2d920013f83acb2ba22b9ea022f12999.ppt
- Количество слайдов: 50
 Neurophysiology and Intracranial Hypertension Mani K. C Vindhya M. D Asst Prof of Anesthesiology Nova Southeastern University
Neurophysiology and Intracranial Hypertension Mani K. C Vindhya M. D Asst Prof of Anesthesiology Nova Southeastern University
 Anesthesia for Craniotomy Three major issues: 1. ICP. The skull is a rigid container, so an increase in intracranial volume (ICV) tends to increase intracranial pressure (ICP). 2. Glucose and oxygen supply and demand. The brain has a high energy requirement with no capacity to store substrates. 3. Venous air emboli. Air can be entrained into cerebral veins, causing venous air emboli. This can happen any time the head is higher than the heart.
Anesthesia for Craniotomy Three major issues: 1. ICP. The skull is a rigid container, so an increase in intracranial volume (ICV) tends to increase intracranial pressure (ICP). 2. Glucose and oxygen supply and demand. The brain has a high energy requirement with no capacity to store substrates. 3. Venous air emboli. Air can be entrained into cerebral veins, causing venous air emboli. This can happen any time the head is higher than the heart.
 Three basic principles of anesthesia for craniotomy surgery 1. Employ techniques to decrease intracranial volume (ICV) and intracranial pressure (ICP). Maintain cerebral perfusion and oxygenation. Monitor for sudden changes in vital signs (as would be caused by venous air emboli).
Three basic principles of anesthesia for craniotomy surgery 1. Employ techniques to decrease intracranial volume (ICV) and intracranial pressure (ICP). Maintain cerebral perfusion and oxygenation. Monitor for sudden changes in vital signs (as would be caused by venous air emboli).
 Introduction to Intracranial Hypertension Intracranial pressure (ICP) is a main concern in many intracranial neuroanesthesia cases: (Sulek CA. Intracranial Pressure, In: Cucchiara RF, Black S, Michenfelder JD, eds. Clinical Neuroanesthesia, 2 nd ed. , Churchhill Livingstone, New York, 1998, pp. 73 -123. ) Brain tumors Head injury Subdural or epidural hematomas Subarachnoid hemorrhage (ruptured aneurysm) Arteriovenous malformation Hydrocephalus
Introduction to Intracranial Hypertension Intracranial pressure (ICP) is a main concern in many intracranial neuroanesthesia cases: (Sulek CA. Intracranial Pressure, In: Cucchiara RF, Black S, Michenfelder JD, eds. Clinical Neuroanesthesia, 2 nd ed. , Churchhill Livingstone, New York, 1998, pp. 73 -123. ) Brain tumors Head injury Subdural or epidural hematomas Subarachnoid hemorrhage (ruptured aneurysm) Arteriovenous malformation Hydrocephalus
 Many other disease processes can also increase ICP: (Sulek CA, ibid. ) Stroke Hypoxic injury Pseudotumor cerebri (impaired CSF reabsorption) Hepatic encephalopathy Eclampsia Venous sinus thrombosis Inflammatory processes Reye syndrome (cerebral edema in children after infection)
Many other disease processes can also increase ICP: (Sulek CA, ibid. ) Stroke Hypoxic injury Pseudotumor cerebri (impaired CSF reabsorption) Hepatic encephalopathy Eclampsia Venous sinus thrombosis Inflammatory processes Reye syndrome (cerebral edema in children after infection)
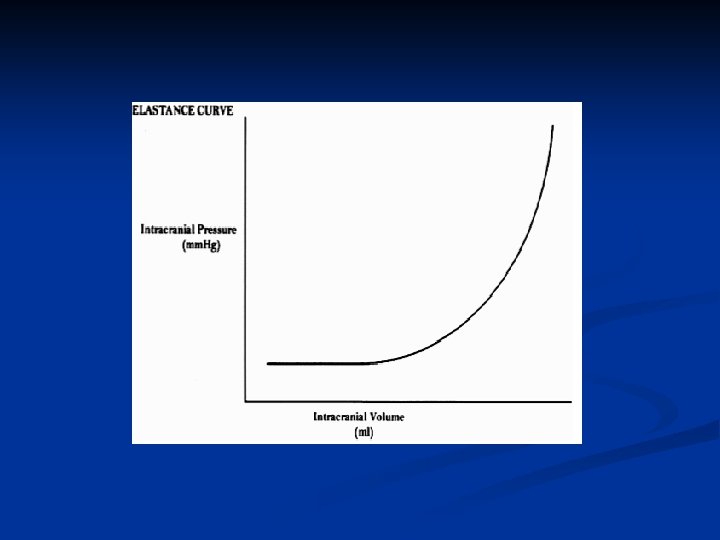
 Why is intracranial pressure (ICP) a main concern in intracranial neuroanesthesia? 1. Skull = a rigid and non-compliant container. This is the Monro-Kellie doctrine or hypothesis (Katz J. Anesthesia for the patient with elevated intracranial pressure. ASA Refresher Course Lecture 152: 1 -7, 1988). 2. Any increase in intracranial volume (ICV) tends to produce an increase in intracranial pressure (ICP). 3. Not exactly a direct 1: 1 relationship between ICV and ICP
Why is intracranial pressure (ICP) a main concern in intracranial neuroanesthesia? 1. Skull = a rigid and non-compliant container. This is the Monro-Kellie doctrine or hypothesis (Katz J. Anesthesia for the patient with elevated intracranial pressure. ASA Refresher Course Lecture 152: 1 -7, 1988). 2. Any increase in intracranial volume (ICV) tends to produce an increase in intracranial pressure (ICP). 3. Not exactly a direct 1: 1 relationship between ICV and ICP
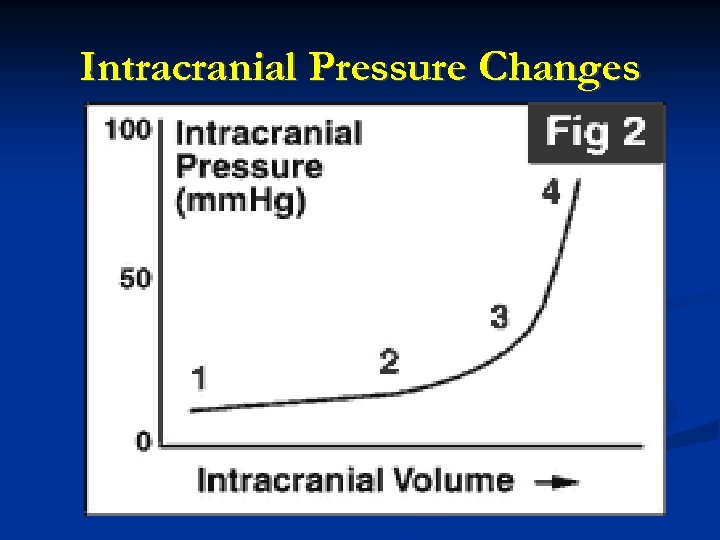 Intracranial Pressure Changes
Intracranial Pressure Changes
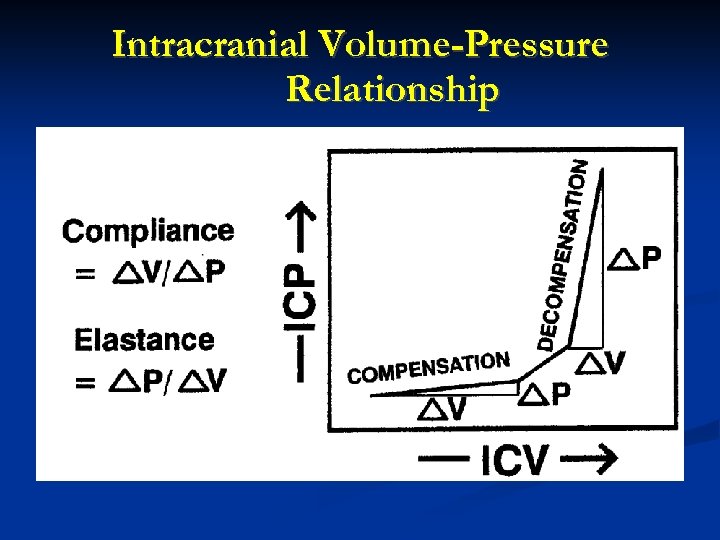 Intracranial Volume-Pressure Relationship
Intracranial Volume-Pressure Relationship
 Intracranial Volume-Pressure Relationship Compliance = change in volume / change in pressure (Think pulmonary compliance) Elastance = change in pressure / change in volume (The intracranial volume-pressure relationship is usually expressed as an elastance curve. ) 3. With compensation, fairly large increases in ICV cause only a small increase in ICP. 4. With decompensation, a further small increase in ICV causes a large increase in ICP.
Intracranial Volume-Pressure Relationship Compliance = change in volume / change in pressure (Think pulmonary compliance) Elastance = change in pressure / change in volume (The intracranial volume-pressure relationship is usually expressed as an elastance curve. ) 3. With compensation, fairly large increases in ICV cause only a small increase in ICP. 4. With decompensation, a further small increase in ICV causes a large increase in ICP.
 Mechanisms that compensate for an increase in intracranial volume Decrease CSF volume in the intracranial vault CSF is displaced into distensible spinal dural sac. Decrease CSF production relative to reabsorption Decrease intracranial blood volume (especially in dural veins)
Mechanisms that compensate for an increase in intracranial volume Decrease CSF volume in the intracranial vault CSF is displaced into distensible spinal dural sac. Decrease CSF production relative to reabsorption Decrease intracranial blood volume (especially in dural veins)
 Monro-Kellie Doctrine
Monro-Kellie Doctrine
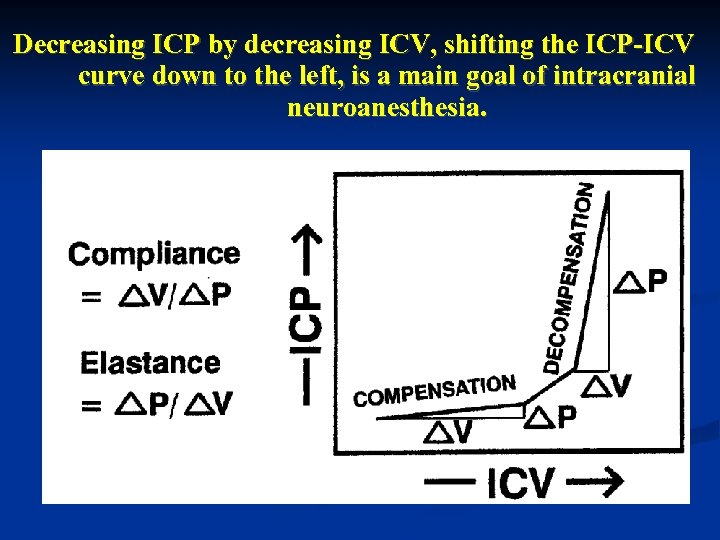 Decreasing ICP by decreasing ICV, shifting the ICP-ICV curve down to the left, is a main goal of intracranial neuroanesthesia.
Decreasing ICP by decreasing ICV, shifting the ICP-ICV curve down to the left, is a main goal of intracranial neuroanesthesia.
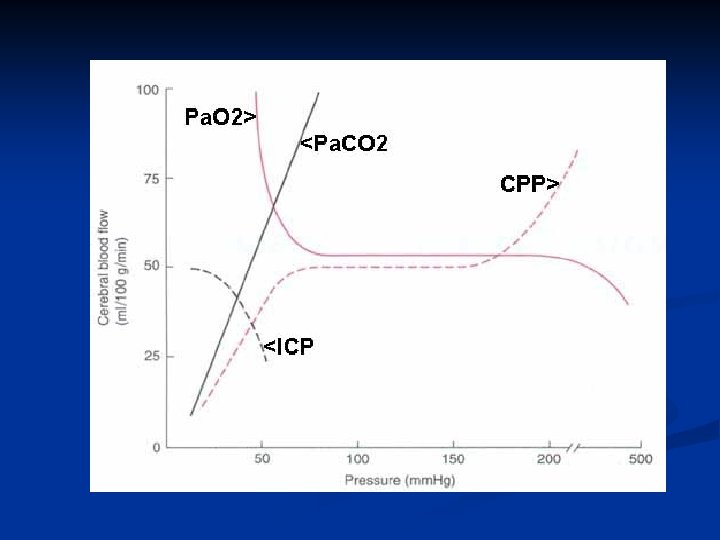
 Two main consequences of decompensation 1. Decreased cerebral perfusion pressure (CPP), resulting in cerebral ischemia. 2. Risk of herniation (Fishman RA. Brain edema. NEJOM 293: 706, 1975)
Two main consequences of decompensation 1. Decreased cerebral perfusion pressure (CPP), resulting in cerebral ischemia. 2. Risk of herniation (Fishman RA. Brain edema. NEJOM 293: 706, 1975)
 Decreased cerebral perfusion pressure (CPP), resulting in cerebral ischemia. CPP = MAP - ICP (or CVP, whichever is greater)
Decreased cerebral perfusion pressure (CPP), resulting in cerebral ischemia. CPP = MAP - ICP (or CVP, whichever is greater)
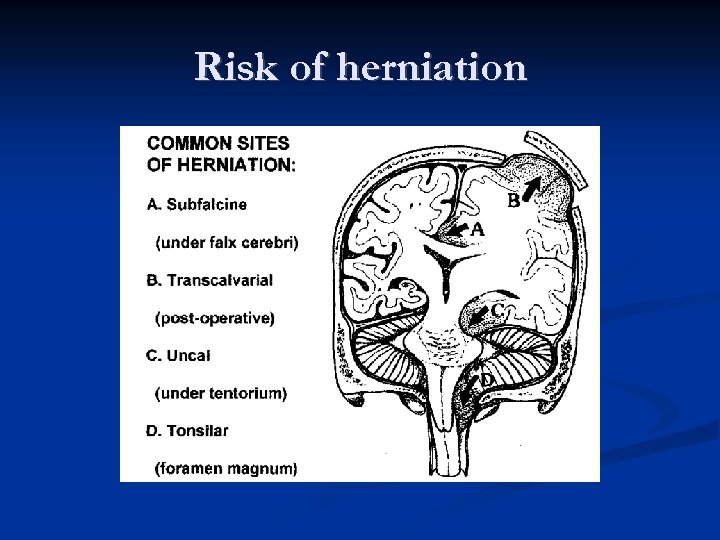 Risk of herniation
Risk of herniation
 Clinical Presentation of Increased ICP Acute vs. chronic process and location Classic clinical manifestations: (Sulek CA, ibid. ) Headache Nausea and vomiting Visual disturbances Altered mentation Papilledema Ocular palsies Other symptoms with worsening ICP include: (Shapiro HM, Drummond JC. Neurosurgical anesthesia and intracranial hypertension. In: Miller RD (ed), Anesthesia, 3 rd. ed. New York: Churchill. Livingstone, 1990, pp. 1737 -9. ) CNS - drowsiness, unconsciousness, decerebrate posturing Cardiovascular (Bendo AA, Luba K. Recent Changes in the Management of Intracranial Hypertension. International Anesthesiology Clinics 38 (4): 69 -85, 2000. ) Respiratory - abnormal breathing, apnea No clinical sign reliably indicates the ICP level.
Clinical Presentation of Increased ICP Acute vs. chronic process and location Classic clinical manifestations: (Sulek CA, ibid. ) Headache Nausea and vomiting Visual disturbances Altered mentation Papilledema Ocular palsies Other symptoms with worsening ICP include: (Shapiro HM, Drummond JC. Neurosurgical anesthesia and intracranial hypertension. In: Miller RD (ed), Anesthesia, 3 rd. ed. New York: Churchill. Livingstone, 1990, pp. 1737 -9. ) CNS - drowsiness, unconsciousness, decerebrate posturing Cardiovascular (Bendo AA, Luba K. Recent Changes in the Management of Intracranial Hypertension. International Anesthesiology Clinics 38 (4): 69 -85, 2000. ) Respiratory - abnormal breathing, apnea No clinical sign reliably indicates the ICP level.
 Cardiovascular Cushing’s response = systemic hypertension + reflex bradycardia Cushing’s triad = intracranial HTN + systemic HTN + reflex bradycardia
Cardiovascular Cushing’s response = systemic hypertension + reflex bradycardia Cushing’s triad = intracranial HTN + systemic HTN + reflex bradycardia
 Diagnosis of increased ICP Indirect diagnosis (CT scan, MRI). Findings suggestive of increased ICP include: (Bendo AA, Luba K, ibid. ) Midline shift Obliteration of basal cisterns Loss of sulci Ventricular effacement (or enlargement, as with hydrocephalus) Edema (region of hypodensity) Direct measurement of ICP Methods: Subdural bolt Subdural catheter Epidural transducer Intraparenchymal fiberoptic catheter Ventriculostomy Findings Normal ICP < 10 mm Hg Elevated ICP > 15 mm Hg
Diagnosis of increased ICP Indirect diagnosis (CT scan, MRI). Findings suggestive of increased ICP include: (Bendo AA, Luba K, ibid. ) Midline shift Obliteration of basal cisterns Loss of sulci Ventricular effacement (or enlargement, as with hydrocephalus) Edema (region of hypodensity) Direct measurement of ICP Methods: Subdural bolt Subdural catheter Epidural transducer Intraparenchymal fiberoptic catheter Ventriculostomy Findings Normal ICP < 10 mm Hg Elevated ICP > 15 mm Hg
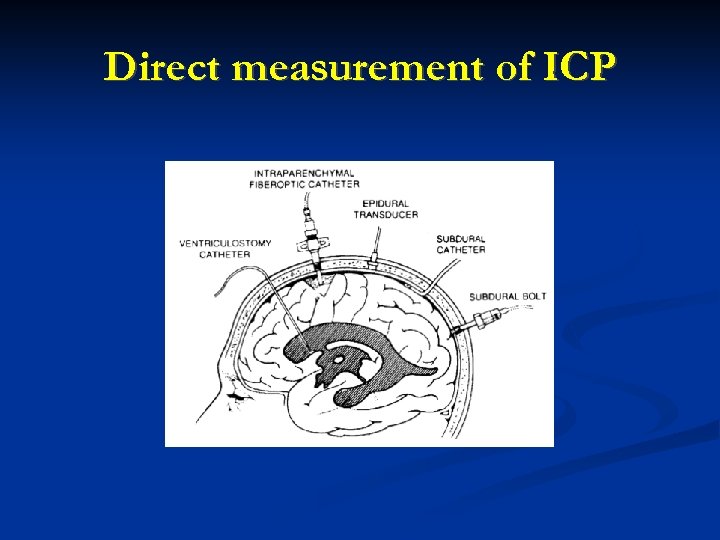 Direct measurement of ICP
Direct measurement of ICP
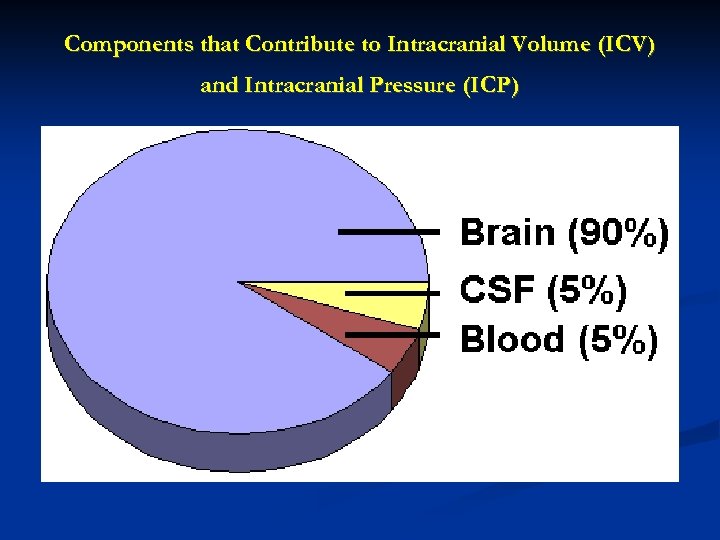 Components that Contribute to Intracranial Volume (ICV) and Intracranial Pressure (ICP)
Components that Contribute to Intracranial Volume (ICV) and Intracranial Pressure (ICP)
 Underlying Assumption Regarding Cerebral Blood Flow (CBF) and ICP • CBF = cerebral blood flow, CBV = cerebral blood volume, • ICV = intracranial volume, and • ICP = intracranial pressure
Underlying Assumption Regarding Cerebral Blood Flow (CBF) and ICP • CBF = cerebral blood flow, CBV = cerebral blood volume, • ICV = intracranial volume, and • ICP = intracranial pressure
 1. You’d assume that anything that increased CBF to the brain would be good. 2. Actually, a large increase in CBF tends to increase ICP and squish the brain, so ultimately it is bad
1. You’d assume that anything that increased CBF to the brain would be good. 2. Actually, a large increase in CBF tends to increase ICP and squish the brain, so ultimately it is bad
 Physiologic Control of Cerebral Blood Flow (CBF) Normal global CBF Average global CBF = 50 cc / 100 gm / min Grey matter CBF = 80 cc / 100 gm / min White matter CBF = 20 cc / 100 gm / min CBF = approximately 750 cc / min in adult Brain weight = 1. 5 kg, about 2% of body weight 15% of cardiac output
Physiologic Control of Cerebral Blood Flow (CBF) Normal global CBF Average global CBF = 50 cc / 100 gm / min Grey matter CBF = 80 cc / 100 gm / min White matter CBF = 20 cc / 100 gm / min CBF = approximately 750 cc / min in adult Brain weight = 1. 5 kg, about 2% of body weight 15% of cardiac output
 Major Factors in Physiologic Control of CBF Metabolic control -- CBF varies directly with brain activity and the cerebral metabolic rate of oxygen consumption, CMRO 2. • Relationships
Major Factors in Physiologic Control of CBF Metabolic control -- CBF varies directly with brain activity and the cerebral metabolic rate of oxygen consumption, CMRO 2. • Relationships
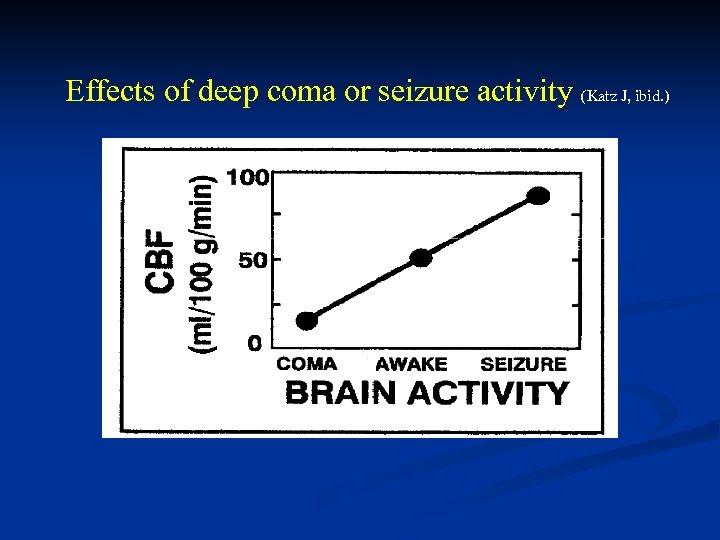 Effects of deep coma or seizure activity (Katz J, ibid. )
Effects of deep coma or seizure activity (Katz J, ibid. )
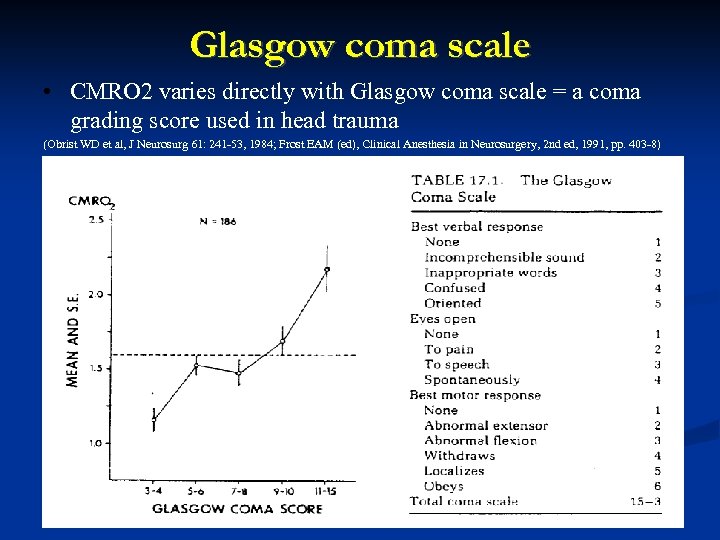 Glasgow coma scale • CMRO 2 varies directly with Glasgow coma scale = a coma grading score used in head trauma (Obrist WD et al, J Neurosurg 61: 241 -53, 1984; Frost EAM (ed), Clinical Anesthesia in Neurosurgery, 2 nd ed, 1991, pp. 403 -8)
Glasgow coma scale • CMRO 2 varies directly with Glasgow coma scale = a coma grading score used in head trauma (Obrist WD et al, J Neurosurg 61: 241 -53, 1984; Frost EAM (ed), Clinical Anesthesia in Neurosurgery, 2 nd ed, 1991, pp. 403 -8)
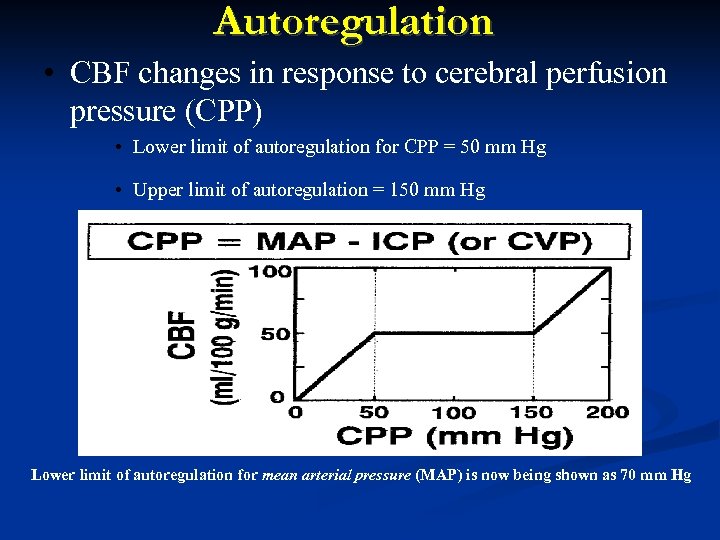 Autoregulation • CBF changes in response to cerebral perfusion pressure (CPP) • Lower limit of autoregulation for CPP = 50 mm Hg • Upper limit of autoregulation = 150 mm Hg Lower limit of autoregulation for mean arterial pressure (MAP) is now being shown as 70 mm Hg
Autoregulation • CBF changes in response to cerebral perfusion pressure (CPP) • Lower limit of autoregulation for CPP = 50 mm Hg • Upper limit of autoregulation = 150 mm Hg Lower limit of autoregulation for mean arterial pressure (MAP) is now being shown as 70 mm Hg
 Autoregulation and Chronic Hypertension
Autoregulation and Chronic Hypertension
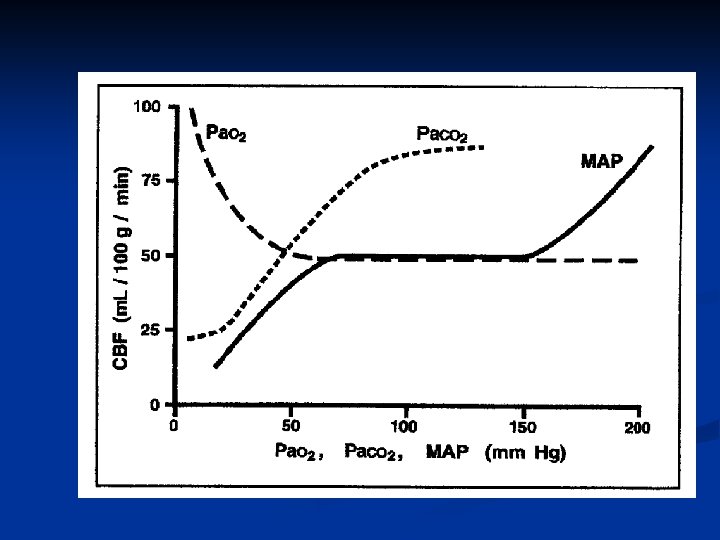
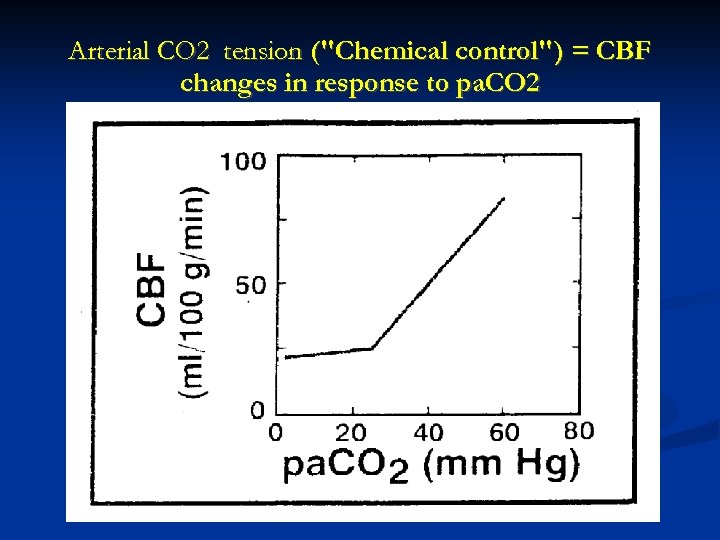 Arterial CO 2 tension ("Chemical control") = CBF changes in response to pa. CO 2
Arterial CO 2 tension ("Chemical control") = CBF changes in response to pa. CO 2
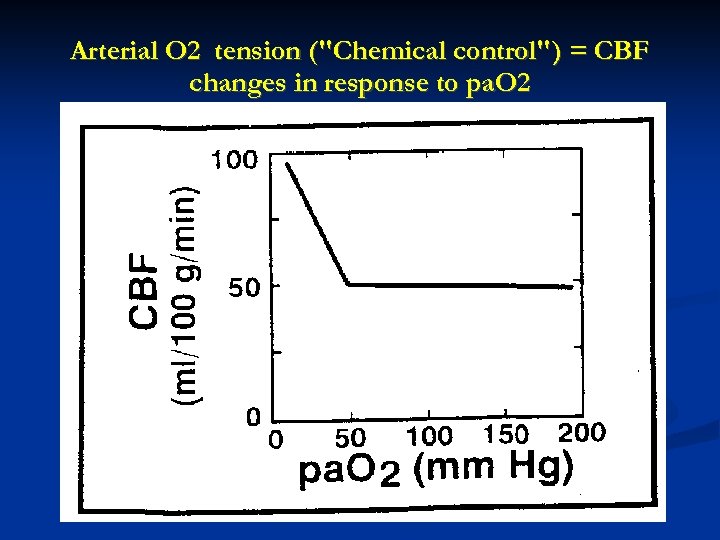 Arterial O 2 tension ("Chemical control") = CBF changes in response to pa. O 2
Arterial O 2 tension ("Chemical control") = CBF changes in response to pa. O 2
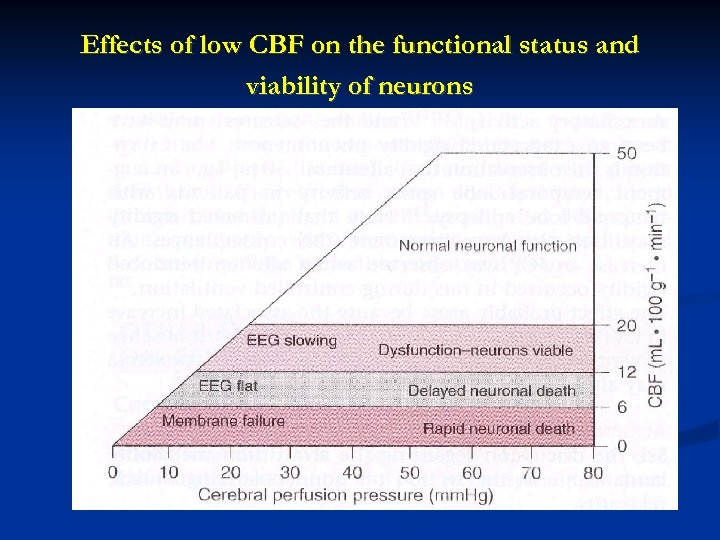 Effects of low CBF on the functional status and viability of neurons
Effects of low CBF on the functional status and viability of neurons
 Other Factors in Control of Cerebral Blood Flow (CBF) Neurogenic Control Autonomic N. S. Sympathetic stimulation Modest cerebral constriction Decrease in CBF Parasympathetic stimulation Modest cerebral dilatation Increase in CBF Central neurogenic control (less well defined) Noradrenergic pathways Cholinergic pathways Serotonergic pathways Temperature CBF changes directly related to brain activity and. CMRO 2
Other Factors in Control of Cerebral Blood Flow (CBF) Neurogenic Control Autonomic N. S. Sympathetic stimulation Modest cerebral constriction Decrease in CBF Parasympathetic stimulation Modest cerebral dilatation Increase in CBF Central neurogenic control (less well defined) Noradrenergic pathways Cholinergic pathways Serotonergic pathways Temperature CBF changes directly related to brain activity and. CMRO 2
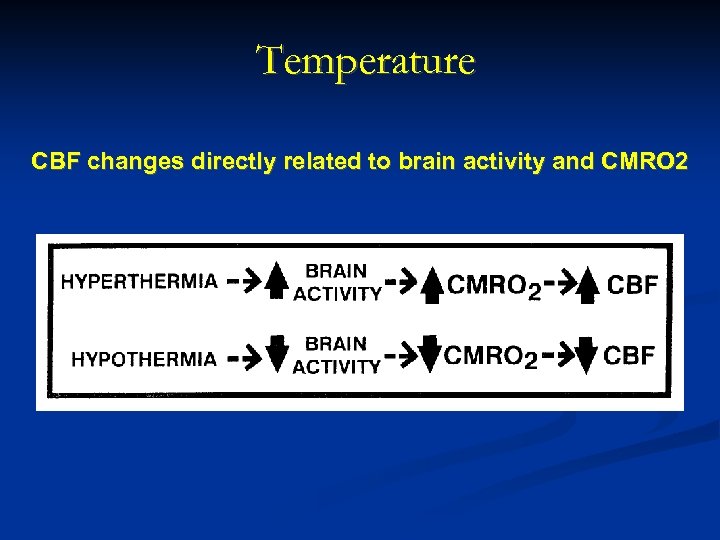 Temperature CBF changes directly related to brain activity and CMRO 2
Temperature CBF changes directly related to brain activity and CMRO 2
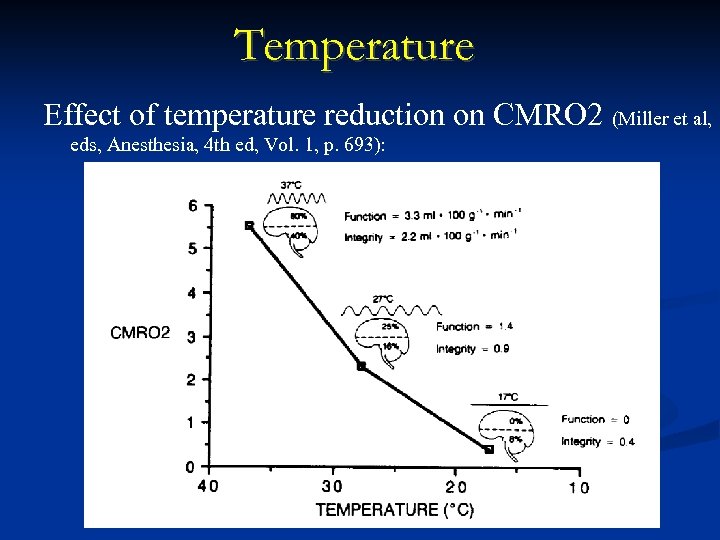 Temperature Effect of temperature reduction on CMRO 2 (Miller et al, eds, Anesthesia, 4 th ed, Vol. 1, p. 693):
Temperature Effect of temperature reduction on CMRO 2 (Miller et al, eds, Anesthesia, 4 th ed, Vol. 1, p. 693):
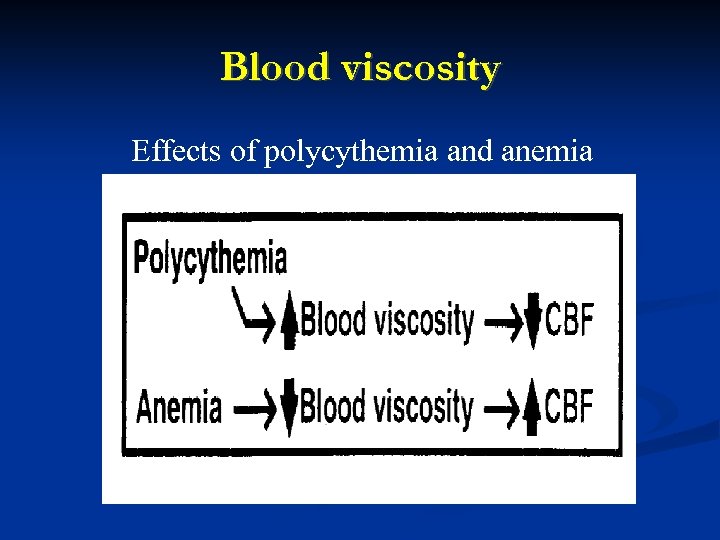 Blood viscosity Effects of polycythemia and anemia
Blood viscosity Effects of polycythemia and anemia
 A decrease in Hgb causes a compensatory: 1) Increase in CBF (according to Poiseuille’s equation, a decrease in viscosity results in a decrease in resistance, and thus an increase in flow)R = 8 Ln / (πr 4) where R = resistance, L = vessel length, n = viscosity, r = radius 2) Decrease in O 2 content (due to decreased Hgb) Ca. O 2 = (1. 39 x Hgb x FSa. O 2) + (0. 003 x pa. O 2) 3) Little change in O 2 delivery until Hgb drops less than 7 O 2 delivery = CBF x Ca. O 2
A decrease in Hgb causes a compensatory: 1) Increase in CBF (according to Poiseuille’s equation, a decrease in viscosity results in a decrease in resistance, and thus an increase in flow)R = 8 Ln / (πr 4) where R = resistance, L = vessel length, n = viscosity, r = radius 2) Decrease in O 2 content (due to decreased Hgb) Ca. O 2 = (1. 39 x Hgb x FSa. O 2) + (0. 003 x pa. O 2) 3) Little change in O 2 delivery until Hgb drops less than 7 O 2 delivery = CBF x Ca. O 2
 Patient Position Head down (Trendelenberg) Increases jugular venous pressure Decreases cerebral venous drainage Head up (reverse Trendelenberg) Decreases jugular venous pressure Increases cerebral venous drainage
Patient Position Head down (Trendelenberg) Increases jugular venous pressure Decreases cerebral venous drainage Head up (reverse Trendelenberg) Decreases jugular venous pressure Increases cerebral venous drainage
 Rules to help remember values for physiologic control of CBF "Rule of 50's" 50 cc / 100 g / min = normal global CBF 80 cc / 100 g / min = grey matter blood flow 20 cc / 100 g / min = white matter blood flow 50 mm Hg = lower limit of autoregulation for cerebral perfusion pressure (CPP) 70 mm Hg = lower limit of autoregulation for MAP 150 mm Hg = upper limit of autoregulation 50% decrease in CBF = approximate change in CBF when you hyperventilate from pa. CO 2 of 40 to 25 50 mm Hg = pa. O 2 at which hypoxic vasodilatation starts to occur 50% (actually 60%) = % of brain metabolic activity devoted to function (EEG activity) ! 50% (actually 40%) = % of brain metabolic activity devoted to maintaining brain integrity (non-EEG, basal activity) 50: 50 = distribution of CSF in skull vs. around spinal cord
Rules to help remember values for physiologic control of CBF "Rule of 50's" 50 cc / 100 g / min = normal global CBF 80 cc / 100 g / min = grey matter blood flow 20 cc / 100 g / min = white matter blood flow 50 mm Hg = lower limit of autoregulation for cerebral perfusion pressure (CPP) 70 mm Hg = lower limit of autoregulation for MAP 150 mm Hg = upper limit of autoregulation 50% decrease in CBF = approximate change in CBF when you hyperventilate from pa. CO 2 of 40 to 25 50 mm Hg = pa. O 2 at which hypoxic vasodilatation starts to occur 50% (actually 60%) = % of brain metabolic activity devoted to function (EEG activity) ! 50% (actually 40%) = % of brain metabolic activity devoted to maintaining brain integrity (non-EEG, basal activity) 50: 50 = distribution of CSF in skull vs. around spinal cord
 "Rule of 3's": 3 = components inside skull that contribute to ICP: Brain (neurons, extracellular fluid, and glia) = 85% Cerebrospinal fluid (CSF) = 10% Blood volume = 5% ! 3 to 3. 5 cc / 100 gm / min =CMRO 2 (cerebral metabolic rate of oxygen consumption) 3. 5 cc / kg / min = BMR (basal metabolic rate) Accordingly, 3. 5 x 70 kg = approx. 250 cc O 2 / min 3 minutes = approx. time to form 1 cc of CSF (in adults) CSF formation rates: 0. 33 cc / min 20 cc / hour 1 cc / 3 min 480 cc / day 3 X = CSF turnover in 1 day 480 cc / 3 X = 160 cc volume of CSF
"Rule of 3's": 3 = components inside skull that contribute to ICP: Brain (neurons, extracellular fluid, and glia) = 85% Cerebrospinal fluid (CSF) = 10% Blood volume = 5% ! 3 to 3. 5 cc / 100 gm / min =CMRO 2 (cerebral metabolic rate of oxygen consumption) 3. 5 cc / kg / min = BMR (basal metabolic rate) Accordingly, 3. 5 x 70 kg = approx. 250 cc O 2 / min 3 minutes = approx. time to form 1 cc of CSF (in adults) CSF formation rates: 0. 33 cc / min 20 cc / hour 1 cc / 3 min 480 cc / day 3 X = CSF turnover in 1 day 480 cc / 3 X = 160 cc volume of CSF
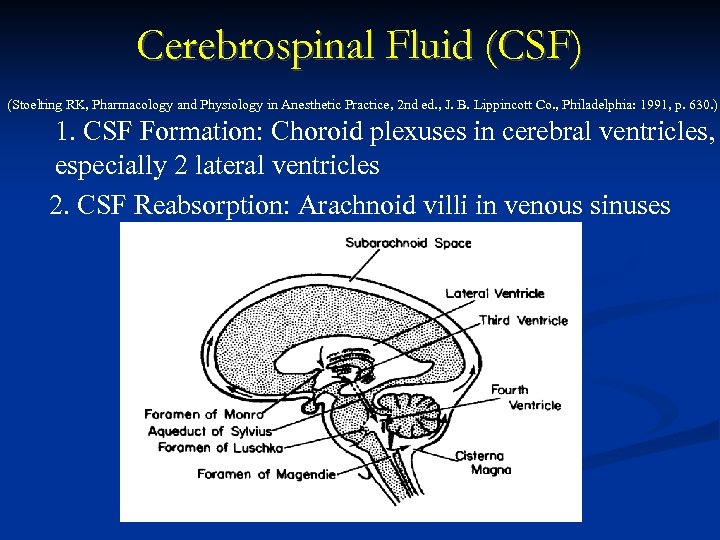 Cerebrospinal Fluid (CSF) (Stoelting RK, Pharmacology and Physiology in Anesthetic Practice, 2 nd ed. , J. B. Lippincott Co. , Philadelphia: 1991, p. 630. ) 1. CSF Formation: Choroid plexuses in cerebral ventricles, especially 2 lateral ventricles 2. CSF Reabsorption: Arachnoid villi in venous sinuses
Cerebrospinal Fluid (CSF) (Stoelting RK, Pharmacology and Physiology in Anesthetic Practice, 2 nd ed. , J. B. Lippincott Co. , Philadelphia: 1991, p. 630. ) 1. CSF Formation: Choroid plexuses in cerebral ventricles, especially 2 lateral ventricles 2. CSF Reabsorption: Arachnoid villi in venous sinuses
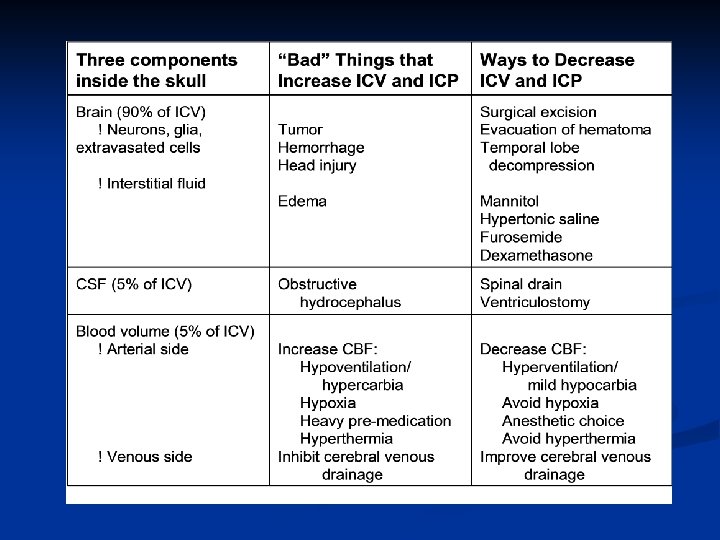
 Ways to Control Intracranial Hypertension Decrease brain bulk Mannitol Mechanism: osmotic diuretic (as is hypertonic saline) Dose: 0. 25 to 1 gm/kg IV Caution: Administer slowly over 10 min. Mannitol-induced hypotension could increase ICP. Onset of action = 10 -15 min Duration. Effects on ICP last only 2 -3 hours. Furosemide Mechanism: loop diuretic Dose: 0. 5 to 1 mg/kg alone or 0. 15 to 0. 3 mg/kg with mannitol Cautions electrolyte abnormalities Hypokalemia may require K+ repletion. Dexamethasone Mechanism: decreases edema around some solid brain tumors (Skjoeth J, Bjerre PK. Acta Neurol Scand 96: 167 -70, 1997). Dose: 10 -20 mg IV Onset of action: Several hours or days to decrease ICP Cautions: Causes hyperglycemia Steroid-treated head-injured patients have a worse out- come (Saul et al, J Neurosurg 54: 596, 1981; Dearden et al, J Neurosurg 64: 80, 1986).
Ways to Control Intracranial Hypertension Decrease brain bulk Mannitol Mechanism: osmotic diuretic (as is hypertonic saline) Dose: 0. 25 to 1 gm/kg IV Caution: Administer slowly over 10 min. Mannitol-induced hypotension could increase ICP. Onset of action = 10 -15 min Duration. Effects on ICP last only 2 -3 hours. Furosemide Mechanism: loop diuretic Dose: 0. 5 to 1 mg/kg alone or 0. 15 to 0. 3 mg/kg with mannitol Cautions electrolyte abnormalities Hypokalemia may require K+ repletion. Dexamethasone Mechanism: decreases edema around some solid brain tumors (Skjoeth J, Bjerre PK. Acta Neurol Scand 96: 167 -70, 1997). Dose: 10 -20 mg IV Onset of action: Several hours or days to decrease ICP Cautions: Causes hyperglycemia Steroid-treated head-injured patients have a worse out- come (Saul et al, J Neurosurg 54: 596, 1981; Dearden et al, J Neurosurg 64: 80, 1986).
 Ways to Control Intracranial Hypertension Decrease CSF Volume Ventriculostomy Lumbar drain Decrease blood volume Optimize hemodynamics (MAP, CVP, PCWP, HR) to maintain CPP = MAP - ICP (or CVP, whichever is greater) Avoid severe anemia Avoid severe hypotension, resulting in cerebral ischemia. Avoid severe hypertension, resulting in: 1) cerebral edema, 2) hemorrhage, or 3) herniation
Ways to Control Intracranial Hypertension Decrease CSF Volume Ventriculostomy Lumbar drain Decrease blood volume Optimize hemodynamics (MAP, CVP, PCWP, HR) to maintain CPP = MAP - ICP (or CVP, whichever is greater) Avoid severe anemia Avoid severe hypotension, resulting in cerebral ischemia. Avoid severe hypertension, resulting in: 1) cerebral edema, 2) hemorrhage, or 3) herniation
 Target normovolemia: Avoid under- or over-hydration. No longer “run ‘em dry. ” For normal craniotomy, Hourly maintenance + Replace urine cc for cc + Cover blood loss 3: 1 with crystalloid Avoid extreme hyperglycemia a. Assumption: If glucose = energy substrate for the brain, then an increase in plasma glucose should be beneficial. b. Actually, elevated plasma glucose levels are deleterious in cerebral ischemia (Wass CT, Lanier WL. Mayo Clin Proc 71: 801 -12, 1996). In animal models, hypoglycemia at the onset of ischemia worsens outcome. In humans, hyperglycemia is associated with a worsening of postischemia brain injury. Why? Intracellular acidosis injures neurons and glia. Common sense goals for glucose management: Monitor glucose levels to maintain “normoglycemia” as rigidly as possible. Avoid IV glucose infusions unless indicated.
Target normovolemia: Avoid under- or over-hydration. No longer “run ‘em dry. ” For normal craniotomy, Hourly maintenance + Replace urine cc for cc + Cover blood loss 3: 1 with crystalloid Avoid extreme hyperglycemia a. Assumption: If glucose = energy substrate for the brain, then an increase in plasma glucose should be beneficial. b. Actually, elevated plasma glucose levels are deleterious in cerebral ischemia (Wass CT, Lanier WL. Mayo Clin Proc 71: 801 -12, 1996). In animal models, hypoglycemia at the onset of ischemia worsens outcome. In humans, hyperglycemia is associated with a worsening of postischemia brain injury. Why? Intracellular acidosis injures neurons and glia. Common sense goals for glucose management: Monitor glucose levels to maintain “normoglycemia” as rigidly as possible. Avoid IV glucose infusions unless indicated.
 NB: Drug infusions Use lactated Ringer’s (LR) with caution. Converted to glucose in the liver Slightly hypotonic (vs. slightly hypertonic NSS) “Big Miller recommends resuscitation with NSS + LR (50: 50) Use insulin to treat hyperglycemia. Don’t allow sustained glucose > 250 (“Big Miller”). Avoid rapid fluid shifts, electrolyte abnormalities, and hypoglycemia Optimize ventilation. Reasonable goals: pa. O 2 > 100 mm Hg pa. CO 2 35 mm Hg Avoid hypoventilation pre-op and intra-op. Hypoxia and hypercarbia increase CBF and ICP, resulting in brain swelling. Hyperventilation on demand? pa. CO 2 of 25 -30 may still be necessary if brain is “tight” Never go below a pa. CO 2 of 25. Can cause cerebral ischemia due to cerebral constriction in some pathologic conditions (von Helden et al, Acta Neurochir Suppl Wien 59: 102 -6, 1993. Hyperventilation is only effective to decrease ICP for 6 -8 hours
NB: Drug infusions Use lactated Ringer’s (LR) with caution. Converted to glucose in the liver Slightly hypotonic (vs. slightly hypertonic NSS) “Big Miller recommends resuscitation with NSS + LR (50: 50) Use insulin to treat hyperglycemia. Don’t allow sustained glucose > 250 (“Big Miller”). Avoid rapid fluid shifts, electrolyte abnormalities, and hypoglycemia Optimize ventilation. Reasonable goals: pa. O 2 > 100 mm Hg pa. CO 2 35 mm Hg Avoid hypoventilation pre-op and intra-op. Hypoxia and hypercarbia increase CBF and ICP, resulting in brain swelling. Hyperventilation on demand? pa. CO 2 of 25 -30 may still be necessary if brain is “tight” Never go below a pa. CO 2 of 25. Can cause cerebral ischemia due to cerebral constriction in some pathologic conditions (von Helden et al, Acta Neurochir Suppl Wien 59: 102 -6, 1993. Hyperventilation is only effective to decrease ICP for 6 -8 hours
 Temperature control Avoid hyperthermia. Increases brain metabolism, CMRO 2, and CBF Worsens outcome after cerebral ischemia in animal experiments (Minamisawa et al, Ann Neurol 28: 26 - 33, 1990) Intra-op moderate hypothermia (34 -35 o. C)? Decreases ICP in head-injured patients by decreasing brain metabolism, CBF, CBV, and CSF production. Multicenter IHAST trial tested if beneficial for intracranial aneurysm surgery (Hindman BJ et al, Neurosurgery 44: 23, 1999). Published results of completed trial indicate no better outcome with hypothermia (33. 5 o. C) than normothermia (36. 5 o. C) (Todd MM et al, N Engl J Med 2005; 352: 135 -45. ) Position to improve cerebral venous return Neutral position Head up
Temperature control Avoid hyperthermia. Increases brain metabolism, CMRO 2, and CBF Worsens outcome after cerebral ischemia in animal experiments (Minamisawa et al, Ann Neurol 28: 26 - 33, 1990) Intra-op moderate hypothermia (34 -35 o. C)? Decreases ICP in head-injured patients by decreasing brain metabolism, CBF, CBV, and CSF production. Multicenter IHAST trial tested if beneficial for intracranial aneurysm surgery (Hindman BJ et al, Neurosurgery 44: 23, 1999). Published results of completed trial indicate no better outcome with hypothermia (33. 5 o. C) than normothermia (36. 5 o. C) (Todd MM et al, N Engl J Med 2005; 352: 135 -45. ) Position to improve cerebral venous return Neutral position Head up
 Summary: Methods to Control Intracranial Hypertension Decrease Brain Bulk Mannitol Furosemide Dexamethasone (solid tumors) Decrease CSF Volume Ventriculostomy Spinal drain Decrease blood volume Optimize hemodynamics Target normovolemia Avoid extreme hyperglycemia Optimize ventilation Avoid hyperthermia Improve cerebral venous return
Summary: Methods to Control Intracranial Hypertension Decrease Brain Bulk Mannitol Furosemide Dexamethasone (solid tumors) Decrease CSF Volume Ventriculostomy Spinal drain Decrease blood volume Optimize hemodynamics Target normovolemia Avoid extreme hyperglycemia Optimize ventilation Avoid hyperthermia Improve cerebral venous return


