Лекции 1 и 2: Cтруктурно-функциональная организация генов и














25546-lecture_1_gene_amp_amp_protein_technologies.ppt
- Количество слайдов: 52
 Лекции 1 и 2: Cтруктурно-функциональная организация генов и белков. Экспрессия генов и биосинтез белка, клеточный контроль этих процессов. Гены продуктивности растений. Курс: Генно-инженерные и белковые технологии в физиологии растений и сельском хозяйстве
Лекции 1 и 2: Cтруктурно-функциональная организация генов и белков. Экспрессия генов и биосинтез белка, клеточный контроль этих процессов. Гены продуктивности растений. Курс: Генно-инженерные и белковые технологии в физиологии растений и сельском хозяйстве
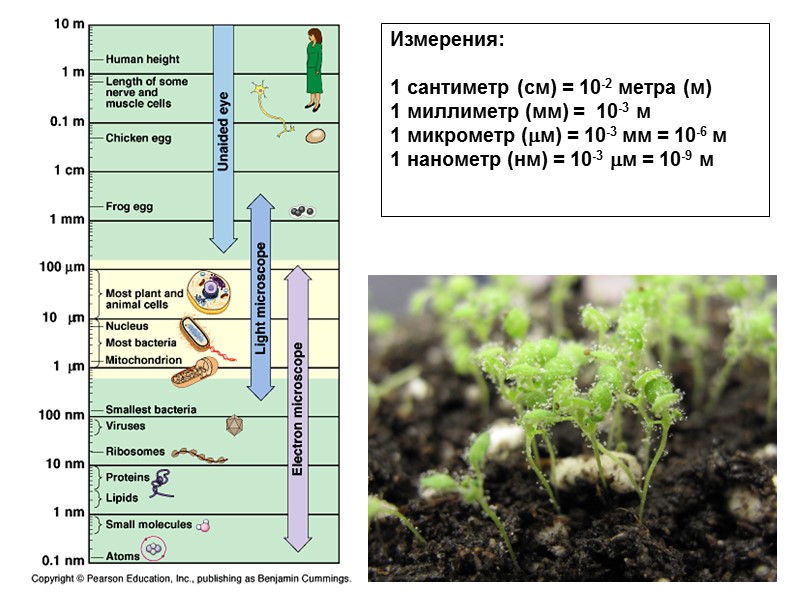
 Измерения: 1 сантиметр (cм) = 10-2 метра (м) 1 миллиметр (мм) = 10-3 м 1 микрометр (м) = 10-3 мм = 10-6 м 1 нанометр (нм) = 10-3 м = 10-9 м
Измерения: 1 сантиметр (cм) = 10-2 метра (м) 1 миллиметр (мм) = 10-3 м 1 микрометр (м) = 10-3 мм = 10-6 м 1 нанометр (нм) = 10-3 м = 10-9 м
 Уровни организации (международная классификация, 2011 год): Atoms Molecules Assemblies Cells Tissue Organ Organ system Organism Population
Уровни организации (международная классификация, 2011 год): Atoms Molecules Assemblies Cells Tissue Organ Organ system Organism Population
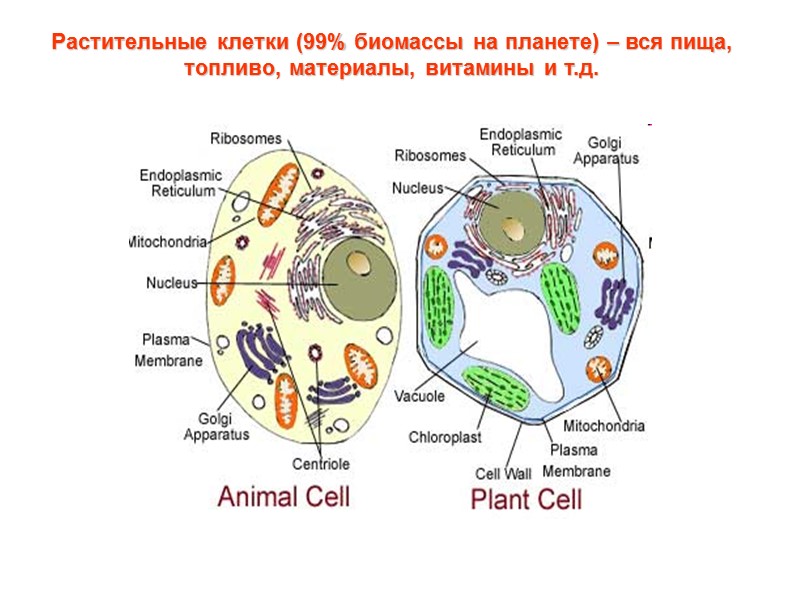
 Растительные клетки (99% биомассы на планете) – вся пища, топливо, материалы, витамины и т.д.
Растительные клетки (99% биомассы на планете) – вся пища, топливо, материалы, витамины и т.д.
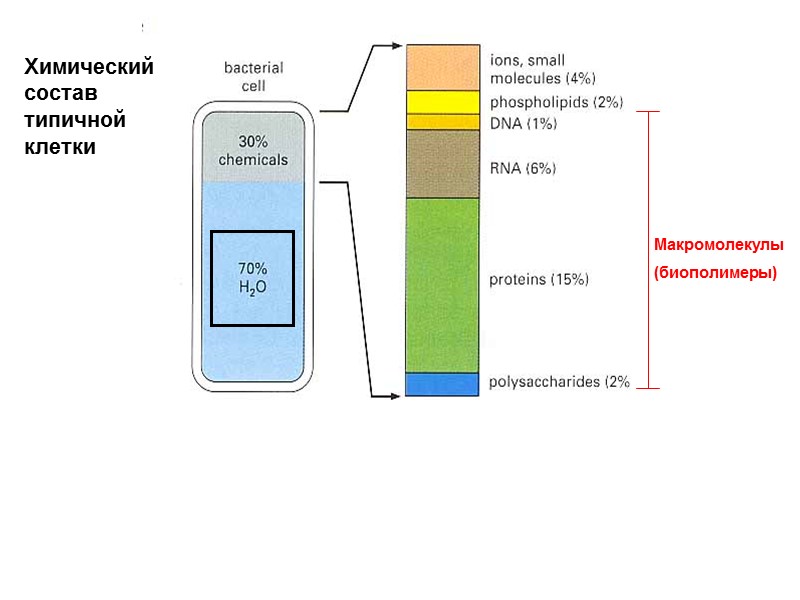
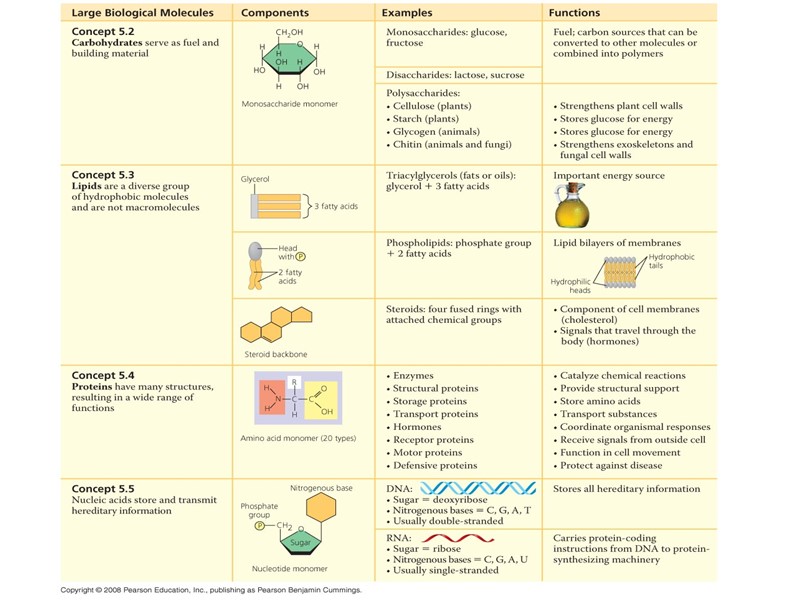
 Химический состав типичной клетки Макромолекулы (биополимеры)
Химический состав типичной клетки Макромолекулы (биополимеры)
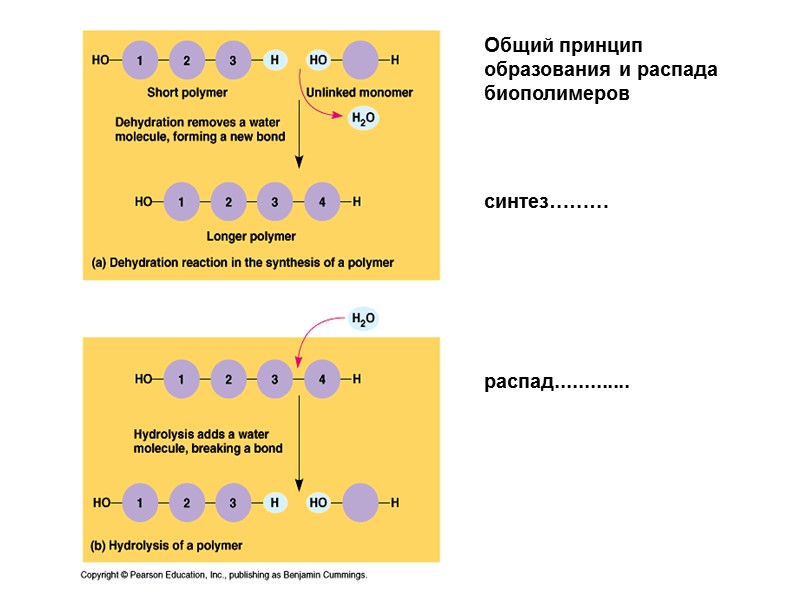
 Общий принцип образования и распада биополимеров синтез……… распад.............
Общий принцип образования и распада биополимеров синтез……… распад.............
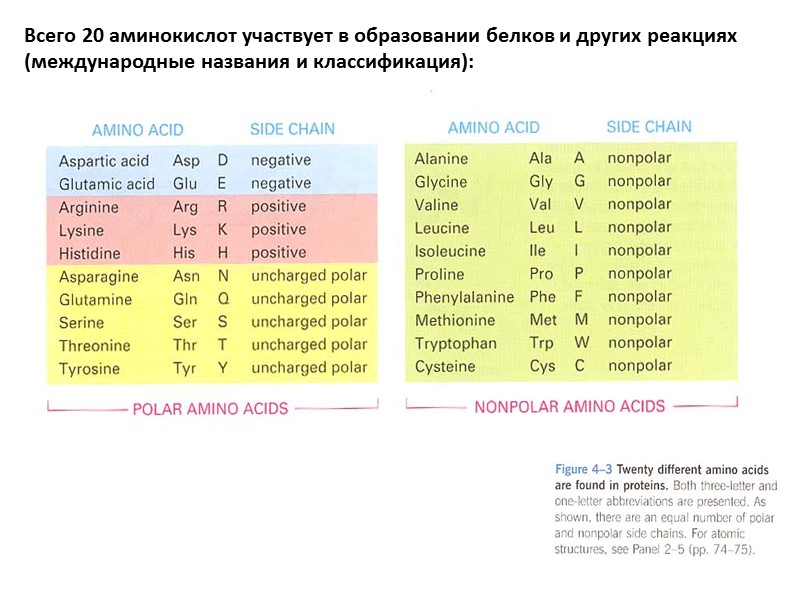
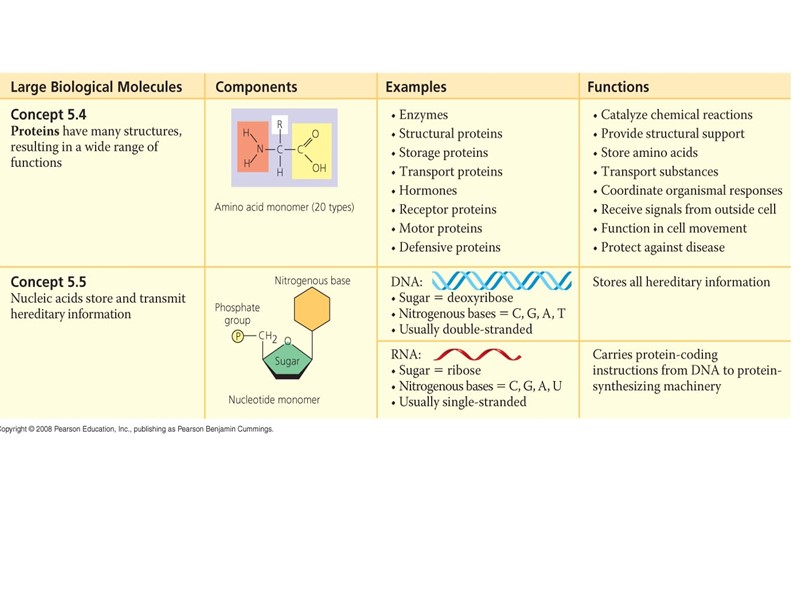
 Всего 20 аминокислот участвует в образовании белков и других реакциях (международные названия и классификация):
Всего 20 аминокислот участвует в образовании белков и других реакциях (международные названия и классификация):
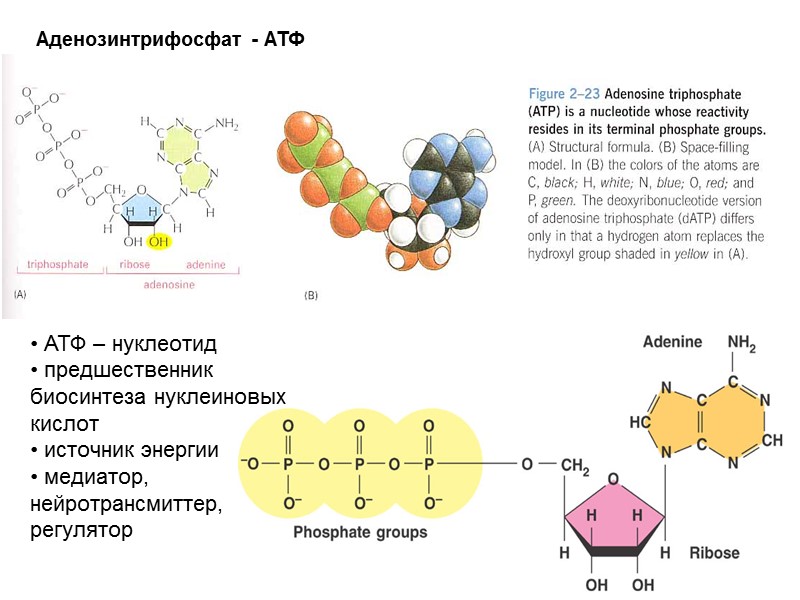
 Аденозинтрифосфат - АТФ АТФ – нуклеотид предшественник биосинтеза нуклеиновых кислот источник энергии медиатор, нейротрансмиттер, регулятор
Аденозинтрифосфат - АТФ АТФ – нуклеотид предшественник биосинтеза нуклеиновых кислот источник энергии медиатор, нейротрансмиттер, регулятор
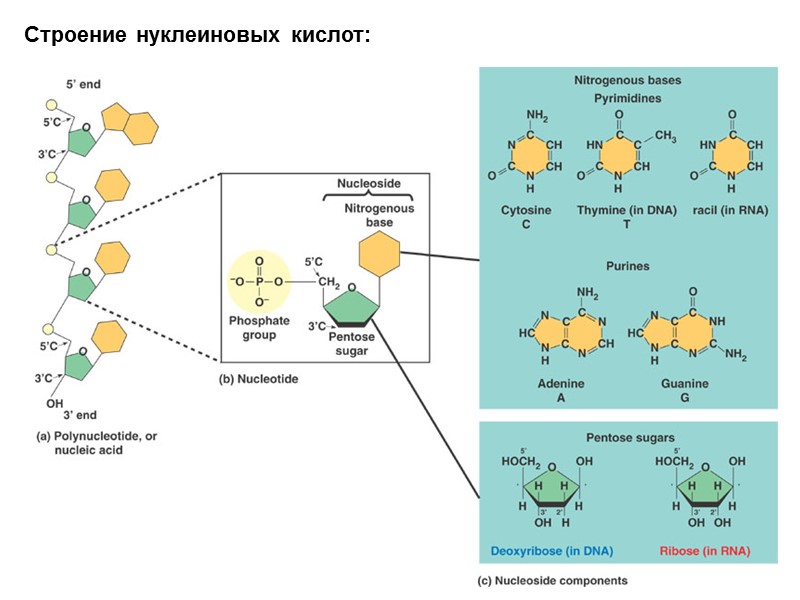
 Строение нуклеиновых кислот:
Строение нуклеиновых кислот:
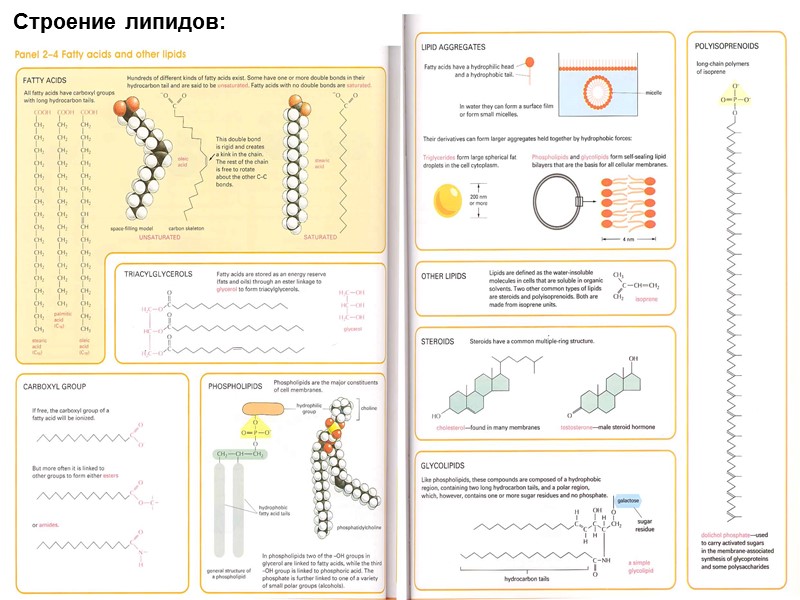
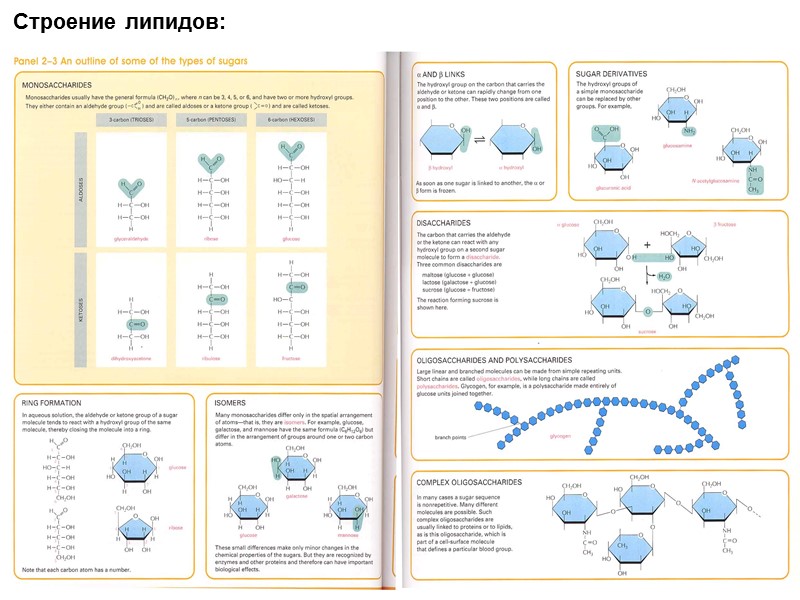
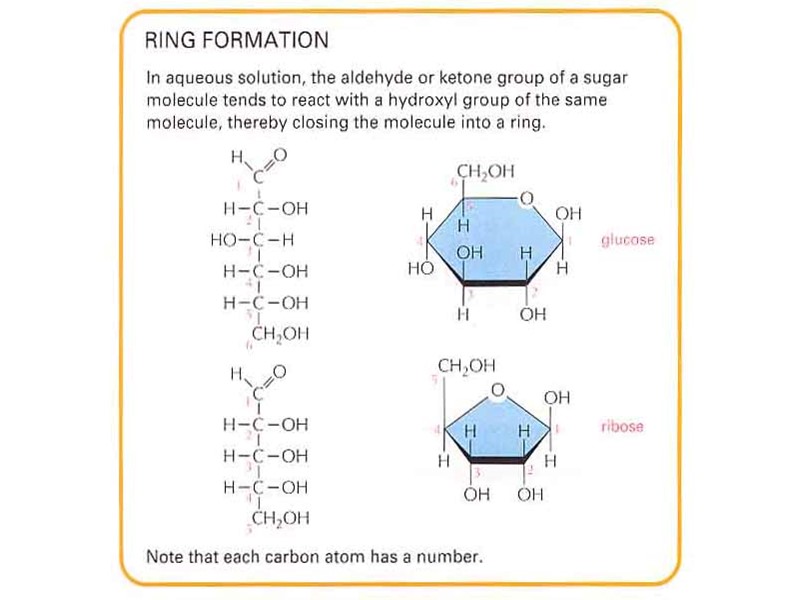
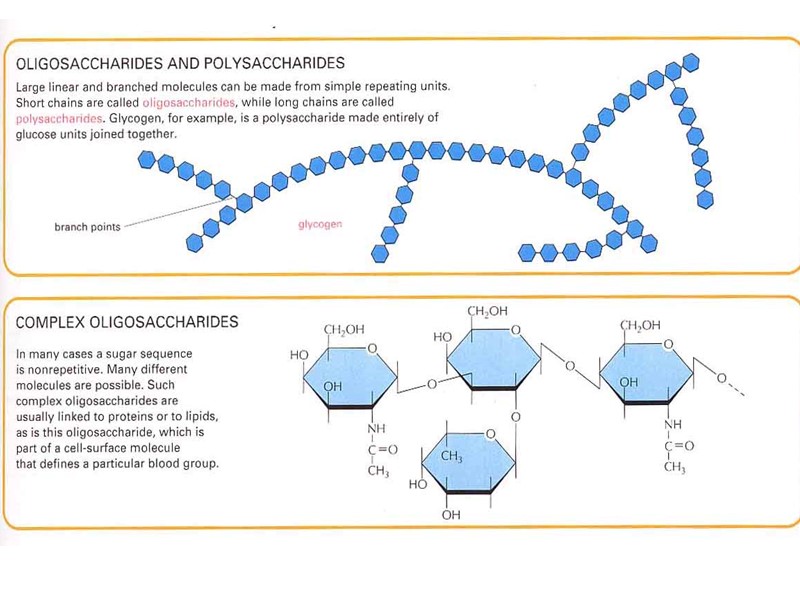
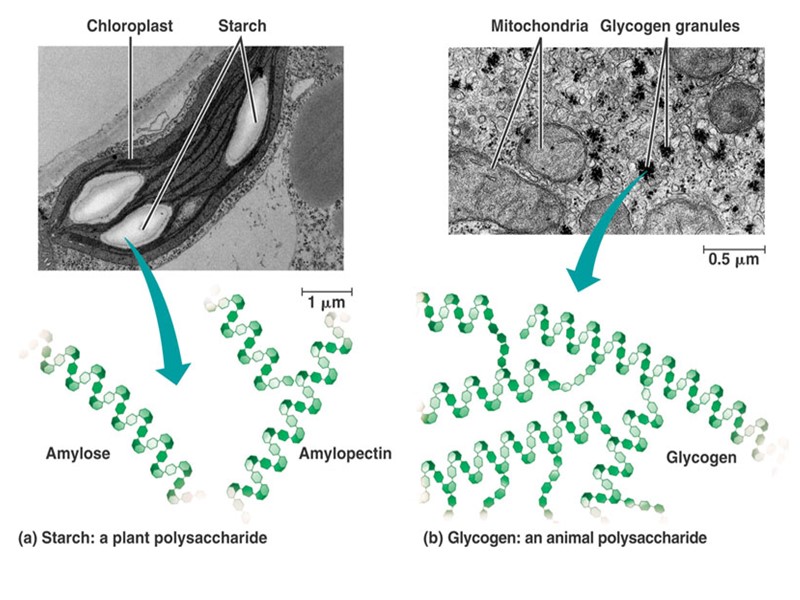
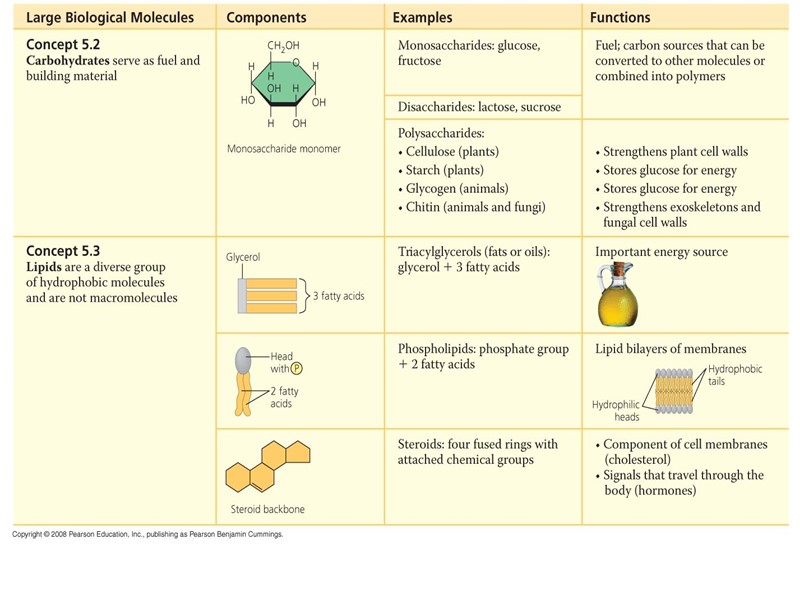
 Строение липидов:
Строение липидов:
 Строение липидов:
Строение липидов:







 Разнообразие комбинаций трех полимеров определяет разнообразие живого на планете. Растения, животные и грибы имеют одинаковые «мономеры» и принципы их соединения в макромолекулы, но благодаря отличиям в молекулярной структуре и организации, они могут сильно отличаться на клеточной и организменном уровне.
Разнообразие комбинаций трех полимеров определяет разнообразие живого на планете. Растения, животные и грибы имеют одинаковые «мономеры» и принципы их соединения в макромолекулы, но благодаря отличиям в молекулярной структуре и организации, они могут сильно отличаться на клеточной и организменном уровне.
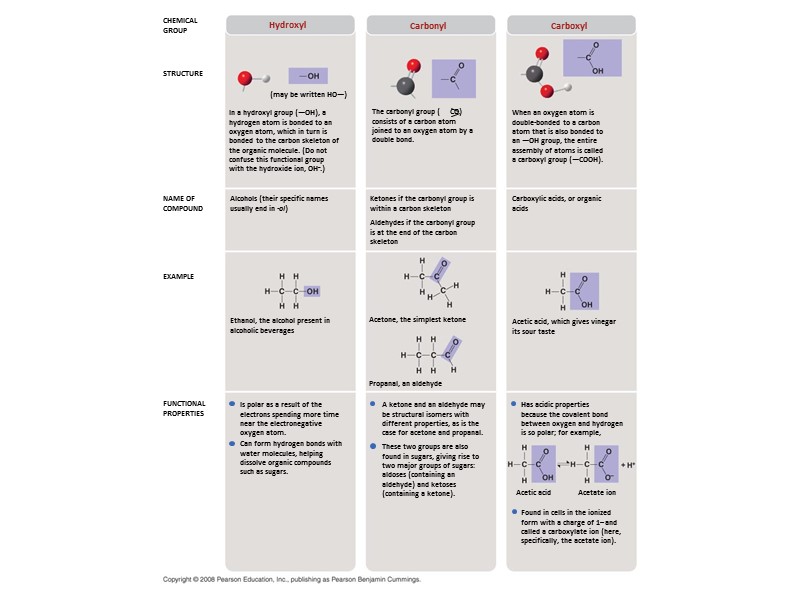
 Семь функциональных групп, которые наиболее важны в биохимических процессах: Гидроксильная Карбонильная Карбоксильная Амино Сульфгидрильная Фосфатная Метильная
Семь функциональных групп, которые наиболее важны в биохимических процессах: Гидроксильная Карбонильная Карбоксильная Амино Сульфгидрильная Фосфатная Метильная
 Hydroxyl CHEMICAL GROUP STRUCTURE NAME OF COMPOUND EXAMPLE FUNCTIONAL PROPERTIES Carbonyl Carboxyl (may be written HO—) In a hydroxyl group (—OH), a hydrogen atom is bonded to an oxygen atom, which in turn is bonded to the carbon skeleton of the organic molecule. (Do not confuse this functional group with the hydroxide ion, OH–.) When an oxygen atom is double-bonded to a carbon atom that is also bonded to an —OH group, the entire assembly of atoms is called a carboxyl group (—COOH). Carboxylic acids, or organic acids Ketones if the carbonyl group is within a carbon skeleton Aldehydes if the carbonyl group is at the end of the carbon skeleton Alcohols (their specific names usually end in -ol) Ethanol, the alcohol present in alcoholic beverages Acetone, the simplest ketone Acetic acid, which gives vinegar its sour taste Propanal, an aldehyde Has acidic properties because the covalent bond between oxygen and hydrogen is so polar; for example, Found in cells in the ionized form with a charge of 1– and called a carboxylate ion (here, specifically, the acetate ion). Acetic acid Acetate ion A ketone and an aldehyde may be structural isomers with different properties, as is the case for acetone and propanal. These two groups are also found in sugars, giving rise to two major groups of sugars: aldoses (containing an aldehyde) and ketoses (containing a ketone). Is polar as a result of the electrons spending more time near the electronegative oxygen atom. Can form hydrogen bonds with water molecules, helping dissolve organic compounds such as sugars. The carbonyl group ( CO) consists of a carbon atom joined to an oxygen atom by a double bond.
Hydroxyl CHEMICAL GROUP STRUCTURE NAME OF COMPOUND EXAMPLE FUNCTIONAL PROPERTIES Carbonyl Carboxyl (may be written HO—) In a hydroxyl group (—OH), a hydrogen atom is bonded to an oxygen atom, which in turn is bonded to the carbon skeleton of the organic molecule. (Do not confuse this functional group with the hydroxide ion, OH–.) When an oxygen atom is double-bonded to a carbon atom that is also bonded to an —OH group, the entire assembly of atoms is called a carboxyl group (—COOH). Carboxylic acids, or organic acids Ketones if the carbonyl group is within a carbon skeleton Aldehydes if the carbonyl group is at the end of the carbon skeleton Alcohols (their specific names usually end in -ol) Ethanol, the alcohol present in alcoholic beverages Acetone, the simplest ketone Acetic acid, which gives vinegar its sour taste Propanal, an aldehyde Has acidic properties because the covalent bond between oxygen and hydrogen is so polar; for example, Found in cells in the ionized form with a charge of 1– and called a carboxylate ion (here, specifically, the acetate ion). Acetic acid Acetate ion A ketone and an aldehyde may be structural isomers with different properties, as is the case for acetone and propanal. These two groups are also found in sugars, giving rise to two major groups of sugars: aldoses (containing an aldehyde) and ketoses (containing a ketone). Is polar as a result of the electrons spending more time near the electronegative oxygen atom. Can form hydrogen bonds with water molecules, helping dissolve organic compounds such as sugars. The carbonyl group ( CO) consists of a carbon atom joined to an oxygen atom by a double bond.
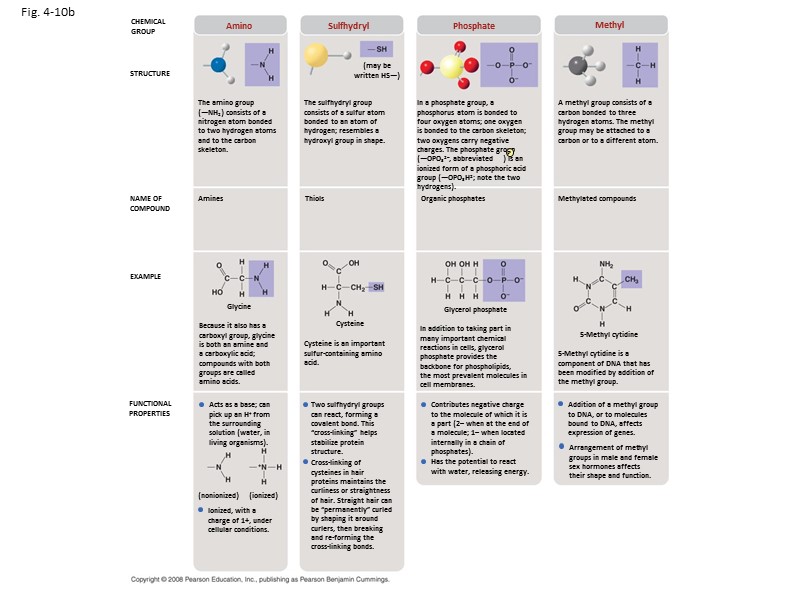
 Fig. 4-10b CHEMICAL GROUP STRUCTURE NAME OF COMPOUND EXAMPLE FUNCTIONAL PROPERTIES Amino Sulfhydryl Phosphate Methyl A methyl group consists of a carbon bonded to three hydrogen atoms. The methyl group may be attached to a carbon or to a different atom. In a phosphate group, a phosphorus atom is bonded to four oxygen atoms; one oxygen is bonded to the carbon skeleton; two oxygens carry negative charges. The phosphate group (—OPO32–, abbreviated ) is an ionized form of a phosphoric acid group (—OPO3H2; note the two hydrogens). P The sulfhydryl group consists of a sulfur atom bonded to an atom of hydrogen; resembles a hydroxyl group in shape. (may be written HS—) The amino group (—NH2) consists of a nitrogen atom bonded to two hydrogen atoms and to the carbon skeleton. Amines Thiols Organic phosphates Methylated compounds 5-Methyl cytidine 5-Methyl cytidine is a component of DNA that has been modified by addition of the methyl group. In addition to taking part in many important chemical reactions in cells, glycerol phosphate provides the backbone for phospholipids, the most prevalent molecules in cell membranes. Glycerol phosphate Cysteine Cysteine is an important sulfur-containing amino acid. Glycine Because it also has a carboxyl group, glycine is both an amine and a carboxylic acid; compounds with both groups are called amino acids. Addition of a methyl group to DNA, or to molecules bound to DNA, affects expression of genes. Arrangement of methyl groups in male and female sex hormones affects their shape and function. Contributes negative charge to the molecule of which it is a part (2– when at the end of a molecule; 1– when located internally in a chain of phosphates). Has the potential to react with water, releasing energy. Two sulfhydryl groups can react, forming a covalent bond. This “cross-linking” helps stabilize protein structure. Cross-linking of cysteines in hair proteins maintains the curliness or straightness of hair. Straight hair can be “permanently” curled by shaping it around curlers, then breaking and re-forming the cross-linking bonds. Acts as a base; can pick up an H+ from the surrounding solution (water, in living organisms). Ionized, with a charge of 1+, under cellular conditions. (nonionized) (ionized)
Fig. 4-10b CHEMICAL GROUP STRUCTURE NAME OF COMPOUND EXAMPLE FUNCTIONAL PROPERTIES Amino Sulfhydryl Phosphate Methyl A methyl group consists of a carbon bonded to three hydrogen atoms. The methyl group may be attached to a carbon or to a different atom. In a phosphate group, a phosphorus atom is bonded to four oxygen atoms; one oxygen is bonded to the carbon skeleton; two oxygens carry negative charges. The phosphate group (—OPO32–, abbreviated ) is an ionized form of a phosphoric acid group (—OPO3H2; note the two hydrogens). P The sulfhydryl group consists of a sulfur atom bonded to an atom of hydrogen; resembles a hydroxyl group in shape. (may be written HS—) The amino group (—NH2) consists of a nitrogen atom bonded to two hydrogen atoms and to the carbon skeleton. Amines Thiols Organic phosphates Methylated compounds 5-Methyl cytidine 5-Methyl cytidine is a component of DNA that has been modified by addition of the methyl group. In addition to taking part in many important chemical reactions in cells, glycerol phosphate provides the backbone for phospholipids, the most prevalent molecules in cell membranes. Glycerol phosphate Cysteine Cysteine is an important sulfur-containing amino acid. Glycine Because it also has a carboxyl group, glycine is both an amine and a carboxylic acid; compounds with both groups are called amino acids. Addition of a methyl group to DNA, or to molecules bound to DNA, affects expression of genes. Arrangement of methyl groups in male and female sex hormones affects their shape and function. Contributes negative charge to the molecule of which it is a part (2– when at the end of a molecule; 1– when located internally in a chain of phosphates). Has the potential to react with water, releasing energy. Two sulfhydryl groups can react, forming a covalent bond. This “cross-linking” helps stabilize protein structure. Cross-linking of cysteines in hair proteins maintains the curliness or straightness of hair. Straight hair can be “permanently” curled by shaping it around curlers, then breaking and re-forming the cross-linking bonds. Acts as a base; can pick up an H+ from the surrounding solution (water, in living organisms). Ionized, with a charge of 1+, under cellular conditions. (nonionized) (ionized)
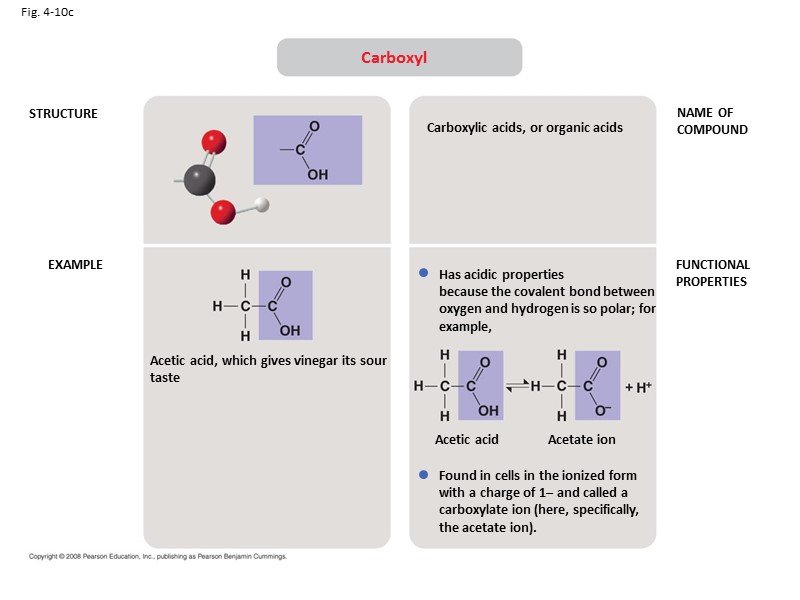
 Fig. 4-10c STRUCTURE EXAMPLE NAME OF COMPOUND FUNCTIONAL PROPERTIES Carboxyl Acetic acid, which gives vinegar its sour taste Carboxylic acids, or organic acids Has acidic properties because the covalent bond between oxygen and hydrogen is so polar; for example, Found in cells in the ionized form with a charge of 1– and called a carboxylate ion (here, specifically, the acetate ion). Acetic acid Acetate ion
Fig. 4-10c STRUCTURE EXAMPLE NAME OF COMPOUND FUNCTIONAL PROPERTIES Carboxyl Acetic acid, which gives vinegar its sour taste Carboxylic acids, or organic acids Has acidic properties because the covalent bond between oxygen and hydrogen is so polar; for example, Found in cells in the ionized form with a charge of 1– and called a carboxylate ion (here, specifically, the acetate ion). Acetic acid Acetate ion
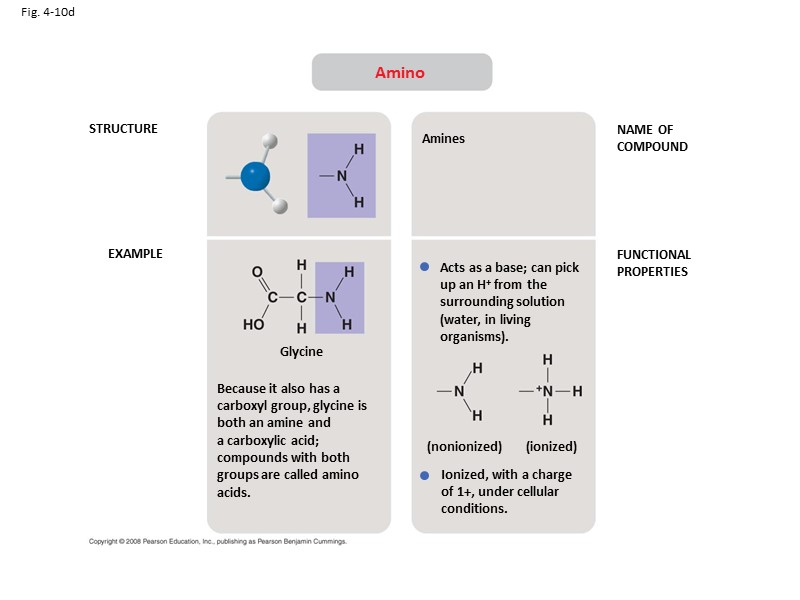
 Fig. 4-10d STRUCTURE EXAMPLE NAME OF COMPOUND FUNCTIONAL PROPERTIES Amino Because it also has a carboxyl group, glycine is both an amine and a carboxylic acid; compounds with both groups are called amino acids. Amines Acts as a base; can pick up an H+ from the surrounding solution (water, in living organisms). Ionized, with a charge of 1+, under cellular conditions. (ionized) (nonionized) Glycine
Fig. 4-10d STRUCTURE EXAMPLE NAME OF COMPOUND FUNCTIONAL PROPERTIES Amino Because it also has a carboxyl group, glycine is both an amine and a carboxylic acid; compounds with both groups are called amino acids. Amines Acts as a base; can pick up an H+ from the surrounding solution (water, in living organisms). Ionized, with a charge of 1+, under cellular conditions. (ionized) (nonionized) Glycine
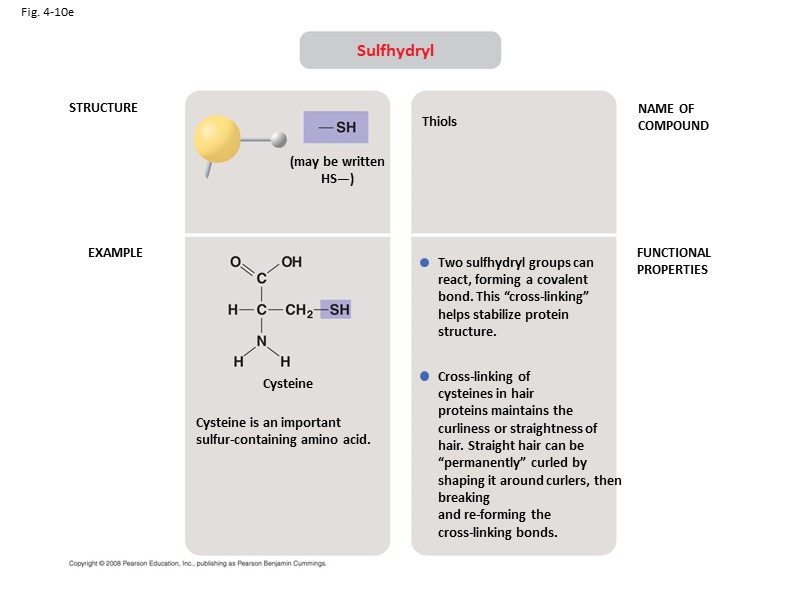
 Fig. 4-10e STRUCTURE EXAMPLE NAME OF COMPOUND FUNCTIONAL PROPERTIES Sulfhydryl (may be written HS—) Cysteine Cysteine is an important sulfur-containing amino acid. Thiols Two sulfhydryl groups can react, forming a covalent bond. This “cross-linking” helps stabilize protein structure. Cross-linking of cysteines in hair proteins maintains the curliness or straightness of hair. Straight hair can be “permanently” curled by shaping it around curlers, then breaking and re-forming the cross-linking bonds.
Fig. 4-10e STRUCTURE EXAMPLE NAME OF COMPOUND FUNCTIONAL PROPERTIES Sulfhydryl (may be written HS—) Cysteine Cysteine is an important sulfur-containing amino acid. Thiols Two sulfhydryl groups can react, forming a covalent bond. This “cross-linking” helps stabilize protein structure. Cross-linking of cysteines in hair proteins maintains the curliness or straightness of hair. Straight hair can be “permanently” curled by shaping it around curlers, then breaking and re-forming the cross-linking bonds.
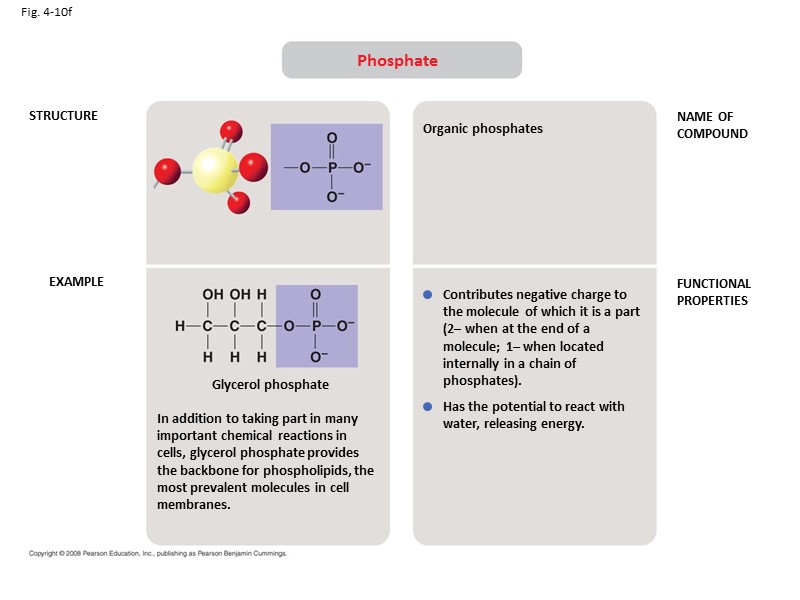
 Fig. 4-10f STRUCTURE EXAMPLE NAME OF COMPOUND FUNCTIONAL PROPERTIES Phosphate In addition to taking part in many important chemical reactions in cells, glycerol phosphate provides the backbone for phospholipids, the most prevalent molecules in cell membranes. Glycerol phosphate Organic phosphates Contributes negative charge to the molecule of which it is a part (2– when at the end of a molecule; 1– when located internally in a chain of phosphates). Has the potential to react with water, releasing energy.
Fig. 4-10f STRUCTURE EXAMPLE NAME OF COMPOUND FUNCTIONAL PROPERTIES Phosphate In addition to taking part in many important chemical reactions in cells, glycerol phosphate provides the backbone for phospholipids, the most prevalent molecules in cell membranes. Glycerol phosphate Organic phosphates Contributes negative charge to the molecule of which it is a part (2– when at the end of a molecule; 1– when located internally in a chain of phosphates). Has the potential to react with water, releasing energy.
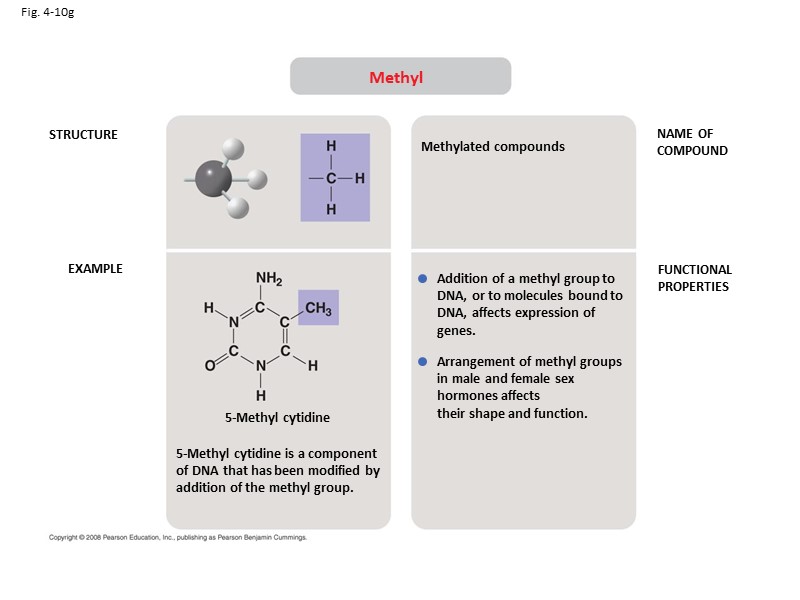
 Fig. 4-10g STRUCTURE EXAMPLE NAME OF COMPOUND FUNCTIONAL PROPERTIES Methyl 5-Methyl cytidine is a component of DNA that has been modified by addition of the methyl group. 5-Methyl cytidine Methylated compounds Addition of a methyl group to DNA, or to molecules bound to DNA, affects expression of genes. Arrangement of methyl groups in male and female sex hormones affects their shape and function.
Fig. 4-10g STRUCTURE EXAMPLE NAME OF COMPOUND FUNCTIONAL PROPERTIES Methyl 5-Methyl cytidine is a component of DNA that has been modified by addition of the methyl group. 5-Methyl cytidine Methylated compounds Addition of a methyl group to DNA, or to molecules bound to DNA, affects expression of genes. Arrangement of methyl groups in male and female sex hormones affects their shape and function.

 James Watson and Francis Crick, UK Erwin Chargaff, Ukraine Linus Pauling, USA Maurice Wilkins, NZ Rosalind Franklin, UK Ученые, которые участвовали в открытии структуры ДНК
James Watson and Francis Crick, UK Erwin Chargaff, Ukraine Linus Pauling, USA Maurice Wilkins, NZ Rosalind Franklin, UK Ученые, которые участвовали в открытии структуры ДНК
 Первое правило Чаргафа: В природной ДНК количество единиц гуанина равно количеству единиц цитозина, тогда как количество единиц аденина равно количеству единиц тимина. В типичной ДНК эукариот (пример: ДНК человека): A=30.9%, T=29.4%; Г=19.9% Ц=19.8%.
Первое правило Чаргафа: В природной ДНК количество единиц гуанина равно количеству единиц цитозина, тогда как количество единиц аденина равно количеству единиц тимина. В типичной ДНК эукариот (пример: ДНК человека): A=30.9%, T=29.4%; Г=19.9% Ц=19.8%.
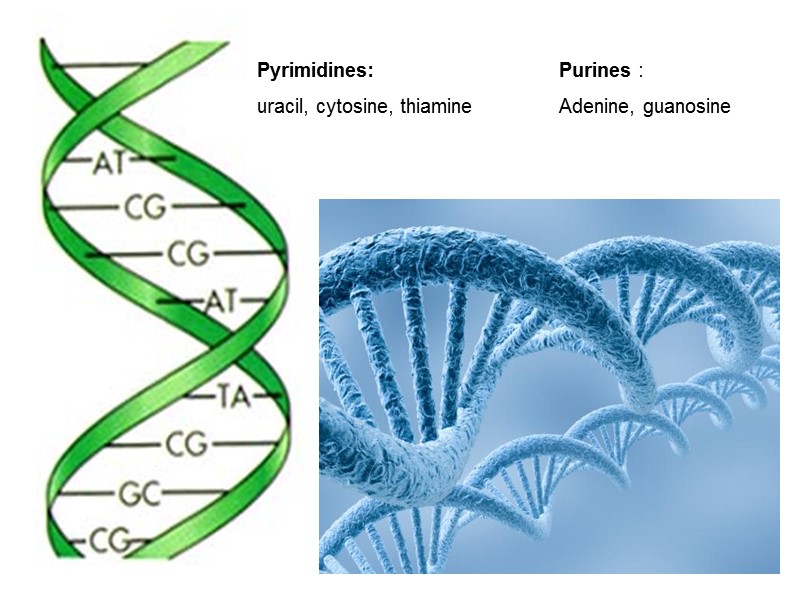
 Pyrimidines: uracil, cytosine, thiamine Purines : Adenine, guanosine
Pyrimidines: uracil, cytosine, thiamine Purines : Adenine, guanosine
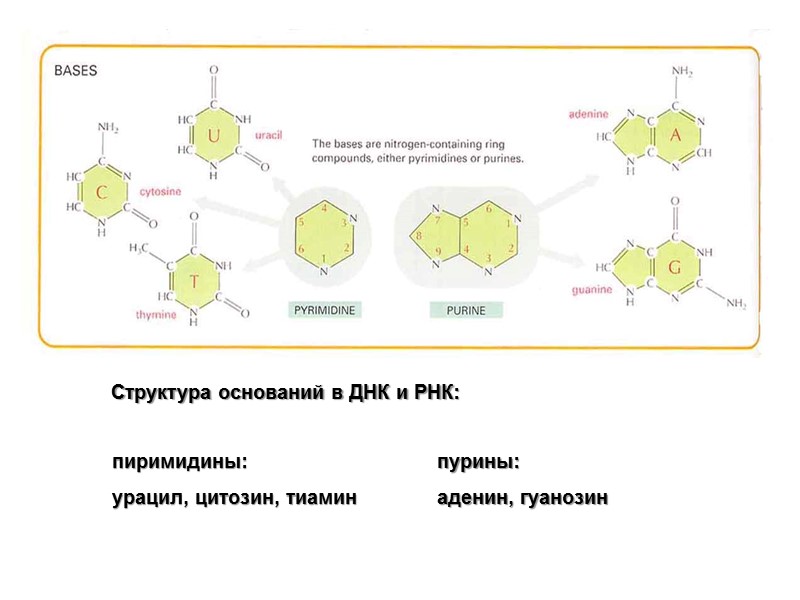
 Структура оснований в ДНК и РНК: пиримидины: урацил, цитозин, тиамин пурины: аденин, гуанозин
Структура оснований в ДНК и РНК: пиримидины: урацил, цитозин, тиамин пурины: аденин, гуанозин
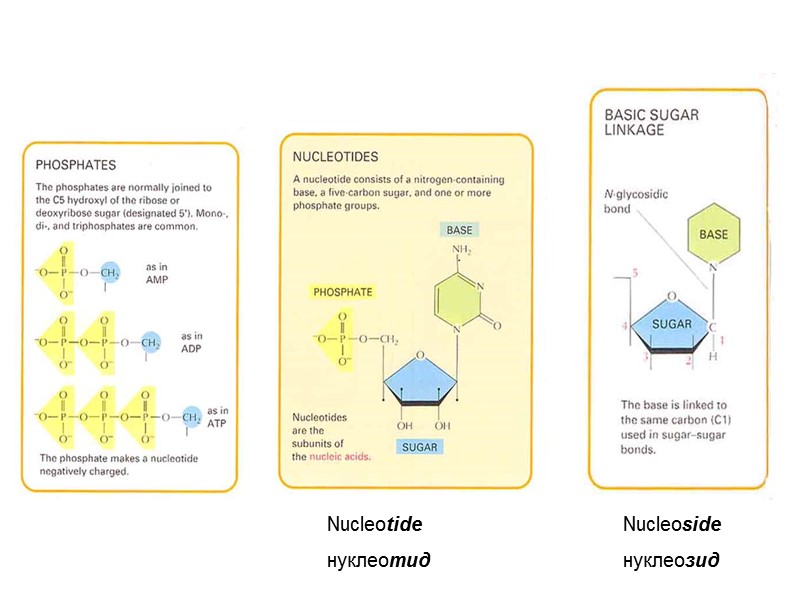
 Nucleoside нуклеозид Nucleotide нуклеотид
Nucleoside нуклеозид Nucleotide нуклеотид
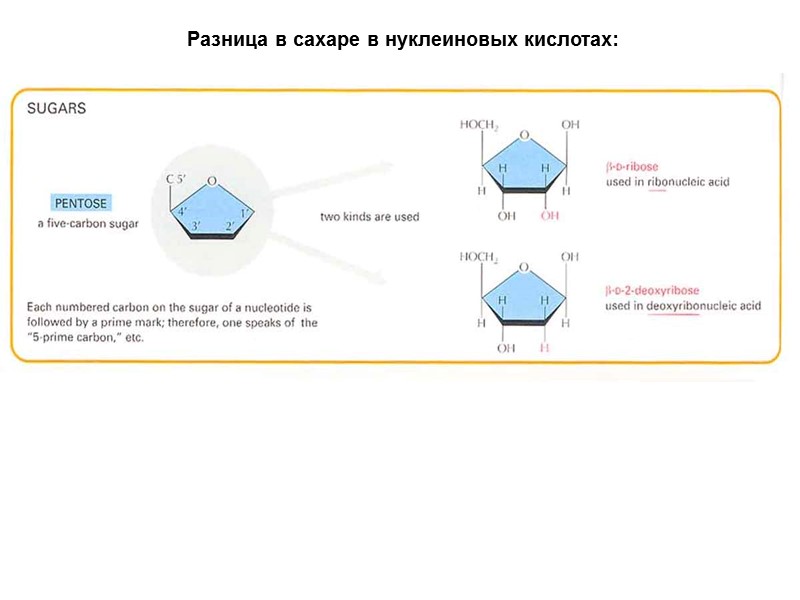
 Разница в сахаре в нуклеиновых кислотах:
Разница в сахаре в нуклеиновых кислотах:
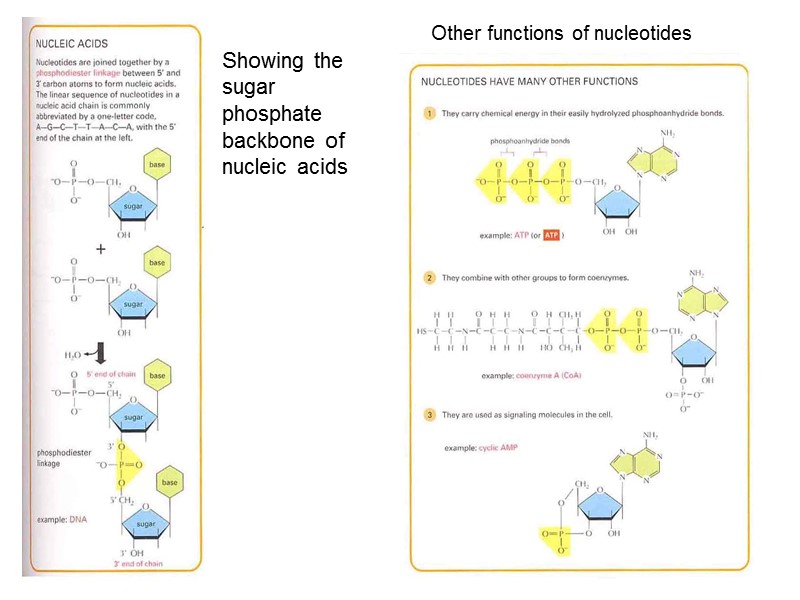
 Other functions of nucleotides Showing the sugar phosphate backbone of nucleic acids
Other functions of nucleotides Showing the sugar phosphate backbone of nucleic acids
 Study this carefully to memorize general structure and nomenclature.
Study this carefully to memorize general structure and nomenclature.
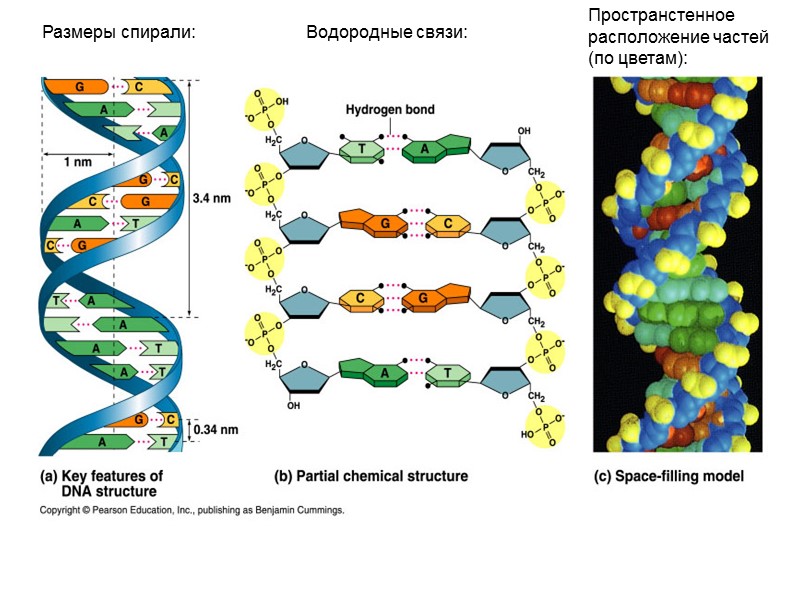
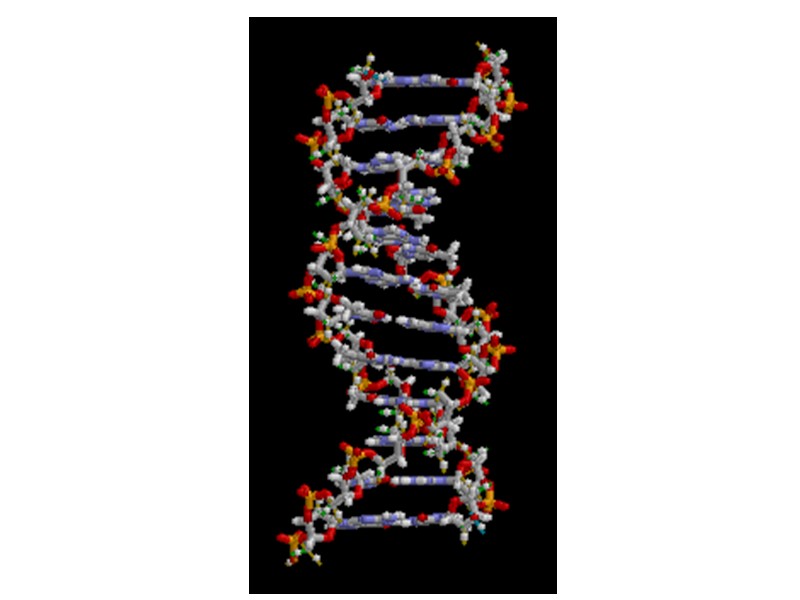
 Размеры спирали: Водородные связи: Пространстенное расположение частей (по цветам):
Размеры спирали: Водородные связи: Пространстенное расположение частей (по цветам):

 Hydrogen bonding between bases important in double helix. Two between A and T; three between G and C.
Hydrogen bonding between bases important in double helix. Two between A and T; three between G and C.
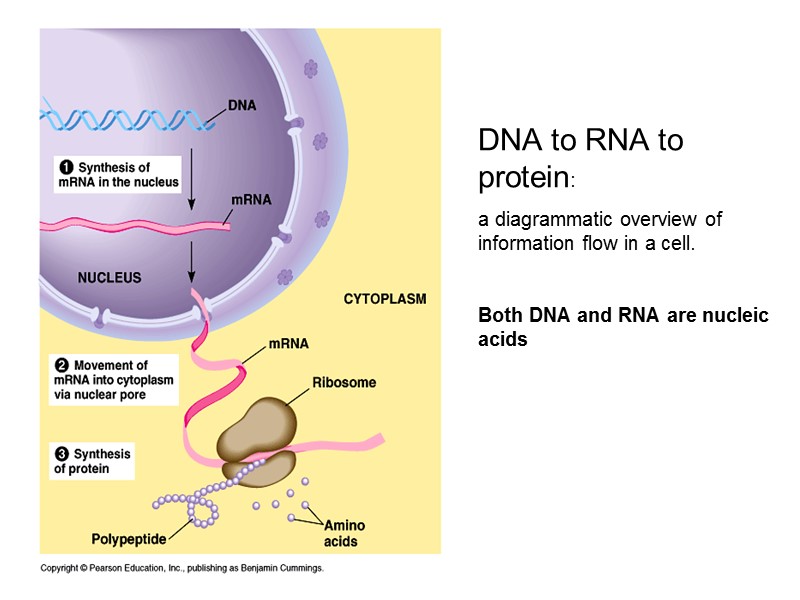
 DNA to RNA to protein: a diagrammatic overview of information flow in a cell. Both DNA and RNA are nucleic acids
DNA to RNA to protein: a diagrammatic overview of information flow in a cell. Both DNA and RNA are nucleic acids
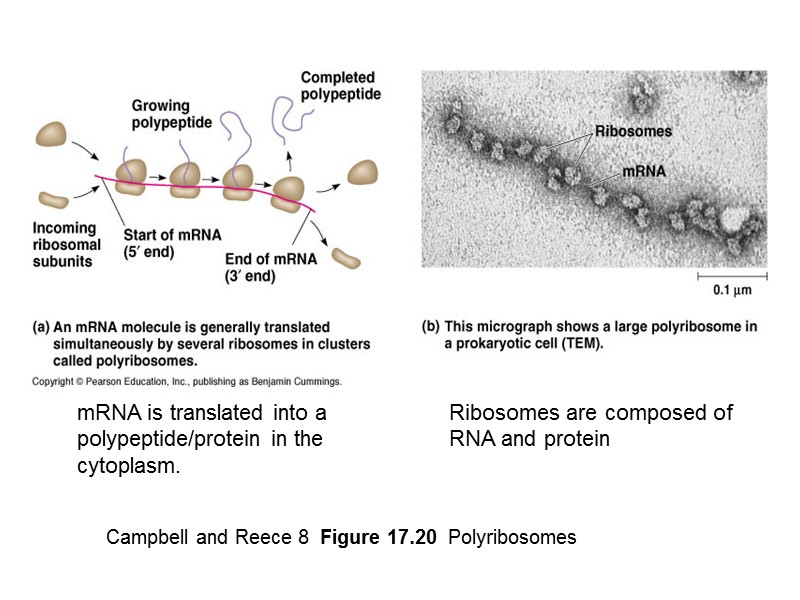
 Campbell and Reece 8 Figure 17.20 Polyribosomes mRNA is translated into a polypeptide/protein in the cytoplasm. Ribosomes are composed of RNA and protein
Campbell and Reece 8 Figure 17.20 Polyribosomes mRNA is translated into a polypeptide/protein in the cytoplasm. Ribosomes are composed of RNA and protein
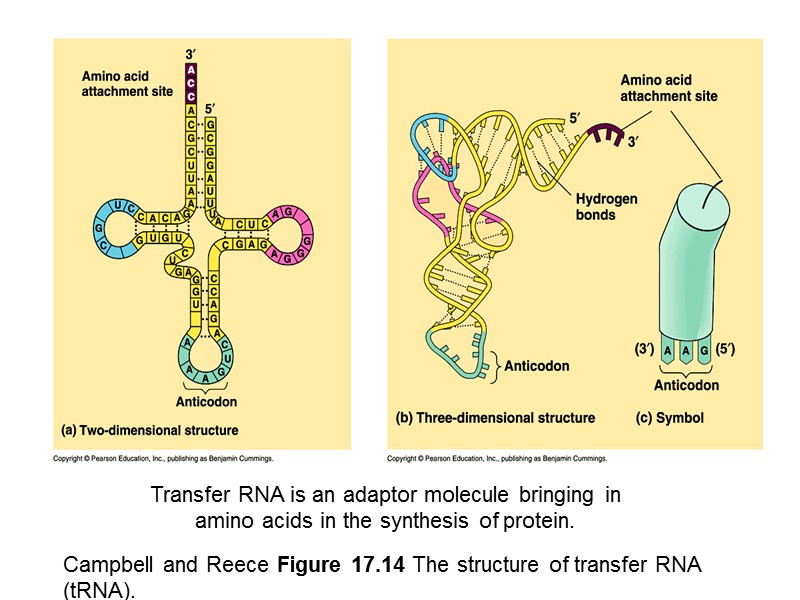
 Campbell and Reece Figure 17.14 The structure of transfer RNA (tRNA). Transfer RNA is an adaptor molecule bringing in amino acids in the synthesis of protein.
Campbell and Reece Figure 17.14 The structure of transfer RNA (tRNA). Transfer RNA is an adaptor molecule bringing in amino acids in the synthesis of protein.
 Campbell and Reece 8 Chapter 17 These first three are the types of RNA that you need to know about: mRNA tRNA rRNA
Campbell and Reece 8 Chapter 17 These first three are the types of RNA that you need to know about: mRNA tRNA rRNA

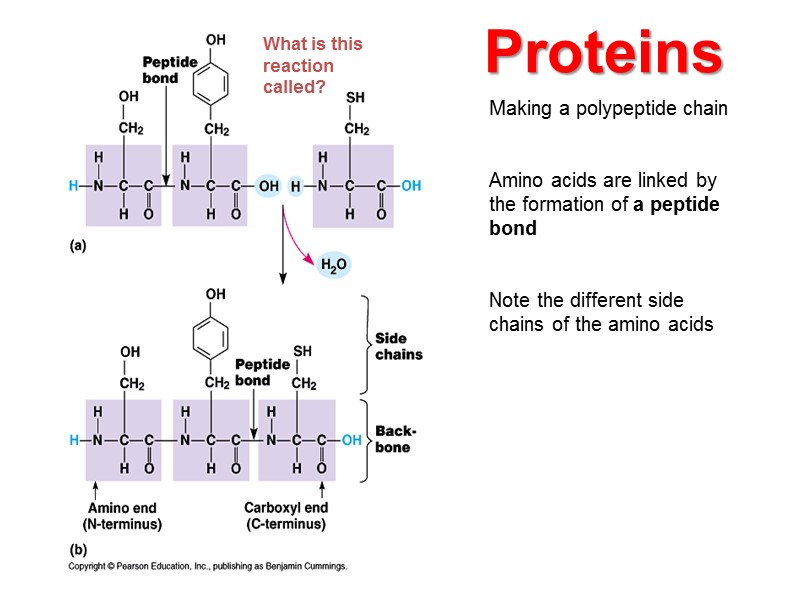
 Making a polypeptide chain Amino acids are linked by the formation of a peptide bond Note the different side chains of the amino acids Proteins What is this reaction called?
Making a polypeptide chain Amino acids are linked by the formation of a peptide bond Note the different side chains of the amino acids Proteins What is this reaction called?
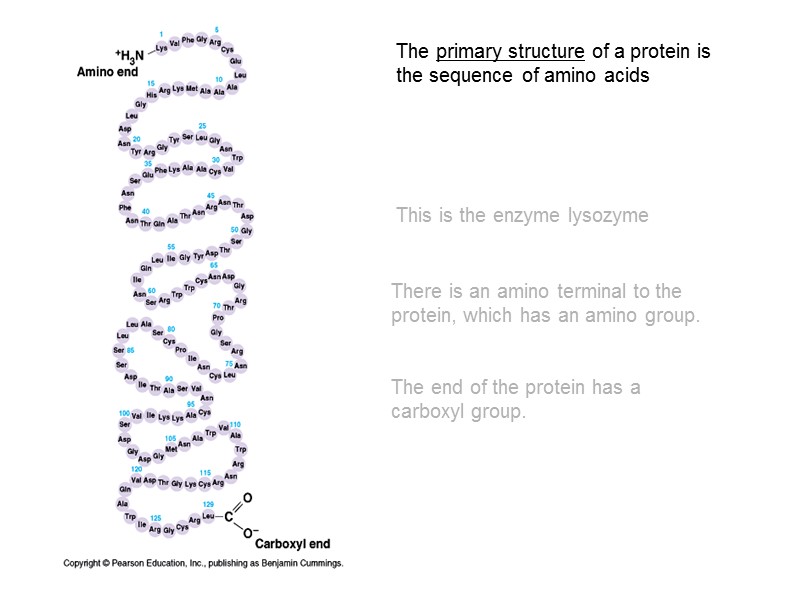
 The primary structure of a protein is the sequence of amino acids This is the enzyme lysozyme There is an amino terminal to the protein, which has an amino group. The end of the protein has a carboxyl group.
The primary structure of a protein is the sequence of amino acids This is the enzyme lysozyme There is an amino terminal to the protein, which has an amino group. The end of the protein has a carboxyl group.
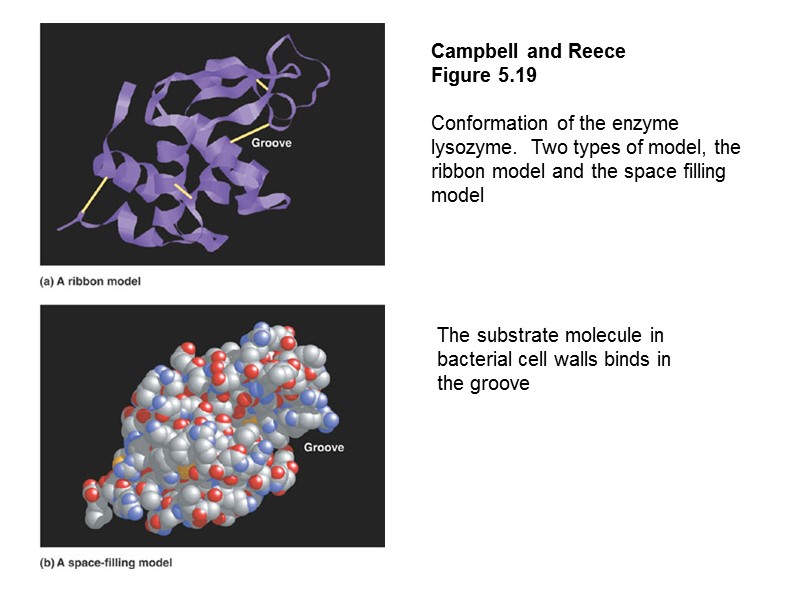
 Campbell and Reece Figure 5.19 Conformation of the enzyme lysozyme. Two types of model, the ribbon model and the space filling model The substrate molecule in bacterial cell walls binds in the groove
Campbell and Reece Figure 5.19 Conformation of the enzyme lysozyme. Two types of model, the ribbon model and the space filling model The substrate molecule in bacterial cell walls binds in the groove
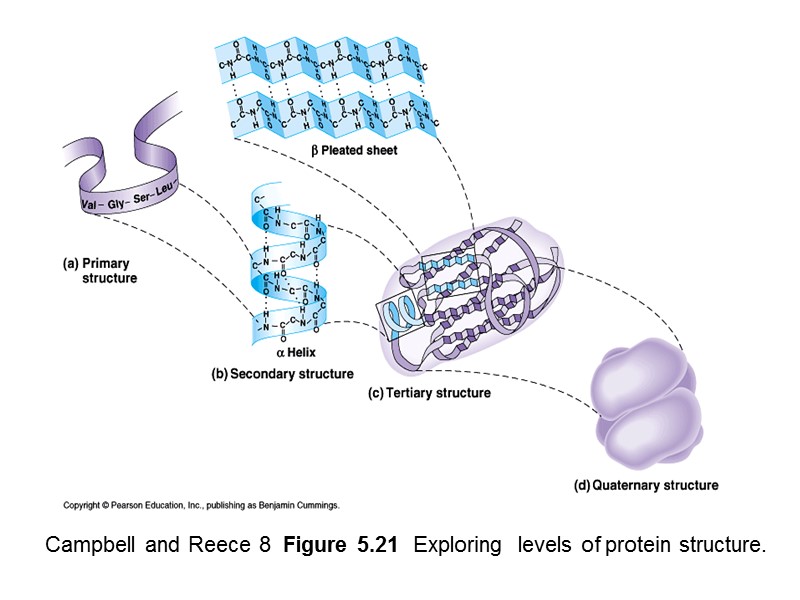
 Campbell and Reece 8 Figure 5.21 Exploring levels of protein structure.
Campbell and Reece 8 Figure 5.21 Exploring levels of protein structure.
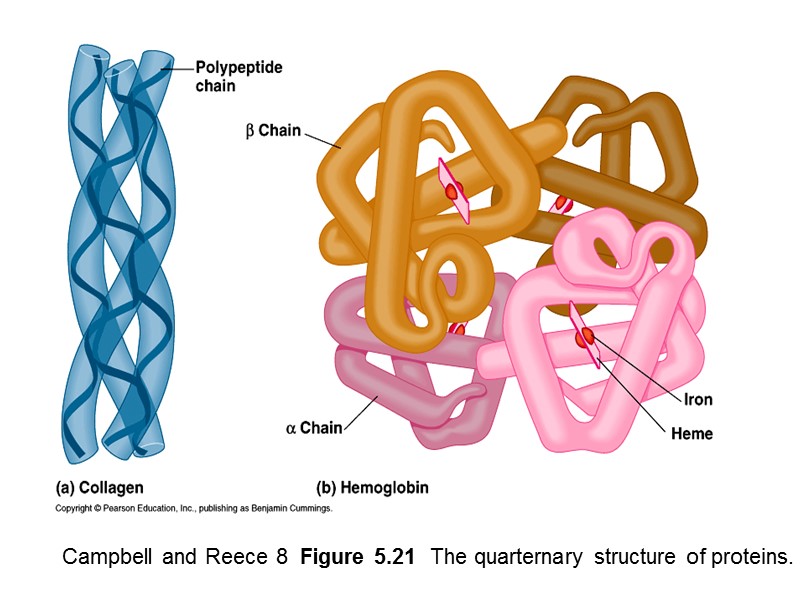
 Campbell and Reece 8 Figure 5.21 The quarternary structure of proteins.
Campbell and Reece 8 Figure 5.21 The quarternary structure of proteins.
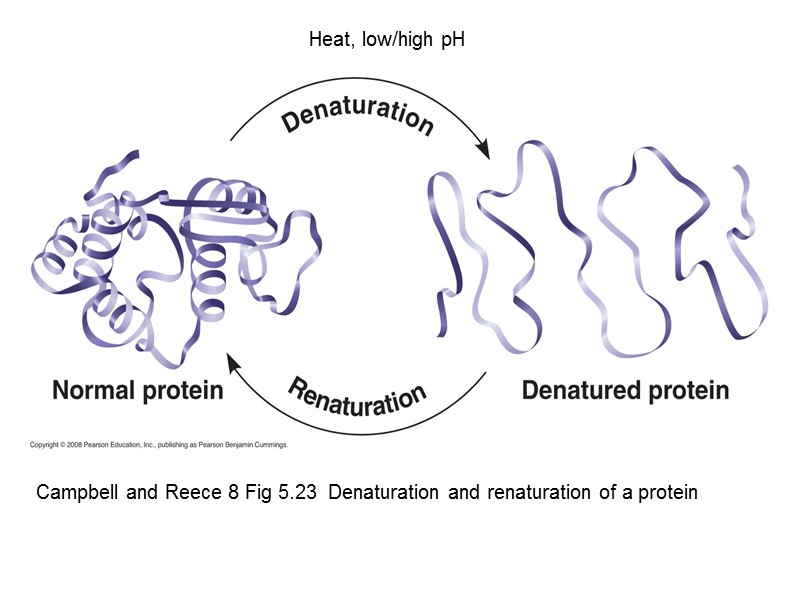
 Campbell and Reece 8 Fig 5.23 Denaturation and renaturation of a protein Heat, low/high pH
Campbell and Reece 8 Fig 5.23 Denaturation and renaturation of a protein Heat, low/high pH
 Essential Cell Biology 2 Protein structure and protein-protein interactions are important in viral particle structure
Essential Cell Biology 2 Protein structure and protein-protein interactions are important in viral particle structure
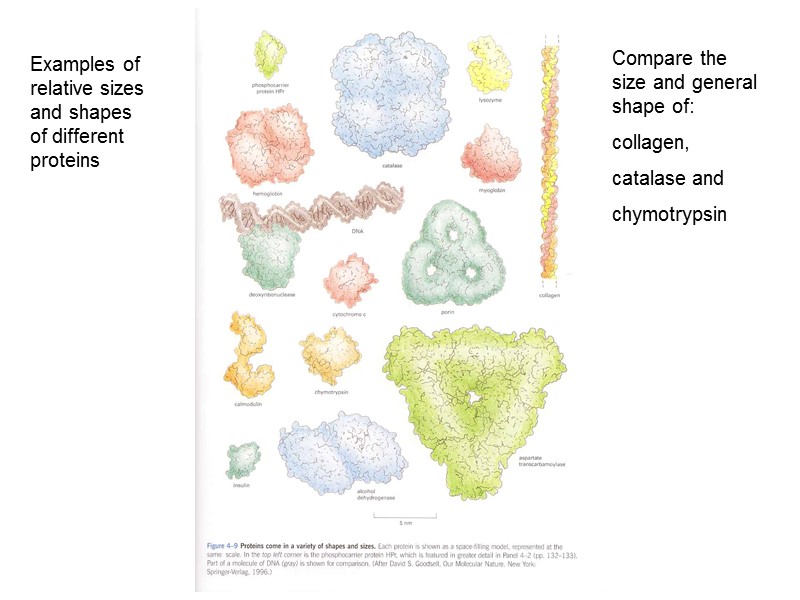
 Examples of relative sizes and shapes of different proteins Compare the size and general shape of: collagen, catalase and chymotrypsin
Examples of relative sizes and shapes of different proteins Compare the size and general shape of: collagen, catalase and chymotrypsin

