consistently included. Gene expression profiles can vary according to the microarray platform and the analytic strategy used.
y. M V Use of microarrays in NSCLC o gli a Sc G tti l Ita S 3. 3 1 • • • Need for complicated methods. Large number of genes used in gene profilings. In most of the studies need of fresh tissue. Lack of both reproducibility and independent validation of the results. Genes varied considerably and only few genes have been consistently included. Gene expression profiles can vary according to the microarray platform and the analytic strategy used.
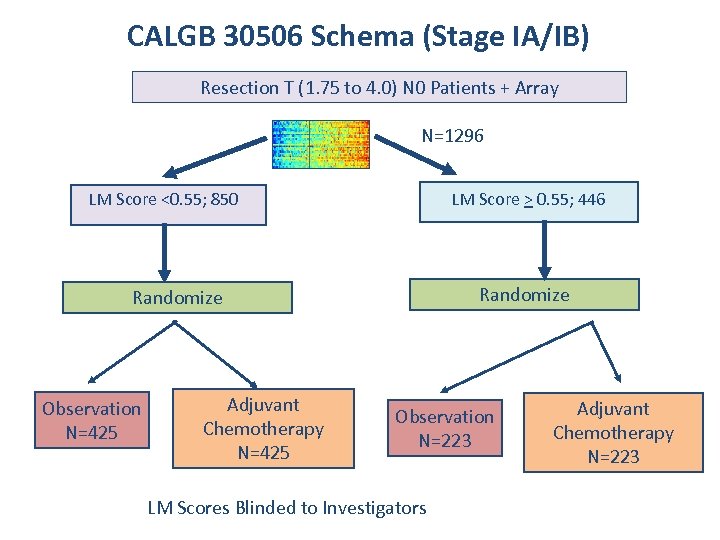 CALGB 30506 Schema (Stage IA/IB) Resection T (1. 75 to 4. 0) N 0 Patients + Array N=1296 LM Score <0. 55; 850 LM Score > 0. 55; 446 Randomize Observation N=425 Adjuvant Chemotherapy N=425 Observation N=223 LM Scores Blinded to Investigators Adjuvant Chemotherapy N=223
CALGB 30506 Schema (Stage IA/IB) Resection T (1. 75 to 4. 0) N 0 Patients + Array N=1296 LM Score <0. 55; 850 LM Score > 0. 55; 446 Randomize Observation N=425 Adjuvant Chemotherapy N=425 Observation N=223 LM Scores Blinded to Investigators Adjuvant Chemotherapy N=223
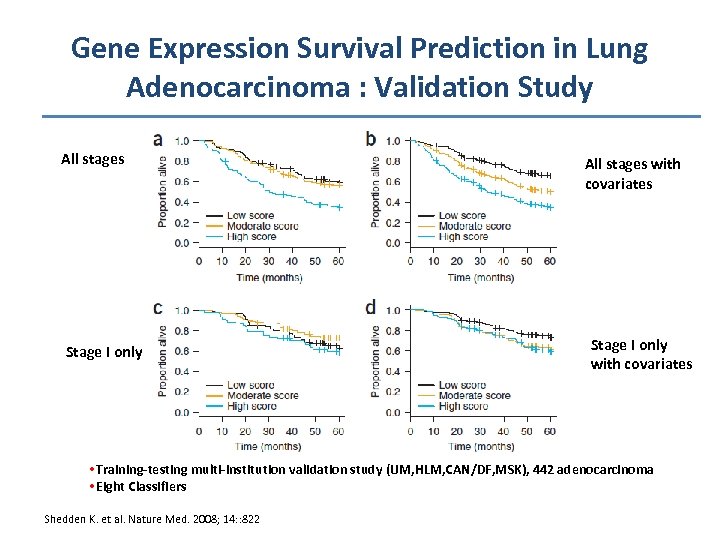 Gene Expression Survival Prediction in Lung Adenocarcinoma : Validation Study All stages Stage I only All stages with covariates Stage I only with covariates • Training-testing multi-institution validation study (UM, HLM, CAN/DF, MSK), 442 adenocarcinoma • Eight Classifiers Shedden K. et al. Nature Med. 2008; 14: : 822
Gene Expression Survival Prediction in Lung Adenocarcinoma : Validation Study All stages Stage I only All stages with covariates Stage I only with covariates • Training-testing multi-institution validation study (UM, HLM, CAN/DF, MSK), 442 adenocarcinoma • Eight Classifiers Shedden K. et al. Nature Med. 2008; 14: : 822
 Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
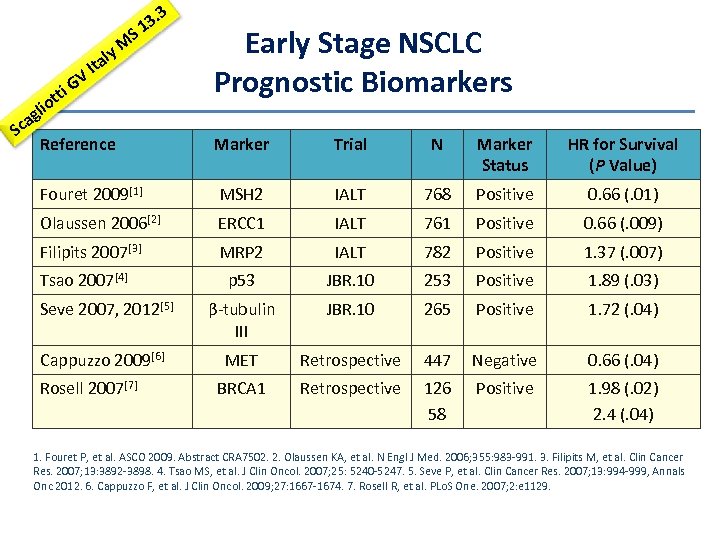 S y. M GV i l Ita 3. 3 1 tt lio g a Sc Reference Early Stage NSCLC Prognostic Biomarkers Marker Trial N Marker Status HR for Survival (P Value) Fouret 2009[1] MSH 2 IALT 768 Positive 0. 66 (. 01) Olaussen 2006[2] ERCC 1 IALT 761 Positive 0. 66 (. 009) Filipits 2007[3] MRP 2 IALT 782 Positive 1. 37 (. 007) p 53 JBR. 10 253 Positive 1. 89 (. 03) Seve 2007, 2012[5] β-tubulin III JBR. 10 265 Positive 1. 72 (. 04) Cappuzzo 2009[6] MET Retrospective 447 Negative 0. 66 (. 04) BRCA 1 Retrospective 126 58 Positive 1. 98 (. 02) 2. 4 (. 04) Tsao 2007[4] Rosell 2007[7] 1. Fouret P, et al. ASCO 2009. Abstract CRA 7502. 2. Olaussen KA, et al. N Engl J Med. 2006; 355: 983 -991. 3. Filipits M, et al. Clin Cancer Res. 2007; 13: 3892 -3898. 4. Tsao MS, et al. J Clin Oncol. 2007; 25: 5240 -5247. 5. Seve P, et al. Clin Cancer Res. 2007; 13: 994 -999, Annals Onc 2012. 6. Cappuzzo F, et al. J Clin Oncol. 2009; 27: 1667 -1674. 7. Rosell R, et al. PLo. S One. 2007; 2: e 1129.
S y. M GV i l Ita 3. 3 1 tt lio g a Sc Reference Early Stage NSCLC Prognostic Biomarkers Marker Trial N Marker Status HR for Survival (P Value) Fouret 2009[1] MSH 2 IALT 768 Positive 0. 66 (. 01) Olaussen 2006[2] ERCC 1 IALT 761 Positive 0. 66 (. 009) Filipits 2007[3] MRP 2 IALT 782 Positive 1. 37 (. 007) p 53 JBR. 10 253 Positive 1. 89 (. 03) Seve 2007, 2012[5] β-tubulin III JBR. 10 265 Positive 1. 72 (. 04) Cappuzzo 2009[6] MET Retrospective 447 Negative 0. 66 (. 04) BRCA 1 Retrospective 126 58 Positive 1. 98 (. 02) 2. 4 (. 04) Tsao 2007[4] Rosell 2007[7] 1. Fouret P, et al. ASCO 2009. Abstract CRA 7502. 2. Olaussen KA, et al. N Engl J Med. 2006; 355: 983 -991. 3. Filipits M, et al. Clin Cancer Res. 2007; 13: 3892 -3898. 4. Tsao MS, et al. J Clin Oncol. 2007; 25: 5240 -5247. 5. Seve P, et al. Clin Cancer Res. 2007; 13: 994 -999, Annals Onc 2012. 6. Cappuzzo F, et al. J Clin Oncol. 2009; 27: 1667 -1674. 7. Rosell R, et al. PLo. S One. 2007; 2: e 1129.
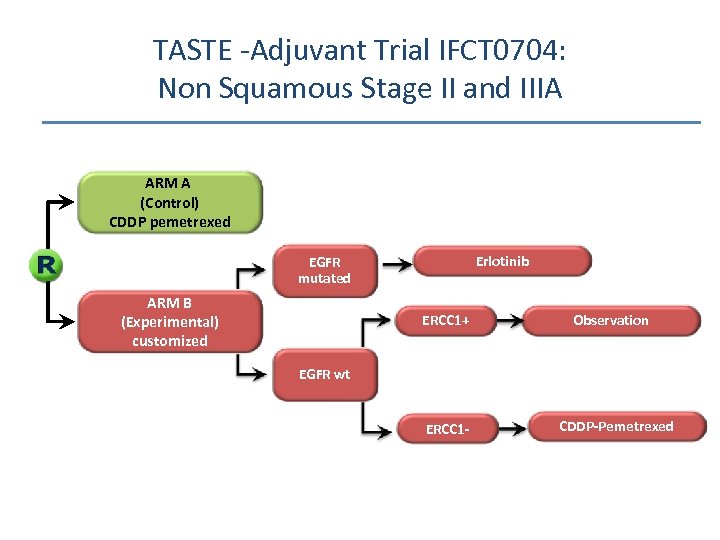 TASTE -Adjuvant Trial IFCT 0704: Non Squamous Stage II and IIIA ARM A (Control) CDDP pemetrexed Erlotinib EGFR mutated ARM B (Experimental) customized ERCC 1+ Observation EGFR wt ERCC 1 - CDDP-Pemetrexed
TASTE -Adjuvant Trial IFCT 0704: Non Squamous Stage II and IIIA ARM A (Control) CDDP pemetrexed Erlotinib EGFR mutated ARM B (Experimental) customized ERCC 1+ Observation EGFR wt ERCC 1 - CDDP-Pemetrexed
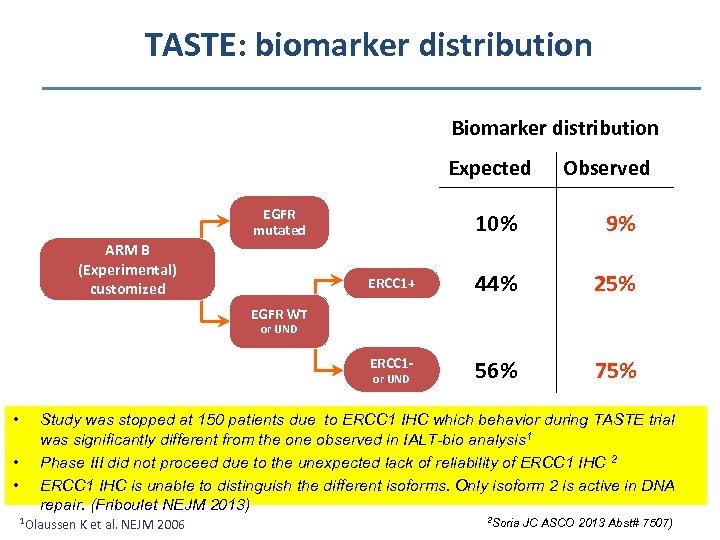 TASTE: biomarker distribution Biomarker distribution Expected EGFR mutated Observed 9% ERCC 1+ 44% 25% ERCC 1 - ARM B (Experimental) customized 10% 56% 75% EGFR WT or UND • • • Study was stopped at 150 patients due to ERCC 1 IHC which behavior during TASTE trial was significantly different from the one observed in IALT-bio analysis 1 Phase III did not proceed due to the unexpected lack of reliability of ERCC 1 IHC 2 ERCC 1 IHC is unable to distinguish the different isoforms. Only isoform 2 is active in DNA repair. (Friboulet NEJM 2013) 1 Olaussen K et al. NEJM 2006 2 Soria JC ASCO 2013 Abst# 7507)
TASTE: biomarker distribution Biomarker distribution Expected EGFR mutated Observed 9% ERCC 1+ 44% 25% ERCC 1 - ARM B (Experimental) customized 10% 56% 75% EGFR WT or UND • • • Study was stopped at 150 patients due to ERCC 1 IHC which behavior during TASTE trial was significantly different from the one observed in IALT-bio analysis 1 Phase III did not proceed due to the unexpected lack of reliability of ERCC 1 IHC 2 ERCC 1 IHC is unable to distinguish the different isoforms. Only isoform 2 is active in DNA repair. (Friboulet NEJM 2013) 1 Olaussen K et al. NEJM 2006 2 Soria JC ASCO 2013 Abst# 7507)
 i t su s in pa BS a M . 01 8 Oo M First analysis of toxicity and treament compliance in customized postoperative chemotherapy based on BRCA 1 levels after NSCLC resection: SCAT (Spanish Customized Adjuvant Therapy) trial. Spanish Lung Cancer Group/GECP Bartomeu Massuti 1, Manuel Cobo 2, Manuel Rodriguez-Paniagua 1, Isabel Ballesteros 3, Teresa Moran 4, Ricardo Arrabal 2, Jose Luis Gonzalez Larriba 5, Isidoro Barneto 6, Yat Wah Pun 3, Javier de. Castro Carpeño 7, Lara Iglesias 8, Carlos Baamonde 6, Miguel Angel Muñoz 9, Guillermo Lopez. Vivanco 10, JJ Rivas de Andres 11, Dolores Isla 12, Rafael Lopez 13, Ramon De Las Peñas 14, Delvis Rodriguez 15, Pedro Lopez De Castro 16, Angel Artal 17, Emilio Esteban Gonzalez 18, Florentino Hernando Trancho 19, Mariano Provencio 20, J Valdivia 21, Prudencio Diaz Agero 7, Jose Luis Martin De Nicolas 8, Eva Pereira 22, Jose Miguel Sanchez 23, Rafael Rosell 16; 1 Alicante University Hospital, Alicante/SPAIN, 2 Hospital Carlos Haya, Malaga/SPAIN, 3 Hospital La Princesa, Madrid/SPAIN, 4 Catalan Institute of Oncology, Badalona/SPAIN, 5 Hospital Clínico San Carlos, Madrid/SPAIN, 6 Hospital Reina Sofia, Cordoba/SPAIN, 7 Hospital Universitario La Paz, Madrid/SPAIN, 8 Hospital 12 de Octubre, Madrid/SPAIN, 9 Instituto Valenciano Oncología, Valencia/SPAIN, 10 Hospital de Cruces de Barakaldo, Vizcaya/SPAIN, 11 Hospital Miguel Servet, Zaragoza/SPAIN, 12 Hospital Lozano Blesa, Zaragoza/SPAIN, 13 Hospital Clinico Universitario de Santiago de Compostela, Santiago De Compostela/SPAIN, 14 Hospital Provincial de Castellón, Castellón/SPAIN, 15 Hospital Universitario Insular de Gran Canaria, Las Palmas De Gran Canaria/SPAIN, 16 Hospital Germans Trias i Pujol, Badalona/SPAIN, 17 Hospital Universitario Miguel Servet, Zaragoza/SPAIN, 18 Hospital Universitario Central de Asturias, Oviedo/SPAIN, 19 Hospital Clinico San Carlos, Madrid/SPAIN, 20 Hospital Puerta de Hierro, Madrid/SPAIN, 21 Hospital Virgen de las Nieves, Granada/SPAIN, 22 Grupo Español de Cancer de Pulmon (GECP), Barcelona/SPAIN, 23 MD Anderson Cancer Center, Madrid/SPAIN
i t su s in pa BS a M . 01 8 Oo M First analysis of toxicity and treament compliance in customized postoperative chemotherapy based on BRCA 1 levels after NSCLC resection: SCAT (Spanish Customized Adjuvant Therapy) trial. Spanish Lung Cancer Group/GECP Bartomeu Massuti 1, Manuel Cobo 2, Manuel Rodriguez-Paniagua 1, Isabel Ballesteros 3, Teresa Moran 4, Ricardo Arrabal 2, Jose Luis Gonzalez Larriba 5, Isidoro Barneto 6, Yat Wah Pun 3, Javier de. Castro Carpeño 7, Lara Iglesias 8, Carlos Baamonde 6, Miguel Angel Muñoz 9, Guillermo Lopez. Vivanco 10, JJ Rivas de Andres 11, Dolores Isla 12, Rafael Lopez 13, Ramon De Las Peñas 14, Delvis Rodriguez 15, Pedro Lopez De Castro 16, Angel Artal 17, Emilio Esteban Gonzalez 18, Florentino Hernando Trancho 19, Mariano Provencio 20, J Valdivia 21, Prudencio Diaz Agero 7, Jose Luis Martin De Nicolas 8, Eva Pereira 22, Jose Miguel Sanchez 23, Rafael Rosell 16; 1 Alicante University Hospital, Alicante/SPAIN, 2 Hospital Carlos Haya, Malaga/SPAIN, 3 Hospital La Princesa, Madrid/SPAIN, 4 Catalan Institute of Oncology, Badalona/SPAIN, 5 Hospital Clínico San Carlos, Madrid/SPAIN, 6 Hospital Reina Sofia, Cordoba/SPAIN, 7 Hospital Universitario La Paz, Madrid/SPAIN, 8 Hospital 12 de Octubre, Madrid/SPAIN, 9 Instituto Valenciano Oncología, Valencia/SPAIN, 10 Hospital de Cruces de Barakaldo, Vizcaya/SPAIN, 11 Hospital Miguel Servet, Zaragoza/SPAIN, 12 Hospital Lozano Blesa, Zaragoza/SPAIN, 13 Hospital Clinico Universitario de Santiago de Compostela, Santiago De Compostela/SPAIN, 14 Hospital Provincial de Castellón, Castellón/SPAIN, 15 Hospital Universitario Insular de Gran Canaria, Las Palmas De Gran Canaria/SPAIN, 16 Hospital Germans Trias i Pujol, Badalona/SPAIN, 17 Hospital Universitario Miguel Servet, Zaragoza/SPAIN, 18 Hospital Universitario Central de Asturias, Oviedo/SPAIN, 19 Hospital Clinico San Carlos, Madrid/SPAIN, 20 Hospital Puerta de Hierro, Madrid/SPAIN, 21 Hospital Virgen de las Nieves, Granada/SPAIN, 22 Grupo Español de Cancer de Pulmon (GECP), Barcelona/SPAIN, 23 MD Anderson Cancer Center, Madrid/SPAIN
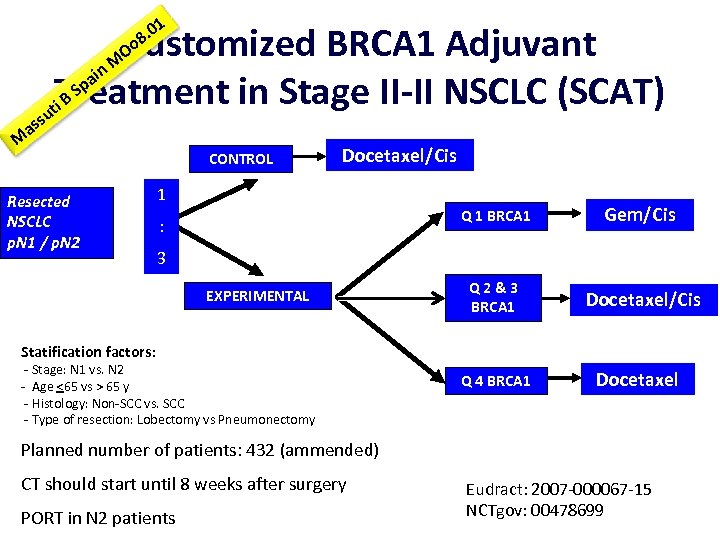 . 01 8 Oo Customized BRCA 1 Adjuvant i pa S Treatment in Stage II-II NSCLC (SCAT) i. B ut n. M s as M Resected NSCLC p. N 1 / p. N 2 CONTROL Docetaxel/Cis 1 Q 1 BRCA 1 : Gem/Cis Q 2&3 BRCA 1 Docetaxel/Cis 3 EXPERIMENTAL Statification factors: - Stage: N 1 vs. N 2 - Age <65 vs > 65 y - Histology: Non-SCC vs. SCC - Type of resection: Lobectomy vs Pneumonectomy Q 4 BRCA 1 Docetaxel Planned number of patients: 432 (ammended) CT should start until 8 weeks after surgery PORT in N 2 patients Eudract: 2007 -000067 -15 NCTgov: 00478699
. 01 8 Oo Customized BRCA 1 Adjuvant i pa S Treatment in Stage II-II NSCLC (SCAT) i. B ut n. M s as M Resected NSCLC p. N 1 / p. N 2 CONTROL Docetaxel/Cis 1 Q 1 BRCA 1 : Gem/Cis Q 2&3 BRCA 1 Docetaxel/Cis 3 EXPERIMENTAL Statification factors: - Stage: N 1 vs. N 2 - Age <65 vs > 65 y - Histology: Non-SCC vs. SCC - Type of resection: Lobectomy vs Pneumonectomy Q 4 BRCA 1 Docetaxel Planned number of patients: 432 (ammended) CT should start until 8 weeks after surgery PORT in N 2 patients Eudract: 2007 -000067 -15 NCTgov: 00478699
 i M a t su s in pa BS M . 01 8 Oo SCAT: BRCA 1 expression • Median m. RNA BRCA 1 levels: 15. 78 (0. 73 -132) • Quartiles distribution: – Q 1: 212 (42. 4%) – Q 2 -3: 150 (30%) – Q 4: 138 (27. 6%) • Mean BRCA 1: – Adenocarcinoma: 6. 95 vs Squamous 20. 29 (p<0. 001) • EGFR mut: 5. 6% (incomplete data)
i M a t su s in pa BS M . 01 8 Oo SCAT: BRCA 1 expression • Median m. RNA BRCA 1 levels: 15. 78 (0. 73 -132) • Quartiles distribution: – Q 1: 212 (42. 4%) – Q 2 -3: 150 (30%) – Q 4: 138 (27. 6%) • Mean BRCA 1: – Adenocarcinoma: 6. 95 vs Squamous 20. 29 (p<0. 001) • EGFR mut: 5. 6% (incomplete data)
 e nc a r lie C Le v he T a Fr , M 6 8. 0 O 0 SCAT trial
e nc a r lie C Le v he T a Fr , M 6 8. 0 O 0 SCAT trial
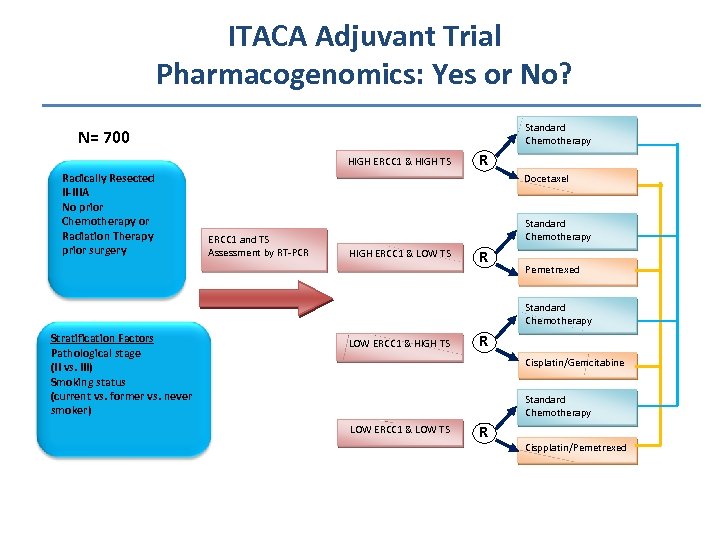 ITACA Adjuvant Trial Pharmacogenomics: Yes or No? Standard Chemotherapy N= 700 HIGH ERCC 1 & HIGH TS Radically Resected II-IIIA No prior Chemotherapy or Radiation Therapy prior surgery R Docetaxel ERCC 1 and TS Assessment by RT-PCR Standard Chemotherapy HIGH ERCC 1 & LOW TS R Pemetrexed Standard Chemotherapy Stratification Factors Pathological stage (II vs. III) Smoking status (current vs. former vs. never smoker) LOW ERCC 1 & HIGH TS R Cisplatin/Gemcitabine Standard Chemotherapy LOW ERCC 1 & LOW TS R Cispplatin/Pemetrexed
ITACA Adjuvant Trial Pharmacogenomics: Yes or No? Standard Chemotherapy N= 700 HIGH ERCC 1 & HIGH TS Radically Resected II-IIIA No prior Chemotherapy or Radiation Therapy prior surgery R Docetaxel ERCC 1 and TS Assessment by RT-PCR Standard Chemotherapy HIGH ERCC 1 & LOW TS R Pemetrexed Standard Chemotherapy Stratification Factors Pathological stage (II vs. III) Smoking status (current vs. former vs. never smoker) LOW ERCC 1 & HIGH TS R Cisplatin/Gemcitabine Standard Chemotherapy LOW ERCC 1 & LOW TS R Cispplatin/Pemetrexed
 Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
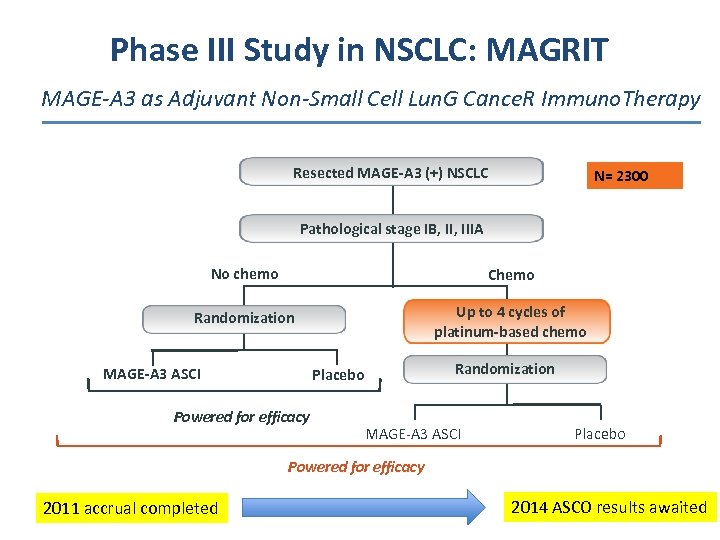 Phase III Study in NSCLC: MAGRIT MAGE-A 3 as Adjuvant Non-Small Cell Lun. G Cance. R Immuno. Therapy I Resected MAGE-A 3 (+) NSCLC N= 2300 Pathological stage IB, IIIA No chemo Chemo Randomization Up to 4 cycles of platinum-based chemo MAGE-A 3 ASCI Randomization Placebo Powered for efficacy MAGE-A 3 ASCI Placebo Powered -for-efficacy 27 2011 accrual completed 2014 ASCO results awaited
Phase III Study in NSCLC: MAGRIT MAGE-A 3 as Adjuvant Non-Small Cell Lun. G Cance. R Immuno. Therapy I Resected MAGE-A 3 (+) NSCLC N= 2300 Pathological stage IB, IIIA No chemo Chemo Randomization Up to 4 cycles of platinum-based chemo MAGE-A 3 ASCI Randomization Placebo Powered for efficacy MAGE-A 3 ASCI Placebo Powered -for-efficacy 27 2011 accrual completed 2014 ASCO results awaited
 Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
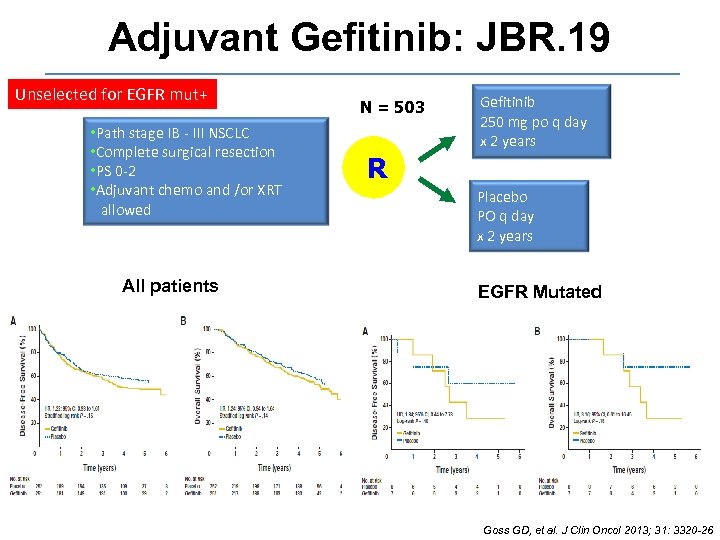 Adjuvant Gefitinib: JBR. 19 Unselected for EGFR mut+ • Path stage IB - III NSCLC • Complete surgical resection • PS 0 -2 • Adjuvant chemo and /or XRT allowed All patients N = 503 R Gefitinib 250 mg po q day x 2 years Placebo PO q day x 2 years EGFR Mutated Goss GD, et al. J Clin Oncol 2013; 31: 3320 -26
Adjuvant Gefitinib: JBR. 19 Unselected for EGFR mut+ • Path stage IB - III NSCLC • Complete surgical resection • PS 0 -2 • Adjuvant chemo and /or XRT allowed All patients N = 503 R Gefitinib 250 mg po q day x 2 years Placebo PO q day x 2 years EGFR Mutated Goss GD, et al. J Clin Oncol 2013; 31: 3320 -26
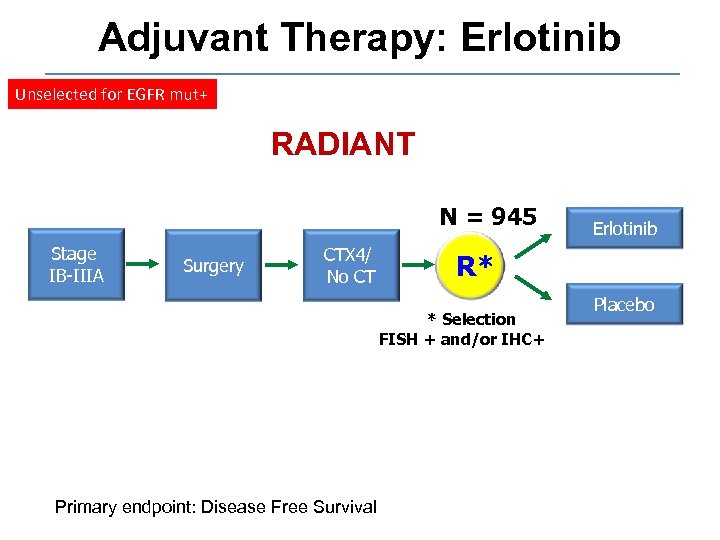 Adjuvant Therapy: Erlotinib Unselected for EGFR mut+ RADIANT N = 945 Stage IB-IIIA Surgery CTX 4/ No CT R* * Selection FISH + and/or IHC+ Primary endpoint: Disease Free Survival Erlotinib Placebo
Adjuvant Therapy: Erlotinib Unselected for EGFR mut+ RADIANT N = 945 Stage IB-IIIA Surgery CTX 4/ No CT R* * Selection FISH + and/or IHC+ Primary endpoint: Disease Free Survival Erlotinib Placebo
 . 4 PC 02 US A, is, Kr k. G M ar EGFR mutation in early stage Lung Cancer: Rationale • • • All stages need more cure Eradicating metastases the goal EGFR as target Results in patients with early stages Trials in progress Seizing the opportunities
. 4 PC 02 US A, is, Kr k. G M ar EGFR mutation in early stage Lung Cancer: Rationale • • • All stages need more cure Eradicating metastases the goal EGFR as target Results in patients with early stages Trials in progress Seizing the opportunities
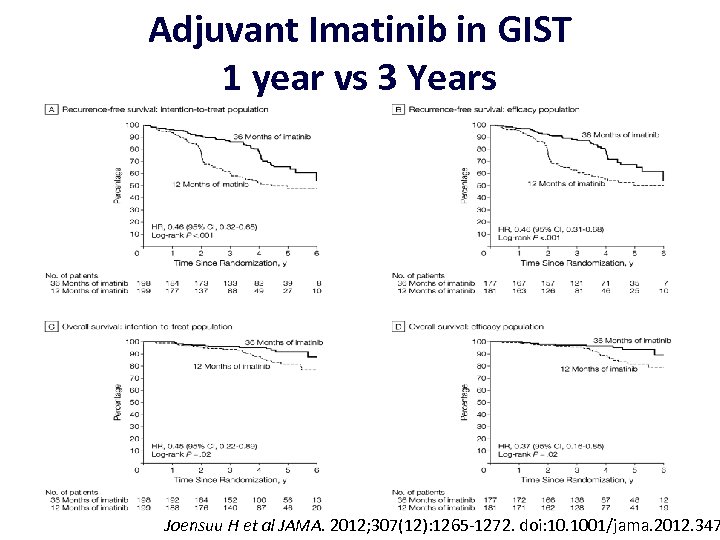 Adjuvant Imatinib in GIST 1 year vs 3 Years Joensuu H et al JAMA. 2012; 307(12): 1265 -1272. doi: 10. 1001/jama. 2012. 347
Adjuvant Imatinib in GIST 1 year vs 3 Years Joensuu H et al JAMA. 2012; 307(12): 1265 -1272. doi: 10. 1001/jama. 2012. 347
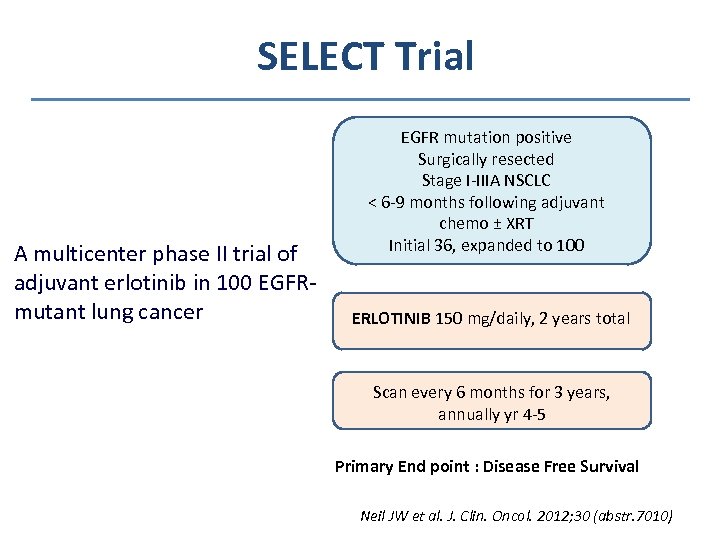 SELECT Trial A multicenter phase II trial of adjuvant erlotinib in 100 EGFRmutant lung cancer EGFR mutation positive Surgically resected Stage I-IIIA NSCLC < 6 -9 months following adjuvant chemo ± XRT Initial 36, expanded to 100 ERLOTINIB 150 mg/daily, 2 years total Scan every 6 months for 3 years, annually yr 4 -5 Primary End point : Disease Free Survival Neil JW et al. J. Clin. Oncol. 2012; 30 (abstr. 7010)
SELECT Trial A multicenter phase II trial of adjuvant erlotinib in 100 EGFRmutant lung cancer EGFR mutation positive Surgically resected Stage I-IIIA NSCLC < 6 -9 months following adjuvant chemo ± XRT Initial 36, expanded to 100 ERLOTINIB 150 mg/daily, 2 years total Scan every 6 months for 3 years, annually yr 4 -5 Primary End point : Disease Free Survival Neil JW et al. J. Clin. Oncol. 2012; 30 (abstr. 7010)
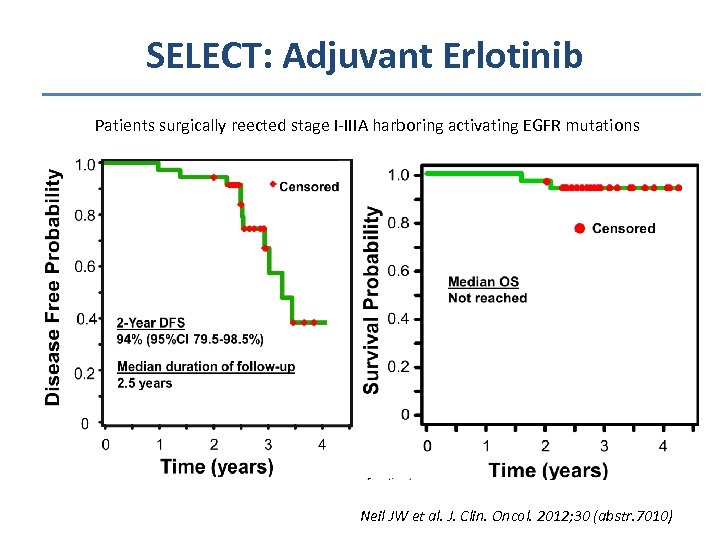 SELECT: Adjuvant Erlotinib Patients surgically reected stage I-IIIA harboring activating EGFR mutations Neil JW et al. J. Clin. Oncol. 2012; 30 (abstr. 7010)
SELECT: Adjuvant Erlotinib Patients surgically reected stage I-IIIA harboring activating EGFR mutations Neil JW et al. J. Clin. Oncol. 2012; 30 (abstr. 7010)
 Further hurdles to be considered with molecular alterations…. . • Is the alteration equally present in early disease? • Is the molecular alteration stable overtime? • Is the targeted treatment equally effective as adjuvant (maintenance) treatment or should be reserved at relapse? • Are long term toxicities tolerable?
Further hurdles to be considered with molecular alterations…. . • Is the alteration equally present in early disease? • Is the molecular alteration stable overtime? • Is the targeted treatment equally effective as adjuvant (maintenance) treatment or should be reserved at relapse? • Are long term toxicities tolerable?
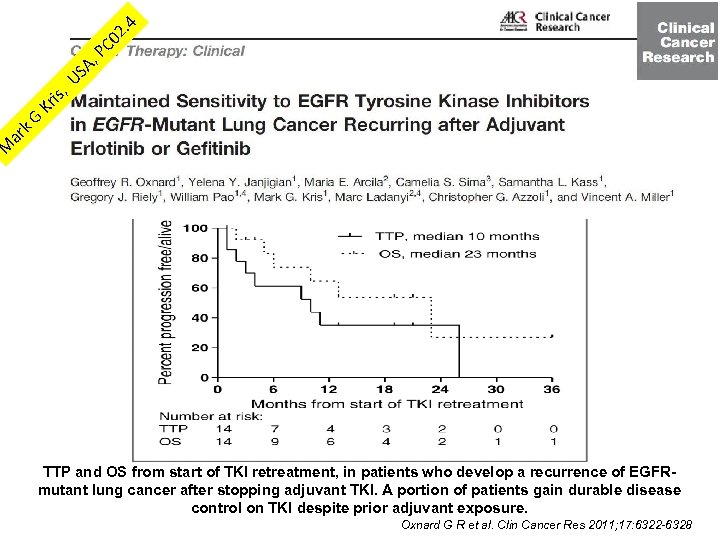 . 4 PC 02 US A, is, Kr k. G ar M TTP and OS from start of TKI retreatment, in patients who develop a recurrence of EGFRmutant lung cancer after stopping adjuvant TKI. A portion of patients gain durable disease control on TKI despite prior adjuvant exposure. Oxnard G R et al. Clin Cancer Res 2011; 17: 6322 -6328
. 4 PC 02 US A, is, Kr k. G ar M TTP and OS from start of TKI retreatment, in patients who develop a recurrence of EGFRmutant lung cancer after stopping adjuvant TKI. A portion of patients gain durable disease control on TKI despite prior adjuvant exposure. Oxnard G R et al. Clin Cancer Res 2011; 17: 6322 -6328
 M ar k. G Kr is, US A, PC 02 . 4 EURECA Erlotinib Used as Adjuvant Therapy in Resected EGFR mutant Lung Carcinoma • Resected stage I-III EGFRmutation positive lung cancer with activating EGFR mutation(exon 19 deletion, L 858 R, L 861 Q, G 719 X) • Perioperative citotoxyc chemotherapy and radiation therapy as indicated • Stratified by staging and perioperative chemotherapy N=286 Adjuvant erlotinib up to 24 months with CT chest every 6 months than yearly N=190 CT chest every 6 months than yearly N=96 • CT scan at 30 months • Follow patients for recurrence or death • Patients with recurrence will be biopsed to confirm recurrence and test for molecular determination of acquired resistance • Record of subsequent chemotherapies Lung Cancer Mutation Consortium PI Cristopher Azzoli
M ar k. G Kr is, US A, PC 02 . 4 EURECA Erlotinib Used as Adjuvant Therapy in Resected EGFR mutant Lung Carcinoma • Resected stage I-III EGFRmutation positive lung cancer with activating EGFR mutation(exon 19 deletion, L 858 R, L 861 Q, G 719 X) • Perioperative citotoxyc chemotherapy and radiation therapy as indicated • Stratified by staging and perioperative chemotherapy N=286 Adjuvant erlotinib up to 24 months with CT chest every 6 months than yearly N=190 CT chest every 6 months than yearly N=96 • CT scan at 30 months • Follow patients for recurrence or death • Patients with recurrence will be biopsed to confirm recurrence and test for molecular determination of acquired resistance • Record of subsequent chemotherapies Lung Cancer Mutation Consortium PI Cristopher Azzoli
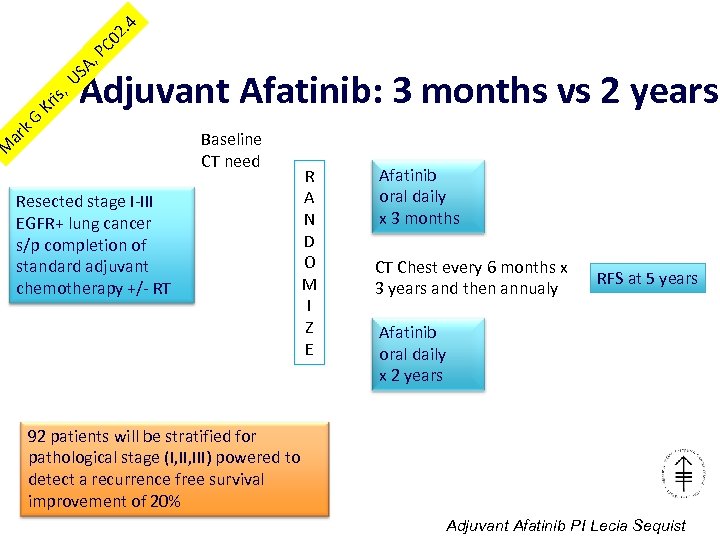 . 4 PC 02 US A, is, Kr M ar k. G Adjuvant Afatinib: 3 months vs 2 years Baseline CT need Resected stage I-III EGFR+ lung cancer s/p completion of standard adjuvant chemotherapy +/- RT R A N D O M I Z E Afatinib oral daily x 3 months CT Chest every 6 months x 3 years and then annualy RFS at 5 years Afatinib oral daily x 2 years 92 patients will be stratified for pathological stage (I, III) powered to detect a recurrence free survival improvement of 20% Adjuvant Afatinib PI Lecia Sequist
. 4 PC 02 US A, is, Kr M ar k. G Adjuvant Afatinib: 3 months vs 2 years Baseline CT need Resected stage I-III EGFR+ lung cancer s/p completion of standard adjuvant chemotherapy +/- RT R A N D O M I Z E Afatinib oral daily x 3 months CT Chest every 6 months x 3 years and then annualy RFS at 5 years Afatinib oral daily x 2 years 92 patients will be stratified for pathological stage (I, III) powered to detect a recurrence free survival improvement of 20% Adjuvant Afatinib PI Lecia Sequist
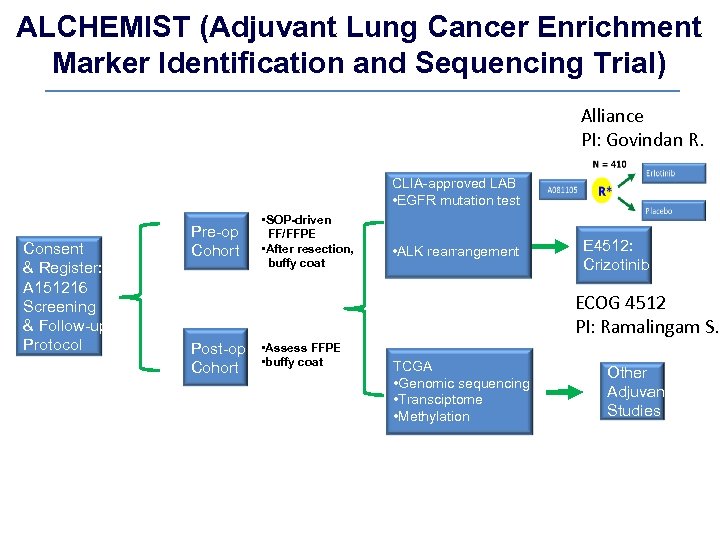 ALCHEMIST (Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trial) Alliance PI: Govindan R. CLIA-approved LAB • EGFR mutation test Consent & Register: A 151216 Screening & Follow-up Protocol Pre-op Cohort Post-op Cohort • SOP-driven FF/FFPE • After resection, buffy coat • ALK rearrangement E 4512: Crizotinib ECOG 4512 PI: Ramalingam S. • Assess FFPE • buffy coat TCGA • Genomic sequencing • Transciptome • Methylation Other Adjuvant Studies
ALCHEMIST (Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trial) Alliance PI: Govindan R. CLIA-approved LAB • EGFR mutation test Consent & Register: A 151216 Screening & Follow-up Protocol Pre-op Cohort Post-op Cohort • SOP-driven FF/FFPE • After resection, buffy coat • ALK rearrangement E 4512: Crizotinib ECOG 4512 PI: Ramalingam S. • Assess FFPE • buffy coat TCGA • Genomic sequencing • Transciptome • Methylation Other Adjuvant Studies
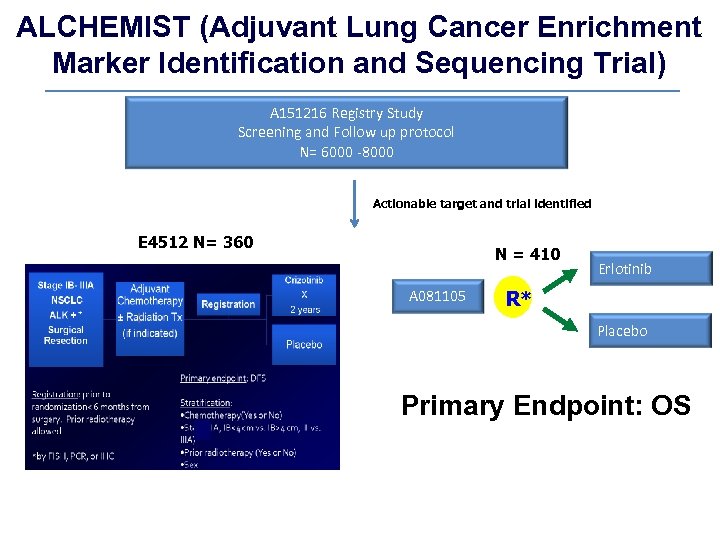 ALCHEMIST (Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trial) A 151216 Registry Study Screening and Follow up protocol N= 6000 -8000 Actionable target and trial identified E 4512 N= 360 N = 410 A 081105 Erlotinib R* Placebo Primary Endpoint: OS
ALCHEMIST (Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trial) A 151216 Registry Study Screening and Follow up protocol N= 6000 -8000 Actionable target and trial identified E 4512 N= 360 N = 410 A 081105 Erlotinib R* Placebo Primary Endpoint: OS
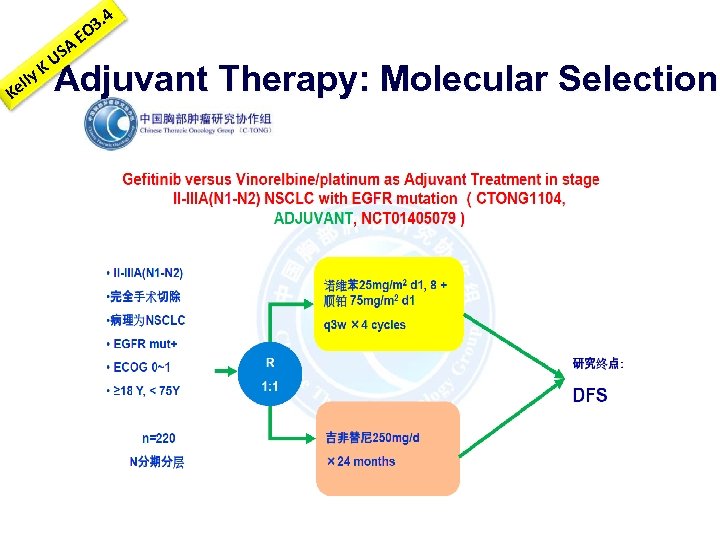 K y ell K A US E 3. 4 O Adjuvant Therapy: Molecular Selection
K y ell K A US E 3. 4 O Adjuvant Therapy: Molecular Selection
 Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
Strategies to select Drugs for use with surgery in Early Stage Lung Cancer • • • Gene expression profiles (microarrays) Repair/Metabolism genotype Immunotherapy Molecular driven mutation New targets Assess radiographic response in induction “window of opportunity”
 O 8. 0 0 s es B M ce n ra F B, e 2 WCLC 2013: what’s new
O 8. 0 0 s es B M ce n ra F B, e 2 WCLC 2013: what’s new
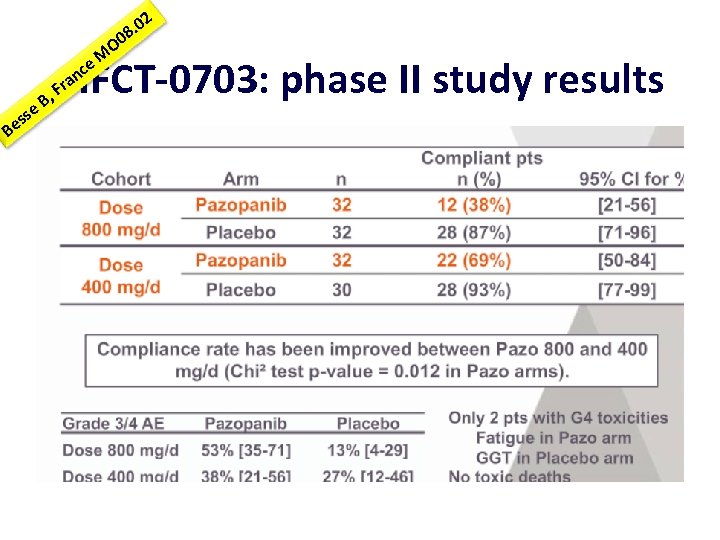 O 8. 0 0 s es B M ce n ra F B, e 2 IFCT-0703: phase II study results
O 8. 0 0 s es B M ce n ra F B, e 2 IFCT-0703: phase II study results
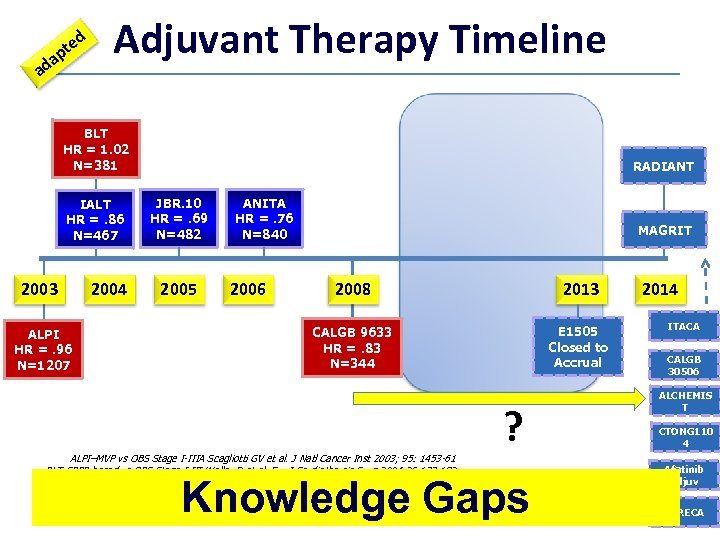 d e pt a ad Adjuvant Therapy Timeline BLT HR = 1. 02 N=381 IALT HR =. 86 N=467 2003 ALPI HR =. 96 N=1207 2004 RADIANT JBR. 10 HR =. 69 N=482~ 6 2005 ANITA HR =. 76 N=840 2006 MAGRIT 2008 2013 CALGB 9633 HR =. 83 N=344 E 1505 Closed to Accrual ALPI–MVP vs OBS Stage I-IIIA Scagliotti GV et al. J Natl Cancer Inst 2003; 95: 1453 -61 BLT-CPPP-based vs OBS Stage I-III Waller D et al. Eur J Cardiothorcic Surg 2004; 26: 173 -182 IALT–CDDP-based vs OBS Stage I-IIIA Arriagada R et al. N Engl J Med 2004; 350: 350 -61 JBR. 10–CDDP-VNR vs OBS Stage IB-II Winton T et al. N Engl J Med 2005; 352: 2589 -97 ANITA–CDDP-VNR vs OBS Stage IB-IIIA Douilland JY et al. Lancet Oncol 2006; 7: 719 -27 CALGB 9633–PAC-CARBO vs OBS Stage IB Strauss GM et al. J Clin Oncol 2008; 26: 5043 -51 ? Knowledge Gaps 2014 ITACA CALGB 30506 ALCHEMIS T CTONG 110 4 Afatinib Adjuv EURECA
d e pt a ad Adjuvant Therapy Timeline BLT HR = 1. 02 N=381 IALT HR =. 86 N=467 2003 ALPI HR =. 96 N=1207 2004 RADIANT JBR. 10 HR =. 69 N=482~ 6 2005 ANITA HR =. 76 N=840 2006 MAGRIT 2008 2013 CALGB 9633 HR =. 83 N=344 E 1505 Closed to Accrual ALPI–MVP vs OBS Stage I-IIIA Scagliotti GV et al. J Natl Cancer Inst 2003; 95: 1453 -61 BLT-CPPP-based vs OBS Stage I-III Waller D et al. Eur J Cardiothorcic Surg 2004; 26: 173 -182 IALT–CDDP-based vs OBS Stage I-IIIA Arriagada R et al. N Engl J Med 2004; 350: 350 -61 JBR. 10–CDDP-VNR vs OBS Stage IB-II Winton T et al. N Engl J Med 2005; 352: 2589 -97 ANITA–CDDP-VNR vs OBS Stage IB-IIIA Douilland JY et al. Lancet Oncol 2006; 7: 719 -27 CALGB 9633–PAC-CARBO vs OBS Stage IB Strauss GM et al. J Clin Oncol 2008; 26: 5043 -51 ? Knowledge Gaps 2014 ITACA CALGB 30506 ALCHEMIS T CTONG 110 4 Afatinib Adjuv EURECA
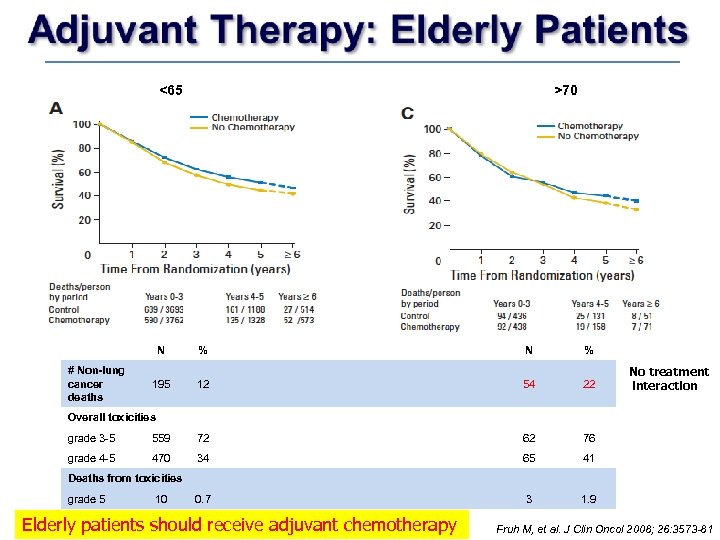 <65 >70 N # Non-lung cancer deaths % N % 195 12 54 22 No treatment interaction Overall toxicities grade 3 -5 559 72 62 76 grade 4 -5 470 34 65 41 0. 7 3 1. 9 Deaths from toxicities grade 5 10 Elderly patients should receive adjuvant chemotherapy Fruh M, et al. J Clin Oncol 2008; 26: 3573 -81
<65 >70 N # Non-lung cancer deaths % N % 195 12 54 22 No treatment interaction Overall toxicities grade 3 -5 559 72 62 76 grade 4 -5 470 34 65 41 0. 7 3 1. 9 Deaths from toxicities grade 5 10 Elderly patients should receive adjuvant chemotherapy Fruh M, et al. J Clin Oncol 2008; 26: 3573 -81
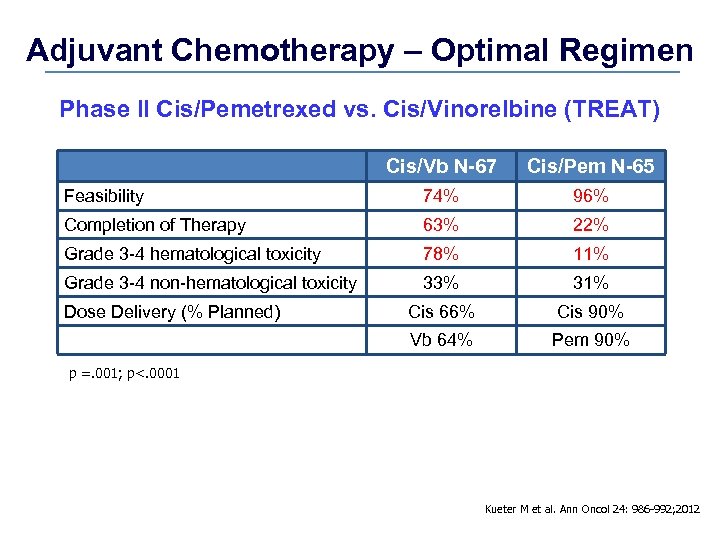 Adjuvant Chemotherapy – Optimal Regimen Phase II Cis/Pemetrexed vs. Cis/Vinorelbine (TREAT) Cis/Vb N-67 Cis/Pem N-65 Feasibility 74% 96% Completion of Therapy 63% 22% Grade 3 -4 hematological toxicity 78% 11% Grade 3 -4 non-hematological toxicity 33% 31% Cis 66% Cis 90% Vb 64% Pem 90% Dose Delivery (% Planned) p =. 001; p<. 0001 Kueter M et al. Ann Oncol 24: 986 -992; 2012
Adjuvant Chemotherapy – Optimal Regimen Phase II Cis/Pemetrexed vs. Cis/Vinorelbine (TREAT) Cis/Vb N-67 Cis/Pem N-65 Feasibility 74% 96% Completion of Therapy 63% 22% Grade 3 -4 hematological toxicity 78% 11% Grade 3 -4 non-hematological toxicity 33% 31% Cis 66% Cis 90% Vb 64% Pem 90% Dose Delivery (% Planned) p =. 001; p<. 0001 Kueter M et al. Ann Oncol 24: 986 -992; 2012
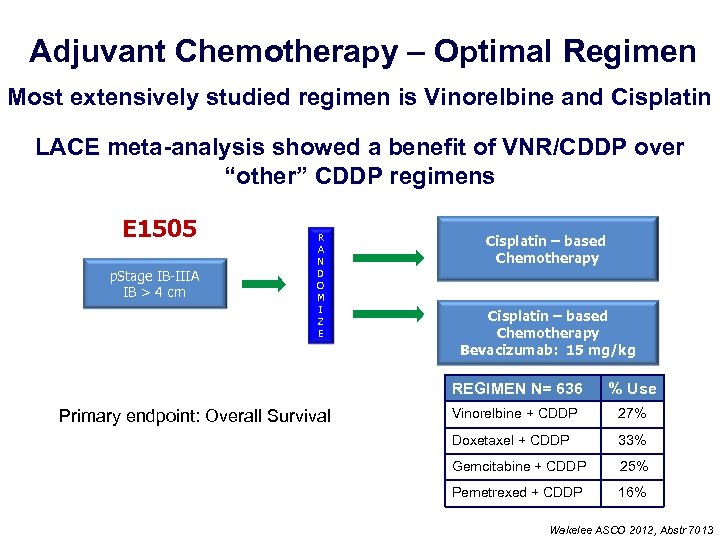 Adjuvant Chemotherapy – Optimal Regimen Most extensively studied regimen is Vinorelbine and Cisplatin LACE meta-analysis showed a benefit of VNR/CDDP over “other” CDDP regimens E 1505 p. Stage IB-IIIA IB > 4 cm R A N D O M I Z E Cisplatin – based Chemotherapy Bevacizumab: 15 mg/kg REGIMEN N= 636 Primary endpoint: Overall Survival % Use Vinorelbine + CDDP 27% Doxetaxel + CDDP 33% Gemcitabine + CDDP 25% Pemetrexed + CDDP 16% Wakelee ASCO 2012, Abstr 7013
Adjuvant Chemotherapy – Optimal Regimen Most extensively studied regimen is Vinorelbine and Cisplatin LACE meta-analysis showed a benefit of VNR/CDDP over “other” CDDP regimens E 1505 p. Stage IB-IIIA IB > 4 cm R A N D O M I Z E Cisplatin – based Chemotherapy Bevacizumab: 15 mg/kg REGIMEN N= 636 Primary endpoint: Overall Survival % Use Vinorelbine + CDDP 27% Doxetaxel + CDDP 33% Gemcitabine + CDDP 25% Pemetrexed + CDDP 16% Wakelee ASCO 2012, Abstr 7013
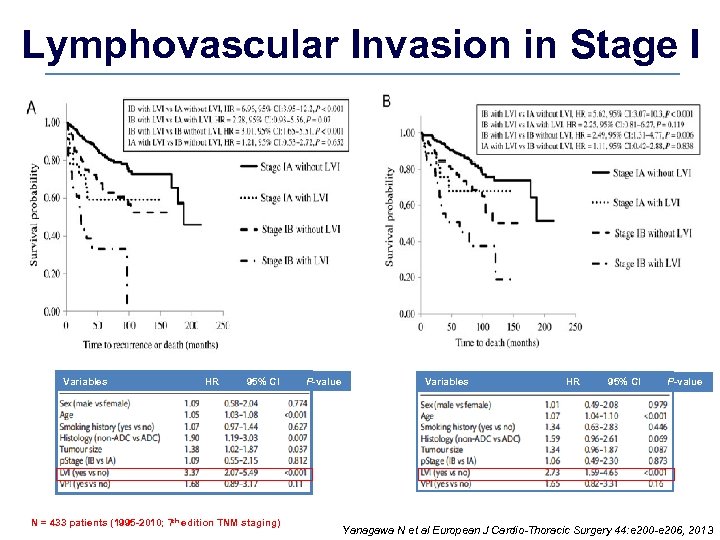 Lymphovascular Invasion in Stage I Variables HR 95% CI N = 433 patients (1995 -2010; 7 th edition TNM staging) P-value Variables HR 95% CI P-value Yanagawa N et al European J Cardio-Thoracic Surgery 44: e 200 -e 206, 2013
Lymphovascular Invasion in Stage I Variables HR 95% CI N = 433 patients (1995 -2010; 7 th edition TNM staging) P-value Variables HR 95% CI P-value Yanagawa N et al European J Cardio-Thoracic Surgery 44: e 200 -e 206, 2013
 Induction (Neoadjuvant) vs Adjuvant Factors Favoring Induction Chemotherapy • Attacks micrometastases at earliest time • Better drug delivery and tolerability • Ability to assess sensitivity of agents used in induction and planned for adjuvant • Platform for new agent testing • Surgical findings an outcome surrogate • Time to identify unsuspected metastases and comorbidities before local therapy • Randomized trials equivalent or better • Provide quick answers
Induction (Neoadjuvant) vs Adjuvant Factors Favoring Induction Chemotherapy • Attacks micrometastases at earliest time • Better drug delivery and tolerability • Ability to assess sensitivity of agents used in induction and planned for adjuvant • Platform for new agent testing • Surgical findings an outcome surrogate • Time to identify unsuspected metastases and comorbidities before local therapy • Randomized trials equivalent or better • Provide quick answers
 Major pathologic response (≤ 10% O 0 SA U viable tumor) following E, J t af Ch neoadjuvant chemotherapy as a surrogate for overall survival in patients with pathologically documented stage IIIA (N 2) lung adenocarcinomas 5. 0 2 Jamie E. Chaft 1, Matthew D. Hellmann 1, William D. Travis 2, Valerie Rusch 3, Mark G. Kris 1 Memorial Sloan-Kettering Departments of 1 Medicine, 2 Pathology, 3 Surgery
Major pathologic response (≤ 10% O 0 SA U viable tumor) following E, J t af Ch neoadjuvant chemotherapy as a surrogate for overall survival in patients with pathologically documented stage IIIA (N 2) lung adenocarcinomas 5. 0 2 Jamie E. Chaft 1, Matthew D. Hellmann 1, William D. Travis 2, Valerie Rusch 3, Mark G. Kris 1 Memorial Sloan-Kettering Departments of 1 Medicine, 2 Pathology, 3 Surgery
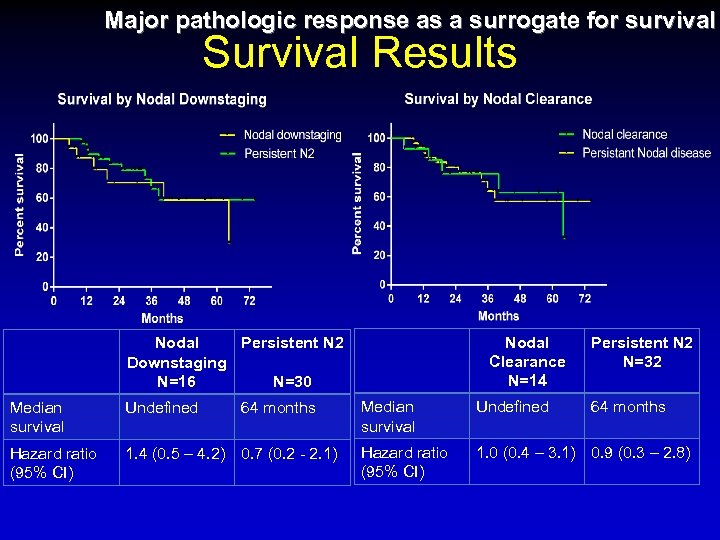 Major pathologic response as a surrogate for survival Survival Results Nodal Clearance N=14 Nodal Persistent N 2 Downstaging N=16 N=30 Persistent N 2 N=32 Median survival Undefined 64 months Hazard ratio (95% CI) 1. 4 (0. 5 – 4. 2) 0. 7 (0. 2 - 2. 1) Hazard ratio (95% CI) 1. 0 (0. 4 – 3. 1) 0. 9 (0. 3 – 2. 8)
Major pathologic response as a surrogate for survival Survival Results Nodal Clearance N=14 Nodal Persistent N 2 Downstaging N=16 N=30 Persistent N 2 N=32 Median survival Undefined 64 months Hazard ratio (95% CI) 1. 4 (0. 5 – 4. 2) 0. 7 (0. 2 - 2. 1) Hazard ratio (95% CI) 1. 0 (0. 4 – 3. 1) 0. 9 (0. 3 – 2. 8)
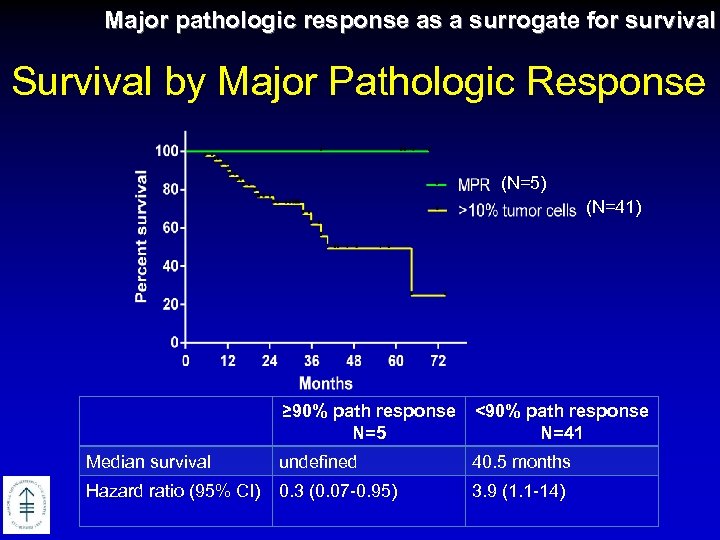 Major pathologic response as a surrogate for survival Survival by Major Pathologic Response (N=5) (N=41) ≥ 90% path response N=5 <90% path response N=41 Median survival undefined 40. 5 months Hazard ratio (95% CI) 0. 3 (0. 07 -0. 95) 3. 9 (1. 1 -14)
Major pathologic response as a surrogate for survival Survival by Major Pathologic Response (N=5) (N=41) ≥ 90% path response N=5 <90% path response N=41 Median survival undefined 40. 5 months Hazard ratio (95% CI) 0. 3 (0. 07 -0. 95) 3. 9 (1. 1 -14)
 Adjuvant Therapy: Molecular Selection
Adjuvant Therapy: Molecular Selection
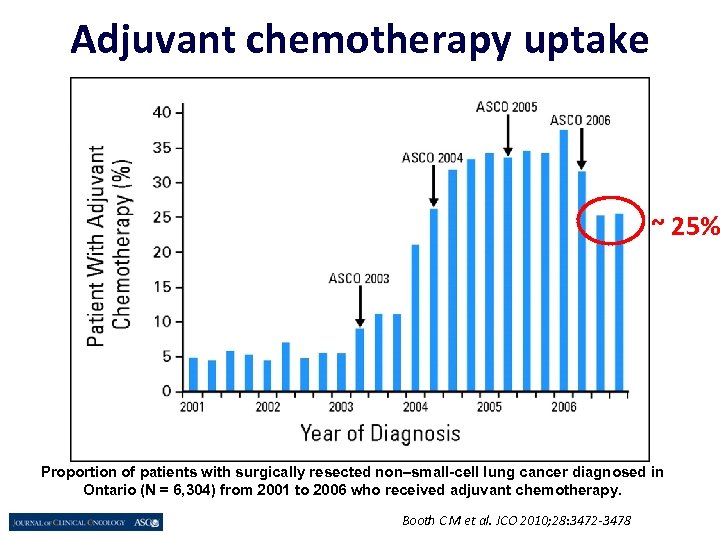 Adjuvant chemotherapy uptake ~ 25% Proportion of patients with surgically resected non–small-cell lung cancer diagnosed in Ontario (N = 6, 304) from 2001 to 2006 who received adjuvant chemotherapy. Booth C M et al. JCO 2010; 28: 3472 -3478
Adjuvant chemotherapy uptake ~ 25% Proportion of patients with surgically resected non–small-cell lung cancer diagnosed in Ontario (N = 6, 304) from 2001 to 2006 who received adjuvant chemotherapy. Booth C M et al. JCO 2010; 28: 3472 -3478
 S y. M t lio g a Sc t V i. G l Ita 3. 3 1 Conclusion • Patient selection & drug selection based on gene expression arrays, gene mutation and amplification and/or proteomics will quite likely improve the efficacy of adjuvant chemotherapy • Phase II studies are marginally appropriate • In this setting alternative efficacy outcomes instead of OS are needed. • Marker-based or marker by treatment interaction are eagerly encouraged. • In the context of rare oncogene-addicted tumors a close multidisciplinary, international collaboration is needed.
S y. M t lio g a Sc t V i. G l Ita 3. 3 1 Conclusion • Patient selection & drug selection based on gene expression arrays, gene mutation and amplification and/or proteomics will quite likely improve the efficacy of adjuvant chemotherapy • Phase II studies are marginally appropriate • In this setting alternative efficacy outcomes instead of OS are needed. • Marker-based or marker by treatment interaction are eagerly encouraged. • In the context of rare oncogene-addicted tumors a close multidisciplinary, international collaboration is needed.


