1bd89e0f38af3035ad09cff5b4b12651.ppt
- Количество слайдов: 14

Ministry of Health Republic of Kazakhstan SALAMATTY KAZAKHSTAN Accessibility of Opioids in the Republic of Kazakhstan G. U. Raimkulova – Head of Pharmaceutical Activities Administration , Medical and Pharmaceutical Activities Committee, Ministry of Health of the Republic of Kazakhstan Almaty 2012 1

State Regulation of Drug Distribution Managed by the following: 1) state regulation of the pharmaceutical activities; 2) licensing the pharmaceutical activities and narcotic, psychotropic substance and precursor distribution activities in the field of health; 3) state marketing authorization of the medicinal products, health products and medical devices (MPHPMD); 4) safety and quality assessment of the marketed medicinal and health products; 5) state price control of medicinal products, purchased under the scope of the guaranteed free medical care. (Code of the Republic of Kazakhstan –On National Health and Health System) 2

Regulatory base of the provision of narcotic, psychotropic substances and precursors 1. Code of the Republic of Kazakhstan (Ro. K) “On National Health and Health care System”; 2. Law of the Republic of Kazakhstan as of July 10, 1998, No 279 -I “On narcotic, psychotropic substances and precursors, and countermeasures to their trafficking and abuse”; 3

State regulation modes for narcotic, psychotropic substances and precursors distribution (NPP) • 1. quota allocation – demand rate for the Republic of Kazakhstan in NPP based on • 2. distribution – Republican internal market provision with specified remedies and • 3. licensing (narcotic, psychotropic substances, precursors distribution activities, • 4. inspections (narcotic, psychotropic substances and precursors distribution order requirement computation. Is approved by Government of the Republic of Kazakhstan and submitted in accordance with international treaties for international quota approval Republic of Kazakhstan UN INCB); professional activities guarantee for different agencies under allocated quota in accordance with regulations: - provision standard is to be approved for importing domestic manufacturers and legal bodies. - consumption standard is to be approved for republican administrativeterritorial units indicating standards for individual consumer organizations (therapeutic, pharmaceutical, veterinarian, academic, and research organization etc. ); NPP import and export); compliance monitoring). 4
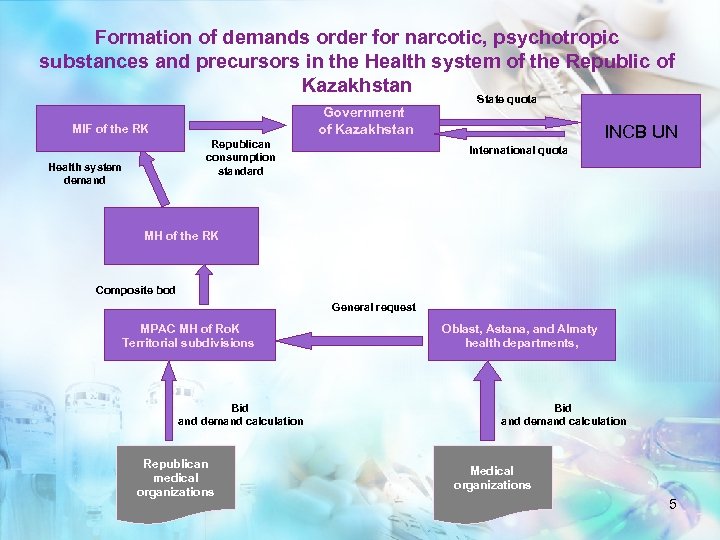
Formation of demands order for narcotic, psychotropic substances and precursors in the Health system of the Republic of Kazakhstan Government of Kazakhstan MIF of the RK Republican consumption standard Health system demand State quota INCB UN International quota MH of the RK Composite bod General request MPAC MH of Ro. K Territorial subdivisions Bid and demand calculation Republican medical organizations Oblast, Astana, and Almaty health departments, Bid and demand calculation Medical organizations 5
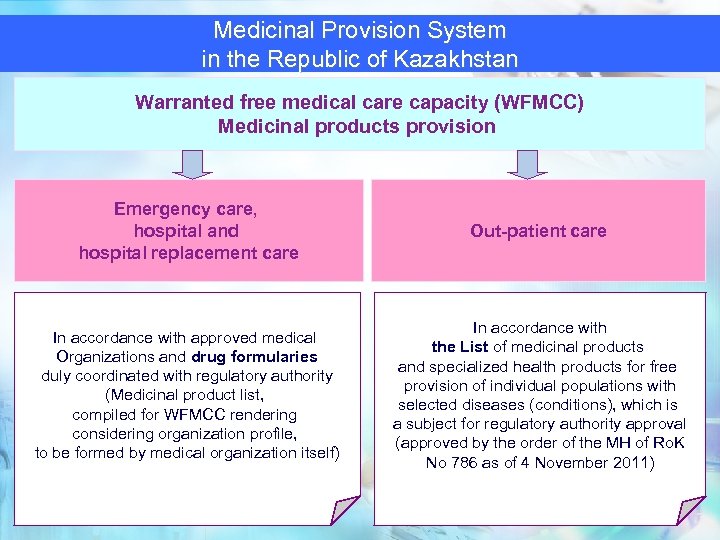
Medicinal Provision System in the Republic of Kazakhstan Warranted free medical care capacity (WFMCC) Medicinal products provision Emergency care, hospital and hospital replacement care Out-patient care In accordance with approved medical Organizations and drug formularies duly coordinated with regulatory authority (Medicinal product list, compiled for WFMCC rendering considering organization profile, to be formed by medical organization itself) In accordance with the List of medicinal products and specialized health products for free provision of individual populations with selected diseases (conditions), which is a subject for regulatory authority approval (approved by the order of the MH of Ro. K No 786 as of 4 November 2011) 66

Republican Drug Formulary International non-proprietary name Pharmaceutical form* No Опиоидные анальгетики 1. Morphine solution for injection 1 % 2. Trimeperidine solution for injection 1 %, 2 % Tramadol solution for injection 100 mg/2 ml, 50 mg/m. L, 5%; oral solution 100 mg/1 m. L; drops 1000 mg/10 m. L; film-coated tablet 50 mg; retard tablet (film-coated tablet) 100 mg, 150 mg, 200 mg; capsules 50 mg; rectal suppositories 4. Fentanyl solution for injection 0, 005 %; transdermal therapeutic system 12, 5 mcg/hour, 25 mcg/hour, 50 mcg/hour, 75 mcg/hour, 100 mcg/hour; sublingual tablet 100 mg, 200 mg, 300 mg, 400 mg, 600 mg, 800 mg 5. Butorphanol solution for injection 2 mg/m. L 3. * All registered in the Republic of Kazakhstan pharmaceutical forms are to be released 7

Drug list in accordance with List (Order No 786) Palliative therapy for malignancies • Morphine, solution for injection; • Trimeperidine hydrochloride, solution for injection; • Tramadol, sustained release tablet, capsule, suppositories, solution for injection; • Fentanyl, transdermal therapeutic system; • Ketoprofen, capsule, tablet, suppositories, solution for injection; • Diazepam, tablet, solution for injection; 8

Opioids availability at the pharmaceutical market of the Republic of Kazakhstan (in accordance with State MPHPMD registry as of 01. 2013) Nowadays, around 77 items of various pharmaceutical forms and dosages for following medicinal products are present in the market of the Republic of Kazakhstan: 1. Morphine hydrochloride – 1 PP (solution for injection); 2. Fentanyl – 16 PP (solution for injection, transdermal therapeutic system and sublingual tablet); 3. Trimeperidine– 1 PP (solution for injection); 4. Tramadol – 41 PP (capsule, drops, tablet, sustained release tablet, solution for injection, suppositories); 5. Combined codein-containing drugs – 18 PP (tablet, syrup, capsule); 9
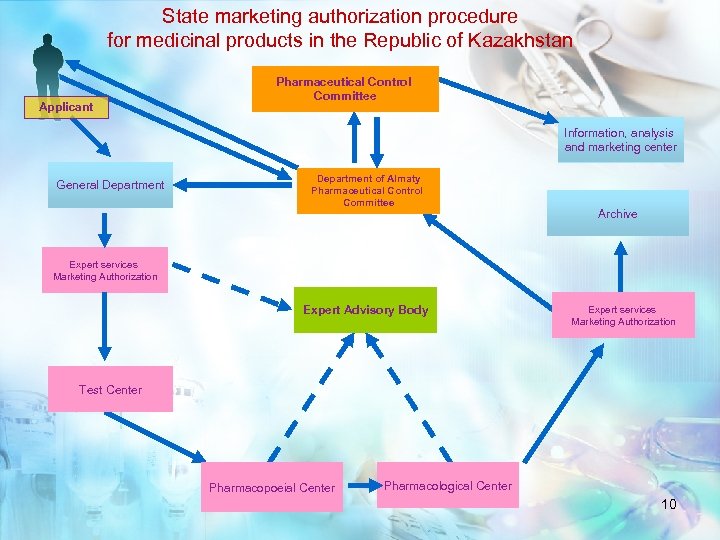
State marketing authorization procedure for medicinal products in the Republic of Kazakhstan Applicant Pharmaceutical Control Committee Information, analysis and marketing center General Department of Almaty Pharmaceutical Control Committee Archive Expert services Marketing Authorization Expert Advisory Body Expert services Marketing Authorization Test Center Pharmacopoeial Center Pharmacological Center 10

State Marketing Authorization for MPHPMD Ø Upon expertise completion the Expert Report on Drug Safety, Efficacy, and Quality is compiled Ø Based on Expert Report the MPAC MH of the Ro. K makes decision and issue an order on medicinal product state marketing authorization Ø All data on registered MP within 24 hours are entered in the State MP Registry An applicant is to be granted by: Ø Ø Marketing Authorization Approved Company Core Data Sheet Medicinal Product Quality Regulatory document (AND) Package design 11
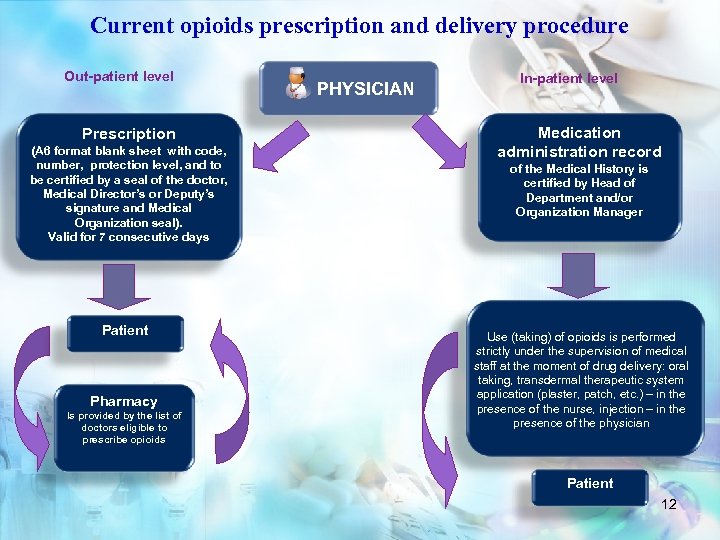
Current opioids prescription and delivery procedure Out-patient level Prescription (А 6 format blank sheet with code, number, protection level, and to be certified by a seal of the doctor, Medical Director’s or Deputy’s signature and Medical Organization seal). Valid for 7 consecutive days Patient Pharmacy Is provided by the list of doctors eligible to prescribe opioids PHYSICIAN In-patient level Medication administration record of the Medical History is certified by Head of Department and/or Organization Manager Use (taking) of opioids is performed strictly under the supervision of medical staff at the moment of drug delivery: oral taking, transdermal therapeutic system application (plaster, patch, etc. ) – in the presence of the nurse, injection – in the presence of the physician Patient 12

Opioids storage and destruction requirements 1. The persons, authorized to work with the opioids, shall prior get the conclusions from psychiatrist and narcologist regarding the absence of narcotic, toxic substances, alcohol abuse, and Internal affairs authorities conclusion regarding relevant inspection. List of Лица, persons authorized to work with narcotic, psychotropic substances and precursors is to be approved by the order of the Health допущенные к работе с опиоидными organization Manager annually. 2. Storage facilities, lock boxes, and cupboards must be in blocking state. Outside office hours these are to be sealed up, keys and seal врачей психиатра and нарколога об press are to be kept in a responsible person. отсутствии заболеваний наркоманией, 3. токсикоманией, хроническим On the inside of the lock box door, cupboards the check-list of kept medicinal products indicating highest single and daily doses 4. All documents on the opioid income side and consumption are to be kept in a filed, sealed form by months and years in responsible выполнению данной деятельности and person in conditions providing their security in the lock boxes and metallic cupboards within storage facility. заключение органов внутренних дел по 5. Documents on the opioid income side and consumption are to be kept for five years. 6. On the inside of the door of the lock box, cupboard the check-list of kept medicinal products containing narcotic, psychotropic substances and precursors indicating highest single and daily doses. 7. Medicinal products, containing narcotic, psychotropic substances and precursors are to be delivered form the storage facility only by the responsible person. 8. In the assistant room stock of narcotic, psychotropic substances from the Table II of the List should not exceed five-day demand, in departments (offices) of health organizations – three-day demand, in pharmacy storage facilities — thirty-day demand, apart from holidays and weekends. 9. Unused special prescriptions given by dead patients relatives are to be kept with following destruction. Prescription destruction is to be performed as far as prescription accumulation, but not less than monthly, by incineration of the ongoing committee, included internal affairs body representative. Unused special prescriptions destruction fact is to be registered in relevant act. 10. To destroy the expired opioids, provided by the deceased patients’ family, breakage, rejects, empty ampules and plasters (transdermal therapeutic systems) as well, and ampules and plasters (transdermal therapeutic systems) with partially used content, under the order of the Health Organization Manager, a constantly functioning committee is to be developed including internal affairs body and sanitary-epidemiological services representatives. анальгетиками должны иметь заключения алкоголизмом, а также о пригодности к проведению соответствующей проверки. 13

Ministry of Health Republic of Kazakhstan SALAMATTY KAZAKHSTAN THANK YOU FOR YOUR ATTENTION 14
1bd89e0f38af3035ad09cff5b4b12651.ppt