d742af530745b5265c5f07817fbbf6cd.ppt
- Количество слайдов: 45
 MECCANISMI DI RILASCIO DI FARMACI DA MATRICI POLIMERICHE MARIO GRASSI UNIVERSITA’ di TRIESTE Dipartimento di Ingegneria Chimica e dei Materiali
MECCANISMI DI RILASCIO DI FARMACI DA MATRICI POLIMERICHE MARIO GRASSI UNIVERSITA’ di TRIESTE Dipartimento di Ingegneria Chimica e dei Materiali
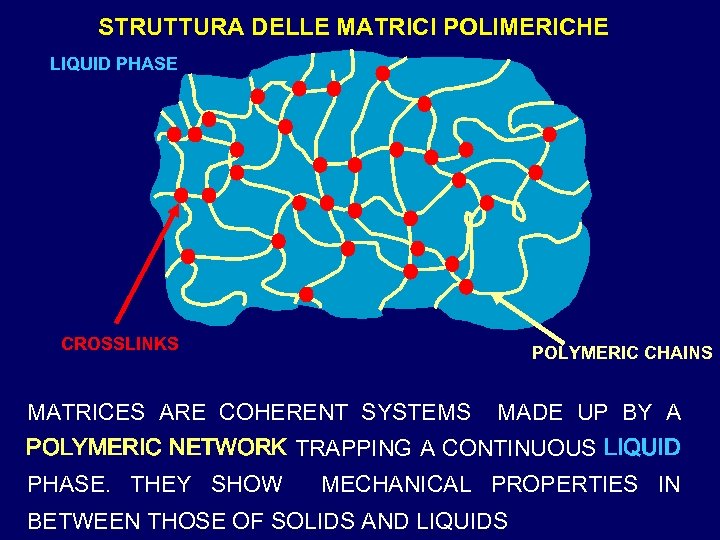 STRUTTURA DELLE MATRICI POLIMERICHE LIQUID PHASE CROSSLINKS POLYMERIC CHAINS MATRICES ARE COHERENT SYSTEMS MADE UP BY A POLYMERIC NETWORK TRAPPING A CONTINUOUS LIQUID PHASE. THEY SHOW MECHANICAL PROPERTIES IN BETWEEN THOSE OF SOLIDS AND LIQUIDS
STRUTTURA DELLE MATRICI POLIMERICHE LIQUID PHASE CROSSLINKS POLYMERIC CHAINS MATRICES ARE COHERENT SYSTEMS MADE UP BY A POLYMERIC NETWORK TRAPPING A CONTINUOUS LIQUID PHASE. THEY SHOW MECHANICAL PROPERTIES IN BETWEEN THOSE OF SOLIDS AND LIQUIDS
 20 mm 0. 2 mm Schneider et al. J. American Chemical Society, 2002. (a) Laser scanning confocal microscopy. Green regions are fluorescently stained self-assembled peptide, and black regions are water-filled pores and channels. (b) Cryo. TEM. Dark structures are selfassembled peptide scaffold, while lighter gray areas are composed of vitrified water.
20 mm 0. 2 mm Schneider et al. J. American Chemical Society, 2002. (a) Laser scanning confocal microscopy. Green regions are fluorescently stained self-assembled peptide, and black regions are water-filled pores and channels. (b) Cryo. TEM. Dark structures are selfassembled peptide scaffold, while lighter gray areas are composed of vitrified water.
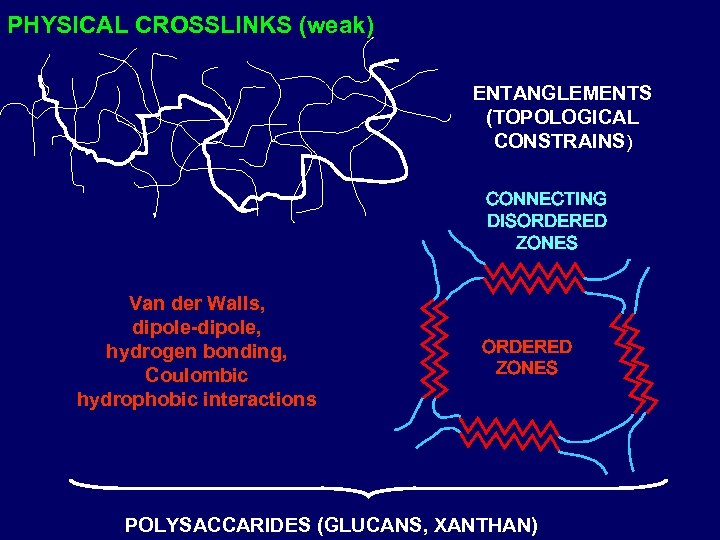 PHYSICAL CROSSLINKS (weak) ENTANGLEMENTS (TOPOLOGICAL CONSTRAINS) CONNECTING DISORDERED ZONES Van der Walls, dipole-dipole, hydrogen bonding, Coulombic hydrophobic interactions ORDERED ZONES POLYSACCARIDES (GLUCANS, XANTHAN)
PHYSICAL CROSSLINKS (weak) ENTANGLEMENTS (TOPOLOGICAL CONSTRAINS) CONNECTING DISORDERED ZONES Van der Walls, dipole-dipole, hydrogen bonding, Coulombic hydrophobic interactions ORDERED ZONES POLYSACCARIDES (GLUCANS, XANTHAN)
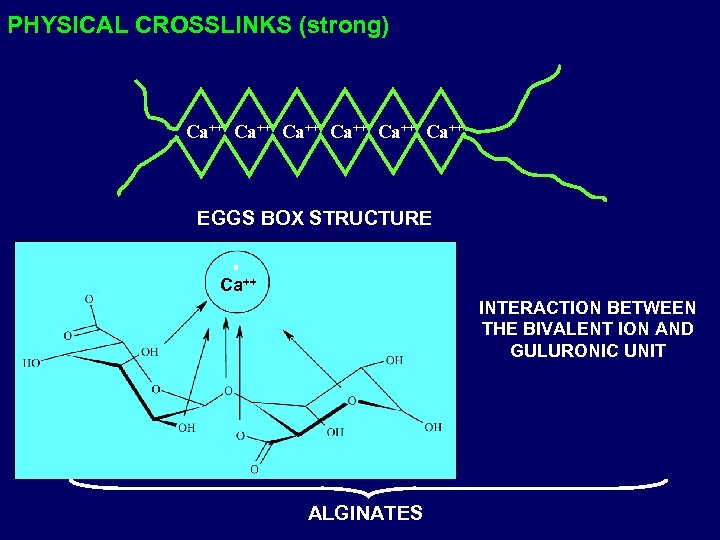 PHYSICAL CROSSLINKS (strong) Ca++ Ca++ EGGS BOX STRUCTURE Ca++ INTERACTION BETWEEN THE BIVALENT ION AND GULURONIC UNIT ALGINATES
PHYSICAL CROSSLINKS (strong) Ca++ Ca++ EGGS BOX STRUCTURE Ca++ INTERACTION BETWEEN THE BIVALENT ION AND GULURONIC UNIT ALGINATES
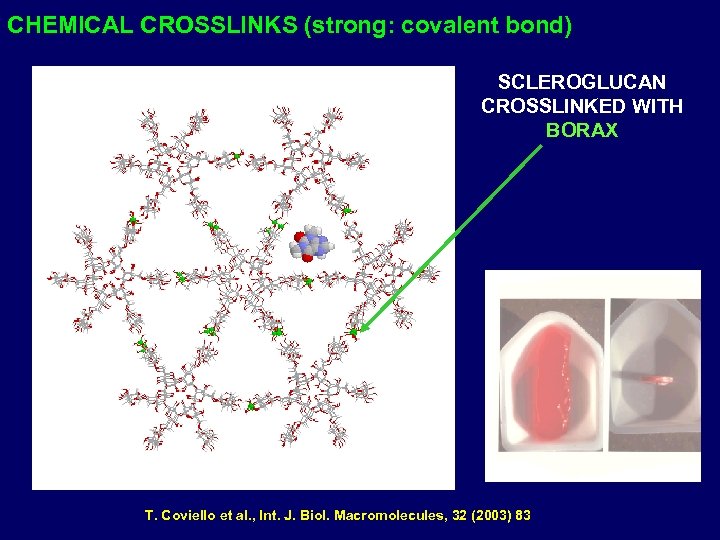 CHEMICAL CROSSLINKS (strong: covalent bond) SCLEROGLUCAN CROSSLINKED WITH BORAX T. Coviello et al. , Int. J. Biol. Macromolecules, 32 (2003) 83
CHEMICAL CROSSLINKS (strong: covalent bond) SCLEROGLUCAN CROSSLINKED WITH BORAX T. Coviello et al. , Int. J. Biol. Macromolecules, 32 (2003) 83
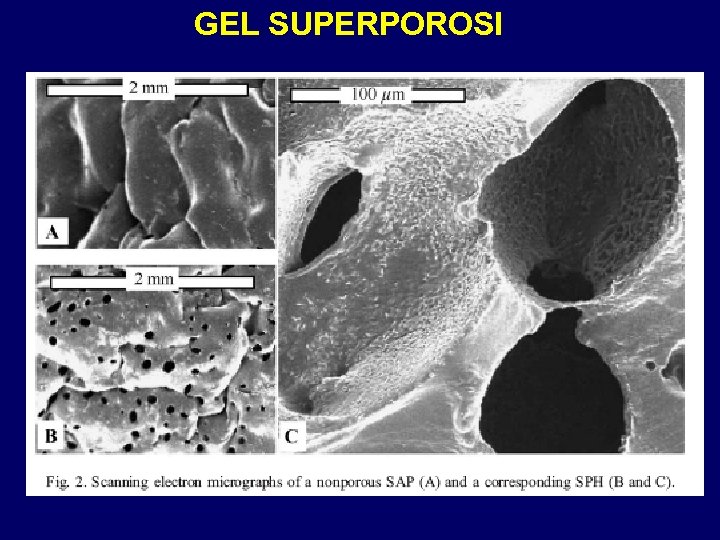 GEL SUPERPOROSI
GEL SUPERPOROSI
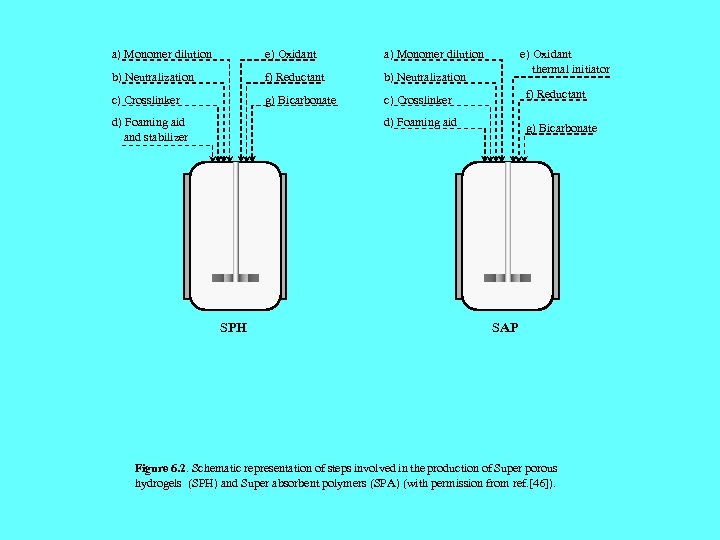 a) Monomer dilution e) Oxidant a) Monomer dilution b) Neutralization f) Reductant b) Neutralization c) Crosslinker g) Bicarbonate c) Crosslinker f) Reductant d) Foaming aid g) Bicarbonate d) Foaming aid and stabilizer SPH e) Oxidant thermal initiator SAP Figure 6. 2. Schematic representation of steps involved in the production of Super porous hydrogels (SPH) and Super absorbent polymers (SPA) (with permission from ref. [46]).
a) Monomer dilution e) Oxidant a) Monomer dilution b) Neutralization f) Reductant b) Neutralization c) Crosslinker g) Bicarbonate c) Crosslinker f) Reductant d) Foaming aid g) Bicarbonate d) Foaming aid and stabilizer SPH e) Oxidant thermal initiator SAP Figure 6. 2. Schematic representation of steps involved in the production of Super porous hydrogels (SPH) and Super absorbent polymers (SPA) (with permission from ref. [46]).
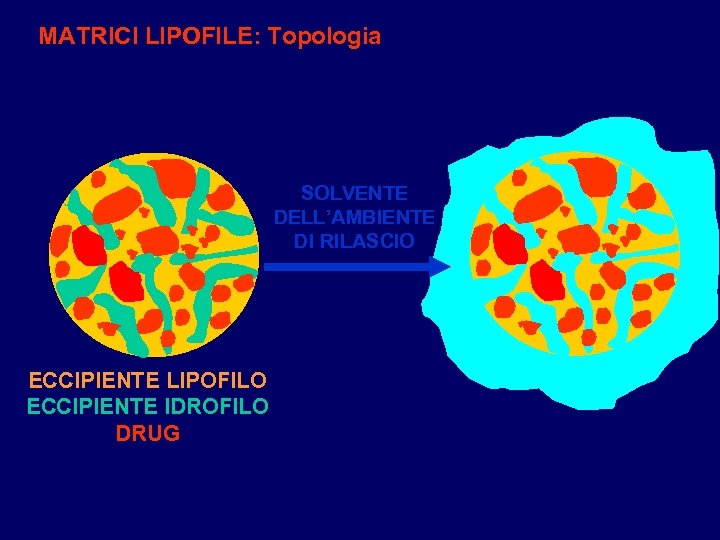 MATRICI LIPOFILE: Topologia SOLVENTE DELL’AMBIENTE DI RILASCIO ECCIPIENTE LIPOFILO ECCIPIENTE IDROFILO DRUG
MATRICI LIPOFILE: Topologia SOLVENTE DELL’AMBIENTE DI RILASCIO ECCIPIENTE LIPOFILO ECCIPIENTE IDROFILO DRUG
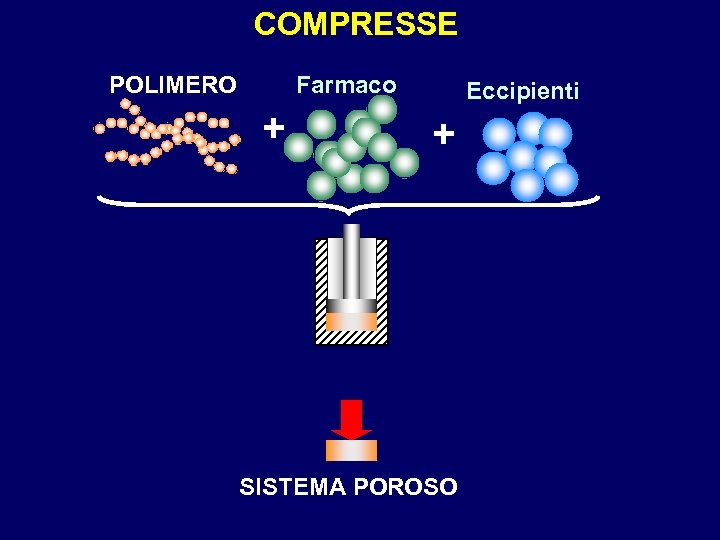 COMPRESSE POLIMERO Farmaco + Eccipienti + SISTEMA POROSO
COMPRESSE POLIMERO Farmaco + Eccipienti + SISTEMA POROSO
 SISTEMI INORGANICI POROSI: ZEOLITI MCM-41 transmission electron micrograph. Hexagonally arranged 4. 0 nm sized pores can be detected
SISTEMI INORGANICI POROSI: ZEOLITI MCM-41 transmission electron micrograph. Hexagonally arranged 4. 0 nm sized pores can be detected
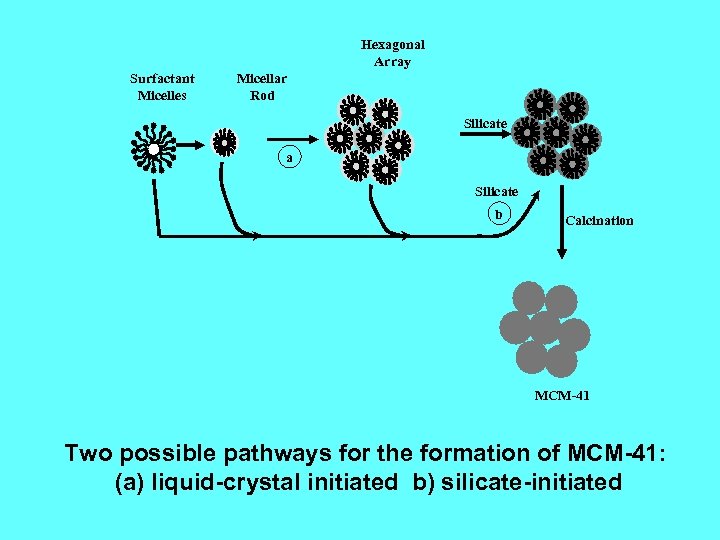 Hexagonal Array Surfactant Micelles Micellar Rod Silicate a Silicate b Calcination MCM-41 Two possible pathways for the formation of MCM-41: (a) liquid-crystal initiated b) silicate-initiated
Hexagonal Array Surfactant Micelles Micellar Rod Silicate a Silicate b Calcination MCM-41 Two possible pathways for the formation of MCM-41: (a) liquid-crystal initiated b) silicate-initiated
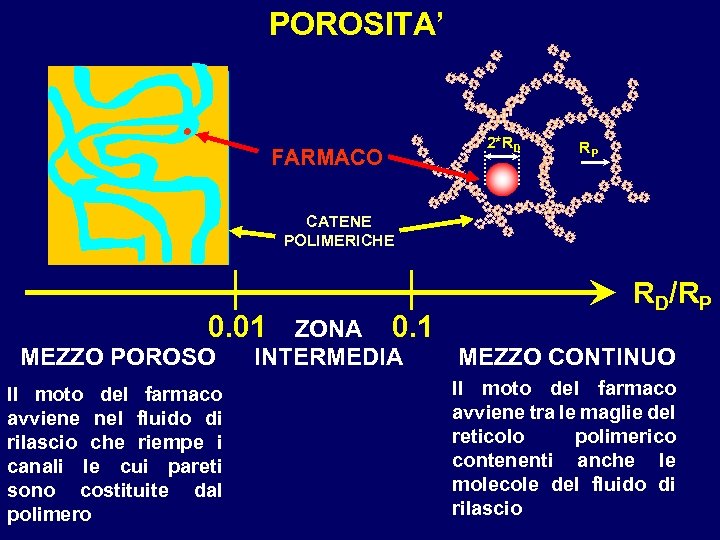 POROSITA’ FARMACO 2*RD RP CATENE POLIMERICHE 0. 01 MEZZO POROSO Il moto del farmaco avviene nel fluido di rilascio che riempe i canali le cui pareti sono costituite dal polimero RD/RP ZONA 0. 1 MEZZO CONTINUO INTERMEDIA Il moto del farmaco avviene tra le maglie del reticolo polimerico contenenti anche le molecole del fluido di rilascio
POROSITA’ FARMACO 2*RD RP CATENE POLIMERICHE 0. 01 MEZZO POROSO Il moto del farmaco avviene nel fluido di rilascio che riempe i canali le cui pareti sono costituite dal polimero RD/RP ZONA 0. 1 MEZZO CONTINUO INTERMEDIA Il moto del farmaco avviene tra le maglie del reticolo polimerico contenenti anche le molecole del fluido di rilascio
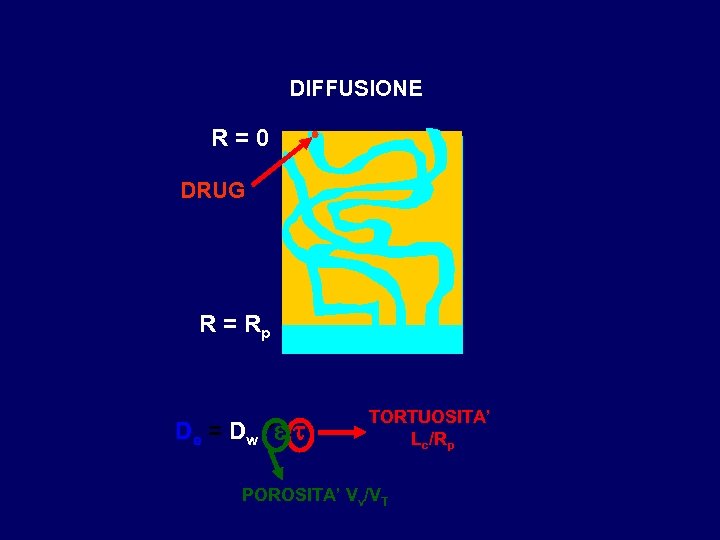 DIFFUSIONE R=0 DRUG R = Rp De = Dw * e / t TORTUOSITA’ Lc/Rp POROSITA’ Vv/VT
DIFFUSIONE R=0 DRUG R = Rp De = Dw * e / t TORTUOSITA’ Lc/Rp POROSITA’ Vv/VT
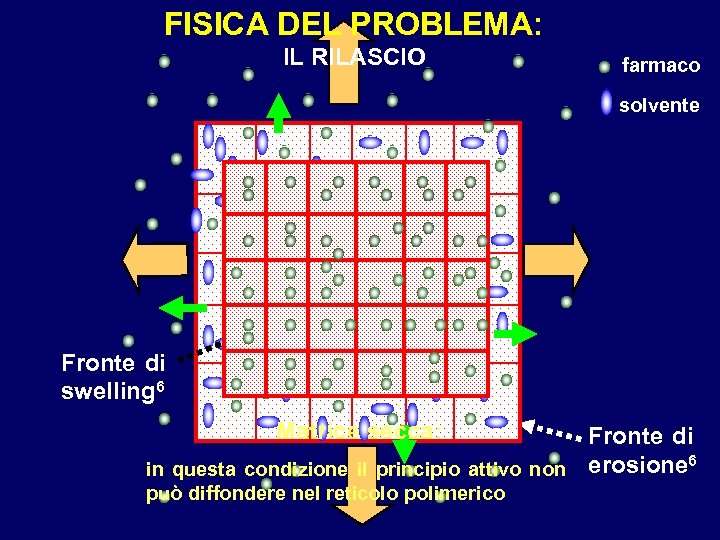 FISICA DEL PROBLEMA: IL RILASCIO farmaco solvente Fronte di swelling 6 Matrice secca: Fronte di in questa condizione il principio attivo non erosione 6 può diffondere nel reticolo polimerico
FISICA DEL PROBLEMA: IL RILASCIO farmaco solvente Fronte di swelling 6 Matrice secca: Fronte di in questa condizione il principio attivo non erosione 6 può diffondere nel reticolo polimerico
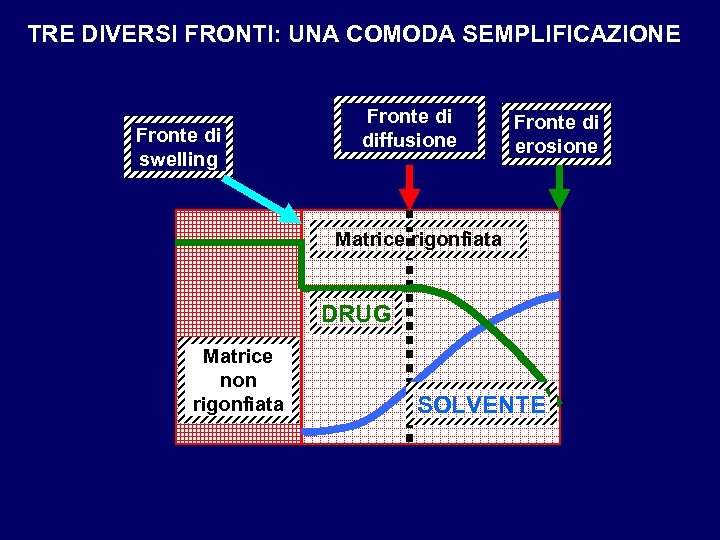 TRE DIVERSI FRONTI: UNA COMODA SEMPLIFICAZIONE Fronte di swelling Fronte di diffusione Fronte di erosione Matrice rigonfiata DRUG Matrice non rigonfiata SOLVENTE
TRE DIVERSI FRONTI: UNA COMODA SEMPLIFICAZIONE Fronte di swelling Fronte di diffusione Fronte di erosione Matrice rigonfiata DRUG Matrice non rigonfiata SOLVENTE
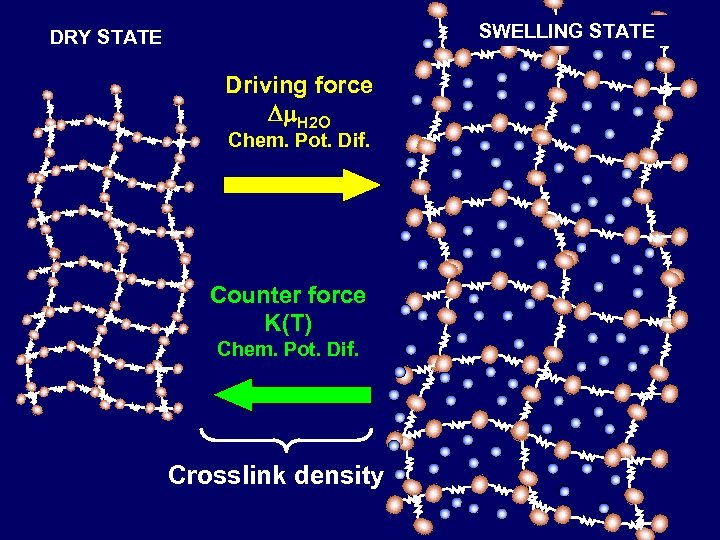 SWELLING STATE DRY STATE Driving force Dm. H 2 O Chem. Pot. Dif. Counter force K(T) Chem. Pot. Dif. Crosslink density
SWELLING STATE DRY STATE Driving force Dm. H 2 O Chem. Pot. Dif. Counter force K(T) Chem. Pot. Dif. Crosslink density
 Polymeric chains pass from one equilibrium state to another one due to the incoming solvent The time required to get the new equilibrium condition is the so called relaxation time tp depending on local solvent concentration and temperature
Polymeric chains pass from one equilibrium state to another one due to the incoming solvent The time required to get the new equilibrium condition is the so called relaxation time tp depending on local solvent concentration and temperature
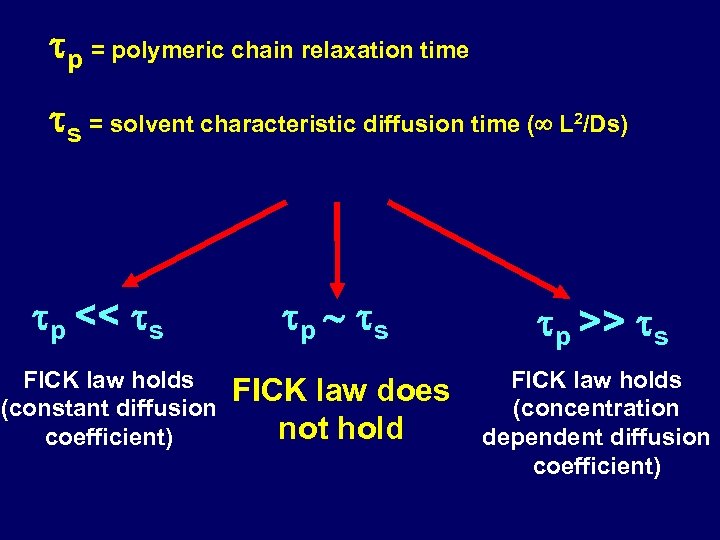 tp = polymeric chain relaxation time ts = solvent characteristic diffusion time ( L /Ds) 2 tp << ts FICK law holds (constant diffusion coefficient) tp ts tp >> ts FICK law does not hold FICK law holds (concentration dependent diffusion coefficient)
tp = polymeric chain relaxation time ts = solvent characteristic diffusion time ( L /Ds) 2 tp << ts FICK law holds (constant diffusion coefficient) tp ts tp >> ts FICK law does not hold FICK law holds (concentration dependent diffusion coefficient)
 C 0 h FICK LAW CL F instantaneously modifies with the concentration gradient C 0 h FICK LAW CL F does not instantaneously modify with the concentration gradient: F is also time dependent (D=D(t))
C 0 h FICK LAW CL F instantaneously modifies with the concentration gradient C 0 h FICK LAW CL F does not instantaneously modify with the concentration gradient: F is also time dependent (D=D(t))
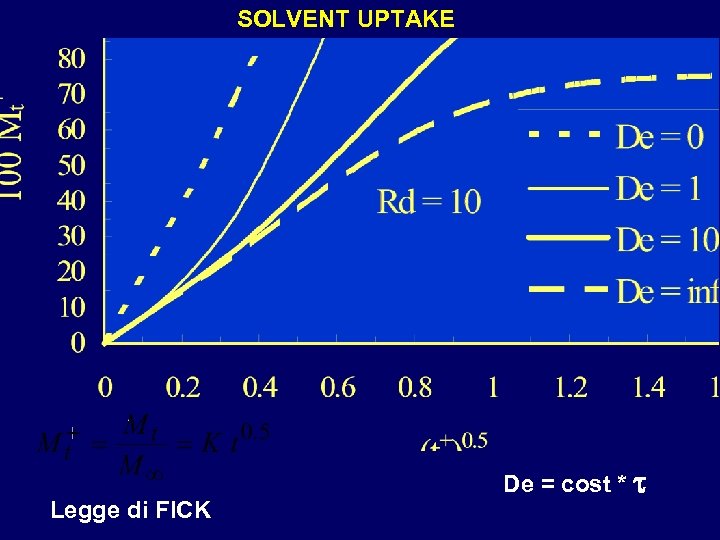 SOLVENT UPTAKE Legge di FICK De = cost * t
SOLVENT UPTAKE Legge di FICK De = cost * t
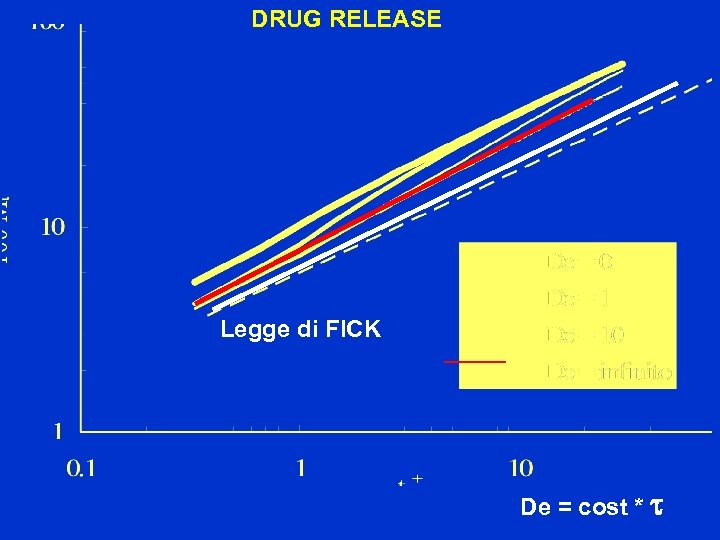 DRUG RELEASE Legge di FICK De = cost * t
DRUG RELEASE Legge di FICK De = cost * t
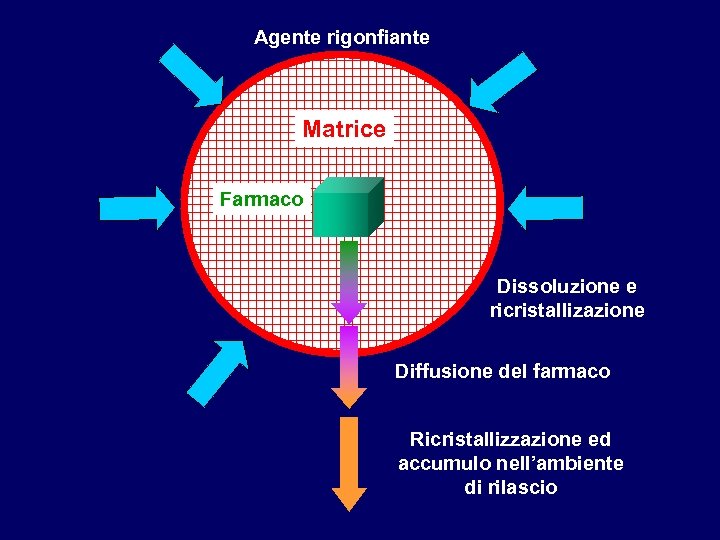 Agente rigonfiante Matrice Farmaco Dissoluzione e ricristallizazione Diffusione del farmaco Ricristallizzazione ed accumulo nell’ambiente di rilascio
Agente rigonfiante Matrice Farmaco Dissoluzione e ricristallizazione Diffusione del farmaco Ricristallizzazione ed accumulo nell’ambiente di rilascio
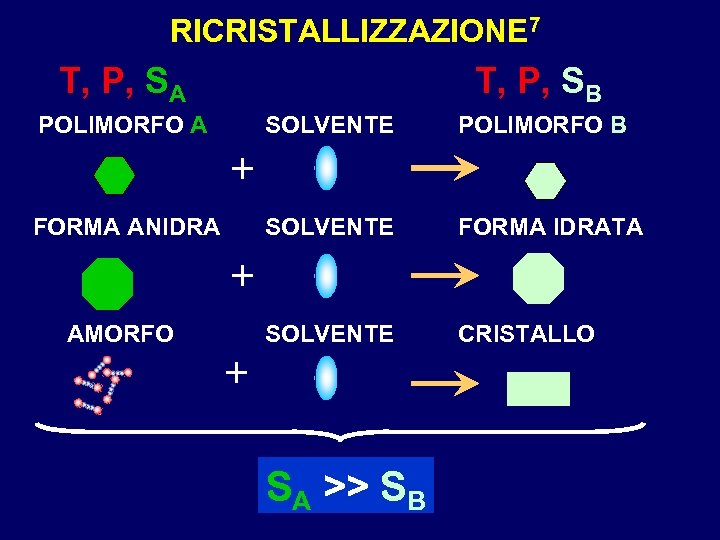 RICRISTALLIZZAZIONE 7 T, P, SA T, P, SB POLIMORFO A SOLVENTE POLIMORFO B SOLVENTE FORMA IDRATA SOLVENTE CRISTALLO + FORMA ANIDRA + AMORFO + SA >> SB
RICRISTALLIZZAZIONE 7 T, P, SA T, P, SB POLIMORFO A SOLVENTE POLIMORFO B SOLVENTE FORMA IDRATA SOLVENTE CRISTALLO + FORMA ANIDRA + AMORFO + SA >> SB
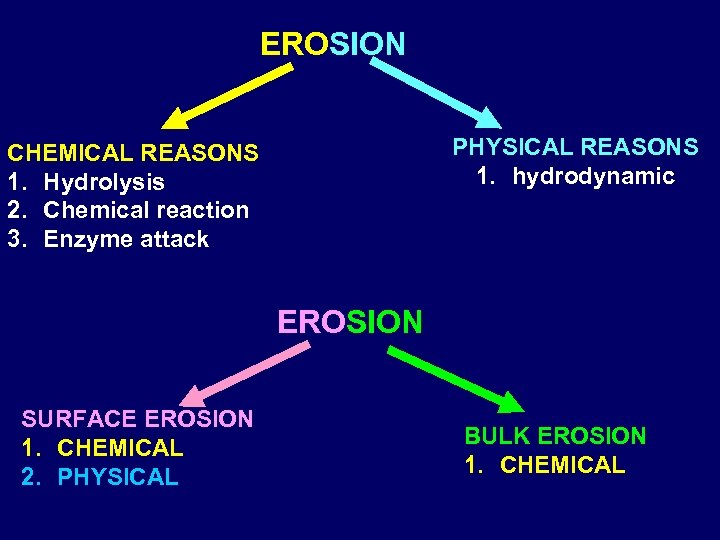 EROSION PHYSICAL REASONS 1. hydrodynamic CHEMICAL REASONS 1. Hydrolysis 2. Chemical reaction 3. Enzyme attack EROSION SURFACE EROSION 1. CHEMICAL 2. PHYSICAL BULK EROSION 1. CHEMICAL
EROSION PHYSICAL REASONS 1. hydrodynamic CHEMICAL REASONS 1. Hydrolysis 2. Chemical reaction 3. Enzyme attack EROSION SURFACE EROSION 1. CHEMICAL 2. PHYSICAL BULK EROSION 1. CHEMICAL
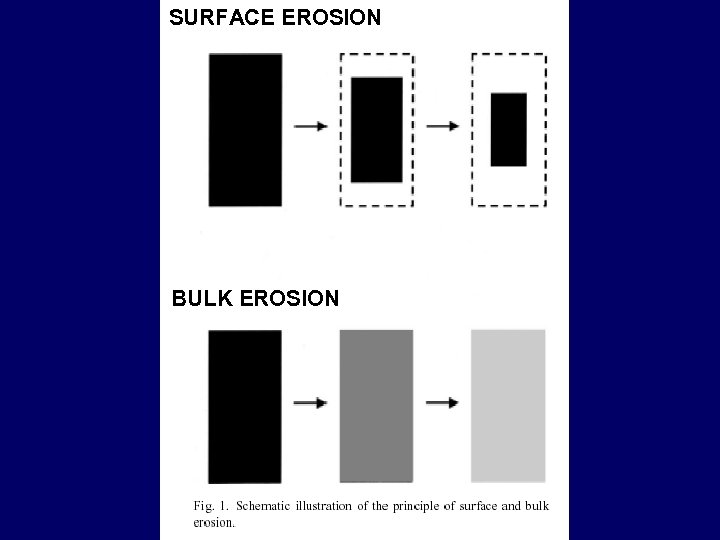 SURFACE EROSION BULK EROSION
SURFACE EROSION BULK EROSION
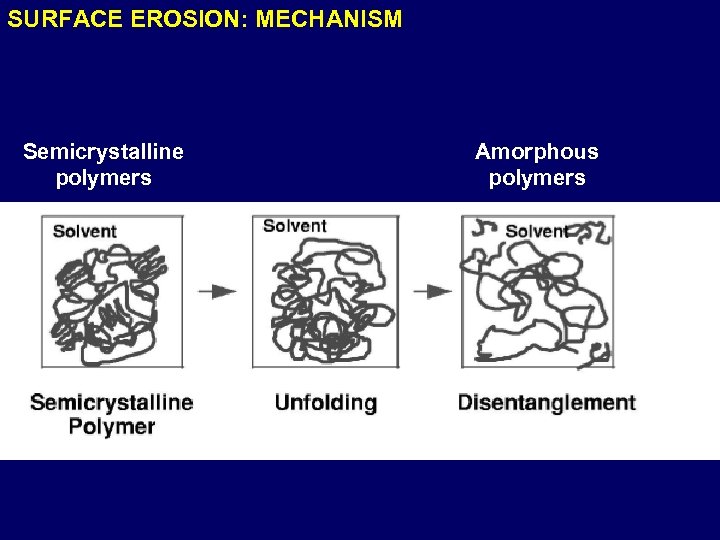 SURFACE EROSION: MECHANISM Semicrystalline polymers Amorphous polymers
SURFACE EROSION: MECHANISM Semicrystalline polymers Amorphous polymers
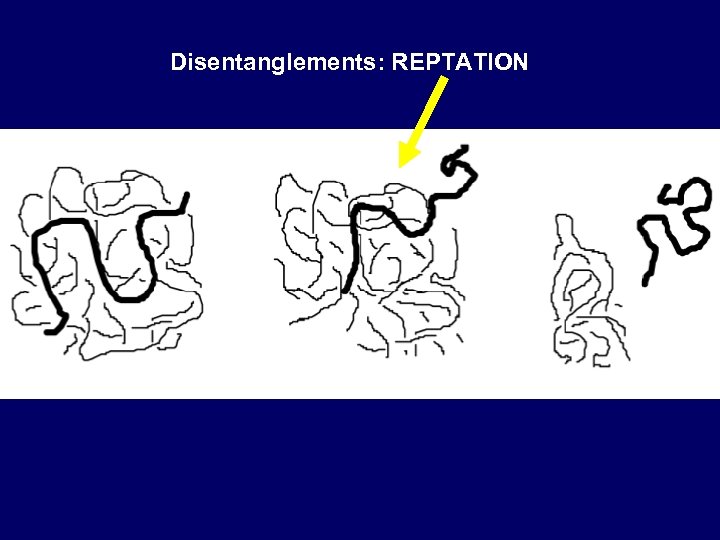 Disentanglements: REPTATION
Disentanglements: REPTATION
 RELEASE FROM ERODING SYSTEM
RELEASE FROM ERODING SYSTEM
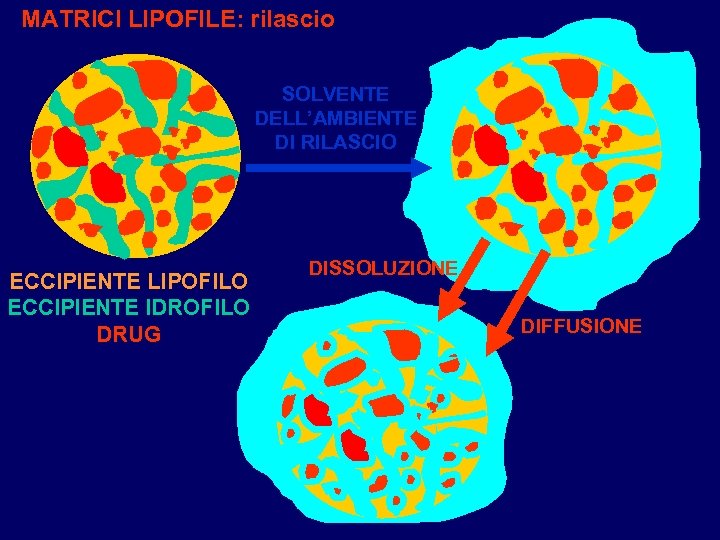 MATRICI LIPOFILE: rilascio SOLVENTE DELL’AMBIENTE DI RILASCIO ECCIPIENTE LIPOFILO ECCIPIENTE IDROFILO DRUG DISSOLUZIONE DIFFUSIONE
MATRICI LIPOFILE: rilascio SOLVENTE DELL’AMBIENTE DI RILASCIO ECCIPIENTE LIPOFILO ECCIPIENTE IDROFILO DRUG DISSOLUZIONE DIFFUSIONE
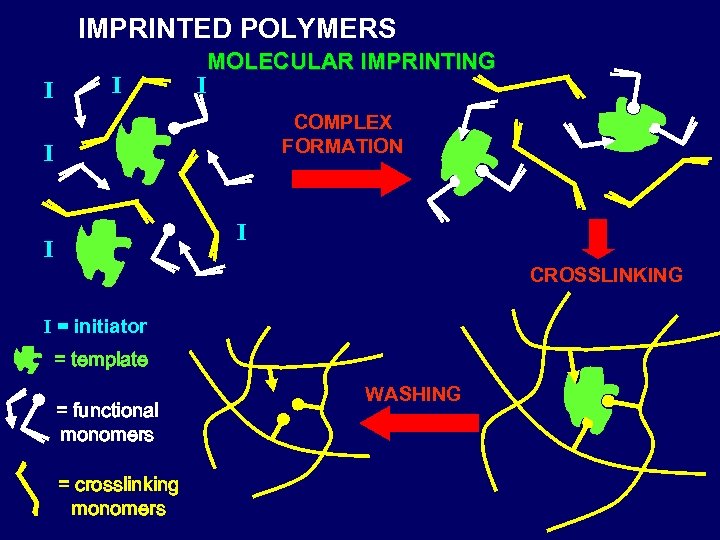 IMPRINTED POLYMERS I I I MOLECULAR IMPRINTING COMPLEX FORMATION I I I CROSSLINKING I = initiator = template = functional monomers = crosslinking monomers WASHING
IMPRINTED POLYMERS I I I MOLECULAR IMPRINTING COMPLEX FORMATION I I I CROSSLINKING I = initiator = template = functional monomers = crosslinking monomers WASHING
 IMPRINTED POLYMERS: CHARACTERISTICS Binding affinity: a measure of how well the template molecule is attracted to the binding site Selectivity : the ability to differentiate between the template and other molecules Binding capacity : the maximum amount of template bound per mass or volume of polymer
IMPRINTED POLYMERS: CHARACTERISTICS Binding affinity: a measure of how well the template molecule is attracted to the binding site Selectivity : the ability to differentiate between the template and other molecules Binding capacity : the maximum amount of template bound per mass or volume of polymer
 BINDING AFFINITY Macromolecular sites concentration Template concentration Forward reaction (binding) Association constant Backward reaction (un-binding)
BINDING AFFINITY Macromolecular sites concentration Template concentration Forward reaction (binding) Association constant Backward reaction (un-binding)
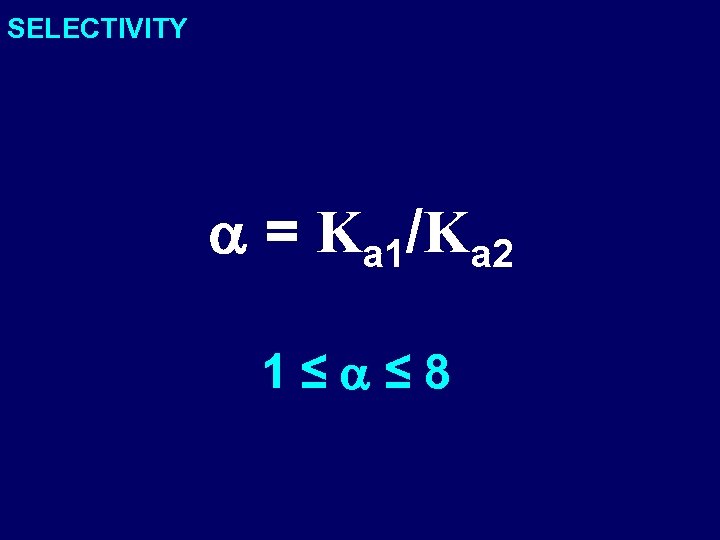 SELECTIVITY a = Ka 1/Ka 2 1≤a≤ 8
SELECTIVITY a = Ka 1/Ka 2 1≤a≤ 8
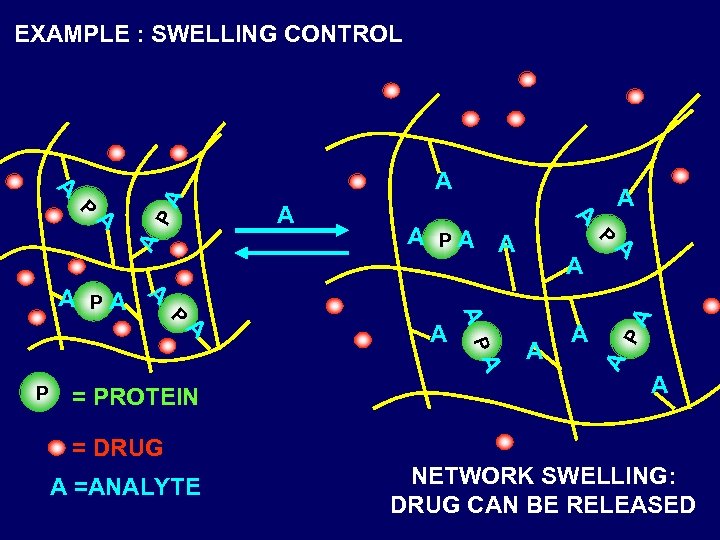 EXAMPLE : SWELLING CONTROL P A = PROTEIN A A A P A PA A A PA P A A = DRUG A =ANALYTE NETWORK SWELLING: DRUG CAN BE RELEASED
EXAMPLE : SWELLING CONTROL P A = PROTEIN A A A P A PA A A PA P A A = DRUG A =ANALYTE NETWORK SWELLING: DRUG CAN BE RELEASED
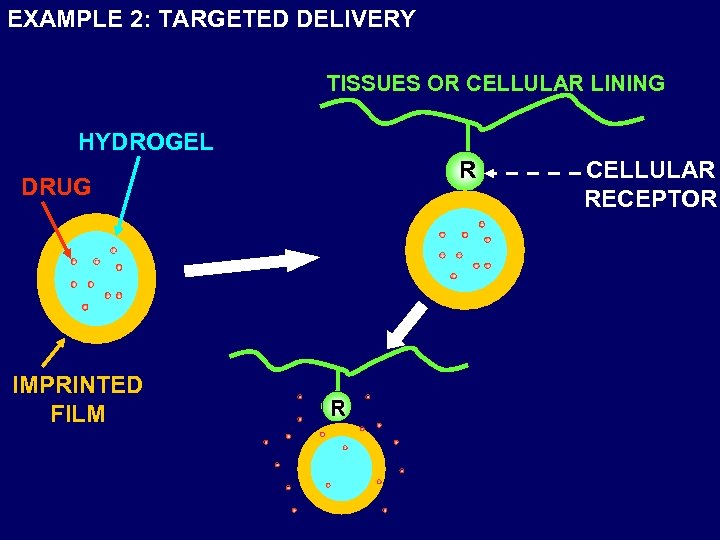 EXAMPLE 2: TARGETED DELIVERY TISSUES OR CELLULAR LINING HYDROGEL R DRUG IMPRINTED FILM R CELLULAR RECEPTOR
EXAMPLE 2: TARGETED DELIVERY TISSUES OR CELLULAR LINING HYDROGEL R DRUG IMPRINTED FILM R CELLULAR RECEPTOR
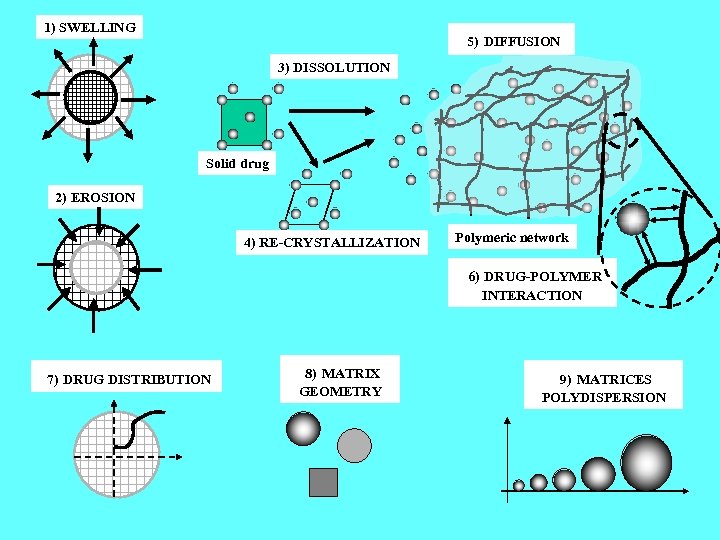 1) SWELLING 5) DIFFUSION 3) DISSOLUTION Solid drug 2) EROSION 4) RE-CRYSTALLIZATION Polymeric network 6) DRUG-POLYMER INTERACTION 7) DRUG DISTRIBUTION 8) MATRIX GEOMETRY 9) MATRICES POLYDISPERSION
1) SWELLING 5) DIFFUSION 3) DISSOLUTION Solid drug 2) EROSION 4) RE-CRYSTALLIZATION Polymeric network 6) DRUG-POLYMER INTERACTION 7) DRUG DISTRIBUTION 8) MATRIX GEOMETRY 9) MATRICES POLYDISPERSION
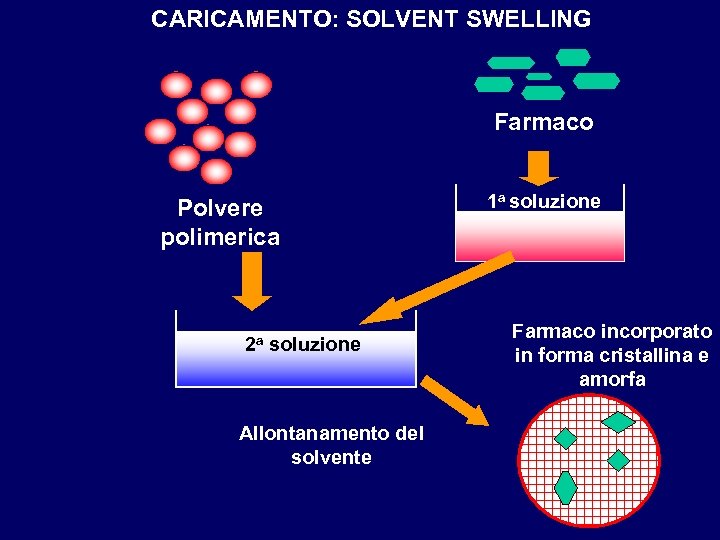 CARICAMENTO: SOLVENT SWELLING Farmaco Polvere polimerica 2 a soluzione Allontanamento del solvente 1 a soluzione Farmaco incorporato in forma cristallina e amorfa
CARICAMENTO: SOLVENT SWELLING Farmaco Polvere polimerica 2 a soluzione Allontanamento del solvente 1 a soluzione Farmaco incorporato in forma cristallina e amorfa
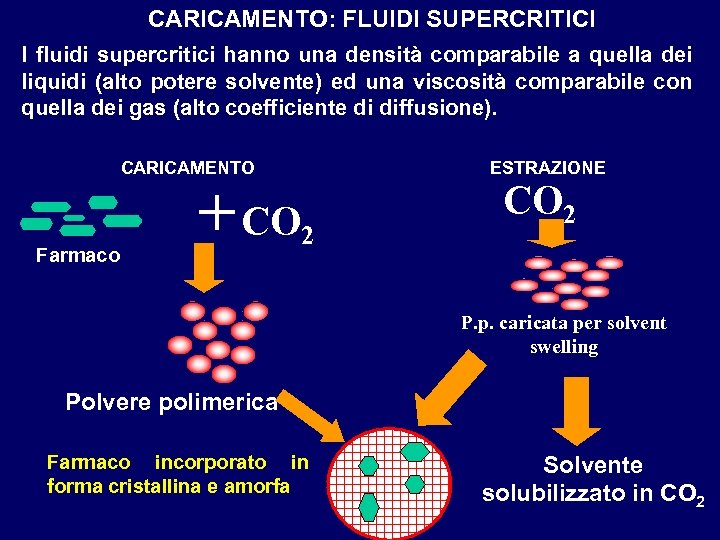 CARICAMENTO: FLUIDI SUPERCRITICI I fluidi supercritici hanno una densità comparabile a quella dei liquidi (alto potere solvente) ed una viscosità comparabile con quella dei gas (alto coefficiente di diffusione). CARICAMENTO Farmaco + CO 2 ESTRAZIONE CO 2 P. p. caricata per solvent swelling Polvere polimerica Farmaco incorporato in forma cristallina e amorfa Solvente solubilizzato in CO 2
CARICAMENTO: FLUIDI SUPERCRITICI I fluidi supercritici hanno una densità comparabile a quella dei liquidi (alto potere solvente) ed una viscosità comparabile con quella dei gas (alto coefficiente di diffusione). CARICAMENTO Farmaco + CO 2 ESTRAZIONE CO 2 P. p. caricata per solvent swelling Polvere polimerica Farmaco incorporato in forma cristallina e amorfa Solvente solubilizzato in CO 2
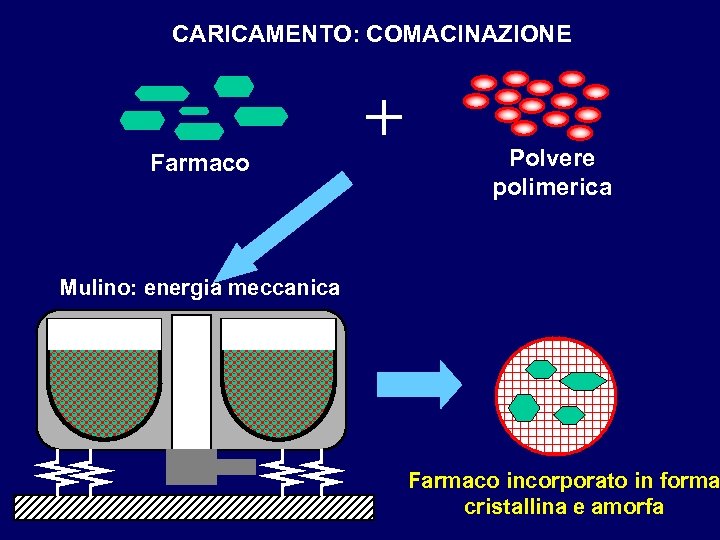 CARICAMENTO: COMACINAZIONE Farmaco + Polvere polimerica Mulino: energia meccanica Farmaco incorporato in forma cristallina e amorfa
CARICAMENTO: COMACINAZIONE Farmaco + Polvere polimerica Mulino: energia meccanica Farmaco incorporato in forma cristallina e amorfa
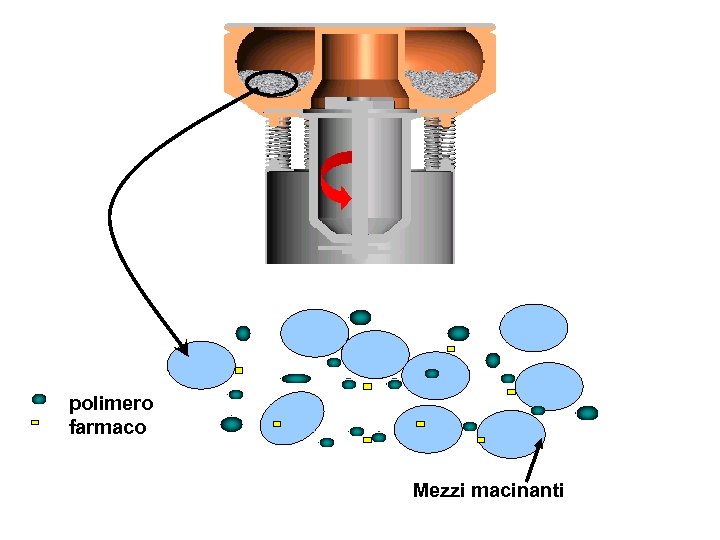 polimero farmaco Mezzi macinanti
polimero farmaco Mezzi macinanti



 BIBLIOGRAFIA 1) Pharmacos 4, Eudralex Collection, Medicinal Products for Human Use: Guidelines. Volume 3 C, p. 234 (internet site: http: //pharmacos. eudra. org/F 2/eudralex/vol 3/home. htm). 2) Israel G. in Modelli Matematici nelle Scienze Biologiche, a cura di P. Freguglia, Edizioni Quattro Venti, Urbino, pag. 134 (1998). 3) Lapasin R, Pricl S, Rheology of Industrial Polysaccharides; Theory and Applications, Chapman and Hall, London, 1995. 4) Coviello T, Grassi M, Rambone G, Santucci E, a Carafa M , Murtas E, Riccieri F M, Franco Alhaique F. Novel hydrogel system from scleroglucan: synthesis and characterization J. Contr. Rel. 60, 367– 378, 1999. 5) A. Kydonieus (Ed. ), Treatise on Controlled Drug Delivery, Marcel Dekker, New York, 1992, pp. 54 -55. 6) Colombo, P. 1993. Swelling-controlled release in hydrogel matrices for oral route. Adv. Drug. Dev. Rev. , 11, 37 – 57 7) Nogami H, Nagai T, Youtsunagi T. Dissolution phenomena of organic medicinals involving simultaneous phase changes. Chem. Pharm. Bull. 17(3), 499 -509, 1969. 8) Lee P I, Initial concentration distribution as a mechanism for regulating drug release from diffusion controlled and surface erosion controlled matrix systems, J. Contr. Rel. 4, 1– 7, 1986. 9) Grassi M, Colombo I, Lapasin R. Drug release from an ensemble of swellable crosslinked polymer particles. J. Contr. Rel. 68, 97 -113, 2000.
BIBLIOGRAFIA 1) Pharmacos 4, Eudralex Collection, Medicinal Products for Human Use: Guidelines. Volume 3 C, p. 234 (internet site: http: //pharmacos. eudra. org/F 2/eudralex/vol 3/home. htm). 2) Israel G. in Modelli Matematici nelle Scienze Biologiche, a cura di P. Freguglia, Edizioni Quattro Venti, Urbino, pag. 134 (1998). 3) Lapasin R, Pricl S, Rheology of Industrial Polysaccharides; Theory and Applications, Chapman and Hall, London, 1995. 4) Coviello T, Grassi M, Rambone G, Santucci E, a Carafa M , Murtas E, Riccieri F M, Franco Alhaique F. Novel hydrogel system from scleroglucan: synthesis and characterization J. Contr. Rel. 60, 367– 378, 1999. 5) A. Kydonieus (Ed. ), Treatise on Controlled Drug Delivery, Marcel Dekker, New York, 1992, pp. 54 -55. 6) Colombo, P. 1993. Swelling-controlled release in hydrogel matrices for oral route. Adv. Drug. Dev. Rev. , 11, 37 – 57 7) Nogami H, Nagai T, Youtsunagi T. Dissolution phenomena of organic medicinals involving simultaneous phase changes. Chem. Pharm. Bull. 17(3), 499 -509, 1969. 8) Lee P I, Initial concentration distribution as a mechanism for regulating drug release from diffusion controlled and surface erosion controlled matrix systems, J. Contr. Rel. 4, 1– 7, 1986. 9) Grassi M, Colombo I, Lapasin R. Drug release from an ensemble of swellable crosslinked polymer particles. J. Contr. Rel. 68, 97 -113, 2000.


