94d2b4021aa4931f1f859b6163f8d167.ppt
- Количество слайдов: 23

Multi-drug Resistant Tuberculosis MDR-TB Leah Erenrich Advisor: Dr. Rapp

MDR-TB Defined • Multi-drug resistant Tuberculosis is defined as “strains of Mycobacterium tuberculosis that are resistant to both isoniazid and rifampin with or without resistance to other drugs” (Blondal 2007). • Second-line antibiotics => much more expensive, more toxic, are difficult to sterilize, and are much less effective
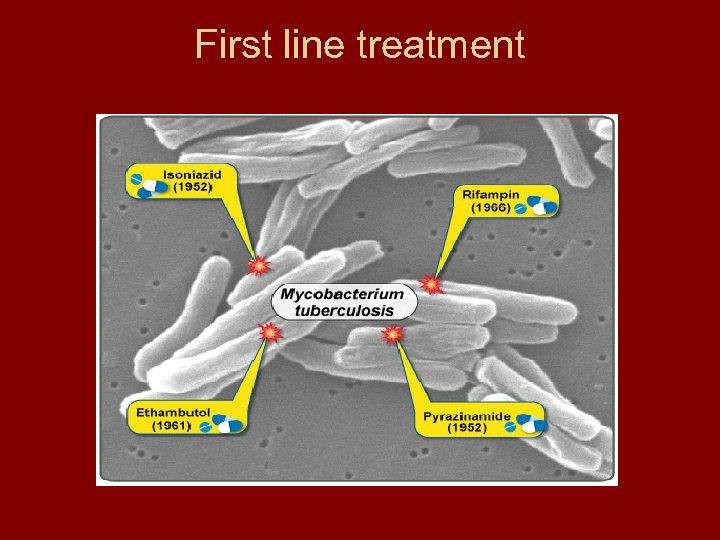
First line treatment
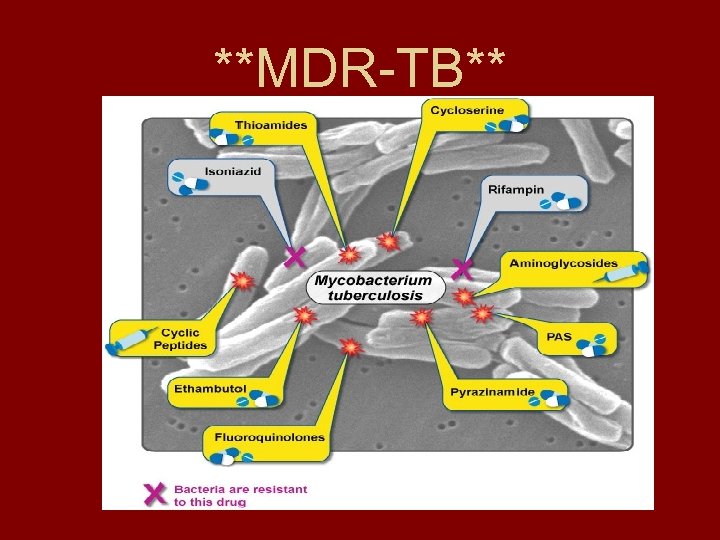
**MDR-TB**

Epidemiology • Globally, the incidence of multi-drug resistant tuberculosis is around 3. 4%. • China, India and the Russian Federation are estimated to account for 62% of the overall incidence of MDR-TB.
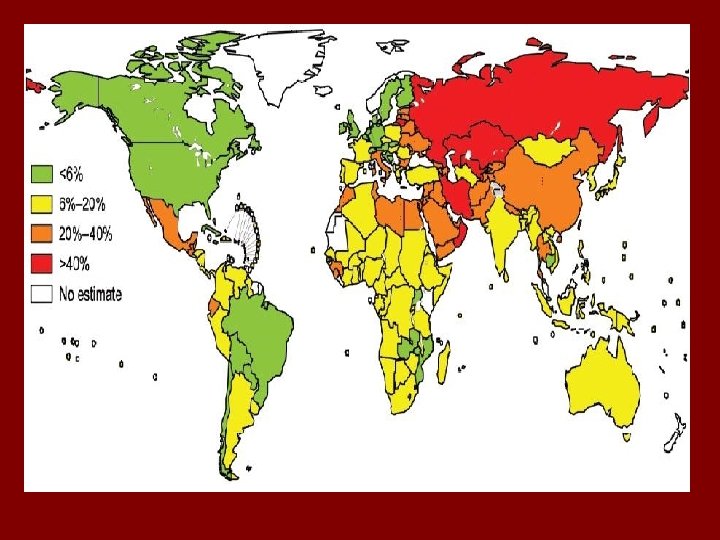

Epidemiology • Some of the highest statistics of HIV and multidrug resistant tuberculosis co-infection are seen in sub-Saharan Africa where HIV/AIDS prevalence is so high. • Currently, world wide MDR-TB mortality rates are at about “ 40 -60% depending on the country of residence and access to immediate care”. These rates are consistent with the rates of untreated tuberculosis (Ducati et al. 2006).

M&M • Morbidity and mortality rates in the United States steadily declined over the years until the 1980’s when a 2. 6% annual increase in cases was discovered. • The sudden increase was attributed to, “deterioration of the tuberculosis program infrastructure, the HIV/AIDS epidemic, drugresistant tuberculosis, tuberculosis among foreign-born persons, and an increase in transmission, especially in congregate and institutional settings” (Schneider et al. 2005).

Drug-Resistance-How it Happens • When the bacilli are exposed to an antituberculosis medication, most are killed. If there are some bacilli present that are resistant to that particular antibiotic, they will mutate and multiply freely. These random chromosomal mutations may occur by nucleotide insertions, deletions, or substitutions (Ducati et al. 2006). • If the patient is given two or three antibiotics, this inhibits the multiplication of resistant mutants due to the unlikelihood that all of the bacilli would be resistant to all three anti-tuberculosis medications (Sharma et al. 2007).

Rifampin and Isoniazid • Mycobacterium tuberculosis becomes resistant to rifampin by an alteration of the B-subunit of RNA polymerase, the rpo. B gene • The genes involved in isoniazid resistance are kat. G, inh. A, ahp. C, and oxy. R

Causes of Drug Resistance • “The most powerful predictor of the presence of MDR-TB is the history of treatment of tuberculosis”. (Sharma 2006) • inadequate treatment is received due to the installment of inadequate programs having poor guidelines that use mono therapy antibiotic treatment • when the mono therapy doesn’t work, practitioners are adding only a single drug to the regimen instead of two or three drugs • poor supply or lack of quality of drugs • lack appropriate storage for the drugs • patient compliance in taking the prescribed medication • failure of equipment, lack of laboratory support, and appropriate facilities for things such as cultures and sensitivity testing

Presentation • • • Sneezing coughing deep cough with bloody sputum chest pain Chills unintended weight loss slight fever night sweats loss of appetite pain with breathing

Diagnosis • • • History is IMPORTANT Mantoux tuberculin skin test (TST) chest radiograph Quanti. FERON-TB Gold test (QFT-G) The presence of acid-fast bacilli on a sputum smear may aid in the diagnosis of tuberculosis, but it is not 100% diagnostic. • The definitive diagnosis is made with a culture of the smear. • susceptibility testing by culture and sensitivity

Treatment • “If the patient’s active-tuberculosis shows resistance to isoniazid and rifampin, begin treatment with a floroquinoline, pyrazinamide, ethambutol, and an injectable agent such as an aminoglycoside (kanamycin, amikacin, streptomycin), or the glyycopeptide, capreomycin” (Nations 2006). Treatment should continue for 18 -24 months

Management • DOTS-plus • Vaccines

DOTS • “Political commitment to effective TB control • Case detection by sputum smear microscopy among symptomatic people • Standardized treatment regimen of 6 -8 months of shortcourse chemotherapy with first-line anti-TB drugs, administered under proper case management conditions, including direct observation • Uninterrupted supply of all essential anti-TB drugs • Standardized recording and reporting system allowing assessment of treatment results” (Frieden 2005)

DOTS-plus • “Sustained political and administrative commitment • Accurate, timely diagnosis through quality-assured culture and drug susceptibility testing • Uninterrupted supply of quality assured first and second-line drugs, appropriate treatment strategies utilizing second-line drugs under strict supervision • Directly observed treatment • Standardized recording and reporting system that enable performance monitoring and evaluation of treatment outcome” (www. who. int/gtb/policyrd/DOTSplus. htm) • In a retrospective study assessing the treatment results in the Latvian DOTS-plus strategy, data has shown itself to be promising. There were 204 MDR-TB cohorts in this study, and of the 204, “ 66% were cured, 7% died, 13% defaulted, and 14% did not respond to treatment” (Sharma et al. 2007).

Vaccines • BCG, the Bacille Calmette-Guerin vaccine • over 100 vaccine candidates • current BCG vaccine in a prime-boost strategy • BCG vaccine in combination with a new vaccine candidate

Vaccine Candidates • MVA-Ag 85 A – The first tuberculosis vaccine candidate that entered into a phase I trial in 2003 was Ag 85 A, an “immuno-dominant protective antigen from M. tuberculosis expressed in a replication-deficient strain of vaccine virus (MVA-Ag 85 A)”. (Ginsburg 2002) • BCG prime/MVA-Ag 85 A

XDR-TB • When Mycobacterium tuberculosis is resistant to “at least 3 of the 6 classes of second-line anti-tuberculosis drugs”, it is identified as extensively drug-resistant tuberculosis, XDR-TB (XDR-TB 2006).
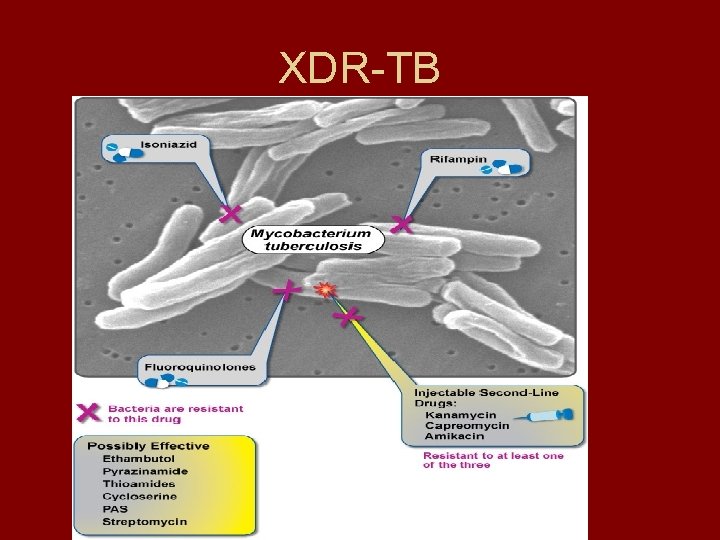
XDR-TB

Conclusion • MDR-TB is a problem! • As PAs we must follow our appropriate procedures for diagnosing, reporting, and treating • We can be effective globally with our time, education, supporting research for vaccines, etc.

Works Cited • • • • • Blondal K. Barriers to reaching the targets for tuberculosis control: multi-drug resistant tuberculosis. Bull World Health Organ. 2007 May; 85(5): 387 -90. Brennan, MJ. Development of new tuberculosis vaccines: a global perspective on regulatory issues. Health in Action. 2007 Aug; 4 (8): 1299 -1302. Center for Disease Control. Trends in Tuberculosis---United States, 2005. MMWR Weekly. 2006 March [cited 2007 Sept]; 55(11): [7 pages]. Available from: http: //www. cdc. gov/mmwr/preview/mmwrhtml/mm 5511 a 3. htm Christensen, D. Another look at TB vaccine: Should the United States use BCG widely? Science News. 2000 Jun 18; 4(12): 23 -35. Ducati RG, Netto AR, Basso LA, Santos DS. The resumption of consumption – A review on tuberculosis. Rio de Janeiro. 2006 November; 101 (7); 697 -714. Extensively drug-resistant tuberculosis (XDR-TB): recommendations for prevention and control. Wkly Epidemiol Rec. 2006 Nov 10; 81(45): 430 -2. Fitz. Gerald JM. Et al. Essentials of tuberculosis control for the practicing physician. Can Med Assoc J 1994; 150 (10): 1561 -1571. Frieden, TR. The DOTS strategy for controlling the global tuberculosis epidemic. Clin Chest Med. 2005 Jun; 26 (2) : 197 -205. Ginsberg, AM. What’s new in tuberculosis vaccines? Bulletin of the World Health Organization. 2002 80 (6): 483 -488. Johnson, MD, Decker, CF. Tuberculosis and HIV infection. Dis-Mon. 2006 Nov; 52 (11 -12): 420 -427. Kidder, Tracy. Mountains Beyond Mountains. Random House Trade Paperbacks. August 31, 2004. Martin, C. The dream of a vaccine against tuberculosis; new vaccines improving or replacing BCG? European Respiratory Journal. 2005; 26 (1): 162 -167. Nations, JA. Drug-resistant tuberculosis. Dis-Mon. 2006 Nov; 52 (11 -12): 435 -440. Sampaio J. Stronger health systems to beat TB. Bull World Health Organ. 2007 May: 85(5): 333. Senior, K. Action needed now to prevent resistant tuberculosis. The Lancet Infectious Diseases. 2007 Aug; 7 (8) : 511. Schneider, E. Epidemiology of tuberculosis in the United s. Sates. Clin Chest Med. 2005 Jun; 26(2) : 183 -195. Sharma SK, Mohan A. Multidrug-Resistant Tuberculosis: A Menace that Threatens to Destabilize Tuberculosis. Chest. 2006 Jul; 130 (1) : 261 -272. Shiferaw G, Woldeamanuel Y, Gebeyehu M, Girmachew F, Demessie D, Lemma E. Evaluation of microscopic observation drug susceptibility assay for detection of multidrug-resistant Mycobacterium tuberculosis. J Clin Microbiol. 2007 Apr: 45(4): 1093 -7. www. stoptb. org/resource_center/assets/documents/DOTS_test 4. pdf http: //www. umdnj. edu/~ntbcweb/history. htm
94d2b4021aa4931f1f859b6163f8d167.ppt