41b3c9a2613136480237800afaff9d66.ppt
- Количество слайдов: 93
 Newer Generation Glaucoma Surgical Procedures
Newer Generation Glaucoma Surgical Procedures
 Why Do We Want or Need New Glaucoma Surgeries ? Medical therapy 1, 2 Drug cost, compliance, side effects Laser trabeculoplasty (ALT/SLT/PLT) Limited efficacy, duration, and questionable repeatibility 3
Why Do We Want or Need New Glaucoma Surgeries ? Medical therapy 1, 2 Drug cost, compliance, side effects Laser trabeculoplasty (ALT/SLT/PLT) Limited efficacy, duration, and questionable repeatibility 3
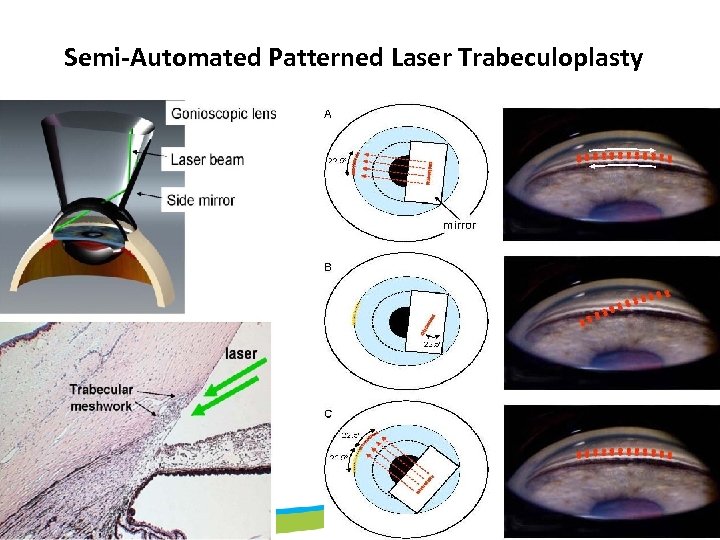
 Semi-Automated Patterned Laser Trabeculoplasty (PLT) - a novel computer-guided laser treatment For open angle glaucoma, using the PASCAL® Photocoagulator system PLT provides nearly uniform coverage of the TM using more than 1000 spots of 100 μm in diameter
Semi-Automated Patterned Laser Trabeculoplasty (PLT) - a novel computer-guided laser treatment For open angle glaucoma, using the PASCAL® Photocoagulator system PLT provides nearly uniform coverage of the TM using more than 1000 spots of 100 μm in diameter
 Semi-Automated Patterned Laser Trabeculoplasty
Semi-Automated Patterned Laser Trabeculoplasty
 – Incisional surgery. Filtering surgery(trabeculectomies and tubes) and postoperative management are technically challenging – Glaucoma surgery carries a significant risk of complications both intra-and postoperative 2 – Long-term risks remain even with a successful result – Moderately good success but gradual decay of effect 2 • • 1. Lee DA et al. Am J Health-Syst Pharm. 2005; 62: 691– 699. 2. Burr J et al. Cochrane Database of Systematic Reviews. 2007; (4): 1– 30. • 3. Stein JD et al. Curr Opin Ophthalmol. 2007; 18: 140– 145 .
– Incisional surgery. Filtering surgery(trabeculectomies and tubes) and postoperative management are technically challenging – Glaucoma surgery carries a significant risk of complications both intra-and postoperative 2 – Long-term risks remain even with a successful result – Moderately good success but gradual decay of effect 2 • • 1. Lee DA et al. Am J Health-Syst Pharm. 2005; 62: 691– 699. 2. Burr J et al. Cochrane Database of Systematic Reviews. 2007; (4): 1– 30. • 3. Stein JD et al. Curr Opin Ophthalmol. 2007; 18: 140– 145 .
 The Ideal Glaucoma Procedure • Intraocular pressure (IOP) reduction is predictable and reproducible • The ideal goal is 8 to 10 mm Hg • Circadian (and upright vs supine) variation of IOP is minimal • Adjunctive medications are not needed • Preoperative visual function is completely preserved • Long-term (>10 years) success is high (> 95%)
The Ideal Glaucoma Procedure • Intraocular pressure (IOP) reduction is predictable and reproducible • The ideal goal is 8 to 10 mm Hg • Circadian (and upright vs supine) variation of IOP is minimal • Adjunctive medications are not needed • Preoperative visual function is completely preserved • Long-term (>10 years) success is high (> 95%)
 The Ideal Glaucoma Procedure • • • Fast Easy Low cost Less frequent follow-up required Less stressful for the patient and surgeon Repeatable
The Ideal Glaucoma Procedure • • • Fast Easy Low cost Less frequent follow-up required Less stressful for the patient and surgeon Repeatable
 The Ideal Glaucoma Procedure • Rare short-or long-term complications, which can include: Hypotony • Flat chambers • Bleb leaks • Blebitis, bleb-related endophthalmitis • Bleb dysesthesia • Cataract formation, progression • Other refractive visual changes—cylinder • Choroidal effusions and hemorrhage • Failure Fantes F et al. In Rhee DJ. Glaucoma: Color Atlas and Synopsis of Clinical Ophthalmology. Mc. Graw Hill; 2003: 338– 362.
The Ideal Glaucoma Procedure • Rare short-or long-term complications, which can include: Hypotony • Flat chambers • Bleb leaks • Blebitis, bleb-related endophthalmitis • Bleb dysesthesia • Cataract formation, progression • Other refractive visual changes—cylinder • Choroidal effusions and hemorrhage • Failure Fantes F et al. In Rhee DJ. Glaucoma: Color Atlas and Synopsis of Clinical Ophthalmology. Mc. Graw Hill; 2003: 338– 362.
 Newer Procedures and Devices • Most of these lack good, large, randomized, peer-reviewed, and published trials with long-term (years) follow-up that have been repeated for confirmation • Some data are available • Some devices have been approved by the Food and Drug Administration (FDA) • Many undergo design or surgical technique modifications to improve outcomes, making them “moving targets” that are hard to evaluate but exciting to follow
Newer Procedures and Devices • Most of these lack good, large, randomized, peer-reviewed, and published trials with long-term (years) follow-up that have been repeated for confirmation • Some data are available • Some devices have been approved by the Food and Drug Administration (FDA) • Many undergo design or surgical technique modifications to improve outcomes, making them “moving targets” that are hard to evaluate but exciting to follow
 The Newer Procedures • • • Viscocanalostomy/ Deep Sclerectomy Ex-Press Shunt Tran. Ciliary filtration Trabectome GMP Eyepass SOLX Gold Shunt i. Stent Canaplasty Others
The Newer Procedures • • • Viscocanalostomy/ Deep Sclerectomy Ex-Press Shunt Tran. Ciliary filtration Trabectome GMP Eyepass SOLX Gold Shunt i. Stent Canaplasty Others
 Transciliary Filtration (TCF) • The new procedure - reduced pressure inside the eye by creating a tiny opening, or micropore, in the sclera and underlying ciliary body to allow intraocular fluid to escape the eye and drain into the eye's lymphatic system. • The micropore is created using a lightweight handheld device called a Fugo Blade (named after its inventor, Richard Fugo, MD, Ph. D)
Transciliary Filtration (TCF) • The new procedure - reduced pressure inside the eye by creating a tiny opening, or micropore, in the sclera and underlying ciliary body to allow intraocular fluid to escape the eye and drain into the eye's lymphatic system. • The micropore is created using a lightweight handheld device called a Fugo Blade (named after its inventor, Richard Fugo, MD, Ph. D)
 • The Fugo Blade generates a cloud of plasma particles around a tiny filament (about as thick as a human hair) at the end of the handpiece, and "cuts" tissue much as a laser beam does • The device leaves a clean margin around the micropore, causing no damage to the surrounding tissue • Video
• The Fugo Blade generates a cloud of plasma particles around a tiny filament (about as thick as a human hair) at the end of the handpiece, and "cuts" tissue much as a laser beam does • The device leaves a clean margin around the micropore, causing no damage to the surrounding tissue • Video
 Advantages of TCF Number of advantages over traditional surgical procedures • TCF is very quick. (On average, it takes less than eight minutes to perform. ) • It causes less tissue damage. • It has less risk of scarring and other complications. • If TCF is ineffective, it can be repeated. • It causes less bleeding during surgery. • TCF minimizes the need for post-operative care
Advantages of TCF Number of advantages over traditional surgical procedures • TCF is very quick. (On average, it takes less than eight minutes to perform. ) • It causes less tissue damage. • It has less risk of scarring and other complications. • If TCF is ineffective, it can be repeated. • It causes less bleeding during surgery. • TCF minimizes the need for post-operative care
 Effectiveness of TCF • Medisurg says nearly 1, 000 transciliary filtration procedures have been performed worldwide. • On average, patients who have received the surgery had preoperative intraocular pressure (IOP) between 30 and 44 mm. Hg. • Three months after TCF, IOP usually stabilizes at 14 to 15 mm Hg
Effectiveness of TCF • Medisurg says nearly 1, 000 transciliary filtration procedures have been performed worldwide. • On average, patients who have received the surgery had preoperative intraocular pressure (IOP) between 30 and 44 mm. Hg. • Three months after TCF, IOP usually stabilizes at 14 to 15 mm Hg
 New Glaucoma Surgical Treatment Modalities SOLX Deep. Light Gold (micro-shunt into supra-choroidal space)
New Glaucoma Surgical Treatment Modalities SOLX Deep. Light Gold (micro-shunt into supra-choroidal space)
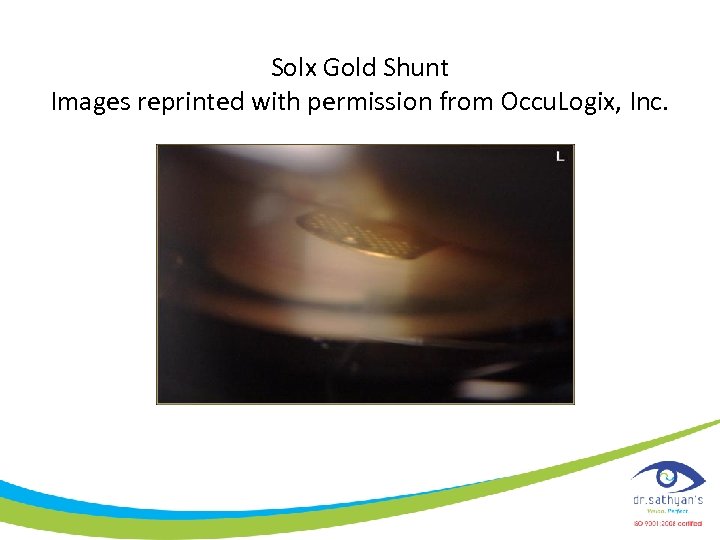
 • It is approximately 3 mm wide, 6 mm long; it is about the thickness of a human hair. • The Gold Shunt's thin structure is designed for implantation through a single micro-incision • It contains numerous micro-channels that bridge the anterior chamber and the suprachoroidal space, controlling aqueous outflow to reduce IOP.
• It is approximately 3 mm wide, 6 mm long; it is about the thickness of a human hair. • The Gold Shunt's thin structure is designed for implantation through a single micro-incision • It contains numerous micro-channels that bridge the anterior chamber and the suprachoroidal space, controlling aqueous outflow to reduce IOP.
 Solx Gold Shunt Images reprinted with permission from Occu. Logix, Inc.
Solx Gold Shunt Images reprinted with permission from Occu. Logix, Inc.
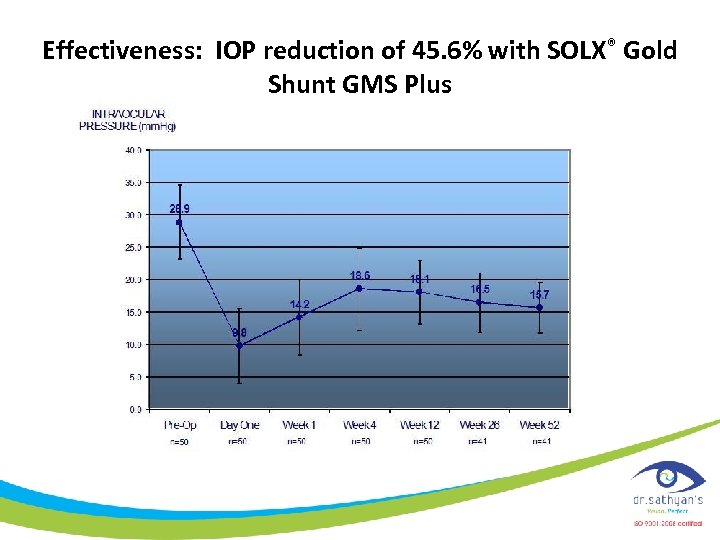 Effectiveness: IOP reduction of 45. 6% with SOLX® Gold Shunt GMS Plus
Effectiveness: IOP reduction of 45. 6% with SOLX® Gold Shunt GMS Plus
 Trabectome™(Ab Interno Trabeculectomy) • FDA approved • 1. 6 -mm clear corneal incision • Micro-electrocautery and aspiration to ablate and remove trabecular meshwork (TM) and inner wall of Schlemm’s Canal (SC) for 30°to 60° • Irrigation and small amount of cohesive viscoelastic help maintain the anterior chamber • Minckler DS et al. Trans Am Ophthalmol Soc. 2006; 104: 40– 50.
Trabectome™(Ab Interno Trabeculectomy) • FDA approved • 1. 6 -mm clear corneal incision • Micro-electrocautery and aspiration to ablate and remove trabecular meshwork (TM) and inner wall of Schlemm’s Canal (SC) for 30°to 60° • Irrigation and small amount of cohesive viscoelastic help maintain the anterior chamber • Minckler DS et al. Trans Am Ophthalmol Soc. 2006; 104: 40– 50.
 Trabectome™(Ab Interno Trabeculectomy) Video.
Trabectome™(Ab Interno Trabeculectomy) Video.
 • Trabectome™(Ab Interno Trabeculectomy)Mean IOP ±SD (mm Hg) • Preop IOP baseline 25. 2 +7. 4; N=204 • Postop IOP 12 months • (Mean % decrease from preop baseline) 16. 7 +2. 6; n=43 (– 47%) • Postop IOP 36 months • (Mean % decrease from preop baseline) 17. 0 +4. 1; n=5 (– 42%)
• Trabectome™(Ab Interno Trabeculectomy)Mean IOP ±SD (mm Hg) • Preop IOP baseline 25. 2 +7. 4; N=204 • Postop IOP 12 months • (Mean % decrease from preop baseline) 16. 7 +2. 6; n=43 (– 47%) • Postop IOP 36 months • (Mean % decrease from preop baseline) 17. 0 +4. 1; n=5 (– 42%)
 Trabectome™(Ab Interno Trabeculectomy) Complications • Back-bleeding from collector channelso 100% clearing over a few days • No long-term vision-threatening occurrences such aso. Prolonged hypotony • Choroidal effusion • Choroidal hemorrhage • Clinical failure resulted in trabeculectomy in 9 of 204 eyes and shunt installation in 3 of 204 eyes • Minckler DS et al. Paper #24 presented at: 17 th. Annual Meeting of the American Glaucoma Society; March 2, 2007; San Francisco, CA.
Trabectome™(Ab Interno Trabeculectomy) Complications • Back-bleeding from collector channelso 100% clearing over a few days • No long-term vision-threatening occurrences such aso. Prolonged hypotony • Choroidal effusion • Choroidal hemorrhage • Clinical failure resulted in trabeculectomy in 9 of 204 eyes and shunt installation in 3 of 204 eyes • Minckler DS et al. Paper #24 presented at: 17 th. Annual Meeting of the American Glaucoma Society; March 2, 2007; San Francisco, CA.
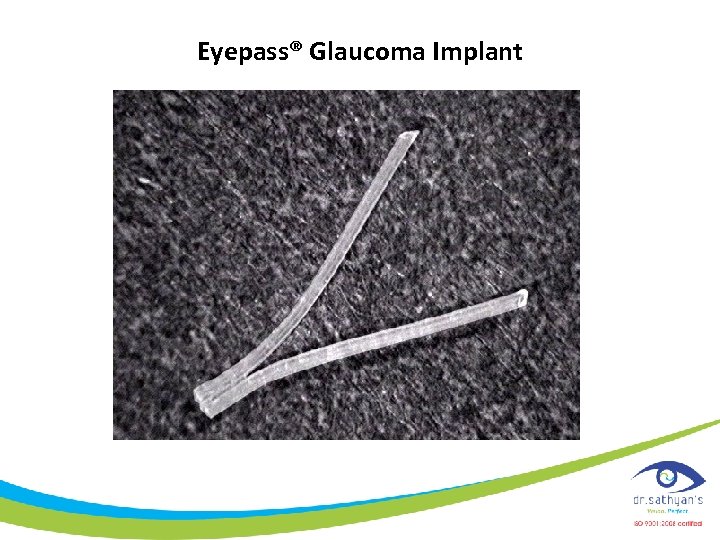 Eyepass® Glaucoma Implant
Eyepass® Glaucoma Implant
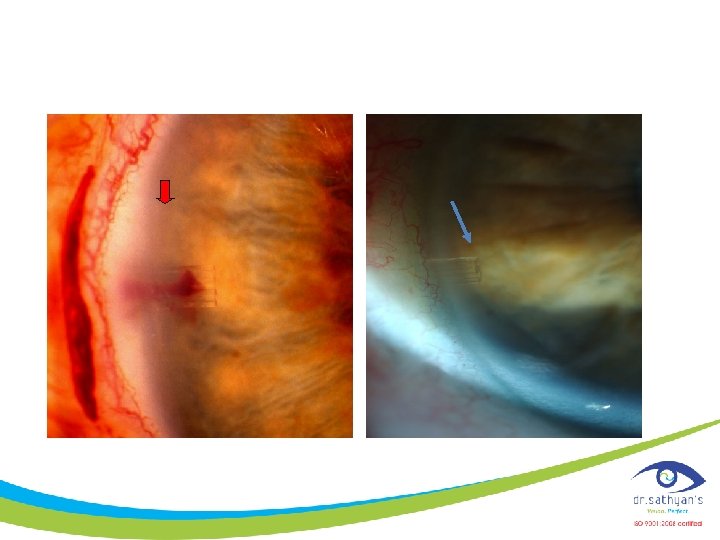
 GMP Eyepass
GMP Eyepass
 Summary and Conclusions • When medical therapy does not adequately control IOP • Ex-PRESS™mini-shunt under a scleral flap • Trabectome™ • Suture canaloplasty • Others
Summary and Conclusions • When medical therapy does not adequately control IOP • Ex-PRESS™mini-shunt under a scleral flap • Trabectome™ • Suture canaloplasty • Others
 Summary and Conclusions • We don’t yet have the perfect glaucoma procedure • Some of these newer procedures appear to have better safety profiles • Until the perfect procedure arrives, perhaps an alternate paradigm to consider may be to use a safer and more predictable procedure and complement it with the adjunctive use of medications for those patients who need low IOPs, but this will need further study first
Summary and Conclusions • We don’t yet have the perfect glaucoma procedure • Some of these newer procedures appear to have better safety profiles • Until the perfect procedure arrives, perhaps an alternate paradigm to consider may be to use a safer and more predictable procedure and complement it with the adjunctive use of medications for those patients who need low IOPs, but this will need further study first
 Thank you
Thank you
 Non – penetrating - terminology • viscocanolostomy – Restores the normal outflow pathway • Deep sclerectomy – Aimed at increasing the outflow at the subconjunctival space
Non – penetrating - terminology • viscocanolostomy – Restores the normal outflow pathway • Deep sclerectomy – Aimed at increasing the outflow at the subconjunctival space
 Ex. Press Shunt • • 3 mm long 400 micron diam; 50 micron lumen Stainless steel Original for impant thro’ limbus Currently being evaluated under scl flap No PI and no removal of Scl tissue South African experience 24 pts Mean IOP decrease 45% at 2 yrs
Ex. Press Shunt • • 3 mm long 400 micron diam; 50 micron lumen Stainless steel Original for impant thro’ limbus Currently being evaluated under scl flap No PI and no removal of Scl tissue South African experience 24 pts Mean IOP decrease 45% at 2 yrs
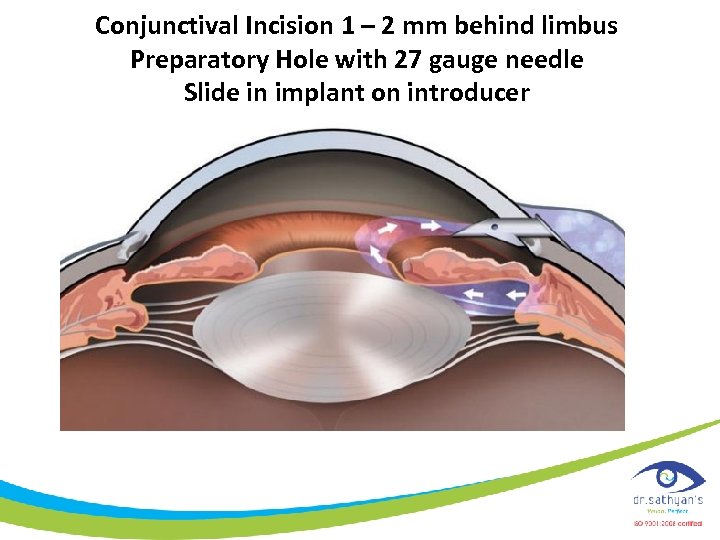 Conjunctival Incision 1 – 2 mm behind limbus Preparatory Hole with 27 gauge needle Slide in implant on introducer
Conjunctival Incision 1 – 2 mm behind limbus Preparatory Hole with 27 gauge needle Slide in implant on introducer
 Express Shunt Video
Express Shunt Video
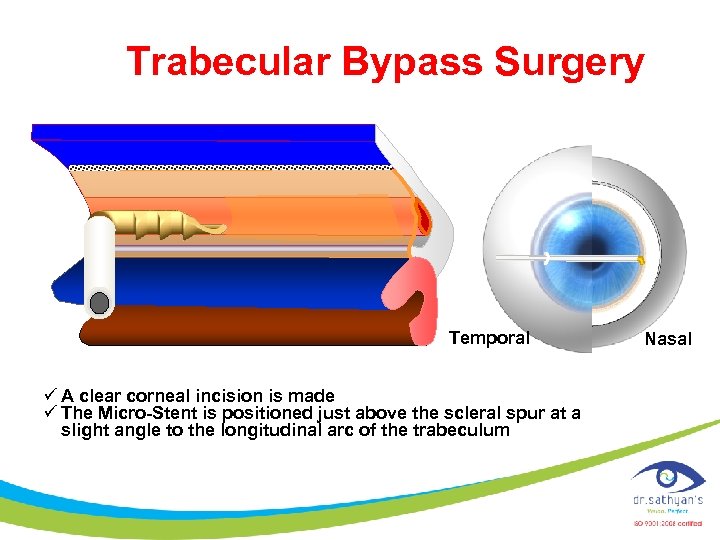 Trabecular Bypass Surgery Temporal ü A clear corneal incision is made ü The Micro-Stent is positioned just above the scleral spur at a slight angle to the longitudinal arc of the trabeculum Nasal
Trabecular Bypass Surgery Temporal ü A clear corneal incision is made ü The Micro-Stent is positioned just above the scleral spur at a slight angle to the longitudinal arc of the trabeculum Nasal
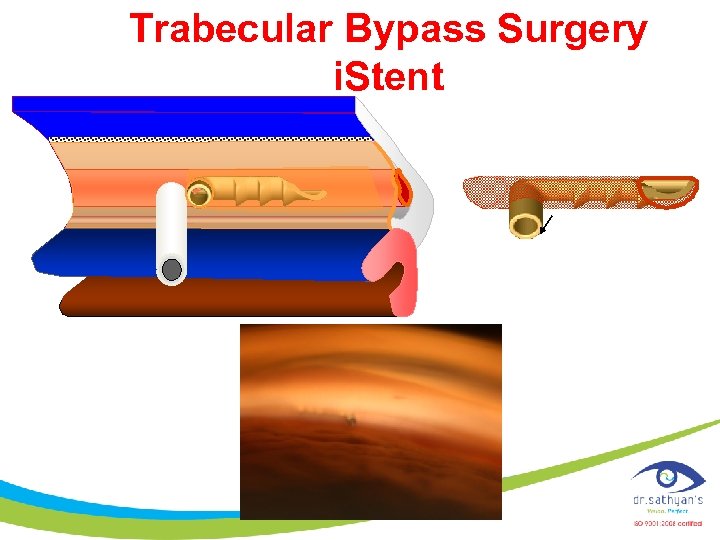 Trabecular Bypass Surgery i. Stent
Trabecular Bypass Surgery i. Stent
 Aqua. Lase Liquefaction Device
Aqua. Lase Liquefaction Device
 The Power Wash
The Power Wash
 Aqua. Lase Function
Aqua. Lase Function
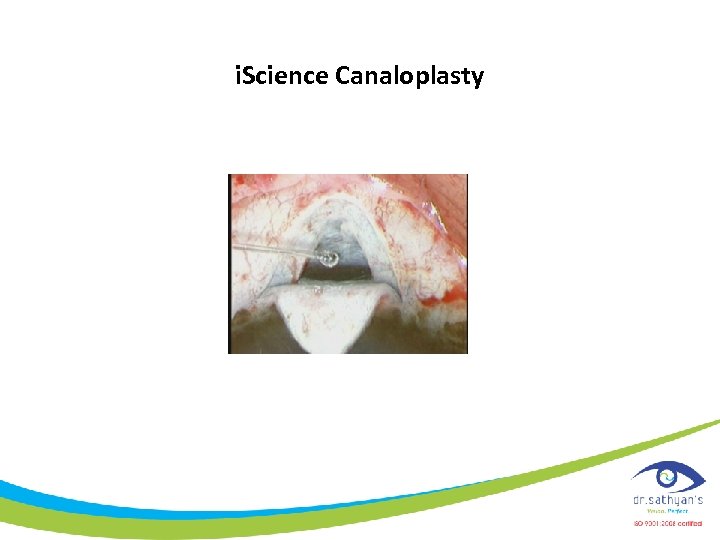 i. Science Canaloplasty
i. Science Canaloplasty
 7$ Canaloplasty
7$ Canaloplasty
 Only Treatable Risk Factor - IOP • Medical • Laser • Surgery
Only Treatable Risk Factor - IOP • Medical • Laser • Surgery
 Surgical Management • Penetrating – Trabeculectomy (anti metabolites, releasable sutures, laser suturolysis) • NPGS – Viscocanolostomy, NPDS (substituted with collagen implant and lasers)
Surgical Management • Penetrating – Trabeculectomy (anti metabolites, releasable sutures, laser suturolysis) • NPGS – Viscocanolostomy, NPDS (substituted with collagen implant and lasers)
 Glaucoma Surgery vs. Cataract Surgery
Glaucoma Surgery vs. Cataract Surgery
 Glaucoma Surgery • Not rewarding for general ophthalmologist – takes longer than cataract – much more post operative care than cataract – longer visual rehabilitation • Unexpected surgical outcome • Negative social marketing
Glaucoma Surgery • Not rewarding for general ophthalmologist – takes longer than cataract – much more post operative care than cataract – longer visual rehabilitation • Unexpected surgical outcome • Negative social marketing
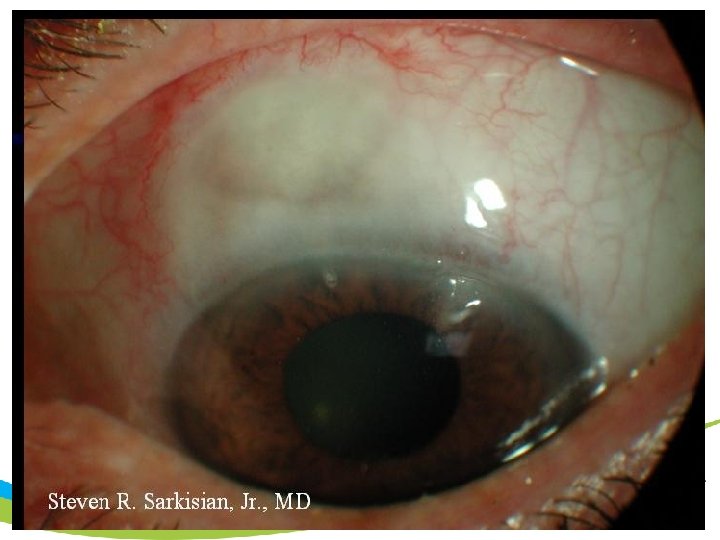
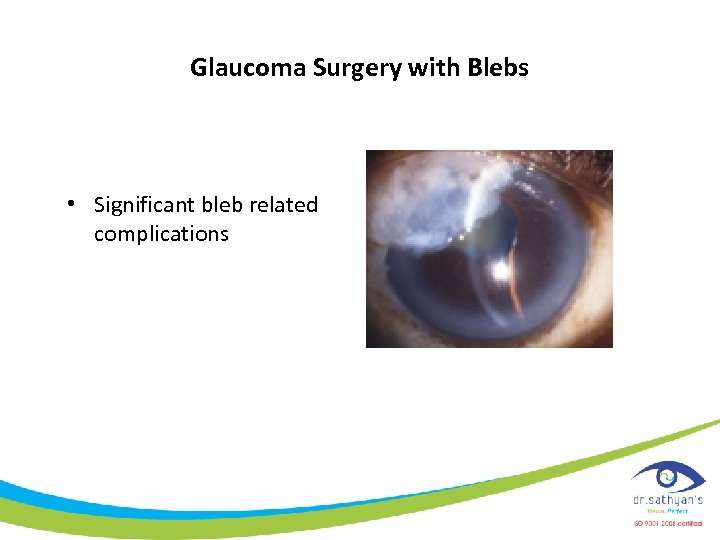 Glaucoma Surgery with Blebs • Significant bleb related complications
Glaucoma Surgery with Blebs • Significant bleb related complications
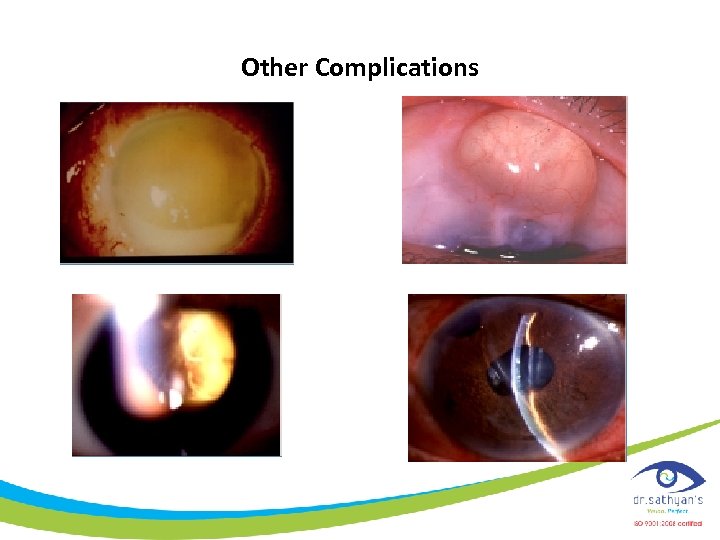 Other Complications
Other Complications
 Blebless Glaucoma Surgery • • • Viscocanolostomy NPDS Express Shunt Glaukos i. Stent Canaloplasty Aqualase power wash
Blebless Glaucoma Surgery • • • Viscocanolostomy NPDS Express Shunt Glaukos i. Stent Canaloplasty Aqualase power wash
 NPGS Video
NPGS Video
 NPGS
NPGS
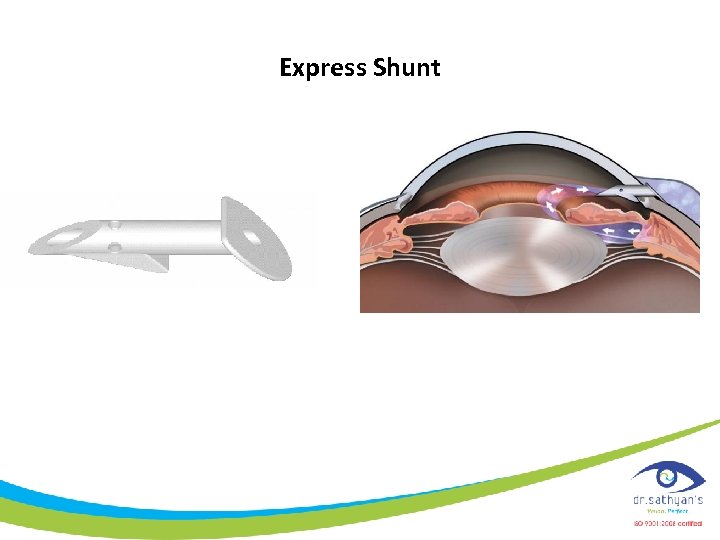 Express Shunt
Express Shunt
 Express Shunt
Express Shunt
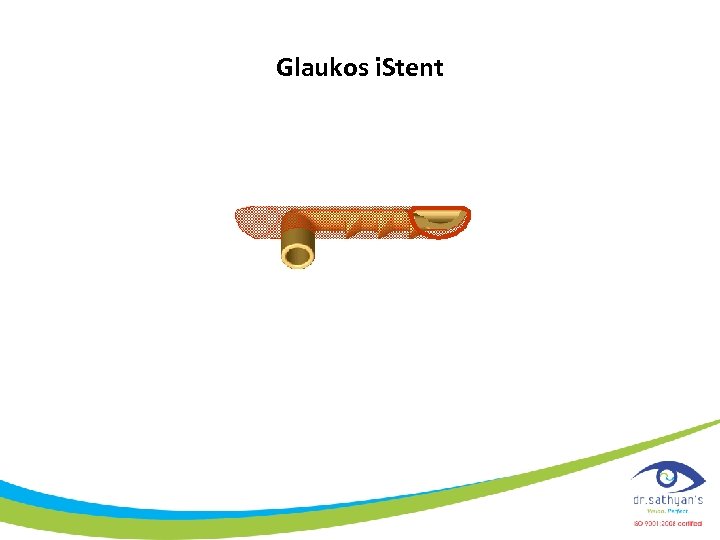 Glaukos i. Stent
Glaukos i. Stent
 Glaukos i. Stent Video
Glaukos i. Stent Video
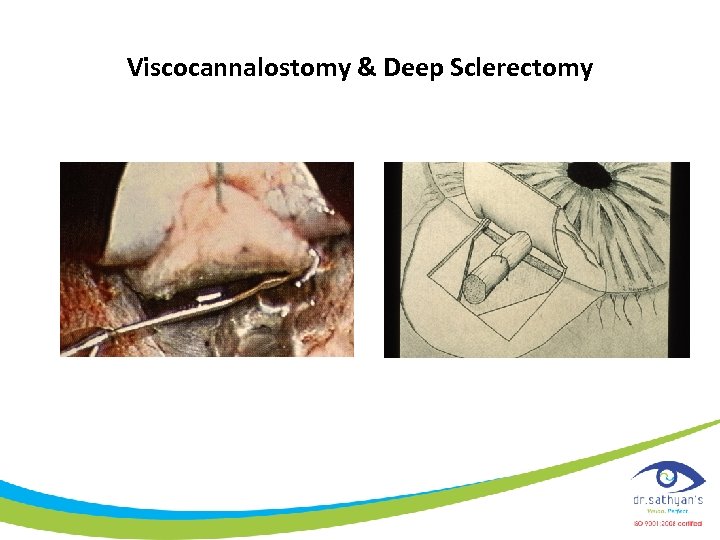 Viscocannalostomy & Deep Sclerectomy
Viscocannalostomy & Deep Sclerectomy
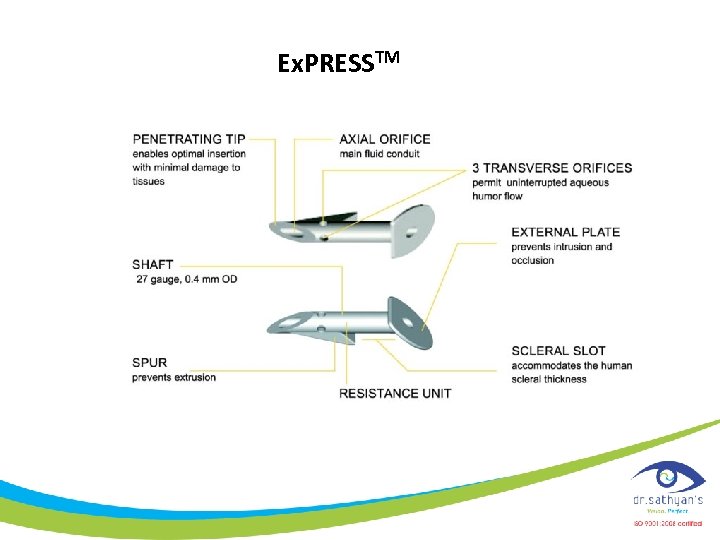 Ex. PRESSTM
Ex. PRESSTM
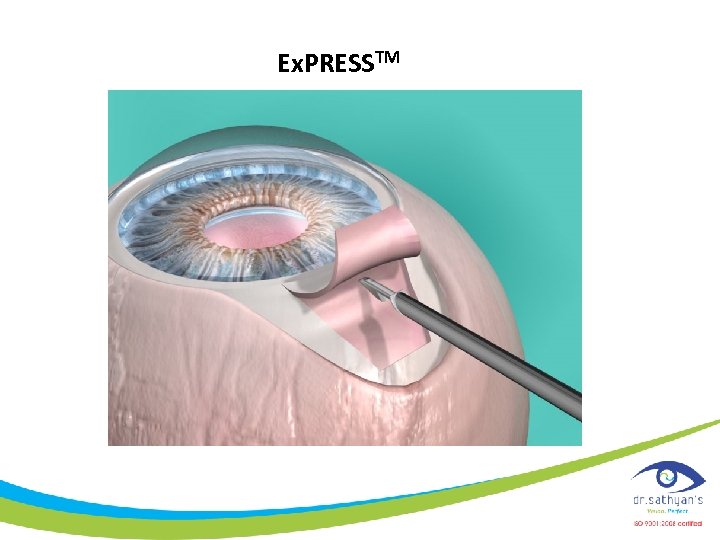 Ex. PRESSTM
Ex. PRESSTM
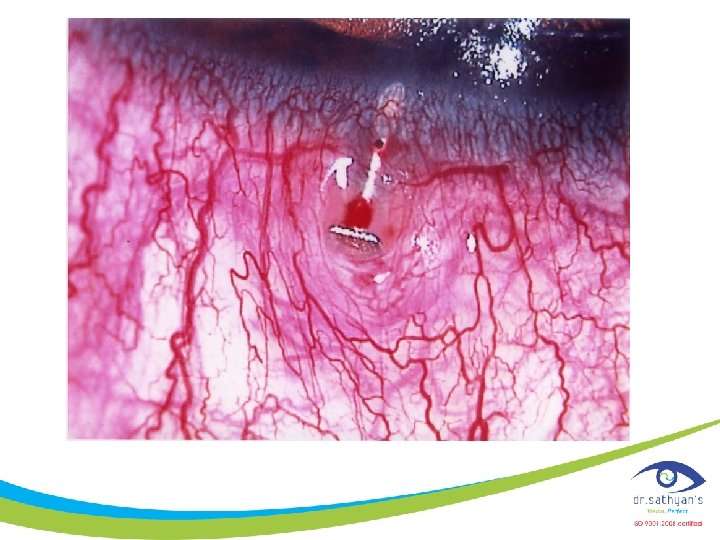
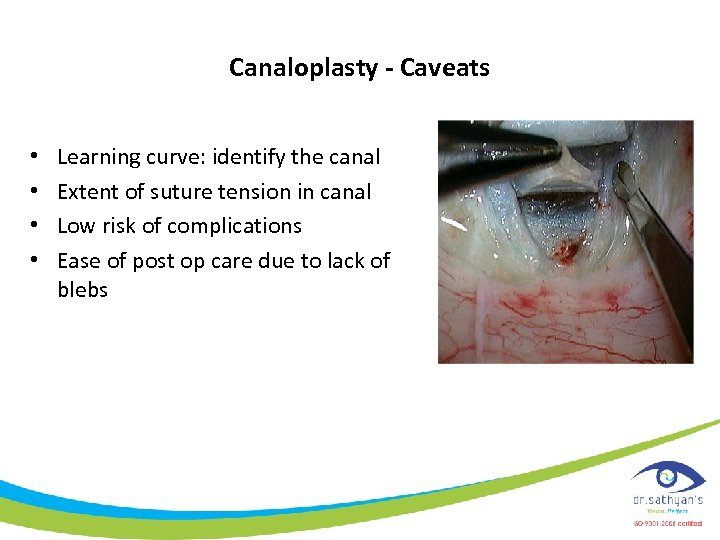 Canaloplasty - Caveats • • Learning curve: identify the canal Extent of suture tension in canal Low risk of complications Ease of post op care due to lack of blebs
Canaloplasty - Caveats • • Learning curve: identify the canal Extent of suture tension in canal Low risk of complications Ease of post op care due to lack of blebs
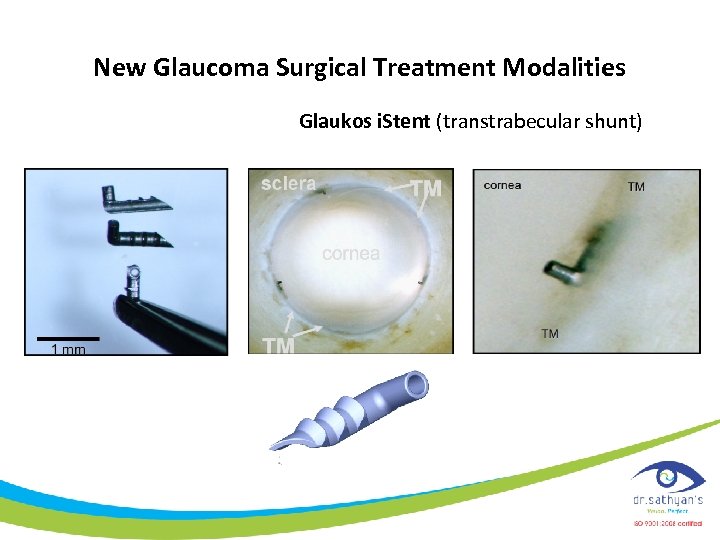 New Glaucoma Surgical Treatment Modalities Glaukos i. Stent (transtrabecular shunt)
New Glaucoma Surgical Treatment Modalities Glaukos i. Stent (transtrabecular shunt)
 Revisiting angle surgery for COAG • Good news: several new options being explored • Bad news: Data are preliminary with short follow-up & no RCTs comparing techniques “Whatever you do in glaucoma surgery will work some of the time!” Robert Shaffer (circa 1950)
Revisiting angle surgery for COAG • Good news: several new options being explored • Bad news: Data are preliminary with short follow-up & no RCTs comparing techniques “Whatever you do in glaucoma surgery will work some of the time!” Robert Shaffer (circa 1950)
 Indications And Perspectives
Indications And Perspectives
 Canaloplasty • Re-establish circumferential flow from the restored canal of Schlemm to the collectors: – Achieve physiologic control of IOP – Without requiring a bleb – Without developing bleb related post operative problems – Uses a standard surgical nonpenetrating approach
Canaloplasty • Re-establish circumferential flow from the restored canal of Schlemm to the collectors: – Achieve physiologic control of IOP – Without requiring a bleb – Without developing bleb related post operative problems – Uses a standard surgical nonpenetrating approach
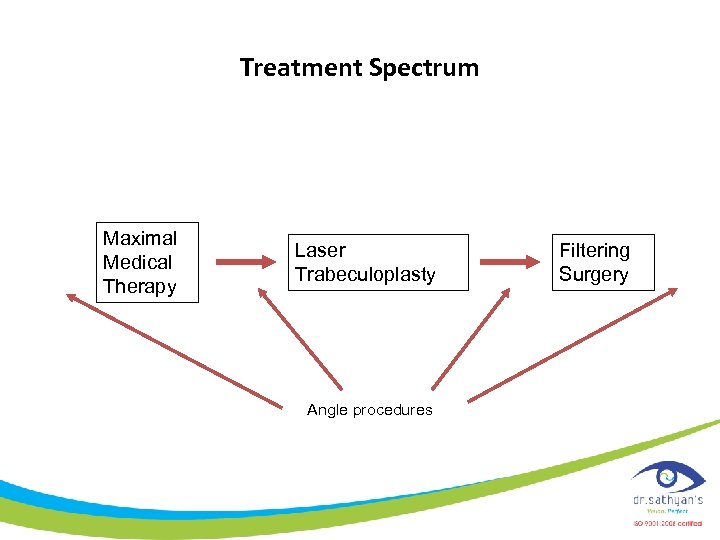 Treatment Spectrum Maximal Medical Therapy Laser Trabeculoplasty Angle procedures Filtering Surgery
Treatment Spectrum Maximal Medical Therapy Laser Trabeculoplasty Angle procedures Filtering Surgery
 Background • Glaucoma drainage devices (GDDs) create an alternate aqueous pathway from the anterior chamber (AC) by channeling aqueous out of the eye through a tube to a subconjunctival bleb or to the suprachoroidal space. • GDDs are being used more frequently in Medication non responders and failed trabeculectomy operations.
Background • Glaucoma drainage devices (GDDs) create an alternate aqueous pathway from the anterior chamber (AC) by channeling aqueous out of the eye through a tube to a subconjunctival bleb or to the suprachoroidal space. • GDDs are being used more frequently in Medication non responders and failed trabeculectomy operations.
 History • The earliest attempt to drain fluid dates back to 1906 when Rollet and Moreau implanted a silk thread connecting the anterior chamber to the subconjunctival space. • Since that time, additional unsuccessful attempts were made, including insertion of a polythene tube by Epstein in 1959 and a silicone tube by Mac. Donald and Pearce in 1965. These operations failed because of excessive scar formation near the limbus, seton migration, and conjunctival erosion.
History • The earliest attempt to drain fluid dates back to 1906 when Rollet and Moreau implanted a silk thread connecting the anterior chamber to the subconjunctival space. • Since that time, additional unsuccessful attempts were made, including insertion of a polythene tube by Epstein in 1959 and a silicone tube by Mac. Donald and Pearce in 1965. These operations failed because of excessive scar formation near the limbus, seton migration, and conjunctival erosion.
 Molteno • In 1969, Molteno introduced concept that a large surface area was needed to disperse the aqueous beneath the conjunctiva. • Inserted a short acrylic tube that was attached to a thin acrylic plate. The plate was sutured to the sclera close to the limbus. • Most of the operations failed after the first 3 -6 months because of plate exposure, tube erosion, and scar formation.
Molteno • In 1969, Molteno introduced concept that a large surface area was needed to disperse the aqueous beneath the conjunctiva. • Inserted a short acrylic tube that was attached to a thin acrylic plate. The plate was sutured to the sclera close to the limbus. • Most of the operations failed after the first 3 -6 months because of plate exposure, tube erosion, and scar formation.
 Molteno • In 1973, Molteno improved device • He introduced the Molteno implant with a long silicone tube attached to a large end plate placed 9 -10 mm posterior to the limbus. • All the currently available GDDs are based on this concept by Molteno.
Molteno • In 1973, Molteno improved device • He introduced the Molteno implant with a long silicone tube attached to a large end plate placed 9 -10 mm posterior to the limbus. • All the currently available GDDs are based on this concept by Molteno.
 Ahmed and Baerveldt • In 1993, Ahmed introduced the Ahmed glaucoma valve (AGV), a pressure-sensitive, unidirectional valve that is designed to open when the intraocular pressure (IOP) is 8 mm Hg. • In 1981, Molteno introduced the double plate implant with a surface area of 270 mm 2. • In 1992, Baerveldt introduced a nonvalved silicone tube attached to a large barium-impregnated silicone plate with a surface area of 250 mm 2, 350 mm 2, or 500 mm 2.
Ahmed and Baerveldt • In 1993, Ahmed introduced the Ahmed glaucoma valve (AGV), a pressure-sensitive, unidirectional valve that is designed to open when the intraocular pressure (IOP) is 8 mm Hg. • In 1981, Molteno introduced the double plate implant with a surface area of 270 mm 2. • In 1992, Baerveldt introduced a nonvalved silicone tube attached to a large barium-impregnated silicone plate with a surface area of 250 mm 2, 350 mm 2, or 500 mm 2.
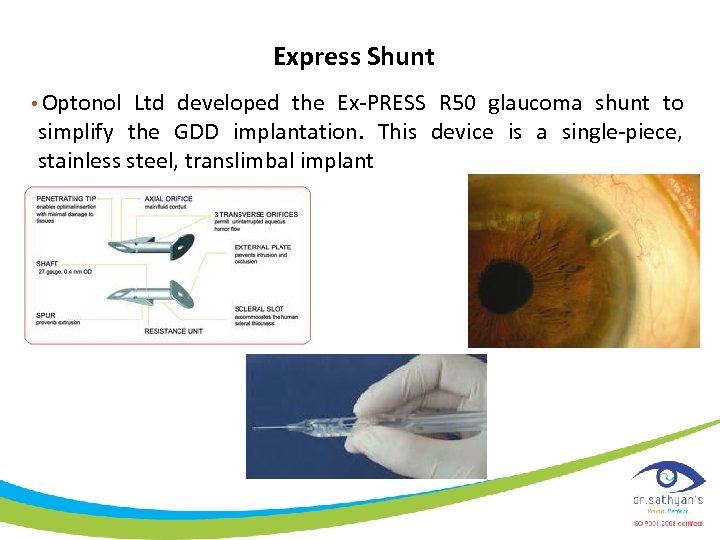 Express Shunt • Optonol Ltd developed the Ex-PRESS R 50 glaucoma shunt to simplify the GDD implantation. This device is a single-piece, stainless steel, translimbal implant
Express Shunt • Optonol Ltd developed the Ex-PRESS R 50 glaucoma shunt to simplify the GDD implantation. This device is a single-piece, stainless steel, translimbal implant
 Glaucoma Drainage Implant GDDs can be classified into • With no resistance, • Those with variable resistance to aqueous outflow.
Glaucoma Drainage Implant GDDs can be classified into • With no resistance, • Those with variable resistance to aqueous outflow.
 GDDs with no resistance • These GDDs consist of a silicone tube attached to an end plate that acts as a surface for bleb formation. Unless the operation is modified with a stent and ripcord technique, • These implants are associated (in the early postoperative period) with a high incidence of overfiltration secondary to no aqueous outflow resistance • This can lead to hypotony, shallow-to-flat anterior chambers, and choroidal effusions.
GDDs with no resistance • These GDDs consist of a silicone tube attached to an end plate that acts as a surface for bleb formation. Unless the operation is modified with a stent and ripcord technique, • These implants are associated (in the early postoperative period) with a high incidence of overfiltration secondary to no aqueous outflow resistance • This can lead to hypotony, shallow-to-flat anterior chambers, and choroidal effusions.
 • The single-plate Molteno implant is a silicone tube attached to a 135 mm 2 polypropylene end plate. • The double-plate Molteno (DPM) is the same as the single-plate Molteno except that a second end plate is attached to the right or left of the original end plate, thus doubling its surface area. It requires a 2 -quadrant dissection.
• The single-plate Molteno implant is a silicone tube attached to a 135 mm 2 polypropylene end plate. • The double-plate Molteno (DPM) is the same as the single-plate Molteno except that a second end plate is attached to the right or left of the original end plate, thus doubling its surface area. It requires a 2 -quadrant dissection.
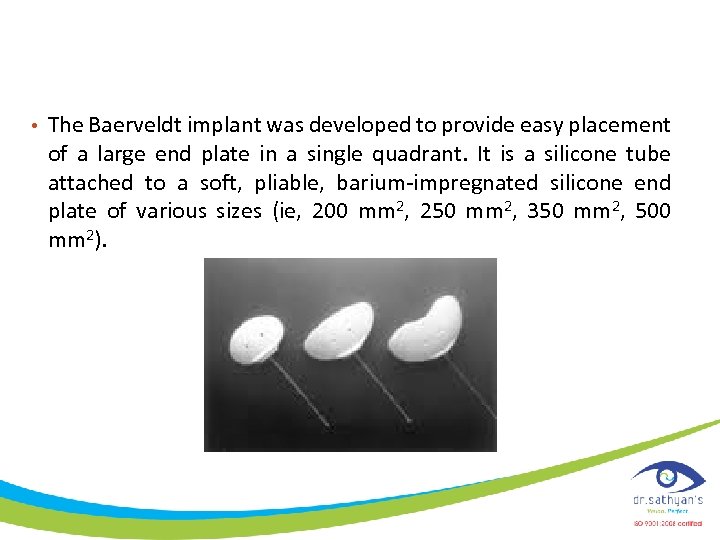 • The Baerveldt implant was developed to provide easy placement of a large end plate in a single quadrant. It is a silicone tube attached to a soft, pliable, barium-impregnated silicone end plate of various sizes (ie, 200 mm 2, 250 mm 2, 350 mm 2, 500 mm 2).
• The Baerveldt implant was developed to provide easy placement of a large end plate in a single quadrant. It is a silicone tube attached to a soft, pliable, barium-impregnated silicone end plate of various sizes (ie, 200 mm 2, 250 mm 2, 350 mm 2, 500 mm 2).
 AADI • Aurolab Aqueous Drainage Implant is a non resistant tube device based on Baerveldt implant. • It reduces intra ocular pressure by draining aqueous from anterior chamber into sub-conjunctival space formed around base of the implant.
AADI • Aurolab Aqueous Drainage Implant is a non resistant tube device based on Baerveldt implant. • It reduces intra ocular pressure by draining aqueous from anterior chamber into sub-conjunctival space formed around base of the implant.
 • The Ex-PRESS R 50 implant • 3 -mm long tube with a 400 µm (27 gauge) external diameter and a 50 µm internal diameter. The penetrating tip is beveled with 3 side orifices and a spurlike projection to prevent extrusion. • The flange is a small (< 1 mm 2) disc like plate that prevents the device from being inserted too deeply. Both the flange and the spurlike projection are angled to conform to the sclera with the distance between them being equal to the scleral thickness at the site of proper implantation.
• The Ex-PRESS R 50 implant • 3 -mm long tube with a 400 µm (27 gauge) external diameter and a 50 µm internal diameter. The penetrating tip is beveled with 3 side orifices and a spurlike projection to prevent extrusion. • The flange is a small (< 1 mm 2) disc like plate that prevents the device from being inserted too deeply. Both the flange and the spurlike projection are angled to conform to the sclera with the distance between them being equal to the scleral thickness at the site of proper implantation.
 GDDs with set resistance • Even though manufacturers claim that these devices contain true valves, independent examinations of the flow characteristics for these devices suggest a wide divergence between observed function and the manufacturers' claims. • The valves appear not to close after initial opening in perfusion tests at physiological flow rates. • AGV has the lowest incidence of hypotony of all GDDs.
GDDs with set resistance • Even though manufacturers claim that these devices contain true valves, independent examinations of the flow characteristics for these devices suggest a wide divergence between observed function and the manufacturers' claims. • The valves appear not to close after initial opening in perfusion tests at physiological flow rates. • AGV has the lowest incidence of hypotony of all GDDs.
 Ahmed Glaucoma Valve • The AGV is a silicone tube connected to a silicone sheet valve held in a polypropylene body. • The end plate measures 185 mm 2 (16 mm long X 13 mm wide X 1. 9 mm thick). The valve consists of thin silicone elastomer membranes (8 mm long X 7 mm wide) that create a venturishaped chamber. • The inlet cross-section of the chamber is wider than the outlet (Bernoulli principle), with a resultant pressure differential between the anterior chamber and the bleb. The valve is designed to open when the IOP is 8 mm Hg. ca
Ahmed Glaucoma Valve • The AGV is a silicone tube connected to a silicone sheet valve held in a polypropylene body. • The end plate measures 185 mm 2 (16 mm long X 13 mm wide X 1. 9 mm thick). The valve consists of thin silicone elastomer membranes (8 mm long X 7 mm wide) that create a venturishaped chamber. • The inlet cross-section of the chamber is wider than the outlet (Bernoulli principle), with a resultant pressure differential between the anterior chamber and the bleb. The valve is designed to open when the IOP is 8 mm Hg. ca
 GDDs with variable resistance • These devices may be modifications of the original Molteno implant and the Baerveldt implant that attempt to incorporate a resistance mechanism dependent on tissue apposition to limit flow. Because the force of tissue apposition is variable, these devices do not function as true valves, and IOP levels remain unpredictable.
GDDs with variable resistance • These devices may be modifications of the original Molteno implant and the Baerveldt implant that attempt to incorporate a resistance mechanism dependent on tissue apposition to limit flow. Because the force of tissue apposition is variable, these devices do not function as true valves, and IOP levels remain unpredictable.
 Indications • Neovascular glaucoma • PKP with glaucoma • Retinal detachment surgery with glaucoma • ICE syndrome • Traumatic glaucoma • Uveitic glaucoma • Open-angle glaucoma with failed trabeculectomy • Epithelial downgrowth • Refractory infantile glaucoma • Contact lens wearers who need glaucoma filtration surgery
Indications • Neovascular glaucoma • PKP with glaucoma • Retinal detachment surgery with glaucoma • ICE syndrome • Traumatic glaucoma • Uveitic glaucoma • Open-angle glaucoma with failed trabeculectomy • Epithelial downgrowth • Refractory infantile glaucoma • Contact lens wearers who need glaucoma filtration surgery
 Contraindications • • • GDDs may have a complicated postoperative course. Excessive scar tissue Thin Conjunctiva Pre existing scleral disease Borderline corneal endothelial function is a relative contraindication for anterior chamber placement of a tube.
Contraindications • • • GDDs may have a complicated postoperative course. Excessive scar tissue Thin Conjunctiva Pre existing scleral disease Borderline corneal endothelial function is a relative contraindication for anterior chamber placement of a tube.
 SURGICAL TECHNIQUE You Tube Link. Express-Shunt Dr Sathyan AADI Dr. Sathyan
SURGICAL TECHNIQUE You Tube Link. Express-Shunt Dr Sathyan AADI Dr. Sathyan
 POST OP • Hypotensive phase • This phase lasts from day 1 to 3 -4 weeks following the operation. • During this phase, the bleb appears to be diffuse and thickwalled with minimally engorged blood vessels. • The IOP is low (ie, from 2 -3 mm Hg to 10 -12 mm Hg). (ANGRY LOW BLEB)
POST OP • Hypotensive phase • This phase lasts from day 1 to 3 -4 weeks following the operation. • During this phase, the bleb appears to be diffuse and thickwalled with minimally engorged blood vessels. • The IOP is low (ie, from 2 -3 mm Hg to 10 -12 mm Hg). (ANGRY LOW BLEB)
 Hypertensive phase • This phase begins 3 -6 weeks after the operation and lasts for 4 -6 months. • The bleb becomes visibly inflamed and dome shaped and, in some cases, is associated with increased IOP to greater than 30 mm Hg. • The incidence of the hypertensive phase appears to be increased with the AGV as compared to the Baerveldt implant • This increased incidence could be explained because of the larger surface area of the Baerveldt implant or because of different biomaterials being used in the different implants.
Hypertensive phase • This phase begins 3 -6 weeks after the operation and lasts for 4 -6 months. • The bleb becomes visibly inflamed and dome shaped and, in some cases, is associated with increased IOP to greater than 30 mm Hg. • The incidence of the hypertensive phase appears to be increased with the AGV as compared to the Baerveldt implant • This increased incidence could be explained because of the larger surface area of the Baerveldt implant or because of different biomaterials being used in the different implants.
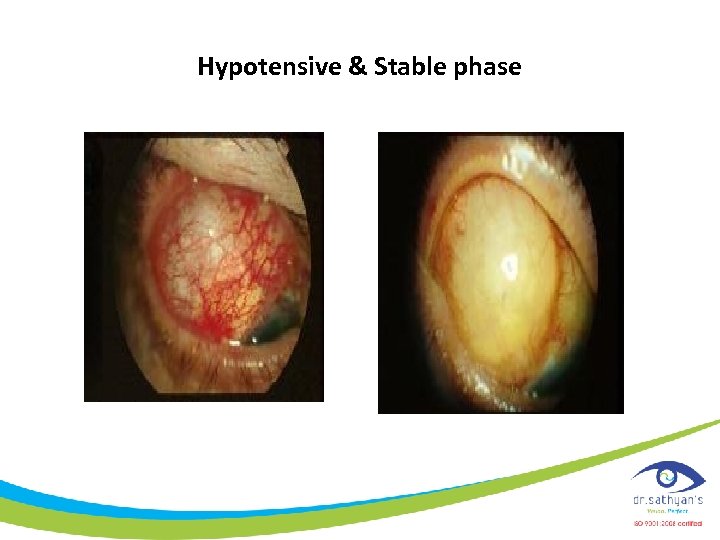
 Stable phase • Following the hypertensive phase, this phase is characterized by stabilization of the IOP in the mid-to-high teens. • At this time, the blebs are supposed to maintain IOP for the rest of the patient's lifetime; however, in reality, more than 50% of blebs fail by the end of 5 years. • The bleb appears as a thick-walled, dome-shaped, elevated area overlying the end plate with no associated inflammation.
Stable phase • Following the hypertensive phase, this phase is characterized by stabilization of the IOP in the mid-to-high teens. • At this time, the blebs are supposed to maintain IOP for the rest of the patient's lifetime; however, in reality, more than 50% of blebs fail by the end of 5 years. • The bleb appears as a thick-walled, dome-shaped, elevated area overlying the end plate with no associated inflammation.
 Hypotensive & Stable phase
Hypotensive & Stable phase
 Complicationsc 1. Hypotony 2. Tube obstruction 3. Tube retraction 4. Diplopia 5. Corneal decompensation 6. Graft failure 7. Tube and end plate exposure 8. Suprachoroidal hemorrhage 9. Late failure 10. Endophthalmitis
Complicationsc 1. Hypotony 2. Tube obstruction 3. Tube retraction 4. Diplopia 5. Corneal decompensation 6. Graft failure 7. Tube and end plate exposure 8. Suprachoroidal hemorrhage 9. Late failure 10. Endophthalmitis
 OUTCOME & PROGNOSIS • Overall, both the success rates and the complication rates following any glaucoma drainage device (GDD) implantation are similar. • The choice of the GDD in the treatment of recalcitrant glaucoma depends upon the patient and the surgeon.
OUTCOME & PROGNOSIS • Overall, both the success rates and the complication rates following any glaucoma drainage device (GDD) implantation are similar. • The choice of the GDD in the treatment of recalcitrant glaucoma depends upon the patient and the surgeon.
 • Currently, 5 GDDs are available. • The Ahmed glaucoma valve (AGV) and the Krupin implant offer resistance to the outflow in the form of a sheet valve and a slit valve, respectively. • The Molteno implant and the Baerveldt implant offer no resistance to the outflow and may lead to hypotony; however, the problem can be overcome using the ripcord technique. • Long-term success and complications associated with the Ex. PRESS shunt have yet to be demonstrated. No peer-reviewed publications concerning the SOLX shunt are available at this time.
• Currently, 5 GDDs are available. • The Ahmed glaucoma valve (AGV) and the Krupin implant offer resistance to the outflow in the form of a sheet valve and a slit valve, respectively. • The Molteno implant and the Baerveldt implant offer no resistance to the outflow and may lead to hypotony; however, the problem can be overcome using the ripcord technique. • Long-term success and complications associated with the Ex. PRESS shunt have yet to be demonstrated. No peer-reviewed publications concerning the SOLX shunt are available at this time.
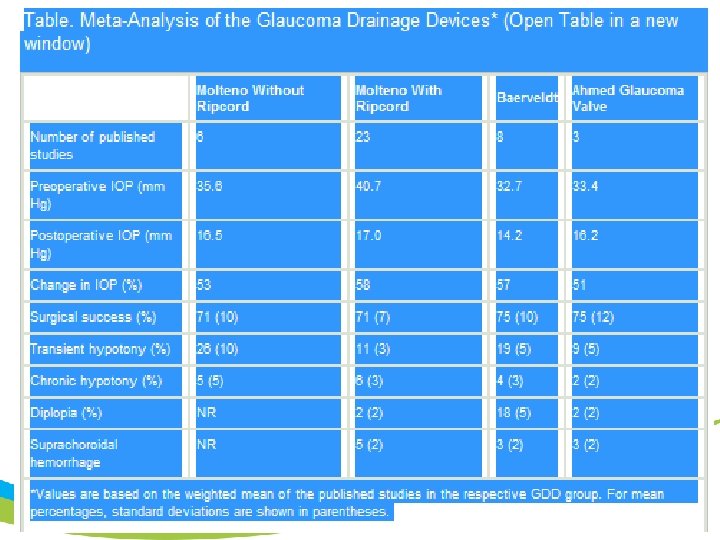
 Recommendations • The AGV is easy to insert, has 1 -quadrant dissection, requires less operative time as compared to other GDD operations, and has a low incidence of hypotony in the postoperative period. • The AGV has a higher incidence of the hypertensive phase postoperatively(80%) that might require additional glaucoma medications or needling of the bleb. • This implant is ideal for patients with diseases presenting with high IOP and minimal damage to the optic nerve, such as neovascular glaucoma, PKP with glaucoma, glaucoma following retinal detachment surgery, and uveitic glaucoma.
Recommendations • The AGV is easy to insert, has 1 -quadrant dissection, requires less operative time as compared to other GDD operations, and has a low incidence of hypotony in the postoperative period. • The AGV has a higher incidence of the hypertensive phase postoperatively(80%) that might require additional glaucoma medications or needling of the bleb. • This implant is ideal for patients with diseases presenting with high IOP and minimal damage to the optic nerve, such as neovascular glaucoma, PKP with glaucoma, glaucoma following retinal detachment surgery, and uveitic glaucoma.
 • GDDs double-plate Molteno implant and the Baerveldt implant, appear to exhibit a lower incidence of the hypertensive phase and may achieve slightly lower IOP. • The Baerveldt implant and the Molteno implant require more extensive dissection, additional operative time, and the use of a stent to avoid postoperative hypotony and a shallow anterior chamber. • The larger surface area of the end plate results in larger blebs and lower IOPs.
• GDDs double-plate Molteno implant and the Baerveldt implant, appear to exhibit a lower incidence of the hypertensive phase and may achieve slightly lower IOP. • The Baerveldt implant and the Molteno implant require more extensive dissection, additional operative time, and the use of a stent to avoid postoperative hypotony and a shallow anterior chamber. • The larger surface area of the end plate results in larger blebs and lower IOPs.
 Thank You www. sathyaneye. com
Thank You www. sathyaneye. com


