89e0dab48483caee05f0affda4bd8ad7.ppt
- Количество слайдов: 43

Lupin Limited Corporate Presentation November 2006

Safe Harbour Statement Materials and information provided during this presentation may contain ‘forward-looking statements’. These statements are based on current expectations, forecasts and assumptions that are subject to risks and uncertainties which could cause actual outcomes and results to differ materially from these statements. Risks and uncertainties include general industry and market conditions, and general domestic and international economic conditions such as interest rate and currency exchange fluctuations. Risks and uncertainties particularly apply with respect to product-related forward-looking statements. Product risks and uncertainties include, but are not limited, to technological advances and patents attained by competitors, challenges inherent in new product development, including completion of clinical trials; claims and concerns about product safety and efficacy; obtaining regulatory approvals; domestic and foreign healthcare reforms; trends toward managed care and healthcare cost containment, and governmental laws and regulations affecting domestic and foreign operations. Also, for products that are approved, there are manufacturing and marketing risks and uncertainties, which include, but are not limited, to inability to build production capacity to meet demand, unavailability of raw materials, and failure to gain market acceptance. The Company disclaims any intention or obligation to update or revise any forward-looking statements whether as a result of new information, future events or otherwise. 2

Contents – – Global Pharmaceutical Industry Overview Indian Pharmaceutical Industry Lupin -Business Update 2006 -07 and Beyond 3

Section 1 Introduction. . . 4

Vision • To be an Innovation led Transnational Pharmaceutical Company Values • • • Superior Performance Entrepreneurship Customer Orientation Working Together Respect for People Integrity 5

Section 2 Pharmaceutical Industry Overview 6

Global Pharmaceutical Industry Overview u. Global Pharmaceutical market at US$600 billion market in 2005 · Market expected to grow at a rate of 6 -7% u. Ten major markets account for almost 81% of the market in terms of revenue · North America is the single largest market accounting for approx. 50% of global sales and is expected to grow at a CAGR of 5 -8% · Japan, the second largest market, at US$60 bn is growing at 6% · EU experiencing growth at 7% at US$ 170 bn u. Growing importance of generics in regulated markets · Pressure from healthcare providers and insurance companies to reduce healthcare costs driving usage of generic drugs · Increasing patent expirations driving generic drug availability u. Stringent testing requirements and declining R&D productivity has led to limited new drug pipeline 7

Indian Pharma – Emerging Scenario Domestic Market: US$ 5 bn Exports: US$ 2. 5 bn Total: US$ 7. 5 bn CONTRACT MANUFACTURING • JV/ Alliances • Competition from China • § § DOMESTIC FORMULATIONS IPR Regime Pricing pressure on ‘older’ molecules Brands/ differentiated products Likely to witness higher growth In Licensing Indian Pharma • § § GENERICS BUSINESS Increasing competitive landscape Pricing pressure Aggressive build up of products Acquisitions US$ 60 bn opportunity INDIGENOUS R&D • ANDA/NDDS/NCE • Out Licensing • High Investment/ Long gestation 8

Section 3 Approach 9

Lupin Strives For Sustainable Earnings Accelerated Growth Fulfilling Stakeholder Aspirations 10

Creating an Edge Technology • • • New Delivery Systems Complex Products Bottom Quartile of costs for generic products Integration NCE Markets • • Expansion & Efficiency in Sales Force ‘Direct to Market’ approach in US Build on Paediatric On-shore presence in 5 select countries Alliances • • Partnerships in Advanced as well as Emerging markets TB Institutional Alliances with Global majors for certain markets New therapies (In-Licensing) 11

Section 4 Advanced Markets 12
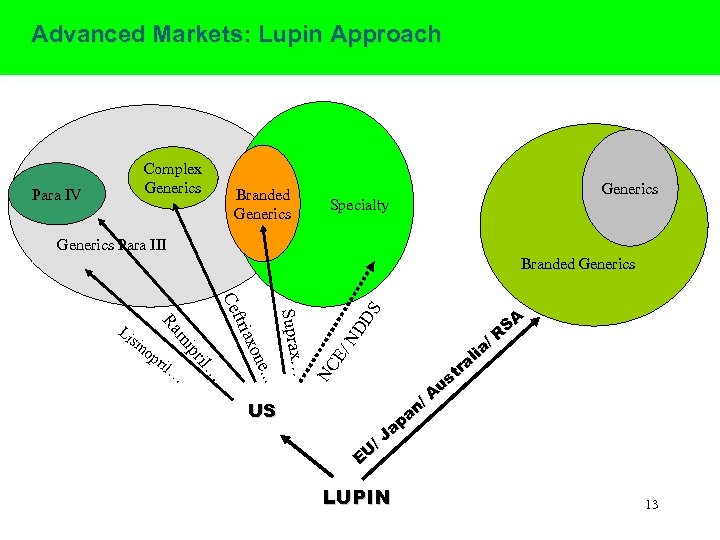
Advanced Markets: Lupin Approach Complex Generics Para IV Branded Generics Specialty Generics Para III US SA R a/ i al r st u A n/ a ap /J EU ND E/ NC Suprax… e. . . … xon tria Cef ril il… op pr mi sin Ra Li DS Branded Generics LUPIN 13

US Branded · Suprax continues its prescription growth: over 5000 Rx per week · Sales force internalised · Copromotion of Atopiclair with Chester Valley · New Products in 2006 -07 Generics · 7 products launched in the US in FY 06; 3 launched till date in FY 07 · Timely launch of Ceftriaxone and Cefprozil immediately on expiry · Launched Lisinopril in Dec 05: Consolidating Market share · Commenced Direct Marketing (DTM) in Dec 05 · Total ANDAs filed: 44 14
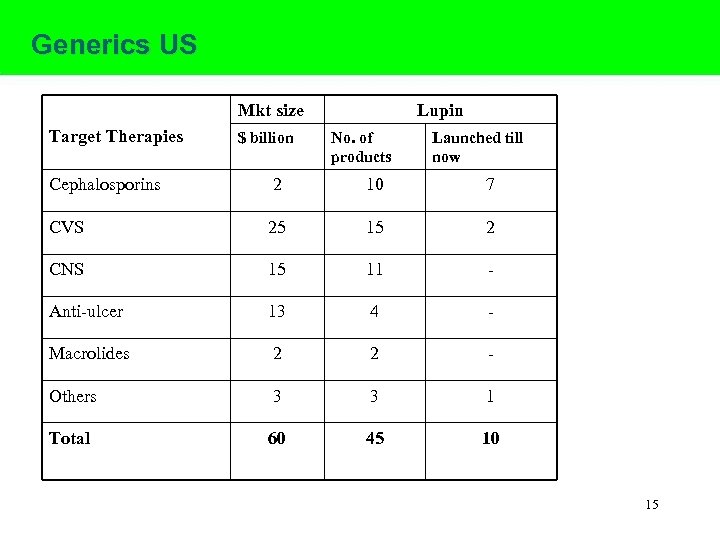
Generics US Mkt size Target Therapies $ billion Lupin No. of products Launched till now Cephalosporins 2 10 7 CVS 25 15 2 CNS 15 11 - Anti-ulcer 13 4 - Macrolides 2 2 - Others 3 3 1 Total 60 45 10 15

Other Markets Europe • Ramping up MAA/ Dossier filings • Alliances in progress • Revenues commenced • Expressions of interest at advanced stages Japan, Australia & LA • Expressions of interest moving forward • MDR-TB sales commenced in LA South Africa • JV with Aspen (Anti-TB FDC) for Africa 16
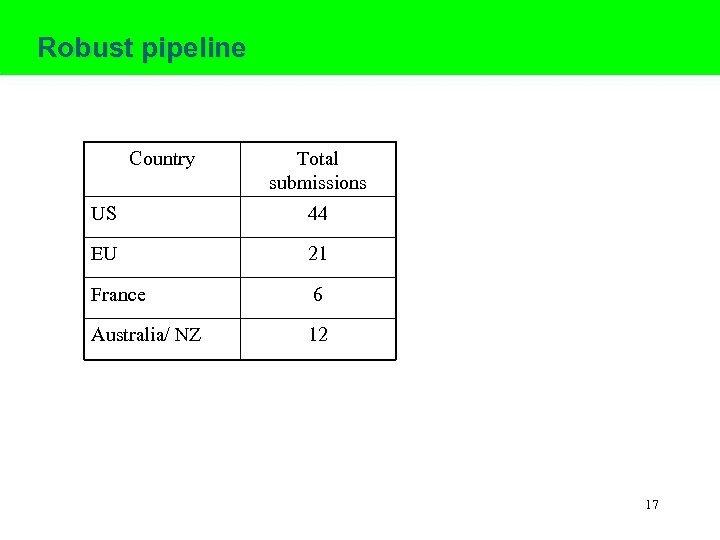
Robust pipeline Country Total submissions US 44 EU 21 France 6 Australia/ NZ 12 17

Section 5 Emerging Markets 18
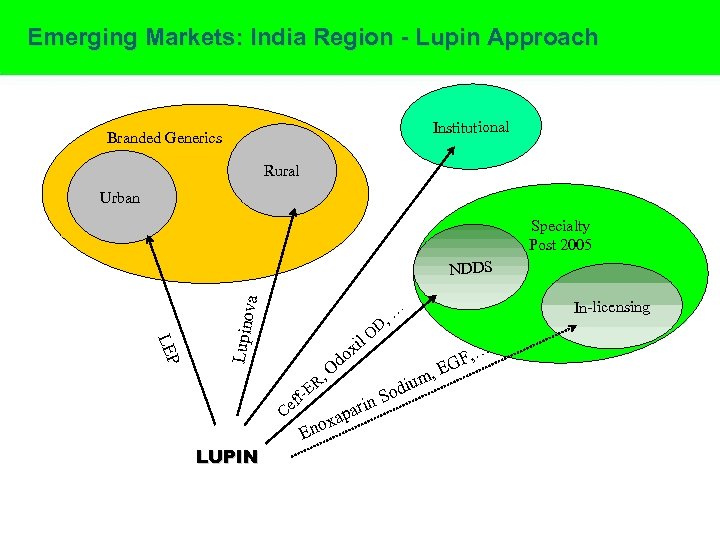
Emerging Markets: India Region - Lupin Approach Institutional Branded Generics Rural Urban Specialty Post 2005 LEP Lupino va NDDS D O il , R f-E f Ce LUPIN In-licensing , … x do O rin pa oxa En i Sod um F , EG , …
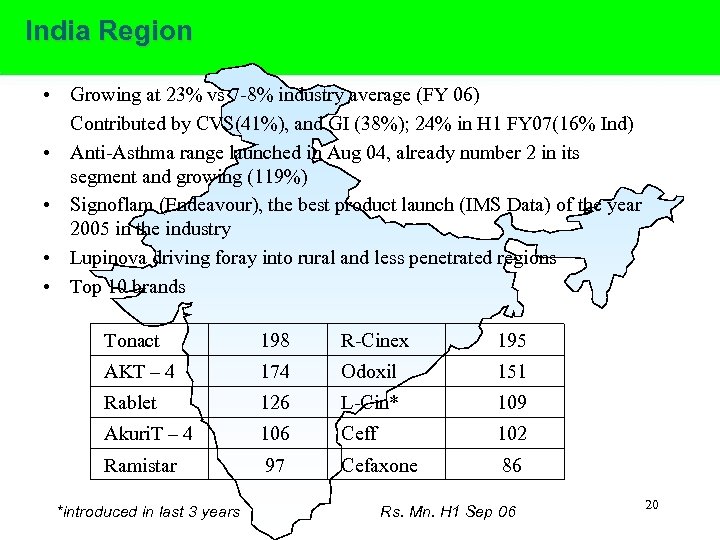
India Region • Growing at 23% vs 7 -8% industry average (FY 06) Contributed by CVS(41%), and GI (38%); 24% in H 1 FY 07(16% Ind) • Anti-Asthma range launched in Aug 04, already number 2 in its segment and growing (119%) • Signoflam (Endeavour), the best product launch (IMS Data) of the year 2005 in the industry • Lupinova driving foray into rural and less penetrated regions • Top 10 brands Tonact 198 R-Cinex 195 AKT – 4 174 Odoxil 151 Rablet 126 L-Cin* 109 Akuri. T – 4 106 Ceff 102 Ramistar 97 Cefaxone 86 *introduced in last 3 years Rs. Mn. H 1 Sep 06 20
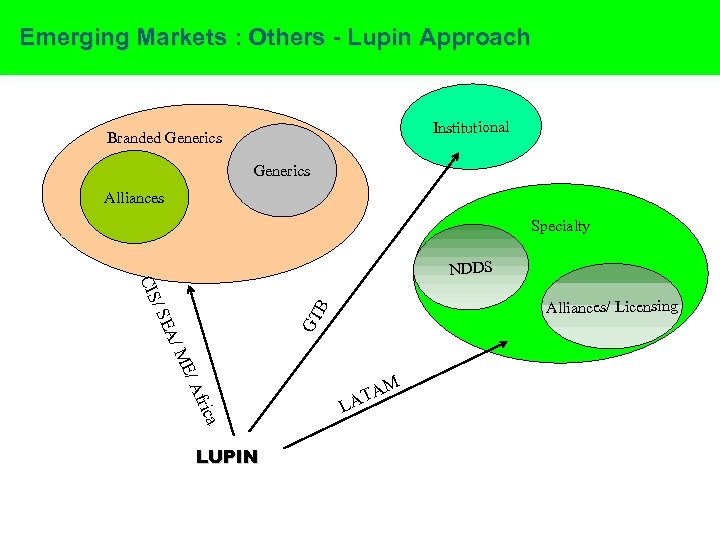
Emerging Markets : Others - Lupin Approach Institutional Branded Generics Alliances Specialty NDDS A/ GT B / SE CIS Alliances/ Licensing /A ME fric a LUPIN AM AT L

Other Markets CIS • Posted a healthy growth – CAGR of 44% over last 4 years • Expanding Field Force – • Russia, Ukraine, Azerbaijan, Kazakhstan, Uzbekistan • Increased market penetration • Around 20 Products Registered in each; around 10 under Registration SE Asia • Alliance with GSK Middle East • Will be shared as Alliances progress Africa • Alliance with Ranbaxy GTB • Posted a healthy growth – 122% 22

Section 6 API & Intermediates 23

API & Intermediates • Sales : 11% growth; 13% in H 1 FY 07 • Focus on Cost & Quality Leadership and Reliability • Long Term Supply Contracts • Global Leadership in areas of therapeutic presence • Future expansion only at SEZs 24
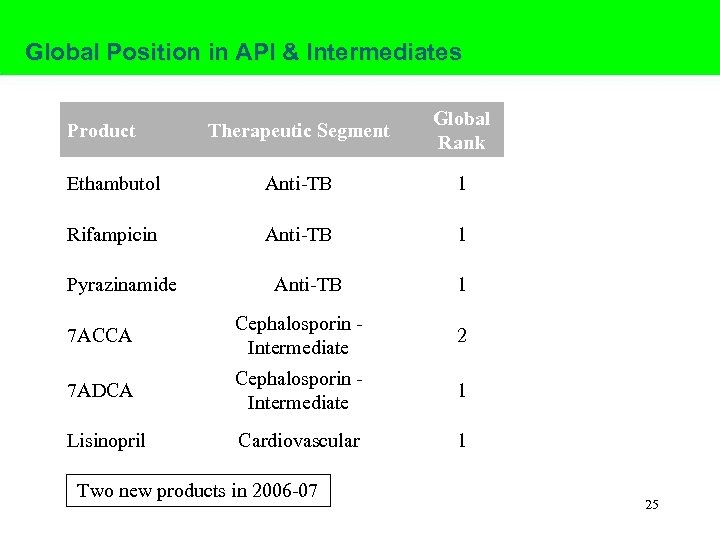
Global Position in API & Intermediates Therapeutic Segment Global Rank Ethambutol Anti-TB 1 Rifampicin Anti-TB 1 Product Pyrazinamide Anti-TB 1 7 ACCA Cephalosporin Intermediate 2 7 ADCA Cephalosporin Intermediate 1 Lisinopril Cardiovascular 1 Two new products in 2006 -07 25

Section 7 Research & Development 26

R&D • Over 400 scientists – Actively engaged in: Process Improvement/ Formulation Development/NCE/ NDDS • Expenditure (FY 0506) : Rs. 1030 mn – 6. 2% of Sales – Continue investing incrementally • Filings rate to continue • Going Forward – – NDDS Paediatrics range Patented Technology platforms Products for paediatric pipeline 27

R&D • NCEs – – Anti Psoriasis (Herbal) Anti Migraine Anti Psoriasis (Chemical) Anti TB - In Phase II - Nearing completion of Phase II - In Phase I 28
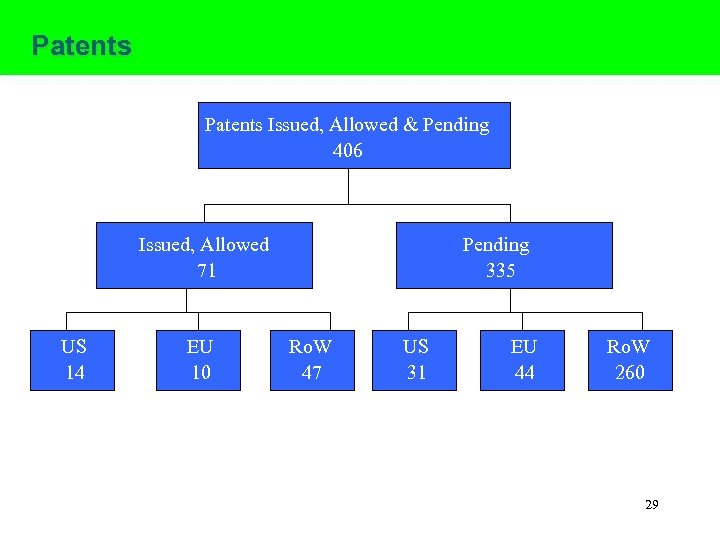
Patents Issued, Allowed & Pending 406 Issued, Allowed 71 US 14 EU 10 Pending 335 Ro. W 47 US 31 EU 44 EU Ro. W 260 29

Section 8 Business Update 30
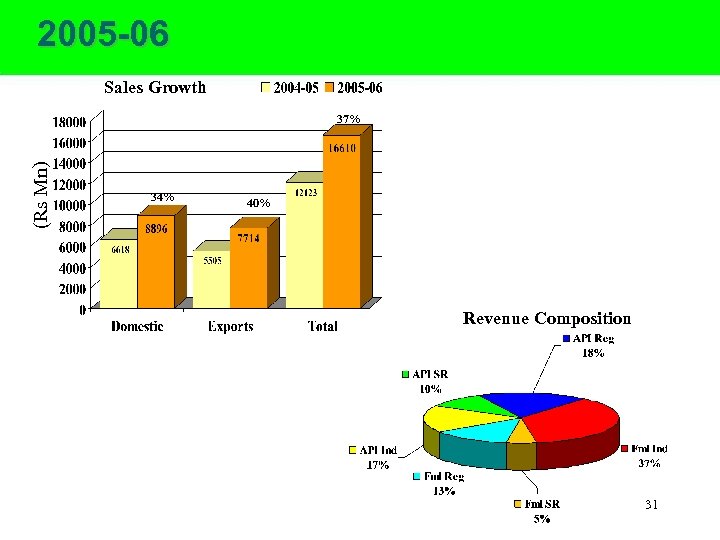
2005 -06 Sales Growth (Rs Mn) 37% 34% 40% Revenue Composition 31
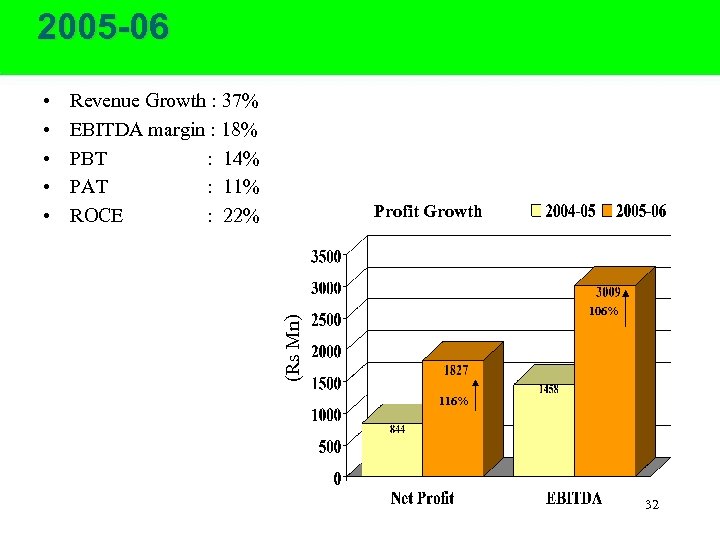
2005 -06 Revenue Growth : 37% EBITDA margin : 18% PBT : 14% PAT : 11% ROCE : 22% Profit Growth 106% (Rs Mn) • • • 116% 32
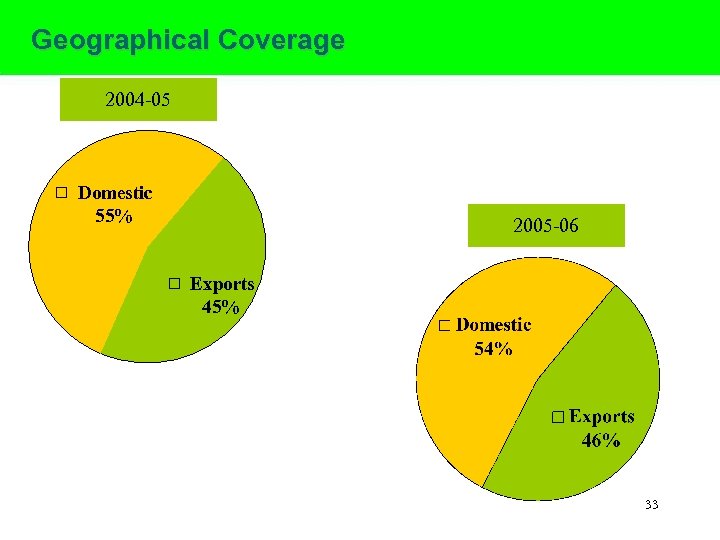
Geographical Coverage 2004 -05 Domestic 55% 2005 -06 Exports 45% 33
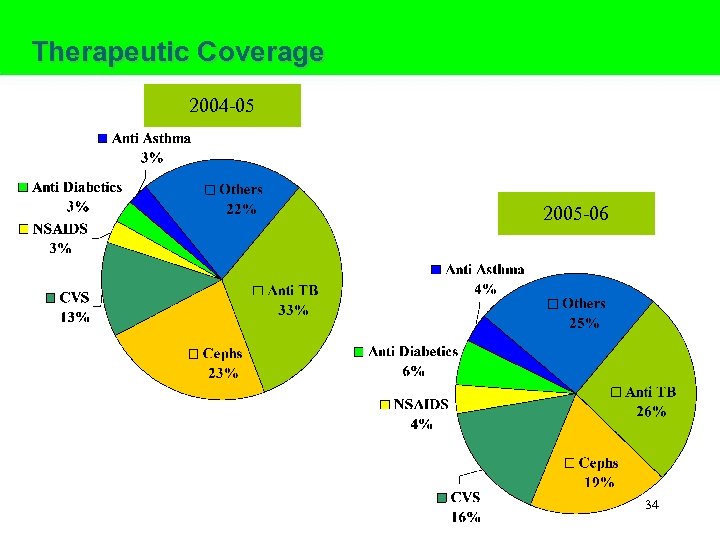
Therapeutic Coverage 2004 -05 2005 -06 34
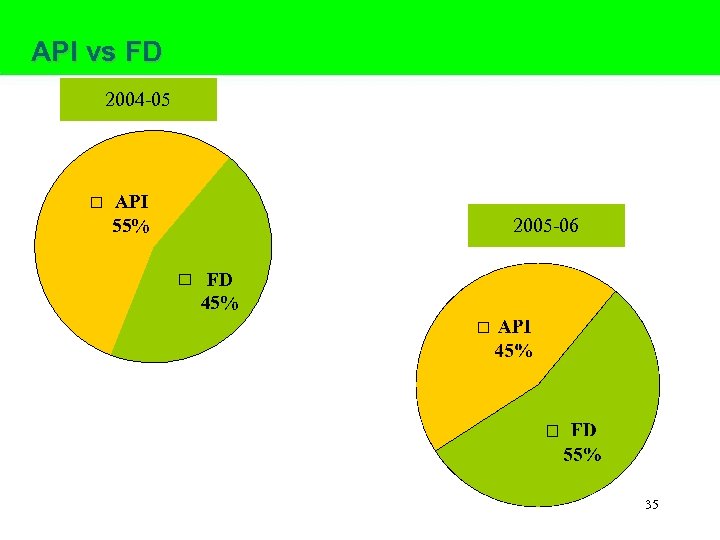
API vs FD 2004 -05 API 55% 2005 -06 FD 45% 35
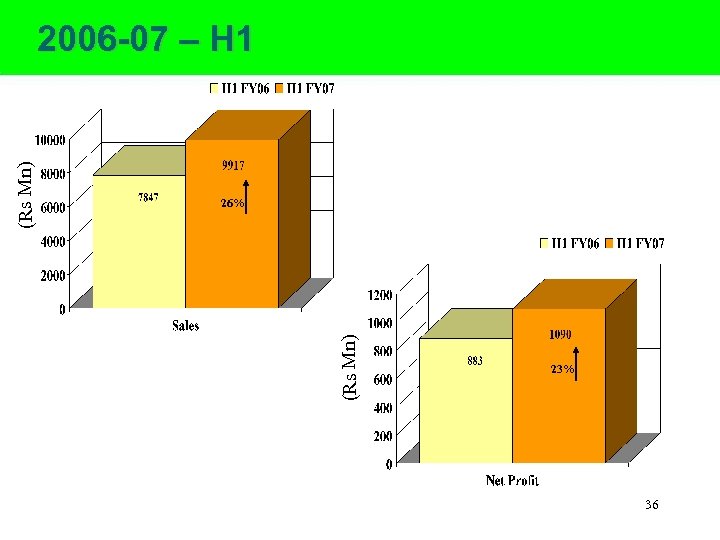
26% (Rs Mn) 2006 -07 – H 1 23% 36

Section 9 Facilities 37
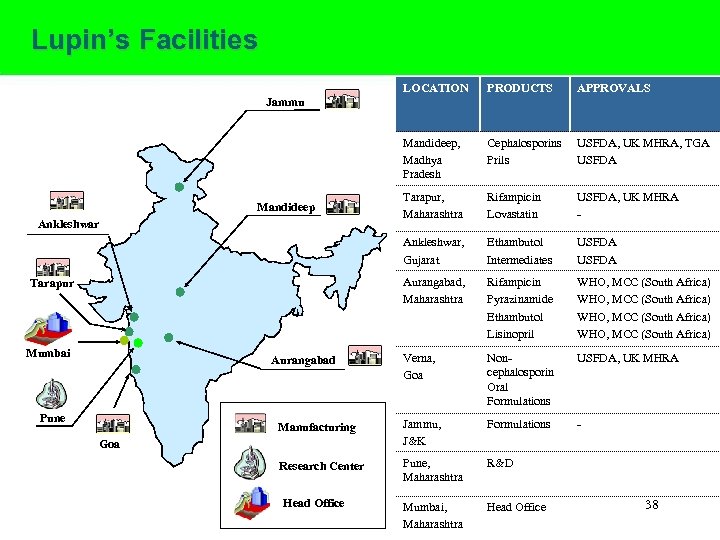
Lupin’s Facilities LOCATION PRODUCTS APPROVALS Mandideep, Madhya Pradesh Cephalosporins Prils USFDA, UK MHRA, TGA USFDA Tarapur, Maharashtra Rifampicin Lovastatin USFDA, UK MHRA - Ankleshwar, Gujarat Ethambutol Intermediates USFDA Aurangabad, Maharashtra Rifampicin Pyrazinamide Ethambutol Lisinopril WHO, MCC (South Africa) Verna, Goa Noncephalosporin Oral Formulations USFDA, UK MHRA Manufacturing Jammu, J&K Formulations - Research Center Pune, Maharashtra R&D Mumbai, Maharashtra Head Office Jammu Mandideep Ankleshwar Tarapur Mumbai Aurangabad Pune Goa Head Office 38

Section 10 Management 39
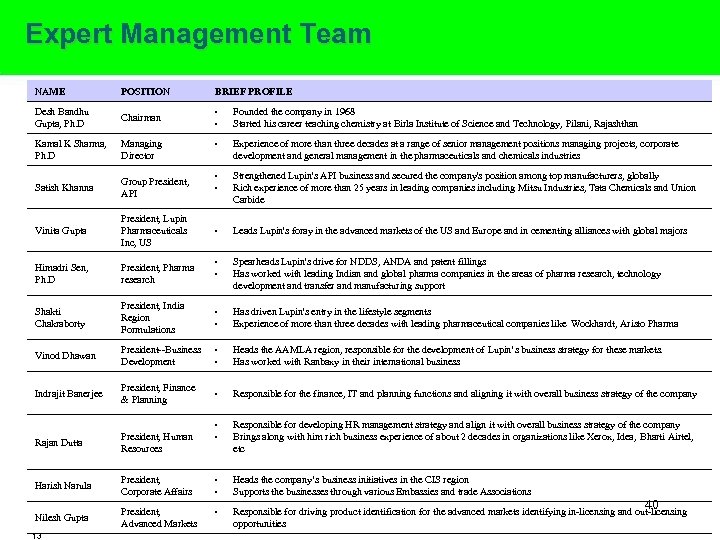
Expert Management Team NAME POSITION BRIEF PROFILE Desh Bandhu Gupta, Ph. D Chairman • • Founded the company in 1968 Started his career teaching chemistry at Birla Institute of Science and Technology, Pilani, Rajashthan Kamal K Sharma, Ph. D Managing Director • Experience of more than three decades at a range of senior management positions managing projects, corporate development and general management in the pharmaceuticals and chemicals industries Satish Khanna Group President, API • • Strengthened Lupin's API business and secured the company's position among top manufacturers, globally Rich experience of more than 25 years in leading companies including Mitsu Industries, Tata Chemicals and Union Carbide Vinita Gupta President, Lupin Pharmaceuticals Inc, US • Leads Lupin's foray in the advanced markets of the US and Europe and in cementing alliances with global majors Himadri Sen, Ph. D President, Pharma research • • Spearheads Lupin's drive for NDDS, ANDA and patent fillings Has worked with leading Indian and global pharma companies in the areas of pharma research, technology development and transfer and manufacturing support Shakti Chakraborty President, India Region Formulations • • Has driven Lupin's entry in the lifestyle segments Experience of more than three decades with leading pharmaceutical companies like Wockhardt, Aristo Pharma Vinod Dhawan President--Business Development • • Heads the AAMLA region, responsible for the development of Lupin’s business strategy for these markets. Has worked with Ranbaxy in their international business Indrajit Banerjee President, Finance & Planning • Responsible for the finance, IT and planning functions and aligning it with overall business strategy of the company Rajan Dutta President, Human Resources • • Responsible for developing HR management strategy and align it with overall business strategy of the company Brings along with him rich business experience of about 2 decades in organizations like Xerox, Idea, Bharti Airtel, etc Harish Narula President, Corporate Affairs • Heads the company’s business initiatives in the CIS region Supports the businesses through various Embassies and trade Associations Nilesh Gupta President, Advanced Markets • 13 • 40 Responsible for driving product identification for the advanced markets identifying in-licensing and out-licensing opportunities

Section 11 Year 2006 -07 and Beyond. . . 41

Going Forward Ø New Markets/ Initiatives • Enter Advanced Markets - EU - Japan - Australia - South Africa • Establish On-shore presence in 5 select markets • Acquisition: - Brands in US - Businesses in select countries of EU - Domestic Ø Explore New Therapies/ Business Segments 42

Thank you 43
89e0dab48483caee05f0affda4bd8ad7.ppt