20f519559f0a171dc49554a35716a5f6.ppt
- Количество слайдов: 98
 Mechanisms of Allergic Immunity crah 1@le. ac. uk
Mechanisms of Allergic Immunity crah 1@le. ac. uk
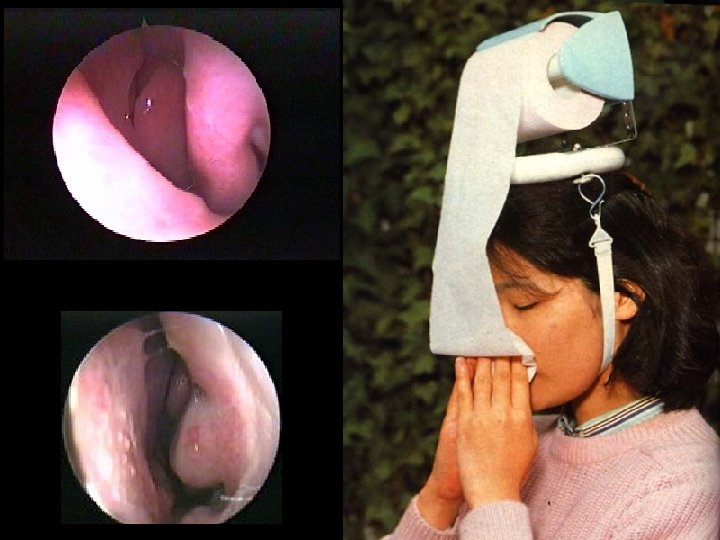
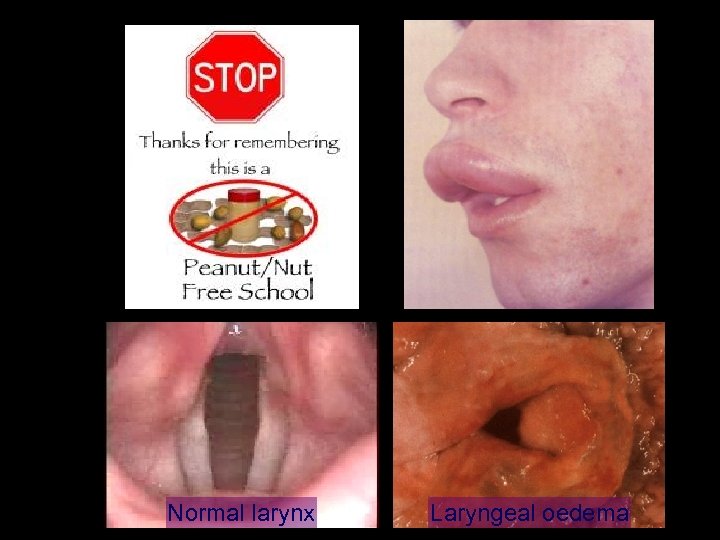 Normal larynx Laryngeal oedema
Normal larynx Laryngeal oedema
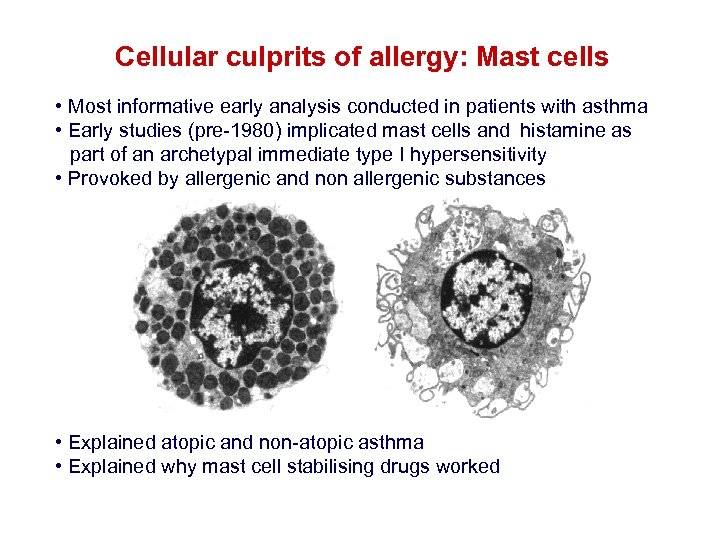 Cellular culprits of allergy: Mast cells • Most informative early analysis conducted in patients with asthma • Early studies (pre-1980) implicated mast cells and histamine as part of an archetypal immediate type I hypersensitivity • Provoked by allergenic and non allergenic substances • Explained atopic and non-atopic asthma • Explained why mast cell stabilising drugs worked
Cellular culprits of allergy: Mast cells • Most informative early analysis conducted in patients with asthma • Early studies (pre-1980) implicated mast cells and histamine as part of an archetypal immediate type I hypersensitivity • Provoked by allergenic and non allergenic substances • Explained atopic and non-atopic asthma • Explained why mast cell stabilising drugs worked
 Cellular culprits of allergy: Mast cells? ? • Corticosteroid treatment worked, but had no effect on histamine release • Anti-histamine treatment had little effect on asthma • Could not explain ‘organ specificity’ of asthma • Could not explain the hyperresponsive airway in asymptomatic asthmatics • Fibreoptic bronchoscopy - immunohistology, biopsy and analysis of bronchoalveolar lavage (BAL) cells (1980’s - present)
Cellular culprits of allergy: Mast cells? ? • Corticosteroid treatment worked, but had no effect on histamine release • Anti-histamine treatment had little effect on asthma • Could not explain ‘organ specificity’ of asthma • Could not explain the hyperresponsive airway in asymptomatic asthmatics • Fibreoptic bronchoscopy - immunohistology, biopsy and analysis of bronchoalveolar lavage (BAL) cells (1980’s - present)
 Cellular culprits of allergy: T cells The early evidence: • Eosinophil & mononuclear cells infiltrate the bronchi of asthmatics • Activated T cells elevated in the peripheral blood of severe acute asthmatics • Activated T cells in peripheral blood correlated with airway narrowing • Bronchial CD 4 lymphocyte numbers correlated with eosinophil numbers • Elevated IL-5 expressing T cells in asthmatic bronchial mucosa and BAL • T cells that release IL-5 co-localise with eosinophils • Eosinophils cause airway hyperresponsiveness, inflammation desquamative bronchitis, mucous hypersecretion and smooth muscle contraction • IL-5 promotes differentiation and regulates the survival of eosinophils • Steroid treatment associated with a decrease in IL-5 producing cells
Cellular culprits of allergy: T cells The early evidence: • Eosinophil & mononuclear cells infiltrate the bronchi of asthmatics • Activated T cells elevated in the peripheral blood of severe acute asthmatics • Activated T cells in peripheral blood correlated with airway narrowing • Bronchial CD 4 lymphocyte numbers correlated with eosinophil numbers • Elevated IL-5 expressing T cells in asthmatic bronchial mucosa and BAL • T cells that release IL-5 co-localise with eosinophils • Eosinophils cause airway hyperresponsiveness, inflammation desquamative bronchitis, mucous hypersecretion and smooth muscle contraction • IL-5 promotes differentiation and regulates the survival of eosinophils • Steroid treatment associated with a decrease in IL-5 producing cells
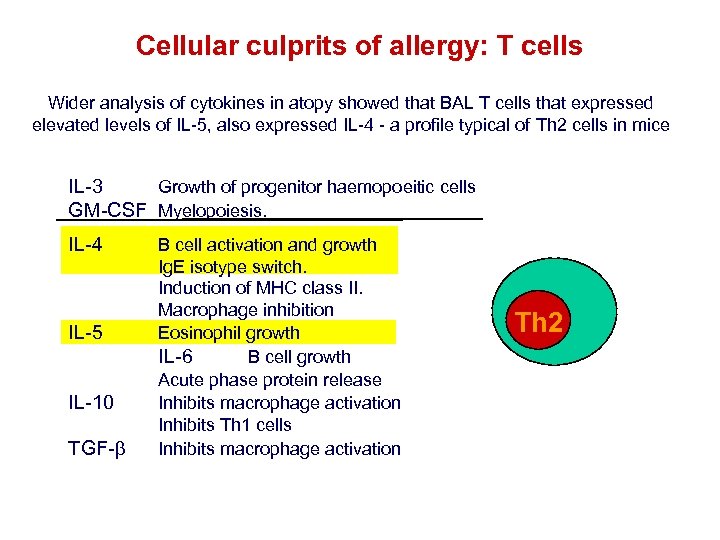 Cellular culprits of allergy: T cells Wider analysis of cytokines in atopy showed that BAL T cells that expressed elevated levels of IL-5, also expressed IL-4 - a profile typical of Th 2 cells in mice IL-3 Growth of progenitor haemopoeitic cells GM-CSF Myelopoiesis. IL-4 IL-5 IL-10 TGF- B cell activation and growth Ig. E isotype switch. Induction of MHC class II. Macrophage inhibition Eosinophil growth IL-6 B cell growth Acute phase protein release Inhibits macrophage activation Inhibits Th 1 cells Inhibits macrophage activation Th 2
Cellular culprits of allergy: T cells Wider analysis of cytokines in atopy showed that BAL T cells that expressed elevated levels of IL-5, also expressed IL-4 - a profile typical of Th 2 cells in mice IL-3 Growth of progenitor haemopoeitic cells GM-CSF Myelopoiesis. IL-4 IL-5 IL-10 TGF- B cell activation and growth Ig. E isotype switch. Induction of MHC class II. Macrophage inhibition Eosinophil growth IL-6 B cell growth Acute phase protein release Inhibits macrophage activation Inhibits Th 1 cells Inhibits macrophage activation Th 2
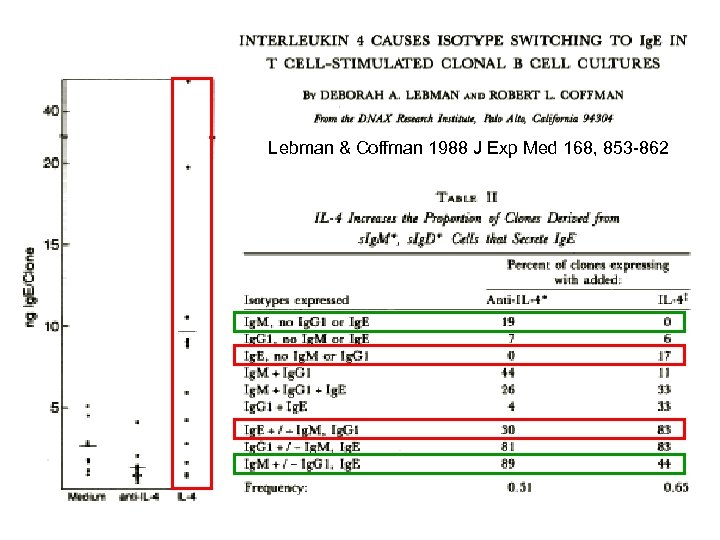 Lebman & Coffman 1988 J Exp Med 168, 853 -862
Lebman & Coffman 1988 J Exp Med 168, 853 -862
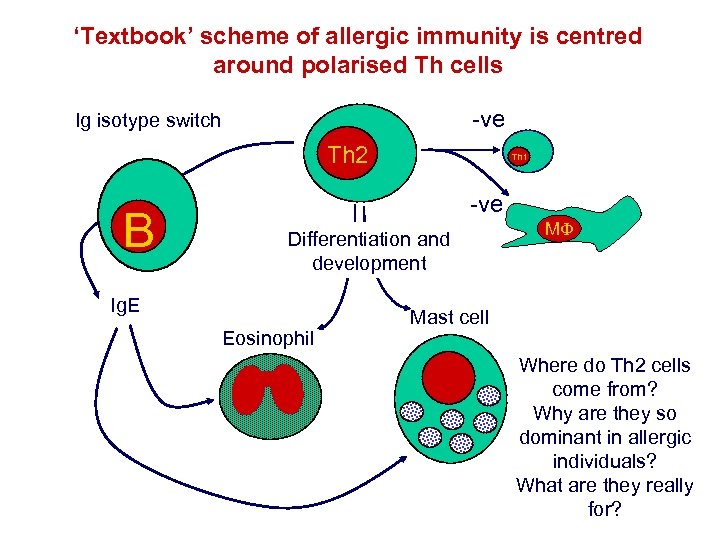 ‘Textbook’ scheme of allergic immunity is centred around polarised Th cells -ve Ig isotype switch Th 2 B Th 1 -ve Differentiation and development Ig. E Eosinophil MF Mast cell Where do Th 2 cells come from? Why are they so dominant in allergic individuals? What are they really for?
‘Textbook’ scheme of allergic immunity is centred around polarised Th cells -ve Ig isotype switch Th 2 B Th 1 -ve Differentiation and development Ig. E Eosinophil MF Mast cell Where do Th 2 cells come from? Why are they so dominant in allergic individuals? What are they really for?
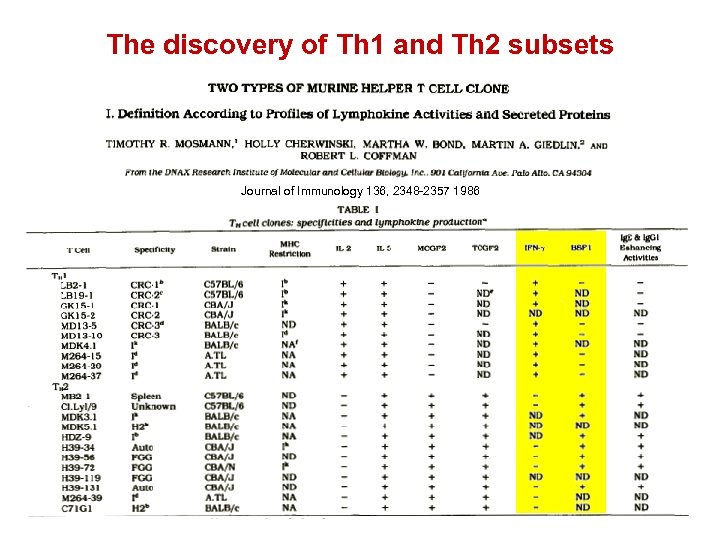 The discovery of Th 1 and Th 2 subsets Journal of Immunology 136, 2348 -2357 1986
The discovery of Th 1 and Th 2 subsets Journal of Immunology 136, 2348 -2357 1986
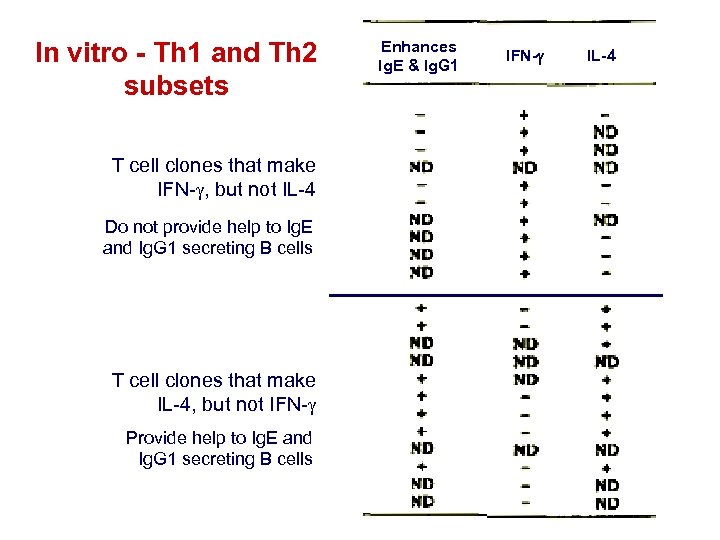 In vitro - Th 1 and Th 2 subsets T cell clones that make IFN- , but not IL-4 Do not provide help to Ig. E and Ig. G 1 secreting B cells T cell clones that make IL-4, but not IFN- Provide help to Ig. E and Ig. G 1 secreting B cells Enhances Ig. E & Ig. G 1 IFN-g IL-4
In vitro - Th 1 and Th 2 subsets T cell clones that make IFN- , but not IL-4 Do not provide help to Ig. E and Ig. G 1 secreting B cells T cell clones that make IL-4, but not IFN- Provide help to Ig. E and Ig. G 1 secreting B cells Enhances Ig. E & Ig. G 1 IFN-g IL-4
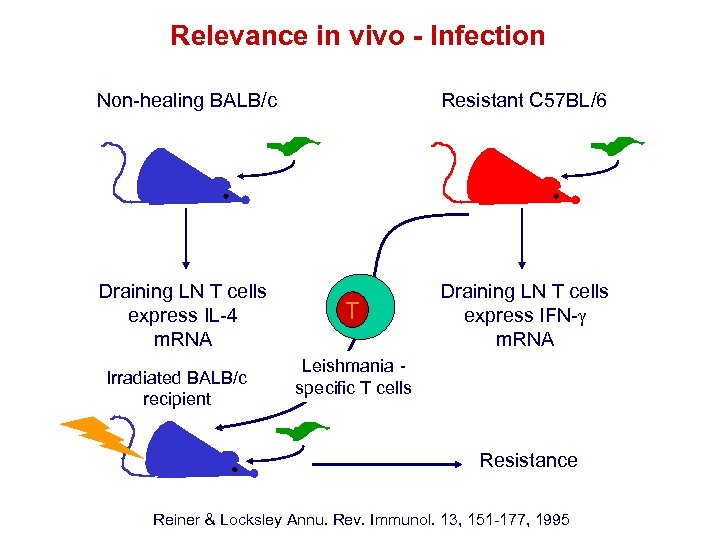 Relevance in vivo - Infection Non-healing BALB/c Resistant C 57 BL/6 Draining LN T cells express IL-4 m. RNA Draining LN T cells express IFN- m. RNA Irradiated BALB/c recipient T Leishmania specific T cells Resistance Reiner & Locksley Annu. Rev. Immunol. 13, 151 -177, 1995
Relevance in vivo - Infection Non-healing BALB/c Resistant C 57 BL/6 Draining LN T cells express IL-4 m. RNA Draining LN T cells express IFN- m. RNA Irradiated BALB/c recipient T Leishmania specific T cells Resistance Reiner & Locksley Annu. Rev. Immunol. 13, 151 -177, 1995
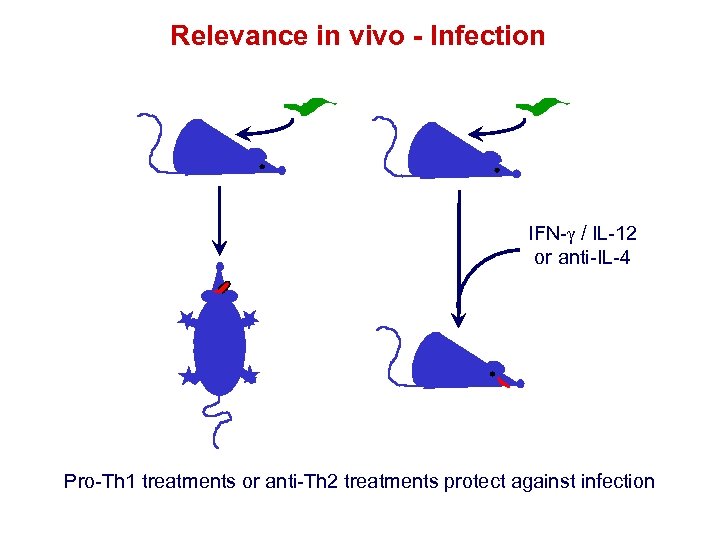 Relevance in vivo - Infection IFN- / IL-12 or anti-IL-4 Pro-Th 1 treatments or anti-Th 2 treatments protect against infection
Relevance in vivo - Infection IFN- / IL-12 or anti-IL-4 Pro-Th 1 treatments or anti-Th 2 treatments protect against infection
 Leishmania resistance - mechanism Th 1 IFN- Inflammatory Th 1 T cell Macrophage and Leishmania Macrophage infected with Leishmania kills pathogen when activated Macrophage activation is dependent upon Th 1 cells
Leishmania resistance - mechanism Th 1 IFN- Inflammatory Th 1 T cell Macrophage and Leishmania Macrophage infected with Leishmania kills pathogen when activated Macrophage activation is dependent upon Th 1 cells
 Relevance of Th subsets in humans Lepromatous and tuberculoid leprosy Infection with Mycobacterium leprae shows two main clinical forms associated with Th 1 and Th 2 responses Tuberculoid leprosy Lepromatous leprosy Low infectivity Localised infection Normal serum Ig Normal T cell response High infectivity Disseminated infection Hypergammaglobulinaemia Unresponsive Th 1 Poor growth of mycobacteria in macrophages Th 2 Florid growth of mycobacteria in macrophages
Relevance of Th subsets in humans Lepromatous and tuberculoid leprosy Infection with Mycobacterium leprae shows two main clinical forms associated with Th 1 and Th 2 responses Tuberculoid leprosy Lepromatous leprosy Low infectivity Localised infection Normal serum Ig Normal T cell response High infectivity Disseminated infection Hypergammaglobulinaemia Unresponsive Th 1 Poor growth of mycobacteria in macrophages Th 2 Florid growth of mycobacteria in macrophages
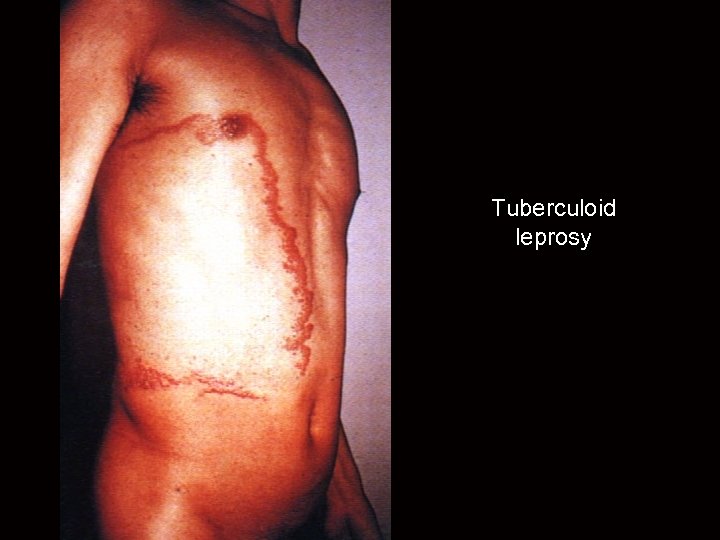 Tuberculoid leprosy
Tuberculoid leprosy
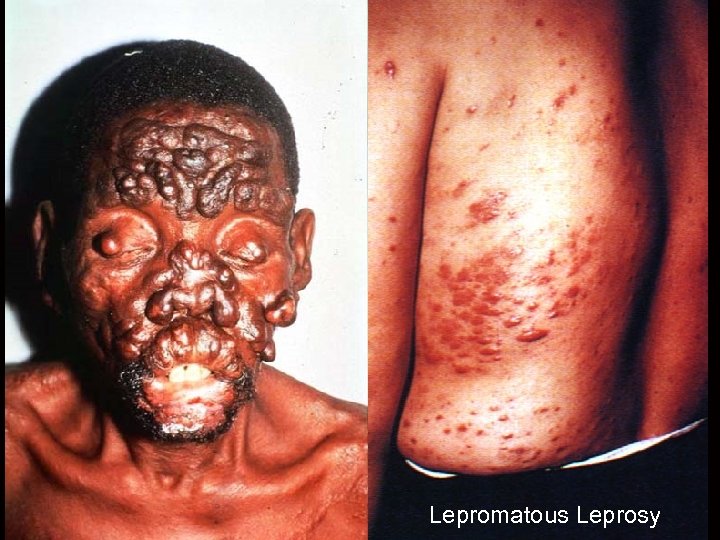 Lepromatous Leprosy
Lepromatous Leprosy
 ‘Textbook’ scheme of allergic immunity is centred around polarised Th cells Immunological fashions • 1960’s & 1970’s Immunoglobulin E • 1970’s & 1980’s Mast cells & Eosinophils • 1980’s & 1990’s Environment – ante-natal & adult, allergens, Th 2 cells • 1990’s & 2000’s Microbial experience, Epithelium, Tregs Although undoubtedly a useful model, the textbook ‘skew to Th 2’ model is too simplistic to explain allergy Allergy is a disease of impaired immune regulation Where is the regulatory lesion?
‘Textbook’ scheme of allergic immunity is centred around polarised Th cells Immunological fashions • 1960’s & 1970’s Immunoglobulin E • 1970’s & 1980’s Mast cells & Eosinophils • 1980’s & 1990’s Environment – ante-natal & adult, allergens, Th 2 cells • 1990’s & 2000’s Microbial experience, Epithelium, Tregs Although undoubtedly a useful model, the textbook ‘skew to Th 2’ model is too simplistic to explain allergy Allergy is a disease of impaired immune regulation Where is the regulatory lesion?
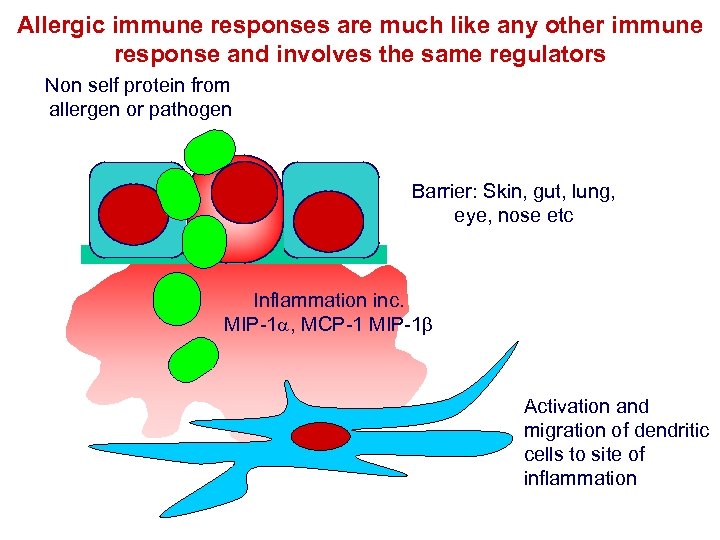 Allergic immune responses are much like any other immune response and involves the same regulators Non self protein from allergen or pathogen Barrier: Skin, gut, lung, eye, nose etc Inflammation inc. MIP-1 a, MCP-1 MIP-1 Activation and migration of dendritic cells to site of inflammation
Allergic immune responses are much like any other immune response and involves the same regulators Non self protein from allergen or pathogen Barrier: Skin, gut, lung, eye, nose etc Inflammation inc. MIP-1 a, MCP-1 MIP-1 Activation and migration of dendritic cells to site of inflammation
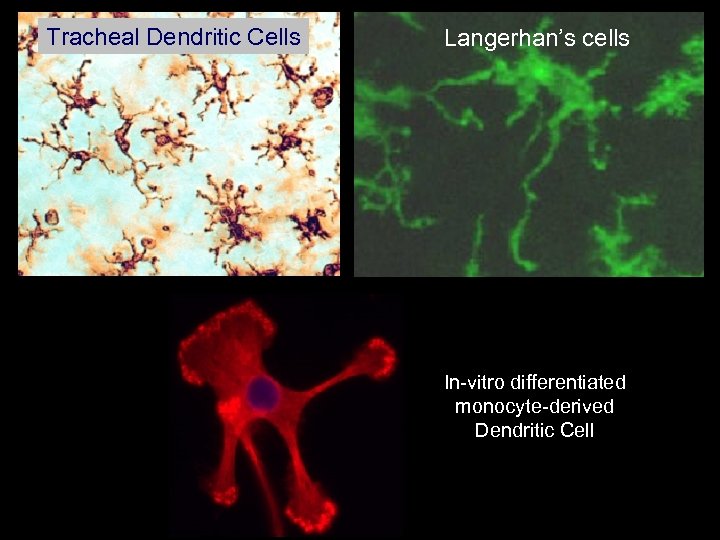 Tracheal Dendritic Cells Langerhan’s cells In-vitro differentiated monocyte-derived Dendritic Cell
Tracheal Dendritic Cells Langerhan’s cells In-vitro differentiated monocyte-derived Dendritic Cell
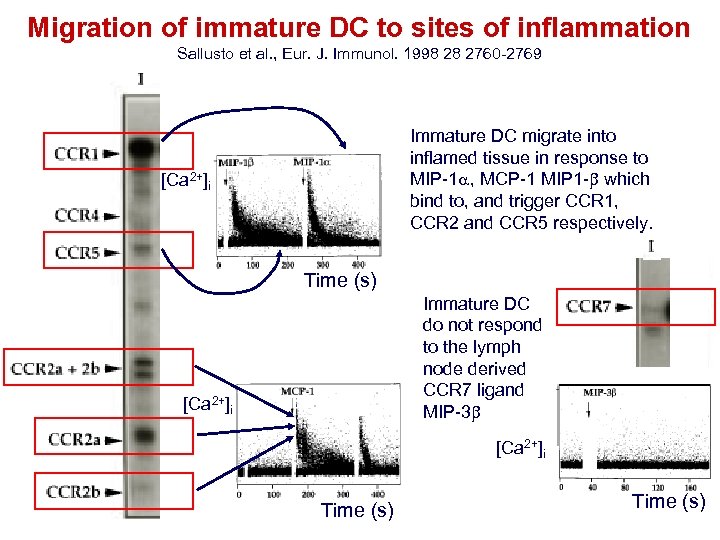 Migration of immature DC to sites of inflammation Sallusto et al. , Eur. J. Immunol. 1998 28 2760 -2769 Immature DC migrate into inflamed tissue in response to MIP-1 a, MCP-1 MIP 1 - which bind to, and trigger CCR 1, CCR 2 and CCR 5 respectively. [Ca 2+]i Time (s) Immature DC do not respond to the lymph node derived CCR 7 ligand MIP-3 [Ca 2+]i Time (s)
Migration of immature DC to sites of inflammation Sallusto et al. , Eur. J. Immunol. 1998 28 2760 -2769 Immature DC migrate into inflamed tissue in response to MIP-1 a, MCP-1 MIP 1 - which bind to, and trigger CCR 1, CCR 2 and CCR 5 respectively. [Ca 2+]i Time (s) Immature DC do not respond to the lymph node derived CCR 7 ligand MIP-3 [Ca 2+]i Time (s)
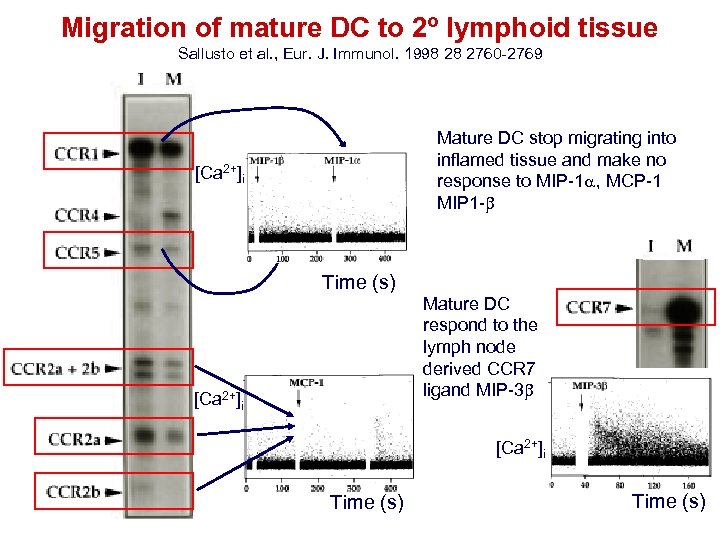 Migration of mature DC to 2º lymphoid tissue Sallusto et al. , Eur. J. Immunol. 1998 28 2760 -2769 Mature DC stop migrating into inflamed tissue and make no response to MIP-1 a, MCP-1 MIP 1 - [Ca 2+]i Time (s) [Ca 2+]i Mature DC respond to the lymph node derived CCR 7 ligand MIP-3 [Ca 2+]i Time (s)
Migration of mature DC to 2º lymphoid tissue Sallusto et al. , Eur. J. Immunol. 1998 28 2760 -2769 Mature DC stop migrating into inflamed tissue and make no response to MIP-1 a, MCP-1 MIP 1 - [Ca 2+]i Time (s) [Ca 2+]i Mature DC respond to the lymph node derived CCR 7 ligand MIP-3 [Ca 2+]i Time (s)
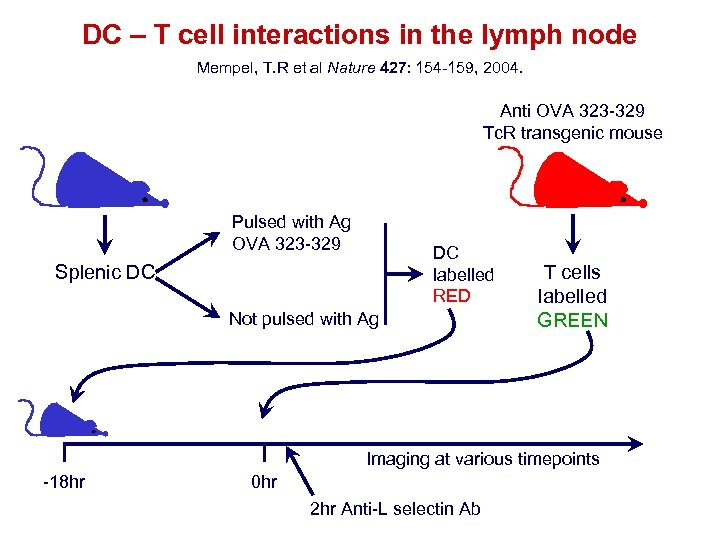 DC – T cell interactions in the lymph node Mempel, T. R et al Nature 427: 154 -159, 2004. Anti OVA 323 -329 Tc. R transgenic mouse Pulsed with Ag OVA 323 -329 DC labelled RED Splenic DC Not pulsed with Ag T cells labelled GREEN Imaging at various timepoints -18 hr 0 hr 2 hr Anti-L selectin Ab
DC – T cell interactions in the lymph node Mempel, T. R et al Nature 427: 154 -159, 2004. Anti OVA 323 -329 Tc. R transgenic mouse Pulsed with Ag OVA 323 -329 DC labelled RED Splenic DC Not pulsed with Ag T cells labelled GREEN Imaging at various timepoints -18 hr 0 hr 2 hr Anti-L selectin Ab
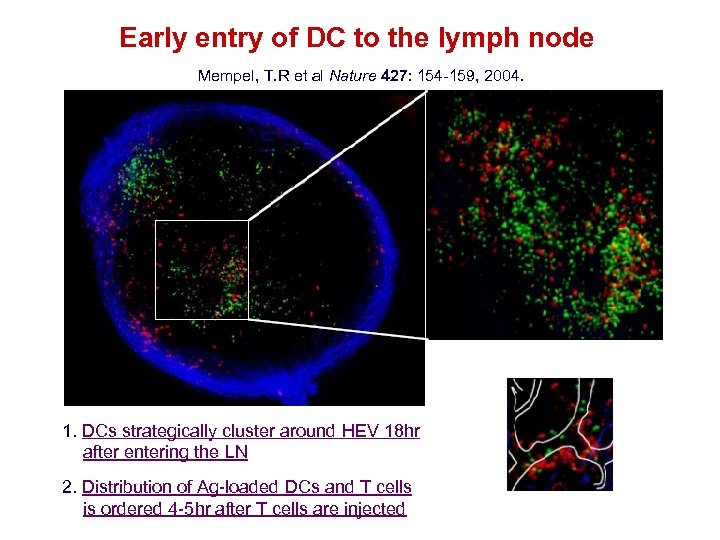 Early entry of DC to the lymph node Mempel, T. R et al Nature 427: 154 -159, 2004. 1. DCs strategically cluster around HEV 18 hr after entering the LN 2. Distribution of Ag-loaded DCs and T cells is ordered 4 -5 hr after T cells are injected
Early entry of DC to the lymph node Mempel, T. R et al Nature 427: 154 -159, 2004. 1. DCs strategically cluster around HEV 18 hr after entering the LN 2. Distribution of Ag-loaded DCs and T cells is ordered 4 -5 hr after T cells are injected
 3. DC become highly migratory & change shape (20 hr)
3. DC become highly migratory & change shape (20 hr)
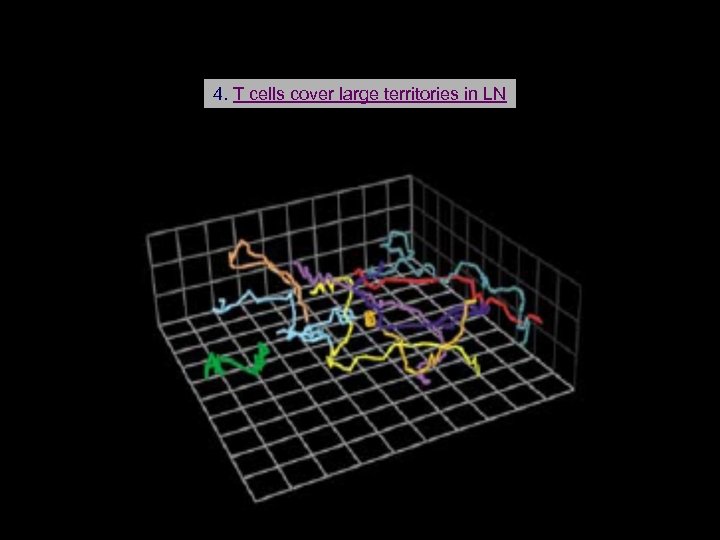 4. T cells cover large territories in LN
4. T cells cover large territories in LN
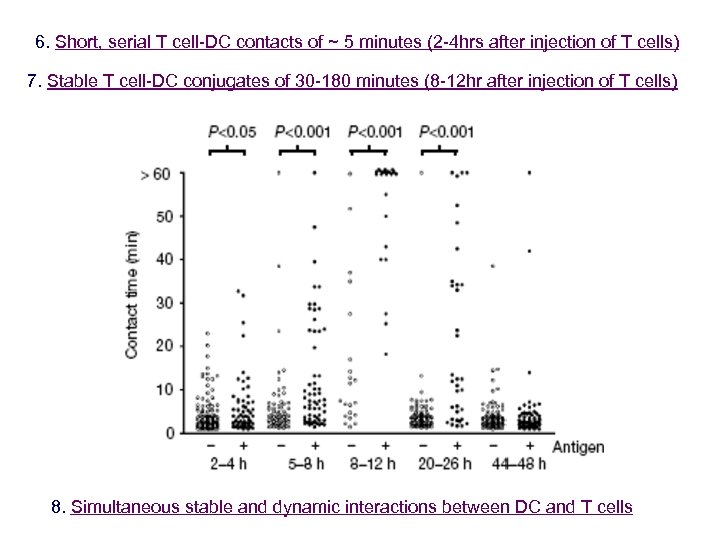 6. Short, serial T cell-DC contacts of ~ 5 minutes (2 -4 hrs after injection of T cells) 7. Stable T cell-DC conjugates of 30 -180 minutes (8 -12 hr after injection of T cells) 8. Simultaneous stable and dynamic interactions between DC and T cells
6. Short, serial T cell-DC contacts of ~ 5 minutes (2 -4 hrs after injection of T cells) 7. Stable T cell-DC conjugates of 30 -180 minutes (8 -12 hr after injection of T cells) 8. Simultaneous stable and dynamic interactions between DC and T cells
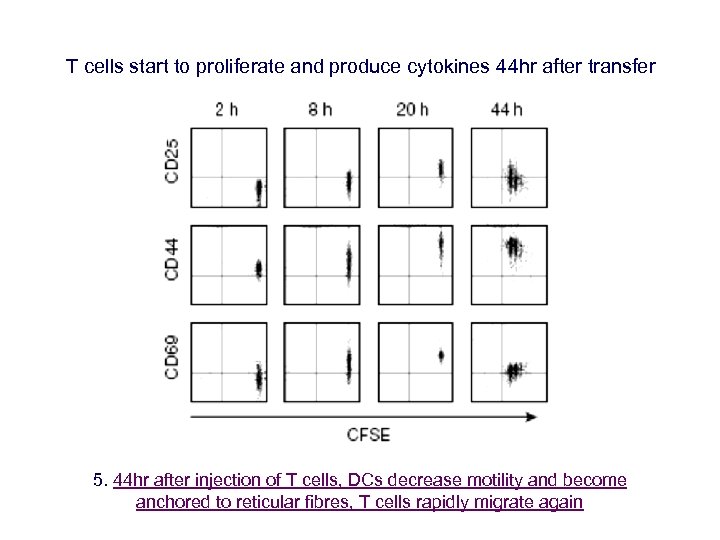 T cells start to proliferate and produce cytokines 44 hr after transfer 5. 44 hr after injection of T cells, DCs decrease motility and become anchored to reticular fibres, T cells rapidly migrate again
T cells start to proliferate and produce cytokines 44 hr after transfer 5. 44 hr after injection of T cells, DCs decrease motility and become anchored to reticular fibres, T cells rapidly migrate again
 More information than is provided by the antigen is exchanged between the DC and T cell DC have a profound influence on the properties of the T cell that develops
More information than is provided by the antigen is exchanged between the DC and T cell DC have a profound influence on the properties of the T cell that develops
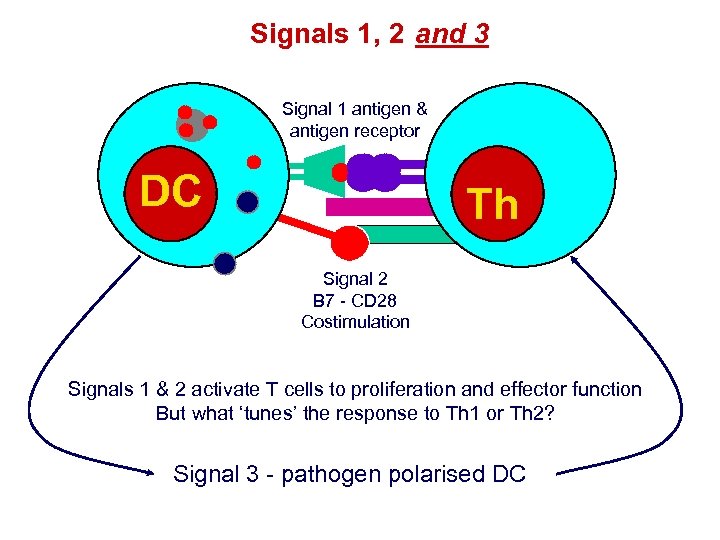 Signals 1, 2 and 3 Signal 1 antigen & antigen receptor DC Th Signal 2 B 7 - CD 28 Costimulation Signals 1 & 2 activate T cells to proliferation and effector function But what ‘tunes’ the response to Th 1 or Th 2? Signal 3 - pathogen polarised DC
Signals 1, 2 and 3 Signal 1 antigen & antigen receptor DC Th Signal 2 B 7 - CD 28 Costimulation Signals 1 & 2 activate T cells to proliferation and effector function But what ‘tunes’ the response to Th 1 or Th 2? Signal 3 - pathogen polarised DC
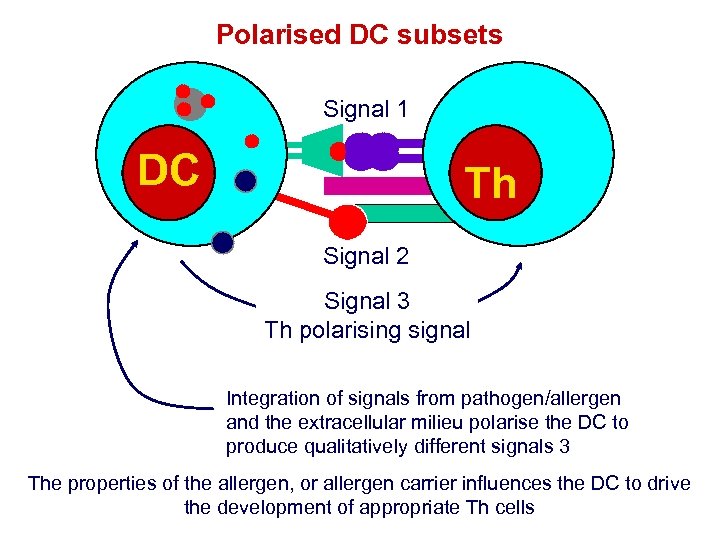 Polarised DC subsets Signal 1 DC Th Signal 2 Signal 3 Th polarising signal Integration of signals from pathogen/allergen and the extracellular milieu polarise the DC to produce qualitatively different signals 3 The properties of the allergen, or allergen carrier influences the DC to drive the development of appropriate Th cells
Polarised DC subsets Signal 1 DC Th Signal 2 Signal 3 Th polarising signal Integration of signals from pathogen/allergen and the extracellular milieu polarise the DC to produce qualitatively different signals 3 The properties of the allergen, or allergen carrier influences the DC to drive the development of appropriate Th cells
 Microbial Patterns Janeway & Medzhitov 2002 Ann Rev Immunol 20 197 -216 Pathogen-associated molecular patterns (PAMPS) • Conserved microbial molecules shared by many pathogens • Include: Bacterial lipopolysaccharides Peptidoglycan Zymosan Flagellin Unmethylated Cp. G DNA Pattern Recognition Receptors (PRR) • Include: Toll like receptors Receptors for apoptotic cells Receptors for opsonins Receptors for coagulation and complement proteins
Microbial Patterns Janeway & Medzhitov 2002 Ann Rev Immunol 20 197 -216 Pathogen-associated molecular patterns (PAMPS) • Conserved microbial molecules shared by many pathogens • Include: Bacterial lipopolysaccharides Peptidoglycan Zymosan Flagellin Unmethylated Cp. G DNA Pattern Recognition Receptors (PRR) • Include: Toll like receptors Receptors for apoptotic cells Receptors for opsonins Receptors for coagulation and complement proteins
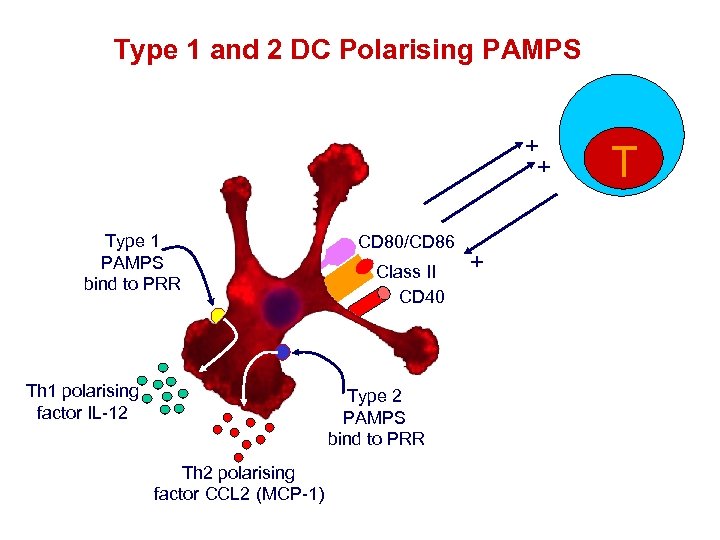 Type 1 and 2 DC Polarising PAMPS + + Type 1 PAMPS bind to PRR Th 1 polarising factor IL-12 CD 80/CD 86 Class II CD 40 Type 2 PAMPS bind to PRR Th 2 polarising factor CCL 2 (MCP-1) + T
Type 1 and 2 DC Polarising PAMPS + + Type 1 PAMPS bind to PRR Th 1 polarising factor IL-12 CD 80/CD 86 Class II CD 40 Type 2 PAMPS bind to PRR Th 2 polarising factor CCL 2 (MCP-1) + T
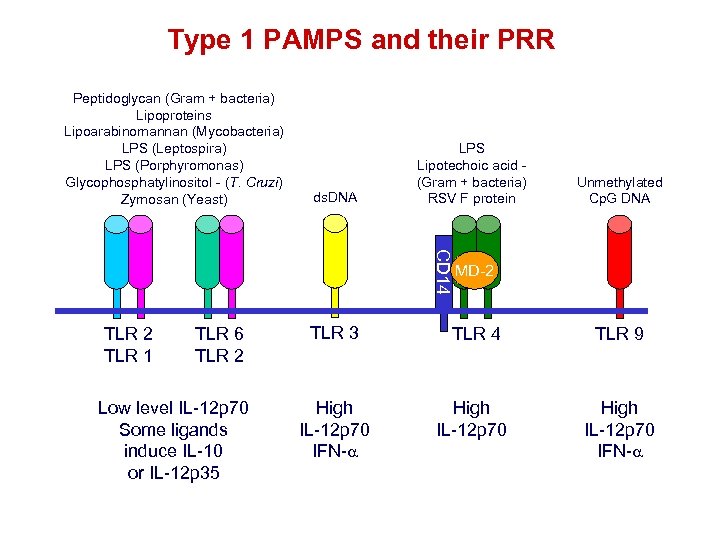 Type 1 PAMPS and their PRR Peptidoglycan (Gram + bacteria) Lipoproteins Lipoarabinomannan (Mycobacteria) LPS (Leptospira) LPS (Porphyromonas) Glycophosphatylinositol - (T. Cruzi) Zymosan (Yeast) ds. DNA LPS Lipotechoic acid (Gram + bacteria) RSV F protein CD 14 TLR 2 TLR 1 TLR 6 TLR 2 Low level IL-12 p 70 Some ligands induce IL-10 or IL-12 p 35 Unmethylated Cp. G DNA MD-2 TLR 3 TLR 4 TLR 9 High IL-12 p 70 IFN-a
Type 1 PAMPS and their PRR Peptidoglycan (Gram + bacteria) Lipoproteins Lipoarabinomannan (Mycobacteria) LPS (Leptospira) LPS (Porphyromonas) Glycophosphatylinositol - (T. Cruzi) Zymosan (Yeast) ds. DNA LPS Lipotechoic acid (Gram + bacteria) RSV F protein CD 14 TLR 2 TLR 1 TLR 6 TLR 2 Low level IL-12 p 70 Some ligands induce IL-10 or IL-12 p 35 Unmethylated Cp. G DNA MD-2 TLR 3 TLR 4 TLR 9 High IL-12 p 70 IFN-a
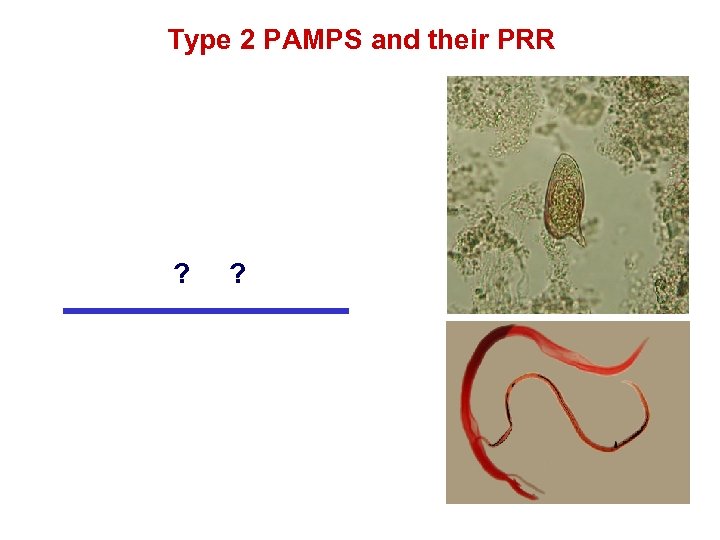 Type 2 PAMPS and their PRR ? ?
Type 2 PAMPS and their PRR ? ?
 Endogenous molecular patterns • Include: Heat shock proteins (HSP 60 HSP 70 GP 96) Extracellular matrix proteins (hyaluronan, fibronectin, fibrinogen) Immune complexes Surfactant protein A Necrotic cell components Pattern Recognition Receptors (PRR) • Include: Toll like receptors Receptors for apoptotic cells Receptors for opsonins Receptors for coagulation and complement proteins
Endogenous molecular patterns • Include: Heat shock proteins (HSP 60 HSP 70 GP 96) Extracellular matrix proteins (hyaluronan, fibronectin, fibrinogen) Immune complexes Surfactant protein A Necrotic cell components Pattern Recognition Receptors (PRR) • Include: Toll like receptors Receptors for apoptotic cells Receptors for opsonins Receptors for coagulation and complement proteins
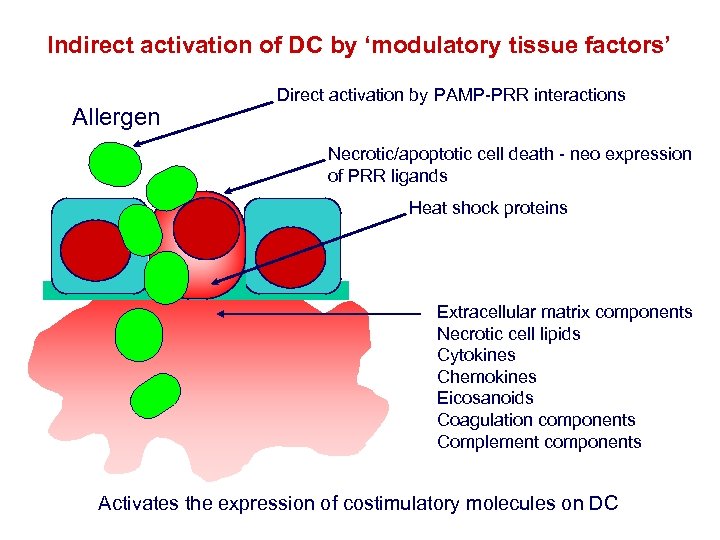 Indirect activation of DC by ‘modulatory tissue factors’ Allergen Direct activation by PAMP-PRR interactions Necrotic/apoptotic cell death - neo expression of PRR ligands Heat shock proteins Extracellular matrix components Necrotic cell lipids Cytokines Chemokines Eicosanoids Coagulation components Complement components Activates the expression of costimulatory molecules on DC
Indirect activation of DC by ‘modulatory tissue factors’ Allergen Direct activation by PAMP-PRR interactions Necrotic/apoptotic cell death - neo expression of PRR ligands Heat shock proteins Extracellular matrix components Necrotic cell lipids Cytokines Chemokines Eicosanoids Coagulation components Complement components Activates the expression of costimulatory molecules on DC
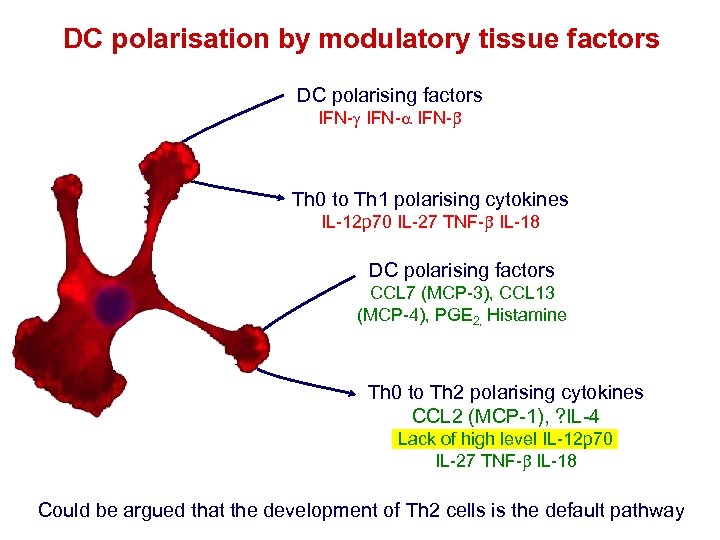 DC polarisation by modulatory tissue factors DC polarising factors IFN-a IFN- Th 0 to Th 1 polarising cytokines IL-12 p 70 IL-27 TNF- IL-18 DC polarising factors CCL 7 (MCP-3), CCL 13 (MCP-4), PGE 2, Histamine Th 0 to Th 2 polarising cytokines CCL 2 (MCP-1), ? IL-4 Lack of high level IL-12 p 70 IL-27 TNF- IL-18 Could be argued that the development of Th 2 cells is the default pathway
DC polarisation by modulatory tissue factors DC polarising factors IFN-a IFN- Th 0 to Th 1 polarising cytokines IL-12 p 70 IL-27 TNF- IL-18 DC polarising factors CCL 7 (MCP-3), CCL 13 (MCP-4), PGE 2, Histamine Th 0 to Th 2 polarising cytokines CCL 2 (MCP-1), ? IL-4 Lack of high level IL-12 p 70 IL-27 TNF- IL-18 Could be argued that the development of Th 2 cells is the default pathway
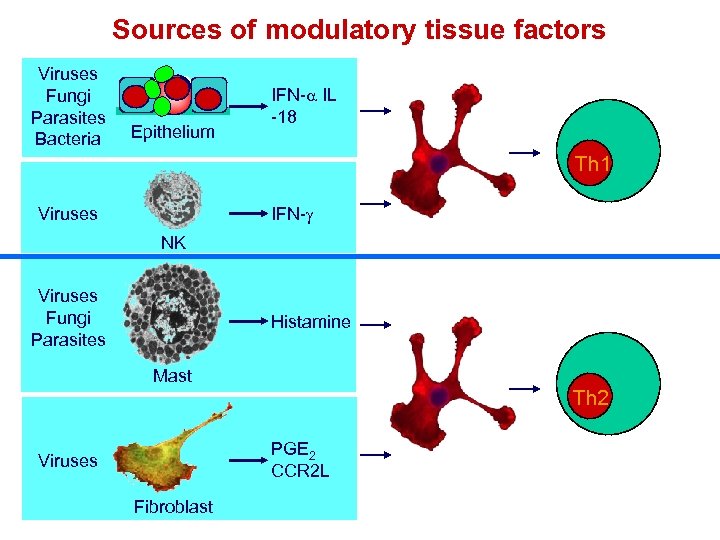 Sources of modulatory tissue factors Viruses Fungi Parasites Bacteria Epithelium IFN-a IL -18 Th 1 Viruses IFN- NK Viruses Fungi Parasites Histamine Mast Th 2 PGE 2 CCR 2 L Viruses Fibroblast
Sources of modulatory tissue factors Viruses Fungi Parasites Bacteria Epithelium IFN-a IL -18 Th 1 Viruses IFN- NK Viruses Fungi Parasites Histamine Mast Th 2 PGE 2 CCR 2 L Viruses Fibroblast
 Explains how Th 2 arise, but… …does not explains why some individuals are allergic and others are not and why the incidence of allergy is increasing. Reduced numbers of IL-12 producing cells? Reduced ability to produce or respond to IL-12? Reduced stimulation of IL-12 by microbial substances? The hygiene hypothesis (Strachan, 1989) Based upon the epidemiology of hay fever “Declining family size, improved household amenities, and higher standards of personal cleanliness have reduced the opportunities for cross-infection in young families This may have resulted in more widespread clinical expression of atopic disease" . . can be interpreted in terms of a failure to microbially modulate default Th 2 responses in childhood
Explains how Th 2 arise, but… …does not explains why some individuals are allergic and others are not and why the incidence of allergy is increasing. Reduced numbers of IL-12 producing cells? Reduced ability to produce or respond to IL-12? Reduced stimulation of IL-12 by microbial substances? The hygiene hypothesis (Strachan, 1989) Based upon the epidemiology of hay fever “Declining family size, improved household amenities, and higher standards of personal cleanliness have reduced the opportunities for cross-infection in young families This may have resulted in more widespread clinical expression of atopic disease" . . can be interpreted in terms of a failure to microbially modulate default Th 2 responses in childhood
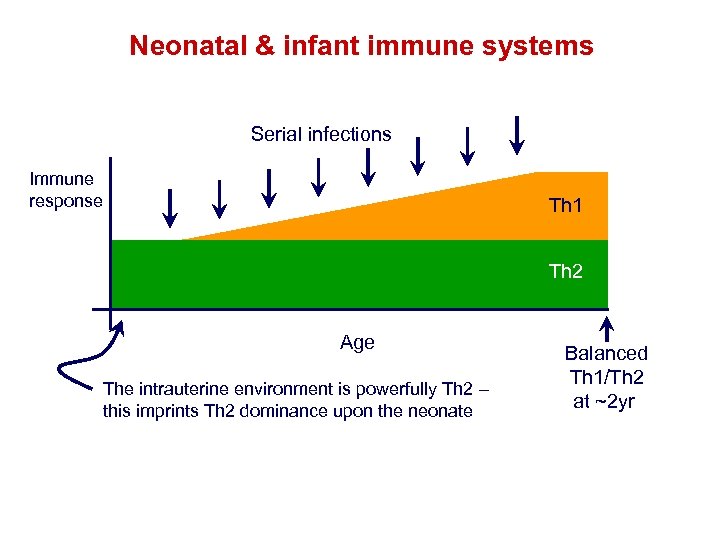 Neonatal & infant immune systems Serial infections Immune response Th 1 Th 2 Age The intrauterine environment is powerfully Th 2 – this imprints Th 2 dominance upon the neonate Balanced Th 1/Th 2 at ~2 yr
Neonatal & infant immune systems Serial infections Immune response Th 1 Th 2 Age The intrauterine environment is powerfully Th 2 – this imprints Th 2 dominance upon the neonate Balanced Th 1/Th 2 at ~2 yr
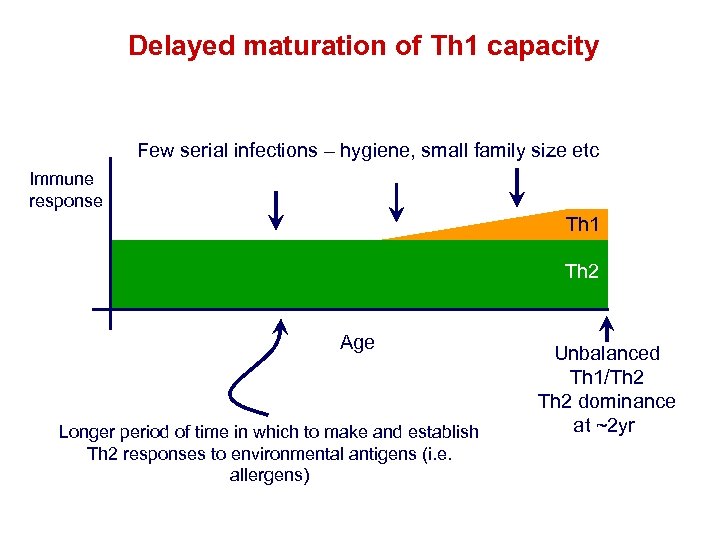 Delayed maturation of Th 1 capacity Few serial infections – hygiene, small family size etc Immune response Th 1 Th 2 Age Longer period of time in which to make and establish Th 2 responses to environmental antigens (i. e. allergens) Unbalanced Th 1/Th 2 dominance at ~2 yr
Delayed maturation of Th 1 capacity Few serial infections – hygiene, small family size etc Immune response Th 1 Th 2 Age Longer period of time in which to make and establish Th 2 responses to environmental antigens (i. e. allergens) Unbalanced Th 1/Th 2 dominance at ~2 yr
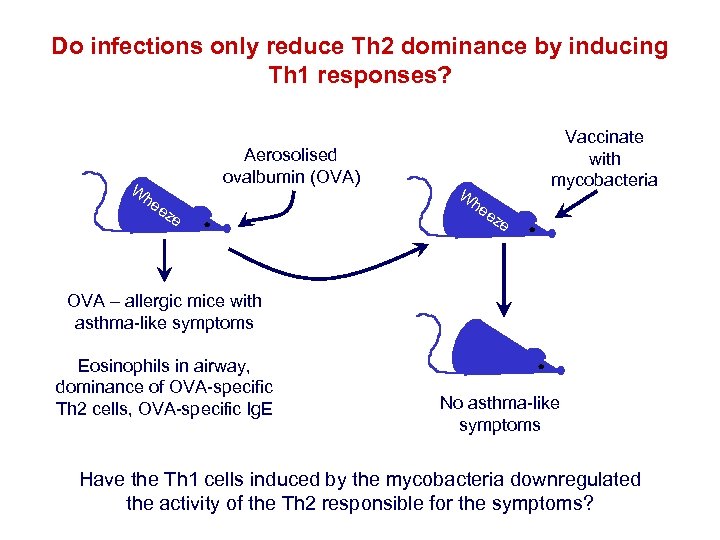 Do infections only reduce Th 2 dominance by inducing Th 1 responses? W Aerosolised ovalbumin (OVA) he ez e Vaccinate with mycobacteria W he ez e OVA – allergic mice with asthma-like symptoms Eosinophils in airway, dominance of OVA-specific Th 2 cells, OVA-specific Ig. E No asthma-like symptoms Have the Th 1 cells induced by the mycobacteria downregulated the activity of the Th 2 responsible for the symptoms?
Do infections only reduce Th 2 dominance by inducing Th 1 responses? W Aerosolised ovalbumin (OVA) he ez e Vaccinate with mycobacteria W he ez e OVA – allergic mice with asthma-like symptoms Eosinophils in airway, dominance of OVA-specific Th 2 cells, OVA-specific Ig. E No asthma-like symptoms Have the Th 1 cells induced by the mycobacteria downregulated the activity of the Th 2 responsible for the symptoms?
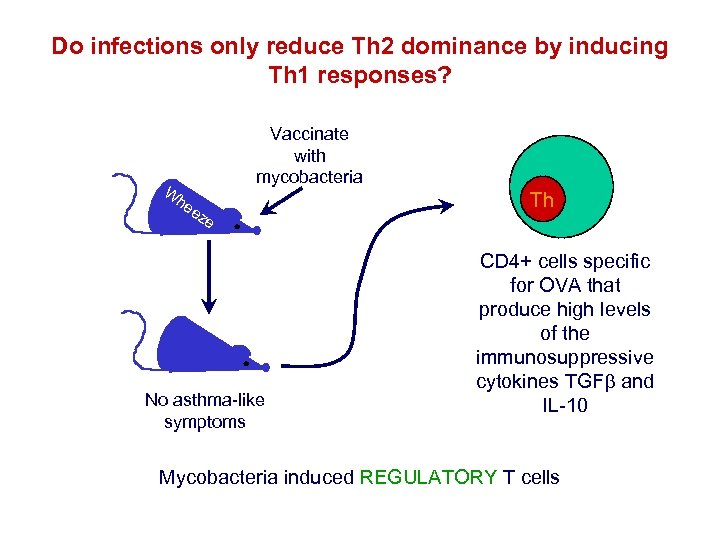 Do infections only reduce Th 2 dominance by inducing Th 1 responses? W Vaccinate with mycobacteria he ez e No asthma-like symptoms Th CD 4+ cells specific for OVA that produce high levels of the immunosuppressive cytokines TGF and IL-10 Mycobacteria induced REGULATORY T cells
Do infections only reduce Th 2 dominance by inducing Th 1 responses? W Vaccinate with mycobacteria he ez e No asthma-like symptoms Th CD 4+ cells specific for OVA that produce high levels of the immunosuppressive cytokines TGF and IL-10 Mycobacteria induced REGULATORY T cells
 Th cell polarisation DC mediated – decision influenced by infection Extracellular milieu - mediated
Th cell polarisation DC mediated – decision influenced by infection Extracellular milieu - mediated
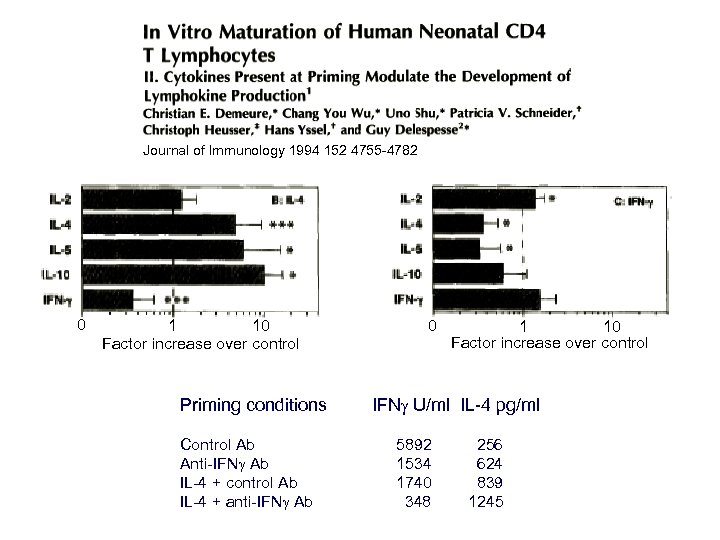 Journal of Immunology 1994 152 4755 -4782 0 1 10 Factor increase over control Priming conditions Control Ab Anti-IFN Ab IL-4 + control Ab IL-4 + anti-IFN Ab 0 1 10 Factor increase over control IFN U/ml IL-4 pg/ml 5892 1534 1740 348 256 624 839 1245
Journal of Immunology 1994 152 4755 -4782 0 1 10 Factor increase over control Priming conditions Control Ab Anti-IFN Ab IL-4 + control Ab IL-4 + anti-IFN Ab 0 1 10 Factor increase over control IFN U/ml IL-4 pg/ml 5892 1534 1740 348 256 624 839 1245
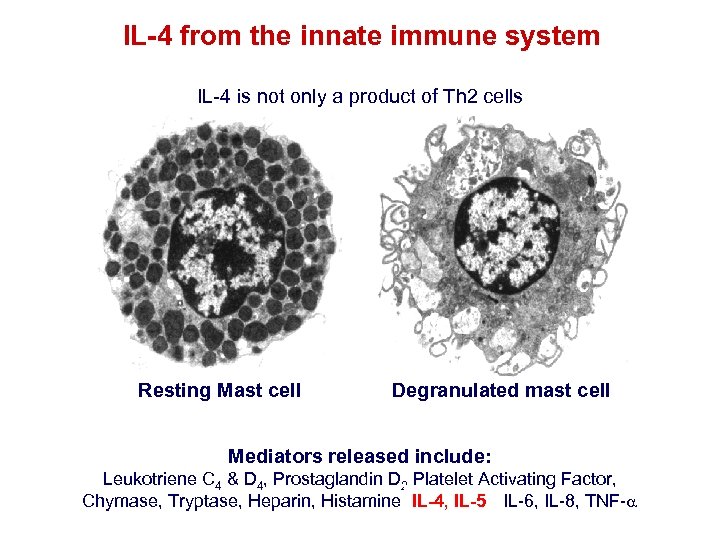 IL-4 from the innate immune system IL-4 is not only a product of Th 2 cells Resting Mast cell Degranulated mast cell Mediators released include: Leukotriene C 4 & D 4, Prostaglandin D 2 Platelet Activating Factor, Chymase, Tryptase, Heparin, Histamine IL-4, IL-5 IL-6, IL-8, TNF-a IL-4, IL-5,
IL-4 from the innate immune system IL-4 is not only a product of Th 2 cells Resting Mast cell Degranulated mast cell Mediators released include: Leukotriene C 4 & D 4, Prostaglandin D 2 Platelet Activating Factor, Chymase, Tryptase, Heparin, Histamine IL-4, IL-5 IL-6, IL-8, TNF-a IL-4, IL-5,
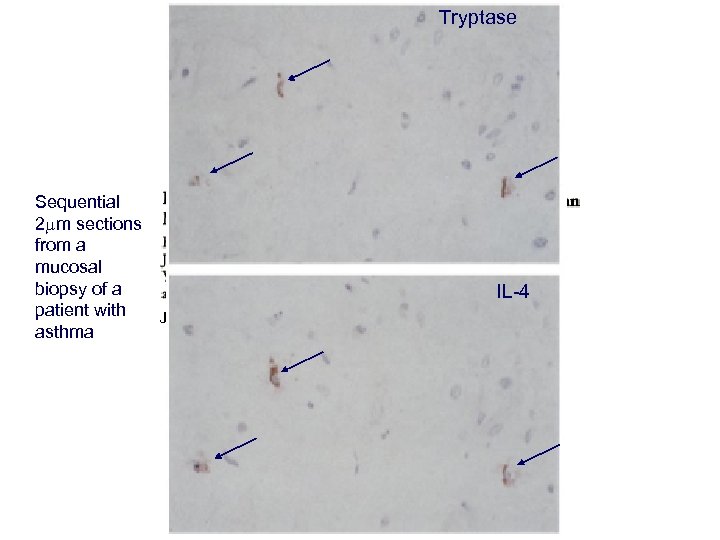 Tryptase Sequential 2 mm sections from a mucosal biopsy of a IL-4 patient with Journal of Experimental Medicine, 1992 176 1381 -1386 asthma
Tryptase Sequential 2 mm sections from a mucosal biopsy of a IL-4 patient with Journal of Experimental Medicine, 1992 176 1381 -1386 asthma
 What properties and characteristics make a substance an allergen? How do these properties disregulate the processes described?
What properties and characteristics make a substance an allergen? How do these properties disregulate the processes described?
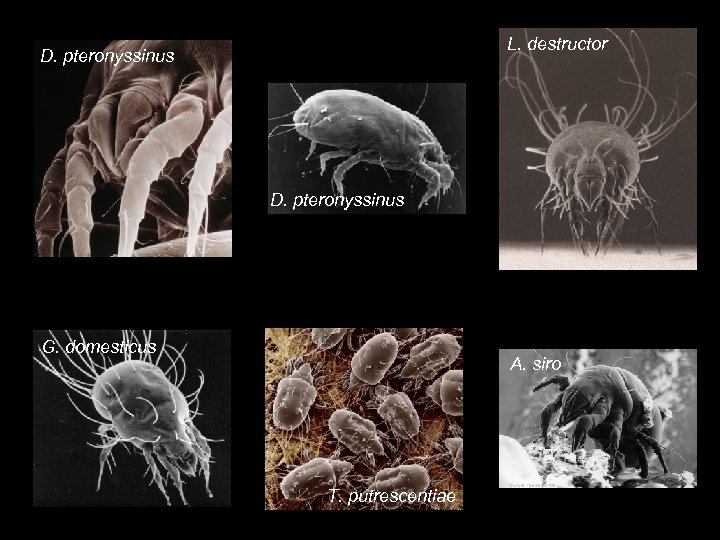 L. destructor D. pteronyssinus G. domesticus A. siro T. putrescentiae
L. destructor D. pteronyssinus G. domesticus A. siro T. putrescentiae
 Allergens of Dermatophagoides pteronyssinus Der p 1 Der p 2 Der p 3 Der p 4 Der p 5 Der p 6 Der p 7 Der p 8 Der p 9 Der p 10 Der p 14 Cysteine protease ? Trypsin (serine protease) Amylase ? Chymotrypsin (serine protease) ? Glutathione transferase Collagenase (serine protease) Tropomyosin Apolipophorin like protein Proteinase allergens are common and widespread: Fungi, insects, plants, parasites, drugs (but…most allergens are not proteases)
Allergens of Dermatophagoides pteronyssinus Der p 1 Der p 2 Der p 3 Der p 4 Der p 5 Der p 6 Der p 7 Der p 8 Der p 9 Der p 10 Der p 14 Cysteine protease ? Trypsin (serine protease) Amylase ? Chymotrypsin (serine protease) ? Glutathione transferase Collagenase (serine protease) Tropomyosin Apolipophorin like protein Proteinase allergens are common and widespread: Fungi, insects, plants, parasites, drugs (but…most allergens are not proteases)
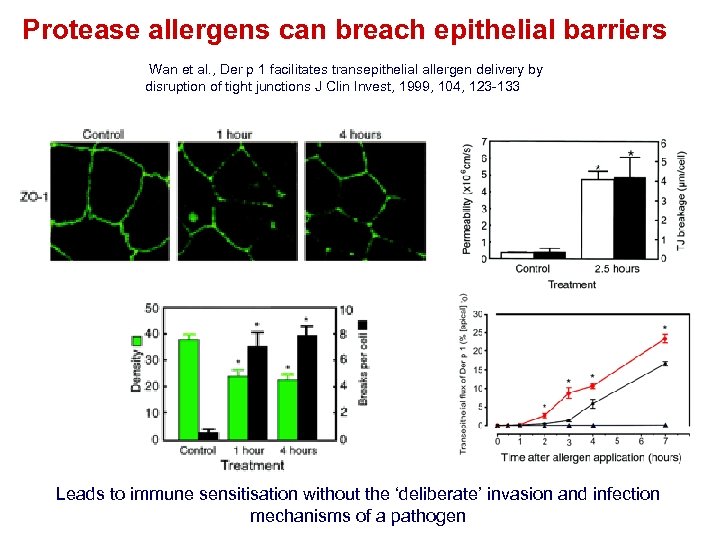 Protease allergens can breach epithelial barriers Wan et al. , Der p 1 facilitates transepithelial allergen delivery by disruption of tight junctions J Clin Invest, 1999, 104, 123 -133 Leads to immune sensitisation without the ‘deliberate’ invasion and infection mechanisms of a pathogen
Protease allergens can breach epithelial barriers Wan et al. , Der p 1 facilitates transepithelial allergen delivery by disruption of tight junctions J Clin Invest, 1999, 104, 123 -133 Leads to immune sensitisation without the ‘deliberate’ invasion and infection mechanisms of a pathogen
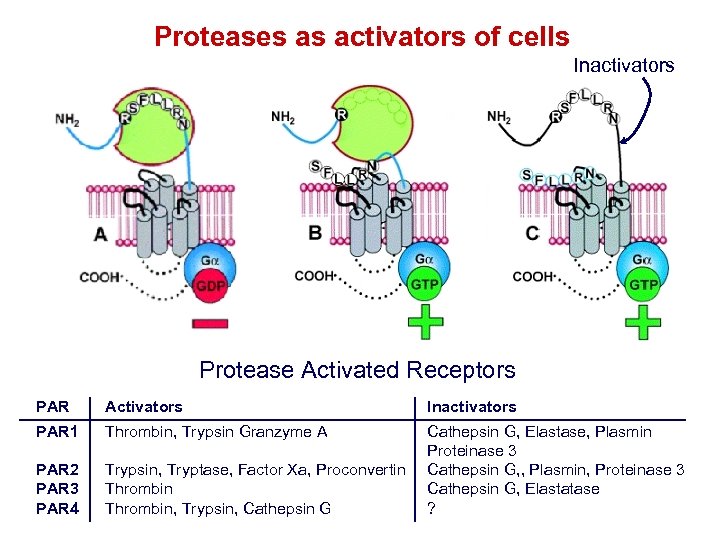 Proteases as activators of cells Inactivators Protease Activated Receptors PAR Activators Inactivators PAR 1 Thrombin, Trypsin Granzyme A PAR 2 PAR 3 PAR 4 Trypsin, Tryptase, Factor Xa, Proconvertin Thrombin, Trypsin, Cathepsin G, Elastase, Plasmin Proteinase 3 Cathepsin G, , Plasmin, Proteinase 3 Cathepsin G, Elastatase ?
Proteases as activators of cells Inactivators Protease Activated Receptors PAR Activators Inactivators PAR 1 Thrombin, Trypsin Granzyme A PAR 2 PAR 3 PAR 4 Trypsin, Tryptase, Factor Xa, Proconvertin Thrombin, Trypsin, Cathepsin G, Elastase, Plasmin Proteinase 3 Cathepsin G, , Plasmin, Proteinase 3 Cathepsin G, Elastatase ?
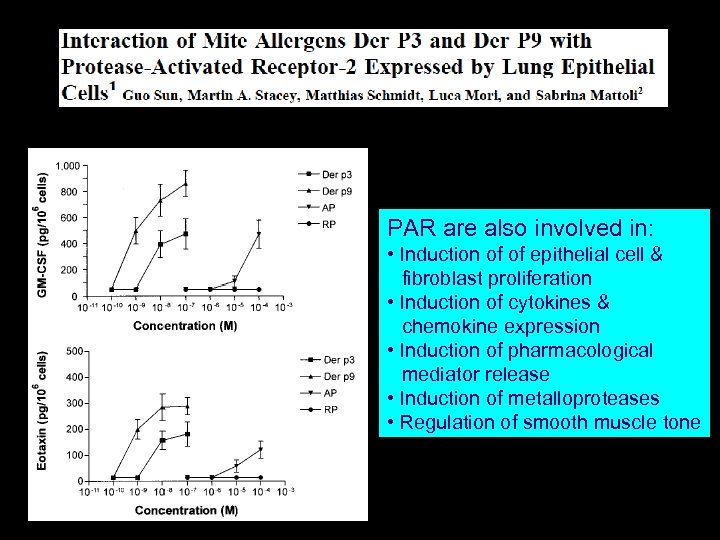 Journal of Immunology 2001 167 1014 -1021 PAR are also involved in: • Induction of of epithelial cell & fibroblast proliferation • Induction of cytokines & chemokine expression • Induction of pharmacological mediator release • Induction of metalloproteases • Regulation of smooth muscle tone
Journal of Immunology 2001 167 1014 -1021 PAR are also involved in: • Induction of of epithelial cell & fibroblast proliferation • Induction of cytokines & chemokine expression • Induction of pharmacological mediator release • Induction of metalloproteases • Regulation of smooth muscle tone
 Do protease allergens induce IL-4 release by Mast cells Resting Mast cell Degranulated mast cell Mediators released include: Leukotriene C 4 & D 4, Prostaglandin D 2 Platelet Activating Factor, Chymase, Tryptase, Heparin, Histamine IL-4, IL-5, IL-6, IL-8, TNF-a IL-4,
Do protease allergens induce IL-4 release by Mast cells Resting Mast cell Degranulated mast cell Mediators released include: Leukotriene C 4 & D 4, Prostaglandin D 2 Platelet Activating Factor, Chymase, Tryptase, Heparin, Histamine IL-4, IL-5, IL-6, IL-8, TNF-a IL-4,
 Journal of Leukocyte Biology 2003, 73 165 -171
Journal of Leukocyte Biology 2003, 73 165 -171
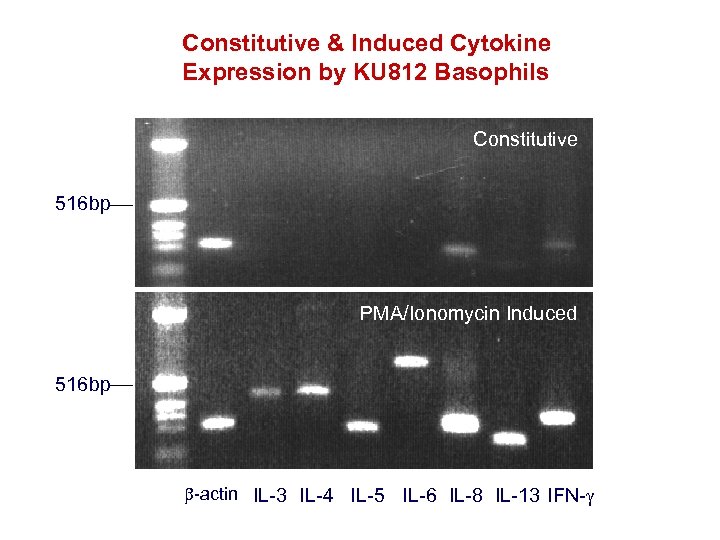 Constitutive & Induced Cytokine Expression by KU 812 Basophils Constitutive 516 bp PMA/Ionomycin Induced 516 bp -actin IL-3 IL-4 IL-5 IL-6 IL-8 IL-13 IFN-
Constitutive & Induced Cytokine Expression by KU 812 Basophils Constitutive 516 bp PMA/Ionomycin Induced 516 bp -actin IL-3 IL-4 IL-5 IL-6 IL-8 IL-13 IFN-
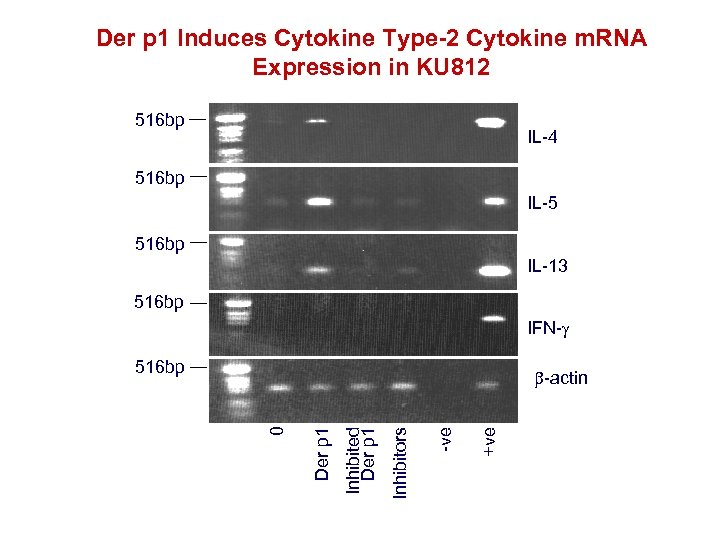 Der p 1 Induces Cytokine Type-2 Cytokine m. RNA Expression in KU 812 516 bp IL-4 516 bp IL-5 516 bp IL-13 516 bp IFN- 516 bp +ve -ve Inhibitors Inhibited Der p 1 0 -actin
Der p 1 Induces Cytokine Type-2 Cytokine m. RNA Expression in KU 812 516 bp IL-4 516 bp IL-5 516 bp IL-13 516 bp IFN- 516 bp +ve -ve Inhibitors Inhibited Der p 1 0 -actin
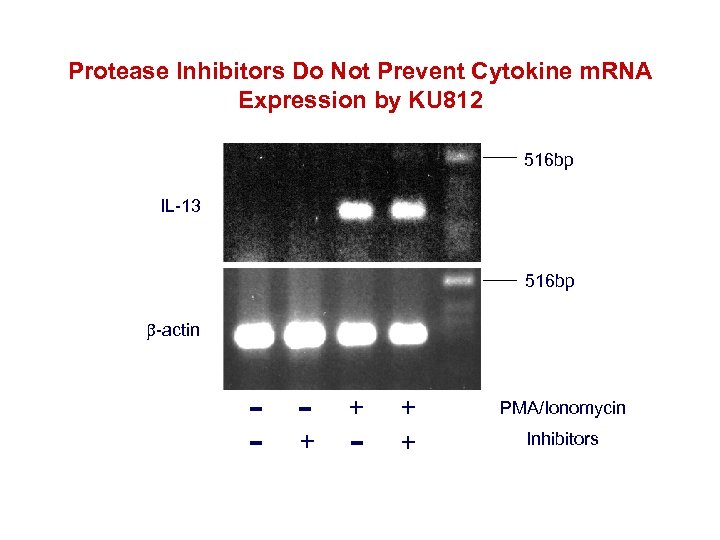 Protease Inhibitors Do Not Prevent Cytokine m. RNA Expression by KU 812 516 bp IL-13 516 bp -actin - - + - + + PMA/Ionomycin Inhibitors
Protease Inhibitors Do Not Prevent Cytokine m. RNA Expression by KU 812 516 bp IL-13 516 bp -actin - - + - + + PMA/Ionomycin Inhibitors
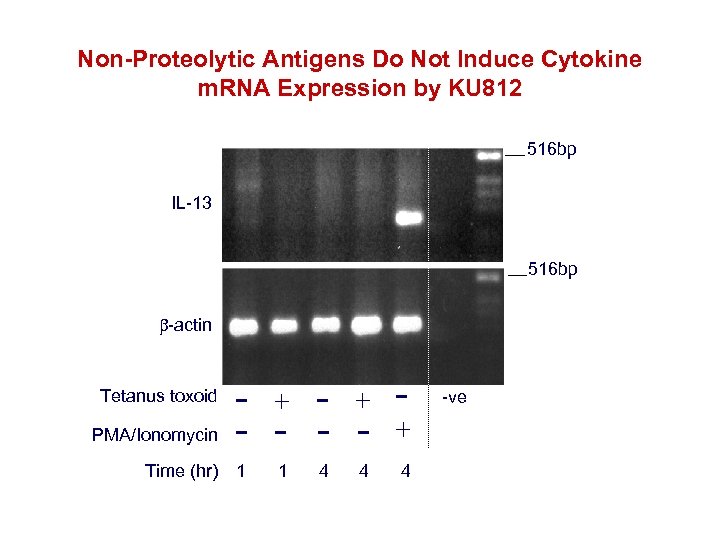 Non-Proteolytic Antigens Do Not Induce Cytokine m. RNA Expression by KU 812 516 bp IL-13 516 bp -actin Time (hr) 1 1 4 4 + PMA/Ionomycin - + - - - Tetanus toxoid 4 -ve
Non-Proteolytic Antigens Do Not Induce Cytokine m. RNA Expression by KU 812 516 bp IL-13 516 bp -actin Time (hr) 1 1 4 4 + PMA/Ionomycin - + - - - Tetanus toxoid 4 -ve
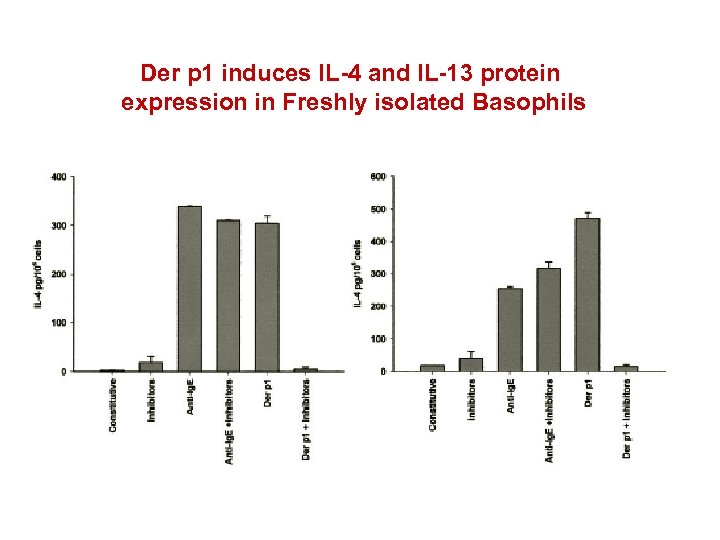 Der p 1 induces IL-4 and IL-13 protein expression in Freshly isolated Basophils
Der p 1 induces IL-4 and IL-13 protein expression in Freshly isolated Basophils
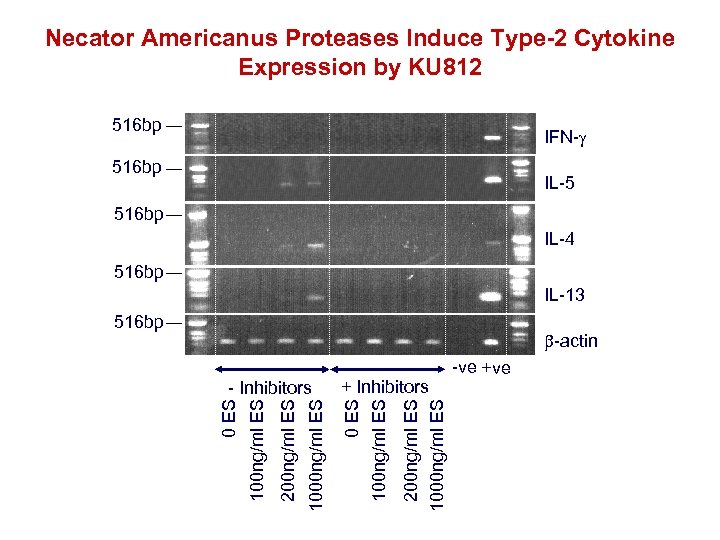 Necator Americanus Proteases Induce Type-2 Cytokine Expression by KU 812 516 bp IFN- 516 bp IL-5 516 bp IL-4 516 bp IL-13 516 bp -actin 200 ng/ml ES 1000 ng/ml ES + Inhibitors 0 ES 100 ng/ml ES 1000 ng/ml ES 200 ng/ml ES 100 ng/ml ES 0 ES - Inhibitors -ve +ve
Necator Americanus Proteases Induce Type-2 Cytokine Expression by KU 812 516 bp IFN- 516 bp IL-5 516 bp IL-4 516 bp IL-13 516 bp -actin 200 ng/ml ES 1000 ng/ml ES + Inhibitors 0 ES 100 ng/ml ES 1000 ng/ml ES 200 ng/ml ES 100 ng/ml ES 0 ES - Inhibitors -ve +ve
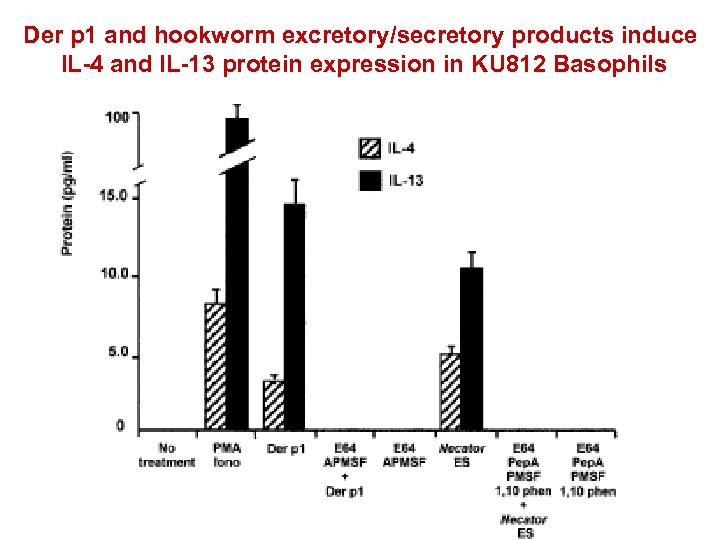 Der p 1 and hookworm excretory/secretory products induce IL-4 and IL-13 protein expression in KU 812 Basophils
Der p 1 and hookworm excretory/secretory products induce IL-4 and IL-13 protein expression in KU 812 Basophils
 The switch to Ig. E
The switch to Ig. E
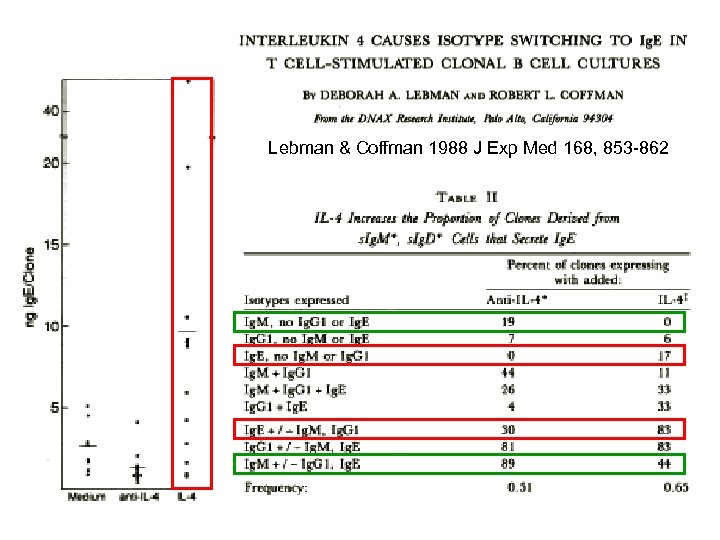 Lebman & Coffman 1988 J Exp Med 168, 853 -862
Lebman & Coffman 1988 J Exp Med 168, 853 -862
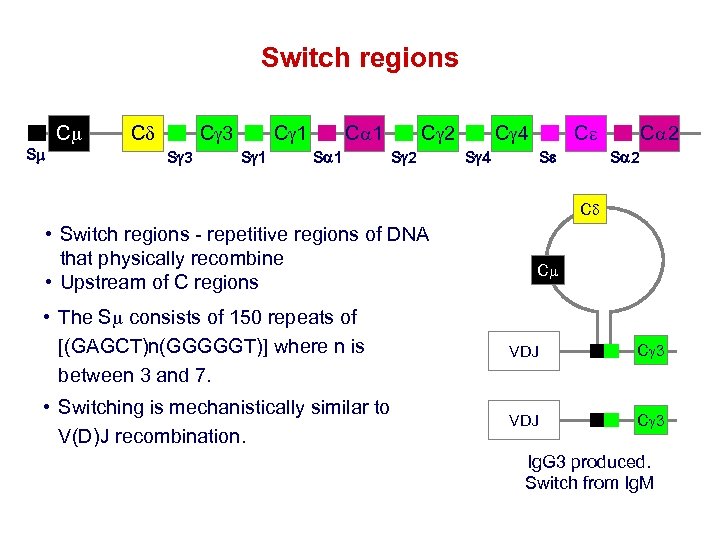 Switch regions Cm Sm Cd C 3 Sg 3 C 1 Sg 1 Ca 1 Sa 1 C 2 Sg 2 C 4 Ce Sg 4 Se Ca 2 Sa 2 Cd • Switch regions - repetitive regions of DNA that physically recombine • Upstream of C regions Cm • The Sm consists of 150 repeats of [(GAGCT)n(GGGGGT)] where n is between 3 and 7. VDJ C 3 • Switching is mechanistically similar to V(D)J recombination. VDJ C 3 Ig. G 3 produced. Switch from Ig. M
Switch regions Cm Sm Cd C 3 Sg 3 C 1 Sg 1 Ca 1 Sa 1 C 2 Sg 2 C 4 Ce Sg 4 Se Ca 2 Sa 2 Cd • Switch regions - repetitive regions of DNA that physically recombine • Upstream of C regions Cm • The Sm consists of 150 repeats of [(GAGCT)n(GGGGGT)] where n is between 3 and 7. VDJ C 3 • Switching is mechanistically similar to V(D)J recombination. VDJ C 3 Ig. G 3 produced. Switch from Ig. M
 Switch recombination to Ig. E A three signal process: 1. Antigen – controls entire process 2. Soluble help via IL-4 or IL-13 from T helper cells 3. Cognate help via CD 40 L from T helper cells
Switch recombination to Ig. E A three signal process: 1. Antigen – controls entire process 2. Soluble help via IL-4 or IL-13 from T helper cells 3. Cognate help via CD 40 L from T helper cells
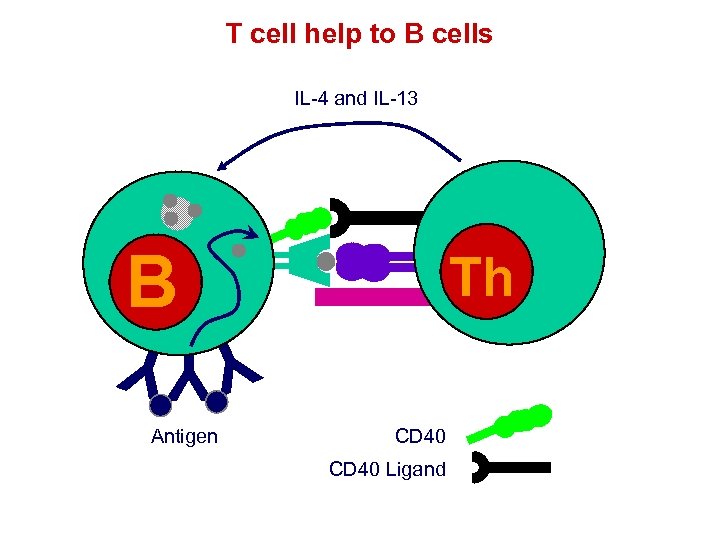 T cell help to B cells IL-4 and IL-13 B Th Th YYY Antigen CD 40 Ligand
T cell help to B cells IL-4 and IL-13 B Th Th YYY Antigen CD 40 Ligand
 Switch recombination to Ig. E A three signal process: 1. Antigen 2. Soluble help via IL-4 or IL-13 from T helper cells 3. Cognate help via CD 40 L from T helper cells
Switch recombination to Ig. E A three signal process: 1. Antigen 2. Soluble help via IL-4 or IL-13 from T helper cells 3. Cognate help via CD 40 L from T helper cells
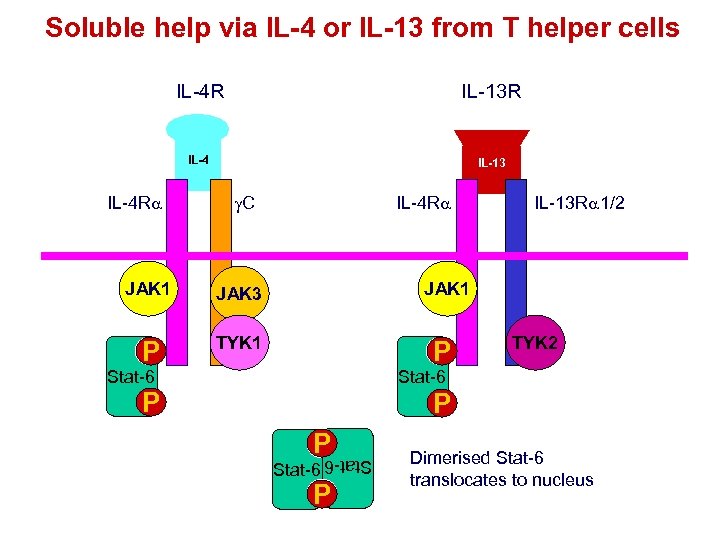 Soluble help via IL-4 or IL-13 from T helper cells IL-4 R IL-13 R IL-4 IL-13 C IL-4 Ra JAK 1 JAK 3 P IL-4 Ra TYK 1 IL-13 Ra 1/2 JAK 1 P Stat-6 P TYK 2 P PDimerised Stat-6 P P P Stat-6 P P Stat-6 translocates to nucleus
Soluble help via IL-4 or IL-13 from T helper cells IL-4 R IL-13 R IL-4 IL-13 C IL-4 Ra JAK 1 JAK 3 P IL-4 Ra TYK 1 IL-13 Ra 1/2 JAK 1 P Stat-6 P TYK 2 P PDimerised Stat-6 P P P Stat-6 P P Stat-6 translocates to nucleus
 Switch recombination to Ig. E A three signal process: 1. Antigen 2. Soluble help via IL-4 or IL-13 from T helper cells 3. Cognate help via CD 40 L from T helper cells
Switch recombination to Ig. E A three signal process: 1. Antigen 2. Soluble help via IL-4 or IL-13 from T helper cells 3. Cognate help via CD 40 L from T helper cells
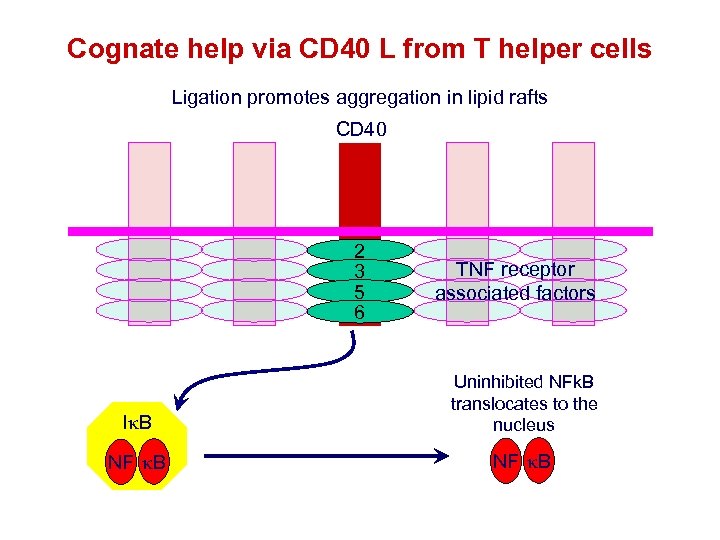 Cognate help via CD 40 L from T helper cells Ligation promotes aggregation in lipid rafts CD 40 2 3 5 6 TNF receptor associated factors Ik. B Uninhibited NFk. B translocates to the Ik. B nucleus NF k. B
Cognate help via CD 40 L from T helper cells Ligation promotes aggregation in lipid rafts CD 40 2 3 5 6 TNF receptor associated factors Ik. B Uninhibited NFk. B translocates to the Ik. B nucleus NF k. B
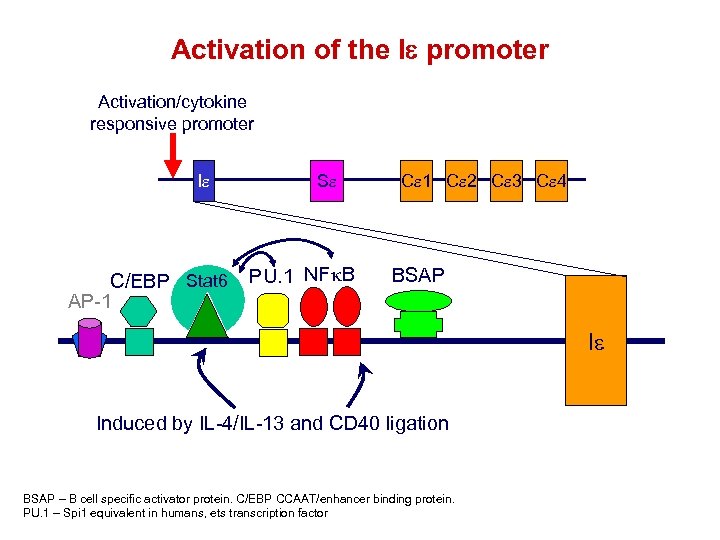 Activation of the Ie promoter Activation/cytokine responsive promoter Ie C/EBP Stat 6 AP-1 Se PU. 1 NFk. B Ce 1 Ce 2 Ce 3 Ce 4 BSAP Ie Induced by IL-4/IL-13 and CD 40 ligation BSAP – B cell specific activator protein. C/EBP CCAAT/enhancer binding protein. PU. 1 – Spi 1 equivalent in humans, ets transcription factor
Activation of the Ie promoter Activation/cytokine responsive promoter Ie C/EBP Stat 6 AP-1 Se PU. 1 NFk. B Ce 1 Ce 2 Ce 3 Ce 4 BSAP Ie Induced by IL-4/IL-13 and CD 40 ligation BSAP – B cell specific activator protein. C/EBP CCAAT/enhancer binding protein. PU. 1 – Spi 1 equivalent in humans, ets transcription factor
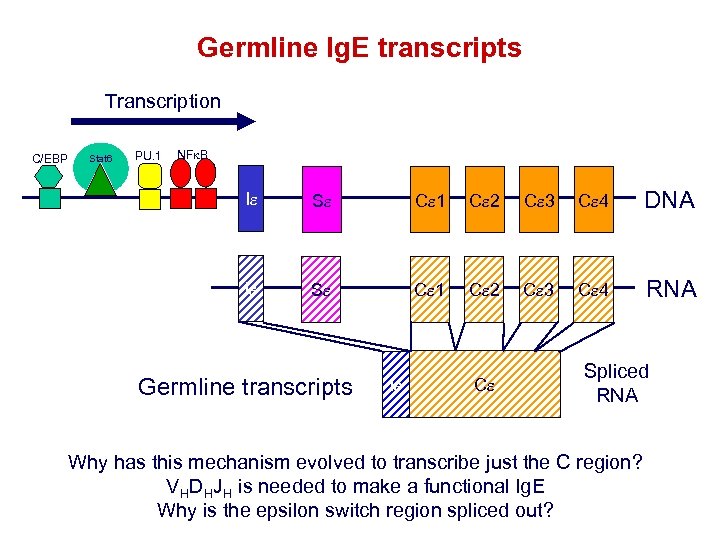 Germline Ig. E transcripts Transcription C/EBP Stat 6 PU. 1 NFk. B Ie Se Ce 1 Ce 2 Ce 3 Ce 4 DNA Ie Se Ce 1 Ce 2 Ce 3 Ce 4 RNA Germline transcripts Ie Ce Spliced RNA Why has this mechanism evolved to transcribe just the C region? VHDHJH is needed to make a functional Ig. E Why is the epsilon switch region spliced out?
Germline Ig. E transcripts Transcription C/EBP Stat 6 PU. 1 NFk. B Ie Se Ce 1 Ce 2 Ce 3 Ce 4 DNA Ie Se Ce 1 Ce 2 Ce 3 Ce 4 RNA Germline transcripts Ie Ce Spliced RNA Why has this mechanism evolved to transcribe just the C region? VHDHJH is needed to make a functional Ig. E Why is the epsilon switch region spliced out?
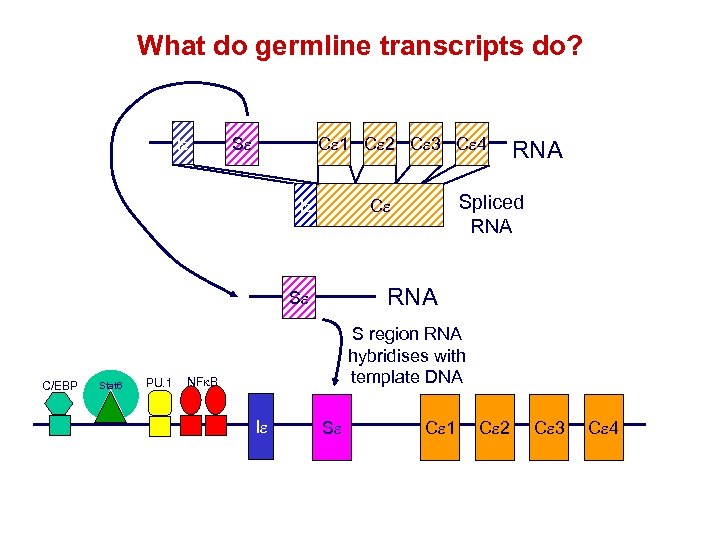 What do germline transcripts do? Ie Se Ce 1 Ce 2 Ce 3 Ce 4 RNA Se C/EBP Stat 6 PU. 1 Spliced RNA Ce Ie RNA S region RNA hybridises with template DNA NFk. B Ie Se Ce 1 Ce 2 Ce 3 Ce 4
What do germline transcripts do? Ie Se Ce 1 Ce 2 Ce 3 Ce 4 RNA Se C/EBP Stat 6 PU. 1 Spliced RNA Ce Ie RNA S region RNA hybridises with template DNA NFk. B Ie Se Ce 1 Ce 2 Ce 3 Ce 4
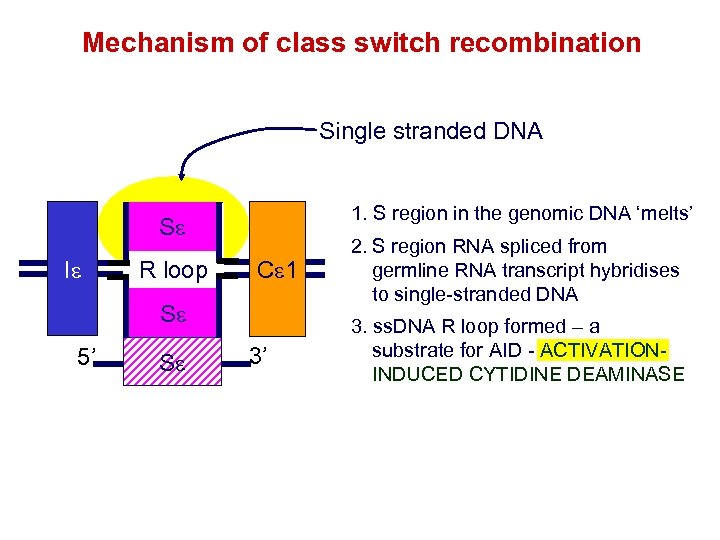 Mechanism of class switch recombination Single stranded DNA 1. S region in the genomic DNA ‘melts’ Se Ie R Se loop Ce 1 Se 5’ Se 3’ 2. S region RNA spliced from germline RNA transcript hybridises to single-stranded DNA 3. ss. DNA R loop formed – a substrate for AID - ACTIVATIONINDUCED CYTIDINE DEAMINASE
Mechanism of class switch recombination Single stranded DNA 1. S region in the genomic DNA ‘melts’ Se Ie R Se loop Ce 1 Se 5’ Se 3’ 2. S region RNA spliced from germline RNA transcript hybridises to single-stranded DNA 3. ss. DNA R loop formed – a substrate for AID - ACTIVATIONINDUCED CYTIDINE DEAMINASE
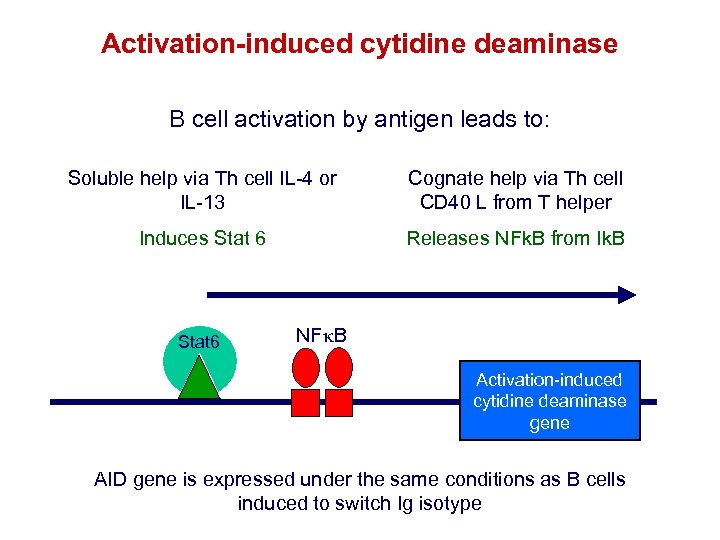 Activation-induced cytidine deaminase B cell activation by antigen leads to: Soluble help via Th cell IL-4 or IL-13 Cognate help via Th cell CD 40 L from T helper Induces Stat 6 Releases NFk. B from Ik. B Stat 6 NFk. B Activation-induced cytidine deaminase gene AID gene is expressed under the same conditions as B cells induced to switch Ig isotype
Activation-induced cytidine deaminase B cell activation by antigen leads to: Soluble help via Th cell IL-4 or IL-13 Cognate help via Th cell CD 40 L from T helper Induces Stat 6 Releases NFk. B from Ik. B Stat 6 NFk. B Activation-induced cytidine deaminase gene AID gene is expressed under the same conditions as B cells induced to switch Ig isotype
 Activation-induced cytidine deaminase • Expressed only in B cells • Involved in isotype class switching & somatic hypermutation • AID knockout mice do not class switch Ig isotype • Ectopic expression in non B cells causes class switch • Mutation in the AID gene can cause hyper Ig. M syndrome • Deaminates cytidine on ss. DNA, i. e. substitutes U for C
Activation-induced cytidine deaminase • Expressed only in B cells • Involved in isotype class switching & somatic hypermutation • AID knockout mice do not class switch Ig isotype • Ectopic expression in non B cells causes class switch • Mutation in the AID gene can cause hyper Ig. M syndrome • Deaminates cytidine on ss. DNA, i. e. substitutes U for C
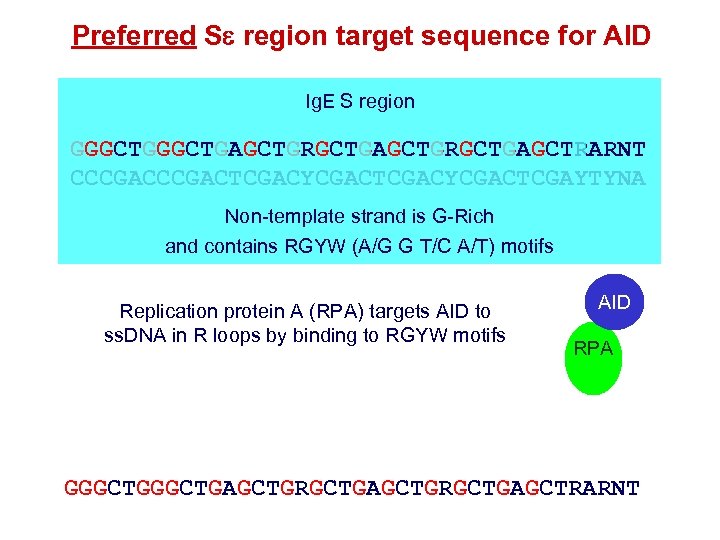 Preferred Se region target sequence for AID Ig. E S region GGGCTGAGCTGRGCTGAGCTRARNT CCCGACTCGACYCGACTCGAYTYNA Non-template strand is G-Rich and contains RGYW (A/G G T/C A/T) motifs Replication protein A (RPA) targets AID to ss. DNA in R loops by binding to RGYW motifs AID RPA GGGCTGAGCTGRGCTGAGCTRARNT
Preferred Se region target sequence for AID Ig. E S region GGGCTGAGCTGRGCTGAGCTRARNT CCCGACTCGACYCGACTCGAYTYNA Non-template strand is G-Rich and contains RGYW (A/G G T/C A/T) motifs Replication protein A (RPA) targets AID to ss. DNA in R loops by binding to RGYW motifs AID RPA GGGCTGAGCTGRGCTGAGCTRARNT
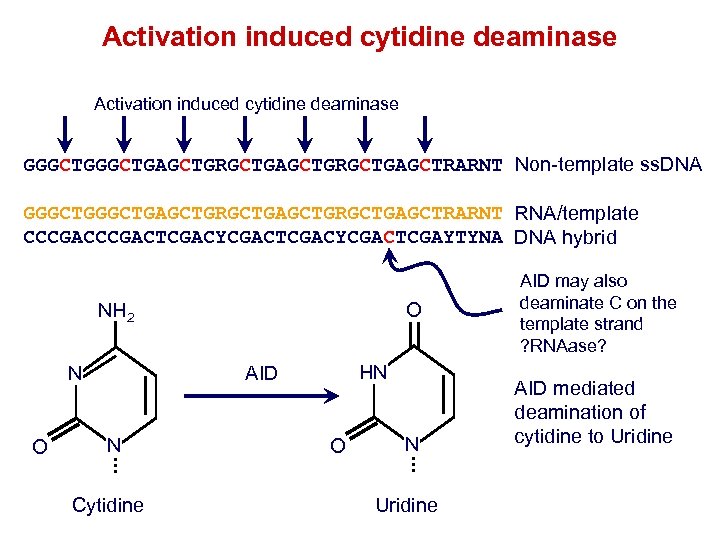 Activation induced cytidine deaminase GGGCTGAGCTGRGCTGAGCTRARNT Non-template ss. DNA GGGCTGAGCTGRGCTGAGCTRARNT RNA/template CCCGACTCGACYCGACTCGAYTYNA DNA hybrid O NH 2 O HN AID N N Cytidine O N Uridine AID may also deaminate C on the template strand ? RNAase? AID mediated deamination of cytidine to Uridine
Activation induced cytidine deaminase GGGCTGAGCTGRGCTGAGCTRARNT Non-template ss. DNA GGGCTGAGCTGRGCTGAGCTRARNT RNA/template CCCGACTCGACYCGACTCGAYTYNA DNA hybrid O NH 2 O HN AID N N Cytidine O N Uridine AID may also deaminate C on the template strand ? RNAase? AID mediated deamination of cytidine to Uridine
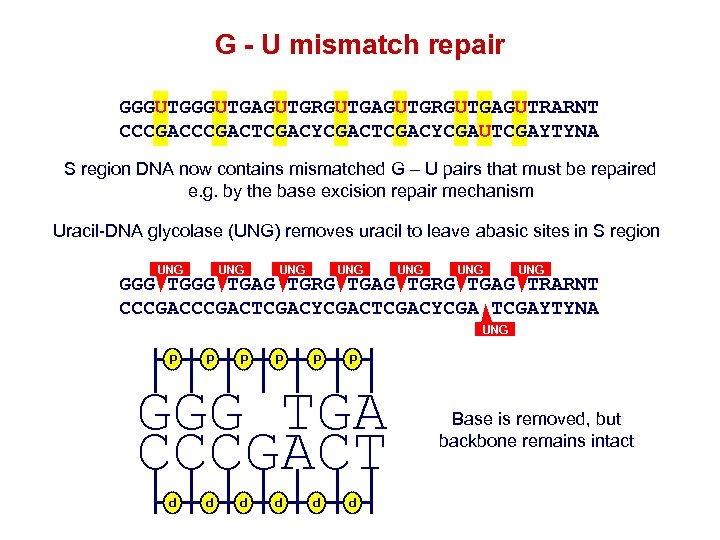 G - U mismatch repair GGGUTGAGUTGRGUTGAGUTRARNT CCCGACTCGACYCGAUTCGAYTYNA S region DNA now contains mismatched G – U pairs that must be repaired e. g. by the base excision repair mechanism Uracil-DNA glycolase (UNG) removes uracil to leave abasic sites in S region UNG UNG GGGUTGAGUTGRGUTGAGUTRARNT CCCGACTCGACYCGAUTCGAYTYNA UNG P P P GGGUTGA CCCGACT Base is removed, but backbone remains intact P P P
G - U mismatch repair GGGUTGAGUTGRGUTGAGUTRARNT CCCGACTCGACYCGAUTCGAYTYNA S region DNA now contains mismatched G – U pairs that must be repaired e. g. by the base excision repair mechanism Uracil-DNA glycolase (UNG) removes uracil to leave abasic sites in S region UNG UNG GGGUTGAGUTGRGUTGAGUTRARNT CCCGACTCGACYCGAUTCGAYTYNA UNG P P P GGGUTGA CCCGACT Base is removed, but backbone remains intact P P P
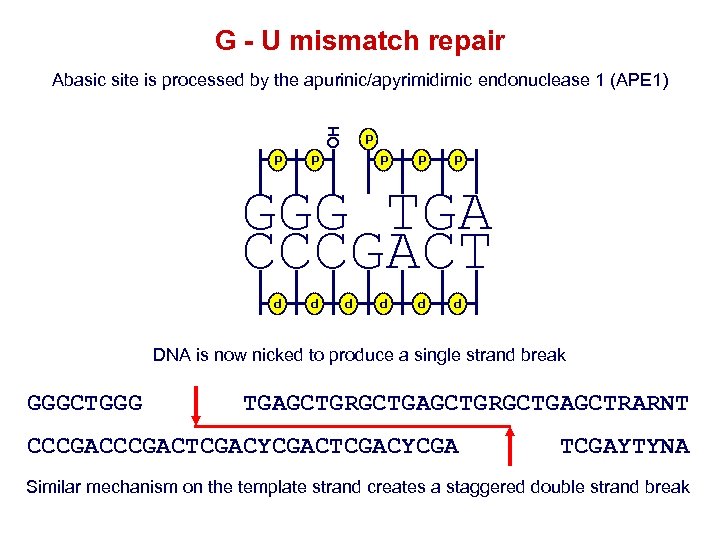 G - U mismatch repair Abasic site is processed by the apurinic/apyrimidimic endonuclease 1 (APE 1) OH APE 1 P P P P GGGUTGA CCCGACT P P P DNA is now nicked to produce a single strand break GGGCTGGGU TGAGCTGRGCTGAGCTRARNT APE 1 CCCGACTCGACYCGAU TCGAYTYNA GGGUTGAGUTGRGUTGAGUTRARNT CCCGACTCGACYCGAUTCGAYTYNA Similar mechanism on the template strand creates a staggered double strand break APE 1
G - U mismatch repair Abasic site is processed by the apurinic/apyrimidimic endonuclease 1 (APE 1) OH APE 1 P P P P GGGUTGA CCCGACT P P P DNA is now nicked to produce a single strand break GGGCTGGGU TGAGCTGRGCTGAGCTRARNT APE 1 CCCGACTCGACYCGAU TCGAYTYNA GGGUTGAGUTGRGUTGAGUTRARNT CCCGACTCGACYCGAUTCGAYTYNA Similar mechanism on the template strand creates a staggered double strand break APE 1
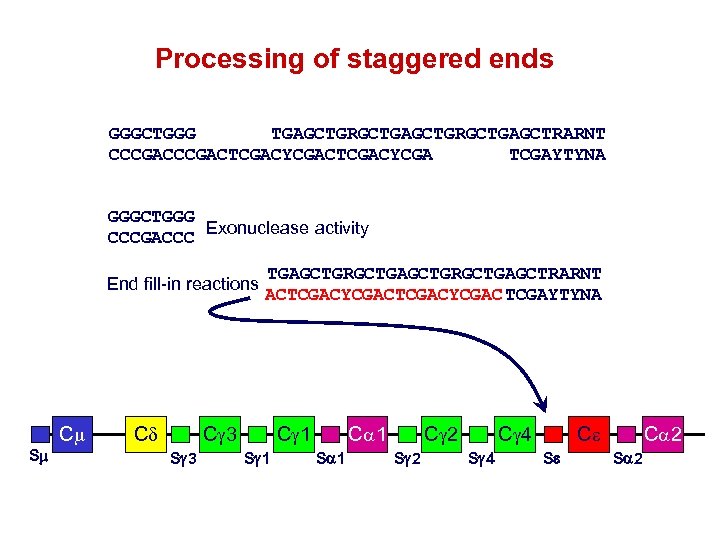 Processing of staggered ends GGGCTGGG TGAGCTGRGCTGAGCTRARNT CCCGACTCGACYCGA TCGAYTYNA GGGCTGGG Exonuclease activity CCCGACTCGACYCGA End fill-in reactions Cm Sm Cd TGAGCTGRGCTGAGCTRARNT ACTCGACYCGAC TCGAYTYNA C 3 Sg 3 C 1 Sg 1 Ca 1 Sa 1 C 2 Sg 2 C 4 Sg 4 Ce Se Ca 2 Sa 2
Processing of staggered ends GGGCTGGG TGAGCTGRGCTGAGCTRARNT CCCGACTCGACYCGA TCGAYTYNA GGGCTGGG Exonuclease activity CCCGACTCGACYCGA End fill-in reactions Cm Sm Cd TGAGCTGRGCTGAGCTRARNT ACTCGACYCGAC TCGAYTYNA C 3 Sg 3 C 1 Sg 1 Ca 1 Sa 1 C 2 Sg 2 C 4 Sg 4 Ce Se Ca 2 Sa 2
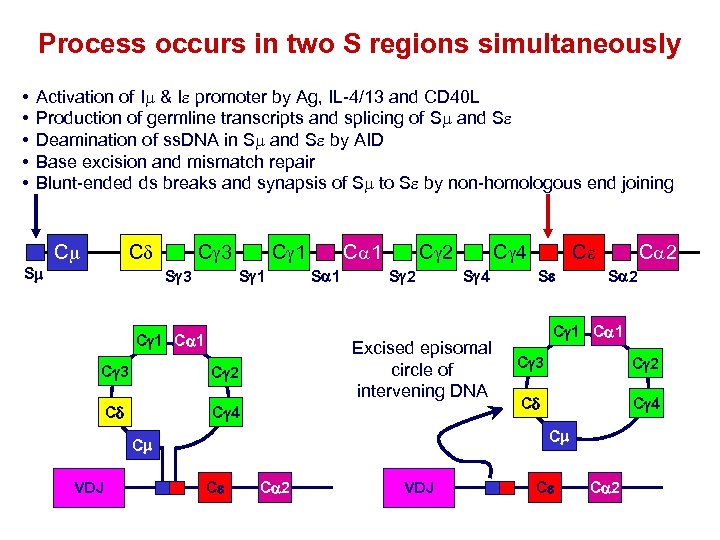 Process occurs in two S regions simultaneously • • • Activation of Im & Ie promoter by Ag, IL-4/13 and CD 40 L Production of germline transcripts and splicing of Sm and Se Deamination of ss. DNA in Sm and Se by AID Base excision and mismatch repair Blunt-ended ds breaks and synapsis of Sm to Se by non-homologous end joining Cm Cd Sm C 3 Sg 3 C 1 Sg 1 Ca 1 Cg 3 Sa 1 C 2 Sg 2 Cg 4 Sg 4 Ce Se Ca 2 Sa 2 Cg 1 Ca 1 Cg 3 Cg 2 Cd Cg 4 Cm Cm VDJ C 4 Excised episomal circle of intervening DNA Cg 2 Cd Ca 1 Ce Ca 2 VDJ Ce Ca 2
Process occurs in two S regions simultaneously • • • Activation of Im & Ie promoter by Ag, IL-4/13 and CD 40 L Production of germline transcripts and splicing of Sm and Se Deamination of ss. DNA in Sm and Se by AID Base excision and mismatch repair Blunt-ended ds breaks and synapsis of Sm to Se by non-homologous end joining Cm Cd Sm C 3 Sg 3 C 1 Sg 1 Ca 1 Cg 3 Sa 1 C 2 Sg 2 Cg 4 Sg 4 Ce Se Ca 2 Sa 2 Cg 1 Ca 1 Cg 3 Cg 2 Cd Cg 4 Cm Cm VDJ C 4 Excised episomal circle of intervening DNA Cg 2 Cd Ca 1 Ce Ca 2 VDJ Ce Ca 2
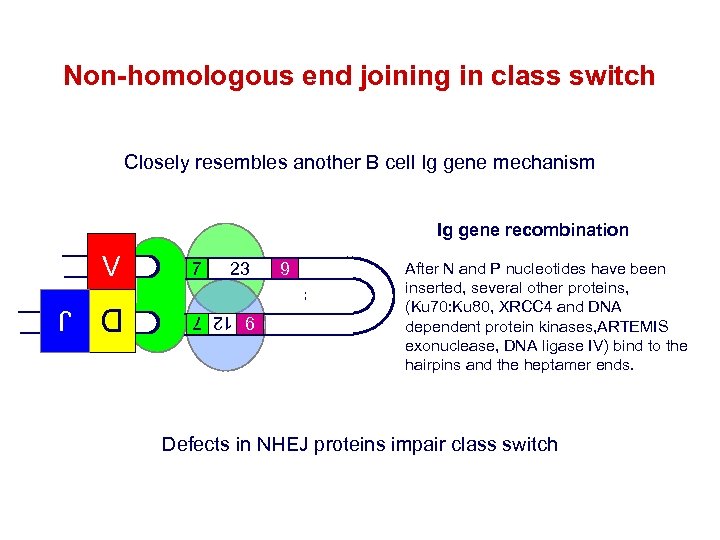 Non-homologous end joining in class switch Closely resembles another B cell Ig gene mechanism Ig gene recombination 7 23 9 12 7 V 9 After N and P nucleotides have been inserted, several other proteins, (Ku 70: Ku 80, XRCC 4 and DNA dependent protein kinases, ARTEMIS exonuclease, DNA ligase IV) bind to the hairpins and the heptamer ends. Defects in NHEJ proteins impair class switch D J
Non-homologous end joining in class switch Closely resembles another B cell Ig gene mechanism Ig gene recombination 7 23 9 12 7 V 9 After N and P nucleotides have been inserted, several other proteins, (Ku 70: Ku 80, XRCC 4 and DNA dependent protein kinases, ARTEMIS exonuclease, DNA ligase IV) bind to the hairpins and the heptamer ends. Defects in NHEJ proteins impair class switch D J
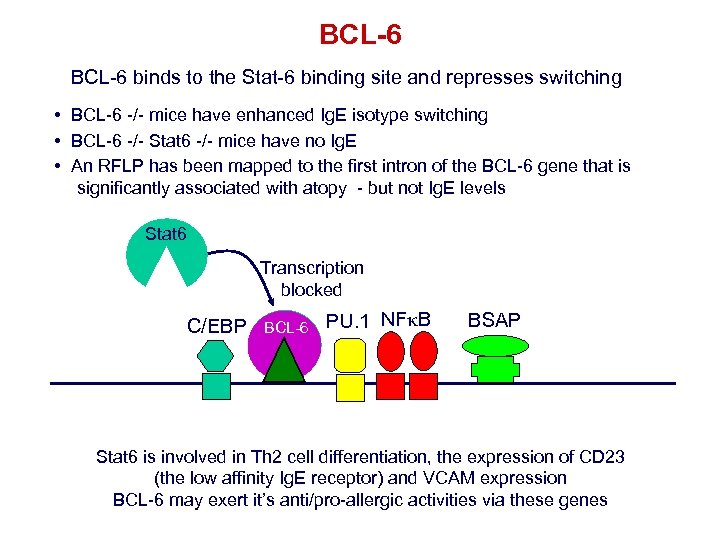 BCL-6 binds to the Stat-6 binding site and represses switching • BCL-6 -/- mice have enhanced Ig. E isotype switching • BCL-6 -/- Stat 6 -/- mice have no Ig. E • An RFLP has been mapped to the first intron of the BCL-6 gene that is significantly associated with atopy - but not Ig. E levels Stat 6 BCL-6 Transcription blocked BCL-6 C/EBP Stat 6 PU. 1 NFk. B BSAP Stat 6 is involved in Th 2 cell differentiation, the expression of CD 23 (the low affinity Ig. E receptor) and VCAM expression BCL-6 may exert it’s anti/pro-allergic activities via these genes
BCL-6 binds to the Stat-6 binding site and represses switching • BCL-6 -/- mice have enhanced Ig. E isotype switching • BCL-6 -/- Stat 6 -/- mice have no Ig. E • An RFLP has been mapped to the first intron of the BCL-6 gene that is significantly associated with atopy - but not Ig. E levels Stat 6 BCL-6 Transcription blocked BCL-6 C/EBP Stat 6 PU. 1 NFk. B BSAP Stat 6 is involved in Th 2 cell differentiation, the expression of CD 23 (the low affinity Ig. E receptor) and VCAM expression BCL-6 may exert it’s anti/pro-allergic activities via these genes
 Additional areas to think about Can’t get over a 2. 2 mark without showing evidence of outside reading in answers
Additional areas to think about Can’t get over a 2. 2 mark without showing evidence of outside reading in answers
 • Relationship between isotype switch, somatic hypermutation and proliferation of B cells in the germinal centre • What is the relationship between the deliberately mutagenic mechanisms of isotype switch and somatic hypermutation in B cells and the propensity of B cells to form tumours • Where are the holes in the ‘skew to Th 2’ model of allergy? • What are allergic responses really for?
• Relationship between isotype switch, somatic hypermutation and proliferation of B cells in the germinal centre • What is the relationship between the deliberately mutagenic mechanisms of isotype switch and somatic hypermutation in B cells and the propensity of B cells to form tumours • Where are the holes in the ‘skew to Th 2’ model of allergy? • What are allergic responses really for?
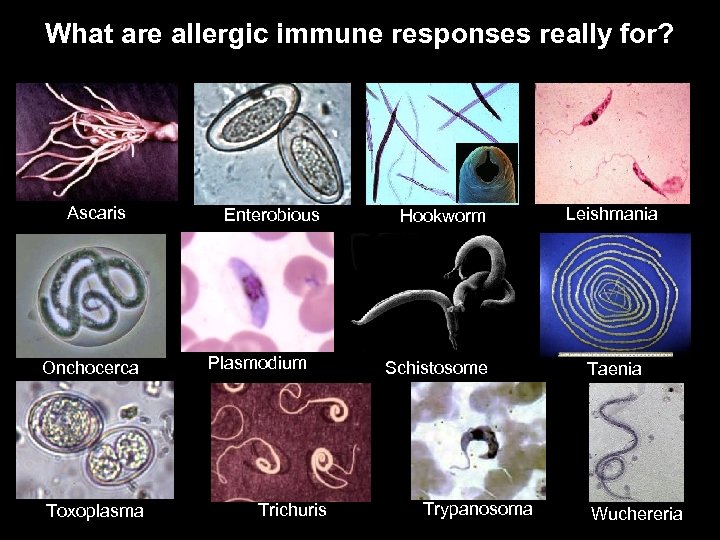 What are allergic immune responses really for? Ascaris Onchocerca Toxoplasma Enterobious Plasmodium Trichuris Hookworm Schistosome Trypanosoma Leishmania Taenia Wuchereria
What are allergic immune responses really for? Ascaris Onchocerca Toxoplasma Enterobious Plasmodium Trichuris Hookworm Schistosome Trypanosoma Leishmania Taenia Wuchereria
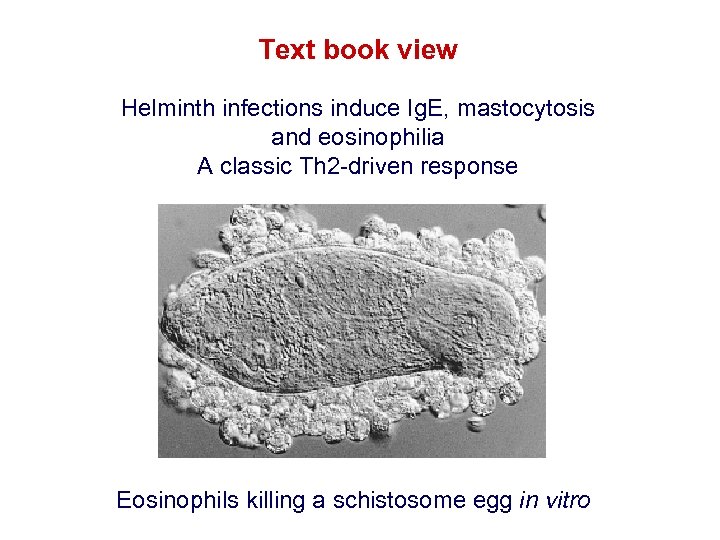 Text book view Helminth infections induce Ig. E, mastocytosis and eosinophilia A classic Th 2 -driven response Eosinophils killing a schistosome egg in vitro
Text book view Helminth infections induce Ig. E, mastocytosis and eosinophilia A classic Th 2 -driven response Eosinophils killing a schistosome egg in vitro
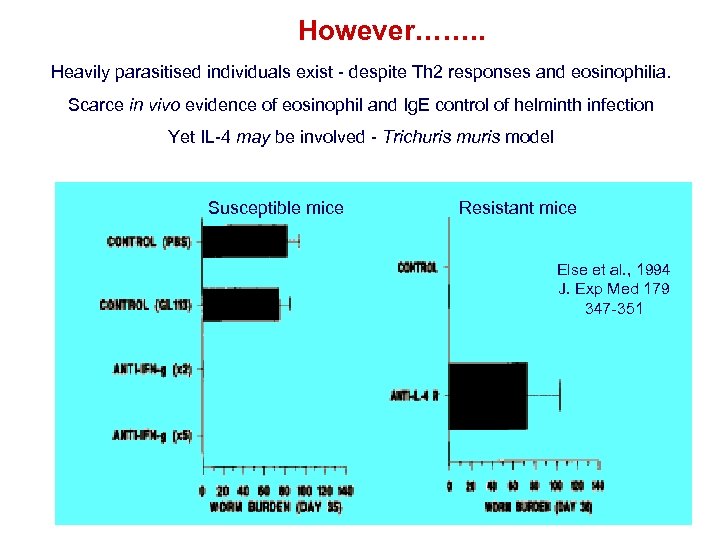 However……. . Heavily parasitised individuals exist - despite Th 2 responses and eosinophilia. Scarce in vivo evidence of eosinophil and Ig. E control of helminth infection Yet IL-4 may be involved - Trichuris model Susceptible mice Resistant mice Else et al. , 1994 J. Exp Med 179 347 -351
However……. . Heavily parasitised individuals exist - despite Th 2 responses and eosinophilia. Scarce in vivo evidence of eosinophil and Ig. E control of helminth infection Yet IL-4 may be involved - Trichuris model Susceptible mice Resistant mice Else et al. , 1994 J. Exp Med 179 347 -351
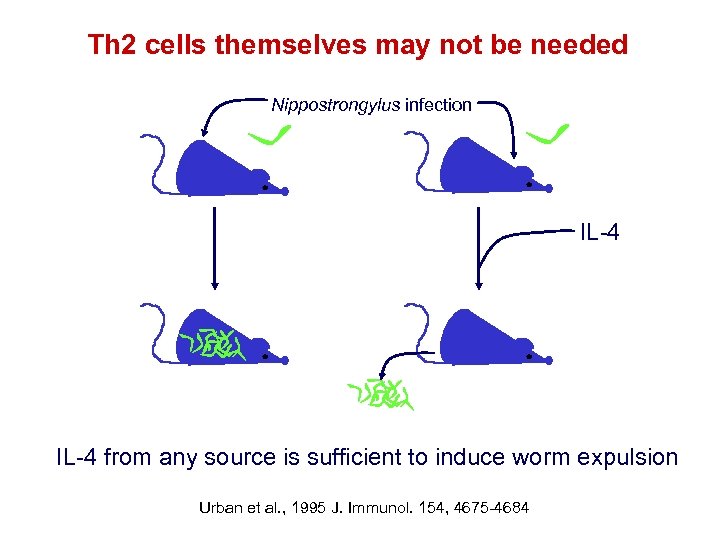 Th 2 cells themselves may not be needed Nippostrongylus infection IL-4 from any source is sufficient to induce worm expulsion Urban et al. , 1995 J. Immunol. 154, 4675 -4684
Th 2 cells themselves may not be needed Nippostrongylus infection IL-4 from any source is sufficient to induce worm expulsion Urban et al. , 1995 J. Immunol. 154, 4675 -4684


