мол_диагностика1_2011.ppt
- Количество слайдов: 74
 Молекулярная диагностика
Молекулярная диагностика
 Молекулярная диагностика: выявление патогенных мутаций в образцах ДНК и РНК с целью диагностики, классификации, прогноза и мониторинга ответа на терапию Методы, основанные на выявлении локализации, определении структуры и функциональной активности генов, опосредующих развитие заболеваний
Молекулярная диагностика: выявление патогенных мутаций в образцах ДНК и РНК с целью диагностики, классификации, прогноза и мониторинга ответа на терапию Методы, основанные на выявлении локализации, определении структуры и функциональной активности генов, опосредующих развитие заболеваний
 Объекты молекулярной диагностики Геномная ДНК Транскрипция м. РНК Трансляция Определение последовательности (секвенирование) Определение мутаций и полиморфизмов с помощью ПЦР Выявление хромосомных аберраций белок Определение состава, количества и локализации белков Определение состава, количества и локализации РНК
Объекты молекулярной диагностики Геномная ДНК Транскрипция м. РНК Трансляция Определение последовательности (секвенирование) Определение мутаций и полиморфизмов с помощью ПЦР Выявление хромосомных аберраций белок Определение состава, количества и локализации белков Определение состава, количества и локализации РНК
 Молекулярная диагностика в медицине Технологии Области применения Технологии ПЦР Профилактика Гибридизация in situ (FISH) Клиническая диагностика Нанотехнологии Судмедэкспертиза Биочипы Фармацевтическая промышленность Пептидные нуклеиновые кислоты Биологическое оружие Протеомные технологии Разработка новых лекарств Электрохимическое выявление ДНК M. Debnath et al. , Molecular Diagnostics: Promises and Possibilities, DOI 10. 1007/97890 -481 -3261 -4_1, C Springer Science+Business Media B. V. 2010
Молекулярная диагностика в медицине Технологии Области применения Технологии ПЦР Профилактика Гибридизация in situ (FISH) Клиническая диагностика Нанотехнологии Судмедэкспертиза Биочипы Фармацевтическая промышленность Пептидные нуклеиновые кислоты Биологическое оружие Протеомные технологии Разработка новых лекарств Электрохимическое выявление ДНК M. Debnath et al. , Molecular Diagnostics: Promises and Possibilities, DOI 10. 1007/97890 -481 -3261 -4_1, C Springer Science+Business Media B. V. 2010
 «Тераностика» Онкология (цетуксимаб, дазатиниб, эрлотиниб, гефитиниб, иматиниб, лапатиниб, трастузумаб и т. д. ) Трансплантология (азатиоприн) ВИЧ /СПИД (абакавир, маара-вирок)
«Тераностика» Онкология (цетуксимаб, дазатиниб, эрлотиниб, гефитиниб, иматиниб, лапатиниб, трастузумаб и т. д. ) Трансплантология (азатиоприн) ВИЧ /СПИД (абакавир, маара-вирок)
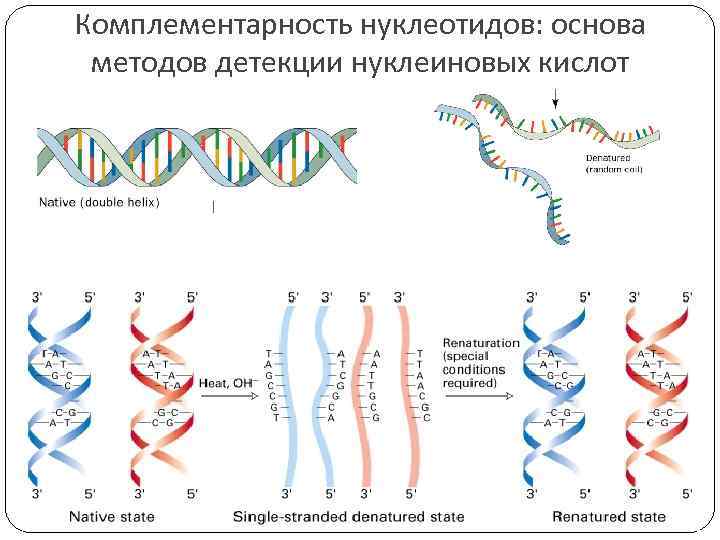 Комплементарность нуклеотидов: основа методов детекции нуклеиновых кислот
Комплементарность нуклеотидов: основа методов детекции нуклеиновых кислот
 Выделение нуклеиновых кислот Экстракция фенолом/хлороформом, переосаждение этанолом выделение всего спектра нуклеиновых кислот С помощью сорбции на микропористый оксид кремния (мембраны, колонки) автоматизация выделения
Выделение нуклеиновых кислот Экстракция фенолом/хлороформом, переосаждение этанолом выделение всего спектра нуклеиновых кислот С помощью сорбции на микропористый оксид кремния (мембраны, колонки) автоматизация выделения
 Секвенирование ДНК Энзиматическая (Sanger, 1977) или химическая (Maxam and Gilbert, 1977) деградация сепарация полученных фрагментов и анализ их длины С помощью синтеза (Ansorge, 1991) детекция вновь встраиваемого флуорохром- меченого нуклеотида – ПЦР-секвенирование с помощью создания библиотек ДНК-фрагментов (ПЦР в эмульсии) с помощью лигирования с помощью детекции встраивания единичного нуклеотида
Секвенирование ДНК Энзиматическая (Sanger, 1977) или химическая (Maxam and Gilbert, 1977) деградация сепарация полученных фрагментов и анализ их длины С помощью синтеза (Ansorge, 1991) детекция вновь встраиваемого флуорохром- меченого нуклеотида – ПЦР-секвенирование с помощью создания библиотек ДНК-фрагментов (ПЦР в эмульсии) с помощью лигирования с помощью детекции встраивания единичного нуклеотида
 Полимеразная цепная реакция Основана на принципе репликации ДНК Цикл ПЦР состоит из: 1. Денатурация мишени 2. Отжиг праймеров 3. Синтез новых цепей
Полимеразная цепная реакция Основана на принципе репликации ДНК Цикл ПЦР состоит из: 1. Денатурация мишени 2. Отжиг праймеров 3. Синтез новых цепей
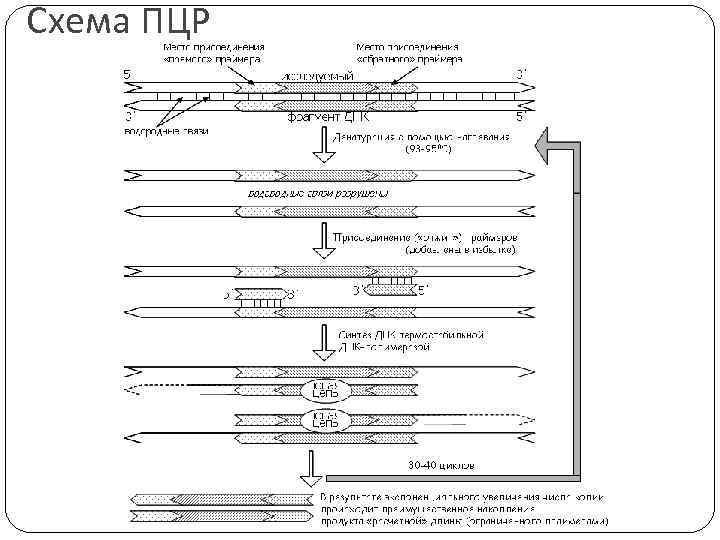 Схема ПЦР
Схема ПЦР
 Виды ПЦР и области применения Секвенирование (cycle sequencing) Клонирование(cloning of gene/gene fragment into plasmid) Аллель-специфичная (polymorphism) In situ (infections and localization) PCR-ELISA (telomerase activity detection) RT-PCR (differential display) «В реальном времени» (reliable qualitative method) Гнездовая ПЦР (gene/transcript in single cell)
Виды ПЦР и области применения Секвенирование (cycle sequencing) Клонирование(cloning of gene/gene fragment into plasmid) Аллель-специфичная (polymorphism) In situ (infections and localization) PCR-ELISA (telomerase activity detection) RT-PCR (differential display) «В реальном времени» (reliable qualitative method) Гнездовая ПЦР (gene/transcript in single cell)
 Дизайн праймеров для ПЦР Define sequence of interest Define method of detection (is labeling or introduction of non-complimentary sequences required? ) Download your sequence of interest into the software Check if sequence suggested by software accomplishes all your requirements Order from company specialized in oligos synthesis
Дизайн праймеров для ПЦР Define sequence of interest Define method of detection (is labeling or introduction of non-complimentary sequences required? ) Download your sequence of interest into the software Check if sequence suggested by software accomplishes all your requirements Order from company specialized in oligos synthesis
 Primer design software http: //www. ebi. ac. uk/biocat. html OR http: //www. chemie. uni marburg. de/~becker/prim gen. html OR http: //norp 5424 b. hsc. usc. edu/genetools. html General hints on primer design http: //alces. med. umn. edu/VGC. html Designing primers for cycle sequencing http: //www. biotech. iastate. edu/Facilities/DSSF/ Designing primers for detecting unknown sequences http: //www. blocks. fhcrc. org/blocks/help/CODEHOP OR http: //www. dartmouth. edu/artsci/bio/ambros/protocols/o ther/ koelle/degenerate_PCR. html
Primer design software http: //www. ebi. ac. uk/biocat. html OR http: //www. chemie. uni marburg. de/~becker/prim gen. html OR http: //norp 5424 b. hsc. usc. edu/genetools. html General hints on primer design http: //alces. med. umn. edu/VGC. html Designing primers for cycle sequencing http: //www. biotech. iastate. edu/Facilities/DSSF/ Designing primers for detecting unknown sequences http: //www. blocks. fhcrc. org/blocks/help/CODEHOP OR http: //www. dartmouth. edu/artsci/bio/ambros/protocols/o ther/ koelle/degenerate_PCR. html
 Primer design CODEHOP http: //blocks. fhcrc. org/blocks/codehop. html Degen. Designer ftp anonymous evolution. bchs. uh. edu Primer Design ftp anonymous ftp. chemie. uni marburg. de Primer Master ftp anonymous ftp. ebi. ac. uk PRIMER MIT ftp anonymous genome. wi. mit. edu Primers! ftp anonymous wuarchive. wustl. edu Primer Selection (VGC) http: //alces. med. umn. edu/vgc. html Primer Selection (USC) http: //norp 5424 b. hsc. usc. edu/genetools. html
Primer design CODEHOP http: //blocks. fhcrc. org/blocks/codehop. html Degen. Designer ftp anonymous evolution. bchs. uh. edu Primer Design ftp anonymous ftp. chemie. uni marburg. de Primer Master ftp anonymous ftp. ebi. ac. uk PRIMER MIT ftp anonymous genome. wi. mit. edu Primers! ftp anonymous wuarchive. wustl. edu Primer Selection (VGC) http: //alces. med. umn. edu/vgc. html Primer Selection (USC) http: //norp 5424 b. hsc. usc. edu/genetools. html
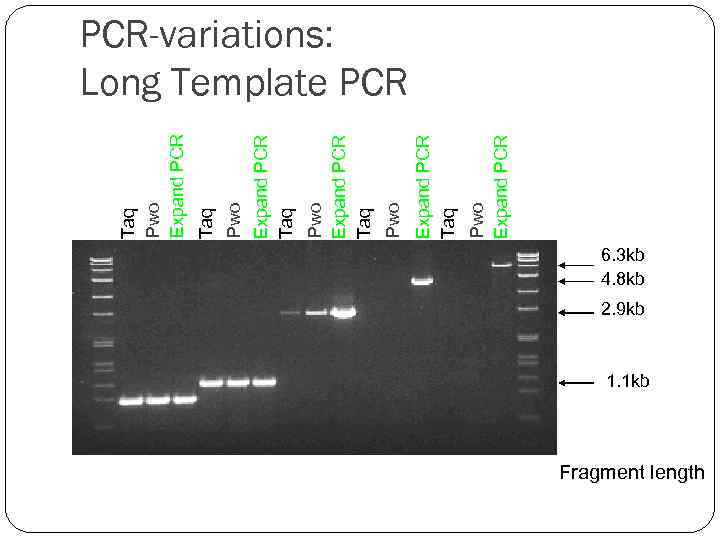 Pwo Expand PCR Taq Pwo Expand PCR Taq Taq Pwo Expand PCR-variations: Long Template PCR 6. 3 kb 4. 8 kb 2. 9 kb 1. 1 kb Fragment length
Pwo Expand PCR Taq Pwo Expand PCR Taq Taq Pwo Expand PCR-variations: Long Template PCR 6. 3 kb 4. 8 kb 2. 9 kb 1. 1 kb Fragment length
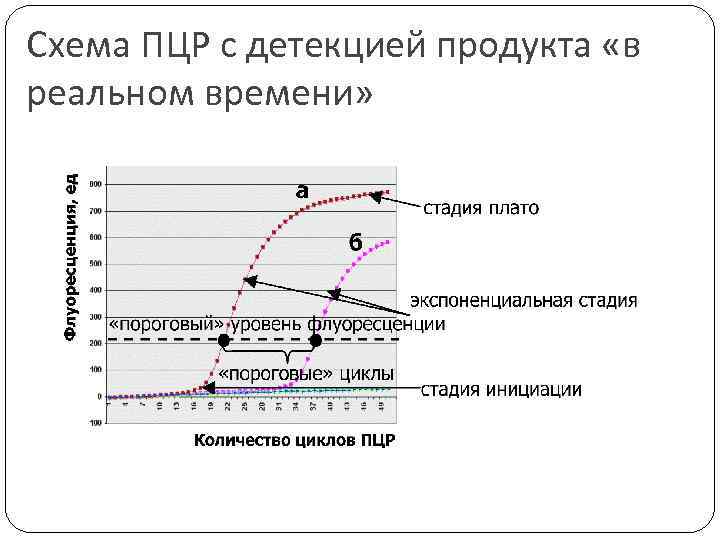 Схема ПЦР с детекцией продукта «в реальном времени»
Схема ПЦР с детекцией продукта «в реальном времени»
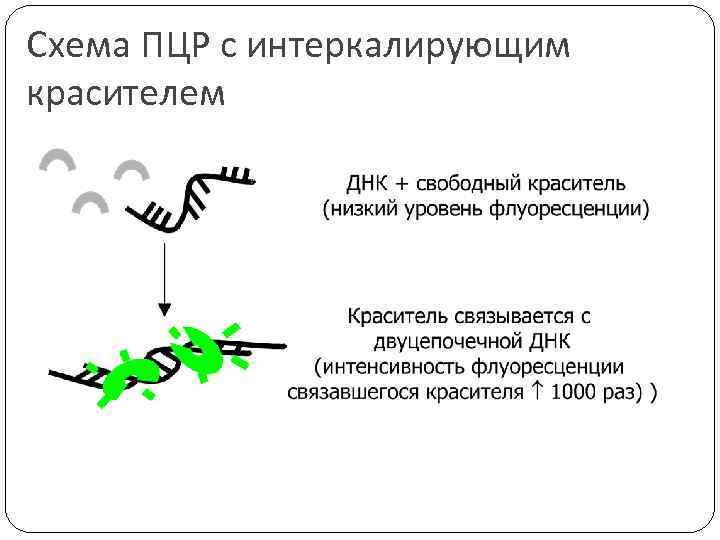 Схема ПЦР с интеркалирующим красителем
Схема ПЦР с интеркалирующим красителем
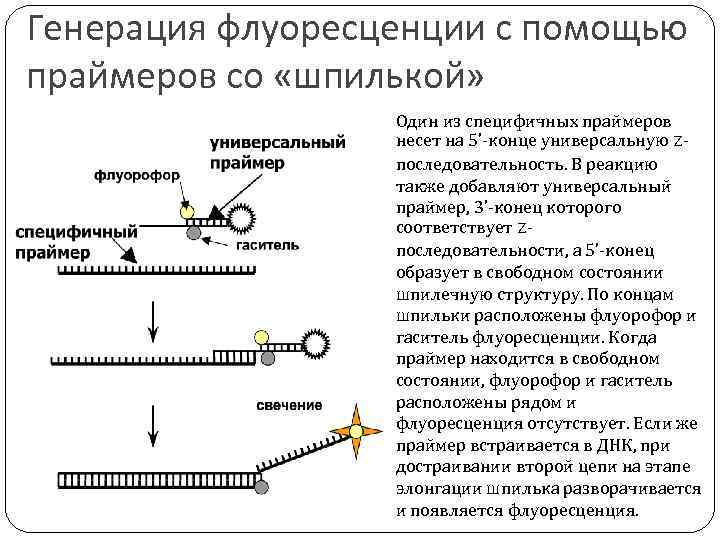 Генерация флуоресценции с помощью праймеров со «шпилькой» Один из специфичных праймеров несет на 5’-конце универсальную Zпоследовательность. В реакцию также добавляют универсальный праймер, 3’-конец которого соответствует Zпоследовательности, а 5’-конец образует в свободном состоянии шпилечную структуру. По концам шпильки расположены флуорофор и гаситель флуоресценции. Когда праймер находится в свободном состоянии, флуорофор и гаситель расположены рядом и флуоресценция отсутствует. Если же праймер встраивается в ДНК, при достраивании второй цепи на этапе элонгации шпилька разворачивается и появляется флуоресценция.
Генерация флуоресценции с помощью праймеров со «шпилькой» Один из специфичных праймеров несет на 5’-конце универсальную Zпоследовательность. В реакцию также добавляют универсальный праймер, 3’-конец которого соответствует Zпоследовательности, а 5’-конец образует в свободном состоянии шпилечную структуру. По концам шпильки расположены флуорофор и гаситель флуоресценции. Когда праймер находится в свободном состоянии, флуорофор и гаситель расположены рядом и флуоресценция отсутствует. Если же праймер встраивается в ДНК, при достраивании второй цепи на этапе элонгации шпилька разворачивается и появляется флуоресценция.
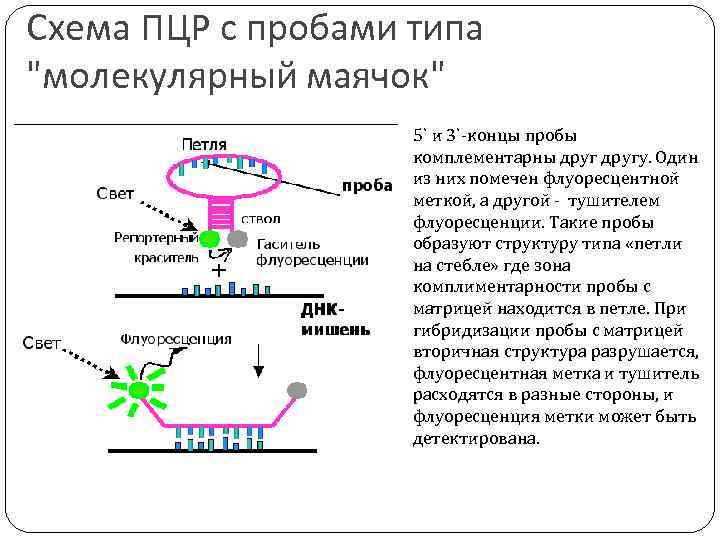 Схема ПЦР с пробами типа "молекулярный маячок" 5` и 3`-концы пробы комплементарны другу. Один из них помечен флуоресцентной меткой, а другой - тушителем флуоресценции. Такие пробы образуют структуру типа «петли на стебле» где зона комплиментарности пробы с матрицей находится в петле. При гибридизации пробы с матрицей вторичная структура разрушается, флуоресцентная метка и тушитель расходятся в разные стороны, и флуоресценция метки может быть детектирована.
Схема ПЦР с пробами типа "молекулярный маячок" 5` и 3`-концы пробы комплементарны другу. Один из них помечен флуоресцентной меткой, а другой - тушителем флуоресценции. Такие пробы образуют структуру типа «петли на стебле» где зона комплиментарности пробы с матрицей находится в петле. При гибридизации пробы с матрицей вторичная структура разрушается, флуоресцентная метка и тушитель расходятся в разные стороны, и флуоресценция метки может быть детектирована.
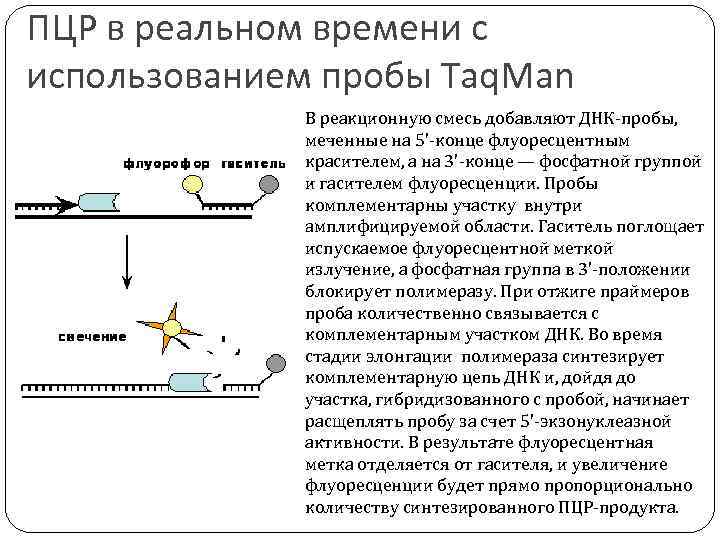 ПЦР в реальном времени с использованием пробы Taq. Man В реакционную смесь добавляют ДНК-пробы, меченные на 5'-конце флуоресцентным красителем, а на 3'-конце — фосфатной группой и гасителем флуоресценции. Пробы комплементарны участку внутри амплифицируемой области. Гаситель поглощает испускаемое флуоресцентной меткой излучение, а фосфатная группа в 3'-положении блокирует полимеразу. При отжиге праймеров проба количественно связывается с комплементарным участком ДНК. Во время стадии элонгации полимераза синтезирует комплементарную цепь ДНК и, дойдя до участка, гибридизованного с пробой, начинает расщеплять пробу за счет 5'-экзонуклеазной активности. В результате флуоресцентная метка отделяется от гасителя, и увеличение флуоресценции будет прямо пропорционально количеству синтезированного ПЦР-продукта.
ПЦР в реальном времени с использованием пробы Taq. Man В реакционную смесь добавляют ДНК-пробы, меченные на 5'-конце флуоресцентным красителем, а на 3'-конце — фосфатной группой и гасителем флуоресценции. Пробы комплементарны участку внутри амплифицируемой области. Гаситель поглощает испускаемое флуоресцентной меткой излучение, а фосфатная группа в 3'-положении блокирует полимеразу. При отжиге праймеров проба количественно связывается с комплементарным участком ДНК. Во время стадии элонгации полимераза синтезирует комплементарную цепь ДНК и, дойдя до участка, гибридизованного с пробой, начинает расщеплять пробу за счет 5'-экзонуклеазной активности. В результате флуоресцентная метка отделяется от гасителя, и увеличение флуоресценции будет прямо пропорционально количеству синтезированного ПЦР-продукта.
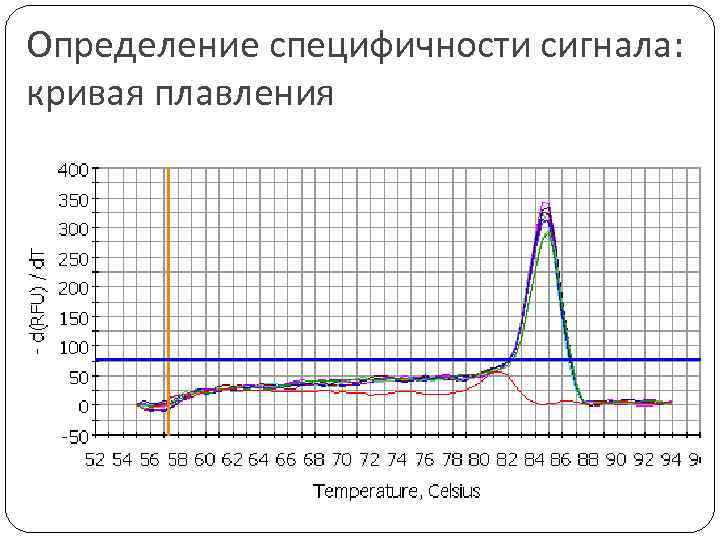 Определение специфичности сигнала: кривая плавления
Определение специфичности сигнала: кривая плавления
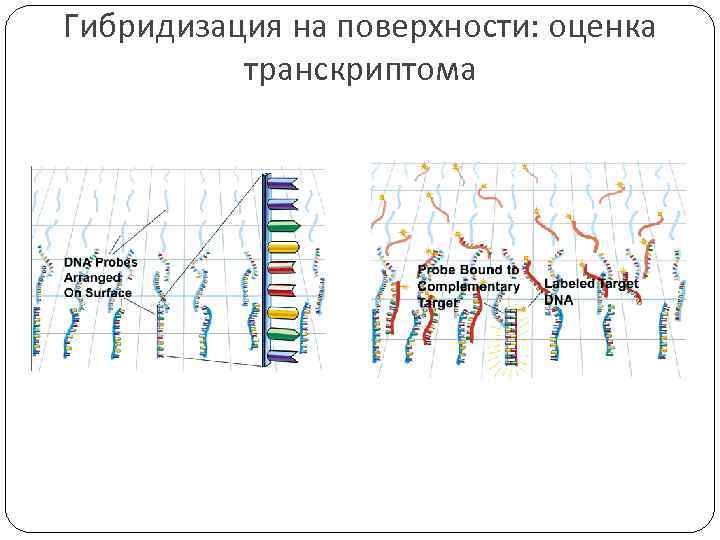 Гибридизация на поверхности: оценка транскриптома
Гибридизация на поверхности: оценка транскриптома
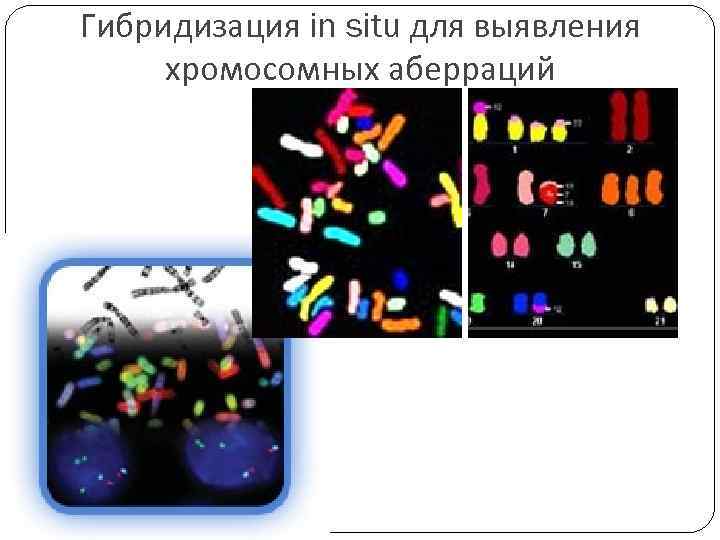 Гибридизация in situ для выявления хромосомных аберраций
Гибридизация in situ для выявления хромосомных аберраций
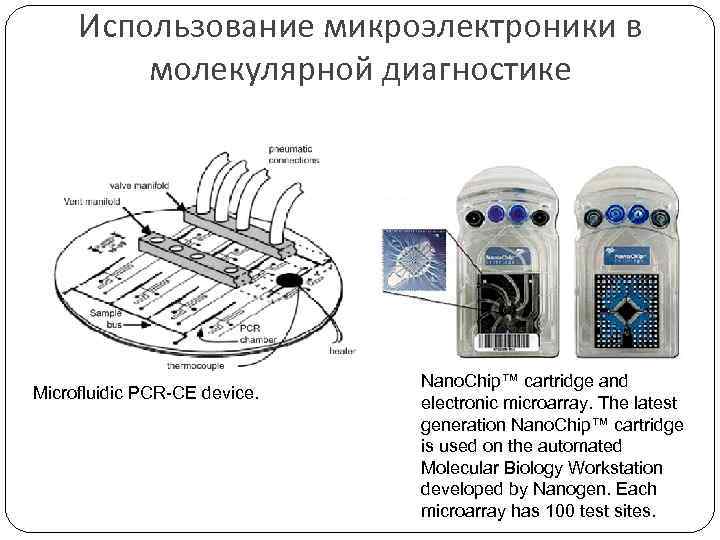 Использование микроэлектроники в молекулярной диагностике Microfluidic PCR-CE device. Nano. Chip™ cartridge and electronic microarray. The latest generation Nano. Chip™ cartridge is used on the automated Molecular Biology Workstation developed by Nanogen. Each microarray has 100 test sites.
Использование микроэлектроники в молекулярной диагностике Microfluidic PCR-CE device. Nano. Chip™ cartridge and electronic microarray. The latest generation Nano. Chip™ cartridge is used on the automated Molecular Biology Workstation developed by Nanogen. Each microarray has 100 test sites.
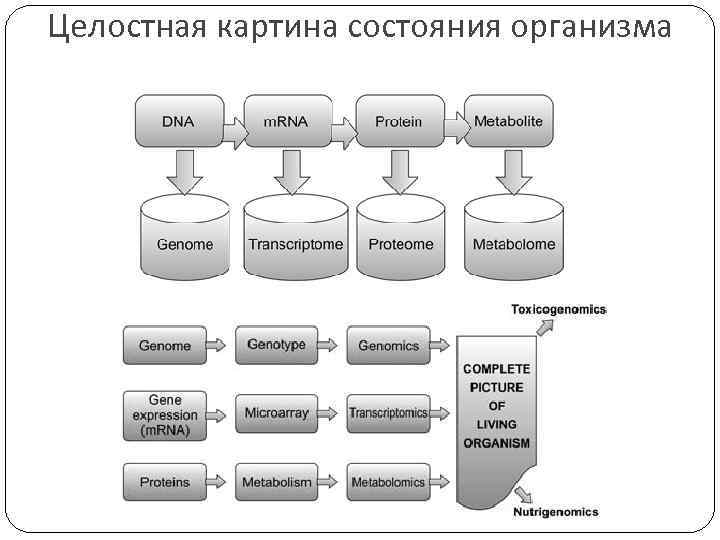 Целостная картина состояния организма
Целостная картина состояния организма
 What is PCR? Polymerase Chain Reaction (PCR) allows for quick replication of DNA. Small quantities of genetic material can be amplified millions of times within a few hours allowing for the rapid and reliable detection of genetic markers (infectious diseases, cancer and genetic disorders). Invented by scientists at Cetus Corporation in 1983. It has enabled significant scientific advances, including DNA fingerprinting, sequencing of the human genome as well as disease diagnosis, particularly in the areas of AIDS and hepatitis C.
What is PCR? Polymerase Chain Reaction (PCR) allows for quick replication of DNA. Small quantities of genetic material can be amplified millions of times within a few hours allowing for the rapid and reliable detection of genetic markers (infectious diseases, cancer and genetic disorders). Invented by scientists at Cetus Corporation in 1983. It has enabled significant scientific advances, including DNA fingerprinting, sequencing of the human genome as well as disease diagnosis, particularly in the areas of AIDS and hepatitis C.
 Where it happens? This process takes place in a thermal cycler, an thermal cycler instrument that automatically controls and quickly alternates the temperatures for programmed periods of time for the appropriate number of PCR cycles (usually between 30 and 40 cycles). The time required by instrument to rapidly decrease and increase the temperature limits the time of the process.
Where it happens? This process takes place in a thermal cycler, an thermal cycler instrument that automatically controls and quickly alternates the temperatures for programmed periods of time for the appropriate number of PCR cycles (usually between 30 and 40 cycles). The time required by instrument to rapidly decrease and increase the temperature limits the time of the process.
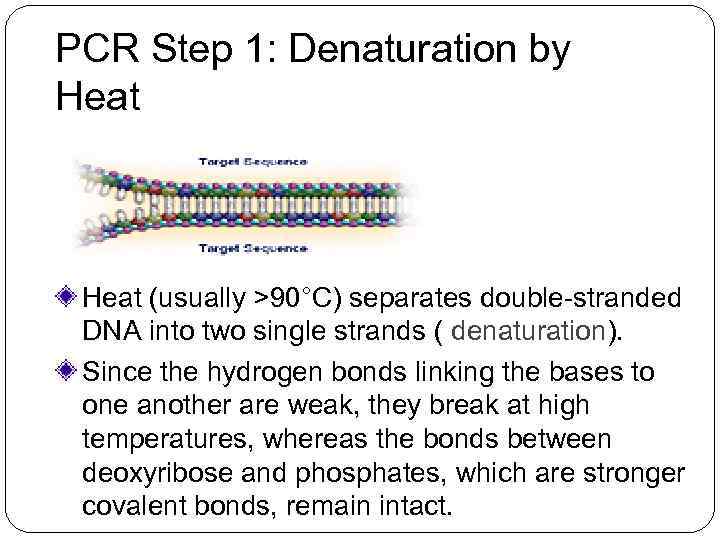 PCR Step 1: Denaturation by Heat (usually >90°C) separates double-stranded DNA into two single strands ( denaturation). Since the hydrogen bonds linking the bases to one another are weak, they break at high temperatures, whereas the bonds between deoxyribose and phosphates, which are stronger covalent bonds, remain intact.
PCR Step 1: Denaturation by Heat (usually >90°C) separates double-stranded DNA into two single strands ( denaturation). Since the hydrogen bonds linking the bases to one another are weak, they break at high temperatures, whereas the bonds between deoxyribose and phosphates, which are stronger covalent bonds, remain intact.
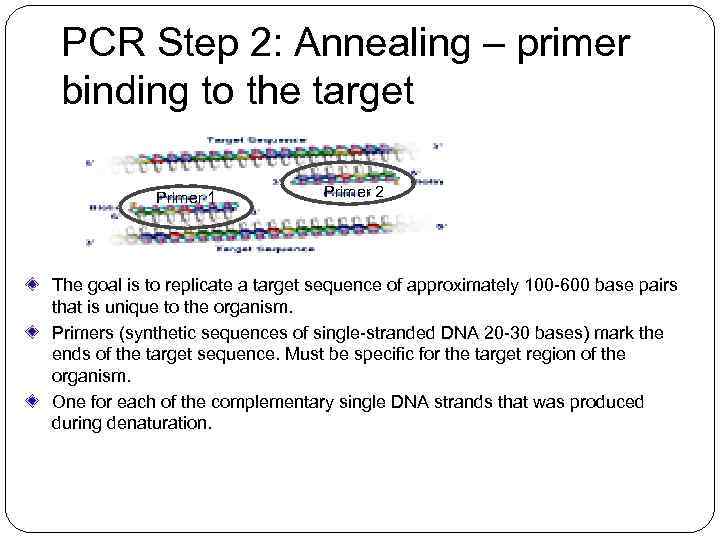 PCR Step 2: Annealing – primer binding to the target Primer 1 Primer 2 The goal is to replicate a target sequence of approximately 100 -600 base pairs that is unique to the organism. Primers (synthetic sequences of single-stranded DNA 20 -30 bases) mark the ends of the target sequence. Must be specific for the target region of the organism. One for each of the complementary single DNA strands that was produced during denaturation.
PCR Step 2: Annealing – primer binding to the target Primer 1 Primer 2 The goal is to replicate a target sequence of approximately 100 -600 base pairs that is unique to the organism. Primers (synthetic sequences of single-stranded DNA 20 -30 bases) mark the ends of the target sequence. Must be specific for the target region of the organism. One for each of the complementary single DNA strands that was produced during denaturation.
 Annealing temperature ensures specific binding of the primer Annealing Temperature: usually 40°C - 65°C, depending on the length and base sequence of the primers (their GC content). This allows the primers to anneal to the target sequence with high specificity. Normally, specific annealing temperature is suggested by primer design software. Primer 1 Primer 2
Annealing temperature ensures specific binding of the primer Annealing Temperature: usually 40°C - 65°C, depending on the length and base sequence of the primers (their GC content). This allows the primers to anneal to the target sequence with high specificity. Normally, specific annealing temperature is suggested by primer design software. Primer 1 Primer 2
 PCR Step 3: Extension – DNA strand replication Once the primers anneal to the complementary DNA sequences, the temperature is raised to approximately 72°C and the enzyme Taq DNA polymerase is used to replicate the DNA strands.
PCR Step 3: Extension – DNA strand replication Once the primers anneal to the complementary DNA sequences, the temperature is raised to approximately 72°C and the enzyme Taq DNA polymerase is used to replicate the DNA strands.
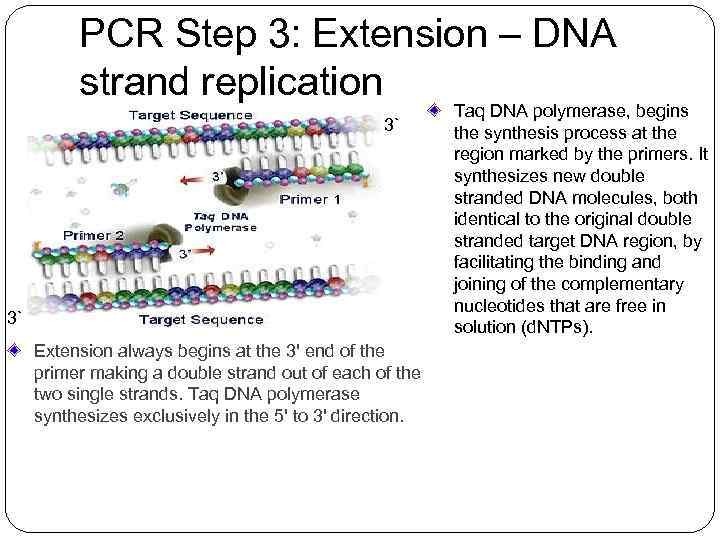 PCR Step 3: Extension – DNA strand replication 3` 3` 3` Extension always begins at the 3' end of the primer making a double strand out of each of the two single strands. Taq DNA polymerase synthesizes exclusively in the 5' to 3' direction. Taq DNA polymerase, begins the synthesis process at the region marked by the primers. It synthesizes new double stranded DNA molecules, both identical to the original double stranded target DNA region, by facilitating the binding and joining of the complementary nucleotides that are free in solution (d. NTPs).
PCR Step 3: Extension – DNA strand replication 3` 3` 3` Extension always begins at the 3' end of the primer making a double strand out of each of the two single strands. Taq DNA polymerase synthesizes exclusively in the 5' to 3' direction. Taq DNA polymerase, begins the synthesis process at the region marked by the primers. It synthesizes new double stranded DNA molecules, both identical to the original double stranded target DNA region, by facilitating the binding and joining of the complementary nucleotides that are free in solution (d. NTPs).
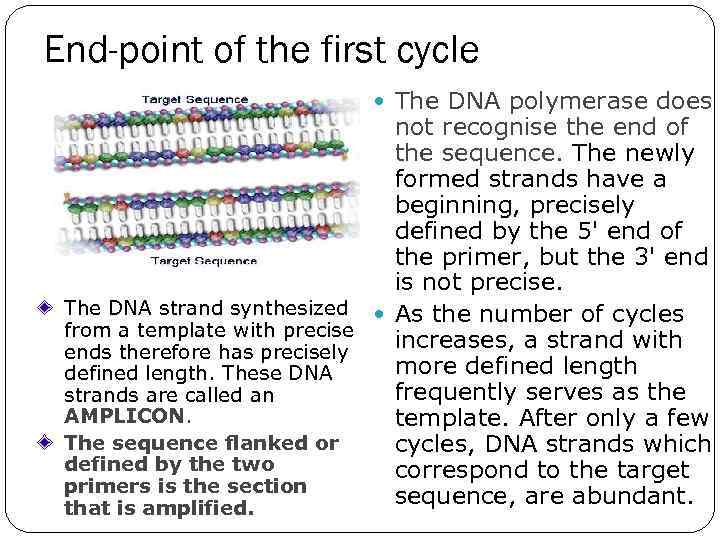 End-point of the first cycle The DNA polymerase does not recognise the end of the sequence. The newly formed strands have a beginning, precisely defined by the 5' end of the primer, but the 3' end is not precise. The DNA strand synthesized As the number of cycles from a template with precise increases, a strand with ends therefore has precisely more defined length. These DNA frequently serves as the strands are called an AMPLICON. template. After only a few The sequence flanked or cycles, DNA strands which defined by the two correspond to the target primers is the section sequence, are abundant. that is amplified.
End-point of the first cycle The DNA polymerase does not recognise the end of the sequence. The newly formed strands have a beginning, precisely defined by the 5' end of the primer, but the 3' end is not precise. The DNA strand synthesized As the number of cycles from a template with precise increases, a strand with ends therefore has precisely more defined length. These DNA frequently serves as the strands are called an AMPLICON. template. After only a few The sequence flanked or cycles, DNA strands which defined by the two correspond to the target primers is the section sequence, are abundant. that is amplified.
 Successful PCR Avoid contamination (usually by setting up laboratory according to PCR standards) Good equipment High quality PCR reaction components Choose of appropriate DNA polymerase Adjust PCR protocol accordingly to every new template
Successful PCR Avoid contamination (usually by setting up laboratory according to PCR standards) Good equipment High quality PCR reaction components Choose of appropriate DNA polymerase Adjust PCR protocol accordingly to every new template
 Cross - contamination: sources Trace amounts of DNA contaminants could serve as templates, resulting in amplification of the wrong template (false positives) Typical sources of contamination Laboratory benches, equipment, and pipetting devices, which can be contaminated by previous DNA preparations, by plasmid DNA, or by purified restriction fragments. Cross contamination between samples. Products from previous PCR amplifications.
Cross - contamination: sources Trace amounts of DNA contaminants could serve as templates, resulting in amplification of the wrong template (false positives) Typical sources of contamination Laboratory benches, equipment, and pipetting devices, which can be contaminated by previous DNA preparations, by plasmid DNA, or by purified restriction fragments. Cross contamination between samples. Products from previous PCR amplifications.
 Cross - contamination: facilities Set up physically separated working places for: Template preparation before PCR Setting up PCR reactions Post PCR analysis. PCR tubes which are DNase and RNase free. Special aerosol resistant pipette tips, and a dedicated used only for PCR) set of pipettes, preferably positive displacement pipettes. If possible, set up PCR reactions under a fume hood.
Cross - contamination: facilities Set up physically separated working places for: Template preparation before PCR Setting up PCR reactions Post PCR analysis. PCR tubes which are DNase and RNase free. Special aerosol resistant pipette tips, and a dedicated used only for PCR) set of pipettes, preferably positive displacement pipettes. If possible, set up PCR reactions under a fume hood.
 Cross - contamination: sample handling Work carefully and maintain sterility. Wear fresh gloves when working in the PCR area and change gloves frequently. Always use new and/or sterilized glassware, plasticware, and pipettes to prepare PCR reagents and template DNA. Autoclave all reagents and solutions that can be autoclaved without affecting their performance. Attention: primers, d. NTPs and DNA Polymerase must not be autoclaved. Have your own set of PCR reagents and solutions that are used only for PCR. Store these reagents in small aliquots. When pipetting DNA, avoid creating aerosols. Always include a negative and positive controls.
Cross - contamination: sample handling Work carefully and maintain sterility. Wear fresh gloves when working in the PCR area and change gloves frequently. Always use new and/or sterilized glassware, plasticware, and pipettes to prepare PCR reagents and template DNA. Autoclave all reagents and solutions that can be autoclaved without affecting their performance. Attention: primers, d. NTPs and DNA Polymerase must not be autoclaved. Have your own set of PCR reagents and solutions that are used only for PCR. Store these reagents in small aliquots. When pipetting DNA, avoid creating aerosols. Always include a negative and positive controls.
 Good equipment: thermal cycler A thermal cycler must, at a minimum, accurately and reproducibly maintain the three PCR incubation temperatures, change from one temperature to another (“ramp”) over a definable time, arrive at the selected temperatures without significant over or undershoot and cycle between the temperatures repeatedly and reproducibly.
Good equipment: thermal cycler A thermal cycler must, at a minimum, accurately and reproducibly maintain the three PCR incubation temperatures, change from one temperature to another (“ramp”) over a definable time, arrive at the selected temperatures without significant over or undershoot and cycle between the temperatures repeatedly and reproducibly.
 Good equipment: reaction tubes The reaction tubes affect the rate at which heat transfers from thermal cycler to the reaction mixture. Therefore, preferably use thin walled reaction tubes that are designed for PCR and that fit precisely into the wells of the particular brand of thermal cycler you are using.
Good equipment: reaction tubes The reaction tubes affect the rate at which heat transfers from thermal cycler to the reaction mixture. Therefore, preferably use thin walled reaction tubes that are designed for PCR and that fit precisely into the wells of the particular brand of thermal cycler you are using.
 High quality template Purity: use as pure template as you can get (by using purification kits). Large amounts of RNA in a DNA template can chelate Mg 2+ and reduce the yield of the PCR. Also, impure templates may contain polymerase inhibitors that decrease the efficiency of the reaction. Integrity: Template DNA should be of high molecular weight (it should not be degraded). Amount: The amount of template in a reaction strongly influences performance in PCR. The recommended amount of template for standard PCR is: The maximum amount of human genomic DNA should be up to 500 ng. 1– 10 ng bacterial DNA. 0. 1– 1 ng plasmid DNA. Low amounts of template, e. g. <10 ng human genomic DNA, will require specific reaction modifications, such as changes in cycle number, redesign of primers, use of “Hot Start”, etc.
High quality template Purity: use as pure template as you can get (by using purification kits). Large amounts of RNA in a DNA template can chelate Mg 2+ and reduce the yield of the PCR. Also, impure templates may contain polymerase inhibitors that decrease the efficiency of the reaction. Integrity: Template DNA should be of high molecular weight (it should not be degraded). Amount: The amount of template in a reaction strongly influences performance in PCR. The recommended amount of template for standard PCR is: The maximum amount of human genomic DNA should be up to 500 ng. 1– 10 ng bacterial DNA. 0. 1– 1 ng plasmid DNA. Low amounts of template, e. g. <10 ng human genomic DNA, will require specific reaction modifications, such as changes in cycle number, redesign of primers, use of “Hot Start”, etc.
 Primer characteristics 18– 24 bases long. Contain no internal secondary structure. Contain 40– 60% G/C. Have a balanced distribution of G/C and A/T rich domains. Are not complementary to each other at the 3' ends (so primer dimers will not form). Have a melting temperature (Tm) that allows annealing temperatures of 55°– 65°C (for maximum specificity use temperatures of 62°– 65°C). For both primers the Tm should be similar. Note: Optimal annealing temperatures are often higher than the Tm of the primers (approx. 5°– 10°C).
Primer characteristics 18– 24 bases long. Contain no internal secondary structure. Contain 40– 60% G/C. Have a balanced distribution of G/C and A/T rich domains. Are not complementary to each other at the 3' ends (so primer dimers will not form). Have a melting temperature (Tm) that allows annealing temperatures of 55°– 65°C (for maximum specificity use temperatures of 62°– 65°C). For both primers the Tm should be similar. Note: Optimal annealing temperatures are often higher than the Tm of the primers (approx. 5°– 10°C).
 Notes to primer characteristics Bases that do not hybridize to the template may be added at the 5' end of a primer, (e. g. for introducing restriction sites into the amplification product for cloning purposes). Primer concentrations between 0. 1 and 0. 6 µM are generally optimal. Higher primer concentrations may promote mispriming and accumulation of non specific product. Lower primer concentrations may be exhausted before the reaction is completed, resulting in lower yields of desired product.
Notes to primer characteristics Bases that do not hybridize to the template may be added at the 5' end of a primer, (e. g. for introducing restriction sites into the amplification product for cloning purposes). Primer concentrations between 0. 1 and 0. 6 µM are generally optimal. Higher primer concentrations may promote mispriming and accumulation of non specific product. Lower primer concentrations may be exhausted before the reaction is completed, resulting in lower yields of desired product.
 Taq DNA polymerase Initial PCR experiments did not utilize Taq polymerase. However, thermostable DNA polymerase, one that could withstand the temperature extremes involved in the cycles of PCR, was necessary in order to automate the PCR process and to maximize its applications. In 1986, such a thermostable DNA polymerase was purified, referred to as native Taq, from the organism Thermus aquaticus. Today, almost all PCR is done using recombinant Taq, a cloned version of the enzyme, as it is less expensive to manufacture that the native form.
Taq DNA polymerase Initial PCR experiments did not utilize Taq polymerase. However, thermostable DNA polymerase, one that could withstand the temperature extremes involved in the cycles of PCR, was necessary in order to automate the PCR process and to maximize its applications. In 1986, such a thermostable DNA polymerase was purified, referred to as native Taq, from the organism Thermus aquaticus. Today, almost all PCR is done using recombinant Taq, a cloned version of the enzyme, as it is less expensive to manufacture that the native form.
 Other DNA polymerases used in PCR The primary requirements for a DNA polymerase used in PCR are optimal activity at temperatures around 75°C and the ability to retain that activity after prolonged incubation at even higher temperatures (95°C). Initially, single DNA polymerases were used in PCR, where Taq DNA Polymerase was used for “standard PCR”, Tth DNA Polymerase for combined RT PCR, and Pwo DNA Polymerase when fidelity of PCR was important.
Other DNA polymerases used in PCR The primary requirements for a DNA polymerase used in PCR are optimal activity at temperatures around 75°C and the ability to retain that activity after prolonged incubation at even higher temperatures (95°C). Initially, single DNA polymerases were used in PCR, where Taq DNA Polymerase was used for “standard PCR”, Tth DNA Polymerase for combined RT PCR, and Pwo DNA Polymerase when fidelity of PCR was important.
 New demands produced modern DNA polymerases As researchers wanted to amplify longer templates, the enzyme blend concept was born, then optimized to produce enzyme blends that could amplify very long DNA (up to 35 kb targets from a genomic template). As the demand for more robust PCR, higher yield and fidelity increased, “blends” of two polymerases, such as the Expand High Fidelity PCR System (Roche Inc. ), were optimized for various applications. These blends actually outperformed the individual DNA polymerases.
New demands produced modern DNA polymerases As researchers wanted to amplify longer templates, the enzyme blend concept was born, then optimized to produce enzyme blends that could amplify very long DNA (up to 35 kb targets from a genomic template). As the demand for more robust PCR, higher yield and fidelity increased, “blends” of two polymerases, such as the Expand High Fidelity PCR System (Roche Inc. ), were optimized for various applications. These blends actually outperformed the individual DNA polymerases.
 DNA polymerases: features to consider - I Processivity: number of bp which can be added to a copy by one molecule of polymerase before it falls off the template. Product length: maximum PCR fragment length which can be amplified from a human genomic DNA template with good yield. Error rate per bp. Sensitivity: Amplification of template present at a low copy number (<500 copies). Specificity: Amplifies only the fragments of interest.
DNA polymerases: features to consider - I Processivity: number of bp which can be added to a copy by one molecule of polymerase before it falls off the template. Product length: maximum PCR fragment length which can be amplified from a human genomic DNA template with good yield. Error rate per bp. Sensitivity: Amplification of template present at a low copy number (<500 copies). Specificity: Amplifies only the fragments of interest.
 DNA polymerases: features to consider - II Robustness: Amplification of templates independent of contaminating agents, GC content, etc. Yield: Produces large amount of products in a given number of PCR cycles. Labeling: Efficient incorporation of modified d. NTPs (e. g. biotin d. UTP, DIG d. UTP, FLUOS d. UTP). Cloning: Taq, Tth, and the Expand blends produce fragments with 3' A overhangs; therefore, TA cloning is the preferred way to clone the products.
DNA polymerases: features to consider - II Robustness: Amplification of templates independent of contaminating agents, GC content, etc. Yield: Produces large amount of products in a given number of PCR cycles. Labeling: Efficient incorporation of modified d. NTPs (e. g. biotin d. UTP, DIG d. UTP, FLUOS d. UTP). Cloning: Taq, Tth, and the Expand blends produce fragments with 3' A overhangs; therefore, TA cloning is the preferred way to clone the products.
 Mg 2+ concentration Mg 2+ forms soluble complexes with d. NTPs to produce the actual substrate that the polymerase recognizes. The concentration of free Mg 2+ depends on the concentrations of compounds that bind the ion, including d. NTP, free pyrophosphate (PPi) and EDTA and must be defined empirically. The optimal concentration is 1 5 m. M. The most commonly used Mg. Cl 2 concentration is 1. 5 m. M (with d. NTPs at a concentration of 200 µM each). Mg 2+ influences enzyme activity and increases the Tm of double stranded DNA. Excess Mg 2+ in the reaction can increase non specific primer binding and increase the non specific background of the reaction.
Mg 2+ concentration Mg 2+ forms soluble complexes with d. NTPs to produce the actual substrate that the polymerase recognizes. The concentration of free Mg 2+ depends on the concentrations of compounds that bind the ion, including d. NTP, free pyrophosphate (PPi) and EDTA and must be defined empirically. The optimal concentration is 1 5 m. M. The most commonly used Mg. Cl 2 concentration is 1. 5 m. M (with d. NTPs at a concentration of 200 µM each). Mg 2+ influences enzyme activity and increases the Tm of double stranded DNA. Excess Mg 2+ in the reaction can increase non specific primer binding and increase the non specific background of the reaction.
 d. NTP concentration Always use balanced solutions of all four d. NTPs to minimize polymerase error rate. Imbalanced d. NTP mixtures will reduce Taq DNA Polymerase fidelity. Provided in PCR kit as premixed combination. The final d. NTP concentration should be 50– 500 µM (each d. NTP). The most commonly used d. NTP concentration is 200 µM. If you increase the concentration of d. NTPs you must also increase Mg 2+ concentration. Increases in d. NTP concentration reduce free Mg 2+, thus interfering with polymerase activity and decreasing primer annealing.
d. NTP concentration Always use balanced solutions of all four d. NTPs to minimize polymerase error rate. Imbalanced d. NTP mixtures will reduce Taq DNA Polymerase fidelity. Provided in PCR kit as premixed combination. The final d. NTP concentration should be 50– 500 µM (each d. NTP). The most commonly used d. NTP concentration is 200 µM. If you increase the concentration of d. NTPs you must also increase Mg 2+ concentration. Increases in d. NTP concentration reduce free Mg 2+, thus interfering with polymerase activity and decreasing primer annealing.
 Optional additives Additives, which can enhance the efficiency or specificity of PCR: Betaine (0. 5– 2 M) Bovine serum albumin (BSA; 100 ng/50 µl) Detergents Dimethylsulfoxide (DMSO; 2– 10%) (v/v) Gelatine Glycerol (1– 5%) (v/v) Pyrophosphatase (0. 001– 0. 1 units/reaction) Spermidine T 4 Gene 32 protein
Optional additives Additives, which can enhance the efficiency or specificity of PCR: Betaine (0. 5– 2 M) Bovine serum albumin (BSA; 100 ng/50 µl) Detergents Dimethylsulfoxide (DMSO; 2– 10%) (v/v) Gelatine Glycerol (1– 5%) (v/v) Pyrophosphatase (0. 001– 0. 1 units/reaction) Spermidine T 4 Gene 32 protein
 Other notes p. H: buffer supplied with the corresponding thermostable DNA polymerase (p. H 8. 3– 9. 0) will give optimal results. However, for some systems, raising the p. H may stabilize the template and enhance results. Reaction overlay A layer of mineral oil is often placed atop the reaction mixture to prevent evaporation during PCR.
Other notes p. H: buffer supplied with the corresponding thermostable DNA polymerase (p. H 8. 3– 9. 0) will give optimal results. However, for some systems, raising the p. H may stabilize the template and enhance results. Reaction overlay A layer of mineral oil is often placed atop the reaction mixture to prevent evaporation during PCR.
 Thermal Cycling Profile for Standard PCR
Thermal Cycling Profile for Standard PCR
 Initial Denaturation It is very important to denature the template DNA completely. Initial heating of the PCR mixture for 2 minutes at 94°– 95°C is enough to completely denature complex genomic DNA so that the primers can anneal to the template as the reaction mix is cooled. If the template DNA is only partially denatured, it will tend to “snap back” very quickly, preventing efficient primer annealing and extension, or leading to “self priming, ” which can lead to false positive results.
Initial Denaturation It is very important to denature the template DNA completely. Initial heating of the PCR mixture for 2 minutes at 94°– 95°C is enough to completely denature complex genomic DNA so that the primers can anneal to the template as the reaction mix is cooled. If the template DNA is only partially denatured, it will tend to “snap back” very quickly, preventing efficient primer annealing and extension, or leading to “self priming, ” which can lead to false positive results.
 Denaturation during cycling Denaturation at 94°– 95°C for 20– 30 seconds is usually sufficient, but this must be adapted for thermal cycler and tubes being used. If the denaturation temperature is too low, the incompletely melted DNA “snaps back”, thus giving no access to the primers. Use a longer denaturation time or higher denaturing temperature for GC rich template DNA. Never use a longer denaturation time than absolutely required for complete denaturation of template DNA. Unnecessarily long denaturation times decerase the activity of DNA Polymerase.
Denaturation during cycling Denaturation at 94°– 95°C for 20– 30 seconds is usually sufficient, but this must be adapted for thermal cycler and tubes being used. If the denaturation temperature is too low, the incompletely melted DNA “snaps back”, thus giving no access to the primers. Use a longer denaturation time or higher denaturing temperature for GC rich template DNA. Never use a longer denaturation time than absolutely required for complete denaturation of template DNA. Unnecessarily long denaturation times decerase the activity of DNA Polymerase.
 Primer 1 Primer 2 Annealing: points to remember Annealing temperature is the most critical factor in designing a high specificity PCR. If the temperature is too high, no annealing occurs, but if it is too low, non specific annealing will increase dramatically. Primer dimers will form if the primers have one or more complementary bases so that base pairing between the 3' ends of the two primers can occur.
Primer 1 Primer 2 Annealing: points to remember Annealing temperature is the most critical factor in designing a high specificity PCR. If the temperature is too high, no annealing occurs, but if it is too low, non specific annealing will increase dramatically. Primer dimers will form if the primers have one or more complementary bases so that base pairing between the 3' ends of the two primers can occur.
 Extension time For fragments up to 3 kb, primer extension is carried out at 72°C. Taq DNA Polymerase can add approximately 60 bases per second at 72°C. A 45 second extension is sufficient for fragments up to 1 kb. For extension of fragments up to 3 kb, allow about 45 seconds per kb. However, these times may need to be adjusted for specific templates. For improved yield, use the cycle extension feature of thermal cycler (e. g. the first 10 cycles at 45 s for a 1 kb product; for the next 20 cycles, increase the extension time by 2– 5 s per cycle). Cycle extension allows the enzyme more time to do its job.
Extension time For fragments up to 3 kb, primer extension is carried out at 72°C. Taq DNA Polymerase can add approximately 60 bases per second at 72°C. A 45 second extension is sufficient for fragments up to 1 kb. For extension of fragments up to 3 kb, allow about 45 seconds per kb. However, these times may need to be adjusted for specific templates. For improved yield, use the cycle extension feature of thermal cycler (e. g. the first 10 cycles at 45 s for a 1 kb product; for the next 20 cycles, increase the extension time by 2– 5 s per cycle). Cycle extension allows the enzyme more time to do its job.
 Cycle number and final extension In an optimal reaction, less than 10 template molecules can be amplified in less than 40 cycles to a product that is easily detectable on a gel stained with ethidium bromide. Most PCRs should, therefore, include only 25 to 35 cycles. As cycle number increases, nonspecific products can accumulate. Usually, after the last cycle, the reaction tubes are held at 72°C for 5– 15 minutes to promote completion of partial extension products and annealing of single stranded complementary products.
Cycle number and final extension In an optimal reaction, less than 10 template molecules can be amplified in less than 40 cycles to a product that is easily detectable on a gel stained with ethidium bromide. Most PCRs should, therefore, include only 25 to 35 cycles. As cycle number increases, nonspecific products can accumulate. Usually, after the last cycle, the reaction tubes are held at 72°C for 5– 15 minutes to promote completion of partial extension products and annealing of single stranded complementary products.
 PCR variations: HOT-START Components essential for polymerase activity is “separated” from the reaction mixture until the temperature in the tubes has exceeded the optimal primer annealing temperature (usually 55°– 65°C). Since the polymerase cannot function in the incomplete reaction mixture, there is no primer elongation during the period when primers can bind non-specifically. “Hot Start” PCR has been shown to significantly improve the specificity, sensitivity, and yield of PCR
PCR variations: HOT-START Components essential for polymerase activity is “separated” from the reaction mixture until the temperature in the tubes has exceeded the optimal primer annealing temperature (usually 55°– 65°C). Since the polymerase cannot function in the incomplete reaction mixture, there is no primer elongation during the period when primers can bind non-specifically. “Hot Start” PCR has been shown to significantly improve the specificity, sensitivity, and yield of PCR
 Hot-start: ways of separation Manually A component such as polymerase or Mg 2+ is omitted from the original reaction mixture, and is only added to the tubes after the temperature is >70°C. Attention: Manual addition of components to the heated tube is generally inconvenient and may lead to problems such as lack of reproducibility and carry-over contamination.
Hot-start: ways of separation Manually A component such as polymerase or Mg 2+ is omitted from the original reaction mixture, and is only added to the tubes after the temperature is >70°C. Attention: Manual addition of components to the heated tube is generally inconvenient and may lead to problems such as lack of reproducibility and carry-over contamination.
 Hot-start: ways of separation Physical separation Reaction components are divided into two mixes, which are separated by a barrier, such as a wax plug. Alternatively, wax beads containing Mg 2+ ions are added to the PCR mix. During the initial denaturation step, the wax melts (at 75°– 80°C) and allows all the reaction components to mix.
Hot-start: ways of separation Physical separation Reaction components are divided into two mixes, which are separated by a barrier, such as a wax plug. Alternatively, wax beads containing Mg 2+ ions are added to the PCR mix. During the initial denaturation step, the wax melts (at 75°– 80°C) and allows all the reaction components to mix.
 Hot-start: ways of separation Polymerase antibodies In a pre-incubation step, a heat-sensitive antibody is added to the polymerase (usually Taq or Tth DNA Polymerase) to keep it inactive in the reaction mixture. As the temperature in the tube rises, the antibody is inactivated, setting the active polymerase free.
Hot-start: ways of separation Polymerase antibodies In a pre-incubation step, a heat-sensitive antibody is added to the polymerase (usually Taq or Tth DNA Polymerase) to keep it inactive in the reaction mixture. As the temperature in the tube rises, the antibody is inactivated, setting the active polymerase free.
 Hot-start: ways of separation Chemical modification of polymerase Addition of heat-labile blocking groups to some of the amino acid residues on Taq DNA Polymerase makes the modified enzyme inactive at room temperature. It is “activated” (by removal of the blocking groups) at high temperatures during a pre-PCR heat step (approx. 2– 4 min at 95°C).
Hot-start: ways of separation Chemical modification of polymerase Addition of heat-labile blocking groups to some of the amino acid residues on Taq DNA Polymerase makes the modified enzyme inactive at room temperature. It is “activated” (by removal of the blocking groups) at high temperatures during a pre-PCR heat step (approx. 2– 4 min at 95°C).
 Hot-start: ways of separation Other inhibitors Other polymerase inhibitors (such as polymerase-binding oligonucleotides) are added to the reaction mixture, keeping the enzyme inactive until increasing temperature dissociates the inhibitor from the enzyme, setting it free to begin polymerization
Hot-start: ways of separation Other inhibitors Other polymerase inhibitors (such as polymerase-binding oligonucleotides) are added to the reaction mixture, keeping the enzyme inactive until increasing temperature dissociates the inhibitor from the enzyme, setting it free to begin polymerization
 PCR-variations: Long Template PCR Taq DNA polymerase amplifies only up to 3 kb genomic templates. Using a mixture of Taq DNA polymerase and a thermostable DNA polymerase with proofreading activity could overcome limitations in the length of fragments amplified. Now there are several expand PCR systems, each of which outperforms conventional polymerases systems by offering greater fidelity, higher yields, and superior results with difficult templates and primers. “basic” PCR “expand” PCR
PCR-variations: Long Template PCR Taq DNA polymerase amplifies only up to 3 kb genomic templates. Using a mixture of Taq DNA polymerase and a thermostable DNA polymerase with proofreading activity could overcome limitations in the length of fragments amplified. Now there are several expand PCR systems, each of which outperforms conventional polymerases systems by offering greater fidelity, higher yields, and superior results with difficult templates and primers. “basic” PCR “expand” PCR
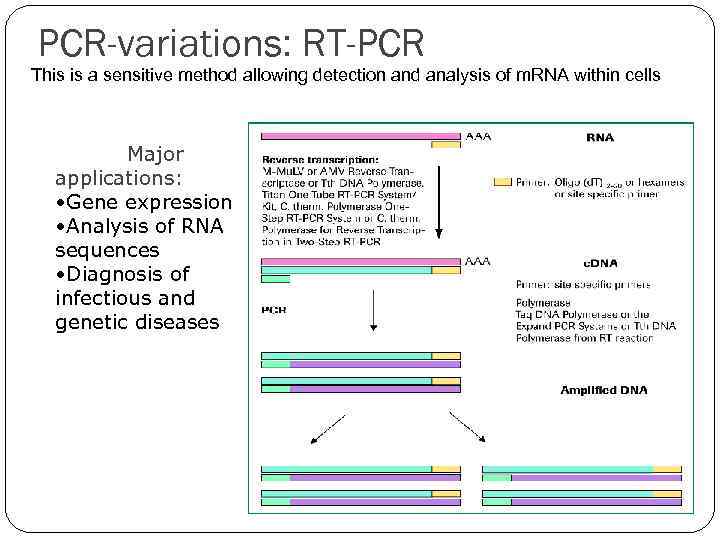 PCR-variations: RT-PCR This is a sensitive method allowing detection and analysis of m. RNA within cells Major applications: • Gene expression • Analysis of RNA sequences • Diagnosis of infectious and genetic diseases
PCR-variations: RT-PCR This is a sensitive method allowing detection and analysis of m. RNA within cells Major applications: • Gene expression • Analysis of RNA sequences • Diagnosis of infectious and genetic diseases
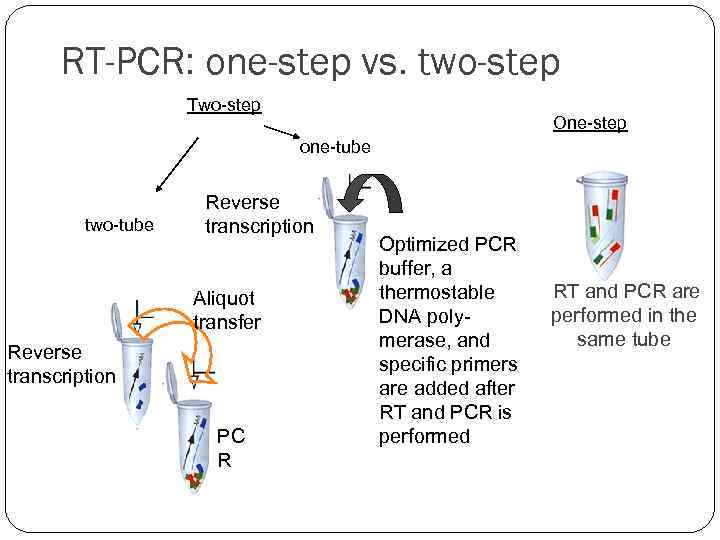 RT-PCR: one-step vs. two-step Two-step One-step one-tube two-tube Reverse transcription Aliquot transfer Reverse transcription PC R Optimized PCR buffer, a thermostable DNA polymerase, and specific primers are added after RT and PCR is performed RT and PCR are performed in the same tube
RT-PCR: one-step vs. two-step Two-step One-step one-tube two-tube Reverse transcription Aliquot transfer Reverse transcription PC R Optimized PCR buffer, a thermostable DNA polymerase, and specific primers are added after RT and PCR is performed RT and PCR are performed in the same tube
 RT-PCR: one-step vs. two-step One-step Minimizes time required Has fewer pipetting steps, significantly reducing the time needed to perform RTPCR and eliminating pipetting errors. Reduces the risk of contamination No transfers required and no need to open the reaction tube. Improves the sensitivity and specificity of c. DNA synthesis (1) the c. DNA reaction is performed at a high temperature (to eliminate problems with RNA secondary structure), and (2) the entire c. DNA sample is used as template for PCR. Two-step Optimizes reaction conditions The two-step format allows both reverse transcription and PCR to be performed under optimal conditions to ensure efficient and accurate amplification. Provides flexibility Product of single c. DNA synthesis reaction can be used for analysis of multiple transcripts. Exampes: rapid amplification of c. DNA ends (RACE) and Differential Display Reverse Transcription (DDRT). Amplifies long sequences With the right combination of reverse transcriptase and thermostable DNA polymerase, two-step RT-PCR can amplify RNA sequences up to 14 kb long.
RT-PCR: one-step vs. two-step One-step Minimizes time required Has fewer pipetting steps, significantly reducing the time needed to perform RTPCR and eliminating pipetting errors. Reduces the risk of contamination No transfers required and no need to open the reaction tube. Improves the sensitivity and specificity of c. DNA synthesis (1) the c. DNA reaction is performed at a high temperature (to eliminate problems with RNA secondary structure), and (2) the entire c. DNA sample is used as template for PCR. Two-step Optimizes reaction conditions The two-step format allows both reverse transcription and PCR to be performed under optimal conditions to ensure efficient and accurate amplification. Provides flexibility Product of single c. DNA synthesis reaction can be used for analysis of multiple transcripts. Exampes: rapid amplification of c. DNA ends (RACE) and Differential Display Reverse Transcription (DDRT). Amplifies long sequences With the right combination of reverse transcriptase and thermostable DNA polymerase, two-step RT-PCR can amplify RNA sequences up to 14 kb long.
 RT-PCR: choice of primers Oligo(d. T)12– 18 , which binds to the endogenous poly(A) + tail at the 3' end of mammalian m. RNA. This primer often produces a full length c. DNA. Random hexanucleotides, which bind to m. RNA at a variety of complementary sites and lead to partial length (short) c. DNAs. Random hexanucleotides may be ideal for overcoming the difficulties presented by extensive secondary structure in the template. These primers may also transcribe more efficiently 5' regions of the m. RNA. Specific oligonucleotide primers, which selectively prime the m. RNA of interest. This type of primer has been used very successfully in diagnostic assays.
RT-PCR: choice of primers Oligo(d. T)12– 18 , which binds to the endogenous poly(A) + tail at the 3' end of mammalian m. RNA. This primer often produces a full length c. DNA. Random hexanucleotides, which bind to m. RNA at a variety of complementary sites and lead to partial length (short) c. DNAs. Random hexanucleotides may be ideal for overcoming the difficulties presented by extensive secondary structure in the template. These primers may also transcribe more efficiently 5' regions of the m. RNA. Specific oligonucleotide primers, which selectively prime the m. RNA of interest. This type of primer has been used very successfully in diagnostic assays.
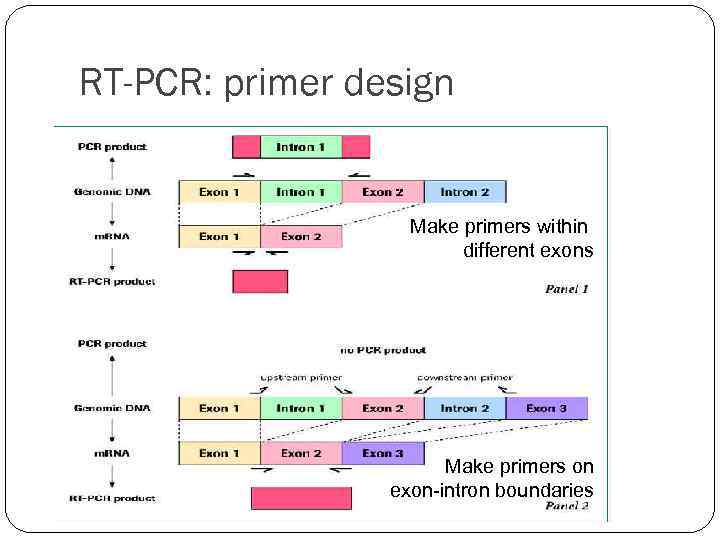 RT-PCR: primer design Make primers within different exons Make primers on exon-intron boundaries
RT-PCR: primer design Make primers within different exons Make primers on exon-intron boundaries
 RT-PCR: template preparation To minimize the activity of RNases that are re-leased during cell lysis, include RNase inhibitors in the lysis mix or use methods that simultane-ously disrupt cells and inactivate RNases. Take steps to eliminate all potential sources of RNase contamination from glassware, plastic-ware, reagents, etc. Use a product specifically designed for nucleic acid purification to prepare starting template RNA. Use purified m. RNA as template, rather than total RNA. Starting with poly(A)+ template m. RNA will greatly increase the likelihood of successful amplification of rare m. RNAs, since the proportion of m. RNA in a total RNA preparation is quite low (typically, 1– 5% of total RNA from a mammalian cell). If using m. RNA as template, check the integrity of the m. RNA by gel electrophoresis before using it in RT-PCR. The m. RNA should appear as a smear between approx. 500 bp and 8 kb. Most of the m. RNA should be between 1. 5 kb and 2 kb.
RT-PCR: template preparation To minimize the activity of RNases that are re-leased during cell lysis, include RNase inhibitors in the lysis mix or use methods that simultane-ously disrupt cells and inactivate RNases. Take steps to eliminate all potential sources of RNase contamination from glassware, plastic-ware, reagents, etc. Use a product specifically designed for nucleic acid purification to prepare starting template RNA. Use purified m. RNA as template, rather than total RNA. Starting with poly(A)+ template m. RNA will greatly increase the likelihood of successful amplification of rare m. RNAs, since the proportion of m. RNA in a total RNA preparation is quite low (typically, 1– 5% of total RNA from a mammalian cell). If using m. RNA as template, check the integrity of the m. RNA by gel electrophoresis before using it in RT-PCR. The m. RNA should appear as a smear between approx. 500 bp and 8 kb. Most of the m. RNA should be between 1. 5 kb and 2 kb.
 PCR Applications Sequencing (cycle sequencing) Cloning (cloning of gene/gene fragment into plasmid) Allele-specific (polymorphism) In situ (infections and localization) PCR-ELISA (telomerase activity detection) RT-PCR (differential display) Real-time (reliable qualitative method) Nested PCR (gene/transcript in single cell)
PCR Applications Sequencing (cycle sequencing) Cloning (cloning of gene/gene fragment into plasmid) Allele-specific (polymorphism) In situ (infections and localization) PCR-ELISA (telomerase activity detection) RT-PCR (differential display) Real-time (reliable qualitative method) Nested PCR (gene/transcript in single cell)
 DNA and RNA have a deoxyribose and ribose sugar backbone, respectively, whereas PNA's backbone is composed of repeating N-(2 -aminoethyl)-glycine units linked by peptide bonds. PNA is not known to occur naturally. The various purine and pyrimidine bases are linked to the backbone by methylene carbonyl bonds. PNAs are depicted like peptides, with the N -terminus at the first (left) position and the C-terminus at the right. [2]
DNA and RNA have a deoxyribose and ribose sugar backbone, respectively, whereas PNA's backbone is composed of repeating N-(2 -aminoethyl)-glycine units linked by peptide bonds. PNA is not known to occur naturally. The various purine and pyrimidine bases are linked to the backbone by methylene carbonyl bonds. PNAs are depicted like peptides, with the N -terminus at the first (left) position and the C-terminus at the right. [2]
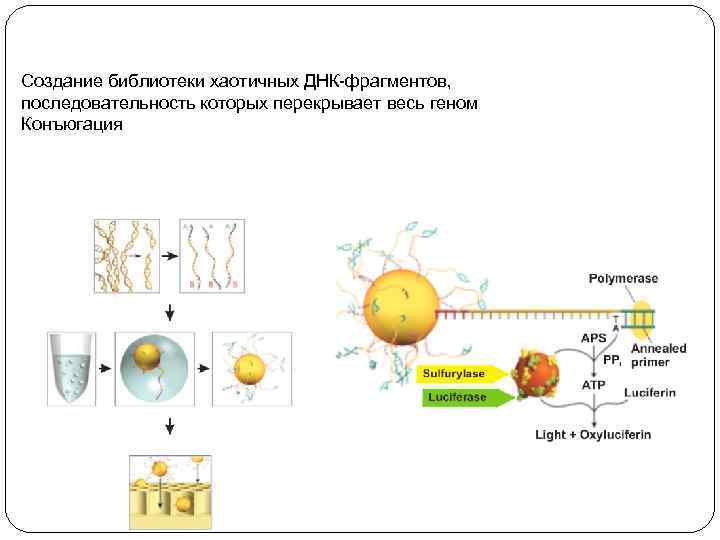 Создание библиотеки хаотичных ДНК-фрагментов, последовательность которых перекрывает весь геном Конъюгация
Создание библиотеки хаотичных ДНК-фрагментов, последовательность которых перекрывает весь геном Конъюгация
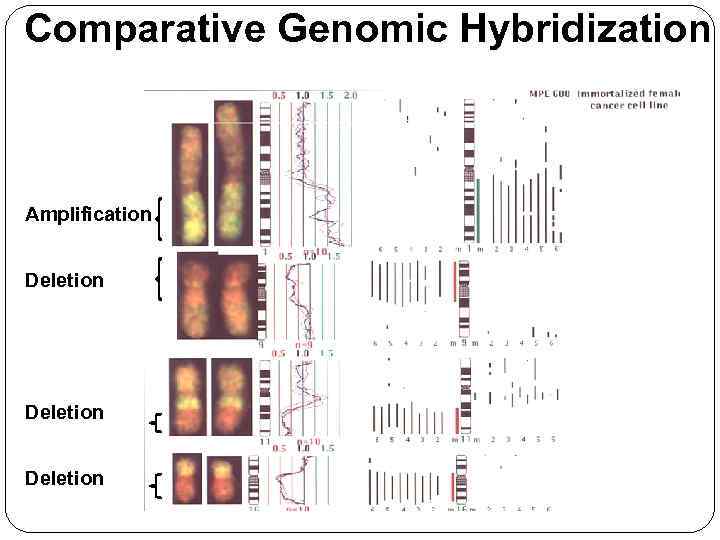 Comparative Genomic Hybridization Amplification Deletion
Comparative Genomic Hybridization Amplification Deletion


