6e107b60205616547a20153242d66990.ppt
- Количество слайдов: 35
 © Medtronic, Inc. 2009 Patient Selection and Outcomes for Implantable Pain Therapies • Faculty Name • Institution • City State
© Medtronic, Inc. 2009 Patient Selection and Outcomes for Implantable Pain Therapies • Faculty Name • Institution • City State
 © Medtronic, Inc. 2009 Learning Objectives • Describe the treatment continuum model. • Define pain types effectively treated with implantable pain therapies. • Identify patient selection criteria for implantable pain therapies. • Describe chronic pain indications, benefits and risks for neurostimulation and implantable drug delivery therapies. • Describe the role of the psychological evaluation. • Explain the importance of patient expectations. 2
© Medtronic, Inc. 2009 Learning Objectives • Describe the treatment continuum model. • Define pain types effectively treated with implantable pain therapies. • Identify patient selection criteria for implantable pain therapies. • Describe chronic pain indications, benefits and risks for neurostimulation and implantable drug delivery therapies. • Describe the role of the psychological evaluation. • Explain the importance of patient expectations. 2
 © Medtronic, Inc. 2009 Definition of Chronic Pain • Definitions include the following: 1 – “It is not the duration of pain that distinguishes acute from chronic pain, but more importantly, the inability of the body to restore its physiological functions to normal homeostatic levels. ” – Chronic pain is commonly triggered by disease or injury. – Stress, environmental, and affective factors may contribute to the pain experience. – Chronic pain will continue when treatment stops. • Chronic pain has only recently been explored as a complex condition that requires individual treatment and a multidisciplinary approach. 1 Loeser JD and Melack R. Pain: an overview. The Lancet 3 1999; 53(9164): 1607 -1609.
© Medtronic, Inc. 2009 Definition of Chronic Pain • Definitions include the following: 1 – “It is not the duration of pain that distinguishes acute from chronic pain, but more importantly, the inability of the body to restore its physiological functions to normal homeostatic levels. ” – Chronic pain is commonly triggered by disease or injury. – Stress, environmental, and affective factors may contribute to the pain experience. – Chronic pain will continue when treatment stops. • Chronic pain has only recently been explored as a complex condition that requires individual treatment and a multidisciplinary approach. 1 Loeser JD and Melack R. Pain: an overview. The Lancet 3 1999; 53(9164): 1607 -1609.
 © Medtronic, Inc. 2009 Pain Treatment Ladder 1 Device therapies are now considered earlier in the treatment continuum 1 Stamatos JM, Forde G, Kechejian P et al. Live Your Life Pain Free: Medical Discoveries that Stop Chronic Pain. Mc. Kinney, TX: Magni 4 Company; 2005.
© Medtronic, Inc. 2009 Pain Treatment Ladder 1 Device therapies are now considered earlier in the treatment continuum 1 Stamatos JM, Forde G, Kechejian P et al. Live Your Life Pain Free: Medical Discoveries that Stop Chronic Pain. Mc. Kinney, TX: Magni 4 Company; 2005.
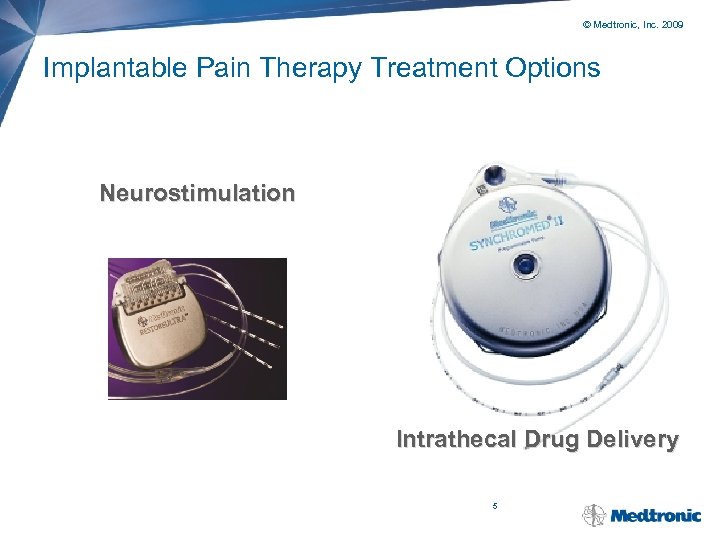 © Medtronic, Inc. 2009 Implantable Pain Therapy Treatment Options Neurostimulation Intrathecal Drug Delivery 5
© Medtronic, Inc. 2009 Implantable Pain Therapy Treatment Options Neurostimulation Intrathecal Drug Delivery 5
 © Medtronic, Inc. 2009 Pain Types Pain types related to implantable pain therapies: • Neuropathic pain • Nociceptive pain • Mixed pain How do pain types impact therapy selection? 6
© Medtronic, Inc. 2009 Pain Types Pain types related to implantable pain therapies: • Neuropathic pain • Nociceptive pain • Mixed pain How do pain types impact therapy selection? 6
 © Medtronic, Inc. 2009 Neuropathic Pain • Associated with injury to the peripheral nervous system or the spinal cord • Perceived as shooting, shock-like pain with severe burning or aching sensations – May have tingling, numbness, or itching – Severe cases have a ripping or tearing sensation • May be responsive to neurostimulation 7
© Medtronic, Inc. 2009 Neuropathic Pain • Associated with injury to the peripheral nervous system or the spinal cord • Perceived as shooting, shock-like pain with severe burning or aching sensations – May have tingling, numbness, or itching – Severe cases have a ripping or tearing sensation • May be responsive to neurostimulation 7
 © Medtronic, Inc. 2009 Nociceptive Pain • Activated in response to tissue damage or inflammation arising from receptors sensitive to noxious stimuli • Can be perceived as: – Well localized, constant, aching, throbbing, dull, vague, or a pressured feeling – Poorly localized and diffuse • Some patients describe as intermittent and sharp pain • Often responsive to opioid treatment; may be responsive to intrathecal drug delivery therapy 8
© Medtronic, Inc. 2009 Nociceptive Pain • Activated in response to tissue damage or inflammation arising from receptors sensitive to noxious stimuli • Can be perceived as: – Well localized, constant, aching, throbbing, dull, vague, or a pressured feeling – Poorly localized and diffuse • Some patients describe as intermittent and sharp pain • Often responsive to opioid treatment; may be responsive to intrathecal drug delivery therapy 8
 © Medtronic, Inc. 2009 Mixed Pain • Most chronic pain is mixed pain. • Mixed pain is a complex condition with neuropathic and nociceptive characteristics. • Pain perception depends on the condition. • Conditions likely to produce mixed pain include: – Chronic low back pain. – Some forms of cancer pain and treatment. • Patient may respond to neurostimulation or intrathecal drug delivery therapy. Clinical judgment and patient characteristics guide therapy selection. 9
© Medtronic, Inc. 2009 Mixed Pain • Most chronic pain is mixed pain. • Mixed pain is a complex condition with neuropathic and nociceptive characteristics. • Pain perception depends on the condition. • Conditions likely to produce mixed pain include: – Chronic low back pain. – Some forms of cancer pain and treatment. • Patient may respond to neurostimulation or intrathecal drug delivery therapy. Clinical judgment and patient characteristics guide therapy selection. 9
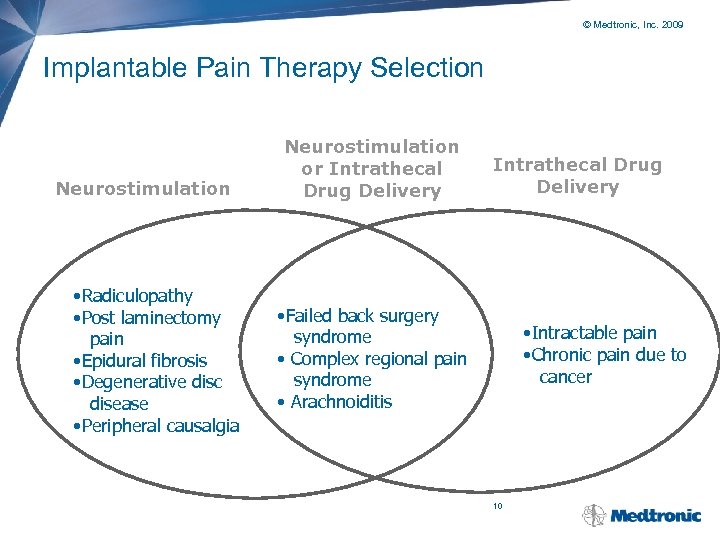 © Medtronic, Inc. 2009 Implantable Pain Therapy Selection Neurostimulation • Radiculopathy • Post laminectomy pain • Epidural fibrosis • Degenerative disc disease • Peripheral causalgia Neurostimulation or Intrathecal Drug Delivery • Failed back surgery syndrome • Complex regional pain syndrome • Arachnoiditis • Intractable pain • Chronic pain due to cancer 10
© Medtronic, Inc. 2009 Implantable Pain Therapy Selection Neurostimulation • Radiculopathy • Post laminectomy pain • Epidural fibrosis • Degenerative disc disease • Peripheral causalgia Neurostimulation or Intrathecal Drug Delivery • Failed back surgery syndrome • Complex regional pain syndrome • Arachnoiditis • Intractable pain • Chronic pain due to cancer 10
 © Medtronic, Inc. 2009 Patient Selection Criteria for Implantable Pain Therapies: Non-malignant Pain • • • Inadequate pain relief and/or intolerable side effects after treatment with more conservative therapies Physiological origin of the pain No current signs of drug abuse or addiction Appropriate surgical candidate Additional surgical interventions are not indicated • • Patient understands therapy limits and risks Patient can articulate goals of therapy and wants to participate in treatment Psychological evaluation and clearance was obtained A screening trial has been successful Patient can properly operate the system Patient is ≥ 18 years old Patient is not pregnant 11
© Medtronic, Inc. 2009 Patient Selection Criteria for Implantable Pain Therapies: Non-malignant Pain • • • Inadequate pain relief and/or intolerable side effects after treatment with more conservative therapies Physiological origin of the pain No current signs of drug abuse or addiction Appropriate surgical candidate Additional surgical interventions are not indicated • • Patient understands therapy limits and risks Patient can articulate goals of therapy and wants to participate in treatment Psychological evaluation and clearance was obtained A screening trial has been successful Patient can properly operate the system Patient is ≥ 18 years old Patient is not pregnant 11
 © Medtronic, Inc. 2009 Patient Selection Criteria for Implantable Pain Therapies: Malignant Pain • Pain unrelieved by more appropriate conservative therapies 1 • Life expectancy >3 months 2 • No cerebrospinal fluid obstruction 2 • Available network for post-surgical management 2 • Trained healthcare providers and pharmacists, home health, or hospice – Ability to change dose or drugs • Appropriate surgical candidate 1 Follett K and Doleys D. Selection of Candidates for Intrathecal Drug Administration to Treat Chronic Pain: Considerations in Pre-implantation Trials. Minneapolis: Medtronic Inc. , 2002: 1 - 19. 2 Smith T. J. and Coyne P. J. How to Use Implantable Drug Delivery Systems for 12 Refractory Cancer Pain. J Supportive Oncology 1: 1: 1 -4.
© Medtronic, Inc. 2009 Patient Selection Criteria for Implantable Pain Therapies: Malignant Pain • Pain unrelieved by more appropriate conservative therapies 1 • Life expectancy >3 months 2 • No cerebrospinal fluid obstruction 2 • Available network for post-surgical management 2 • Trained healthcare providers and pharmacists, home health, or hospice – Ability to change dose or drugs • Appropriate surgical candidate 1 Follett K and Doleys D. Selection of Candidates for Intrathecal Drug Administration to Treat Chronic Pain: Considerations in Pre-implantation Trials. Minneapolis: Medtronic Inc. , 2002: 1 - 19. 2 Smith T. J. and Coyne P. J. How to Use Implantable Drug Delivery Systems for 12 Refractory Cancer Pain. J Supportive Oncology 1: 1: 1 -4.
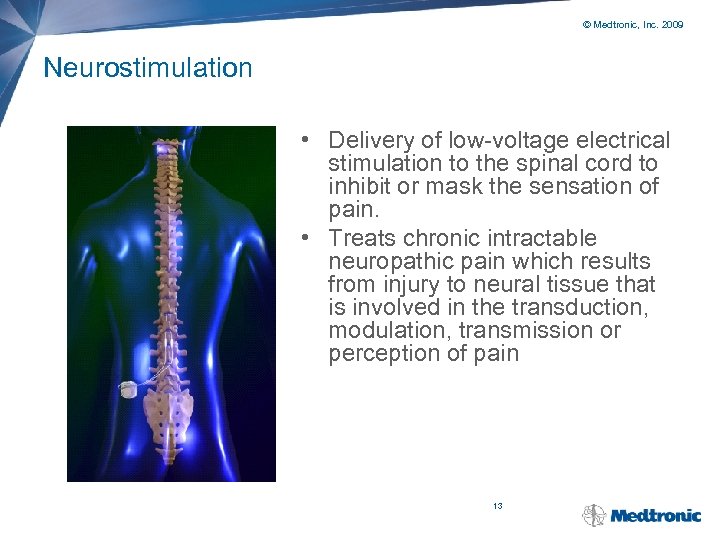 © Medtronic, Inc. 2009 Neurostimulation • Delivery of low-voltage electrical stimulation to the spinal cord to inhibit or mask the sensation of pain. • Treats chronic intractable neuropathic pain which results from injury to neural tissue that is involved in the transduction, modulation, transmission or perception of pain 13
© Medtronic, Inc. 2009 Neurostimulation • Delivery of low-voltage electrical stimulation to the spinal cord to inhibit or mask the sensation of pain. • Treats chronic intractable neuropathic pain which results from injury to neural tissue that is involved in the transduction, modulation, transmission or perception of pain 13
 © Medtronic, Inc. 2009 History of Neurostimulation • • 1962 - First implant of carotid sinus nerve stimulator 1967 - Development of first “dorsal column” stimulator 1970 s - Spinal cord stimulator becomes widely used 1980 - Medtronic introduces first programmable electrode system in U. S. • 1982 - First clinical implant of Itrel® totally implantable neurostimulator • Today - More than 100, 000 patients have received Medtronic neurostimulation therapy for chronic pain 14
© Medtronic, Inc. 2009 History of Neurostimulation • • 1962 - First implant of carotid sinus nerve stimulator 1967 - Development of first “dorsal column” stimulator 1970 s - Spinal cord stimulator becomes widely used 1980 - Medtronic introduces first programmable electrode system in U. S. • 1982 - First clinical implant of Itrel® totally implantable neurostimulator • Today - More than 100, 000 patients have received Medtronic neurostimulation therapy for chronic pain 14
 © Medtronic, Inc. 2009 Neurostimulation Indications Indicated as an aid in the management of chronic intractable pain of the trunk and/or limbs, including unilateral or bilateral pain associated with the following conditions: • • • Failed Back Syndrome or Low Back Syndrome or Failed Back Radicular Pain Syndrome or Radiculopathies resulting in pain secondary to Failed Back Syndrome or Herniated Disk Post-laminectomy Pain Multiple Back Operations Unsuccessful Disk Surgery • • • Degenerative Disk Disease (DDD)/Herniated Disk pain refractory to conservative and surgical interventions Peripheral Causalgia Epidural Fibrosis Arachnoiditis or Lumbar Adhesive Arachnoiditis Complex Regional Pain Syndrome (CRPS) or Reflex Sympathetic Dystrophy (RSD) or Causalgia Refer to the package labeling for a complete list of indications. 15
© Medtronic, Inc. 2009 Neurostimulation Indications Indicated as an aid in the management of chronic intractable pain of the trunk and/or limbs, including unilateral or bilateral pain associated with the following conditions: • • • Failed Back Syndrome or Low Back Syndrome or Failed Back Radicular Pain Syndrome or Radiculopathies resulting in pain secondary to Failed Back Syndrome or Herniated Disk Post-laminectomy Pain Multiple Back Operations Unsuccessful Disk Surgery • • • Degenerative Disk Disease (DDD)/Herniated Disk pain refractory to conservative and surgical interventions Peripheral Causalgia Epidural Fibrosis Arachnoiditis or Lumbar Adhesive Arachnoiditis Complex Regional Pain Syndrome (CRPS) or Reflex Sympathetic Dystrophy (RSD) or Causalgia Refer to the package labeling for a complete list of indications. 15
 © Medtronic, Inc. 2009 Potential Benefits of Neurostimulation for Chronic Pain 1 • • • May result in a 50% - 70% reduction in pain May reduce use of narcotics May increase ability to perform activities of daily living Increases ability to work May be trialed to evaluate pain relief and functional improvement from therapy • May be reversed • May achieve long-term cost-effectiveness for batteries that last over one year 1 Taylor R, Caraway D, Van Buyten JP et al. eds. Spinal Cord Stimulation: Overcoming Barriers to Successful Outcomes for the Complex Pain Patient. 16 Minneapolis: Medtronic Inc. , 2006: 1 -10.
© Medtronic, Inc. 2009 Potential Benefits of Neurostimulation for Chronic Pain 1 • • • May result in a 50% - 70% reduction in pain May reduce use of narcotics May increase ability to perform activities of daily living Increases ability to work May be trialed to evaluate pain relief and functional improvement from therapy • May be reversed • May achieve long-term cost-effectiveness for batteries that last over one year 1 Taylor R, Caraway D, Van Buyten JP et al. eds. Spinal Cord Stimulation: Overcoming Barriers to Successful Outcomes for the Complex Pain Patient. 16 Minneapolis: Medtronic Inc. , 2006: 1 -10.
 © Medtronic, Inc. 2009 Risks of Neurostimulation for Chronic Pain 1 • The most frequently reported problems: – – – Infection Lead movement Pain at the implant site Loss of therapy effect Therapy which did not meet the patient's expectations • Some of the most severe reported problems: – Epidural hemorrhage – Spinal fluid leakage – Paralysis For a complete list of adverse events which have been associated with neurostimulation, please refer to the product labeling and information for prescribers. 1 Medtronic Implantable Systems Performance Registry (ISPR). 17 2007 Annual Report. Protocol NSP 0010 -10000. Report date: October 2, 2008.
© Medtronic, Inc. 2009 Risks of Neurostimulation for Chronic Pain 1 • The most frequently reported problems: – – – Infection Lead movement Pain at the implant site Loss of therapy effect Therapy which did not meet the patient's expectations • Some of the most severe reported problems: – Epidural hemorrhage – Spinal fluid leakage – Paralysis For a complete list of adverse events which have been associated with neurostimulation, please refer to the product labeling and information for prescribers. 1 Medtronic Implantable Systems Performance Registry (ISPR). 17 2007 Annual Report. Protocol NSP 0010 -10000. Report date: October 2, 2008.
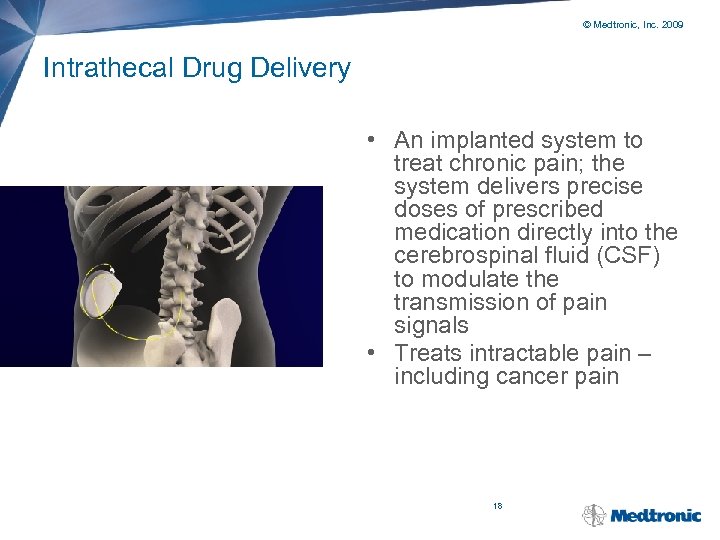 © Medtronic, Inc. 2009 Intrathecal Drug Delivery • An implanted system to treat chronic pain; the system delivers precise doses of prescribed medication directly into the cerebrospinal fluid (CSF) to modulate the transmission of pain signals • Treats intractable pain – including cancer pain 18
© Medtronic, Inc. 2009 Intrathecal Drug Delivery • An implanted system to treat chronic pain; the system delivers precise doses of prescribed medication directly into the cerebrospinal fluid (CSF) to modulate the transmission of pain signals • Treats intractable pain – including cancer pain 18
 © Medtronic, Inc. 2009 History of Intrathecal Drug Delivery • 1982 - First clinical implant of programmable pump (became Synchro. Med pump) for intrathecal morphine • 1988 - Market release of implantable programmable Synchro. Med Pump (IV use for cancer chemotherapy) • 1991 - Market release of Synchro. Med Pump (intrathecal morphine for cancer pain and non-malignant pain) • Today - More than 100, 000 patients worldwide have received a Medtronic pump for chronic pain 19
© Medtronic, Inc. 2009 History of Intrathecal Drug Delivery • 1982 - First clinical implant of programmable pump (became Synchro. Med pump) for intrathecal morphine • 1988 - Market release of implantable programmable Synchro. Med Pump (IV use for cancer chemotherapy) • 1991 - Market release of Synchro. Med Pump (intrathecal morphine for cancer pain and non-malignant pain) • Today - More than 100, 000 patients worldwide have received a Medtronic pump for chronic pain 19
 © Medtronic, Inc. 2009 Intrathecal Drug Delivery Indications • The Synchro. Med® Pump is indicated for use when patient therapy requires the chronic intrathecal infusion of medications. • Approved for use with: – Preservative-free morphine sulfate sterile solution in the treatment of chronic intractable pain. – Preservative-free ziconotide sterile solution for the management of severe chronic pain. – Lioresal® Intrathecal (baclofen injection) in the management of severe spasticity. – Chronic intravascular infusion of floxuridine (FUDR), methotrexate, doxorubicin or cisplatin for the treatment of primary or metastatic cancer. Refer to the package labeling for a complete list of indications. Lioresal® is a registered trademark of Novartis Pharmaceuticals, Inc. 20
© Medtronic, Inc. 2009 Intrathecal Drug Delivery Indications • The Synchro. Med® Pump is indicated for use when patient therapy requires the chronic intrathecal infusion of medications. • Approved for use with: – Preservative-free morphine sulfate sterile solution in the treatment of chronic intractable pain. – Preservative-free ziconotide sterile solution for the management of severe chronic pain. – Lioresal® Intrathecal (baclofen injection) in the management of severe spasticity. – Chronic intravascular infusion of floxuridine (FUDR), methotrexate, doxorubicin or cisplatin for the treatment of primary or metastatic cancer. Refer to the package labeling for a complete list of indications. Lioresal® is a registered trademark of Novartis Pharmaceuticals, Inc. 20
 © Medtronic, Inc. 2009 Intrathecal Drug Delivery Contraindications • IDD system implant is contraindicated when: – Infection is present. – When the pump cannot be implanted at the appropriate depth from the surface of the skin. – Body size is insufficient to accommodate the pump. – Contraindications exist relating to the drug (e. g. , allergic reaction). • Do not use the patient control device to administer opioid to opioid-naïve patients or to administer ziconotide or baclofen. Refer to the package labeling for a complete list of contraindications. 21
© Medtronic, Inc. 2009 Intrathecal Drug Delivery Contraindications • IDD system implant is contraindicated when: – Infection is present. – When the pump cannot be implanted at the appropriate depth from the surface of the skin. – Body size is insufficient to accommodate the pump. – Contraindications exist relating to the drug (e. g. , allergic reaction). • Do not use the patient control device to administer opioid to opioid-naïve patients or to administer ziconotide or baclofen. Refer to the package labeling for a complete list of contraindications. 21
 © Medtronic, Inc. 2009 Potential Benefits of Intrathecal Drug Delivery for Chronic Pain • Site-specific drug delivery 1 – Faster onset and longer lasting analgesia than other routes of administration – Equivalent analgesia with lower doses – Decreased likelihood of side effects because of lower doses • Noninvasive programmable therapy – Minimizes patient discomfort – Can be programmed to meet a patient’s changing needs • Can be trialed to evaluate pain relief and functional improvement of therapy • Reversible 1 Follett K and Doleys D. Selection of Candidates for Intrathecal Drug Administration to Treat Chronic Pain: Considerations in Pre-implantation Trials. Minneapolis: Medtronic 22 Inc. , 2002: 1 - 19.
© Medtronic, Inc. 2009 Potential Benefits of Intrathecal Drug Delivery for Chronic Pain • Site-specific drug delivery 1 – Faster onset and longer lasting analgesia than other routes of administration – Equivalent analgesia with lower doses – Decreased likelihood of side effects because of lower doses • Noninvasive programmable therapy – Minimizes patient discomfort – Can be programmed to meet a patient’s changing needs • Can be trialed to evaluate pain relief and functional improvement of therapy • Reversible 1 Follett K and Doleys D. Selection of Candidates for Intrathecal Drug Administration to Treat Chronic Pain: Considerations in Pre-implantation Trials. Minneapolis: Medtronic 22 Inc. , 2002: 1 - 19.
 © Medtronic, Inc. 2009 Risks of Intrathecal Drug Delivery 1, 2 • The most frequently reported problems: – Infection – Spinal fluid leak – Pump inversion – Skin erosion – Drug side-effects – Loss of therapy effect – Therapy which did not meet the patient's expectations • Some of the most severe reported problems: – Inflammatory mass – Spinal cord damage – Meningitis – Life threatening drug adverse effects due to over infusion as a result of programming or patient monitoring errors or device malfunction – Complications due to use of unapproved drugs and/or not using drugs in accordance with drug labeling For a complete list of adverse events which have been associated with intrathecal drug delivery, please refer to the product labeling and information for prescribers. 1 Medtronic Implantable Systems Performance Registry (ISPR). 2007 Annual Report. Protocol NSP 001010000. Report date: October 2, 2008. 2 Coffey RJ, Owens, M, Broste, Steven K, et al. Mortality Associated with Intrathecal Opioid Drug Infusion to 23 Treat Non-Cancer Pain: Risk Factors and Mitigation. Late breaking paper presented at: North American Neuromodulation Society 12 th Annual Meeting; December 4 -7; 2008; Las Vegas, NV.
© Medtronic, Inc. 2009 Risks of Intrathecal Drug Delivery 1, 2 • The most frequently reported problems: – Infection – Spinal fluid leak – Pump inversion – Skin erosion – Drug side-effects – Loss of therapy effect – Therapy which did not meet the patient's expectations • Some of the most severe reported problems: – Inflammatory mass – Spinal cord damage – Meningitis – Life threatening drug adverse effects due to over infusion as a result of programming or patient monitoring errors or device malfunction – Complications due to use of unapproved drugs and/or not using drugs in accordance with drug labeling For a complete list of adverse events which have been associated with intrathecal drug delivery, please refer to the product labeling and information for prescribers. 1 Medtronic Implantable Systems Performance Registry (ISPR). 2007 Annual Report. Protocol NSP 001010000. Report date: October 2, 2008. 2 Coffey RJ, Owens, M, Broste, Steven K, et al. Mortality Associated with Intrathecal Opioid Drug Infusion to 23 Treat Non-Cancer Pain: Risk Factors and Mitigation. Late breaking paper presented at: North American Neuromodulation Society 12 th Annual Meeting; December 4 -7; 2008; Las Vegas, NV.
 © Medtronic, Inc. 2009 Patient Evaluation • • Obtain patient history and conduct physical examination Assess and document patient’s pain-related symptoms Evaluate other appropriate and less invasive therapies Assess patient motivation and commitment to therapy • Establish reasonable goals for therapy: – – Reduce pain Reduce use of oral medication and side effects Improve function Improve quality of life 24
© Medtronic, Inc. 2009 Patient Evaluation • • Obtain patient history and conduct physical examination Assess and document patient’s pain-related symptoms Evaluate other appropriate and less invasive therapies Assess patient motivation and commitment to therapy • Establish reasonable goals for therapy: – – Reduce pain Reduce use of oral medication and side effects Improve function Improve quality of life 24
 © Medtronic, Inc. 2009 Psychological Evaluation A psychological evaluation can be used to identify patients with characteristics that are positive indicators to a successful outcome. 1 – Assess existence of significant psychological signs, drug addiction or behavioral problems and unresolved issues of secondary gain. – Identify and treat psychological co-morbidities prior to therapy. – Evaluate patient willingness, understanding, and competency to actively participate in their therapy. – Establish a baseline against which to measure improvement. 1 Doleys DM. . Psychological factors in spinal cord stimulation therapy: brief 25 review and discussion. Neurosurgical Focus 2006: 21(6): 1 -6.
© Medtronic, Inc. 2009 Psychological Evaluation A psychological evaluation can be used to identify patients with characteristics that are positive indicators to a successful outcome. 1 – Assess existence of significant psychological signs, drug addiction or behavioral problems and unresolved issues of secondary gain. – Identify and treat psychological co-morbidities prior to therapy. – Evaluate patient willingness, understanding, and competency to actively participate in their therapy. – Establish a baseline against which to measure improvement. 1 Doleys DM. . Psychological factors in spinal cord stimulation therapy: brief 25 review and discussion. Neurosurgical Focus 2006: 21(6): 1 -6.
 © Medtronic, Inc. 2009 Patient Expectations Candidates must: • Be able to determine specific, measurable goals, and expectations for therapy. • Want pain to decrease and ability to function to improve. • Understand that pain has multiple components. • Express realistic expectations about treatment outcomes. • Be able and willing to participate in therapy and rehabilitation. 26
© Medtronic, Inc. 2009 Patient Expectations Candidates must: • Be able to determine specific, measurable goals, and expectations for therapy. • Want pain to decrease and ability to function to improve. • Understand that pain has multiple components. • Express realistic expectations about treatment outcomes. • Be able and willing to participate in therapy and rehabilitation. 26
 © Medtronic, Inc. 2009 References • • • Coffey RJ, Owens, M, Broste, Steven K, et al. Mortality Associated with Intrathecal Opioid Drug Infusion to Treat Non-Cancer Pain: Risk Factors and Mitigation. Late breaking paper presented at: North American Neuromodulation Society 12 th Annual Meeting; December 4 -7; 2008; Las Vegas, NV. de Lissovoy G, Brown RE, Halpern M et al. Cost Effectiveness of Long-term Intrathecal Morphine for Pain Associated with Failed Back Surgery Syndrome. Clinical Therapeutics 1997; 19(1): 96– 112. Deer T, Chapple I, Classen A, et al. Intrathecal drug delivery for treatment of chronic low back pain: report from the National Outcomes Registry for low back pain. Pain Med 2004; 5(1): 6 -13. Doleys DM. Psychological factors in spinal cord stimulation therapy: brief review and discussion. Neurosurgical Focus 2006: 21(6): 1 -6. Doleys DM and Olson K ed. Psychological assessment and intervention in implantable pain therapies. Minneapolis: Medtronic Inc. , 1996: 1 -20. Follett K and Doleys D. Selection of Candidates for Intrathecal Drug Administration to Treat Chronic Pain: Considerations in Pre-implantation Trials. Minneapolis: Medtronic Inc. , 2002: 1 - 19. 27
© Medtronic, Inc. 2009 References • • • Coffey RJ, Owens, M, Broste, Steven K, et al. Mortality Associated with Intrathecal Opioid Drug Infusion to Treat Non-Cancer Pain: Risk Factors and Mitigation. Late breaking paper presented at: North American Neuromodulation Society 12 th Annual Meeting; December 4 -7; 2008; Las Vegas, NV. de Lissovoy G, Brown RE, Halpern M et al. Cost Effectiveness of Long-term Intrathecal Morphine for Pain Associated with Failed Back Surgery Syndrome. Clinical Therapeutics 1997; 19(1): 96– 112. Deer T, Chapple I, Classen A, et al. Intrathecal drug delivery for treatment of chronic low back pain: report from the National Outcomes Registry for low back pain. Pain Med 2004; 5(1): 6 -13. Doleys DM. Psychological factors in spinal cord stimulation therapy: brief review and discussion. Neurosurgical Focus 2006: 21(6): 1 -6. Doleys DM and Olson K ed. Psychological assessment and intervention in implantable pain therapies. Minneapolis: Medtronic Inc. , 1996: 1 -20. Follett K and Doleys D. Selection of Candidates for Intrathecal Drug Administration to Treat Chronic Pain: Considerations in Pre-implantation Trials. Minneapolis: Medtronic Inc. , 2002: 1 - 19. 27
 © Medtronic, Inc. 2009 References continued • • Kumar K, Hunter G, Demaria D. Spinal cord stimulation in treatment of chronic benign pain: challenges in treatment planning and present status, a 22 -year experience. Neurosurgery. 2006; 58(3): 481 -496. Loeser JD and Melack R. Pain: an overview. The Lancet 1999; 353(9164): 1607 -1609. North RB, Kidd DH, Farrokhi F, Piantadosi SA. Spinal cord stimulation versus repeated lumbosacral spine surgery for chronic pain: a randomized, controlled trial. Neurosurgery. 2005; 56(1): 98 -107. Medtronic Implantable Systems Performance Registry (ISPR). 2007 Annual Report. Protocol NSP 0010 -10000. Report date: October 2, 2008. Roberts LJ, Finch PM, Goucke CR, Price LM. Outcome of intrathecal opioids in chronic non-cancer pain. Eur J Pain. 2001; 5(4): 353 -361. Smith T. J. and Coyne P. J. How to Use Implantable Drug Delivery Systems for Refractory Cancer Pain. J Supportive Oncology 1: 1: 1 -4. Taylor R, Caraway D, Van Buyten JP et al. eds. Spinal Cord Stimulation: Overcoming Barriers to Successful Outcomes for the Complex Pain Patient. Minneapolis: Medtronic Inc. , 2006: 1 -10. Van Buyten JP, Van Zundert J, Vueghs P, Vanduffel L. Efficacy of spinal cord stimulation: 10 years of experience in a pain centre in Belgium. Eur J Pain. 2001; 5(3): 299 -307. 28
© Medtronic, Inc. 2009 References continued • • Kumar K, Hunter G, Demaria D. Spinal cord stimulation in treatment of chronic benign pain: challenges in treatment planning and present status, a 22 -year experience. Neurosurgery. 2006; 58(3): 481 -496. Loeser JD and Melack R. Pain: an overview. The Lancet 1999; 353(9164): 1607 -1609. North RB, Kidd DH, Farrokhi F, Piantadosi SA. Spinal cord stimulation versus repeated lumbosacral spine surgery for chronic pain: a randomized, controlled trial. Neurosurgery. 2005; 56(1): 98 -107. Medtronic Implantable Systems Performance Registry (ISPR). 2007 Annual Report. Protocol NSP 0010 -10000. Report date: October 2, 2008. Roberts LJ, Finch PM, Goucke CR, Price LM. Outcome of intrathecal opioids in chronic non-cancer pain. Eur J Pain. 2001; 5(4): 353 -361. Smith T. J. and Coyne P. J. How to Use Implantable Drug Delivery Systems for Refractory Cancer Pain. J Supportive Oncology 1: 1: 1 -4. Taylor R, Caraway D, Van Buyten JP et al. eds. Spinal Cord Stimulation: Overcoming Barriers to Successful Outcomes for the Complex Pain Patient. Minneapolis: Medtronic Inc. , 2006: 1 -10. Van Buyten JP, Van Zundert J, Vueghs P, Vanduffel L. Efficacy of spinal cord stimulation: 10 years of experience in a pain centre in Belgium. Eur J Pain. 2001; 5(3): 299 -307. 28
 © Medtronic, Inc. 2009 Neurostimulation Systems for Pain Therapy Brief Summary (continued) Contraindications Diathermy - Do not use shortwave diathermy, microwave or therapeutic ultrasound diathermy (all now referred to as diathermy) on patients implanted with a neurostimulation system. Energy from diathermy can be transferred through the implanted system and cause tissue damage at the locations of the implanted electrodes, resulting in severe injury or death. Warnings Sources of strong electromagnetic interference (eg, defibrillation, diathermy, electrocautery, MRI, RF ablation, and therapeutic ultrasound) can interact with the neurostimulation system, resulting in serious patient injury or death. These and other sources of EMI can also result in system damage, operational changes to the neurostimulator or unexpected changes in stimulation. Rupture or piercing of the neurostimulator can result in severe burns. An implanted cardiac device (eg, pacemaker, defibrillator) may damage a neurostimulator, and the electrical pulses from the neurostimulator may result in an inappropriate response of the cardiac device. Precautions The safety and effectiveness of this therapy has not been established for pediatric use (patients under the age of 18), pregnancy, unborn fetus, or delivery. Patients should be detoxified from narcotics prior to lead placement. Clinicians and patients should follow programming guidelines and precautions provided in product manuals. Patients should avoid activities that may put undue stress on the implanted neurostimulation system components. Patients should not scuba dive below 10 meters of water or enter hyperbaric chambers above 2. 0 atmosphere absolute (ATA). Electromagnetic interference, postural changes, and other activities may cause shocking or jolting.
© Medtronic, Inc. 2009 Neurostimulation Systems for Pain Therapy Brief Summary (continued) Contraindications Diathermy - Do not use shortwave diathermy, microwave or therapeutic ultrasound diathermy (all now referred to as diathermy) on patients implanted with a neurostimulation system. Energy from diathermy can be transferred through the implanted system and cause tissue damage at the locations of the implanted electrodes, resulting in severe injury or death. Warnings Sources of strong electromagnetic interference (eg, defibrillation, diathermy, electrocautery, MRI, RF ablation, and therapeutic ultrasound) can interact with the neurostimulation system, resulting in serious patient injury or death. These and other sources of EMI can also result in system damage, operational changes to the neurostimulator or unexpected changes in stimulation. Rupture or piercing of the neurostimulator can result in severe burns. An implanted cardiac device (eg, pacemaker, defibrillator) may damage a neurostimulator, and the electrical pulses from the neurostimulator may result in an inappropriate response of the cardiac device. Precautions The safety and effectiveness of this therapy has not been established for pediatric use (patients under the age of 18), pregnancy, unborn fetus, or delivery. Patients should be detoxified from narcotics prior to lead placement. Clinicians and patients should follow programming guidelines and precautions provided in product manuals. Patients should avoid activities that may put undue stress on the implanted neurostimulation system components. Patients should not scuba dive below 10 meters of water or enter hyperbaric chambers above 2. 0 atmosphere absolute (ATA). Electromagnetic interference, postural changes, and other activities may cause shocking or jolting.
 Neurostimulation Systems for Pain Therapy Brief Summary © Medtronic, Inc. 2009 Product manuals must be reviewed prior to use for detailed disclosure. Indications Implantable neurostimulation systems - A Medtronic implantable neurostimulation system is indicated for spinal cord stimulation (SCS) as an aid in the management of chronic, intractable pain of the trunk and/or limbs-including unilateral or bilateral pain associated with the following conditions: > Failed Back Syndrome (FBS) or low back syndrome or failed back > Radicular pain syndrome or radiculopathies resulting in pain secondary to FBS or herniated disk > Postlaminectomy pain > Multiple back operations > Unsuccessful disk surgery > Degenerative Disk Disease (DDD)/herniated disk pain refractory to conservative and surgical interventions > Peripheral causalgia > Epidural fibrosis > Arachnoiditis or lumbar adhesive arachnoiditis > Complex Regional Pain Syndrome (CRPS), Reflex Sympathetic Dystrophy (RSD), or causalgia
Neurostimulation Systems for Pain Therapy Brief Summary © Medtronic, Inc. 2009 Product manuals must be reviewed prior to use for detailed disclosure. Indications Implantable neurostimulation systems - A Medtronic implantable neurostimulation system is indicated for spinal cord stimulation (SCS) as an aid in the management of chronic, intractable pain of the trunk and/or limbs-including unilateral or bilateral pain associated with the following conditions: > Failed Back Syndrome (FBS) or low back syndrome or failed back > Radicular pain syndrome or radiculopathies resulting in pain secondary to FBS or herniated disk > Postlaminectomy pain > Multiple back operations > Unsuccessful disk surgery > Degenerative Disk Disease (DDD)/herniated disk pain refractory to conservative and surgical interventions > Peripheral causalgia > Epidural fibrosis > Arachnoiditis or lumbar adhesive arachnoiditis > Complex Regional Pain Syndrome (CRPS), Reflex Sympathetic Dystrophy (RSD), or causalgia
 © Medtronic, Inc. 2009 Neurostimulation Systems for Pain Therapy Brief Summary (continued) Adverse Events Adverse events may include: undesirable change in stimulation described by some patients as uncomfortable, jolting or shocking; hematoma, epidural hemorrhage, paralysis, seroma, CSF leakage, infection, erosion, allergic response, hardware malfunction or migration, pain at implant site, loss of pain relief, chest wall stimulation, and surgical risks. For further information, please call Medtronic at 1 -800 -328 -0810 and/or consult Medtronic’s website at www. medtronic. com. USA Rx Only Rev 0209
© Medtronic, Inc. 2009 Neurostimulation Systems for Pain Therapy Brief Summary (continued) Adverse Events Adverse events may include: undesirable change in stimulation described by some patients as uncomfortable, jolting or shocking; hematoma, epidural hemorrhage, paralysis, seroma, CSF leakage, infection, erosion, allergic response, hardware malfunction or migration, pain at implant site, loss of pain relief, chest wall stimulation, and surgical risks. For further information, please call Medtronic at 1 -800 -328 -0810 and/or consult Medtronic’s website at www. medtronic. com. USA Rx Only Rev 0209
 © Medtronic, Inc. 2009 Synchro. Med® II Drug Infusion System Brief Summary: Product technical manuals and the appropriate drug labeling must be reviewed prior to use for detailed disclosure. Indications: US: Chronic intraspinal (epidural and intrathecal) infusion of preservative-free morphine sulfate sterile solution in the treatment of chronic intractable pain, chronic intrathecal infusion of preservative-free ziconotide sterile solution for the management of severe chronic pain, and chronic intrathecal infusion of Lioresal® Intrathecal (baclofen injection) for the management of severe spasticity; chronic intravascular infusion of floxuridine (FUDR) or methotrexate for the treatment of primary or metastatic cancer. Outside of US: Chronic infusion of drugs or fluids tested as compatible and listed in the product labeling. Contraindications: Infection; implant depth greater than 2. 5 cm below skin; insufficient body size; spinal anomalies; drugs with preservatives, drug contraindications, drug formulations with p. H < 3, use of catheter access port (CAP) kit for refills or of refill kit for catheter access, blood sampling through CAP in vascular applications, use of Personal Therapy Manager to administer opioid to opioid-naïve patients or to administer ziconotide.
© Medtronic, Inc. 2009 Synchro. Med® II Drug Infusion System Brief Summary: Product technical manuals and the appropriate drug labeling must be reviewed prior to use for detailed disclosure. Indications: US: Chronic intraspinal (epidural and intrathecal) infusion of preservative-free morphine sulfate sterile solution in the treatment of chronic intractable pain, chronic intrathecal infusion of preservative-free ziconotide sterile solution for the management of severe chronic pain, and chronic intrathecal infusion of Lioresal® Intrathecal (baclofen injection) for the management of severe spasticity; chronic intravascular infusion of floxuridine (FUDR) or methotrexate for the treatment of primary or metastatic cancer. Outside of US: Chronic infusion of drugs or fluids tested as compatible and listed in the product labeling. Contraindications: Infection; implant depth greater than 2. 5 cm below skin; insufficient body size; spinal anomalies; drugs with preservatives, drug contraindications, drug formulations with p. H < 3, use of catheter access port (CAP) kit for refills or of refill kit for catheter access, blood sampling through CAP in vascular applications, use of Personal Therapy Manager to administer opioid to opioid-naïve patients or to administer ziconotide.
 © Medtronic, Inc. 2009 Synchro. Med® II Drug Infusion System Brief Summary: (continued) Warnings: Non-indicated formulations may contain neurotoxic preservatives, antimicrobials, or antioxidants, or may be incompatible with and damage the system. Failure to comply with all product instructions, including use of drugs or fluids not indicated for use with system, or of questionable sterility or quality, or use of non-Medtronic components or inappropriate kits, can result in improper use, technical errors, increased risks to patient, tissue damage, damage to the system requiring revision or replacement, and/or change in therapy, and may result in additional surgical procedures, a return of underlying symptoms, and/or a clinically significant or fatal drug under- or overdose. Refer to appropriate drug labeling for indications, contraindications, warnings, precautions, dosage and administration information, screening procedures and underdose and overdose symptoms and methods of management. Physicians must be familiar with the drug stability information in the product technical manuals and must understand the dose relationship to drug concentration and pump flow rate before prescribing pump infusion. Implantation and ongoing system management must be performed by individuals trained in the operation and handling of the infusion system. An inflammatory mass that can result in serious neurological impairment, including paralysis, may occur at the tip of the implanted catheter. Clinicians should monitor patients on intraspinal therapy carefully for any new neurological signs or symptoms, change in underlying symptoms, or need for rapid dose escalation.
© Medtronic, Inc. 2009 Synchro. Med® II Drug Infusion System Brief Summary: (continued) Warnings: Non-indicated formulations may contain neurotoxic preservatives, antimicrobials, or antioxidants, or may be incompatible with and damage the system. Failure to comply with all product instructions, including use of drugs or fluids not indicated for use with system, or of questionable sterility or quality, or use of non-Medtronic components or inappropriate kits, can result in improper use, technical errors, increased risks to patient, tissue damage, damage to the system requiring revision or replacement, and/or change in therapy, and may result in additional surgical procedures, a return of underlying symptoms, and/or a clinically significant or fatal drug under- or overdose. Refer to appropriate drug labeling for indications, contraindications, warnings, precautions, dosage and administration information, screening procedures and underdose and overdose symptoms and methods of management. Physicians must be familiar with the drug stability information in the product technical manuals and must understand the dose relationship to drug concentration and pump flow rate before prescribing pump infusion. Implantation and ongoing system management must be performed by individuals trained in the operation and handling of the infusion system. An inflammatory mass that can result in serious neurological impairment, including paralysis, may occur at the tip of the implanted catheter. Clinicians should monitor patients on intraspinal therapy carefully for any new neurological signs or symptoms, change in underlying symptoms, or need for rapid dose escalation.
 © Medtronic, Inc. 2009 Synchro. Med® II Drug Infusion System Brief Summary: (continued) Inform patients of the signs and symptoms of drug under- or overdose, appropriate drug warnings and precautions regarding drug interactions, potential side effects, and signs and symptoms that require medical attention, including prodromal signs and symptoms of inflammatory mass. Failure to recognize signs and symptoms and seek appropriate medical intervention can result in serious injury or death. Instruct patients to notify their healthcare professionals of the implanted pump before medical tests/procedures, to return for refills at prescribed times, to carry their Medtronic device identification card, to avoid manipulating the pump through the skin, to consult with their clinician if the pump alarms and before traveling or engaging in activities that can stress the infusion system or involve pressure or temperature changes. Strong sources of electromagnetic interference (EMI), such as short wave (RF) diathermy and MRI, can negatively interact with the pump and cause heating of the implanted pump, system damage, or changes in pump operation or flow rate, that can result in patient injury from tissue heating, additional surgical procedures, a return of underlying symptoms, and/or a clinically significant or fatal drug underdose or overdose. Avoid using shortwave (RF) diathermy within 30 cm of the pump or catheter. Effects of other types of diathermy (microwave, ultrasonic, etc. ) on the pump are unknown. Drug infusion is suspended during MRI; for patients who can not safely tolerate suspension, use alternative drug delivery method during MRI. Patients receiving intrathecal baclofen therapy are at higher risk for adverse events, as baclofen withdrawal can lead to a life threatening condition if not treated promptly and effectively. Confirm pump status before and after MRI. Reference product labeling for information on sources of EMI, effects on patient and system, and steps to reduce risks from EMI.
© Medtronic, Inc. 2009 Synchro. Med® II Drug Infusion System Brief Summary: (continued) Inform patients of the signs and symptoms of drug under- or overdose, appropriate drug warnings and precautions regarding drug interactions, potential side effects, and signs and symptoms that require medical attention, including prodromal signs and symptoms of inflammatory mass. Failure to recognize signs and symptoms and seek appropriate medical intervention can result in serious injury or death. Instruct patients to notify their healthcare professionals of the implanted pump before medical tests/procedures, to return for refills at prescribed times, to carry their Medtronic device identification card, to avoid manipulating the pump through the skin, to consult with their clinician if the pump alarms and before traveling or engaging in activities that can stress the infusion system or involve pressure or temperature changes. Strong sources of electromagnetic interference (EMI), such as short wave (RF) diathermy and MRI, can negatively interact with the pump and cause heating of the implanted pump, system damage, or changes in pump operation or flow rate, that can result in patient injury from tissue heating, additional surgical procedures, a return of underlying symptoms, and/or a clinically significant or fatal drug underdose or overdose. Avoid using shortwave (RF) diathermy within 30 cm of the pump or catheter. Effects of other types of diathermy (microwave, ultrasonic, etc. ) on the pump are unknown. Drug infusion is suspended during MRI; for patients who can not safely tolerate suspension, use alternative drug delivery method during MRI. Patients receiving intrathecal baclofen therapy are at higher risk for adverse events, as baclofen withdrawal can lead to a life threatening condition if not treated promptly and effectively. Confirm pump status before and after MRI. Reference product labeling for information on sources of EMI, effects on patient and system, and steps to reduce risks from EMI.
 © Medtronic, Inc. 2009 Synchro. Med® II Drug Infusion System Brief Summary: (continued) Precautions: Monitor patients after device or catheter replacement for signs of underdose/overdose. Infuse preservative-free (intraspinal) saline or, for vascular applications, infuse heparinized solutions therapy at minimum flow rate if therapy is discontinued for an extended period of time to avoid system damage. EMI may interfere with programmer telemetry during pump programming sessions. EMI from the Synchro. Med programmer may interfere with other active implanted devices (e. g. , pacemaker, defibrillator, neurostimulator). Adverse Events: Include, but are not limited to, spinal/vascular procedure risks; infection; bleeding; tissue damage, damage to the system or loss of, or change in, therapy that may result in additional surgical procedures, a return of underlying symptoms, and/or a clinically significant or fatal drug underdose or overdose, due to end of device service life, failure of the catheter, pump or other system component, pump inversion, technical/programming errors, or improper use, including use of non-indicated formulations and/or not using drugs or system in accordance with labeling; pocket seroma, hematoma, erosion, infection; post-lumbar puncture (spinal headache); CSF leak and rare central nervous system pressure-related problems; hygroma; radiculitis; arachnoiditis; spinal cord bleeding/damage; meningitis; neurological impairment (including paralysis) due to inflammatory mass; potential serious adverse effects from catheter fragments in intrathecal space, including potential to compromise antibiotic effectiveness for CSF infection; anesthesia complications; body rejection phenomena; local and systemic drug toxicity and related side effects; potential serious adverse effects from catheter placement in intravascular applications. USA Rx Only Rev 10/09
© Medtronic, Inc. 2009 Synchro. Med® II Drug Infusion System Brief Summary: (continued) Precautions: Monitor patients after device or catheter replacement for signs of underdose/overdose. Infuse preservative-free (intraspinal) saline or, for vascular applications, infuse heparinized solutions therapy at minimum flow rate if therapy is discontinued for an extended period of time to avoid system damage. EMI may interfere with programmer telemetry during pump programming sessions. EMI from the Synchro. Med programmer may interfere with other active implanted devices (e. g. , pacemaker, defibrillator, neurostimulator). Adverse Events: Include, but are not limited to, spinal/vascular procedure risks; infection; bleeding; tissue damage, damage to the system or loss of, or change in, therapy that may result in additional surgical procedures, a return of underlying symptoms, and/or a clinically significant or fatal drug underdose or overdose, due to end of device service life, failure of the catheter, pump or other system component, pump inversion, technical/programming errors, or improper use, including use of non-indicated formulations and/or not using drugs or system in accordance with labeling; pocket seroma, hematoma, erosion, infection; post-lumbar puncture (spinal headache); CSF leak and rare central nervous system pressure-related problems; hygroma; radiculitis; arachnoiditis; spinal cord bleeding/damage; meningitis; neurological impairment (including paralysis) due to inflammatory mass; potential serious adverse effects from catheter fragments in intrathecal space, including potential to compromise antibiotic effectiveness for CSF infection; anesthesia complications; body rejection phenomena; local and systemic drug toxicity and related side effects; potential serious adverse effects from catheter placement in intravascular applications. USA Rx Only Rev 10/09


