12757f0a7af76cd1c938084aa1ac5695.ppt
- Количество слайдов: 60
 Mine Drainage Control and Treatment Options Region 10 Webinar Workshops – Hardrock mine geochemistry and hydrology Workshop #3: Preparing for unexpected outcomes at mine sites and means to control contamination Barbara A. Butler March 5, 2013 1
Mine Drainage Control and Treatment Options Region 10 Webinar Workshops – Hardrock mine geochemistry and hydrology Workshop #3: Preparing for unexpected outcomes at mine sites and means to control contamination Barbara A. Butler March 5, 2013 1
 Disclaimer Mention of trade names or products does not constitute the U. S. EPA’s endorsement. The information presented is the sole responsibility of the author and does not necessarily represent the views of the U. S. EPA 2
Disclaimer Mention of trade names or products does not constitute the U. S. EPA’s endorsement. The information presented is the sole responsibility of the author and does not necessarily represent the views of the U. S. EPA 2
 Overview n n n Definitions How does mine-drainage form? Mitigation measures n n What happens when drainage enters a stream? n n Design and operation Precipitation and sorption reactions Remedial options n n Active treatment Passive (semi-passive) treatment 3
Overview n n n Definitions How does mine-drainage form? Mitigation measures n n What happens when drainage enters a stream? n n Design and operation Precipitation and sorption reactions Remedial options n n Active treatment Passive (semi-passive) treatment 3
 Definitions n Mitigation n All steps taken to prevent (avoid), reduce (minimize), treat, and compensate for any potential adverse impacts (risk) on the environment from a given activity (hazard) n Proper planning, design, construction, operation, management, and closure of waste and water containment and treatment facilities, monitoring and maintenance over all mine-life phases, including after mine closure. 4
Definitions n Mitigation n All steps taken to prevent (avoid), reduce (minimize), treat, and compensate for any potential adverse impacts (risk) on the environment from a given activity (hazard) n Proper planning, design, construction, operation, management, and closure of waste and water containment and treatment facilities, monitoring and maintenance over all mine-life phases, including after mine closure. 4
 Definitions n Reclamation Refers to restoring 1 a disturbed area to an acceptable form and planned use, following active mining n Includes actions taken to mitigate against future risks n 1 Rarely is a site able to be restored to what it was prior to mining, which is the strictest definition of restoration. 5
Definitions n Reclamation Refers to restoring 1 a disturbed area to an acceptable form and planned use, following active mining n Includes actions taken to mitigate against future risks n 1 Rarely is a site able to be restored to what it was prior to mining, which is the strictest definition of restoration. 5
 Definitions n Remediation n Refers to correcting an issue that has become evident, such as cleaning up a spill or an abandoned mine site 6
Definitions n Remediation n Refers to correcting an issue that has become evident, such as cleaning up a spill or an abandoned mine site 6
 Definitions n Best Management Practice (BMP) n With respect to the CWA, this term generally applies to specific measures for managing non-point source runoff from storm water (40 CFR Part 130. 2(m)). 7
Definitions n Best Management Practice (BMP) n With respect to the CWA, this term generally applies to specific measures for managing non-point source runoff from storm water (40 CFR Part 130. 2(m)). 7
 Overview n n n Definitions How does mine-drainage form? Mitigation measures n n What happens when drainage enters a stream? n n Design and operation Precipitation and sorption reactions Remedial options n n Active treatment Passive (semi-passive) treatment 8
Overview n n n Definitions How does mine-drainage form? Mitigation measures n n What happens when drainage enters a stream? n n Design and operation Precipitation and sorption reactions Remedial options n n Active treatment Passive (semi-passive) treatment 8
 How Does Mine-Drainage Form? n Mining Process Exposes pyrite and other minerals to the atmosphere n Grinding processes n n Larger surface areas for exposure (weather more quickly) Pyrite http: //en. wikipedia. org/wiki/File: Pyrite_3. jpg 9
How Does Mine-Drainage Form? n Mining Process Exposes pyrite and other minerals to the atmosphere n Grinding processes n n Larger surface areas for exposure (weather more quickly) Pyrite http: //en. wikipedia. org/wiki/File: Pyrite_3. jpg 9
 How Does Mine-Drainage Form? Initiator Reaction Pyrite Propagation Cycle 106 x faster Pyrite http: //en. wikipedia. org/wiki/File: Pyrite_3. jpg 10
How Does Mine-Drainage Form? Initiator Reaction Pyrite Propagation Cycle 106 x faster Pyrite http: //en. wikipedia. org/wiki/File: Pyrite_3. jpg 10
 How Does Mine-Drainage Form? n Acidic, neutral or alkaline 1? n Acid-producing reactions n n Pyritic minerals; oxyhydroxide formation Acid-consuming reactions n Carbonate minerals (produce alkalinity) n n Aluminosilicate (clay minerals) n n E. g. , limestone and dolomite E. g. , muscovite, kaolinite Formation from non-pyritic minerals 1 Mining-influenced waters (MIW) coined by Dr. Ron Schmiermund in 1997 to encompass all types 11
How Does Mine-Drainage Form? n Acidic, neutral or alkaline 1? n Acid-producing reactions n n Pyritic minerals; oxyhydroxide formation Acid-consuming reactions n Carbonate minerals (produce alkalinity) n n Aluminosilicate (clay minerals) n n E. g. , limestone and dolomite E. g. , muscovite, kaolinite Formation from non-pyritic minerals 1 Mining-influenced waters (MIW) coined by Dr. Ron Schmiermund in 1997 to encompass all types 11
 Overview n n n Definitions How does mine-drainage form? Mitigation measures n n What happens when drainage enters a stream? n n Design and operation Precipitation and sorption reactions Remedial options n n Active treatment Passive (semi-passive) treatment 12
Overview n n n Definitions How does mine-drainage form? Mitigation measures n n What happens when drainage enters a stream? n n Design and operation Precipitation and sorption reactions Remedial options n n Active treatment Passive (semi-passive) treatment 12
 Mitigation Measures n Waste rock piles n Tailings storage facilities Measures presented are not inclusive of all options available. 13
Mitigation Measures n Waste rock piles n Tailings storage facilities Measures presented are not inclusive of all options available. 13
 Mitigation Measures Waste Rock Piles n Minimize leaching of acidity and ions n Minimize exposure of water bodies to seepage/drainage n Minimize acid generation 14
Mitigation Measures Waste Rock Piles n Minimize leaching of acidity and ions n Minimize exposure of water bodies to seepage/drainage n Minimize acid generation 14
 Mitigation Measures Waste Rock Piles n Minimize leaching of acidity and ions n Encapsulation with impermeable layer n n Compaction n n Minimize infiltration, seepage, and oxygen transfer Minimizes voids Progressive reclamation 15
Mitigation Measures Waste Rock Piles n Minimize leaching of acidity and ions n Encapsulation with impermeable layer n n Compaction n n Minimize infiltration, seepage, and oxygen transfer Minimizes voids Progressive reclamation 15
 Mitigation Measures Waste Rock Piles n Minimize exposure of water bodies to seepage/drainage n Locating piles within cone of depression n n Drainage flows to pit and is pumped out with groundwater (GW) Under-drains, diversions, GW monitoring wells, liners (clay and/or geosynthetic), 16
Mitigation Measures Waste Rock Piles n Minimize exposure of water bodies to seepage/drainage n Locating piles within cone of depression n n Drainage flows to pit and is pumped out with groundwater (GW) Under-drains, diversions, GW monitoring wells, liners (clay and/or geosynthetic), 16
 Mitigation Measures Waste Rock Piles n Minimize acid generation Segregation of potentially acid-generating (PAG) rock with non-acid-generating (NAG) rock n Mining of sub-economic ore (PAG) as blending material, or at end of operations n Layering of PAG rock NAG rock n Blending with acid-consuming materials n Bactericide n Isolation (encapsulation) n 17
Mitigation Measures Waste Rock Piles n Minimize acid generation Segregation of potentially acid-generating (PAG) rock with non-acid-generating (NAG) rock n Mining of sub-economic ore (PAG) as blending material, or at end of operations n Layering of PAG rock NAG rock n Blending with acid-consuming materials n Bactericide n Isolation (encapsulation) n 17
 Mitigation Measures Tailings Storage Facilities n Wet storage n Most common n Mining uses a lot of water and facility provides dual purpose as storage Seismic and flood considerations Dry storage Filtered tailings removes maximum amount of water n Not appropriate for PAG n 18
Mitigation Measures Tailings Storage Facilities n Wet storage n Most common n Mining uses a lot of water and facility provides dual purpose as storage Seismic and flood considerations Dry storage Filtered tailings removes maximum amount of water n Not appropriate for PAG n 18
 Mitigation Measures Tailings Storage Facilities n Isolation of PAG material Selective processing methods n Sub-aqueous disposal to minimize oxidation n Encapsulation n 19
Mitigation Measures Tailings Storage Facilities n Isolation of PAG material Selective processing methods n Sub-aqueous disposal to minimize oxidation n Encapsulation n 19
 Mitigation Measures Tailings Storage Facilities n Minimize exposure of water bodies to seepage/drainage Liners, under-drains, decant systems n Diversions for clean water n Seepage collection systems n Monitoring wells downstream n Caps on beaches, drawdown water, erosion control – closure n 20
Mitigation Measures Tailings Storage Facilities n Minimize exposure of water bodies to seepage/drainage Liners, under-drains, decant systems n Diversions for clean water n Seepage collection systems n Monitoring wells downstream n Caps on beaches, drawdown water, erosion control – closure n 20
 Overview n n n Definitions How does mine-drainage form? Mitigation measures n n What happens when drainage enters a stream? n n Design and operation Precipitation and sorption reactions Remedial options n n Active treatment Passive (semi-passive) treatment 21
Overview n n n Definitions How does mine-drainage form? Mitigation measures n n What happens when drainage enters a stream? n n Design and operation Precipitation and sorption reactions Remedial options n n Active treatment Passive (semi-passive) treatment 21
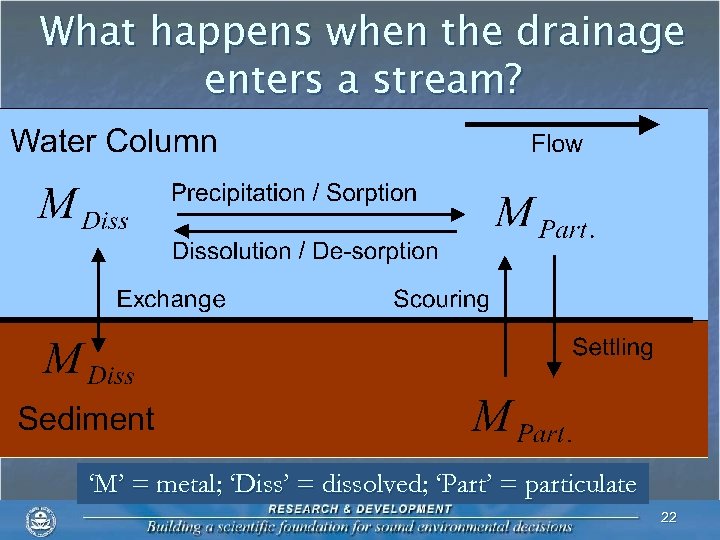 What happens when the drainage enters a stream? ‘M’ = metal; ‘Diss’ = dissolved; ‘Part’ = particulate 22
What happens when the drainage enters a stream? ‘M’ = metal; ‘Diss’ = dissolved; ‘Part’ = particulate 22
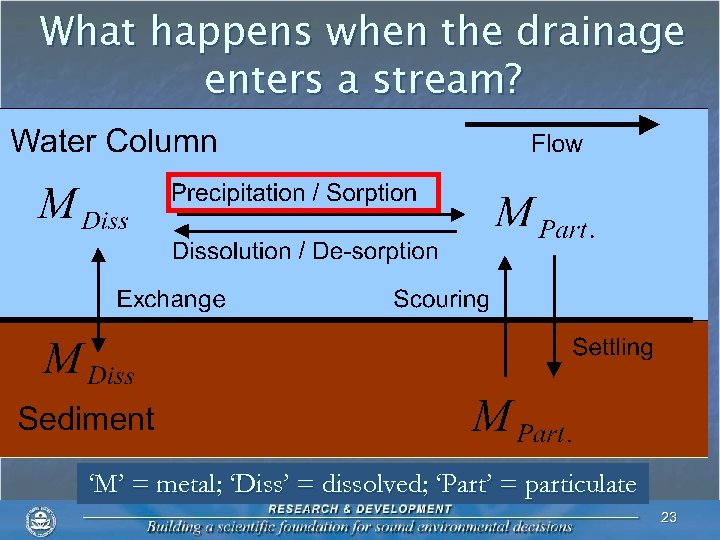 What happens when the drainage enters a stream? ‘M’ = metal; ‘Diss’ = dissolved; ‘Part’ = particulate 23
What happens when the drainage enters a stream? ‘M’ = metal; ‘Diss’ = dissolved; ‘Part’ = particulate 23
 What happens when the drainage enters a stream? n Iron (HFO) n Rapid formation in water column at p. H > ~ 3. 5 n Oxidation of ferrous iron precedes precipitation 24
What happens when the drainage enters a stream? n Iron (HFO) n Rapid formation in water column at p. H > ~ 3. 5 n Oxidation of ferrous iron precedes precipitation 24
 What happens when the drainage enters a stream? n Aluminum (ALO) n Rapid formation in water column at p. H > ~ 5 n No oxidation necessary 25
What happens when the drainage enters a stream? n Aluminum (ALO) n Rapid formation in water column at p. H > ~ 5 n No oxidation necessary 25
 What happens when the drainage enters a stream? n Manganese (HMO) n Requires oxidation n Requires a surface n Formation at p. H > ~ 9 26
What happens when the drainage enters a stream? n Manganese (HMO) n Requires oxidation n Requires a surface n Formation at p. H > ~ 9 26
 What happens when the drainage enters a stream? n Manganese (HMO) n Formation may be facilitated by periphyton 27
What happens when the drainage enters a stream? n Manganese (HMO) n Formation may be facilitated by periphyton 27
 What happens when the drainage enters a stream? n Manganese (HMO) n Formation may be facilitated by periphyton n Algal photosynthesis 28
What happens when the drainage enters a stream? n Manganese (HMO) n Formation may be facilitated by periphyton n Algal photosynthesis 28
 Overview n n n Definitions How does mine-drainage form? Mitigation measures n n What happens when drainage enters a stream? n n Design and operation Precipitation and sorption reactions Remedial options n n Active treatment Passive (semi-passive) treatment 29
Overview n n n Definitions How does mine-drainage form? Mitigation measures n n What happens when drainage enters a stream? n n Design and operation Precipitation and sorption reactions Remedial options n n Active treatment Passive (semi-passive) treatment 29
 Remedial Options n Isolation, removal, treatment of source Physical removal n Grouting reactive rock surfaces n Redirection of water flow n Adding neutralizing agents n 30
Remedial Options n Isolation, removal, treatment of source Physical removal n Grouting reactive rock surfaces n Redirection of water flow n Adding neutralizing agents n 30
 Remedial Options n Water treatment Removal of acidity n Removal of metals n Removal of salts (Na, Mg, Ca, K) n Removal of sulfate n Ultimately – removal of toxicity n 31
Remedial Options n Water treatment Removal of acidity n Removal of metals n Removal of salts (Na, Mg, Ca, K) n Removal of sulfate n Ultimately – removal of toxicity n 31
 Remedial Options Water Treatment n Active n Passive 32
Remedial Options Water Treatment n Active n Passive 32
 Remedial Options Water Treatment n Active n Ongoing humanly operated n Frequent maintenance and monitoring n Generally requires external energy source n Generally type chosen for active mining 33
Remedial Options Water Treatment n Active n Ongoing humanly operated n Frequent maintenance and monitoring n Generally requires external energy source n Generally type chosen for active mining 33
 Remedial Options Water Treatment n Passive (semi-passive) n Constant human intervention not required n Natural processes n Natural materials n Regeneration of materials n Gravity feed versus pumping 34
Remedial Options Water Treatment n Passive (semi-passive) n Constant human intervention not required n Natural processes n Natural materials n Regeneration of materials n Gravity feed versus pumping 34
 Remedial Options Active Treatment n Chemical Reaction n Oxidation n n Aeration to increase oxygen content or use of oxidizers Neutralization, precipitation, and sorption Hydroxide, Ca(OH)2, Ca. O, Na. OH; Mg. O, Carbonate, Ca. CO 3, Na 2 CO 3 n Fly ash; slag; kiln dust n n Coagulation, flocculation 35
Remedial Options Active Treatment n Chemical Reaction n Oxidation n n Aeration to increase oxygen content or use of oxidizers Neutralization, precipitation, and sorption Hydroxide, Ca(OH)2, Ca. O, Na. OH; Mg. O, Carbonate, Ca. CO 3, Na 2 CO 3 n Fly ash; slag; kiln dust n n Coagulation, flocculation 35
 Remedial Options Active Treatment n Physical Membrane n Reverse osmosis n Micro, ultra, or nano-filtration n Ion exchange n 36
Remedial Options Active Treatment n Physical Membrane n Reverse osmosis n Micro, ultra, or nano-filtration n Ion exchange n 36
 Remedial Options Passive/Semi-passive Treatment n Oxidation n Aeration Neutralization and precipitation n Ion exchange n Sorption (organic and inorganic) n Plant uptake n 37
Remedial Options Passive/Semi-passive Treatment n Oxidation n Aeration Neutralization and precipitation n Ion exchange n Sorption (organic and inorganic) n Plant uptake n 37
 Remedial Options Passive/Semi-passive Treatment Aerobic wetland n Anoxic limestone drain (ALD) n Anaerobic wetland n n Sulfate-reducing bioreactor (SRBR) n Biochemical reactor (BCR) n Permeable reactive barrier (PRB) 38
Remedial Options Passive/Semi-passive Treatment Aerobic wetland n Anoxic limestone drain (ALD) n Anaerobic wetland n n Sulfate-reducing bioreactor (SRBR) n Biochemical reactor (BCR) n Permeable reactive barrier (PRB) 38
 Remedial Options Passive/Semi-passive Treatment n Aerobic wetland Best for net alkaline drainage n Precipitation and sorption n Shallow – aeration n Plants – uptake n 39
Remedial Options Passive/Semi-passive Treatment n Aerobic wetland Best for net alkaline drainage n Precipitation and sorption n Shallow – aeration n Plants – uptake n 39
 Remedial Options Passive/Semi-passive Treatment n Anoxic limestone drain (ALD) Best for net acidic, low Fe 3+, low Al 3+, and low dissolved oxygen n Neutralize acidity n Effluent to aerobic pond n 40
Remedial Options Passive/Semi-passive Treatment n Anoxic limestone drain (ALD) Best for net acidic, low Fe 3+, low Al 3+, and low dissolved oxygen n Neutralize acidity n Effluent to aerobic pond n 40
 Remedial Options Passive/Semi-passive Treatment n Reducing alkalinity producing system (RAPS) n Similar to ALD, but reduces the water then adds alkalinity 41
Remedial Options Passive/Semi-passive Treatment n Reducing alkalinity producing system (RAPS) n Similar to ALD, but reduces the water then adds alkalinity 41
 Remedial Options Passive/Semi-passive Treatment n Anaerobic wetland (BCR) Best for net acidic, low Fe 3+, low Al 3+, high metals n Reduction of redox active elements n Microbial reduction of sulfate to sulfide n Metal sulfide precipitation n Sorption of metals n n Organic / inorganic 42
Remedial Options Passive/Semi-passive Treatment n Anaerobic wetland (BCR) Best for net acidic, low Fe 3+, low Al 3+, high metals n Reduction of redox active elements n Microbial reduction of sulfate to sulfide n Metal sulfide precipitation n Sorption of metals n n Organic / inorganic 42
 Remedial Options Passive/Semi-passive Treatment n Anaerobic wetland (BCR) n Natural materials n Hay, straw, wood chips, manure, crushed limestone Layer of water n Generally vertical flow n Effluent to aerobic pond or weir n 43
Remedial Options Passive/Semi-passive Treatment n Anaerobic wetland (BCR) n Natural materials n Hay, straw, wood chips, manure, crushed limestone Layer of water n Generally vertical flow n Effluent to aerobic pond or weir n 43
 Remedial Options Passive/Semi-passive Treatment n Permeable Reactive Barrier (PRB) n Similar to BCR, but treat groundwater 44
Remedial Options Passive/Semi-passive Treatment n Permeable Reactive Barrier (PRB) n Similar to BCR, but treat groundwater 44
 Remedial Options What to Choose? n Choice depends on Amounts - money, time, land, metals, acid, flow n Site access n Active or abandoned mine n Specific contaminants n 45
Remedial Options What to Choose? n Choice depends on Amounts - money, time, land, metals, acid, flow n Site access n Active or abandoned mine n Specific contaminants n 45
 Passive Treatment Case Study (Standard Mine, Colorado) n Operation began in the 1930’s Ag, Au, Pb, Zn n Abandoned in 1966 n Adit drains into Elk Creek n n Devoid of aquatic life n n Cd, Cu, Fe, Pb, Mn, and Zn exceed WQS Elk Creek feeds into Coal Creek n Source of DW for Crested Butte 46
Passive Treatment Case Study (Standard Mine, Colorado) n Operation began in the 1930’s Ag, Au, Pb, Zn n Abandoned in 1966 n Adit drains into Elk Creek n n Devoid of aquatic life n n Cd, Cu, Fe, Pb, Mn, and Zn exceed WQS Elk Creek feeds into Coal Creek n Source of DW for Crested Butte 46
 Standard Mine Crested Butte – remote location Constructed in 2007 11, 000 ft 1. 2 gpm Cd, Cu, Fe, Pb, Mn, Zn No electricity Photo by Scott Jacobs - NRMRL 47
Standard Mine Crested Butte – remote location Constructed in 2007 11, 000 ft 1. 2 gpm Cd, Cu, Fe, Pb, Mn, Zn No electricity Photo by Scott Jacobs - NRMRL 47
![Aerobic Polishing Cell(s) [2008] Remove biological oxygen demand Increase dissolved oxygen Degas sulfide and Aerobic Polishing Cell(s) [2008] Remove biological oxygen demand Increase dissolved oxygen Degas sulfide and](https://present5.com/presentation/12757f0a7af76cd1c938084aa1ac5695/image-48.jpg) Aerobic Polishing Cell(s) [2008] Remove biological oxygen demand Increase dissolved oxygen Degas sulfide and ammonia Reduce total suspended solids Oxidize and precipitate any Fe 48
Aerobic Polishing Cell(s) [2008] Remove biological oxygen demand Increase dissolved oxygen Degas sulfide and ammonia Reduce total suspended solids Oxidize and precipitate any Fe 48
 Passive Treatment Case Study (Standard Mine, Colorado) Year-round treatment n Automated sampling n n p. H, temperature, ORP Satellite reporting n Protection from weather n 49
Passive Treatment Case Study (Standard Mine, Colorado) Year-round treatment n Automated sampling n n p. H, temperature, ORP Satellite reporting n Protection from weather n 49
 33 -58 feet annual snowfall http: //dept. ca. uky. edu/asmr/W/Full%20 Papers%202008/0892 -Reisman-OH. pdf 50
33 -58 feet annual snowfall http: //dept. ca. uky. edu/asmr/W/Full%20 Papers%202008/0892 -Reisman-OH. pdf 50
 Passive Treatment Case Study (Standard Mine, Colorado) n Methods n Whole effluent toxicity tests (WET) Ceriodaphnia dubia (water flea) n Pimephales promelas (fathead minnow) n n Acute 48 -hr LC 50 n n Percentage of water Control survival > 90% 51
Passive Treatment Case Study (Standard Mine, Colorado) n Methods n Whole effluent toxicity tests (WET) Ceriodaphnia dubia (water flea) n Pimephales promelas (fathead minnow) n n Acute 48 -hr LC 50 n n Percentage of water Control survival > 90% 51
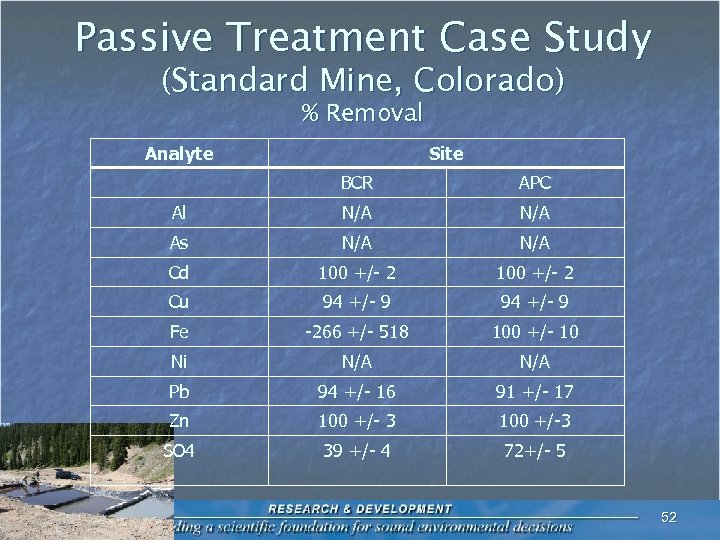 Passive Treatment Case Study (Standard Mine, Colorado) % Removal Analyte Site BCR APC Al N/A As N/A Cd 100 +/- 2 Cu 94 +/- 9 Fe -266 +/- 518 100 +/- 10 Ni N/A Pb 94 +/- 16 91 +/- 17 Zn 100 +/- 3 100 +/-3 SO 4 39 +/- 4 72+/- 5 52
Passive Treatment Case Study (Standard Mine, Colorado) % Removal Analyte Site BCR APC Al N/A As N/A Cd 100 +/- 2 Cu 94 +/- 9 Fe -266 +/- 518 100 +/- 10 Ni N/A Pb 94 +/- 16 91 +/- 17 Zn 100 +/- 3 100 +/-3 SO 4 39 +/- 4 72+/- 5 52
 Gray = water flea Black = fathead minnow 1% 2% 53
Gray = water flea Black = fathead minnow 1% 2% 53
 35% mortality Gray = water flea Black = fathead minnow 54
35% mortality Gray = water flea Black = fathead minnow 54
 Gray = water flea Black = fathead minnow Not different from control 55
Gray = water flea Black = fathead minnow Not different from control 55
 Effluent samples more toxic to fathead minnow than to the water flea 56
Effluent samples more toxic to fathead minnow than to the water flea 56
 100% <20% 57
100% <20% 57
 2 ug/l H 2 S. 2 to 5 mg/l NH 3 58
2 ug/l H 2 S. 2 to 5 mg/l NH 3 58
 Passive Treatment Case Study (Standard Mine, Colorado) n Concluding remarks Results strongly suggest toxicity from dissolved hydrogen sulfide gas n Other BCRs may have different toxicants, depending on: n Contaminants present and efficiency of removal n Concentrations of dissolved gases n p. H of effluent n 59
Passive Treatment Case Study (Standard Mine, Colorado) n Concluding remarks Results strongly suggest toxicity from dissolved hydrogen sulfide gas n Other BCRs may have different toxicants, depending on: n Contaminants present and efficiency of removal n Concentrations of dissolved gases n p. H of effluent n 59
 Thank you! Study at Standard Mine and 3 other sites: Butler, BA, Smith, ME, Reisman DJ, Lazorchak JM. 2011. Metal , , removal efficiency and ecotoxicological assessment of field-scale passive treatment biochemical reactors. Environmental Toxicology & Chemistry. 30(2): 385 -392. 60
Thank you! Study at Standard Mine and 3 other sites: Butler, BA, Smith, ME, Reisman DJ, Lazorchak JM. 2011. Metal , , removal efficiency and ecotoxicological assessment of field-scale passive treatment biochemical reactors. Environmental Toxicology & Chemistry. 30(2): 385 -392. 60


