5477a7bff25d19aaa51c98243be0b368.ppt
- Количество слайдов: 20
MEDICAL DEVICES DRUGS ECUADOR REGULATION
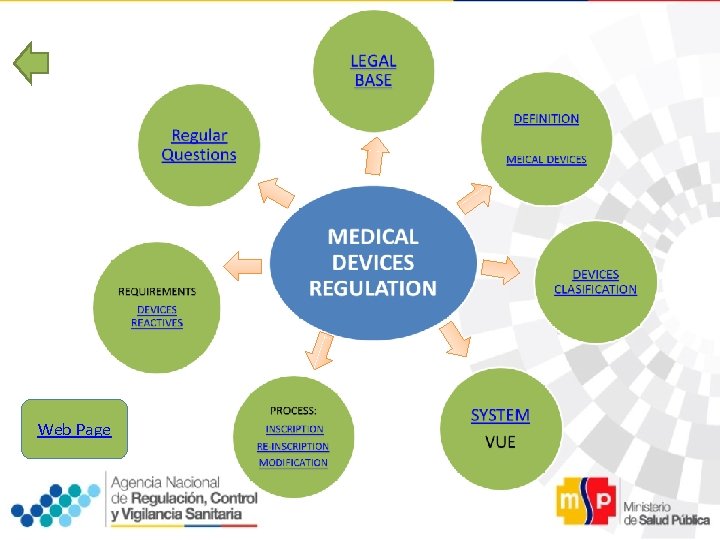
Web Page
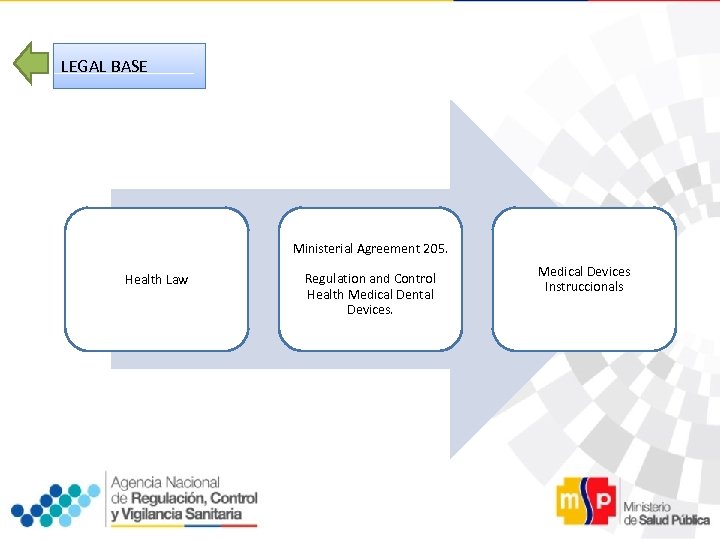
LEGAL BASE Ministerial Agreement 205. Health Law Regulation and Control Health Medical Dental Devices. Medical Devices Instruccionals
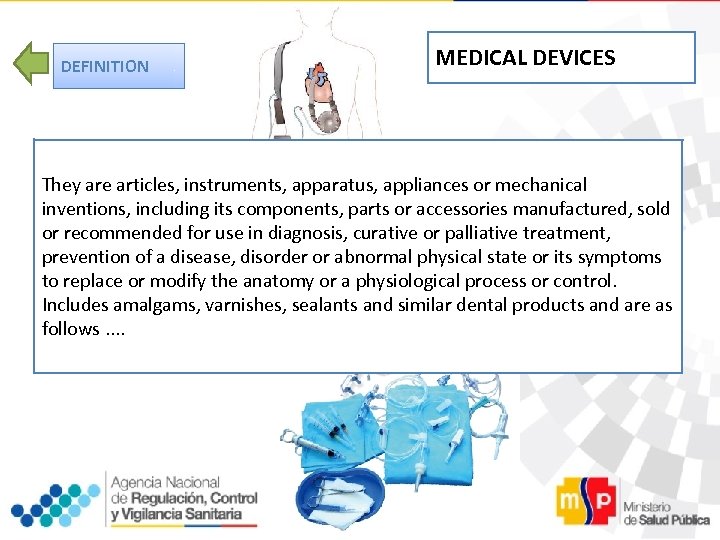
DEFINITION MEDICAL DEVICES They are articles, instruments, apparatus, appliances or mechanical inventions, including its components, parts or accessories manufactured, sold or recommended for use in diagnosis, curative or palliative treatment, prevention of a disease, disorder or abnormal physical state or its symptoms to replace or modify the anatomy or a physiological process or control. Includes amalgams, varnishes, sealants and similar dental products and are as follows. .
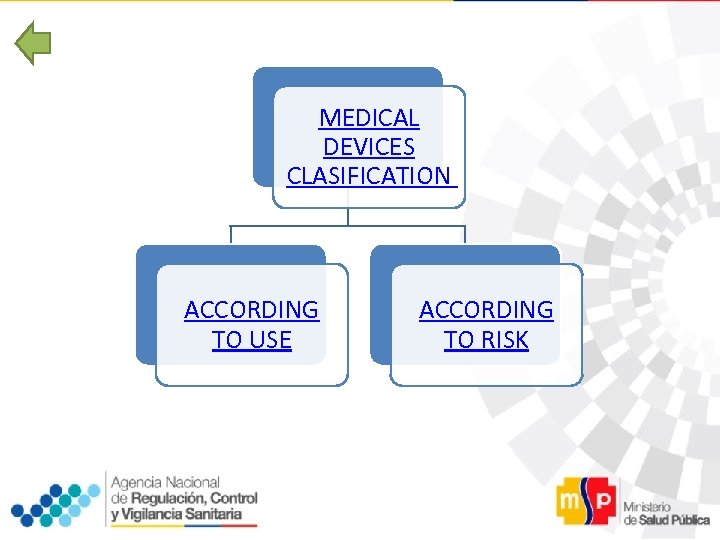
MEDICAL DEVICES CLASIFICATION ACCORDING TO USE ACCORDING TO RISK

MEDICAL DEVICES CLASIFICATION ü Codificación de la UMDNS (Nomenclatura Universal de Dispositivos Médicos del Instituto ECRI) ü Global Harmonization Task Force
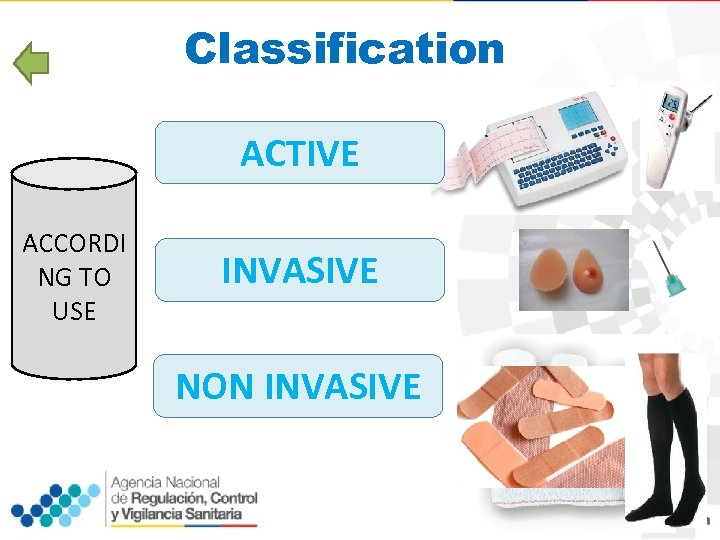
Classification ACTIVE ACCORDI NG TO USE INVASIVE NON INVASIVE
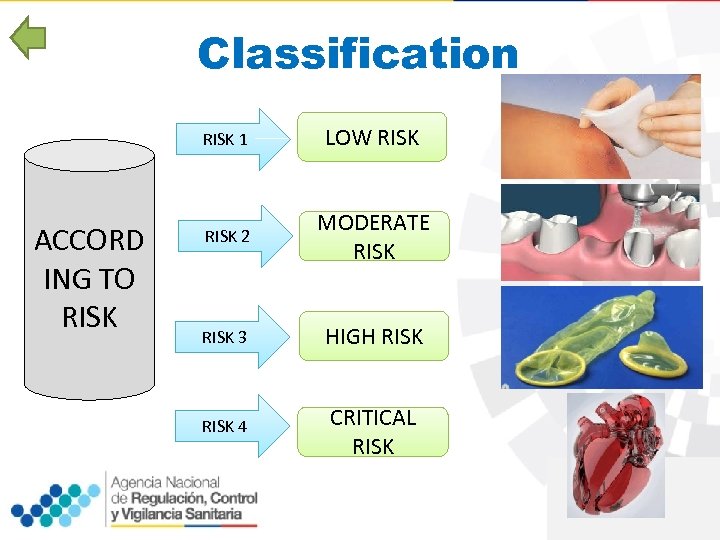
Classification RISK 1 ACCORD ING TO RISK LOW RISK 2 MODERATE RISK 3 HIGH RISK 4 CRITICAL RISK

SYSTEM TO ISSUE CERTIFICATES OF HEALTH RECORDS FOR EXTERNAL USERS VUE FOR INTERNAL USERS SIVC Ecuadorian Single Window for Foreign Trade (VUE) Informatics' System, Sanitary Surveillance and Control (SIVC) It is an electronic tool through which all users of customs services and, in general, all foreign trade operators will present the requirements, procedures and documents required for conducting foreign trade operations. The overall objective of VUE system is to optimize and integrate foreign trade processes. HEALTH RECORD CAN BE VALIDATED ONLINE WHAT IS? PRINCIPAL OBJETIVE?
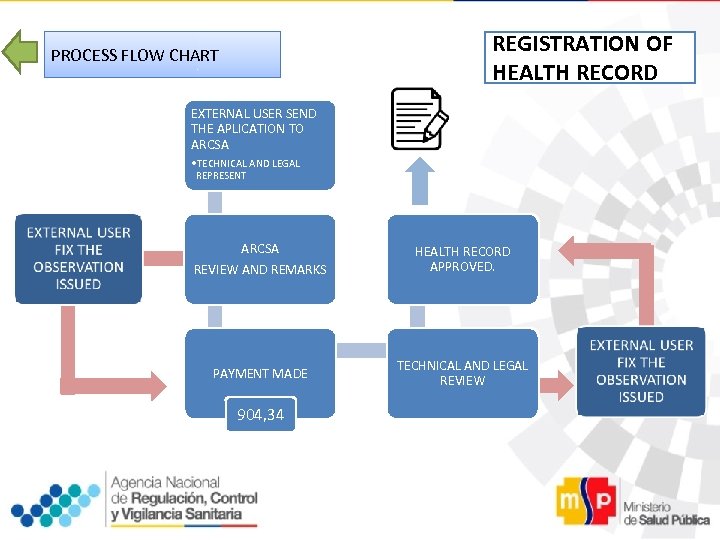
REGISTRATION OF HEALTH RECORD PROCESS FLOW CHART EXTERNAL USER SEND THE APLICATION TO ARCSA • TECHNICAL AND LEGAL REPRESENT ARCSA REVIEW AND REMARKS HEALTH RECORD APPROVED. PAYMENT MADE TECHNICAL AND LEGAL REVIEW 904, 34
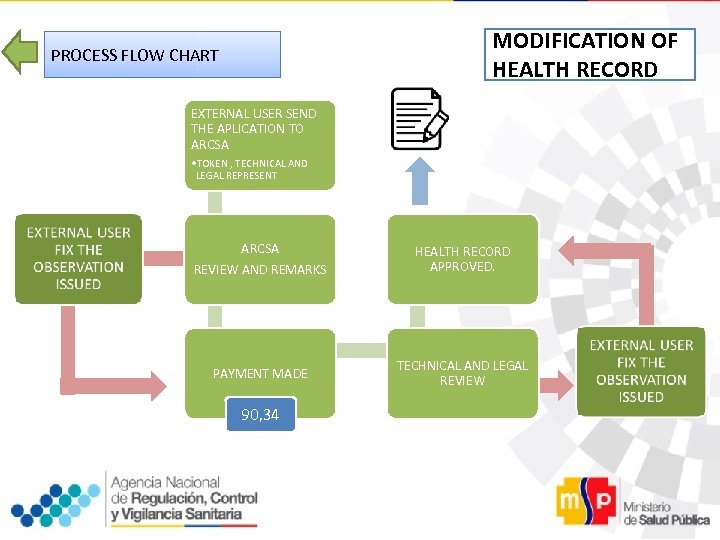
MODIFICATION OF HEALTH RECORD PROCESS FLOW CHART EXTERNAL USER SEND THE APLICATION TO ARCSA • TOKEN , TECHNICAL AND LEGAL REPRESENT ARCSA REVIEW AND REMARKS HEALTH RECORD APPROVED. PAYMENT MADE TECHNICAL AND LEGAL REVIEW 90, 34
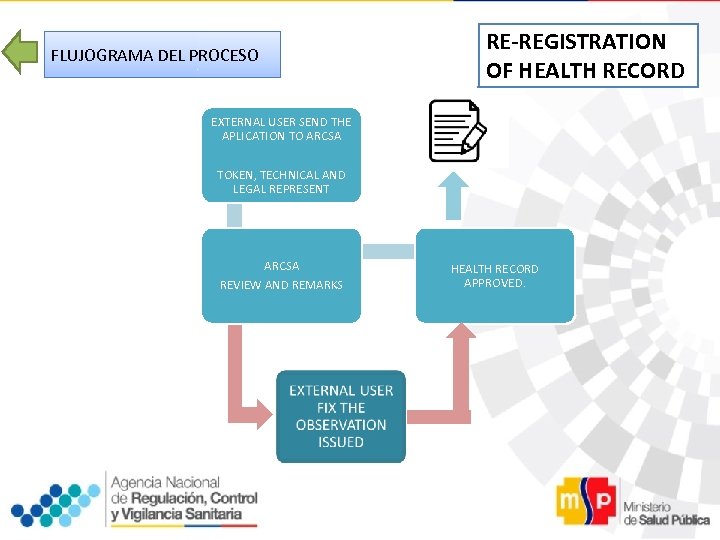
FLUJOGRAMA DEL PROCESO RE-REGISTRATION OF HEALTH RECORD EXTERNAL USER SEND THE APLICATION TO ARCSA TOKEN, TECHNICAL AND LEGAL REPRESENT ARCSA REVIEW AND REMARKS HEALTH RECORD APPROVED.

REGISTRATION OF HEALTH RECORD REQUIREMENTS • Finished Product Specifications • Product Authorization Holder to • Quality Certificate or Certificate of Analysis MEDICAL DEVICE request medical record Ecuador. • Description of Use • Certificate of Sterility • Scientific Literature Product • Certificate of Stability • Original Product Tags • Interpretation of the Lot • Formats interim Tags, Inserts for use in • Certificate of Free Sale • Certificate of GMP or: – ISO 9001 (RISK 1) – ISO 13485(RISK>1) Spanish. NOTE: The applicant must submit a separate application for medical devices belonging to the same class, group, risk, trademark, owner and manufacturer (single plant).

REQUISITOS/INSTRUCTIVOS Página web: http: //www. controlsanitario. gob. ec/

DRUG REGULATION

DRUG REGULATIONS LEGAL BASE REQUIERMENTS
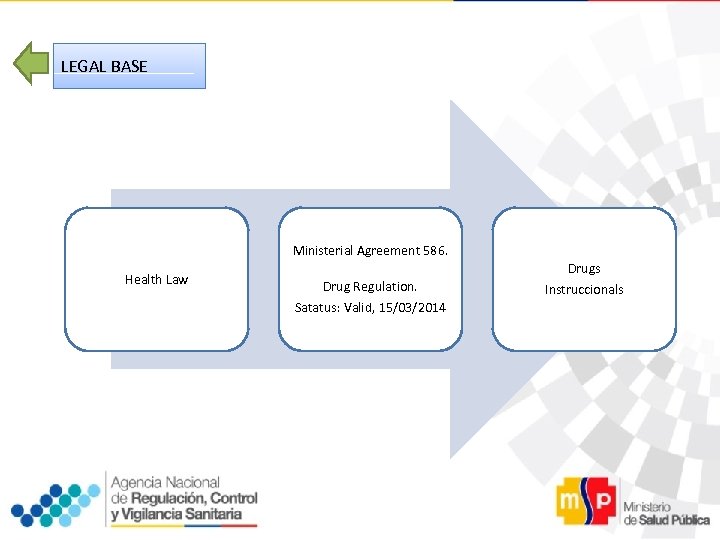
LEGAL BASE Ministerial Agreement 586. Health Law Drug Regulation. Satatus: Valid, 15/03/2014 Drugs Instruccionals

REGISTRATION OF HEALTH RECORD REQUIREMENTS DRUG REGULATION 1. Current certificate of pharmaceutical product marketed. 2. Authorization of the owner or duly authenticated Product power. 3. Format internal and external product labels to market in Ecuador. 4. Memorandum addressed to the user written in Castilian language. 5. Certificate of Good Manufacturing Practices in force and duly authenticated. 6. Specifications of finished product quality. 7. Interpretation of batch code 8. Justification of the composition formula 9. Description of the manufacturing process of the drug 10. Analytical methodology of the finished product.

REGISTRATION OF HEALTH RECORD REQUIREMENTS DRUG REGULATION 11. Internal and external of how the drug is marketed in the country of origin Original tags. 12. Specifications of quality raw materials. 13. Stability Study Product 14. Description of the nature of the primary container and / or secondary 15. Documentation of safety and efficacy of the finished product. 16. A copy of the authorization of the owner of the product for the use of technical documentation. 17. Working pharmacological and toxicological nature (for new drugs). 18. Justification for the pharmaceutical equivalence (for alternate manufacturers). 19. Studies of equivalence in vivo / in vitro bioequivalence (where applicable).

Very thanks GRACIAS
5477a7bff25d19aaa51c98243be0b368.ppt