UNIVERSITY 4 2011.ppt
- Количество слайдов: 147
 Лекции 7 -8 Каталитические аутоантитела в медецине и биотехнологии Габибов А. Г.
Лекции 7 -8 Каталитические аутоантитела в медецине и биотехнологии Габибов А. Г.
 Ферменты – биокатализаторы жизни – были отобраны естественной эволюцией для обеспечения развития живых клеток и целых организмов в их естественной среде. Поэтому, природные ферменты не всегда обладают адекватными свойствами, специфичностью, стабильностью и каталитической активностью для применения их в биотехнологии. Jacques Fastrez
Ферменты – биокатализаторы жизни – были отобраны естественной эволюцией для обеспечения развития живых клеток и целых организмов в их естественной среде. Поэтому, природные ферменты не всегда обладают адекватными свойствами, специфичностью, стабильностью и каталитической активностью для применения их в биотехнологии. Jacques Fastrez
 Каталитические антитела Anti. Bodies ENzymes Abzymes (Абзимы)
Каталитические антитела Anti. Bodies ENzymes Abzymes (Абзимы)
 Каталитические антитела. Emil Fisher, Berichte Deutsh. Chem. Ges. , 1894, vol. 27, p. 2985 “The Lock and Key” approach William Jencks, Catalysis in Chemistry and Enzymology, Mc. Grow Hill, 1969 Catalytic properties of anti-transition state antibodies Richard Lerner, Stephen Benkovich, Peter Shultz Science, 1991, 252(5006): 659 -67. At the crossroads of chemistry and immunology: catalytic antibodies. Linus Pauling, Am. Sci. , 1948, vol. 36, p. 51 ”Transition state” concept Niels Jerne, Ann. Inst. Pasteur. Immunol. , 125 C, 1974, p. 373 Network theory of Immune System Paul S et al. Catalytic hydrolysis of vasoactive intestinal peptide by human autoantibody. Science. 1989; v. 244, pp. 1158 -62 Shuster AM et al. DNA hydrolyzing autoantibodies. Science. 1992 v. 256 pp. 665 -7
Каталитические антитела. Emil Fisher, Berichte Deutsh. Chem. Ges. , 1894, vol. 27, p. 2985 “The Lock and Key” approach William Jencks, Catalysis in Chemistry and Enzymology, Mc. Grow Hill, 1969 Catalytic properties of anti-transition state antibodies Richard Lerner, Stephen Benkovich, Peter Shultz Science, 1991, 252(5006): 659 -67. At the crossroads of chemistry and immunology: catalytic antibodies. Linus Pauling, Am. Sci. , 1948, vol. 36, p. 51 ”Transition state” concept Niels Jerne, Ann. Inst. Pasteur. Immunol. , 125 C, 1974, p. 373 Network theory of Immune System Paul S et al. Catalytic hydrolysis of vasoactive intestinal peptide by human autoantibody. Science. 1989; v. 244, pp. 1158 -62 Shuster AM et al. DNA hydrolyzing autoantibodies. Science. 1992 v. 256 pp. 665 -7
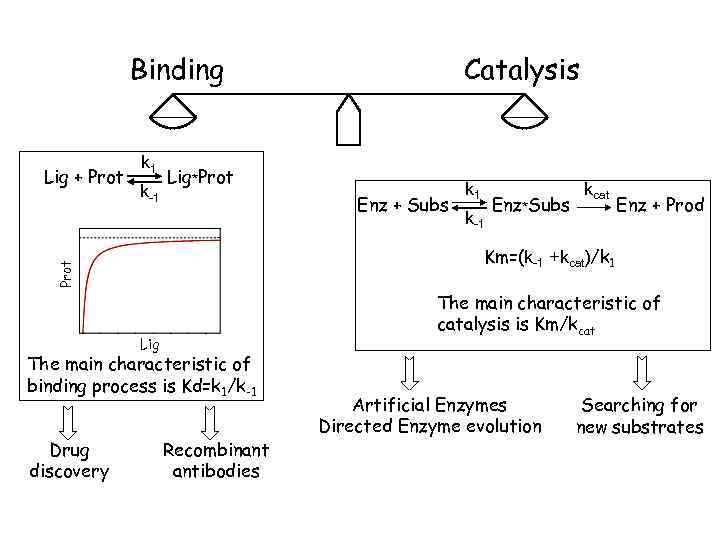 Binding k 1 k-1 Lig*Prot Enz + Subs k-1 Enz*Subs kcat Enz + Prod The main characteristic of catalysis is Km/kcat Lig The main characteristic of binding process is Kd=k 1/k-1 Drug discovery k 1 Km=(k-1 +kcat)/k 1 Prot Lig + Prot Catalysis Recombinant antibodies Artificial Enzymes Directed Enzyme evolution Searching for new substrates
Binding k 1 k-1 Lig*Prot Enz + Subs k-1 Enz*Subs kcat Enz + Prod The main characteristic of catalysis is Km/kcat Lig The main characteristic of binding process is Kd=k 1/k-1 Drug discovery k 1 Km=(k-1 +kcat)/k 1 Prot Lig + Prot Catalysis Recombinant antibodies Artificial Enzymes Directed Enzyme evolution Searching for new substrates
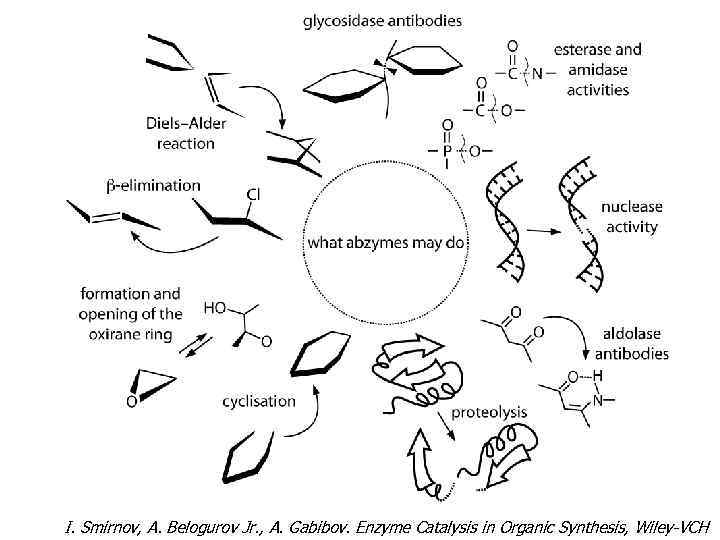 I. Smirnov, A. Belogurov Jr. , A. Gabibov. Enzyme Catalysis in Organic Synthesis, Wiley-VCH
I. Smirnov, A. Belogurov Jr. , A. Gabibov. Enzyme Catalysis in Organic Synthesis, Wiley-VCH
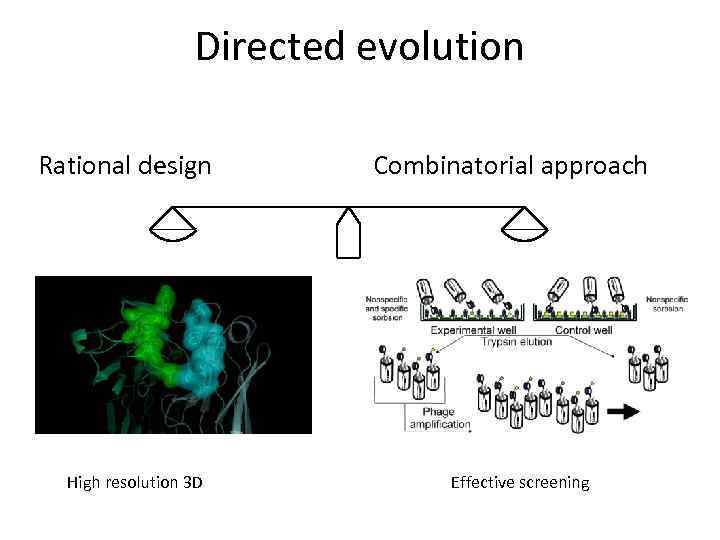 Directed evolution Rational design High resolution 3 D Combinatorial approach Effective screening
Directed evolution Rational design High resolution 3 D Combinatorial approach Effective screening
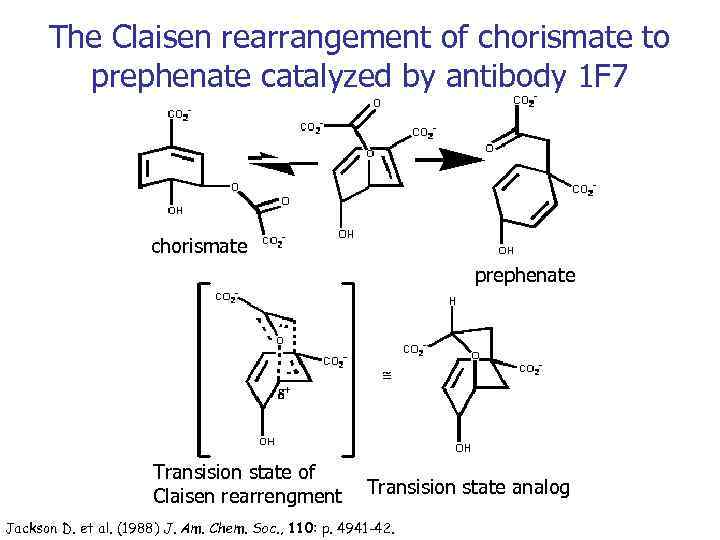 The Claisen rearrangement of chorismate to prephenate catalyzed by antibody 1 F 7 chorismate prephenate Transision state of Claisen rearrengment Transision state analog Jackson D. et al. (1988) J. Am. Chem. Soc. , 110: p. 4941 -42.
The Claisen rearrangement of chorismate to prephenate catalyzed by antibody 1 F 7 chorismate prephenate Transision state of Claisen rearrengment Transision state analog Jackson D. et al. (1988) J. Am. Chem. Soc. , 110: p. 4941 -42.
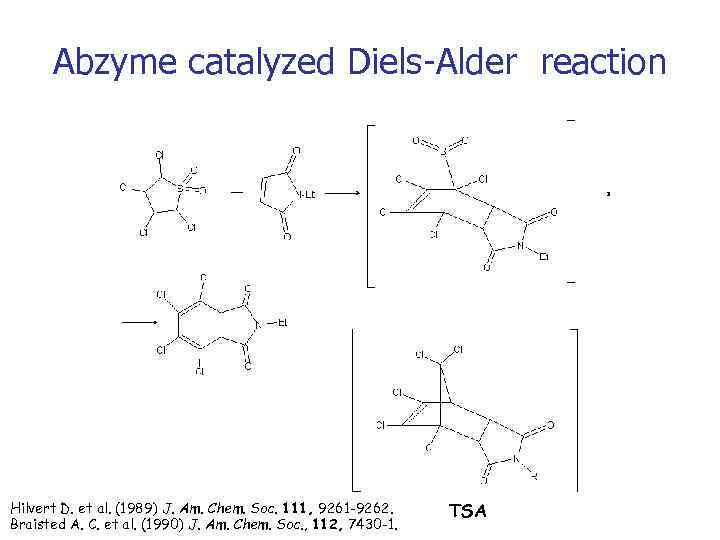 Abzyme catalyzed Diels-Alder reaction Hilvert D. et al. (1989) J. Am. Chem. Soc. 111, 9261 -9262. Braisted A. C. et al. (1990) J. Am. Chem. Soc. , 112, 7430 -1. TSA
Abzyme catalyzed Diels-Alder reaction Hilvert D. et al. (1989) J. Am. Chem. Soc. 111, 9261 -9262. Braisted A. C. et al. (1990) J. Am. Chem. Soc. , 112, 7430 -1. TSA
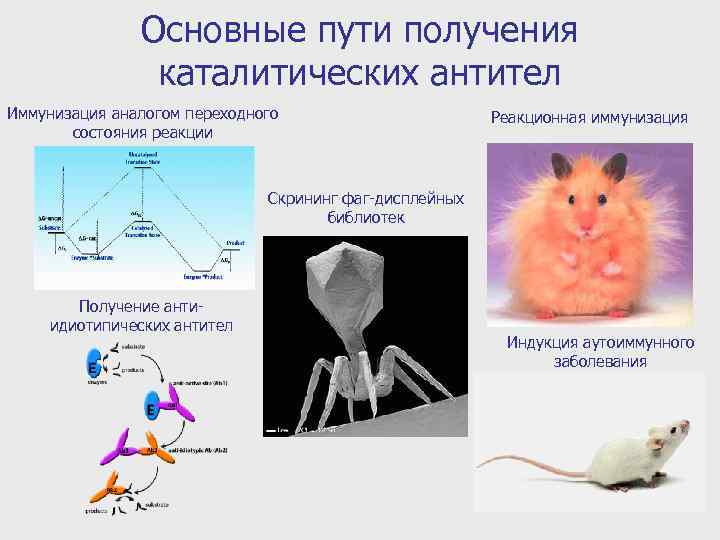 Основные пути получения каталитических антител Иммунизация аналогом переходного состояния реакции Реакционная иммунизация Скрининг фаг-дисплейных библиотек Получение антиидиотипических антител Индукция аутоиммунного заболевания
Основные пути получения каталитических антител Иммунизация аналогом переходного состояния реакции Реакционная иммунизация Скрининг фаг-дисплейных библиотек Получение антиидиотипических антител Индукция аутоиммунного заболевания
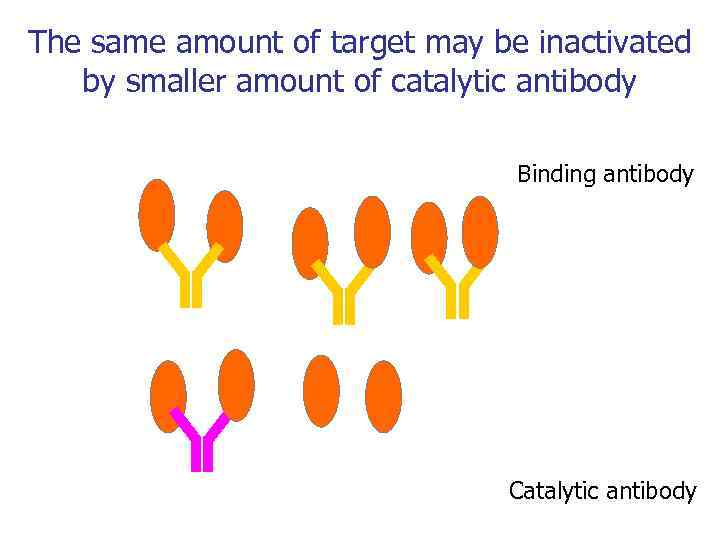 The same amount of target may be inactivated by smaller amount of catalytic antibody Binding antibody Catalytic antibody
The same amount of target may be inactivated by smaller amount of catalytic antibody Binding antibody Catalytic antibody
 Почему терапия каталитическими антителами может быть эффективна? • Долгое время жизни в кровотоке • Высокая специфичность • Одно и то же количество антигена может быть инактивированно меньшим количеством каталитического антитела + Богатый мировой опыт в антительной терапии + Широкие возможности по созданию и производству м. Ат + Мышиный репертуар может быть заменен на человеческий - Низкая скорость
Почему терапия каталитическими антителами может быть эффективна? • Долгое время жизни в кровотоке • Высокая специфичность • Одно и то же количество антигена может быть инактивированно меньшим количеством каталитического антитела + Богатый мировой опыт в антительной терапии + Широкие возможности по созданию и производству м. Ат + Мышиный репертуар может быть заменен на человеческий - Низкая скорость
 Каталитические антитела: • Способны акцептировать или каталитически уничтожать: фосфорорганические вещества, химическое оружие, пестициды (Smirnov et al. PNAS 2011) • Способны каталитически уничтожать: белки-патогены белки оболочки вирусов и бактерий высокий уровень цитокинов при инфекциях • Способны гидролизовать препараты в кровотоке кокаин (Mets B. et al PNAS (1998), 95, 10176 -81) • Способны превращать пролекарства в их активную форму раковая химеотерапия (Wentworth P. et al PNAS (1996), 93, 799 -83)
Каталитические антитела: • Способны акцептировать или каталитически уничтожать: фосфорорганические вещества, химическое оружие, пестициды (Smirnov et al. PNAS 2011) • Способны каталитически уничтожать: белки-патогены белки оболочки вирусов и бактерий высокий уровень цитокинов при инфекциях • Способны гидролизовать препараты в кровотоке кокаин (Mets B. et al PNAS (1998), 95, 10176 -81) • Способны превращать пролекарства в их активную форму раковая химеотерапия (Wentworth P. et al PNAS (1996), 93, 799 -83)
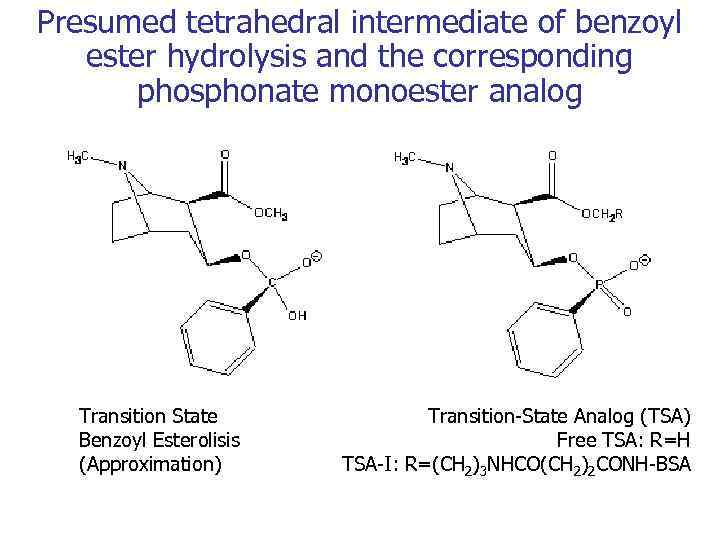 Presumed tetrahedral intermediate of benzoyl ester hydrolysis and the corresponding phosphonate monoester analog Transition State Benzoyl Esterolisis (Approximation) Transition-State Analog (TSA) Free TSA: R=H TSA-I: R=(CH 2)3 NHCO(CH 2)2 CONH-BSA
Presumed tetrahedral intermediate of benzoyl ester hydrolysis and the corresponding phosphonate monoester analog Transition State Benzoyl Esterolisis (Approximation) Transition-State Analog (TSA) Free TSA: R=H TSA-I: R=(CH 2)3 NHCO(CH 2)2 CONH-BSA
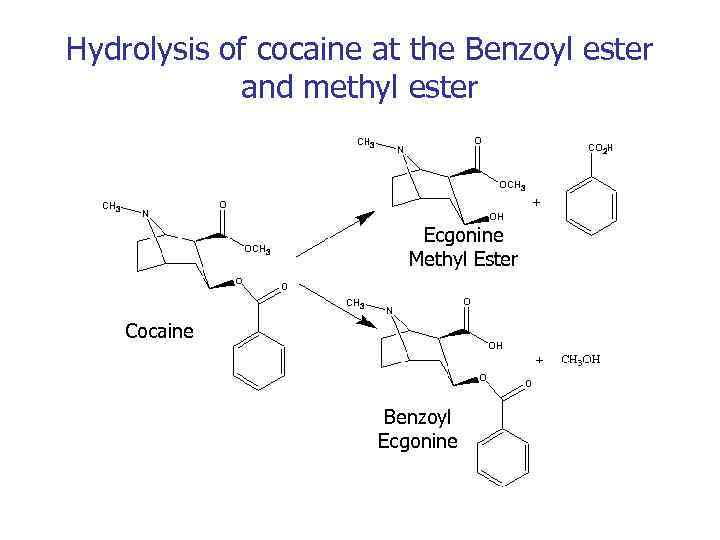 Hydrolysis of cocaine at the Benzoyl ester and methyl ester Ecgonine Methyl Ester Cocaine Benzoyl Ecgonine
Hydrolysis of cocaine at the Benzoyl ester and methyl ester Ecgonine Methyl Ester Cocaine Benzoyl Ecgonine
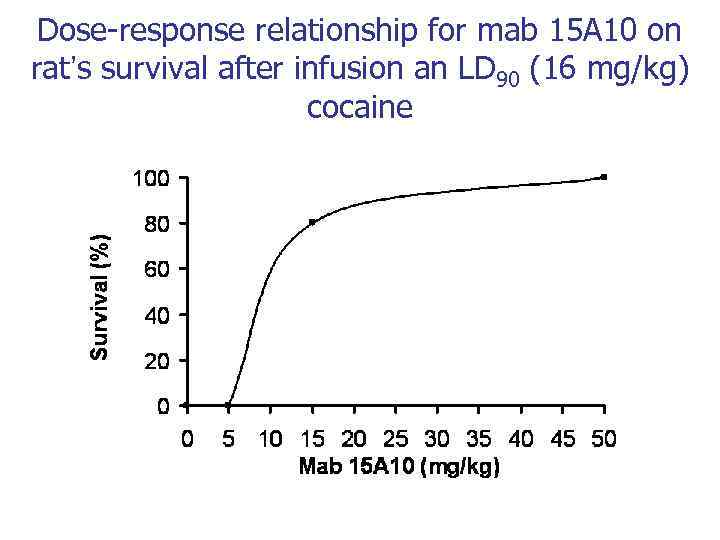 Dose-response relationship for mab 15 A 10 on rat’s survival after infusion an LD 90 (16 mg/kg) cocaine
Dose-response relationship for mab 15 A 10 on rat’s survival after infusion an LD 90 (16 mg/kg) cocaine
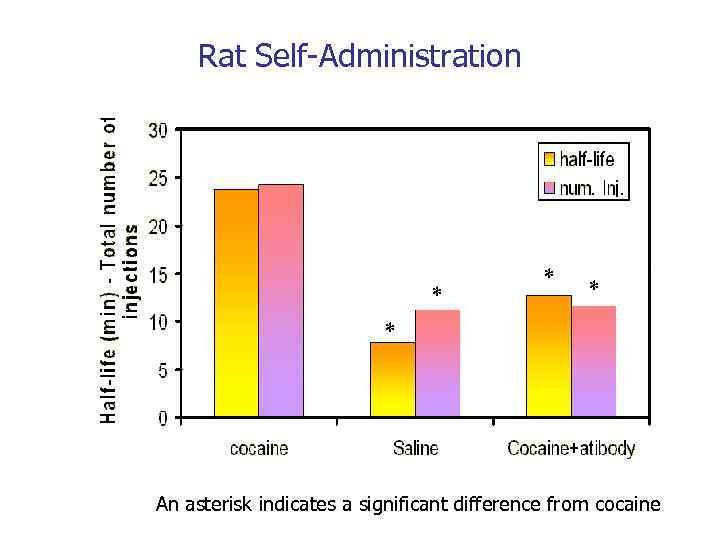 Rat Self-Administration * * An asterisk indicates a significant difference from cocaine
Rat Self-Administration * * An asterisk indicates a significant difference from cocaine
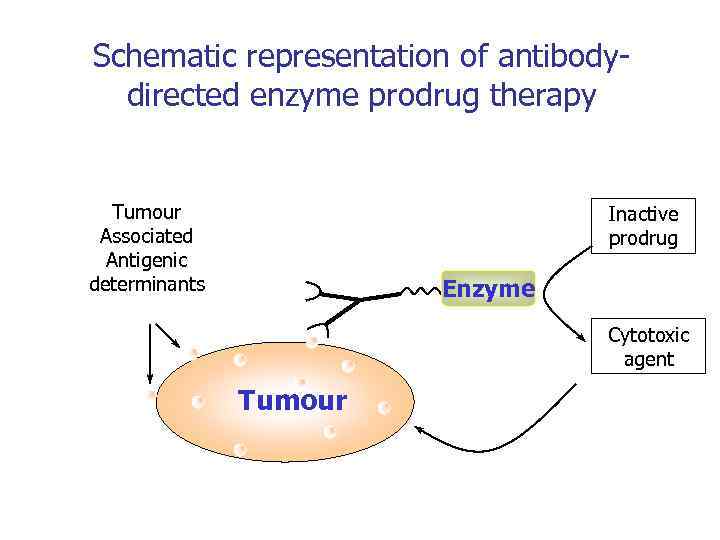 Schematic representation of antibodydirected enzyme prodrug therapy Tumour Associated Antigenic determinants Inactive prodrug Enzyme Cytotoxic agent Tumour
Schematic representation of antibodydirected enzyme prodrug therapy Tumour Associated Antigenic determinants Inactive prodrug Enzyme Cytotoxic agent Tumour
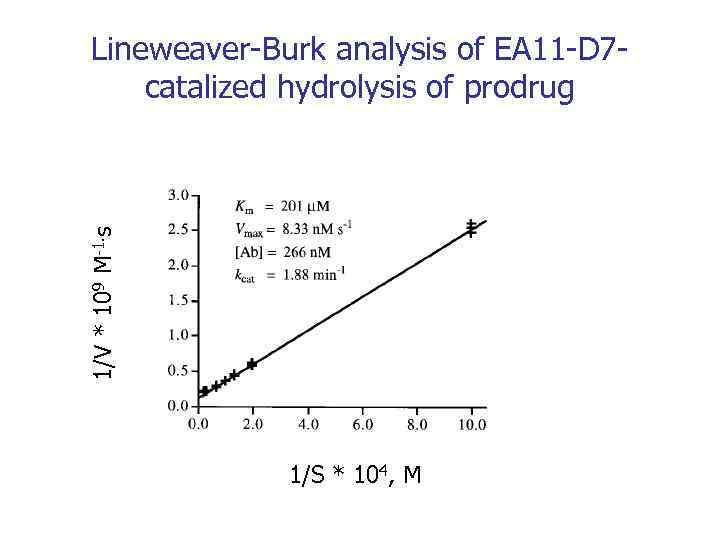 1/V * 109 M-1. s Lineweaver-Burk analysis of EA 11 -D 7 catalized hydrolysis of prodrug 1/S * 104, M
1/V * 109 M-1. s Lineweaver-Burk analysis of EA 11 -D 7 catalized hydrolysis of prodrug 1/S * 104, M
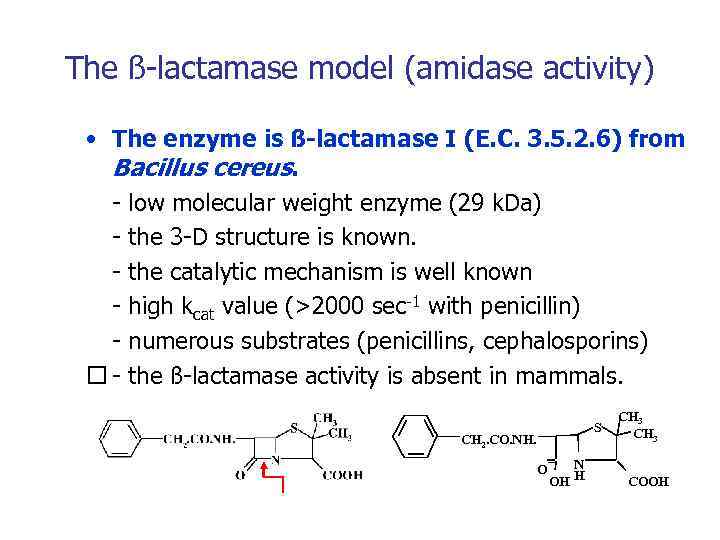 The ß-lactamase model (amidase activity) • The enzyme is ß-lactamase I (E. C. 3. 5. 2. 6) from Bacillus cereus. - low molecular weight enzyme (29 k. Da) - the 3 -D structure is known. - the catalytic mechanism is well known - high kcat value (>2000 sec-1 with penicillin) - numerous substrates (penicillins, cephalosporins) - the ß-lactamase activity is absent in mammals. S CH 2. CO. NH. O N H OH CH 3 COOH
The ß-lactamase model (amidase activity) • The enzyme is ß-lactamase I (E. C. 3. 5. 2. 6) from Bacillus cereus. - low molecular weight enzyme (29 k. Da) - the 3 -D structure is known. - the catalytic mechanism is well known - high kcat value (>2000 sec-1 with penicillin) - numerous substrates (penicillins, cephalosporins) - the ß-lactamase activity is absent in mammals. S CH 2. CO. NH. O N H OH CH 3 COOH
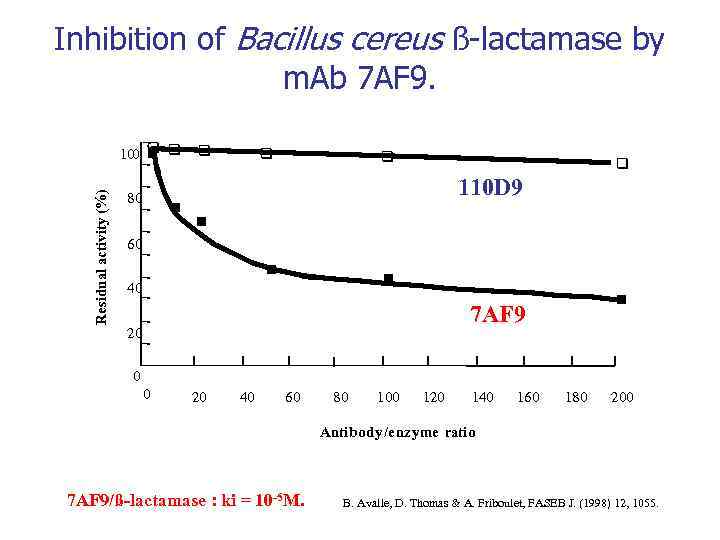 Inhibition of Bacillus cereus ß-lactamase by m. Ab 7 AF 9. Residual activity (%) n 100 q q n 80 n q q 110 D 9 n 60 n n 40 n 7 AF 9 20 0 0 20 40 60 80 100 120 140 160 180 200 Antibody/enzyme ratio 7 AF 9/ß-lactamase : ki = 10 -5 M. B. Avalle, D. Thomas & A. Friboulet, FASEB J. (1998) 12, 1055.
Inhibition of Bacillus cereus ß-lactamase by m. Ab 7 AF 9. Residual activity (%) n 100 q q n 80 n q q 110 D 9 n 60 n n 40 n 7 AF 9 20 0 0 20 40 60 80 100 120 140 160 180 200 Antibody/enzyme ratio 7 AF 9/ß-lactamase : ki = 10 -5 M. B. Avalle, D. Thomas & A. Friboulet, FASEB J. (1998) 12, 1055.
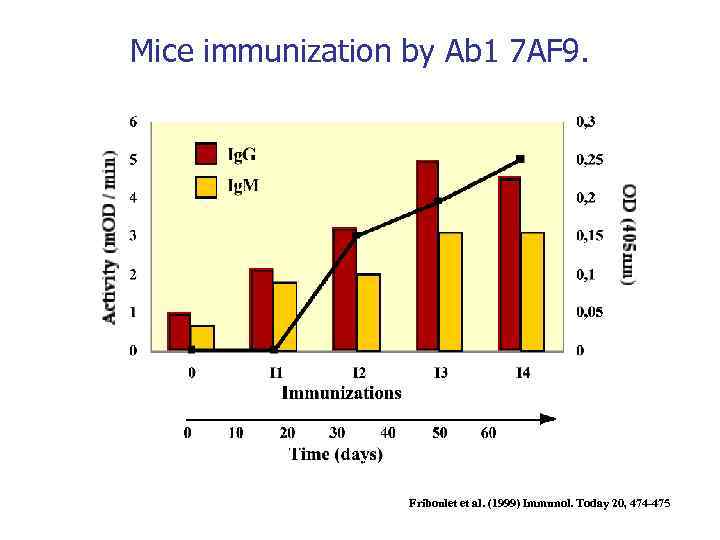 Mice immunization by Ab 1 7 AF 9. Friboulet et al. (1999) Immunol. Today 20, 474 -475
Mice immunization by Ab 1 7 AF 9. Friboulet et al. (1999) Immunol. Today 20, 474 -475
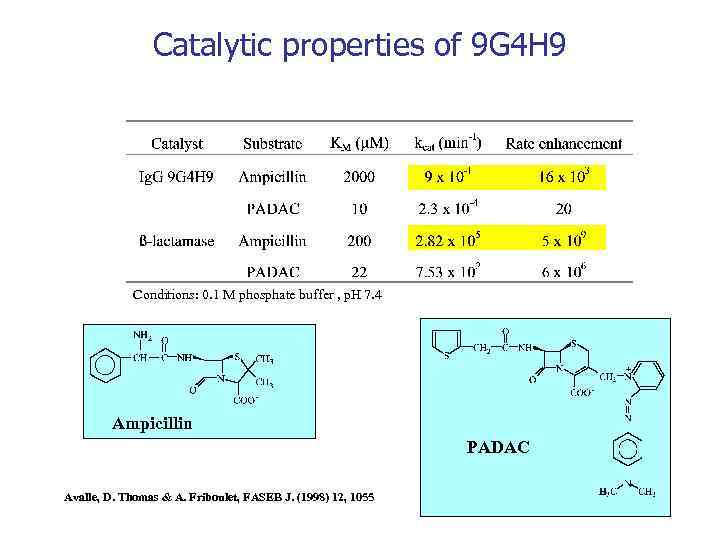 Catalytic properties of 9 G 4 H 9 Conditions: 0. 1 M phosphate buffer , p. H 7. 4 Ampicillin PADAC Avalle, D. Thomas & A. Friboulet, FASEB J. (1998) 12, 1055
Catalytic properties of 9 G 4 H 9 Conditions: 0. 1 M phosphate buffer , p. H 7. 4 Ampicillin PADAC Avalle, D. Thomas & A. Friboulet, FASEB J. (1998) 12, 1055
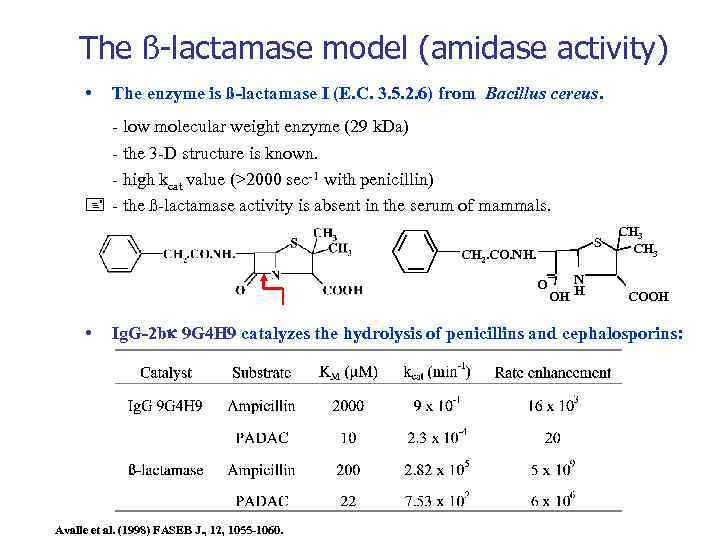 The ß-lactamase model (amidase activity) • The enzyme is ß-lactamase I (E. C. 3. 5. 2. 6) from Bacillus cereus. - low molecular weight enzyme (29 k. Da) - the 3 -D structure is known. - high kcat value (>2000 sec-1 with penicillin) - the ß-lactamase activity is absent in the serum of mammals. S CH 2. CO. NH. O • N H OH CH 3 COOH Ig. G-2 b 9 G 4 H 9 catalyzes the hydrolysis of penicillins and cephalosporins: Avalle et al. (1998) FASEB J. , 12, 1055 -1060.
The ß-lactamase model (amidase activity) • The enzyme is ß-lactamase I (E. C. 3. 5. 2. 6) from Bacillus cereus. - low molecular weight enzyme (29 k. Da) - the 3 -D structure is known. - high kcat value (>2000 sec-1 with penicillin) - the ß-lactamase activity is absent in the serum of mammals. S CH 2. CO. NH. O • N H OH CH 3 COOH Ig. G-2 b 9 G 4 H 9 catalyzes the hydrolysis of penicillins and cephalosporins: Avalle et al. (1998) FASEB J. , 12, 1055 -1060.
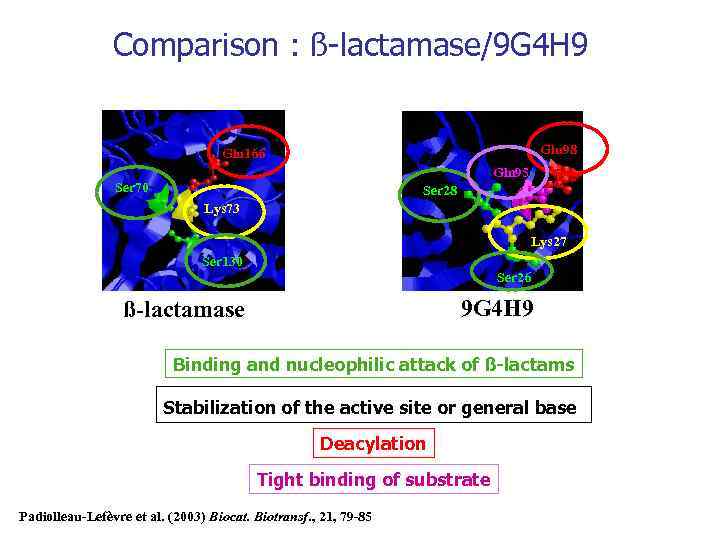 Comparison : ß-lactamase/9 G 4 H 9 Glu 98 Glu 166 Gln 95 Ser 70 Ser 28 Lys 73 Lys 27 Ser 130 Ser 26 9 G 4 H 9 ß-lactamase Binding and nucleophilic attack of ß-lactams Stabilization of the active site or general base Deacylation Tight binding of substrate Padiolleau-Lefèvre et al. (2003) Biocat. Biotransf. , 21, 79 -85
Comparison : ß-lactamase/9 G 4 H 9 Glu 98 Glu 166 Gln 95 Ser 70 Ser 28 Lys 73 Lys 27 Ser 130 Ser 26 9 G 4 H 9 ß-lactamase Binding and nucleophilic attack of ß-lactams Stabilization of the active site or general base Deacylation Tight binding of substrate Padiolleau-Lefèvre et al. (2003) Biocat. Biotransf. , 21, 79 -85
 Exo-amylolytic And A-glucosidase Activities Of Human Abzymes Exo-amylase action: a-Glucosidase action:
Exo-amylolytic And A-glucosidase Activities Of Human Abzymes Exo-amylase action: a-Glucosidase action:
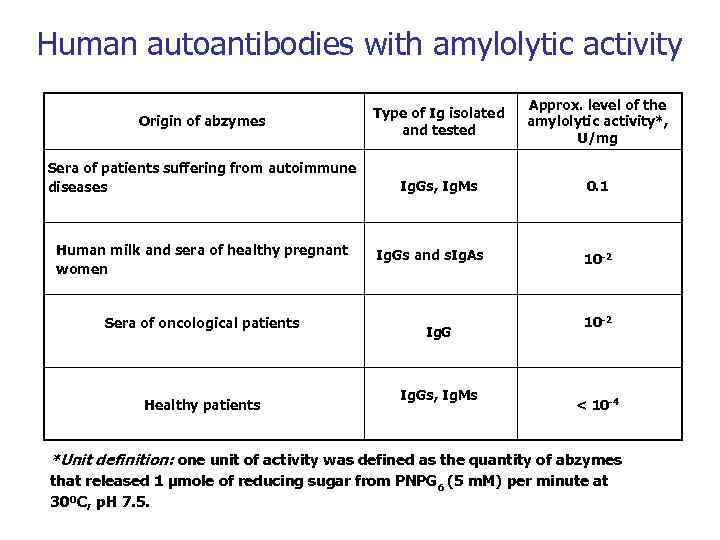 Human autoantibodies with amylolytic activity Origin of abzymes Sera of patients suffering from autoimmune diseases Human milk and sera of healthy pregnant women Sera of oncological patients Healthy patients Type of Ig isolated and tested Approx. level of the amylolytic activity*, U/mg Ig. Gs, Ig. Ms 0. 1 Ig. Gs and s. Ig. As Ig. Gs, Ig. Ms 10 -2 < 10 -4 *Unit definition: one unit of activity was defined as the quantity of abzymes that released 1 μmole of reducing sugar from PNPG 6 (5 m. M) per minute at 30ºC, p. H 7. 5.
Human autoantibodies with amylolytic activity Origin of abzymes Sera of patients suffering from autoimmune diseases Human milk and sera of healthy pregnant women Sera of oncological patients Healthy patients Type of Ig isolated and tested Approx. level of the amylolytic activity*, U/mg Ig. Gs, Ig. Ms 0. 1 Ig. Gs and s. Ig. As Ig. Gs, Ig. Ms 10 -2 < 10 -4 *Unit definition: one unit of activity was defined as the quantity of abzymes that released 1 μmole of reducing sugar from PNPG 6 (5 m. M) per minute at 30ºC, p. H 7. 5.
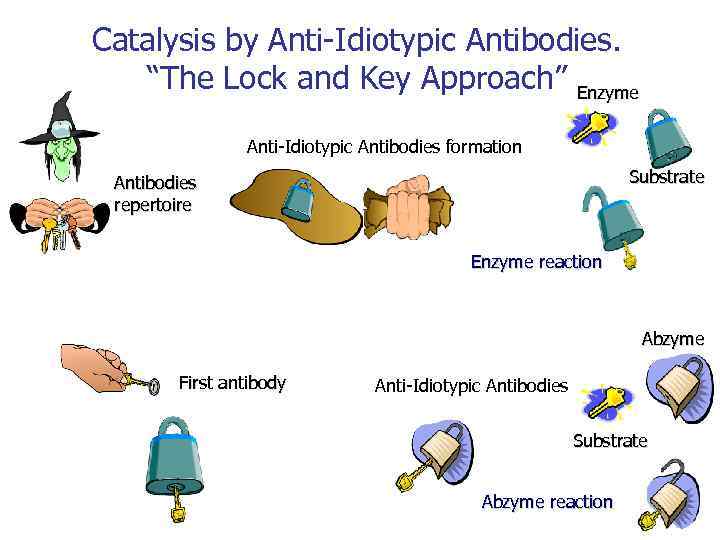 Catalysis by Anti-Idiotypic Antibodies. “The Lock and Key Approach” Enzyme Anti-Idiotypic Antibodies formation Substrate Antibodies repertoire Enzyme reaction Abzyme First antibody Anti-Idiotypic Antibodies Substrate Abzyme reaction
Catalysis by Anti-Idiotypic Antibodies. “The Lock and Key Approach” Enzyme Anti-Idiotypic Antibodies formation Substrate Antibodies repertoire Enzyme reaction Abzyme First antibody Anti-Idiotypic Antibodies Substrate Abzyme reaction
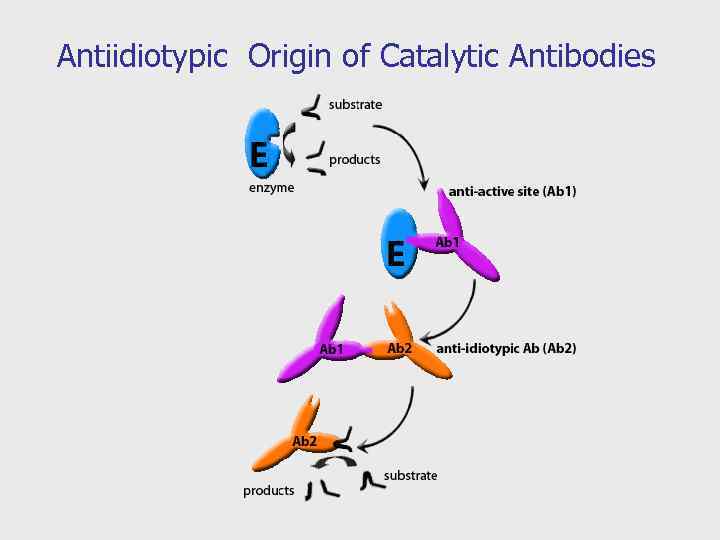 Antiidiotypic Origin of Catalytic Antibodies
Antiidiotypic Origin of Catalytic Antibodies
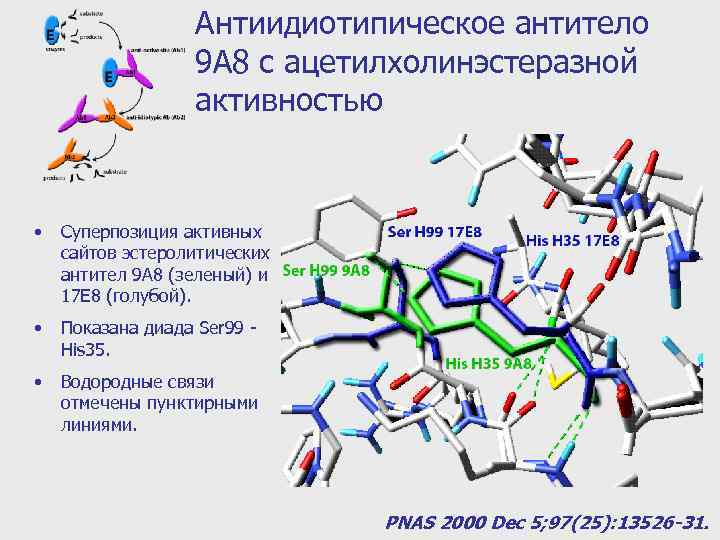 Антиидиотипическое антитело 9 A 8 с ацетилхолинэстеразной активностью • Суперпозиция активных сайтов эстеролитических антител 9 A 8 (зеленый) и 17 E 8 (голубой). • Показана диада Ser 99 His 35. • Водородные связи отмечены пунктирными линиями. PNAS 2000 Dec 5; 97(25): 13526 -31.
Антиидиотипическое антитело 9 A 8 с ацетилхолинэстеразной активностью • Суперпозиция активных сайтов эстеролитических антител 9 A 8 (зеленый) и 17 E 8 (голубой). • Показана диада Ser 99 His 35. • Водородные связи отмечены пунктирными линиями. PNAS 2000 Dec 5; 97(25): 13526 -31.
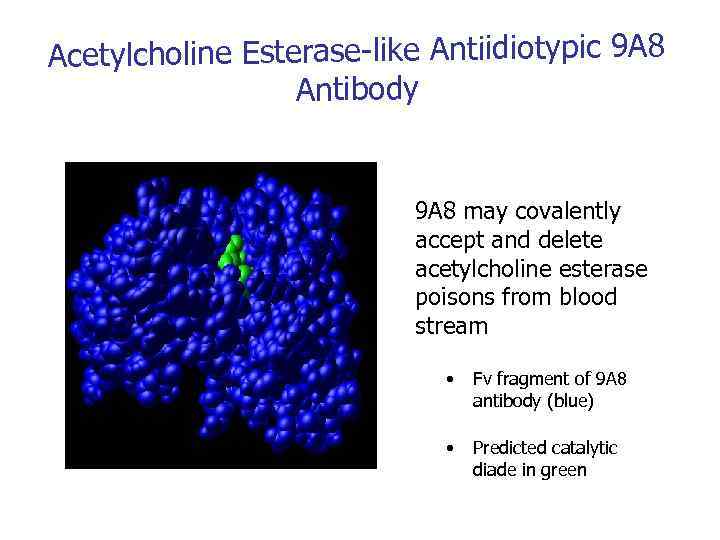 Acetylcholine Esterase-like Antiidiotypic 9 A 8 Antibody 9 A 8 may covalently accept and delete acetylcholine esterase poisons from blood stream • Fv fragment of 9 А 8 antibody (blue) • Predicted catalytic diade in green
Acetylcholine Esterase-like Antiidiotypic 9 A 8 Antibody 9 A 8 may covalently accept and delete acetylcholine esterase poisons from blood stream • Fv fragment of 9 А 8 antibody (blue) • Predicted catalytic diade in green
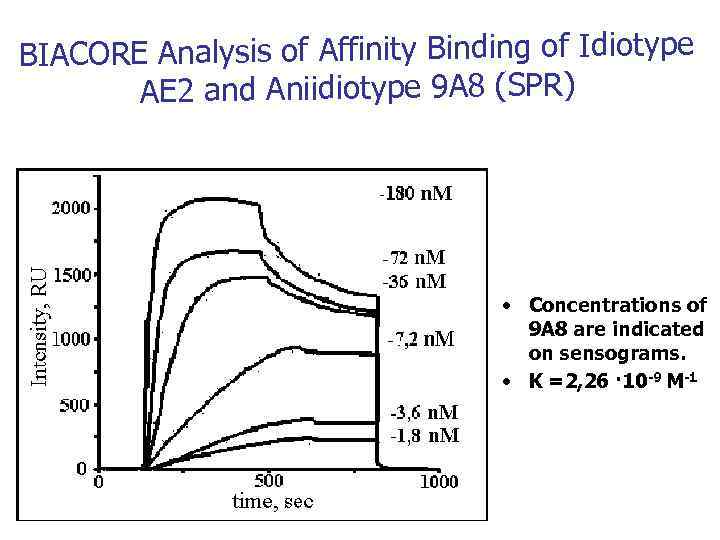 BIACORE Analysis of Affinity Binding of Idiotype AE 2 and Aniidiotype 9 A 8 (SPR) • Concentrations of 9 A 8 are indicated on sensograms. • K =2, 26 · 10 -9 М-1
BIACORE Analysis of Affinity Binding of Idiotype AE 2 and Aniidiotype 9 A 8 (SPR) • Concentrations of 9 A 8 are indicated on sensograms. • K =2, 26 · 10 -9 М-1
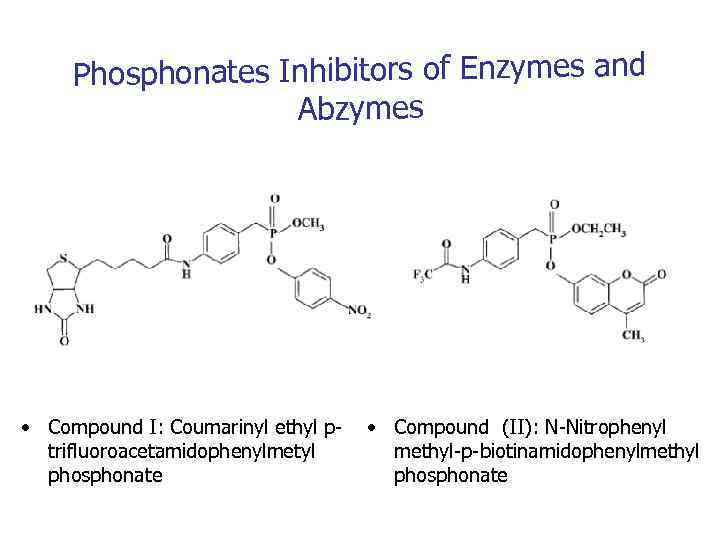 Phosphonates Inhibitors of Enzymes and Abzymes • Compound I: Coumarinyl ethyl ptrifluoroacetamidophenylmetyl phosphonate • Compound (II): N-Nitrophenyl methyl-p-biotinamidophenylmethyl phosphonate
Phosphonates Inhibitors of Enzymes and Abzymes • Compound I: Coumarinyl ethyl ptrifluoroacetamidophenylmetyl phosphonate • Compound (II): N-Nitrophenyl methyl-p-biotinamidophenylmethyl phosphonate
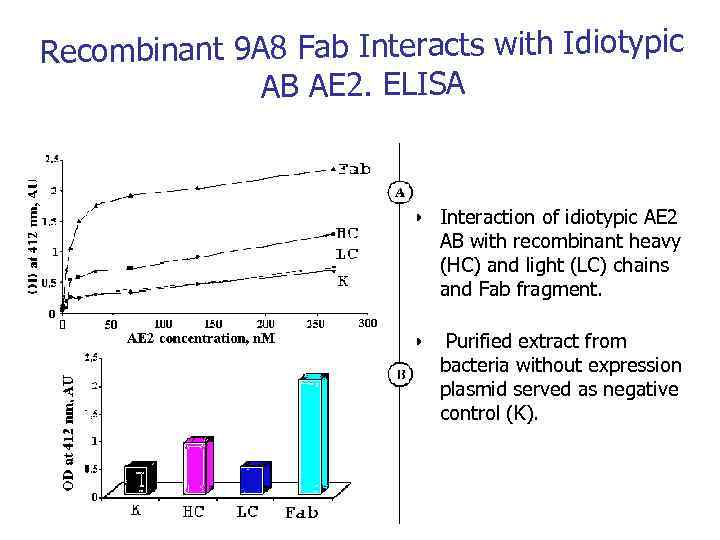 Recombinant 9 A 8 Fab Interacts with Idiotypic AB AE 2. ELISA • Interaction of idiotypic AE 2 AB with recombinant heavy (HC) and light (LC) chains and Fab fragment. • Purified extract from bacteria without expression plasmid served as negative control (K).
Recombinant 9 A 8 Fab Interacts with Idiotypic AB AE 2. ELISA • Interaction of idiotypic AE 2 AB with recombinant heavy (HC) and light (LC) chains and Fab fragment. • Purified extract from bacteria without expression plasmid served as negative control (K).
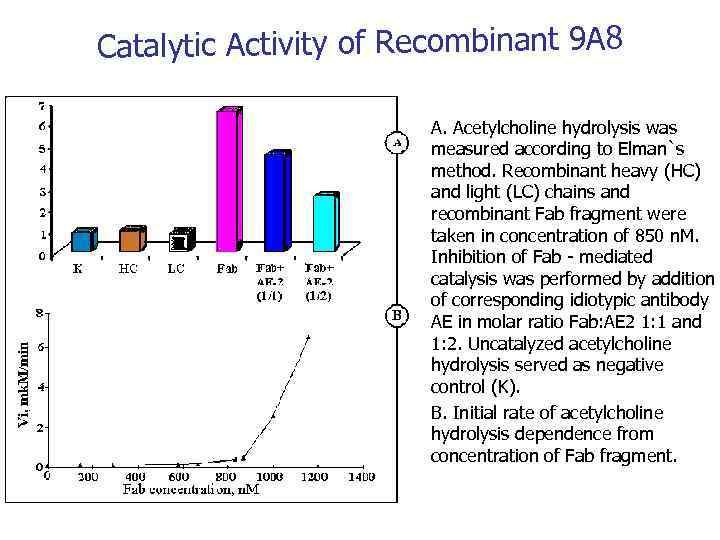 Catalytic Activity of Recombinant 9 А 8 • • A. Acetylcholine hydrolysis was measured according to Elman`s method. Recombinant heavy (HC) and light (LC) chains and recombinant Fab fragment were taken in concentration of 850 n. M. Inhibition of Fab - mediated catalysis was performed by addition of corresponding idiotypic antibody AE in molar ratio Fab: AE 2 1: 1 and 1: 2. Uncatalyzed acetylcholine hydrolysis served as negative control (K). B. Initial rate of acetylcholine hydrolysis dependence from concentration of Fab fragment.
Catalytic Activity of Recombinant 9 А 8 • • A. Acetylcholine hydrolysis was measured according to Elman`s method. Recombinant heavy (HC) and light (LC) chains and recombinant Fab fragment were taken in concentration of 850 n. M. Inhibition of Fab - mediated catalysis was performed by addition of corresponding idiotypic antibody AE in molar ratio Fab: AE 2 1: 1 and 1: 2. Uncatalyzed acetylcholine hydrolysis served as negative control (K). B. Initial rate of acetylcholine hydrolysis dependence from concentration of Fab fragment.
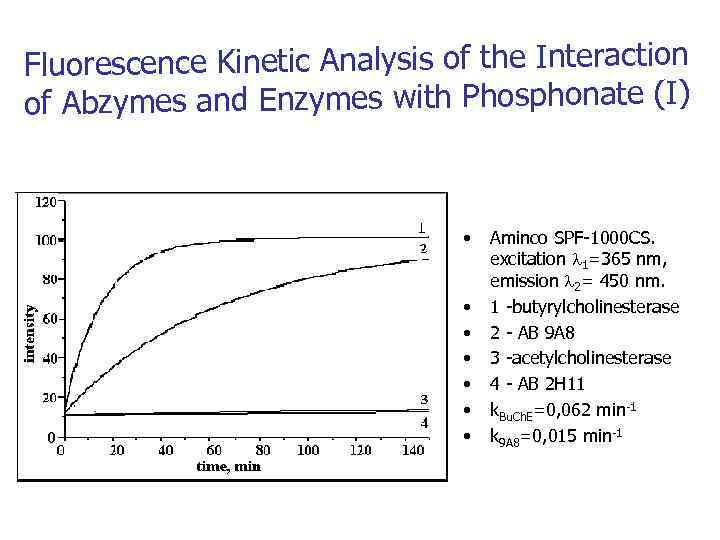 Fluorescence Kinetic Analysis of the Interaction of Abzymes and Enzymes with Phosphonate (I) • • Aminco SPF-1000 CS. excitation 1=365 nm, emission 2= 450 nm. 1 -butyrylcholinesterase 2 - AB 9 А 8 3 -acetylcholinesterase 4 - AB 2 Н 11 k. Bu. Ch. E=0, 062 min-1 k 9 A 8=0, 015 min-1
Fluorescence Kinetic Analysis of the Interaction of Abzymes and Enzymes with Phosphonate (I) • • Aminco SPF-1000 CS. excitation 1=365 nm, emission 2= 450 nm. 1 -butyrylcholinesterase 2 - AB 9 А 8 3 -acetylcholinesterase 4 - AB 2 Н 11 k. Bu. Ch. E=0, 062 min-1 k 9 A 8=0, 015 min-1
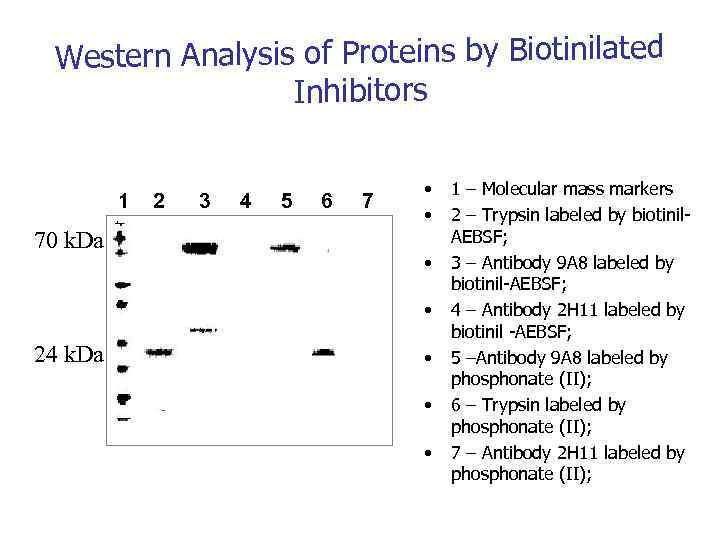 Western Analysis of Proteins by Biotinilated Inhibitors 1 70 k. Da 2 3 4 5 6 7 • • 24 k. Da • • • 1 – Molecular mass markers 2 – Trypsin labeled by biotinil. AEBSF; 3 – Antibody 9 А 8 labeled by biotinil-AEBSF; 4 – Antibody 2 H 11 labeled by biotinil -AEBSF; 5 –Antibody 9 А 8 labeled by phosphonate (II); 6 – Trypsin labeled by phosphonate (II); 7 – Antibody 2 H 11 labeled by phosphonate (II);
Western Analysis of Proteins by Biotinilated Inhibitors 1 70 k. Da 2 3 4 5 6 7 • • 24 k. Da • • • 1 – Molecular mass markers 2 – Trypsin labeled by biotinil. AEBSF; 3 – Antibody 9 А 8 labeled by biotinil-AEBSF; 4 – Antibody 2 H 11 labeled by biotinil -AEBSF; 5 –Antibody 9 А 8 labeled by phosphonate (II); 6 – Trypsin labeled by phosphonate (II); 7 – Antibody 2 H 11 labeled by phosphonate (II);
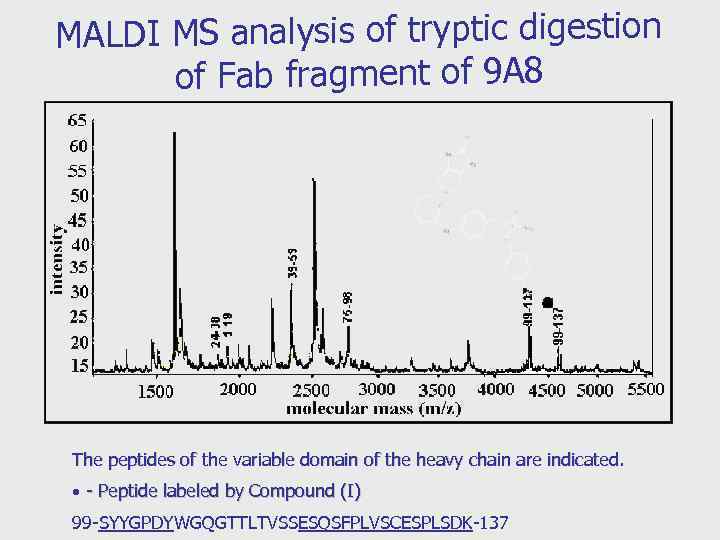 MALDI MS analysis of tryptic digestion of Fab fragment of 9 A 8 The peptides of the variable domain of the heavy chain are indicated. - Peptide labeled by Compound (I) 99 -SYYGPDYWGQGTTLTVSSESQSFPLVSCESPLSDK-137
MALDI MS analysis of tryptic digestion of Fab fragment of 9 A 8 The peptides of the variable domain of the heavy chain are indicated. - Peptide labeled by Compound (I) 99 -SYYGPDYWGQGTTLTVSSESQSFPLVSCESPLSDK-137
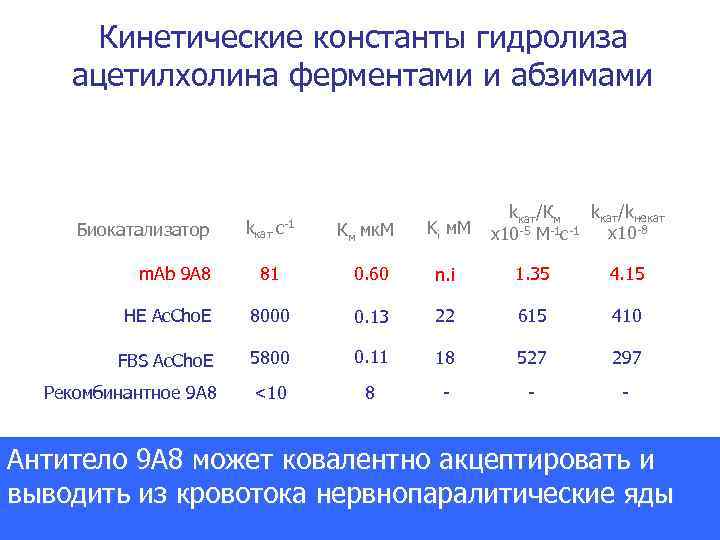 Кинетические константы гидролиза ацетилхолина ферментами и абзимами Биокатализатор kкат с-1 Км мк. М Ki м. М kкат/kнекат kкат/Км x 10 -8 х10 -5 М-1 с-1 81 0. 60 n. i 1. 35 4. 15 HE Ac. Cho. E 8000 0. 13 22 615 410 FBS Ac. Cho. E 5800 0. 11 18 527 297 Рекомбинантное 9 А 8 <10 8 - - - m. Ab 9 A 8 Антитело 9 A 8 может ковалентно акцептировать и выводить из кровотока нервнопаралитические яды PNAS 2000 Dec 5; 97(25): 13526 -31.
Кинетические константы гидролиза ацетилхолина ферментами и абзимами Биокатализатор kкат с-1 Км мк. М Ki м. М kкат/kнекат kкат/Км x 10 -8 х10 -5 М-1 с-1 81 0. 60 n. i 1. 35 4. 15 HE Ac. Cho. E 8000 0. 13 22 615 410 FBS Ac. Cho. E 5800 0. 11 18 527 297 Рекомбинантное 9 А 8 <10 8 - - - m. Ab 9 A 8 Антитело 9 A 8 может ковалентно акцептировать и выводить из кровотока нервнопаралитические яды PNAS 2000 Dec 5; 97(25): 13526 -31.
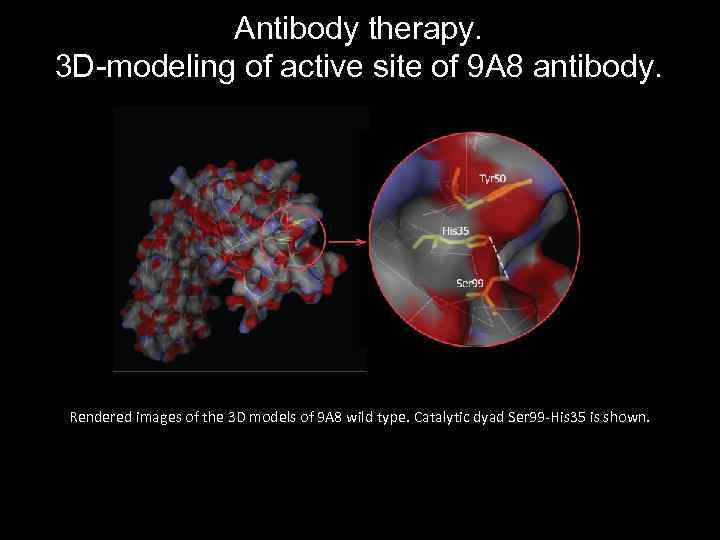 Antibody therapy. 3 D-modeling of active site of 9 A 8 antibody. Rendered images of the 3 D models of 9 A 8 wild type. Catalytic dyad Ser 99 -His 35 is shown.
Antibody therapy. 3 D-modeling of active site of 9 A 8 antibody. Rendered images of the 3 D models of 9 A 8 wild type. Catalytic dyad Ser 99 -His 35 is shown.
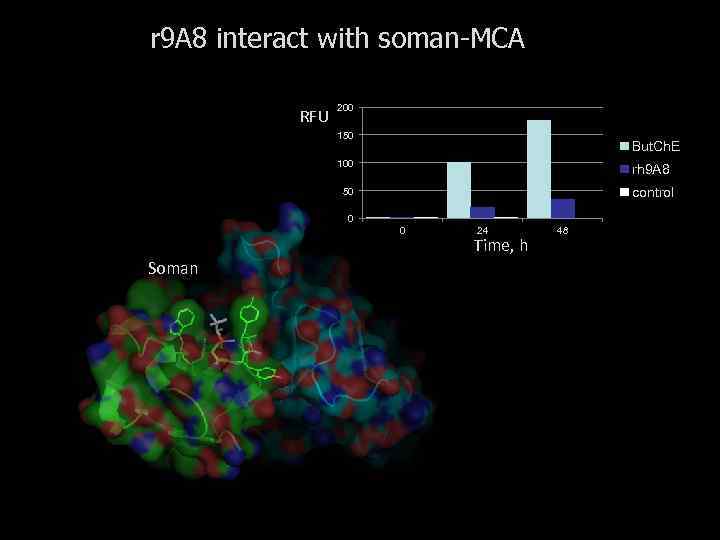 r 9 A 8 interact with soman-MCA RFU 200 150 But. Ch. E 100 rh 9 A 8 50 control 0 0 Soman 24 Time, h 48
r 9 A 8 interact with soman-MCA RFU 200 150 But. Ch. E 100 rh 9 A 8 50 control 0 0 Soman 24 Time, h 48
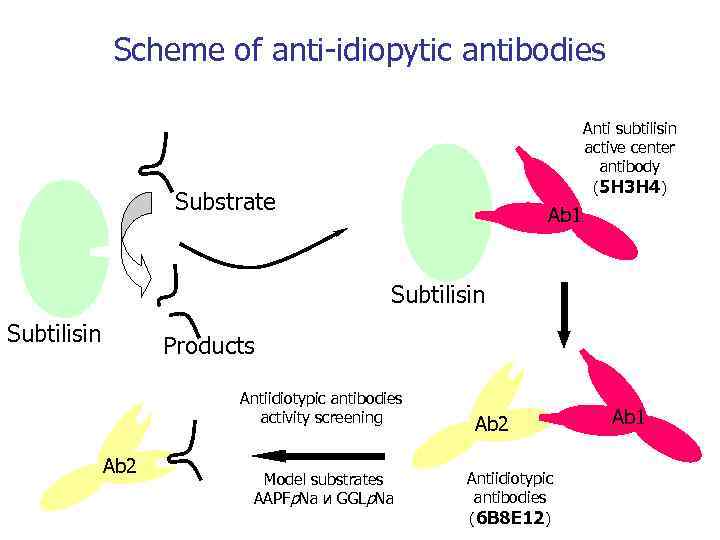 Scheme of anti-idiopytic antibodies Anti subtilisin active center antibody (5 H 3 H 4) Substrate Ab 1 Subtilisin Products Antiidiotypic antibodies activity screening Ab 2 Model substrates AAPFp. Na и GGLp. Na Ab 2 Antiidiotypic antibodies (6 B 8 E 12) Ab 1
Scheme of anti-idiopytic antibodies Anti subtilisin active center antibody (5 H 3 H 4) Substrate Ab 1 Subtilisin Products Antiidiotypic antibodies activity screening Ab 2 Model substrates AAPFp. Na и GGLp. Na Ab 2 Antiidiotypic antibodies (6 B 8 E 12) Ab 1
 Proteolytic activity of 6 B 8 -E 12 anti-idiotypic m. Ab The anti-idiotypic antibody 6 B 8 -E 12 (100 and 200 ng, lanes 2 and 3, respectively) was separated by 10% SDS-PAGE with FBSA impregnated in the gel during polymerization (see Materials and Methods for details). After electrophoresis, proteins were in-gel renatured by Triton X-100 washes. Proteolytic degradation was visualized by the increase in fluorescence intensity, seen as bright bands at the dark background. Mouse anti-c-myc antibody 9 E 10 was used as the negative control (lane 1). Bovine trypsin (100 pg and 1 ng, lanes 4 and 5, respectively) was used as the positive control.
Proteolytic activity of 6 B 8 -E 12 anti-idiotypic m. Ab The anti-idiotypic antibody 6 B 8 -E 12 (100 and 200 ng, lanes 2 and 3, respectively) was separated by 10% SDS-PAGE with FBSA impregnated in the gel during polymerization (see Materials and Methods for details). After electrophoresis, proteins were in-gel renatured by Triton X-100 washes. Proteolytic degradation was visualized by the increase in fluorescence intensity, seen as bright bands at the dark background. Mouse anti-c-myc antibody 9 E 10 was used as the negative control (lane 1). Bovine trypsin (100 pg and 1 ng, lanes 4 and 5, respectively) was used as the positive control.
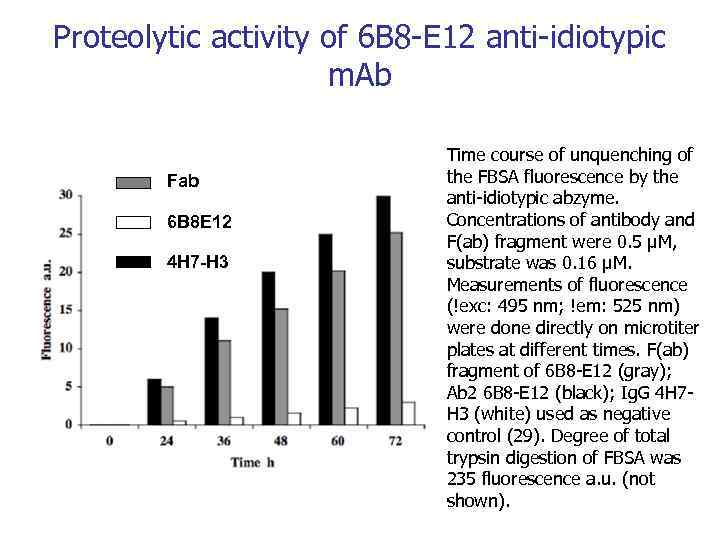 Proteolytic activity of 6 B 8 -E 12 anti-idiotypic m. Ab Fab 6 B 8 E 12 4 H 7 -H 3 Time course of unquenching of the FBSA fluorescence by the anti-idiotypic abzyme. Concentrations of antibody and F(ab) fragment were 0. 5 μM, substrate was 0. 16 μM. Measurements of fluorescence (!exc: 495 nm; !em: 525 nm) were done directly on microtiter plates at different times. F(ab) fragment of 6 B 8 -E 12 (gray); Ab 2 6 B 8 -E 12 (black); Ig. G 4 H 7 H 3 (white) used as negative control (29). Degree of total trypsin digestion of FBSA was 235 fluorescence a. u. (not shown).
Proteolytic activity of 6 B 8 -E 12 anti-idiotypic m. Ab Fab 6 B 8 E 12 4 H 7 -H 3 Time course of unquenching of the FBSA fluorescence by the anti-idiotypic abzyme. Concentrations of antibody and F(ab) fragment were 0. 5 μM, substrate was 0. 16 μM. Measurements of fluorescence (!exc: 495 nm; !em: 525 nm) were done directly on microtiter plates at different times. F(ab) fragment of 6 B 8 -E 12 (gray); Ab 2 6 B 8 -E 12 (black); Ig. G 4 H 7 H 3 (white) used as negative control (29). Degree of total trypsin digestion of FBSA was 235 fluorescence a. u. (not shown).
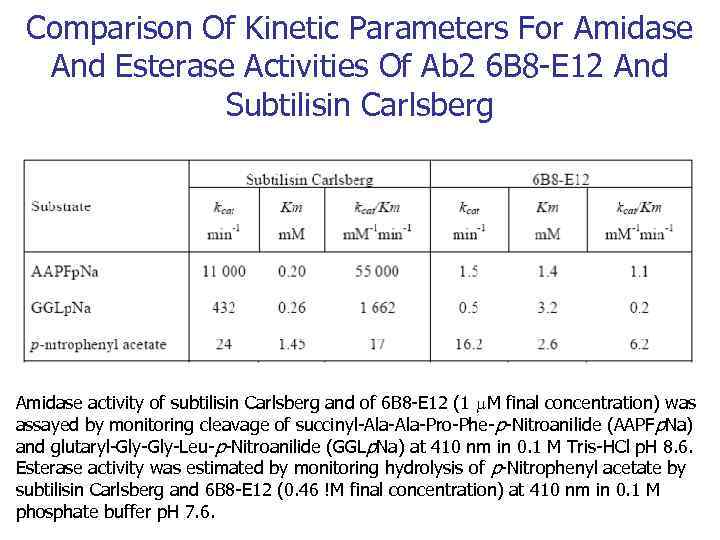 Comparison Of Kinetic Parameters For Amidase And Esterase Activities Of Ab 2 6 B 8 -E 12 And Subtilisin Carlsberg Amidase activity of subtilisin Carlsberg and of 6 B 8 -E 12 (1 M final concentration) was assayed by monitoring cleavage of succinyl-Ala-Pro-Phe- p-Nitroanilide (AAPFp. Na) and glutaryl-Gly-Leu-p-Nitroanilide (GGLp. Na) at 410 nm in 0. 1 M Tris-HCl p. H 8. 6. Esterase activity was estimated by monitoring hydrolysis of p-Nitrophenyl acetate by subtilisin Carlsberg and 6 B 8 -E 12 (0. 46 !M final concentration) at 410 nm in 0. 1 M phosphate buffer p. H 7. 6.
Comparison Of Kinetic Parameters For Amidase And Esterase Activities Of Ab 2 6 B 8 -E 12 And Subtilisin Carlsberg Amidase activity of subtilisin Carlsberg and of 6 B 8 -E 12 (1 M final concentration) was assayed by monitoring cleavage of succinyl-Ala-Pro-Phe- p-Nitroanilide (AAPFp. Na) and glutaryl-Gly-Leu-p-Nitroanilide (GGLp. Na) at 410 nm in 0. 1 M Tris-HCl p. H 8. 6. Esterase activity was estimated by monitoring hydrolysis of p-Nitrophenyl acetate by subtilisin Carlsberg and 6 B 8 -E 12 (0. 46 !M final concentration) at 410 nm in 0. 1 M phosphate buffer p. H 7. 6.
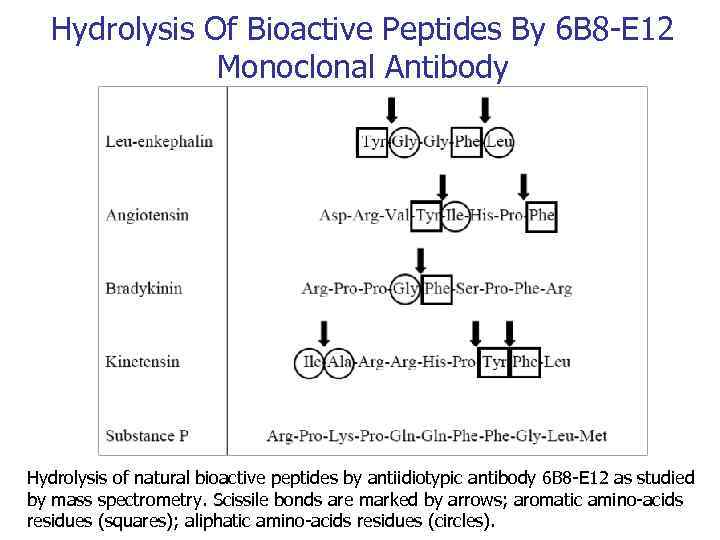 Hydrolysis Of Bioactive Peptides By 6 B 8 -E 12 Monoclonal Antibody Hydrolysis of natural bioactive peptides by antiidiotypic antibody 6 B 8 -E 12 as studied by mass spectrometry. Scissile bonds are marked by arrows; aromatic amino-acids residues (squares); aliphatic amino-acids residues (circles).
Hydrolysis Of Bioactive Peptides By 6 B 8 -E 12 Monoclonal Antibody Hydrolysis of natural bioactive peptides by antiidiotypic antibody 6 B 8 -E 12 as studied by mass spectrometry. Scissile bonds are marked by arrows; aromatic amino-acids residues (squares); aliphatic amino-acids residues (circles).
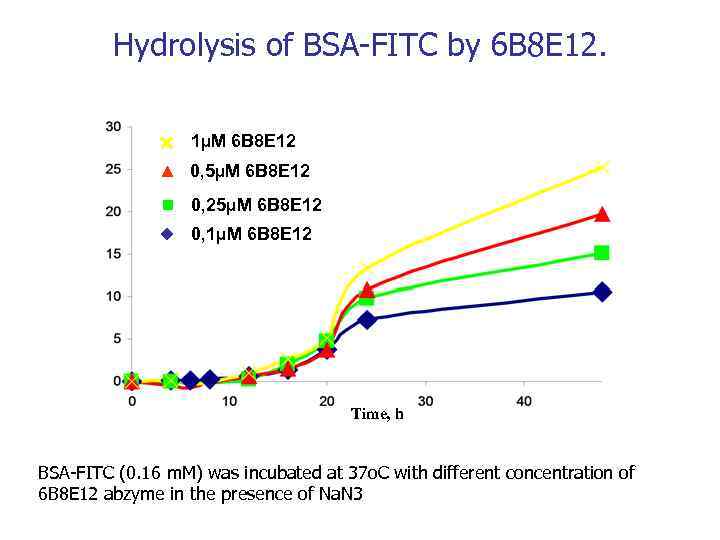 Hydrolysis of BSA-FITC by 6 B 8 E 12. 1µM 6 B 8 E 12 0, 5µM 6 B 8 E 12 0, 25µM 6 B 8 E 12 0, 1µM 6 B 8 E 12 Time, h BSA-FITC (0. 16 m. M) was incubated at 37 o. C with different concentration of 6 B 8 E 12 abzyme in the presence of Na. N 3
Hydrolysis of BSA-FITC by 6 B 8 E 12. 1µM 6 B 8 E 12 0, 5µM 6 B 8 E 12 0, 25µM 6 B 8 E 12 0, 1µM 6 B 8 E 12 Time, h BSA-FITC (0. 16 m. M) was incubated at 37 o. C with different concentration of 6 B 8 E 12 abzyme in the presence of Na. N 3
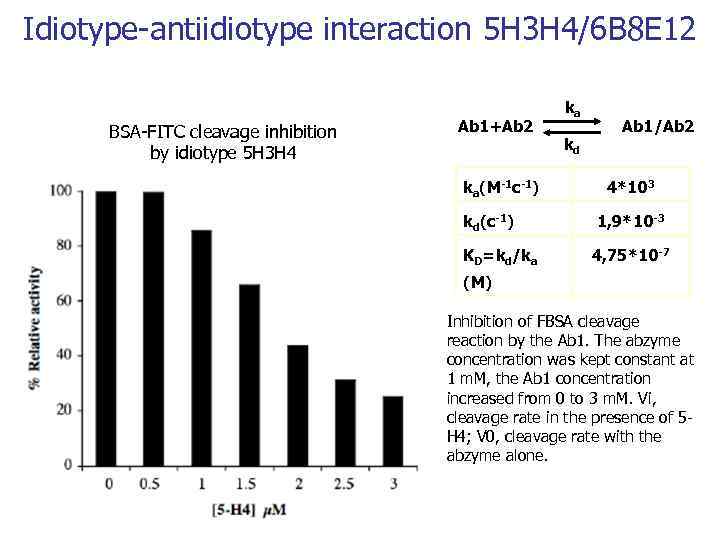 Idiotype-antiidiotype interaction 5 H 3 H 4/6 B 8 E 12 BSA-FITC cleavage inhibition by idiotype 5 H 3 H 4 Ab 1+Ab 2 ka(M-1 c-1) kd(с-1) KD=kd/ka ka kd Ab 1/Ab 2 4*103 1, 9*10 -3 4, 75*10 -7 (M) Inhibition of FBSA cleavage reaction by the Ab 1. The abzyme concentration was kept constant at 1 m. M, the Ab 1 concentration increased from 0 to 3 m. M. Vi, cleavage rate in the presence of 5 H 4; V 0, cleavage rate with the abzyme alone.
Idiotype-antiidiotype interaction 5 H 3 H 4/6 B 8 E 12 BSA-FITC cleavage inhibition by idiotype 5 H 3 H 4 Ab 1+Ab 2 ka(M-1 c-1) kd(с-1) KD=kd/ka ka kd Ab 1/Ab 2 4*103 1, 9*10 -3 4, 75*10 -7 (M) Inhibition of FBSA cleavage reaction by the Ab 1. The abzyme concentration was kept constant at 1 m. M, the Ab 1 concentration increased from 0 to 3 m. M. Vi, cleavage rate in the presence of 5 H 4; V 0, cleavage rate with the abzyme alone.
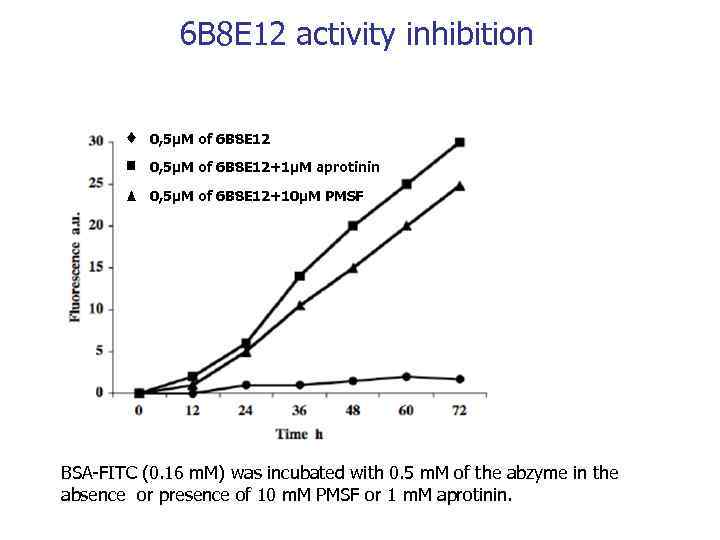 6 B 8 E 12 activity inhibition 0, 5µM of 6 B 8 E 12+1µM aprotinin 0, 5µM of 6 B 8 E 12+10µM PMSF BSA-FITC (0. 16 m. M) was incubated with 0. 5 m. M of the abzyme in the absence or presence of 10 m. M PMSF or 1 m. M aprotinin.
6 B 8 E 12 activity inhibition 0, 5µM of 6 B 8 E 12+1µM aprotinin 0, 5µM of 6 B 8 E 12+10µM PMSF BSA-FITC (0. 16 m. M) was incubated with 0. 5 m. M of the abzyme in the absence or presence of 10 m. M PMSF or 1 m. M aprotinin.
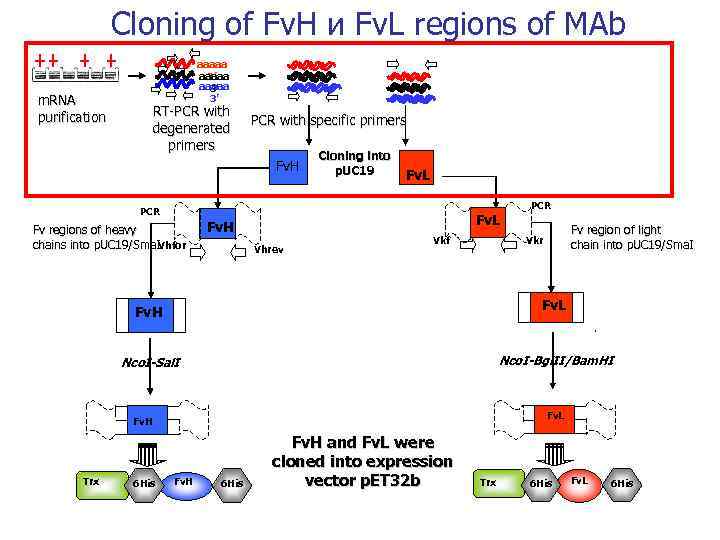 Cloning of Fv. Н и Fv. L regions of MAb m. RNA purification aaaaa 3’ 3’ RT-PCR with degenerated primers PCR with specific primers Fv. H Cloning into p. UC 19 Fv. L PCR Fv regions of heavy chains into p. UC 19/Sma. IVhfor Fv. L Fv. H Vhrev Vkf Fv. L Nco. I-Bgl. II/Bam. HI Nco. I-Sal. I Fv. L Fv. H 6 His Fv region of light chain into p. UC 19/Sma. I Vkr Fv. H Trx PCR Fv. H 6 His Fv. H and Fv. L were cloned into expression vector p. ET 32 b Trx 6 His Fv. L 6 His
Cloning of Fv. Н и Fv. L regions of MAb m. RNA purification aaaaa 3’ 3’ RT-PCR with degenerated primers PCR with specific primers Fv. H Cloning into p. UC 19 Fv. L PCR Fv regions of heavy chains into p. UC 19/Sma. IVhfor Fv. L Fv. H Vhrev Vkf Fv. L Nco. I-Bgl. II/Bam. HI Nco. I-Sal. I Fv. L Fv. H 6 His Fv region of light chain into p. UC 19/Sma. I Vkr Fv. H Trx PCR Fv. H 6 His Fv. H and Fv. L were cloned into expression vector p. ET 32 b Trx 6 His Fv. L 6 His
 Catalytic activity of recombinant Sc. Fv 6 B 8 E 12 Biocatalysts Substrate kcat, s-1 Km, M kcat/Km, Sc. Fv 6 B 8 E 12 Met-MCA 1, 50*10 -2 6, 53*10 -5 230 Sc. Fv 6 B 8 E 12 Phe-MCA 7, 56*10 -3 8, 60*10 -6 879 Sc. Fv 6 B 8 E 12 Leu-MCA 8, 70*10 -3 5, 00*10 -5 174 M-1 s-1
Catalytic activity of recombinant Sc. Fv 6 B 8 E 12 Biocatalysts Substrate kcat, s-1 Km, M kcat/Km, Sc. Fv 6 B 8 E 12 Met-MCA 1, 50*10 -2 6, 53*10 -5 230 Sc. Fv 6 B 8 E 12 Phe-MCA 7, 56*10 -3 8, 60*10 -6 879 Sc. Fv 6 B 8 E 12 Leu-MCA 8, 70*10 -3 5, 00*10 -5 174 M-1 s-1
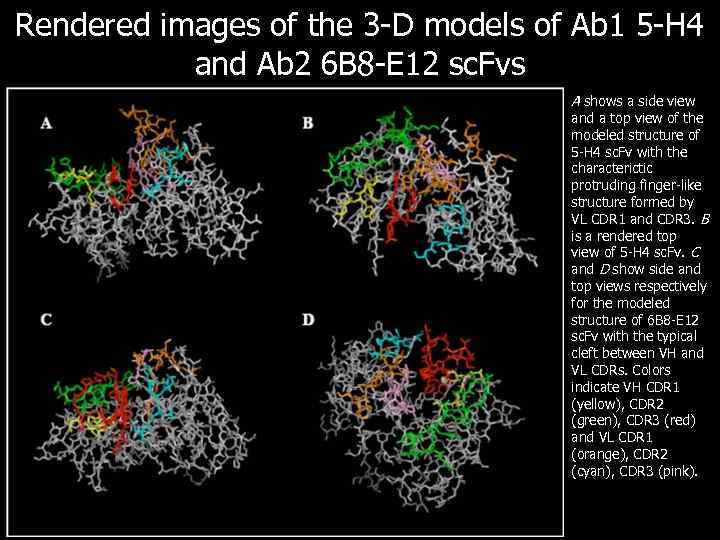 Rendered images of the 3 -D models of Ab 1 5 -H 4 and Ab 2 6 B 8 -E 12 sc. Fvs A shows a side view and a top view of the modeled structure of 5 -H 4 sc. Fv with the characterictic protruding finger-like structure formed by VL CDR 1 and CDR 3. B is a rendered top view of 5 -H 4 sc. Fv. C and D show side and top views respectively for the modeled structure of 6 B 8 -E 12 sc. Fv with the typical cleft between VH and VL CDRs. Colors indicate VH CDR 1 (yellow), CDR 2 (green), CDR 3 (red) and VL CDR 1 (orange), CDR 2 (cyan), CDR 3 (pink).
Rendered images of the 3 -D models of Ab 1 5 -H 4 and Ab 2 6 B 8 -E 12 sc. Fvs A shows a side view and a top view of the modeled structure of 5 -H 4 sc. Fv with the characterictic protruding finger-like structure formed by VL CDR 1 and CDR 3. B is a rendered top view of 5 -H 4 sc. Fv. C and D show side and top views respectively for the modeled structure of 6 B 8 -E 12 sc. Fv with the typical cleft between VH and VL CDRs. Colors indicate VH CDR 1 (yellow), CDR 2 (green), CDR 3 (red) and VL CDR 1 (orange), CDR 2 (cyan), CDR 3 (pink).
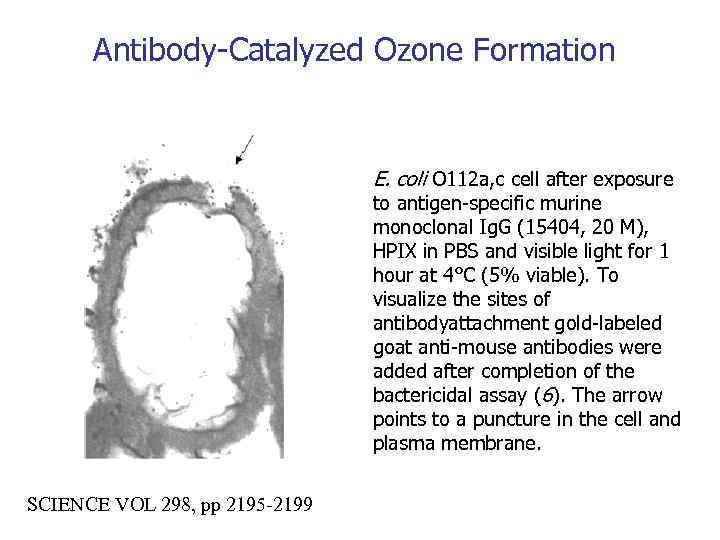 Antibody-Catalyzed Ozone Formation E. coli O 112 a, c cell after exposure to antigen-specific murine monoclonal Ig. G (15404, 20 M), HPIX in PBS and visible light for 1 hour at 4°C (5% viable). To visualize the sites of antibodyattachment gold-labeled goat anti-mouse antibodies were added after completion of the bactericidal assay (6). The arrow points to a puncture in the cell and plasma membrane. SCIENCE VOL 298, pp 2195 -2199
Antibody-Catalyzed Ozone Formation E. coli O 112 a, c cell after exposure to antigen-specific murine monoclonal Ig. G (15404, 20 M), HPIX in PBS and visible light for 1 hour at 4°C (5% viable). To visualize the sites of antibodyattachment gold-labeled goat anti-mouse antibodies were added after completion of the bactericidal assay (6). The arrow points to a puncture in the cell and plasma membrane. SCIENCE VOL 298, pp 2195 -2199
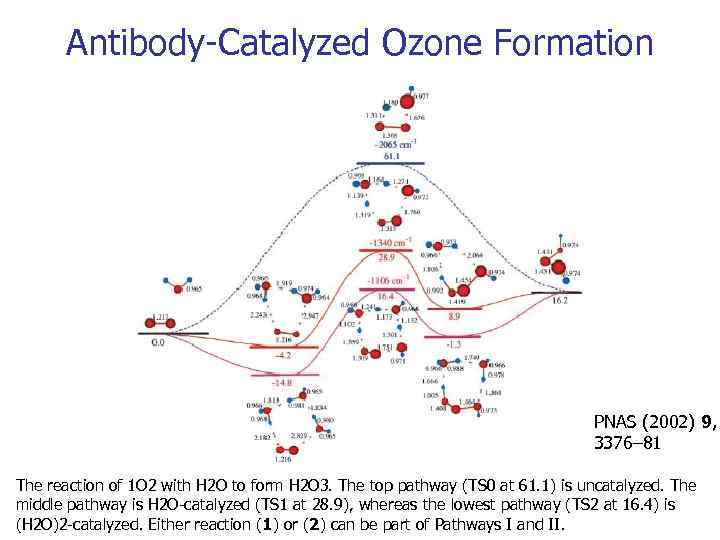 Antibody-Catalyzed Ozone Formation PNAS (2002) 9, 3376– 81 The reaction of 1 O 2 with H 2 O to form H 2 O 3. The top pathway (TS 0 at 61. 1) is uncatalyzed. The middle pathway is H 2 O-catalyzed (TS 1 at 28. 9), whereas the lowest pathway (TS 2 at 16. 4) is (H 2 O)2 -catalyzed. Either reaction (1) or (2) can be part of Pathways I and II.
Antibody-Catalyzed Ozone Formation PNAS (2002) 9, 3376– 81 The reaction of 1 O 2 with H 2 O to form H 2 O 3. The top pathway (TS 0 at 61. 1) is uncatalyzed. The middle pathway is H 2 O-catalyzed (TS 1 at 28. 9), whereas the lowest pathway (TS 2 at 16. 4) is (H 2 O)2 -catalyzed. Either reaction (1) or (2) can be part of Pathways I and II.
 Methods for generation and screening of combinatorial chemical libraries Tagged Methodologies: Based on a physical link between a protein and its encoded gene – the best known system is phage display. üPhage Technology üPeptides on Plasmids üPeptide coded Libraries üElectrophoric Polyhalobenzene Coded Libraries üEncoded Combinatorial Libraries The product of gene 8 (g 8 p) – is a small protein that forms the cylinder of the capsid; its number of copies (2700 for the wild-type phage). The other coat proteins (g 3 p, g 6 p, g 7 p and g 9 p) close the extremities of the cylinder. The product of gene 3 – present in three to five copies – is responsible for phage infectivity. Proteins of interest are usually fused by their C-terminal to g 3 p or to g 8 p. Filamentous phage
Methods for generation and screening of combinatorial chemical libraries Tagged Methodologies: Based on a physical link between a protein and its encoded gene – the best known system is phage display. üPhage Technology üPeptides on Plasmids üPeptide coded Libraries üElectrophoric Polyhalobenzene Coded Libraries üEncoded Combinatorial Libraries The product of gene 8 (g 8 p) – is a small protein that forms the cylinder of the capsid; its number of copies (2700 for the wild-type phage). The other coat proteins (g 3 p, g 6 p, g 7 p and g 9 p) close the extremities of the cylinder. The product of gene 3 – present in three to five copies – is responsible for phage infectivity. Proteins of interest are usually fused by their C-terminal to g 3 p or to g 8 p. Filamentous phage
 Three critical aspects in any combinatorial library: üThe chemical units that go to the library üThe technique for generating the library üIdentification of the library members that interact with the biological target of interest
Three critical aspects in any combinatorial library: üThe chemical units that go to the library üThe technique for generating the library üIdentification of the library members that interact with the biological target of interest
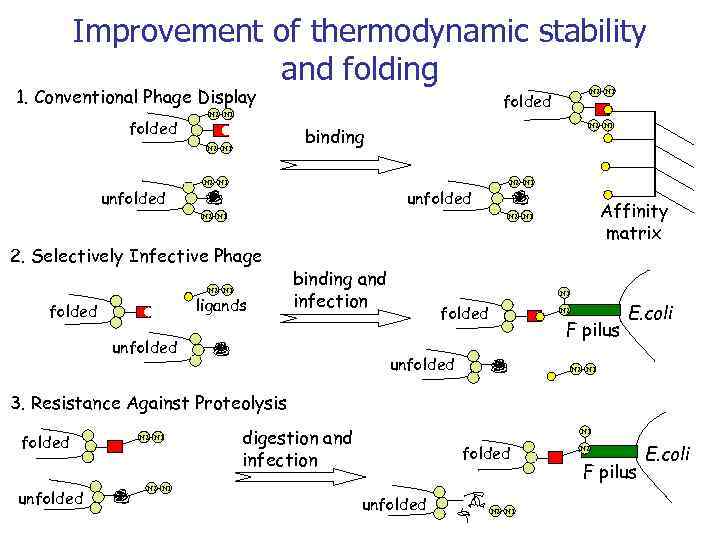 Improvement of thermodynamic stability and folding 1. Conventional Phage Display folded N 2 N 1 binding N 2 N 1 unfolded N 2 N 1 Affinity matrix N 2 N 1 2. Selectively Infective Phage N 2 N 1 ligands folded N 2 N 1 folded binding and infection unfolded N 1 folded N 2 F pilus unfolded E. coli N 2 N 1 3. Resistance Against Proteolysis folded unfolded N 2 N 1 digestion and infection N 1 folded unfolded N 2 N 1 N 2 F pilus E. coli
Improvement of thermodynamic stability and folding 1. Conventional Phage Display folded N 2 N 1 binding N 2 N 1 unfolded N 2 N 1 Affinity matrix N 2 N 1 2. Selectively Infective Phage N 2 N 1 ligands folded N 2 N 1 folded binding and infection unfolded N 1 folded N 2 F pilus unfolded E. coli N 2 N 1 3. Resistance Against Proteolysis folded unfolded N 2 N 1 digestion and infection N 1 folded unfolded N 2 N 1 N 2 F pilus E. coli
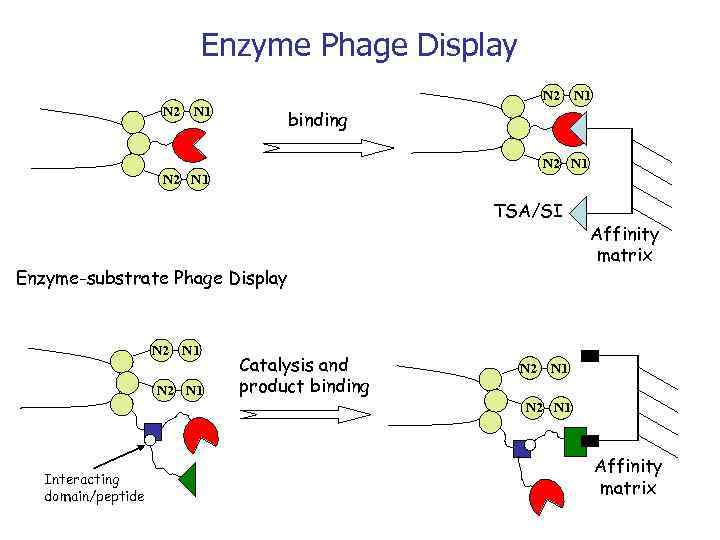 Enzyme Phage Display N 2 N 1 binding N 2 N 1 TSA/SI Enzyme-substrate Phage Display N 2 N 1 Catalysis and product binding Affinity matrix N 2 N 1 Interacting domain/peptide Affinity matrix
Enzyme Phage Display N 2 N 1 binding N 2 N 1 TSA/SI Enzyme-substrate Phage Display N 2 N 1 Catalysis and product binding Affinity matrix N 2 N 1 Interacting domain/peptide Affinity matrix
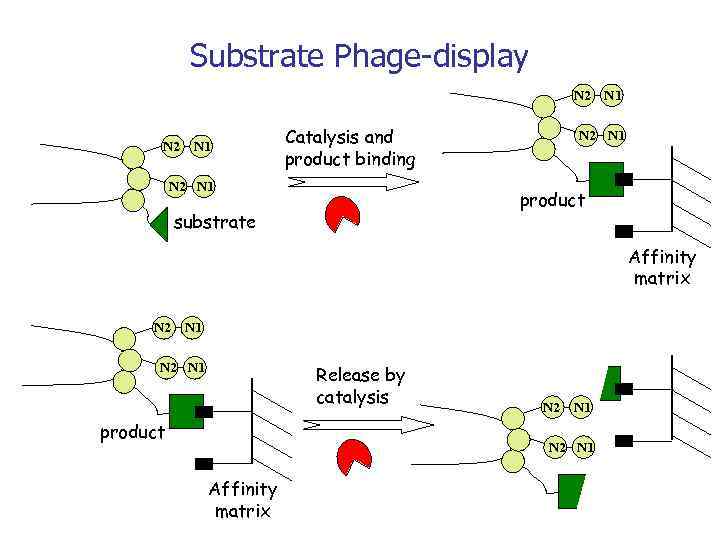 Substrate Phage-display N 2 N 1 Catalysis and product binding N 2 N 1 product substrate Affinity matrix N 2 N 1 Release by catalysis product N 2 N 1 Affinity matrix
Substrate Phage-display N 2 N 1 Catalysis and product binding N 2 N 1 product substrate Affinity matrix N 2 N 1 Release by catalysis product N 2 N 1 Affinity matrix
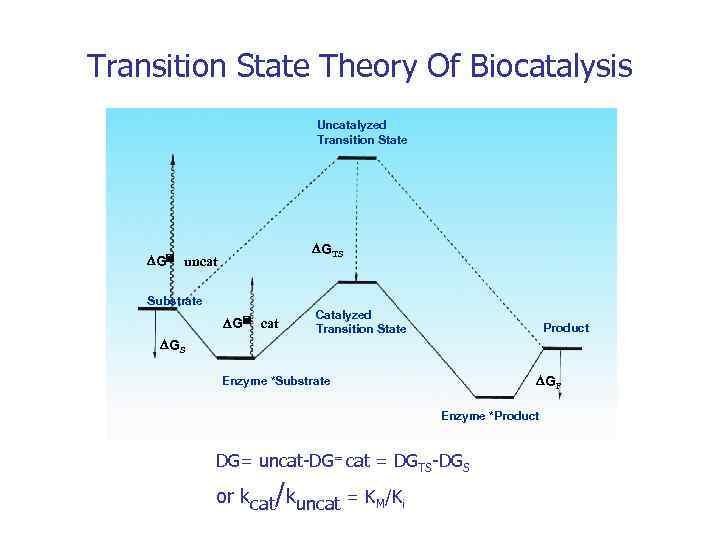 Transition State Theory Of Biocatalysis Uncatalyzed Transition State DGTS DG= uncat Substrate DG= cat Catalyzed Transition State Product DGS DGP Enzyme *Substrate Enzyme *Product DG= uncat-DG= cat = DGTS-DGS or kcat/kuncat = KM/Ki
Transition State Theory Of Biocatalysis Uncatalyzed Transition State DGTS DG= uncat Substrate DG= cat Catalyzed Transition State Product DGS DGP Enzyme *Substrate Enzyme *Product DG= uncat-DG= cat = DGTS-DGS or kcat/kuncat = KM/Ki
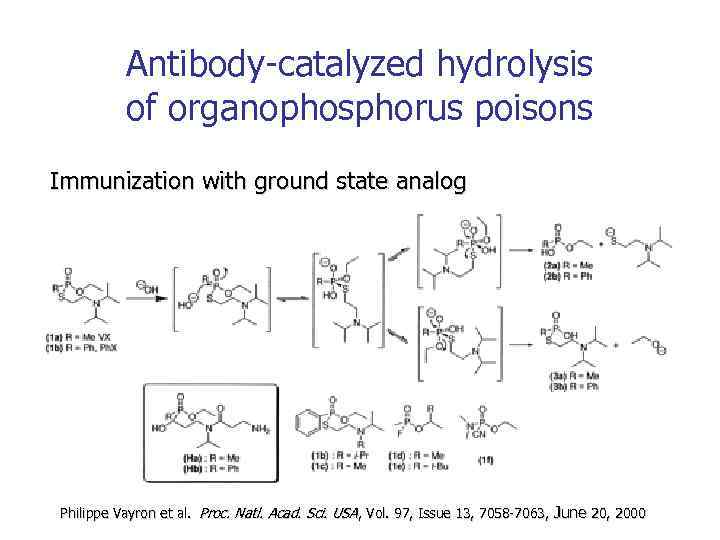 Antibody-catalyzed hydrolysis of organophosphorus poisons Immunization with ground state analog Philippe Vayron et al. Proc. Natl. Acad. Sci. USA, Vol. 97, Issue 13, 7058 -7063, June 20, 2000
Antibody-catalyzed hydrolysis of organophosphorus poisons Immunization with ground state analog Philippe Vayron et al. Proc. Natl. Acad. Sci. USA, Vol. 97, Issue 13, 7058 -7063, June 20, 2000
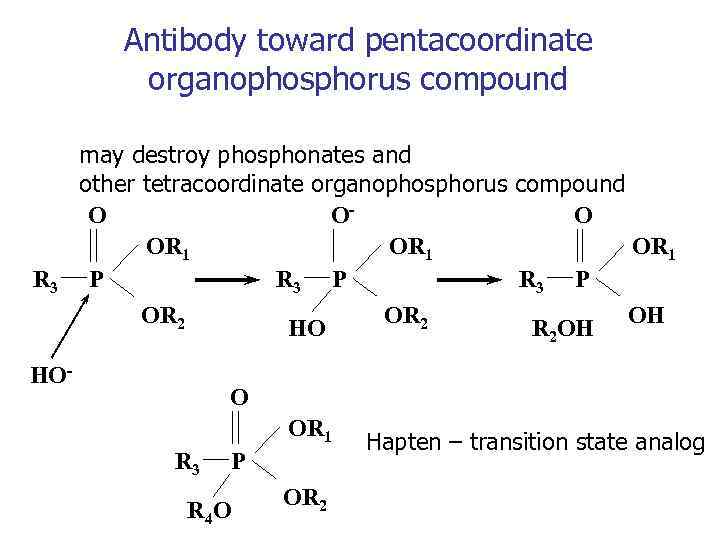 Antibody toward pentacoordinate organophosphorus compound R 3 may destroy phosphonates and other tetracoordinate organophosphorus compound O OO OR 1 R 3 P P OR 2 OH HO R 2 OH HO- O OR 1 R 3 P R 4 O OR 2 Hapten – transition state analog
Antibody toward pentacoordinate organophosphorus compound R 3 may destroy phosphonates and other tetracoordinate organophosphorus compound O OO OR 1 R 3 P P OR 2 OH HO R 2 OH HO- O OR 1 R 3 P R 4 O OR 2 Hapten – transition state analog
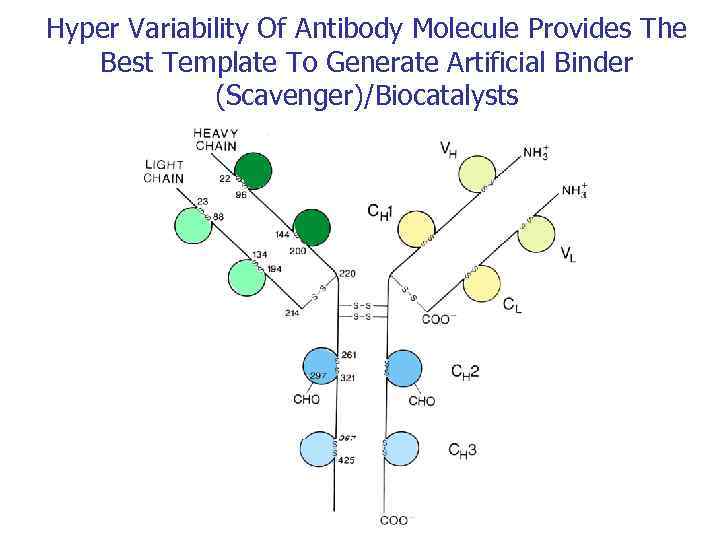 Hyper Variability Of Antibody Molecule Provides The Best Template To Generate Artificial Binder (Scavenger)/Biocatalysts
Hyper Variability Of Antibody Molecule Provides The Best Template To Generate Artificial Binder (Scavenger)/Biocatalysts
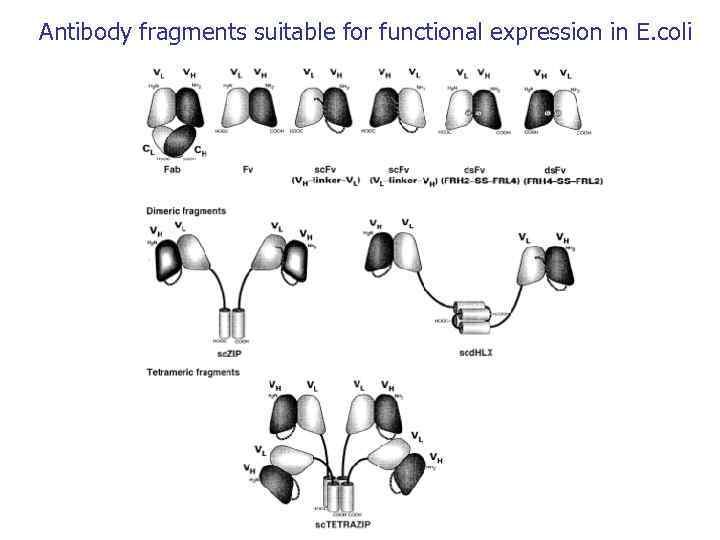 Antibody fragments suitable for functional expression in E. coli
Antibody fragments suitable for functional expression in E. coli
 Химическая селекция фагдисплейных библиотек Ферменты Миллионы лет эволюции Искусственные биокатализаторы, полученные комбинаторными способами Несколько месяцев или недель эволюции Отбор из 108÷ 109 индивидуальных клонов Классы ферментов могут быть заменены суперсемейством иммуноглобулинов
Химическая селекция фагдисплейных библиотек Ферменты Миллионы лет эволюции Искусственные биокатализаторы, полученные комбинаторными способами Несколько месяцев или недель эволюции Отбор из 108÷ 109 индивидуальных клонов Классы ферментов могут быть заменены суперсемейством иммуноглобулинов
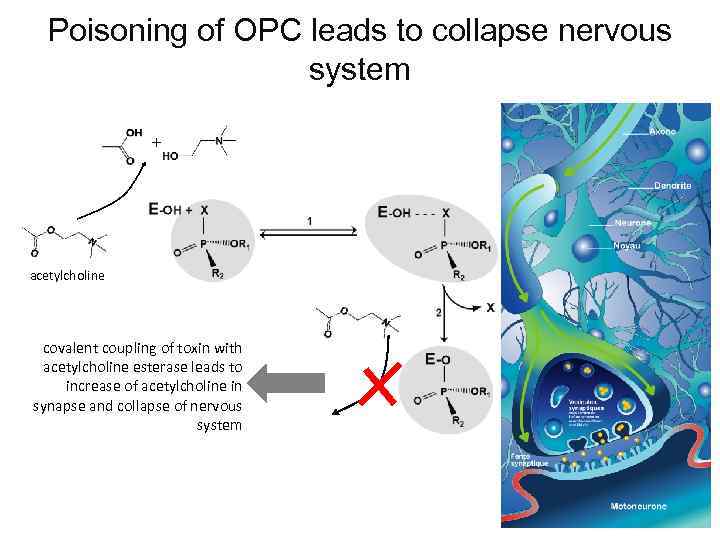 Poisoning of OPC leads to collapse nervous system + acetylcholine covalent coupling of toxin with acetylcholine esterase leads to increase of acetylcholine in synapse and collapse of nervous system
Poisoning of OPC leads to collapse nervous system + acetylcholine covalent coupling of toxin with acetylcholine esterase leads to increase of acetylcholine in synapse and collapse of nervous system
 Question of OPC-poising therapy The major target of organophosphorous toxins are cholinesterase-like enzymes; Extremely low LD 50 value OPC-associated mortality is 200000 people per year; There are real threats of acts of terrorism, for example sarin attack in Tokyo underground at 20 March of 1995
Question of OPC-poising therapy The major target of organophosphorous toxins are cholinesterase-like enzymes; Extremely low LD 50 value OPC-associated mortality is 200000 people per year; There are real threats of acts of terrorism, for example sarin attack in Tokyo underground at 20 March of 1995
 Risk group Soldier Stuff of OP poisons utilization plant Farmers Users
Risk group Soldier Stuff of OP poisons utilization plant Farmers Users
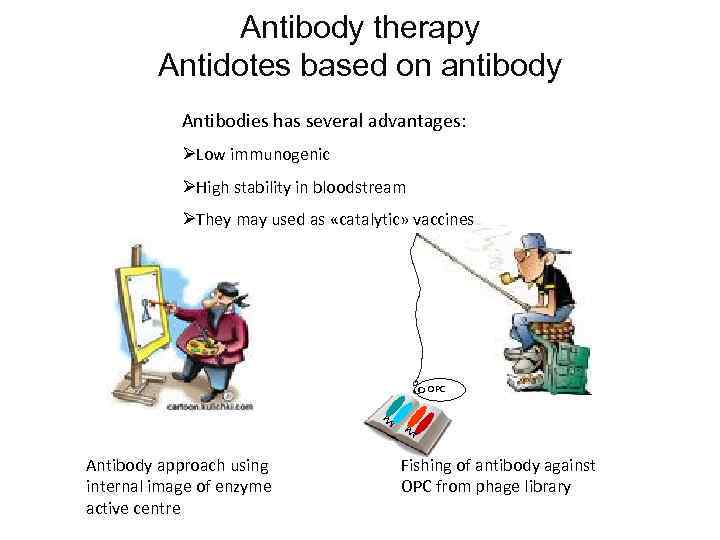 Antibody therapy Antidotes based on antibody Antibodies has several advantages: ØLow immunogenic ØHigh stability in bloodstream ØThey may used as «catalytic» vaccines OPC Antibody approach using internal image of enzyme active centre Fishing of antibody against OPC from phage library
Antibody therapy Antidotes based on antibody Antibodies has several advantages: ØLow immunogenic ØHigh stability in bloodstream ØThey may used as «catalytic» vaccines OPC Antibody approach using internal image of enzyme active centre Fishing of antibody against OPC from phage library
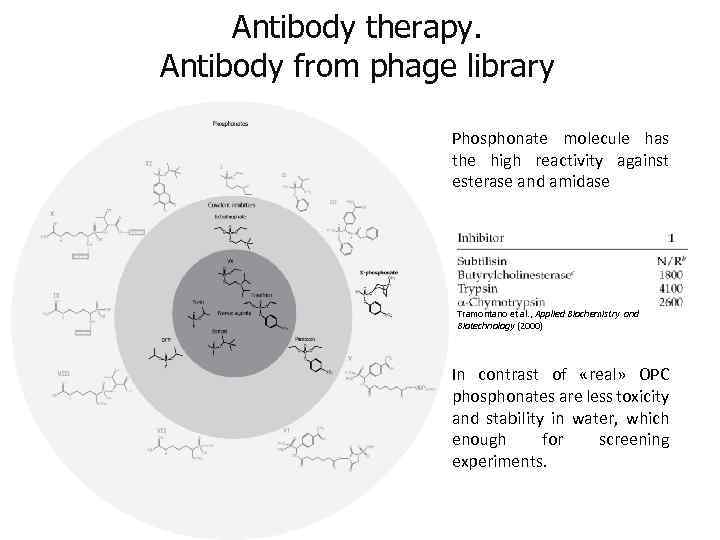 Antibody therapy. Antibody from phage library Phosphonate molecule has the high reactivity against esterase and amidase Tramontano et al. , Applied Biochemistry and Biotechnology (2000) In contrast of «real» OPC phosphonates are less toxicity and stability in water, which enough for screening experiments.
Antibody therapy. Antibody from phage library Phosphonate molecule has the high reactivity against esterase and amidase Tramontano et al. , Applied Biochemistry and Biotechnology (2000) In contrast of «real» OPC phosphonates are less toxicity and stability in water, which enough for screening experiments.
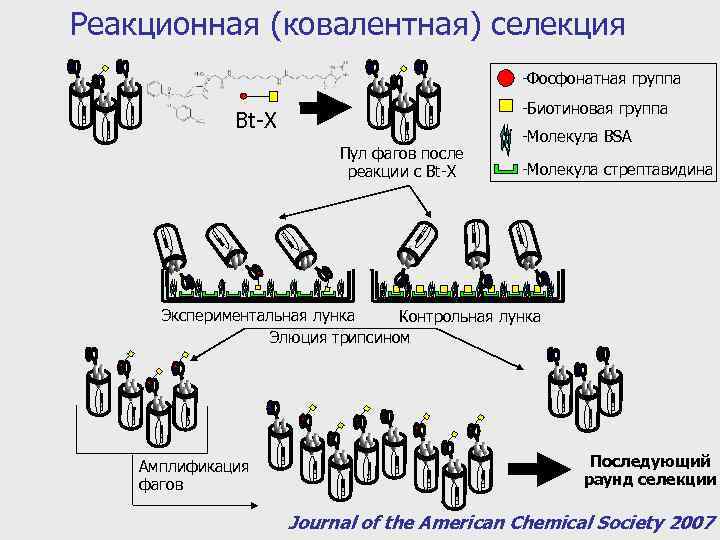 Реакционная (ковалентная) селекция -Фосфонатная группа -Биотиновая группа Bt-X Пул фагов после реакции с Bt-X -Молекула BSA -Молекула стрептавидина Экспериментальная лунка Контрольная лунка Элюция трипсином Амплификация фагов Последующий раунд селекции Journal of the American Chemical Society 2007
Реакционная (ковалентная) селекция -Фосфонатная группа -Биотиновая группа Bt-X Пул фагов после реакции с Bt-X -Молекула BSA -Молекула стрептавидина Экспериментальная лунка Контрольная лунка Элюция трипсином Амплификация фагов Последующий раунд селекции Journal of the American Chemical Society 2007
 Alignment of covalent and non-covalent binding clones All selected clones are highly conservative, are highlighted in yellow and those of The conserved amino acid residues of the heavy chain but those clones which could light phosphonate non-covalently deviates more significant. bind chain in green. CDR regions are enclosed in blue boxes. Reactive clones are shown in. bold font.
Alignment of covalent and non-covalent binding clones All selected clones are highly conservative, are highlighted in yellow and those of The conserved amino acid residues of the heavy chain but those clones which could light phosphonate non-covalently deviates more significant. bind chain in green. CDR regions are enclosed in blue boxes. Reactive clones are shown in. bold font.
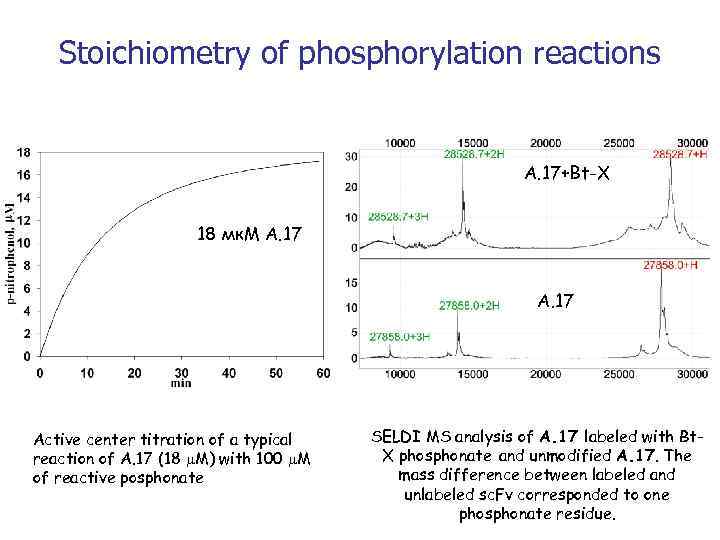 Stoichiometry of phosphorylation reactions А. 17+Bt-X 18 мк. М А. 17 Active center titration of a typical reaction of A. 17 (18 M) with 100 M of reactive posphonate SELDI MS analysis of A. 17 labeled with Bt. X phosphonate and unmodified A. 17. The mass difference between labeled and unlabeled sc. Fv corresponded to one phosphonate residue.
Stoichiometry of phosphorylation reactions А. 17+Bt-X 18 мк. М А. 17 Active center titration of a typical reaction of A. 17 (18 M) with 100 M of reactive posphonate SELDI MS analysis of A. 17 labeled with Bt. X phosphonate and unmodified A. 17. The mass difference between labeled and unlabeled sc. Fv corresponded to one phosphonate residue.
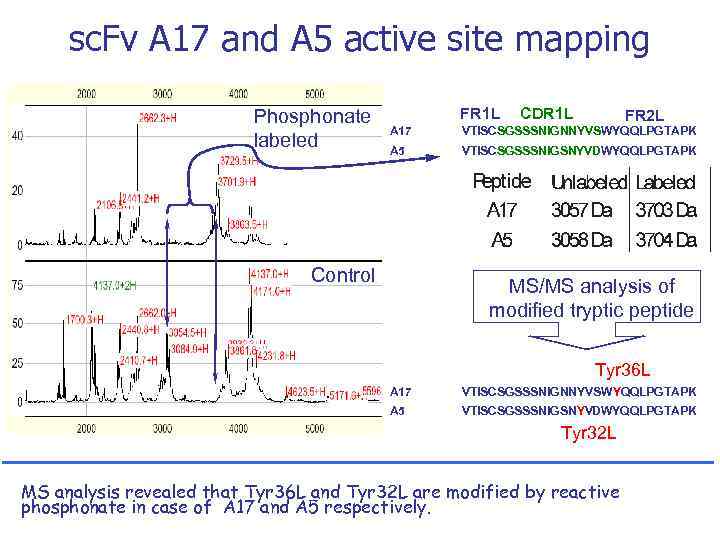 sc. Fv A 17 and A 5 active site mapping Phosphonate labeled FR 1 L CDR 1 L FR 2 L A 17 VTISCSGSSSNIGNNYVSWYQQLPGTAPK A 5 VTISCSGSSSNIGSNYVDWYQQLPGTAPK Control MS/MS analysis of modified tryptic peptide Tyr 36 L A 17 VTISCSGSSSNIGNNYVSWYQQLPGTAPK A 5 VTISCSGSSSNIGSNYVDWYQQLPGTAPK Tyr 32 L MS analysis revealed that Tyr 36 L and Tyr 32 L are modified by reactive phosphonate in case of A 17 and A 5 respectively.
sc. Fv A 17 and A 5 active site mapping Phosphonate labeled FR 1 L CDR 1 L FR 2 L A 17 VTISCSGSSSNIGNNYVSWYQQLPGTAPK A 5 VTISCSGSSSNIGSNYVDWYQQLPGTAPK Control MS/MS analysis of modified tryptic peptide Tyr 36 L A 17 VTISCSGSSSNIGNNYVSWYQQLPGTAPK A 5 VTISCSGSSSNIGSNYVDWYQQLPGTAPK Tyr 32 L MS analysis revealed that Tyr 36 L and Tyr 32 L are modified by reactive phosphonate in case of A 17 and A 5 respectively.
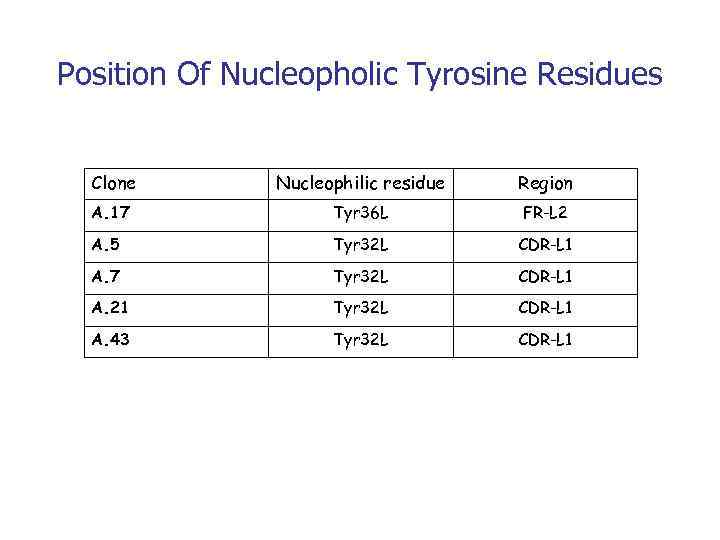 Position Of Nucleopholic Tyrosine Residues Clone Nucleophilic residue Region А. 17 Tyr 36 L FR-L 2 А. 5 Tyr 32 L CDR-L 1 А. 7 Tyr 32 L CDR-L 1 А. 21 Tyr 32 L CDR-L 1 А. 43 Tyr 32 L CDR-L 1
Position Of Nucleopholic Tyrosine Residues Clone Nucleophilic residue Region А. 17 Tyr 36 L FR-L 2 А. 5 Tyr 32 L CDR-L 1 А. 7 Tyr 32 L CDR-L 1 А. 21 Tyr 32 L CDR-L 1 А. 43 Tyr 32 L CDR-L 1
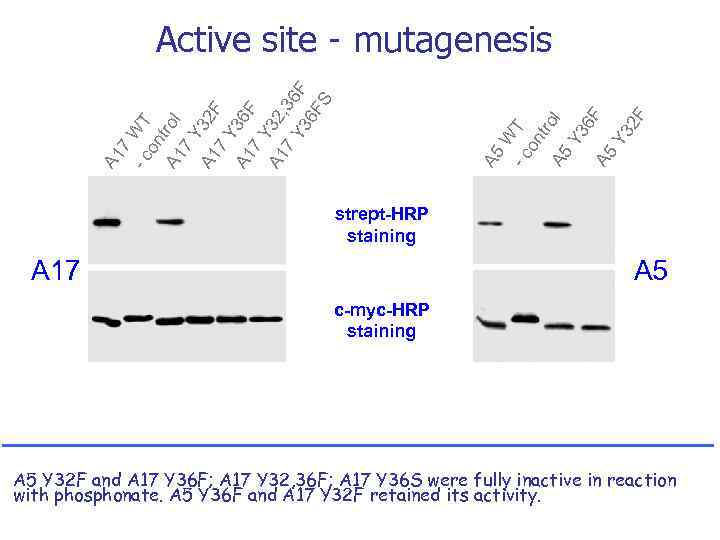 -c A 1 A 5 7 W T on tr A 5 ol Y 3 6 F A 5 Y 3 2 F - c WT on A 1 trol 7 Y A 1 32 F 7 Y A 1 36 F 7 Y A 1 32 , 3 7 Y 3 6 F 6 F S Active site - mutagenesis strept-HRP staining A 17 A 5 c-myc-HRP staining A 5 Y 32 F and A 17 Y 36 F; A 17 Y 32, 36 F; A 17 Y 36 S were fully inactive in reaction with phosphonate. A 5 Y 36 F and A 17 Y 32 F retained its activity.
-c A 1 A 5 7 W T on tr A 5 ol Y 3 6 F A 5 Y 3 2 F - c WT on A 1 trol 7 Y A 1 32 F 7 Y A 1 36 F 7 Y A 1 32 , 3 7 Y 3 6 F 6 F S Active site - mutagenesis strept-HRP staining A 17 A 5 c-myc-HRP staining A 5 Y 32 F and A 17 Y 36 F; A 17 Y 32, 36 F; A 17 Y 36 S were fully inactive in reaction with phosphonate. A 5 Y 36 F and A 17 Y 32 F retained its activity.
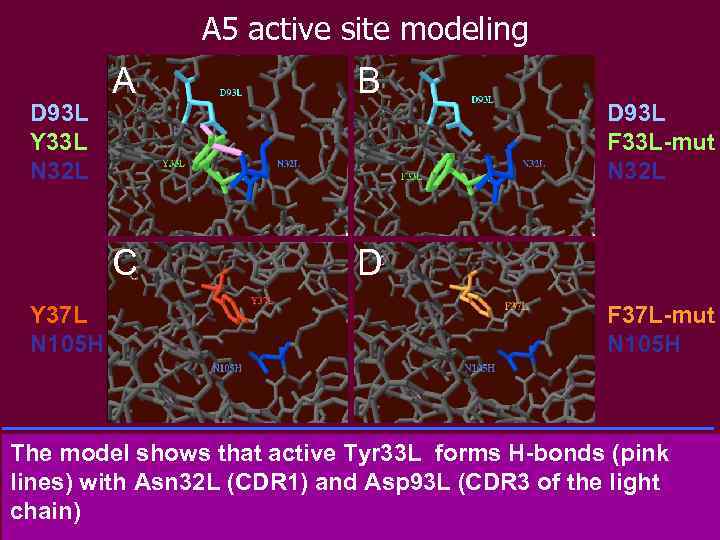 A 5 active site modeling Y 37 L N 105 H B C D 93 L Y 33 L N 32 L A D D 93 L F 33 L-mut N 32 L F 37 L-mut N 105 H The model shows that active Tyr 33 L forms H-bonds (pink lines) with Asn 32 L (CDR 1) and Asp 93 L (CDR 3 of the light chain) A: The model shows that Tyr 33 L, which is located on the surface of the protein, in CDR 1 of the light chain, forms H-bonds (pink lines) with Asn 32 L (CDR 1) and Asp 93 L (CDR 3 of the light chain). B: When Tyr 33 L is mutated in Phe 33 L no H-bonds were found. C: Tyr 37 L does not form any H-bond with Asn 105 H (CDR 3 of the heavy chain). The distance between the Tyr and Asn residues is 3. 28 angstroms. D: When Tyr 37 L is mutated into Phe 37 L, the distance between the Tyr and Phe residues is found to increase to 4. 84 angstroms
A 5 active site modeling Y 37 L N 105 H B C D 93 L Y 33 L N 32 L A D D 93 L F 33 L-mut N 32 L F 37 L-mut N 105 H The model shows that active Tyr 33 L forms H-bonds (pink lines) with Asn 32 L (CDR 1) and Asp 93 L (CDR 3 of the light chain) A: The model shows that Tyr 33 L, which is located on the surface of the protein, in CDR 1 of the light chain, forms H-bonds (pink lines) with Asn 32 L (CDR 1) and Asp 93 L (CDR 3 of the light chain). B: When Tyr 33 L is mutated in Phe 33 L no H-bonds were found. C: Tyr 37 L does not form any H-bond with Asn 105 H (CDR 3 of the heavy chain). The distance between the Tyr and Asn residues is 3. 28 angstroms. D: When Tyr 37 L is mutated into Phe 37 L, the distance between the Tyr and Phe residues is found to increase to 4. 84 angstroms
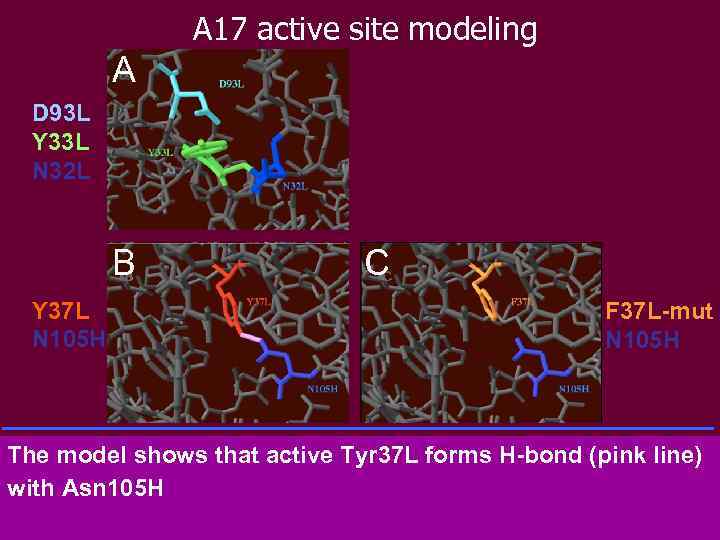 A 17 active site modeling A D 93 L Y 33 L N 32 L B Y 37 L N 105 H C F 37 L-mut N 105 H A: The model shows that Tyr 33 L does not form any H-bond with Asn 32 L (CDR 1) and Asp 93 L (CDR 3 of the light chain). The distances between the Tyr residue and the Asp and Asn residues are found to be 3. 33 and 3. 80 angstroms respectively (to be compared with the 2. 18 and 2. 04 found for sc. Fv#5). B: At the contrary to sc. Fv#5, in sc. Fv#17, Tyr 37 L forms an H-bond with Asn 105 H. The distance between the two residues is found to be 1. 79 angstroms. C: When Tyr 37 L is mutated to Phe 37 L there is no longer any H-bond, the distance between the two residues is found to be 2. 93 angstroms The model shows that active Tyr 37 L forms H-bond (pink line) with Asn 105 H
A 17 active site modeling A D 93 L Y 33 L N 32 L B Y 37 L N 105 H C F 37 L-mut N 105 H A: The model shows that Tyr 33 L does not form any H-bond with Asn 32 L (CDR 1) and Asp 93 L (CDR 3 of the light chain). The distances between the Tyr residue and the Asp and Asn residues are found to be 3. 33 and 3. 80 angstroms respectively (to be compared with the 2. 18 and 2. 04 found for sc. Fv#5). B: At the contrary to sc. Fv#5, in sc. Fv#17, Tyr 37 L forms an H-bond with Asn 105 H. The distance between the two residues is found to be 1. 79 angstroms. C: When Tyr 37 L is mutated to Phe 37 L there is no longer any H-bond, the distance between the two residues is found to be 2. 93 angstroms The model shows that active Tyr 37 L forms H-bond (pink line) with Asn 105 H
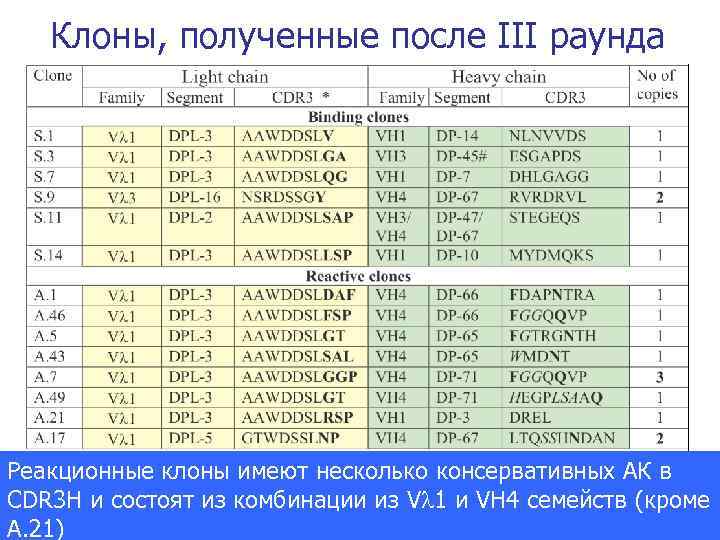 Клоны, полученные после III раунда Реакционные клоны имеют несколько консервативных АК в CDR 3 H и состоят из комбинации из V 1 и VH 4 семейств (кроме Journal of the American Chemical Society 2007 A. 21)
Клоны, полученные после III раунда Реакционные клоны имеют несколько консервативных АК в CDR 3 H и состоят из комбинации из V 1 и VH 4 семейств (кроме Journal of the American Chemical Society 2007 A. 21)
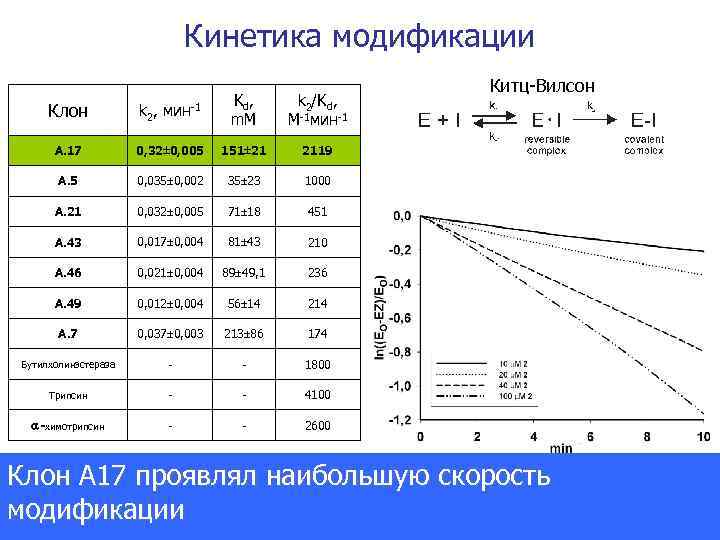 Кинетика модификации Клон k 2, мин-1 Kd , m. M k 2/Kd, М-1 мин-1 А. 17 0, 32 0, 005 151 21 2119 А. 5 0, 035 0, 002 35 23 1000 А. 21 0, 032 0, 005 71 18 451 А. 43 0, 017 0, 004 81 43 210 А. 46 0, 021 0, 004 89 49, 1 236 А. 49 0, 012 0, 004 56 14 214 А. 7 0, 037 0, 003 213 86 174 Бутилхолинэстераза - - 1800 Трипсин - - 4100 -химотрипсин - - Китц-Вилсон 2600 Клон А 17 проявлял наибольшую скорость модификации Journal of the American Chemical Society 2007
Кинетика модификации Клон k 2, мин-1 Kd , m. M k 2/Kd, М-1 мин-1 А. 17 0, 32 0, 005 151 21 2119 А. 5 0, 035 0, 002 35 23 1000 А. 21 0, 032 0, 005 71 18 451 А. 43 0, 017 0, 004 81 43 210 А. 46 0, 021 0, 004 89 49, 1 236 А. 49 0, 012 0, 004 56 14 214 А. 7 0, 037 0, 003 213 86 174 Бутилхолинэстераза - - 1800 Трипсин - - 4100 -химотрипсин - - Китц-Вилсон 2600 Клон А 17 проявлял наибольшую скорость модификации Journal of the American Chemical Society 2007
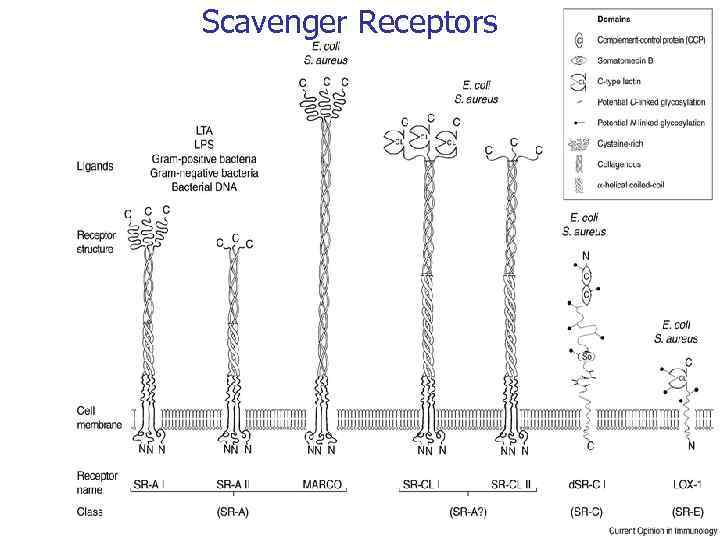 Scavenger Receptors
Scavenger Receptors
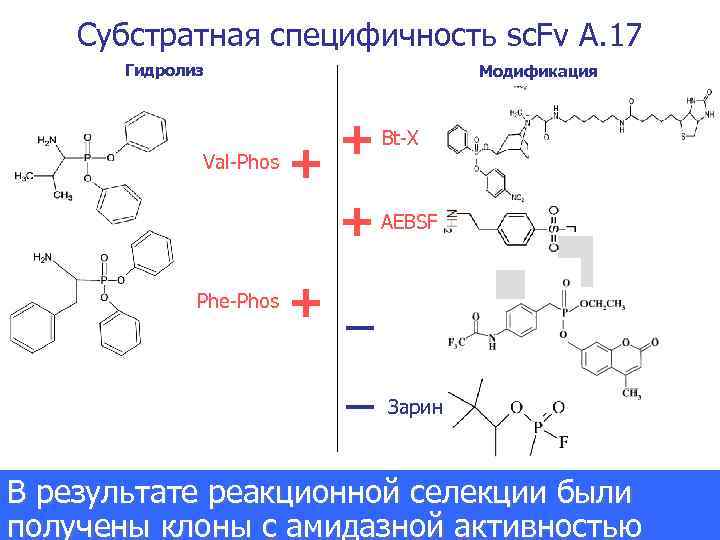 Субстратная специфичность sc. Fv A. 17 Гидролиз Val-Phos Модификация + + Bt-X + AEBSF Phe-Phos + _ _ Зарин В результате реакционной селекции были Journal of the American Chemical Society получены клоны с амидазной активностью 2007
Субстратная специфичность sc. Fv A. 17 Гидролиз Val-Phos Модификация + + Bt-X + AEBSF Phe-Phos + _ _ Зарин В результате реакционной селекции были Journal of the American Chemical Society получены клоны с амидазной активностью 2007
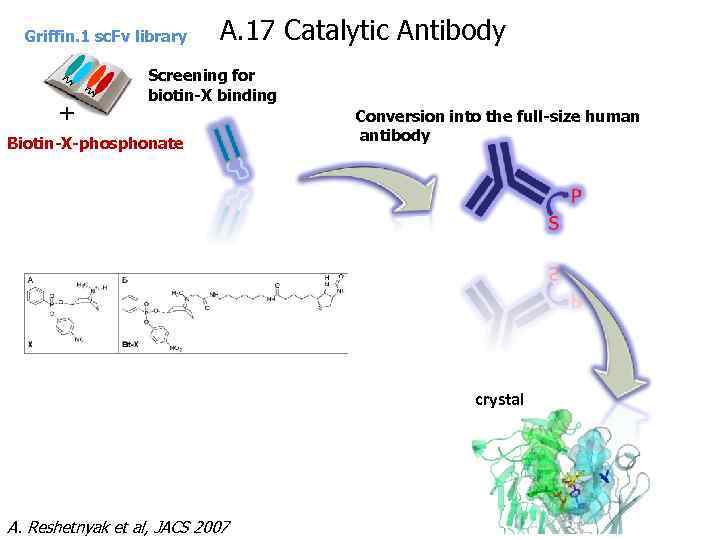 Griffin. 1 sc. Fv library + A. 17 Catalytic Antibody Screening for biotin-X binding Biotin-X-phosphonate Conversion into the full-size human antibody crystal A. Reshetnyak et al, JACS 2007
Griffin. 1 sc. Fv library + A. 17 Catalytic Antibody Screening for biotin-X binding Biotin-X-phosphonate Conversion into the full-size human antibody crystal A. Reshetnyak et al, JACS 2007
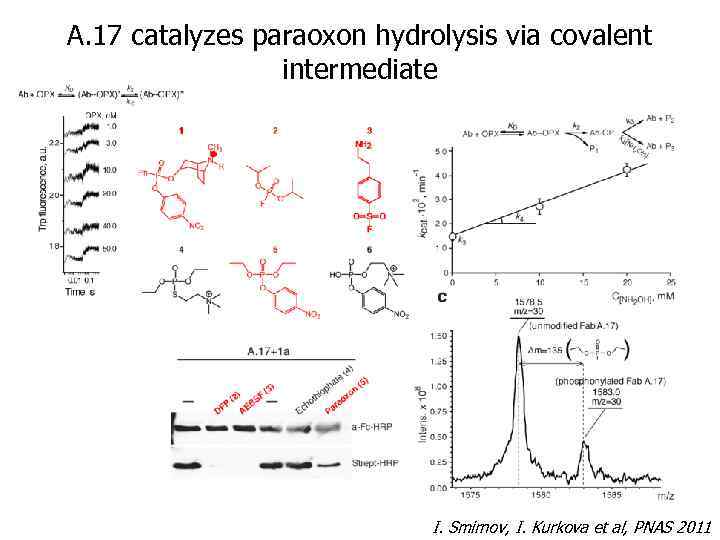 A. 17 catalyzes paraoxon hydrolysis via covalent intermediate I. Smirnov, I. Kurkova et al, PNAS 2011
A. 17 catalyzes paraoxon hydrolysis via covalent intermediate I. Smirnov, I. Kurkova et al, PNAS 2011
 A. 17 antibody has unusual deep cavity with nucleophilic tyrosine at its base I. Smirnov, I. Kurkova et al, PNAS 2011
A. 17 antibody has unusual deep cavity with nucleophilic tyrosine at its base I. Smirnov, I. Kurkova et al, PNAS 2011
 A. 17 antibody has unusual deep cavity with nucleophilic tyrosine at its base I. Smirnov, I. Kurkova et al, PNAS 2011
A. 17 antibody has unusual deep cavity with nucleophilic tyrosine at its base I. Smirnov, I. Kurkova et al, PNAS 2011
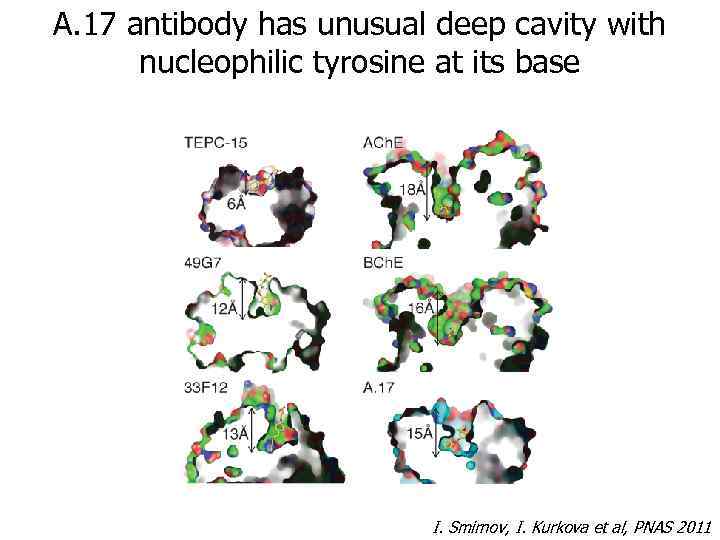 A. 17 antibody has unusual deep cavity with nucleophilic tyrosine at its base I. Smirnov, I. Kurkova et al, PNAS 2011
A. 17 antibody has unusual deep cavity with nucleophilic tyrosine at its base I. Smirnov, I. Kurkova et al, PNAS 2011
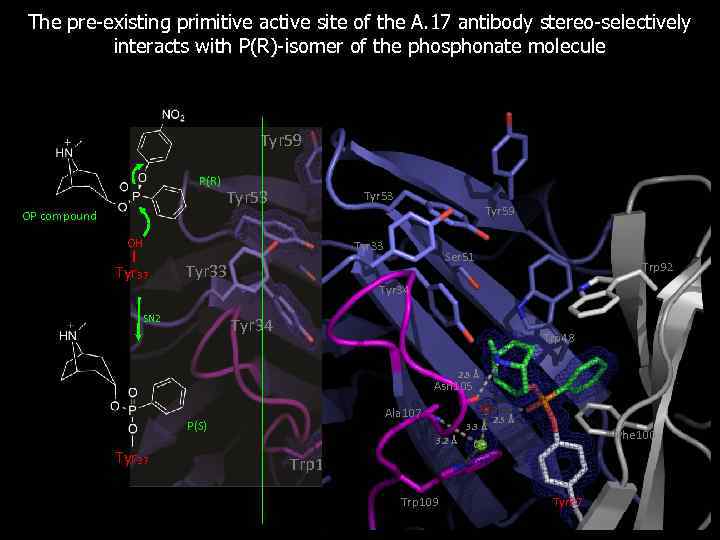 The pre-existing primitive active site of the A. 17 antibody stereo-selectively interacts with P(R)-isomer of the phosphonate molecule Tyr 59 P(R) OP compound Tyr 53 OH Tyr 37 Tyr 59 Trp 92 Tyr 33 Ser 51 Tyr 33 Trp 48 Tyr 34 SN 2 Trp 92 Tyr 34 Phe 100 Trp 48 2. 8 Å Asn 105 Ala 107 P(S) 3. 3 Å 3. 2 Å Tyr 37 Trp 109 2. 5 Å Phe 100 Cl- Tyr 37
The pre-existing primitive active site of the A. 17 antibody stereo-selectively interacts with P(R)-isomer of the phosphonate molecule Tyr 59 P(R) OP compound Tyr 53 OH Tyr 37 Tyr 59 Trp 92 Tyr 33 Ser 51 Tyr 33 Trp 48 Tyr 34 SN 2 Trp 92 Tyr 34 Phe 100 Trp 48 2. 8 Å Asn 105 Ala 107 P(S) 3. 3 Å 3. 2 Å Tyr 37 Trp 109 2. 5 Å Phe 100 Cl- Tyr 37
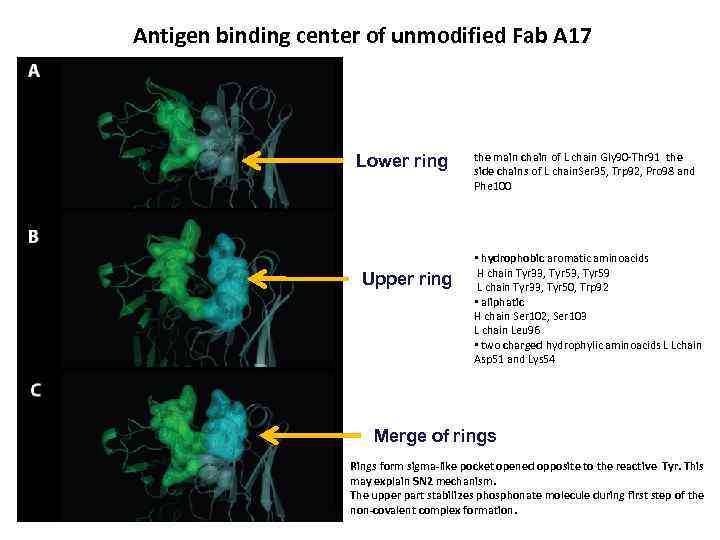 Antigen binding center of unmodified Fab A 17 Lower ring Upper ring the main chain of L chain Gly 90 -Thr 91 the side chains of L chain. Ser 35, Trp 92, Pro 98 and Phe 100 • hydrophobic aromatic aminoacids H chain Tyr 33, Tyr 59 L chain Tyr 33, Tyr 50, Trp 92 • aliphatic H chain Ser 102, Ser 103 L chain Leu 96 • two charged hydrophylic aminoacids L Lchain Asp 51 and Lys 54 Merge of rings Rings form sigma-like pocket opened opposite to the reactive Tyr. This may explain SN 2 mechanism. The upper part stabilizes phosphonate molecule during first step of the non-covalent complex formation.
Antigen binding center of unmodified Fab A 17 Lower ring Upper ring the main chain of L chain Gly 90 -Thr 91 the side chains of L chain. Ser 35, Trp 92, Pro 98 and Phe 100 • hydrophobic aromatic aminoacids H chain Tyr 33, Tyr 59 L chain Tyr 33, Tyr 50, Trp 92 • aliphatic H chain Ser 102, Ser 103 L chain Leu 96 • two charged hydrophylic aminoacids L Lchain Asp 51 and Lys 54 Merge of rings Rings form sigma-like pocket opened opposite to the reactive Tyr. This may explain SN 2 mechanism. The upper part stabilizes phosphonate molecule during first step of the non-covalent complex formation.
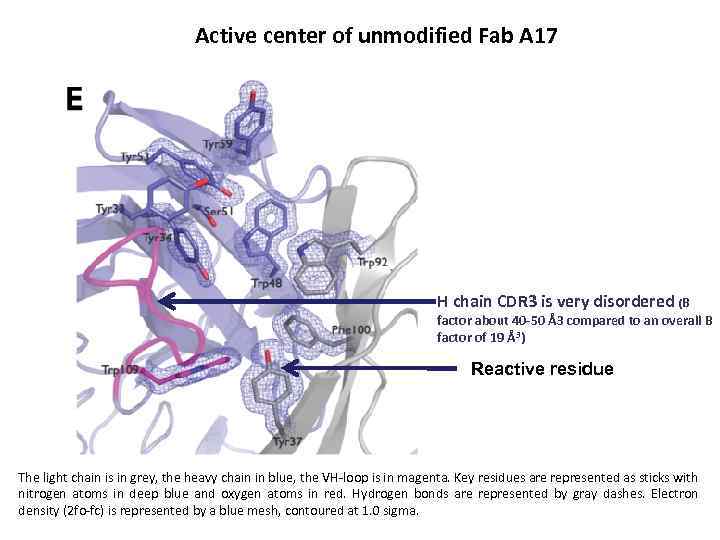 Active center of unmodified Fab A 17 H chain CDR 3 is very disordered (B factor about 40 -50 Å3 compared to an overall B factor of 19 Å3) Reactive residue The light chain is in grey, the heavy chain in blue, the VH-loop is in magenta. Key residues are represented as sticks with nitrogen atoms in deep blue and oxygen atoms in red. Hydrogen bonds are represented by gray dashes. Electron density (2 fo-fc) is represented by a blue mesh, contoured at 1. 0 sigma.
Active center of unmodified Fab A 17 H chain CDR 3 is very disordered (B factor about 40 -50 Å3 compared to an overall B factor of 19 Å3) Reactive residue The light chain is in grey, the heavy chain in blue, the VH-loop is in magenta. Key residues are represented as sticks with nitrogen atoms in deep blue and oxygen atoms in red. Hydrogen bonds are represented by gray dashes. Electron density (2 fo-fc) is represented by a blue mesh, contoured at 1. 0 sigma.
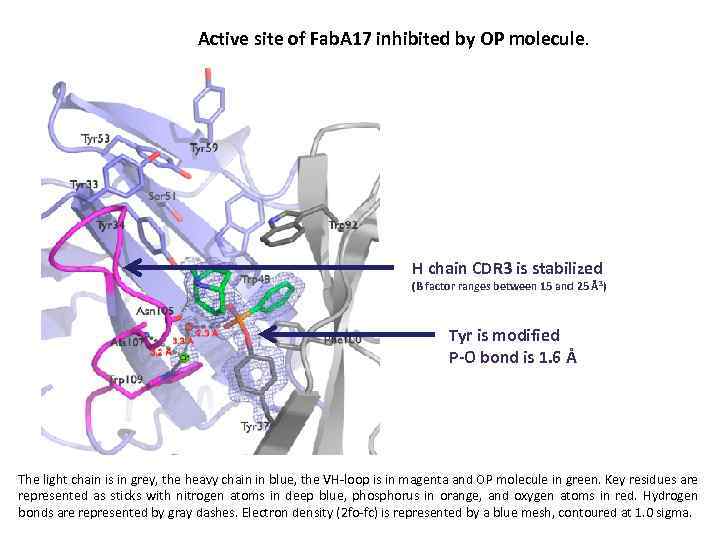 Active site of Fab. A 17 inhibited by OP molecule. H chain CDR 3 is stabilized (B factor ranges between 15 and 25 Å 3) Tyr is modified P-O bond is 1. 6 Å The light chain is in grey, the heavy chain in blue, the VH-loop is in magenta and OP molecule in green. Key residues are represented as sticks with nitrogen atoms in deep blue, phosphorus in orange, and oxygen atoms in red. Hydrogen bonds are represented by gray dashes. Electron density (2 fo-fc) is represented by a blue mesh, contoured at 1. 0 sigma.
Active site of Fab. A 17 inhibited by OP molecule. H chain CDR 3 is stabilized (B factor ranges between 15 and 25 Å 3) Tyr is modified P-O bond is 1. 6 Å The light chain is in grey, the heavy chain in blue, the VH-loop is in magenta and OP molecule in green. Key residues are represented as sticks with nitrogen atoms in deep blue, phosphorus in orange, and oxygen atoms in red. Hydrogen bonds are represented by gray dashes. Electron density (2 fo-fc) is represented by a blue mesh, contoured at 1. 0 sigma.
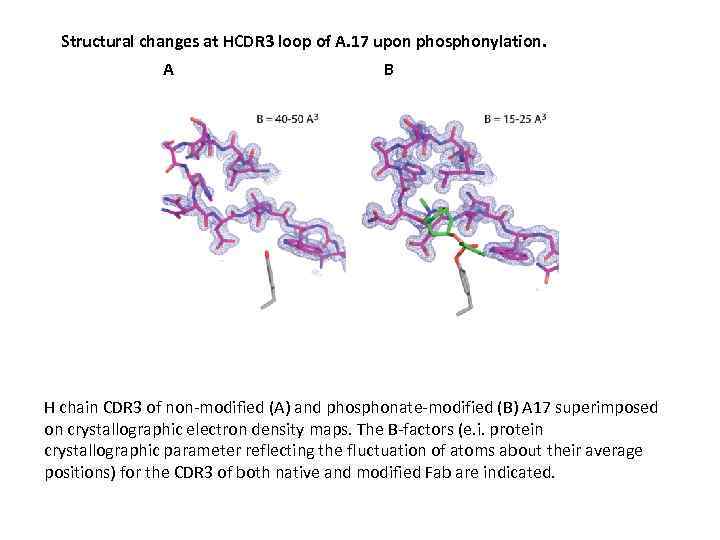 Structural changes at HCDR 3 loop of A. 17 upon phosphonylation. A B H chain CDR 3 of non-modified (A) and phosphonate-modified (B) A 17 superimposed on crystallographic electron density maps. The B-factors (e. i. protein crystallographic parameter reflecting the fluctuation of atoms about their average positions) for the CDR 3 of both native and modified Fab are indicated.
Structural changes at HCDR 3 loop of A. 17 upon phosphonylation. A B H chain CDR 3 of non-modified (A) and phosphonate-modified (B) A 17 superimposed on crystallographic electron density maps. The B-factors (e. i. protein crystallographic parameter reflecting the fluctuation of atoms about their average positions) for the CDR 3 of both native and modified Fab are indicated.
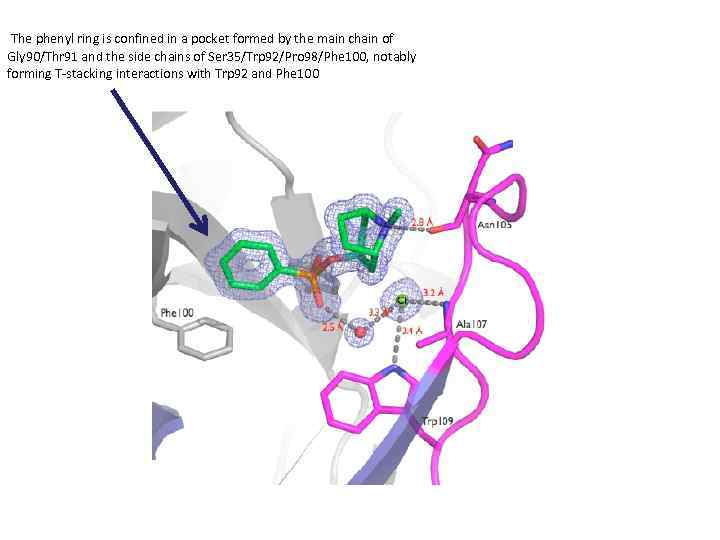 The phenyl ring is confined in a pocket formed by the main chain of Gly 90/Thr 91 and the side chains of Ser 35/Trp 92/Pro 98/Phe 100, notably forming T-stacking interactions with Trp 92 and Phe 100
The phenyl ring is confined in a pocket formed by the main chain of Gly 90/Thr 91 and the side chains of Ser 35/Trp 92/Pro 98/Phe 100, notably forming T-stacking interactions with Trp 92 and Phe 100
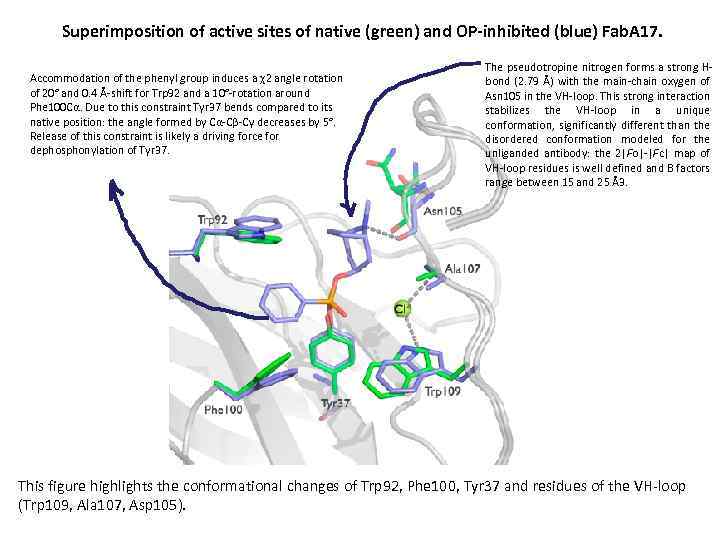 Superimposition of active sites of native (green) and OP-inhibited (blue) Fab. A 17. Accommodation of the phenyl group induces a χ2 angle rotation of 20° and 0. 4 Å-shift for Trp 92 and a 10°-rotation around Phe 100 Cα. Due to this constraint Tyr 37 bends compared to its native position: the angle formed by Cα-Cβ-Cγ decreases by 5°. Release of this constraint is likely a driving force for dephosphonylation of Tyr 37. The pseudotropine nitrogen forms a strong Hbond (2. 79 Å) with the main-chain oxygen of Asn 105 in the VH-loop. This strong interaction stabilizes the VH-loop in a unique conformation, significantly different than the disordered conformation modeled for the unliganded antibody: the 2|Fo|-|Fc| map of VH-loop residues is well defined and B factors range between 15 and 25 Å3. This figure highlights the conformational changes of Trp 92, Phe 100, Tyr 37 and residues of the VH-loop (Trp 109, Ala 107, Asp 105).
Superimposition of active sites of native (green) and OP-inhibited (blue) Fab. A 17. Accommodation of the phenyl group induces a χ2 angle rotation of 20° and 0. 4 Å-shift for Trp 92 and a 10°-rotation around Phe 100 Cα. Due to this constraint Tyr 37 bends compared to its native position: the angle formed by Cα-Cβ-Cγ decreases by 5°. Release of this constraint is likely a driving force for dephosphonylation of Tyr 37. The pseudotropine nitrogen forms a strong Hbond (2. 79 Å) with the main-chain oxygen of Asn 105 in the VH-loop. This strong interaction stabilizes the VH-loop in a unique conformation, significantly different than the disordered conformation modeled for the unliganded antibody: the 2|Fo|-|Fc| map of VH-loop residues is well defined and B factors range between 15 and 25 Å3. This figure highlights the conformational changes of Trp 92, Phe 100, Tyr 37 and residues of the VH-loop (Trp 109, Ala 107, Asp 105).
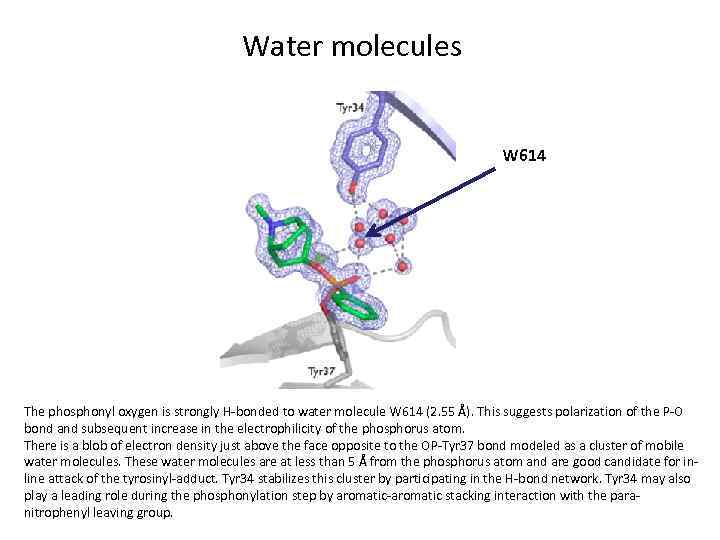 Water molecules W 614 The phosphonyl oxygen is strongly H-bonded to water molecule W 614 (2. 55 Å). This suggests polarization of the P-O bond and subsequent increase in the electrophilicity of the phosphorus atom. There is a blob of electron density just above the face opposite to the OP-Tyr 37 bond modeled as a cluster of mobile water molecules. These water molecules are at less than 5 Å from the phosphorus atom and are good candidate for inline attack of the tyrosinyl-adduct. Tyr 34 stabilizes this cluster by participating in the H-bond network. Tyr 34 may also play a leading role during the phosphonylation step by aromatic-aromatic stacking interaction with the paranitrophenyl leaving group.
Water molecules W 614 The phosphonyl oxygen is strongly H-bonded to water molecule W 614 (2. 55 Å). This suggests polarization of the P-O bond and subsequent increase in the electrophilicity of the phosphorus atom. There is a blob of electron density just above the face opposite to the OP-Tyr 37 bond modeled as a cluster of mobile water molecules. These water molecules are at less than 5 Å from the phosphorus atom and are good candidate for inline attack of the tyrosinyl-adduct. Tyr 34 stabilizes this cluster by participating in the H-bond network. Tyr 34 may also play a leading role during the phosphonylation step by aromatic-aromatic stacking interaction with the paranitrophenyl leaving group.
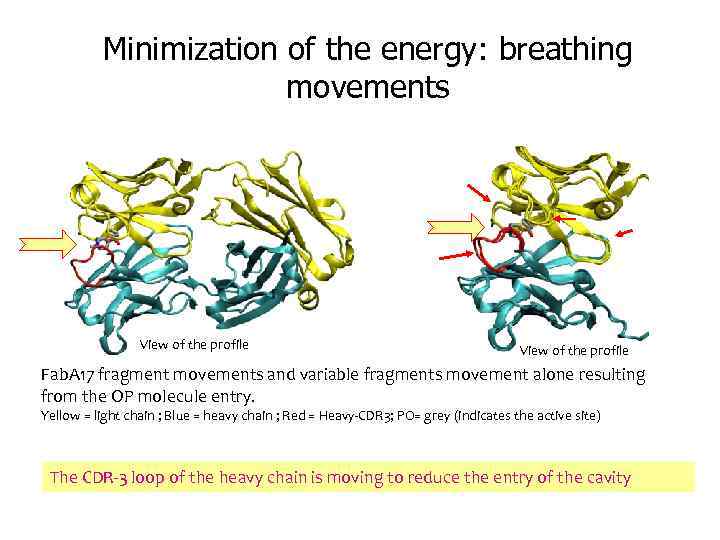 Minimization of the energy: breathing movements View of the profile Fab. A 17 fragment movements and variable fragments movement alone resulting from the OP molecule entry. Yellow = light chain ; Blue = heavy chain ; Red = Heavy-CDR 3; PO= grey (indicates the active site) The CDR-3 loop of the heavy chain is moving to reduce the entry of the cavity
Minimization of the energy: breathing movements View of the profile Fab. A 17 fragment movements and variable fragments movement alone resulting from the OP molecule entry. Yellow = light chain ; Blue = heavy chain ; Red = Heavy-CDR 3; PO= grey (indicates the active site) The CDR-3 loop of the heavy chain is moving to reduce the entry of the cavity
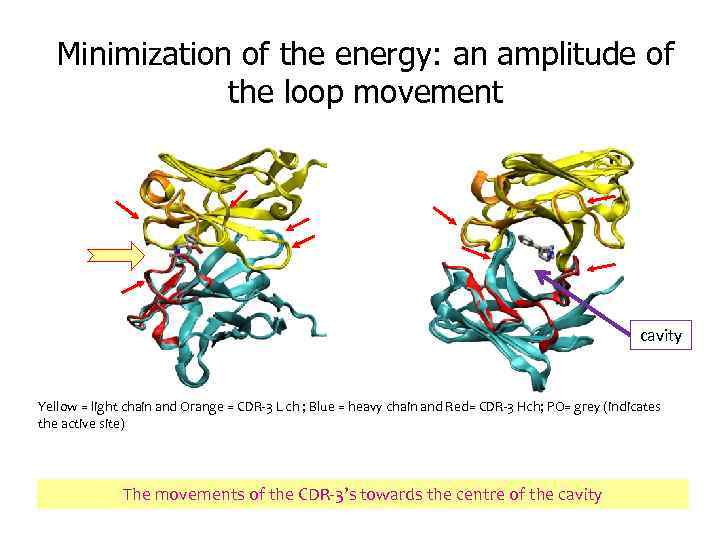 Minimization of the energy: an amplitude of the loop movement cavity Yellow = light chain and Orange = CDR-3 L ch ; Blue = heavy chain and Red= CDR-3 Hch; PO= grey (indicates the active site) The movements of the CDR-3’s towards the centre of the cavity
Minimization of the energy: an amplitude of the loop movement cavity Yellow = light chain and Orange = CDR-3 L ch ; Blue = heavy chain and Red= CDR-3 Hch; PO= grey (indicates the active site) The movements of the CDR-3’s towards the centre of the cavity
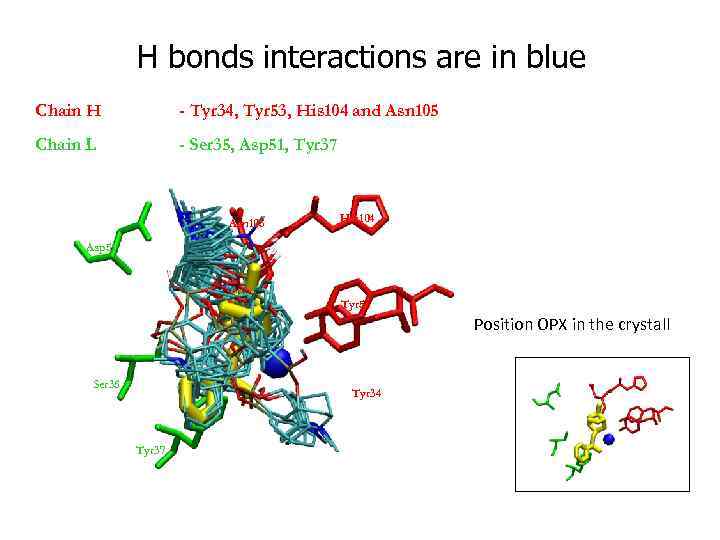 H bonds interactions are in blue Chain H - Tyr 34, Tyr 53, His 104 and Asn 105 Chain L - Ser 35, Asp 51, Tyr 37 Asn 105 His 104 Asp 51 Tyr 53 Position OPX in the crystall Ser 35 Tyr 34 Tyr 37
H bonds interactions are in blue Chain H - Tyr 34, Tyr 53, His 104 and Asn 105 Chain L - Ser 35, Asp 51, Tyr 37 Asn 105 His 104 Asp 51 Tyr 53 Position OPX in the crystall Ser 35 Tyr 34 Tyr 37
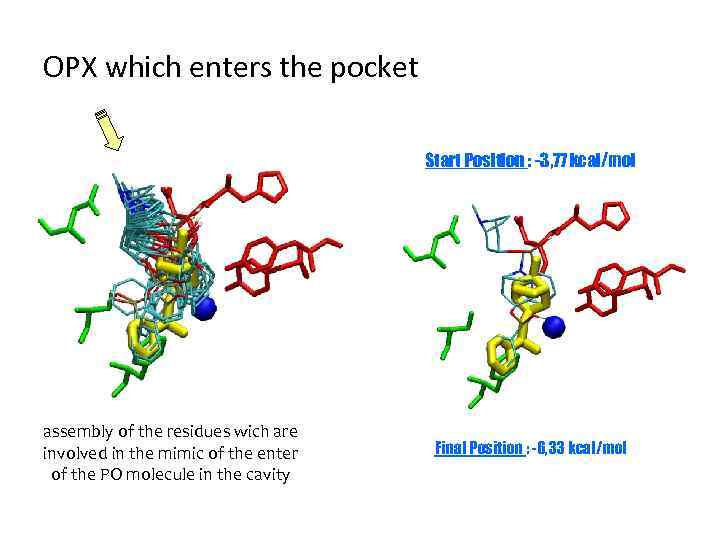 OPX which enters the pocket Start Position : -3, 77 kcal/mol assembly of the residues wich are involved in the mimic of the enter of the PO molecule in the cavity Final Position : -6, 33 kcal/mol
OPX which enters the pocket Start Position : -3, 77 kcal/mol assembly of the residues wich are involved in the mimic of the enter of the PO molecule in the cavity Final Position : -6, 33 kcal/mol
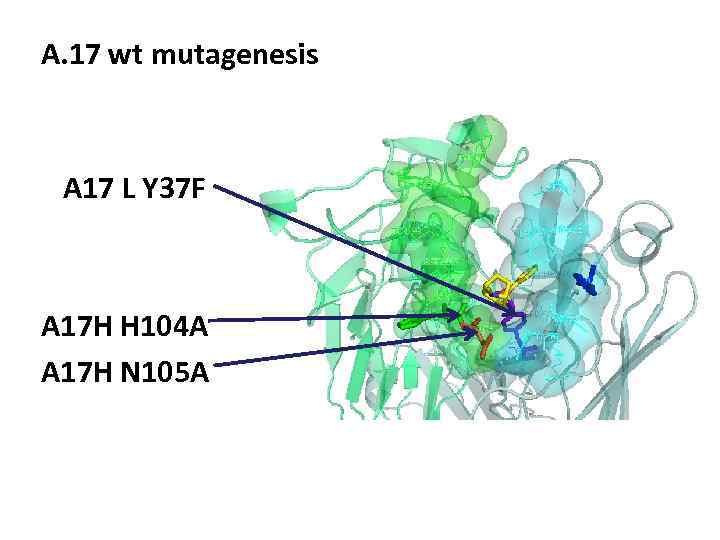 A. 17 wt mutagenesis A 17 L Y 37 F A 17 H H 104 A A 17 H N 105 A
A. 17 wt mutagenesis A 17 L Y 37 F A 17 H H 104 A A 17 H N 105 A
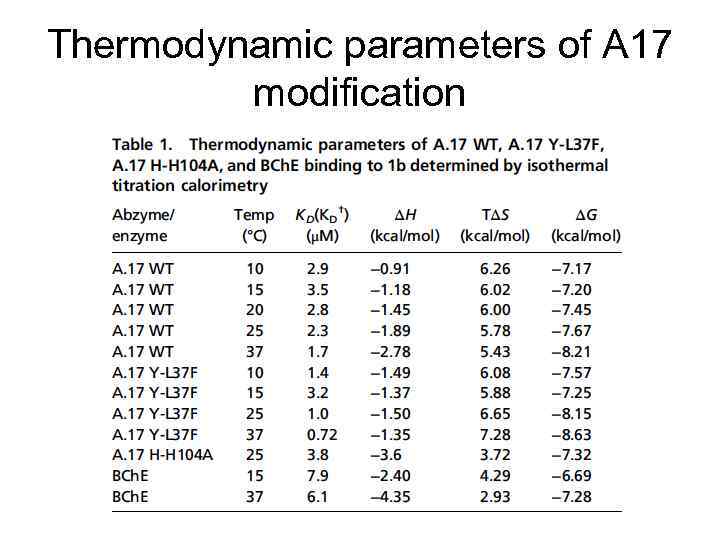 Thermodynamic parameters of A 17 modification
Thermodynamic parameters of A 17 modification
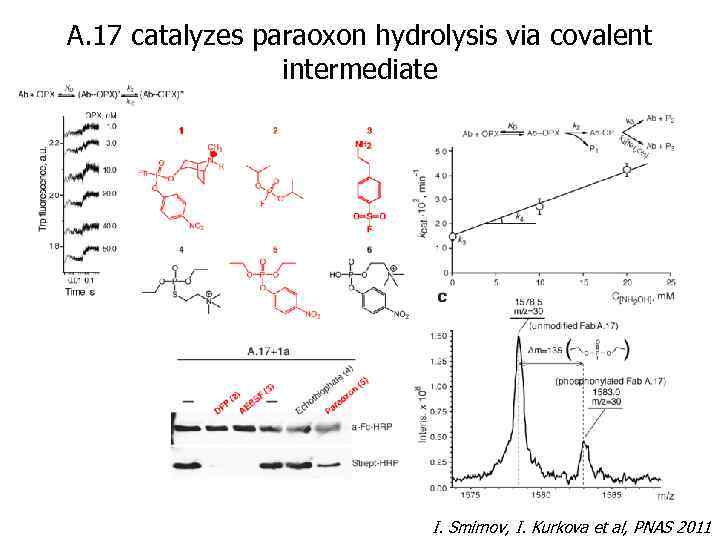 A. 17 catalyzes paraoxon hydrolysis via covalent intermediate I. Smirnov, I. Kurkova et al, PNAS 2011
A. 17 catalyzes paraoxon hydrolysis via covalent intermediate I. Smirnov, I. Kurkova et al, PNAS 2011
 Titration of active sites A 17 and Mutants with X phosphonate (Kitz-Wilson)
Titration of active sites A 17 and Mutants with X phosphonate (Kitz-Wilson)
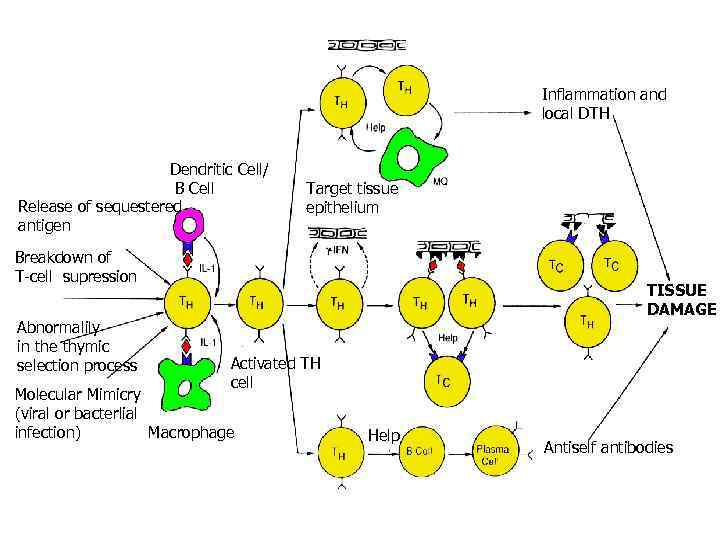 Inflammation and local DTH Dendritic Cell/ B Cell Release of sequestered antigen Target tissue epithelium Breakdown of T-cell supression Abnormalily in the thymic selection process TISSUE DAMAGE Activated TH cell Molecular Mimicry (viral or bacterlial Macrophage infection) Help Antiself antibodies
Inflammation and local DTH Dendritic Cell/ B Cell Release of sequestered antigen Target tissue epithelium Breakdown of T-cell supression Abnormalily in the thymic selection process TISSUE DAMAGE Activated TH cell Molecular Mimicry (viral or bacterlial Macrophage infection) Help Antiself antibodies
 Hypotheses ON THE INCREASED ABZYME OCCURRENCE IN AUTOIMMUNITY Expanded repertoire of underevolved antibodies may favor the emergence of abzyme activity by following possible routes: 1. Spontaneous evolution of the existing enzyme plethora. 2. Expanded repertoire can result in the increased production of anti-idiotypes. Biological mechanisms of this phenomenon are still unknown.
Hypotheses ON THE INCREASED ABZYME OCCURRENCE IN AUTOIMMUNITY Expanded repertoire of underevolved antibodies may favor the emergence of abzyme activity by following possible routes: 1. Spontaneous evolution of the existing enzyme plethora. 2. Expanded repertoire can result in the increased production of anti-idiotypes. Biological mechanisms of this phenomenon are still unknown.
 Autoimmunity and Antibody Mediated Tissue Destructions Catalytic activity of Bence Jones Proteins 1845 W. Mac. Intire recognized the first case of Multiple myeloma Henry Bence Jones discussed the peculiarities of urinary proteins Gulstonian Lectures on chemical pathology to the Royal College of Physicians in London 1962 Edelman and Gally found that light chain of plasma immunoglobulin from patients with myeloma is identical with Bence Jones urine protein from the same patient. 1974 Erhan and Greller pointed out the statistically significant amino acid identity between immunoglobulin light chain and active cite of serine proteinase 1994 Matsuura et al, 1995 Paul et al. demonstrated catalytic activity of Bence Jones protein Catalytic Activity of Bence Jones Proteins is a possible Cause of Renal Failure
Autoimmunity and Antibody Mediated Tissue Destructions Catalytic activity of Bence Jones Proteins 1845 W. Mac. Intire recognized the first case of Multiple myeloma Henry Bence Jones discussed the peculiarities of urinary proteins Gulstonian Lectures on chemical pathology to the Royal College of Physicians in London 1962 Edelman and Gally found that light chain of plasma immunoglobulin from patients with myeloma is identical with Bence Jones urine protein from the same patient. 1974 Erhan and Greller pointed out the statistically significant amino acid identity between immunoglobulin light chain and active cite of serine proteinase 1994 Matsuura et al, 1995 Paul et al. demonstrated catalytic activity of Bence Jones protein Catalytic Activity of Bence Jones Proteins is a possible Cause of Renal Failure
 Role of B cells in the pathogenesis of rheumatoid arthritis The rheumatoid factors form large antigen/antibody complexes which may enhance the process of joint destruction. The poor prognosis of rheumatoid factor-positive patients is indicative of the critical role of immunoglobulin complexes in the continuous stimulation of the immune system and thus of the inflammatory processes. Gause A, Berek C. Bio. Drugs 2001; 15(2): 73 -9 Sjogren's syndrome. i) defective apoptosis could lead to lymphoid cell accumulation and chronic inflammation in exocrine glands; ii) increased apoptosis of epithelial cells might explain the loss of secreting epithelium; and iii) orderly destruction of cellular components might induce autoantibody production. Bolstad AI, Jonsson R. Antineutrophil cytoplasmic antibodies induce human monocytes to produce oxygen radicals in vitro. Antineutrophil cytoplasmic antibodies (ANCA) are believed to play a pathogenetic role in necrotizing small-vessel vasculitis. Normal nonspecific Ig. G or their corresponding F(ab')2 fragments induced no release or very little release of oxygen radicals. Preincubation of monocytes with the Fcy receptor type II-blocking monoclonal antibody IV. 3 before addition of ANCA greatly reduced formation of oxygen radicals Weidner S, et al. Arthritis Rheum 2001 Jul; 44(7): 1698 -706 Immunopathogenic and clinical relevance of antibodies against myelin oligodendrocyte glycoprotein (MOG) in Multiple Sclerosis Immunopathogenic role of antibodies against myelin oligodendrocyte glycoprotein (MOG) and other myelin/nonmyelin targets in multiple sclerosis, as well as possible clinical implications for prognosis and therapy in the future. Berger T, Reindl M. J Neural Transm Suppl 2000; (60): 351 -60
Role of B cells in the pathogenesis of rheumatoid arthritis The rheumatoid factors form large antigen/antibody complexes which may enhance the process of joint destruction. The poor prognosis of rheumatoid factor-positive patients is indicative of the critical role of immunoglobulin complexes in the continuous stimulation of the immune system and thus of the inflammatory processes. Gause A, Berek C. Bio. Drugs 2001; 15(2): 73 -9 Sjogren's syndrome. i) defective apoptosis could lead to lymphoid cell accumulation and chronic inflammation in exocrine glands; ii) increased apoptosis of epithelial cells might explain the loss of secreting epithelium; and iii) orderly destruction of cellular components might induce autoantibody production. Bolstad AI, Jonsson R. Antineutrophil cytoplasmic antibodies induce human monocytes to produce oxygen radicals in vitro. Antineutrophil cytoplasmic antibodies (ANCA) are believed to play a pathogenetic role in necrotizing small-vessel vasculitis. Normal nonspecific Ig. G or their corresponding F(ab')2 fragments induced no release or very little release of oxygen radicals. Preincubation of monocytes with the Fcy receptor type II-blocking monoclonal antibody IV. 3 before addition of ANCA greatly reduced formation of oxygen radicals Weidner S, et al. Arthritis Rheum 2001 Jul; 44(7): 1698 -706 Immunopathogenic and clinical relevance of antibodies against myelin oligodendrocyte glycoprotein (MOG) in Multiple Sclerosis Immunopathogenic role of antibodies against myelin oligodendrocyte glycoprotein (MOG) and other myelin/nonmyelin targets in multiple sclerosis, as well as possible clinical implications for prognosis and therapy in the future. Berger T, Reindl M. J Neural Transm Suppl 2000; (60): 351 -60
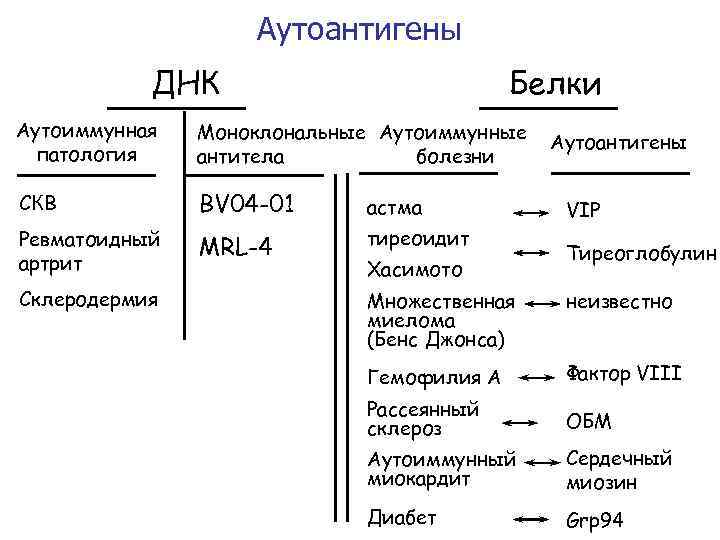 Аутоантигены ДНК Белки Аутоиммунная патология Моноклональные Аутоиммунные антитела болезни СКВ BV 04 -01 астма Ревматоидный артрит MRL-4 тиреоидит Склеродермия Хасимото Аутоантигены VIP Тиреоглобулин Множественная миелома (Бенс Джонса) неизвестно Гемофилия A Фактор VIII Рассеянный склероз ОБМ Аутоиммунный миокардит Сердечный миозин Диабет Grp 94
Аутоантигены ДНК Белки Аутоиммунная патология Моноклональные Аутоиммунные антитела болезни СКВ BV 04 -01 астма Ревматоидный артрит MRL-4 тиреоидит Склеродермия Хасимото Аутоантигены VIP Тиреоглобулин Множественная миелома (Бенс Джонса) неизвестно Гемофилия A Фактор VIII Рассеянный склероз ОБМ Аутоиммунный миокардит Сердечный миозин Диабет Grp 94
 Рассеянный склероз
Рассеянный склероз
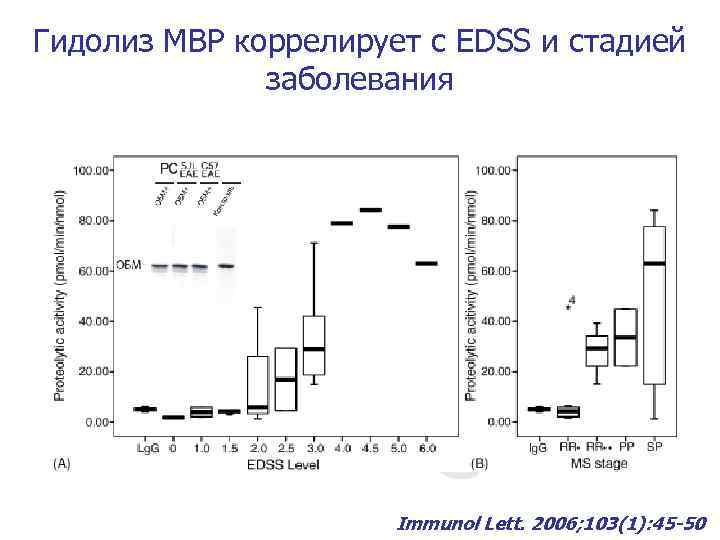 Гидолиз MBP коррелирует с EDSS и стадией заболевания Immunol Lett. 2006; 103(1): 45 -50
Гидолиз MBP коррелирует с EDSS и стадией заболевания Immunol Lett. 2006; 103(1): 45 -50
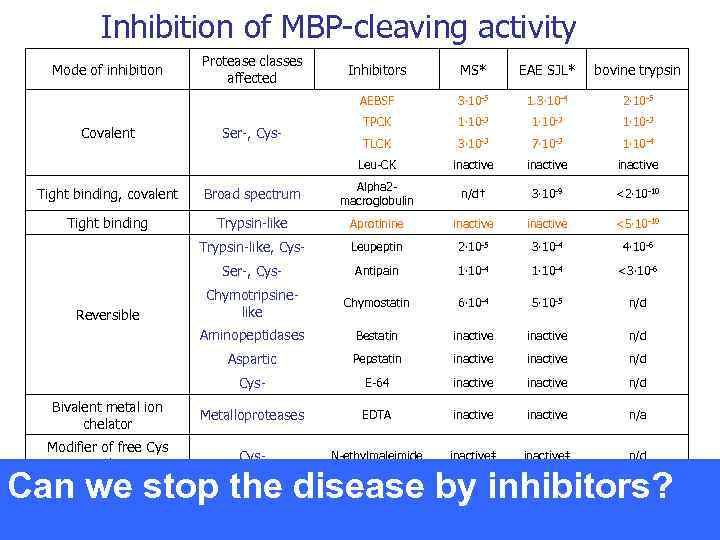 Inhibition of MBP-cleaving activity Mode of inhibition Protease classes affected EAE SJL* bovine trypsin 3∙ 10 -5 1. 3∙ 10 -4 2∙ 10 -5 TPCK 1∙ 10 -3 TLCK 3∙ 10 -3 7∙ 10 -3 1∙ 10 -4 Leu-CK Ser-, Cys- MS* AEBSF Covalent Inhibitors inactive Tight binding, covalent Broad spectrum Alpha 2 macroglobulin n/d† 3∙ 10 -9 <2∙ 10 -10 Tight binding Trypsin-like Aprotinine inactive <5∙ 10 -10 Trypsin-like, Cys- Leupeptin 2∙ 10 -5 3∙ 10 -4 4∙ 10 -6 Ser-, Cys- Antipain 1∙ 10 -4 <3∙ 10 -6 Chymotripsinelike Chymostatin 6∙ 10 -4 5∙ 10 -5 n/d Aminopeptidases Bestatin inactive n/d Aspartic Pepstatin inactive n/d Cys- E-64 inactive n/d Bivalent metal ion chelator Metalloproteases EDTA inactive n/a Modifier of free Cys residues Cys- N-ethylmaleimide inactive‡ n/d Reversible Can we stop the disease by inhibitors?
Inhibition of MBP-cleaving activity Mode of inhibition Protease classes affected EAE SJL* bovine trypsin 3∙ 10 -5 1. 3∙ 10 -4 2∙ 10 -5 TPCK 1∙ 10 -3 TLCK 3∙ 10 -3 7∙ 10 -3 1∙ 10 -4 Leu-CK Ser-, Cys- MS* AEBSF Covalent Inhibitors inactive Tight binding, covalent Broad spectrum Alpha 2 macroglobulin n/d† 3∙ 10 -9 <2∙ 10 -10 Tight binding Trypsin-like Aprotinine inactive <5∙ 10 -10 Trypsin-like, Cys- Leupeptin 2∙ 10 -5 3∙ 10 -4 4∙ 10 -6 Ser-, Cys- Antipain 1∙ 10 -4 <3∙ 10 -6 Chymotripsinelike Chymostatin 6∙ 10 -4 5∙ 10 -5 n/d Aminopeptidases Bestatin inactive n/d Aspartic Pepstatin inactive n/d Cys- E-64 inactive n/d Bivalent metal ion chelator Metalloproteases EDTA inactive n/a Modifier of free Cys residues Cys- N-ethylmaleimide inactive‡ n/d Reversible Can we stop the disease by inhibitors?
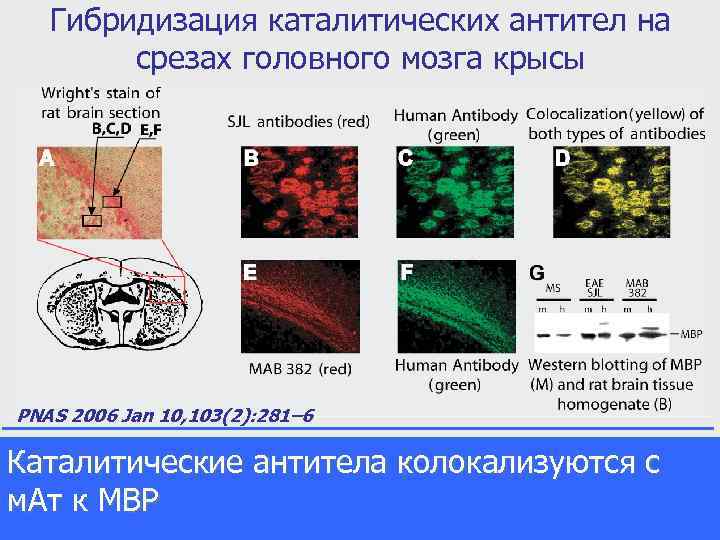 Гибридизация каталитических антител на срезах головного мозга крысы PNAS 2006 Jan 10, 103(2): 281– 6 Каталитические антитела колокализуются с м. Ат к MBP Human autoantibodies and EAE SJL antibodies recognize myelin basic protein in rat brain. A - Wright's stain of rat brain section. B, C, D - double-label immunofluorescence of EAE SJL and human ab. B - SJL antibodies (red), C - Human Antibody (green), D - colocalization (yellow) of both antibodies detected by fluorecsent microscopy. E, F - double-label immunofluorescence of MAB 382 and human ab. E - MAB 382 (red), F - human ab (green). G - Western blotting of MBP (M) and rat brain tissue homogenate (B)
Гибридизация каталитических антител на срезах головного мозга крысы PNAS 2006 Jan 10, 103(2): 281– 6 Каталитические антитела колокализуются с м. Ат к MBP Human autoantibodies and EAE SJL antibodies recognize myelin basic protein in rat brain. A - Wright's stain of rat brain section. B, C, D - double-label immunofluorescence of EAE SJL and human ab. B - SJL antibodies (red), C - Human Antibody (green), D - colocalization (yellow) of both antibodies detected by fluorecsent microscopy. E, F - double-label immunofluorescence of MAB 382 and human ab. E - MAB 382 (red), F - human ab (green). G - Western blotting of MBP (M) and rat brain tissue homogenate (B)
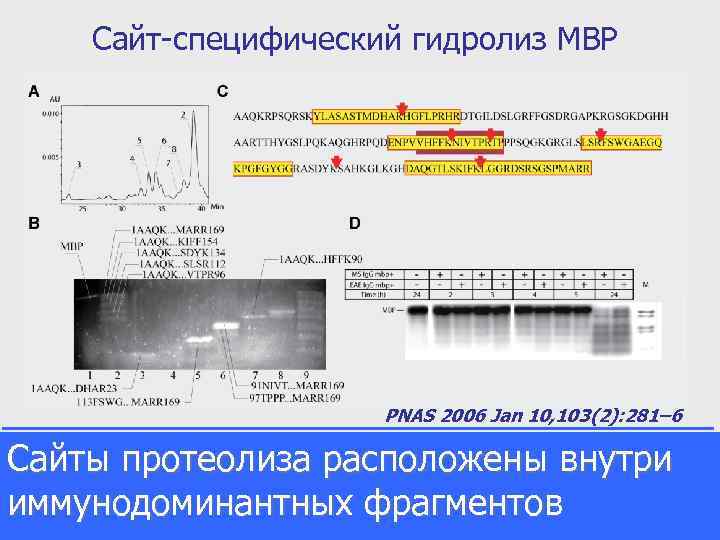 Сайт-специфический гидролиз MBP PNAS 2006 Jan 10, 103(2): 281– 6 Сайты протеолиза расположены внутри иммунодоминантных фрагментов Analysis of major MBP cleavage products. Reverse-phase HPLC-MS analysis of major MBP cleavage products. Column eluate fractions, corresponding to dominant chromatography peaks (A), were collected, freeze dried, redissolved, and applied to SELDI H 4 chip and tricine-SDSPAGE (B). Gel was stained by Sypro Orange. Peptides, unambiguously identified by SELDI and clearly seen in corresponding gel lanes, are indicated. (C) Schematic description of the preferential antibody cleavage sites in the MBP sequence. Sequence fragments identical to the immunodominant MBP-derived peptides (12– 31, 82– 98, 110– 128, and 144– 169) are shown in yellow rectangles, and the encephalitogenic peptide region (86– 98) is marked by the red box. (D) 32 P-MBP degradation by autoantibodies. Autoradiography of 32 P-phosphorylated MBP hydrolysis by proteolytic mouse (EAE SJL) and human (MS) Ig. G. Line M, molecular mass markers (range 2. 5– 16. 9 k. Da, Amersham Pharmacia).
Сайт-специфический гидролиз MBP PNAS 2006 Jan 10, 103(2): 281– 6 Сайты протеолиза расположены внутри иммунодоминантных фрагментов Analysis of major MBP cleavage products. Reverse-phase HPLC-MS analysis of major MBP cleavage products. Column eluate fractions, corresponding to dominant chromatography peaks (A), were collected, freeze dried, redissolved, and applied to SELDI H 4 chip and tricine-SDSPAGE (B). Gel was stained by Sypro Orange. Peptides, unambiguously identified by SELDI and clearly seen in corresponding gel lanes, are indicated. (C) Schematic description of the preferential antibody cleavage sites in the MBP sequence. Sequence fragments identical to the immunodominant MBP-derived peptides (12– 31, 82– 98, 110– 128, and 144– 169) are shown in yellow rectangles, and the encephalitogenic peptide region (86– 98) is marked by the red box. (D) 32 P-MBP degradation by autoantibodies. Autoradiography of 32 P-phosphorylated MBP hydrolysis by proteolytic mouse (EAE SJL) and human (MS) Ig. G. Line M, molecular mass markers (range 2. 5– 16. 9 k. Da, Amersham Pharmacia).
 Конструкция эпитопной библиотеки PNAS 2006 Jan 10, 103(2): 281– 6 Корректная презентация фрагментов MBP возможна лишь в составе слитного белка с тиоредоксином
Конструкция эпитопной библиотеки PNAS 2006 Jan 10, 103(2): 281– 6 Корректная презентация фрагментов MBP возможна лишь в составе слитного белка с тиоредоксином
 Деградация эпитопной библиотеки модельными ферментами и абзимами Journal of Immunology 2007 (in press)
Деградация эпитопной библиотеки модельными ферментами и абзимами Journal of Immunology 2007 (in press)
 Метод измерения специфической абзиматической активности на основе FRET-пары Journal of Immunology 2008
Метод измерения специфической абзиматической активности на основе FRET-пары Journal of Immunology 2008
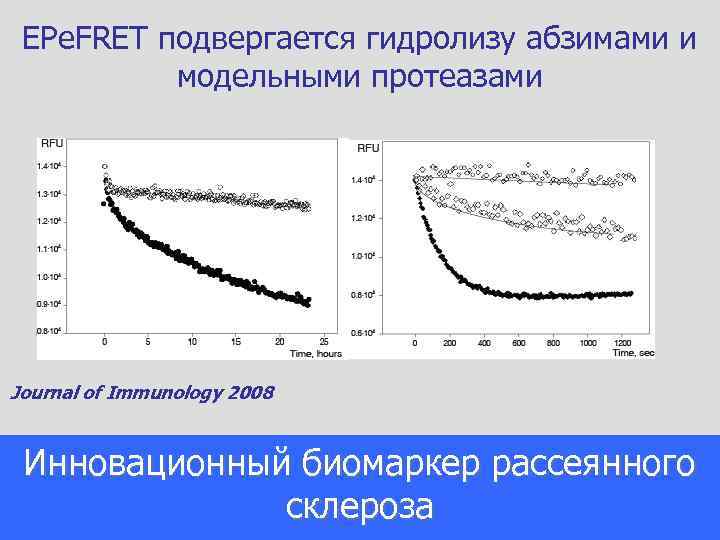 EPe. FRET подвергается гидролизу абзимами и модельными протеазами Journal of Immunology 2008 Инновационный биомаркер рассеянного склероза
EPe. FRET подвергается гидролизу абзимами и модельными протеазами Journal of Immunology 2008 Инновационный биомаркер рассеянного склероза
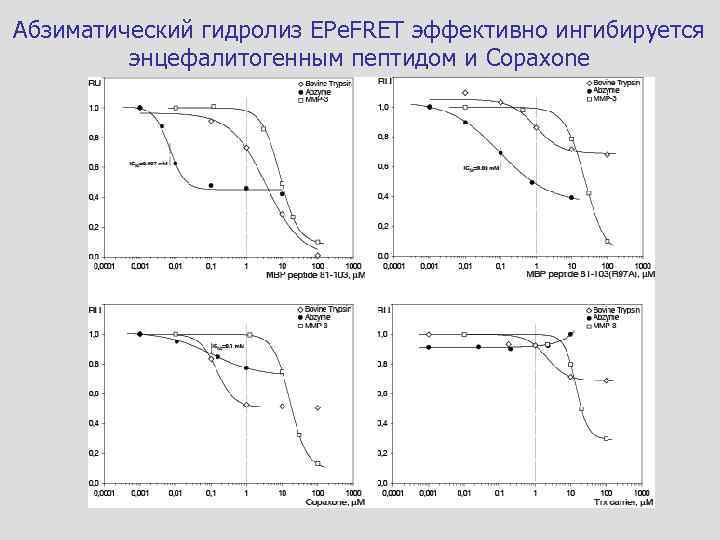 Абзиматический гидролиз EPe. FRET эффективно ингибируется энцефалитогенным пептидом и Copaxone
Абзиматический гидролиз EPe. FRET эффективно ингибируется энцефалитогенным пептидом и Copaxone
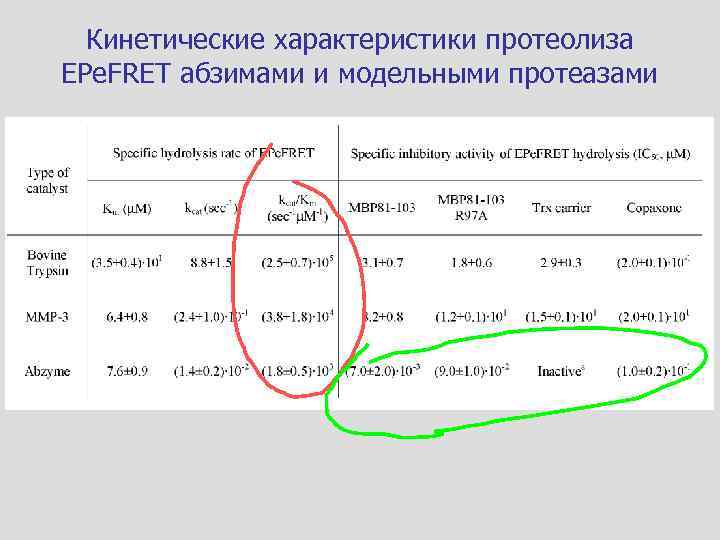 Кинетические характеристики протеолиза EPe. FRET абзимами и модельными протеазами
Кинетические характеристики протеолиза EPe. FRET абзимами и модельными протеазами
 Паттерн связывания аутоантител при PC и других нейродегенеративных заболеваниях с эпитопной библиотекой MBP Новые подходы к дифференциальной диагностике РС
Паттерн связывания аутоантител при PC и других нейродегенеративных заболеваниях с эпитопной библиотекой MBP Новые подходы к дифференциальной диагностике РС
 Паттерн связывания аутоантител с эпитопной библиотекой MBP при РС и различных его животных моделях
Паттерн связывания аутоантител с эпитопной библиотекой MBP при РС и различных его животных моделях
 Каталитические антитела к gp 120 в качестве терапии ВИЧ Разрушение gp 120 абзимами, пассивная вакцинация Мутация вариабельных регионов Протеолитическое разрушение молекулы gp 120 приводит к ее поверхностной реорганизации. Это может помочь иммунной системе бороться с ВИЧ.
Каталитические антитела к gp 120 в качестве терапии ВИЧ Разрушение gp 120 абзимами, пассивная вакцинация Мутация вариабельных регионов Протеолитическое разрушение молекулы gp 120 приводит к ее поверхностной реорганизации. Это может помочь иммунной системе бороться с ВИЧ.
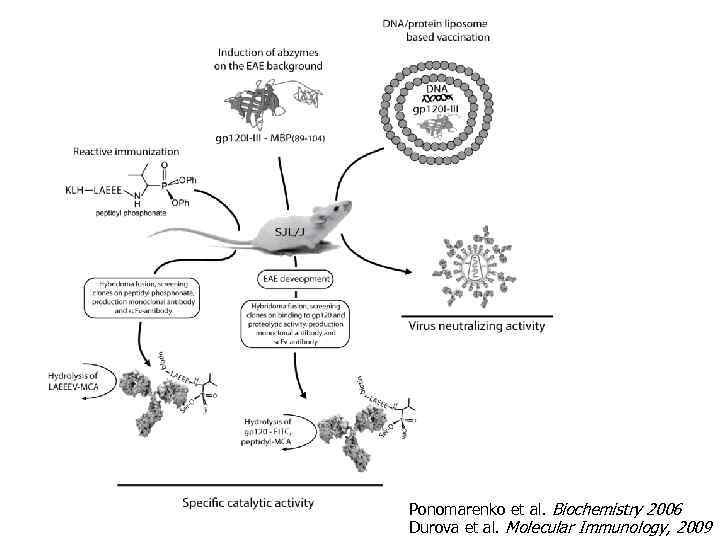 Ponomarenko et al. Biochemistry 2006 Durova et al. Molecular Immunology, 2009
Ponomarenko et al. Biochemistry 2006 Durova et al. Molecular Immunology, 2009
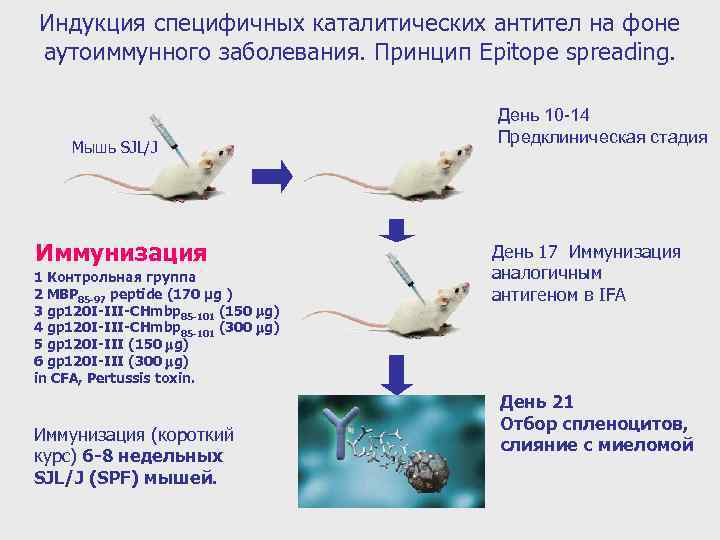 Индукция специфичных каталитических антител на фоне аутоиммунного заболевания. Принцип Epitope spreading. Мышь SJL/J Иммунизация 1 Контрольная группа 2 MBP 85 -97 peptide (170 μg ) 3 gp 120 I-III-CHmbp 85 -101 (150 mg) 4 gp 120 I-III-CHmbp 85 -101 (300 mg) 5 gp 120 I-III (150 mg) 6 gp 120 I-III (300 mg) in CFA, Pertussis toxin. Иммунизация (короткий курс) 6 -8 недельных SJL/J (SPF) мышей. День 10 -14 Предклиническая стадия День 17 Иммунизация аналогичным антигеном в IFA День 21 Отбор спленоцитов, слияние с миеломой
Индукция специфичных каталитических антител на фоне аутоиммунного заболевания. Принцип Epitope spreading. Мышь SJL/J Иммунизация 1 Контрольная группа 2 MBP 85 -97 peptide (170 μg ) 3 gp 120 I-III-CHmbp 85 -101 (150 mg) 4 gp 120 I-III-CHmbp 85 -101 (300 mg) 5 gp 120 I-III (150 mg) 6 gp 120 I-III (300 mg) in CFA, Pertussis toxin. Иммунизация (короткий курс) 6 -8 недельных SJL/J (SPF) мышей. День 10 -14 Предклиническая стадия День 17 Иммунизация аналогичным антигеном в IFA День 21 Отбор спленоцитов, слияние с миеломой
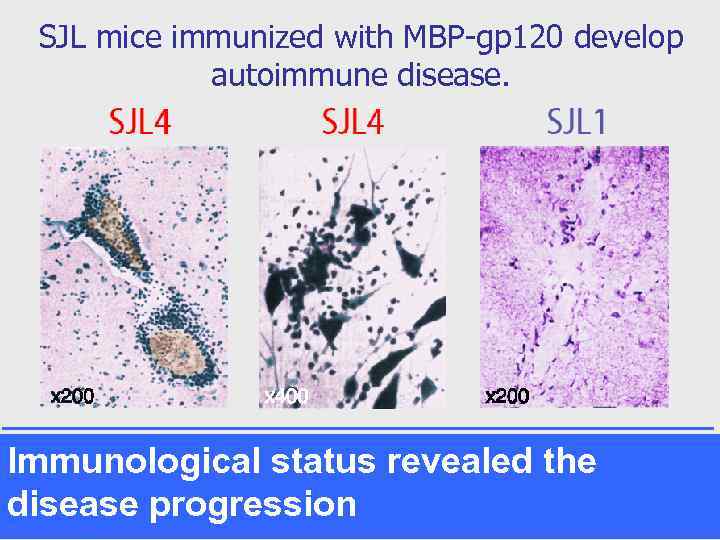 SJL mice immunized with MBP-gp 120 develop autoimmune disease. Immunological status revealed the disease progression
SJL mice immunized with MBP-gp 120 develop autoimmune disease. Immunological status revealed the disease progression
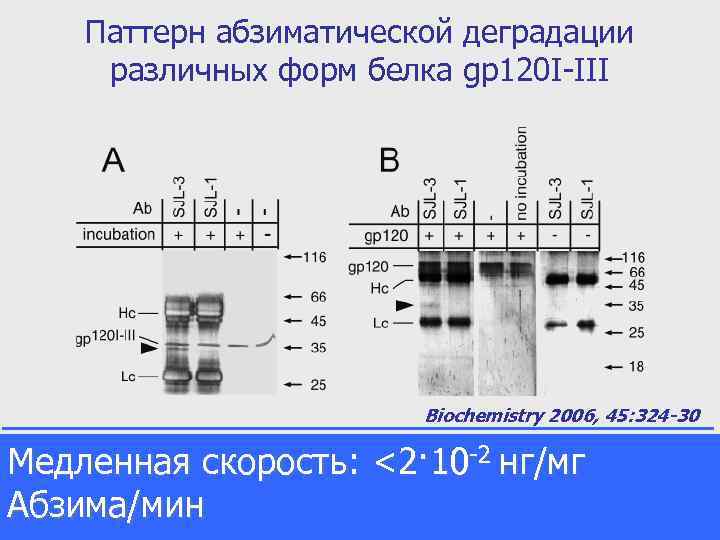 Паттерн абзиматической деградации различных форм белка gp 120 I-III Biochemistry 2006, 45: 324 -30 Proteolytic degradation pattern of Медленная of gp 120 I-IIInon-glycosilated (A) and glycosilated baculovirusскорость: Antibodies heavy нг/мг chains, <2· 10 -2 chains, light expressed (B) forms protein. gp 120 I-III and it`s degradation products are marked as Hc, Lc, gp and arrow sign, respectively. Абзима/мин
Паттерн абзиматической деградации различных форм белка gp 120 I-III Biochemistry 2006, 45: 324 -30 Proteolytic degradation pattern of Медленная of gp 120 I-IIInon-glycosilated (A) and glycosilated baculovirusскорость: Antibodies heavy нг/мг chains, <2· 10 -2 chains, light expressed (B) forms protein. gp 120 I-III and it`s degradation products are marked as Hc, Lc, gp and arrow sign, respectively. Абзима/мин
 Сайт-специфический гидролиз gp 120 был показан с помощью SELDI MS Абзим -P 484 -LGVAPTKAKLDPHHHHHHGSGEQKLISEEDLNSSSVDKLAAALEHHHHHH Приблизительная масса 5528 Да Biochemistry 2006, 45: 324 -30
Сайт-специфический гидролиз gp 120 был показан с помощью SELDI MS Абзим -P 484 -LGVAPTKAKLDPHHHHHHGSGEQKLISEEDLNSSSVDKLAAALEHHHHHH Приблизительная масса 5528 Да Biochemistry 2006, 45: 324 -30
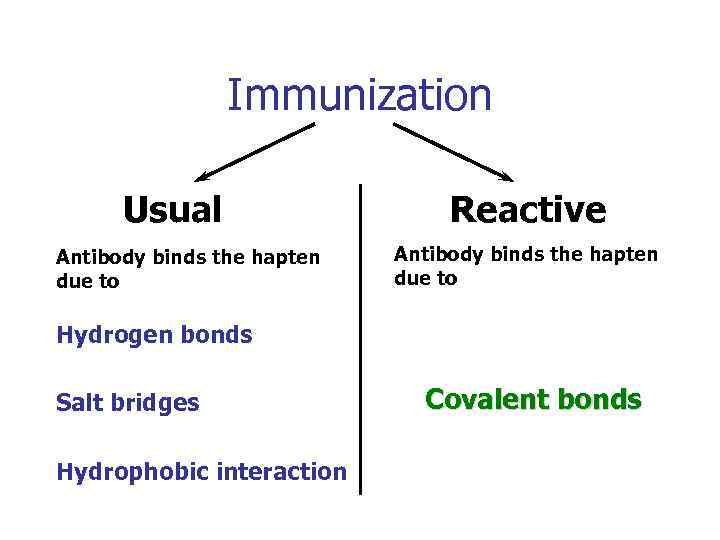 Immunization Usual Antibody binds the hapten due to Reactive Antibody binds the hapten due to Hydrogen bonds Salt bridges Hydrophobic interaction Covalent bonds
Immunization Usual Antibody binds the hapten due to Reactive Antibody binds the hapten due to Hydrogen bonds Salt bridges Hydrophobic interaction Covalent bonds
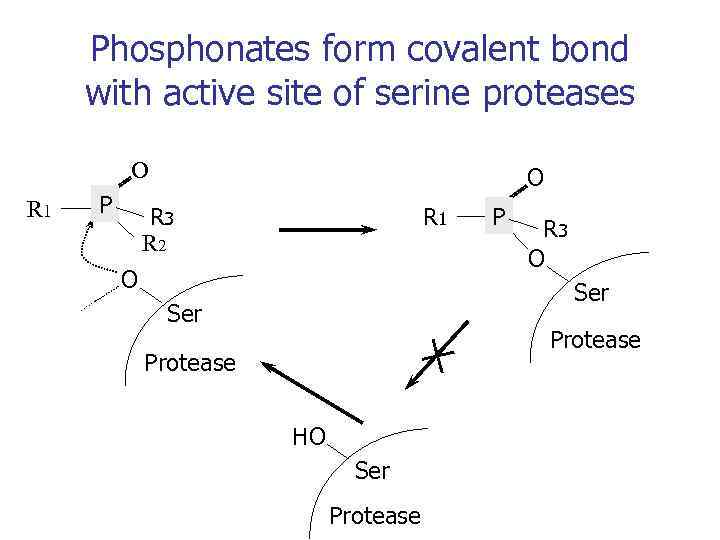 Phosphonates form covalent bond with active site of serine proteases O R 1 P C O R 3 R 2 R 1 O P C R 3 O Ser Protease HO Ser Protease
Phosphonates form covalent bond with active site of serine proteases O R 1 P C O R 3 R 2 R 1 O P C R 3 O Ser Protease HO Ser Protease
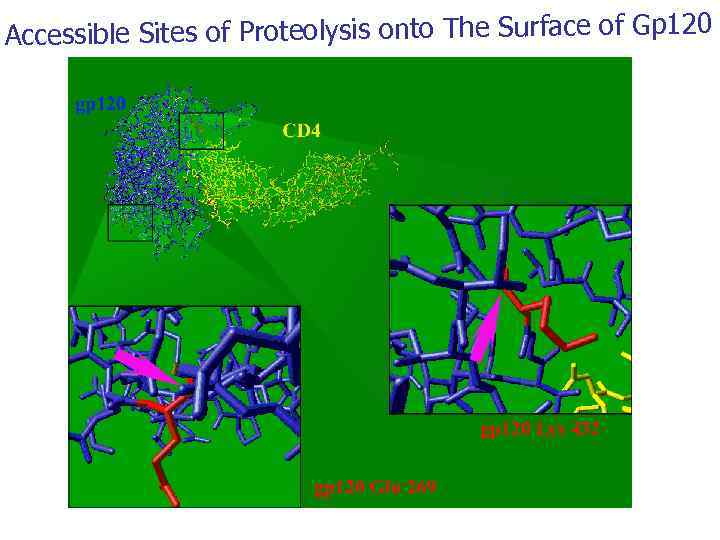 Accessible Sites of Proteolysis onto The Surface of Gp 120
Accessible Sites of Proteolysis onto The Surface of Gp 120
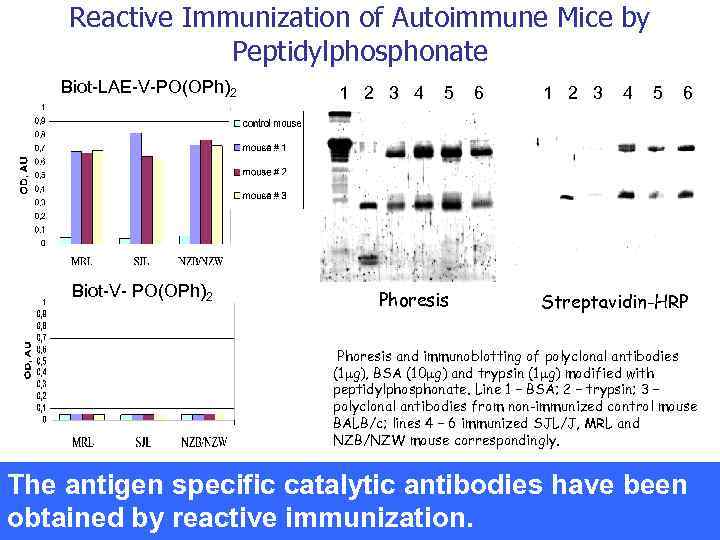 Reactive Immunization of Autoimmune Mice by Peptidylphosphonate Biot-LAE-V-PO(OPh)2 Biot-V- PO(OPh)2 1 2 3 4 5 Phoresis 6 1 2 3 4 5 6 Streptavidin-HRP Phoresis and immunoblotting of polyclonal antibodies (1 g), BSA (10 g) and trypsin (1 g) modified with peptidylphosphonate. Line 1 – BSA; 2 – trypsin; 3 – polyclonal antibodies from non-immunized control mouse BALB/c; lines 4 – 6 immunized SJL/J, MRL and NZB/NZW mouse correspondingly. The antigen specific catalytic antibodies have been obtained by reactive immunization.
Reactive Immunization of Autoimmune Mice by Peptidylphosphonate Biot-LAE-V-PO(OPh)2 Biot-V- PO(OPh)2 1 2 3 4 5 Phoresis 6 1 2 3 4 5 6 Streptavidin-HRP Phoresis and immunoblotting of polyclonal antibodies (1 g), BSA (10 g) and trypsin (1 g) modified with peptidylphosphonate. Line 1 – BSA; 2 – trypsin; 3 – polyclonal antibodies from non-immunized control mouse BALB/c; lines 4 – 6 immunized SJL/J, MRL and NZB/NZW mouse correspondingly. The antigen specific catalytic antibodies have been obtained by reactive immunization.
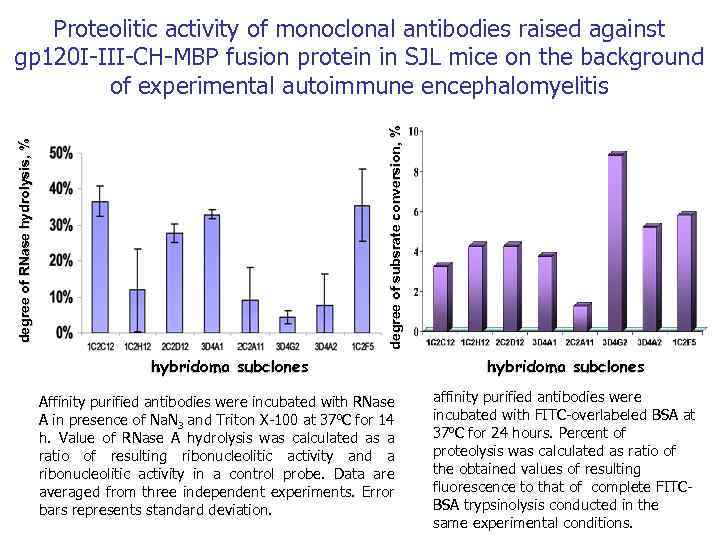 degree of RNase hydrolysis, % degree of subsrate conversion, % Proteolitic activity of monoclonal antibodies raised against gp 120 I-III-CH-MBP fusion protein in SJL mice on the background of experimental autoimmune encephalomyelitis hybridoma subclones Affinity purified antibodies were incubated with RNase A in presence of Na. N 3 and Triton X-100 at 37 o. C for 14 h. Value of RNase A hydrolysis was calculated as a ratio of resulting ribonucleolitic activity and a ribonucleolitic activity in a control probe. Data are averaged from three independent experiments. Error bars represents standard deviation. hybridoma subclones affinity purified antibodies were incubated with FITC-overlabeled BSA at 37 o. C for 24 hours. Percent of proteolysis was calculated as ratio of the obtained values of resulting fluorescence to that of complete FITCBSA trypsinolysis conducted in the same experimental conditions.
degree of RNase hydrolysis, % degree of subsrate conversion, % Proteolitic activity of monoclonal antibodies raised against gp 120 I-III-CH-MBP fusion protein in SJL mice on the background of experimental autoimmune encephalomyelitis hybridoma subclones Affinity purified antibodies were incubated with RNase A in presence of Na. N 3 and Triton X-100 at 37 o. C for 14 h. Value of RNase A hydrolysis was calculated as a ratio of resulting ribonucleolitic activity and a ribonucleolitic activity in a control probe. Data are averaged from three independent experiments. Error bars represents standard deviation. hybridoma subclones affinity purified antibodies were incubated with FITC-overlabeled BSA at 37 o. C for 24 hours. Percent of proteolysis was calculated as ratio of the obtained values of resulting fluorescence to that of complete FITCBSA trypsinolysis conducted in the same experimental conditions.
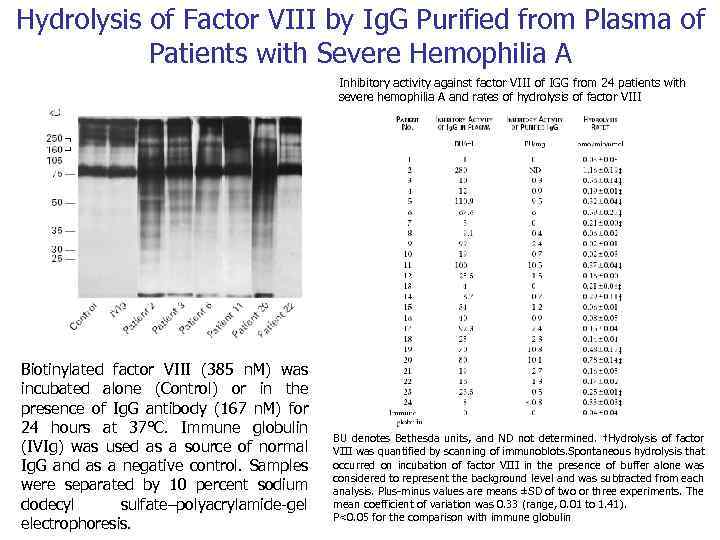 Hydrolysis of Factor VIII by Ig. G Purified from Plasma of Patients with Severe Hemophilia A Inhibitory activity against factor VIII of IGG from 24 patients with severe hemophilia A and rates of hydrolysis of factor VIII Biotinylated factor VIII (385 n. M) was incubated alone (Control) or in the presence of Ig. G antibody (167 n. M) for 24 hours at 37°C. Immune globulin (IVIg) was used as a source of normal Ig. G and as a negative control. Samples were separated by 10 percent sodium dodecyl sulfate–polyacrylamide-gel electrophoresis. BU denotes Bethesda units, and ND not determined. †Hydrolysis of factor VIII was quantified by scanning of immunoblots. Spontaneous hydrolysis that occurred on incubation of factor VIII in the presence of buffer alone was considered to represent the background level and was subtracted from each analysis. Plus-minus values are means ±SD of two or three experiments. The mean coefficient of variation was 0. 33 (range, 0. 01 to 1. 41). P<0. 05 for the comparison with immune globulin
Hydrolysis of Factor VIII by Ig. G Purified from Plasma of Patients with Severe Hemophilia A Inhibitory activity against factor VIII of IGG from 24 patients with severe hemophilia A and rates of hydrolysis of factor VIII Biotinylated factor VIII (385 n. M) was incubated alone (Control) or in the presence of Ig. G antibody (167 n. M) for 24 hours at 37°C. Immune globulin (IVIg) was used as a source of normal Ig. G and as a negative control. Samples were separated by 10 percent sodium dodecyl sulfate–polyacrylamide-gel electrophoresis. BU denotes Bethesda units, and ND not determined. †Hydrolysis of factor VIII was quantified by scanning of immunoblots. Spontaneous hydrolysis that occurred on incubation of factor VIII in the presence of buffer alone was considered to represent the background level and was subtracted from each analysis. Plus-minus values are means ±SD of two or three experiments. The mean coefficient of variation was 0. 33 (range, 0. 01 to 1. 41). P<0. 05 for the comparison with immune globulin
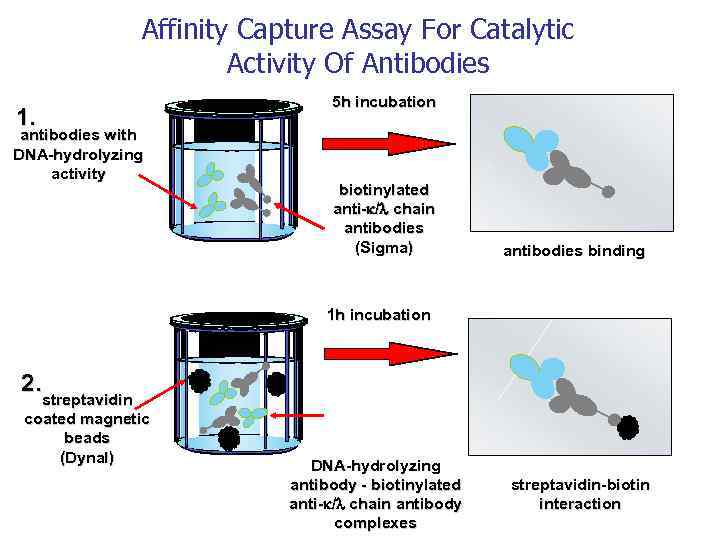 Affinity Capture Assay For Catalytic Activity Of Antibodies 1. antibodies with DNA-hydrolyzing activity 5 h incubation biotinylated anti- / chain antibodies (Sigma) antibodies binding 1 h incubation 2. streptavidin coated magnetic beads (Dynal) DNA-hydrolyzing antibody - biotinylated anti- / chain antibody complexes streptavidin-biotin interaction
Affinity Capture Assay For Catalytic Activity Of Antibodies 1. antibodies with DNA-hydrolyzing activity 5 h incubation biotinylated anti- / chain antibodies (Sigma) antibodies binding 1 h incubation 2. streptavidin coated magnetic beads (Dynal) DNA-hydrolyzing antibody - biotinylated anti- / chain antibody complexes streptavidin-biotin interaction
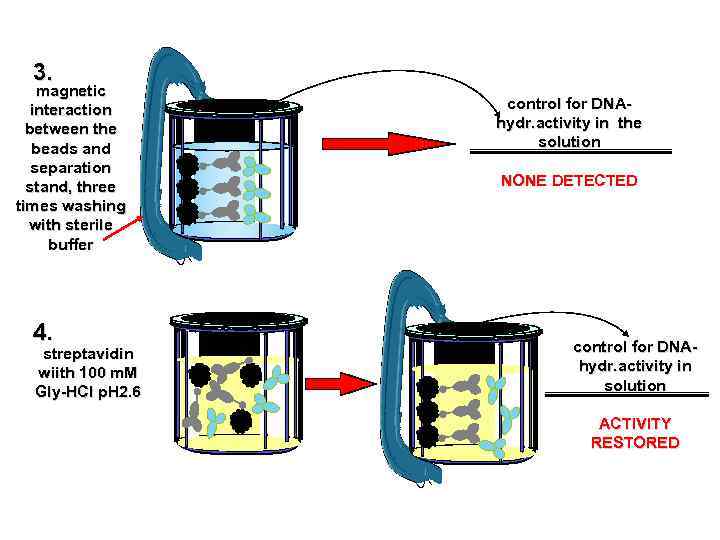 3. magnetic interaction between the beads and separation stand, three times washing with sterile buffer 4. streptavidin wiith 100 m. M Gly-HCl p. H 2. 6 control for DNAhydr. activity in the solution NONE DETECTED control for DNAhydr. activity in solution ACTIVITY RESTORED
3. magnetic interaction between the beads and separation stand, three times washing with sterile buffer 4. streptavidin wiith 100 m. M Gly-HCl p. H 2. 6 control for DNAhydr. activity in the solution NONE DETECTED control for DNAhydr. activity in solution ACTIVITY RESTORED
 Possible Ways Of Dna Cleavage nucleotide 3’ 3’ P P 5’ 5’ DNA-hydrolyzing Abs, Nuclease S 1, Phosphodiesterase Micrococcal nuclease
Possible Ways Of Dna Cleavage nucleotide 3’ 3’ P P 5’ 5’ DNA-hydrolyzing Abs, Nuclease S 1, Phosphodiesterase Micrococcal nuclease
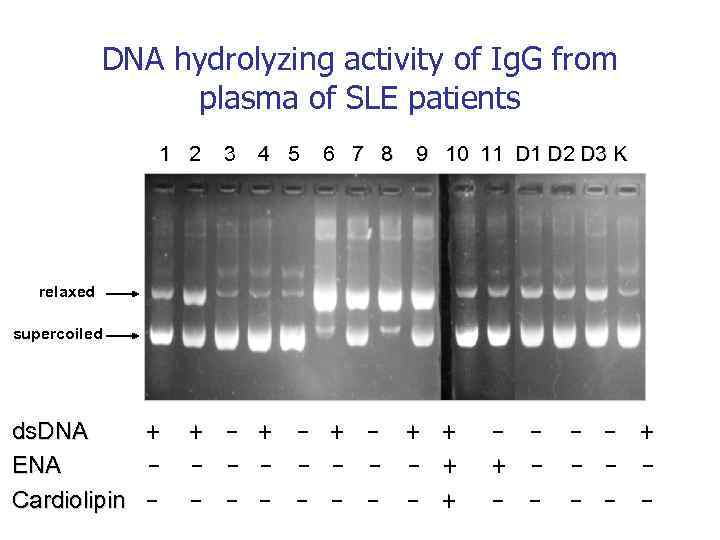 DNA hydrolyzing activity of Ig. G from plasma of SLE patients 1 2 3 4 5 6 7 8 9 10 11 D 2 D 3 K relaxed supercoiled ds. DNA + ENA Cardiolipin - + - + + - - - - + - - -
DNA hydrolyzing activity of Ig. G from plasma of SLE patients 1 2 3 4 5 6 7 8 9 10 11 D 2 D 3 K relaxed supercoiled ds. DNA + ENA Cardiolipin - + - + + - - - - + - - -
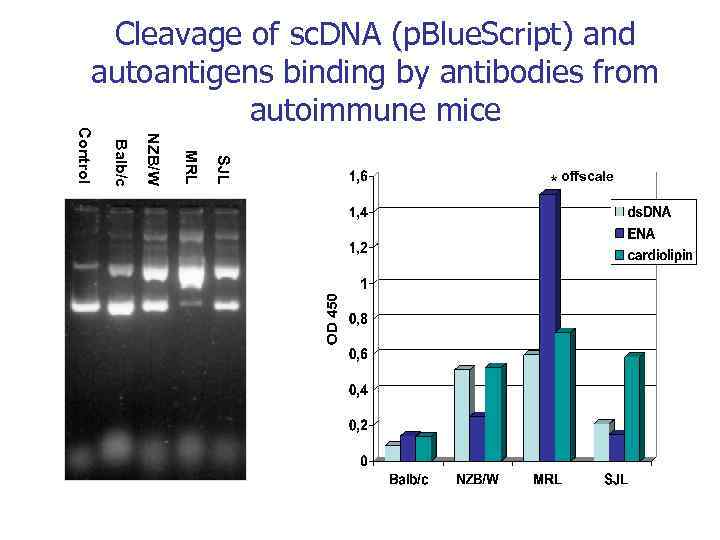 SJL MRL NZB/W Balb/c Control Cleavage of sc. DNA (p. Blue. Script) and autoantigens binding by antibodies from autoimmune mice * offscale
SJL MRL NZB/W Balb/c Control Cleavage of sc. DNA (p. Blue. Script) and autoantigens binding by antibodies from autoimmune mice * offscale
 • MESSAGE Basically ALL Autoantigens may serve as substrates for autoantibodies. Shuster et al. Science, 1992 Kolesnikov et al. PNAS, 2000 Ponomarenko et al. Meth. Immunol. , 2002 Kozyr et al. Imm. Lett. , 2002 Ponomarenko et al. PNAS, 2006 Ponomarenko et al. Biochemistry, 2007 Belogurov et al. J. Immunology, 2008 Durova et al. Molecular Immunology, 2009 Belogurov et al. Autoimmunity, 2009 A. Belogurov Jr. et al, Bio. Essays 2009
• MESSAGE Basically ALL Autoantigens may serve as substrates for autoantibodies. Shuster et al. Science, 1992 Kolesnikov et al. PNAS, 2000 Ponomarenko et al. Meth. Immunol. , 2002 Kozyr et al. Imm. Lett. , 2002 Ponomarenko et al. PNAS, 2006 Ponomarenko et al. Biochemistry, 2007 Belogurov et al. J. Immunology, 2008 Durova et al. Molecular Immunology, 2009 Belogurov et al. Autoimmunity, 2009 A. Belogurov Jr. et al, Bio. Essays 2009
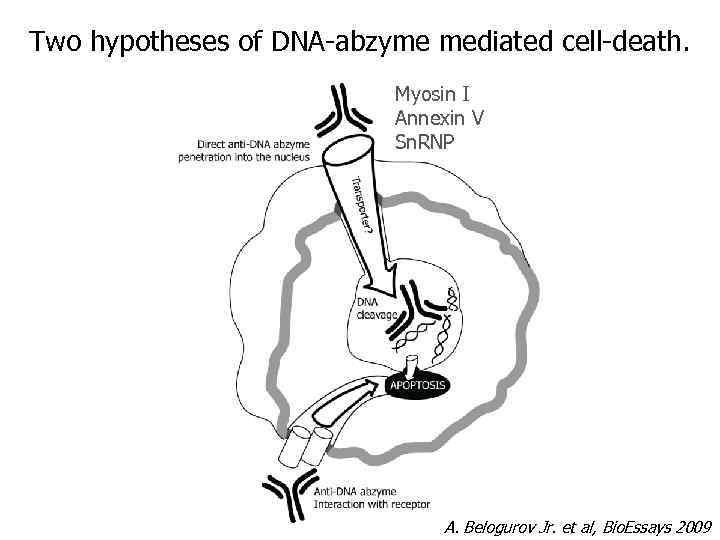 Two hypotheses of DNA-abzyme mediated cell-death. Myosin I Annexin V Sn. RNP A. Belogurov Jr. et al, Bio. Essays 2009
Two hypotheses of DNA-abzyme mediated cell-death. Myosin I Annexin V Sn. RNP A. Belogurov Jr. et al, Bio. Essays 2009
 Anti-DNA Autoantibodies Frequency In Several Diseases Disease DNA-binding Abs frequency DNA-hydrolyzing Abs frequency SLE +++ RA ++ ++ Scleroderma AIDS (early stage) +/- +/++ +/- AIDS (late stage) ++ ++ Healthy donors +/- - - CLL Gammaglobulinemia +++ - more than in 30% cases analyzed ++ - 15 -30% - - never detected + - 5 -15% +/- - 0 -5%
Anti-DNA Autoantibodies Frequency In Several Diseases Disease DNA-binding Abs frequency DNA-hydrolyzing Abs frequency SLE +++ RA ++ ++ Scleroderma AIDS (early stage) +/- +/++ +/- AIDS (late stage) ++ ++ Healthy donors +/- - - CLL Gammaglobulinemia +++ - more than in 30% cases analyzed ++ - 15 -30% - - never detected + - 5 -15% +/- - 0 -5%
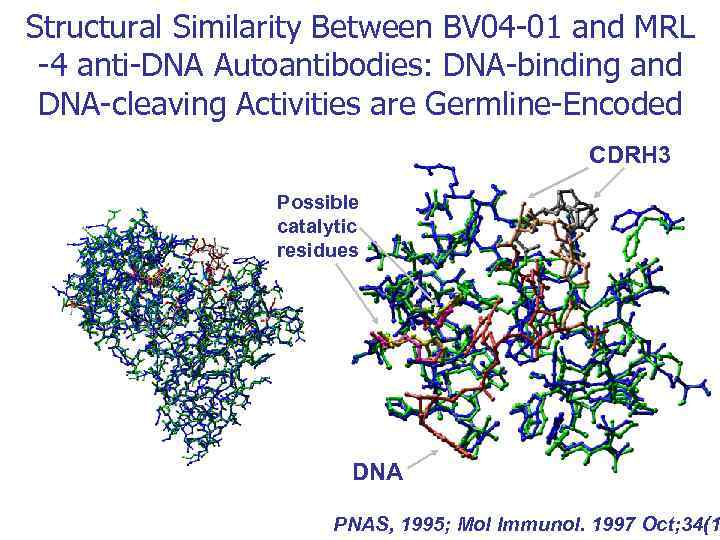 Structural Similarity Between BV 04 -01 and MRL -4 anti-DNA Autoantibodies: DNA-binding and DNA-cleaving Activities are Germline-Encoded CDRH 3 Possible catalytic residues DNA PNAS, 1995; Mol Immunol. 1997 Oct; 34(1
Structural Similarity Between BV 04 -01 and MRL -4 anti-DNA Autoantibodies: DNA-binding and DNA-cleaving Activities are Germline-Encoded CDRH 3 Possible catalytic residues DNA PNAS, 1995; Mol Immunol. 1997 Oct; 34(1
 Catalytic activity of mutants of DNA-abzyme Sca Variant Km k cat /Km -8 2. 5 x 10 M L 32 Tyr/L 27 d. His (WT) 1. 3 x 10 -2 min-1 5. 2 x 105 min-1 M-1 L 32 Phe/L 27 d. His -9 6. 0 x 10 M 1. 1 x 10 -3 min-1 1. 8 x 105 min-1 M-1 L 32 Ala/L 27 d. His 2. 4 x 10 -7 M 1. 0 x 10 -1 min-1 4. 2 x 105 min-1 M-1 L 32 Tyr/L 27 d. Ala 4. 1 x 10 -8 M 4. 9 x 10 -4 min-1 1. 2 x 104 min-1 M-1 Site-directed mutagenesis shows that DNA-abzyme participates in the chemical
Catalytic activity of mutants of DNA-abzyme Sca Variant Km k cat /Km -8 2. 5 x 10 M L 32 Tyr/L 27 d. His (WT) 1. 3 x 10 -2 min-1 5. 2 x 105 min-1 M-1 L 32 Phe/L 27 d. His -9 6. 0 x 10 M 1. 1 x 10 -3 min-1 1. 8 x 105 min-1 M-1 L 32 Ala/L 27 d. His 2. 4 x 10 -7 M 1. 0 x 10 -1 min-1 4. 2 x 105 min-1 M-1 L 32 Tyr/L 27 d. Ala 4. 1 x 10 -8 M 4. 9 x 10 -4 min-1 1. 2 x 104 min-1 M-1 Site-directed mutagenesis shows that DNA-abzyme participates in the chemical
 Перспективы: создание медленных биокатализаторов in vivo IL 6 ? ? ? Ab P gp 120 TNF-a
Перспективы: создание медленных биокатализаторов in vivo IL 6 ? ? ? Ab P gp 120 TNF-a
 Der Mensch als Industriepalast (Man as Industrial Palace) Stuttgart, 1926. Chromolithograph. National Library of Medicine. Fritz Kahn (1888 -1968) Kahn’s modernist visualization of the digestive and respiratory system as "industrial palace, " really a chemical plant
Der Mensch als Industriepalast (Man as Industrial Palace) Stuttgart, 1926. Chromolithograph. National Library of Medicine. Fritz Kahn (1888 -1968) Kahn’s modernist visualization of the digestive and respiratory system as "industrial palace, " really a chemical plant
 Институт биоорганической химии РАН им. Академиков М. М. Шемякина и Ю. А. Овчинникова Авторы: Александр Габибов, Алексей Белогуров, Иван Смирнов, Андрей Решетняк, Инна Куркова, Мария Захарова, Иван Воробьев, Оксана Дурова, Наталья Пономаренко.
Институт биоорганической химии РАН им. Академиков М. М. Шемякина и Ю. А. Овчинникова Авторы: Александр Габибов, Алексей Белогуров, Иван Смирнов, Андрей Решетняк, Инна Куркова, Мария Захарова, Иван Воробьев, Оксана Дурова, Наталья Пономаренко.


