a878f9247a056841246e59bd06d4196c.ppt
- Количество слайдов: 62
 Mevacor Daily® Merck NDA 21 -213 Nonprescription Drugs Advisory Committee and Endocrinologic and Metabolic Advisory Committee Meeting Silver Spring, Maryland December 13, 2007 Linda Hu, M. D. Division of Nonprescription Clinical Evaluation Center for Drug Evaluation and Research
Mevacor Daily® Merck NDA 21 -213 Nonprescription Drugs Advisory Committee and Endocrinologic and Metabolic Advisory Committee Meeting Silver Spring, Maryland December 13, 2007 Linda Hu, M. D. Division of Nonprescription Clinical Evaluation Center for Drug Evaluation and Research
 Self Evaluation of Lovastatin to Enhance Cholesterol Treatment (SELECT) NDAC December 13, 2007
Self Evaluation of Lovastatin to Enhance Cholesterol Treatment (SELECT) NDAC December 13, 2007
 Overview • Regulatory Background • SELECT Labeling Paradigms • SELECT Study Design • SELECT Results – Analyses of Mitigations – Analysis Of Eligibility Criteria Subsets (Hierarchies) – Analysis Of User Population • Summary NDAC December 13, 2007 3
Overview • Regulatory Background • SELECT Labeling Paradigms • SELECT Study Design • SELECT Results – Analyses of Mitigations – Analysis Of Eligibility Criteria Subsets (Hierarchies) – Analysis Of User Population • Summary NDAC December 13, 2007 3
 Regulatory Background • Initial submission 1999 • Response to October 2000 non-approval letter, which included – CUSTOM actual use study, “A Consumer Use Study of Over. The-Counter MEVACOR” – Label comprehension study – Other efficacy data and safety data • Led to non-approval letter in February, 2005 NDAC December 13, 2007 4
Regulatory Background • Initial submission 1999 • Response to October 2000 non-approval letter, which included – CUSTOM actual use study, “A Consumer Use Study of Over. The-Counter MEVACOR” – Label comprehension study – Other efficacy data and safety data • Led to non-approval letter in February, 2005 NDAC December 13, 2007 4
 CUSTOM Actual-Use Study • 10% of CUSTOM users correctly self-selected – Required criteria age, LDL-C, CHD risk factors, absence of certain conditions (conditions = liver disease, high triglycerides, history of statin-induced muscle pain) • 54% (571/1059) of CUSTOM users considered appropriate by sponsor – 416 said they talked to their doctor NDAC December 13, 2007 5
CUSTOM Actual-Use Study • 10% of CUSTOM users correctly self-selected – Required criteria age, LDL-C, CHD risk factors, absence of certain conditions (conditions = liver disease, high triglycerides, history of statin-induced muscle pain) • 54% (571/1059) of CUSTOM users considered appropriate by sponsor – 416 said they talked to their doctor NDAC December 13, 2007 5
 Deficiencies in Feb 2005 Non Approval Letter • Concern about inability of consumers, on their own, to make decisions on the appropriateness of statin therapy for their own personal use • Remaining safety concerns – Use by pregnant women and women of childbearing potential – Compliance with muscle pain warning – Use by patients with asymptomatic liver disease NDAC December 13, 2007 6
Deficiencies in Feb 2005 Non Approval Letter • Concern about inability of consumers, on their own, to make decisions on the appropriateness of statin therapy for their own personal use • Remaining safety concerns – Use by pregnant women and women of childbearing potential – Compliance with muscle pain warning – Use by patients with asymptomatic liver disease NDAC December 13, 2007 6
 Recommendations from Feb 2005 Non Approval Letter • A self-selection/use study or studies • Label comprehension studies • Address the risk in subjects with asymptomatic liver disease NDAC December 13, 2007 7
Recommendations from Feb 2005 Non Approval Letter • A self-selection/use study or studies • Label comprehension studies • Address the risk in subjects with asymptomatic liver disease NDAC December 13, 2007 7
 SELECT Study Label Paradigms LDL-C Label Total-C Label NDAC December 13, 2007
SELECT Study Label Paradigms LDL-C Label Total-C Label NDAC December 13, 2007
 LDL-C Label Target Population • Males ≥ 45 years; females ≥ 55 years • LDL in range 130 - 170 mg/d. L • One or more additional cardiac risk factors NDAC December 13, 2007 9
LDL-C Label Target Population • Males ≥ 45 years; females ≥ 55 years • LDL in range 130 - 170 mg/d. L • One or more additional cardiac risk factors NDAC December 13, 2007 9
 Cardiac Risk Factors • Cigarette smoking • Hypertension or on BP medication • Family history of premature CHD (below age of 55 years in a male parent or sibling or below 65 in female relative) • HDL-C < 40 mg/d. L • Age (men > 45 years, women > 55 years). NDAC December 13, 2007 10
Cardiac Risk Factors • Cigarette smoking • Hypertension or on BP medication • Family history of premature CHD (below age of 55 years in a male parent or sibling or below 65 in female relative) • HDL-C < 40 mg/d. L • Age (men > 45 years, women > 55 years). NDAC December 13, 2007 10
 Total-C Label Target Population • Total-C in range 200 -240 mg/d. L • Males – > 45 years • Females – HDL < 60 mg/d. L – > 55 years – One or more additional cardiac risk factors NDAC December 13, 2007 11
Total-C Label Target Population • Total-C in range 200 -240 mg/d. L • Males – > 45 years • Females – HDL < 60 mg/d. L – > 55 years – One or more additional cardiac risk factors NDAC December 13, 2007 11
 SELECT Label Paradigms • Label should be consistent with NCEP ATP III guidelines • LDL-C label conforms better to ATP III than Total-C label • Focus on results from LDL-C label arm NDAC December 13, 2007 12
SELECT Label Paradigms • Label should be consistent with NCEP ATP III guidelines • LDL-C label conforms better to ATP III than Total-C label • Focus on results from LDL-C label arm NDAC December 13, 2007 12
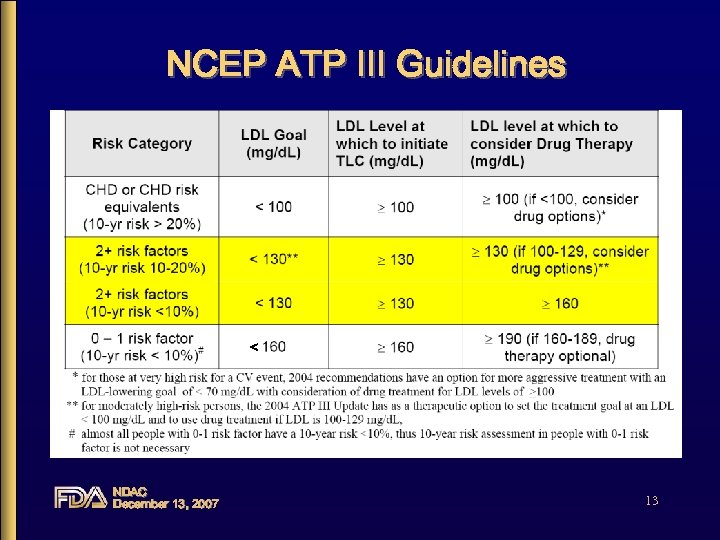 NCEP ATP III Guidelines < NDAC December 13, 2007 13
NCEP ATP III Guidelines < NDAC December 13, 2007 13
 Self Evaluation of Lovastatin to Enhance Cholesterol Treatment (SELECT) Study Design and Study Population NDAC December 13, 2007
Self Evaluation of Lovastatin to Enhance Cholesterol Treatment (SELECT) Study Design and Study Population NDAC December 13, 2007
 SELECT Self-Selection Study Self-selection: A decision by a consumer whether or not to choose a product for use based on two things: (1) his or her unique medical history and (2) the product labeling. • How many consumers will decide correctly that Mevacor is OK for them to use (“selfselection”)? • Study should be naturalistic NDAC December 13, 2007 15
SELECT Self-Selection Study Self-selection: A decision by a consumer whether or not to choose a product for use based on two things: (1) his or her unique medical history and (2) the product labeling. • How many consumers will decide correctly that Mevacor is OK for them to use (“selfselection”)? • Study should be naturalistic NDAC December 13, 2007 15
 SELECT Self-Selection Study • Consumers were asked – Self-assessment (SA) question – Purchase decision (PD) question – Reasons why, if incorrect • Subjects randomized to two labels – LDL-C – Total-C NDAC December 13, 2007 Focus on LDL-C 16
SELECT Self-Selection Study • Consumers were asked – Self-assessment (SA) question – Purchase decision (PD) question – Reasons why, if incorrect • Subjects randomized to two labels – LDL-C – Total-C NDAC December 13, 2007 Focus on LDL-C 16
 Focus on Self-Assessment • Purchase Decision is influenced by potentially confounding economic and marketing issues – cost – consumer appeal of product • Our interest is in whether the consumer understands – who should use – why should one use – how to use NDAC December 13, 2007 17
Focus on Self-Assessment • Purchase Decision is influenced by potentially confounding economic and marketing issues – cost – consumer appeal of product • Our interest is in whether the consumer understands – who should use – why should one use – how to use NDAC December 13, 2007 17
 Directions Prior to Self-Assessment Question • Participants were told – The product is not appropriate for everyone – You will be asked whether the product was appropriate for you – You will be asked whether you wish to purchase – Concentrate and to take as much time as needed NDAC December 13, 2007 18
Directions Prior to Self-Assessment Question • Participants were told – The product is not appropriate for everyone – You will be asked whether the product was appropriate for you – You will be asked whether you wish to purchase – Concentrate and to take as much time as needed NDAC December 13, 2007 18
 Self-Assessment (SA) • “Based on this label, is this product appropriate for you to use right now or not? ” – Yes – No – Other (unsure or wanted to ask a doctor) NDAC December 13, 2007 19
Self-Assessment (SA) • “Based on this label, is this product appropriate for you to use right now or not? ” – Yes – No – Other (unsure or wanted to ask a doctor) NDAC December 13, 2007 19
 Purchase Decision (PD) • “Would you like to pay for this right now for your own use or put it back in the display? ” • If the participant wants to purchase, then ask, “Is there anything you plan to do before you start using it? ” NDAC December 13, 2007 20
Purchase Decision (PD) • “Would you like to pay for this right now for your own use or put it back in the display? ” • If the participant wants to purchase, then ask, “Is there anything you plan to do before you start using it? ” NDAC December 13, 2007 20
 Recruitment • Recruitment by mass media advertising • Ads told subjects to know: – total cholesterol, LDL, HDL, and triglycerides • Advertising may have recruited more informed subjects who were better prepared for selfselection NDAC December 13, 2007 21
Recruitment • Recruitment by mass media advertising • Ads told subjects to know: – total cholesterol, LDL, HDL, and triglycerides • Advertising may have recruited more informed subjects who were better prepared for selfselection NDAC December 13, 2007 21
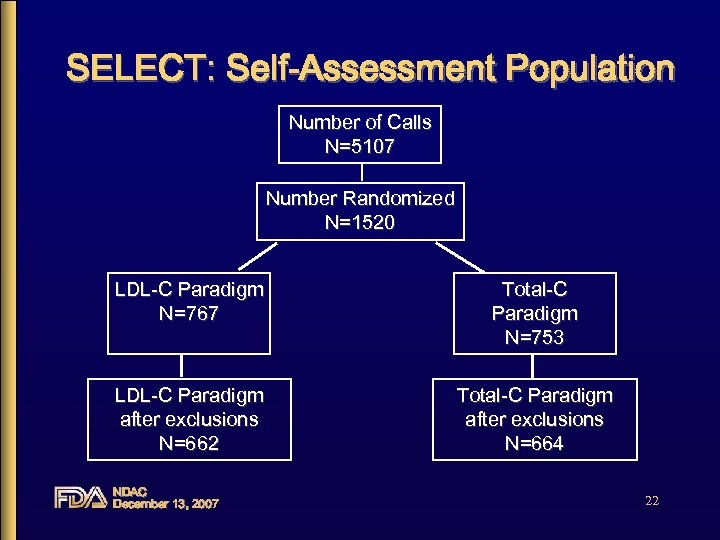 SELECT: Self-Assessment Population Number of Calls N=5107 Number Randomized N=1520 LDL-C Paradigm N=767 Total-C Paradigm N=753 LDL-C Paradigm after exclusions N=662 Total-C Paradigm after exclusions N=664 NDAC December 13, 2007 22
SELECT: Self-Assessment Population Number of Calls N=5107 Number Randomized N=1520 LDL-C Paradigm N=767 Total-C Paradigm N=753 LDL-C Paradigm after exclusions N=662 Total-C Paradigm after exclusions N=664 NDAC December 13, 2007 22
 SELECT Study Population • 1520 subjects randomized – typically well educated, middle-class – 90% high school graduates and 60% had some college – 70% had insurance – 14% low literate NDAC December 13, 2007 23
SELECT Study Population • 1520 subjects randomized – typically well educated, middle-class – 90% high school graduates and 60% had some college – 70% had insurance – 14% low literate NDAC December 13, 2007 23
 Areas Targeted For Improvement in SELECT • Decrease the proportion of women <55 • Decrease the proportion of women of childbearing potential • Decrease the proportion of low CHD risk (<5% risk of CHD in 10 years) NDAC December 13, 2007 24
Areas Targeted For Improvement in SELECT • Decrease the proportion of women <55 • Decrease the proportion of women of childbearing potential • Decrease the proportion of low CHD risk (<5% risk of CHD in 10 years) NDAC December 13, 2007 24
 SELECT RESULTS NDAC December 13, 2007 25
SELECT RESULTS NDAC December 13, 2007 25
 Eligibility Assessment Criteria • Age • Allergy to drug • Pregnant or breastfeeding • Lipid-lowering • May become pregnant medications • Heart disease • Interacting drugs • Stroke • LDL-C • Diabetes • HDL-C • Liver disease • Grapefruit juice • Listed risk factors • Fasting lipid profile NDAC December 13, 2007 26
Eligibility Assessment Criteria • Age • Allergy to drug • Pregnant or breastfeeding • Lipid-lowering • May become pregnant medications • Heart disease • Interacting drugs • Stroke • LDL-C • Diabetes • HDL-C • Liver disease • Grapefruit juice • Listed risk factors • Fasting lipid profile NDAC December 13, 2007 26
 Self-Assessment Results LDL-C Is it appropriate for you? SA population N=662 Not appropriate for me (SA =No) N=448 (68%) Correct 439 (98%) Incorrect 9 (2%) Correct 473 (71. 5%) NDAC December 13, 2007 Appropriate for me (SA =Yes) N=214 (32%) Correct 34 (16%) Incorrect 180 (84%) Incorrect 189 (28. 5%) 27
Self-Assessment Results LDL-C Is it appropriate for you? SA population N=662 Not appropriate for me (SA =No) N=448 (68%) Correct 439 (98%) Incorrect 9 (2%) Correct 473 (71. 5%) NDAC December 13, 2007 Appropriate for me (SA =Yes) N=214 (32%) Correct 34 (16%) Incorrect 180 (84%) Incorrect 189 (28. 5%) 27
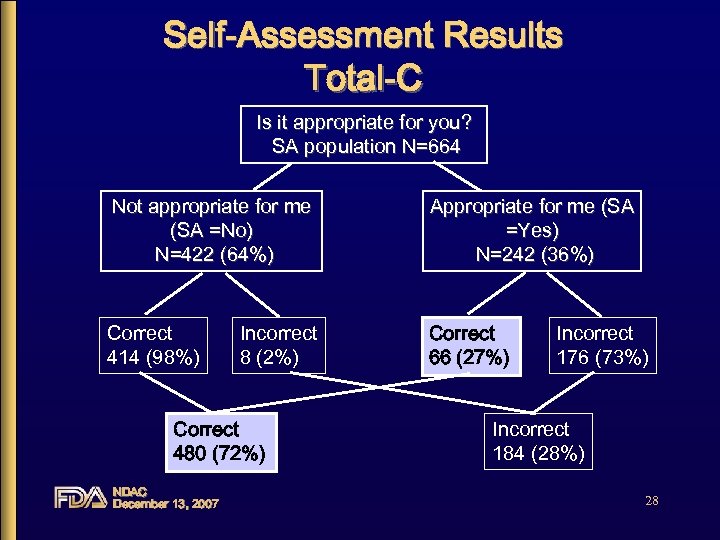 Self-Assessment Results Total-C Is it appropriate for you? SA population N=664 Not appropriate for me (SA =No) N=422 (64%) Appropriate for me (SA =Yes) N=242 (36%) Correct 414 (98%) Correct 66 (27%) Incorrect 8 (2%) Correct 480 (72%) NDAC December 13, 2007 Incorrect 176 (73%) Incorrect 184 (28%) 28
Self-Assessment Results Total-C Is it appropriate for you? SA population N=664 Not appropriate for me (SA =No) N=422 (64%) Appropriate for me (SA =Yes) N=242 (36%) Correct 414 (98%) Correct 66 (27%) Incorrect 8 (2%) Correct 480 (72%) NDAC December 13, 2007 Incorrect 176 (73%) Incorrect 184 (28%) 28
 Mitigations for Incorrect Self-Assessment • Some subjects who incorrectly self-selected to use gave open-ended responses that were acceptable and could be “mitigated” – Would talk to doctor – Gave reasonable rationale for treatment – Gave evidence of not understanding the selfassessment question NDAC December 13, 2007 29
Mitigations for Incorrect Self-Assessment • Some subjects who incorrectly self-selected to use gave open-ended responses that were acceptable and could be “mitigated” – Would talk to doctor – Gave reasonable rationale for treatment – Gave evidence of not understanding the selfassessment question NDAC December 13, 2007 29
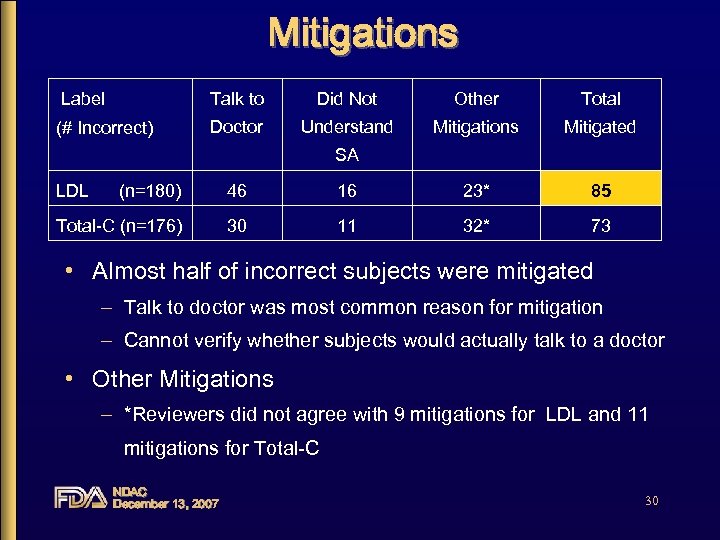 Mitigations Label Talk to Did Not Other Total (# Incorrect) Doctor Understand Mitigations Mitigated SA LDL (n=180) 46 16 23* 85 Total-C (n=176) 30 11 32* 73 • Almost half of incorrect subjects were mitigated – Talk to doctor was most common reason for mitigation – Cannot verify whether subjects would actually talk to a doctor • Other Mitigations – *Reviewers did not agree with 9 mitigations for LDL and 11 mitigations for Total-C NDAC December 13, 2007 30
Mitigations Label Talk to Did Not Other Total (# Incorrect) Doctor Understand Mitigations Mitigated SA LDL (n=180) 46 16 23* 85 Total-C (n=176) 30 11 32* 73 • Almost half of incorrect subjects were mitigated – Talk to doctor was most common reason for mitigation – Cannot verify whether subjects would actually talk to a doctor • Other Mitigations – *Reviewers did not agree with 9 mitigations for LDL and 11 mitigations for Total-C NDAC December 13, 2007 30
 Examples of Other Mitigations Made by Sponsor • Substitution of Mevacor for current lipid medication without talking to doctor • High risk subject who self-selected to use without talking to doctor • Self-selected yes when had prior side effects on statins NDAC December 13, 2007 31
Examples of Other Mitigations Made by Sponsor • Substitution of Mevacor for current lipid medication without talking to doctor • High risk subject who self-selected to use without talking to doctor • Self-selected yes when had prior side effects on statins NDAC December 13, 2007 31
 Self-Selection Percent Completely Correct, and Correct After Mitigations Label Self % Completely % Correct After Assessment Correct Mitigations LDL Yes 16% ~50% Total-C Yes 27% ~50% NDAC December 13, 2007 32
Self-Selection Percent Completely Correct, and Correct After Mitigations Label Self % Completely % Correct After Assessment Correct Mitigations LDL Yes 16% ~50% Total-C Yes 27% ~50% NDAC December 13, 2007 32
 SELECT RESULTS: ANALYSIS OF ELIGIBILITY CRITERIA SUBSETS HIERARCHIES NDAC December 13, 2007 33
SELECT RESULTS: ANALYSIS OF ELIGIBILITY CRITERIA SUBSETS HIERARCHIES NDAC December 13, 2007 33
 Hierarchies • Many eligibility criteria – Difficult to get all eligibility criteria correct – Some criteria of more clinical importance then others • New analyses where % correct was calculated for subsets of eligibility criteria (“hierarchies”) • Self-assessment hierarchies will be presented NDAC December 13, 2007 34
Hierarchies • Many eligibility criteria – Difficult to get all eligibility criteria correct – Some criteria of more clinical importance then others • New analyses where % correct was calculated for subsets of eligibility criteria (“hierarchies”) • Self-assessment hierarchies will be presented NDAC December 13, 2007 34
 Hierarchies for LDL-C Arm • Self-Selection Results Using ALL Label Criteria – % entirely correct (16%) – % correct after mitigation (~50%) • Using “hierarchies” or subsets of label criteria – % correct before mitigation – % correct after mitigation • % correct depends on which criteria are used in hierarchy NDAC December 13, 2007 35
Hierarchies for LDL-C Arm • Self-Selection Results Using ALL Label Criteria – % entirely correct (16%) – % correct after mitigation (~50%) • Using “hierarchies” or subsets of label criteria – % correct before mitigation – % correct after mitigation • % correct depends on which criteria are used in hierarchy NDAC December 13, 2007 35
 Sponsor Safety Hierarchy • Pregnant/breast feeding • May become pregnant • Allergy to lovastatin • On interacting medications • On lipid-lowering medications • Liver problem NDAC December 13, 2007 36
Sponsor Safety Hierarchy • Pregnant/breast feeding • May become pregnant • Allergy to lovastatin • On interacting medications • On lipid-lowering medications • Liver problem NDAC December 13, 2007 36
 SAFETY Hierarchy SA=Yes (LDL arm) N=214 Of 2 Pregnant/breastfeeding No Correct N=0 N=214 Of 12 may become pregnant No Correct N=1 N=213 Of 4 allergy to lovastatin No Correct N=0 N=213 Of 12 on interacting medicine No Correct NDAC December 13, 2007 N=210 N=3 37
SAFETY Hierarchy SA=Yes (LDL arm) N=214 Of 2 Pregnant/breastfeeding No Correct N=0 N=214 Of 12 may become pregnant No Correct N=1 N=213 Of 4 allergy to lovastatin No Correct N=0 N=213 Of 12 on interacting medicine No Correct NDAC December 13, 2007 N=210 N=3 37
 SAFETY Hierarchy SA=Yes N=210 Of 140 on lipid lowering medicine No Correct N=36 N=174 Of 23 liver problem No Correct N=0 N=174 Percent Correct 81. 3% NDAC December 13, 2007 38
SAFETY Hierarchy SA=Yes N=210 Of 140 on lipid lowering medicine No Correct N=36 N=174 Of 23 liver problem No Correct N=0 N=174 Percent Correct 81. 3% NDAC December 13, 2007 38
 Sponsor Safety Hierarchy Sponsor Safety: Pregnant/breast-feeding, may become pregnant, allergy to lovastatin, interacting medications, lipid lowering medications, and liver problems LDL (N=214) Hierarchy Sponsor Safety* Total-C (N=242) % Correct Before Mitigation % Correct After Mitigation 81. 3% 89. 3% 83. 1% 90. 1% For all eligibility criteria 16% correct NDAC December 13, 2007 39
Sponsor Safety Hierarchy Sponsor Safety: Pregnant/breast-feeding, may become pregnant, allergy to lovastatin, interacting medications, lipid lowering medications, and liver problems LDL (N=214) Hierarchy Sponsor Safety* Total-C (N=242) % Correct Before Mitigation % Correct After Mitigation 81. 3% 89. 3% 83. 1% 90. 1% For all eligibility criteria 16% correct NDAC December 13, 2007 39
 Eligibility Assessment Criteria • Age • Allergy to drug • Pregnant or breastfeeding • Lipid-lowering • May become pregnant medications • Heart disease • Interacting drugs • Stroke • LDL-C • Diabetes • HDL-C • Liver disease • Grapefruit juice • Listed risk factors • Fasting lipid profile NDAC December 13, 2007 40
Eligibility Assessment Criteria • Age • Allergy to drug • Pregnant or breastfeeding • Lipid-lowering • May become pregnant medications • Heart disease • Interacting drugs • Stroke • LDL-C • Diabetes • HDL-C • Liver disease • Grapefruit juice • Listed risk factors • Fasting lipid profile NDAC December 13, 2007 40
 Sponsor Hierarchies LDL (N=214) Total-C (N=242) % Correct Before Mitigation % Correct After Mitigation Safety* 81. 3% 89. 3% 83. 1% 90. 1% Benefit** 25. 7% 57. 0% 41. 7% 62. 4% Safety & Benefit 21. 0% 52. 8% 35. 1% 58. 3% Hierarchy * Pregnant/breast-feeding, may become pregnant, allergy to lovastatin, interacting medications, lipid lowering medications, liver problems ** Age, lipid values (LDL-C or Total-C), and risk factors NDAC December 13, 2007 41
Sponsor Hierarchies LDL (N=214) Total-C (N=242) % Correct Before Mitigation % Correct After Mitigation Safety* 81. 3% 89. 3% 83. 1% 90. 1% Benefit** 25. 7% 57. 0% 41. 7% 62. 4% Safety & Benefit 21. 0% 52. 8% 35. 1% 58. 3% Hierarchy * Pregnant/breast-feeding, may become pregnant, allergy to lovastatin, interacting medications, lipid lowering medications, liver problems ** Age, lipid values (LDL-C or Total-C), and risk factors NDAC December 13, 2007 41
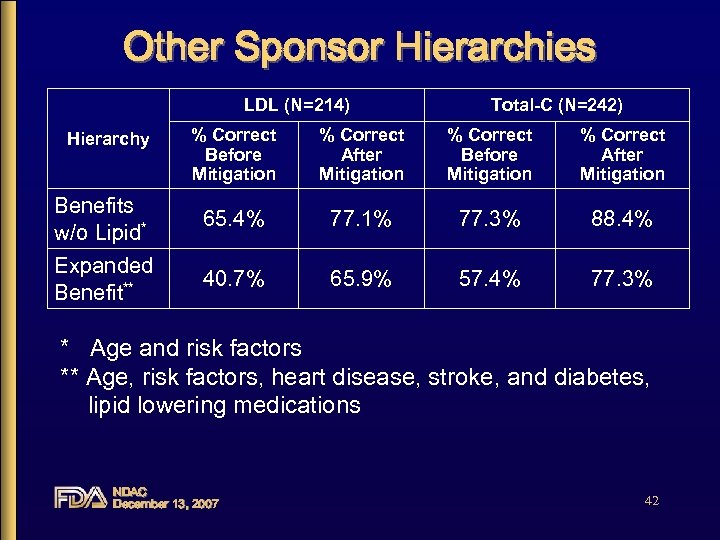 Other Sponsor Hierarchies LDL (N=214) Total-C (N=242) % Correct Before Mitigation % Correct After Mitigation Benefits w/o Lipid* 65. 4% 77. 1% 77. 3% 88. 4% Expanded Benefit** 40. 7% 65. 9% 57. 4% 77. 3% Hierarchy * Age and risk factors ** Age, risk factors, heart disease, stroke, and diabetes, lipid lowering medications NDAC December 13, 2007 42
Other Sponsor Hierarchies LDL (N=214) Total-C (N=242) % Correct Before Mitigation % Correct After Mitigation Benefits w/o Lipid* 65. 4% 77. 1% 77. 3% 88. 4% Expanded Benefit** 40. 7% 65. 9% 57. 4% 77. 3% Hierarchy * Age and risk factors ** Age, risk factors, heart disease, stroke, and diabetes, lipid lowering medications NDAC December 13, 2007 42
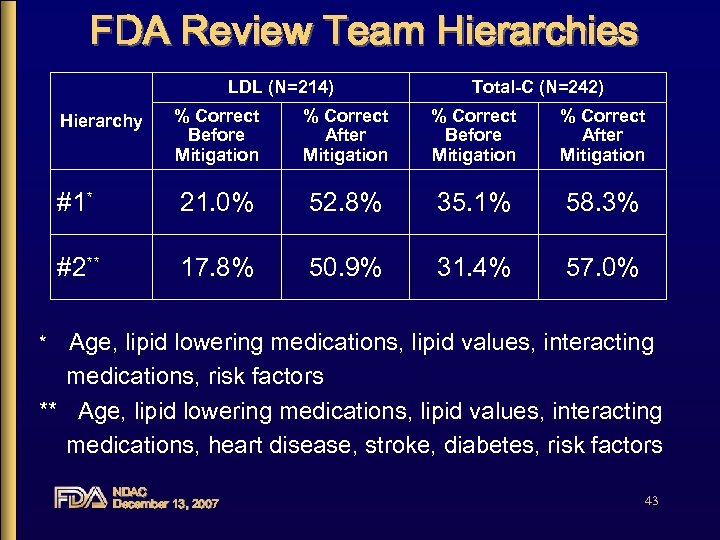 FDA Review Team Hierarchies LDL (N=214) Total-C (N=242) % Correct Before Mitigation % Correct After Mitigation #1* 21. 0% 52. 8% 35. 1% 58. 3% #2** 17. 8% 50. 9% 31. 4% 57. 0% Hierarchy Age, lipid lowering medications, lipid values, interacting medications, risk factors ** Age, lipid lowering medications, lipid values, interacting medications, heart disease, stroke, diabetes, risk factors * NDAC December 13, 2007 43
FDA Review Team Hierarchies LDL (N=214) Total-C (N=242) % Correct Before Mitigation % Correct After Mitigation #1* 21. 0% 52. 8% 35. 1% 58. 3% #2** 17. 8% 50. 9% 31. 4% 57. 0% Hierarchy Age, lipid lowering medications, lipid values, interacting medications, risk factors ** Age, lipid lowering medications, lipid values, interacting medications, heart disease, stroke, diabetes, risk factors * NDAC December 13, 2007 43
 Hierarchies • None of the hierarchies was defined a priori in the study protocol • Depending on which criteria are included, the percent correct in the hierarchies before mitigations ranged from 20 -80% NDAC December 13, 2007 44
Hierarchies • None of the hierarchies was defined a priori in the study protocol • Depending on which criteria are included, the percent correct in the hierarchies before mitigations ranged from 20 -80% NDAC December 13, 2007 44
 SELECT RESULTS: Profiles of User Population NDAC December 13, 2007 45
SELECT RESULTS: Profiles of User Population NDAC December 13, 2007 45
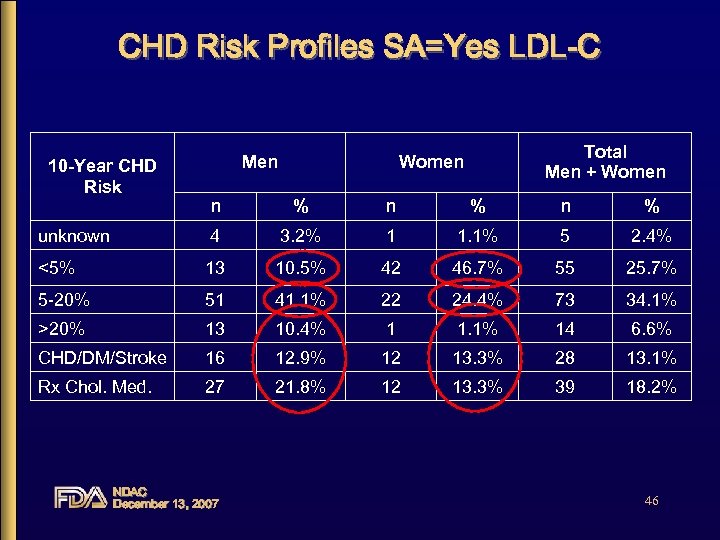 CHD Risk Profiles SA=Yes LDL-C 10 -Year CHD Risk Men Total Men + Women n % n % unknown 4 3. 2% 1 1. 1% 5 2. 4% <5% 13 10. 5% 42 46. 7% 55 25. 7% 5 -20% 51 41. 1% 22 24. 4% 73 34. 1% >20% 13 10. 4% 1 1. 1% 14 6. 6% CHD/DM/Stroke 16 12. 9% 12 13. 3% 28 13. 1% Rx Chol. Med. 27 21. 8% 12 13. 3% 39 18. 2% NDAC December 13, 2007 46
CHD Risk Profiles SA=Yes LDL-C 10 -Year CHD Risk Men Total Men + Women n % n % unknown 4 3. 2% 1 1. 1% 5 2. 4% <5% 13 10. 5% 42 46. 7% 55 25. 7% 5 -20% 51 41. 1% 22 24. 4% 73 34. 1% >20% 13 10. 4% 1 1. 1% 14 6. 6% CHD/DM/Stroke 16 12. 9% 12 13. 3% 28 13. 1% Rx Chol. Med. 27 21. 8% 12 13. 3% 39 18. 2% NDAC December 13, 2007 46
 Women Too Young (<55 yo) LDL arm • 391 women were asked the self-assessment question in the LDL arm, 220 (56. 3%) were too young. – 13. 2% (29/220) self-selected to use • Of 391 women asked the SA question, 101 women of all ages self-selected to use – Of these 101 women, ~ 29% were < 55 years old • Reasons for their decision were: age is close, lower my cholesterol, family history NDAC December 13, 2007 47
Women Too Young (<55 yo) LDL arm • 391 women were asked the self-assessment question in the LDL arm, 220 (56. 3%) were too young. – 13. 2% (29/220) self-selected to use • Of 391 women asked the SA question, 101 women of all ages self-selected to use – Of these 101 women, ~ 29% were < 55 years old • Reasons for their decision were: age is close, lower my cholesterol, family history NDAC December 13, 2007 47
 Pregnant/May Become Pregnant • Pregnant (n=4) – 1 out of 4: SA=Yes, – sponsor mitigated – reviewers agreed • May become pregnant (n=22) – 9% (2/22) chose SA=Yes, – sponsor mitigated – reviewers did not agree NDAC December 13, 2007 48
Pregnant/May Become Pregnant • Pregnant (n=4) – 1 out of 4: SA=Yes, – sponsor mitigated – reviewers agreed • May become pregnant (n=22) – 9% (2/22) chose SA=Yes, – sponsor mitigated – reviewers did not agree NDAC December 13, 2007 48
 Heart Disease (LDL Arm) • 48. 5% (33/68) with heart disease self -selected yes • Subjects at high CHD risk might be undertreated by 20 mg of lovastatin NDAC December 13, 2007 49
Heart Disease (LDL Arm) • 48. 5% (33/68) with heart disease self -selected yes • Subjects at high CHD risk might be undertreated by 20 mg of lovastatin NDAC December 13, 2007 49
 Already on Lipid Medications (LDL arm) • 140/750 (18. 7%) in the self-selection population were already taking lipidlowering medication • 44/140 (31. 4%) of these participants self -selected yes NDAC December 13, 2007 50
Already on Lipid Medications (LDL arm) • 140/750 (18. 7%) in the self-selection population were already taking lipidlowering medication • 44/140 (31. 4%) of these participants self -selected yes NDAC December 13, 2007 50
 Reasons Why Subjects on Rx Selected Incorrectly • The most frequent reasons for choosing to use or purchasing, when the subject was already taking lipid-lowering medication – to replace the prescription medication – specifically to replace it because of lower cost NDAC December 13, 2007 51
Reasons Why Subjects on Rx Selected Incorrectly • The most frequent reasons for choosing to use or purchasing, when the subject was already taking lipid-lowering medication – to replace the prescription medication – specifically to replace it because of lower cost NDAC December 13, 2007 51
 Substitution Rate of OTC for Rx Medications CHD Reported Action LDL-C (N=27) N(%) Take along with it 8 (29. 6%) Take in place 14 (51. 9%) Don’t know 1 (3. 7%) Other 3 (11. 1%) Missing 1 (3. 7%) NDAC December 13, 2007 52
Substitution Rate of OTC for Rx Medications CHD Reported Action LDL-C (N=27) N(%) Take along with it 8 (29. 6%) Take in place 14 (51. 9%) Don’t know 1 (3. 7%) Other 3 (11. 1%) Missing 1 (3. 7%) NDAC December 13, 2007 52
 Reasons for Preferring OTC Med Rather than Rx Med for Cholesterol • The most frequent reasons for preferring OTC medications, for those who self-selected yes : – less expensive (50%) – convenience (29%) – don’t have to see the doctor (15%) – feels safer/less side effects (11. 3%) NDAC December 13, 2007 53
Reasons for Preferring OTC Med Rather than Rx Med for Cholesterol • The most frequent reasons for preferring OTC medications, for those who self-selected yes : – less expensive (50%) – convenience (29%) – don’t have to see the doctor (15%) – feels safer/less side effects (11. 3%) NDAC December 13, 2007 53
 High Risk, CHD, Diabetes, Stroke: LDL-C arm • On average, ~30% of subjects with CHD, DM or stroke wanted to buy • 2/3 of these subjects were not on any lipidlowering medications NDAC December 13, 2007 54
High Risk, CHD, Diabetes, Stroke: LDL-C arm • On average, ~30% of subjects with CHD, DM or stroke wanted to buy • 2/3 of these subjects were not on any lipidlowering medications NDAC December 13, 2007 54
 What Medications Would Be Substituted: LDL arm NDAC December 13, 2007 55
What Medications Would Be Substituted: LDL arm NDAC December 13, 2007 55
 Knowing LDL-C versus Total-C • In the LDL-C arm, 37. 5% (268/714) did not know their LDL-C, whereas in the Total-C arm, 21% (149/708) did not know their Total-C. • Of those who did not know their cholesterol numbers – LDL-C: 22. 4% ( 60/268) self-selected yes – Total-C: 17. 4% (26/149) self- selected yes NDAC December 13, 2007 56
Knowing LDL-C versus Total-C • In the LDL-C arm, 37. 5% (268/714) did not know their LDL-C, whereas in the Total-C arm, 21% (149/708) did not know their Total-C. • Of those who did not know their cholesterol numbers – LDL-C: 22. 4% ( 60/268) self-selected yes – Total-C: 17. 4% (26/149) self- selected yes NDAC December 13, 2007 56
 Ineligible Subjects for Lipid Values • Many of the subjects who had LDL outside the eligible range nevertheless selected yes in the LDL-C arm – 43% (52/122) of subjects w/ LDL-C too high (above 170) self-selected yes – 17% (26/153) of subject whose LDL-C too low (below 130) self-selected yes NDAC December 13, 2007 57
Ineligible Subjects for Lipid Values • Many of the subjects who had LDL outside the eligible range nevertheless selected yes in the LDL-C arm – 43% (52/122) of subjects w/ LDL-C too high (above 170) self-selected yes – 17% (26/153) of subject whose LDL-C too low (below 130) self-selected yes NDAC December 13, 2007 57
 SELECT Summary • ~20% self-selected yes entirely correctly – With mitigations, % correct ~ 50% • Only 4 pregnant women in the study • Of those who may become pregnant – 2/22 (9%) were incorrect (SA=Yes) • ~ 30% of participants with CHD, diabetes mellitus, or stroke wanted to buy – 2/3 were not on cholesterol medications NDAC December 13, 2007 58
SELECT Summary • ~20% self-selected yes entirely correctly – With mitigations, % correct ~ 50% • Only 4 pregnant women in the study • Of those who may become pregnant – 2/22 (9%) were incorrect (SA=Yes) • ~ 30% of participants with CHD, diabetes mellitus, or stroke wanted to buy – 2/3 were not on cholesterol medications NDAC December 13, 2007 58
 SELECT Summary (LDL paradigm) • ~ 30% of subjects on lipid-lowering medication self- selected to use • Of those who would purchase and were on lipid-lowering medication – 50% would take Mevacor “in place of” Rx med – 30% would take Mevacor along with their Rx Med – The most commonly taken lipid-lowering medications were atorvastatin, simvastatin, rosuvastatin, and lovastatin NDAC December 13, 2007 59
SELECT Summary (LDL paradigm) • ~ 30% of subjects on lipid-lowering medication self- selected to use • Of those who would purchase and were on lipid-lowering medication – 50% would take Mevacor “in place of” Rx med – 30% would take Mevacor along with their Rx Med – The most commonly taken lipid-lowering medications were atorvastatin, simvastatin, rosuvastatin, and lovastatin NDAC December 13, 2007 59
 SELECT Summary (LDL paradigm) Those who self-selected to use in the LDL arm included: • 101 of 391 women who were asked the SA question – 29% were < 55 years old • 13. 2% of women < 55 years old • 22% of the participants who did not know their LDL-C value • 43% of subjects who had LDL too high (above 170) • 17% of subject whose LDL was too low (below 130) NDAC December 13, 2007 60
SELECT Summary (LDL paradigm) Those who self-selected to use in the LDL arm included: • 101 of 391 women who were asked the SA question – 29% were < 55 years old • 13. 2% of women < 55 years old • 22% of the participants who did not know their LDL-C value • 43% of subjects who had LDL too high (above 170) • 17% of subject whose LDL was too low (below 130) NDAC December 13, 2007 60
 SELECT Summary 10 -Year CHD risk: LDL-C Label (SA=Yes) • 41. 1% men were in the targeted CHD risk range – Most men outside the target range were of high risk, CHD equivalent or on lipid lowering meds • 24. 4% of women were in the targeted range – Most women outside this range were low risk • ~11% of men and over 45% of women were of low CHD risk (<5% risk of CHD in 10 years) NDAC December 13, 2007 61
SELECT Summary 10 -Year CHD risk: LDL-C Label (SA=Yes) • 41. 1% men were in the targeted CHD risk range – Most men outside the target range were of high risk, CHD equivalent or on lipid lowering meds • 24. 4% of women were in the targeted range – Most women outside this range were low risk • ~11% of men and over 45% of women were of low CHD risk (<5% risk of CHD in 10 years) NDAC December 13, 2007 61
 Acknowledgements • Division of Nonprescription Clinical Evaluation Review Team – Daiva Shetty, M. D. – Joel Schiffenbauer, M. D. – Andrea Leonard Segal, M. D. – Laura Shay, CNP – Stan Lin, Ph. D. – Michael Koenig, Ph. D. – Matthew Holman, Ph. D. – Mary Lewis, PM NDAC December 13, 2007 62
Acknowledgements • Division of Nonprescription Clinical Evaluation Review Team – Daiva Shetty, M. D. – Joel Schiffenbauer, M. D. – Andrea Leonard Segal, M. D. – Laura Shay, CNP – Stan Lin, Ph. D. – Michael Koenig, Ph. D. – Matthew Holman, Ph. D. – Mary Lewis, PM NDAC December 13, 2007 62


