804744bffbab4f23a53251a109219c05.ppt
- Количество слайдов: 46
 Molecular diagnosis of respiratory viruses and its impact on clinical management Prof G Kudesia Sheffield Teaching Hospitals NHS Trust
Molecular diagnosis of respiratory viruses and its impact on clinical management Prof G Kudesia Sheffield Teaching Hospitals NHS Trust
 Cell Culture u u Widely used Result in 7 -14 days or longer
Cell Culture u u Widely used Result in 7 -14 days or longer
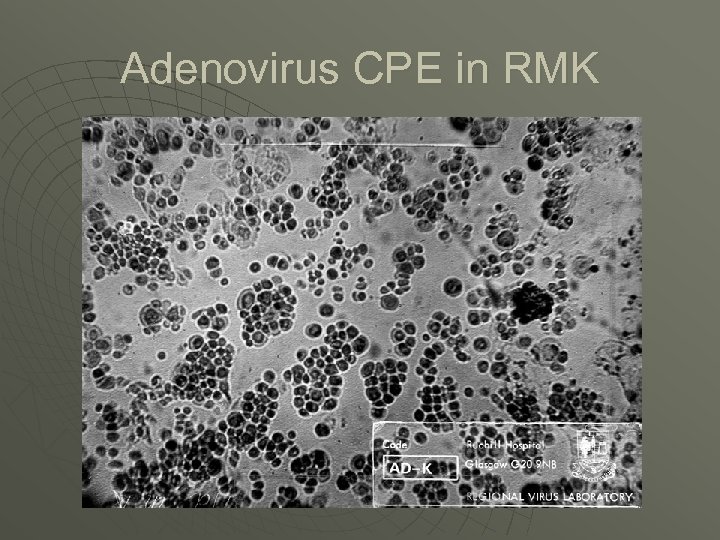 Adenovirus CPE in RMK
Adenovirus CPE in RMK
 Un-infected RMK
Un-infected RMK
 Live cells required Cytopathic effect needs to be confirmed by specific tests. Technical expertise Time delay Catch ‘all’
Live cells required Cytopathic effect needs to be confirmed by specific tests. Technical expertise Time delay Catch ‘all’
 Antigen detection by Immunofluorescence u u Rapid Relatively insensitive Not suitable for all speciemn types Subjective
Antigen detection by Immunofluorescence u u Rapid Relatively insensitive Not suitable for all speciemn types Subjective
 Serology Technically demanding Insensitive Acute and convalescent serum sample
Serology Technically demanding Insensitive Acute and convalescent serum sample
 Respiratory Viruses: Diagnosis Pre 1990’s. Advantages Disadvantage s Tissue Culture u“Open” Technique u. Sensitive u. Further characterisation u. Evidence of active infection u. Not Serology u. Detects current and past infection (immunity) u. Important for fastidious viruses u. Prolonged Antigen detection u. Rapid u. Not u. Detects non-viable virus u. Can test large numbers of samples applicable to all u. Time consuming uviable virus only u. Contamination/to xins time testing applicable to all uinterference
Respiratory Viruses: Diagnosis Pre 1990’s. Advantages Disadvantage s Tissue Culture u“Open” Technique u. Sensitive u. Further characterisation u. Evidence of active infection u. Not Serology u. Detects current and past infection (immunity) u. Important for fastidious viruses u. Prolonged Antigen detection u. Rapid u. Not u. Detects non-viable virus u. Can test large numbers of samples applicable to all u. Time consuming uviable virus only u. Contamination/to xins time testing applicable to all uinterference
 Polymerase Chain Reaction (PCR)-Xeroxing DNA! u u Kary Mullis Won the Nobel. Prize in 1993 for describing the methodology in 1985 to replicate DNA in a test tube.
Polymerase Chain Reaction (PCR)-Xeroxing DNA! u u Kary Mullis Won the Nobel. Prize in 1993 for describing the methodology in 1985 to replicate DNA in a test tube.
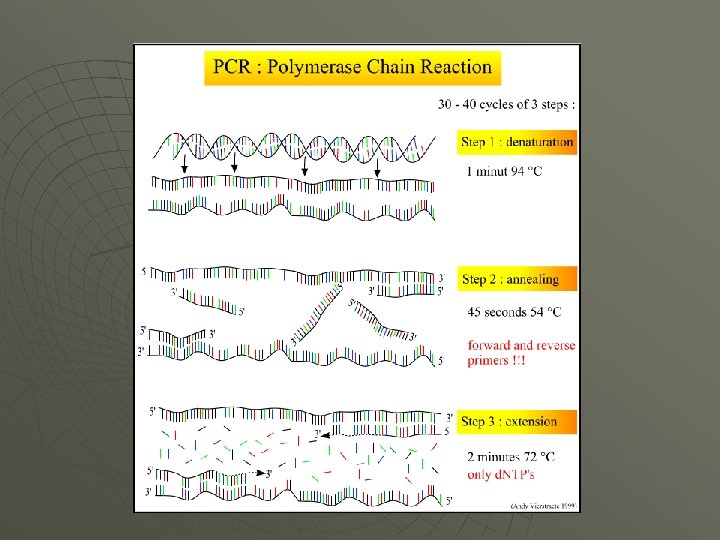
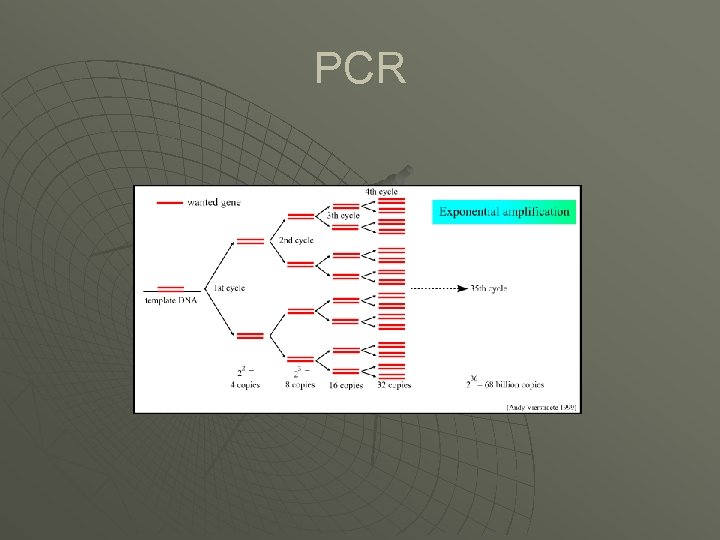 PCR
PCR
 Impact of PCR testing on respiratory virus investigations
Impact of PCR testing on respiratory virus investigations
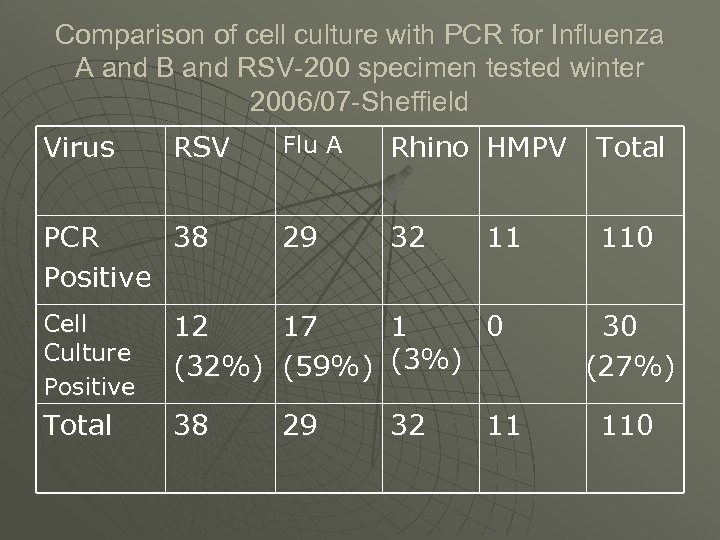 Comparison of cell culture with PCR for Influenza A and B and RSV-200 specimen tested winter 2006/07 -Sheffield Virus RSV PCR 38 Positive Flu A Rhino HMPV Total 29 32 110 11 Cell Culture Positive 12 17 1 0 (32%) (59%) (3%) Total 38 29 32 11 30 (27%) 110
Comparison of cell culture with PCR for Influenza A and B and RSV-200 specimen tested winter 2006/07 -Sheffield Virus RSV PCR 38 Positive Flu A Rhino HMPV Total 29 32 110 11 Cell Culture Positive 12 17 1 0 (32%) (59%) (3%) Total 38 29 32 11 30 (27%) 110
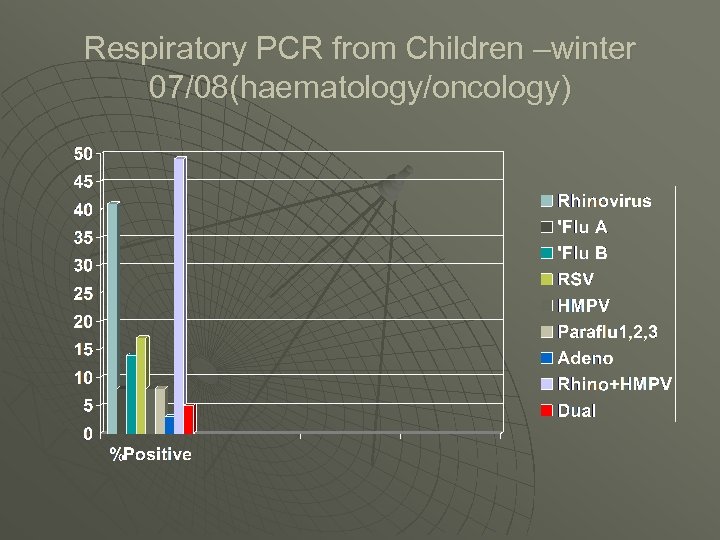 Respiratory PCR from Children –winter 07/08(haematology/oncology)
Respiratory PCR from Children –winter 07/08(haematology/oncology)
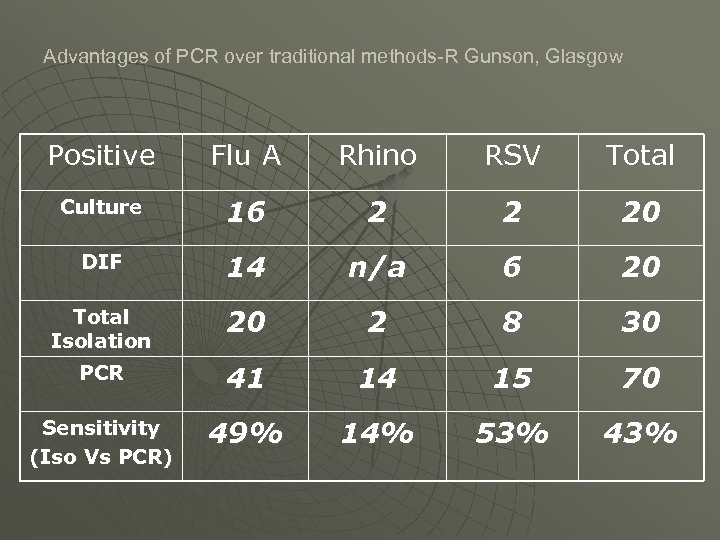 Advantages of PCR over traditional methods-R Gunson, Glasgow Positive Flu A Rhino RSV Total Culture 16 2 2 20 DIF 14 n/a 6 20 Total Isolation 20 2 8 30 PCR 41 14 15 70 Sensitivity (Iso Vs PCR) 49% 14% 53% 43%
Advantages of PCR over traditional methods-R Gunson, Glasgow Positive Flu A Rhino RSV Total Culture 16 2 2 20 DIF 14 n/a 6 20 Total Isolation 20 2 8 30 PCR 41 14 15 70 Sensitivity (Iso Vs PCR) 49% 14% 53% 43%
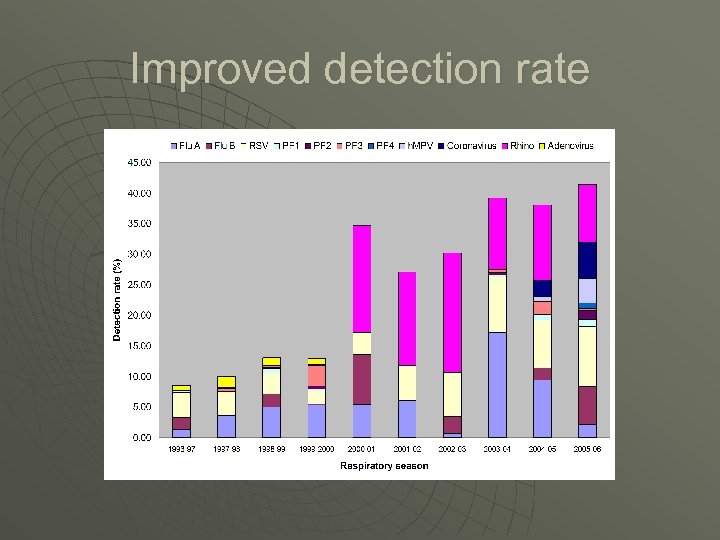 Improved detection rate
Improved detection rate
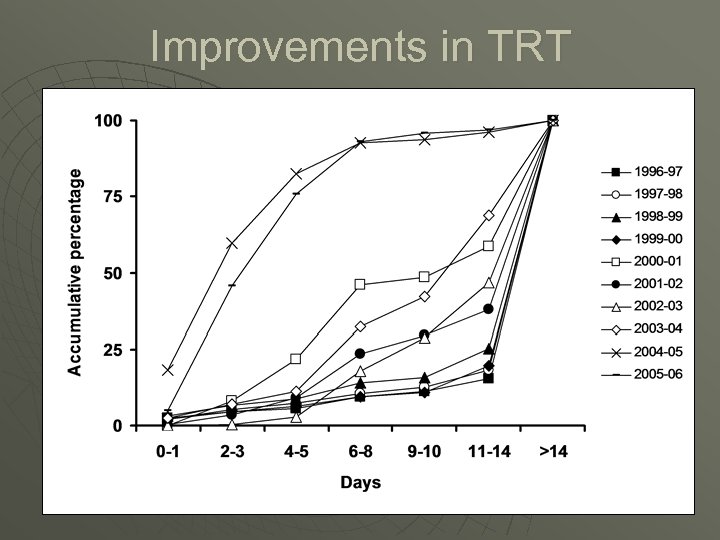 Improvements in TRT
Improvements in TRT
 Clinical impact u Influenza Treatment u Prophylaxis u Outbreak Management u Control of infection u u Immunocompromised Treatment u Control of infection u
Clinical impact u Influenza Treatment u Prophylaxis u Outbreak Management u Control of infection u u Immunocompromised Treatment u Control of infection u
 Treatment/prophylaxis for influenza -start within 48 hours u Oseltamivir • Treat- 75 mg twice a day x 5 days • Prophylaxis- 75 mg once a day x 10 days u u Speed for laboratory confirmation of essence PCR testing was invaluable in the late influenza B activity this winter- both for outbreak and individual patient management
Treatment/prophylaxis for influenza -start within 48 hours u Oseltamivir • Treat- 75 mg twice a day x 5 days • Prophylaxis- 75 mg once a day x 10 days u u Speed for laboratory confirmation of essence PCR testing was invaluable in the late influenza B activity this winter- both for outbreak and individual patient management
 Flu outbreak- SVC west Scotland Occurred out with flu season Hospital X Flu virus sequenced Index case Patient A – flu positive Doctor B – flu positive Phylogenetic tree created Nurse – flu positive Patient B – flu positive Patient C – flu positive
Flu outbreak- SVC west Scotland Occurred out with flu season Hospital X Flu virus sequenced Index case Patient A – flu positive Doctor B – flu positive Phylogenetic tree created Nurse – flu positive Patient B – flu positive Patient C – flu positive
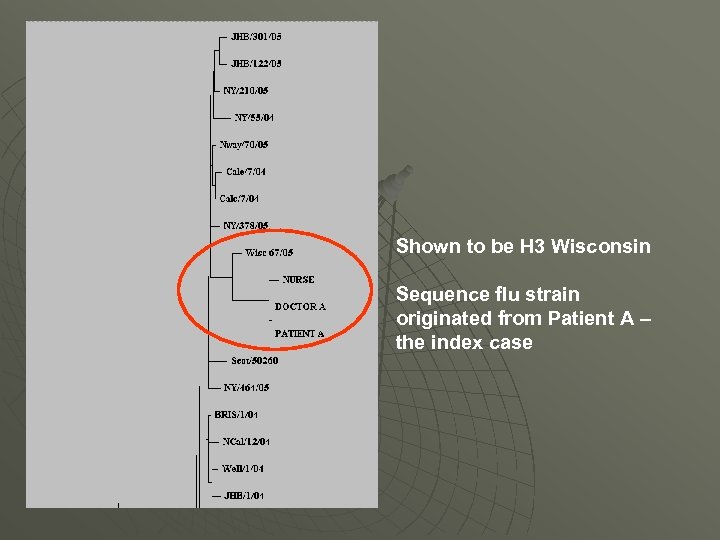 Shown to be H 3 Wisconsin Sequence flu strain originated from Patient A – the index case
Shown to be H 3 Wisconsin Sequence flu strain originated from Patient A – the index case
 Flu outbreak Hospital Y Flu virus sequenced Patient D – flu positive Doctor C – flu positive Phylogenetic tree created Patient E – flu positive Was there a connection with hospital X?
Flu outbreak Hospital Y Flu virus sequenced Patient D – flu positive Doctor C – flu positive Phylogenetic tree created Patient E – flu positive Was there a connection with hospital X?
 Hospital X No connection between Hospital X and Y Tree may have looked like this Hospital Y BUT THERE WAS A CONNECTION
Hospital X No connection between Hospital X and Y Tree may have looked like this Hospital Y BUT THERE WAS A CONNECTION
 Showed both flu outbreaks were connected All were H 3 Wisconsin What was the connection?
Showed both flu outbreaks were connected All were H 3 Wisconsin What was the connection?
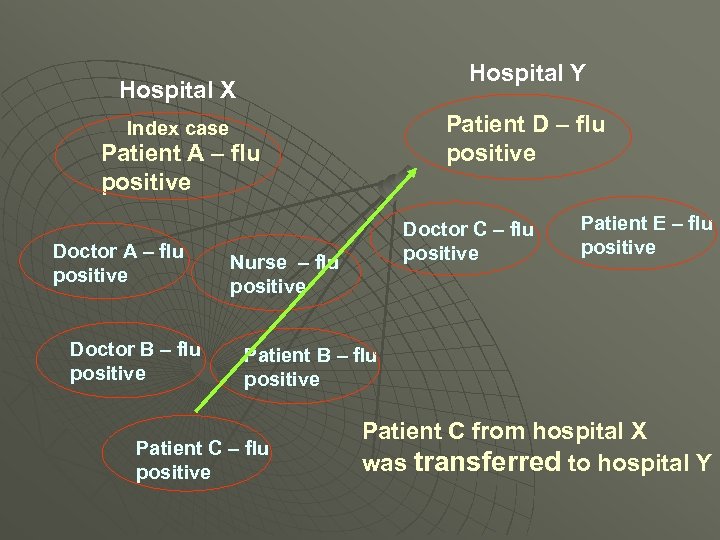 Hospital Y Hospital X Patient D – flu positive Index case Patient A – flu positive Doctor B – flu positive Doctor C – flu positive Nurse – flu positive Patient E – flu positive Patient B – flu positive Patient C from hospital X was transferred to hospital Y
Hospital Y Hospital X Patient D – flu positive Index case Patient A – flu positive Doctor B – flu positive Doctor C – flu positive Nurse – flu positive Patient E – flu positive Patient B – flu positive Patient C from hospital X was transferred to hospital Y
 Molecular epidemiology for outbreak sequencing u Implications • Shows connections between patients/staff • Raises infection control issues Patient transferred while ill u Why were staff infected u • Re-evaluate hospital procedures u E. g. masks, gowns, gloves, hand washing
Molecular epidemiology for outbreak sequencing u Implications • Shows connections between patients/staff • Raises infection control issues Patient transferred while ill u Why were staff infected u • Re-evaluate hospital procedures u E. g. masks, gowns, gloves, hand washing
 A case of Respiratory infection in BMT-Sheffield u u u 37 year old male post BMT Presented with GVHD in December 07 Third week of march 08 - respiratory symptoms- ? Infection, ? Respiratory GVHD Respiratory and PCP PCRs- HMPV PCR positive 25/3, 7/4 Not treated initially but subsequently treated with I/V and nebulised Ribavirin due to deterioration in respiratory symptoms. Died 14/4
A case of Respiratory infection in BMT-Sheffield u u u 37 year old male post BMT Presented with GVHD in December 07 Third week of march 08 - respiratory symptoms- ? Infection, ? Respiratory GVHD Respiratory and PCP PCRs- HMPV PCR positive 25/3, 7/4 Not treated initially but subsequently treated with I/V and nebulised Ribavirin due to deterioration in respiratory symptoms. Died 14/4
 Post-mortem histology of lung u u Sections from both the lungs show fibrin and macrophages in the alveolar spaces along with focal squamous metaplasia. There are scattered large bizzare cells with basophilic inclusions in the cytoplasm. The features are those of an organizing pneumonia with virocytopathic effect suggesting of viral aetiology.
Post-mortem histology of lung u u Sections from both the lungs show fibrin and macrophages in the alveolar spaces along with focal squamous metaplasia. There are scattered large bizzare cells with basophilic inclusions in the cytoplasm. The features are those of an organizing pneumonia with virocytopathic effect suggesting of viral aetiology.
 Human Metapneumovirus Discovered in 2000. u Paramyxoviridae u Negative sense, Single stranded RNA u Two genotypes A and B u
Human Metapneumovirus Discovered in 2000. u Paramyxoviridae u Negative sense, Single stranded RNA u Two genotypes A and B u
 Clinical Problems Upper respiratory infection u Lower respiratory infection u Non-specific symptoms u Fatalities reported in BMT patients u
Clinical Problems Upper respiratory infection u Lower respiratory infection u Non-specific symptoms u Fatalities reported in BMT patients u
 Objective To determine the incidence u 1 st September 2005 to 31 May 2006 u
Objective To determine the incidence u 1 st September 2005 to 31 May 2006 u
 Methods Data collection-retrospectively u Descriptive methods u
Methods Data collection-retrospectively u Descriptive methods u
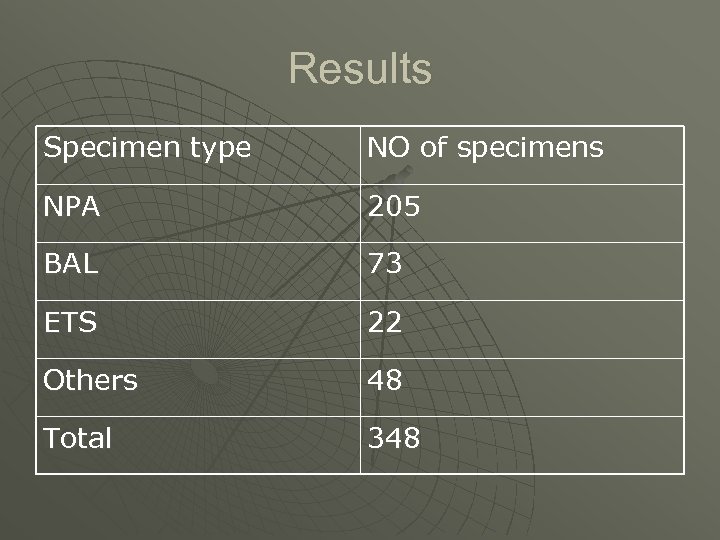 Results Specimen type NO of specimens NPA 205 BAL 73 ETS 22 Others 48 Total 348
Results Specimen type NO of specimens NPA 205 BAL 73 ETS 22 Others 48 Total 348
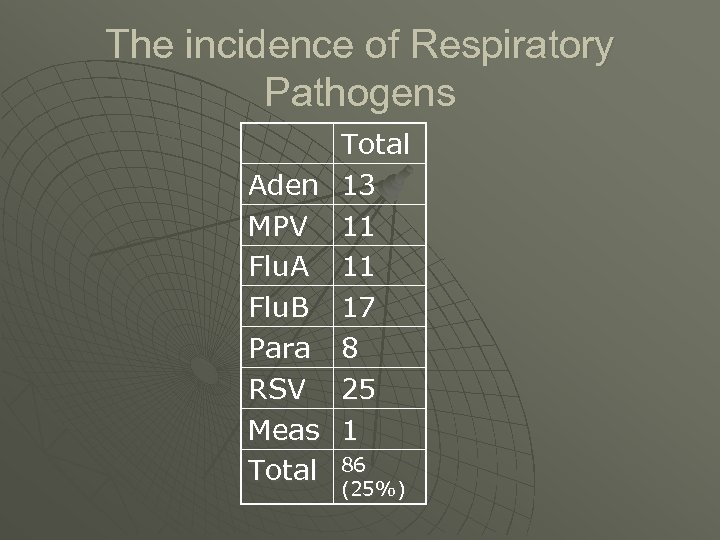 The incidence of Respiratory Pathogens Aden MPV Flu. A Flu. B Para RSV Meas Total 13 11 11 17 8 25 1 86 (25%)
The incidence of Respiratory Pathogens Aden MPV Flu. A Flu. B Para RSV Meas Total 13 11 11 17 8 25 1 86 (25%)
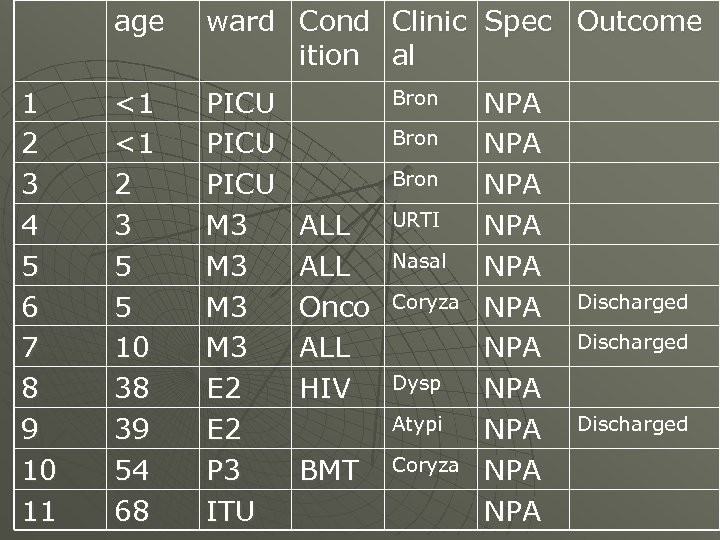 age 1 2 3 4 5 6 7 8 9 10 11 ward Cond Clinic Spec Outcome ition al <1 <1 2 3 5 5 10 38 39 54 68 PICU M 3 M 3 E 2 P 3 ITU Bron ALL Onco ALL HIV URTI Nasal Coryza Dysp Atypi BMT Coryza NPA NPA NPA Discharged
age 1 2 3 4 5 6 7 8 9 10 11 ward Cond Clinic Spec Outcome ition al <1 <1 2 3 5 5 10 38 39 54 68 PICU M 3 M 3 E 2 P 3 ITU Bron ALL Onco ALL HIV URTI Nasal Coryza Dysp Atypi BMT Coryza NPA NPA NPA Discharged
 Finding HMPV -4 th commonest respiratory pathogen u Affected all age groups u Detected in patients with both upper and lower respiratory tract infections u Some patients discharged before results were available u Further studies for clinical significance u
Finding HMPV -4 th commonest respiratory pathogen u Affected all age groups u Detected in patients with both upper and lower respiratory tract infections u Some patients discharged before results were available u Further studies for clinical significance u
 New viruses- human Bocavirus (HBo. V) Identified in 2005 u DNA virus belonging to family Parvoviridae u Found in respiratory secretions from children with and with out respiratory symptoms u Exact role in respiratory infections to be still worked out u
New viruses- human Bocavirus (HBo. V) Identified in 2005 u DNA virus belonging to family Parvoviridae u Found in respiratory secretions from children with and with out respiratory symptoms u Exact role in respiratory infections to be still worked out u
 u How feasible is it to introduce PCR in routine diagnosis
u How feasible is it to introduce PCR in routine diagnosis
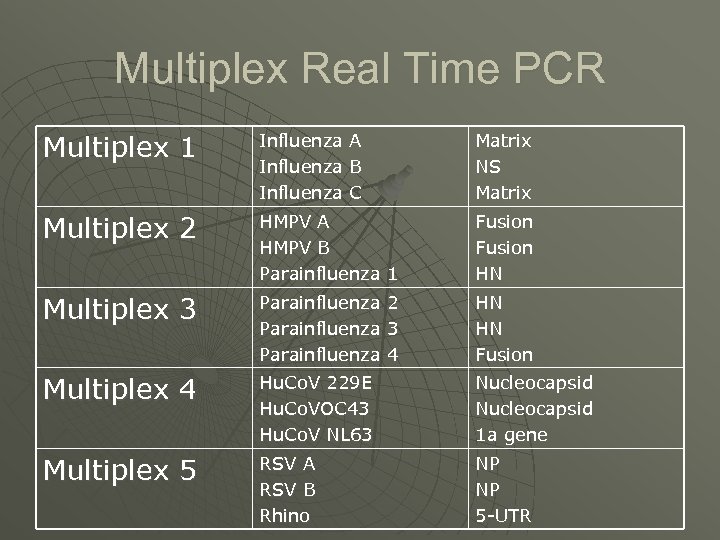 Multiplex Real Time PCR Multiplex 1 Influenza Multiplex 2 HMPV A HMPV B Parainfluenza 1 Fusion HN Multiplex 3 Parainfluenza 2 Parainfluenza 3 Parainfluenza 4 HN HN Fusion Multiplex 4 Hu. Co. V 229 E Hu. Co. VOC 43 Hu. Co. V NL 63 Nucleocapsid 1 a gene Multiplex 5 RSV A RSV B Rhino NP NP 5 -UTR A B C Matrix NS Matrix
Multiplex Real Time PCR Multiplex 1 Influenza Multiplex 2 HMPV A HMPV B Parainfluenza 1 Fusion HN Multiplex 3 Parainfluenza 2 Parainfluenza 3 Parainfluenza 4 HN HN Fusion Multiplex 4 Hu. Co. V 229 E Hu. Co. VOC 43 Hu. Co. V NL 63 Nucleocapsid 1 a gene Multiplex 5 RSV A RSV B Rhino NP NP 5 -UTR A B C Matrix NS Matrix
 Small numbers of tests/More pathogens detected Year Format No of tests 2000 -01 2001 -02 Tests performed (cumulative) influenza A, B; RSV; adenovirus; picornavirus Gel based nested 4 2002 -03 influenza A, B; RSV; adenovirus; picornavirus; 2003 -04 5 influenza A, B; RSV; adenovirus; Rhinovirus; PF 1, 2, 3 2004 -05 5 influenza A, B; RSV A + B; adenovirus; rhinovirus; PF 1, 2, 3; coronavirus NL 63, 229 e, OC 43; Hu. MPV 6 influenza A, B, C; RSV A + B; adenovirus; rhinovirus; PF 1, 2, 3, 4; coronavirus NL 63, 229 e, OC 43; Hu. MPV A + B 2005 -06 2007 -08 Real time PCR 5 influenza A, B, C; RSV A + B; adenovirus; rhinovirus; PF 1, 2, 3, 4; coronavirus NL 63, 229 e, OC 43; Hu. MPV A + B; M pneumoniae
Small numbers of tests/More pathogens detected Year Format No of tests 2000 -01 2001 -02 Tests performed (cumulative) influenza A, B; RSV; adenovirus; picornavirus Gel based nested 4 2002 -03 influenza A, B; RSV; adenovirus; picornavirus; 2003 -04 5 influenza A, B; RSV; adenovirus; Rhinovirus; PF 1, 2, 3 2004 -05 5 influenza A, B; RSV A + B; adenovirus; rhinovirus; PF 1, 2, 3; coronavirus NL 63, 229 e, OC 43; Hu. MPV 6 influenza A, B, C; RSV A + B; adenovirus; rhinovirus; PF 1, 2, 3, 4; coronavirus NL 63, 229 e, OC 43; Hu. MPV A + B 2005 -06 2007 -08 Real time PCR 5 influenza A, B, C; RSV A + B; adenovirus; rhinovirus; PF 1, 2, 3, 4; coronavirus NL 63, 229 e, OC 43; Hu. MPV A + B; M pneumoniae
 Advantages of PCR The utilisation of PCR conferred many advantages: – Highly sensitive/specific • • Applicable to RNA or DNA viruses Rapid (turn around time of 24 -48 hours) Can detect multiple viruses Products can be sequenced for epidemiological/resistance study. • Improved patient management and disease surveillance But……………. . : – Post amplification processing – Contamination – Prolonged testing time – Non-automated – Expensive to implement/expertise needed – Qualitative (difficult to quantify) Submitted by R Gunson June 2003
Advantages of PCR The utilisation of PCR conferred many advantages: – Highly sensitive/specific • • Applicable to RNA or DNA viruses Rapid (turn around time of 24 -48 hours) Can detect multiple viruses Products can be sequenced for epidemiological/resistance study. • Improved patient management and disease surveillance But……………. . : – Post amplification processing – Contamination – Prolonged testing time – Non-automated – Expensive to implement/expertise needed – Qualitative (difficult to quantify) Submitted by R Gunson June 2003
 Real time PCR: Unlike conventional PCR: – – Amplicon is visualised as the amplification progresses. Exponential rather than endpoint analysis – Closed system n. No post-amplification processing n. Rapid n. Reduced contamination n. Automation/high throughput/Cost effective – Increased sensitivity/specificity – More tests /less reagents/standardised cycling conditions Disadvantages of real time PCR: – – – Submitted by R Gunson June 2003 Risk of false negative reactions (due to miss-matches). Number of amplicons detected is limited by the number of fluorophores. Expensive to implement
Real time PCR: Unlike conventional PCR: – – Amplicon is visualised as the amplification progresses. Exponential rather than endpoint analysis – Closed system n. No post-amplification processing n. Rapid n. Reduced contamination n. Automation/high throughput/Cost effective – Increased sensitivity/specificity – More tests /less reagents/standardised cycling conditions Disadvantages of real time PCR: – – – Submitted by R Gunson June 2003 Risk of false negative reactions (due to miss-matches). Number of amplicons detected is limited by the number of fluorophores. Expensive to implement
 Examples of the benefits of real time PCR assays in viral respiratory infection u Gueudin et al: • Developed a real time PCR to detect, subgroup, and quantitate RSV A and B u u u RSV A and B to be responsible to differing disease severities Found higher viral loads in more severe infections Elden et al: • Developed a real time PCR for simultaneous detection of influenza A and B. u Rapid diagnosis allowed timely therapeutic and infection control intervention u Quantitation could be used to examine the effects of antiviral therapy u Mackay et al: • Developed a sensitive real time PCR for Human metapneumovirus u Most sensitive assay currently available u Puhakka et al: • Examined the effect of zanamivir on the viral load of influenza R Gunson Submitted by u Viral loads were reduced significantly June 2003
Examples of the benefits of real time PCR assays in viral respiratory infection u Gueudin et al: • Developed a real time PCR to detect, subgroup, and quantitate RSV A and B u u u RSV A and B to be responsible to differing disease severities Found higher viral loads in more severe infections Elden et al: • Developed a real time PCR for simultaneous detection of influenza A and B. u Rapid diagnosis allowed timely therapeutic and infection control intervention u Quantitation could be used to examine the effects of antiviral therapy u Mackay et al: • Developed a sensitive real time PCR for Human metapneumovirus u Most sensitive assay currently available u Puhakka et al: • Examined the effect of zanamivir on the viral load of influenza R Gunson Submitted by u Viral loads were reduced significantly June 2003
 Summary PCR for respiratory viruses are sensitive and specific u Positivity rate of 50% or greater u Cell culture sensitivity 30 -50% compared to PCR (for viruses that can be cultured) u Detection rate of PCR improved further as many viruses not culturable. u
Summary PCR for respiratory viruses are sensitive and specific u Positivity rate of 50% or greater u Cell culture sensitivity 30 -50% compared to PCR (for viruses that can be cultured) u Detection rate of PCR improved further as many viruses not culturable. u
 Summary- continued Several viruses can be tested for at the same time by multiplex PCR u In-house PCR cost effective compared to cell culture u PCR effective epidemiological tool in investigation of outbreaks u Rapid and sensitive assay aids in clinical management of respiratory infections. u
Summary- continued Several viruses can be tested for at the same time by multiplex PCR u In-house PCR cost effective compared to cell culture u PCR effective epidemiological tool in investigation of outbreaks u Rapid and sensitive assay aids in clinical management of respiratory infections. u



