ccdd73c6836e5549d0524b79fe5790a0.ppt
- Количество слайдов: 20

Multiple Myeloma in the Non-transplant Setting Antonio Palumbo, MD University of Torino, I, EU

Standard of Care for Elderly Patients

Meta-Analysis: MPT vs MP • Meta-analysis of randomized clinical trials (GIMEMA, IFM, HOVON, NMSG and TMSG) N = 1, 682 (MP, n = 868 vs MPT, n = 814) – Median overall survival (OS), 32. 7 mo vs 39. 3 mo Overall hazard ratio for OS = 0. 82 – Median progression-free survival (PFS), 14. 9 mo vs 20. 4 mo Overall hazard ratio for PFS = 0. 67 • Test of heterogeneity between studies was statistically significant for OS (p = 0. 26) and for PFS (p = 0. 23). MPT: melphalan-prednisone-thalidomide; MP: melphalan-prednisone Waage A et al. EHA 2010. Abstract 0567.
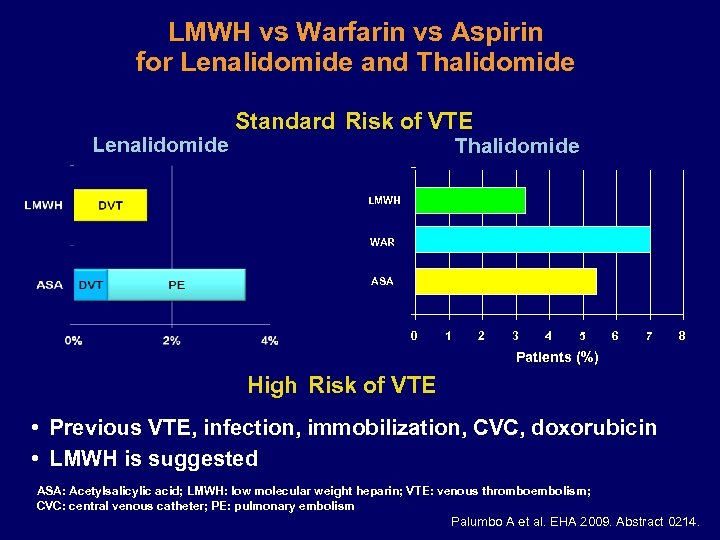
LMWH vs Warfarin vs Aspirin for Lenalidomide and Thalidomide Lenalidomide Standard Risk of VTE Thalidomide LMWH WAR ASA 0 1 2 3 4 5 6 7 8 Patients (%) High Risk of VTE • Previous VTE, infection, immobilization, CVC, doxorubicin • LMWH is suggested ASA: Acetylsalicylic acid; LMWH: low molecular weight heparin; VTE: venous thromboembolism; CVC: central venous catheter; PE: pulmonary embolism Palumbo A et al. EHA 2009. Abstract 0214.

VMP (Bortezomib/Melphalan/Prednisone) – Current Standard of Care • ~52% reduced risk of progression • ~36% reduced risk of death Progression-free survival VMP 24. 0 mos (83 events) MP 16. 6 mos (146 events) HR = 0. 483 p <. 000001 * Median follow-up 25. 9 months, median OS not reached San Miguel JF et al. ASH 2008. Abstract 650. 3 -year overall survival* VMP MP 72% 59% HR = 0. 644 p =. 0032
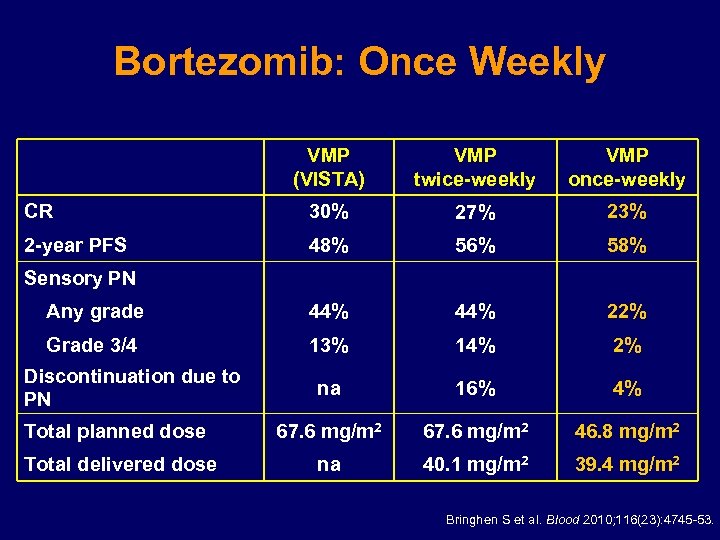
Bortezomib: Once Weekly VMP (VISTA) VMP twice-weekly VMP once-weekly CR 30% 27% 23% 2 -year PFS 48% 56% 58% Any grade 44% 22% Grade 3/4 13% 14% 2% na 16% 4% Total planned dose 67. 6 mg/m 2 46. 8 mg/m 2 Total delivered dose na 40. 1 mg/m 2 39. 4 mg/m 2 Sensory PN Discontinuation due to PN Bringhen S et al. Blood 2010; 116(23): 4745 -53.

New Treatment Options

Bortezomib-Melphalan-Prednisone-Thalidomide VMPT-VT vs VMP • 511 patients (older than 65 years) randomized from 61 Italian centers • Patients: Symptomatic multiple myeloma/end-organ damage with measurable disease • ≥ 65 yrs or < 65 yrs and not transplant-eligible; creatinine < 2. 5 mg/d. L R A N D O M I Z E VMP Cycles 1 -9 Bortezomib 1. 3 mg/m 2 IV Days 1, 8, 15, 22* Melphalan 9 mg/m 2 and prednisone 60 mg/m 2 Days 1 -4 9 x 5 -week cycles in both arms NO MAINTENANCE Until relapse VMPT MAINTENANCE Cycles 1 -9 Bortezomib 1. 3 mg/m 2 IV Days 1, 8, 15, 22* Melphalan 9 mg/m 2 and prednisone 60 mg/m 2 Days 1 -4 Thalidomide 50 mg/day continuously Bortezomib 1. 3 mg/m 2 IV Days 1, 15 Thalidomide 50 mg/day continuously *66 VMP patients and 73 VMPT-VT patients were treated with twice weekly infusions of bortezomib Palumbo A et al. J Clin Oncol 2010; 28(34): 5101 -9.

Bortezomib-Melphalan-Prednisone-Thalidomide Time to First Response and Time to CR VMPT VT VMP 100 PR: VMPT VT % of patients 80 PR: VMP 60 CR: VMPT VT 40 CR: VMP 20 0 0 5 10 15 20 25 30 Months Palumbo A et al. Proc ASH 2010; Abstract 620.

Bortezomib-Melphalan-Prednisone-Thalidomide Median follow-up 32 months Progression-free survival Time to next therapy 41% Reduced Risk of Progression 48% Reduced Risk of Progression 3 -year TNT 3 -year PFS Median PFS VMPT 51% 37. 2 months VMPT 70% Not reached VMP 32% 27. 4 months VMP 51% 37. 6 months HR 0. 59 p < 0. 0001 Median TTNT HR 0. 52 p < 0. 0001 Palumbo A et al. J Clin Oncol 2010; 28(34): 5101 -9.
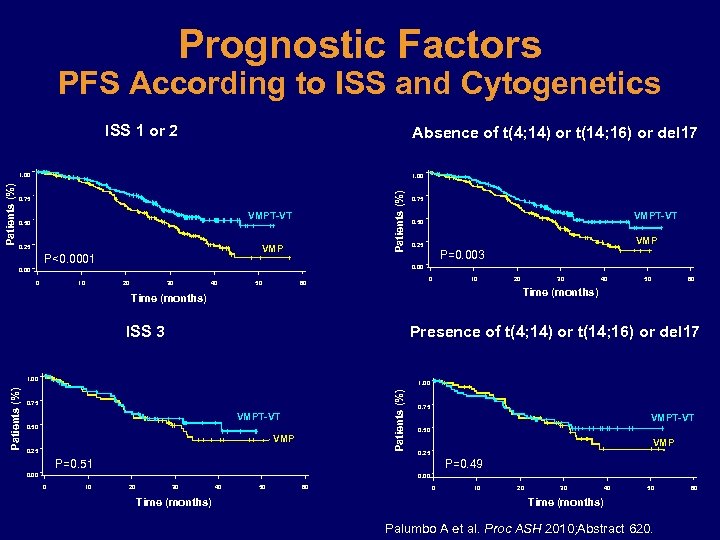
Prognostic Factors PFS According to ISS and Cytogenetics ISS 1 or 2 Absence of t(4; 14) or t(14; 16) or del 17 1. 00 Patients (%) 1. 00 0. 75 VMPT-VT 0. 50 VMP 0. 25 P<0. 0001 10 VMPT-VT 0. 50 VMP 0. 25 P=0. 003 0. 00 0 0. 75 20 30 40 50 0 60 10 30 40 50 60 Time (months) ISS 3 Presence of t(4; 14) or t(14; 16) or del 17 1. 00 Patients (%) 20 0. 75 VMPT-VT 0. 50 VMP 0. 25 P=0. 51 P=0. 49 0. 00 0 10 20 30 Time (months) 40 50 60 0 10 20 30 40 50 Time (months) Palumbo A et al. Proc ASH 2010; Abstract 620. 60
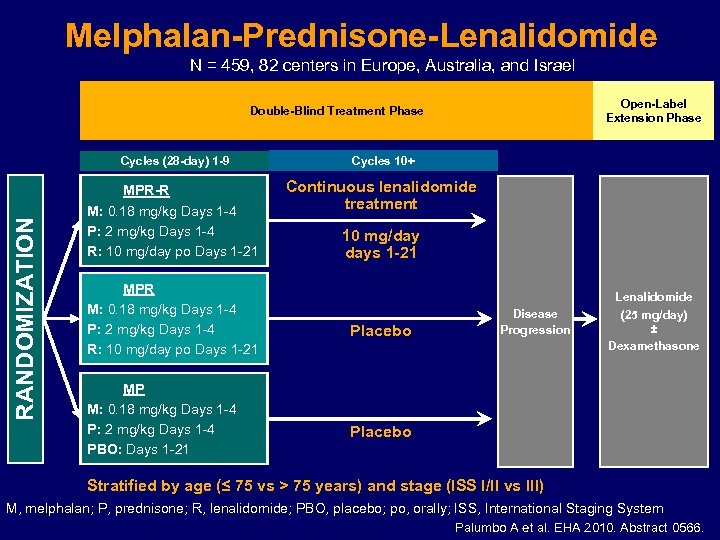
Melphalan-Prednisone-Lenalidomide N = 459, 82 centers in Europe, Australia, and Israel Open-Label Extension Phase Double-Blind Treatment Phase RANDOMIZATION Cycles (28 -day) 1 -9 Cycles 10+ MPR-R M: 0. 18 mg/kg Days 1 -4 P: 2 mg/kg Days 1 -4 R: 10 mg/day po Days 1 -21 Continuous lenalidomide treatment MPR M: 0. 18 mg/kg Days 1 -4 P: 2 mg/kg Days 1 -4 R: 10 mg/day po Days 1 -21 MP M: 0. 18 mg/kg Days 1 -4 P: 2 mg/kg Days 1 -4 PBO: Days 1 -21 10 mg/day days 1 -21 Placebo Disease Progression Lenalidomide (25 mg/day) ± Dexamethasone Placebo Stratified by age (≤ 75 vs > 75 years) and stage (ISS I/II vs III) M, melphalan; P, prednisone; R, lenalidomide; PBO, placebo; po, orally; ISS, International Staging System Palumbo A et al. EHA 2010. Abstract 0566.
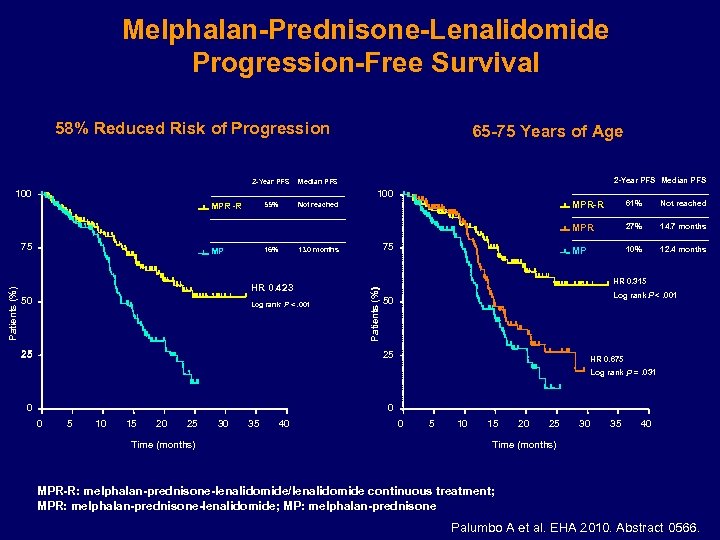
Melphalan-Prednisone-Lenalidomide Progression-Free Survival 58% Reduced Risk of Progression 2 -Year PFS Median PFS 55% 65 -75 Years of Age Not reached 100 16% MP HR 0. 423 50 Log rank P <. 001 25 61% Not reached 27% 14. 7 months MP 75 13. 0 months 10% 12. 4 months HR 0. 315 Patients (%) 75 MPR-R Log rank P <. 001 50 25 HR 0. 675 Log rank P =. 031 0 0 0 5 10 15 20 25 Time (months) 30 35 40 0 5 10 15 20 25 30 35 40 Time (months) MPR-R: melphalan-prednisone-lenalidomide/lenalidomide continuous treatment; MPR: melphalan-prednisone-lenalidomide; MP: melphalan-prednisone Palumbo A et al. EHA 2010. Abstract 0566.
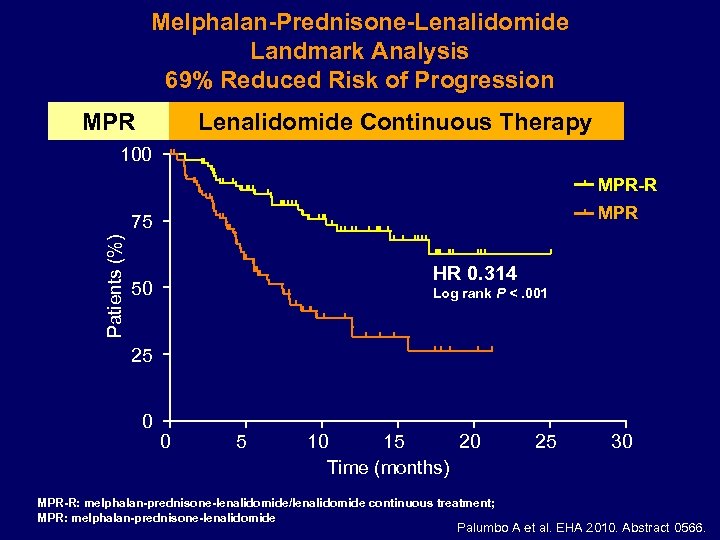
Melphalan-Prednisone-Lenalidomide Landmark Analysis 69% Reduced Risk of Progression MPR Lenalidomide Continuous Therapy 100 MPR-R MPR Patients (%) 75 HR 0. 314 50 Log rank P <. 001 25 0 0 5 10 15 20 Time (months) MPR-R: melphalan-prednisone-lenalidomide/lenalidomide continuous treatment; MPR: melphalan-prednisone-lenalidomide 25 30 Palumbo A et al. EHA 2010. Abstract 0566.

Age-Adjusted Therapies

Impact of AEs on Outcome Safety meta-analysis of 6 MPT trials 1 Median OS, MP vs MPT: 32. 7 mo vs 39. 3 mo HR = 0. 83 p = 0. 004 Median PFS, MP vs MPT: 14. 9 mo vs 20. 3 mo Estimated HR = 0. 68 p < 0. 0001 – In practice, clinicians would vary the actual dose according to patient's status, evidence of response or relapse and occurrence of side effects or toxicity. – A substantial proportion of patients in all studies either stopped thalidomide prematurely or dose reduced. MPT, melphalan-prednisone-thalidomide; AE, adverse event 1 Fayers P et al Blood 2011, in press

Frail Patients: Treatment Algorithm RISK FACTORS - Age over 75 years - Mild, moderate or severe frailty: Help needed for household and personal care - Comorbidities and organ dysfunction: Cardiac Pulmonary Hepatic Renal Go-go Moderate-go Dose level 0 Dose level -1 Dose level -2 At least one risk factor + any G 3 -4 non-hematologic AE No risk factors Slow-go Palumbo personal communication

Frail Patients: Treatment Algorithm Dose Reductions Agent Dose level 0 Dose level -1 Dose level -2 Bortezomib 1. 3 mg/m 2 twice / wk d 1, 4, 8, 11 / 3 wks 1. 3 mg/m 2 once / wk d 1, 8, 15, 22 / 5 wks 1. 0 mg/m 2 once / wk d 1, 8, 15, 22 / 5 wks Thalidomide 100 mg/d 50 mg qod Lenalidomide 25 mg/d d 1 -21 / 4 wks 10 mg/d d 1 -21 / 4 wks 40 mg/d d 1, 8, 15, 22 / 4 wks 0. 25 mg/kg d 1 -4 / 4 -6 wks 20 mg/d d 1, 8, 15, 22 / 4 wks 0. 18 mg/kg d 1 -4 / 4 -6 wks 10 mg/d d 1, 8, 15, 22 / 4 wks 0. 13 mg/kg d 1 -4 / 4 -6 wks 50 mg qod 25 mg qod 12. 5 mg qod 100 mg/d d 1 -21 / 4 wks 50 mg qod d 1 -21 / 4 wks Dexamethasone Melphalan Prednisone Cyclophosphamide Wk, week; d, day; qod, every other day Palumbo & Anderson, New Engl J Med 2011

For how many patients with MM have you used subcutaneous (SQ) bortezomib? Copyright © 2011 Research To Practice. All rights reserved.

What Clinicians Want to Know A Live CME Event Addressing the Most Common Questions and Controversies in the Current Clinical Management of Select Hematologic Cancers Sunday, June 5, 2011 7: 00 PM – 9: 30 PM Chicago, Illinois Moderator Neil Love, MD Faculty Sergio Giralt, MD John P Leonard, MD Lauren C Pinter-Brown, MD Antonio Palumbo, MD Susan M O’Brien, MD Professor Michael Hallek Copyright © 2011 Research To Practice. All rights reserved.
ccdd73c6836e5549d0524b79fe5790a0.ppt