287832bbac2e09058e65d70602089b85.ppt
- Количество слайдов: 29
 Mystery of the Crooked Cell: Investigate Sickle Cell Anemia Using Gel Electrophoresis Module developed at Boston University School of Medicine Presented by Dr. Dan Murray
Mystery of the Crooked Cell: Investigate Sickle Cell Anemia Using Gel Electrophoresis Module developed at Boston University School of Medicine Presented by Dr. Dan Murray
 Outline Sickle Cell Anemia Central Dogma of Biology Genetic Code Hemoglobin Electrophoresis
Outline Sickle Cell Anemia Central Dogma of Biology Genetic Code Hemoglobin Electrophoresis
 Sickle Cell Anemia
Sickle Cell Anemia
 Sickle Cell Anemia Genetic Disease l Heterozygous individuals – carriers l Homozygous individuals – diseased Hemoglobin l Found in red blood cells l Carries oxygen to tissues SCA Results from Defective Hemoglobin l Hemoglobins stick together l Red blood cells damaged Complications from low oxygen supply to tissues l Pain, organ damage, strokes, increased infections, etc. Incidence highest among Africans and Indians l Heterozygotes protected from Malaria
Sickle Cell Anemia Genetic Disease l Heterozygous individuals – carriers l Homozygous individuals – diseased Hemoglobin l Found in red blood cells l Carries oxygen to tissues SCA Results from Defective Hemoglobin l Hemoglobins stick together l Red blood cells damaged Complications from low oxygen supply to tissues l Pain, organ damage, strokes, increased infections, etc. Incidence highest among Africans and Indians l Heterozygotes protected from Malaria
 Central Dogma of Biology
Central Dogma of Biology
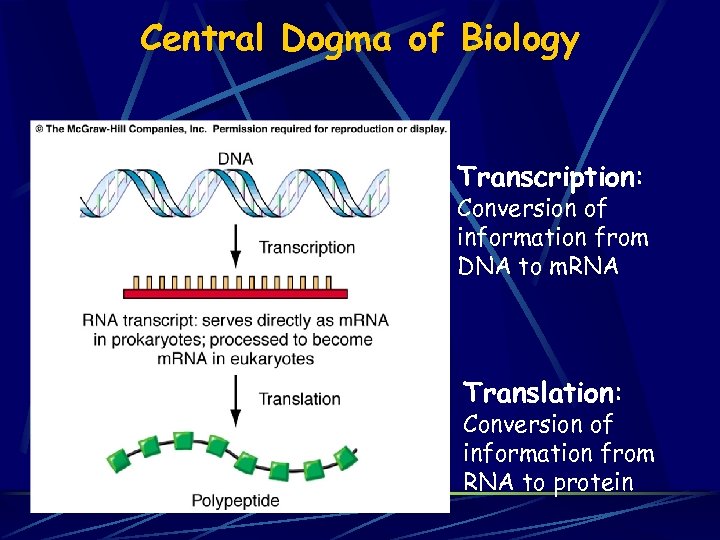 Central Dogma of Biology Transcription: Conversion of information from DNA to m. RNA Translation: Conversion of information from RNA to protein
Central Dogma of Biology Transcription: Conversion of information from DNA to m. RNA Translation: Conversion of information from RNA to protein
 The Genetic Code
The Genetic Code
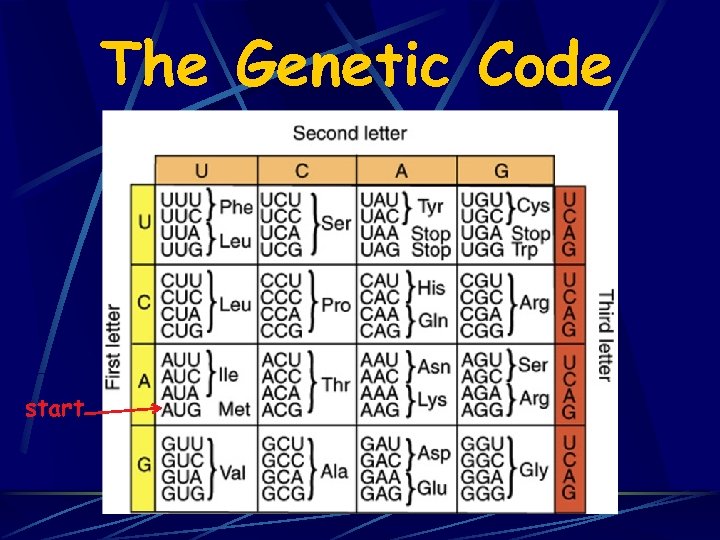 The Genetic Code start
The Genetic Code start
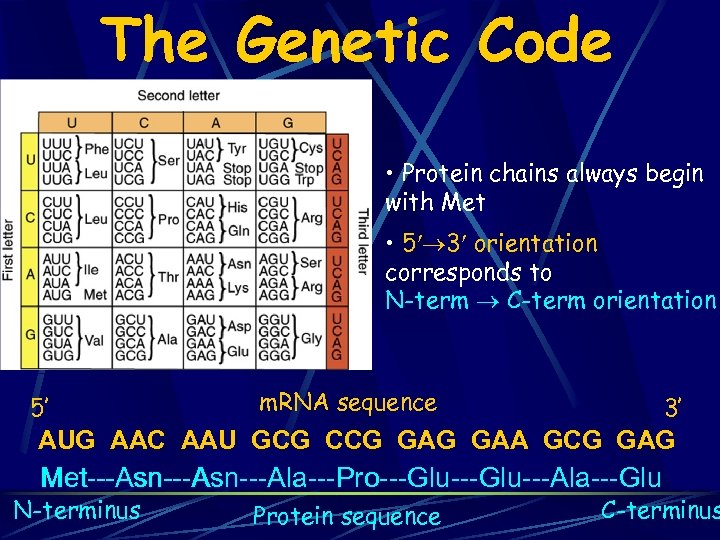 The Genetic Code • Protein chains always begin with Met • 5 3 orientation corresponds to N-term C-term orientation 5’ m. RNA sequence 3’ AUG AAC AAU GCG CCG GAA GCG GAG Met---Asn---Asn---Ala---Pro---Glu---Ala---Glu Met---Asn---Asn Met N-terminus Protein sequence C-terminus
The Genetic Code • Protein chains always begin with Met • 5 3 orientation corresponds to N-term C-term orientation 5’ m. RNA sequence 3’ AUG AAC AAU GCG CCG GAA GCG GAG Met---Asn---Asn---Ala---Pro---Glu---Ala---Glu Met---Asn---Asn Met N-terminus Protein sequence C-terminus
 Hemoglobin
Hemoglobin
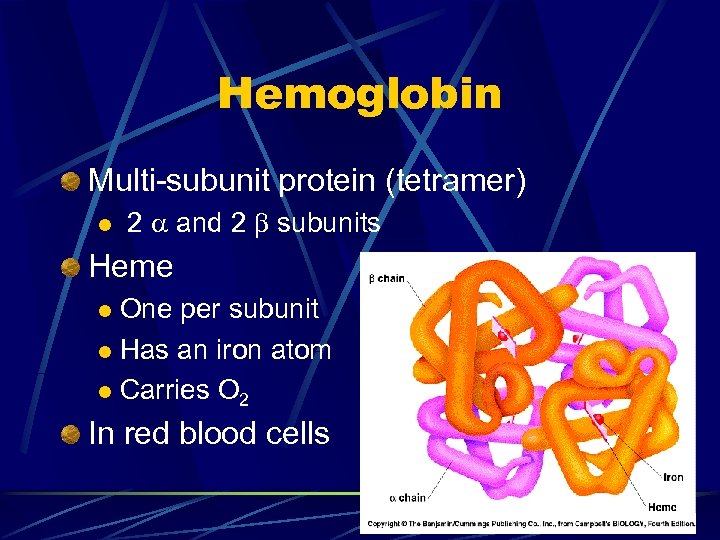 Hemoglobin Multi-subunit protein (tetramer) l 2 and 2 subunits Heme One per subunit l Has an iron atom l Carries O 2 l In red blood cells
Hemoglobin Multi-subunit protein (tetramer) l 2 and 2 subunits Heme One per subunit l Has an iron atom l Carries O 2 l In red blood cells
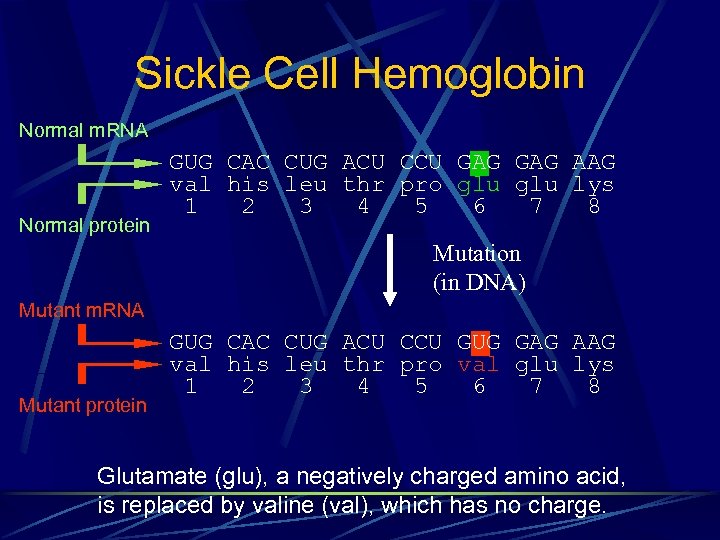 Sickle Cell Hemoglobin Normal m. RNA Normal protein GUG CAC CUG ACU CCU GAG AAG val his leu thr pro glu lys 1 2 3 4 5 6 7 8 Mutation (in DNA) Mutant m. RNA Mutant protein GUG CAC CUG ACU CCU GUG GAG AAG val his leu thr pro val glu lys 1 2 3 4 5 6 7 8 Glutamate (glu), a negatively charged amino acid, is replaced by valine (val), which has no charge.
Sickle Cell Hemoglobin Normal m. RNA Normal protein GUG CAC CUG ACU CCU GAG AAG val his leu thr pro glu lys 1 2 3 4 5 6 7 8 Mutation (in DNA) Mutant m. RNA Mutant protein GUG CAC CUG ACU CCU GUG GAG AAG val his leu thr pro val glu lys 1 2 3 4 5 6 7 8 Glutamate (glu), a negatively charged amino acid, is replaced by valine (val), which has no charge.
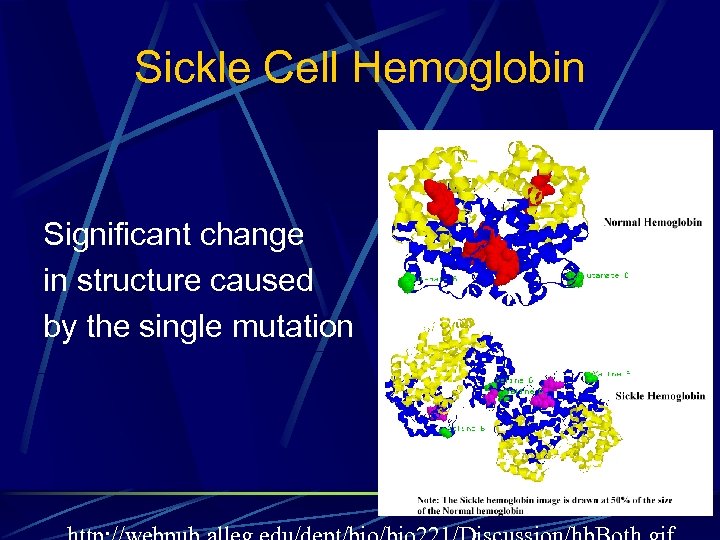 Sickle Cell Hemoglobin Significant change in structure caused by the single mutation
Sickle Cell Hemoglobin Significant change in structure caused by the single mutation
 A Possible Cure for Sickle Cell Anemia? During fetal development, a different gene (gamma) produces hemoglobin Expression of gamma gene stops naturally during development Research efforts focused on stopping silencing of gamma gene Would provide sickle cell patients with good hemoglobin
A Possible Cure for Sickle Cell Anemia? During fetal development, a different gene (gamma) produces hemoglobin Expression of gamma gene stops naturally during development Research efforts focused on stopping silencing of gamma gene Would provide sickle cell patients with good hemoglobin
 Electrophoresis
Electrophoresis
 Gel Electrophoresis Method for separating molecules (DNA, proteins, etc. ) on the basis of physical or chemical properties such as: (1) size (2) shape (3) electrical charge
Gel Electrophoresis Method for separating molecules (DNA, proteins, etc. ) on the basis of physical or chemical properties such as: (1) size (2) shape (3) electrical charge
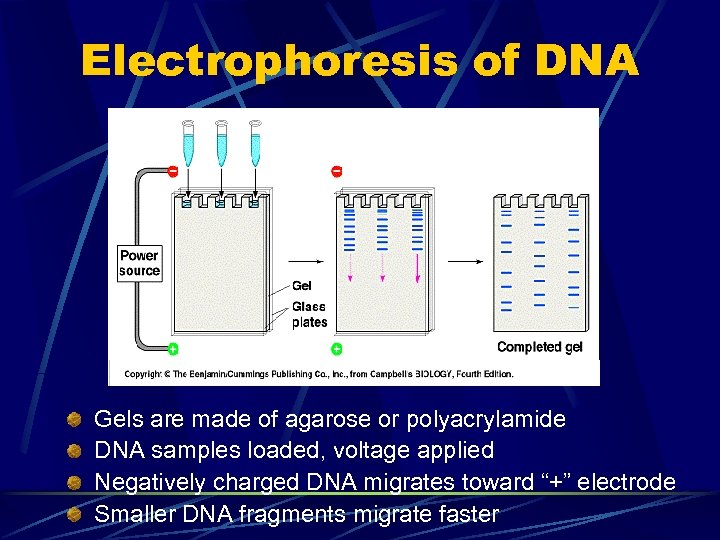 Electrophoresis of DNA Gels are made of agarose or polyacrylamide DNA samples loaded, voltage applied Negatively charged DNA migrates toward “+” electrode Smaller DNA fragments migrate faster
Electrophoresis of DNA Gels are made of agarose or polyacrylamide DNA samples loaded, voltage applied Negatively charged DNA migrates toward “+” electrode Smaller DNA fragments migrate faster
 Electrophoresis of Proteins More complex than DNA electrophoresis Different proteins have different charges l Proteins vary widely in shape l Polyacrylamide is usually the gel medium
Electrophoresis of Proteins More complex than DNA electrophoresis Different proteins have different charges l Proteins vary widely in shape l Polyacrylamide is usually the gel medium
 Protein Electrophoresis Non-Denaturing conditions Non-denaturing (native): no pretreatment of proteins before electrophoresis l l l Proteins retain normal shape Proteins retain normal charge Proteins separated on basis of charge, size, and shape Name Charge Mass Protein Q +2 30 k. D Protein R 4 42 k. D Shape
Protein Electrophoresis Non-Denaturing conditions Non-denaturing (native): no pretreatment of proteins before electrophoresis l l l Proteins retain normal shape Proteins retain normal charge Proteins separated on basis of charge, size, and shape Name Charge Mass Protein Q +2 30 k. D Protein R 4 42 k. D Shape
 Non-Denaturing Electrophoresis of Normal and Mutant Hemoglobin Charge, Size, Shape Q. Which of the above properties will be different for normal Hemoglobin (Hg. A) and mutant Hemoglobin (Hg. S)? A. Charge: Yes, Hg. A has one “–” more than Hg. S. Size: No, Hg. A and Hg. S are the same size. Shape: Yes, the shapes are different.
Non-Denaturing Electrophoresis of Normal and Mutant Hemoglobin Charge, Size, Shape Q. Which of the above properties will be different for normal Hemoglobin (Hg. A) and mutant Hemoglobin (Hg. S)? A. Charge: Yes, Hg. A has one “–” more than Hg. S. Size: No, Hg. A and Hg. S are the same size. Shape: Yes, the shapes are different.
 Migration Rates of Normal and Mutant Hemoglobin Which Hg migrates faster during electrophoresis? Normal Mutant (Hg. A) (Hg. S) Charge Size Shape Reason Hg. A has one more “ ” than Hg. S NA Amino acids Val and Glu about same size NA Hg. A more compact than Hg. S
Migration Rates of Normal and Mutant Hemoglobin Which Hg migrates faster during electrophoresis? Normal Mutant (Hg. A) (Hg. S) Charge Size Shape Reason Hg. A has one more “ ” than Hg. S NA Amino acids Val and Glu about same size NA Hg. A more compact than Hg. S
 Protein Structure
Protein Structure
 Protein Structure 1 = Primary Structure 2 = Secondary Structure 3 = Tertiary Structure 4 = Quaternary Structure
Protein Structure 1 = Primary Structure 2 = Secondary Structure 3 = Tertiary Structure 4 = Quaternary Structure
 Primary Structure Definition - Sequence of amino acids in a protein Example – Primary structure of the enzyme lysozyme: 1 2 3 4 5 126 127 128 129 Lys-Val-Phe-Gly-Arg. . . Gly-Cys-Arg-Leu Note: By convention, amino acid sequences are written starting with the amino terminus.
Primary Structure Definition - Sequence of amino acids in a protein Example – Primary structure of the enzyme lysozyme: 1 2 3 4 5 126 127 128 129 Lys-Val-Phe-Gly-Arg. . . Gly-Cys-Arg-Leu Note: By convention, amino acid sequences are written starting with the amino terminus.
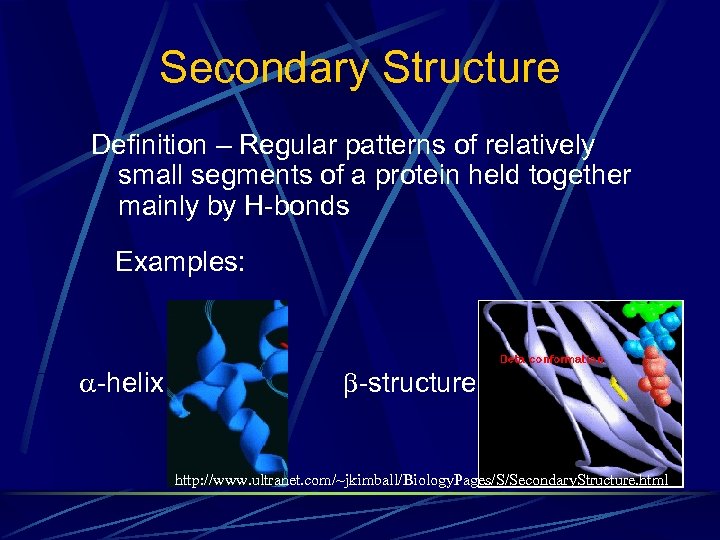 Secondary Structure Definition – Regular patterns of relatively small segments of a protein held together mainly by H-bonds Examples: -helix -structure http: //www. ultranet. com/~jkimball/Biology. Pages/S/Secondary. Structure. html
Secondary Structure Definition – Regular patterns of relatively small segments of a protein held together mainly by H-bonds Examples: -helix -structure http: //www. ultranet. com/~jkimball/Biology. Pages/S/Secondary. Structure. html
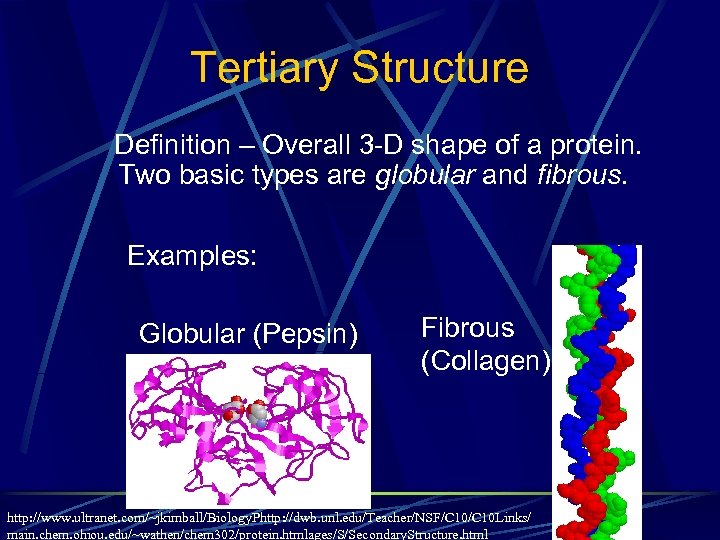 Tertiary Structure Definition – Overall 3 -D shape of a protein. Two basic types are globular and fibrous. Examples: Globular (Pepsin) Fibrous (Collagen) http: //www. ultranet. com/~jkimball/Biology. Phttp: //dwb. unl. edu/Teacher/NSF/C 10 Links/ main. chem. ohiou. edu/~wathen/chem 302/protein. htmlages/S/Secondary. Structure. html
Tertiary Structure Definition – Overall 3 -D shape of a protein. Two basic types are globular and fibrous. Examples: Globular (Pepsin) Fibrous (Collagen) http: //www. ultranet. com/~jkimball/Biology. Phttp: //dwb. unl. edu/Teacher/NSF/C 10 Links/ main. chem. ohiou. edu/~wathen/chem 302/protein. htmlages/S/Secondary. Structure. html
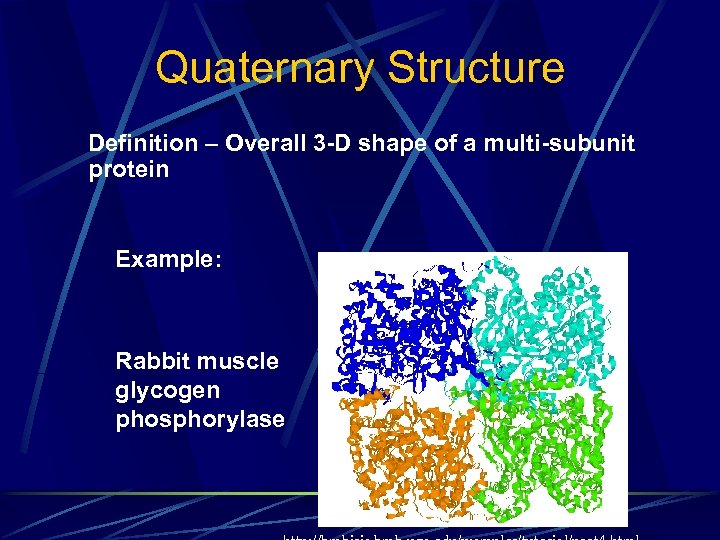 Quaternary Structure Definition – Overall 3 -D shape of a multi-subunit protein Example: Rabbit muscle glycogen phosphorylase
Quaternary Structure Definition – Overall 3 -D shape of a multi-subunit protein Example: Rabbit muscle glycogen phosphorylase
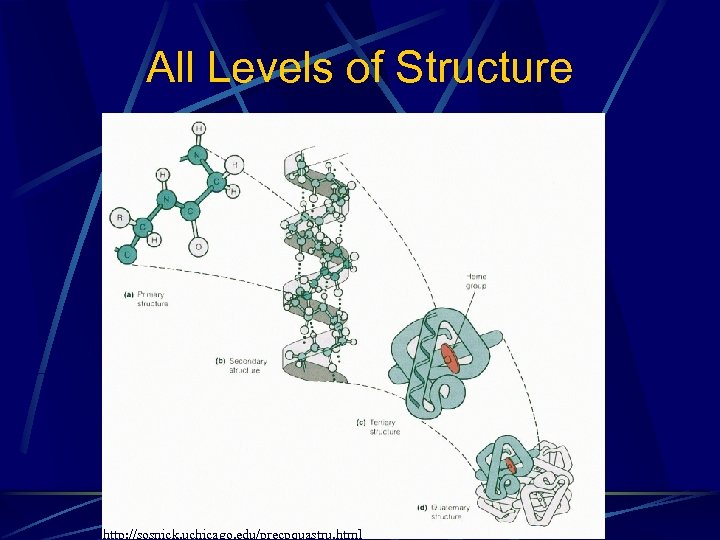 All Levels of Structure http: //sosnick. uchicago. edu/precpquastru. html
All Levels of Structure http: //sosnick. uchicago. edu/precpquastru. html
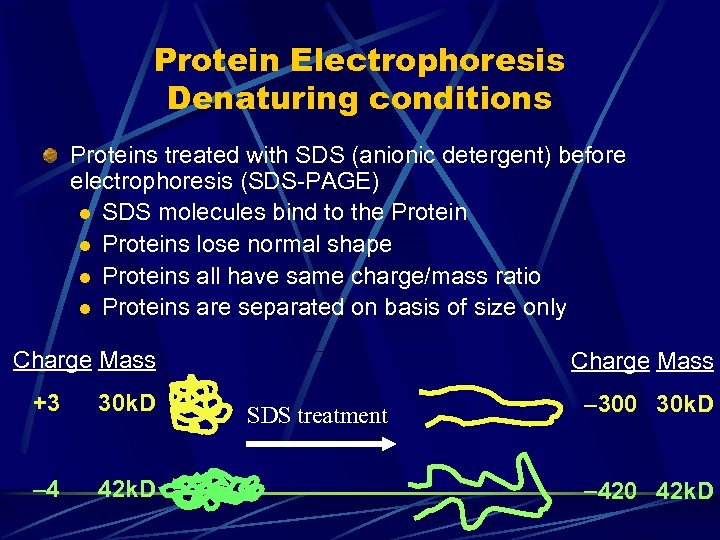 Protein Electrophoresis Denaturing conditions Proteins treated with SDS (anionic detergent) before electrophoresis (SDS-PAGE) l SDS molecules bind to the Protein l Proteins lose normal shape l Proteins all have same charge/mass ratio l Proteins are separated on basis of size only Charge Mass +3 30 k. D 4 42 k. D Charge Mass SDS treatment 300 30 k. D 420 42 k. D
Protein Electrophoresis Denaturing conditions Proteins treated with SDS (anionic detergent) before electrophoresis (SDS-PAGE) l SDS molecules bind to the Protein l Proteins lose normal shape l Proteins all have same charge/mass ratio l Proteins are separated on basis of size only Charge Mass +3 30 k. D 4 42 k. D Charge Mass SDS treatment 300 30 k. D 420 42 k. D


