9c0d7d5bbad2c6b99d2c6d3f3d71e771.ppt
- Количество слайдов: 59
 MANAGEMENT OF YSLIPIDEMIA : D WHEN STATIN ALONE ISINSUFFICIENT ! Sompongse Suwanwalaikorn, MD
MANAGEMENT OF YSLIPIDEMIA : D WHEN STATIN ALONE ISINSUFFICIENT ! Sompongse Suwanwalaikorn, MD
 Options to achieve low LDL-C targets Use maximal doses of currently available statins Consider new mega-statins Consider combination therapy
Options to achieve low LDL-C targets Use maximal doses of currently available statins Consider new mega-statins Consider combination therapy
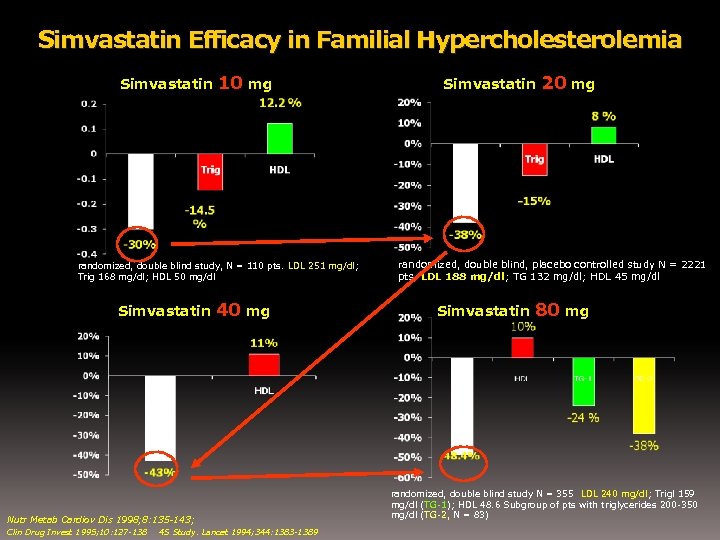 Simvastatin Efficacy in Familial Hypercholesterolemia Simvastatin 10 mg randomized, double blind study, N = 110 pts. LDL 251 mg/dl; Trig 168 mg/dl; HDL 50 mg/dl Simvastatin 40 mg Nutr Metab Cardiov Dis 1998; 8: 135 -143; Clin Drug Invest 1995; 10: 127 -138 4 S Study. Lancet 1994; 344: 1383 -1389 Simvastatin 20 mg randomized, double blind, placebo controlled study N = 2221 pts. LDL 188 mg/dl; TG 132 mg/dl; HDL 45 mg/dl Simvastatin 80 mg randomized, double blind study N = 355 LDL 240 mg/dl; Trigl 159 mg/dl (TG-1); HDL 48. 6 Subgroup of pts with triglycerides 200 -350 mg/dl (TG-2, N = 83)
Simvastatin Efficacy in Familial Hypercholesterolemia Simvastatin 10 mg randomized, double blind study, N = 110 pts. LDL 251 mg/dl; Trig 168 mg/dl; HDL 50 mg/dl Simvastatin 40 mg Nutr Metab Cardiov Dis 1998; 8: 135 -143; Clin Drug Invest 1995; 10: 127 -138 4 S Study. Lancet 1994; 344: 1383 -1389 Simvastatin 20 mg randomized, double blind, placebo controlled study N = 2221 pts. LDL 188 mg/dl; TG 132 mg/dl; HDL 45 mg/dl Simvastatin 80 mg randomized, double blind study N = 355 LDL 240 mg/dl; Trigl 159 mg/dl (TG-1); HDL 48. 6 Subgroup of pts with triglycerides 200 -350 mg/dl (TG-2, N = 83)
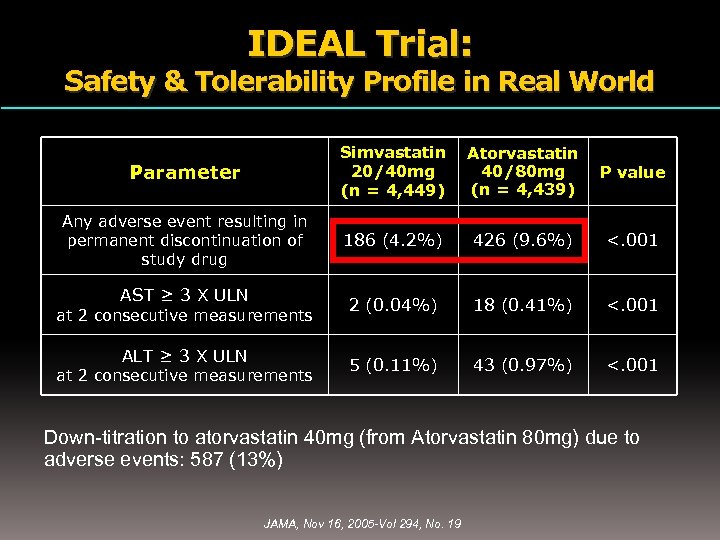 IDEAL Trial: Safety & Tolerability Profile in Real World Parameter Simvastatin 20/40 mg (n = 4, 449) Atorvastatin 40/80 mg (n = 4, 439) P value Any adverse event resulting in permanent discontinuation of study drug 186 (4. 2%) 426 (9. 6%) <. 001 2 (0. 04%) 18 (0. 41%) <. 001 5 (0. 11%) 43 (0. 97%) <. 001 AST ≥ 3 X ULN at 2 consecutive measurements ALT ≥ 3 X ULN at 2 consecutive measurements Down-titration to atorvastatin 40 mg (from Atorvastatin 80 mg) due to adverse events: 587 (13%) JAMA, Nov 16, 2005 -Vol 294, No. 19
IDEAL Trial: Safety & Tolerability Profile in Real World Parameter Simvastatin 20/40 mg (n = 4, 449) Atorvastatin 40/80 mg (n = 4, 439) P value Any adverse event resulting in permanent discontinuation of study drug 186 (4. 2%) 426 (9. 6%) <. 001 2 (0. 04%) 18 (0. 41%) <. 001 5 (0. 11%) 43 (0. 97%) <. 001 AST ≥ 3 X ULN at 2 consecutive measurements ALT ≥ 3 X ULN at 2 consecutive measurements Down-titration to atorvastatin 40 mg (from Atorvastatin 80 mg) due to adverse events: 587 (13%) JAMA, Nov 16, 2005 -Vol 294, No. 19
 Adverse event withdrawal rates of statins Br J Cardiol 2004
Adverse event withdrawal rates of statins Br J Cardiol 2004
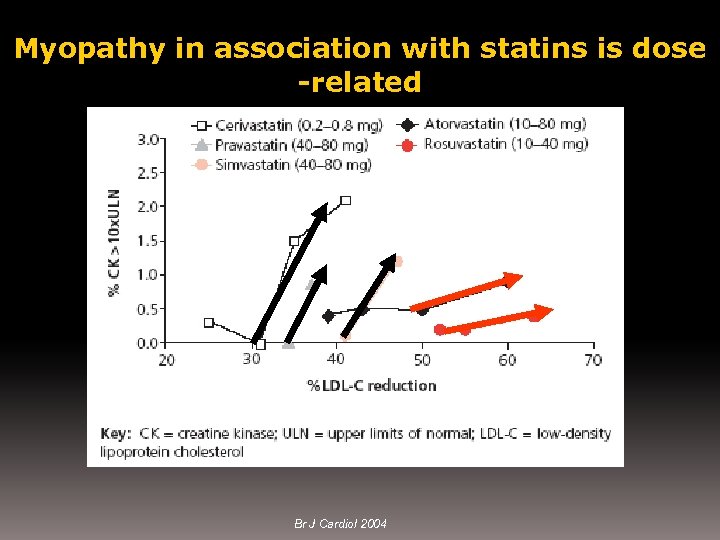 Myopathy in association with statins is dose -related Br J Cardiol 2004
Myopathy in association with statins is dose -related Br J Cardiol 2004
 Options to achieve low LDL-C targets Use maximal doses of currently available statins Consider new mega-statins Consider combination therapy
Options to achieve low LDL-C targets Use maximal doses of currently available statins Consider new mega-statins Consider combination therapy
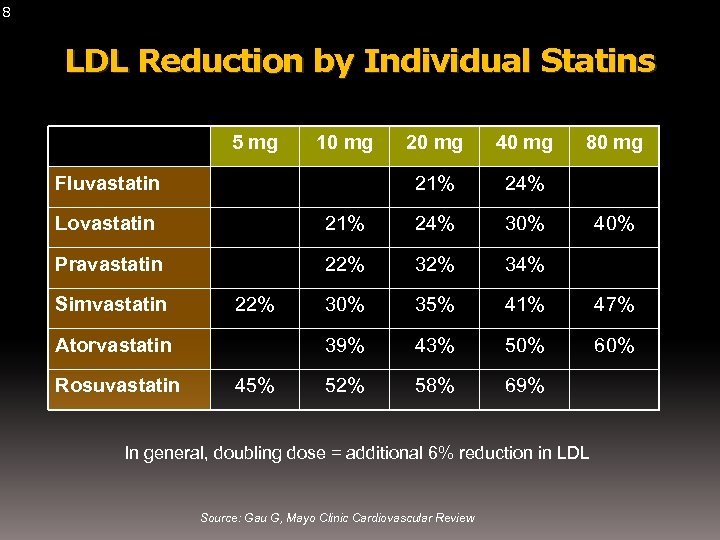 8 LDL Reduction by Individual Statins 5 mg 10 mg 40 mg 21% Fluvastatin 20 mg 80 mg 24% Lovastatin 21% 24% 30% Pravastatin 22% 34% 30% 35% 41% 47% 39% 43% 50% 60% 52% 58% 69% Simvastatin 22% Atorvastatin Rosuvastatin 45% In general, doubling dose = additional 6% reduction in LDL Source: Gau G, Mayo Clinic Cardiovascular Review 40%
8 LDL Reduction by Individual Statins 5 mg 10 mg 40 mg 21% Fluvastatin 20 mg 80 mg 24% Lovastatin 21% 24% 30% Pravastatin 22% 34% 30% 35% 41% 47% 39% 43% 50% 60% 52% 58% 69% Simvastatin 22% Atorvastatin Rosuvastatin 45% In general, doubling dose = additional 6% reduction in LDL Source: Gau G, Mayo Clinic Cardiovascular Review 40%
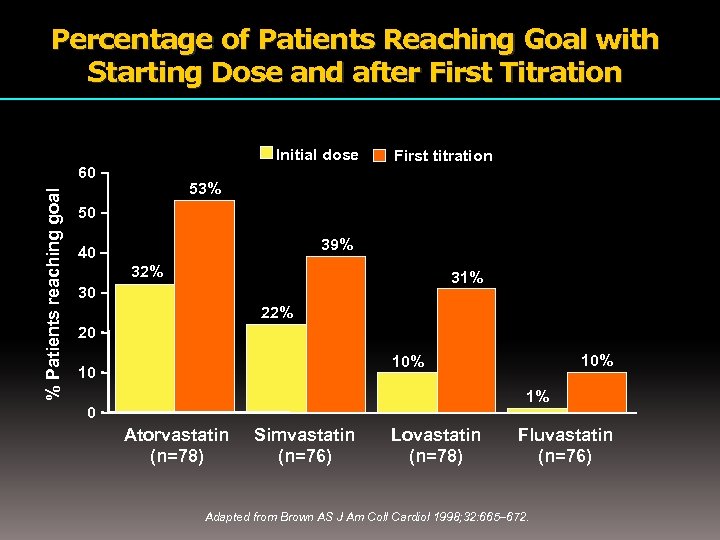 Percentage of Patients Reaching Goal with Starting Dose and after First Titration Initial dose % Patients reaching goal 60 First titration 53% 50 39% 40 32% 31% 30 22% 20 10% 10 1% 0 Atorvastatin (n=78) Simvastatin (n=76) Lovastatin (n=78) Fluvastatin (n=76) Adapted from Brown AS J Am Coll Cardiol 1998; 32: 665– 672.
Percentage of Patients Reaching Goal with Starting Dose and after First Titration Initial dose % Patients reaching goal 60 First titration 53% 50 39% 40 32% 31% 30 22% 20 10% 10 1% 0 Atorvastatin (n=78) Simvastatin (n=76) Lovastatin (n=78) Fluvastatin (n=76) Adapted from Brown AS J Am Coll Cardiol 1998; 32: 665– 672.
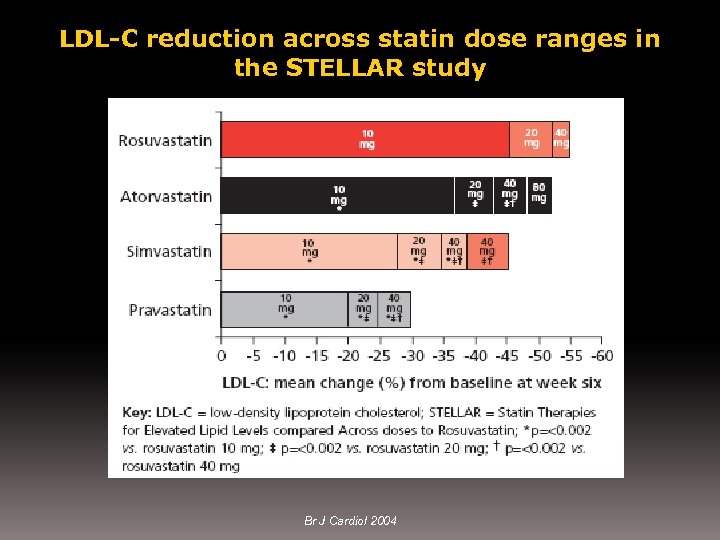 LDL-C reduction across statin dose ranges in the STELLAR study Br J Cardiol 2004
LDL-C reduction across statin dose ranges in the STELLAR study Br J Cardiol 2004
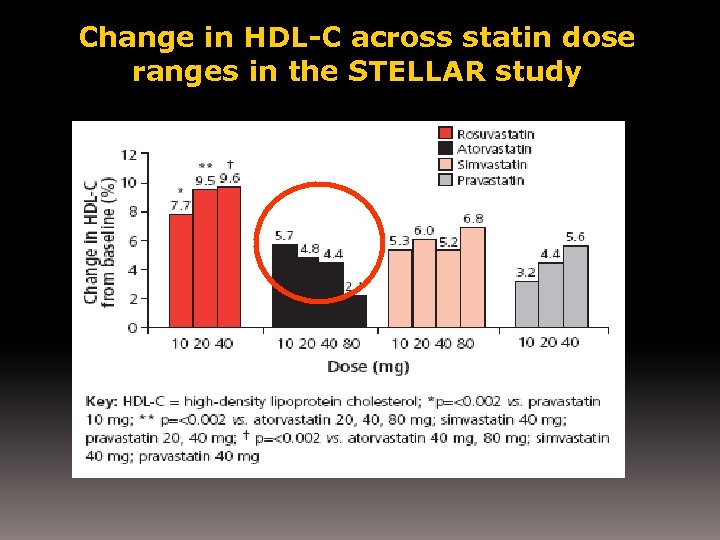 Change in HDL-C across statin dose ranges in the STELLAR study
Change in HDL-C across statin dose ranges in the STELLAR study
 Options to achieve low LDL-C targets Use maximal doses of currently available statins Consider new mega-statins Consider combination therapy
Options to achieve low LDL-C targets Use maximal doses of currently available statins Consider new mega-statins Consider combination therapy
 13 Theoretical Basis for Combination Rx Multiple pathogenetic pathways of cholesterol in development of CAD Differential effects of various lipid- lowering agents: Act on different pathways Target different lipid particles
13 Theoretical Basis for Combination Rx Multiple pathogenetic pathways of cholesterol in development of CAD Differential effects of various lipid- lowering agents: Act on different pathways Target different lipid particles
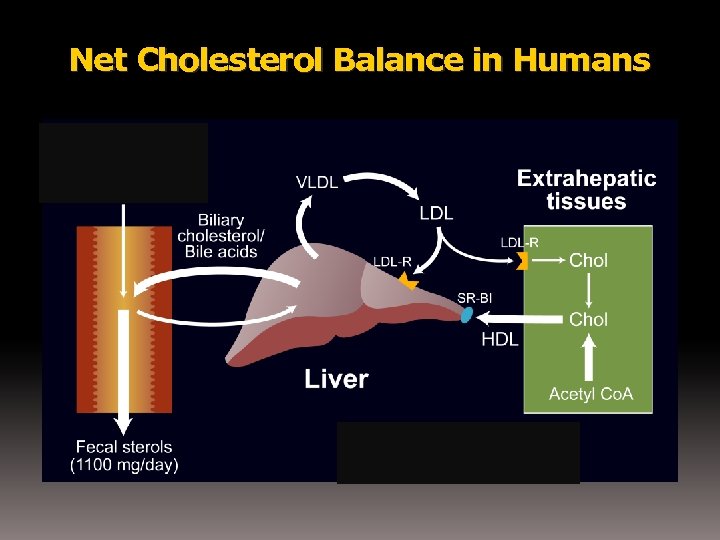 Net Cholesterol Balance in Humans
Net Cholesterol Balance in Humans
 Indications for Combination Rx Treatment goals not met with single agent Risk of intolerance, toxicity, or adverse drug interactions with higher dose of single agent Perceived benefit from ≥ 2 agents due to complementary: • Mechanisms of action • Effects on different lipids • Effects on CHD risk
Indications for Combination Rx Treatment goals not met with single agent Risk of intolerance, toxicity, or adverse drug interactions with higher dose of single agent Perceived benefit from ≥ 2 agents due to complementary: • Mechanisms of action • Effects on different lipids • Effects on CHD risk
 Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
 17 Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
17 Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
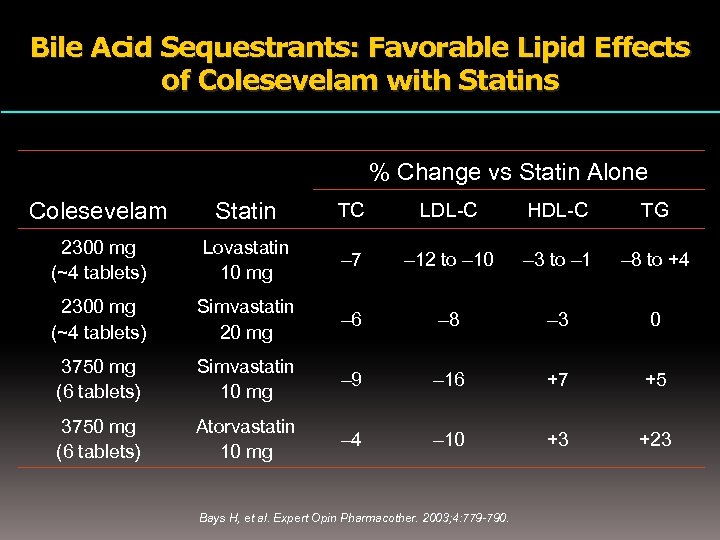 Bile Acid Sequestrants: Favorable Lipid Effects of Colesevelam with Statins % Change vs Statin Alone Colesevelam Statin TC LDL-C HDL-C TG 2300 mg (~4 tablets) Lovastatin 10 mg – 7 – 12 to – 10 – 3 to – 1 – 8 to +4 2300 mg (~4 tablets) Simvastatin 20 mg – 6 – 8 – 3 0 3750 mg (6 tablets) Simvastatin 10 mg – 9 – 16 +7 +5 3750 mg (6 tablets) Atorvastatin 10 mg – 4 – 10 +3 +23 Bays H, et al. Expert Opin Pharmacother. 2003; 4: 779 -790.
Bile Acid Sequestrants: Favorable Lipid Effects of Colesevelam with Statins % Change vs Statin Alone Colesevelam Statin TC LDL-C HDL-C TG 2300 mg (~4 tablets) Lovastatin 10 mg – 7 – 12 to – 10 – 3 to – 1 – 8 to +4 2300 mg (~4 tablets) Simvastatin 20 mg – 6 – 8 – 3 0 3750 mg (6 tablets) Simvastatin 10 mg – 9 – 16 +7 +5 3750 mg (6 tablets) Atorvastatin 10 mg – 4 – 10 +3 +23 Bays H, et al. Expert Opin Pharmacother. 2003; 4: 779 -790.
 Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
 PPAR- Agonists (Fibrates) and Statins Combination may significantly improve TG, LDL-C, and HDL-C levels Fibrates plus statins associated with increased risk for myopathy and rhabdomyolysis Probably not due to cytochrome P 450 drug interaction Gemfibrozil may impair glucuronidation of statins Fenofibrate may have relatively less potential for impairment of statin metabolism Ballantyne CM, et al. Arch Intern Med. 2003; 163: 553 -564. Bays H. Am J Cardiol. 2002; 90: 30 K-43 K. Bays HE, et al. Expert Opin Pharmacother. 2003; 4: 1901 -1938.
PPAR- Agonists (Fibrates) and Statins Combination may significantly improve TG, LDL-C, and HDL-C levels Fibrates plus statins associated with increased risk for myopathy and rhabdomyolysis Probably not due to cytochrome P 450 drug interaction Gemfibrozil may impair glucuronidation of statins Fenofibrate may have relatively less potential for impairment of statin metabolism Ballantyne CM, et al. Arch Intern Med. 2003; 163: 553 -564. Bays H. Am J Cardiol. 2002; 90: 30 K-43 K. Bays HE, et al. Expert Opin Pharmacother. 2003; 4: 1901 -1938.
 Adding Fibrate to Statin Rx Improves Profile of Atherogenic Dyslipidemia in Patients with Metabolic Sydrome simvastatin fenofibrate simvastatin+fenofibrate *† *† * † Mean ratio large/small LDL Mean mg/d. L * P ≤ 0. 05 vs. placebo; † P ≤ 0. 05 vs. simvastatin alone Vega. Am J Cardiol. 2003; 91: 956.
Adding Fibrate to Statin Rx Improves Profile of Atherogenic Dyslipidemia in Patients with Metabolic Sydrome simvastatin fenofibrate simvastatin+fenofibrate *† *† * † Mean ratio large/small LDL Mean mg/d. L * P ≤ 0. 05 vs. placebo; † P ≤ 0. 05 vs. simvastatin alone Vega. Am J Cardiol. 2003; 91: 956.
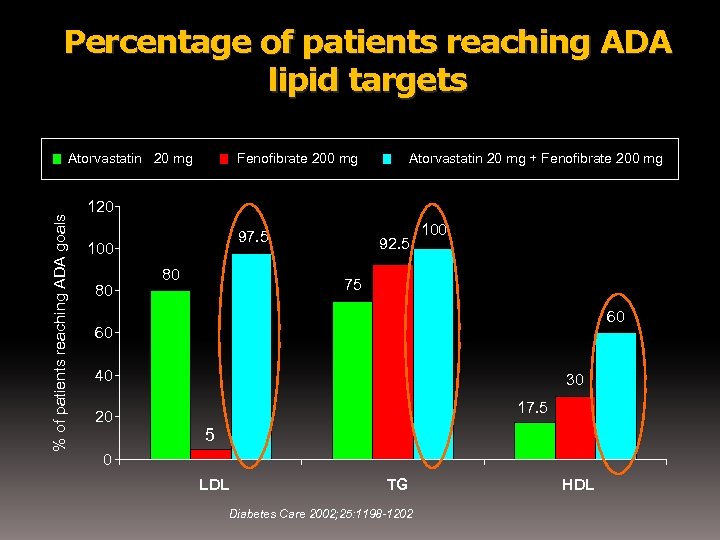 Percentage of patients reaching ADA lipid targets % of patients reaching ADA goals Atorvastatin 20 mg Fenofibrate 200 mg Atorvastatin 20 mg + Fenofibrate 200 mg 120 97. 5 100 80 80 92. 5 100 75 60 60 40 20 30 17. 5 5 0 LDL TG Diabetes Care 2002; 25: 1198 -1202 HDL
Percentage of patients reaching ADA lipid targets % of patients reaching ADA goals Atorvastatin 20 mg Fenofibrate 200 mg Atorvastatin 20 mg + Fenofibrate 200 mg 120 97. 5 100 80 80 92. 5 100 75 60 60 40 20 30 17. 5 5 0 LDL TG Diabetes Care 2002; 25: 1198 -1202 HDL
 Clinical Trials of Fibrates Statins FIELD (Fenofibrate Intervention and Event Lowering in Diabetes) 9, 000 patients with diabetes Fenofibrate vs. placebo No statistically significant difference in the primary composite endpoint of CHD death or nonfatal MI. A significant reduction in nonfatal MI ACCORD (Action to Control Cardiovascular Risk in Diabetes) 5800 patients with type 2 diabetes in fibrate substudy Fenofibrate + statin vs. placebo + statin Study completion - 2010 FIELD. Available at: http: //www. cvm. controlled-trials. com/content/2/5/218. Accessed March 2, 2005. ACCORD. Available at: http: //www. accordtrial. org/public/index. cfm. Accessed March 2, 2005.
Clinical Trials of Fibrates Statins FIELD (Fenofibrate Intervention and Event Lowering in Diabetes) 9, 000 patients with diabetes Fenofibrate vs. placebo No statistically significant difference in the primary composite endpoint of CHD death or nonfatal MI. A significant reduction in nonfatal MI ACCORD (Action to Control Cardiovascular Risk in Diabetes) 5800 patients with type 2 diabetes in fibrate substudy Fenofibrate + statin vs. placebo + statin Study completion - 2010 FIELD. Available at: http: //www. cvm. controlled-trials. com/content/2/5/218. Accessed March 2, 2005. ACCORD. Available at: http: //www. accordtrial. org/public/index. cfm. Accessed March 2, 2005.
 PPAR- Agonists (TZDs) and Statins Metabolic effects of TZDs may be complementary to lipid-altering effects of statins: Increased LDL-C particle size Reduced TG levels (with pioglitazone) Increased HDL-C levels (with pioglitazone and rosiglitazone) Number of LDL particles Unchanged with pioglitazone Potentially worsened with rosiglitazone Bays HE. Br J Diabetes Vascular Dis. 2003; 3: 356 -360. Dis. Ginsberg HN. Am J Cardiol. 2003; 91: 29 E-39 E. Cardiol.
PPAR- Agonists (TZDs) and Statins Metabolic effects of TZDs may be complementary to lipid-altering effects of statins: Increased LDL-C particle size Reduced TG levels (with pioglitazone) Increased HDL-C levels (with pioglitazone and rosiglitazone) Number of LDL particles Unchanged with pioglitazone Potentially worsened with rosiglitazone Bays HE. Br J Diabetes Vascular Dis. 2003; 3: 356 -360. Dis. Ginsberg HN. Am J Cardiol. 2003; 91: 29 E-39 E. Cardiol.
 Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
 Fish Oils and Statins Marine fish oils rich in omega-3 fatty acids: • Lower TG levels • May be alternative to fibrate plus statin Other CV effects complementary to statins: • Reduction in malignant ventricular dysrhythmias • Increased heart rate variability • Antithrombotic effects • Improved endothelial reactivity/relaxation • Anti-inflammatory effects • Slight lowering of blood pressure Bays HE, et al. Expert Opin Pharmacother. 2003; 4: 1901 -1938. Pharmacother. Kris-Etherton PM, et al. Circulation. 2002; 106: 2747 -2757. Circulation.
Fish Oils and Statins Marine fish oils rich in omega-3 fatty acids: • Lower TG levels • May be alternative to fibrate plus statin Other CV effects complementary to statins: • Reduction in malignant ventricular dysrhythmias • Increased heart rate variability • Antithrombotic effects • Improved endothelial reactivity/relaxation • Anti-inflammatory effects • Slight lowering of blood pressure Bays HE, et al. Expert Opin Pharmacother. 2003; 4: 1901 -1938. Pharmacother. Kris-Etherton PM, et al. Circulation. 2002; 106: 2747 -2757. Circulation.
 27 Commercial Fish Oil Preparations Capsules available containing >1000 mg omega 3 fatty acids, including: Eicosapentaenoic acid (EPA) Docosahexaenoic acid (DHA) TG benefits usually associated with combined dose of EPA + DHA of 4 g/d to 9 g/d Side effects include fishy aftertaste and dyspepsia Inconsistent reports of early, short-lived, potential increase in blood glucose levels in patients with diabetes mellitus Bays HE, et al. Expert Opin Pharmacother. 2003; 4: 1901 -1938. Pharmacother. Kris-Etherton PM, et al. Circulation. 2002; 106: 2747 -2757. Circulation.
27 Commercial Fish Oil Preparations Capsules available containing >1000 mg omega 3 fatty acids, including: Eicosapentaenoic acid (EPA) Docosahexaenoic acid (DHA) TG benefits usually associated with combined dose of EPA + DHA of 4 g/d to 9 g/d Side effects include fishy aftertaste and dyspepsia Inconsistent reports of early, short-lived, potential increase in blood glucose levels in patients with diabetes mellitus Bays HE, et al. Expert Opin Pharmacother. 2003; 4: 1901 -1938. Pharmacother. Kris-Etherton PM, et al. Circulation. 2002; 106: 2747 -2757. Circulation.
 Fish Oil and Hematologic Factors May impair platelet aggregation and increase bleeding time Potentially reduces risk for thrombosis Concomitant use with anticoagulants (aspirin or warfarin): No significant increase in risk of bleeding in clinical trials Bays HE, et al. Expert Opin Pharmacother. 2003; 4: 1901 -1938. Pharmacother. Kris-Etherton PM, et al. Circulation. 2002; 106: 2747 -2757. Circulation.
Fish Oil and Hematologic Factors May impair platelet aggregation and increase bleeding time Potentially reduces risk for thrombosis Concomitant use with anticoagulants (aspirin or warfarin): No significant increase in risk of bleeding in clinical trials Bays HE, et al. Expert Opin Pharmacother. 2003; 4: 1901 -1938. Pharmacother. Kris-Etherton PM, et al. Circulation. 2002; 106: 2747 -2757. Circulation.
 Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
 Role of Niacin in Combination Therapy As monotherapy, niacin effects include: Increases in HDL-C Decreases in LDL-C, triglycerides, lipoprotein(a) Converts small LDL into more buoyant (less atherogenic) particles Safe and effective in combination with statins or bile acid sequestrants Consider tolerability profiles of different formulations May adversely effect glucose, insulin sensitivity Miller M. Mayo Clin Proc. 2003; 78: 735.
Role of Niacin in Combination Therapy As monotherapy, niacin effects include: Increases in HDL-C Decreases in LDL-C, triglycerides, lipoprotein(a) Converts small LDL into more buoyant (less atherogenic) particles Safe and effective in combination with statins or bile acid sequestrants Consider tolerability profiles of different formulations May adversely effect glucose, insulin sensitivity Miller M. Mayo Clin Proc. 2003; 78: 735.
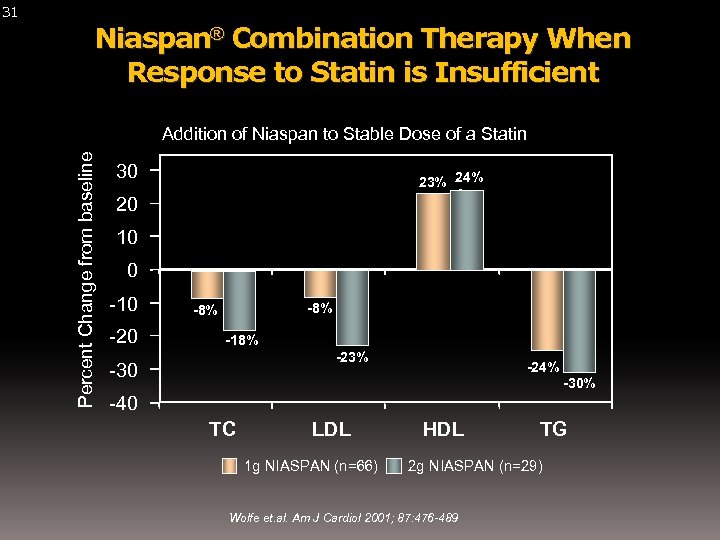 Niaspan® Combination Therapy When Response to Statin is Insufficient Addition of Niaspan to Stable Dose of a Statin Percent Change from baseline 31 30 23% 24% 20 10 0 -10 -20 -8% -18% -23% -30 -24% -30% -40 TC LDL 1 g NIASPAN (n=66) HDL TG 2 g NIASPAN (n=29) Wolfe et. al. Am J Cardiol 2001; 87: 476 -489
Niaspan® Combination Therapy When Response to Statin is Insufficient Addition of Niaspan to Stable Dose of a Statin Percent Change from baseline 31 30 23% 24% 20 10 0 -10 -20 -8% -18% -23% -30 -24% -30% -40 TC LDL 1 g NIASPAN (n=66) HDL TG 2 g NIASPAN (n=29) Wolfe et. al. Am J Cardiol 2001; 87: 476 -489
 Niacin in Combination with Statins Stein et al— 17 -week randomized trial; N=180 Immediate-release niacin plus simvastatin more effective than either drug alone at raising HLD-C and lowering VLDL-C Guyton et al— 48 -week open-label trial; N=269 ER niacin plus statin (lovastatin, pravastatin, simvastatin): Reduced TC 23%; LDL-C 32%; TG 30%; Lp(a) 19% Increased HDL-C 26% Stein et al. J Cardiovasc Pharmacol Ther. 1996; 1: 107. Guyton et al. Am J Cardiol. 1998; 82: 737.
Niacin in Combination with Statins Stein et al— 17 -week randomized trial; N=180 Immediate-release niacin plus simvastatin more effective than either drug alone at raising HLD-C and lowering VLDL-C Guyton et al— 48 -week open-label trial; N=269 ER niacin plus statin (lovastatin, pravastatin, simvastatin): Reduced TC 23%; LDL-C 32%; TG 30%; Lp(a) 19% Increased HDL-C 26% Stein et al. J Cardiovasc Pharmacol Ther. 1996; 1: 107. Guyton et al. Am J Cardiol. 1998; 82: 737.
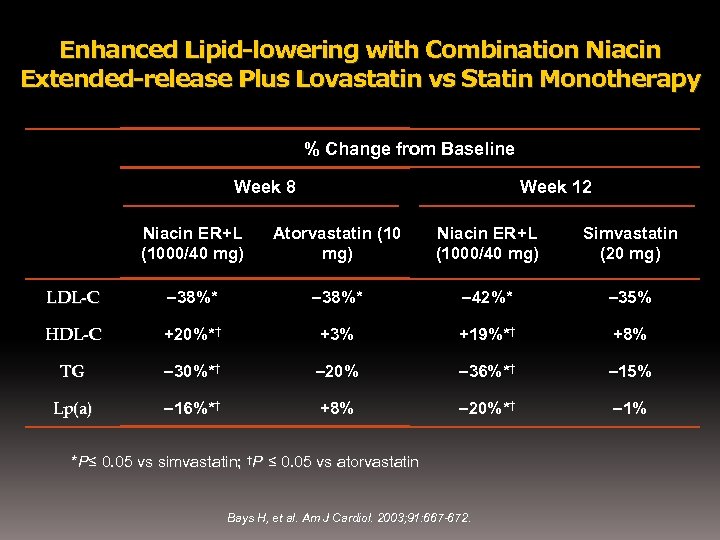 Enhanced Lipid-lowering with Combination Niacin Extended-release Plus Lovastatin vs Statin Monotherapy % Change from Baseline Week 8 Week 12 Niacin ER+L (1000/40 mg) Atorvastatin (10 mg) Niacin ER+L (1000/40 mg) Simvastatin (20 mg) LDL-C – 38%* – 42%* – 35% HDL-C +20%*† +3% +19%*† +8% TG – 30%*† – 20% – 36%*† – 15% Lp(a) – 16%*† +8% – 20%*† – 1% *P≤ 0. 05 vs simvastatin; †P ≤ 0. 05 vs atorvastatin Bays H, et al. Am J Cardiol. 2003; 91: 667 -672.
Enhanced Lipid-lowering with Combination Niacin Extended-release Plus Lovastatin vs Statin Monotherapy % Change from Baseline Week 8 Week 12 Niacin ER+L (1000/40 mg) Atorvastatin (10 mg) Niacin ER+L (1000/40 mg) Simvastatin (20 mg) LDL-C – 38%* – 42%* – 35% HDL-C +20%*† +3% +19%*† +8% TG – 30%*† – 20% – 36%*† – 15% Lp(a) – 16%*† +8% – 20%*† – 1% *P≤ 0. 05 vs simvastatin; †P ≤ 0. 05 vs atorvastatin Bays H, et al. Am J Cardiol. 2003; 91: 667 -672.
 Issues of Safety and Tolerability of Niacin and Fibrates Niacin Chief complaint is flushing, intolerable in 10% of patients Also conjunctivitis, nasal stuffiness, loose stools/diarrhea, acanthosis nigricans, ichthyosis, hepatitis Fibrates Abdominal discomfort, possible gallstones Myositis with impaired renal function Increased risk of rhabdomyolysis with fibrate or niacin + statin Statin Atorvastatin Fluvastatin Lovastatin Pravastatin Simvastatin No. of Cases of Rhabdomyolysis Associated With: Fibrates Niacin 10 Not reported 4 Not reported 5 1 6 1 33 2 Knopp RH. N Engl J Med. 1999; 341: 498 -511; Jones PH, Davidson MH. Am J Cardiol. 2005; 95: 120 -122; Bellosta S, et al. Circulation. 2004; 109(23 Suppl 1): III 50 -57
Issues of Safety and Tolerability of Niacin and Fibrates Niacin Chief complaint is flushing, intolerable in 10% of patients Also conjunctivitis, nasal stuffiness, loose stools/diarrhea, acanthosis nigricans, ichthyosis, hepatitis Fibrates Abdominal discomfort, possible gallstones Myositis with impaired renal function Increased risk of rhabdomyolysis with fibrate or niacin + statin Statin Atorvastatin Fluvastatin Lovastatin Pravastatin Simvastatin No. of Cases of Rhabdomyolysis Associated With: Fibrates Niacin 10 Not reported 4 Not reported 5 1 6 1 33 2 Knopp RH. N Engl J Med. 1999; 341: 498 -511; Jones PH, Davidson MH. Am J Cardiol. 2005; 95: 120 -122; Bellosta S, et al. Circulation. 2004; 109(23 Suppl 1): III 50 -57
 Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
Agents Commonly Used in Combination with Statins Bile acid sequestrants PPAR agonists (fibrates) Fish oils Niacin Ezetimibe
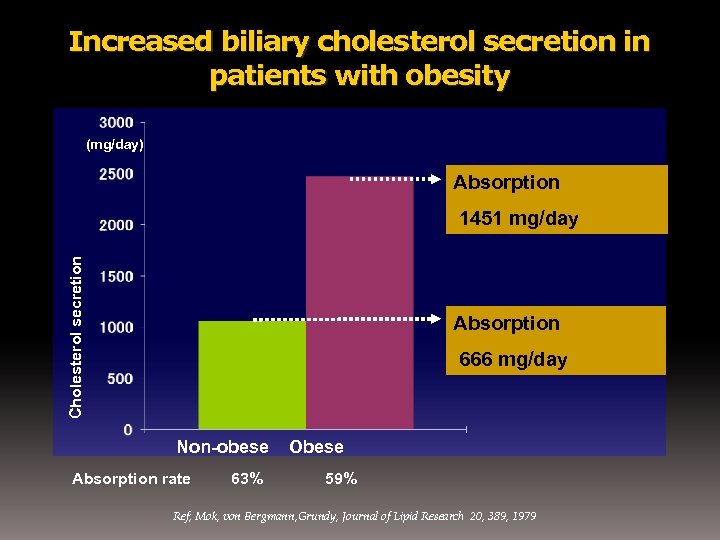 Increased biliary cholesterol secretion in patients with obesity (mg/day) Absorption Cholesterol secretion 1451 mg/day Absorption 666 mg/day Non-obese Absorption rate 63% Obese 59% Ref; Mok, von Bergmann, Grundy, Journal of Lipid Research 20, 389, 1979
Increased biliary cholesterol secretion in patients with obesity (mg/day) Absorption Cholesterol secretion 1451 mg/day Absorption 666 mg/day Non-obese Absorption rate 63% Obese 59% Ref; Mok, von Bergmann, Grundy, Journal of Lipid Research 20, 389, 1979
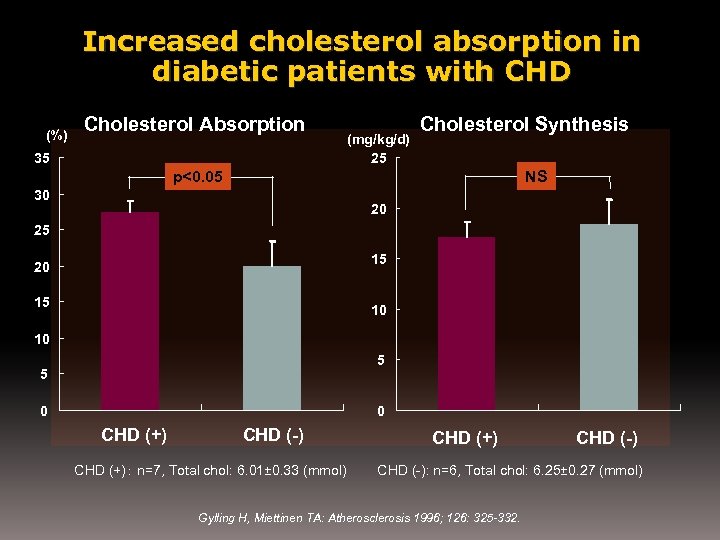 Increased cholesterol absorption in diabetic patients with CHD (%) Cholesterol Absorption 35 (mg/kg/d) 25 Cholesterol Synthesis NS p<0. 05 30 20 25 15 20 15 10 10 5 5 0 0 CHD (+) CHD (-) CHD (+): n=7, Total chol: 6. 01± 0. 33 (mmol) CHD (+) CHD (-): n=6, Total chol: 6. 25± 0. 27 (mmol) Gylling H, Miettinen TA: Atherosclerosis 1996; 126: 325 -332.
Increased cholesterol absorption in diabetic patients with CHD (%) Cholesterol Absorption 35 (mg/kg/d) 25 Cholesterol Synthesis NS p<0. 05 30 20 25 15 20 15 10 10 5 5 0 0 CHD (+) CHD (-) CHD (+): n=7, Total chol: 6. 01± 0. 33 (mmol) CHD (+) CHD (-): n=6, Total chol: 6. 25± 0. 27 (mmol) Gylling H, Miettinen TA: Atherosclerosis 1996; 126: 325 -332.
 Ezetimibe Blocks Cholesterol Metabolism at a New Site Bile acid sequestrants Bile acids X Free cholesterol Brush Border XX Sterols/ Micelles Remnants Statins Unstirred water layer Cholesterol DIET stanols X CE Synthesis Plaque formation ENTEROCYTE BLOOD FC biosynthesis FC X ACAT Cholesteryl Ester (CE) CE Chylomicrons Cholesterol Absorption Inhibitors (e. g. , ezetimibe)
Ezetimibe Blocks Cholesterol Metabolism at a New Site Bile acid sequestrants Bile acids X Free cholesterol Brush Border XX Sterols/ Micelles Remnants Statins Unstirred water layer Cholesterol DIET stanols X CE Synthesis Plaque formation ENTEROCYTE BLOOD FC biosynthesis FC X ACAT Cholesteryl Ester (CE) CE Chylomicrons Cholesterol Absorption Inhibitors (e. g. , ezetimibe)
 Ezetimibe: Indications Hypercholesterolemia Monotherapy Combination therapy with statins Homozygous familial hypercholesterolemia Homozygous sitosterolemia Note: no data are yet available on effects of ezetimibe (alone or in combination therapy) on CHD morbidity and mortality Bays H. Expert Opin Investig Drugs. 2002; 11: 1587 -1604. Drugs.
Ezetimibe: Indications Hypercholesterolemia Monotherapy Combination therapy with statins Homozygous familial hypercholesterolemia Homozygous sitosterolemia Note: no data are yet available on effects of ezetimibe (alone or in combination therapy) on CHD morbidity and mortality Bays H. Expert Opin Investig Drugs. 2002; 11: 1587 -1604. Drugs.
 Ezetimibe: Mechanism of Action Selective inhibition of intestinal cholesterol absorption • Intestinal delivery of cholesterol to liver • Expression of hepatic LDL receptors • Cholesterol content of atherogenic particles Enterohepatic circulation of ezetimibe and active glucuronide metabolite • Delivers agent back to site of action • Limits systemic exposure Bays H. Expert Opin Investig Drugs. 2002; 11: 1587 -1604. Drugs.
Ezetimibe: Mechanism of Action Selective inhibition of intestinal cholesterol absorption • Intestinal delivery of cholesterol to liver • Expression of hepatic LDL receptors • Cholesterol content of atherogenic particles Enterohepatic circulation of ezetimibe and active glucuronide metabolite • Delivers agent back to site of action • Limits systemic exposure Bays H. Expert Opin Investig Drugs. 2002; 11: 1587 -1604. Drugs.
 Low Systemic Exposure of Ezetimibe May Reduce Potential for Adverse Effects/ Drug Interactions 3 H-DPM (x 10– 6) 3 h post-3 H ezetimibe IV dosing Intestinal lumen Intestinal wall Plasma Liver van Heek M, et al. Br J Pharmacol. 2000; 129: 1748 -1754. Bile
Low Systemic Exposure of Ezetimibe May Reduce Potential for Adverse Effects/ Drug Interactions 3 H-DPM (x 10– 6) 3 h post-3 H ezetimibe IV dosing Intestinal lumen Intestinal wall Plasma Liver van Heek M, et al. Br J Pharmacol. 2000; 129: 1748 -1754. Bile
 Ezetimibe: Dosage and Administration 10 mg once daily Any time of day, with or without food May be taken concomitantly with statin Dosage adjustment not necessary according to Gender Mild hepatic or renal insufficiency Age (ie, elderly) Ezetimibe not recommended in children <10 years old Bays H. Expert Opin Investig Drugs. 2002; 11: 1587 -1604. Drugs.
Ezetimibe: Dosage and Administration 10 mg once daily Any time of day, with or without food May be taken concomitantly with statin Dosage adjustment not necessary according to Gender Mild hepatic or renal insufficiency Age (ie, elderly) Ezetimibe not recommended in children <10 years old Bays H. Expert Opin Investig Drugs. 2002; 11: 1587 -1604. Drugs.
 Ezetimibe: Contraindications Hypersensitivity to any component of agent Combination with statins in patients with active liver disease or unexplained persistent elevations in serum transaminases Pregnancy C category as monotherapy (no adequate studies in pregnant women) All statins are contraindicated in pregnant and nursing women Precaution: Moderate or severe hepatic insufficiency Bays H. Expert Opin Investig Drugs. 2002; 11: 1587 -1604.
Ezetimibe: Contraindications Hypersensitivity to any component of agent Combination with statins in patients with active liver disease or unexplained persistent elevations in serum transaminases Pregnancy C category as monotherapy (no adequate studies in pregnant women) All statins are contraindicated in pregnant and nursing women Precaution: Moderate or severe hepatic insufficiency Bays H. Expert Opin Investig Drugs. 2002; 11: 1587 -1604.
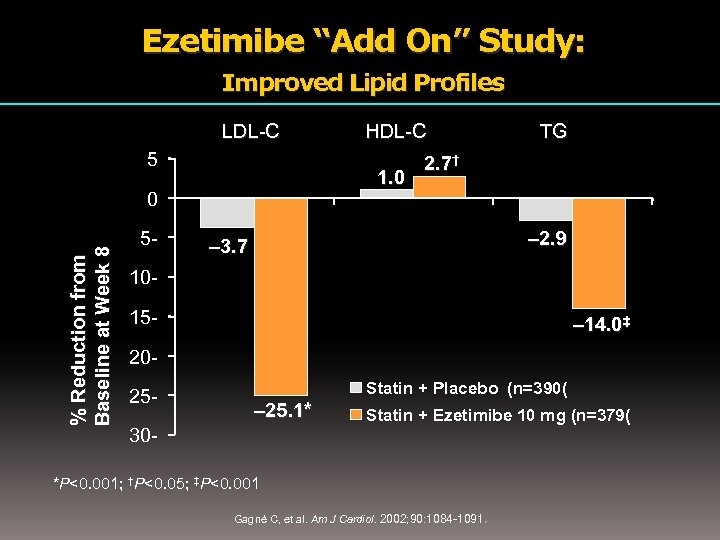 Ezetimibe “Add On” Study: Improved Lipid Profiles LDL-C 5 1. 0 % Reduction from Baseline at Week 8 0 5 - HDL-C TG 2. 7† – 2. 9 – 3. 7 1015 - – 14. 0‡ 2025 - Statin + Placebo (n=390( – 25. 1* 30 - Statin + Ezetimibe 10 mg (n=379( *P<0. 001; †P<0. 05; ‡P<0. 001 . Gagné C, et al. Am J Cardiol. 2002; 90: 1084 -1091 Cardiol.
Ezetimibe “Add On” Study: Improved Lipid Profiles LDL-C 5 1. 0 % Reduction from Baseline at Week 8 0 5 - HDL-C TG 2. 7† – 2. 9 – 3. 7 1015 - – 14. 0‡ 2025 - Statin + Placebo (n=390( – 25. 1* 30 - Statin + Ezetimibe 10 mg (n=379( *P<0. 001; †P<0. 05; ‡P<0. 001 . Gagné C, et al. Am J Cardiol. 2002; 90: 1084 -1091 Cardiol.
 LDL-C Reduction across the Dose Range VYTORIN™ vs. Simvastatin Efficacy Study 10/20 mg Mean % change from baseline to week 12 0 20 mg 10/40 mg 10/80 mg (n=86) (n=89) (n=90) (n=91) (n=87) – 10 – 20 – 35% – 40 – 50 – 60 – 70 – 42% – 51%* – 46% – 55%* – 61%* VYTORIN Simvastatin *p<0. 001 vs. corresponding dose of simvastatin Adapted from Goldberg AC et al Mayo Clin Proc 2004; 79: 620– 629. VYTORIN™ (ezetimibe/simvastatin) is a trademark of MSP Singapore Company, LLC.
LDL-C Reduction across the Dose Range VYTORIN™ vs. Simvastatin Efficacy Study 10/20 mg Mean % change from baseline to week 12 0 20 mg 10/40 mg 10/80 mg (n=86) (n=89) (n=90) (n=91) (n=87) – 10 – 20 – 35% – 40 – 50 – 60 – 70 – 42% – 51%* – 46% – 55%* – 61%* VYTORIN Simvastatin *p<0. 001 vs. corresponding dose of simvastatin Adapted from Goldberg AC et al Mayo Clin Proc 2004; 79: 620– 629. VYTORIN™ (ezetimibe/simvastatin) is a trademark of MSP Singapore Company, LLC.
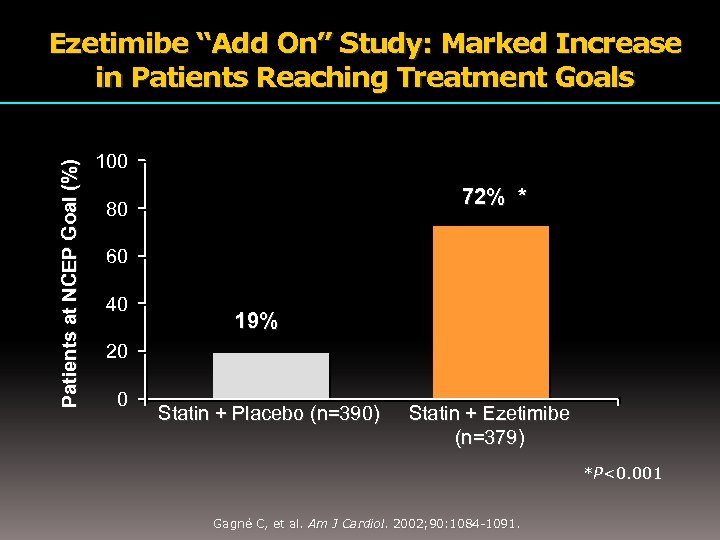 Patients at NCEP Goal (%) Ezetimibe “Add On” Study: Marked Increase in Patients Reaching Treatment Goals 100 72% * 80 60 40 19% 20 0 Statin + Placebo (n=390) Statin + Ezetimibe (n=379) *P<0. 001 Gagné C, et al. Am J Cardiol. 2002; 90: 1084 -1091.
Patients at NCEP Goal (%) Ezetimibe “Add On” Study: Marked Increase in Patients Reaching Treatment Goals 100 72% * 80 60 40 19% 20 0 Statin + Placebo (n=390) Statin + Ezetimibe (n=379) *P<0. 001 Gagné C, et al. Am J Cardiol. 2002; 90: 1084 -1091.
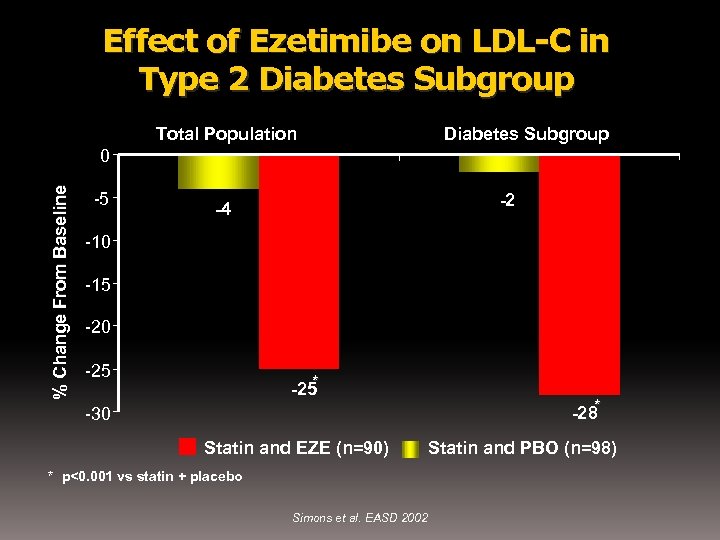 Effect of Ezetimibe on LDL-C in Type 2 Diabetes Subgroup Total Population Diabetes Subgroup % Change From Baseline 0 -5 -2 -4 -10 -15 -20 -25 * -25 -30 Statin and EZE (n=90) * p<0. 001 vs statin + placebo Simons et al. EASD 2002 -28* Statin and PBO (n=98)
Effect of Ezetimibe on LDL-C in Type 2 Diabetes Subgroup Total Population Diabetes Subgroup % Change From Baseline 0 -5 -2 -4 -10 -15 -20 -25 * -25 -30 Statin and EZE (n=90) * p<0. 001 vs statin + placebo Simons et al. EASD 2002 -28* Statin and PBO (n=98)
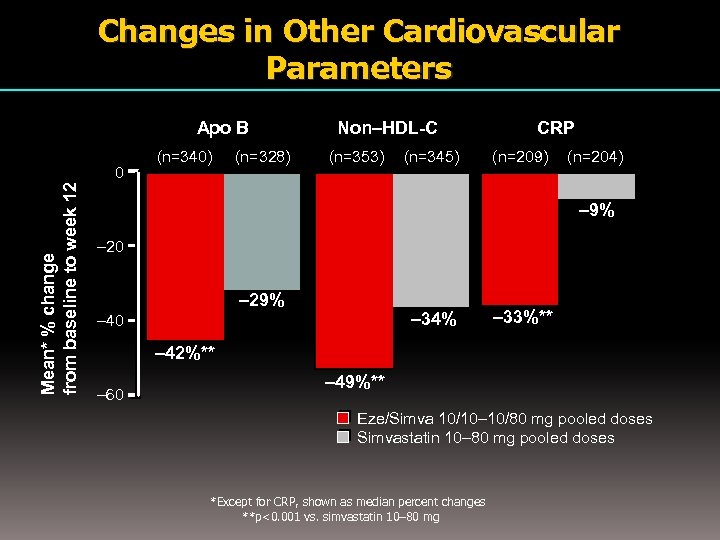 Changes in Other Cardiovascular Parameters Apo B Mean* % change from baseline to week 12 0 (n=340) (n=328) Non–HDL-C (n=353) (n=345) CRP (n=209) (n=204) – 9% – 20 – 29% – 34% – 40 – 33%** – 42%** – 60 – 49%** Eze/Simva 10/10– 10/80 mg pooled doses Simvastatin 10– 80 mg pooled doses *Except for CRP, shown as median percent changes **p<0. 001 vs. simvastatin 10– 80 mg
Changes in Other Cardiovascular Parameters Apo B Mean* % change from baseline to week 12 0 (n=340) (n=328) Non–HDL-C (n=353) (n=345) CRP (n=209) (n=204) – 9% – 20 – 29% – 34% – 40 – 33%** – 42%** – 60 – 49%** Eze/Simva 10/10– 10/80 mg pooled doses Simvastatin 10– 80 mg pooled doses *Except for CRP, shown as median percent changes **p<0. 001 vs. simvastatin 10– 80 mg
 Statin and Complementary GI-Acting Drugs vs Statin Titration 6% Statin at starting dose 6% 1 st 6% 2 nd 3 rd 3 -STEP TITRATION Doubling 18% Statin at starting dose + GI-acting drug % Reduction in LDL-C Bays H, et al. Expert Opin Pharmacother. 2003; 4: 779 -790. Pharmacother. 1 -STEP COADMINISTRATION
Statin and Complementary GI-Acting Drugs vs Statin Titration 6% Statin at starting dose 6% 1 st 6% 2 nd 3 rd 3 -STEP TITRATION Doubling 18% Statin at starting dose + GI-acting drug % Reduction in LDL-C Bays H, et al. Expert Opin Pharmacother. 2003; 4: 779 -790. Pharmacother. 1 -STEP COADMINISTRATION
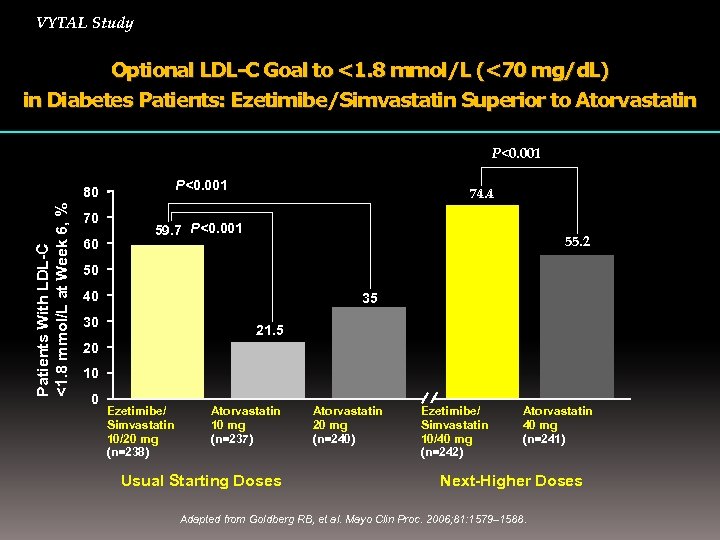 VYTAL Study Optional LDL-C Goal to <1. 8 mmol/L (<70 mg/d. L) in Diabetes Patients: Ezetimibe/Simvastatin Superior to Atorvastatin P<0. 001 Patients With LDL-C <1. 8 mmol/L at Week 6, % 80 70 60 74. 4 59. 7 P<0. 001 55. 2 50 40 35 30 21. 5 20 10 0 Ezetimibe/ Simvastatin 10/20 mg (n=238) Atorvastatin 10 mg (n=237) Usual Starting Doses Atorvastatin 20 mg (n=240) Ezetimibe/ Simvastatin 10/40 mg (n=242) Atorvastatin 40 mg (n=241) Next-Higher Doses Adapted from Goldberg RB, et al. Mayo Clin Proc. 2006; 81: 1579– 1588.
VYTAL Study Optional LDL-C Goal to <1. 8 mmol/L (<70 mg/d. L) in Diabetes Patients: Ezetimibe/Simvastatin Superior to Atorvastatin P<0. 001 Patients With LDL-C <1. 8 mmol/L at Week 6, % 80 70 60 74. 4 59. 7 P<0. 001 55. 2 50 40 35 30 21. 5 20 10 0 Ezetimibe/ Simvastatin 10/20 mg (n=238) Atorvastatin 10 mg (n=237) Usual Starting Doses Atorvastatin 20 mg (n=240) Ezetimibe/ Simvastatin 10/40 mg (n=242) Atorvastatin 40 mg (n=241) Next-Higher Doses Adapted from Goldberg RB, et al. Mayo Clin Proc. 2006; 81: 1579– 1588.
 52 Large Clinical Outcomes Program in >21, 000 Patients at High-Risk of CV Events Study, N Study Population Treatments Primary Endpoint ENHANCE, ~725 He. FH Ezetimibe/ simvastatin 10/80 mg Simvastatin 80 mg CA IMT SHARP, ~9000 Chronic kidney disease Ezetimibe/ simvastatin 10/20 mg Placebo CV outcomes (MI, stroke, coronary revascularization) SEAS, ~1400 Asymptomatic aortic stenosis with LDL-C <6 mmol/L Ezetimibe/ simvastatin 10/40 mg Placebo CV death, aortic surgery, CV outcomes Ezetimibe/ simvastatin 10/40 mg Simvastatin 40 mg CV death, major coronary events, stroke IMPROVE-IT, ACS ~10, 000 CA IMT = Carotid artery intima-media thickness Adapted from Kastelein JJP, et al. Am Heart J. 2005; 149: 234– 239; Baigent C, Landry M. Kidney Int. 2003; 63(suppl 84): S 207–S 210; Oxford Clinical Trial Service Unit. The Study of Heart and Renal Protection (SHARP). Available at: http: //www. ctsu. ox. ac. uk/ ~sharp/. Accessed June 2005; Rossebo A, et al, for the SEAS Steering Committee. Presented at: XIII International Symposium on Atherosclerosis; September 23–October 2, 2003; Kyoto, Japan. Poster 3 P-0870; Schering-Plough. IMPROVE -IT: Examining outcomes in subjects with acute coronary syndrome: Vytorin (ezetimibe/simvastatin) vs simvastatin (Study P 04103). Available at: http: //www. clinicaltrials. gov/ct/show/NCT 00202878. Accessed November 2006.
52 Large Clinical Outcomes Program in >21, 000 Patients at High-Risk of CV Events Study, N Study Population Treatments Primary Endpoint ENHANCE, ~725 He. FH Ezetimibe/ simvastatin 10/80 mg Simvastatin 80 mg CA IMT SHARP, ~9000 Chronic kidney disease Ezetimibe/ simvastatin 10/20 mg Placebo CV outcomes (MI, stroke, coronary revascularization) SEAS, ~1400 Asymptomatic aortic stenosis with LDL-C <6 mmol/L Ezetimibe/ simvastatin 10/40 mg Placebo CV death, aortic surgery, CV outcomes Ezetimibe/ simvastatin 10/40 mg Simvastatin 40 mg CV death, major coronary events, stroke IMPROVE-IT, ACS ~10, 000 CA IMT = Carotid artery intima-media thickness Adapted from Kastelein JJP, et al. Am Heart J. 2005; 149: 234– 239; Baigent C, Landry M. Kidney Int. 2003; 63(suppl 84): S 207–S 210; Oxford Clinical Trial Service Unit. The Study of Heart and Renal Protection (SHARP). Available at: http: //www. ctsu. ox. ac. uk/ ~sharp/. Accessed June 2005; Rossebo A, et al, for the SEAS Steering Committee. Presented at: XIII International Symposium on Atherosclerosis; September 23–October 2, 2003; Kyoto, Japan. Poster 3 P-0870; Schering-Plough. IMPROVE -IT: Examining outcomes in subjects with acute coronary syndrome: Vytorin (ezetimibe/simvastatin) vs simvastatin (Study P 04103). Available at: http: //www. clinicaltrials. gov/ct/show/NCT 00202878. Accessed November 2006.
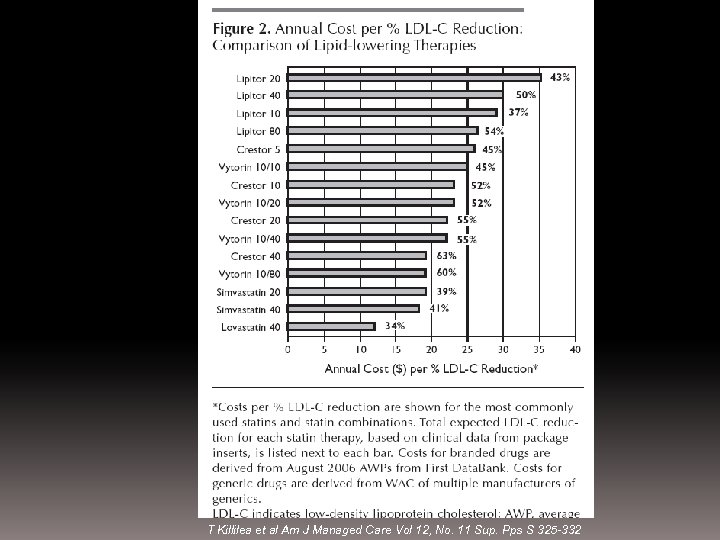 T Killilea et al Am J Managed Care Vol 12, No. 11 Sup. Pps S 325 -332
T Killilea et al Am J Managed Care Vol 12, No. 11 Sup. Pps S 325 -332
 54 Conclusion: Different lipid-lowering agents affect different lipid parameters Combination therapy may further reduce CHD risk vs monotherapy Consider combination therapy if: Treatment goals cannot be met with statins alone Risk for intolerance, toxicity, or adverse drug interactions with higher dose of single lipidlowering agent
54 Conclusion: Different lipid-lowering agents affect different lipid parameters Combination therapy may further reduce CHD risk vs monotherapy Consider combination therapy if: Treatment goals cannot be met with statins alone Risk for intolerance, toxicity, or adverse drug interactions with higher dose of single lipidlowering agent
 55 Conclusions Inhibition of cholesterol absorption and production via coadministration of ezetimibe with a statin or ezetimibe/simvastatin in the single tablet Is helping to set a new standard Provides greater efficacy than statin therapy alone to get patients to a lower LDL-C goal Outcomes studies will determine if the risk of cardiac endpoints decreases as LDL-C levels decrease with ezetimibe/simvastatin
55 Conclusions Inhibition of cholesterol absorption and production via coadministration of ezetimibe with a statin or ezetimibe/simvastatin in the single tablet Is helping to set a new standard Provides greater efficacy than statin therapy alone to get patients to a lower LDL-C goal Outcomes studies will determine if the risk of cardiac endpoints decreases as LDL-C levels decrease with ezetimibe/simvastatin
 Treatment Paradigm Combination therapy-Statin plus what? For LDL Cholesterol absorption inhibitor – Ezetimibe/Ezetrol® Bile acid sequestrant For HDL Niacin/Niaspan®/Fibrate For TG Fibrate (fenofibrate)/Niacin/Niaspan®
Treatment Paradigm Combination therapy-Statin plus what? For LDL Cholesterol absorption inhibitor – Ezetimibe/Ezetrol® Bile acid sequestrant For HDL Niacin/Niaspan®/Fibrate For TG Fibrate (fenofibrate)/Niacin/Niaspan®
 Thank you
Thank you
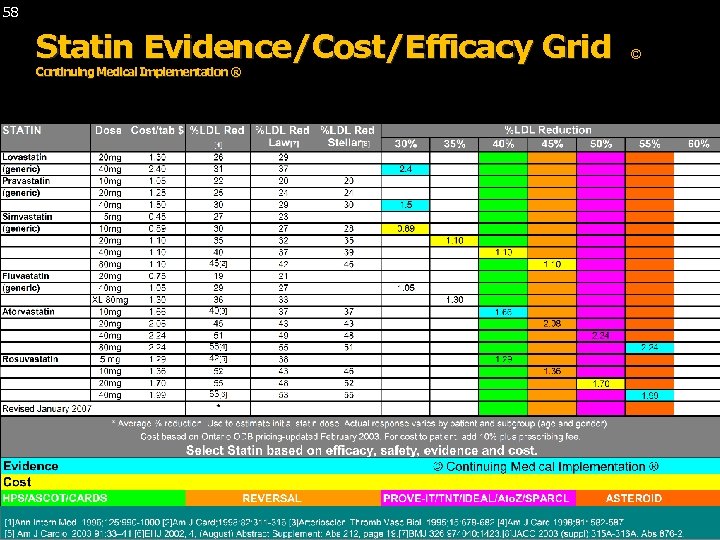 58 Statin Evidence/Cost/Efficacy Grid Continuing Medical Implementation ® © Continuing Medical Implementation ® …. . . bridging the care gap ©
58 Statin Evidence/Cost/Efficacy Grid Continuing Medical Implementation ® © Continuing Medical Implementation ® …. . . bridging the care gap ©
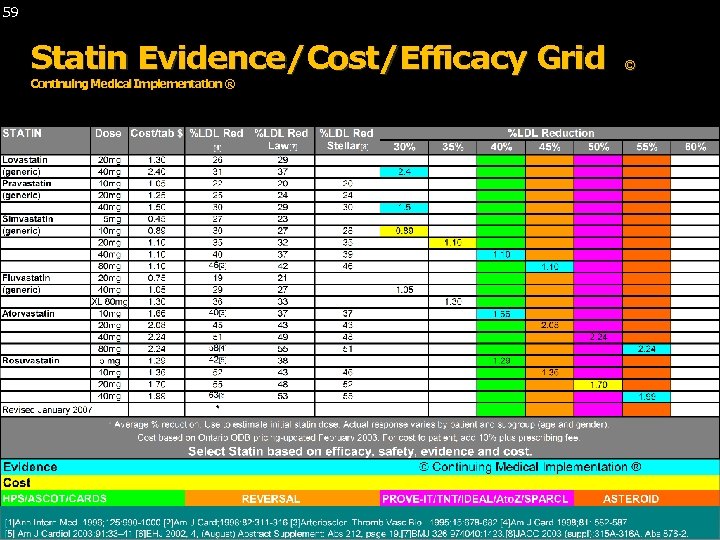 59 Statin Evidence/Cost/Efficacy Grid Continuing Medical Implementation ® © Continuing Medical Implementation ® …. . . bridging the care gap ©
59 Statin Evidence/Cost/Efficacy Grid Continuing Medical Implementation ® © Continuing Medical Implementation ® …. . . bridging the care gap ©
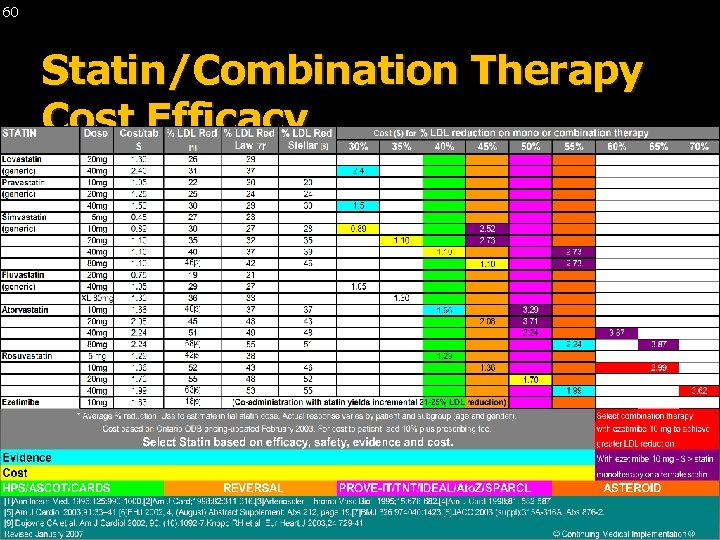 60 Statin/Combination Therapy Cost Efficacy © Continuing Medical Implementation ® …. . . bridging the care gap
60 Statin/Combination Therapy Cost Efficacy © Continuing Medical Implementation ® …. . . bridging the care gap


