b78c08705b2b04baf8fb99c397c83991.ppt
- Количество слайдов: 59
 New and Emerging Therapies for the Clinical Management of HIV Infection Sponsored for CME credit by Rush University Medical Center Supported by an independent educational grant from Gilead Sciences Medical Affairs
New and Emerging Therapies for the Clinical Management of HIV Infection Sponsored for CME credit by Rush University Medical Center Supported by an independent educational grant from Gilead Sciences Medical Affairs
 CME Disclaimer, Disclosure Information, and Slide Handouts ● CME Disclaimer - These slides may not be videotaped, published, posted online, and/or presented for Continuing Medical Education credit without written permission from Rush University Medical Center and Practice Point Communications ● Disclosure Information - - - It is the policy of the Rush University Medical Center Office of Continuing Medical Education to ensure that its CME activities are independent, free of commercial bias and beyond the control of persons or organizations with an economic interest in influencing the content of CME Everyone who is in a position to control the content of an educational activity must disclose all relevant financial relationships with any commercial interest (including but not limited to pharmaceutical companies, biomedical device manufacturers, or other corporations whose products or services are related to the subject matter of the presentation topic) within the preceding 12 months If there are relationships that create a conflict of interest, these must be resolved by the CME Course Director in consultation with the Office of Continuing Medical Education prior to the participation of the faculty member in the development or presentation of course content ● Slide Handouts 2 The enclosed slide handouts are provided for reference purposes only The faculty presenter may have customized the slides through reordering or deleting and thus the handouts may not exactly match the presentation
CME Disclaimer, Disclosure Information, and Slide Handouts ● CME Disclaimer - These slides may not be videotaped, published, posted online, and/or presented for Continuing Medical Education credit without written permission from Rush University Medical Center and Practice Point Communications ● Disclosure Information - - - It is the policy of the Rush University Medical Center Office of Continuing Medical Education to ensure that its CME activities are independent, free of commercial bias and beyond the control of persons or organizations with an economic interest in influencing the content of CME Everyone who is in a position to control the content of an educational activity must disclose all relevant financial relationships with any commercial interest (including but not limited to pharmaceutical companies, biomedical device manufacturers, or other corporations whose products or services are related to the subject matter of the presentation topic) within the preceding 12 months If there are relationships that create a conflict of interest, these must be resolved by the CME Course Director in consultation with the Office of Continuing Medical Education prior to the participation of the faculty member in the development or presentation of course content ● Slide Handouts 2 The enclosed slide handouts are provided for reference purposes only The faculty presenter may have customized the slides through reordering or deleting and thus the handouts may not exactly match the presentation
 Educator Lisa Hightow-Weidman, MD, MPH Associate Professor University of North Carolina at Chapel Hill ● Disclosures - 3 Grants/Research Support: n/a Consultant: n/a Speakers’ Bureau: Gilead, Janssen Stock Shareholder: n/a Other Financial or Material Support: n/a
Educator Lisa Hightow-Weidman, MD, MPH Associate Professor University of North Carolina at Chapel Hill ● Disclosures - 3 Grants/Research Support: n/a Consultant: n/a Speakers’ Bureau: Gilead, Janssen Stock Shareholder: n/a Other Financial or Material Support: n/a
 Accreditation and Designation Rush University Medical Center is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians. Rush University Medical Center designates this live activity for a maximum of 1 AMA PRA Category 1 Credit™. Physicians should claim only credit commensurate with the extent of their participation in the activity. ANAC is an approved provider of continuing nursing education (CNE) by the Virginia Nurses Association, an accredited approver by the American Nurses Credentialing Center’s Commission on Accreditation. This activity is approved for 1. 0 contact hour by the Association of Nurses in AIDS Care. The University of Florida College of Pharmacy is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education (UAN #0012 -9999 -13 -043 -L 02 -P). This activity is accredited for 1 hour of continuing pharmacy education (CPE) credit. The University of Florida College of Pharmacy will report all credit to CPE Monitor within 30 working days after receiving evidence of successful completion of the course. Successful completion means that you must attend the entire program and complete an evaluation form. Supported by an independent educational grant from Gilead Sciences Medical Affairs. 4
Accreditation and Designation Rush University Medical Center is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians. Rush University Medical Center designates this live activity for a maximum of 1 AMA PRA Category 1 Credit™. Physicians should claim only credit commensurate with the extent of their participation in the activity. ANAC is an approved provider of continuing nursing education (CNE) by the Virginia Nurses Association, an accredited approver by the American Nurses Credentialing Center’s Commission on Accreditation. This activity is approved for 1. 0 contact hour by the Association of Nurses in AIDS Care. The University of Florida College of Pharmacy is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education (UAN #0012 -9999 -13 -043 -L 02 -P). This activity is accredited for 1 hour of continuing pharmacy education (CPE) credit. The University of Florida College of Pharmacy will report all credit to CPE Monitor within 30 working days after receiving evidence of successful completion of the course. Successful completion means that you must attend the entire program and complete an evaluation form. Supported by an independent educational grant from Gilead Sciences Medical Affairs. 4
 Faculty CME Course Director Harold A. Kessler, MD Professor of Medicine and Immunology/Microbiology Associate Director, Section of Infectious Diseases Rush University Medical Center Chicago, Illinois Chief, Division of HIV Medicine Harbor-UCLA Medical Center Torrance, California Professor of Medicine David Geffen School of Medicine at UCLA Los Angeles, California CME Reviewer David M. Simon, MD, Ph. D CNE Reviewer Allison R. Webel, RN, Ph. D Associate Professor Section of Infectious Diseases Rush University Medical Center Chicago, Illinois 5 Content Development and Training Eric S. Daar, MD Instructor and KL 2 Clinical Research Scholar Frances Payne Bolton School of Nursing Case Western Reserve University Cleveland, Ohio
Faculty CME Course Director Harold A. Kessler, MD Professor of Medicine and Immunology/Microbiology Associate Director, Section of Infectious Diseases Rush University Medical Center Chicago, Illinois Chief, Division of HIV Medicine Harbor-UCLA Medical Center Torrance, California Professor of Medicine David Geffen School of Medicine at UCLA Los Angeles, California CME Reviewer David M. Simon, MD, Ph. D CNE Reviewer Allison R. Webel, RN, Ph. D Associate Professor Section of Infectious Diseases Rush University Medical Center Chicago, Illinois 5 Content Development and Training Eric S. Daar, MD Instructor and KL 2 Clinical Research Scholar Frances Payne Bolton School of Nursing Case Western Reserve University Cleveland, Ohio
 Faculty Disclosures CME Course Director: Harold A. Kessler, MD Content Development and Training: Eric S. Daar, MD Grants/research support None Abbott, Gilead Sciences, Merck, Pfizer, Vii. V Consultant None Bristol-Myers Squibb, Gilead Sciences, Merck, Vii. V Speakers’ bureau None Abbott Laboratories, Glaxo. Smith. Kline, Merck None Stock shareholder Other financial or material support 6
Faculty Disclosures CME Course Director: Harold A. Kessler, MD Content Development and Training: Eric S. Daar, MD Grants/research support None Abbott, Gilead Sciences, Merck, Pfizer, Vii. V Consultant None Bristol-Myers Squibb, Gilead Sciences, Merck, Vii. V Speakers’ bureau None Abbott Laboratories, Glaxo. Smith. Kline, Merck None Stock shareholder Other financial or material support 6
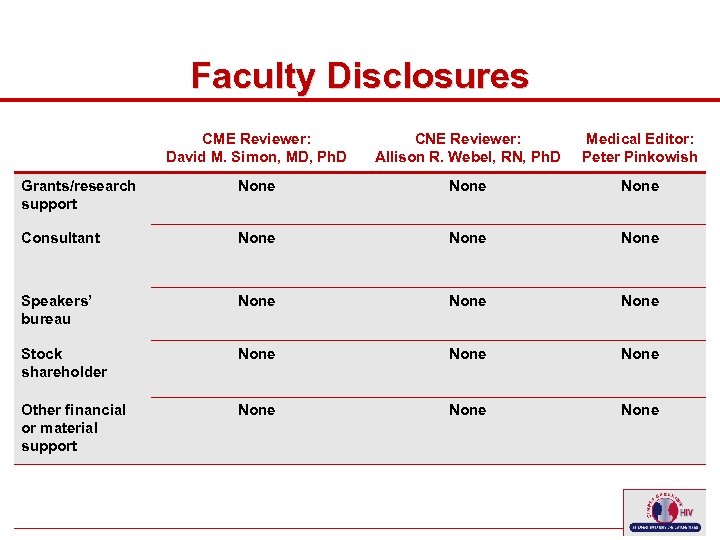 Faculty Disclosures CME Reviewer: David M. Simon, MD, Ph. D Medical Editor: Peter Pinkowish Grants/research support None Consultant None Speakers’ bureau None Stock shareholder None Other financial or material support 7 CNE Reviewer: Allison R. Webel, RN, Ph. D None
Faculty Disclosures CME Reviewer: David M. Simon, MD, Ph. D Medical Editor: Peter Pinkowish Grants/research support None Consultant None Speakers’ bureau None Stock shareholder None Other financial or material support 7 CNE Reviewer: Allison R. Webel, RN, Ph. D None
 Opinions and Off-Label Discussions The opinions or views expressed in this educational program are those of the participants and do not necessarily reflect the opinions or recommendations of Gilead Sciences Medical Affairs, Rush University Medical Center, the Association of Nurses in AIDS Care, or the University of Florida College of Pharmacy The faculty may have included discussion on unlabeled uses of a commercial product or an investigational use of a product not yet approved for this purpose Please consult the full prescribing information before using any medication mentioned in this program 8
Opinions and Off-Label Discussions The opinions or views expressed in this educational program are those of the participants and do not necessarily reflect the opinions or recommendations of Gilead Sciences Medical Affairs, Rush University Medical Center, the Association of Nurses in AIDS Care, or the University of Florida College of Pharmacy The faculty may have included discussion on unlabeled uses of a commercial product or an investigational use of a product not yet approved for this purpose Please consult the full prescribing information before using any medication mentioned in this program 8
 New Electronic Evaluation Process ● Please clearly print your information on the Sign-in Sheet ● You will receive an electronic evaluation to the email address provided within 1 business day ● Reminder email communications will be sent up to 5 days post lecture until the evaluation is completed ● Completion Is Required for CME/CNE/CPE credit and future attendance ● Incomplete evaluations will preclude attendees from receiving their CME/CNE/CPE certificate & future communications about lectures in your area 9
New Electronic Evaluation Process ● Please clearly print your information on the Sign-in Sheet ● You will receive an electronic evaluation to the email address provided within 1 business day ● Reminder email communications will be sent up to 5 days post lecture until the evaluation is completed ● Completion Is Required for CME/CNE/CPE credit and future attendance ● Incomplete evaluations will preclude attendees from receiving their CME/CNE/CPE certificate & future communications about lectures in your area 9
 Learning Objectives (CME/CNE and CPE) CPE CME/CNE ● Upon completion of this activity, the participant intends to incorporate the following objectives into their practice of medicine and/or advance practice nursing: - Appropriately select antiretroviral therapy for my HIV-infected patients according to the guideline recommendations by the Department of Health and Human Services ● Upon completion of this activity, the pharmacist should be able to: - Recommend antiretroviral therapy for my HIV-infected patients according to the guideline recommendations by the Department of Health and Human Services - Counsel my HIV-infected patients on the benefits and risks associated with antiretroviral therapy - - Counsel my HIV-infected patients on new potential drug targets against HIV infection - - 10 Counsel my HIV-infected patients on the benefits and risks associated with antiretroviral therapy Counsel my HIV-infected patients how HIV agents in late-stage clinical development may impact future management of HIV-infected patients
Learning Objectives (CME/CNE and CPE) CPE CME/CNE ● Upon completion of this activity, the participant intends to incorporate the following objectives into their practice of medicine and/or advance practice nursing: - Appropriately select antiretroviral therapy for my HIV-infected patients according to the guideline recommendations by the Department of Health and Human Services ● Upon completion of this activity, the pharmacist should be able to: - Recommend antiretroviral therapy for my HIV-infected patients according to the guideline recommendations by the Department of Health and Human Services - Counsel my HIV-infected patients on the benefits and risks associated with antiretroviral therapy - - Counsel my HIV-infected patients on new potential drug targets against HIV infection - - 10 Counsel my HIV-infected patients on the benefits and risks associated with antiretroviral therapy Counsel my HIV-infected patients how HIV agents in late-stage clinical development may impact future management of HIV-infected patients
 Program Overview ● Treatment challenges/clinical needs ● New antiviral drugs/formulations 11
Program Overview ● Treatment challenges/clinical needs ● New antiviral drugs/formulations 11
 Worldwide Treatment and Prevention Gaps (2011) ● On ART: 8 million ● Number needing ART: 15 million ● New infections: 2 million ● People were waiting to become treatmenteligible, sicken, or die: ~24 million ● Estimated coverage of ART in low- and middleincome countries: 36% Granich R, et al. Curr Opin HIV AIDS. 2013; 8: 41 -49. 12
Worldwide Treatment and Prevention Gaps (2011) ● On ART: 8 million ● Number needing ART: 15 million ● New infections: 2 million ● People were waiting to become treatmenteligible, sicken, or die: ~24 million ● Estimated coverage of ART in low- and middleincome countries: 36% Granich R, et al. Curr Opin HIV AIDS. 2013; 8: 41 -49. 12
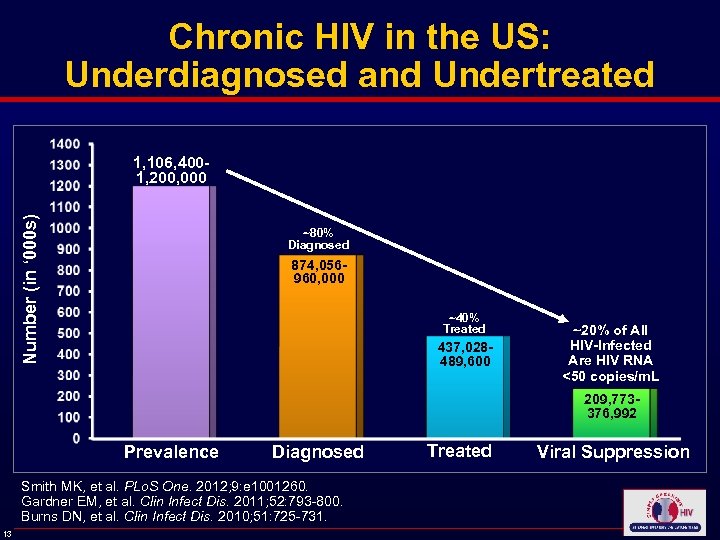 Chronic HIV in the US: Underdiagnosed and Undertreated Number (in ‘ 000 s) 1, 106, 4001, 200, 000 ~80% Diagnosed 874, 056960, 000 ~40% Treated 437, 028489, 600 ~20% of All HIV-Infected Are HIV RNA <50 copies/m. L 209, 773376, 992 Prevalence Diagnosed Smith MK, et al. PLo. S One. 2012; 9: e 1001260. Gardner EM, et al. Clin Infect Dis. 2011; 52: 793 -800. Burns DN, et al. Clin Infect Dis. 2010; 51: 725 -731. 13 Treated Viral Suppression
Chronic HIV in the US: Underdiagnosed and Undertreated Number (in ‘ 000 s) 1, 106, 4001, 200, 000 ~80% Diagnosed 874, 056960, 000 ~40% Treated 437, 028489, 600 ~20% of All HIV-Infected Are HIV RNA <50 copies/m. L 209, 773376, 992 Prevalence Diagnosed Smith MK, et al. PLo. S One. 2012; 9: e 1001260. Gardner EM, et al. Clin Infect Dis. 2011; 52: 793 -800. Burns DN, et al. Clin Infect Dis. 2010; 51: 725 -731. 13 Treated Viral Suppression
 No Single, Stand-Alone HIV Prevention Intervention Will Halt the HIV Pandemic ● Over the past 30 years, existing prevention strategies have had limited to no success - 14 Education about risks Behavioral interventions to decrease risk Harm reduction Vaccines
No Single, Stand-Alone HIV Prevention Intervention Will Halt the HIV Pandemic ● Over the past 30 years, existing prevention strategies have had limited to no success - 14 Education about risks Behavioral interventions to decrease risk Harm reduction Vaccines
 The Shift Towards Earlier Initiation of Antiretroviral Therapy ● Newer ART regimens - Generally better tolerated, more convenient, and more potent than older regimens ● Survival benefit - Randomized controlled trials - Observational cohort data ● Untreated HIV - Maybe associated the development of non-AIDS-defining illness ● Biologic rationale ● Effective ART reduces HIV transmission 15
The Shift Towards Earlier Initiation of Antiretroviral Therapy ● Newer ART regimens - Generally better tolerated, more convenient, and more potent than older regimens ● Survival benefit - Randomized controlled trials - Observational cohort data ● Untreated HIV - Maybe associated the development of non-AIDS-defining illness ● Biologic rationale ● Effective ART reduces HIV transmission 15
 Simultaneous Use of Different Classes of Prevention Strategies Biomedical Interventions Structural Interventions Combination HIV Prevention Community Interventions 16 HIV Testing, Linkage to Care, Expanded ART Coverage Individual and Small Group Behavioral Interventions
Simultaneous Use of Different Classes of Prevention Strategies Biomedical Interventions Structural Interventions Combination HIV Prevention Community Interventions 16 HIV Testing, Linkage to Care, Expanded ART Coverage Individual and Small Group Behavioral Interventions
 FDA Approves First Drug for Reducing the Risk of Sexually Acquired HIV Infection (July 16, 2012) ● Emtricitabine/tenofovir DF - Indicated in combination with safer sex practices for pre-exposure prophylaxis (Pr. EP) to reduce the risk of sexually acquired HIV-1 infection in adults at high risk • ● Other prevention methods (eg, safe sex practices, risk reduction counseling, and regular HIV testing) Revised Pr. EP Boxed Warning ● Use in those who are confirmed HIV-negative prior to prescribing the drug and at least every 3 months during use Contraindicated in those with unknown or positive HIV status Gilead Sciences conditions of Pr. EP approval - Collect viral isolates from individuals who acquire HIV while taking emtricitabine/tenofovir DF and to evaluate these isolates for the presence of resistance - Collect pregnancy outcomes data for women who become pregnant while taking emtricitabine/tenofovir DF for Pr. EP - Conduct a trial to evaluate drug adherence and its relationship to adverse events, risk of seroconversion, and resistance development in seroconverters Available at: http: //www. fda. gov/News. Events/Newsroom/Press. Announcements/ucm 312210. htm. 17
FDA Approves First Drug for Reducing the Risk of Sexually Acquired HIV Infection (July 16, 2012) ● Emtricitabine/tenofovir DF - Indicated in combination with safer sex practices for pre-exposure prophylaxis (Pr. EP) to reduce the risk of sexually acquired HIV-1 infection in adults at high risk • ● Other prevention methods (eg, safe sex practices, risk reduction counseling, and regular HIV testing) Revised Pr. EP Boxed Warning ● Use in those who are confirmed HIV-negative prior to prescribing the drug and at least every 3 months during use Contraindicated in those with unknown or positive HIV status Gilead Sciences conditions of Pr. EP approval - Collect viral isolates from individuals who acquire HIV while taking emtricitabine/tenofovir DF and to evaluate these isolates for the presence of resistance - Collect pregnancy outcomes data for women who become pregnant while taking emtricitabine/tenofovir DF for Pr. EP - Conduct a trial to evaluate drug adherence and its relationship to adverse events, risk of seroconversion, and resistance development in seroconverters Available at: http: //www. fda. gov/News. Events/Newsroom/Press. Announcements/ucm 312210. htm. 17
 CDC Interim Guidance: Pr. EP in Heterosexually Active Adults and MSM ● Interim guidance as part of a comprehensive set of HIV prevention services ● Pr. EP has the potential to contribute to effective and safe HIV prevention for if it is: - Targeted to those at high risk for HIV acquisition Delivered as part of a comprehensive set of prevention services • • • - Risk reduction and Pr. EP adherence counseling Ready access to condoms Diagnosis and treatment of STIs Accompanied by monitoring HIV status, side effects, adherence, and risk behavior CDC. MMWR Morb Mortal Wkly Rep. 2011; 60: 65 -68. CDC. MMWR Morb Mortal Wkly Rep. 2012; 61: 586 -589. 18
CDC Interim Guidance: Pr. EP in Heterosexually Active Adults and MSM ● Interim guidance as part of a comprehensive set of HIV prevention services ● Pr. EP has the potential to contribute to effective and safe HIV prevention for if it is: - Targeted to those at high risk for HIV acquisition Delivered as part of a comprehensive set of prevention services • • • - Risk reduction and Pr. EP adherence counseling Ready access to condoms Diagnosis and treatment of STIs Accompanied by monitoring HIV status, side effects, adherence, and risk behavior CDC. MMWR Morb Mortal Wkly Rep. 2011; 60: 65 -68. CDC. MMWR Morb Mortal Wkly Rep. 2012; 61: 586 -589. 18
 Pr. EP Trial Results ● Proof of efficacy study of topical tenofovir gel in women - CAPRISA 004 ● First oral Pr. EP study of emtricitabine/tenofovir DF for MSM - i. Pr. Ex ● Proof of efficacy studies in young, heterosexual adults in Africa - Partners Pr. EP - TDF 2 (CDC 4940) ● Early termination due to futility of Pr. EP in women - FEM-Pr. EP - VOICE (oral tenofovir DF and topical tenofovir arms only) 19
Pr. EP Trial Results ● Proof of efficacy study of topical tenofovir gel in women - CAPRISA 004 ● First oral Pr. EP study of emtricitabine/tenofovir DF for MSM - i. Pr. Ex ● Proof of efficacy studies in young, heterosexual adults in Africa - Partners Pr. EP - TDF 2 (CDC 4940) ● Early termination due to futility of Pr. EP in women - FEM-Pr. EP - VOICE (oral tenofovir DF and topical tenofovir arms only) 19
 CDC Interim Guidance for Healthcare Providers: Beginning Pr. EP Medication Regimen ● Prescribe emtricitabine/tenofovir DF (200/300 mg) - 1 tablet daily ● In general, prescribe no more than a 90 -day supply - Renew only after confirming patient remains HIV uninfected ● If HBV infected - Consider emtricitabine/tenofovir DF for HBV and HIV prevention ● Provide risk-reduction and Pr. EP medication adherence counseling and condoms CDC. MMWR Morb Mortal Wkly Rep. 2011; 60: 65 -68. CDC. MMWR Morb Mortal Wkly Rep. 2012; 61: 586 -589. 20
CDC Interim Guidance for Healthcare Providers: Beginning Pr. EP Medication Regimen ● Prescribe emtricitabine/tenofovir DF (200/300 mg) - 1 tablet daily ● In general, prescribe no more than a 90 -day supply - Renew only after confirming patient remains HIV uninfected ● If HBV infected - Consider emtricitabine/tenofovir DF for HBV and HIV prevention ● Provide risk-reduction and Pr. EP medication adherence counseling and condoms CDC. MMWR Morb Mortal Wkly Rep. 2011; 60: 65 -68. CDC. MMWR Morb Mortal Wkly Rep. 2012; 61: 586 -589. 20
 CDC Interim Guidance for Healthcare Providers: Follow-Up While on Pr. EP ● Evaluate and support Pr. EP medication adherence at each follow-up visit (more often if needed) - For women, conduct pregnancy test ● Every 2 to 3 months - HIV antibody test (document negative result) Assess • • Risk behaviors and provide risk-reduction counseling and condoms STI symptoms (if present, test and treat as needed) ● Every 6 months - Test for STI regardless of symptomatology (treat as needed) ● Every 3 months after initiation, then yearly while on Pr. EP - Blood urea nitrogen Serum creatinine CDC. MMWR Morb Mortal Wkly Rep. 2011; 60: 65 -68. CDC. MMWR Morb Mortal Wkly Rep. 2012; 61: 586 -589. 21
CDC Interim Guidance for Healthcare Providers: Follow-Up While on Pr. EP ● Evaluate and support Pr. EP medication adherence at each follow-up visit (more often if needed) - For women, conduct pregnancy test ● Every 2 to 3 months - HIV antibody test (document negative result) Assess • • Risk behaviors and provide risk-reduction counseling and condoms STI symptoms (if present, test and treat as needed) ● Every 6 months - Test for STI regardless of symptomatology (treat as needed) ● Every 3 months after initiation, then yearly while on Pr. EP - Blood urea nitrogen Serum creatinine CDC. MMWR Morb Mortal Wkly Rep. 2011; 60: 65 -68. CDC. MMWR Morb Mortal Wkly Rep. 2012; 61: 586 -589. 21
 CDC Interim Guidance for Healthcare Providers: Discontinuing Pr. EP ● Perform HIV test(s) to confirm HIV status - If positive • • • - Order and document results of resistance testing Establish linkage to care For pregnant women, inform prenatal-care provider and coordinate care to maintain HIV prevention during pregnancy and breastfeeding If negative • Establish linkage to risk-reduction support services as indicated ● If active HBV infection at initiation of Pr. EP - Consider appropriate medication for continued treatment of HBV infection CDC. MMWR Morb Mortal Wkly Rep. 2011; 60: 65 -68. CDC. MMWR Morb Mortal Wkly Rep. 2012; 61: 586 -589. 22
CDC Interim Guidance for Healthcare Providers: Discontinuing Pr. EP ● Perform HIV test(s) to confirm HIV status - If positive • • • - Order and document results of resistance testing Establish linkage to care For pregnant women, inform prenatal-care provider and coordinate care to maintain HIV prevention during pregnancy and breastfeeding If negative • Establish linkage to risk-reduction support services as indicated ● If active HBV infection at initiation of Pr. EP - Consider appropriate medication for continued treatment of HBV infection CDC. MMWR Morb Mortal Wkly Rep. 2011; 60: 65 -68. CDC. MMWR Morb Mortal Wkly Rep. 2012; 61: 586 -589. 22
 Program Overview ● Treatment challenges/clinical needs ● New antiviral drugs/formulations 23
Program Overview ● Treatment challenges/clinical needs ● New antiviral drugs/formulations 23
 DHHS Guidelines: When To Start Perspectives ● Untreated HIV infection may have detrimental effects at all stages of infection - Effects of immune deficiency, direct effects of HIV on specific end organs, and the indirect effects of HIV-associated inflammation on these organs ● Earlier treatment may prevent the damage associated with HIV replication during early stages of infection - Sustaining viral suppression and maintaining higher CD 4 count via ART delays or prevents some non-AIDS-defining complications and disorders ● Success of ART hinges on avoiding treatment interruptions DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 24
DHHS Guidelines: When To Start Perspectives ● Untreated HIV infection may have detrimental effects at all stages of infection - Effects of immune deficiency, direct effects of HIV on specific end organs, and the indirect effects of HIV-associated inflammation on these organs ● Earlier treatment may prevent the damage associated with HIV replication during early stages of infection - Sustaining viral suppression and maintaining higher CD 4 count via ART delays or prevents some non-AIDS-defining complications and disorders ● Success of ART hinges on avoiding treatment interruptions DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 24
 When to Start Treatment 2/2013 DHHS Guidelines 2012 IAS-USA Guidelines CD 4 Count HIV RNA (cells/mm 3) (copies/m. L) Any value Treat <500 Any value Treat >500 Any value Treat Pregnant women Any value Treat HIV-associated nephropathy Any value Treat HIV/HBV coinfection when HBV treatment is indicated Any value Treat Clinical Category AIDS-defining illness or severe symptoms Asymptomatic The IAS-USA guidelines also recommends initiating antiretroviral therapy in HIV-infected patients with active hepatitis C virus infection, active or high risk for cardiovascular disease, and symptomatic primary HIV infection. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013; Thompson MA, et al. JAMA. 2012; 308: 387 -402. 25
When to Start Treatment 2/2013 DHHS Guidelines 2012 IAS-USA Guidelines CD 4 Count HIV RNA (cells/mm 3) (copies/m. L) Any value Treat <500 Any value Treat >500 Any value Treat Pregnant women Any value Treat HIV-associated nephropathy Any value Treat HIV/HBV coinfection when HBV treatment is indicated Any value Treat Clinical Category AIDS-defining illness or severe symptoms Asymptomatic The IAS-USA guidelines also recommends initiating antiretroviral therapy in HIV-infected patients with active hepatitis C virus infection, active or high risk for cardiovascular disease, and symptomatic primary HIV infection. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013; Thompson MA, et al. JAMA. 2012; 308: 387 -402. 25
 DHHS Guidelines: Preferred Regimens NNRTI PI Efavirenz 1/emtricitabine 2/tenofovir DF 3 Atazanavir 4 + ritonavir + emtricitabine 2/tenofovir DF 3 Darunavir + ritonavir (qd) + emtricitabine 2/tenofovir DF 3 INSTI Raltegravir + emtricitabine 2/tenofovir DF 3 Pregnant Lopinavir/r bid + zidovudine/lamivudine 2 women INSTI: Integrase strand transfer inhibitors. 1 Efavirenz should not be used during the first trimester of pregnancy or in women trying to conceive or not using effective and consistent contraception. 2 Lamivudine may substitute for emtricitabine or visa versa. 3 Tenofovir DF should be used with caution in patients with renal insufficiency. 4 Atazanavir + RTV should not be used in patients who require >20 mg omeprazole equivalent/day. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 26
DHHS Guidelines: Preferred Regimens NNRTI PI Efavirenz 1/emtricitabine 2/tenofovir DF 3 Atazanavir 4 + ritonavir + emtricitabine 2/tenofovir DF 3 Darunavir + ritonavir (qd) + emtricitabine 2/tenofovir DF 3 INSTI Raltegravir + emtricitabine 2/tenofovir DF 3 Pregnant Lopinavir/r bid + zidovudine/lamivudine 2 women INSTI: Integrase strand transfer inhibitors. 1 Efavirenz should not be used during the first trimester of pregnancy or in women trying to conceive or not using effective and consistent contraception. 2 Lamivudine may substitute for emtricitabine or visa versa. 3 Tenofovir DF should be used with caution in patients with renal insufficiency. 4 Atazanavir + RTV should not be used in patients who require >20 mg omeprazole equivalent/day. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 26
 DHHS Guidelines: Alternative Regimens NNRTI Efavirenz + (abacavir 1 or zidovudine)/lamivudine 2 Rilpivirine 3/emtricitabine 2/tenofovir DF Rilpivirine 3 + abacavir/lamivudine 2 PI Atazanavir + ritonavir + abacavir/lamivudine 2 Darunavir + ritonavir + abacavir/lamivudine 2 Fosamprenavir + ritonavir (qd or bid) + abacavir/lamivudine 2 or emtricitabine 2/tenofovir DF Lopinavir/r 4 (qd or bid) + abacavir/lamivudine 2 or emtricitabine 2/tenofovir DF INSTI Raltegravir + abacavir/lamivudine 2 Elvitegravir/cobicistat/emtricitabine/tenofovir DF 5 1 Abacavir should not be used in patients who test positive for HLA-B*5701. Use abacavir with caution in patients with high risk of cardiovascular disease or pretreatment HIV RNA >100, 000 copies/m. L. 2 Lamivudine may substitute for emtricitabine or visa versa. 3 Use rilpivirine with caution in patients with pretreatment HIV RNA >100, 000 copies/m. L. 4 Once-daily lopinavir/r is not recommended in pregnant women. 5 Patients with creatinine clearance >70 m. L/min. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 27
DHHS Guidelines: Alternative Regimens NNRTI Efavirenz + (abacavir 1 or zidovudine)/lamivudine 2 Rilpivirine 3/emtricitabine 2/tenofovir DF Rilpivirine 3 + abacavir/lamivudine 2 PI Atazanavir + ritonavir + abacavir/lamivudine 2 Darunavir + ritonavir + abacavir/lamivudine 2 Fosamprenavir + ritonavir (qd or bid) + abacavir/lamivudine 2 or emtricitabine 2/tenofovir DF Lopinavir/r 4 (qd or bid) + abacavir/lamivudine 2 or emtricitabine 2/tenofovir DF INSTI Raltegravir + abacavir/lamivudine 2 Elvitegravir/cobicistat/emtricitabine/tenofovir DF 5 1 Abacavir should not be used in patients who test positive for HLA-B*5701. Use abacavir with caution in patients with high risk of cardiovascular disease or pretreatment HIV RNA >100, 000 copies/m. L. 2 Lamivudine may substitute for emtricitabine or visa versa. 3 Use rilpivirine with caution in patients with pretreatment HIV RNA >100, 000 copies/m. L. 4 Once-daily lopinavir/r is not recommended in pregnant women. 5 Patients with creatinine clearance >70 m. L/min. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 27
 No One Right Option for Everyone: Limitations of Current First-Line Regimens ● Efavirenz-based regimens - Not recommended for women at risk of becoming pregnant CNS toxicity Rash Low barrier to resistance ● Raltegravir-based regimens - Twice-daily administration Relatively low barrier to resistance Lack of second-line integrase inhibitor option ● Ritonavir-boosted PI-based regimens 28 Higher pill count Gastrointestinal toxicity
No One Right Option for Everyone: Limitations of Current First-Line Regimens ● Efavirenz-based regimens - Not recommended for women at risk of becoming pregnant CNS toxicity Rash Low barrier to resistance ● Raltegravir-based regimens - Twice-daily administration Relatively low barrier to resistance Lack of second-line integrase inhibitor option ● Ritonavir-boosted PI-based regimens 28 Higher pill count Gastrointestinal toxicity
 Simplified and Convenient ART: Achieving Goals of Therapy ● Treatment goals - Reduce HIV-associated morbidity and prolong the duration and quality of survival - Restore and preserve immunologic function Maximally and durably suppress plasma HIV viral load Prevent HIV transmission ● Individualize strategies to achieve goals - Tailor regimens to enhance adherence - Pretreatment genotypic resistance testing - Maximize conditions to promote ART adherence DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 29
Simplified and Convenient ART: Achieving Goals of Therapy ● Treatment goals - Reduce HIV-associated morbidity and prolong the duration and quality of survival - Restore and preserve immunologic function Maximally and durably suppress plasma HIV viral load Prevent HIV transmission ● Individualize strategies to achieve goals - Tailor regimens to enhance adherence - Pretreatment genotypic resistance testing - Maximize conditions to promote ART adherence DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 29
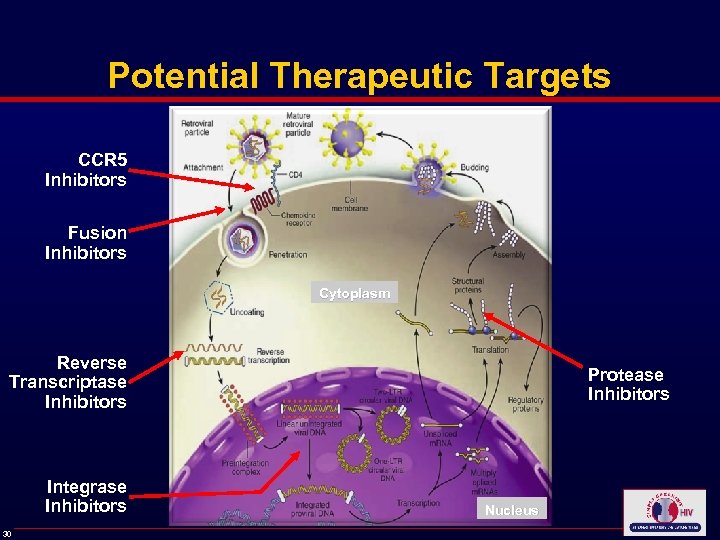 Potential Therapeutic Targets CCR 5 Inhibitors Fusion Inhibitors Cytoplasm Reverse Transcriptase Inhibitors Integrase Inhibitors 30 Protease Inhibitors Nucleus
Potential Therapeutic Targets CCR 5 Inhibitors Fusion Inhibitors Cytoplasm Reverse Transcriptase Inhibitors Integrase Inhibitors 30 Protease Inhibitors Nucleus
 Program Overview ● Treatment challenges/clinical needs ● New antiviral drugs/formulations 31
Program Overview ● Treatment challenges/clinical needs ● New antiviral drugs/formulations 31
 Study 102: QUAD Versus Efavirenz/Emtricitabine/Tenofovir DF Single-Tablet, Once-Daily Regimens Phase 3 study (192 weeks) Treatment-naïve Double-blind HIV RNA >5000 copies/m. L Any CD 4 count Non-inferiority (12% margin) Randomization 1: 1 Elvitegravir 150 mg/Cobicistat 150 mg/ Emtricitabine/Tenofovir DF (n=348) Efavirenz 600 mg/ Emtricitabine/Tenofovir DF (n=352) Primary Endpoint Week 48 HIV RNA <50 Copies/m. L Sax PE, et al. Lancet. 2012; 379: 2439 -2448. Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 32
Study 102: QUAD Versus Efavirenz/Emtricitabine/Tenofovir DF Single-Tablet, Once-Daily Regimens Phase 3 study (192 weeks) Treatment-naïve Double-blind HIV RNA >5000 copies/m. L Any CD 4 count Non-inferiority (12% margin) Randomization 1: 1 Elvitegravir 150 mg/Cobicistat 150 mg/ Emtricitabine/Tenofovir DF (n=348) Efavirenz 600 mg/ Emtricitabine/Tenofovir DF (n=352) Primary Endpoint Week 48 HIV RNA <50 Copies/m. L Sax PE, et al. Lancet. 2012; 379: 2439 -2448. Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 32
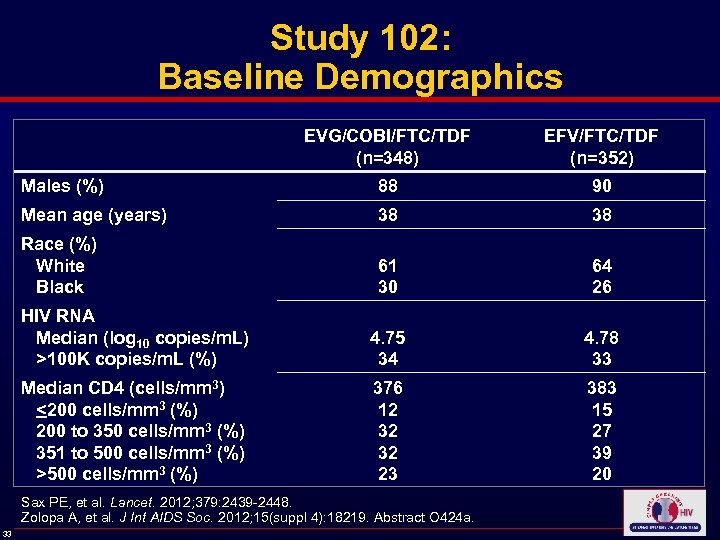 Study 102: Baseline Demographics EVG/COBI/FTC/TDF (n=348) EFV/FTC/TDF (n=352) Males (%) 88 90 Mean age (years) 38 38 Race (%) White Black 61 30 64 26 HIV RNA Median (log 10 copies/m. L) >100 K copies/m. L (%) 4. 75 34 4. 78 33 Median CD 4 (cells/mm 3) <200 cells/mm 3 (%) 200 to 350 cells/mm 3 (%) 351 to 500 cells/mm 3 (%) >500 cells/mm 3 (%) 376 12 32 32 23 383 15 27 39 20 Sax PE, et al. Lancet. 2012; 379: 2439 -2448. Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 33
Study 102: Baseline Demographics EVG/COBI/FTC/TDF (n=348) EFV/FTC/TDF (n=352) Males (%) 88 90 Mean age (years) 38 38 Race (%) White Black 61 30 64 26 HIV RNA Median (log 10 copies/m. L) >100 K copies/m. L (%) 4. 75 34 4. 78 33 Median CD 4 (cells/mm 3) <200 cells/mm 3 (%) 200 to 350 cells/mm 3 (%) 351 to 500 cells/mm 3 (%) >500 cells/mm 3 (%) 376 12 32 32 23 383 15 27 39 20 Sax PE, et al. Lancet. 2012; 379: 2439 -2448. Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 33
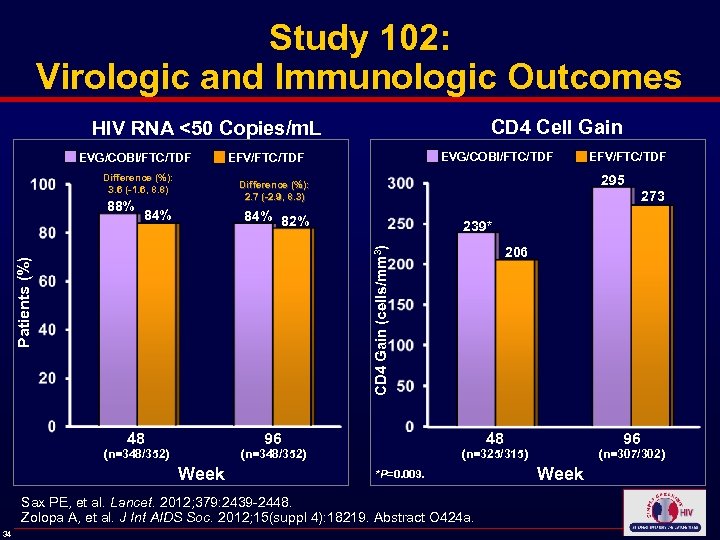 Study 102: Virologic and Immunologic Outcomes CD 4 Cell Gain HIV RNA <50 Copies/m. L EVG/COBI/FTC/TDF EFV/FTC/TDF Difference (%): 3. 6 (-1. 6, 8. 8) 84% 273 84% 82% 239* Patients (%) CD 4 Gain (cells/mm 3) 88% 295 Difference (%): 2. 7 (-2. 9, 8. 3) 96 48 Week (n=307/302) (n=325/315) *P=0. 009. Sax PE, et al. Lancet. 2012; 379: 2439 -2448. Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 34 96 48 (n=348/352) 206 Week
Study 102: Virologic and Immunologic Outcomes CD 4 Cell Gain HIV RNA <50 Copies/m. L EVG/COBI/FTC/TDF EFV/FTC/TDF Difference (%): 3. 6 (-1. 6, 8. 8) 84% 273 84% 82% 239* Patients (%) CD 4 Gain (cells/mm 3) 88% 295 Difference (%): 2. 7 (-2. 9, 8. 3) 96 48 Week (n=307/302) (n=325/315) *P=0. 009. Sax PE, et al. Lancet. 2012; 379: 2439 -2448. Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 34 96 48 (n=348/352) 206 Week
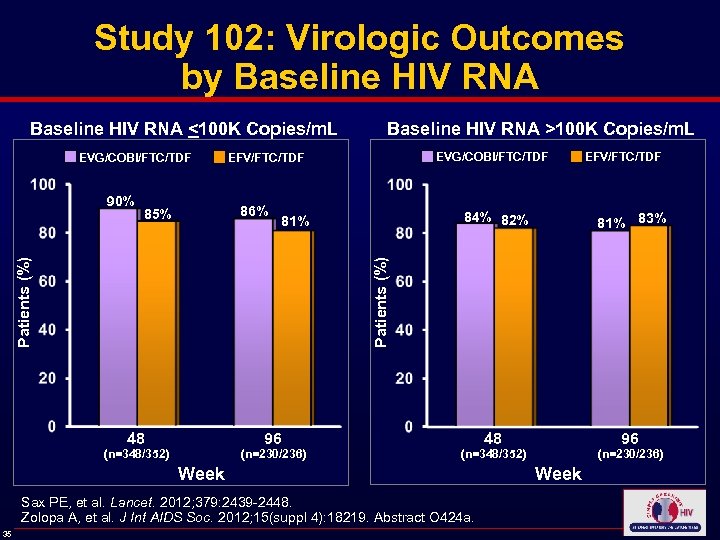 Study 102: Virologic Outcomes by Baseline HIV RNA <100 K Copies/m. L Baseline HIV RNA >100 K Copies/m. L EVG/COBI/FTC/TDF EFV/FTC/TDF 86% 85% 84% 82% 81% 83% 48 96 Patients (%) 90% 96 48 (n=230/236) (n=348/352) Week Sax PE, et al. Lancet. 2012; 379: 2439 -2448. Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 35 (n=230/236) (n=348/352) Week
Study 102: Virologic Outcomes by Baseline HIV RNA <100 K Copies/m. L Baseline HIV RNA >100 K Copies/m. L EVG/COBI/FTC/TDF EFV/FTC/TDF 86% 85% 84% 82% 81% 83% 48 96 Patients (%) 90% 96 48 (n=230/236) (n=348/352) Week Sax PE, et al. Lancet. 2012; 379: 2439 -2448. Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 35 (n=230/236) (n=348/352) Week
 Study 102 (Week 96): NNRTI, Integrase, and NRTI Resistance EVG/COBI/FTC/TDF (n=348) EFV/FTC/TDF (n=352) Number of patients analyzed for resistance With resistance to ART regimen 14 10 17 10 Any primary integrase resistance (number of patients) E 92 Q T 66 I Q 148 R N 155 H 9 7 1 1 3 Any primary NNRTI resistance (number of patients) K 103 N K 101 E/K Y 188 Y/F/H/L G 190 A/S Any primary NRTI resistance (number of patients) M 184 V/I K 65 R 10 9 3 2 1 10 10 4 Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 36 3 3 3
Study 102 (Week 96): NNRTI, Integrase, and NRTI Resistance EVG/COBI/FTC/TDF (n=348) EFV/FTC/TDF (n=352) Number of patients analyzed for resistance With resistance to ART regimen 14 10 17 10 Any primary integrase resistance (number of patients) E 92 Q T 66 I Q 148 R N 155 H 9 7 1 1 3 Any primary NNRTI resistance (number of patients) K 103 N K 101 E/K Y 188 Y/F/H/L G 190 A/S Any primary NRTI resistance (number of patients) M 184 V/I K 65 R 10 9 3 2 1 10 10 4 Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 36 3 3 3
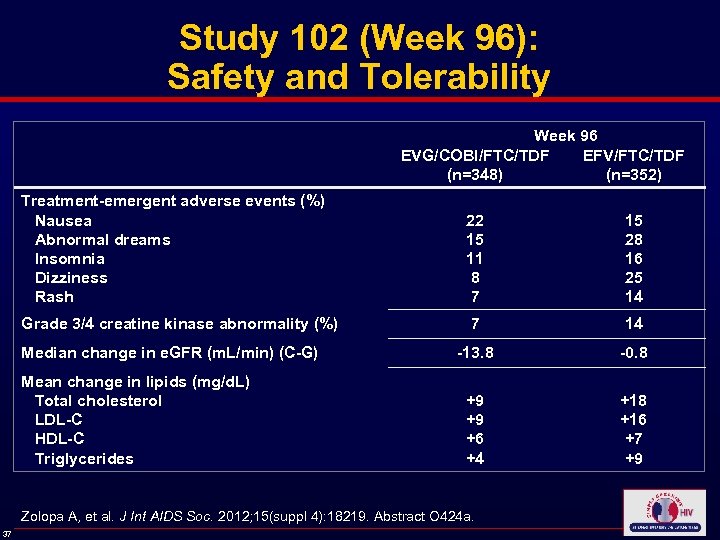 Study 102 (Week 96): Safety and Tolerability Week 96 EVG/COBI/FTC/TDF EFV/FTC/TDF (n=348) (n=352) Treatment-emergent adverse events (%) Nausea Abnormal dreams Insomnia Dizziness Rash 22 15 11 8 7 15 28 16 25 14 Grade 3/4 creatine kinase abnormality (%) 7 14 -13. 8 -0. 8 +9 +9 +6 +4 +18 +16 +7 +9 Median change in e. GFR (m. L/min) (C-G) Mean change in lipids (mg/d. L) Total cholesterol LDL-C HDL-C Triglycerides Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 37
Study 102 (Week 96): Safety and Tolerability Week 96 EVG/COBI/FTC/TDF EFV/FTC/TDF (n=348) (n=352) Treatment-emergent adverse events (%) Nausea Abnormal dreams Insomnia Dizziness Rash 22 15 11 8 7 15 28 16 25 14 Grade 3/4 creatine kinase abnormality (%) 7 14 -13. 8 -0. 8 +9 +9 +6 +4 +18 +16 +7 +9 Median change in e. GFR (m. L/min) (C-G) Mean change in lipids (mg/d. L) Total cholesterol LDL-C HDL-C Triglycerides Zolopa A, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18219. Abstract O 424 a. 37
 Study 103: QUAD Versus Atazanavir/r + Emtricitabine/Tenofovir DF Once-Daily Regimens Phase 3 study (192 weeks) Treatment-naïve Double-blind HIV RNA >5000 copies/m. L Any CD 4 count Non-inferiority (12% margin) Randomization 1: 1 Elvitegravir/Cobicistat/ Emtricitabine/Tenofovir DF (n=353) Atazanavir/r + Emtricitabine/Tenofovir DF (n=355) Primary Endpoint Week 48 HIV RNA <50 Copies/m. L De. Jesus E, et al. Lancet. 2012; 379: 2429 -2438. Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 38
Study 103: QUAD Versus Atazanavir/r + Emtricitabine/Tenofovir DF Once-Daily Regimens Phase 3 study (192 weeks) Treatment-naïve Double-blind HIV RNA >5000 copies/m. L Any CD 4 count Non-inferiority (12% margin) Randomization 1: 1 Elvitegravir/Cobicistat/ Emtricitabine/Tenofovir DF (n=353) Atazanavir/r + Emtricitabine/Tenofovir DF (n=355) Primary Endpoint Week 48 HIV RNA <50 Copies/m. L De. Jesus E, et al. Lancet. 2012; 379: 2429 -2438. Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 38
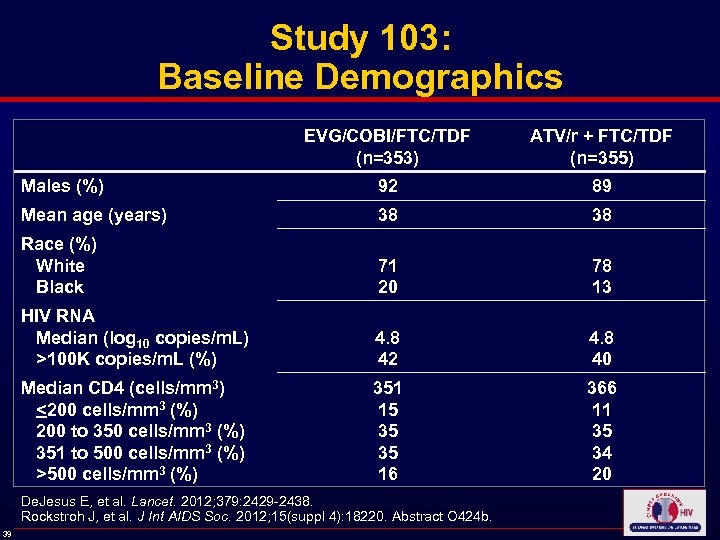 Study 103: Baseline Demographics EVG/COBI/FTC/TDF (n=353) ATV/r + FTC/TDF (n=355) Males (%) 92 89 Mean age (years) 38 38 Race (%) White Black 71 20 78 13 HIV RNA Median (log 10 copies/m. L) >100 K copies/m. L (%) 4. 8 42 4. 8 40 Median CD 4 (cells/mm 3) <200 cells/mm 3 (%) 200 to 350 cells/mm 3 (%) 351 to 500 cells/mm 3 (%) >500 cells/mm 3 (%) 351 15 35 35 16 366 11 35 34 20 De. Jesus E, et al. Lancet. 2012; 379: 2429 -2438. Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 39
Study 103: Baseline Demographics EVG/COBI/FTC/TDF (n=353) ATV/r + FTC/TDF (n=355) Males (%) 92 89 Mean age (years) 38 38 Race (%) White Black 71 20 78 13 HIV RNA Median (log 10 copies/m. L) >100 K copies/m. L (%) 4. 8 42 4. 8 40 Median CD 4 (cells/mm 3) <200 cells/mm 3 (%) 200 to 350 cells/mm 3 (%) 351 to 500 cells/mm 3 (%) >500 cells/mm 3 (%) 351 15 35 35 16 366 11 35 34 20 De. Jesus E, et al. Lancet. 2012; 379: 2429 -2438. Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 39
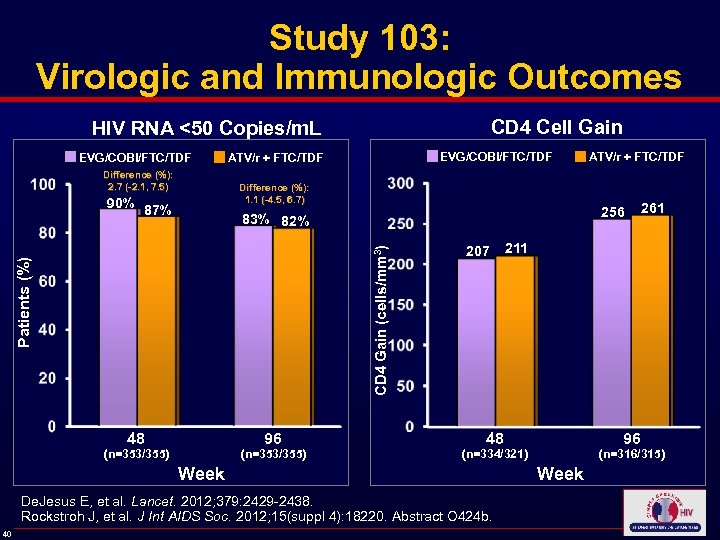 Study 103: Virologic and Immunologic Outcomes CD 4 Cell Gain HIV RNA <50 Copies/m. L EVG/COBI/FTC/TDF ATV/r + FTC/TDF Difference (%): 2. 7 (-2. 1, 7. 5) 87% 256 83% 82% Patients (%) CD 4 Gain (cells/mm 3) 90% Difference (%): 1. 1 (-4. 5, 6. 7) 96 48 (n=353/355) 207 96 (n=316/315) (n=334/321) De. Jesus E, et al. Lancet. 2012; 379: 2429 -2438. Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 40 211 48 Week 261 Week
Study 103: Virologic and Immunologic Outcomes CD 4 Cell Gain HIV RNA <50 Copies/m. L EVG/COBI/FTC/TDF ATV/r + FTC/TDF Difference (%): 2. 7 (-2. 1, 7. 5) 87% 256 83% 82% Patients (%) CD 4 Gain (cells/mm 3) 90% Difference (%): 1. 1 (-4. 5, 6. 7) 96 48 (n=353/355) 207 96 (n=316/315) (n=334/321) De. Jesus E, et al. Lancet. 2012; 379: 2429 -2438. Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 40 211 48 Week 261 Week
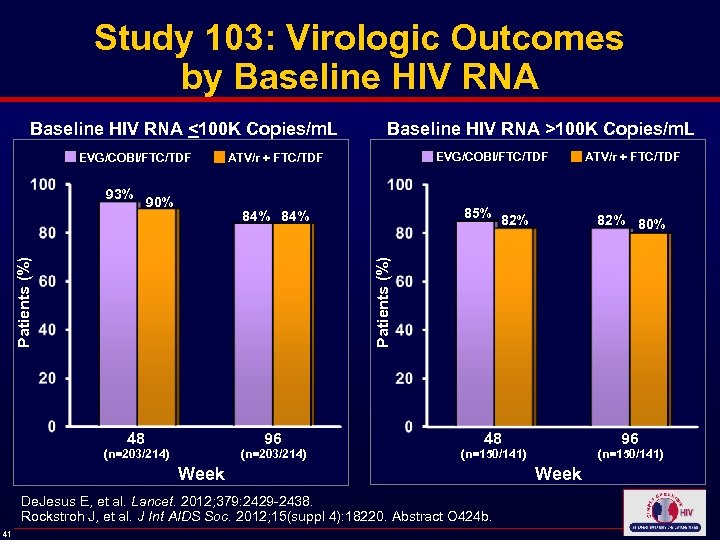 Study 103: Virologic Outcomes by Baseline HIV RNA <100 K Copies/m. L Baseline HIV RNA >100 K Copies/m. L EVG/COBI/FTC/TDF ATV/r + FTC/TDF 90% 85% 84% Patients (%) 96 48 (n=203/214) 82% 80% 96 48 (n=150/141) Week De. Jesus E, et al. Lancet. 2012; 379: 2429 -2438. Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 41 82% Patients (%) 93% Week
Study 103: Virologic Outcomes by Baseline HIV RNA <100 K Copies/m. L Baseline HIV RNA >100 K Copies/m. L EVG/COBI/FTC/TDF ATV/r + FTC/TDF 90% 85% 84% Patients (%) 96 48 (n=203/214) 82% 80% 96 48 (n=150/141) Week De. Jesus E, et al. Lancet. 2012; 379: 2429 -2438. Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 41 82% Patients (%) 93% Week
 Study 103 (Week 96): Resistance EVG/COBI/FTC/TDF (n=353) ATV/r + FTC/TDF (n=355) Number of patients Analyzed for resistance With resistance to ART regimen 12 6 8 0 Any primary integrase resistance (number of patients) E 92 Q T 66 I Q 148 R N 155 H 5 2 1 2 2 Any primary NRTI resistance (number of patients) M 184 V/I K 65 R 5 5 1 Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 42 0
Study 103 (Week 96): Resistance EVG/COBI/FTC/TDF (n=353) ATV/r + FTC/TDF (n=355) Number of patients Analyzed for resistance With resistance to ART regimen 12 6 8 0 Any primary integrase resistance (number of patients) E 92 Q T 66 I Q 148 R N 155 H 5 2 1 2 2 Any primary NRTI resistance (number of patients) M 184 V/I K 65 R 5 5 1 Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 42 0
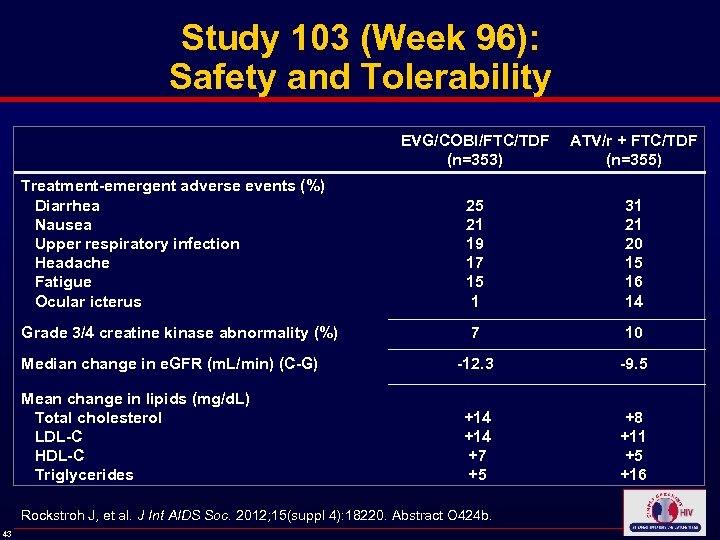 Study 103 (Week 96): Safety and Tolerability EVG/COBI/FTC/TDF (n=353) ATV/r + FTC/TDF (n=355) Treatment-emergent adverse events (%) Diarrhea Nausea Upper respiratory infection Headache Fatigue Ocular icterus 25 21 19 17 15 1 31 21 20 15 16 14 Grade 3/4 creatine kinase abnormality (%) 7 10 Median change in e. GFR (m. L/min) (C-G) -12. 3 -9. 5 Mean change in lipids (mg/d. L) Total cholesterol LDL-C HDL-C Triglycerides +14 +7 +5 +8 +11 +5 +16 Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 43
Study 103 (Week 96): Safety and Tolerability EVG/COBI/FTC/TDF (n=353) ATV/r + FTC/TDF (n=355) Treatment-emergent adverse events (%) Diarrhea Nausea Upper respiratory infection Headache Fatigue Ocular icterus 25 21 19 17 15 1 31 21 20 15 16 14 Grade 3/4 creatine kinase abnormality (%) 7 10 Median change in e. GFR (m. L/min) (C-G) -12. 3 -9. 5 Mean change in lipids (mg/d. L) Total cholesterol LDL-C HDL-C Triglycerides +14 +7 +5 +8 +11 +5 +16 Rockstroh J, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18220. Abstract O 424 b. 43
 Elvitegravir/Cobicistat/Emtricitabine/Tenofovir DF: Dosing and Safety Considerations ● Meal restrictions - Take with meal ● Adverse events - Diarrhea, nausea • - Early decrease in estimated GFR from cobicistat • - Comparable with ATV/r, usually mild and rarely leads to drug discontinuation Generally benign if <0. 4 mg/d. L increase in creatinine Drug-drug interactions: may be similar to ritonavir-boosted PI, do not use with other PIs ● Elvitegravir and raltegravir share similar resistance pathways (cross resistant) DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. Johnson VA, et al. Top Antivir Med. 2011; 19: 156 -164. 44
Elvitegravir/Cobicistat/Emtricitabine/Tenofovir DF: Dosing and Safety Considerations ● Meal restrictions - Take with meal ● Adverse events - Diarrhea, nausea • - Early decrease in estimated GFR from cobicistat • - Comparable with ATV/r, usually mild and rarely leads to drug discontinuation Generally benign if <0. 4 mg/d. L increase in creatinine Drug-drug interactions: may be similar to ritonavir-boosted PI, do not use with other PIs ● Elvitegravir and raltegravir share similar resistance pathways (cross resistant) DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. Johnson VA, et al. Top Antivir Med. 2011; 19: 156 -164. 44
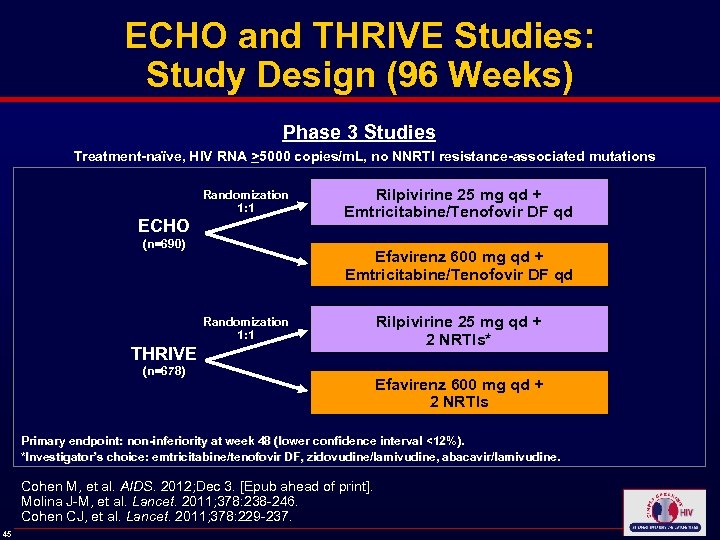 ECHO and THRIVE Studies: Study Design (96 Weeks) Phase 3 Studies Treatment-naïve, HIV RNA >5000 copies/m. L, no NNRTI resistance-associated mutations Randomization 1: 1 ECHO (n=690) Rilpivirine 25 mg qd + Emtricitabine/Tenofovir DF qd Efavirenz 600 mg qd + Emtricitabine/Tenofovir DF qd Randomization 1: 1 THRIVE (n=678) Rilpivirine 25 mg qd + 2 NRTIs* Efavirenz 600 mg qd + 2 NRTIs Primary endpoint: non-inferiority at week 48 (lower confidence interval <12%). *Investigator’s choice: emtricitabine/tenofovir DF, zidovudine/lamivudine, abacavir/lamivudine. *Investigator’ Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. Molina J-M, et al. Lancet. 2011; 378: 238 -246. Cohen CJ, et al. Lancet. 2011; 378: 229 -237. 45
ECHO and THRIVE Studies: Study Design (96 Weeks) Phase 3 Studies Treatment-naïve, HIV RNA >5000 copies/m. L, no NNRTI resistance-associated mutations Randomization 1: 1 ECHO (n=690) Rilpivirine 25 mg qd + Emtricitabine/Tenofovir DF qd Efavirenz 600 mg qd + Emtricitabine/Tenofovir DF qd Randomization 1: 1 THRIVE (n=678) Rilpivirine 25 mg qd + 2 NRTIs* Efavirenz 600 mg qd + 2 NRTIs Primary endpoint: non-inferiority at week 48 (lower confidence interval <12%). *Investigator’s choice: emtricitabine/tenofovir DF, zidovudine/lamivudine, abacavir/lamivudine. *Investigator’ Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. Molina J-M, et al. Lancet. 2011; 378: 238 -246. Cohen CJ, et al. Lancet. 2011; 378: 229 -237. 45
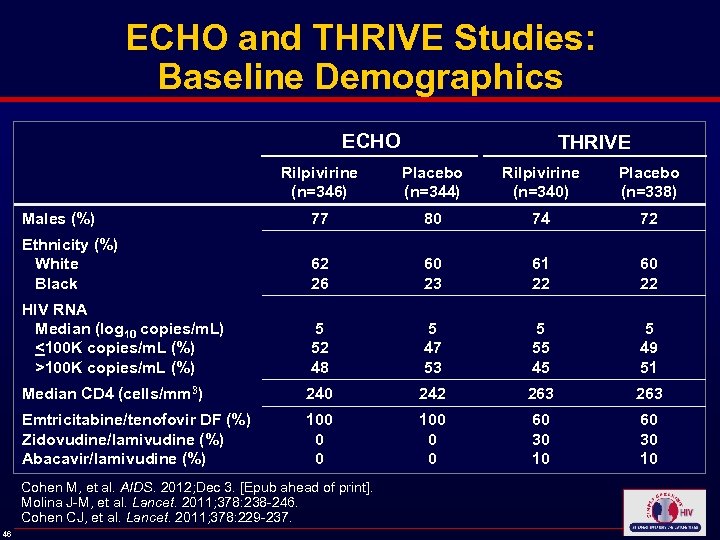 ECHO and THRIVE Studies: Baseline Demographics ECHO THRIVE Rilpivirine (n=346) Placebo (n=344) Rilpivirine (n=340) Placebo (n=338) Males (%) 77 80 74 72 Ethnicity (%) White Black 62 26 60 23 61 22 60 22 HIV RNA Median (log 10 copies/m. L) <100 K copies/m. L (%) >100 K copies/m. L (%) 5 52 48 5 47 53 5 55 45 5 49 51 Median CD 4 (cells/mm 3) 240 242 263 Emtricitabine/tenofovir DF (%) Zidovudine/lamivudine (%) Abacavir/lamivudine (%) 100 0 0 60 30 10 Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. Molina J-M, et al. Lancet. 2011; 378: 238 -246. Cohen CJ, et al. Lancet. 2011; 378: 229 -237. 46
ECHO and THRIVE Studies: Baseline Demographics ECHO THRIVE Rilpivirine (n=346) Placebo (n=344) Rilpivirine (n=340) Placebo (n=338) Males (%) 77 80 74 72 Ethnicity (%) White Black 62 26 60 23 61 22 60 22 HIV RNA Median (log 10 copies/m. L) <100 K copies/m. L (%) >100 K copies/m. L (%) 5 52 48 5 47 53 5 55 45 5 49 51 Median CD 4 (cells/mm 3) 240 242 263 Emtricitabine/tenofovir DF (%) Zidovudine/lamivudine (%) Abacavir/lamivudine (%) 100 0 0 60 30 10 Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. Molina J-M, et al. Lancet. 2011; 378: 238 -246. Cohen CJ, et al. Lancet. 2011; 378: 229 -237. 46
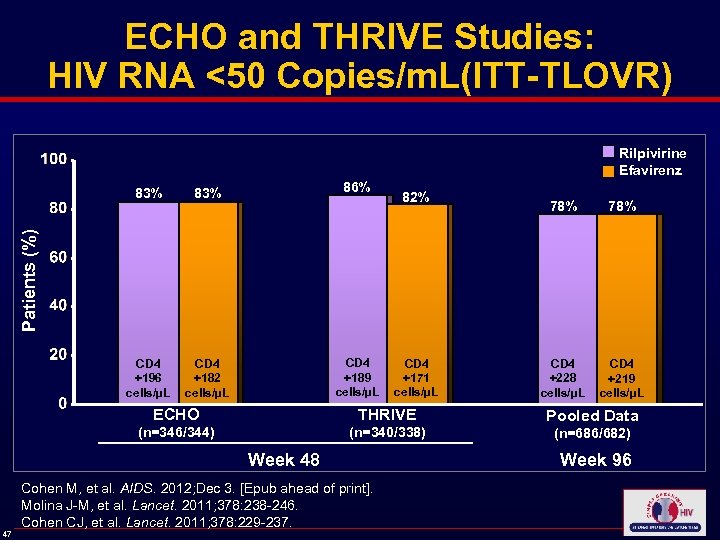 ECHO and THRIVE Studies: HIV RNA <50 Copies/m. L(ITT-TLOVR) Rilpivirine Efavirenz CD 4 +196 cells/µL 86% 83% CD 4 +182 cells/µL 82% 78% CD 4 +228 cells/µL CD 4 +219 cells/µL Patients (%) 83% CD 4 +189 cells/µL CD 4 +171 cells/µL ECHO THRIVE Pooled Data (n=346/344) (n=340/338) (n=686/682) Week 48 47 Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. Molina J-M, et al. Lancet. 2011; 378: 238 -246. Cohen CJ, et al. Lancet. 2011; 378: 229 -237. Week 96
ECHO and THRIVE Studies: HIV RNA <50 Copies/m. L(ITT-TLOVR) Rilpivirine Efavirenz CD 4 +196 cells/µL 86% 83% CD 4 +182 cells/µL 82% 78% CD 4 +228 cells/µL CD 4 +219 cells/µL Patients (%) 83% CD 4 +189 cells/µL CD 4 +171 cells/µL ECHO THRIVE Pooled Data (n=346/344) (n=340/338) (n=686/682) Week 48 47 Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. Molina J-M, et al. Lancet. 2011; 378: 238 -246. Cohen CJ, et al. Lancet. 2011; 378: 229 -237. Week 96
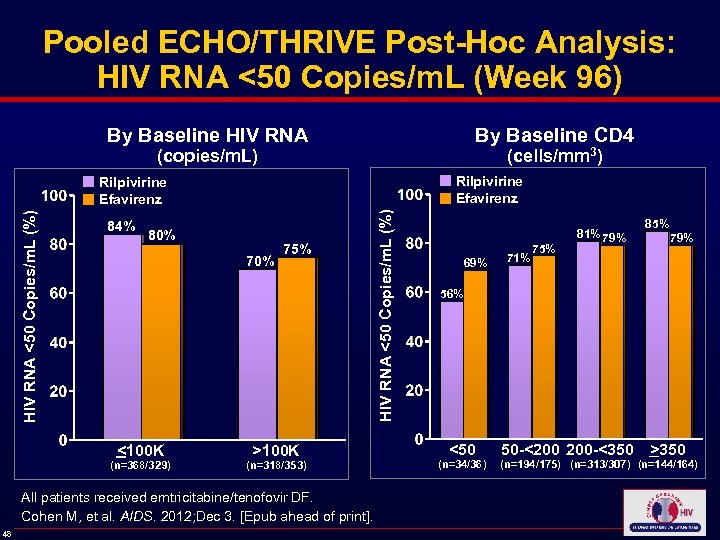 Pooled ECHO/THRIVE Post-Hoc Analysis: HIV RNA <50 Copies/m. L (Week 96) By Baseline HIV RNA By Baseline CD 4 (copies/m. L) (cells/mm 3) Rilpivirine Efavirenz 84% 80% 70% <100 K (n=368/329) 75% >100 K (n=318/353) All patients received emtricitabine/tenofovir DF. Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. 48 HIV RNA <50 Copies/m. L (%) Rilpivirine Efavirenz 69% 71% 75% 81% 79% 85% 79% 56% <50 (n=34/36) 50 -<200 200 -<350 >350 (n=194/175) (n=313/307) (n=144/164)
Pooled ECHO/THRIVE Post-Hoc Analysis: HIV RNA <50 Copies/m. L (Week 96) By Baseline HIV RNA By Baseline CD 4 (copies/m. L) (cells/mm 3) Rilpivirine Efavirenz 84% 80% 70% <100 K (n=368/329) 75% >100 K (n=318/353) All patients received emtricitabine/tenofovir DF. Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. 48 HIV RNA <50 Copies/m. L (%) Rilpivirine Efavirenz 69% 71% 75% 81% 79% 85% 79% 56% <50 (n=34/36) 50 -<200 200 -<350 >350 (n=194/175) (n=313/307) (n=144/164)
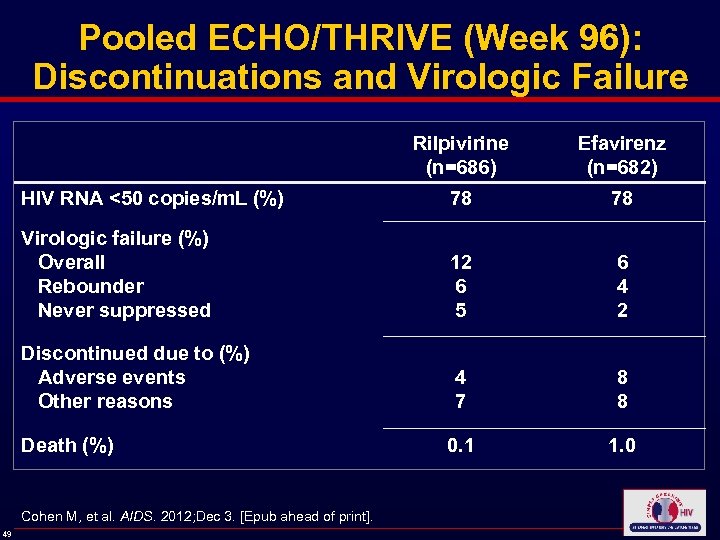 Pooled ECHO/THRIVE (Week 96): Discontinuations and Virologic Failure Rilpivirine (n=686) Efavirenz (n=682) HIV RNA <50 copies/m. L (%) 78 78 Virologic failure (%) Overall Rebounder Never suppressed 12 6 5 6 4 2 Discontinued due to (%) Adverse events Other reasons 4 7 8 8 0. 1 1. 0 Death (%) Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. 49
Pooled ECHO/THRIVE (Week 96): Discontinuations and Virologic Failure Rilpivirine (n=686) Efavirenz (n=682) HIV RNA <50 copies/m. L (%) 78 78 Virologic failure (%) Overall Rebounder Never suppressed 12 6 5 6 4 2 Discontinued due to (%) Adverse events Other reasons 4 7 8 8 0. 1 1. 0 Death (%) Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. 49
 Pooled ECHO/THRIVE (Week 96): Safety Rilpivirine (n=686) Efavirenz (n=682) Most common adverse events of interest (%) Any neurologic Dizziness Any psychiatric Abnormal dreams/nightmares Rash (any type) 17 8 16 8 4 38* 27* 22* 13† 15* Grade 2 -4 laboratory abnormality (%) Total cholesterol LDL-C AST ALT 7 7 6 6 22* 18* 10 11 *P<0. 0001 and †P=0. 0039 versus rilpivirine. Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. 50
Pooled ECHO/THRIVE (Week 96): Safety Rilpivirine (n=686) Efavirenz (n=682) Most common adverse events of interest (%) Any neurologic Dizziness Any psychiatric Abnormal dreams/nightmares Rash (any type) 17 8 16 8 4 38* 27* 22* 13† 15* Grade 2 -4 laboratory abnormality (%) Total cholesterol LDL-C AST ALT 7 7 6 6 22* 18* 10 11 *P<0. 0001 and †P=0. 0039 versus rilpivirine. Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. 50
 ECHO/THRIVE: Conclusions ● Efficacy - Week 0 -48: rilpivirine was non-inferior to efavirenz Week 48 -96: comparable between rilpivirine and efavirenz arms Better rilpivirine response: baseline HIV RNA <100 K versus >100 K copies/m. L ● Overall virologic failure rate - Week 0 -48: higher in the rilpivirine arm Weeks 48 -96: similar increases in rilpivirine and efavirenz arms ● Resistance with virologic failure - Rilpivirine: 6. 4%; efavirenz: 2. 3% ● Safety - 51 Lower incidence of adverse events of interest compared with efavirenz Most adverse events emerge during the first 4 weeks of therapy Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. Molina J-M, et al. Lancet. 2011; 378: 238 -246. Cohen CJ, et al. Lancet. 2011; 378: 229 -237. Rashbaum B, et al. 51 st ICAAC. Chicago, 2011. Abstract H 2 -805. Rimsky L, et al. JAIDS. 2012; 59: 39 -46.
ECHO/THRIVE: Conclusions ● Efficacy - Week 0 -48: rilpivirine was non-inferior to efavirenz Week 48 -96: comparable between rilpivirine and efavirenz arms Better rilpivirine response: baseline HIV RNA <100 K versus >100 K copies/m. L ● Overall virologic failure rate - Week 0 -48: higher in the rilpivirine arm Weeks 48 -96: similar increases in rilpivirine and efavirenz arms ● Resistance with virologic failure - Rilpivirine: 6. 4%; efavirenz: 2. 3% ● Safety - 51 Lower incidence of adverse events of interest compared with efavirenz Most adverse events emerge during the first 4 weeks of therapy Cohen M, et al. AIDS. 2012; Dec 3. [Epub ahead of print]. Molina J-M, et al. Lancet. 2011; 378: 238 -246. Cohen CJ, et al. Lancet. 2011; 378: 229 -237. Rashbaum B, et al. 51 st ICAAC. Chicago, 2011. Abstract H 2 -805. Rimsky L, et al. JAIDS. 2012; 59: 39 -46.
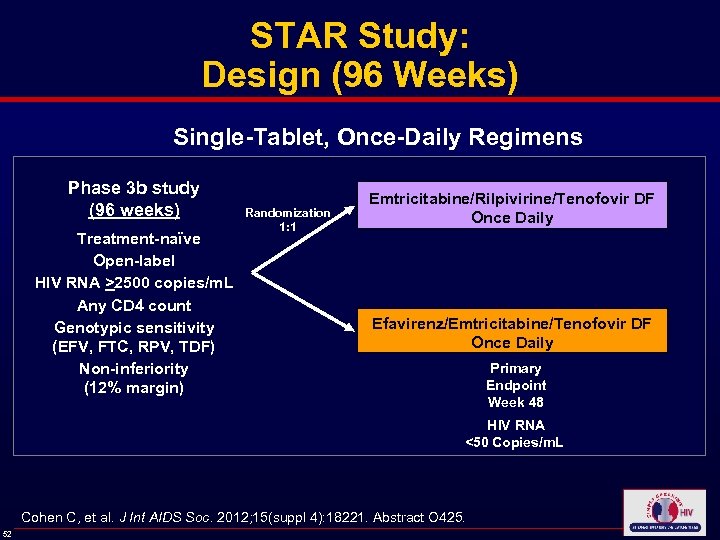 STAR Study: Design (96 Weeks) Single-Tablet, Once-Daily Regimens Phase 3 b study (96 weeks) Treatment-naïve Open-label HIV RNA >2500 copies/m. L Any CD 4 count Genotypic sensitivity (EFV, FTC, RPV, TDF) Non-inferiority (12% margin) Randomization 1: 1 Emtricitabine/Rilpivirine/Tenofovir DF Once Daily Efavirenz/Emtricitabine/Tenofovir DF Once Daily Primary Endpoint Week 48 HIV RNA <50 Copies/m. L Cohen C, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18221. Abstract O 425. 52
STAR Study: Design (96 Weeks) Single-Tablet, Once-Daily Regimens Phase 3 b study (96 weeks) Treatment-naïve Open-label HIV RNA >2500 copies/m. L Any CD 4 count Genotypic sensitivity (EFV, FTC, RPV, TDF) Non-inferiority (12% margin) Randomization 1: 1 Emtricitabine/Rilpivirine/Tenofovir DF Once Daily Efavirenz/Emtricitabine/Tenofovir DF Once Daily Primary Endpoint Week 48 HIV RNA <50 Copies/m. L Cohen C, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18221. Abstract O 425. 52
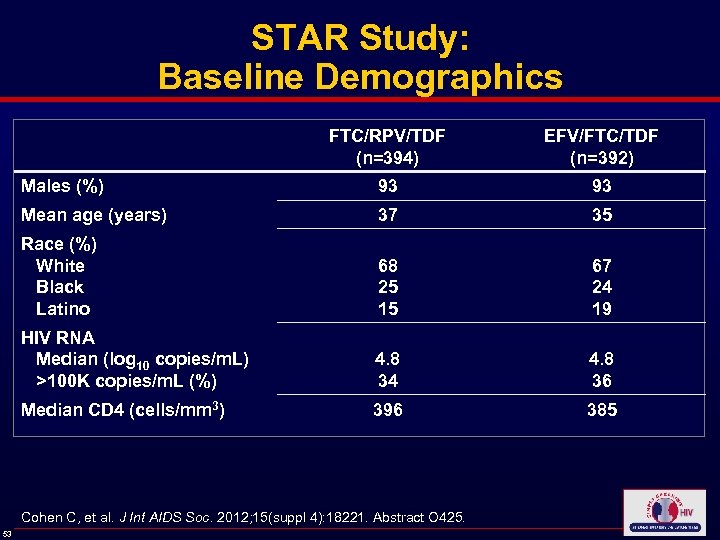 STAR Study: Baseline Demographics FTC/RPV/TDF (n=394) EFV/FTC/TDF (n=392) Males (%) 93 93 Mean age (years) 37 35 Race (%) White Black Latino 68 25 15 67 24 19 HIV RNA Median (log 10 copies/m. L) >100 K copies/m. L (%) 4. 8 34 4. 8 36 Median CD 4 (cells/mm 3) 396 385 Cohen C, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18221. Abstract O 425. 53
STAR Study: Baseline Demographics FTC/RPV/TDF (n=394) EFV/FTC/TDF (n=392) Males (%) 93 93 Mean age (years) 37 35 Race (%) White Black Latino 68 25 15 67 24 19 HIV RNA Median (log 10 copies/m. L) >100 K copies/m. L (%) 4. 8 34 4. 8 36 Median CD 4 (cells/mm 3) 396 385 Cohen C, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18221. Abstract O 425. 53
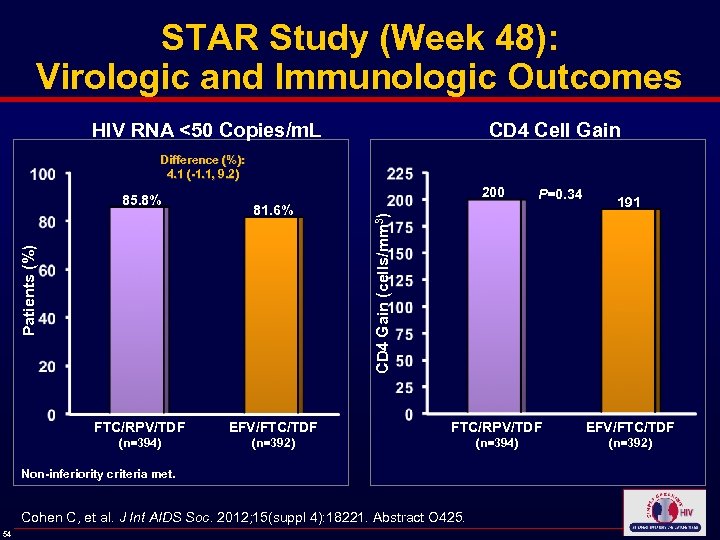 STAR Study (Week 48): Virologic and Immunologic Outcomes HIV RNA <50 Copies/m. L CD 4 Cell Gain Difference (%): 4. 1 (-1. 1, 9. 2) 81. 6% P=0. 34 191 CD 4 Gain (cells/mm 3) 200 Patients (%) 85. 8% FTC/RPV/TDF EFV/FTC/TDF (n=394) (n=392) Non-inferiority criteria met. Cohen C, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18221. Abstract O 425. 54
STAR Study (Week 48): Virologic and Immunologic Outcomes HIV RNA <50 Copies/m. L CD 4 Cell Gain Difference (%): 4. 1 (-1. 1, 9. 2) 81. 6% P=0. 34 191 CD 4 Gain (cells/mm 3) 200 Patients (%) 85. 8% FTC/RPV/TDF EFV/FTC/TDF (n=394) (n=392) Non-inferiority criteria met. Cohen C, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18221. Abstract O 425. 54
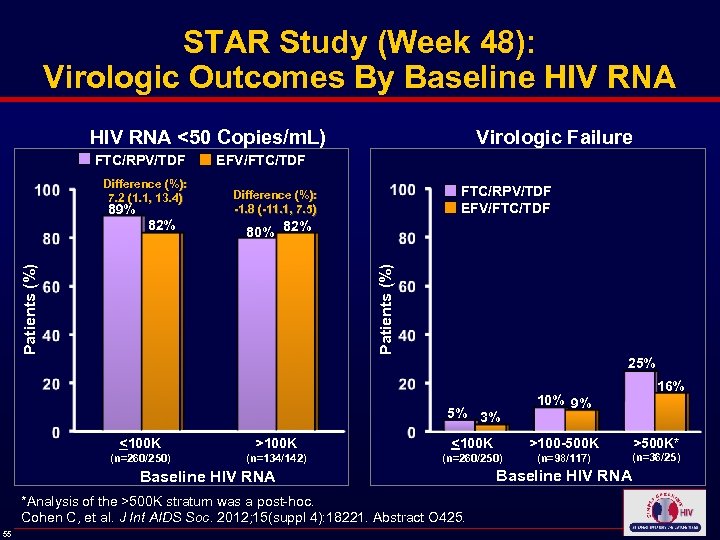 STAR Study (Week 48): Virologic Outcomes By Baseline HIV RNA <50 Copies/m. L) Virologic Failure FTC/RPV/TDF EFV/FTC/TDF Difference (%): 7. 2 (1. 1, 13. 4) 89% 80% 82% Patients (%) 82% FTC/RPV/TDF EFV/FTC/TDF Difference (%): -1. 8 (-11. 1, 7. 5) 25% 5% 3% 10% 9% 16% <100 K >100 K <100 K >100 -500 K >500 K* (n=260/250) (n=134/142) (n=260/250) (n=98/117) (n=36/25) Baseline HIV RNA *Analysis of the >500 K stratum was a post-hoc. Cohen C, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18221. Abstract O 425. 55 Baseline HIV RNA
STAR Study (Week 48): Virologic Outcomes By Baseline HIV RNA <50 Copies/m. L) Virologic Failure FTC/RPV/TDF EFV/FTC/TDF Difference (%): 7. 2 (1. 1, 13. 4) 89% 80% 82% Patients (%) 82% FTC/RPV/TDF EFV/FTC/TDF Difference (%): -1. 8 (-11. 1, 7. 5) 25% 5% 3% 10% 9% 16% <100 K >100 K <100 K >100 -500 K >500 K* (n=260/250) (n=134/142) (n=260/250) (n=98/117) (n=36/25) Baseline HIV RNA *Analysis of the >500 K stratum was a post-hoc. Cohen C, et al. J Int AIDS Soc. 2012; 15(suppl 4): 18221. Abstract O 425. 55 Baseline HIV RNA
 Emtricitabine/Rilpivirine/Tenofovir DF: Dosing and Safety Considerations ● Meal restrictions - Take with meal ● Drugs that increase gastric p. H (eg, proton-pump inhibitors) may decrease plasma concentrations of rilpivirine ● Adverse events (less common with rilpivirine compared with efavirenz) - Rash Neuropsychiatric symptoms ● Resistance (rilpivirine) - Most common RAM is E 138 K (leads to cross resistance to etravirine) DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. Johnson VA, et al. Top Antivir Med. 2011; 19: 156 -164. 56
Emtricitabine/Rilpivirine/Tenofovir DF: Dosing and Safety Considerations ● Meal restrictions - Take with meal ● Drugs that increase gastric p. H (eg, proton-pump inhibitors) may decrease plasma concentrations of rilpivirine ● Adverse events (less common with rilpivirine compared with efavirenz) - Rash Neuropsychiatric symptoms ● Resistance (rilpivirine) - Most common RAM is E 138 K (leads to cross resistance to etravirine) DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. Johnson VA, et al. Top Antivir Med. 2011; 19: 156 -164. 56
 Evidence Supports Combination ART for Prevention of HIV Transmission ● Transmission only occurs from persons with HIV ● HIV RNA level is single greatest risk factor for HIV transmission ● Combination ART can lower HIV RNA level to undetectable levels ● Observational evidence in heterosexual couples ● Previous modeling work suggests considerable potential ● Knowing one’s HIV status is key to prevention with combination ART ● When to start combination ART is not known for certainty 57
Evidence Supports Combination ART for Prevention of HIV Transmission ● Transmission only occurs from persons with HIV ● HIV RNA level is single greatest risk factor for HIV transmission ● Combination ART can lower HIV RNA level to undetectable levels ● Observational evidence in heterosexual couples ● Previous modeling work suggests considerable potential ● Knowing one’s HIV status is key to prevention with combination ART ● When to start combination ART is not known for certainty 57
 New Electronic Evaluation Process ● Please clearly print your information on the Sign-in Sheet ● You will receive an electronic evaluation to the email address provided within 1 business day ● Reminder email communications will be sent up to 5 days post lecture until the evaluation is completed ● Completion Is Required for CME/CNE/CPE credit and future attendance ● Incomplete evaluations will preclude attendees from receiving their CME/CNE/CPE certificate & future communications about lectures in your area 58
New Electronic Evaluation Process ● Please clearly print your information on the Sign-in Sheet ● You will receive an electronic evaluation to the email address provided within 1 business day ● Reminder email communications will be sent up to 5 days post lecture until the evaluation is completed ● Completion Is Required for CME/CNE/CPE credit and future attendance ● Incomplete evaluations will preclude attendees from receiving their CME/CNE/CPE certificate & future communications about lectures in your area 58
 Outcomes Measurement Reminder ● We are required to assess “changes in learners’ competence, performance or patient outcomes achieved as a result of their participation in a CME/CNE/CPE sponsored educational activity” ● As a result of this requirement you will receive a short survey via email 8 to 12 weeks after completing this course - We consider the survey to be an additional component of your overall participation in this educational activity and would urge you to reflect on what you learned in the activity and then complete this survey 59
Outcomes Measurement Reminder ● We are required to assess “changes in learners’ competence, performance or patient outcomes achieved as a result of their participation in a CME/CNE/CPE sponsored educational activity” ● As a result of this requirement you will receive a short survey via email 8 to 12 weeks after completing this course - We consider the survey to be an additional component of your overall participation in this educational activity and would urge you to reflect on what you learned in the activity and then complete this survey 59


