b1dba2ee9b533b650339ea9fae93982e.ppt
- Количество слайдов: 95

NDA 21 -600 Marqibo (Vincristine Sulfate Liposomes Injection) Treatment of patients with aggressive non -Hodgkin’s lymphoma previously treated with at least two combination chemotherapy regimens P-1

External Consultants Fernando Cabanillas, MD Clinical Professor of Medicine MD Anderson Cancer Center Clinician and Presenter Medical Director Auxilio Mutuo Cancer Center Jane Winter, MD Professor of Medicine Department of Hematology/Oncology Northwestern University Clinician 2 nd largest site 2

External Consultants (Continued) Randy Gascoyne, MD Pathologist and Clinical Professor British Columbia Cancer Agency Lymphoma Pathologist Scott Gazelle, MD, MPH, Ph. D Associate Professor Massachusetts General Hospital Radiologist, Independent Review Panel Sandra Chica, MD Medical Director - Radiologist Perceptive Informatics, Inc. (Parexel) Radiologist 3

External Consultants (Continued) Shayne Gad, Ph. D, DABT, AST Gad Consulting Services Toxicologist Jean-Marie Houle, Ph. D Houlemiron BC Enterprises Inc. Pharmacokineticist Louis Gura, MS Three Flags Consulting Statistician 4

Unmet Medical Need in Aggressive NHL Fernando Cabanillas, MD Clinical Professor of Medicine MD Anderson Cancer Center Medical Director Auxilio Mutuo Cancer Center P-5

Overview of NHL Ÿ NHLs broadly classified as aggressive vs. indolent Ÿ Aggressive NHL – 35 -40% of NHL – Diffuse large B-cell lymphoma, peripheral T-cell lymphoma Ÿ DLBCL frequently presents with divergent histologies – Treatment is driven by the most aggressive histology – Response is measured the same way – At relapse, life expectancy measured in months Ÿ Indolent NHL – At relapse, life expectancy measured in years Ÿ No new agents approved for aggressive NHL in last 17 years 6

Overview of Aggressive NHL ŸFirst-line therapy – R-CHOP therapy cures 50% of aggressive B-cell NHL ŸSecond-line therapy – If <65 years, 20% cured with high dose chemo and ASCT (only if response to salvage therapy) – If 65 years or if ASCT not feasible, 10% curable Ÿ Median survival 6 months ŸResponse rates and duration drop with each relapse 7

Overview of Aggressive NHL (Continued) ŸThird-line or later therapy (indicated population) – 10, 000 – 15, 000 patient prevalence in 2001 – No standard therapy – Bone marrow frequently compromised, thus fewer options – Reduction in tumor burden associated with symptom improvement – Results are dismal, complete responses are rarely achieved and survival is short 8
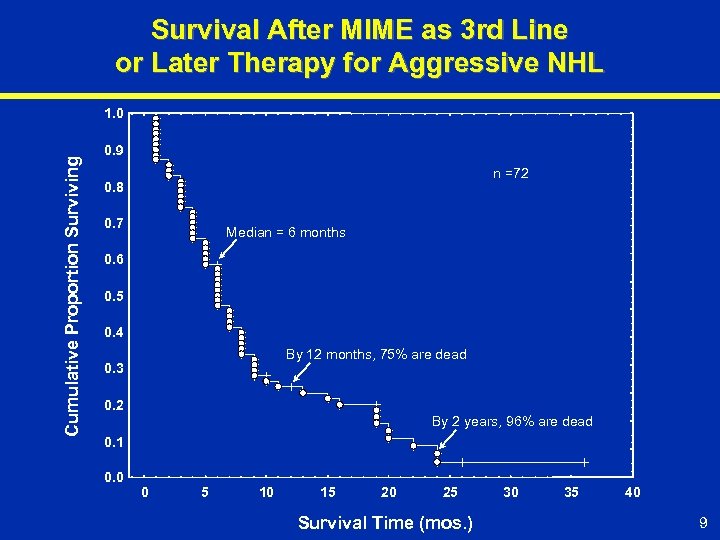
Survival After MIME as 3 rd Line or Later Therapy for Aggressive NHL Cumulative Proportion Surviving 1. 0 0. 9 n =72 0. 8 0. 7 Median = 6 months 0. 6 0. 5 0. 4 By 12 months, 75% are dead 0. 3 0. 2 By 2 years, 96% are dead 0. 1 0. 0 0 5 10 15 20 25 Survival Time (mos. ) 30 35 40 9

FDA Single Agent Papers Not Adequate for Comparison to VSLI P-10

FDA Papers for Comparison to VSLI 11

Single Agent Rituximab in Aggressive NHL 12

Combination Regimens as 3 rd Line or Later Therapy 13

Unmet Clinical Needs Ÿ Patients who don’t qualify for aggressive combination regimens or who have relapsed after ASCT – – – Poor marrow function Age >65 Poor performance status No response to pre-transplant salvage therapy Co-morbidities Ÿ Patients with compromised marrow function – Rituximab no longer a viable alternative Ÿ No compelling literature evidence for “available therapy” after 2 nd relapse Ÿ Need an agent that can provide clinically meaningful benefit without excessive toxicity 14

Pharmacology Presentation Tom Madden, Ph. D Senior Director, Technology Development and Licensing Inex Pharmaceuticals Corporation P-15
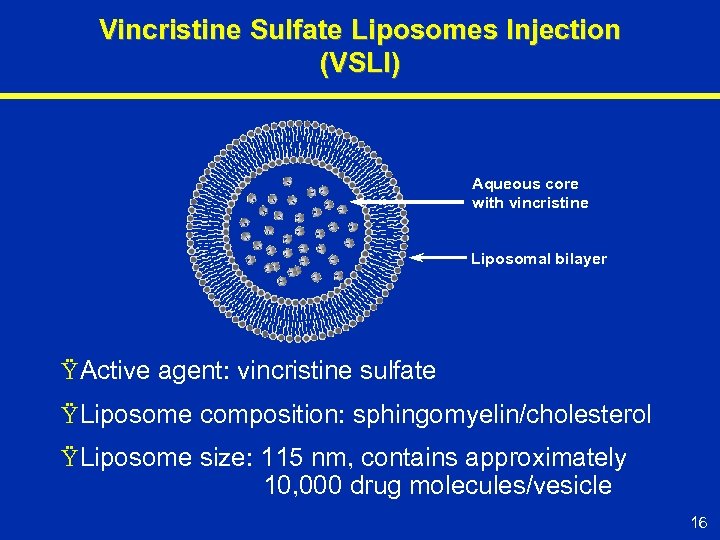
Vincristine Sulfate Liposomes Injection (VSLI) Aqueous core with vincristine Liposomal bilayer Ÿ Active agent: vincristine sulfate Ÿ Liposome composition: sphingomyelin/cholesterol Ÿ Liposome size: 115 nm, contains approximately 10, 000 drug molecules/vesicle 16

VSLI: Product Rationale VSLI Increases Tumor Exposure to Vincristine • Higher vincristine tumor levels due to preferential delivery • Longer duration of exposure due to slow vincristine release 17
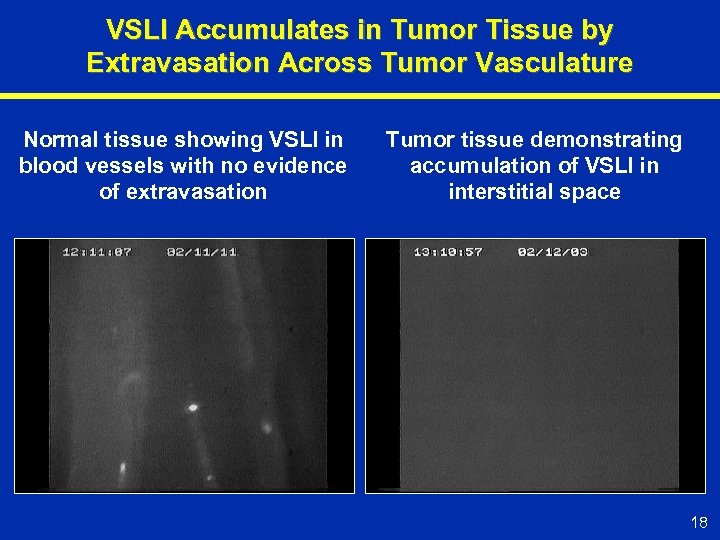
VSLI Accumulates in Tumor Tissue by Extravasation Across Tumor Vasculature Normal tissue showing VSLI in blood vessels with no evidence of extravasation Tumor tissue demonstrating accumulation of VSLI in interstitial space 18
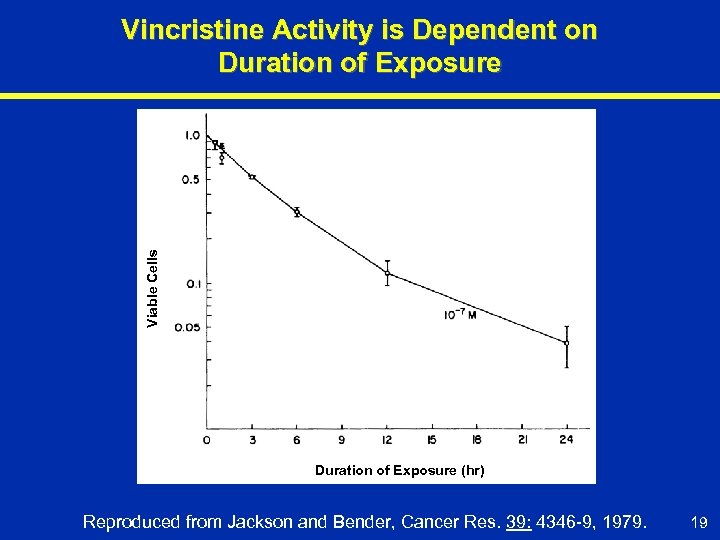
Viable Cells Vincristine Activity is Dependent on Duration of Exposure (hr) Reproduced from Jackson and Bender, Cancer Res. 39: 4346 -9, 1979. 19

Vincristine is Slowly Released from VSLI in Vivo (Rat Plasma) VSLI provides prolonged exposure to vincristine 20

Antitumor Activity of VSLI Is Significantly Greater Than VCR in the Namalwa Lymphoma Xenograft Model 21
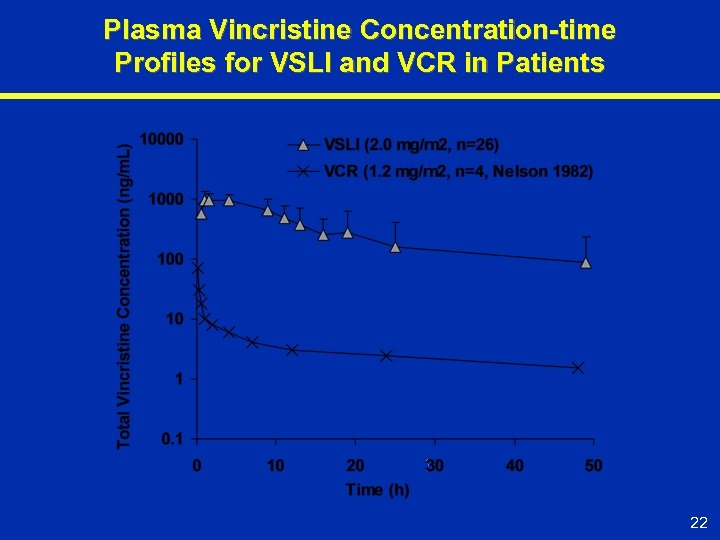
Plasma Vincristine Concentration-time Profiles for VSLI and VCR in Patients 22

VSLI Nonclinical Summary ŸCompared to conventional vincristine – VSLI provides increased tumor exposure – VSLI provides increased antitumor activity in nonclinical studies – VSLI elicits the same toxicities 23

Clinical Efficacy and Safety Alexandra Mancini, MSc. Senior Vice President, Clinical and Regulatory Affairs Inex Pharmaceuticals Corporation P-24

Efficacy Trials in Relapsed Aggressive NHL Ÿ Supportive Phase IIa Study (DM 97 -162) – NHL or ALL – Investigator-sponsored at MD Anderson Cancer Center – 92 patients with relapsed aggressive NHL Ÿ Primary Phase IIb Study (CA 99002) – International multicenter: 42 sites enrolled – 119 patients enrolled Ÿ Two largest trials in multiply relapsed aggressive NHL Ÿ Similar study designs and response criteria Ÿ Consistent results in 211 patients 25

Randomized Controlled Trial ŸPost-approval commitment to confirm clinical benefit – 3 meetings and SPA comments from FDA – Revised protocol to be resubmitted shortly – Study to start in first half 2005 26

Study Conduct Issues Raised by FDA P-27

Study Conduct Issues Raised by FDA Low Number of Eligible Patients Ÿ Numerous protocol amendments and exemptions Ÿ Low histologic eligibility rate Ÿ Incomplete staging in 19% of patients Conduct of Independent Review Panel (IRP) Ÿ Wording of response criteria Ÿ Operations of core imaging lab Ÿ Amendments to IRP Charter 28

Protocol Amendments 29

Protocol Version 5. 0 7. 0 Key Inclusion Criteria ŸPatients with a CR or CRu to 1 st line chemotherapy at least a minor response ŸPatients with at least a PR to most recent therapy deleted A poorer prognosis population 30

Other Amendments Version 7. 0 8. 0 Ÿ Peripheral T-cell lymphoma (1 pt) Ÿ Anaplastic large null-/T-cell lymphoma (2 pts) Ÿ Transformed NHL (11 pts) A poorer prognosis population Ÿ No further changes in eligibility criteria Ÿ With each amendment FDA agreed that trial population suitable for accelerated approval 31

Protocol Amendments Version 8. 0 to 9. 0 ŸAdditional CT scans scheduled 4 weeks after first response instead of the original 8 weeks ŸA clarification changed wording that these confirmatory CT scans “should” instead of “must” be obtained 32

Enrollment Exemptions ŸCareful to not allow exemptions that would have enhanced apparent VSLI response rate A poorer prognosis population 33

Histologic Eligibility Ÿ 19% ineligible by retrospective Central Review – Mostly indolent lymphomas ŸFDA excluded additional 7 patients described as ‘probably eligible’ by Central Review ŸNot protocol violations or due to amendments – Site pathology defined them as eligible for enrollment 34

Histologic Eligibility by Central Pathology Review (ITT) 35

Other FDA Eligibility Exclusions ŸStage of disease was not an eligibility criterion 1 0 0 0 36

Response Criteria Wording ŸIn some situations the criteria are ambiguous or silent ŸThese clarifications were undertaken to uphold the rigor of the criteria ŸTo ensure consistent interpretation in this multicenter study 37

Response Criteria Wording April 3, 2000 Meeting with FDA ŸProtocol Version 5. 0 with clarified response criteria wording ŸFDA agreed with the protocol wording ŸNo changes to response criteria since that meeting 38

Operations of Core Imaging Lab ŸFDA review noted that procedures manual was dated 1 year after review of images began ŸEarlier version in place before reviews began ŸNo changes to core lab procedures for entire IRP process 39

Amendments to IRP Charter ŸNo changes to conduct of IRP radiology and oncology reviews ŸAmendments in place before reviews began ŸA few clarifications for situations not previously anticipated requested by Dr. Scott Gazelle – Amendments documented what was done ŸAll images read in chronologic sequence and locked 40

Conclusions Regarding Study Conduct Issues Ÿ Protocol amendments and exemptions defined a population with a poorer prognosis Ÿ Histologic eligibility comparable to literature rates Ÿ Only 9 patients (8%) ineligible for efficacy evaluation due to protocol violations Ÿ IRP process was well conducted Ÿ Well defined and reliable assessment of objective response in the indicated population Ÿ Adequate and well-controlled trial 41

Pivotal Study Presentation P-42

Key Eligibility Criteria ŸAggressive de novo or transformed NHL ŸAt least 2 prior combination regimens including an anthracycline ŸAt least a minor response to 1 st line therapy 43

Key Eligibility Criteria (Continued) ŸNo maximum number of prior regimens ŸNo requirement of response to prior salvage therapies ŸNo upper limit on age ŸECOG PS 0 -3 accepted ŸGrade 1 -2 neuropathy permitted ŸGranulocytes 0. 5 x 109/L ŸPlatelets 50 x 109/L 44

VSLI Monotherapy Regimen Ÿ 2 mg/m 2 without dose capping, 1 hr IV infusion ŸRepeat every 2 weeks Ÿ 12 cycles maximum, 2 cycles after CR At least 2 x dose intensity of vincristine 45

Efficacy Endpoints and Populations Efficacy Endpoints Ÿ Primary – Objective response rate (CR + CRu + PR) Ÿ Secondary – Duration of response – Time to progression – Overall survival Efficacy Populations Ÿ Intent-to-Treat Population (ITT) (n=119) Ÿ Per-Protocol Population (PP) (n=77) 46

Efficacy Evaluations ŸInternational Workshop Criteria (Cheson et al 1999) – CTs of chest, abdomen, pelvis – 6 indicator lesions – Response does not require confirmation ŸIndependent Review Panel (IRP) – Primary efficacy assessment – Blinded to site opinion of response – Independent selection of indicator lesions 47

Patient Population P-48

Extent of Prior Therapy (ITT) • Mean: 3. 8; Median: 3 • Predominantly at 4 th-5 th line • 33% had prior ASCT 49

Response to Prior Therapy Ÿ 75% received a combination regimen as last therapy 50

Sensitivity to Last Qualifying Therapy (ITT) 51

Efficacy Data (ITT) P-52

Objective Response Rate (ITT) by IRP 53

Objective Response Rate Comparisons 54

Is Objective Response Likely to Predict Clinical Benefit? P-55

Symptom Improvement in Responders Ÿ 8 patients with CR or CRu – 3 patients asymptomatic – Remaining 5 patients either had resolution of symptoms or improved ECOG PS Ÿ 22 patients with PR – 15 had improvements in symptoms or ECOG PS 56

Time-to-Event Endpoints P-57

Duration of Response 58

Time to Progression (ITT) by IRP 59

Survival (ITT) Censored Observations Median 6. 7 months 25% alive at 2 years Patients at Risk 119 68 47 41 35 26 15 5 0 60

Subgroup Analyses P-61

ORR by Number of Prior Regimens and Sensitivity to Last Qualifying Regimen 62

Consistent Results in Both Studies (n=211) 63

Univariate Analyses Objective Response Rate 64

VSLI Efficacy Compared to Single-Agent Rituximab 65

Objective Tumor Response by IRP Single-Agent Rituximab as Last Therapy 66

Safety Data P-67

Extent of Treatment with VSLI (ITT) 68

Safety – Major Endpoints Ÿ 14% of patients withdrawn due to associated AEs, mostly neuropathy ŸNo treatment-associated deaths 69

Neuropathy P-70

Prior Neurotoxic Therapies (ITT) 71

Worst Neuropathy on Study by Grade at Study Entry (Pain, Paresthesia, Numbness, Weakness, Constipation) 72

Mean Change from Baseline for Cycles 1 to 6 for Hand Numbness 73

Dose to Grade 3 or 4 Neuropathy (Pain, Paresthesia, Numbness, Weakness, Constipation) • Equivalent to 15 doses of conventional vincristine 74

Comparison of Neuropathy in Responders vs. Nonresponders (Numbness) 75

Timing of Antitumor Effect vs. Neuropathy ŸAntitumor activity evident early in patients who responded, usually within 2 weeks (1 st dose) – Symptomatic improvements – Reduced palpable adenopathy – Decreased LDH ŸDevelopment of neuropathy is gradual and predictable ŸInformed treatment decisions can be made before significant neuropathy develops 76

Hematologic Abnormalities P-77

Hematologic Abnormalities at Study Entry 78

Hematology CTC Grade Changes from Baseline to Worst Grade 79

Hematologic Toxicity Neutropenia • 8% Grade 4 neutropenia (<0. 5 ANC) • 3% febrile neutropenia • 2% prophylactic filgrastim usage Thrombocytopenia • 1% Grade 4 thrombocytopenia (<10 x 109/L) • 6% platelet transfusions 80

Patients with Net Clinical Benefit Fernando Cabanillas, MD Clinical Professor of Medicine MD Anderson Cancer Center Medical Director Auxilio Mutuo Cancer Center P-81

Patient Benefit Summaries ŸFDA requested patient benefit summaries to facilitate review for clinical benefit Ÿ 38 patients considered to be responders by either IRP or Investigator Ÿ 5 patients with SD (minor response) had evidence of clinical benefit ŸTotal of 43 individual patient benefit-risk assessments 82

41 Patients Had Evidence of Net Clinical Benefit Ÿ Improvement in symptoms or ECOG PS Ÿ Tumor response to VSLI that permitted stem cell transplant Ÿ Durable complete response Ÿ Durable PR or prolonged SD Ÿ Better response than with prior regimen In case studies Ÿ Improvement in laboratory parameters 83

Clinical Benefit: Symptomatic and ECOG PS Improvements 84

Clinical Benefit: Stem Cell Transplant Ÿ 6 patients were able to receive transplants after VSLI study, 5 allogeneic, 1 autologous ŸResponsiveness to VSLI therapy and maintenance of good performance status enabled consideration for transplant Ÿ 5 patients alive (1 died at 29 months) – 1 with disease (survival of 28+ months) – 4 with no evidence of disease (27+, 29+, 31+, 39+ months) 85

Case Studies of Selected Patients with Clinical Benefit P-86

Patient 35 -01 Ÿ 56 y/o F, Stage IV-B, IPI=1 Ÿ Primary mediastinal DLCL, weight loss, fever, night sweats, anemia Ÿ Prior Rx: 1) CHOP PR for 3 months; 2) ESHAP PD; 3) RICE PD Ÿ 20 cycles VSLI/38 weeks CRu of ~1 yr Ÿ Transient Grade 4 neutropenia at Cycle 5 Ÿ Attained CRu after being refractory to all prior Rx; resolution of B symptoms and anemia 87

Patient 40 -01 Ÿ 76 y/o F, DLCL-B, IPI=3 Ÿ Multiple pulmonary metastases Ÿ Low platelets (72 k) Ÿ Prior Rx: 1) CHOP CR; 2) CTX-VP 16 -DTIC-Rituxan- Pred CR Ÿ 8 cycles VSLI/14 weeks PR 8+ mos, platelets normalized Ÿ No Grade 3 -4 toxicities Ÿ Residual pulmonary nodules not changed @ 2. 5 years (fibrotic tissue? ) Ÿ Chemo-free interval of 27+ mos, a longer remission than with any prior therapies 88

Patient 33 -06 Ÿ 47 y/o M, Stage IV-B, DLCL-B, IPI=1 Ÿ Mediastinal mass and marrow involvement Ÿ Prior Rx: 1) CHOP MR; 2) RICE PD Ÿ 8 cycles VSLI/14 weeks PR 9+ mos (IRP); CR 14+ mos (INV) Ÿ No Grade 3 -4 toxicities Ÿ Alive with no evidence of disease at 30+ mos, with no subsequent therapies Ÿ Attained CR after being refractory to all prior Rx; resolution of B symptoms and anemia 89

Benefit-Risk Conclusions P-90

Summary of Patient Population ŸMedian 3 prior regimens 4 th-5 th line therapy Ÿ 33% had prior ASCT Ÿ 33% with low blood counts Ÿ 50% refractory to last therapy Ÿ 24% >70 years Ÿ 66% with elevated LDH 91

Summary of VSLI Benefits Ÿ 25% ORR in heavily pretreated patients with highly resistant disease and compromised marrow – 46% ORR in those treated on 2 nd relapse ŸClinically important ORR for this population Ÿ 22% of patients with symptomatic or ECOG PS improvement ŸMedian duration of response of 3 months, 4 months for time to progression for a population with a median survival of 7 months 92

Summary of Risks ŸNeuropathy is dose-limiting toxicity – Gradual cumulative development – Only 13% withdrew for neuropathy ŸWell tolerated compared to other agents – Low incidence of severe myelotoxicity and hospitalizations – Low incidence of severe nausea and vomiting or alopecia 93

Favorable Benefit-Risk Profile ŸSymptomatic improvement and antitumor activity evident early, allowing informed treatment decisions before significant neuropathy develops ŸFavorable benefit-risk profile for this population with no standard treatment options 94

Why Do We Need VSLI? ŸEffective and well-tolerated agent for – Patients at 3 rd line or later – Patients who don’t qualify for aggressive combination regimens or who have relapsed after ASCT – Patients with compromised marrow function ŸBenefits 1 in 4 patients with minimal toxicity 95
b1dba2ee9b533b650339ea9fae93982e.ppt