f7616f1301d50e3d40f74aa9320946f8.ppt
- Количество слайдов: 28
 Medtronic Core. Valve US Pivotal Trial: Update Jeffrey J. Popma, MD Director, Interventional Cardiology Clinical Services Beth Israel Deaconess Medical Center Associate Professor of Medicine Harvard Medical School Boston, MA
Medtronic Core. Valve US Pivotal Trial: Update Jeffrey J. Popma, MD Director, Interventional Cardiology Clinical Services Beth Israel Deaconess Medical Center Associate Professor of Medicine Harvard Medical School Boston, MA
 Jeffrey J. Popma, MD § Honoraria – Boston Scientific Corporation – Cordis Corporation § Grants/Contracted Research – Boston Scientific Corporation – Cordis Corporation – Medtronic, Inc. – Abbott Laboratories
Jeffrey J. Popma, MD § Honoraria – Boston Scientific Corporation – Cordis Corporation § Grants/Contracted Research – Boston Scientific Corporation – Cordis Corporation – Medtronic, Inc. – Abbott Laboratories
 I intend to reference off label or unapproved uses of drugs or devices in my presentation. I intend to discuss Transcatheter aortic valves that are not approved in the USA
I intend to reference off label or unapproved uses of drugs or devices in my presentation. I intend to discuss Transcatheter aortic valves that are not approved in the USA
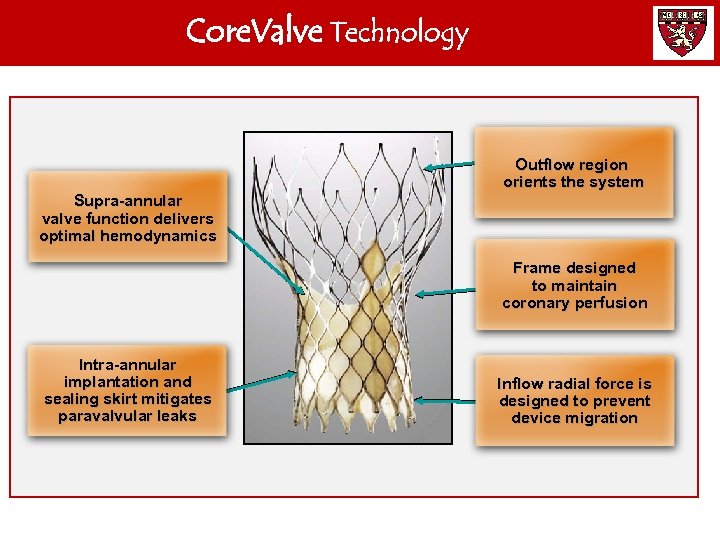 Core. Valve Technology Outflow region orients the system Supra-annular valve function delivers optimal hemodynamics Frame designed to maintain coronary perfusion Intra-annular implantation and sealing skirt mitigates paravalvular leaks Inflow radial force is designed to prevent device migration
Core. Valve Technology Outflow region orients the system Supra-annular valve function delivers optimal hemodynamics Frame designed to maintain coronary perfusion Intra-annular implantation and sealing skirt mitigates paravalvular leaks Inflow radial force is designed to prevent device migration
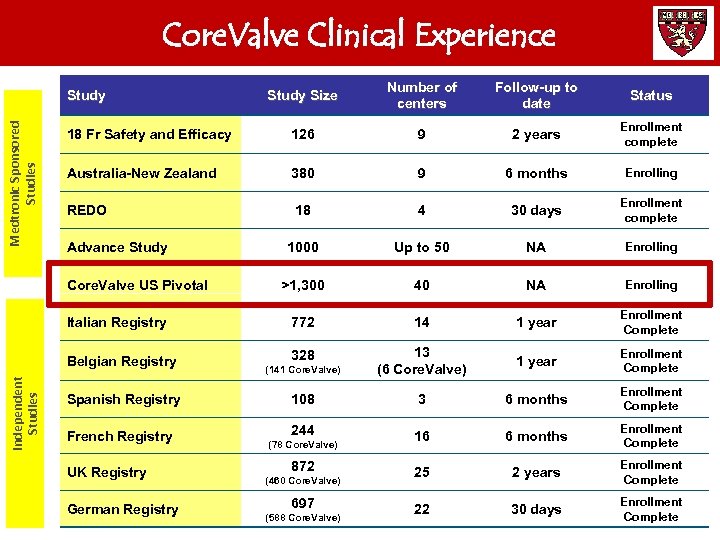 Core. Valve Clinical Experience Study Size Number of centers Follow-up to date Status 18 Fr Safety and Efficacy 126 9 2 years Enrollment complete Australia-New Zealand 380 9 6 months Enrolling REDO 18 4 30 days Enrollment complete 1000 Up to 50 NA Enrolling >1, 300 40 NA Enrolling Italian Registry 772 14 1 year Enrollment Complete Belgian Registry 328 (141 Core. Valve) 13 (6 Core. Valve) 1 year Enrollment Complete 108 3 6 months Enrollment Complete 244 16 6 months Enrollment Complete 25 2 years Enrollment Complete 22 30 days Enrollment Complete Medtronic Sponsored Studies Study Advance Study Independent Studies Core. Valve US Pivotal Spanish Registry French Registry UK Registry German Registry (78 Core. Valve) 872 (460 Core. Valve) 697 (588 Core. Valve)
Core. Valve Clinical Experience Study Size Number of centers Follow-up to date Status 18 Fr Safety and Efficacy 126 9 2 years Enrollment complete Australia-New Zealand 380 9 6 months Enrolling REDO 18 4 30 days Enrollment complete 1000 Up to 50 NA Enrolling >1, 300 40 NA Enrolling Italian Registry 772 14 1 year Enrollment Complete Belgian Registry 328 (141 Core. Valve) 13 (6 Core. Valve) 1 year Enrollment Complete 108 3 6 months Enrollment Complete 244 16 6 months Enrollment Complete 25 2 years Enrollment Complete 22 30 days Enrollment Complete Medtronic Sponsored Studies Study Advance Study Independent Studies Core. Valve US Pivotal Spanish Registry French Registry UK Registry German Registry (78 Core. Valve) 872 (460 Core. Valve) 697 (588 Core. Valve)
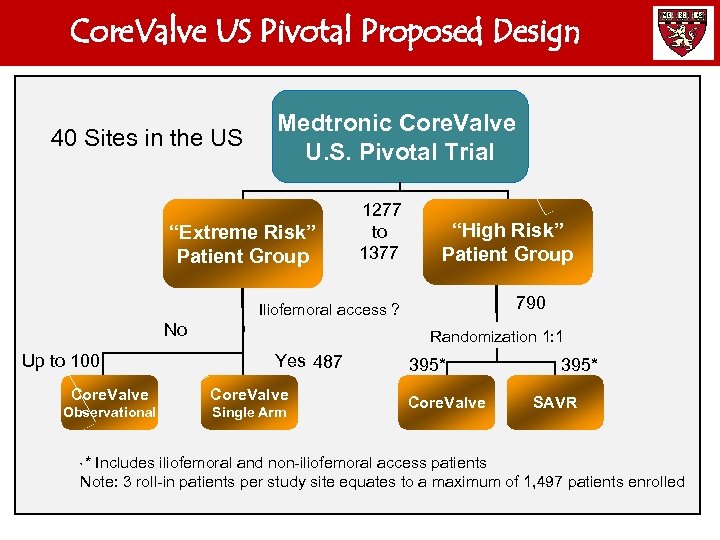 Core. Valve US Pivotal Proposed Design 40 Sites in the US Medtronic Core. Valve U. S. Pivotal Trial “Extreme Risk” Patient Group 1277 to 1377 “High Risk” Patient Group 790 Iliofemoral access ? No Up to 100 Randomization 1: 1 Yes 487 Core. Valve Observational Single Arm 395* Core. Valve 395* SAVR * Includes iliofemoral and non-iliofemoral access patients Note: 3 roll-in patients per study site equates to a maximum of 1, 497 patients enrolled *
Core. Valve US Pivotal Proposed Design 40 Sites in the US Medtronic Core. Valve U. S. Pivotal Trial “Extreme Risk” Patient Group 1277 to 1377 “High Risk” Patient Group 790 Iliofemoral access ? No Up to 100 Randomization 1: 1 Yes 487 Core. Valve Observational Single Arm 395* Core. Valve 395* SAVR * Includes iliofemoral and non-iliofemoral access patients Note: 3 roll-in patients per study site equates to a maximum of 1, 497 patients enrolled *
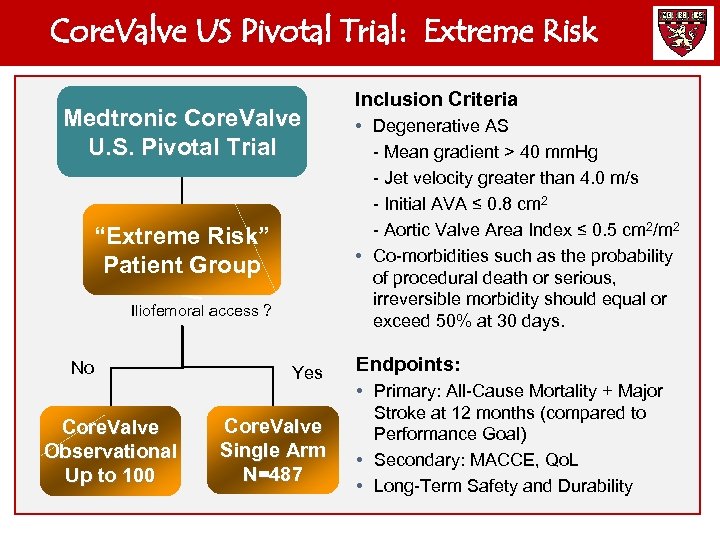 Core. Valve US Pivotal Trial: Extreme Risk Medtronic Core. Valve U. S. Pivotal Trial “Extreme Risk” Patient Group Iliofemoral access ? No Core. Valve Observational Up to 100 Yes Core. Valve Single Arm N=487 Inclusion Criteria • Degenerative AS - Mean gradient > 40 mm. Hg - Jet velocity greater than 4. 0 m/s - Initial AVA ≤ 0. 8 cm 2 - Aortic Valve Area Index ≤ 0. 5 cm 2/m 2 • Co-morbidities such as the probability of procedural death or serious, irreversible morbidity should equal or exceed 50% at 30 days. Endpoints: • Primary: All-Cause Mortality + Major Stroke at 12 months (compared to Performance Goal) • Secondary: MACCE, Qo. L • Long-Term Safety and Durability
Core. Valve US Pivotal Trial: Extreme Risk Medtronic Core. Valve U. S. Pivotal Trial “Extreme Risk” Patient Group Iliofemoral access ? No Core. Valve Observational Up to 100 Yes Core. Valve Single Arm N=487 Inclusion Criteria • Degenerative AS - Mean gradient > 40 mm. Hg - Jet velocity greater than 4. 0 m/s - Initial AVA ≤ 0. 8 cm 2 - Aortic Valve Area Index ≤ 0. 5 cm 2/m 2 • Co-morbidities such as the probability of procedural death or serious, irreversible morbidity should equal or exceed 50% at 30 days. Endpoints: • Primary: All-Cause Mortality + Major Stroke at 12 months (compared to Performance Goal) • Secondary: MACCE, Qo. L • Long-Term Safety and Durability
 Core. Valve US Pivotal Trial: High Risk Medtronic Core. Valve U. S. Pivotal Trial “High Risk” Patient Group N=790 Randomization 1: 1 Core. Valve N=395 SAVR N=395 Inclusion Criteria • Degenerative AS - Mean gradient > 40 mm. Hg - Jet velocity greater than 4. 0 m/s - Initial AVA ≤ 0. 8 cm 2 - Aortic Valve Area Index ≤ 0. 5 cm 2/m 2 • Estimated surgical mortality > 15% at 30 days Endpoints: • Primary: All-Cause Mortality at 12 months (non-inferiority) • Secondary: Pre-specified Hierarchical Testing • Long-Term Safety and Durability
Core. Valve US Pivotal Trial: High Risk Medtronic Core. Valve U. S. Pivotal Trial “High Risk” Patient Group N=790 Randomization 1: 1 Core. Valve N=395 SAVR N=395 Inclusion Criteria • Degenerative AS - Mean gradient > 40 mm. Hg - Jet velocity greater than 4. 0 m/s - Initial AVA ≤ 0. 8 cm 2 - Aortic Valve Area Index ≤ 0. 5 cm 2/m 2 • Estimated surgical mortality > 15% at 30 days Endpoints: • Primary: All-Cause Mortality at 12 months (non-inferiority) • Secondary: Pre-specified Hierarchical Testing • Long-Term Safety and Durability
 Core. Valve US Pivotal Trial: Alternative Access Extreme Risk Cohort High Risk Surgical Cohort Patients with no appropriate iliofemoral access will be evaluated for alternative access. Those who do have alternative access will be enrolled in an observational arm that will contain up to 100 patients. Those who do have alternative access will be randomized against SAVR and implanted either with SAVR or with TAVI through alternative access These patients will be part of the 790 patients enrolled in the randomized study (maximum 100 patients).
Core. Valve US Pivotal Trial: Alternative Access Extreme Risk Cohort High Risk Surgical Cohort Patients with no appropriate iliofemoral access will be evaluated for alternative access. Those who do have alternative access will be enrolled in an observational arm that will contain up to 100 patients. Those who do have alternative access will be randomized against SAVR and implanted either with SAVR or with TAVI through alternative access These patients will be part of the 790 patients enrolled in the randomized study (maximum 100 patients).
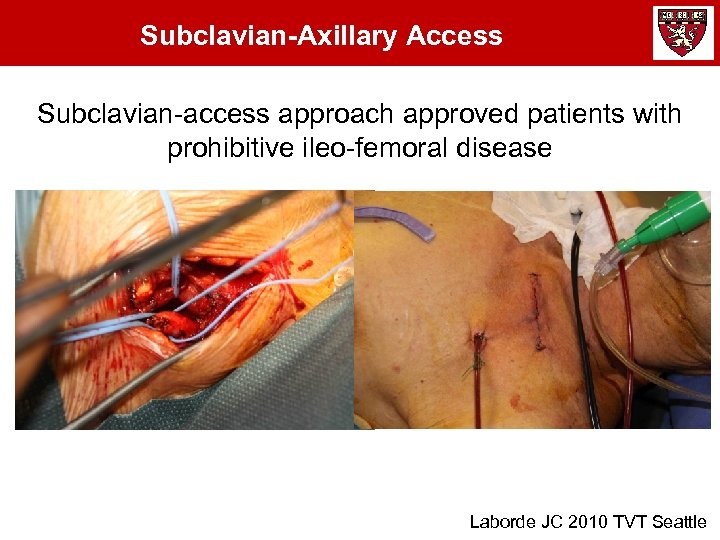 Subclavian-Axillary Access Subclavian-access approach approved patients with prohibitive ileo-femoral disease Laborde JC 2010 TVT Seattle
Subclavian-Axillary Access Subclavian-access approach approved patients with prohibitive ileo-femoral disease Laborde JC 2010 TVT Seattle
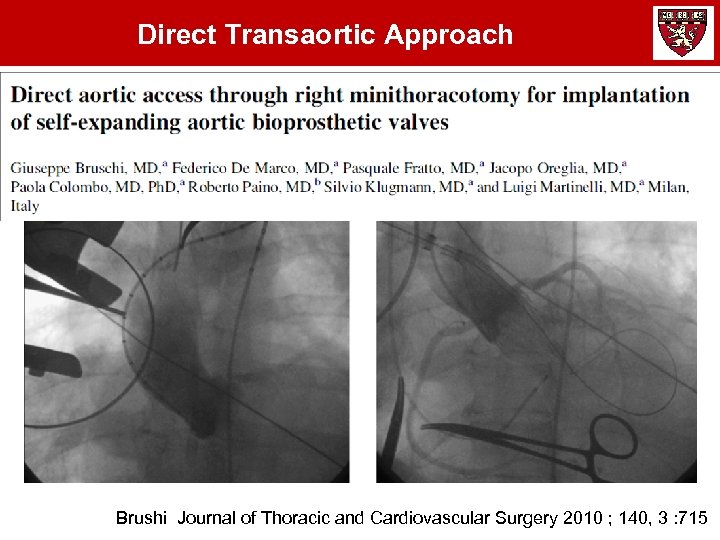 Direct Transaortic Approach Brushi Journal of Thoracic and Cardiovascular Surgery 2010 ; 140, 3 : 715
Direct Transaortic Approach Brushi Journal of Thoracic and Cardiovascular Surgery 2010 ; 140, 3 : 715
 Difference from other US TAVI trials Extreme Risk Study High Risk Study Single arm with performance Inclusion estimated surgical mortality >15% at 30 days goal. All patients enrolled are implanted with 18 F Core. Valve All-cause mortality as single All-cause mortality + major endoint stroke as single endpoint Alternative access possible (e. g. subclavian/axillary) in observational arm Alternative access (e. g. subclavian/axillary) included but no transapical.
Difference from other US TAVI trials Extreme Risk Study High Risk Study Single arm with performance Inclusion estimated surgical mortality >15% at 30 days goal. All patients enrolled are implanted with 18 F Core. Valve All-cause mortality as single All-cause mortality + major endoint stroke as single endpoint Alternative access possible (e. g. subclavian/axillary) in observational arm Alternative access (e. g. subclavian/axillary) included but no transapical.
 Core. Valve US Pivotal Trial: Current Status 40 Sites Trained Roll-ins Ongoing 16 Site Extreme Risk Registry “High-Risk” RCT We are on our way. . .
Core. Valve US Pivotal Trial: Current Status 40 Sites Trained Roll-ins Ongoing 16 Site Extreme Risk Registry “High-Risk” RCT We are on our way. . .
 Interdisciplinary Committees • Screening Committee – National PIs and designated investigators (voting) as well as non voting Core. Valve proctors and analysts – Detailed review patient screening and imaging data to determine patient eligibility – Establish and maintain consistency in the designation of “Extreme Risk” and “High Risk” patients – Minimum of two surgeons and one interventionalist provide a screening committee quorum
Interdisciplinary Committees • Screening Committee – National PIs and designated investigators (voting) as well as non voting Core. Valve proctors and analysts – Detailed review patient screening and imaging data to determine patient eligibility – Establish and maintain consistency in the designation of “Extreme Risk” and “High Risk” patients – Minimum of two surgeons and one interventionalist provide a screening committee quorum
 Screening Committee Challenges • Patients required to have critical aortic stenosis that is severe enough to impact their chance of survival over the next 12 months. Low-gradient, low-output AS DSE • Patients required to have substantial (> 15%) 30 -day surgical mortality risk No minimum STS risk score; new indices for frailty • Patients required to have a good likelihood of surviving for the next year after their aortic stenosis has been corrected Exclude “mortal” co-morbidities
Screening Committee Challenges • Patients required to have critical aortic stenosis that is severe enough to impact their chance of survival over the next 12 months. Low-gradient, low-output AS DSE • Patients required to have substantial (> 15%) 30 -day surgical mortality risk No minimum STS risk score; new indices for frailty • Patients required to have a good likelihood of surviving for the next year after their aortic stenosis has been corrected Exclude “mortal” co-morbidities
 Screening Committee Considerations Clinical Data Summary • • • Clinical History STS and related Co-Morbidities Frailty Indices: Gait Speed, Grip Strength, ADLs 100% Central Imaging Lab Review (Intelemage) § § 2 D Echocardiography Coronary and Graft Angiography CTA Chest: Annular dimensions, SOV Dimensions CTA Torso: Peripheral Vessel Diameter, Calcification, Tortuousity
Screening Committee Considerations Clinical Data Summary • • • Clinical History STS and related Co-Morbidities Frailty Indices: Gait Speed, Grip Strength, ADLs 100% Central Imaging Lab Review (Intelemage) § § 2 D Echocardiography Coronary and Graft Angiography CTA Chest: Annular dimensions, SOV Dimensions CTA Torso: Peripheral Vessel Diameter, Calcification, Tortuousity
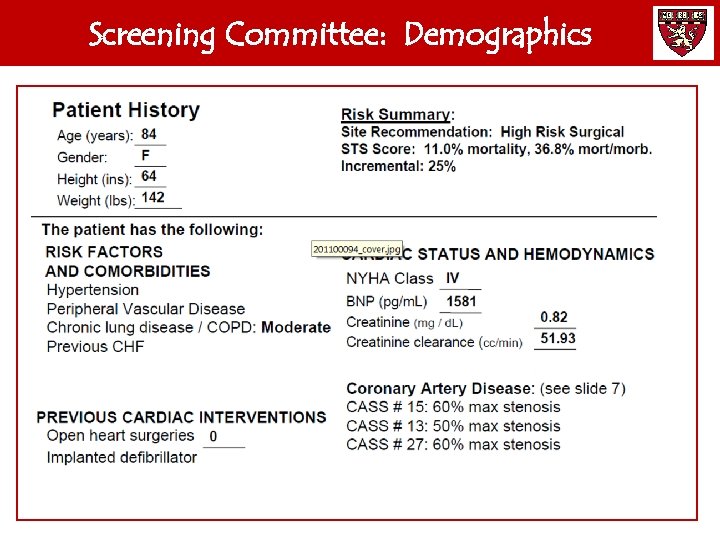 Screening Committee: Demographics
Screening Committee: Demographics
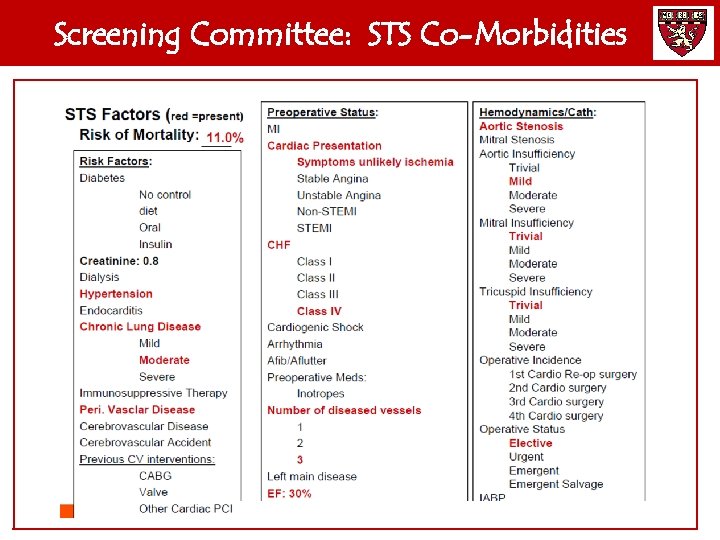 Screening Committee: STS Co-Morbidities
Screening Committee: STS Co-Morbidities
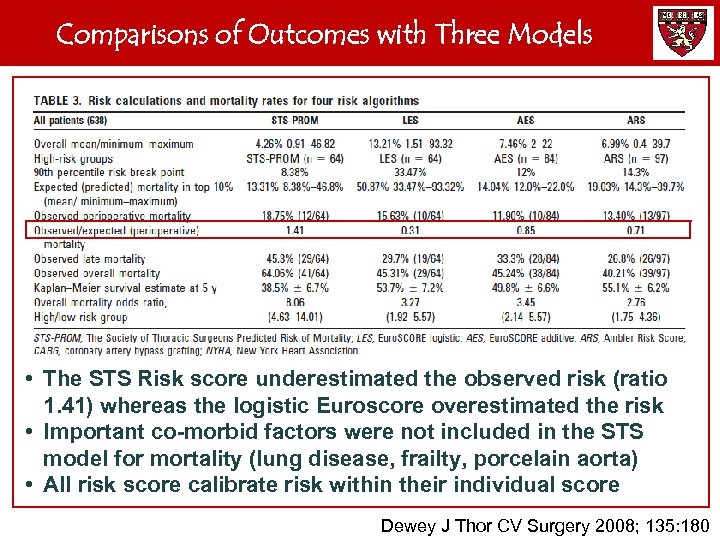 Comparisons of Outcomes with Three Models • The STS Risk score underestimated the observed risk (ratio 1. 41) whereas the logistic Euroscore overestimated the risk • Important co-morbid factors were not included in the STS model for mortality (lung disease, frailty, porcelain aorta) • All risk score calibrate risk within their individual score Dewey J Thor CV Surgery 2008; 135: 180
Comparisons of Outcomes with Three Models • The STS Risk score underestimated the observed risk (ratio 1. 41) whereas the logistic Euroscore overestimated the risk • Important co-morbid factors were not included in the STS model for mortality (lung disease, frailty, porcelain aorta) • All risk score calibrate risk within their individual score Dewey J Thor CV Surgery 2008; 135: 180
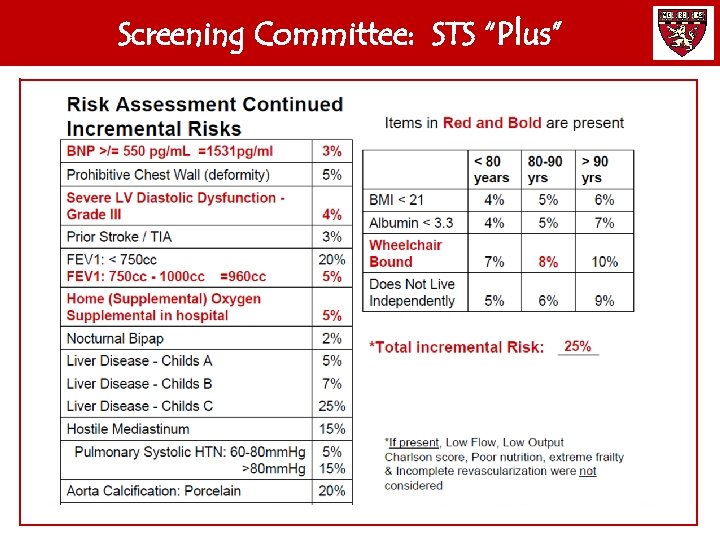 Screening Committee: STS “Plus”
Screening Committee: STS “Plus”
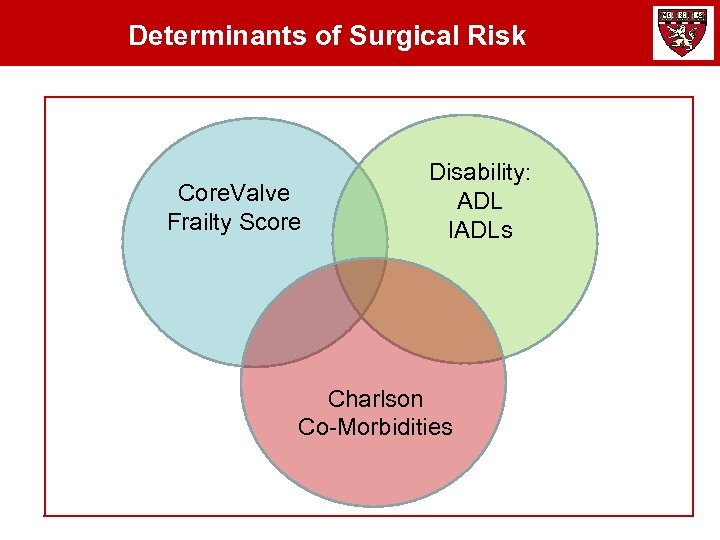 Determinants of Surgical Risk Core. Valve Frailty Score Disability: ADL IADLs Charlson Co-Morbidities
Determinants of Surgical Risk Core. Valve Frailty Score Disability: ADL IADLs Charlson Co-Morbidities
 Quantitating “Frailty” for the Core. Valve Pivotal The Core. Valve Frailty Index includes measures of nutritional assessment, strength and balance, energy level, average daily living, mental status, and medical co-mordibities. The measures will be collected prospectively to assess the patients overall frailty for this study. Nutritional Assessment Serum Albumin (≤ 3. 3 g/d. L) Hematocrit (≤ 35%) BMI (< 19) Unintentional Weight Loss Average Daily Living Bathing, Dressing, Toileting, Transferring, Continence, Feeding Charlson Co-morbidity Score Strength and Balance Wheelchair-requirement (0 = No; 1 = Yes) Grip strength adjusted for weight-BMI (kgs) 15 -foot walk test (m/s) (Get-Up and Go) Falls (≥ 1 in past 6 mos Energy Level Poor endurance - self report Low activity - kilocalorie expenditure Men (< 383 Kcals/week Women (< 270 Kcal/week)
Quantitating “Frailty” for the Core. Valve Pivotal The Core. Valve Frailty Index includes measures of nutritional assessment, strength and balance, energy level, average daily living, mental status, and medical co-mordibities. The measures will be collected prospectively to assess the patients overall frailty for this study. Nutritional Assessment Serum Albumin (≤ 3. 3 g/d. L) Hematocrit (≤ 35%) BMI (< 19) Unintentional Weight Loss Average Daily Living Bathing, Dressing, Toileting, Transferring, Continence, Feeding Charlson Co-morbidity Score Strength and Balance Wheelchair-requirement (0 = No; 1 = Yes) Grip strength adjusted for weight-BMI (kgs) 15 -foot walk test (m/s) (Get-Up and Go) Falls (≥ 1 in past 6 mos Energy Level Poor endurance - self report Low activity - kilocalorie expenditure Men (< 383 Kcals/week Women (< 270 Kcal/week)
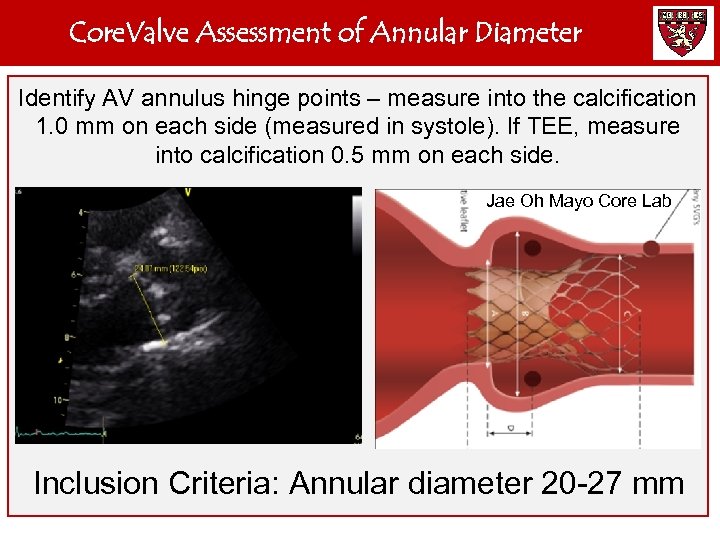 Core. Valve Assessment of Annular Diameter Identify AV annulus hinge points – measure into the calcification 1. 0 mm on each side (measured in systole). If TEE, measure into calcification 0. 5 mm on each side. Jae Oh Mayo Core Lab Inclusion Criteria: Annular diameter 20 -27 mm
Core. Valve Assessment of Annular Diameter Identify AV annulus hinge points – measure into the calcification 1. 0 mm on each side (measured in systole). If TEE, measure into calcification 0. 5 mm on each side. Jae Oh Mayo Core Lab Inclusion Criteria: Annular diameter 20 -27 mm
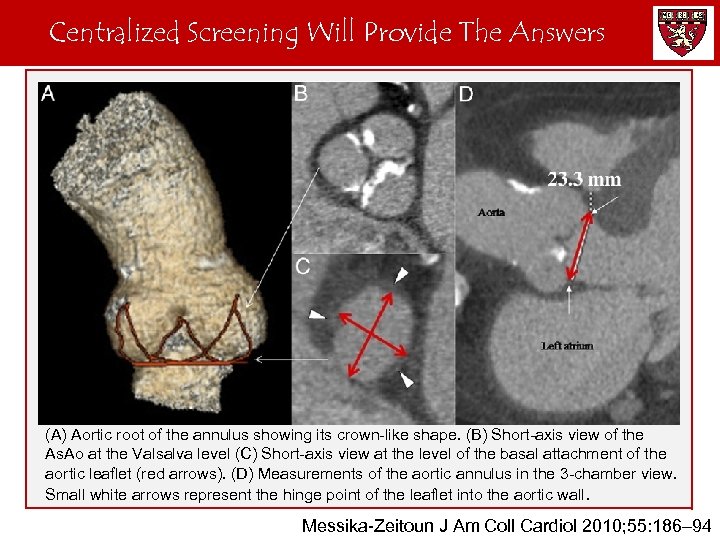 Centralized Screening Will Provide The Answers CT Measurement of the Aortic Annulus (A) Aortic root of the annulus showing its crown-like shape. (B) Short-axis view of the As. Ao at the Valsalva level (C) Short-axis view at the level of the basal attachment of the aortic leaflet (red arrows). (D) Measurements of the aortic annulus in the 3 -chamber view. Small white arrows represent the hinge point of the leaflet into the aortic wall. Messika-Zeitoun J Am Coll Cardiol 2010; 55: 186– 94
Centralized Screening Will Provide The Answers CT Measurement of the Aortic Annulus (A) Aortic root of the annulus showing its crown-like shape. (B) Short-axis view of the As. Ao at the Valsalva level (C) Short-axis view at the level of the basal attachment of the aortic leaflet (red arrows). (D) Measurements of the aortic annulus in the 3 -chamber view. Small white arrows represent the hinge point of the leaflet into the aortic wall. Messika-Zeitoun J Am Coll Cardiol 2010; 55: 186– 94
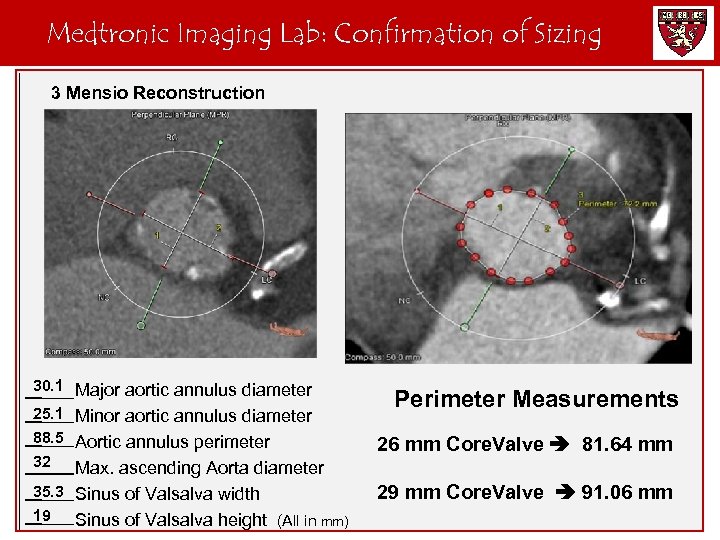 Medtronic Imaging Lab: Confirmation of Sizing 3 Mensio Reconstruction 30. 1 Major aortic annulus diameter 25. 1 Minor aortic annulus diameter 88. 5 Aortic annulus perimeter 32 Max. ascending Aorta diameter 35. 3 Sinus of Valsalva width 19 Sinus of Valsalva height (All in mm) Perimeter Measurements 26 mm Core. Valve 81. 64 mm 29 mm Core. Valve 91. 06 mm
Medtronic Imaging Lab: Confirmation of Sizing 3 Mensio Reconstruction 30. 1 Major aortic annulus diameter 25. 1 Minor aortic annulus diameter 88. 5 Aortic annulus perimeter 32 Max. ascending Aorta diameter 35. 3 Sinus of Valsalva width 19 Sinus of Valsalva height (All in mm) Perimeter Measurements 26 mm Core. Valve 81. 64 mm 29 mm Core. Valve 91. 06 mm
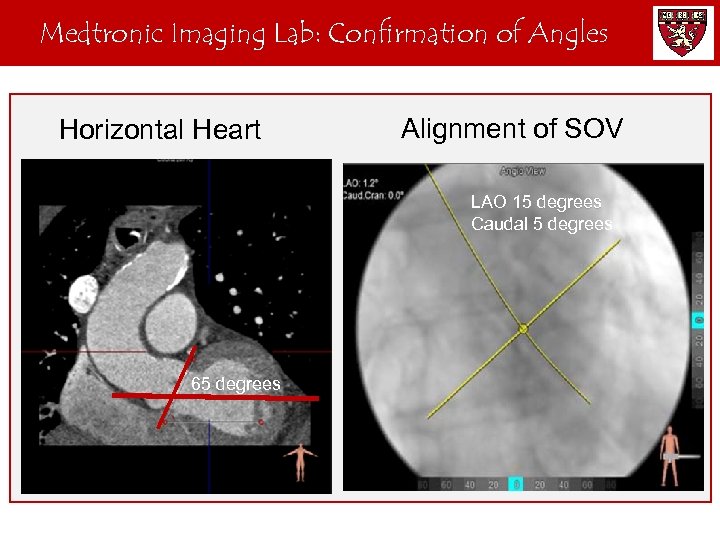 Medtronic Imaging Lab: Confirmation of Angles Horizontal Heart Alignment of SOV LAO 15 degrees Caudal 5 degrees 65 degrees
Medtronic Imaging Lab: Confirmation of Angles Horizontal Heart Alignment of SOV LAO 15 degrees Caudal 5 degrees 65 degrees
 US Pivotal Trial: Summary • Progressive activation of clinical sites • Meticulous oversight by Screening Committee to assess: - Patients co-morbidities and surgical risk - Echocardiographic-CTA analysis in all patients to confirmed anatomy - Surgeon qualification § “High-Risk” RCT will likely have lower STS scores but comparable co-mordidities for surgery § “Extreme Risk” analysis v. PG
US Pivotal Trial: Summary • Progressive activation of clinical sites • Meticulous oversight by Screening Committee to assess: - Patients co-morbidities and surgical risk - Echocardiographic-CTA analysis in all patients to confirmed anatomy - Surgeon qualification § “High-Risk” RCT will likely have lower STS scores but comparable co-mordidities for surgery § “Extreme Risk” analysis v. PG


