Презентация англ.ppt
- Количество слайдов: 15
 Ministry of Education youth and sports of Ukraine Chernivtsi National University named Yuri Fedkovych Chemical Faculty Department of Inorganic Chemistry Term paper on: "Radioactivity. Families of radioactive elements" Fulfilled second-year student 202 groups Shevchenko Victoria Supervisor: Korov'yanko О. О.
Ministry of Education youth and sports of Ukraine Chernivtsi National University named Yuri Fedkovych Chemical Faculty Department of Inorganic Chemistry Term paper on: "Radioactivity. Families of radioactive elements" Fulfilled second-year student 202 groups Shevchenko Victoria Supervisor: Korov'yanko О. О.
 Content: l l l l 1. Radioactivity 1. 1. Alpha radioactivity 1. 2. Alpha barrier penetration 1. 3. Alpha, beta and gamma 1. 4. Penetration of matter 2. Radioactive decay 3. Radioactive series 3. 1. Thorium series 3. 2. Neptunium series 3. 3. Radium series 3. 4. Actinium series 4. Uses of radioactivity 5. Literature
Content: l l l l 1. Radioactivity 1. 1. Alpha radioactivity 1. 2. Alpha barrier penetration 1. 3. Alpha, beta and gamma 1. 4. Penetration of matter 2. Radioactive decay 3. Radioactive series 3. 1. Thorium series 3. 2. Neptunium series 3. 3. Radium series 3. 4. Actinium series 4. Uses of radioactivity 5. Literature
 Radioactivity l l Radioactivity refers to the particles which are emitted from nuclei as a result of nuclear instability. The most common types of radiation are called alpha, beta, and gamma radiation, but there are several other varieties of radioactive decay. The different types of radioactivity lead to different decay paths which transmute the nuclei into other chemical elements.
Radioactivity l l Radioactivity refers to the particles which are emitted from nuclei as a result of nuclear instability. The most common types of radiation are called alpha, beta, and gamma radiation, but there are several other varieties of radioactive decay. The different types of radioactivity lead to different decay paths which transmute the nuclei into other chemical elements.
 Alpha radioactivity l Alpha Radioactivity сomposed of two protons and two neutrons, the alpha particle is a nucleus of the element helium.
Alpha radioactivity l Alpha Radioactivity сomposed of two protons and two neutrons, the alpha particle is a nucleus of the element helium.
 Alpha barrier penetration l The energy of emitted alpha particles was a mystery to early investigators because it was evident that they did not have enough energy, according to classical physics, to escape the nucleus. Once an approximate size of the nucleus was obtained by Rutherford scattering, one could calculate the height of the Coulomb barrier at the radius of the nucleus. It was evident that this energy was several times higher than the observed alpha particle energies. There was also an incredible range of half lives for the alpha particle which could not be explained by anything in classical physics. Alpha particle emission is modeled as a barrier penetration process. The alpha particle is the nucleus of the helium atom and is the nucleus of highest stability.
Alpha barrier penetration l The energy of emitted alpha particles was a mystery to early investigators because it was evident that they did not have enough energy, according to classical physics, to escape the nucleus. Once an approximate size of the nucleus was obtained by Rutherford scattering, one could calculate the height of the Coulomb barrier at the radius of the nucleus. It was evident that this energy was several times higher than the observed alpha particle energies. There was also an incredible range of half lives for the alpha particle which could not be explained by anything in classical physics. Alpha particle emission is modeled as a barrier penetration process. The alpha particle is the nucleus of the helium atom and is the nucleus of highest stability.
 Alpha, beta and gamma l Historically, the products of radioactivity were called alpha, beta, and gamma when it was found that they could be analyzed into three distinct species by either a magnetic field or an electric field.
Alpha, beta and gamma l Historically, the products of radioactivity were called alpha, beta, and gamma when it was found that they could be analyzed into three distinct species by either a magnetic field or an electric field.
 Penetration of matter l Though the most massive and most energetic of radioactive emissions, the alpha particle is the shortest in range because of its strong interaction with matter. The electromagnetic gamma ray is extremely penetrating, even penetrating considerable thicknesses of concrete. The electron of beta radioactivity strongly interacts with matter and has a short range.
Penetration of matter l Though the most massive and most energetic of radioactive emissions, the alpha particle is the shortest in range because of its strong interaction with matter. The electromagnetic gamma ray is extremely penetrating, even penetrating considerable thicknesses of concrete. The electron of beta radioactivity strongly interacts with matter and has a short range.
 Radioactive decay l l l Radioactive decay is the process by which an atomic nucleus of an unstable atom loses energy by emitting ionizing particles (ionizing radiation). The first decay processes to be discovered were alpha decay, beta decay, and gamma decay. Radioactive decay is a stochastic (i. e. , random) process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay.
Radioactive decay l l l Radioactive decay is the process by which an atomic nucleus of an unstable atom loses energy by emitting ionizing particles (ionizing radiation). The first decay processes to be discovered were alpha decay, beta decay, and gamma decay. Radioactive decay is a stochastic (i. e. , random) process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay.
 Radioactive series l In nuclear science, the decay chain refers to the radioactive decay of different discrete radioactive decay products as a chained series of transformations. Most radioactive elements do not decay directly to a stable state, but rather undergo a series of decays until eventually a stable isotope is reached.
Radioactive series l In nuclear science, the decay chain refers to the radioactive decay of different discrete radioactive decay products as a chained series of transformations. Most radioactive elements do not decay directly to a stable state, but rather undergo a series of decays until eventually a stable isotope is reached.
 Thorium series l The 4 n chain of Th-232 is commonly called the "thorium series. " Beginning with naturally occurring thorium-232, this series includes the following elements: actinium, bismuth, lead, polonium, radium, and radon. All are present, at least transiently, in any natural thorium-containing sample, whether metal, compound, or mineral.
Thorium series l The 4 n chain of Th-232 is commonly called the "thorium series. " Beginning with naturally occurring thorium-232, this series includes the following elements: actinium, bismuth, lead, polonium, radium, and radon. All are present, at least transiently, in any natural thorium-containing sample, whether metal, compound, or mineral.
 Neptunium series l The 4 n + 1 chain of Np-237 are commonly called the "neptunium series. " In this series, only two of the isotopes involved are found naturally, namely those of bismuth and thallium. A smoke detector containing an americium-241 ionization chamber accumulates a significant amount of neptunium-237 as its americium decays; the following elements are also present in it, at least transiently, as decay products of the neptunium: actinium, astatine, bismuth, francium, lead, polonium, protactinium, radium, thallium, thorium, and uranium. Since this series was only studied more recently, its nuclides do not have historic names.
Neptunium series l The 4 n + 1 chain of Np-237 are commonly called the "neptunium series. " In this series, only two of the isotopes involved are found naturally, namely those of bismuth and thallium. A smoke detector containing an americium-241 ionization chamber accumulates a significant amount of neptunium-237 as its americium decays; the following elements are also present in it, at least transiently, as decay products of the neptunium: actinium, astatine, bismuth, francium, lead, polonium, protactinium, radium, thallium, thorium, and uranium. Since this series was only studied more recently, its nuclides do not have historic names.
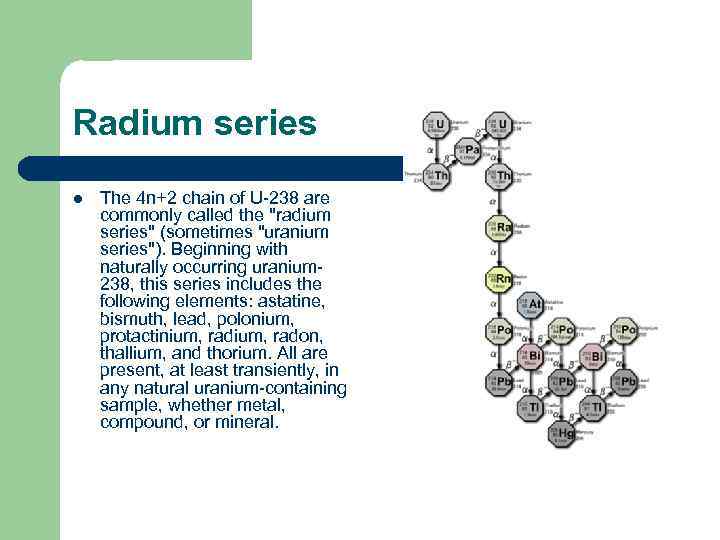 Radium series l The 4 n+2 chain of U-238 are commonly called the "radium series" (sometimes "uranium series"). Beginning with naturally occurring uranium 238, this series includes the following elements: astatine, bismuth, lead, polonium, protactinium, radon, thallium, and thorium. All are present, at least transiently, in any natural uranium-containing sample, whether metal, compound, or mineral.
Radium series l The 4 n+2 chain of U-238 are commonly called the "radium series" (sometimes "uranium series"). Beginning with naturally occurring uranium 238, this series includes the following elements: astatine, bismuth, lead, polonium, protactinium, radon, thallium, and thorium. All are present, at least transiently, in any natural uranium-containing sample, whether metal, compound, or mineral.
 Actinium series l The 4 n+3 chain of uranium-235 are commonly called the "actinium series". Beginning with the naturally-occurring isotope U-235, this decay series includes the following elements: Actinium, astatine, bismuth, francium, lead, polonium, protactinium, radium, radon, thallium, and thorium. All are present, at least transiently, in any sample containing uranium-235, whether metal, compound, ore, or mineral. This series terminates with the stable isotope lead-207.
Actinium series l The 4 n+3 chain of uranium-235 are commonly called the "actinium series". Beginning with the naturally-occurring isotope U-235, this decay series includes the following elements: Actinium, astatine, bismuth, francium, lead, polonium, protactinium, radium, radon, thallium, and thorium. All are present, at least transiently, in any sample containing uranium-235, whether metal, compound, ore, or mineral. This series terminates with the stable isotope lead-207.
 Uses of radioactivity Nuclear reactors l l There are many practical applications to the use of radioactivity/radiation. Radioactive sources are used to study living organisms, to diagnose and treat diseases, to sterilize medical instruments and food, to produce energy for heat and electric power, and to monitor various steps in all types of industrial processes. Radiation sources are extremely important to the manufacturing industries throughout the world. They are commonly employed by nondestructive testing personnel to monitor materials and processes in the making of the products we see and use every day. Trained technicians use radiography to image materials and products much like a dentist uses radiation to x-ray your teeth for cavities. There are many industrial applications that rely on radioactivity to assist in determining if the material or product is internally sound and fit for its application. Medical Uses for Radioactivity
Uses of radioactivity Nuclear reactors l l There are many practical applications to the use of radioactivity/radiation. Radioactive sources are used to study living organisms, to diagnose and treat diseases, to sterilize medical instruments and food, to produce energy for heat and electric power, and to monitor various steps in all types of industrial processes. Radiation sources are extremely important to the manufacturing industries throughout the world. They are commonly employed by nondestructive testing personnel to monitor materials and processes in the making of the products we see and use every day. Trained technicians use radiography to image materials and products much like a dentist uses radiation to x-ray your teeth for cavities. There are many industrial applications that rely on radioactivity to assist in determining if the material or product is internally sound and fit for its application. Medical Uses for Radioactivity
 Literature l l l 1. http: //hyperphysics. phy-astr. gsu. edu/hbase/nuclear/radact. html 2. http: //en. wikipedia. org 3. "Decay and Half Life". Retrieved 2009 -12 -14. 4. http: //www. epa. gov/radiation/understand/chain. html 5. http: //muller. lbl. gov/teaching/physics 10/old%20 physics%2010/physics%2010%20 notes/Ra dioisotope. Dating. html 6. http: //library. thinkquest. org/17940/texts/radioactivity. html 7. http: //www. ndted. org/Education. Resources/High. School/Radiography/producearadiograph. htm 8. Біленко І. І. Фізичний словник. — К. : Вища школа, Головне видав. 1979. — 336 с. 9. Романова Н. В. загальна та неорганічна хімія: Підруч. для студ. вищ. навч. закл. – К. ; Ірпінь: ВТФ «Перун» , 2007. – 480 с. 10. О. Панчук, Неорганічна хімія, ч. І, конспект лекцій, – Чернівці: Рута, 2007, 140 с. 11. Ф. М. Боднарюк, Загальна та неорганічна хімія, част. І, – Рівне: НУВГП, 2006, 241 с.
Literature l l l 1. http: //hyperphysics. phy-astr. gsu. edu/hbase/nuclear/radact. html 2. http: //en. wikipedia. org 3. "Decay and Half Life". Retrieved 2009 -12 -14. 4. http: //www. epa. gov/radiation/understand/chain. html 5. http: //muller. lbl. gov/teaching/physics 10/old%20 physics%2010/physics%2010%20 notes/Ra dioisotope. Dating. html 6. http: //library. thinkquest. org/17940/texts/radioactivity. html 7. http: //www. ndted. org/Education. Resources/High. School/Radiography/producearadiograph. htm 8. Біленко І. І. Фізичний словник. — К. : Вища школа, Головне видав. 1979. — 336 с. 9. Романова Н. В. загальна та неорганічна хімія: Підруч. для студ. вищ. навч. закл. – К. ; Ірпінь: ВТФ «Перун» , 2007. – 480 с. 10. О. Панчук, Неорганічна хімія, ч. І, конспект лекцій, – Чернівці: Рута, 2007, 140 с. 11. Ф. М. Боднарюк, Загальна та неорганічна хімія, част. І, – Рівне: НУВГП, 2006, 241 с.


