991dd6318c42f3daf99879fd4c6c9857.ppt
- Количество слайдов: 49
 Medical University of Sofia, Faculty of Me Department of Pharmacology and Toxic An(a)esthetics © Assoc. Prof. Iv. Lambev
Medical University of Sofia, Faculty of Me Department of Pharmacology and Toxic An(a)esthetics © Assoc. Prof. Iv. Lambev
 General anesthetics (GAs) History General anesthesia was introduced into clinical practice in the 19 th centu with the use of volatile liquids as diethyl ether and chloroform. Cardiac and hepatic toxicity limited the usefulness of chloroform (out of
General anesthetics (GAs) History General anesthesia was introduced into clinical practice in the 19 th centu with the use of volatile liquids as diethyl ether and chloroform. Cardiac and hepatic toxicity limited the usefulness of chloroform (out of
 William Morton (Boston, 1846) used ether successfully to extract a tooth. Pirogoff (Russia, 1847) Simpson (Glasgow, 1847) used chloroform in obstetrics. Queen Victoria gave birth to her children under chloroform anesthesia.
William Morton (Boston, 1846) used ether successfully to extract a tooth. Pirogoff (Russia, 1847) Simpson (Glasgow, 1847) used chloroform in obstetrics. Queen Victoria gave birth to her children under chloroform anesthesia.
 W. Morton (1846) 4
W. Morton (1846) 4
 General anesthesia caus • loss of consciousness • analgesia • amnesia • muscle relaxation (expressed in different extent) • loss of homeostatic control of respiration and cardiovascular function
General anesthesia caus • loss of consciousness • analgesia • amnesia • muscle relaxation (expressed in different extent) • loss of homeostatic control of respiration and cardiovascular function
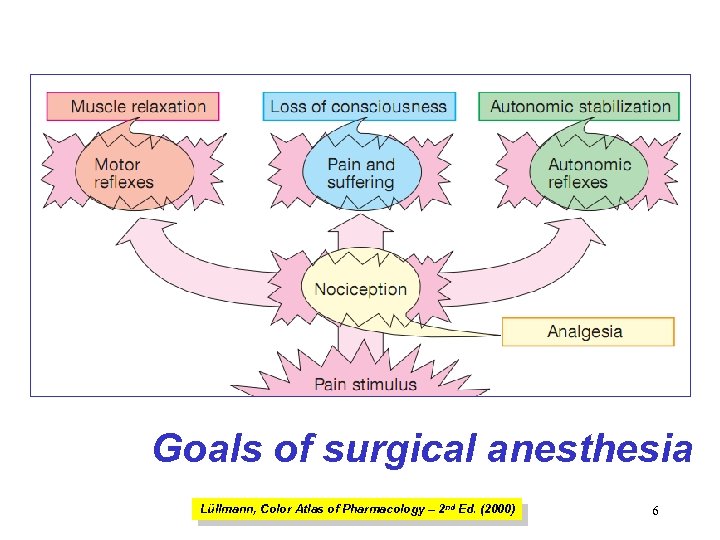 Goals of surgical anesthesia Lüllmann, Color Atlas of Pharmacology – 2 nd Ed. (2000) 6
Goals of surgical anesthesia Lüllmann, Color Atlas of Pharmacology – 2 nd Ed. (2000) 6
 The mode of action of GAs is still debated. • All GAs act on the mid-brain reticula activating system and cerebral cor to produce complete but reversible loss of consciousness. • The principle site of their action is probably the neuronal lipid membrane or hydrophobic domains of membrane proteins.
The mode of action of GAs is still debated. • All GAs act on the mid-brain reticula activating system and cerebral cor to produce complete but reversible loss of consciousness. • The principle site of their action is probably the neuronal lipid membrane or hydrophobic domains of membrane proteins.
 According to the new olysynaptolytic theory general anesthetics inhibit reversible neurotransmission in many synapses of CNS.
According to the new olysynaptolytic theory general anesthetics inhibit reversible neurotransmission in many synapses of CNS.
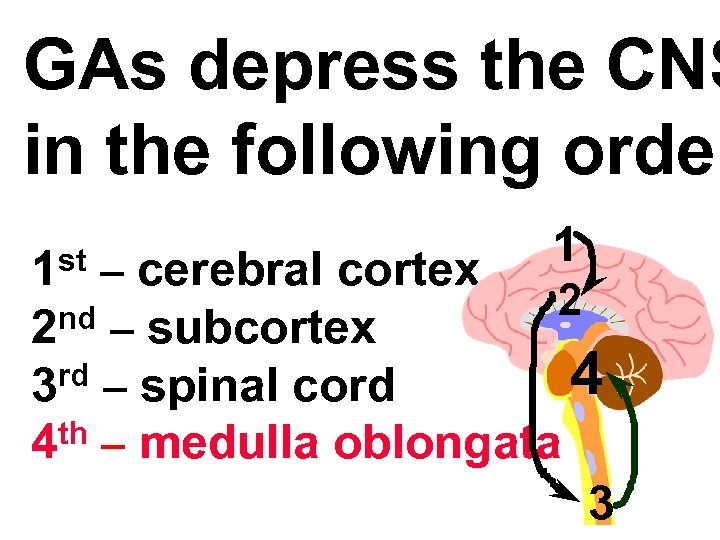 GAs depress the CNS in the following order st 1 1 – cerebral cortex 2 2 nd – subcortex rd – spinal cord 4 3 th – medulla oblongata 4 3
GAs depress the CNS in the following order st 1 1 – cerebral cortex 2 2 nd – subcortex rd – spinal cord 4 3 th – medulla oblongata 4 3
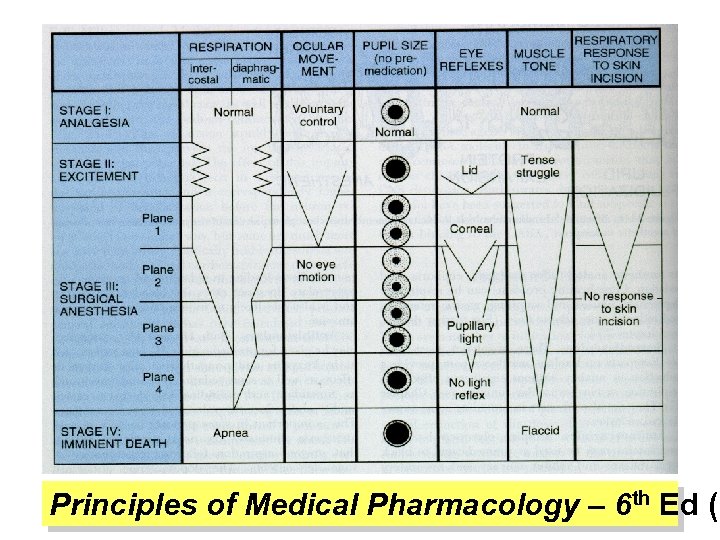 Principles of Medical Pharmacology – 6 th Ed (1
Principles of Medical Pharmacology – 6 th Ed (1
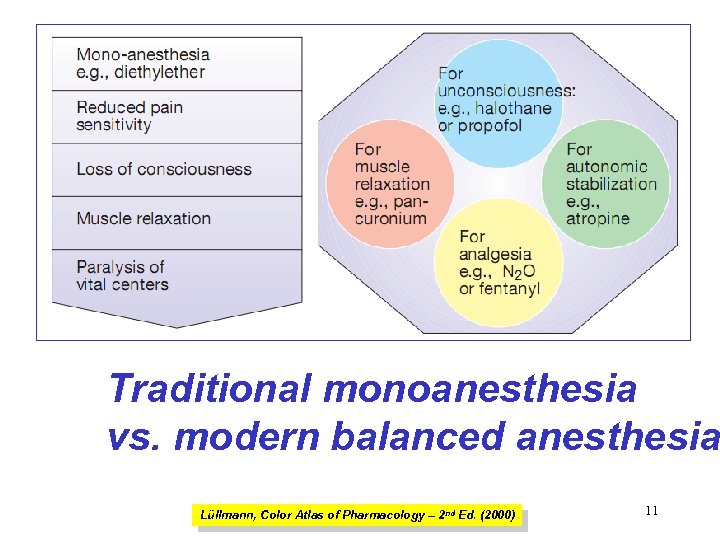 Traditional monoanesthesia vs. modern balanced anesthesia Lüllmann, Color Atlas of Pharmacology – 2 nd Ed. (2000) 11
Traditional monoanesthesia vs. modern balanced anesthesia Lüllmann, Color Atlas of Pharmacology – 2 nd Ed. (2000) 11
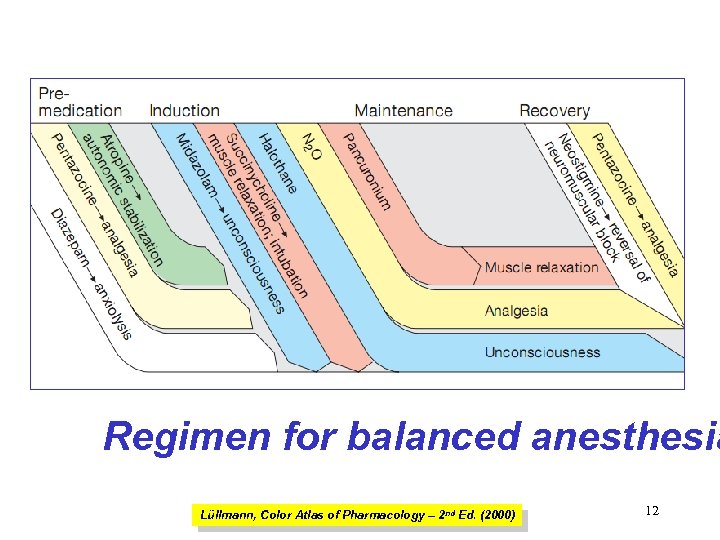 Regimen for balanced anesthesia Lüllmann, Color Atlas of Pharmacology – 2 nd Ed. (2000) 12
Regimen for balanced anesthesia Lüllmann, Color Atlas of Pharmacology – 2 nd Ed. (2000) 12
 Pharmacokinetic aspect For practical purposes GAs can b regarded physicochemicaly as id gases: their solubility in different media can be expressed as partit coefficients (PC), defined as the ratio of the concentration of the agent in two phases at equilibrium
Pharmacokinetic aspect For practical purposes GAs can b regarded physicochemicaly as id gases: their solubility in different media can be expressed as partit coefficients (PC), defined as the ratio of the concentration of the agent in two phases at equilibrium
 • The minimum alveolar concentration (MAC) indicates the minimum concentration of an anesthetic required to prevent reflex response to surgical stimulation.
• The minimum alveolar concentration (MAC) indicates the minimum concentration of an anesthetic required to prevent reflex response to surgical stimulation.
 GENERAL ANAESTHETIC 1. Inhalational GAs • Volatile liquids Desflurane Isoflurane halogenated Enflurane Halothane anesthetics Sevoflurane Diethyl ether (Aether anaestheticus – out of date • Gases Nitrous oxide
GENERAL ANAESTHETIC 1. Inhalational GAs • Volatile liquids Desflurane Isoflurane halogenated Enflurane Halothane anesthetics Sevoflurane Diethyl ether (Aether anaestheticus – out of date • Gases Nitrous oxide
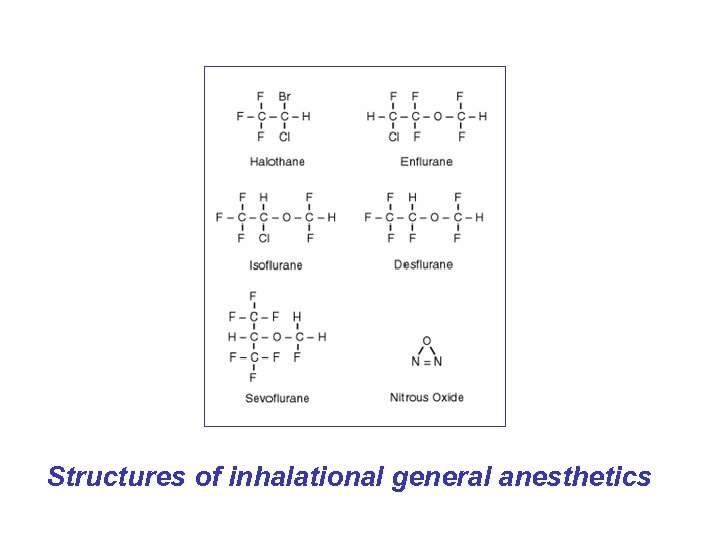 Structures of inhalational general anesthetics
Structures of inhalational general anesthetics
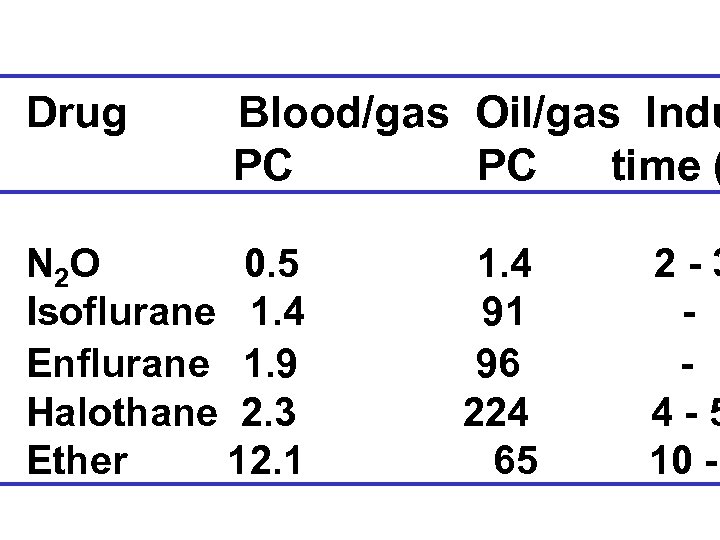 Drug Blood/gas Oil/gas Indu PC PC time ( N 2 O 0. 5 Isoflurane 1. 4 Enflurane 1. 9 Halothane 2. 3 Ether 12. 1 1. 4 91 96 224 65 2 -3 4 -5 10 -
Drug Blood/gas Oil/gas Indu PC PC time ( N 2 O 0. 5 Isoflurane 1. 4 Enflurane 1. 9 Halothane 2. 3 Ether 12. 1 1. 4 91 96 224 65 2 -3 4 -5 10 -
 Drug MAC Metabo(%) lism (%) N 2 O >100 0 Isoflurane 1. 2 0. 2 Enflurane 1. 7 2 -10 Halothane 0. 8 15 Ether 2 5 -10 Flameаbility ++
Drug MAC Metabo(%) lism (%) N 2 O >100 0 Isoflurane 1. 2 0. 2 Enflurane 1. 7 2 -10 Halothane 0. 8 15 Ether 2 5 -10 Flameаbility ++
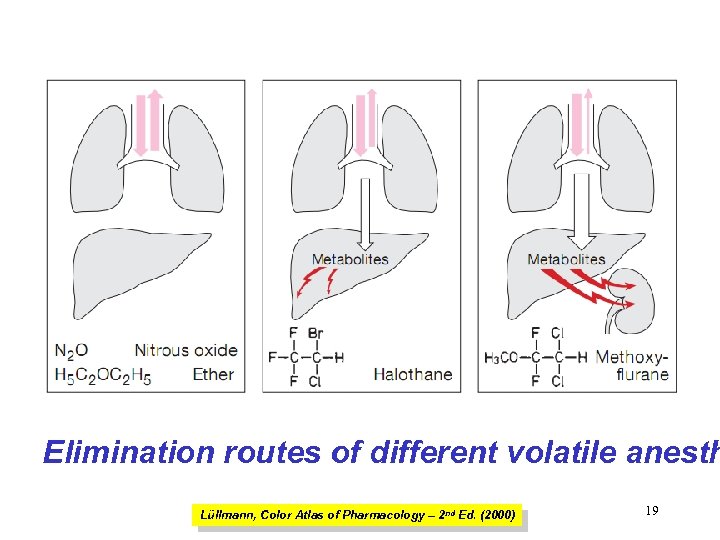 Elimination routes of different volatile anesth Lüllmann, Color Atlas of Pharmacology – 2 nd Ed. (2000) 19
Elimination routes of different volatile anesth Lüllmann, Color Atlas of Pharmacology – 2 nd Ed. (2000) 19
 Isoflurane is a less soluble is of enflurane, and is widely use. It potentiates the action of neuromu blockers. It produces dose-depen peripheral vasodilatation and hyp sion but with less myocardial dep sion than enflurane and halothane
Isoflurane is a less soluble is of enflurane, and is widely use. It potentiates the action of neuromu blockers. It produces dose-depen peripheral vasodilatation and hyp sion but with less myocardial dep sion than enflurane and halothane
 • Cerebral blood flow is little affected by isoflurane which makes it an agent of choice during neurosurgery. • Uterine tone is well maintain compared with halothane or enflurane and thereby isoflura reduces postpartum hemorrha
• Cerebral blood flow is little affected by isoflurane which makes it an agent of choice during neurosurgery. • Uterine tone is well maintain compared with halothane or enflurane and thereby isoflura reduces postpartum hemorrha
 Particular aspects of the use of Halothane relate to the following • Moderate muscular relaxation is produced, but is rarely sufficient fo major abdominal surgery. It potentiates the action of neuromuscula blockers. • Heat loss is accelerated. • It is useful in bronchitic and asthm tic patients.
Particular aspects of the use of Halothane relate to the following • Moderate muscular relaxation is produced, but is rarely sufficient fo major abdominal surgery. It potentiates the action of neuromuscula blockers. • Heat loss is accelerated. • It is useful in bronchitic and asthm tic patients.
 Adverse effects of halothane • Increased myocardial excitability (ventricular exstrasystoles, tachycardia and fibrillation). Extrasystoles can be controlled by beta-blockers. • Blood pressure usually falls, due to central vasomotor depression and myocardial depression. • Cerebral blood flow is increased which is an contraindication for use in head injury and intracranial tumors.
Adverse effects of halothane • Increased myocardial excitability (ventricular exstrasystoles, tachycardia and fibrillation). Extrasystoles can be controlled by beta-blockers. • Blood pressure usually falls, due to central vasomotor depression and myocardial depression. • Cerebral blood flow is increased which is an contraindication for use in head injury and intracranial tumors.
 • Halothane is not good analgesic and also may lead to convulsions. • It can produce massive hepatic necrosis or subclinical hepatitis following anesthesia. The liver damage appears to be a hypersensitivity type of hepatitis which is independent of dose. • Halothane can cause malignant hyperth (which need treatment with Dantrolene uterine atony and postpartum hemorrha It has a teratogenic activity.
• Halothane is not good analgesic and also may lead to convulsions. • It can produce massive hepatic necrosis or subclinical hepatitis following anesthesia. The liver damage appears to be a hypersensitivity type of hepatitis which is independent of dose. • Halothane can cause malignant hyperth (which need treatment with Dantrolene uterine atony and postpartum hemorrha It has a teratogenic activity.
 • N 2 O uses to reduce pain during childbirth. • Concomitant administration of N 2 O with one of the volatile GA reduces the MAC value of the volatile drug by up to 75%. • Risk of bone marrow depressio occurs with prolonged adminis ration of N 2 O.
• N 2 O uses to reduce pain during childbirth. • Concomitant administration of N 2 O with one of the volatile GA reduces the MAC value of the volatile drug by up to 75%. • Risk of bone marrow depressio occurs with prolonged adminis ration of N 2 O.
 2. Parenteral GAs Barbiturates and thiobarbiturat • Methohexital i. v. • Thiopental (Thiopenthone) i. v Other preparations • Ketamine i. v. /i. m. • Propofol i. v. • Etomidate i. v.
2. Parenteral GAs Barbiturates and thiobarbiturat • Methohexital i. v. • Thiopental (Thiopenthone) i. v Other preparations • Ketamine i. v. /i. m. • Propofol i. v. • Etomidate i. v.
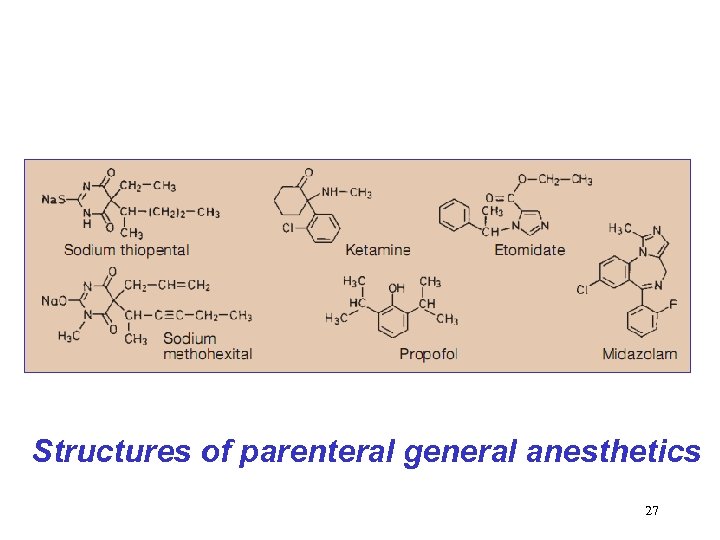 Structures of parenteral general anesthetics 27
Structures of parenteral general anesthetics 27
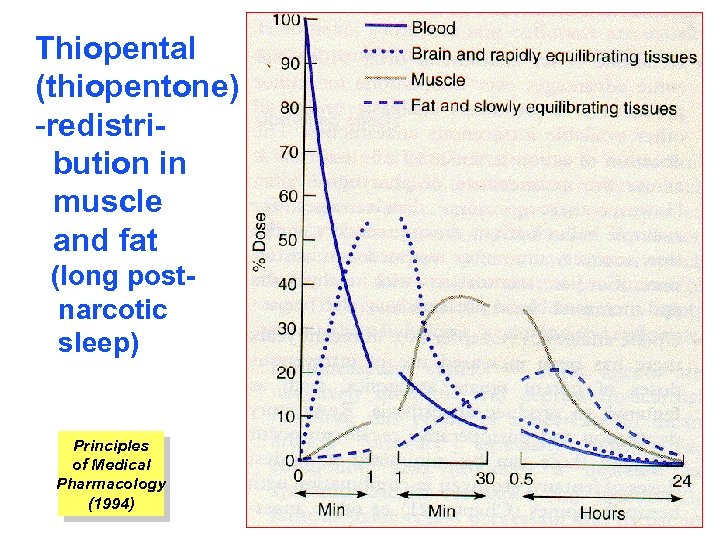 Thiopental (thiopentone) -redistribution in muscle and fat (long postnarcotic sleep) Principles of Medical Pharmacology (1994)
Thiopental (thiopentone) -redistribution in muscle and fat (long postnarcotic sleep) Principles of Medical Pharmacology (1994)
 Thiopental use i. v. for induc- tion of anesthesia, which is main tained with an inhalation agents. Propofol. The onset of its action begins after 30 s. After a single dose patient recover after 5 min with a clear head and no hangover.
Thiopental use i. v. for induc- tion of anesthesia, which is main tained with an inhalation agents. Propofol. The onset of its action begins after 30 s. After a single dose patient recover after 5 min with a clear head and no hangover.
 Propofol is a donor of NO with amnesic and antiemetic acti Indications: • i. v. induction (2– 2. 5 mg/kg) • maintenance of anaesthesia in doses of 6– 12 mg/kg • sedation (2– 3 mg/kg) in intensive care or during intensive procedu
Propofol is a donor of NO with amnesic and antiemetic acti Indications: • i. v. induction (2– 2. 5 mg/kg) • maintenance of anaesthesia in doses of 6– 12 mg/kg • sedation (2– 3 mg/kg) in intensive care or during intensive procedu
 Ketamine is an antagonist of NMDA-recepto • It produces dissosiative anaesthesia (sedation, amnesia, dissosiation, analgesia). • Ketamine can cause hallucinations and unpleasant, brightly coloured dreams in 15% of patients during recovery, which are very often accompanied by delirium. • Its use widespread in countries where there are few skilled specialists. • Usually it applied mainly for minor procedures in children (10 mg/kg i. m. ).
Ketamine is an antagonist of NMDA-recepto • It produces dissosiative anaesthesia (sedation, amnesia, dissosiation, analgesia). • Ketamine can cause hallucinations and unpleasant, brightly coloured dreams in 15% of patients during recovery, which are very often accompanied by delirium. • Its use widespread in countries where there are few skilled specialists. • Usually it applied mainly for minor procedures in children (10 mg/kg i. m. ).
 Local anesthetics (LAs) • LAs are drugs which reversibly pr vent the transmission of pain stimu locally at their site of administratio • The clinical uses and responses of depend both on the drug selected the site of administration.
Local anesthetics (LAs) • LAs are drugs which reversibly pr vent the transmission of pain stimu locally at their site of administratio • The clinical uses and responses of depend both on the drug selected the site of administration.
 LAs are weak bases (p. Kb 7– 8) They exist as an equilibrium between nized (LAH+) and unionized (LA) form The unionized forms are lipid soluble and cross the axonal membranes. Aft that the part of the unionized forms protonates intracellulary into the ionized forms. The ionized forms bind to the intracellular receptors, obstruc and block Na+ channel (see figure).
LAs are weak bases (p. Kb 7– 8) They exist as an equilibrium between nized (LAH+) and unionized (LA) form The unionized forms are lipid soluble and cross the axonal membranes. Aft that the part of the unionized forms protonates intracellulary into the ionized forms. The ionized forms bind to the intracellular receptors, obstruc and block Na+ channel (see figure).
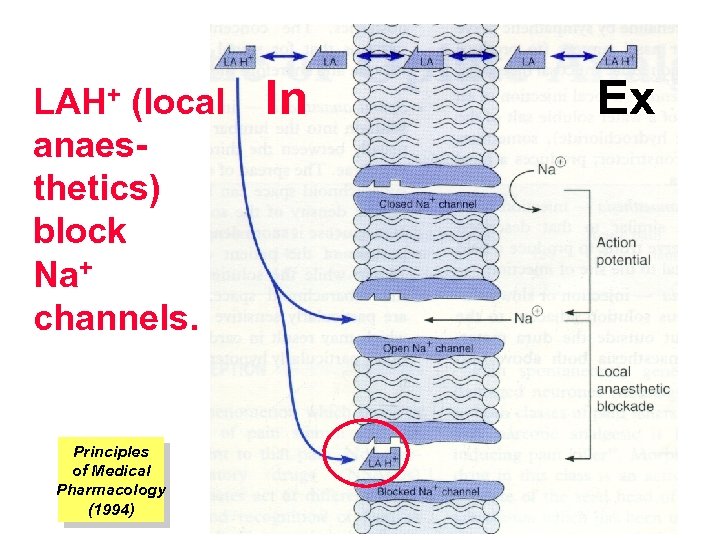 LAH+ (local anaesthetics) block Na+ channels. Principles of Medical Pharmacology (1994) In Ex
LAH+ (local anaesthetics) block Na+ channels. Principles of Medical Pharmacology (1994) In Ex
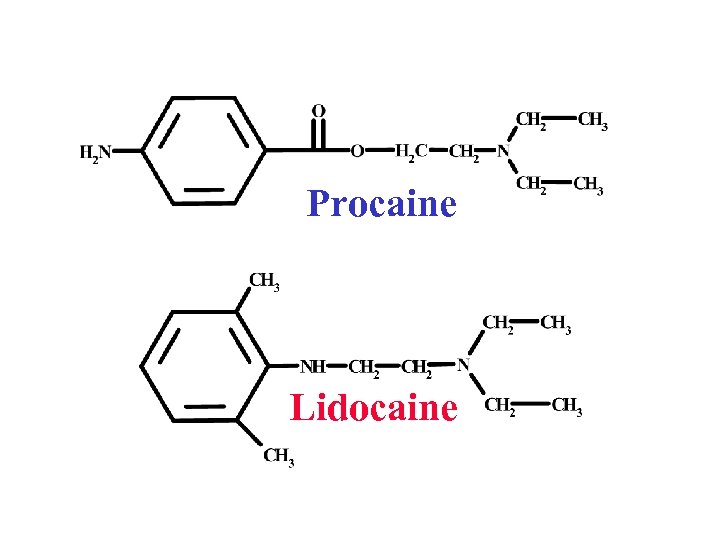 Procaine Lidocaine
Procaine Lidocaine
 Anesthetic potenc Lidocaine Bupivacaine Procaine Articaine 4 16 1 4
Anesthetic potenc Lidocaine Bupivacaine Procaine Articaine 4 16 1 4
 LAs from the group of ester (proc tetracaine, benzocaine) in plasma liver hydrolyze to the para-aminobenzoic acid, which is a competit antagonist of the sulfonamides. T the co-administration of esters an sulfonamides is not rational.
LAs from the group of ester (proc tetracaine, benzocaine) in plasma liver hydrolyze to the para-aminobenzoic acid, which is a competit antagonist of the sulfonamides. T the co-administration of esters an sulfonamides is not rational.
 Unwanted effects Local effects at the site of administ tion: irritation and inflammation; local hypoxia (if co-administered wi vasoconstrictor); tissue damage (so times necrosis) following inappropr ate administration (e. g accidental intra-arterial administration or spina administration of an epidural dose)
Unwanted effects Local effects at the site of administ tion: irritation and inflammation; local hypoxia (if co-administered wi vasoconstrictor); tissue damage (so times necrosis) following inappropr ate administration (e. g accidental intra-arterial administration or spina administration of an epidural dose)
 Systemic effects. High systemic dos may affect other excitable membran such as the heart (e. g. lidocaine can cause AV block and cardiovascular collapse; bupivacaine can cause ser arrhythmias) or the CNS (tetracaine can cause convulsions and eyes disturbances; cocaine – euphoria, hallucinations and drug abuse).
Systemic effects. High systemic dos may affect other excitable membran such as the heart (e. g. lidocaine can cause AV block and cardiovascular collapse; bupivacaine can cause ser arrhythmias) or the CNS (tetracaine can cause convulsions and eyes disturbances; cocaine – euphoria, hallucinations and drug abuse).
 Procaine sometimes causes urticaria. Some systemic unwanted effe due to the vasoconstrictor NA or adrenaline. They include hypertension and tachycardia.
Procaine sometimes causes urticaria. Some systemic unwanted effe due to the vasoconstrictor NA or adrenaline. They include hypertension and tachycardia.
 Clinical uses The extent of local anesthesia depe largely on technique of administrati Surface administration (anesthes - high concentrations (2– 5%) of the LAs can slowly penetrate the skin a mucous membranes to give a smal localized anesthesia.
Clinical uses The extent of local anesthesia depe largely on technique of administrati Surface administration (anesthes - high concentrations (2– 5%) of the LAs can slowly penetrate the skin a mucous membranes to give a smal localized anesthesia.
 Benzocaine and tetracaine are su table for these purposes. They produ useful anesthesia of mucous membr of the throat. Cocaine and tetracaine are used b re painful ophthalmological procedu Propipocaine widely use in dentistr dermatology, and obstetrics to prod surface anesthesia.
Benzocaine and tetracaine are su table for these purposes. They produ useful anesthesia of mucous membr of the throat. Cocaine and tetracaine are used b re painful ophthalmological procedu Propipocaine widely use in dentistr dermatology, and obstetrics to prod surface anesthesia.
 Infiltration anesthesia can produce with 0. 25– 0. 5% aqueous solution of lidocaine or procaine (usually with co-administration of adrenaline).
Infiltration anesthesia can produce with 0. 25– 0. 5% aqueous solution of lidocaine or procaine (usually with co-administration of adrenaline).
 The other main types of local anesthesia are: • nerve trunk block anesthesia; • epidural anesthesia (injection the L to the spinal column but outside the dura mater), used in obstetrics • spinal anesthesia (injection the LA into the lumbar subarachnoid space, usually between the 3 rd and lumbar vertebrae).
The other main types of local anesthesia are: • nerve trunk block anesthesia; • epidural anesthesia (injection the L to the spinal column but outside the dura mater), used in obstetrics • spinal anesthesia (injection the LA into the lumbar subarachnoid space, usually between the 3 rd and lumbar vertebrae).
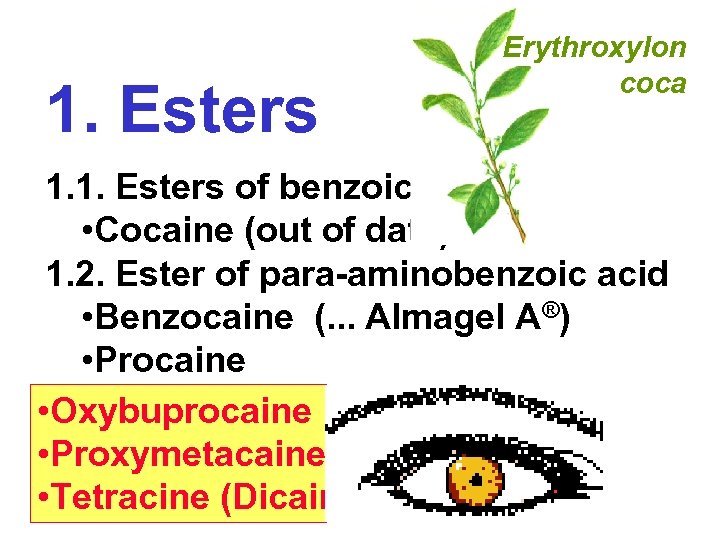 1. Esters Erythroxylon coca 1. 1. Esters of benzoic acid • Cocaine (out of date) 1. 2. Ester of para-aminobenzoic acid • Benzocaine (. . . Almagel A®) • Procaine • Oxybuprocaine • Proxymetacaine • Tetracine (Dicain®)
1. Esters Erythroxylon coca 1. 1. Esters of benzoic acid • Cocaine (out of date) 1. 2. Ester of para-aminobenzoic acid • Benzocaine (. . . Almagel A®) • Procaine • Oxybuprocaine • Proxymetacaine • Tetracine (Dicain®)
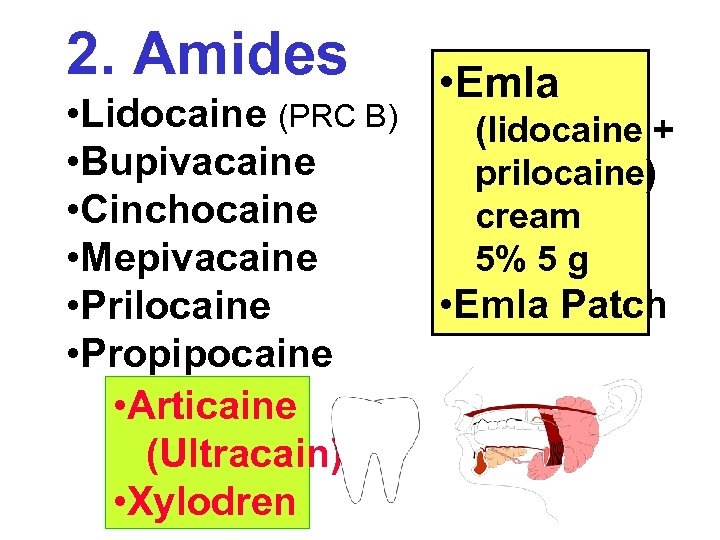 2. Amides • Lidocaine (PRC B) • Bupivacaine • Cinchocaine • Mepivacaine • Prilocaine • Propipocaine • Articaine (Ultracain) • Xylodren • Emla (lidocaine + prilocaine) cream 5% 5 g • Emla Patch
2. Amides • Lidocaine (PRC B) • Bupivacaine • Cinchocaine • Mepivacaine • Prilocaine • Propipocaine • Articaine (Ultracain) • Xylodren • Emla (lidocaine + prilocaine) cream 5% 5 g • Emla Patch
 Lidocaine (Lignocaine has also antiarrhythmic action It is an antidysrrhythmic agen from class IB, used for treatm of ventricular tachyarrhythmia from myocardial infarction, ventricular tachycardia and ventricular fibrillation.
Lidocaine (Lignocaine has also antiarrhythmic action It is an antidysrrhythmic agen from class IB, used for treatm of ventricular tachyarrhythmia from myocardial infarction, ventricular tachycardia and ventricular fibrillation.
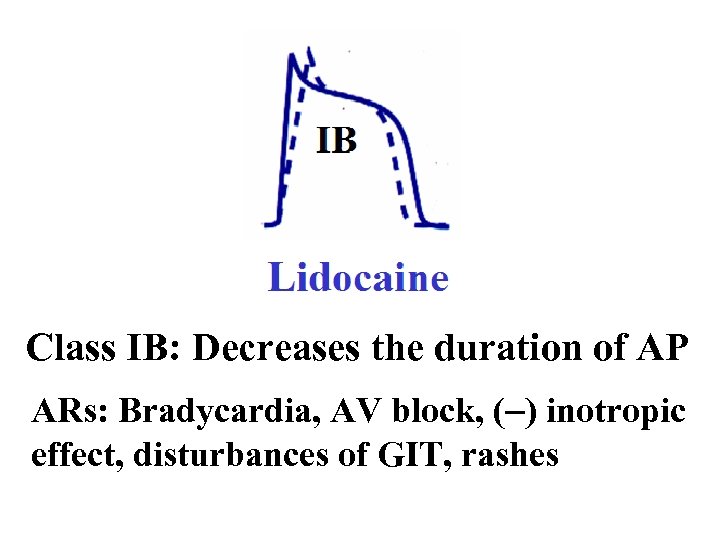 Class IB: Decreases the duration of AP ARs: Bradycardia, AV block, (-) inotropic effect, disturbances of GIT, rashes
Class IB: Decreases the duration of AP ARs: Bradycardia, AV block, (-) inotropic effect, disturbances of GIT, rashes
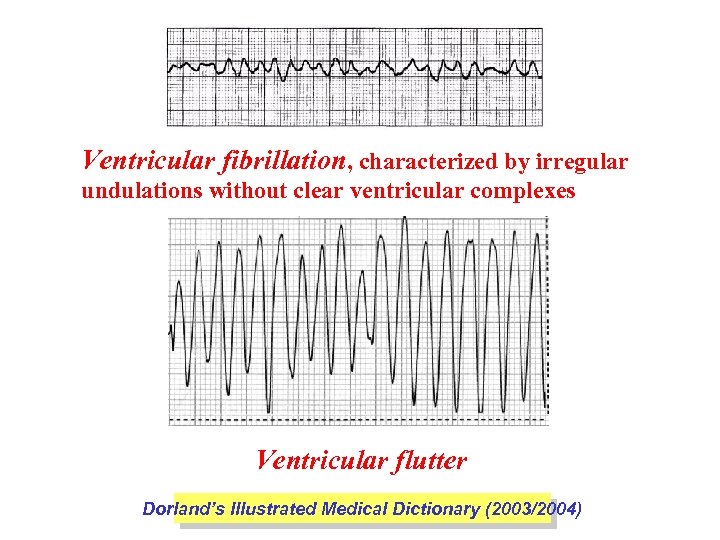 Ventricular fibrillation, characterized by irregular undulations without clear ventricular complexes Ventricular flutter Dorland’s Illustrated Medical Dictionary (2003/2004)
Ventricular fibrillation, characterized by irregular undulations without clear ventricular complexes Ventricular flutter Dorland’s Illustrated Medical Dictionary (2003/2004)


