5ed47466792d1da4b85ebbfd602be094.ppt
- Количество слайдов: 19
 Molecular and clinical determinants of survival following relapse after curative treatment of stage IIIII colon cancer (CC). Results of the translational study on the PETACC 3 - EORTC 40993 - SAKK 6000 trial A. D. Roth, D. Klingbiel, P. Yan, R. Fiocca, M. Delorenzi, R. Labianca, D. Cunningham, E. Van Cutsem, F. Bosman, S. Tejpar Taiwan 2000 PETACC 2009 3 ASCO 2010
Molecular and clinical determinants of survival following relapse after curative treatment of stage IIIII colon cancer (CC). Results of the translational study on the PETACC 3 - EORTC 40993 - SAKK 6000 trial A. D. Roth, D. Klingbiel, P. Yan, R. Fiocca, M. Delorenzi, R. Labianca, D. Cunningham, E. Van Cutsem, F. Bosman, S. Tejpar Taiwan 2000 PETACC 2009 3 ASCO 2010
 Rationale • Our previous results showed that stage II and III colon cancers harbour different biomarker alteration frequencies with different prognostic effects according to disease stage (ASCO proc. 2009, 27 abstr. #4002) • There is a lack of data regarding biomarkers prognostic for disease behaviour after relapse • Early observations suggest that marker sets prognostic for the risk of relapse might be different from those prognostic for survival after relapse (SAR) PETACC 3 ASCO 2010
Rationale • Our previous results showed that stage II and III colon cancers harbour different biomarker alteration frequencies with different prognostic effects according to disease stage (ASCO proc. 2009, 27 abstr. #4002) • There is a lack of data regarding biomarkers prognostic for disease behaviour after relapse • Early observations suggest that marker sets prognostic for the risk of relapse might be different from those prognostic for survival after relapse (SAR) PETACC 3 ASCO 2010
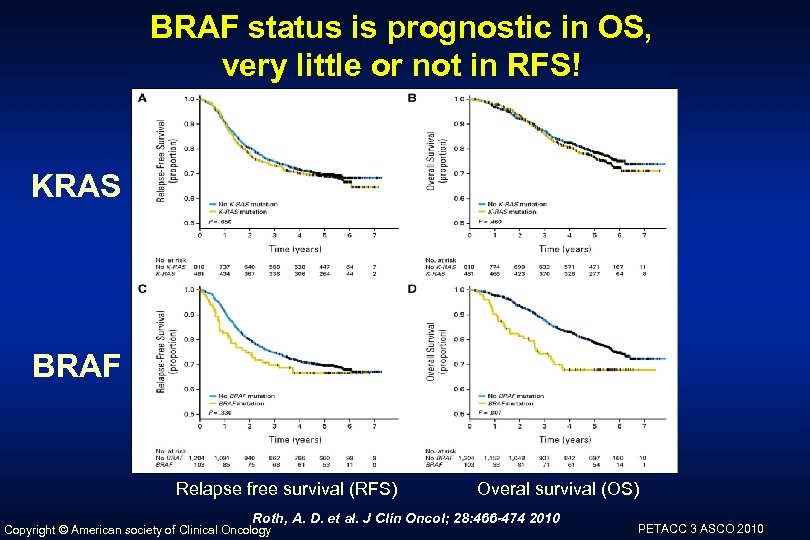 BRAF status is prognostic in OS, very little or not in RFS! KRAS BRAF Relapse free survival (RFS) Overal survival (OS) Roth, A. D. et al. J Clin Oncol; 28: 466 -474 2010 Copyright © American society of Clinical Oncology PETACC 3 ASCO 2010
BRAF status is prognostic in OS, very little or not in RFS! KRAS BRAF Relapse free survival (RFS) Overal survival (OS) Roth, A. D. et al. J Clin Oncol; 28: 466 -474 2010 Copyright © American society of Clinical Oncology PETACC 3 ASCO 2010
 Objectives • To look for clinical and molecular markers prognostic for survival after relapse (SAR). • To assess their respective and relative prognostic relevance in the relapsing patient population using SAR as endpoint. • To assess possible differences between markers prognostic for risk of relapse and markers prognostic for SAR. PETACC 3 ASCO 2010
Objectives • To look for clinical and molecular markers prognostic for survival after relapse (SAR). • To assess their respective and relative prognostic relevance in the relapsing patient population using SAR as endpoint. • To assess possible differences between markers prognostic for risk of relapse and markers prognostic for SAR. PETACC 3 ASCO 2010
 PETACC 3 trial: Patient Distribution Total Accrual: 3278 NO suitable material for biomarkers # relapsing 3241 Patients treated 1837 1404 598 392 (32. 5%) With suitable material for biomarkers (27. 9%) # relapsing PETACC 3 ASCO 2010
PETACC 3 trial: Patient Distribution Total Accrual: 3278 NO suitable material for biomarkers # relapsing 3241 Patients treated 1837 1404 598 392 (32. 5%) With suitable material for biomarkers (27. 9%) # relapsing PETACC 3 ASCO 2010
 Methods (1) • FFPE tissue blocks prospectively collected and cut in 5 -20µ sections • Immunohistochemistry (IHC) – P 53: mouse m. Ab clone D 07, ABC Basic DAB Detection (Ventana medical systems) – SMAD 4: mouse m. Ab clone B 8 (Ig. G 1, Santa Cruz Biotechnology). Novocastra polymer detection kit – Thymidylate Synthetase (TS): Monoclonal antibody TS 106/4 H 4 B 1 (Ig. G 1, Zymed). DAKO En. Vision detection system – Telomerase (HTERT): Monoclonal antibody NCL-h. TERT (Ig. G 2, Novocastra). DAKO En. Vision detection system PETACC 3 ASCO 2010
Methods (1) • FFPE tissue blocks prospectively collected and cut in 5 -20µ sections • Immunohistochemistry (IHC) – P 53: mouse m. Ab clone D 07, ABC Basic DAB Detection (Ventana medical systems) – SMAD 4: mouse m. Ab clone B 8 (Ig. G 1, Santa Cruz Biotechnology). Novocastra polymer detection kit – Thymidylate Synthetase (TS): Monoclonal antibody TS 106/4 H 4 B 1 (Ig. G 1, Zymed). DAKO En. Vision detection system – Telomerase (HTERT): Monoclonal antibody NCL-h. TERT (Ig. G 2, Novocastra). DAKO En. Vision detection system PETACC 3 ASCO 2010
 Methods (2) • DNA was extracted with phenol/chloroform from normal and tumoral tissues after macrodissection of FFPE sections. • Molecular analysis: – Microsatellite Instability (MSI): assessed on 10 markers (BAT-25, BAT-26, D 5 S 346, D 2 S 123, D 17 S 250, BAT 40, TGF-ß RII, D 18 S 58, D 18 S 69, D 17 S 787) < 3 positive markers = MSI stable (MSS) ≥ 3 positive markers = MSI high (MSI-H) – 18 q. LOH: multiple SNPs typing by pyrosequencing on Normal and tumor DNA – KRAS exon 2 and BRAF exon 15: Allele specific real time PCR on Tumor DNA PETACC 3 ASCO 2010
Methods (2) • DNA was extracted with phenol/chloroform from normal and tumoral tissues after macrodissection of FFPE sections. • Molecular analysis: – Microsatellite Instability (MSI): assessed on 10 markers (BAT-25, BAT-26, D 5 S 346, D 2 S 123, D 17 S 250, BAT 40, TGF-ß RII, D 18 S 58, D 18 S 69, D 17 S 787) < 3 positive markers = MSI stable (MSS) ≥ 3 positive markers = MSI high (MSI-H) – 18 q. LOH: multiple SNPs typing by pyrosequencing on Normal and tumor DNA – KRAS exon 2 and BRAF exon 15: Allele specific real time PCR on Tumor DNA PETACC 3 ASCO 2010
 Statistical methods • Prognostic value of the markers was analyzed by Cox regression for SAR and adjusted for stage • p values of the multivariate analysis taken from the full model (unless indicated otherwise) • Time to recurrence (TTR) of ≤ 18 months defined early relapse (ER), >18 months late relapse (LR) • Survival curves were computed according to the Kaplan-Meier method and compared using the logrank test PETACC 3 ASCO 2010
Statistical methods • Prognostic value of the markers was analyzed by Cox regression for SAR and adjusted for stage • p values of the multivariate analysis taken from the full model (unless indicated otherwise) • Time to recurrence (TTR) of ≤ 18 months defined early relapse (ER), >18 months late relapse (LR) • Survival curves were computed according to the Kaplan-Meier method and compared using the logrank test PETACC 3 ASCO 2010
 PETACC 3 trial: Relapser Distribution With material (biomarker set) Full set 392 990 Ø material 598 221 ER 171 LR 43. 6% 343 ER 255 LR 42. 6% PETACC 3 ASCO 2010
PETACC 3 trial: Relapser Distribution With material (biomarker set) Full set 392 990 Ø material 598 221 ER 171 LR 43. 6% 343 ER 255 LR 42. 6% PETACC 3 ASCO 2010
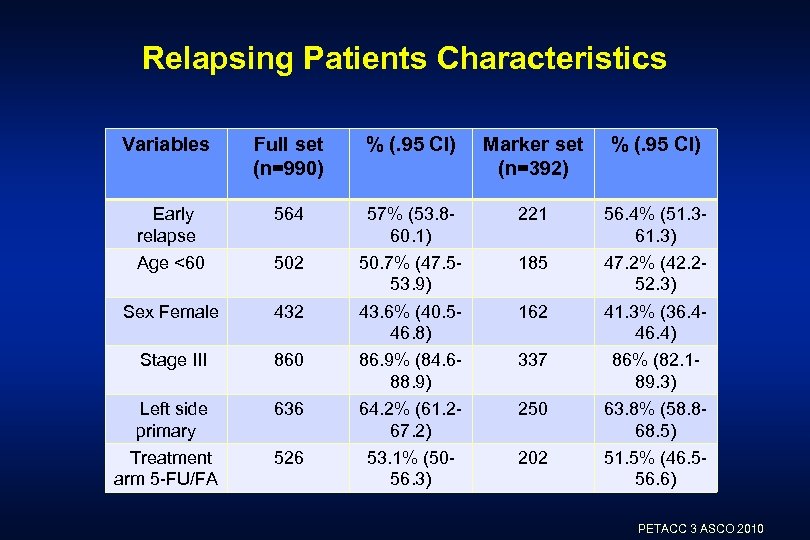 Relapsing Patients Characteristics Variables Full set (n=990) % (. 95 CI) Marker set (n=392) % (. 95 CI) Early relapse 564 57% (53. 860. 1) 221 56. 4% (51. 361. 3) Age <60 502 50. 7% (47. 553. 9) 185 47. 2% (42. 252. 3) Sex Female 432 43. 6% (40. 546. 8) 162 41. 3% (36. 446. 4) Stage III 860 86. 9% (84. 688. 9) 337 86% (82. 189. 3) Left side primary 636 64. 2% (61. 267. 2) 250 63. 8% (58. 868. 5) 526 53. 1% (5056. 3) 202 51. 5% (46. 556. 6) Treatment arm 5 -FU/FA PETACC 3 ASCO 2010
Relapsing Patients Characteristics Variables Full set (n=990) % (. 95 CI) Marker set (n=392) % (. 95 CI) Early relapse 564 57% (53. 860. 1) 221 56. 4% (51. 361. 3) Age <60 502 50. 7% (47. 553. 9) 185 47. 2% (42. 252. 3) Sex Female 432 43. 6% (40. 546. 8) 162 41. 3% (36. 446. 4) Stage III 860 86. 9% (84. 688. 9) 337 86% (82. 189. 3) Left side primary 636 64. 2% (61. 267. 2) 250 63. 8% (58. 868. 5) 526 53. 1% (5056. 3) 202 51. 5% (46. 556. 6) Treatment arm 5 -FU/FA PETACC 3 ASCO 2010
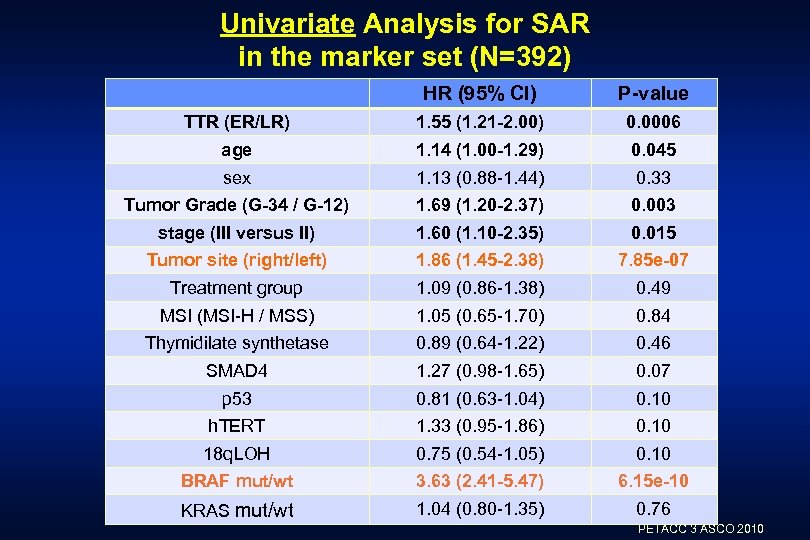 Univariate Analysis for SAR in the marker set (N=392) HR (95% CI) P-value TTR (ER/LR) 1. 55 (1. 21 -2. 00) 0. 0006 age 1. 14 (1. 00 -1. 29) 0. 045 sex 1. 13 (0. 88 -1. 44) 0. 33 Tumor Grade (G-34 / G-12) 1. 69 (1. 20 -2. 37) 0. 003 stage (III versus II) 1. 60 (1. 10 -2. 35) 0. 015 Tumor site (right/left) 1. 86 (1. 45 -2. 38) 7. 85 e-07 Treatment group 1. 09 (0. 86 -1. 38) 0. 49 MSI (MSI-H / MSS) 1. 05 (0. 65 -1. 70) 0. 84 Thymidilate synthetase 0. 89 (0. 64 -1. 22) 0. 46 SMAD 4 1. 27 (0. 98 -1. 65) 0. 07 p 53 0. 81 (0. 63 -1. 04) 0. 10 h. TERT 1. 33 (0. 95 -1. 86) 0. 10 18 q. LOH 0. 75 (0. 54 -1. 05) 0. 10 BRAF mut/wt 3. 63 (2. 41 -5. 47) 6. 15 e-10 KRAS mut/wt 1. 04 (0. 80 -1. 35) 0. 76 PETACC 3 ASCO 2010
Univariate Analysis for SAR in the marker set (N=392) HR (95% CI) P-value TTR (ER/LR) 1. 55 (1. 21 -2. 00) 0. 0006 age 1. 14 (1. 00 -1. 29) 0. 045 sex 1. 13 (0. 88 -1. 44) 0. 33 Tumor Grade (G-34 / G-12) 1. 69 (1. 20 -2. 37) 0. 003 stage (III versus II) 1. 60 (1. 10 -2. 35) 0. 015 Tumor site (right/left) 1. 86 (1. 45 -2. 38) 7. 85 e-07 Treatment group 1. 09 (0. 86 -1. 38) 0. 49 MSI (MSI-H / MSS) 1. 05 (0. 65 -1. 70) 0. 84 Thymidilate synthetase 0. 89 (0. 64 -1. 22) 0. 46 SMAD 4 1. 27 (0. 98 -1. 65) 0. 07 p 53 0. 81 (0. 63 -1. 04) 0. 10 h. TERT 1. 33 (0. 95 -1. 86) 0. 10 18 q. LOH 0. 75 (0. 54 -1. 05) 0. 10 BRAF mut/wt 3. 63 (2. 41 -5. 47) 6. 15 e-10 KRAS mut/wt 1. 04 (0. 80 -1. 35) 0. 76 PETACC 3 ASCO 2010
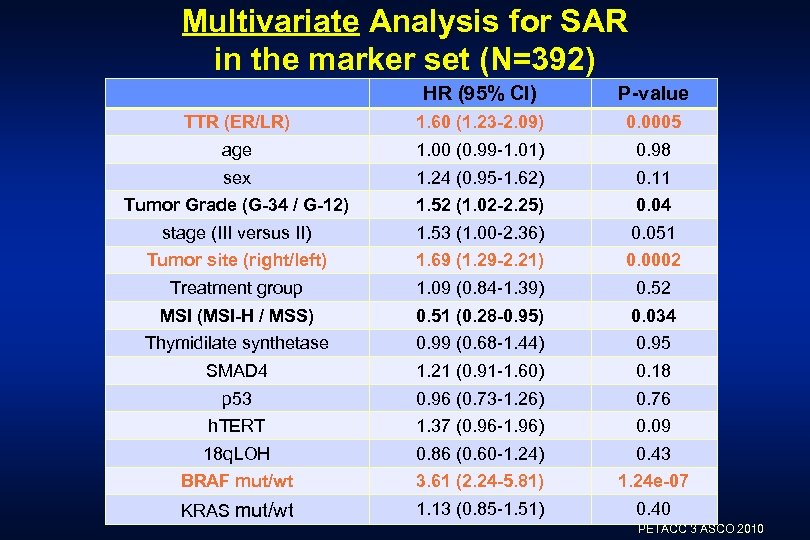 Multivariate Analysis for SAR in the marker set (N=392) HR (95% CI) P-value TTR (ER/LR) 1. 60 (1. 23 -2. 09) 0. 0005 age 1. 00 (0. 99 -1. 01) 0. 98 sex 1. 24 (0. 95 -1. 62) 0. 11 Tumor Grade (G-34 / G-12) 1. 52 (1. 02 -2. 25) 0. 04 stage (III versus II) 1. 53 (1. 00 -2. 36) 0. 051 Tumor site (right/left) 1. 69 (1. 29 -2. 21) 0. 0002 Treatment group 1. 09 (0. 84 -1. 39) 0. 52 MSI (MSI-H / MSS) 0. 51 (0. 28 -0. 95) 0. 034 Thymidilate synthetase 0. 99 (0. 68 -1. 44) 0. 95 SMAD 4 1. 21 (0. 91 -1. 60) 0. 18 p 53 0. 96 (0. 73 -1. 26) 0. 76 h. TERT 1. 37 (0. 96 -1. 96) 0. 09 18 q. LOH 0. 86 (0. 60 -1. 24) 0. 43 BRAF mut/wt 3. 61 (2. 24 -5. 81) 1. 24 e-07 KRAS mut/wt 1. 13 (0. 85 -1. 51) 0. 40 PETACC 3 ASCO 2010
Multivariate Analysis for SAR in the marker set (N=392) HR (95% CI) P-value TTR (ER/LR) 1. 60 (1. 23 -2. 09) 0. 0005 age 1. 00 (0. 99 -1. 01) 0. 98 sex 1. 24 (0. 95 -1. 62) 0. 11 Tumor Grade (G-34 / G-12) 1. 52 (1. 02 -2. 25) 0. 04 stage (III versus II) 1. 53 (1. 00 -2. 36) 0. 051 Tumor site (right/left) 1. 69 (1. 29 -2. 21) 0. 0002 Treatment group 1. 09 (0. 84 -1. 39) 0. 52 MSI (MSI-H / MSS) 0. 51 (0. 28 -0. 95) 0. 034 Thymidilate synthetase 0. 99 (0. 68 -1. 44) 0. 95 SMAD 4 1. 21 (0. 91 -1. 60) 0. 18 p 53 0. 96 (0. 73 -1. 26) 0. 76 h. TERT 1. 37 (0. 96 -1. 96) 0. 09 18 q. LOH 0. 86 (0. 60 -1. 24) 0. 43 BRAF mut/wt 3. 61 (2. 24 -5. 81) 1. 24 e-07 KRAS mut/wt 1. 13 (0. 85 -1. 51) 0. 40 PETACC 3 ASCO 2010
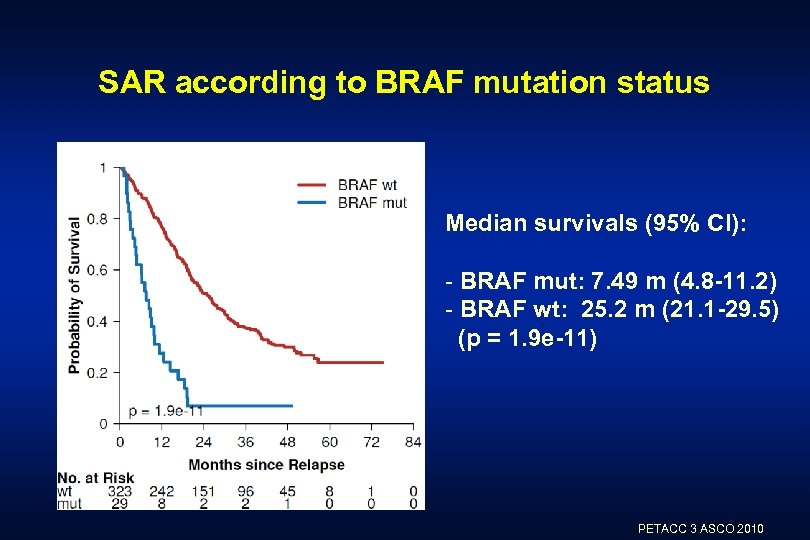 SAR according to BRAF mutation status Median survivals (95% CI): - BRAF mut: 7. 49 m (4. 8 -11. 2) - BRAF wt: 25. 2 m (21. 1 -29. 5) (p = 1. 9 e-11) PETACC 3 ASCO 2010
SAR according to BRAF mutation status Median survivals (95% CI): - BRAF mut: 7. 49 m (4. 8 -11. 2) - BRAF wt: 25. 2 m (21. 1 -29. 5) (p = 1. 9 e-11) PETACC 3 ASCO 2010
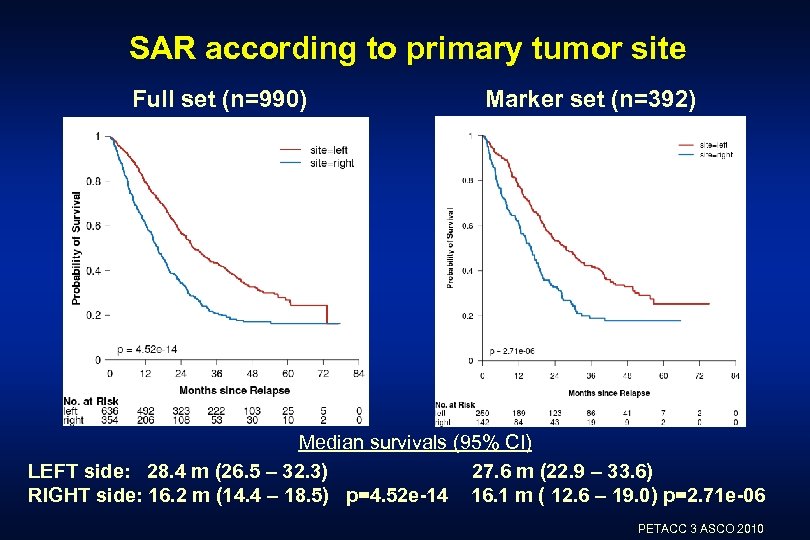 SAR according to primary tumor site Full set (n=990) Marker set (n=392) Median survivals (95% CI) LEFT side: 28. 4 m (26. 5 – 32. 3) 27. 6 m (22. 9 – 33. 6) RIGHT side: 16. 2 m (14. 4 – 18. 5) p=4. 52 e-14 16. 1 m ( 12. 6 – 19. 0) p=2. 71 e-06 PETACC 3 ASCO 2010
SAR according to primary tumor site Full set (n=990) Marker set (n=392) Median survivals (95% CI) LEFT side: 28. 4 m (26. 5 – 32. 3) 27. 6 m (22. 9 – 33. 6) RIGHT side: 16. 2 m (14. 4 – 18. 5) p=4. 52 e-14 16. 1 m ( 12. 6 – 19. 0) p=2. 71 e-06 PETACC 3 ASCO 2010
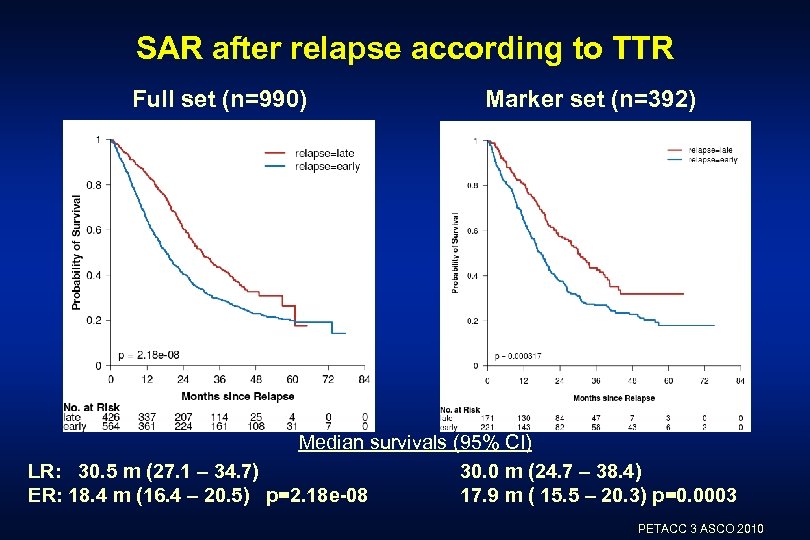 SAR after relapse according to TTR Full set (n=990) Marker set (n=392) Median survivals (95% CI) LR: 30. 5 m (27. 1 – 34. 7) 30. 0 m (24. 7 – 38. 4) ER: 18. 4 m (16. 4 – 20. 5) p=2. 18 e-08 17. 9 m ( 15. 5 – 20. 3) p=0. 0003 PETACC 3 ASCO 2010
SAR after relapse according to TTR Full set (n=990) Marker set (n=392) Median survivals (95% CI) LR: 30. 5 m (27. 1 – 34. 7) 30. 0 m (24. 7 – 38. 4) ER: 18. 4 m (16. 4 – 20. 5) p=2. 18 e-08 17. 9 m ( 15. 5 – 20. 3) p=0. 0003 PETACC 3 ASCO 2010
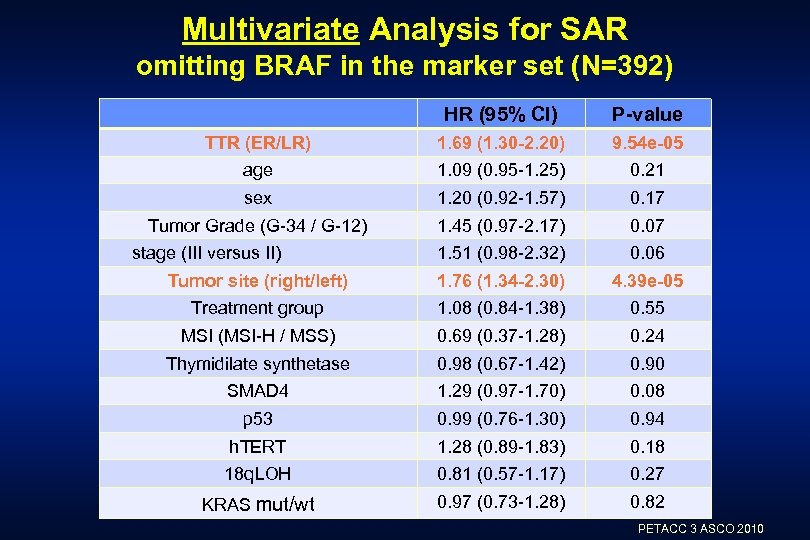 Multivariate Analysis for SAR omitting BRAF in the marker set (N=392) HR (95% CI) P-value TTR (ER/LR) 1. 69 (1. 30 -2. 20) 9. 54 e-05 age 1. 09 (0. 95 -1. 25) 0. 21 sex 1. 20 (0. 92 -1. 57) 0. 17 Tumor Grade (G-34 / G-12) 1. 45 (0. 97 -2. 17) 0. 07 1. 51 (0. 98 -2. 32) 0. 06 Tumor site (right/left) 1. 76 (1. 34 -2. 30) 4. 39 e-05 Treatment group 1. 08 (0. 84 -1. 38) 0. 55 MSI (MSI-H / MSS) 0. 69 (0. 37 -1. 28) 0. 24 Thymidilate synthetase 0. 98 (0. 67 -1. 42) 0. 90 SMAD 4 1. 29 (0. 97 -1. 70) 0. 08 p 53 0. 99 (0. 76 -1. 30) 0. 94 h. TERT 1. 28 (0. 89 -1. 83) 0. 18 18 q. LOH 0. 81 (0. 57 -1. 17) 0. 27 KRAS mut/wt 0. 97 (0. 73 -1. 28) 0. 82 stage (III versus II) PETACC 3 ASCO 2010
Multivariate Analysis for SAR omitting BRAF in the marker set (N=392) HR (95% CI) P-value TTR (ER/LR) 1. 69 (1. 30 -2. 20) 9. 54 e-05 age 1. 09 (0. 95 -1. 25) 0. 21 sex 1. 20 (0. 92 -1. 57) 0. 17 Tumor Grade (G-34 / G-12) 1. 45 (0. 97 -2. 17) 0. 07 1. 51 (0. 98 -2. 32) 0. 06 Tumor site (right/left) 1. 76 (1. 34 -2. 30) 4. 39 e-05 Treatment group 1. 08 (0. 84 -1. 38) 0. 55 MSI (MSI-H / MSS) 0. 69 (0. 37 -1. 28) 0. 24 Thymidilate synthetase 0. 98 (0. 67 -1. 42) 0. 90 SMAD 4 1. 29 (0. 97 -1. 70) 0. 08 p 53 0. 99 (0. 76 -1. 30) 0. 94 h. TERT 1. 28 (0. 89 -1. 83) 0. 18 18 q. LOH 0. 81 (0. 57 -1. 17) 0. 27 KRAS mut/wt 0. 97 (0. 73 -1. 28) 0. 82 stage (III versus II) PETACC 3 ASCO 2010
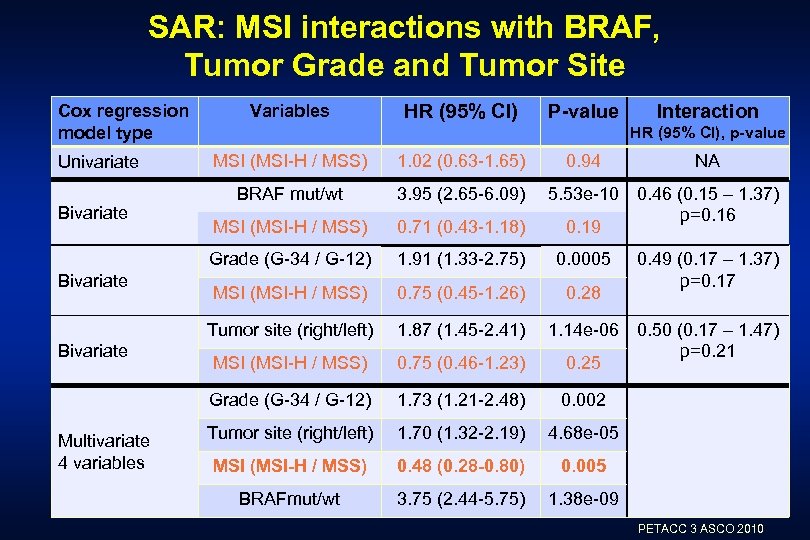 SAR: MSI interactions with BRAF, Tumor Grade and Tumor Site Cox regression model type Variables HR (95% CI) P-value HR (95% CI), p-value Bivariate Multivariate 4 variables BRAF mut/wt 3. 95 (2. 65 -6. 09) MSI (MSI-H / MSS) 0. 71 (0. 43 -1. 18) 1. 91 (1. 33 -2. 75) 0. 0005 MSI (MSI-H / MSS) 0. 75 (0. 45 -1. 26) 0. 28 1. 87 (1. 45 -2. 41) MSI (MSI-H / MSS) 0. 75 (0. 46 -1. 23) Grade (G-34 / G-12) Bivariate 1. 02 (0. 63 -1. 65) Tumor site (right/left) Bivariate MSI (MSI-H / MSS) Grade (G-34 / G-12) Univariate Interaction 0. 94 1. 73 (1. 21 -2. 48) 0. 002 Tumor site (right/left) 1. 70 (1. 32 -2. 19) 4. 68 e-05 MSI (MSI-H / MSS) 0. 48 (0. 28 -0. 80) 0. 005 BRAFmut/wt 3. 75 (2. 44 -5. 75) NA 1. 38 e-09 5. 53 e-10 0. 46 (0. 15 – 1. 37) p=0. 16 0. 19 0. 49 (0. 17 – 1. 37) p=0. 17 1. 14 e-06 0. 50 (0. 17 – 1. 47) p=0. 21 0. 25 PETACC 3 ASCO 2010
SAR: MSI interactions with BRAF, Tumor Grade and Tumor Site Cox regression model type Variables HR (95% CI) P-value HR (95% CI), p-value Bivariate Multivariate 4 variables BRAF mut/wt 3. 95 (2. 65 -6. 09) MSI (MSI-H / MSS) 0. 71 (0. 43 -1. 18) 1. 91 (1. 33 -2. 75) 0. 0005 MSI (MSI-H / MSS) 0. 75 (0. 45 -1. 26) 0. 28 1. 87 (1. 45 -2. 41) MSI (MSI-H / MSS) 0. 75 (0. 46 -1. 23) Grade (G-34 / G-12) Bivariate 1. 02 (0. 63 -1. 65) Tumor site (right/left) Bivariate MSI (MSI-H / MSS) Grade (G-34 / G-12) Univariate Interaction 0. 94 1. 73 (1. 21 -2. 48) 0. 002 Tumor site (right/left) 1. 70 (1. 32 -2. 19) 4. 68 e-05 MSI (MSI-H / MSS) 0. 48 (0. 28 -0. 80) 0. 005 BRAFmut/wt 3. 75 (2. 44 -5. 75) NA 1. 38 e-09 5. 53 e-10 0. 46 (0. 15 – 1. 37) p=0. 16 0. 19 0. 49 (0. 17 – 1. 37) p=0. 17 1. 14 e-06 0. 50 (0. 17 – 1. 47) p=0. 21 0. 25 PETACC 3 ASCO 2010
 Conclusions • Whereas BRAF status and tumor site were not prognostic for RFS of stage II-III colon cancer (Roth, A. D. et al. J Clin Oncol; 28: 466 -474 2010), BRAF status, tumor site and TTR are highly prognostic for colon cancer survival after relapse • Tumor stage at diagnosis and MSI status have a weaker impact on survival after relapse • BRAF status, tumor site and TTR should imperatively be used to stratify studies in metastatic colon cancer • The independent effects of tumor site and TTR might be indicative of additional as yet non-identified prognostically significant molecular markers PETACC 3 ASCO 2010
Conclusions • Whereas BRAF status and tumor site were not prognostic for RFS of stage II-III colon cancer (Roth, A. D. et al. J Clin Oncol; 28: 466 -474 2010), BRAF status, tumor site and TTR are highly prognostic for colon cancer survival after relapse • Tumor stage at diagnosis and MSI status have a weaker impact on survival after relapse • BRAF status, tumor site and TTR should imperatively be used to stratify studies in metastatic colon cancer • The independent effects of tumor site and TTR might be indicative of additional as yet non-identified prognostically significant molecular markers PETACC 3 ASCO 2010
 Thank You For All Your Efforts!! Austria, Belgium, Bulgaria, Croatia, Czech Republic, Denmark, Egypt, Finland, France, Germany, Greece & Cyprus, Hungary, Ireland, Iceland, Israel, Italy, Netherlands, Norway, Poland, Portugal, Russia, South Africa, Slovakia, Slovenia, Spain, Sweden, Switzerland, Taiwan, Turkey & UK We would also like to thank Pfizer for facilitating the execution and analysis of the PETACC 3 study PETACC 3 ASCO 2010
Thank You For All Your Efforts!! Austria, Belgium, Bulgaria, Croatia, Czech Republic, Denmark, Egypt, Finland, France, Germany, Greece & Cyprus, Hungary, Ireland, Iceland, Israel, Italy, Netherlands, Norway, Poland, Portugal, Russia, South Africa, Slovakia, Slovenia, Spain, Sweden, Switzerland, Taiwan, Turkey & UK We would also like to thank Pfizer for facilitating the execution and analysis of the PETACC 3 study PETACC 3 ASCO 2010


