a1636fa376302a97813ffa8122a6fc16.ppt
- Количество слайдов: 27
 Lecture 6 Kjemisk reaksjonsteknikk Chemical Reaction Engineering • Review of previous lectures • Pressure drop in fixed bed reactor • PFR reactor design with pressure drop (ε=0) Department of Chemical Engineering 1 - 3/17/2018
Lecture 6 Kjemisk reaksjonsteknikk Chemical Reaction Engineering • Review of previous lectures • Pressure drop in fixed bed reactor • PFR reactor design with pressure drop (ε=0) Department of Chemical Engineering 1 - 3/17/2018
 Reaction Engineering Stoichiometry Rate Laws Department of Chemical Engineering 2 Mole Balance Isothermal reactor design These topics build upon one another 2 - 3/17/2018
Reaction Engineering Stoichiometry Rate Laws Department of Chemical Engineering 2 Mole Balance Isothermal reactor design These topics build upon one another 2 - 3/17/2018
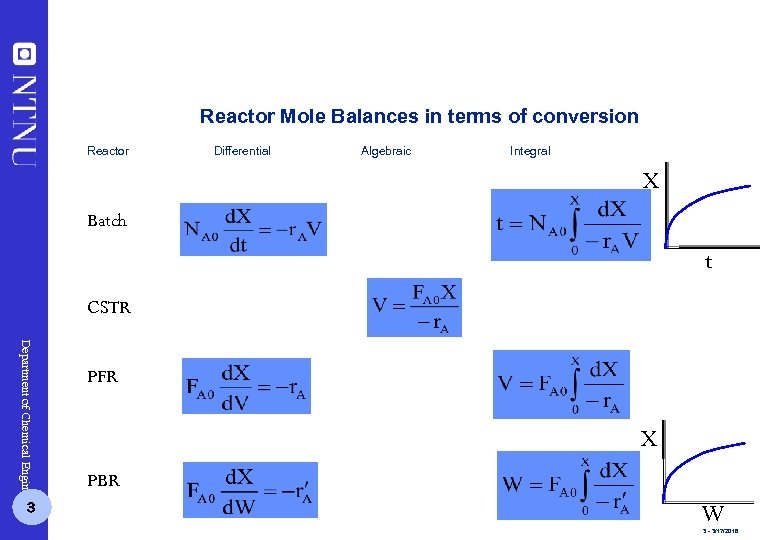 Reactor Mole Balances in terms of conversion Reactor Differential Algebraic Integral X Batch t CSTR Department of Chemical Engineering 3 PFR X PBR W 3 - 3/17/2018
Reactor Mole Balances in terms of conversion Reactor Differential Algebraic Integral X Batch t CSTR Department of Chemical Engineering 3 PFR X PBR W 3 - 3/17/2018
 Concentration Flow System: Gas Phase Flow System: Department of Chemical Engineering 4 4 - 3/17/2018
Concentration Flow System: Gas Phase Flow System: Department of Chemical Engineering 4 4 - 3/17/2018
 Pressure Drop in Packed Bed Reactors Note: Pressure drop does NOT affect liquid phase reactions Sample Question: Department of Chemical Engineering 5 Analyze the following second order gas phase reaction that occurs isothermally in a PBR: 2 A B Mole Balance: Must use the differential form of the mole balance to separate variables: Rate Law: Second order in A and irreversible: 5 - 3/17/2018
Pressure Drop in Packed Bed Reactors Note: Pressure drop does NOT affect liquid phase reactions Sample Question: Department of Chemical Engineering 5 Analyze the following second order gas phase reaction that occurs isothermally in a PBR: 2 A B Mole Balance: Must use the differential form of the mole balance to separate variables: Rate Law: Second order in A and irreversible: 5 - 3/17/2018
 Pressure Drop in Packed Bed Reactors Stoichiometry: Isothermal, T=T 0 Department of Chemical Engineering 6 Combine: Need to find (P/P 0) as a function of W (or V if you have a PFR) 6 - 3/17/2018
Pressure Drop in Packed Bed Reactors Stoichiometry: Isothermal, T=T 0 Department of Chemical Engineering 6 Combine: Need to find (P/P 0) as a function of W (or V if you have a PFR) 6 - 3/17/2018
 Pressure Drop in Packed Bed Reactors Ergun Equation: Department of Chemical Engineering 7 P pressure, Φ porosity (volume of void/total bed volume) 1 - Φ (volume of solid/total bed volume) gc conversion factor. 1. 0 for metric system Dp diameter of particle in bed μ viscosity of gas passing through the bed Z length down the packed bed u, superficial velocity ρ gas density G= ρu superficial mass velocity k. Pa m kg/m. s m m/s kg/m 3 kg/m 2, s 7 - 3/17/2018
Pressure Drop in Packed Bed Reactors Ergun Equation: Department of Chemical Engineering 7 P pressure, Φ porosity (volume of void/total bed volume) 1 - Φ (volume of solid/total bed volume) gc conversion factor. 1. 0 for metric system Dp diameter of particle in bed μ viscosity of gas passing through the bed Z length down the packed bed u, superficial velocity ρ gas density G= ρu superficial mass velocity k. Pa m kg/m. s m m/s kg/m 3 kg/m 2, s 7 - 3/17/2018
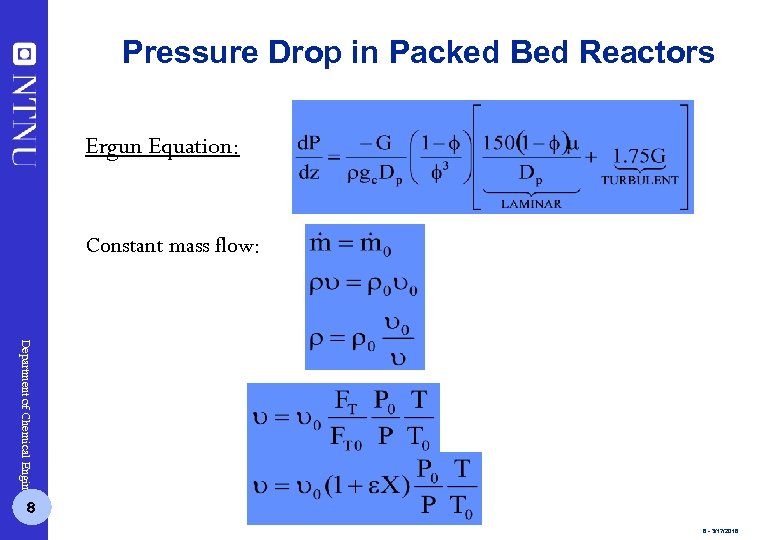 Pressure Drop in Packed Bed Reactors Ergun Equation: Constant mass flow: Department of Chemical Engineering 8 8 - 3/17/2018
Pressure Drop in Packed Bed Reactors Ergun Equation: Constant mass flow: Department of Chemical Engineering 8 8 - 3/17/2018
 Pressure Drop in Packed Bed Reactors Variable Density Department of Chemical Engineering Let 9 9 - 3/17/2018
Pressure Drop in Packed Bed Reactors Variable Density Department of Chemical Engineering Let 9 9 - 3/17/2018
 Pressure Drop in Packed Bed Reactors Catalyst Weight Where Ac, cross section area Department of Chemical Engineering Let 10 10 - 3/17/2018
Pressure Drop in Packed Bed Reactors Catalyst Weight Where Ac, cross section area Department of Chemical Engineering Let 10 10 - 3/17/2018
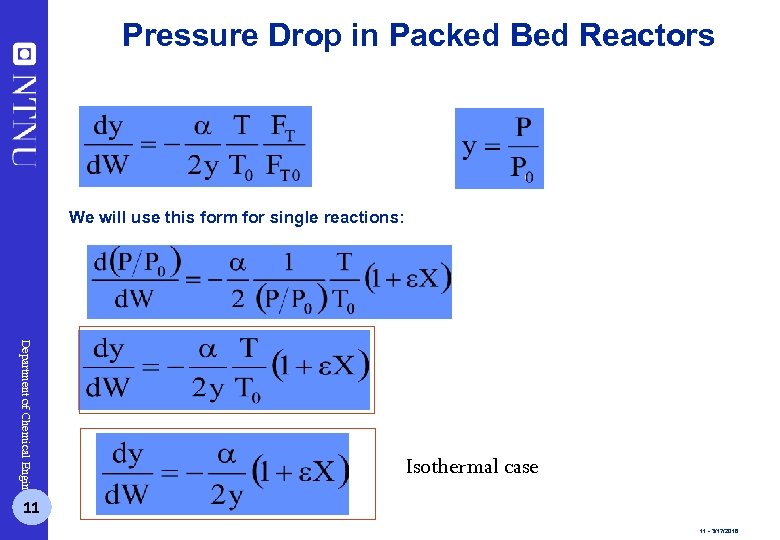 Pressure Drop in Packed Bed Reactors We will use this form for single reactions: Department of Chemical Engineering Isothermal case 11 11 - 3/17/2018
Pressure Drop in Packed Bed Reactors We will use this form for single reactions: Department of Chemical Engineering Isothermal case 11 11 - 3/17/2018
 Pressure Drop in Packed Bed Reactors and or Department of Chemical Engineering The two expressions are coupled ordinary differential equations. We can only solve them simultaneously using an ODE solver such as Polymath. For the special case of isothermal operation and epsilon = 0, we can obtain an analytical solution. Polymath will combine the mole balance, rate law and stoichiometry. 12 12 - 3/17/2018
Pressure Drop in Packed Bed Reactors and or Department of Chemical Engineering The two expressions are coupled ordinary differential equations. We can only solve them simultaneously using an ODE solver such as Polymath. For the special case of isothermal operation and epsilon = 0, we can obtain an analytical solution. Polymath will combine the mole balance, rate law and stoichiometry. 12 12 - 3/17/2018
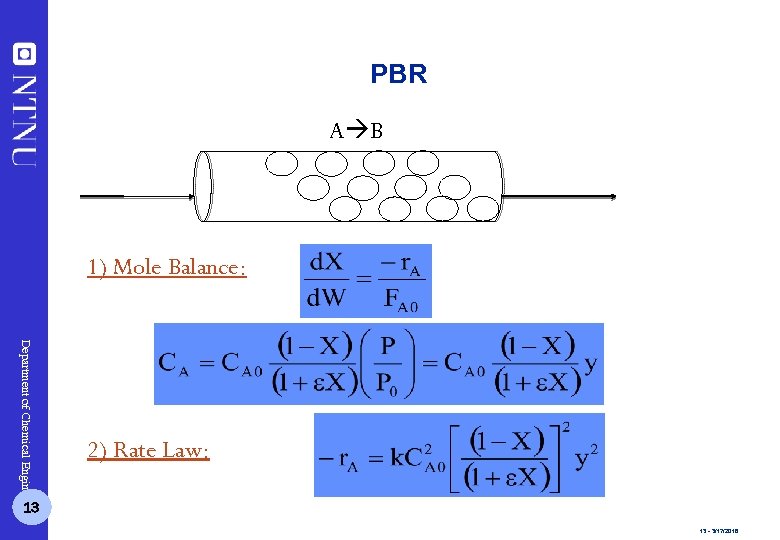 PBR A B 1) Mole Balance: Department of Chemical Engineering 2) Rate Law: 13 13 - 3/17/2018
PBR A B 1) Mole Balance: Department of Chemical Engineering 2) Rate Law: 13 13 - 3/17/2018
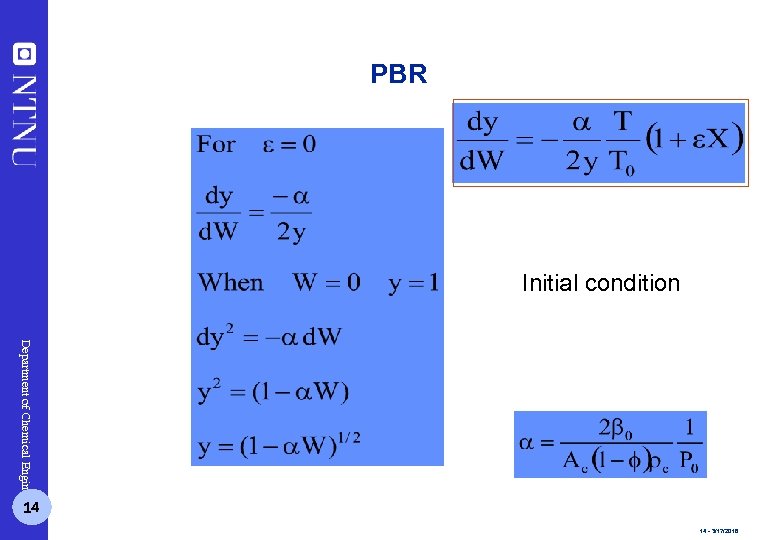 PBR Initial condition Department of Chemical Engineering 14 14 - 3/17/2018
PBR Initial condition Department of Chemical Engineering 14 14 - 3/17/2018
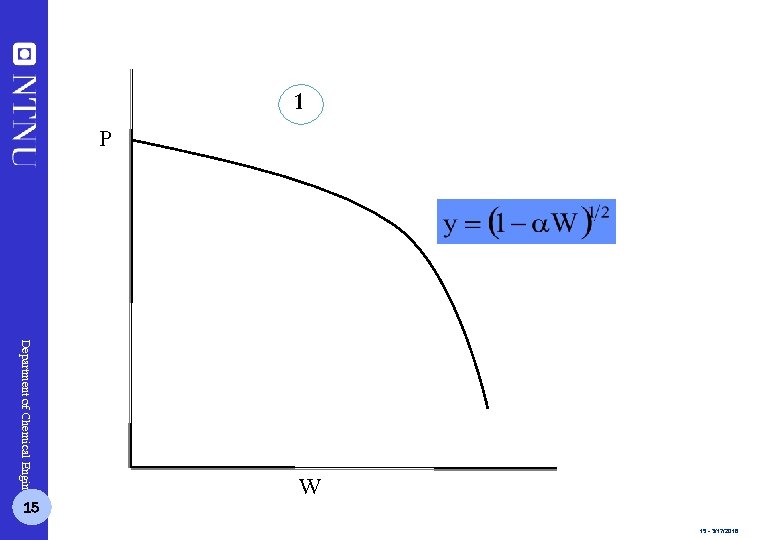 1 P Department of Chemical Engineering 15 W 15 - 3/17/2018
1 P Department of Chemical Engineering 15 W 15 - 3/17/2018
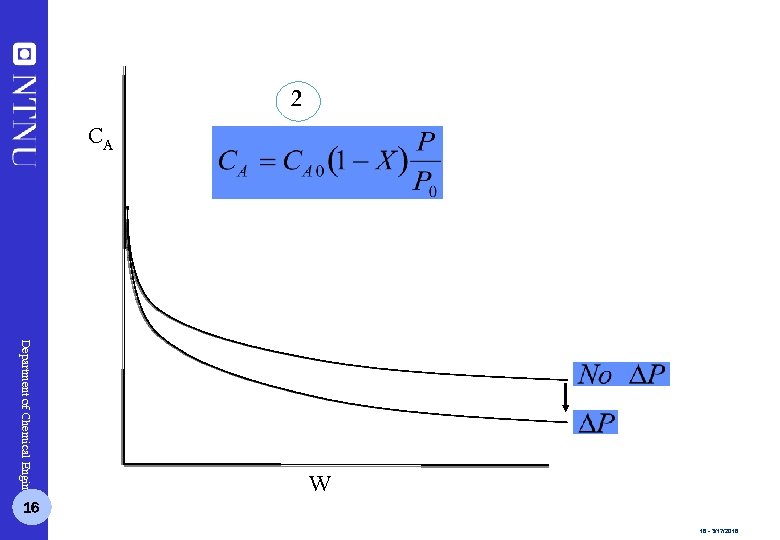 2 CA Department of Chemical Engineering 16 W 16 - 3/17/2018
2 CA Department of Chemical Engineering 16 W 16 - 3/17/2018
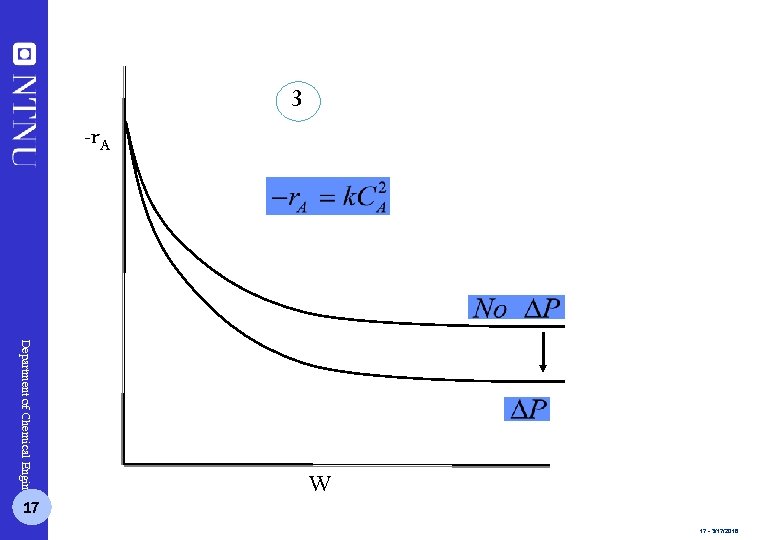 3 -r. A Department of Chemical Engineering 17 W 17 - 3/17/2018
3 -r. A Department of Chemical Engineering 17 W 17 - 3/17/2018
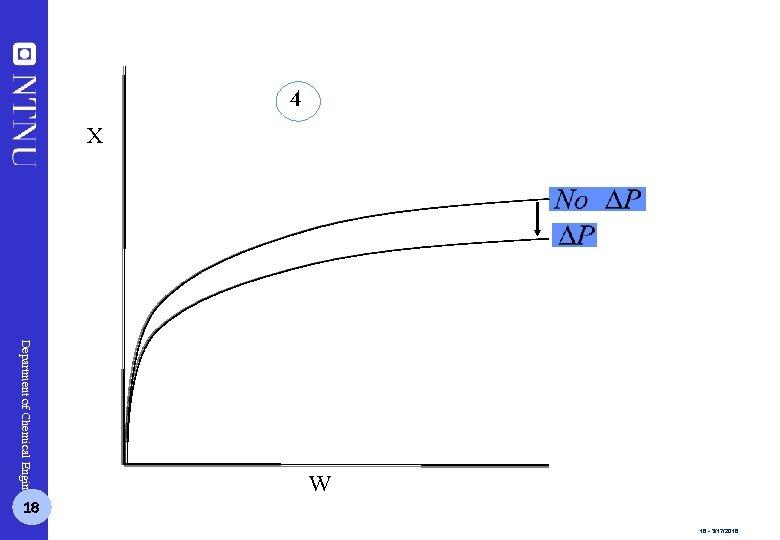 4 X Department of Chemical Engineering 18 W 18 - 3/17/2018
4 X Department of Chemical Engineering 18 W 18 - 3/17/2018
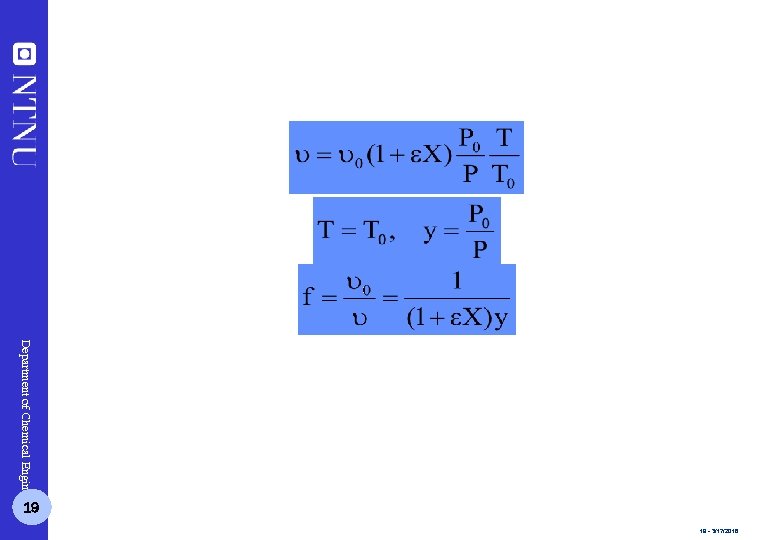 Department of Chemical Engineering 19 19 - 3/17/2018
Department of Chemical Engineering 19 19 - 3/17/2018
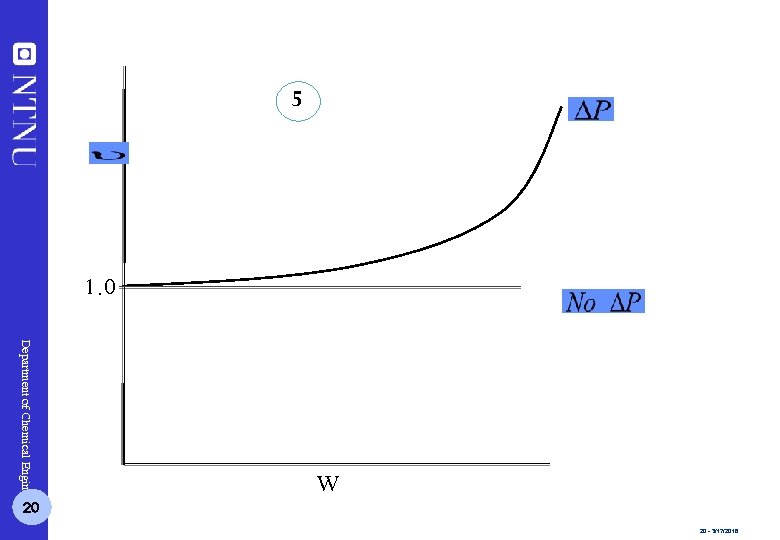 5 1. 0 Department of Chemical Engineering 20 W 20 - 3/17/2018
5 1. 0 Department of Chemical Engineering 20 W 20 - 3/17/2018
 Example 1: Gas Phase Reaction in PBR for δ = 0 Gas Phase Reaction in PBR with δ = 0 (Polymath Solution) A + B 2 C Repeat the previous one with equil molar feed of A and B and k. A = 1. 5 dm 9/mol 2/kg/min α = 0. 0099 kg-1 Find X at 100 kg Department of Chemical Engineering 21 21 - 3/17/2018
Example 1: Gas Phase Reaction in PBR for δ = 0 Gas Phase Reaction in PBR with δ = 0 (Polymath Solution) A + B 2 C Repeat the previous one with equil molar feed of A and B and k. A = 1. 5 dm 9/mol 2/kg/min α = 0. 0099 kg-1 Find X at 100 kg Department of Chemical Engineering 21 21 - 3/17/2018
 Example 1: Gas Phase Reaction in PBR for δ = 0 A + B 2 C Case 1: Department of Chemical Engineering Case 2: 22 22 - 3/17/2018
Example 1: Gas Phase Reaction in PBR for δ = 0 A + B 2 C Case 1: Department of Chemical Engineering Case 2: 22 22 - 3/17/2018
 Example 1: Gas Phase Reaction in PBR for δ = 0 1) Mole Balance: 2) Rate Law: 3) Department of Chemical Engineering 4) 23 23 - 3/17/2018
Example 1: Gas Phase Reaction in PBR for δ = 0 1) Mole Balance: 2) Rate Law: 3) Department of Chemical Engineering 4) 23 23 - 3/17/2018
 Example 1: Gas Phase Reaction in PBR for δ = 0 5) Department of Chemical Engineering 24 24 - 3/17/2018
Example 1: Gas Phase Reaction in PBR for δ = 0 5) Department of Chemical Engineering 24 24 - 3/17/2018
 Example 1: Gas Phase Reaction in PBR for δ = 0 Department of Chemical Engineering 25 25 - 3/17/2018
Example 1: Gas Phase Reaction in PBR for δ = 0 Department of Chemical Engineering 25 25 - 3/17/2018
 Example A + B → 2 C Department of Chemical Engineering 26 26 - 3/17/2018
Example A + B → 2 C Department of Chemical Engineering 26 26 - 3/17/2018
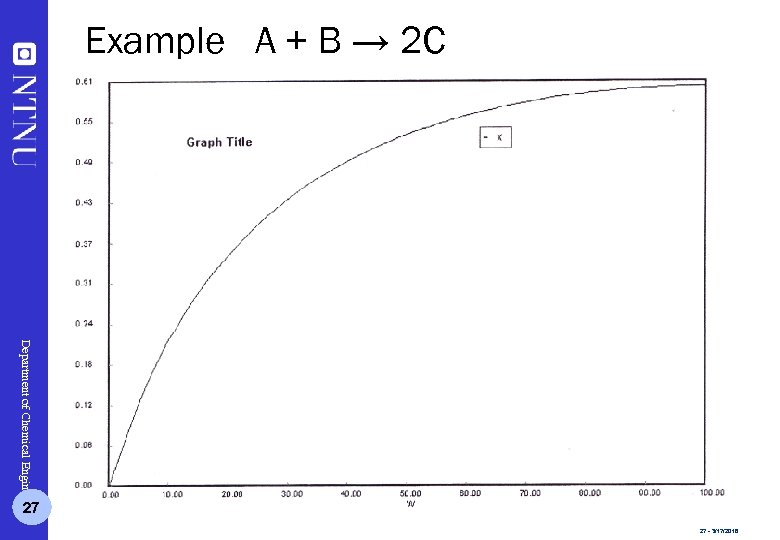 Example A + B → 2 C Department of Chemical Engineering 27 27 - 3/17/2018
Example A + B → 2 C Department of Chemical Engineering 27 27 - 3/17/2018


