88fc17b97386a46e806e9e8aa709a231.ppt
- Количество слайдов: 71
 Novel Targets and Therapies for GI Malignancies: What does the future hold? David H. Ilson, M. D. , Ph. D. GI Oncology Service Memorial Sloan-Kettering Cancer Center New York, NY
Novel Targets and Therapies for GI Malignancies: What does the future hold? David H. Ilson, M. D. , Ph. D. GI Oncology Service Memorial Sloan-Kettering Cancer Center New York, NY
 GI Cancers: US Incidence in 2013 l 292, 200 new cases and 144, 570 deaths (49%) l Case Fatality Rate: – Colorectal: 48% – Esophagogastric: 66% – Pancreatic: 85% – HCC: 70% l Male > Female l Ongoing rise in Esophageal and GEJ Adenocarcinoma, HCC Siegel et al, CA 63: 11 -30; 2013
GI Cancers: US Incidence in 2013 l 292, 200 new cases and 144, 570 deaths (49%) l Case Fatality Rate: – Colorectal: 48% – Esophagogastric: 66% – Pancreatic: 85% – HCC: 70% l Male > Female l Ongoing rise in Esophageal and GEJ Adenocarcinoma, HCC Siegel et al, CA 63: 11 -30; 2013
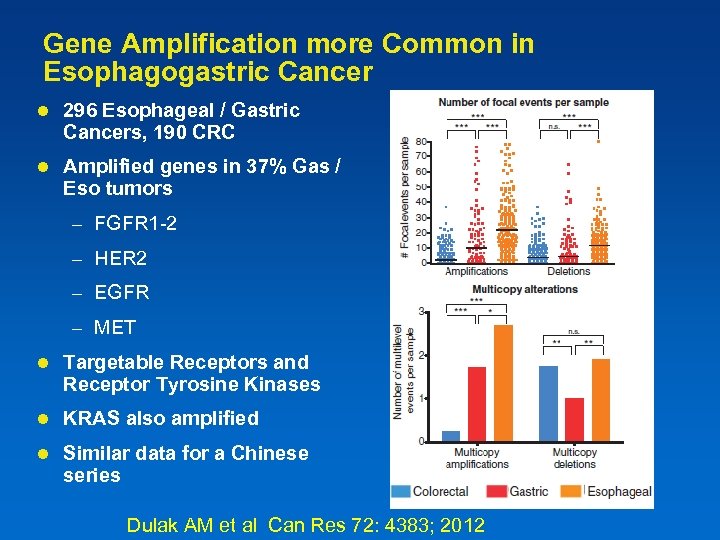 Gene Amplification more Common in Esophagogastric Cancer l 296 Esophageal / Gastric Cancers, 190 CRC l Amplified genes in 37% Gas / Eso tumors – FGFR 1 -2 – HER 2 – EGFR – MET l Targetable Receptors and Receptor Tyrosine Kinases l KRAS also amplified l Similar data for a Chinese series Dulak AM et al Can Res 72: 4383; 2012
Gene Amplification more Common in Esophagogastric Cancer l 296 Esophageal / Gastric Cancers, 190 CRC l Amplified genes in 37% Gas / Eso tumors – FGFR 1 -2 – HER 2 – EGFR – MET l Targetable Receptors and Receptor Tyrosine Kinases l KRAS also amplified l Similar data for a Chinese series Dulak AM et al Can Res 72: 4383; 2012
 Molecular Targets: Esophageal and Gastric Cancer • KRAS mutation: <5% • BRAF mutation: <5% • EGFr mutation: <5% • HER 2 over expression / amplification: 10% to 25% – Trastuzumab + chemo improves OS in HER 2+ disease • CMET amplification: 10% – IHC over expression 40% Dulak AM, et al. Cancer Res. 2012; 72(17): 4383 -4393. Dulak AM, et al. Nat Genet. 2013; 45(5): 478 -486. Lordick F, et al. Lancet Oncol. 2013; 14(6): 490 -499. Bang YJ, et al. Lancet. 2010; 376(9742): 687 -697.
Molecular Targets: Esophageal and Gastric Cancer • KRAS mutation: <5% • BRAF mutation: <5% • EGFr mutation: <5% • HER 2 over expression / amplification: 10% to 25% – Trastuzumab + chemo improves OS in HER 2+ disease • CMET amplification: 10% – IHC over expression 40% Dulak AM, et al. Cancer Res. 2012; 72(17): 4383 -4393. Dulak AM, et al. Nat Genet. 2013; 45(5): 478 -486. Lordick F, et al. Lancet Oncol. 2013; 14(6): 490 -499. Bang YJ, et al. Lancet. 2010; 376(9742): 687 -697.
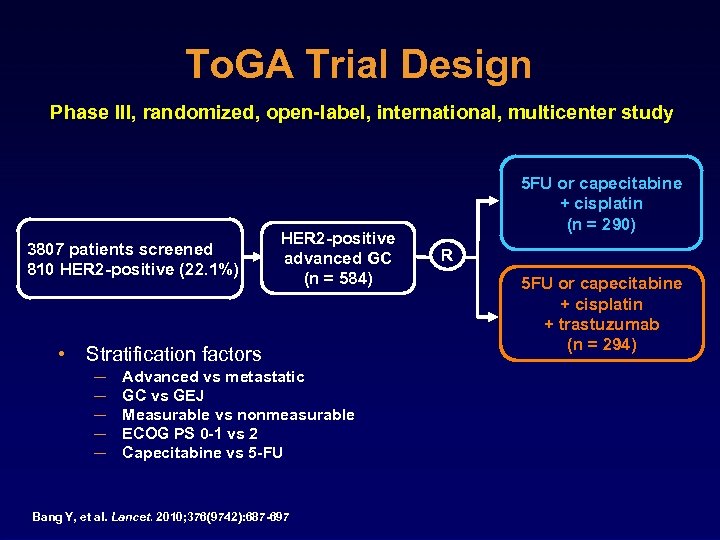 To. GA Trial Design Phase III, randomized, open-label, international, multicenter study 3807 patients screened 810 HER 2 -positive (22. 1%) • HER 2 -positive advanced GC (n = 584) Stratification factors ─ ─ ─ Advanced vs metastatic GC vs GEJ Measurable vs nonmeasurable ECOG PS 0 -1 vs 2 Capecitabine vs 5 -FU Bang Y, et al. Lancet. 2010; 376(9742): 687 -697 5 FU or capecitabine + cisplatin (n = 290) R 5 FU or capecitabine + cisplatin + trastuzumab (n = 294)
To. GA Trial Design Phase III, randomized, open-label, international, multicenter study 3807 patients screened 810 HER 2 -positive (22. 1%) • HER 2 -positive advanced GC (n = 584) Stratification factors ─ ─ ─ Advanced vs metastatic GC vs GEJ Measurable vs nonmeasurable ECOG PS 0 -1 vs 2 Capecitabine vs 5 -FU Bang Y, et al. Lancet. 2010; 376(9742): 687 -697 5 FU or capecitabine + cisplatin (n = 290) R 5 FU or capecitabine + cisplatin + trastuzumab (n = 294)
 To. GA: Efficacy Outcome Chemotherapy + Trastuzumab Alone (n = 294) (n = 290) HR (95% CI) P Value Primary endpoint Median OS, months 13. 8 11. 1 0. 74 (0. 60 -0. 91) . 0046 Median PFS, months 6. 7 5. 5 0. 71 (0. 59 -0. 85) . 0002 ORR, % 47. 3 34. 6 - . 0017 • • CR 5. 4 2. 4 - . 0599 PR 41. 8 32. 1 - . 0145 Secondary endpoints • Preplanned subgroup analysis indicated improved OS benefit with increasing HER 2 expression by IHC • Exploratory analysis of IHC 2+/FISH+ and IHC 3+ cohort demonstrated a 4 -month increase in OS with trastuzumab − HR: 0. 65 (95% CI: 0. 51 -0. 83) ORR, overall response rate Bang Y, et al. Lancet. 2010; 376(9742): 687 -697.
To. GA: Efficacy Outcome Chemotherapy + Trastuzumab Alone (n = 294) (n = 290) HR (95% CI) P Value Primary endpoint Median OS, months 13. 8 11. 1 0. 74 (0. 60 -0. 91) . 0046 Median PFS, months 6. 7 5. 5 0. 71 (0. 59 -0. 85) . 0002 ORR, % 47. 3 34. 6 - . 0017 • • CR 5. 4 2. 4 - . 0599 PR 41. 8 32. 1 - . 0145 Secondary endpoints • Preplanned subgroup analysis indicated improved OS benefit with increasing HER 2 expression by IHC • Exploratory analysis of IHC 2+/FISH+ and IHC 3+ cohort demonstrated a 4 -month increase in OS with trastuzumab − HR: 0. 65 (95% CI: 0. 51 -0. 83) ORR, overall response rate Bang Y, et al. Lancet. 2010; 376(9742): 687 -697.
 Targeted Agents Phase III: HER 2: Met Disease l LOGIC: Cape-Ox + / - Lapatinib (HER 2+) – First line – Negative trial for OS – Benefit in Asian pts l TYTAN: Paclitaxel + / - Lapatinib (HER 2+) – Second Line: Negative Trial – PFS and Survival Benefit in subset of patients IHC 3+ for lapatinib Hecht JR, et al. J Clin Oncol. 2013; 31(Suppl): Abstract LBA 4001 Bang et al GI Cancers Symposium 2013 Abstract 11
Targeted Agents Phase III: HER 2: Met Disease l LOGIC: Cape-Ox + / - Lapatinib (HER 2+) – First line – Negative trial for OS – Benefit in Asian pts l TYTAN: Paclitaxel + / - Lapatinib (HER 2+) – Second Line: Negative Trial – PFS and Survival Benefit in subset of patients IHC 3+ for lapatinib Hecht JR, et al. J Clin Oncol. 2013; 31(Suppl): Abstract LBA 4001 Bang et al GI Cancers Symposium 2013 Abstract 11
 RTOG 1010: Phase III Study of Neoadjuvant Trastuzumab and Chemoradiation for Esophageal Adenocarcinoma (Siewert I, II) CHEMORADIATION SURGERY HER-2 (+) (FISH) TRASTUZUMAB + CHEMORADIATION HER-2 (-) (FISH) SURGERY + TRASTUZUMAB (1 YR) ALTERNATIVE STUDIES §Chemoradiation: Carboplatin, Paclitaxel + RT 5040 c. Gy Surge §Maintenance trastuzumab post op §OS Primary Endpoint
RTOG 1010: Phase III Study of Neoadjuvant Trastuzumab and Chemoradiation for Esophageal Adenocarcinoma (Siewert I, II) CHEMORADIATION SURGERY HER-2 (+) (FISH) TRASTUZUMAB + CHEMORADIATION HER-2 (-) (FISH) SURGERY + TRASTUZUMAB (1 YR) ALTERNATIVE STUDIES §Chemoradiation: Carboplatin, Paclitaxel + RT 5040 c. Gy Surge §Maintenance trastuzumab post op §OS Primary Endpoint
 HER 2 -Directed Therapy Trials • Ongoing HER 2 Trials – First-line - JACOB: Cape-Cis-Trastuzumab + / - Pertuzumab, 780 patients - HELOISE: Cape-Cis + 2 dose levels of Trastuzumab, 400 patients – Second-line: - GATSBY: Paclitaxel vs TDM-1
HER 2 -Directed Therapy Trials • Ongoing HER 2 Trials – First-line - JACOB: Cape-Cis-Trastuzumab + / - Pertuzumab, 780 patients - HELOISE: Cape-Cis + 2 dose levels of Trastuzumab, 400 patients – Second-line: - GATSBY: Paclitaxel vs TDM-1
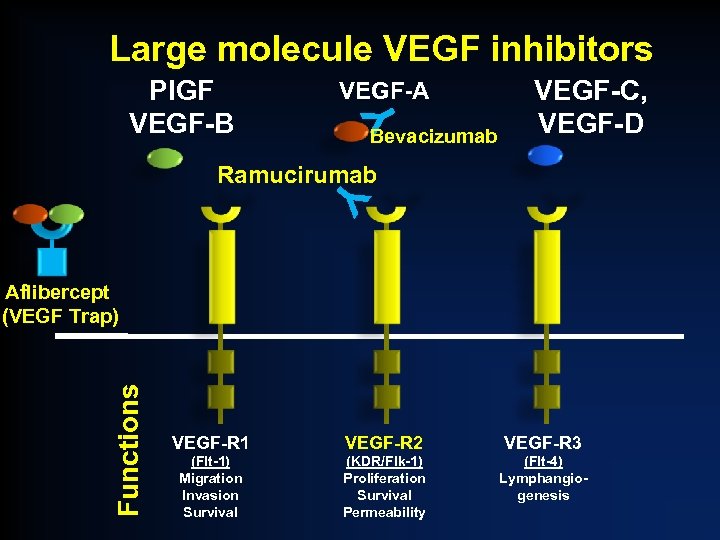 Large molecule VEGF inhibitors VEGF-A Y Pl. GF VEGF-B Bevacizumab VEGF-C, VEGF-D Y Ramucirumab Functions Aflibercept (VEGF Trap) VEGF-R 1 VEGF-R 2 VEGF-R 3 (Flt-1) Migration Invasion Survival (KDR/Flk-1) Proliferation Survival Permeability (Flt-4) Lymphangiogenesis
Large molecule VEGF inhibitors VEGF-A Y Pl. GF VEGF-B Bevacizumab VEGF-C, VEGF-D Y Ramucirumab Functions Aflibercept (VEGF Trap) VEGF-R 1 VEGF-R 2 VEGF-R 3 (Flt-1) Migration Invasion Survival (KDR/Flk-1) Proliferation Survival Permeability (Flt-4) Lymphangiogenesis
 Targeted Agents Phase III: Negative Trials for VEGF, m. TOR, and EGFr l AVAGAST: Cape-Cisplatin + / - Bevacizumab – Negative trial for OS l m. TOR GRANITE: BSC vs Everolimus – Negative trial for OS l REAL 3: ECX + / - Panitumumab (U. K. ) – Negative: Panitumumab had inferior outcomes l EXPAND: Cape-Cis + / Cetuximab (E. U. ) – Negative: Cetuximab trended inferior l COG: BSC vs Gefitinib (U. K. ): Negative Ohtsu A, et al. J Clin Oncol. 2011; 29(30): 3968 -3976 Ohtsu A, et al. J Clin Oncol. 2013; 31(31): 3935 -3943 Waddell T Lance Oncol 14: 481; 2013 Lordick F et al Lancet 14: 490; 2013 Sutton JCO 30: 2012 (suppl 34 abstr
Targeted Agents Phase III: Negative Trials for VEGF, m. TOR, and EGFr l AVAGAST: Cape-Cisplatin + / - Bevacizumab – Negative trial for OS l m. TOR GRANITE: BSC vs Everolimus – Negative trial for OS l REAL 3: ECX + / - Panitumumab (U. K. ) – Negative: Panitumumab had inferior outcomes l EXPAND: Cape-Cis + / Cetuximab (E. U. ) – Negative: Cetuximab trended inferior l COG: BSC vs Gefitinib (U. K. ): Negative Ohtsu A, et al. J Clin Oncol. 2011; 29(30): 3968 -3976 Ohtsu A, et al. J Clin Oncol. 2013; 31(31): 3935 -3943 Waddell T Lance Oncol 14: 481; 2013 Lordick F et al Lancet 14: 490; 2013 Sutton JCO 30: 2012 (suppl 34 abstr
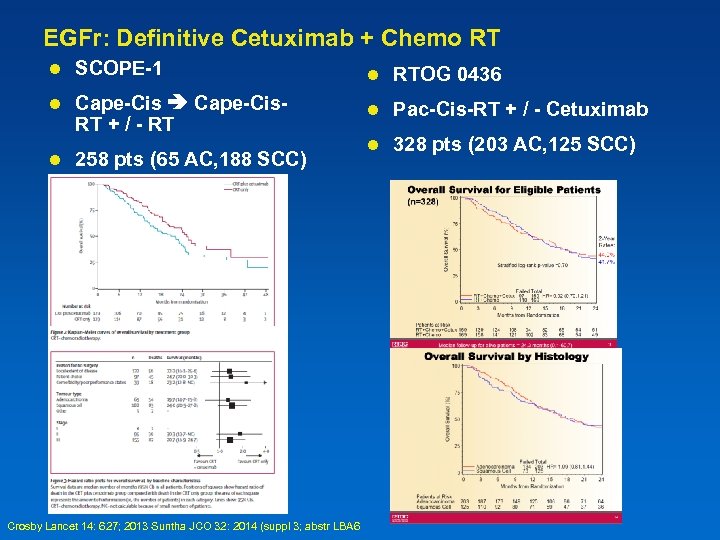 EGFr: Definitive Cetuximab + Chemo RT l SCOPE-1 l RTOG 0436 l Cape-Cis. RT + / - RT l Pac-Cis-RT + / - Cetuximab l 328 pts (203 AC, 125 SCC) l 258 pts (65 AC, 188 SCC) Crosby Lancet 14: 627; 2013 Suntha JCO 32: 2014 (suppl 3; abstr LBA 6
EGFr: Definitive Cetuximab + Chemo RT l SCOPE-1 l RTOG 0436 l Cape-Cis. RT + / - RT l Pac-Cis-RT + / - Cetuximab l 328 pts (203 AC, 125 SCC) l 258 pts (65 AC, 188 SCC) Crosby Lancet 14: 627; 2013 Suntha JCO 32: 2014 (suppl 3; abstr LBA 6
 VEGF Revisited? • Apatinib – Small-molecule multitargeted TKI with activity against VEGFR – China – 144 patients, placebo vs 850 mg/d or 425 mg BID – OS 2. 5 months 4. 0 months, 4. 5 months – RR 10% • Phase III Trial Planned • Ramucirumab: Humanized mo. Ab Targeting VEGr 2 receptor TKI, tyrosine kinase inhibitor; VEGFR, VEGF receptor Li J, et al. J Clin Oncol. 2013; 31(26): 3219 -3225. Fuchs CS, et al. Lancet. 2014; 383(9911): 31 -39.
VEGF Revisited? • Apatinib – Small-molecule multitargeted TKI with activity against VEGFR – China – 144 patients, placebo vs 850 mg/d or 425 mg BID – OS 2. 5 months 4. 0 months, 4. 5 months – RR 10% • Phase III Trial Planned • Ramucirumab: Humanized mo. Ab Targeting VEGr 2 receptor TKI, tyrosine kinase inhibitor; VEGFR, VEGF receptor Li J, et al. J Clin Oncol. 2013; 31(26): 3219 -3225. Fuchs CS, et al. Lancet. 2014; 383(9911): 31 -39.
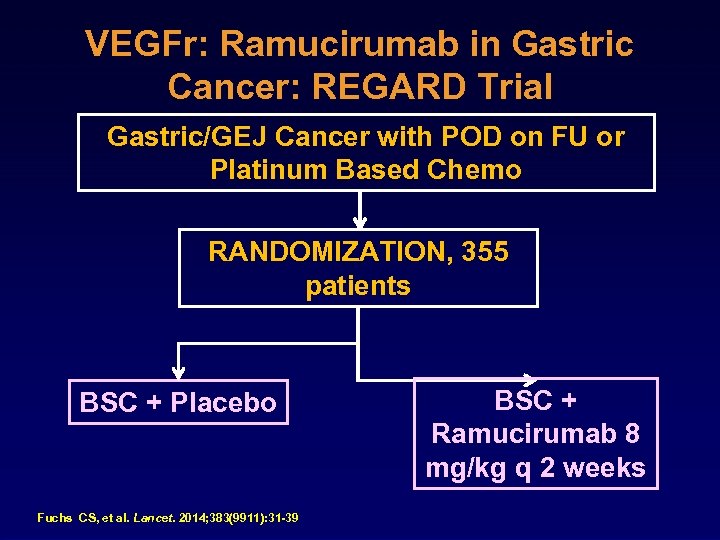 VEGFr: Ramucirumab in Gastric Cancer: REGARD Trial Gastric/GEJ Cancer with POD on FU or Platinum Based Chemo RANDOMIZATION, 355 patients BSC + Placebo Fuchs CS, et al. Lancet. 2014; 383(9911): 31 -39 BSC + Ramucirumab 8 mg/kg q 2 weeks
VEGFr: Ramucirumab in Gastric Cancer: REGARD Trial Gastric/GEJ Cancer with POD on FU or Platinum Based Chemo RANDOMIZATION, 355 patients BSC + Placebo Fuchs CS, et al. Lancet. 2014; 383(9911): 31 -39 BSC + Ramucirumab 8 mg/kg q 2 weeks
 VEGF Revisited? Ramucirumab: REGARD Trial • PFS improved 2. 1 months 3. 8 months (HR 0. 483, P<. 0001) • OS improved 3. 8 months 5. 2 months (HR 0. 776, P =. 047) • Disease control improved from 23% to 49% (P<. 0001) • Essentially no toxicity (rare grade ≥ 3 hypertension 8%) Fuchs CS, et al. Lancet. 2014; 383(9911): 31 -39.
VEGF Revisited? Ramucirumab: REGARD Trial • PFS improved 2. 1 months 3. 8 months (HR 0. 483, P<. 0001) • OS improved 3. 8 months 5. 2 months (HR 0. 776, P =. 047) • Disease control improved from 23% to 49% (P<. 0001) • Essentially no toxicity (rare grade ≥ 3 hypertension 8%) Fuchs CS, et al. Lancet. 2014; 383(9911): 31 -39.
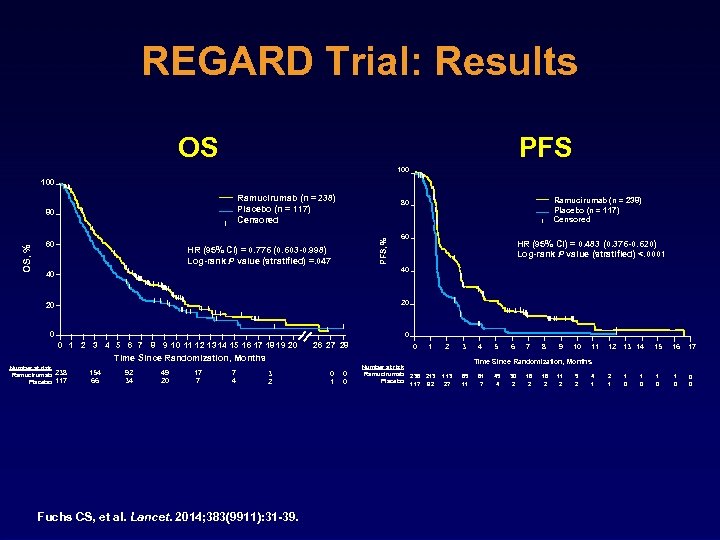 REGARD Trial: Results OS PFS 100 Ramucirumab (n = 238) Placebo (n = 117) Censored 60 Ramucirumab (n = 238) Placebo (n = 117) Censored 80 PFS, % OS, % 80 HR (95% CI) = 0. 776 (0. 603 -0. 998) Log-rank P value (stratified) =. 047 60 HR (95% CI) = 0. 483 (0. 376 -0. 620) Log-rank P value (stratified) <. 0001 40 40 20 20 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 26 27 28 0 1 2 3 113 27 65 11 Time Since Randomization, Months Number at risk Ramucirumab 238 Placebo 117 154 66 92 34 49 20 17 7 7 4 3 2 Fuchs CS, et al. Lancet. 2014; 383(9911): 31 -39. 0 1 0 0 Number at risk Ramucirumab Placebo 4 5 6 7 8 9 10 11 12 13 14 15 16 17 1 0 0 0 Time Since Randomization, Months 238 213 117 92 61 7 45 4 30 2 18 2 11 2 5 2 4 1 2 1 1 0
REGARD Trial: Results OS PFS 100 Ramucirumab (n = 238) Placebo (n = 117) Censored 60 Ramucirumab (n = 238) Placebo (n = 117) Censored 80 PFS, % OS, % 80 HR (95% CI) = 0. 776 (0. 603 -0. 998) Log-rank P value (stratified) =. 047 60 HR (95% CI) = 0. 483 (0. 376 -0. 620) Log-rank P value (stratified) <. 0001 40 40 20 20 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 26 27 28 0 1 2 3 113 27 65 11 Time Since Randomization, Months Number at risk Ramucirumab 238 Placebo 117 154 66 92 34 49 20 17 7 7 4 3 2 Fuchs CS, et al. Lancet. 2014; 383(9911): 31 -39. 0 1 0 0 Number at risk Ramucirumab Placebo 4 5 6 7 8 9 10 11 12 13 14 15 16 17 1 0 0 0 Time Since Randomization, Months 238 213 117 92 61 7 45 4 30 2 18 2 11 2 5 2 4 1 2 1 1 0
 VEGFr: RAINBOW: Study Design 1: 1 R A N D O M I Z E S C R E E N • Ramucirumab 8 mg/kg day 1&15 + Paclitaxel 80 mg/m 2 day 1, 8 &15 of a 28 -day cycle N = 330 Placebo day 1&15 + Paclitaxel 80 mg/m 2 day 1, 8 &15 N = 335 Important inclusion criteria: - Metastatic or loc. adv. unresectable gastric or GEJ* adenocarcinoma - Progression after 1 st line platinum/fluoropyrimidine based chemotherapy • Stratification factors: - Geographic region, - Measurable vs non-measurable disease, - Time to progression on 1 st line therapy (< 6 mos vs. ≥ 6 mos) Wilke GI Symposium 2014 LBA 7 Treat until disease progression or intolerable toxicity Survival and safety follow -up
VEGFr: RAINBOW: Study Design 1: 1 R A N D O M I Z E S C R E E N • Ramucirumab 8 mg/kg day 1&15 + Paclitaxel 80 mg/m 2 day 1, 8 &15 of a 28 -day cycle N = 330 Placebo day 1&15 + Paclitaxel 80 mg/m 2 day 1, 8 &15 N = 335 Important inclusion criteria: - Metastatic or loc. adv. unresectable gastric or GEJ* adenocarcinoma - Progression after 1 st line platinum/fluoropyrimidine based chemotherapy • Stratification factors: - Geographic region, - Measurable vs non-measurable disease, - Time to progression on 1 st line therapy (< 6 mos vs. ≥ 6 mos) Wilke GI Symposium 2014 LBA 7 Treat until disease progression or intolerable toxicity Survival and safety follow -up
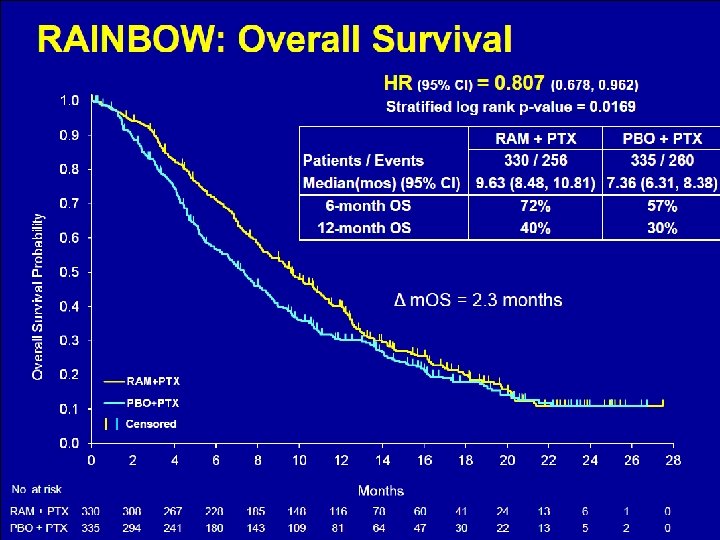
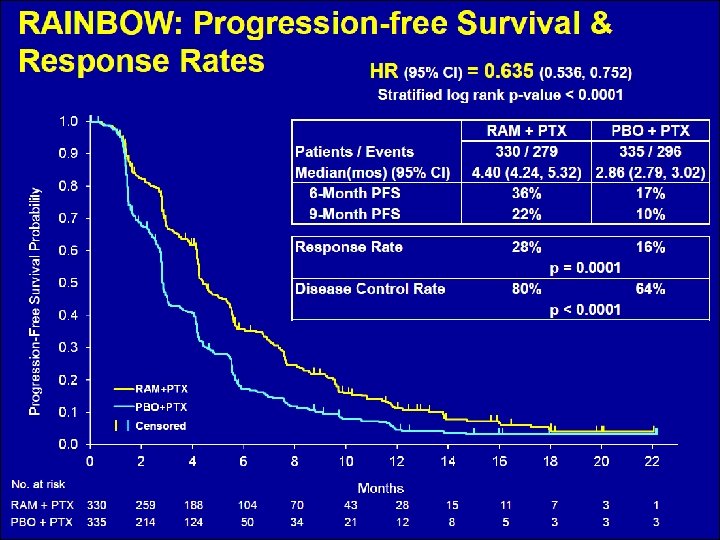
 RAINBOW: Ramucirumab + Paclitaxel for Metastatic Gastric Cancer • 665 pts with POD on fluorinated pyrimidine + platinum • Weekly paclitaxel 80 mg/m 2 + /- Ram 8 mg/kg • PFS improved 2. 9 months 4. 4 months (HR 0. 635, P<. 0001) • OS improved 7. 4 months 9. 6 months (HR 0. 807, P =. 0169) • RR improved from 16% to 28% (p = 0. 0010 (P<. 0001) • Increased toxicity neutropenia and hypertension Wilke GI Symposium 2014 LBA 7
RAINBOW: Ramucirumab + Paclitaxel for Metastatic Gastric Cancer • 665 pts with POD on fluorinated pyrimidine + platinum • Weekly paclitaxel 80 mg/m 2 + /- Ram 8 mg/kg • PFS improved 2. 9 months 4. 4 months (HR 0. 635, P<. 0001) • OS improved 7. 4 months 9. 6 months (HR 0. 807, P =. 0169) • RR improved from 16% to 28% (p = 0. 0010 (P<. 0001) • Increased toxicity neutropenia and hypertension Wilke GI Symposium 2014 LBA 7
 Ramucirumab • First-line: FOLFOX + / - Ramucirumab – Randomized phase II • Other VEGF agents – FOLFOX + / - Pazopanib (TKI)
Ramucirumab • First-line: FOLFOX + / - Ramucirumab – Randomized phase II • Other VEGF agents – FOLFOX + / - Pazopanib (TKI)
 VEGF Adjuvant Trials Gastric Cancer • MAGIC 2 Trial: EOX + / - Bevacizumab – Amended for HER 2 + patients – Randomized to + / - Lapatinib (HER 1 -2 TKI)
VEGF Adjuvant Trials Gastric Cancer • MAGIC 2 Trial: EOX + / - Bevacizumab – Amended for HER 2 + patients – Randomized to + / - Lapatinib (HER 1 -2 TKI)
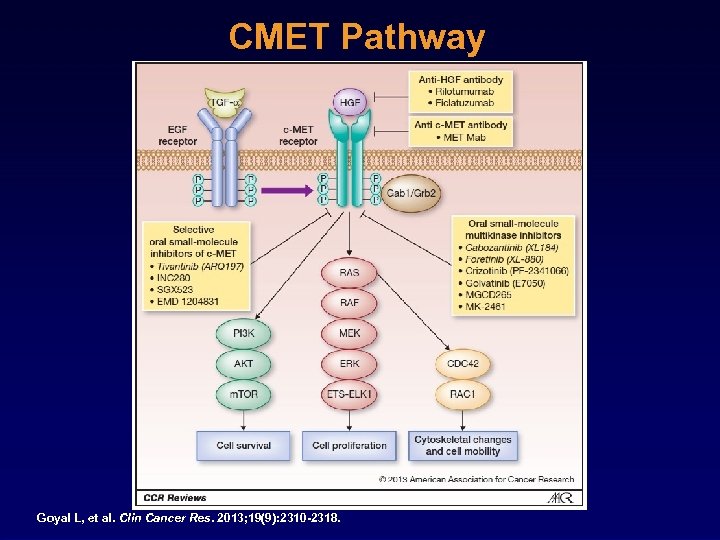 CMET Pathway Goyal L, et al. Clin Cancer Res. 2013; 19(9): 2310 -2318.
CMET Pathway Goyal L, et al. Clin Cancer Res. 2013; 19(9): 2310 -2318.
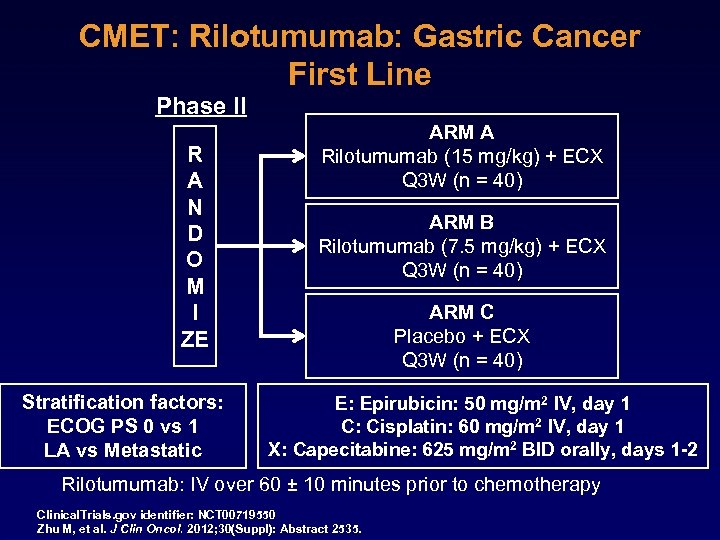 CMET: Rilotumumab: Gastric Cancer First Line Phase II R A N D O M I ZE Stratification factors: ECOG PS 0 vs 1 LA vs Metastatic ARM A Rilotumumab (15 mg/kg) + ECX Q 3 W (n = 40) ARM B Rilotumumab (7. 5 mg/kg) + ECX Q 3 W (n = 40) ARM C Placebo + ECX Q 3 W (n = 40) E: Epirubicin: 50 mg/m 2 IV, day 1 C: Cisplatin: 60 mg/m 2 IV, day 1 X: Capecitabine: 625 mg/m 2 BID orally, days 1 -2 Rilotumumab: IV over 60 ± 10 minutes prior to chemotherapy Clinical. Trials. gov identifier: NCT 00719550 Zhu M, et al. J Clin Oncol. 2012; 30(Suppl): Abstract 2535.
CMET: Rilotumumab: Gastric Cancer First Line Phase II R A N D O M I ZE Stratification factors: ECOG PS 0 vs 1 LA vs Metastatic ARM A Rilotumumab (15 mg/kg) + ECX Q 3 W (n = 40) ARM B Rilotumumab (7. 5 mg/kg) + ECX Q 3 W (n = 40) ARM C Placebo + ECX Q 3 W (n = 40) E: Epirubicin: 50 mg/m 2 IV, day 1 C: Cisplatin: 60 mg/m 2 IV, day 1 X: Capecitabine: 625 mg/m 2 BID orally, days 1 -2 Rilotumumab: IV over 60 ± 10 minutes prior to chemotherapy Clinical. Trials. gov identifier: NCT 00719550 Zhu M, et al. J Clin Oncol. 2012; 30(Suppl): Abstract 2535.
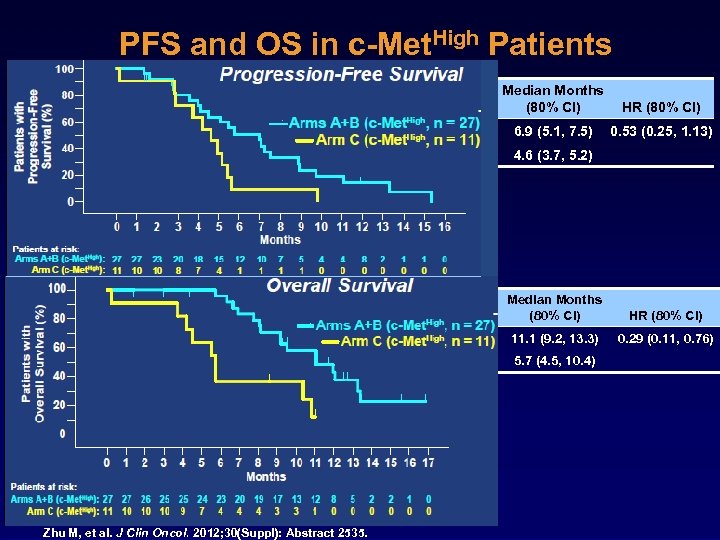 PFS and OS in c-Met. High Patients Median Months (80% CI) HR (80% CI) 6. 9 (5. 1, 7. 5) 0. 53 (0. 25, 1. 13) 4. 6 (3. 7, 5. 2) Median Months (80% CI) HR (80% CI) 11. 1 (9. 2, 13. 3) 0. 29 (0. 11, 0. 76) 5. 7 (4. 5, 10. 4) Zhu M, et al. J Clin Oncol. 2012; 30(Suppl): Abstract 2535.
PFS and OS in c-Met. High Patients Median Months (80% CI) HR (80% CI) 6. 9 (5. 1, 7. 5) 0. 53 (0. 25, 1. 13) 4. 6 (3. 7, 5. 2) Median Months (80% CI) HR (80% CI) 11. 1 (9. 2, 13. 3) 0. 29 (0. 11, 0. 76) 5. 7 (4. 5, 10. 4) Zhu M, et al. J Clin Oncol. 2012; 30(Suppl): Abstract 2535.
 Ongoing Trials: Met Inhibitors l Targeting CMET, + IHC l RILOMET-1 – ECX + / - Rilotumumab (targeting ligand HGF) l Met. Gastric – FOLFOX + / - Met. Mab (targeting receptor) l Tyrosine Kinase Inhibitors – Promising phase I activity in CMET amplified
Ongoing Trials: Met Inhibitors l Targeting CMET, + IHC l RILOMET-1 – ECX + / - Rilotumumab (targeting ligand HGF) l Met. Gastric – FOLFOX + / - Met. Mab (targeting receptor) l Tyrosine Kinase Inhibitors – Promising phase I activity in CMET amplified
 Fibroblast growth factor receptor l Ligand activated trans membrane growth factor receptor l Signals via RAS Map kinase and PI 3 K-AKT but also via Hedgehog and Notch pathways, and WNT l Phase II Trials – Dovitinib (TKI) in FGFR gene amplified gastric cancer – Dovitinib + Docetaxel in gastric cancer
Fibroblast growth factor receptor l Ligand activated trans membrane growth factor receptor l Signals via RAS Map kinase and PI 3 K-AKT but also via Hedgehog and Notch pathways, and WNT l Phase II Trials – Dovitinib (TKI) in FGFR gene amplified gastric cancer – Dovitinib + Docetaxel in gastric cancer
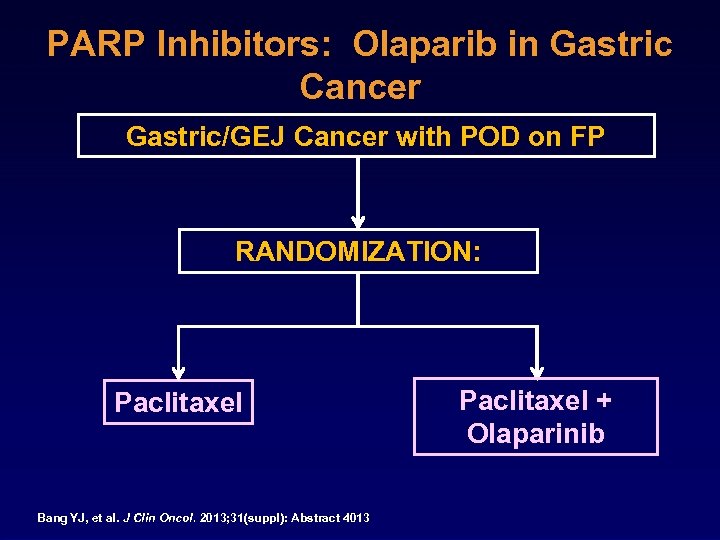 PARP Inhibitors: Olaparib in Gastric Cancer Gastric/GEJ Cancer with POD on FP RANDOMIZATION: Paclitaxel Bang YJ, et al. J Clin Oncol. 2013; 31(suppl): Abstract 4013 Paclitaxel + Olaparinib
PARP Inhibitors: Olaparib in Gastric Cancer Gastric/GEJ Cancer with POD on FP RANDOMIZATION: Paclitaxel Bang YJ, et al. J Clin Oncol. 2013; 31(suppl): Abstract 4013 Paclitaxel + Olaparinib
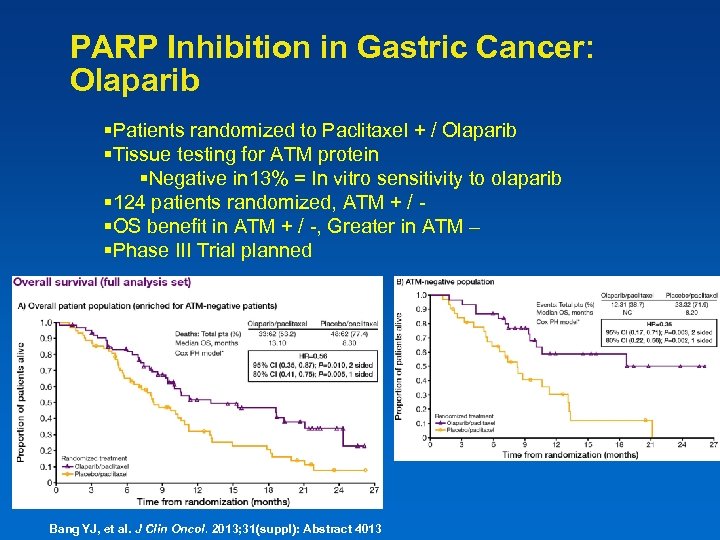 PARP Inhibition in Gastric Cancer: Olaparib §Patients randomized to Paclitaxel + / Olaparib §Tissue testing for ATM protein §Negative in 13% = In vitro sensitivity to olaparib § 124 patients randomized, ATM + / §OS benefit in ATM + / -, Greater in ATM – §Phase III Trial planned Bang YJ, et al. J Clin Oncol. 2013; 31(suppl): Abstract 4013
PARP Inhibition in Gastric Cancer: Olaparib §Patients randomized to Paclitaxel + / Olaparib §Tissue testing for ATM protein §Negative in 13% = In vitro sensitivity to olaparib § 124 patients randomized, ATM + / §OS benefit in ATM + / -, Greater in ATM – §Phase III Trial planned Bang YJ, et al. J Clin Oncol. 2013; 31(suppl): Abstract 4013
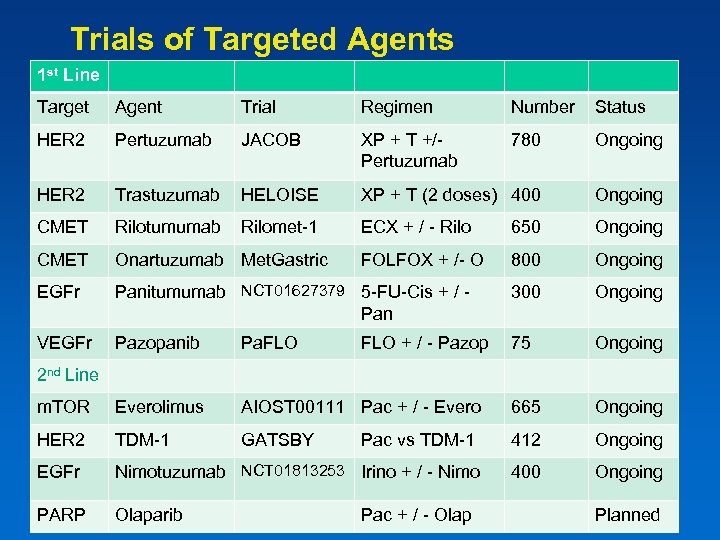 Trials of Targeted Agents 1 st Line Target Agent Trial Regimen Number Status HER 2 Pertuzumab JACOB XP + T +/Pertuzumab 780 Ongoing HER 2 Trastuzumab HELOISE XP + T (2 doses) 400 Ongoing CMET Rilotumumab Rilomet-1 ECX + / - Rilo 650 Ongoing CMET Onartuzumab Met. Gastric FOLFOX + /- O 800 Ongoing EGFr Panitumumab NCT 01627379 5 -FU-Cis + / Pan 300 Ongoing VEGFr Pazopanib Pa. FLO 75 Ongoing m. TOR Everolimus AIOST 00111 Pac + / - Evero 665 Ongoing HER 2 TDM-1 GATSBY Pac vs TDM-1 412 Ongoing EGFr Nimotuzumab NCT 01813253 Irino + / - Nimo 400 Ongoing PARP Olaparib FLO + / - Pazop 2 nd Line Pac + / - Olap Planned
Trials of Targeted Agents 1 st Line Target Agent Trial Regimen Number Status HER 2 Pertuzumab JACOB XP + T +/Pertuzumab 780 Ongoing HER 2 Trastuzumab HELOISE XP + T (2 doses) 400 Ongoing CMET Rilotumumab Rilomet-1 ECX + / - Rilo 650 Ongoing CMET Onartuzumab Met. Gastric FOLFOX + /- O 800 Ongoing EGFr Panitumumab NCT 01627379 5 -FU-Cis + / Pan 300 Ongoing VEGFr Pazopanib Pa. FLO 75 Ongoing m. TOR Everolimus AIOST 00111 Pac + / - Evero 665 Ongoing HER 2 TDM-1 GATSBY Pac vs TDM-1 412 Ongoing EGFr Nimotuzumab NCT 01813253 Irino + / - Nimo 400 Ongoing PARP Olaparib FLO + / - Pazop 2 nd Line Pac + / - Olap Planned
 Esophagogastric Cancer: Immunotherapies • Agents that deregulate immune suppression • Anti PD-1 phase I: – PD-1: T cell programmed cell death receptor, blockade may enhance immune responses – Active in NSCLC, RCC – 7 gastric cancers, no activity • Anti PDL-1 phase I – MPDL 3280 A: Blocks ligand – PR in 1/1 Gastric Cancer, 26/29 responses ongoing – Enhanced activity in PDL-1 + patients • Ipilimumab – Anti CTLA-4 antibody – Phase II – FOLFOX capecitabine maintenance vs ipilumimab Ribas A, et al. N Eng J Med. 2012; 366: 2443 Herbst R et al. JCO 31 (supp): Abstract 3000
Esophagogastric Cancer: Immunotherapies • Agents that deregulate immune suppression • Anti PD-1 phase I: – PD-1: T cell programmed cell death receptor, blockade may enhance immune responses – Active in NSCLC, RCC – 7 gastric cancers, no activity • Anti PDL-1 phase I – MPDL 3280 A: Blocks ligand – PR in 1/1 Gastric Cancer, 26/29 responses ongoing – Enhanced activity in PDL-1 + patients • Ipilimumab – Anti CTLA-4 antibody – Phase II – FOLFOX capecitabine maintenance vs ipilumimab Ribas A, et al. N Eng J Med. 2012; 366: 2443 Herbst R et al. JCO 31 (supp): Abstract 3000
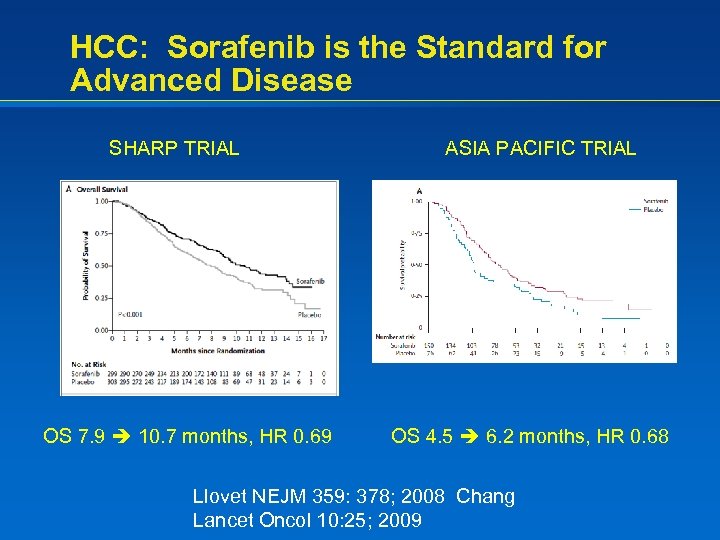 HCC: Sorafenib is the Standard for Advanced Disease SHARP TRIAL OS 7. 9 10. 7 months, HR 0. 69 ASIA PACIFIC TRIAL OS 4. 5 6. 2 months, HR 0. 68 Llovet NEJM 359: 378; 2008 Chang Lancet Oncol 10: 25; 2009
HCC: Sorafenib is the Standard for Advanced Disease SHARP TRIAL OS 7. 9 10. 7 months, HR 0. 69 ASIA PACIFIC TRIAL OS 4. 5 6. 2 months, HR 0. 68 Llovet NEJM 359: 378; 2008 Chang Lancet Oncol 10: 25; 2009
 Sorafenib in HCC l Modest single agent activity in Child’s A pts with HCC l Toxicity monitoring and dose reduction are key l Outcomes vary depending on geographic region, etiology and severity of cirrhosis l No biomarker has been identified
Sorafenib in HCC l Modest single agent activity in Child’s A pts with HCC l Toxicity monitoring and dose reduction are key l Outcomes vary depending on geographic region, etiology and severity of cirrhosis l No biomarker has been identified
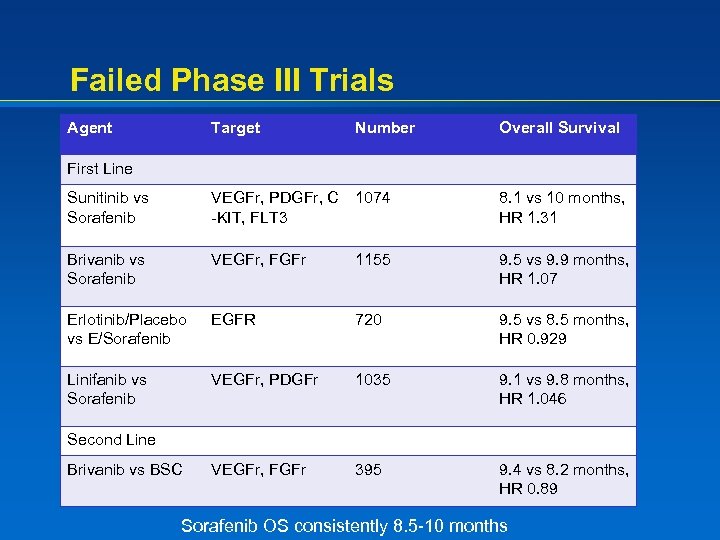 Failed Phase III Trials Agent Target Number Overall Survival Sunitinib vs Sorafenib VEGFr, PDGFr, C -KIT, FLT 3 1074 8. 1 vs 10 months, HR 1. 31 Brivanib vs Sorafenib VEGFr, FGFr 1155 9. 5 vs 9. 9 months, HR 1. 07 Erlotinib/Placebo vs E/Sorafenib EGFR 720 9. 5 vs 8. 5 months, HR 0. 929 Linifanib vs Sorafenib VEGFr, PDGFr 1035 9. 1 vs 9. 8 months, HR 1. 046 VEGFr, FGFr 395 9. 4 vs 8. 2 months, HR 0. 89 First Line Second Line Brivanib vs BSC Sorafenib OS consistently 8. 5 -10 months
Failed Phase III Trials Agent Target Number Overall Survival Sunitinib vs Sorafenib VEGFr, PDGFr, C -KIT, FLT 3 1074 8. 1 vs 10 months, HR 1. 31 Brivanib vs Sorafenib VEGFr, FGFr 1155 9. 5 vs 9. 9 months, HR 1. 07 Erlotinib/Placebo vs E/Sorafenib EGFR 720 9. 5 vs 8. 5 months, HR 0. 929 Linifanib vs Sorafenib VEGFr, PDGFr 1035 9. 1 vs 9. 8 months, HR 1. 046 VEGFr, FGFr 395 9. 4 vs 8. 2 months, HR 0. 89 First Line Second Line Brivanib vs BSC Sorafenib OS consistently 8. 5 -10 months
 Ongoing Single Agent Studies l Angiogenesis: – Ramucirumab, TSU-68, Cedirinab, Pazopanib, lenvatinib, Axitinib l CMET: – Tivantinib, cabozantinib, foretinib, METmab, IMC-280, LY 2875358 l EGFR: – Lapatinib, cetuximab l m. TOR: – Everolimus, temsirolimus, CC-23
Ongoing Single Agent Studies l Angiogenesis: – Ramucirumab, TSU-68, Cedirinab, Pazopanib, lenvatinib, Axitinib l CMET: – Tivantinib, cabozantinib, foretinib, METmab, IMC-280, LY 2875358 l EGFR: – Lapatinib, cetuximab l m. TOR: – Everolimus, temsirolimus, CC-23
 Ongoing Single Agent Studies l MEK – Selumetinib, rafametinib l HDAC – Belinostat, resminostat l HSP-90 – Genetespib l Oncolytic viruses – JX-594 l Immunotherapy – Tremelimumab, PD-1, PD-L 1
Ongoing Single Agent Studies l MEK – Selumetinib, rafametinib l HDAC – Belinostat, resminostat l HSP-90 – Genetespib l Oncolytic viruses – JX-594 l Immunotherapy – Tremelimumab, PD-1, PD-L 1
 CMET Targeted Therapy in HCC l Tivantinib vs Placebo in HCC – CMET TKI l 107 pts, Child’s Pugh A, PS 0 -1, most failed sorafenib – 160 mg tivantinib vs placebo – Cross over permitted at POD l TTP HR 0. 64, p = 0. 04 – OS not different, given cross over (6. 2 - 6. 6 months) l CMET IHC low, better prognosis, no benefit from tivantinib l CMET IHC high, OS 3. 8 to 7. 2 months (HR 0. 38, p = 0. 01) with tivantinib l Phase III Trial planned in CMET high pts OS CMET High Santoro Lancet Oncol 14: 55; 2013
CMET Targeted Therapy in HCC l Tivantinib vs Placebo in HCC – CMET TKI l 107 pts, Child’s Pugh A, PS 0 -1, most failed sorafenib – 160 mg tivantinib vs placebo – Cross over permitted at POD l TTP HR 0. 64, p = 0. 04 – OS not different, given cross over (6. 2 - 6. 6 months) l CMET IHC low, better prognosis, no benefit from tivantinib l CMET IHC high, OS 3. 8 to 7. 2 months (HR 0. 38, p = 0. 01) with tivantinib l Phase III Trial planned in CMET high pts OS CMET High Santoro Lancet Oncol 14: 55; 2013
 CMET Targeted Therapy in HCC l Cabozantinib vs Placebo in HCC (4007) – CMET and VEGR 2 TKI, most patients failed sorafenib l 107 pts, Child’s Pugh A, PS 0 -1, most failed sorafenib – 100 mg cabozantinib, stable disease randomized to placebo or continuation – Cross over permitted at POD l 41 treated l PFS 4. 4 mos, OS 15 mos in all pts l RR 5%, Stable disease 78% l Larger phase II trial planned Verslype et al. J Clin Oncol 30: 2012 (suppl Abst 4007)
CMET Targeted Therapy in HCC l Cabozantinib vs Placebo in HCC (4007) – CMET and VEGR 2 TKI, most patients failed sorafenib l 107 pts, Child’s Pugh A, PS 0 -1, most failed sorafenib – 100 mg cabozantinib, stable disease randomized to placebo or continuation – Cross over permitted at POD l 41 treated l PFS 4. 4 mos, OS 15 mos in all pts l RR 5%, Stable disease 78% l Larger phase II trial planned Verslype et al. J Clin Oncol 30: 2012 (suppl Abst 4007)
 Promising Signals l Ramucirumab – Anti VEGFr 2 – RR 10%, PFS 4 months, OS 12 months l Lenvatinib – VEGFr 1 -3, FGFr 1 -4, RET, KIT, PDGFrβ TKI – 37% modified RECIST response rate – TTP 12. 8 months, OS 18. 7 months l Immunotherapy – Anti CTLA-4 – RR 17%, PFS 6 months
Promising Signals l Ramucirumab – Anti VEGFr 2 – RR 10%, PFS 4 months, OS 12 months l Lenvatinib – VEGFr 1 -3, FGFr 1 -4, RET, KIT, PDGFrβ TKI – 37% modified RECIST response rate – TTP 12. 8 months, OS 18. 7 months l Immunotherapy – Anti CTLA-4 – RR 17%, PFS 6 months
 Ongoing Trials l First Line – Sorafenib vs Sorafenib + Doxorubicin (CALGB 80802) – Lenvantinib vs Sorafenib l Sorafenib + Local Regional Therapy – Sorafenib + / - SBRT (RTOG 1112) – Sorafenib + / - TACE (ECOG) – Sorafenib vs Y 90 l Second Line – Ramucirumab vs BSC – ADI-PEG 20 vs BSC – Tivantinib and Cabozantinib vs BSC – Regorafenib vs BSC
Ongoing Trials l First Line – Sorafenib vs Sorafenib + Doxorubicin (CALGB 80802) – Lenvantinib vs Sorafenib l Sorafenib + Local Regional Therapy – Sorafenib + / - SBRT (RTOG 1112) – Sorafenib + / - TACE (ECOG) – Sorafenib vs Y 90 l Second Line – Ramucirumab vs BSC – ADI-PEG 20 vs BSC – Tivantinib and Cabozantinib vs BSC – Regorafenib vs BSC
 Pancreatic Cancer l Improvements in Chemotherapy l Gemcitabine G + Nab-Paclitaxel FOLFIRINOX l OS 6 months 8. 5 months 11. 1 months l Response: 6% 23% 32% l Targeted Agents – Only approved agent is EGFr TKI Erlotinib
Pancreatic Cancer l Improvements in Chemotherapy l Gemcitabine G + Nab-Paclitaxel FOLFIRINOX l OS 6 months 8. 5 months 11. 1 months l Response: 6% 23% 32% l Targeted Agents – Only approved agent is EGFr TKI Erlotinib
 NCI PA. 3 Phase III Trial Untreated Advanced Pancreas Ca Stratify N= 569 • LA vs M 1 • Center • PS 0 -1 vs 2 Primary Enpoint OS 80% power, 33% increase R A N D O M I Z E Moore, et al. J Clin Oncol, 2007 Gemcitabine + Erlotinib Gemcitabine + Placebo
NCI PA. 3 Phase III Trial Untreated Advanced Pancreas Ca Stratify N= 569 • LA vs M 1 • Center • PS 0 -1 vs 2 Primary Enpoint OS 80% power, 33% increase R A N D O M I Z E Moore, et al. J Clin Oncol, 2007 Gemcitabine + Erlotinib Gemcitabine + Placebo
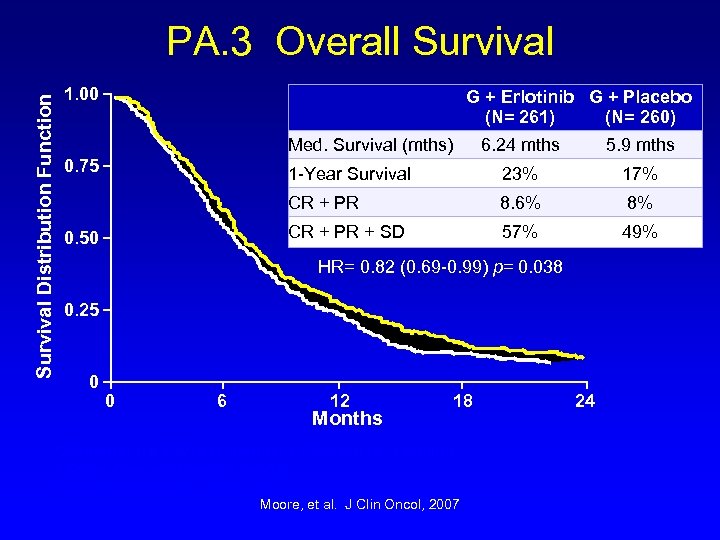 Survival Distribution Function PA. 3 Overall Survival 1. 00 G + Erlotinib G + Placebo (N= 261) (N= 260) Med. Survival (mths) 23% 17% CR + PR 8. 6% 8% CR + PR + SD 0. 50 5. 9 mths 1 -Year Survival 0. 75 6. 24 mths 57% 49% HR= 0. 82 (0. 69 -0. 99) p= 0. 038 0. 25 0 0 6 12 Months 18 *Adjusted for PS and extent of disease at baseline † From Cox regression model ‡ From 2 -sided log-rank test Moore, et al. J Clin Oncol, 2007 24
Survival Distribution Function PA. 3 Overall Survival 1. 00 G + Erlotinib G + Placebo (N= 261) (N= 260) Med. Survival (mths) 23% 17% CR + PR 8. 6% 8% CR + PR + SD 0. 50 5. 9 mths 1 -Year Survival 0. 75 6. 24 mths 57% 49% HR= 0. 82 (0. 69 -0. 99) p= 0. 038 0. 25 0 0 6 12 Months 18 *Adjusted for PS and extent of disease at baseline † From Cox regression model ‡ From 2 -sided log-rank test Moore, et al. J Clin Oncol, 2007 24
 Molecular Correlates Gemcitabine +/- Erlotinib PA. 3 • N= 569 pts – 117 samples (21%) – EGFR (+) or (–) no correlation with outcome – Post-hoc K-ras mutational status analysis Trend to OS benefit in the pts with wild-type K-ras Gem + E HR P-value K-ras mutant 7. 4 mths 6. 0 mths 1. 07 0. 78 K-ras WT 4. 5 mths 6. 1 mths 0. 66 0. 34 Moore, et al. ASCO, 2007 (Abst #4521)
Molecular Correlates Gemcitabine +/- Erlotinib PA. 3 • N= 569 pts – 117 samples (21%) – EGFR (+) or (–) no correlation with outcome – Post-hoc K-ras mutational status analysis Trend to OS benefit in the pts with wild-type K-ras Gem + E HR P-value K-ras mutant 7. 4 mths 6. 0 mths 1. 07 0. 78 K-ras WT 4. 5 mths 6. 1 mths 0. 66 0. 34 Moore, et al. ASCO, 2007 (Abst #4521)
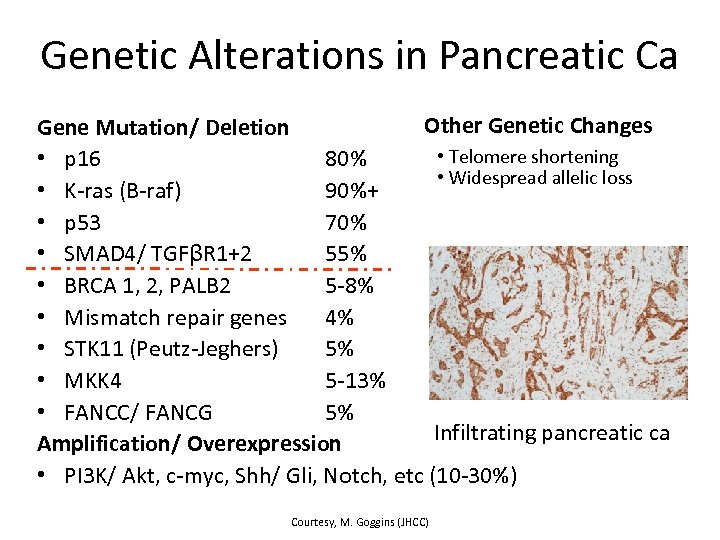 Genetic Alterations in Pancreatic Ca Other Genetic Changes Gene Mutation/ Deletion • Telomere shortening • p 16 80% • Widespread allelic loss • K-ras (B-raf) 90%+ • p 53 70% • SMAD 4/ TGFβR 1+2 55% • BRCA 1, 2, PALB 2 5 -8% • Mismatch repair genes 4% • STK 11 (Peutz-Jeghers) 5% • MKK 4 5 -13% • FANCC/ FANCG 5% Infiltrating pancreatic ca Amplification/ Overexpression • PI 3 K/ Akt, c-myc, Shh/ Gli, Notch, etc (10 -30%) Courtesy, M. Goggins (JHCC)
Genetic Alterations in Pancreatic Ca Other Genetic Changes Gene Mutation/ Deletion • Telomere shortening • p 16 80% • Widespread allelic loss • K-ras (B-raf) 90%+ • p 53 70% • SMAD 4/ TGFβR 1+2 55% • BRCA 1, 2, PALB 2 5 -8% • Mismatch repair genes 4% • STK 11 (Peutz-Jeghers) 5% • MKK 4 5 -13% • FANCC/ FANCG 5% Infiltrating pancreatic ca Amplification/ Overexpression • PI 3 K/ Akt, c-myc, Shh/ Gli, Notch, etc (10 -30%) Courtesy, M. Goggins (JHCC)
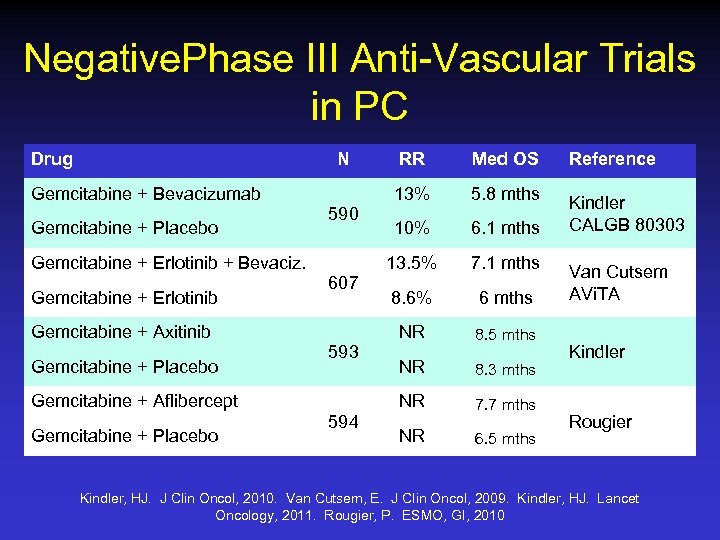 Negative. Phase III Anti-Vascular Trials in PC Drug N Gemcitabine + Placebo Gemcitabine + Erlotinib 607 Gemcitabine + Axitinib Gemcitabine + Placebo 593 6. 1 mths 7. 1 mths 8. 6% 6 mths 8. 5 mths 594 NR 8. 3 mths NR Gemcitabine + Aflibercept Gemcitabine + Placebo 10% NR Gemcitabine + Erlotinib + Bevaciz. 5. 8 mths 13. 5% 590 Med OS 13% Gemcitabine + Bevacizumab RR 7. 7 mths NR 6. 5 mths Reference Kindler CALGB 80303 Van Cutsem AVi. TA Kindler Rougier Kindler, HJ. J Clin Oncol, 2010. Van Cutsem, E. J Clin Oncol, 2009. Kindler, HJ. Lancet Oncology, 2011. Rougier, P. ESMO, GI, 2010
Negative. Phase III Anti-Vascular Trials in PC Drug N Gemcitabine + Placebo Gemcitabine + Erlotinib 607 Gemcitabine + Axitinib Gemcitabine + Placebo 593 6. 1 mths 7. 1 mths 8. 6% 6 mths 8. 5 mths 594 NR 8. 3 mths NR Gemcitabine + Aflibercept Gemcitabine + Placebo 10% NR Gemcitabine + Erlotinib + Bevaciz. 5. 8 mths 13. 5% 590 Med OS 13% Gemcitabine + Bevacizumab RR 7. 7 mths NR 6. 5 mths Reference Kindler CALGB 80303 Van Cutsem AVi. TA Kindler Rougier Kindler, HJ. J Clin Oncol, 2010. Van Cutsem, E. J Clin Oncol, 2009. Kindler, HJ. Lancet Oncology, 2011. Rougier, P. ESMO, GI, 2010
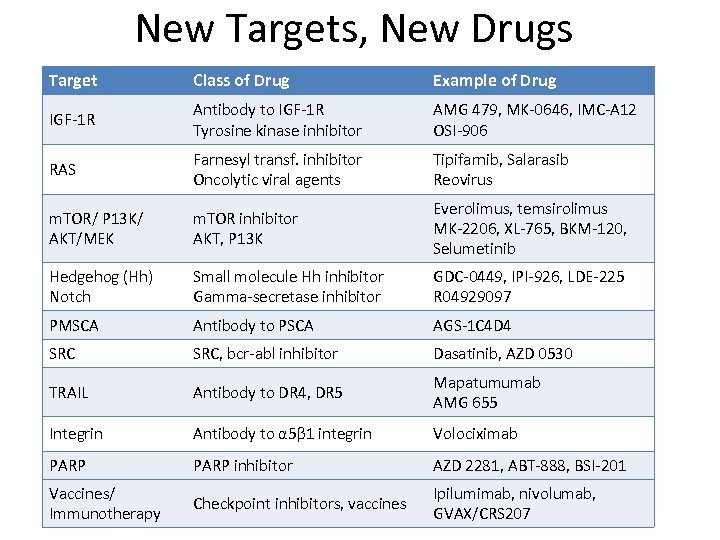 New Targets, New Drugs Target Class of Drug Example of Drug IGF-1 R Antibody to IGF-1 R Tyrosine kinase inhibitor AMG 479, MK-0646, IMC-A 12 OSI-906 RAS Farnesyl transf. inhibitor Oncolytic viral agents Tipifarnib, Salarasib Reovirus m. TOR/ P 13 K/ AKT/MEK m. TOR inhibitor AKT, P 13 K Everolimus, temsirolimus MK-2206, XL-765, BKM-120, Selumetinib Hedgehog (Hh) Notch Small molecule Hh inhibitor Gamma-secretase inhibitor GDC-0449, IPI-926, LDE-225 R 04929097 PMSCA Antibody to PSCA AGS-1 C 4 D 4 SRC, bcr-abl inhibitor Dasatinib, AZD 0530 TRAIL Antibody to DR 4, DR 5 Mapatumumab AMG 655 Integrin Antibody to α 5β 1 integrin Volociximab PARP inhibitor AZD 2281, ABT-888, BSI-201 Vaccines/ Immunotherapy Checkpoint inhibitors, vaccines Ipilumimab, nivolumab, GVAX/CRS 207
New Targets, New Drugs Target Class of Drug Example of Drug IGF-1 R Antibody to IGF-1 R Tyrosine kinase inhibitor AMG 479, MK-0646, IMC-A 12 OSI-906 RAS Farnesyl transf. inhibitor Oncolytic viral agents Tipifarnib, Salarasib Reovirus m. TOR/ P 13 K/ AKT/MEK m. TOR inhibitor AKT, P 13 K Everolimus, temsirolimus MK-2206, XL-765, BKM-120, Selumetinib Hedgehog (Hh) Notch Small molecule Hh inhibitor Gamma-secretase inhibitor GDC-0449, IPI-926, LDE-225 R 04929097 PMSCA Antibody to PSCA AGS-1 C 4 D 4 SRC, bcr-abl inhibitor Dasatinib, AZD 0530 TRAIL Antibody to DR 4, DR 5 Mapatumumab AMG 655 Integrin Antibody to α 5β 1 integrin Volociximab PARP inhibitor AZD 2281, ABT-888, BSI-201 Vaccines/ Immunotherapy Checkpoint inhibitors, vaccines Ipilumimab, nivolumab, GVAX/CRS 207
 Phase II: GVAX + / - CRS-207 l GVAX: Irradiated, GM-CSF secreting allogeneic pancreatic cancer cell lines given intradermally, preceded by Cytoxan to reduce T regs l CRS-207: Live-attenuated Listeria monocytogenes which expresses mesothelin immune stimulant l 90 pts previous treated randomized 2: 1 to C/GVAX + CRS-207 or C/GVAX alone l OS 6. 1 vs 3. 9 months (HR 0. 54, p = 0. 011) l More CA 19 -9 stabilization with combination Le J Clin Oncol 32: 2014 (suppl 3; abstr 177)
Phase II: GVAX + / - CRS-207 l GVAX: Irradiated, GM-CSF secreting allogeneic pancreatic cancer cell lines given intradermally, preceded by Cytoxan to reduce T regs l CRS-207: Live-attenuated Listeria monocytogenes which expresses mesothelin immune stimulant l 90 pts previous treated randomized 2: 1 to C/GVAX + CRS-207 or C/GVAX alone l OS 6. 1 vs 3. 9 months (HR 0. 54, p = 0. 011) l More CA 19 -9 stabilization with combination Le J Clin Oncol 32: 2014 (suppl 3; abstr 177)
 Recent Negative Phase II-III Targeted Therapy Drug Trial Gem + AMG-479 (IGF-1 R) Phase III GAMMA Med PFS or OS No Change Press Release Gem + Placebo Gem + Sorafenib Phase III BAYPAN Gem + Placebo Gem Phase II LEAP Phase III Gem (Shh) 7. 7 mos 6. 0 mos Phase II Gem + Vismodegib OS 6. 0 mos Gem + Placebo Gem + IPI-926 (Shh) PFS 5. 7 mos 3. 8 mos CO-1. 01 (h. ENT 1) Gem + Masitinib (CKIT, PDGF, FGF) Reference 6. 0 mos Goncalves Ann Oncol 2012 Poplin ASCO 2013 Deplanqa GI Symposium 2013 Press Release 5. 9 mos Phase II 6. 3 mos 5. 4 mos Catenacci ASCO 2012
Recent Negative Phase II-III Targeted Therapy Drug Trial Gem + AMG-479 (IGF-1 R) Phase III GAMMA Med PFS or OS No Change Press Release Gem + Placebo Gem + Sorafenib Phase III BAYPAN Gem + Placebo Gem Phase II LEAP Phase III Gem (Shh) 7. 7 mos 6. 0 mos Phase II Gem + Vismodegib OS 6. 0 mos Gem + Placebo Gem + IPI-926 (Shh) PFS 5. 7 mos 3. 8 mos CO-1. 01 (h. ENT 1) Gem + Masitinib (CKIT, PDGF, FGF) Reference 6. 0 mos Goncalves Ann Oncol 2012 Poplin ASCO 2013 Deplanqa GI Symposium 2013 Press Release 5. 9 mos Phase II 6. 3 mos 5. 4 mos Catenacci ASCO 2012
 Novel Targets l Stroma and Microenvironment – PEGPH 20: degrades hyaluronic acic, may increase drug delivery – Onoing phase II: n Nab-P + / - PEGPH 20 n FOLFIRINOX + / - PEGPH 20 l BRCA mutant pancreatic cancers (5%) – Deficient homologous DS DNA repair – Results in genomic instability, chromosomal deletion and exchange – PARP inhibition n Cis-Gem + / Veliparib, randomized phase II
Novel Targets l Stroma and Microenvironment – PEGPH 20: degrades hyaluronic acic, may increase drug delivery – Onoing phase II: n Nab-P + / - PEGPH 20 n FOLFIRINOX + / - PEGPH 20 l BRCA mutant pancreatic cancers (5%) – Deficient homologous DS DNA repair – Results in genomic instability, chromosomal deletion and exchange – PARP inhibition n Cis-Gem + / Veliparib, randomized phase II
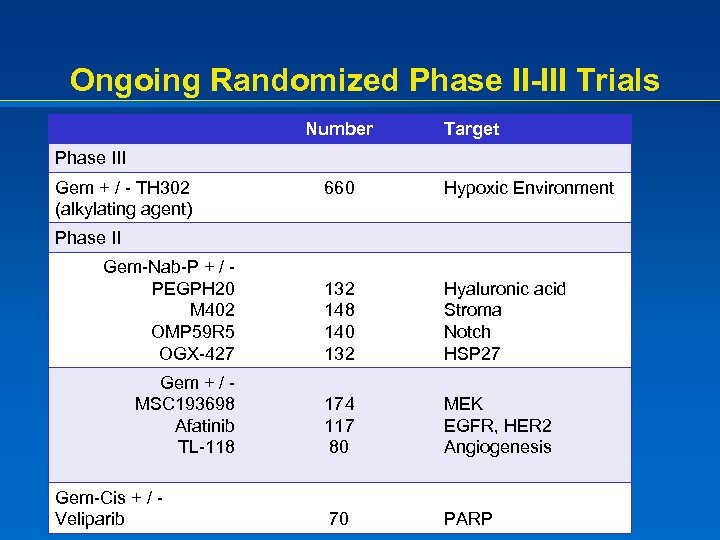 Ongoing Randomized Phase II-III Trials Number Target Phase III Gem + / - TH 302 (alkylating agent) 660 Hypoxic Environment Gem-Nab-P + / PEGPH 20 M 402 OMP 59 R 5 OGX-427 132 148 140 132 Hyaluronic acid Stroma Notch HSP 27 Gem + / MSC 193698 Afatinib TL-118 174 117 80 MEK EGFR, HER 2 Angiogenesis 70 PARP Phase II Gem-Cis + / Veliparib
Ongoing Randomized Phase II-III Trials Number Target Phase III Gem + / - TH 302 (alkylating agent) 660 Hypoxic Environment Gem-Nab-P + / PEGPH 20 M 402 OMP 59 R 5 OGX-427 132 148 140 132 Hyaluronic acid Stroma Notch HSP 27 Gem + / MSC 193698 Afatinib TL-118 174 117 80 MEK EGFR, HER 2 Angiogenesis 70 PARP Phase II Gem-Cis + / Veliparib
 Approved Targeted Agents in CRC l Growth factor receptor inhibitors: – VEGFr/ VEGF n Anti VEGF A ligand antibody: Bevacizumab n Soluble VEGF receptor: Aflibercept n VEGFr TKI: Regorafenib – EGFr: n Anti EGFr antibodies: § Cetuximab § Panitumumab
Approved Targeted Agents in CRC l Growth factor receptor inhibitors: – VEGFr/ VEGF n Anti VEGF A ligand antibody: Bevacizumab n Soluble VEGF receptor: Aflibercept n VEGFr TKI: Regorafenib – EGFr: n Anti EGFr antibodies: § Cetuximab § Panitumumab
 Integration of VEGF Targeted Agents into Chemotherapy l Bevacizumab can be used with first line FOLFIRI, 5 -FU/capecitabine, FOLFOX l Bevacizumab can be continued into second line chemotherapy, with FOLFOX or FOLFIRI l Alfibercept can be used second line with FOLFIRI after POD on FOLFOX/Bev l Regorafenib: Late line therapy after POD on all conventional lines of therapy
Integration of VEGF Targeted Agents into Chemotherapy l Bevacizumab can be used with first line FOLFIRI, 5 -FU/capecitabine, FOLFOX l Bevacizumab can be continued into second line chemotherapy, with FOLFOX or FOLFIRI l Alfibercept can be used second line with FOLFIRI after POD on FOLFOX/Bev l Regorafenib: Late line therapy after POD on all conventional lines of therapy
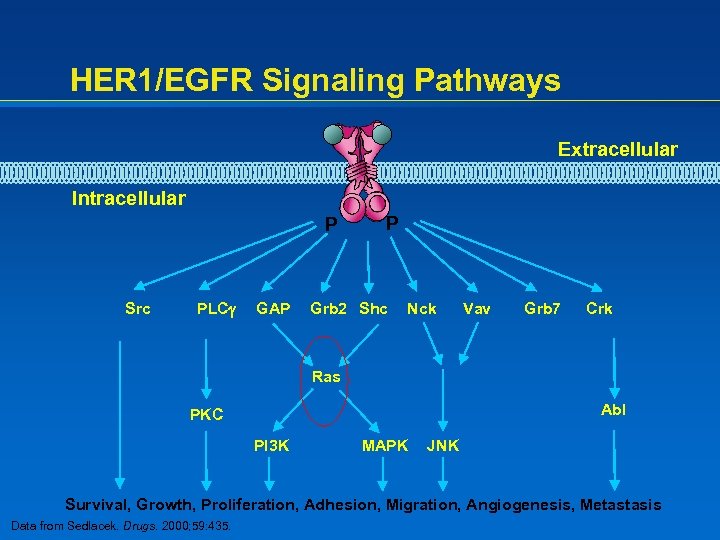 HER 1/EGFR Signaling Pathways Extracellular Intracellular P Src PLC GAP P Grb 2 Shc Nck Vav Grb 7 Crk Ras Abl PKC PI 3 K MAPK JNK Survival, Growth, Proliferation, Adhesion, Migration, Angiogenesis, Metastasis Data from Sedlacek. Drugs. 2000; 59: 435.
HER 1/EGFR Signaling Pathways Extracellular Intracellular P Src PLC GAP P Grb 2 Shc Nck Vav Grb 7 Crk Ras Abl PKC PI 3 K MAPK JNK Survival, Growth, Proliferation, Adhesion, Migration, Angiogenesis, Metastasis Data from Sedlacek. Drugs. 2000; 59: 435.
 Integration of EGFR Agents in Colorectal Cancer: KRAS WT tumors l First Line: – Cetuximab, Panitumumab approved to combine with FOLFIRI or FOLFOX – Capecitabine based trials with Cetuximab failed toxicity l Second, Third Line – Cetuximab, Panitumumab approved as monotherapy – Suggested added benefit when combined with irinotecan or FOLFIRI second line l BRAF mutant patients are eligible for EGFR therapy
Integration of EGFR Agents in Colorectal Cancer: KRAS WT tumors l First Line: – Cetuximab, Panitumumab approved to combine with FOLFIRI or FOLFOX – Capecitabine based trials with Cetuximab failed toxicity l Second, Third Line – Cetuximab, Panitumumab approved as monotherapy – Suggested added benefit when combined with irinotecan or FOLFIRI second line l BRAF mutant patients are eligible for EGFR therapy
 All KRAS Mutant Patients Should Not Get Cetuximab l Current testing looks at KRAS exon 2 mutations l Trial of FOLFOX + / - Cetuximab l An additional 17% had KRAS exon 3/4, NRAS exon 2/3/4 Douillard NEJM 369: 11; 2013
All KRAS Mutant Patients Should Not Get Cetuximab l Current testing looks at KRAS exon 2 mutations l Trial of FOLFOX + / - Cetuximab l An additional 17% had KRAS exon 3/4, NRAS exon 2/3/4 Douillard NEJM 369: 11; 2013
 CALGB 80405: Bevacizumab vs Cetuximab in First-line KRAS WT m. CRC Untreated KRAS WT m. CRC (n=1500) n n Bevacizumab + FOLFOX or FOLFIRI PD Cetuximab + FOLFOX or FOLFIRI PD R Primary endpoint: OS Secondary endpoints: ORR, PFS, TTF, DOR, and safety NCT identifier: NCT 00265850. 57 57
CALGB 80405: Bevacizumab vs Cetuximab in First-line KRAS WT m. CRC Untreated KRAS WT m. CRC (n=1500) n n Bevacizumab + FOLFOX or FOLFIRI PD Cetuximab + FOLFOX or FOLFIRI PD R Primary endpoint: OS Secondary endpoints: ORR, PFS, TTF, DOR, and safety NCT identifier: NCT 00265850. 57 57
 Phase III study design FOLFIRI + Cetuximab m. CRC 1 st-line therapy KRAS wild-type N= 592 Cetuximab: Randomize 1: 1 400 mg/m 2 250 mg/m 2 i. v. 120 min i. v. 60 min initial dose q 1 w FOLFIRI + Bevacizumab: 5 mg/kg i. v. 30 -90 min q 2 w FOLFIRI: 5 -FU: 400 mg/m 2 (i. v. bolus); folinic acid: 400 mg/m 2 irinotecan: 180 mg/m 2 5 -FU: 2, 400 mg/m 2 (i. v. 46 h) • Key inclusion criteria – Patients ≥ 18 years with histologically confirmed diagnosis of m. CRC – ECOG PS 0 -2 – prior adjuvant chemotherapy allowed if completed >6 month before inclusion • Amendment in October 2008 to include only KRAS wildtype patients • 150 active centers in Germany and Austria
Phase III study design FOLFIRI + Cetuximab m. CRC 1 st-line therapy KRAS wild-type N= 592 Cetuximab: Randomize 1: 1 400 mg/m 2 250 mg/m 2 i. v. 120 min i. v. 60 min initial dose q 1 w FOLFIRI + Bevacizumab: 5 mg/kg i. v. 30 -90 min q 2 w FOLFIRI: 5 -FU: 400 mg/m 2 (i. v. bolus); folinic acid: 400 mg/m 2 irinotecan: 180 mg/m 2 5 -FU: 2, 400 mg/m 2 (i. v. 46 h) • Key inclusion criteria – Patients ≥ 18 years with histologically confirmed diagnosis of m. CRC – ECOG PS 0 -2 – prior adjuvant chemotherapy allowed if completed >6 month before inclusion • Amendment in October 2008 to include only KRAS wildtype patients • 150 active centers in Germany and Austria
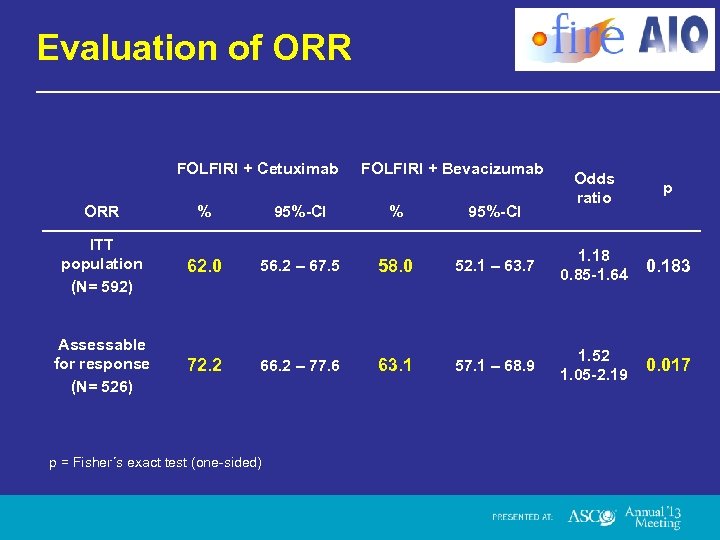 Evaluation of ORR FOLFIRI + Cetuximab FOLFIRI + Bevacizumab Odds ratio p ORR % 95%-CI ITT population (N= 592) 62. 0 56. 2 – 67. 5 58. 0 52. 1 – 63. 7 1. 18 0. 85 -1. 64 0. 183 Assessable for response (N= 526) 72. 2 66. 2 – 77. 6 63. 1 57. 1 – 68. 9 1. 52 1. 05 -2. 19 0. 017 p = Fisher´s exact test (one-sided)
Evaluation of ORR FOLFIRI + Cetuximab FOLFIRI + Bevacizumab Odds ratio p ORR % 95%-CI ITT population (N= 592) 62. 0 56. 2 – 67. 5 58. 0 52. 1 – 63. 7 1. 18 0. 85 -1. 64 0. 183 Assessable for response (N= 526) 72. 2 66. 2 – 77. 6 63. 1 57. 1 – 68. 9 1. 52 1. 05 -2. 19 0. 017 p = Fisher´s exact test (one-sided)
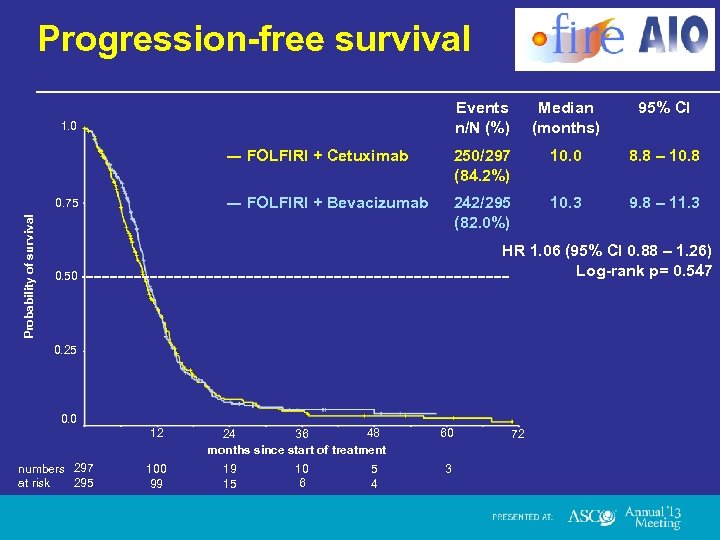 Progression-free survival Events n/N (%) Median (months) 95% CI ― FOLFIRI + Cetuximab 250/297 (84. 2%) 10. 0 8. 8 – 10. 8 ― FOLFIRI + Bevacizumab 242/295 (82. 0%) 10. 3 9. 8 – 11. 3 1. 0 Probability of survival 0. 75 HR 1. 06 (95% CI 0. 88 – 1. 26) Log-rank p= 0. 547 0. 50 0. 25 0. 0 12 numbers 297 295 at risk 100 99 48 36 24 months since start of treatment 19 15 10 6 5 4 60 3 72
Progression-free survival Events n/N (%) Median (months) 95% CI ― FOLFIRI + Cetuximab 250/297 (84. 2%) 10. 0 8. 8 – 10. 8 ― FOLFIRI + Bevacizumab 242/295 (82. 0%) 10. 3 9. 8 – 11. 3 1. 0 Probability of survival 0. 75 HR 1. 06 (95% CI 0. 88 – 1. 26) Log-rank p= 0. 547 0. 50 0. 25 0. 0 12 numbers 297 295 at risk 100 99 48 36 24 months since start of treatment 19 15 10 6 5 4 60 3 72
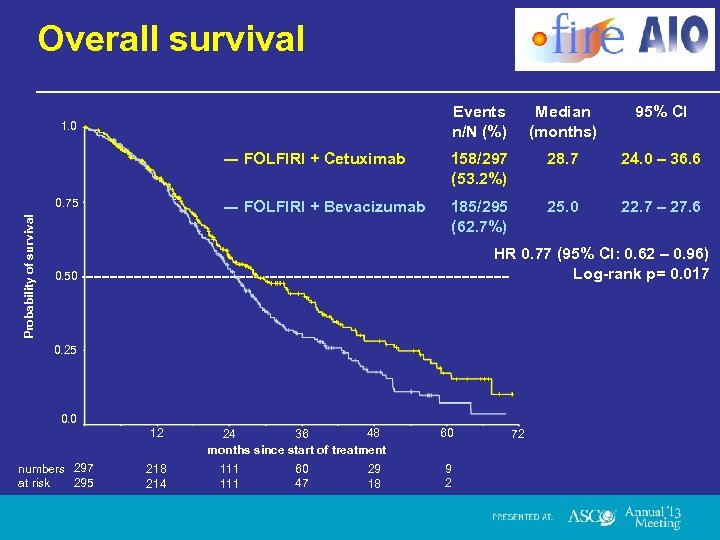 Overall survival Events n/N (%) Median (months) 95% CI ― FOLFIRI + Cetuximab 158/297 (53. 2%) 28. 7 24. 0 – 36. 6 ― FOLFIRI + Bevacizumab 185/295 (62. 7%) 25. 0 22. 7 – 27. 6 1. 0 Probability of survival 0. 75 HR 0. 77 (95% CI: 0. 62 – 0. 96) Log-rank p= 0. 017 0. 50 0. 25 0. 0 12 numbers 297 295 at risk 218 214 48 36 24 months since start of treatment 111 60 47 29 18 60 9 2 72
Overall survival Events n/N (%) Median (months) 95% CI ― FOLFIRI + Cetuximab 158/297 (53. 2%) 28. 7 24. 0 – 36. 6 ― FOLFIRI + Bevacizumab 185/295 (62. 7%) 25. 0 22. 7 – 27. 6 1. 0 Probability of survival 0. 75 HR 0. 77 (95% CI: 0. 62 – 0. 96) Log-rank p= 0. 017 0. 50 0. 25 0. 0 12 numbers 297 295 at risk 218 214 48 36 24 months since start of treatment 111 60 47 29 18 60 9 2 72
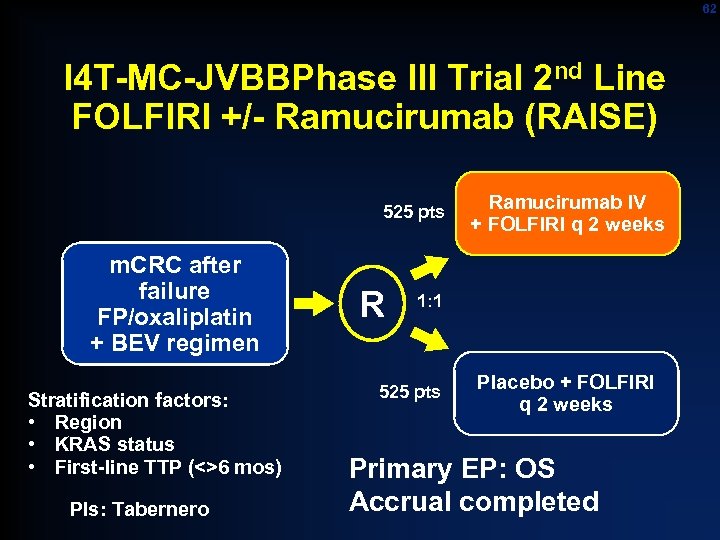 62 I 4 T-MC-JVBBPhase III Trial 2 nd Line FOLFIRI +/- Ramucirumab (RAISE) 525 pts m. CRC after failure FP/oxaliplatin + BEV regimen Stratification factors: • Region • KRAS status • First-line TTP (<>6 mos) PIs: Tabernero R Ramucirumab IV + FOLFIRI q 2 weeks 1: 1 525 pts Placebo + FOLFIRI q 2 weeks Primary EP: OS Accrual completed
62 I 4 T-MC-JVBBPhase III Trial 2 nd Line FOLFIRI +/- Ramucirumab (RAISE) 525 pts m. CRC after failure FP/oxaliplatin + BEV regimen Stratification factors: • Region • KRAS status • First-line TTP (<>6 mos) PIs: Tabernero R Ramucirumab IV + FOLFIRI q 2 weeks 1: 1 525 pts Placebo + FOLFIRI q 2 weeks Primary EP: OS Accrual completed
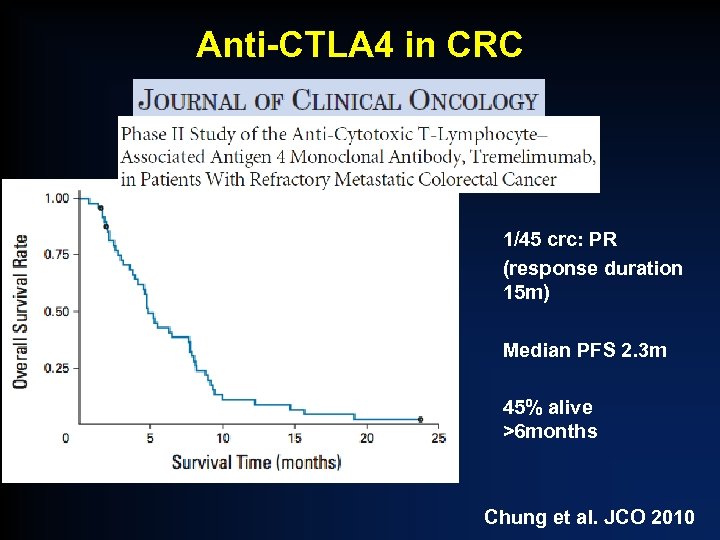 Anti-CTLA 4 in CRC 1/45 crc: PR (response duration 15 m) Median PFS 2. 3 m 45% alive >6 months Chung et al. JCO 2010
Anti-CTLA 4 in CRC 1/45 crc: PR (response duration 15 m) Median PFS 2. 3 m 45% alive >6 months Chung et al. JCO 2010
 Anti-PDL 1 Anti-PD 1 BMS-936559 BMS-936558 RESPONSE RATE 0/18 crc RESPONSE RATE 1/14 crc: CR MPDL 3280 A RESPONSE RATE 1/4 crc RESPONSE RATE 0/19 crc Net N =55 CRC patients
Anti-PDL 1 Anti-PD 1 BMS-936559 BMS-936558 RESPONSE RATE 0/18 crc RESPONSE RATE 1/14 crc: CR MPDL 3280 A RESPONSE RATE 1/4 crc RESPONSE RATE 0/19 crc Net N =55 CRC patients
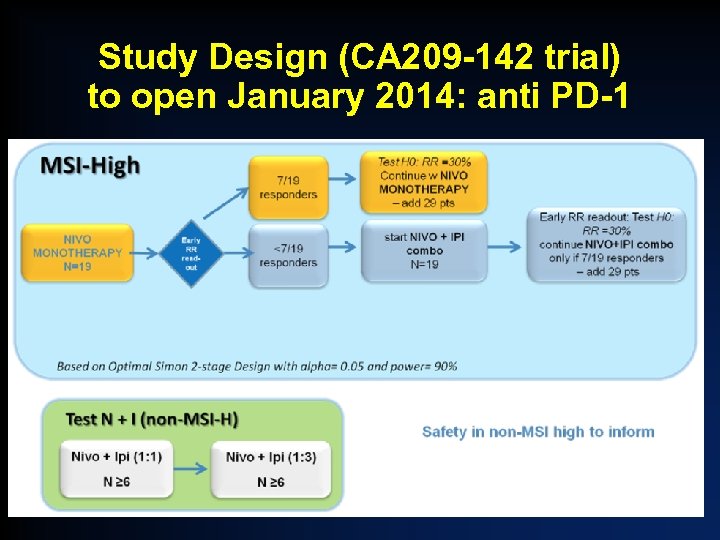 Study Design (CA 209 -142 trial) to open January 2014: anti PD-1
Study Design (CA 209 -142 trial) to open January 2014: anti PD-1
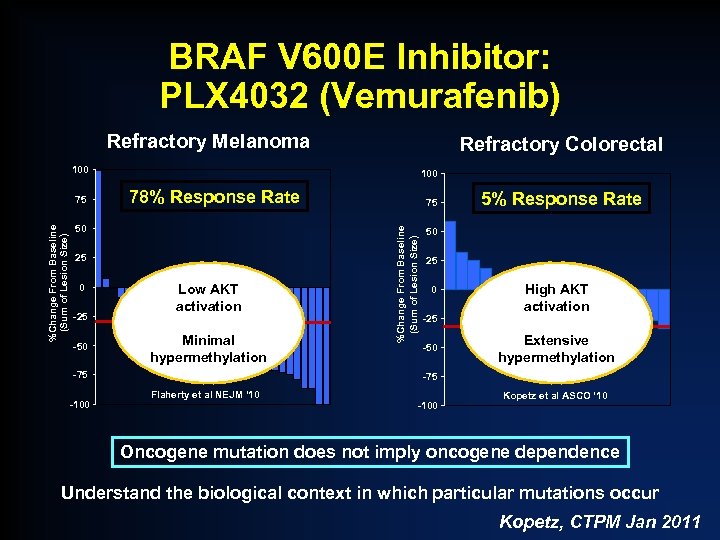 BRAF V 600 E Inhibitor: PLX 4032 (Vemurafenib) Refractory Melanoma Refractory Colorectal 100 78% Response Rate 50 25 0 -25 -50 Low AKT activation Minimal hypermethylation -75 -100 75 %Change From Baseline (Sum of Lesion Size) 75 100 5% Response Rate 50 25 0 -25 -50 High AKT activation Extensive hypermethylation -75 Flaherty et al NEJM ‘ 10 -100 Kopetz et al ASCO ‘ 10 Oncogene mutation does not imply oncogene dependence Understand the biological context in which particular mutations occur Kopetz, CTPM Jan 2011
BRAF V 600 E Inhibitor: PLX 4032 (Vemurafenib) Refractory Melanoma Refractory Colorectal 100 78% Response Rate 50 25 0 -25 -50 Low AKT activation Minimal hypermethylation -75 -100 75 %Change From Baseline (Sum of Lesion Size) 75 100 5% Response Rate 50 25 0 -25 -50 High AKT activation Extensive hypermethylation -75 Flaherty et al NEJM ‘ 10 -100 Kopetz et al ASCO ‘ 10 Oncogene mutation does not imply oncogene dependence Understand the biological context in which particular mutations occur Kopetz, CTPM Jan 2011
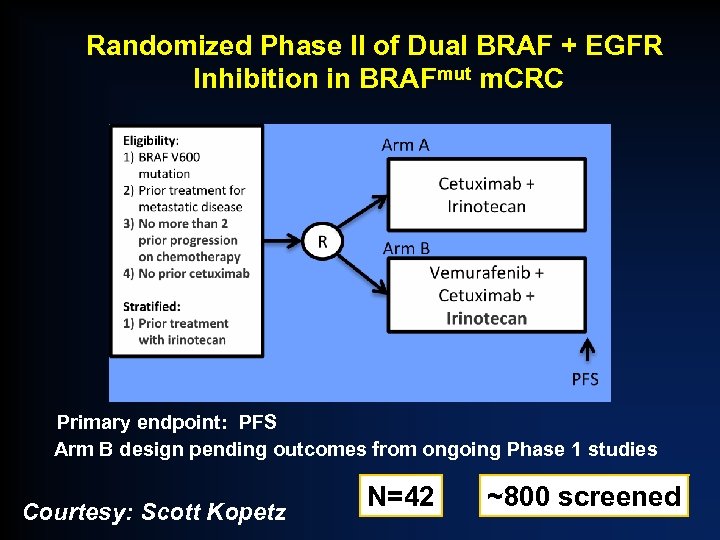 Randomized Phase II of Dual BRAF + EGFR Inhibition in BRAFmut m. CRC Primary endpoint: PFS Arm B design pending outcomes from ongoing Phase 1 studies Courtesy: Scott Kopetz N=42 ~800 screened
Randomized Phase II of Dual BRAF + EGFR Inhibition in BRAFmut m. CRC Primary endpoint: PFS Arm B design pending outcomes from ongoing Phase 1 studies Courtesy: Scott Kopetz N=42 ~800 screened
 Prominent c. MET / HGF Inhibitors Agent Structure Target Rilotumumab Human monoclonal antibody HGF Onartuzumab (met. Mab) Humanized monovalent c-MET antibody Tivantinib (ARQ 197) Small molecule c-MET kinase Cabozantinib (XL 184) Small molecule c-MET kinase
Prominent c. MET / HGF Inhibitors Agent Structure Target Rilotumumab Human monoclonal antibody HGF Onartuzumab (met. Mab) Humanized monovalent c-MET antibody Tivantinib (ARQ 197) Small molecule c-MET kinase Cabozantinib (XL 184) Small molecule c-MET kinase
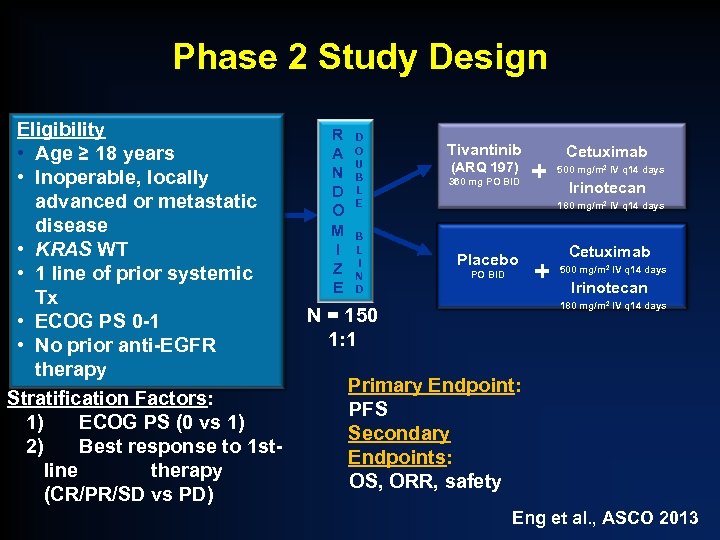 Phase 2 Study Design Eligibility • Age ≥ 18 years • Inoperable, locally advanced or metastatic disease • KRAS WT • 1 line of prior systemic Tx • ECOG PS 0 -1 • No prior anti-EGFR therapy Stratification Factors: 1) ECOG PS (0 vs 1) 2) Best response to 1 stline therapy (CR/PR/SD vs PD) R A N D O M I Z E D O U B L E B L I N D Tivantinib (ARQ 197) + 360 mg PO BID Cetuximab 500 mg/m 2 IV q 14 days Irinotecan 180 mg/m 2 IV q 14 days Placebo PO BID + Cetuximab 500 mg/m 2 IV q 14 days Irinotecan 180 mg/m 2 IV q 14 days N = 150 1: 1 Primary Endpoint: PFS Secondary Endpoints: OS, ORR, safety Eng et al. , ASCO 2013
Phase 2 Study Design Eligibility • Age ≥ 18 years • Inoperable, locally advanced or metastatic disease • KRAS WT • 1 line of prior systemic Tx • ECOG PS 0 -1 • No prior anti-EGFR therapy Stratification Factors: 1) ECOG PS (0 vs 1) 2) Best response to 1 stline therapy (CR/PR/SD vs PD) R A N D O M I Z E D O U B L E B L I N D Tivantinib (ARQ 197) + 360 mg PO BID Cetuximab 500 mg/m 2 IV q 14 days Irinotecan 180 mg/m 2 IV q 14 days Placebo PO BID + Cetuximab 500 mg/m 2 IV q 14 days Irinotecan 180 mg/m 2 IV q 14 days N = 150 1: 1 Primary Endpoint: PFS Secondary Endpoints: OS, ORR, safety Eng et al. , ASCO 2013
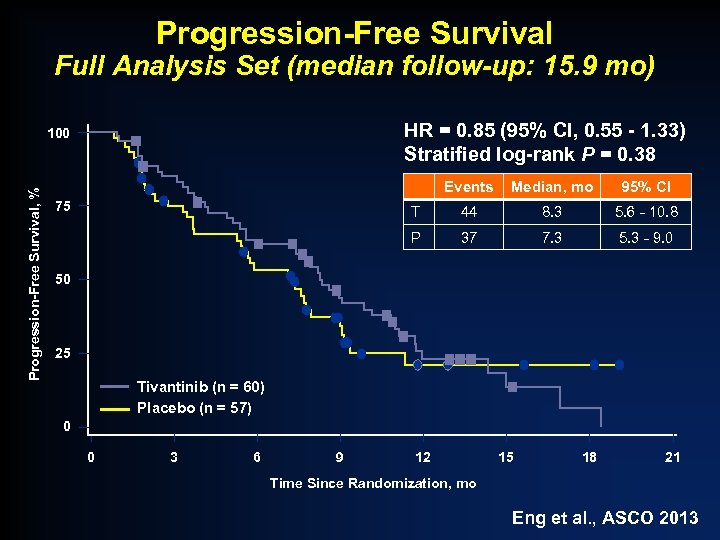 Progression-Free Survival Full Analysis Set (median follow-up: 15. 9 mo) HR = 0. 85 (95% CI, 0. 55 - 1. 33) Stratified log-rank P = 0. 38 Progression-Free Survival, % 100 Events 95% CI T 44 8. 3 5. 6 - 10. 8 P 75 Median, mo 37 7. 3 5. 3 - 9. 0 50 25 Tivantinib (n = 60) Placebo (n = 57) 0 0 3 6 9 12 15 18 21 Time Since Randomization, mo Eng et al. , ASCO 2013
Progression-Free Survival Full Analysis Set (median follow-up: 15. 9 mo) HR = 0. 85 (95% CI, 0. 55 - 1. 33) Stratified log-rank P = 0. 38 Progression-Free Survival, % 100 Events 95% CI T 44 8. 3 5. 6 - 10. 8 P 75 Median, mo 37 7. 3 5. 3 - 9. 0 50 25 Tivantinib (n = 60) Placebo (n = 57) 0 0 3 6 9 12 15 18 21 Time Since Randomization, mo Eng et al. , ASCO 2013
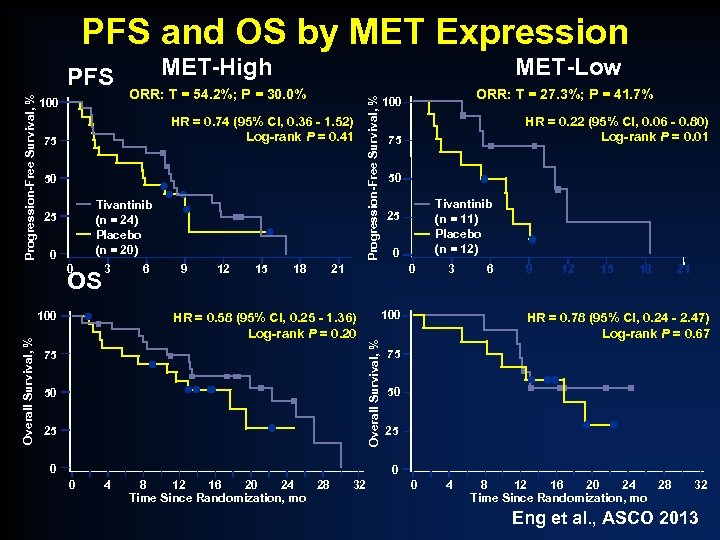 PFS and OS by MET Expression ORR: T = 54. 2%; P = 30. 0% HR = 0. 74 (95% CI, 0. 36 - 1. 52) Log-rank P = 0. 41 75 50 Tivantinib (n = 24) Placebo (n = 20) 25 0 0 OS 3 100 Overall Survival, % MET-Low 6 9 12 15 18 Progression-Free Survival, % 100 MET-High ORR: T = 27. 3%; P = 41. 7% 100 HR = 0. 22 (95% CI, 0. 06 - 0. 80) Log-rank P = 0. 01 75 50 Tivantinib (n = 11) Placebo (n = 12) 25 0 21 0 HR = 0. 58 (95% CI, 0. 25 - 1. 36) Log-rank P = 0. 20 75 50 25 0 3 100 Overall Survival, % Progression-Free Survival, % PFS 6 9 12 15 18 21 HR = 0. 78 (95% CI, 0. 24 - 2. 47) Log-rank P = 0. 67 75 50 25 0 0 4 8 12 16 20 24 28 Time Since Randomization, mo 32 Eng et al. , ASCO 2013
PFS and OS by MET Expression ORR: T = 54. 2%; P = 30. 0% HR = 0. 74 (95% CI, 0. 36 - 1. 52) Log-rank P = 0. 41 75 50 Tivantinib (n = 24) Placebo (n = 20) 25 0 0 OS 3 100 Overall Survival, % MET-Low 6 9 12 15 18 Progression-Free Survival, % 100 MET-High ORR: T = 27. 3%; P = 41. 7% 100 HR = 0. 22 (95% CI, 0. 06 - 0. 80) Log-rank P = 0. 01 75 50 Tivantinib (n = 11) Placebo (n = 12) 25 0 21 0 HR = 0. 58 (95% CI, 0. 25 - 1. 36) Log-rank P = 0. 20 75 50 25 0 3 100 Overall Survival, % Progression-Free Survival, % PFS 6 9 12 15 18 21 HR = 0. 78 (95% CI, 0. 24 - 2. 47) Log-rank P = 0. 67 75 50 25 0 0 4 8 12 16 20 24 28 Time Since Randomization, mo 32 Eng et al. , ASCO 2013


