6902524c9b064fc972a0fb3a73de5268.ppt
- Количество слайдов: 48
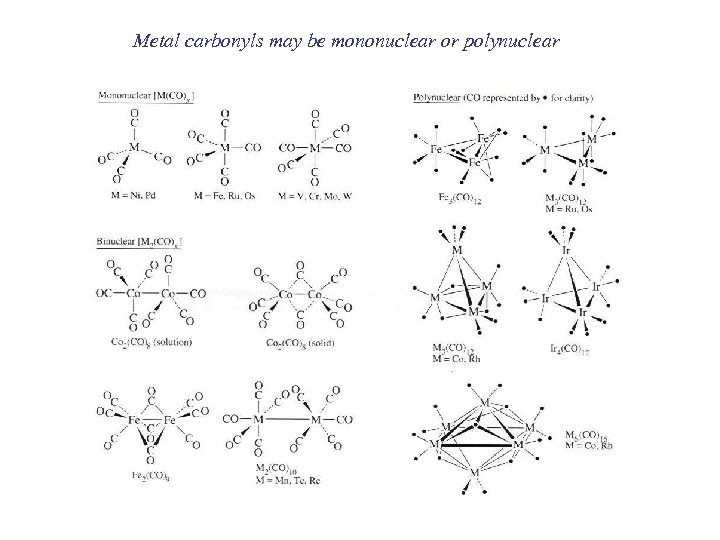
Metal carbonyls may be mononuclear or polynuclear
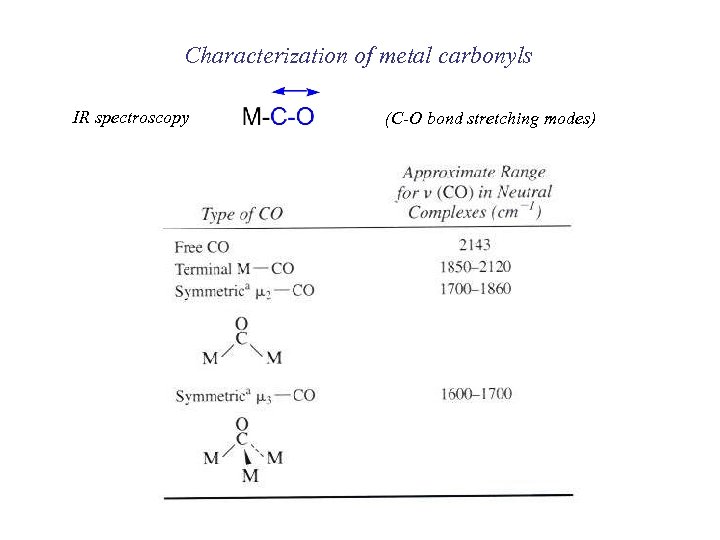
Characterization of metal carbonyls IR spectroscopy (C-O bond stretching modes)
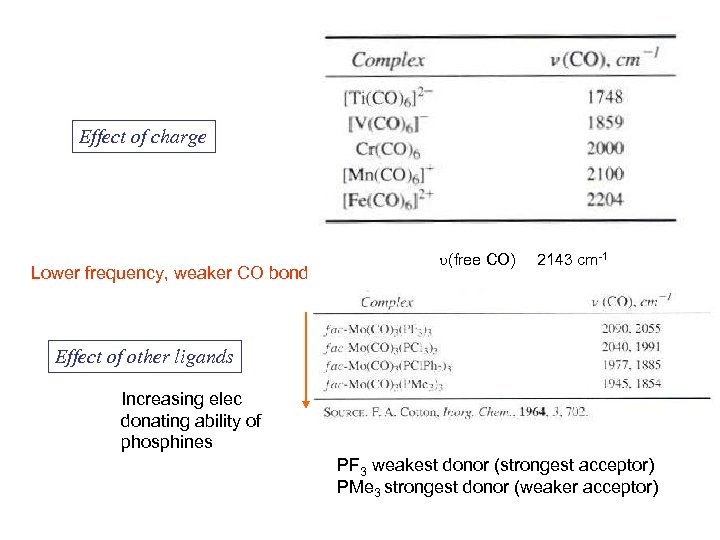
Effect of charge Lower frequency, weaker CO bond u(free CO) 2143 cm-1 Effect of other ligands Increasing elec donating ability of phosphines PF 3 weakest donor (strongest acceptor) PMe 3 strongest donor (weaker acceptor)
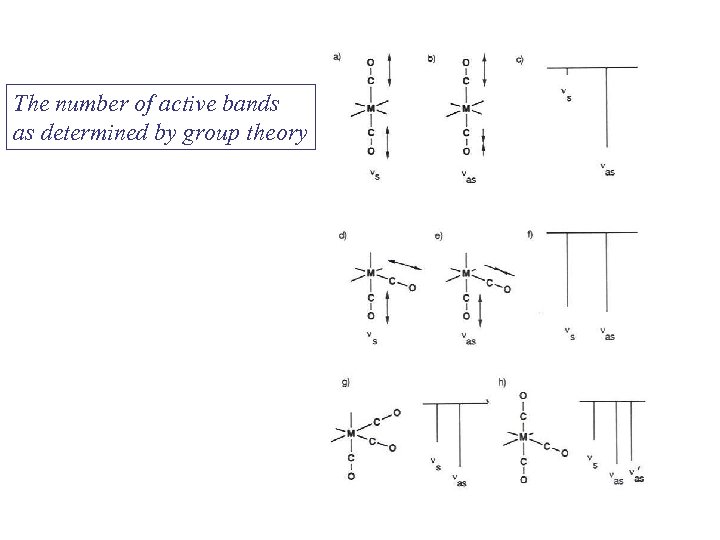
The number of active bands as determined by group theory
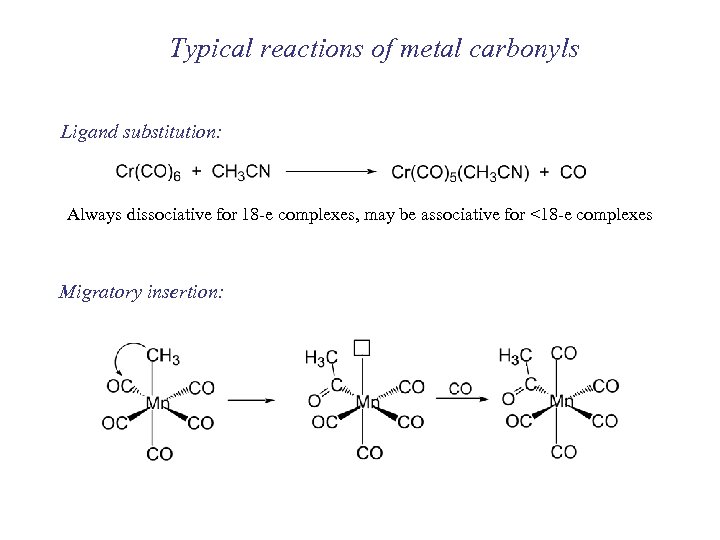
Typical reactions of metal carbonyls Ligand substitution: Always dissociative for 18 -e complexes, may be associative for <18 -e complexes Migratory insertion:
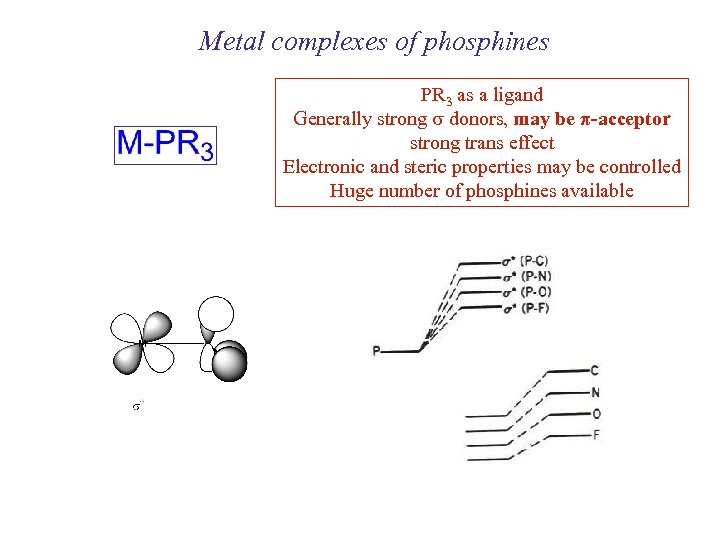
Metal complexes of phosphines PR 3 as a ligand Generally strong s donors, may be π-acceptor strong trans effect Electronic and steric properties may be controlled Huge number of phosphines available
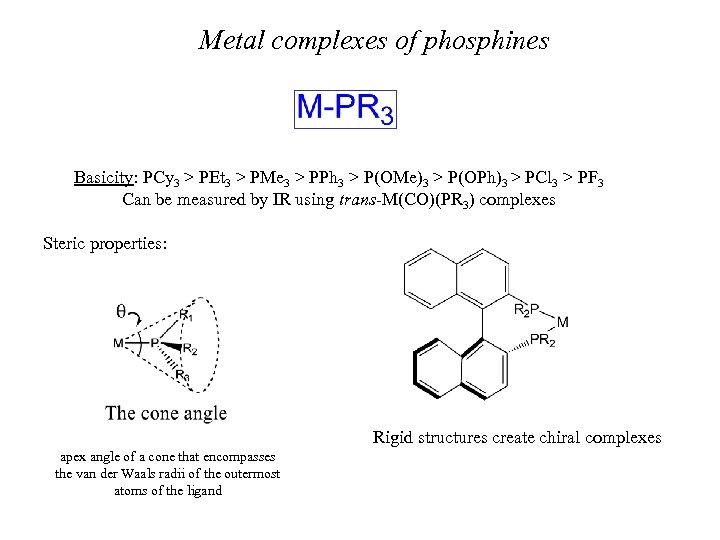
Metal complexes of phosphines Basicity: PCy 3 > PEt 3 > PMe 3 > PPh 3 > P(OMe)3 > P(OPh)3 > PCl 3 > PF 3 Can be measured by IR using trans-M(CO)(PR 3) complexes Steric properties: Rigid structures create chiral complexes apex angle of a cone that encompasses the van der Waals radii of the outermost atoms of the ligand
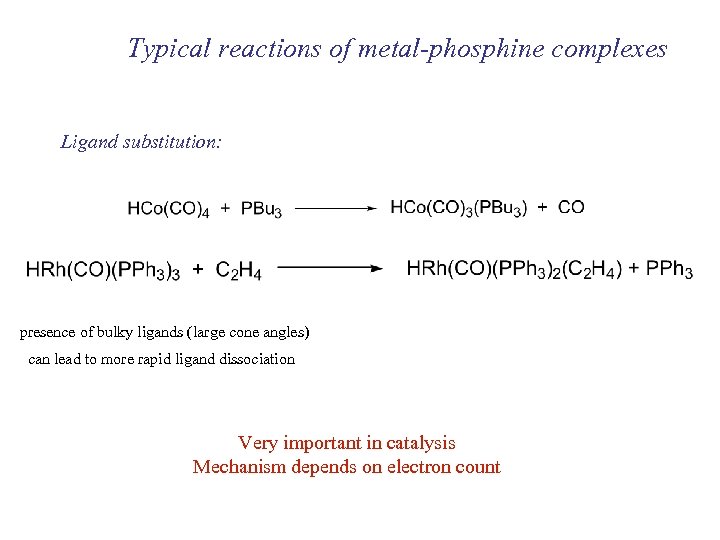
Typical reactions of metal-phosphine complexes Ligand substitution: presence of bulky ligands (large cone angles) can lead to more rapid ligand dissociation Very important in catalysis Mechanism depends on electron count
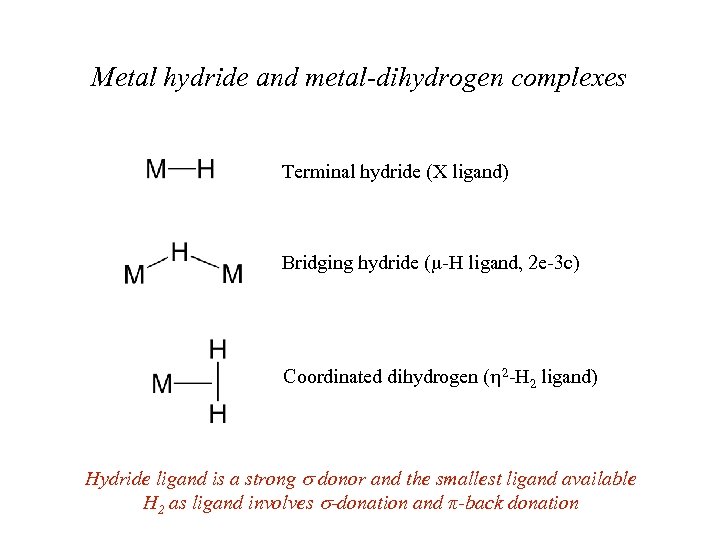
Metal hydride and metal-dihydrogen complexes Terminal hydride (X ligand) Bridging hydride (m-H ligand, 2 e-3 c) Coordinated dihydrogen (h 2 -H 2 ligand) Hydride ligand is a strong donor and the smallest ligand available H 2 as ligand involves -donation and π-back donation
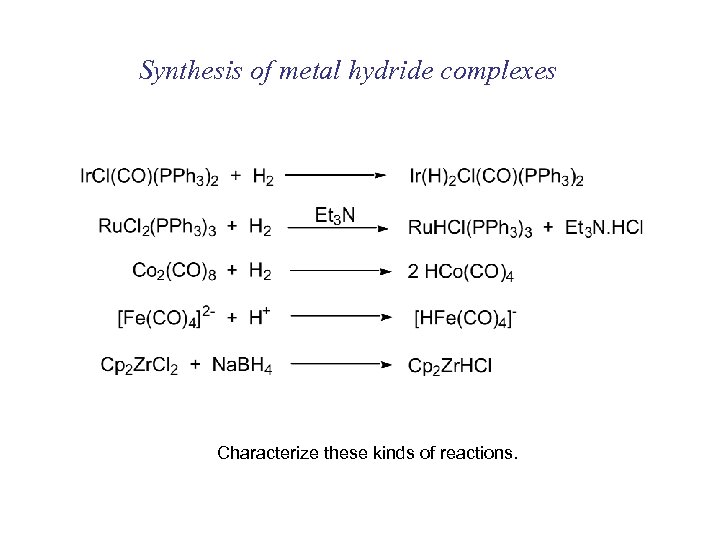
Synthesis of metal hydride complexes Characterize these kinds of reactions.

Characterization of metal hydride complexes 1 H NMR spectroscopy High field chemical shifts (d 0 to -25 ppm usual, up to -70 ppm possible) Coupling to metal nuclei (101 Rh, 183 W, 195 Pt) J(M-H) = 35 -1370 Hz Coupling between inequivalent hydrides J(H-H) = 1 -10 Hz Coupling to 31 P of phosphines J(H-P) = 10 -40 Hz cis; 90 -150 Hz trans IR spectroscopy n(M-H) = 1500 -2000 cm-1 (terminal); 800 -1600 cm-1 bridging n(M-H)/n(M-D) = √ 2 Weak bands, not very reliable
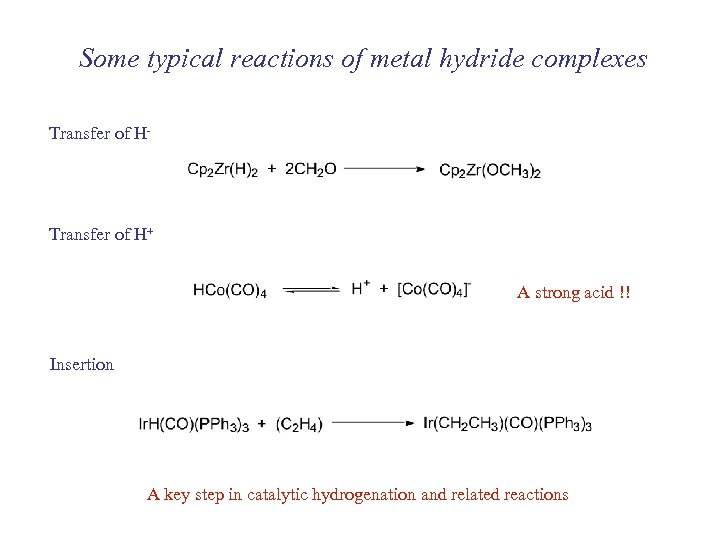
Some typical reactions of metal hydride complexes Transfer of H- Transfer of H+ A strong acid !! Insertion A key step in catalytic hydrogenation and related reactions
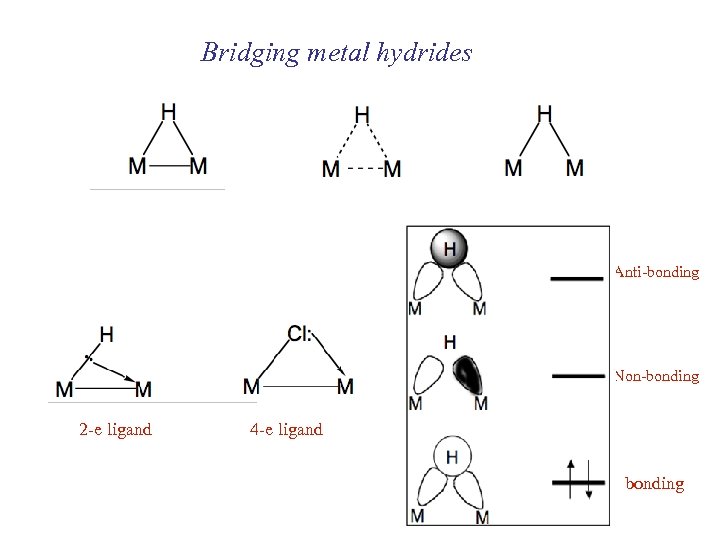
Bridging metal hydrides Anti-bonding Non-bonding 2 -e ligand 4 -e ligand bonding
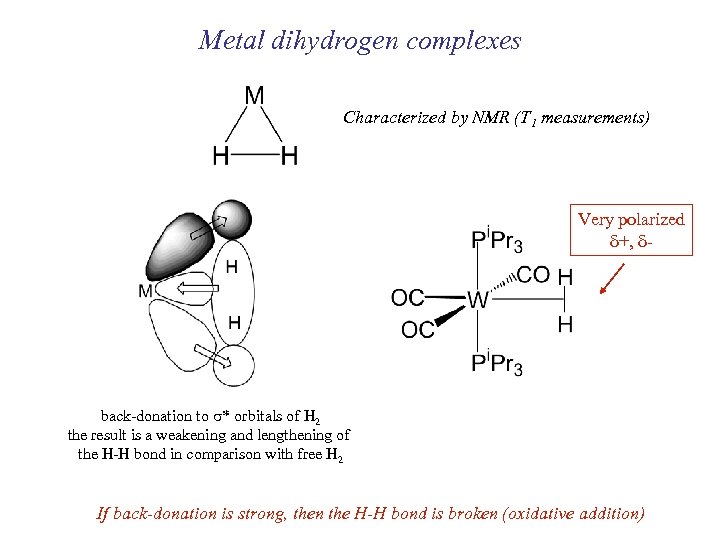
Metal dihydrogen complexes Characterized by NMR (T 1 measurements) Very polarized d+, d- back-donation to s* orbitals of H 2 the result is a weakening and lengthening of the H-H bond in comparison with free H 2 If back-donation is strong, then the H-H bond is broken (oxidative addition)
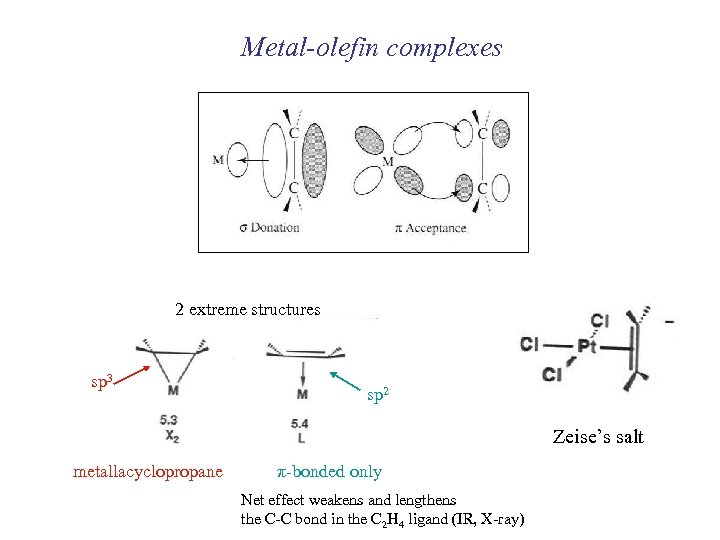
Metal-olefin complexes 2 extreme structures sp 3 sp 2 Zeise’s salt metallacyclopropane π-bonded only Net effect weakens and lengthens the C-C bond in the C 2 H 4 ligand (IR, X-ray)
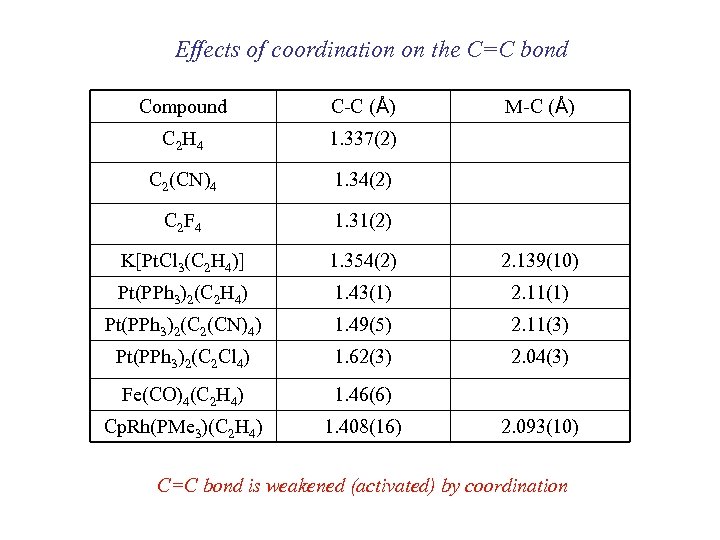
Effects of coordination on the C=C bond Compound C-C (Å) M-C (Å) C 2 H 4 1. 337(2) C 2(CN)4 1. 34(2) C 2 F 4 1. 31(2) K[Pt. Cl 3(C 2 H 4)] 1. 354(2) 2. 139(10) Pt(PPh 3)2(C 2 H 4) 1. 43(1) 2. 11(1) Pt(PPh 3)2(C 2(CN)4) 1. 49(5) 2. 11(3) Pt(PPh 3)2(C 2 Cl 4) 1. 62(3) 2. 04(3) Fe(CO)4(C 2 H 4) 1. 46(6) Cp. Rh(PMe 3)(C 2 H 4) 1. 408(16) 2. 093(10) C=C bond is weakened (activated) by coordination
Characterization of metal-olefin complexes IR n(C=C) ~ 1500 cm-1 (w) NMR 1 H and 13 C, d < free ligand X-rays C=C and M-C bond lengths indicate strength of bond
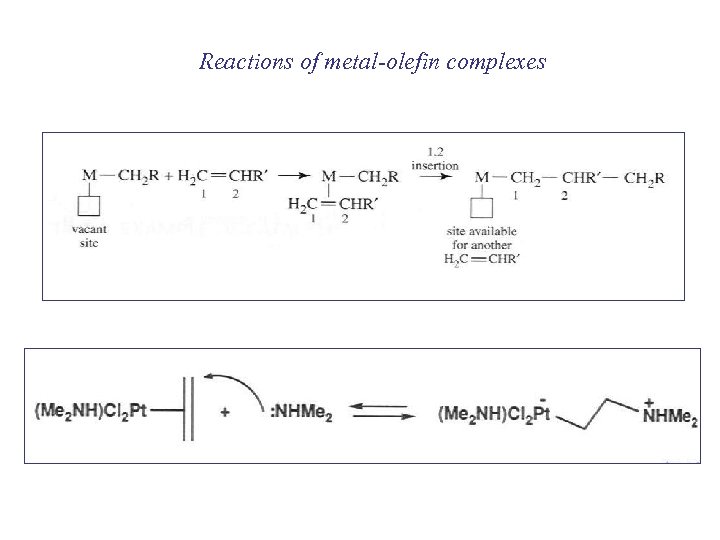
Reactions of metal-olefin complexes
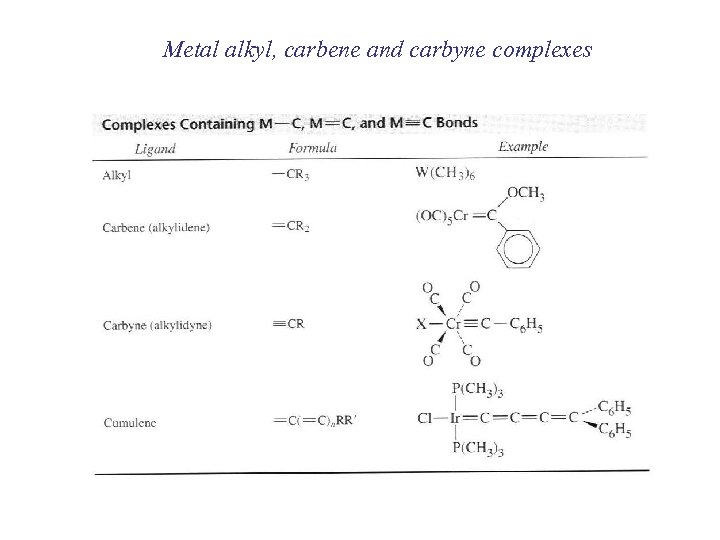
Metal alkyl, carbene and carbyne complexes

Metal-alkyl complexes Main group metal-alkyls known since old times (Et 2 Zn, Frankland 1857; R-Mg-X, Grignard, 1903)) Transition-metal alkyls mainly from the 1960’s onward W(CH 3)6 Ti(CH 3)6 Cp(CO)2 Fe(CH 2 CH 3)6 Pt. H(C CH)L 2 [Cr(H 2 O)5(CH 2 CH 3)6]2+ Why were they so elusive? Kinetically unstable (although thermodynamically stable)
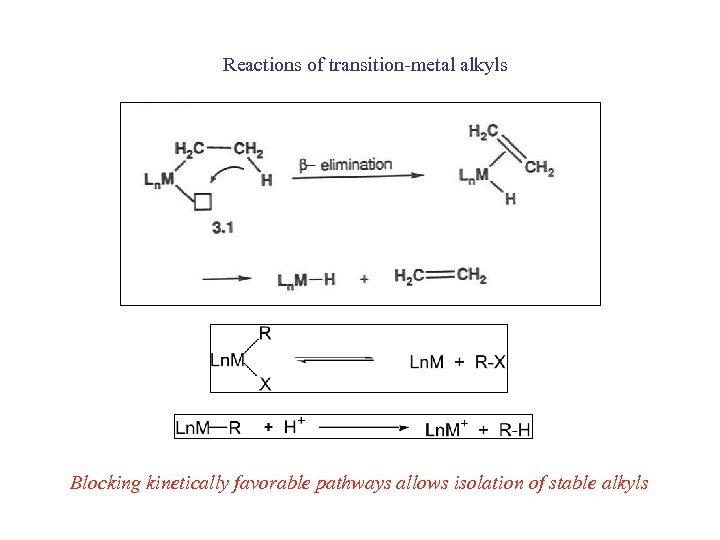
Reactions of transition-metal alkyls Blocking kinetically favorable pathways allows isolation of stable alkyls
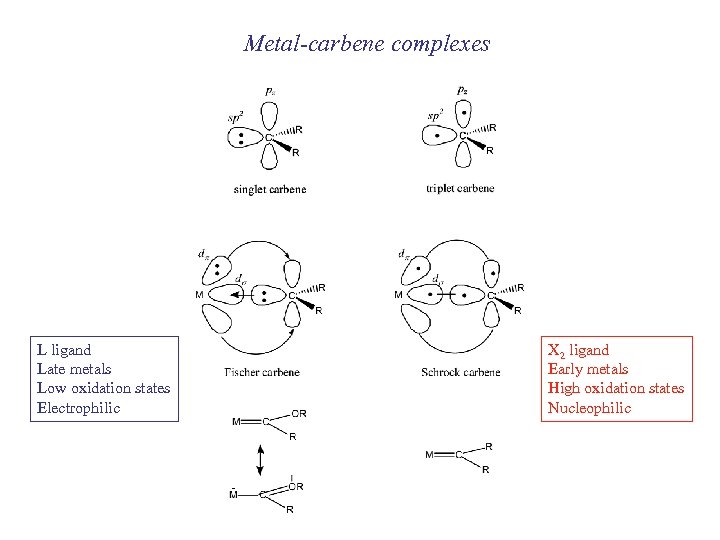
Metal-carbene complexes L ligand Late metals Low oxidation states Electrophilic X 2 ligand Early metals High oxidation states Nucleophilic
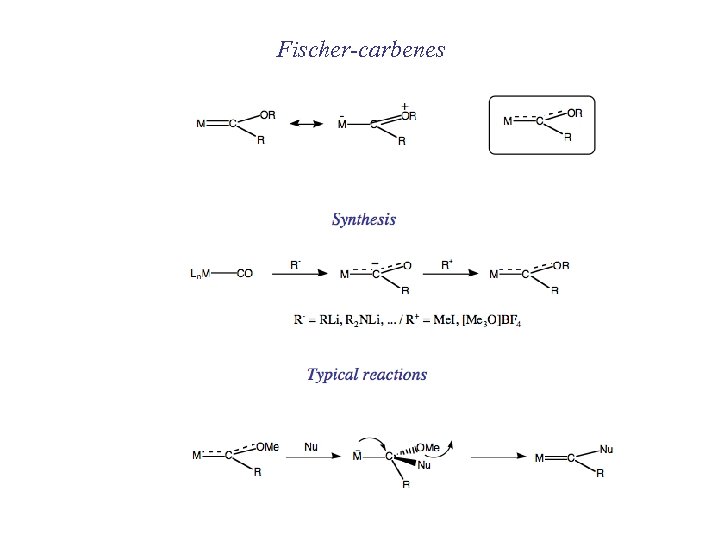
Fischer-carbenes
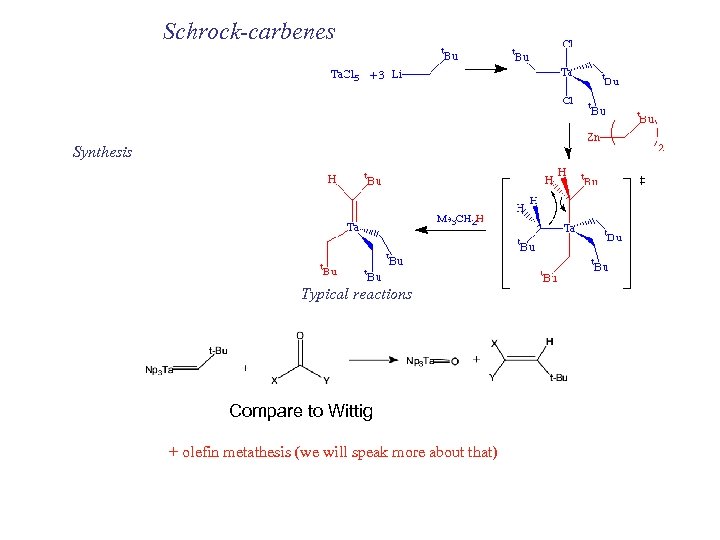
Schrock-carbenes Synthesis Typical reactions Compare to Wittig + olefin metathesis (we will speak more about that)
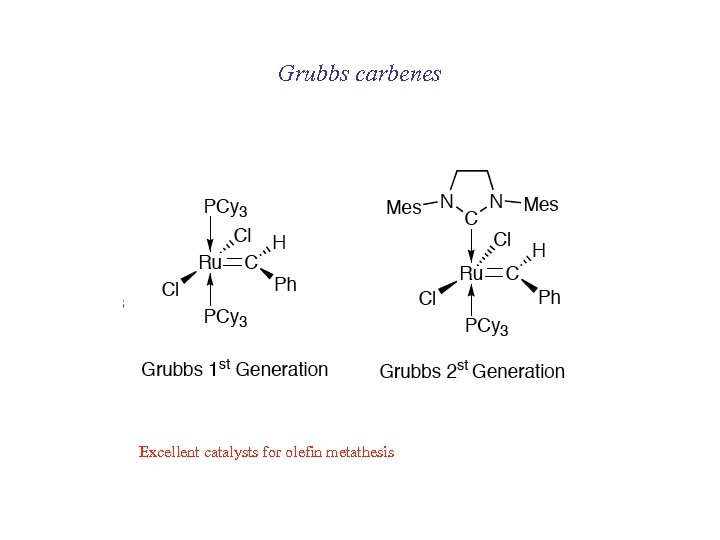
Grubbs carbenes Excellent catalysts for olefin metathesis
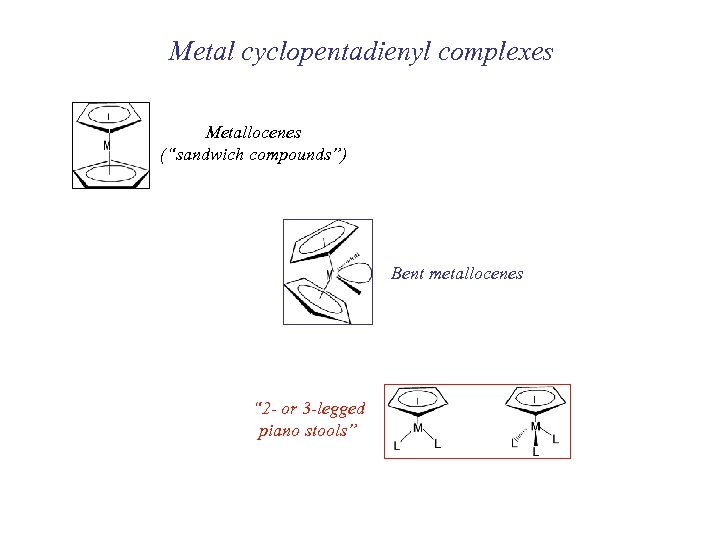
Metal cyclopentadienyl complexes Metallocenes (“sandwich compounds”) Bent metallocenes “ 2 - or 3 -legged piano stools”
Homogeneous catalysis: an important application of organometallic compounds Catalysis in a homogeneous liquid phase Very important fundamentally Many synthetic and industrial applications
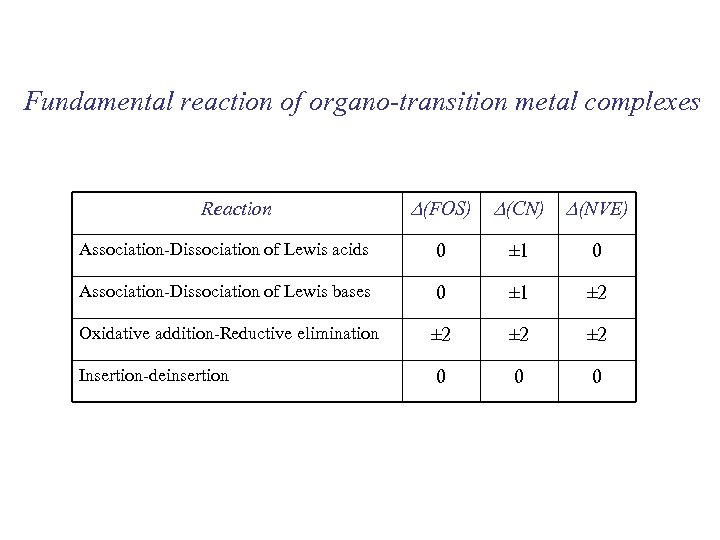
Fundamental reaction of organo-transition metal complexes Reaction D(FOS) D(CN) D(NVE) Association-Dissociation of Lewis acids 0 ± 1 0 Association-Dissociation of Lewis bases 0 ± 1 ± 2 Oxidative addition-Reductive elimination ± 2 ± 2 0 0 0 Insertion-deinsertion
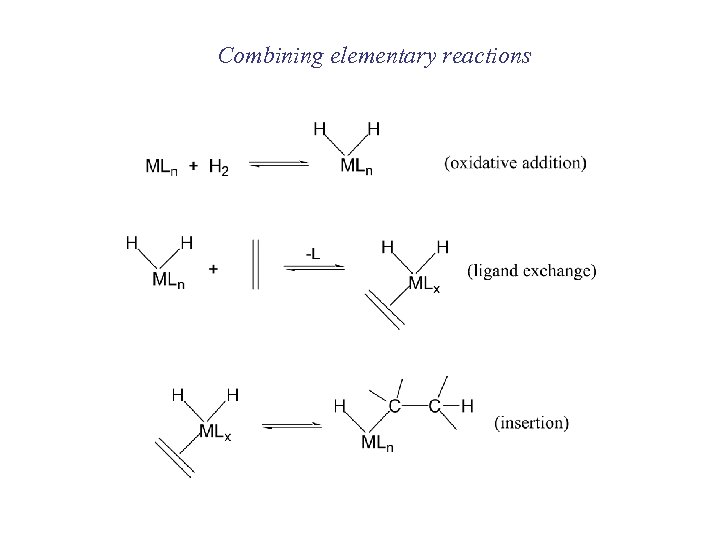
Combining elementary reactions
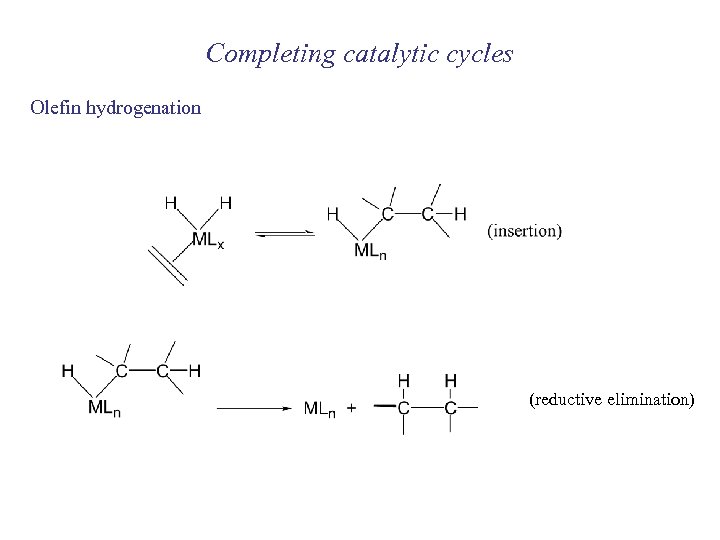
Completing catalytic cycles Olefin hydrogenation (reductive elimination)
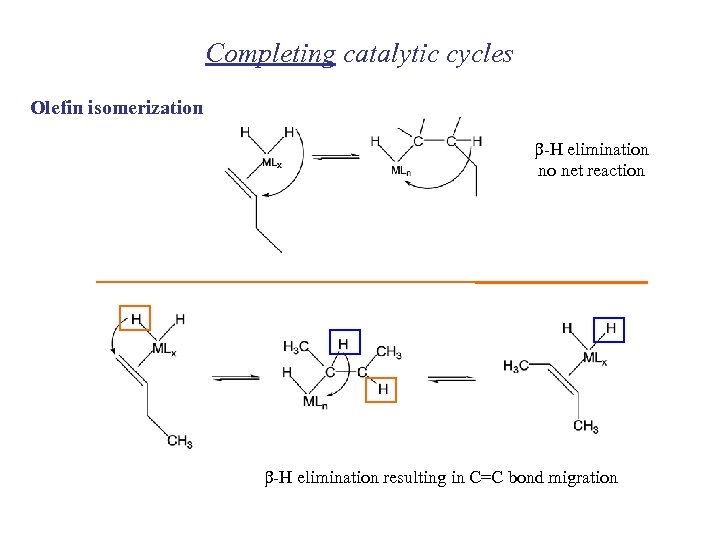
Completing catalytic cycles Olefin isomerization b-H elimination no net reaction b-H elimination resulting in C=C bond migration
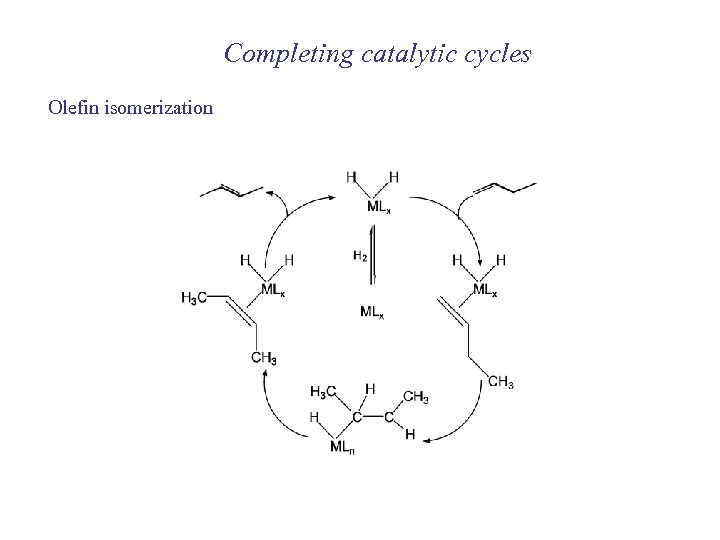
Completing catalytic cycles Olefin isomerization
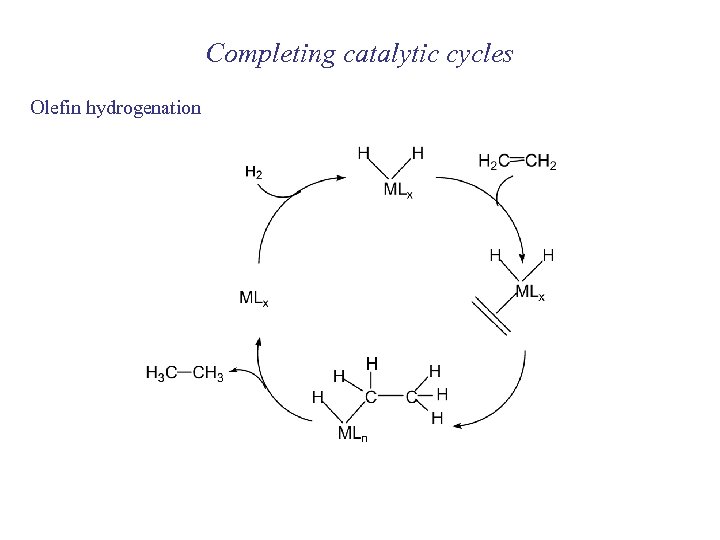
Completing catalytic cycles Olefin hydrogenation
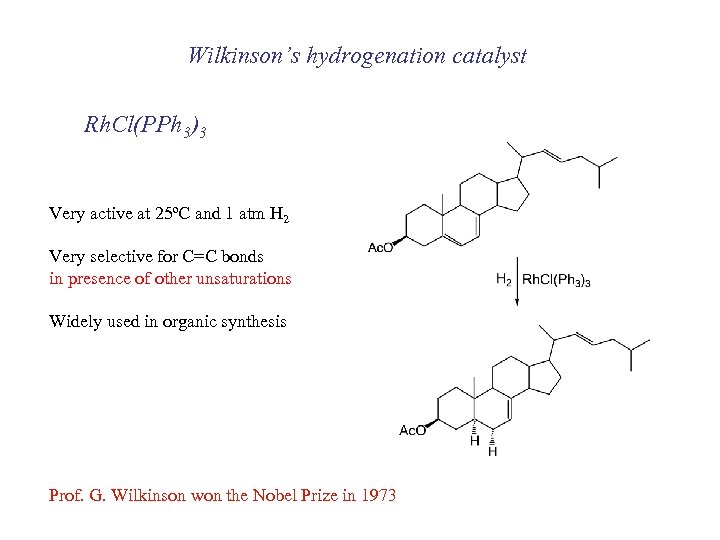
Wilkinson’s hydrogenation catalyst Rh. Cl(PPh 3)3 Very active at 25ºC and 1 atm H 2 Very selective for C=C bonds in presence of other unsaturations Widely used in organic synthesis Prof. G. Wilkinson won the Nobel Prize in 1973
![Other hydrogenation catalysts [Rh(H)2(PR 3)2(solv)2]+ With a large variety of phosphines including chiral ones Other hydrogenation catalysts [Rh(H)2(PR 3)2(solv)2]+ With a large variety of phosphines including chiral ones](https://present5.com/presentation/6902524c9b064fc972a0fb3a73de5268/image-35.jpg)
Other hydrogenation catalysts [Rh(H)2(PR 3)2(solv)2]+ With a large variety of phosphines including chiral ones for enantioselective hydrogenation Ru. II/(chiral diphosphine)/diamine Extremely efficient catalysts for the enantioselective hydrogenation of C=C and C=O bonds Profs. Noyori, Sharpless and Knowles won the Nobel Prize in 2001
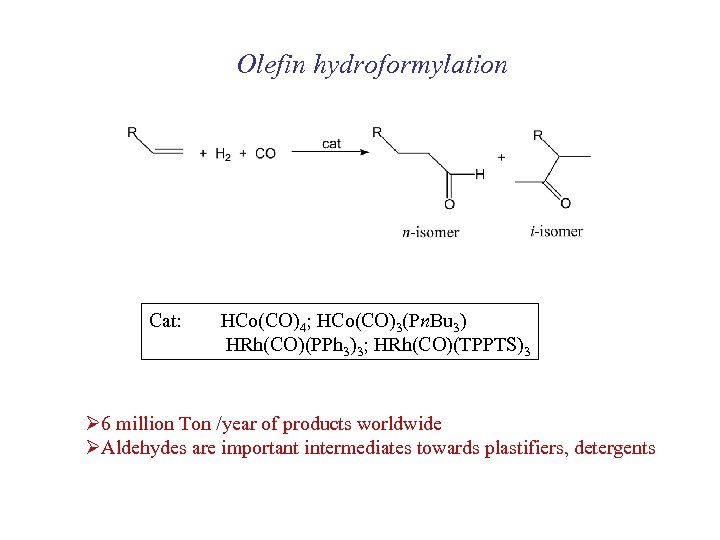
Olefin hydroformylation Cat: HCo(CO)4; HCo(CO)3(Pn. Bu 3) HRh(CO)(PPh 3)3; HRh(CO)(TPPTS)3 Ø 6 million Ton /year of products worldwide ØAldehydes are important intermediates towards plastifiers, detergents
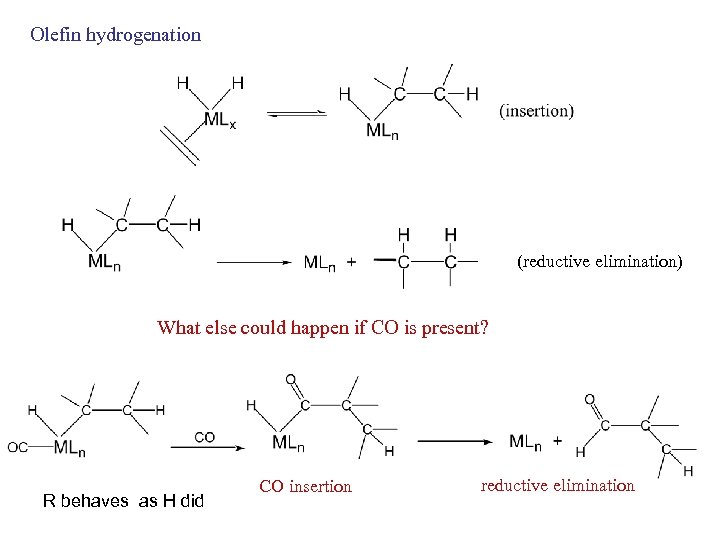
Olefin hydrogenation (reductive elimination) What else could happen if CO is present? R behaves as H did CO insertion reductive elimination
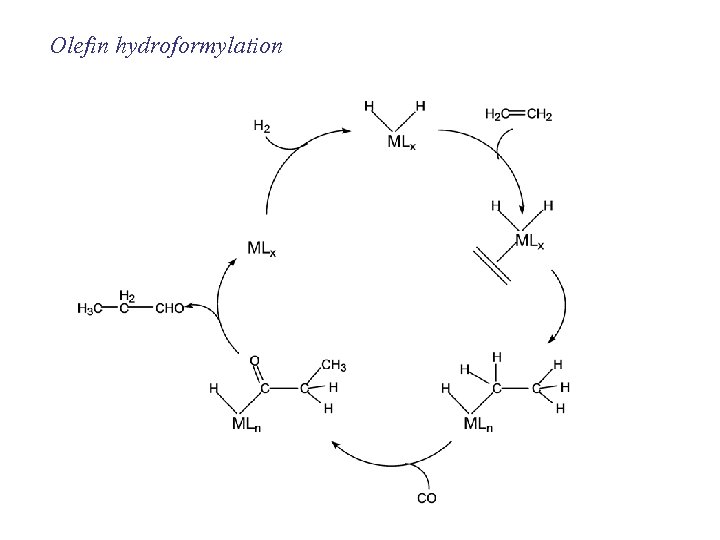
Olefin hydroformylation

Catalysts for polyolefin synthesis Polyolefins are the most important products of organometallic catalysis (> 60 million Tons per year) • Polyethylene (low, medium, high, ultrahigh density) used in packaging, containers, toys, house ware items, wire insulators, bags, pipes. • Polypropylene (food and beverage containers, medical tubing, bumpers, foot ware, thermal insulation, mats)
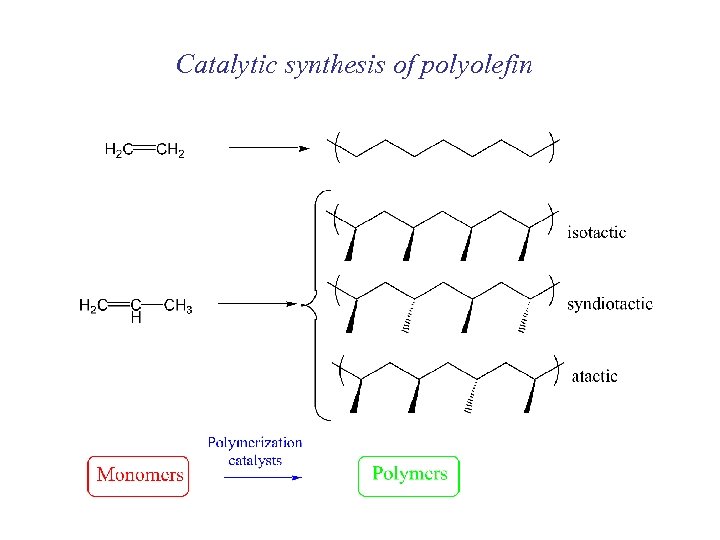
Catalytic synthesis of polyolefin
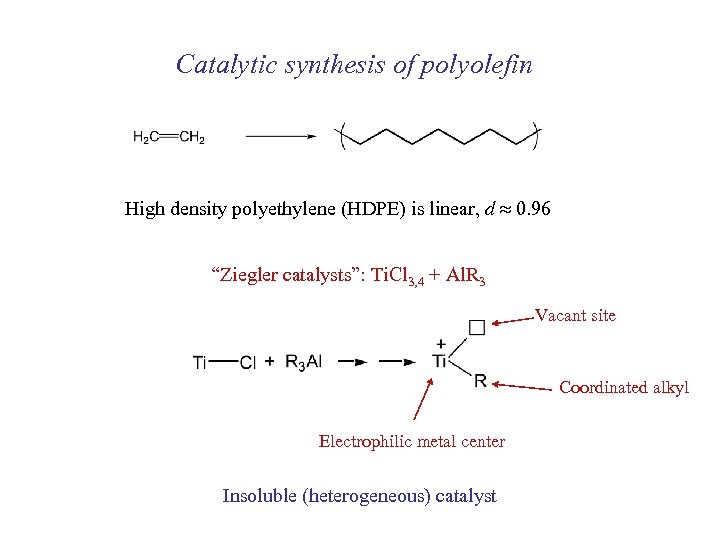
Catalytic synthesis of polyolefin High density polyethylene (HDPE) is linear, d 0. 96 “Ziegler catalysts”: Ti. Cl 3, 4 + Al. R 3 Vacant site Coordinated alkyl Electrophilic metal center Insoluble (heterogeneous) catalyst
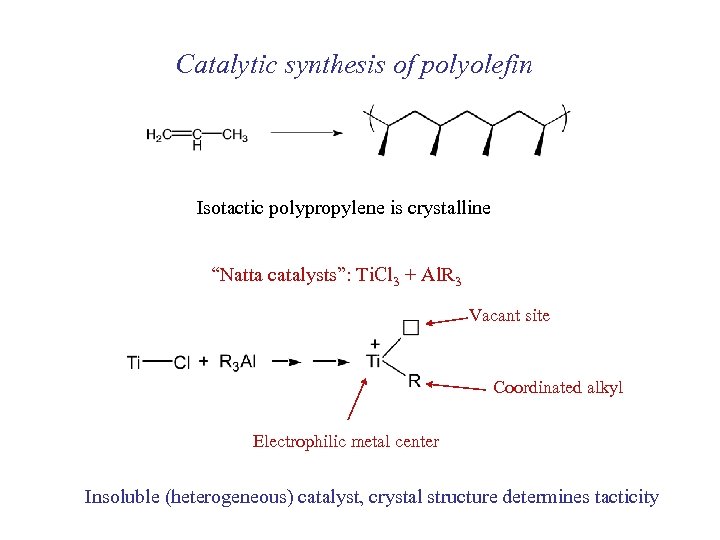
Catalytic synthesis of polyolefin Isotactic polypropylene is crystalline “Natta catalysts”: Ti. Cl 3 + Al. R 3 Vacant site Coordinated alkyl Electrophilic metal center Insoluble (heterogeneous) catalyst, crystal structure determines tacticity
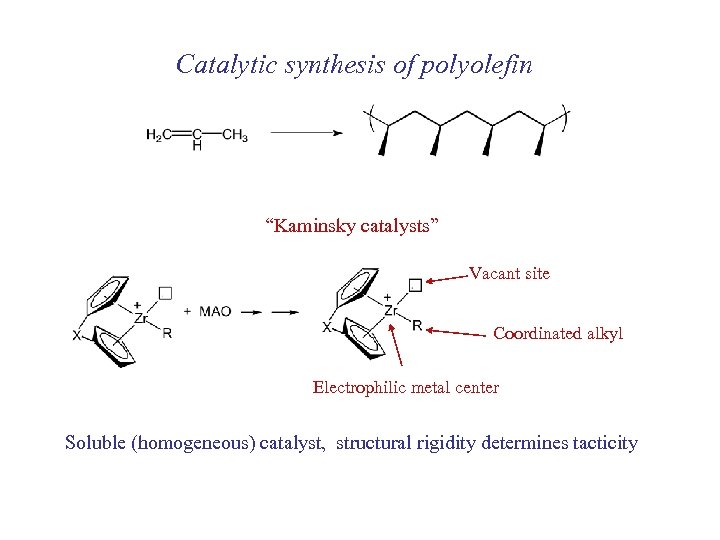
Catalytic synthesis of polyolefin “Kaminsky catalysts” Vacant site Coordinated alkyl Electrophilic metal center Soluble (homogeneous) catalyst, structural rigidity determines tacticity
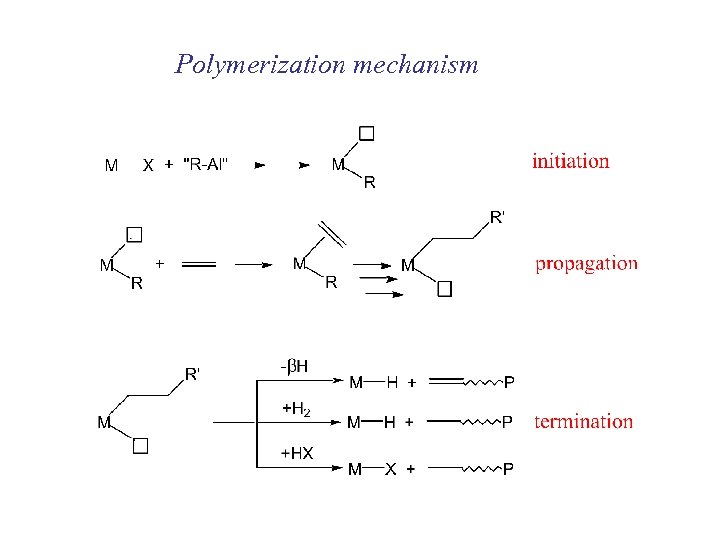
Polymerization mechanism
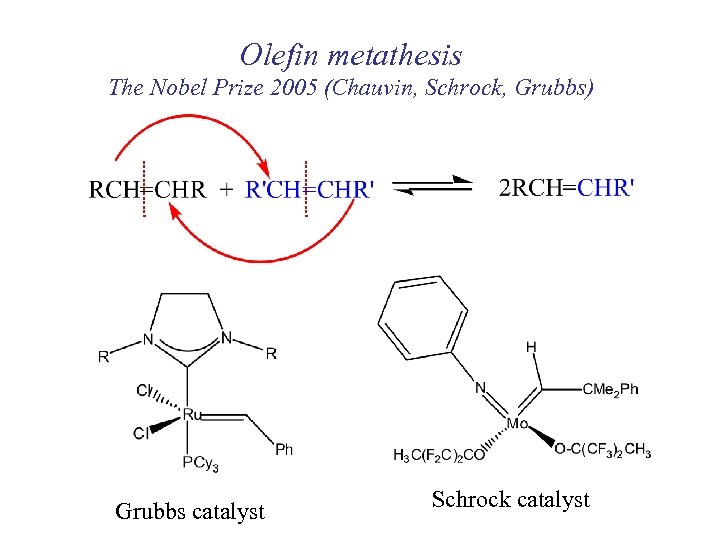
Olefin metathesis The Nobel Prize 2005 (Chauvin, Schrock, Grubbs) Grubbs catalyst Schrock catalyst
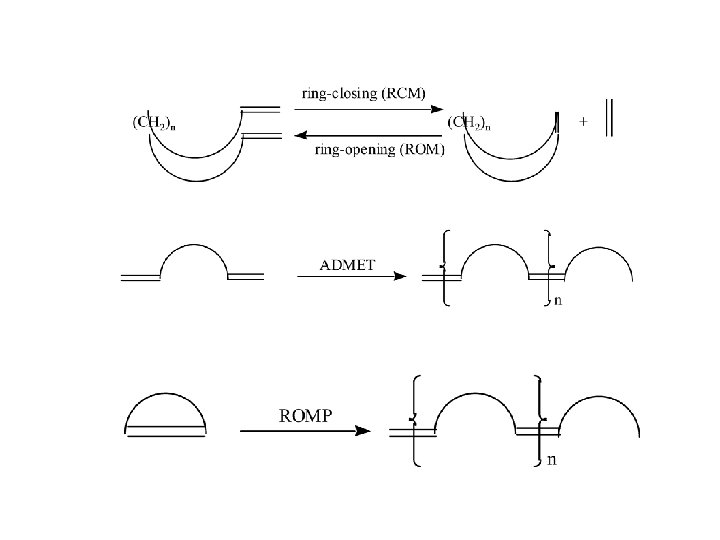
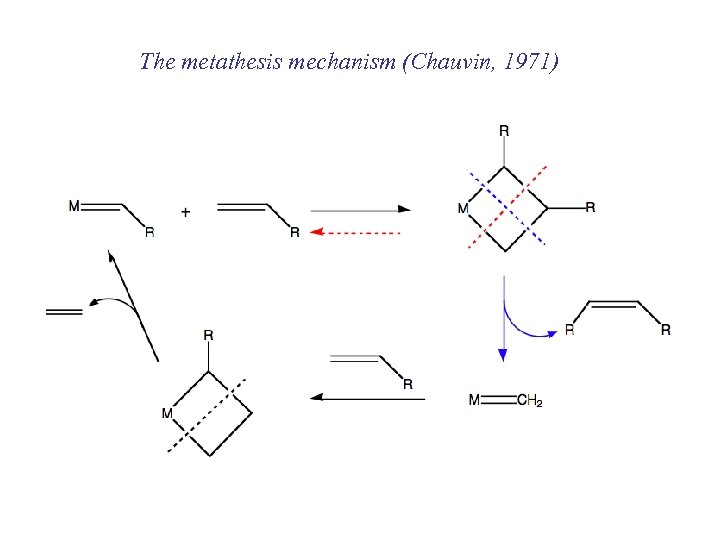
The metathesis mechanism (Chauvin, 1971)

6902524c9b064fc972a0fb3a73de5268.ppt