61df4a6dfec470d379f680ab67366271.ppt
- Количество слайдов: 11
 Non North American Committee Meeting Philadelphia, PA Friday, June 17, 2011
Non North American Committee Meeting Philadelphia, PA Friday, June 17, 2011
 Agenda • • Current International Members International Quality Control Audit Updates International Accrual for 2011 EU Representation RTOG 0848 EORTC participation RTOG Website Changes Drug Distribution Issues You’re Invited 2
Agenda • • Current International Members International Quality Control Audit Updates International Accrual for 2011 EU Representation RTOG 0848 EORTC participation RTOG Website Changes Drug Distribution Issues You’re Invited 2
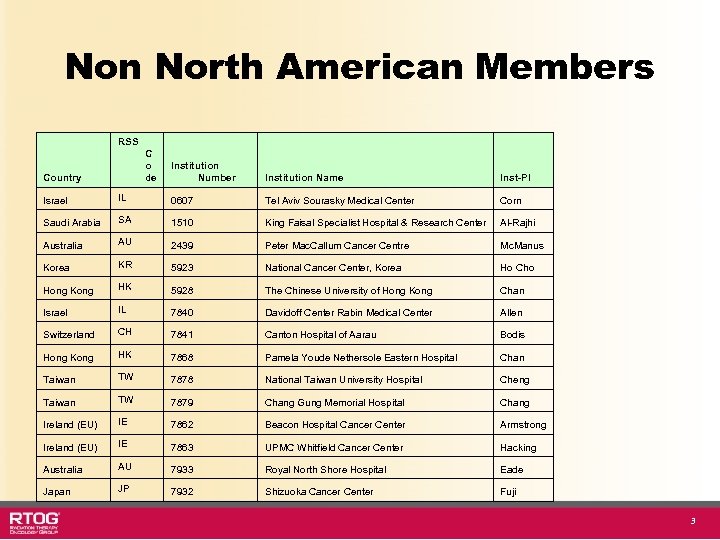 Non North American Members RSS C o de Country Institution Number Institution Name Inst-PI Israel IL 0607 Tel Aviv Sourasky Medical Center Corn Saudi Arabia SA 1510 King Faisal Specialist Hospital & Research Center Al-Rajhi Australia AU 2439 Peter Mac. Callum Cancer Centre Mc. Manus Korea KR 5923 National Cancer Center, Korea Ho Cho Hong Kong HK 5928 The Chinese University of Hong Kong Chan Israel IL 7840 Davidoff Center Rabin Medical Center Allen Switzerland CH 7841 Canton Hospital of Aarau Bodis Hong Kong HK 7868 Pamela Youde Nethersole Eastern Hospital Chan Taiwan TW 7878 National Taiwan University Hospital Cheng Taiwan TW 7879 Chang Gung Memorial Hospital Chang Ireland (EU) IE 7862 Beacon Hospital Cancer Center Armstrong Ireland (EU) IE 7863 UPMC Whitfield Cancer Center Hacking Australia AU 7933 Royal North Shore Hospital Eade Japan JP 7932 Shizuoka Cancer Center Fuji 3
Non North American Members RSS C o de Country Institution Number Institution Name Inst-PI Israel IL 0607 Tel Aviv Sourasky Medical Center Corn Saudi Arabia SA 1510 King Faisal Specialist Hospital & Research Center Al-Rajhi Australia AU 2439 Peter Mac. Callum Cancer Centre Mc. Manus Korea KR 5923 National Cancer Center, Korea Ho Cho Hong Kong HK 5928 The Chinese University of Hong Kong Chan Israel IL 7840 Davidoff Center Rabin Medical Center Allen Switzerland CH 7841 Canton Hospital of Aarau Bodis Hong Kong HK 7868 Pamela Youde Nethersole Eastern Hospital Chan Taiwan TW 7878 National Taiwan University Hospital Cheng Taiwan TW 7879 Chang Gung Memorial Hospital Chang Ireland (EU) IE 7862 Beacon Hospital Cancer Center Armstrong Ireland (EU) IE 7863 UPMC Whitfield Cancer Center Hacking Australia AU 7933 Royal North Shore Hospital Eade Japan JP 7932 Shizuoka Cancer Center Fuji 3
 Quality Control Audit Update • Presented by Elaine Boyle 4
Quality Control Audit Update • Presented by Elaine Boyle 4
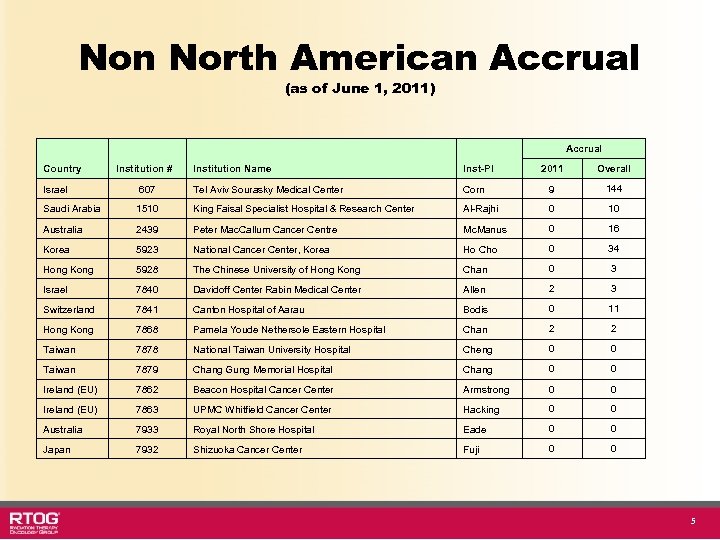 Non North American Accrual (as of June 1, 2011) Accrual Country Institution # Institution Name Inst-PI 2011 Overall Israel 607 Tel Aviv Sourasky Medical Center Corn 9 144 Saudi Arabia 1510 King Faisal Specialist Hospital & Research Center Al-Rajhi 0 10 Australia 2439 Peter Mac. Callum Cancer Centre Mc. Manus 0 16 Korea 5923 National Cancer Center, Korea Ho Cho 0 34 Hong Kong 5928 The Chinese University of Hong Kong Chan 0 3 Israel 7840 Davidoff Center Rabin Medical Center Allen 2 3 Switzerland 7841 Canton Hospital of Aarau Bodis 0 11 Hong Kong 7868 Pamela Youde Nethersole Eastern Hospital Chan 2 2 Taiwan 7878 National Taiwan University Hospital Cheng 0 0 Taiwan 7879 Chang Gung Memorial Hospital Chang 0 0 Ireland (EU) 7862 Beacon Hospital Cancer Center Armstrong 0 0 Ireland (EU) 7863 UPMC Whitfield Cancer Center Hacking 0 0 Australia 7933 Royal North Shore Hospital Eade 0 0 Japan 7932 Shizuoka Cancer Center Fuji 0 0 5
Non North American Accrual (as of June 1, 2011) Accrual Country Institution # Institution Name Inst-PI 2011 Overall Israel 607 Tel Aviv Sourasky Medical Center Corn 9 144 Saudi Arabia 1510 King Faisal Specialist Hospital & Research Center Al-Rajhi 0 10 Australia 2439 Peter Mac. Callum Cancer Centre Mc. Manus 0 16 Korea 5923 National Cancer Center, Korea Ho Cho 0 34 Hong Kong 5928 The Chinese University of Hong Kong Chan 0 3 Israel 7840 Davidoff Center Rabin Medical Center Allen 2 3 Switzerland 7841 Canton Hospital of Aarau Bodis 0 11 Hong Kong 7868 Pamela Youde Nethersole Eastern Hospital Chan 2 2 Taiwan 7878 National Taiwan University Hospital Cheng 0 0 Taiwan 7879 Chang Gung Memorial Hospital Chang 0 0 Ireland (EU) 7862 Beacon Hospital Cancer Center Armstrong 0 0 Ireland (EU) 7863 UPMC Whitfield Cancer Center Hacking 0 0 Australia 7933 Royal North Shore Hospital Eade 0 0 Japan 7932 Shizuoka Cancer Center Fuji 0 0 5
 EU Representation • The EU Clinical Trial Directive (2001) states that a clinical trial sponsor must be resident /located within the European Community. • An alternative to this is to have a nominee/representative (usually an investigator/organization) to conduct the study in the European Union. • Ireland is the first RTOG Member from the EU • RTOG is exploring the possibility of the (ICORG) Ireland Cooperative Oncology Research Group assuming this role for the EU 6
EU Representation • The EU Clinical Trial Directive (2001) states that a clinical trial sponsor must be resident /located within the European Community. • An alternative to this is to have a nominee/representative (usually an investigator/organization) to conduct the study in the European Union. • Ireland is the first RTOG Member from the EU • RTOG is exploring the possibility of the (ICORG) Ireland Cooperative Oncology Research Group assuming this role for the EU 6
 RTOG 0848 “A PHASE III TRIAL EVALUATING BOTH ERLOTINIB AND CHEMORADIATION AS ADJUVANT TREATMENT FOR PATIENTS WITH RESECTED HEAD OF PANCREAS ADENOCARCINOMA” • 0848 will open to Non North American participation via the EORTC later this year. • The MOU still pending • Non North American RTOG Members will still enroll through RTOG 7
RTOG 0848 “A PHASE III TRIAL EVALUATING BOTH ERLOTINIB AND CHEMORADIATION AS ADJUVANT TREATMENT FOR PATIENTS WITH RESECTED HEAD OF PANCREAS ADENOCARCINOMA” • 0848 will open to Non North American participation via the EORTC later this year. • The MOU still pending • Non North American RTOG Members will still enroll through RTOG 7
 New Look For RTOG Website 8
New Look For RTOG Website 8
 Drug Availability Issues • Is becoming more complicated for Canadian & Non North American sites • RTOG, CTEP/PMB & GIPC (Group International Planning Committee) are working together to overcome the current hurdles • Using your own local source of drug will likely now require a CMC certification declaring agents are identical 9
Drug Availability Issues • Is becoming more complicated for Canadian & Non North American sites • RTOG, CTEP/PMB & GIPC (Group International Planning Committee) are working together to overcome the current hurdles • Using your own local source of drug will likely now require a CMC certification declaring agents are identical 9
 You’re Invited • To attend a presentation by the study chair, Dr. Vicini regarding: – RTOG 1005 “A Phase III Trial Of Accelerated Whole Breast Irradiation With Hypofractionation Plus Concurrent Boost Versus Standard Whole Breast Irradiation Plus Sequential Boost For Early-Stage Breast Cancer” • When: Tomorrow, Saturday, June 18 at 3 PM • Where: Congress B • We hope to be able to provide more topics of interest to our Non North American & Canadian Members at each semi annual meeting 10
You’re Invited • To attend a presentation by the study chair, Dr. Vicini regarding: – RTOG 1005 “A Phase III Trial Of Accelerated Whole Breast Irradiation With Hypofractionation Plus Concurrent Boost Versus Standard Whole Breast Irradiation Plus Sequential Boost For Early-Stage Breast Cancer” • When: Tomorrow, Saturday, June 18 at 3 PM • Where: Congress B • We hope to be able to provide more topics of interest to our Non North American & Canadian Members at each semi annual meeting 10
 Regulatory Contact Information 11
Regulatory Contact Information 11


