da94676851e08ca4c7119b0b9daf27c4.ppt
- Количество слайдов: 109

Lecture 4, January 14, 2011 aufbau principle atoms Nature of the Chemical Bond with applications to catalysis, materials science, nanotechnology, surface science, bioinorganic chemistry, and energy William A. Goddard, III, wag@wag. caltech. edu 316 Beckman Institute, x 3093 Charles and Mary Ferkel Professor of Chemistry, Materials Science, and Applied Physics, California Institute of Technology Teaching Assistants: Wei-Guang Liu <wgliu@wag. caltech. edu> Caitlin Scott <cescott@caltech. edu> Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 1

The excited states of H atom z hφk = ekφk for all excited states k θ We will use spherical polar coordinates, r, θ, φ φ where z=r cosθ, x=r sinθ cosφ, y=r sinθ sinφ x h = - ½ 2 – Z/r is independent of θ and φ which can be seen by noting that 2 = d 2/dx 2 + d 2/dy 2 is independent of θ and φ (it transforms like r 2 = x 2 + y 2 + z 2). Consequently the eigenfunctions of h can be written as a factor depending only on r and a factor depending only on θ and φ φnlm = Rnl(r) Zlm(θ, φ) where the reason for the numbers nlm will become apparent later Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 y 2

The ground state of H atom h = - ½ 2 – Z/r φnlm = Rnl(r) Zlm(θ, φ) z θ φ For the ground state, x R 1 s = exp(-Zr) and Z 1 s = Z 00 = 1 (a constant), where l=0 and m=0 again ignoring normalization. The radial wavefunction is nodeless, as expected The angular function is a constant, which is clearly the best for KE. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 y 3
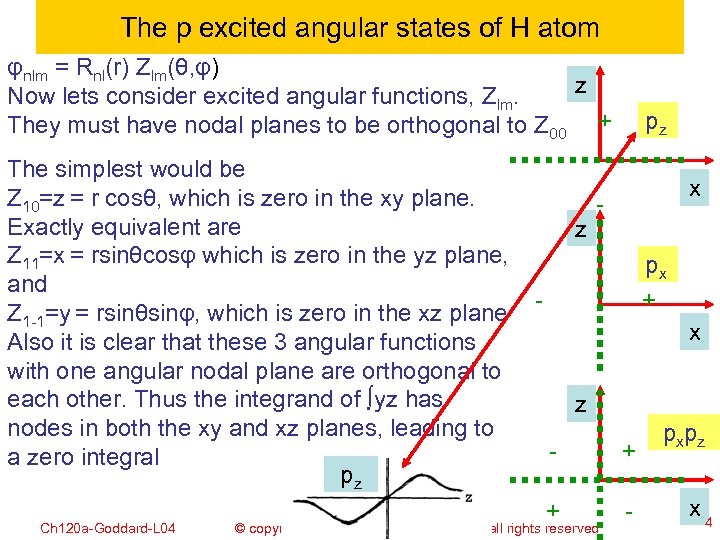
The p excited angular states of H atom φnlm = Rnl(r) Zlm(θ, φ) z Now lets consider excited angular functions, Zlm. They must have nodal planes to be orthogonal to Z 00 + The simplest would be Z 10=z = r cosθ, which is zero in the xy plane. Exactly equivalent are Z 11=x = rsinθcosφ which is zero in the yz plane, and Z 1 -1=y = rsinθsinφ, which is zero in the xz plane Also it is clear that these 3 angular functions with one angular nodal plane are orthogonal to each other. Thus the integrand of ∫yz has nodes in both the xy and xz planes, leading to a zero integral pz Ch 120 a-Goddard-L 04 z pz x px + - x z - + + - © copyright 2010 William A. Goddard III, all rights reserved 2011 p xp z x 4
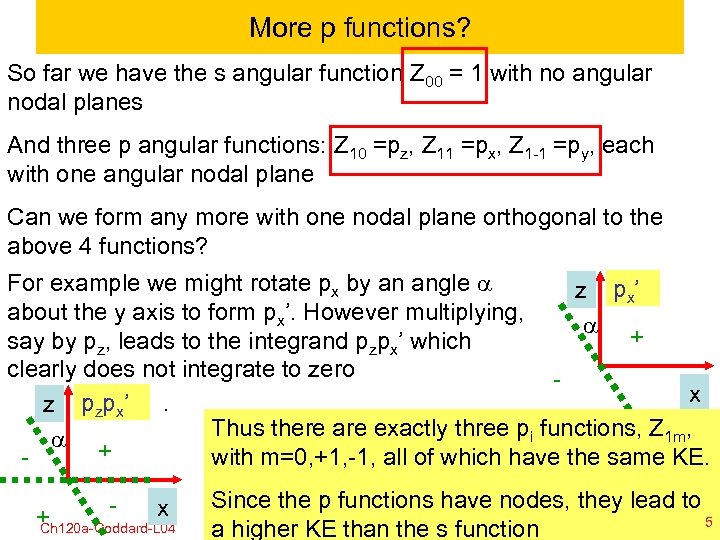
More p functions? So far we have the s angular function Z 00 = 1 with no angular nodal planes And three p angular functions: Z 10 =pz, Z 11 =px, Z 1 -1 =py, each with one angular nodal plane Can we form any more with one nodal plane orthogonal to the above 4 functions? For example we might rotate px by an angle a z p x’ about the y axis to form px’. However multiplying, a + say by pz, leads to the integrand pzpx’ which clearly does not integrate to zero x z pzpx’. Thus there are exactly three pi functions, Z 1 m, a + with m=0, +1, -1, all of which have the same KE. - x + Ch 120 a-Goddard-L 04 Since the p functions have nodes, they lead to 5 © copyright 2010 William the s function 2011 a higher KE than A. Goddard III, all rights reserved
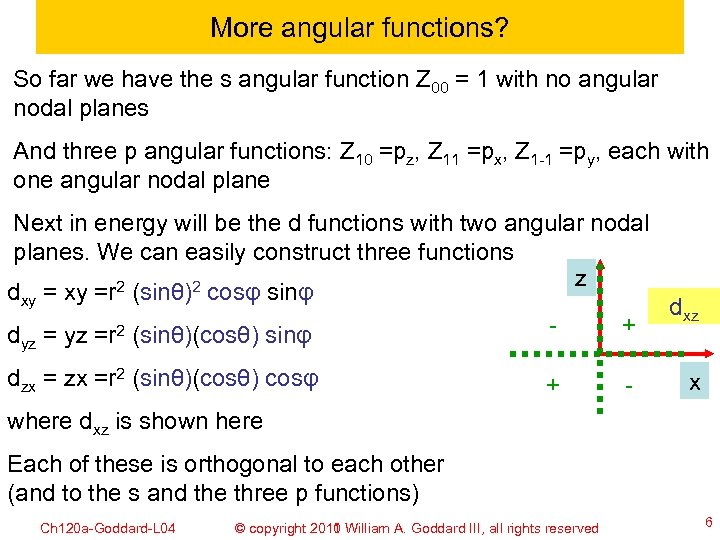
More angular functions? So far we have the s angular function Z 00 = 1 with no angular nodal planes And three p angular functions: Z 10 =pz, Z 11 =px, Z 1 -1 =py, each with one angular nodal plane Next in energy will be the d functions with two angular nodal planes. We can easily construct three functions z dxy =r 2 (sinθ)2 cosφ sinφ dxz + d = yz =r 2 (sinθ)(cosθ) sinφ yz dzx =r 2 (sinθ)(cosθ) cosφ + - x where dxz is shown here Each of these is orthogonal to each other (and to the s and the three p functions) Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 6

More d angular functions? In addition we can construct three other functions with two nodal planes z dx 2 -y 2 = x 2 – y 2 = r 2 (sinθ)2 [(cosφ)2 – (sinφ)2] dz 2 -x 2 + dy 2 -z 2 = y 2 – z 2 = r 2 [(sinθ)2(sinφ)2 – (cosθ)2] dz 2 -x 2 = z 2 – x 2 = r 2 [(cosθ)2 -(sinθ)2(cosφ)2] x where dz 2 -x 2 is shown here, + Each of these three is orthogonal to the previous three d functions (and to the s and the three p functions) This leads to six functions with 2 nodal planes Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 7

More d angular functions? In addition we can construct three other functions with two nodal planes z dx 2 -y 2 = x 2 – y 2 = r 2 (sinθ)2 [(cosφ)2 – (sinφ)2] dz 2 -x 2 + dy 2 -z 2 = y 2 – z 2 = r 2 [(sinθ)2(sinφ)2 – (cosθ)2] dz 2 -x 2 = z 2 – x 2 = r 2 [(cosθ)2 -(sinθ)2(cosφ)2] x where dz 2 -x 2 is shown here, + Each of these three is orthogonal to the previous three d functions (and to the s and the three p functions) This leads to six functions with 2 nodal planes However (x 2 – y 2) + (y 2 – z 2) + (z 2 – x 2) = 0 Which indicates that there are only two independent such functions. We combine the 2 nd two as (z 2 – x 2) - (y 2 – z 2) = [2 z 2 – x 2 - y 2 ] = [3 z 2 – x 2 - y 2 –z 2] = = [3 z 2 – r 2 ] which we denote as dz 2 Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 8

Summarizing we get 5 d angular functions z dz 2 Z 20 = dz 2 = [3 – ] m=0, ds + Z 21 = dzx =r 2 (sinθ)(cosθ) cosφ m = 1, dp - 57º Z 2 -1 = dyz =r 2 (sinθ)(cosθ) sinφ x + Z 22 = dx 2 -y 2 = x 2 – y 2 = r 2 (sinθ)2 [(cosφ)2 – (sinφ)2] m = 2, dd Z 22 = dxy =r 2 (sinθ)2 cosφ sinφ z 2 r 2 We find it useful to keep track of how often the wavefunction changes sign as the φ coordinate is increased by 2 p = 360º When this number, m=0 we call it a s function When m=1 we call it a p function When m=2 we call it a d function When m=3 we call it a copyright 2010 William A. Goddard III, all rights reserved f function Ch 120 a-Goddard-L 04 © 2011 9

Summarizing the angular functions So far we have • one s angular function (no angular nodes) called ℓ=0 • three p angular functions (one angular node) called ℓ=1 • five d angular functions (two angular nodes) called ℓ=2 Continuing we can form • seven f angular functions (three angular nodes) called ℓ=3 • nine g angular functions (four angular nodes) called ℓ=4 where ℓ is referred to as the angular momentum quantum number And there are (2ℓ+1) m values for each ℓ Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 10

The real (Zlm) and complex (Ylm) momentum functions Here the bar over m negative Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 11
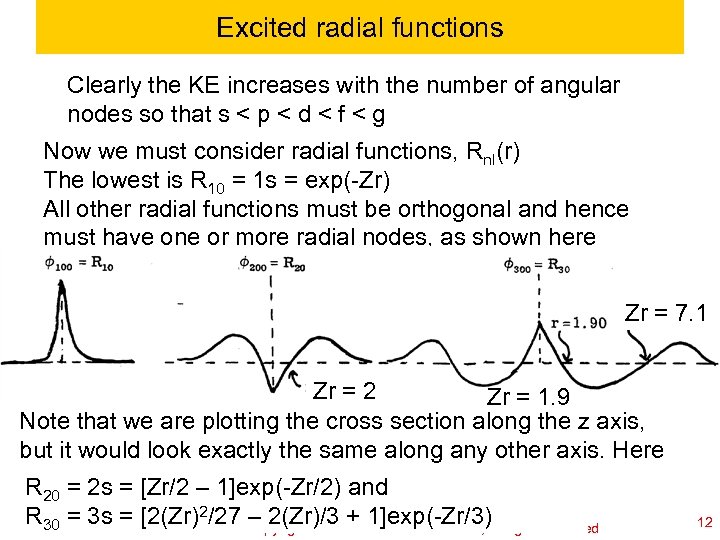
Excited radial functions Clearly the KE increases with the number of angular nodes so that s < p < d < f < g Now we must consider radial functions, Rnl(r) The lowest is R 10 = 1 s = exp(-Zr) All other radial functions must be orthogonal and hence must have one or more radial nodes, as shown here Zr = 7. 1 Zr = 2 Zr = 1. 9 Note that we are plotting the cross section along the z axis, but it would look exactly the same along any other axis. Here R 20 = 2 s = [Zr/2 – 1]exp(-Zr/2) and RCh 120 a-Goddard-L 04 2/27 – 2(Zr)/3 William A. Goddard III, all rights reserved + 1]exp(-Zr/3) 30 = 3 s = [2(Zr) © copyright 2010 2011 12

Size (a 0) name total nodal planes radial nodal planes angular nodal planes Combination of radial and angular nodal planes Combining radial and angular functions gives the various excited states of the H atom. They are named as shown where the n quantum number is the total number of nodal planes plus 1 The nodal theorem does not specify how 2 s and 1 s 0 0 0 1. 0 2 p are related, but it turns out that the total energy 2 s 1 1 0 4. 0 depends only on n. 2 p 1 0 1 4. 0 3 s 2 2 0 9. 0 Enlm = - Z 2/2 n 2 3 p 2 1 1 9. 0 The potential energy is given by 3 d 2 0 2 9. 0 ˉ ˉ 4 s 3 3 0 16. 0 PE = - Z 2/2 n 2 = -Z/ R , where R =n 2/Z 4 p 3 2 1 16. 0 Thus Enlm = - Z/2 Rˉ 4 d 3 1 2 16. 0 13 4 f Ch 120 a-Goddard-L 04 3 0 3 16. 0 © copyright 2010 William A. Goddard III, all rights reserved 2011
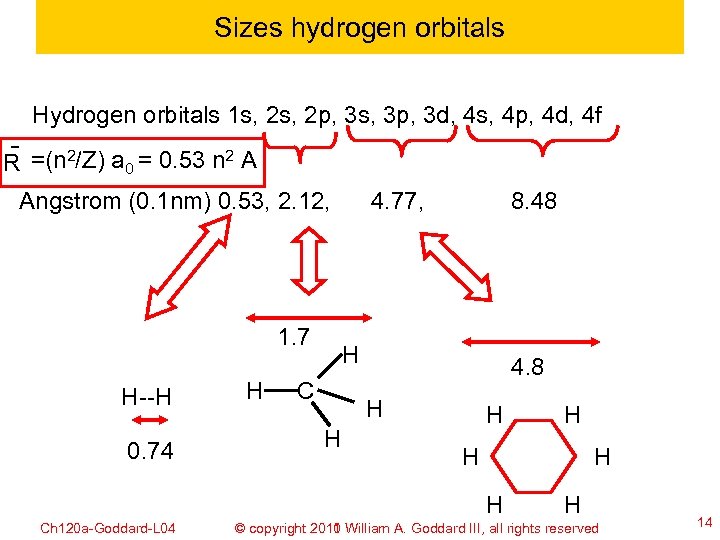
Sizes hydrogen orbitals Hydrogen orbitals 1 s, 2 p, 3 s, 3 p, 3 d, 4 s, 4 p, 4 d, 4 f ˉ R =(n 2/Z) a 0 = 0. 53 n 2 A Angstrom (0. 1 nm) 0. 53, 2. 12, 1. 7 H--H 0. 74 H 4. 77, 8. 48 H C 4. 8 H H H Ch 120 a-Goddard-L 04 H H © copyright 2010 William A. Goddard III, all rights reserved 2011 14

Hydrogen atom excited states Energy Enlm = - Z 2/2 n 2 = -(1/2 n 2)h 0 = 13. 6 e. V/n 2 zero -0. 033 h 0 = -0. 9 e. V -0. 056 h 0 = -1. 5 e. V -0. 125 h 0 = -3. 4 e. V -0. 5 h 0 = -13. 6 e. V Ch 120 a-Goddard-L 04 4 s 4 p 4 d 3 s 3 p 3 d 2 s 2 p 1 s 4 f © copyright 2010 William A. Goddard III, all rights reserved 2011 15
Plotting of orbitals: line cross-section vs. contour Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 16
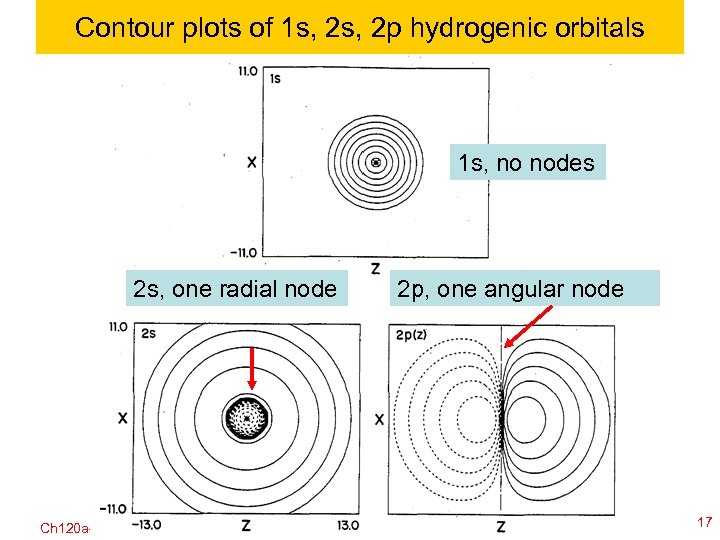
Contour plots of 1 s, 2 p hydrogenic orbitals 1 s, no nodes 2 s, one radial node Ch 120 a-Goddard-L 04 2 p, one angular node © copyright 2010 William A. Goddard III, all rights reserved 2011 17
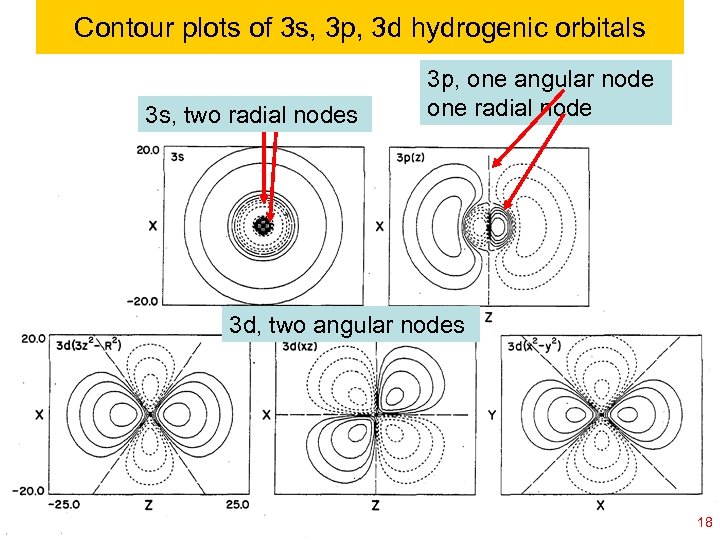
Contour plots of 3 s, 3 p, 3 d hydrogenic orbitals 3 s, two radial nodes 3 p, one angular node one radial node 3 d, two angular nodes Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 18
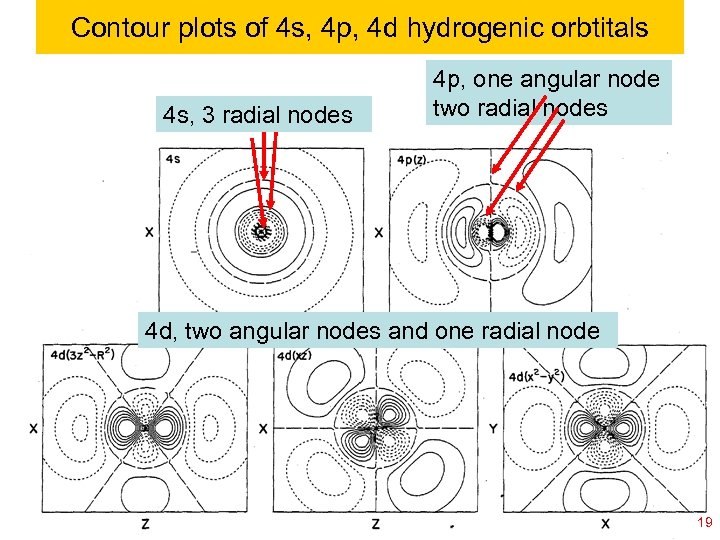
Contour plots of 4 s, 4 p, 4 d hydrogenic orbtitals 4 s, 3 radial nodes 4 p, one angular node two radial nodes 4 d, two angular nodes and one radial node Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 19
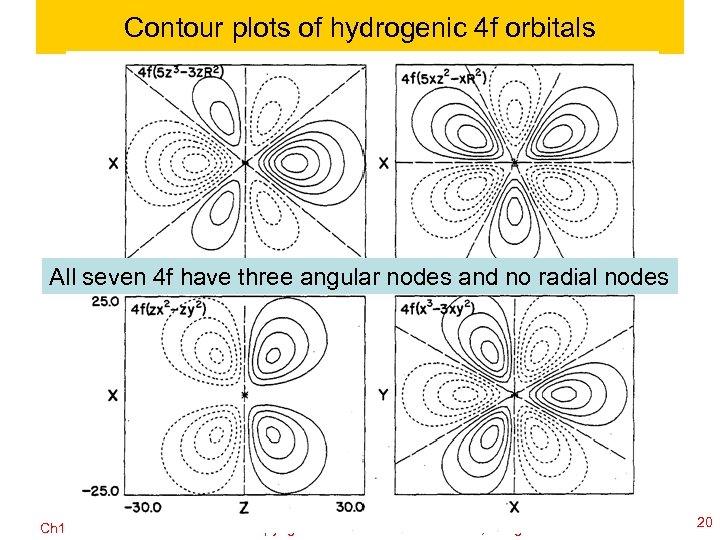
Contour plots of hydrogenic 4 f orbitals All seven 4 f have three angular nodes and no radial nodes Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 20

He atom – describe using He+ orbitals With 2 electrons, we can form the ground state of He by putting both electrons in the He+ 1 s orbital, just like the MO state of H 2 ΨHe(1, 2) = A[(Φ 1 sa)(Φ 1 sb)]= Φ 1 s(1)Φ 1 s(2) (ab-ba) EHe= 2<1 s|h|1 s> + J 1 s, 1 s two one-electron energies one Coulomb repulsion Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 21

He atom – describe using He+ orbitals With 2 electrons, we can form the ground state of He by putting both electrons in the He+ 1 s orbital, just like the MO state of H 2 ΨHe(1, 2) = A[(Φ 1 sa)(Φ 1 sb)]= Φ 1 s(1)Φ 1 s(2) (ab-ba) EHe= 2<1 s|h|1 s> + J 1 s, 1 s First lets review the energy for He+. Writing Φ 1 s = exp(-zr) we determine the optimum z for He+ as follows <1 s|KE|1 s> = + ½ z 2 (goes as the square of 1/size) <1 s|PE|1 s> = - Zz (linear in 1/size) Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 22

He atom – describe using He+ orbitals With 2 electrons, we can form the ground state of He by putting both electrons in the He+ 1 s orbital, just like the MO state of H 2 ΨHe(1, 2) = A[(Φ 1 sa)(Φ 1 sb)]= Φ 1 s(1)Φ 1 s(2) (ab-ba) EHe= 2<1 s|h|1 s> + J 1 s, 1 s First lets review the energy for He+. Writing Φ 1 s = exp(-zr) we determine the optimum z for He+ as follows <1 s|KE|1 s> = + ½ z 2 (goes as the square of 1/size) <1 s|PE|1 s> = - Zz (linear in 1/size) Applying the variational principle, the optimum z must satisfy d. E/dz = z - Z = 0 leading to z = Z, This value of z leads to KE = ½ Z 2, PE = -Z 2, E=-Z 2/2 = -2 h 0. write PE=-Z/R 0, so that the average radius is R 0=1/z Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 23

J 1 s, 1 s = e-e energy of He atom – He+ orbitals Now consider He atom: EHe = 2(½ z 2) – 2 Zz + J 1 s, 1 s How can we estimate J 1 s, 1 s e 1 R 0 e 2 Assume that each electron moves on a sphere With the average radius R 0 = 1/z And assume that e 1 at along the z axis (θ=0) Neglecting correlation in the electron motions, e 2 will on the average have θ=90º so that the average e 1 -e 2 distance is ~sqrt(2)*R 0 Thus J 1 s, 1 s ~ 1/[sqrt(2)*R 0] = 0. 71 z A rigorous calculation (notes chapter 3, appendix 3 -C page 6) Gives J 1 s, 1 s = (5/8) z Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 24

The optimum atomic orbital for He atom: EHe = 2(½ z 2) – 2 Zz + (5/8)z Applying the variational principle, the optimum z must satisfy d. E/dz = 0 leading to 2 z - 2 Z + (5/8) = 0 Thus z = (Z – 5/16) = 1. 6875 KE = 2(½ z 2) = z 2 PE = - 2 Zz + (5/8)z = -2 z 2 E= - z 2 = -2. 8477 h 0 Ignoring e-e interactions the energy would have been E = -4 h 0 The exact energy is E = -2. 9037 h 0 (from memory, TA please check). Thus this simple approximation accounts for 98. 1% of the exact result. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 25

Interpretation: The optimum atomic orbital for He atom ΨHe(1, 2) = Φ 1 s(1)Φ 1 s(2) with Φ 1 s = exp(-zr) We find that the optimum z = (Z – 5/16) = 1. 6875 With this value of z, the total energy is E= - z 2 = -2. 8477 h 0 This wavefunction can be interpreted as two electrons moving independently in the orbital Φ 1 s = exp(-zr) which has been adjusted to account for the average shielding due to the other electron in this orbital. On the average this other electron is closer to the nucleus about 31% of the time so that the effective charge seen by each electron is 2. 00 -0. 31=1. 69 The total energy is just the sum of the individual energies. Ionizing the 2 nd electron, the 1 st electron readjusts to z = Z = 2 With E(He+) = -Z 2/2 = - 2 h 0. thus the ionization potential (IP) is. Ch 120 a-Goddard-L 04 23. 1 e. V (exact value. A. = 24. 6 III, all rights reserved 0. 8477 h 0 = © copyright 2010 William Goddard e. V) 2011 26

Now lets add a 3 rd electron to form Li ΨLi(1, 2, 3) = A[(Φ 1 sa)(Φ 1 sb)(Φ 1 sg)] Problem with either g = a or g = b, we get ΨLi(1, 2, 3) = 0 This is an essential result of the Pauli principle Thus the 3 rd electron must go into an excited orbital ΨLi(1, 2, 3) = A[(Φ 1 sa)(Φ 1 sb) )(Φ 2 sa)] or ΨLi(1, 2, 3) = A[(Φ 1 sa)(Φ 1 sb) )(Φ 2 pza)] (or 2 px or 2 py) First consider Li+ with ΨLi(1, 2, 3) = A[(Φ 1 sa)(Φ 1 sb)] Here Φ 1 s = exp(-zr) with z = Z-0. 3125 = 2. 69 and E = -z 2 = -7. 2226 h 0. Since the E (Li 2+) = -9/2 = -4. 5 h 0 the IP = 2. 7226 h 0 = 74. 1 e. V The size of the 1 s orbital is R 0 = 1/z = 0. 372 a 0 = 0. 2 A Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 27
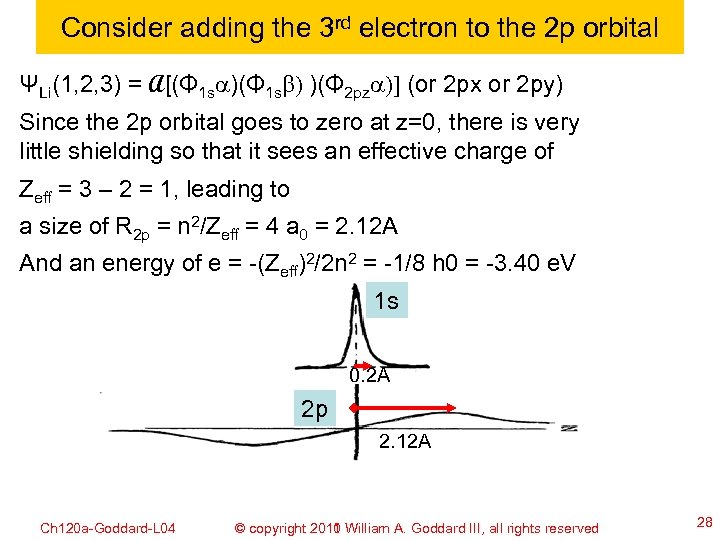
Consider adding the 3 rd electron to the 2 p orbital ΨLi(1, 2, 3) = A[(Φ 1 sa)(Φ 1 sb) )(Φ 2 pza)] (or 2 px or 2 py) Since the 2 p orbital goes to zero at z=0, there is very little shielding so that it sees an effective charge of Zeff = 3 – 2 = 1, leading to a size of R 2 p = n 2/Zeff = 4 a 0 = 2. 12 A And an energy of e = -(Zeff)2/2 n 2 = -1/8 h 0 = -3. 40 e. V 1 s 0. 2 A 2 p 2. 12 A Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 28
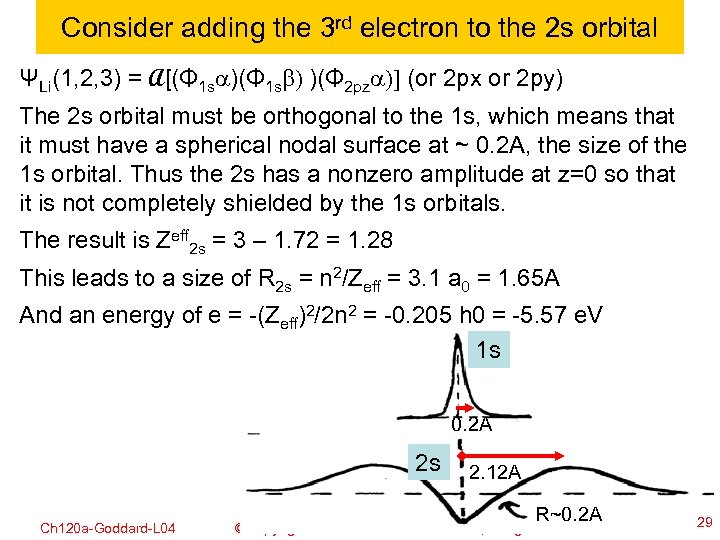
Consider adding the 3 rd electron to the 2 s orbital ΨLi(1, 2, 3) = A[(Φ 1 sa)(Φ 1 sb) )(Φ 2 pza)] (or 2 px or 2 py) The 2 s orbital must be orthogonal to the 1 s, which means that it must have a spherical nodal surface at ~ 0. 2 A, the size of the 1 s orbital. Thus the 2 s has a nonzero amplitude at z=0 so that it is not completely shielded by the 1 s orbitals. The result is Zeff 2 s = 3 – 1. 72 = 1. 28 This leads to a size of R 2 s = n 2/Zeff = 3. 1 a 0 = 1. 65 A And an energy of e = -(Zeff)2/2 n 2 = -0. 205 h 0 = -5. 57 e. V 1 s 0. 2 A 2 s Ch 120 a-Goddard-L 04 2. 12 A R~0. 2 A © copyright 2010 William A. Goddard III, all rights reserved 2011 29
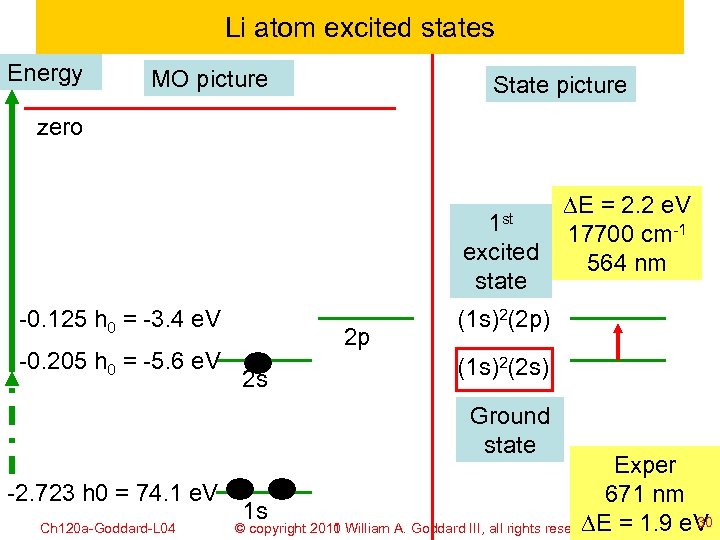
Li atom excited states Energy MO picture State picture zero 1 st excited state -0. 125 h 0 = -3. 4 e. V -0. 205 h 0 = -5. 6 e. V 2 p 2 s DE = 2. 2 e. V 17700 cm-1 564 nm (1 s)2(2 p) (1 s)2(2 s) Ground state Exper -2. 723 h 0 = 74. 1 e. V 671 nm 1 s 30 DE Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved = 1. 9 e. V 2011
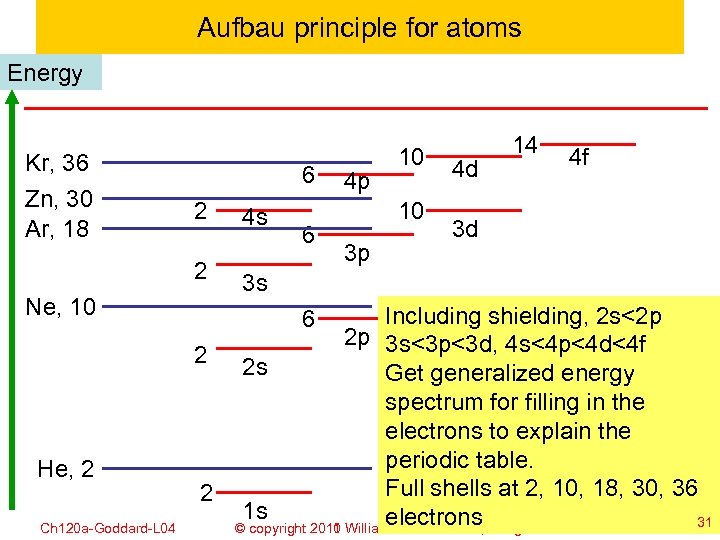
Aufbau principle for atoms Energy Kr, 36 Zn, 30 Ar, 18 6 2 2 Ne, 10 He, 2 Ch 120 a-Goddard-L 04 4 s 6 4 p 10 10 3 p 4 d 14 4 f 3 d 3 s Including shielding, 2 s<2 p 2 p 3 s<3 p<3 d, 4 s<4 p<4 d<4 f 2 2 s Get generalized energy spectrum for filling in the electrons to explain the periodic table. Full shells at 2, 10, 18, 30, 36 2 1 s electrons 31 © copyright 2010 William A. Goddard III, all rights reserved 2011 6
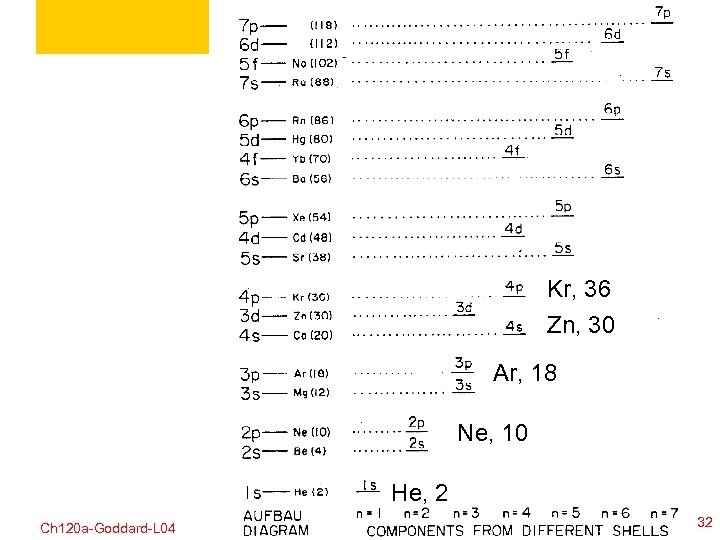
Kr, 36 Zn, 30 Ar, 18 Ne, 10 He, 2 Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 32

General trends along a row of the periodic table As we fill a shell, thus B(2 s)2(2 p)1 to Ne (2 s)2(2 p)6 For each atom we add one more proton to the nucleus and one more electron to the valence shell But the valence electrons only partially shield each other. Thus Zeff increases leading to a decrease in the radius ~ n 2/Zeff And an increase in the IP ~ (Zeff)2/2 n 2 Example Zeff 2 s= 1. 28 Li, 1. 92 Be, 2. 28 B, 2. 64 C, 3. 00 N, 3. 36 O, 4. 00 F, 4. 64 Ne Thus (2 s Li)/(2 s Ne) ~ 4. 64/1. 28 = 3. 6 Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 33

Many-electron configurations General aufbau ordering Ch 120 a-Goddard-L 04 Particularly stable © copyright 2010 William A. Goddard III, all rights reserved 2011 34

General trends along a column of the periodic table As we go down a column Li [He}(2 s) to Na [Ne]3 s to K [Ar]4 s to Rb [Kr]5 s to Cs[Xe]6 s Things get more complicated The radius ~ n 2/Zeff And the IP ~ (Zeff)2/2 n 2 But the Zeff tends to increase, partially compensating for the change in n so that the atomic sizes increase only slowly as we go down the periodic table and The IP decrease only slowly (in e. V): 5. 39 Li, 5. 14 Na, 4. 34 K, 4. 18 Rb, 3. 89 Cs (13. 6 H), 17. 4 F, 13. 0 Cl, 11. 8 Br, 10. 5 I, 9. 5 At Ch 120 a-Goddard-L 04 © copyright 2011 24. 5 He, 21. 6 Ne, 15. 8 Ar, 2010 William A. Goddard III, all rights reserved 14. 0 Kr, 12. 1 Xe, 10. 7 Rn 35
Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 36
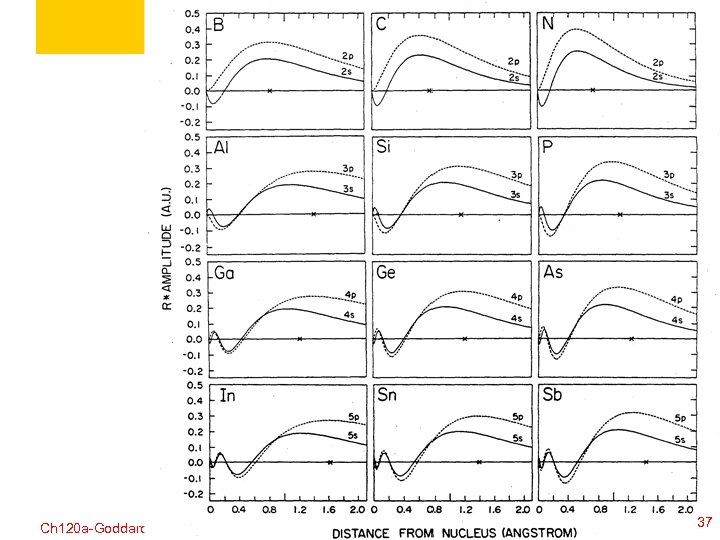
Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 37
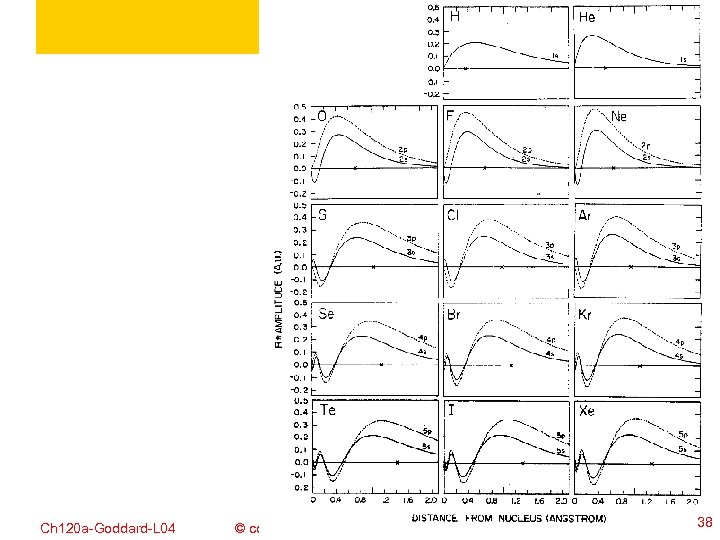
Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 38
![Transition metals; consider [Ar] plus one electron [IP 4 s = (Zeff 4 s Transition metals; consider [Ar] plus one electron [IP 4 s = (Zeff 4 s](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-39.jpg)
Transition metals; consider [Ar] plus one electron [IP 4 s = (Zeff 4 s )2/2 n 2 = 4. 34 e. V Zeff 4 s = 2. 26; 4 s<4 p<3 d K IP 4 p = (Zeff 4 p )2/2 n 2 = 2. 73 e. V Zeff 4 p = 1. 79; IP 3 d = (Zeff 3 d )2/2 n 2 = 1. 67 e. V Zeff 3 d = 1. 05; IP 4 s = (Zeff 4 s )2/2 n 2 = 11. 87 e. V Zeff 4 s = 3. 74; 4 s<3 d<4 p Ca+ IP 3 d = (Zeff 3 d )2/2 n 2 = 10. 17 e. V Zeff 3 d = 2. 59; IP 4 p = (Zeff 4 p )2/2 n 2 = 8. 73 e. V Zeff 4 p = 3. 20; IP 3 d = (Zeff 3 d )2/2 n 2 = 24. 75 e. V Zeff 3 d = 4. 05; 3 d<4 s<4 p Sc++ IP 4 s = (Zeff 4 s )2/2 n 2 = 21. 58 e. V Zeff 4 s = 5. 04; IP 4 p = (Zeff 4 p )2/2 n 2 = 17. 01 e. V Zeff 4 p = 4. 47; As the net charge increases the differential shielding for 4 s vs 3 d is less important than the difference in n quantum number 3 vs 4 39 Ch 120 a-Goddard-L 04 copyright 2010 William A. 2011 Thus charged system©prefers 3 d vs 4 s. Goddard III, all rights reserved

Transition metals; consider Sc 0, Sc+, Sc 2+ 3 d: IP 3 d = (Zeff 3 d )2/2 n 2 = 24. 75 e. V Zeff 3 d = 4. 05; Sc++ 4 s: IP 4 s = (Zeff 4 s )2/2 n 2 = 21. 58 e. V Zeff 4 s = 5. 04; 4 p: IP 4 p = (Zeff 4 p )2/2 n 2 = 17. 01 e. V Zeff 4 p = 4. 47; (3 d)(4 s): IP 4 s = (Zeff 4 s )2/2 n 2 = 12. 89 e. V Zeff 4 s = 3. 89; Sc+ (3 d)2: IP 3 d = (Zeff 3 d )2/2 n 2 = 12. 28 e. V Zeff 3 d = 2. 85; (3 d)(4 p): IP 4 p = (Zeff 4 p )2/2 n 2 = 9. 66 e. V Zeff 4 p = 3. 37; (3 d)(4 s)2: IP 4 s = (Zeff 4 s )2/2 n 2 = 6. 56 e. V Zeff 4 s = 2. 78; Sc (4 s)(3 d)2: IP 3 d = (Zeff 3 d )2/2 n 2 = 5. 12 e. V Zeff 3 d = 1. 84; (3 d)(4 s)(4 p): IP 4 p = (Zeff 4 p )2/2 n 2 = 4. 59 e. V Zeff 4 p = 2. 32; As increase net charge the increases in the differential shielding for 4 s vs 3 d is less important than the difference in n quantum number 3 vs 4. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 40

Implications on transition metals The simple Aufbau principle puts 4 s below 3 d But increasing the charge tends to prefers 3 d vs 4 s. Thus Ground state of Sc 2+ , Ti 2+ …. . Zn 2+ are all (3 d)n For all neutral elements K through Zn the 4 s orbital is easiest to ionize. This is because of increase in relative stability of 3 d for higher ions Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 41
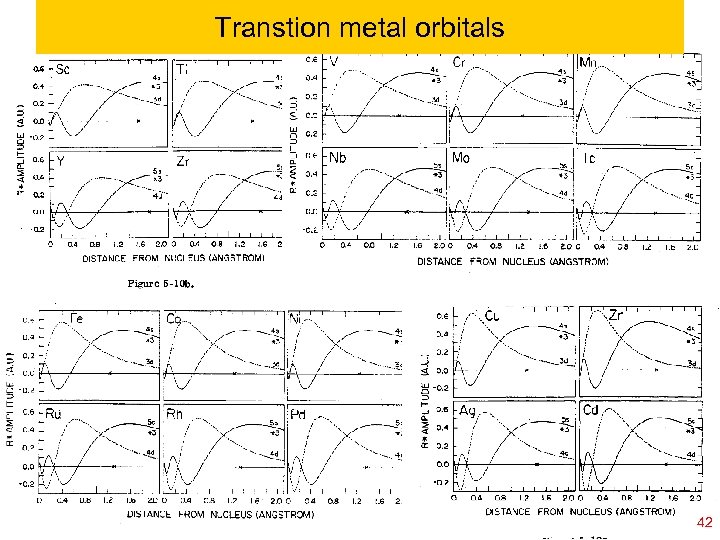
Transtion metal orbitals Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 42

More detailed description of first row atoms Li: (2 s) Be: (2 s)2 B: [Be](2 p)1 C: [Be](2 p)2 N: [Be](2 p)3 O: [Be](2 p)4 F: [Be](2 p)5 Ne: [Be](2 p)6 Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 43
x Ignore the [Be] core then 1 Consider the ground state of B: [Be](2 p)x Ignore the [Be] core then](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-44.jpg)
1 Consider the ground state of B: [Be](2 p)x Ignore the [Be] core then Can put 1 electron in 2 px, 2 py, or 2 pz each with either up or down spin. Thus get 6 states. 2 px We will depict these states by simplified contour diagrams in the xz plane, as at the right. 2 pz Of course 2 py is zero on this plane. Instead we show it as a circle as if you can see just the front part of the lobe sticking out of the paper. z 2 py Because there are 3 degenerate states we denote this as a P state. Because the spin can be +½ or –½, we call it a spin doublet 2 44 and we denote the overall state William. P Goddard III, all rights reserved Ch 120 a-Goddard-L 04 © copyright 2010 as 2011 A.
2 x Ignore the [Be] core then Consider the ground state of C: [Be](2 p)2 x Ignore the [Be] core then](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-45.jpg)
Consider the ground state of C: [Be](2 p)2 x Ignore the [Be] core then Can put 2 electrons in 2 px, 2 py, or 2 pz each with both up and down spin. x Or can put one electron in each of two orbitals: (2 px)(2 py), (2 px), (2 py)(2 pz), We will depict these states by simplified contour diagrams in the xz plane, as at the right. z (2 px)2 (2 pz)2 z x (2 px)(2 pz) Which state is better? The difference is in the electron-electron repulsion: 1/r 12 Clearly two electrons in the same orbital have a much smaller average r 12 and hence a much higher e-e repusion. Thus the Ch 120 a-Goddard-L 04 © electron William A. Goddard III, all orbital 2011 ground state has each copyright 2010 in a different 2 prights reserved z 45

Consider the states of C: formed from (x)(y), (x)(z), (y)(z) Consider first (x)(y): can form two spatial products: Φx(1)Φy(2) and Φy(1)Φx(2) x (2 px)(2 py) These are not spatially symmetric, thus combine Φ(1, 2)s=φx(1) φy(2) + φy(1) φx(2) y Φ(1, 2)a= φx(1) φy(2) - φy(1) φx(2) Which state is better? The difference is in the electron-electron repulsion: 1/r 12 To analyze this, expand the orbitals in terms of the angular coordinates, r, θ, φ Φ(1, 2)s= f(r 1)f(r 2)(sinθ 1)(sinθ 2)[(cosφ1)(sinφ2)+(sinφ1) (cosφ2)] =f(r 1)f(r 2)(sinθ 1)(sinθ 2)[sin(φ1+φ2)] Φ(1, 2)a= f(r 1)f(r 2)(sinθ 1)(sinθ 2)[(cosφ1)(sinφ2)-(sinφ1) (cosφ2)] © copyright 2010 William A. )] 2011 =f(r )(sinθ )[sin(φ -φ Goddard III, all rights reserved Ch 120 a-Goddard-L 04 46

Consider the symmetric and antisymmetric combinations of (x)(y) Φ(1, 2)s= f(r 1)f(r 2)(sinθ 1)(sinθ 2)[sin(φ1+φ2)] x Φ(1, 2)a= f(r 1)f(r 2)(sinθ 1)(sinθ 2)[sin(φ2 -φ1)] (2 px)(2 py) The big difference is that Φ(1, 2)a = 0 when φ2 = φ1 and is a maximum for φ2 and φ1 out of phase by p/2. y But for Φ(1, 2)s the probability of φ2 = φ1 is comparable to that of being out of phase by p/2. Thus the best combination is Φ(1, 2)a Combining with the spin parts we get [φx(1) φy(2) + φy(1) φx(2)](ab-ba) or spin = S = 0 [φx(1) φy(2) - φy(1) φx(2)](ab+ba), also aa and bb or spin = S = 1 Thus for 2 electrons in orthogonal orbitals, high spin is best because the electrons can never be at same spot at the same time Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 47

Summarizing the states for C atom Ground state: three triplet states=2 L=1. Thus L=1, denote as 3 P x (xy-yx) ≡ [x(1)y(2)-y(1)x(2)] (2 px)(2 py) (xz-zx) (yz-zy) y Next state: five singlet states=2 L+1. Thus L=2, denote as 1 D (xy+yx) (xz+zx) (yz+zy) (xx-yy) (2 zz-xx-yy) Highest state: one singlet=2 L+1. thus L=0. Denote as 1 S (zz+xx+yy) Hund’s rule. Given n electrons distributed among m equivalent orgthogonal orbitals, the ground state is the one with the highest possible spin. Given more than one state with the highest spin, the. Ch 120 a-Goddard-L 04 angular momentum Goddard III, GS reserved highest orbital is the all rights © copyright 2010 William A. 2011 48

Calculating energies for C atom The energy of xy is Exy = hxx + hyy + Jxy = 2 hpp +Jxy x (2 px)(2 py) Thus the energy of the 3 P state is E(3 P) = Exy – Kxy = 2 hpp +Jxy - Kxy y For the (xy+yx) component of the 1 D state, we get E(1 D) = Exy + Kxy = 2 hpp +Jxy + Kxy Whereas for the (xx-yy) component of the 1 D state, we get E(1 D) = Exx - Kxy = 2 hpp +Jxx - Kxy This means that Jxx - Kxy = Jxy + Kxy so that Jxx = Jxy + 2 Kxy Also for (2 zz-xx-yy) we obtain E = 2 hpp +Jxx - Kxy For (zz+xx+yy) we obtain E(1 Ch 120 a-Goddard-L 04 + 2© Kxy 2010 William A. Goddard III, all rights reserved S) = 2 hpp + Jxx copyright 2011 49

Summarizing the energies for C atom E(1 S) = 2 hpp + Jxx + 2 Kxy 3 Kxy E(1 D) = 2 hpp +Jxx - Kxy = 2 hpp +Jxy + Kxy 2 Kxy E(3 P) = 2 hpp + Exy – Kxy = 2 hpp +Jxy - Kxy Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 50

Comparison with experiment E(1 S) 3 Kxy E(1 D) 2 Kxy E(3 P) C Si Ge Sn Pb TA’s look up data and list excitation energies in e. V and Kxy in e. V. Get data from Moore’s tables or paper by Harding and Goddard Ann Rev Phys Chem ~1985) Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 51

Summary ground state for C atom Ψ(1, 2, 3, 4, 5, 6)xz= A[(1 sa)(1 sb)(2 sa)(2 sb)(2 pxa)(2 pza)] = = A[(1 s)2(2 pxa)(2 pza)] =A[(Be)(2 pxa)(2 pza)] = (2 px)(2 pz) = A[(xa)(za)] = which we visualize as Ψ(1, 2, 3, 4, 5, 6)xy = A[(xa)(ya)] x x z which we visualize as z Ψ(1, 2, 3, 4, 5, 6)yz = A[(ya)(za)] which we visualize as x z Ch 120 a-Goddard-L 04 Note that we choose to use the xz plane for all 3 wavefunctions, so that the py orbitals look like circles (seeing only the + lobe out of the plane © copyright 2010 William A. Goddard III, all rights reserved 2011 52
3 Ignore the [Be] core then Can Consider the ground state of N: [Be](2 p)3 Ignore the [Be] core then Can](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-53.jpg)
Consider the ground state of N: [Be](2 p)3 Ignore the [Be] core then Can put one electron in each of three orbitals: (2 px)(2 py)(2 pz) Or can put 2 in 1 and 1 in another: (x)2(y), (x)2(z), (y)2(x), (y)2(z), (z)2(x), (z)2(y) As we saw for C, the best state is (x)(y)(z) because of the lowest ee repulsion. xyz can be combined with various spin functions, but from Hund’s rule we expect A[(xa)(ya)(za)] = [Axyz]aaa to be the ground state. (here Axyz is the antisymmetric combination of x(1)y(2)x(3)] The four symmetric spin functions are aaa, aab + aba + baa, abb + bba + bab, bbb With Ms = 3/2, 1/2, -1/2 -3/2, which refer to as S=3/2 or quartet Since there is only one xyz state = 2 L+1 with L=0, we denote it as 53 Ch 120 a-Goddard-L 04 copyright 2011 L=0, leading to the 4 S©state. 2010 William A. Goddard III, all rights reserved
![Energy of the ground state of N: A[(xa)(ya)(za)] = [Axyz]aaa x Simple product xyz Energy of the ground state of N: A[(xa)(ya)(za)] = [Axyz]aaa x Simple product xyz](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-54.jpg)
Energy of the ground state of N: A[(xa)(ya)(za)] = [Axyz]aaa x Simple product xyz leads to Exyz = 3 hpp + Jxy + Jxz + Jyz E(4 S) = <xyz|H|A[xyz]>/ <xyz|A[xyz]> z Denominator = <xyz|A[xyz]> = 1 Numerator = Exyz - Kxy - Kxz – Kyz = E(4 S) = 3 hpp + (Jxy - Kxy) + (Jxz - Kxz) + (Jyz – Kyz) E(2 P) =3 hpp + 2 Jxy + Jxx + 1 Kxy Pictorial representation of the N ground state 2 Kxy TA’s check this E(2 D) =3 hpp + 3 Jxy + 1 Kxy = 3 hpp + 2 Jxy + Jxx - 1 Kxy 4 Kxy Since Jxy = Jxz = Jyz E(4 S) =3 hpp + 3 Jxy - 3 Kxy Ch 120 a-Goddard-L 04 and K = Kyz © copyright 2010 William A. Goddard III, allxy 2011 rights reserved xz 54

Comparison with experiment E(1 S) 2 Kxy E(1 D) 4 Kxy E(3 P) N P As Sb Bi TA’s look up data and list excitation energies in e. V and Kxy in e. V. Get data from Moore’s tables or paper by Harding and Goddard Ann Rev Phys Chem ~1985) Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 55
4 (2 px)2(2 py)(2 pz) x Only Consider the ground state of O: [Be](2 p)4 (2 px)2(2 py)(2 pz) x Only](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-56.jpg)
Consider the ground state of O: [Be](2 p)4 (2 px)2(2 py)(2 pz) x Only have 3 orbitals, x, y, and z. thus must have a least one doubly occupied Choices: (z)2(x)(y), (y)2(x)(z), (x)2(z)(y) z and (z)2(x)2, (y)2(z)2 Clearly it is better to have two singly occupied orbitals. (2 pz)2(2 px)2 Just as for C atom, two singly occupied orbitals lead to both a triplet state and a singlet state, but the high spin triplet with the same spin for the two singly occupied orbitals is best Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 x z 56
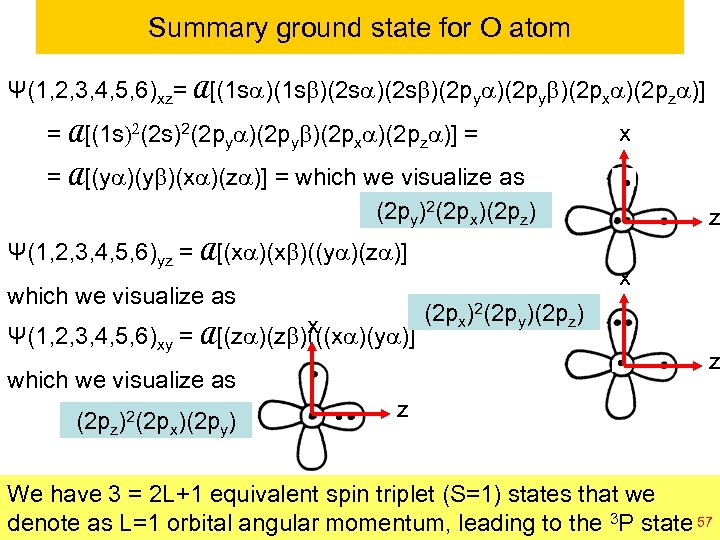
Summary ground state for O atom Ψ(1, 2, 3, 4, 5, 6)xz= A[(1 sa)(1 sb)(2 sa)(2 sb)(2 pya)(2 pyb)(2 pxa)(2 pza)] = A[(1 s)2(2 pya)(2 pyb)(2 pxa)(2 pza)] = x = A[(ya)(yb)(xa)(za)] = which we visualize as (2 py)2(2 px)(2 pz) Ψ(1, 2, 3, 4, 5, 6)yz = A[(xa)(xb)((ya)(za)] which we visualize as x Ψ(1, 2, 3, 4, 5, 6)xy = A[(za)(zb)(((xa)(ya)] which we visualize as (2 pz)2(2 px)(2 py) z x (2 px)2(2 py)(2 pz) z z We have 3 = 2 L+1 equivalent spin triplet (S=1) states that we denote as L=1 orbital © copyright 2010 William A. Goddard III, all rights to the 3 P state 57 angular momentum, leading reserved Ch 120 a-Goddard-L 04 2011

Calculating energies for O atom The energy of Ψ(1, 2, 3, 4, 5, 6)xz= A[(1 sa)(1 sb)(2 sa)(2 sb)(2 pya)(2 pyb)(2 pxa)(2 pza)] (2 py)2(2 px)(2 pz) = A[[Be](ya)(yb)(xa)(za)] is Exz = E(Be) + 2 hyy + hxx + hzz + Jyy+ 2 Jxy+ 2 Jyx+ Jxz – Kxy – Kyz – Kxz Check: 4 electrons, therefore 4 x 3/2 = 6 coulomb interactions 3 up-spin electrons, therefor 3 x 2/2 = 3 exchange interactions Other ways to group energy terms Exz = 4 hpp + Jyy + (2 Jxy – Kxy) + (2 Jyx – Kyz) + (Jxz– Kxz) Same energy for other two components of 3 P state Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 58
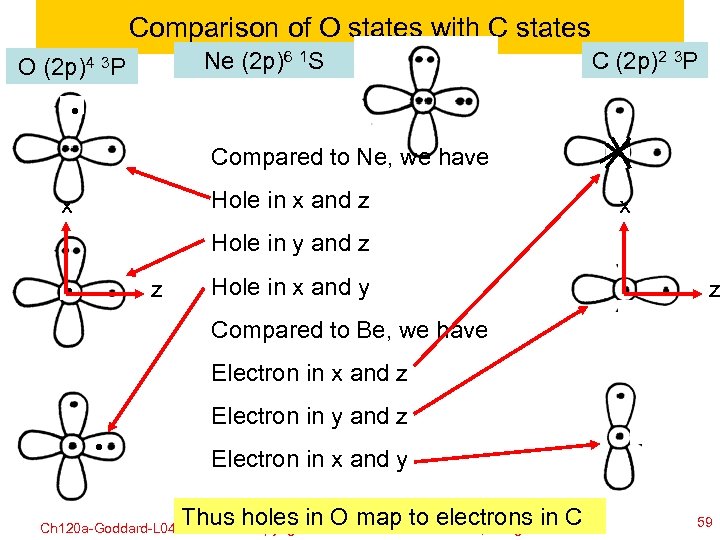
Comparison of O states with C states Ne (2 p)6 1 S O (2 p)4 3 P C (2 p)2 3 P Compared to Ne, we have Hole in x and z x x Hole in y and z z Hole in x and y z Compared to Be, we have Electron in x and z Electron in y and z Electron in x and y Ch 120 a-Goddard-L 04 Thus©holes in OWilliam A. to electrons in C map Goddard III, all rights reserved copyright 2010 2011 59

Summarizing the energies for O atom E(1 S) 3 Kxy E(1 D) 2 Kxy O S Se Te Po TA’s look up data and list excitation energies in e. V and Kxy in e. V. Get data from Moore’s tables or paper by Harding and Goddard Ann Rev Phys Chem ~1985) E(3 P) Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 60
5 Only have 3 orbitals, x, y, Consider the ground state of F: [Be](2 p)5 Only have 3 orbitals, x, y,](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-61.jpg)
Consider the ground state of F: [Be](2 p)5 Only have 3 orbitals, x, y, and z. thus must have two doubly occupied Choices: (x)2(y)2 (z), (x)2(y)(z)2, (x)(y)2(z)2 Clearly all three equivalent give rise to spin doublet. Since 3 = 2 L+1 denote as L=1 or 2 P Ψ(1 -9)z= A[(1 sa)(1 sb)(2 sa)(2 sb)(2 pya)(2 pyb)(2 pxa)(2 pza)] = A[(1 s)2(2 pya)(2 pyb)(2 pxa)(2 pza)] = = A{[Be](xa)(xb)(ya)(yb)(za)} which we visualize as x z Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 61

Calculating energies for F atom (2 px)2(2 py)2(2 pz) The energy of Ψ(1 -9)z= A{[Be](2 pxa)(2 pxb)(2 pya)(2 pyb)(2 pza)] is Exz = 5 hpp + Jxx+ Jyy+ (4 Jxy – 2 Kxy) + (2 Jxz – Kxz) + (2 Jyx– Kyz) Check: 5 electrons, therefore 5 x 4/2 = 10 coulomb interactions 3 up-spin electrons, therefore 3 x 2/2 = 3 exchange interactions 2 down-spin electrons, therefore 2 x 1/2 = 1 exchange interaction Other ways to group energy terms Same energy for other two components of 2 P state Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 62
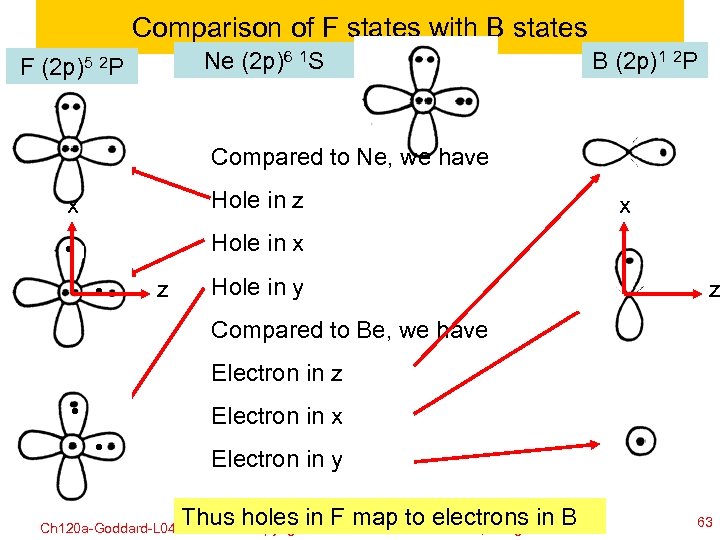
Comparison of F states with B states Ne (2 p)6 1 S F (2 p)5 2 P B (2 p)1 2 P Compared to Ne, we have Hole in z x x Hole in x z Hole in y z Compared to Be, we have Electron in z Electron in x Electron in y Ch 120 a-Goddard-L 04 Thus©holes in FWilliam A. to electrons in B map Goddard III, all rights reserved copyright 2010 2011 63

Calculating energies for F atom (2 px)2(2 py)2(2 pz) The energy of Ψ(1 -9)z= A{[Be](2 pxa)(2 pxb)(2 pya)(2 pyb)(2 pza)] is Exz = 5 hpp + Jxx+ Jyy+ (4 Jxy – 2 Kxy) + (2 Jxz – Kxz) + (2 Jyx– Kyz) Check: 5 electrons, therefore 5 x 4/2 = 10 coulomb interactions 3 up-spin electrons, therefore 3 x 2/2 = 3 exchange interactions 2 down-spin electrons, therefore 2 x 1/2 = 1 exchange interaction Other ways to group energy terms Same energy for other two components of 2 P state Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 64
6 Only have 3 orbitals, x, y, Consider the ground state of Ne: [Be](2 p)6 Only have 3 orbitals, x, y,](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-65.jpg)
Consider the ground state of Ne: [Be](2 p)6 Only have 3 orbitals, x, y, and z. thus must have all three doubly occupied Choices: (x)2(y)2(z)2 Thus get spin singlet, S=0 Since just one spatial state, 1=2 L+1 L=0. denote as 1 S Ψ(1 -10)z = A[[Be](xa)(xb)(ya)(yb)(za)(zb)] x which we visualize as Ne (2 p)6 1 S z Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 65

Calculating energy for Ne atom The energy of (2 px)2(2 py)2(2 pz)2 Ψ(1 -9)z= A{[Be](2 pxa)(2 pxb)(2 pya)(2 pyb)(2 pza)(2 pzb)} is Exz = 6 hpp +Jxx+Jyy+ Jzz + (4 Jxy – 2 Kxy) + (4 Jxz – 2 Kxz) + (4 Jyx– 2 Kyz) Check: 6 p electrons, therefore 6 x 5/2 = 15 coulomb interactions 3 up-spin electrons, therefore 3 x 2/2 = 3 exchange interactions 3 down-spin electrons, therefore 3 x 2/2 = 3 exchange interaction Since Jxx = Kxx we can rewrite this as Exz = 6 hpp +(2 Jxx-Kxx ) +(2 Jyy-Kyy ) +(2 Jzz-Kzz ) + 2(2 Jxy – Kxy) + 2(2 Jxz – Kxz) + 2(2 Jyx– Kyz) Which we will find later to be more convenient for calculating the wavefunctions using the variational principle all rights reserved Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, 2011 66
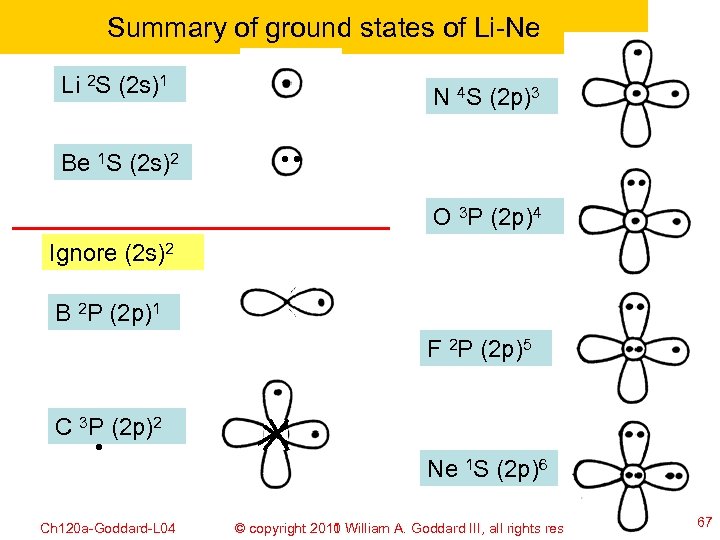
Summary of ground states of Li-Ne Li 2 S (2 s)1 N 4 S (2 p)3 Be 1 S (2 s)2 O 3 P (2 p)4 Ignore (2 s)2 B 2 P (2 p)1 F 2 P (2 p)5 C 3 P (2 p)2 Ne 1 S (2 p)6 Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 67
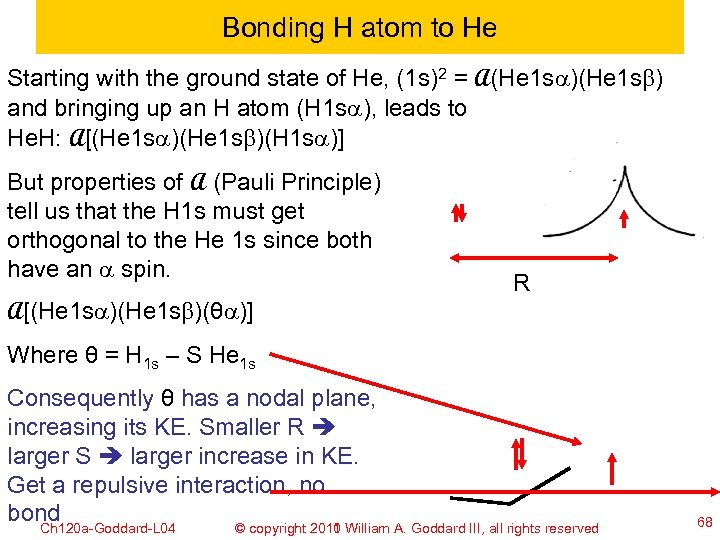
Bonding H atom to He Starting with the ground state of He, (1 s)2 = A(He 1 sa)(He 1 sb) and bringing up an H atom (H 1 sa), leads to He. H: A[(He 1 sa)(He 1 sb)(H 1 sa)] But properties of A (Pauli Principle) tell us that the H 1 s must get orthogonal to the He 1 s since both have an a spin. R A[(He 1 sa)(He 1 sb)(θa)] Where θ = H 1 s – S He 1 s Consequently θ has a nodal plane, increasing its KE. Smaller R larger S larger increase in KE. Get a repulsive interaction, no bond Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 68
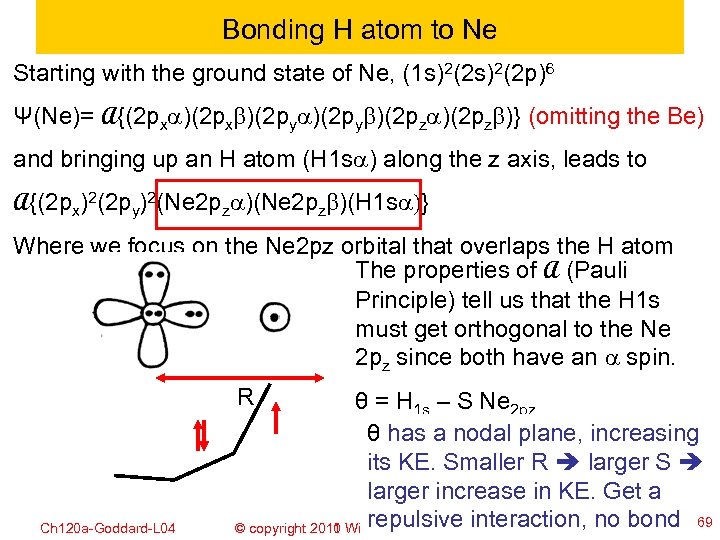
Bonding H atom to Ne Starting with the ground state of Ne, (1 s)2(2 p)6 Ψ(Ne)= A{(2 pxa)(2 pxb)(2 pya)(2 pyb)(2 pza)(2 pzb)} (omitting the Be) and bringing up an H atom (H 1 sa) along the z axis, leads to A{(2 px)2(2 py)2(Ne 2 pza)(Ne 2 pzb)(H 1 sa)} Where we focus on the Ne 2 pz orbital that overlaps the H atom The properties of A (Pauli Principle) tell us that the H 1 s must get orthogonal to the Ne 2 pz since both have an a spin. R Ch 120 a-Goddard-L 04 θ = H 1 s – S Ne 2 pz θ has a nodal plane, increasing its KE. Smaller R larger S larger increase in KE. Get a repulsive interaction, no bond 69 © copyright 2010 William A. Goddard III, all rights reserved 2011
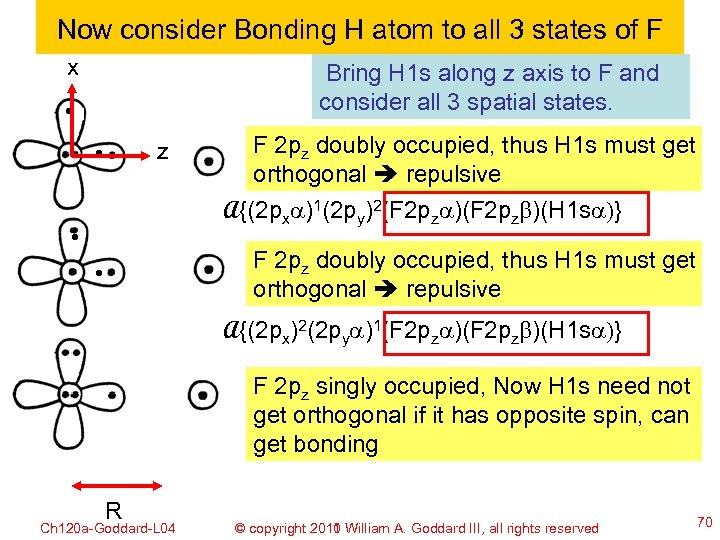
Now consider Bonding H atom to all 3 states of F x Bring H 1 s along z axis to F and consider all 3 spatial states. z F 2 pz doubly occupied, thus H 1 s must get orthogonal repulsive A{(2 pxa)1(2 py)2(F 2 pza)(F 2 pzb)(H 1 sa)} F 2 pz doubly occupied, thus H 1 s must get orthogonal repulsive A{(2 px)2(2 pya)1(F 2 pza)(F 2 pzb)(H 1 sa)} F 2 pz singly occupied, Now H 1 s need not get orthogonal if it has opposite spin, can get bonding R Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 70
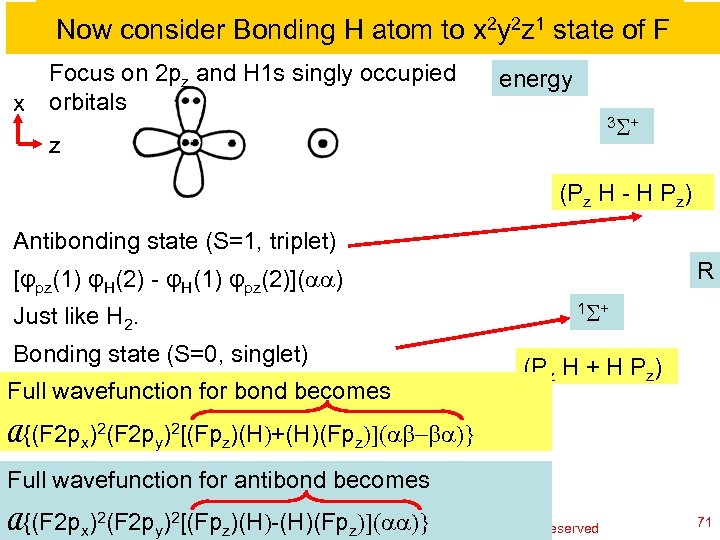
Now consider Bonding H atom to x 2 y 2 z 1 state of F Focus on 2 pz and H 1 s singly occupied x orbitals energy 3 S + z (Pz H - H Pz) Antibonding state (S=1, triplet) R [φpz(1) φH(2) - φH(1) φpz(2)](aa) Just like H 2. Bonding state (S=0, singlet) Full (1) φH(2) + φH(1) bond becomes [φpzwavefunction for φpz(2)](ab-ba) 1 S + (Pz H + H Pz) A{(F 2 px)2(F 2 py)2[(Fpz)(H)+(H)(Fpz)](ab-ba)} Full wavefunction for antibond becomes A{(F 2 px)2(F 2 py)2[(Fpz)(H)-(H)(Fp. William A. Goddard III, all rights reserved Ch 120 a-Goddard-L 04 © copyright 2010 z)](aa)} 2011 71

Schematic depiction of HF Denote the ground state of HF as Where the line connecting the two singly occupied orbitals covalent bonding We will not generally be interested in the antibonding state, but if we were it would be denoted as Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 72
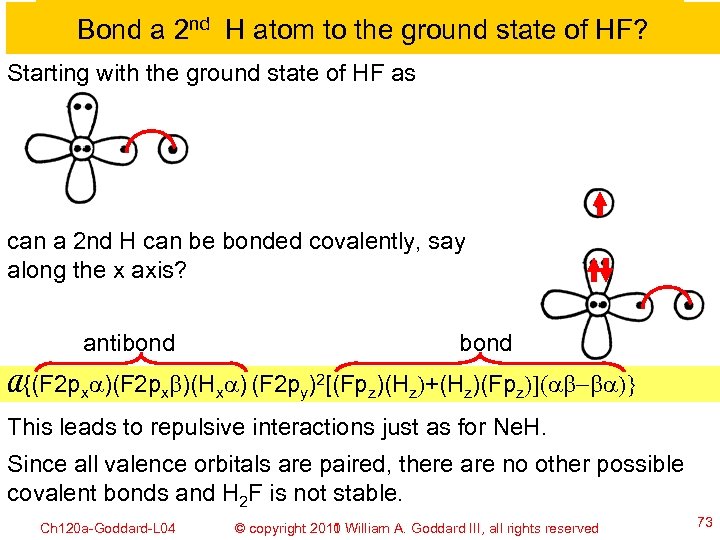
Bond a 2 nd H atom to the ground state of HF? Starting with the ground state of HF as can a 2 nd H can be bonded covalently, say along the x axis? antibond A{(F 2 pxa)(F 2 pxb)(Hxa) (F 2 py)2[(Fpz)(Hz)+(Hz)(Fpz)](ab-ba)} This leads to repulsive interactions just as for Ne. H. Since all valence orbitals are paired, there are no other possible covalent bonds and H 2 F is not stable. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 73
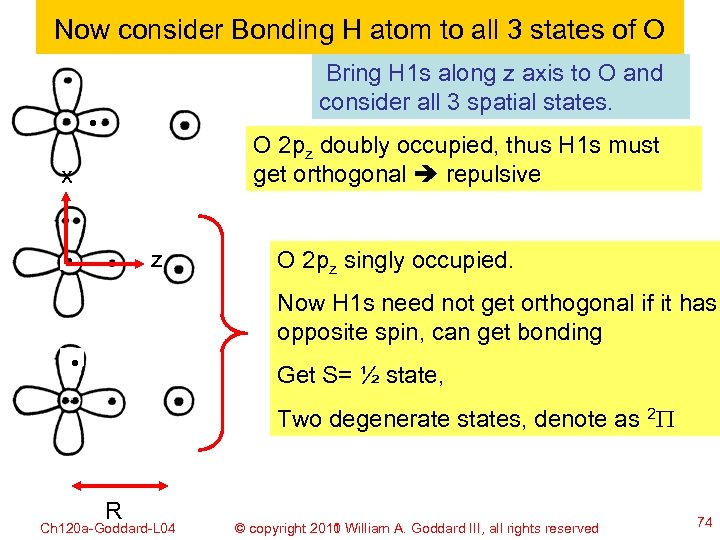
Now consider Bonding H atom to all 3 states of O Bring H 1 s along z axis to O and consider all 3 spatial states. O 2 pz doubly occupied, thus H 1 s must get orthogonal repulsive x z O 2 pz singly occupied. Now H 1 s need not get orthogonal if it has opposite spin, can get bonding Get S= ½ state, Two degenerate states, denote as 2 P R Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 74

Bonding H atom to x 1 y 2 z 1 and x 2 y 1 z 1 states of O The full wavefunction for the bonding state 2 P bond y A{(O 2 px)2(O 2 pya)1[(Opz)(H)+(H)(Opz)](ab-ba)} 2 P x bond A{(O 2 pxa)1(O 2 py)2[(Opz)(H)+(H)(Opz)](ab-ba)} x z R 2 P Ch 120 a-Goddard-L 04 (P H + H P ) © copyright 2010 William A. Goddard III, all z 2011 rights reserved z 75

Bond a 2 nd H atom to the ground state of OH Starting with the ground state of OH, we can ask whether a 2 nd H can be bonded covalently, say along the x axis. 2 P x y Bonding a 2 nd H along the x axis to the 2 Py state leads to repulsive interactions just as for Ne. H. No bond antibond A {(O 2 pxa)(O 2 pxb)(Hxa) (O 2 pya)1[(Opz)(H)+(H)(Opz)](ab-ba)} z 2 P x Bonding a 2 nd H along the x axis to the 2 Px state leads to a covalent bond A{(O 2 py)2[(Opx )(Hx)+(Hx)(Opx)](ab-ba)[(Opz)(Hz)+(HIII, all rights reserved Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard z)(Opz)](ab-ba)} 2011 76

In fact the bond 90º for H 2 O, but 90º for S Se Analize Bond in the ground state of H 2 O bond A{(O 2 py)2[(Opx )(Hx)+(Hx)(Opx)](ab-ba)[(Opz)(Hz)+(Hz)(Opz)](ab-ba)} e For optimum bonding, the pz orbital should point at the Hz while the px orbital should point at the Hx e This state of H 2 O is a spin singlet state, which we denote as 1 A 1. Thus the bond angle should be 90º. x z Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011

What is origin of large distorsion in bond angle of H 2 O A{(O 2 py)2[(Opx )(Hx)+(Hx)(Opx)](ab-ba)[(Opz)(Hz)+(Hz)(Opz)](ab-ba)} OHx bond x z OHz bond Bonding Hz to pz leaves the Hz orbital orthogonal to px and py while bonding Hx to px leaves the Hx orbital orthogonal to py and pz, so that there should be little interference in the bonds, except that the Hz orbital can overlap the Hx orbital. Since the spin on Hx is a half the time and b the other half while the same is true for Hz, then ¼ the time both are a and ¼ the time both are b. Thus the Pauli Principle (the antisymmetrizer) forces these orbitals to become orthogonal. This increases the energy as the overlap of the 1 s orbitals increases. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 Increasing the bond angle reduces this repulsive interaction 78

Testing the origin of bond angle distorsion in H 2 O x z If the increase in bond angle is a reponse to the overlap of the Hz orbital with the Hx orbital, then it should increase as the H---H distance decreases. θe Re In fact: Thus the distortion increases as Re decreases, becoming very large for R=1 A (H—H of 1. 4 A, which leads to large overlap ~0. 5). To test this interpretation, Emily Carter and wag (1983) carried out calculations of the optimum θe as a function of R for H 2 O and found that increasing R from 0. 96 A to 1. 34 A, decreases in θe by 11. 5º (from 106. 5º to 95º). Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 79

Validation of concept that the bond angle increase is due to H---H overlap Although the driving force for distorting the bond angle from 90º to 104. 5º is H—H overlap, the increase in the bond angle causes many changes in the wavefunction that can obscure the origin. Thus for the H’s to overlap the O orbitals best, the pz and px orbtials mix in some 2 s character, that opens up the angle between them. This causes the O 2 s orbital to build in p character to remain orthonal to the bonding orbitals. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 80

Bond a 3 rd H atom to the ground state of H 2 O? Starting with the ground state of H 2 O We can a 3 rd H along the y axis. This leads to A{(O 2 pya)(O 2 pyb)(Hya) [(Opx )(Hx)+(Hx)(Opx)](ab-ba)[(Opz)(Hz)+(Hz)(Opz)](ab-ba)} OHx bond OHz bond OHy antibond This leads to repulsive interactions just as for Ne. H. Since all valence orbitals are paired, there are no other possible covalent bonds and H 3 O is not stable. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 81
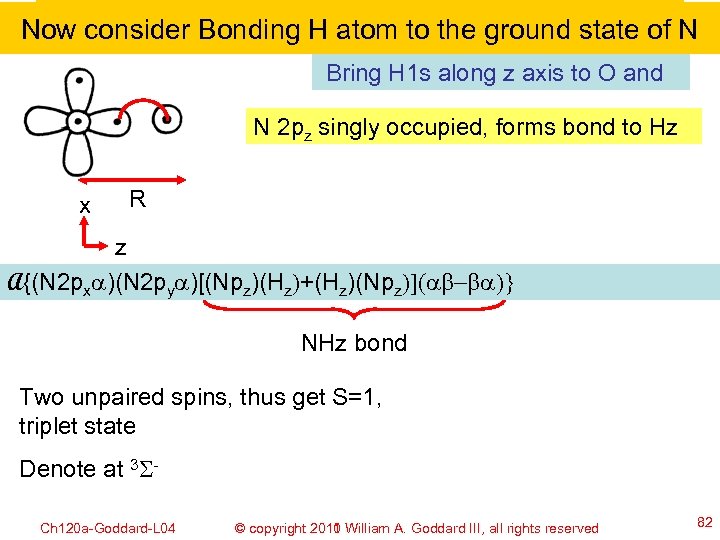
Now consider Bonding H atom to the ground state of N Bring H 1 s along z axis to O and N 2 pz singly occupied, forms bond to Hz x R z A{(N 2 pxa)(N 2 pya)[(Npz)(Hz)+(Hz)(Npz)](ab-ba)} NHz bond Two unpaired spins, thus get S=1, triplet state Denote at 3 SCh 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 82

Bond a 2 nd H atom to the ground state of NH Starting with the ground state of NH, bring a 2 nd H along the x axis. This leads to a 2 nd covalent bond. A{(N 2 pya)[(Npx )(Hx)+(Hx)(Npx)](ab-ba)[(Npz)(Hz)+(Hz)(Npz)](ab-ba)} bond Denote this as 2 B 1 state. Again expect 90º bond angle. x z Ch 120 a-Goddard-L 04 θe Re Indeed as for H 2 O we find big deviations for the 1 st row, but because N is bigger than O, the deviations are smaller. © copyright 2010 William A. Goddard III, all rights reserved 2011 83
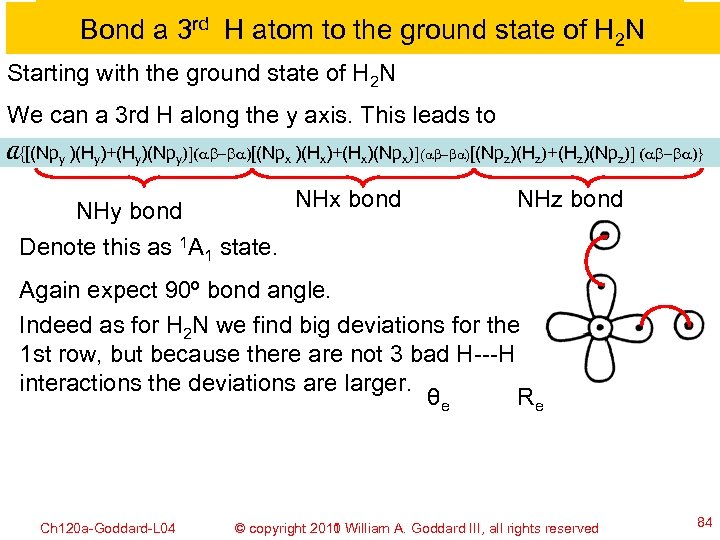
Bond a 3 rd H atom to the ground state of H 2 N Starting with the ground state of H 2 N We can a 3 rd H along the y axis. This leads to A{[(Npy )(Hy)+(Hy)(Npy)](ab-ba)[(Npx )(Hx)+(Hx)(Npx)](ab-ba)[(Npz)(Hz)+(Hz)(Npz)] (ab-ba)} NHx bond NHy bond NHz bond Denote this as 1 A 1 state. Again expect 90º bond angle. Indeed as for H 2 N we find big deviations for the 1 st row, but because there are not 3 bad H---H interactions the deviations are larger. θe Re Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 84

Bond a 4 th H atom to the ground state of H 3 N? The ground state of H 3 N has all valence orbitals are paired, there are no other possible covalent bonds and H 4 N is not stable. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 85

More on symmetry We saw in previous lectures that symmetry often has a profound effect on the solutions of the Schrödinger Equation For example inversion symmetry g or u states Permutational symmetry in electrons symmetric and antisymmetric wavefunctions for transposition of space, spin, and space-spin coordinates. More general discussions make use of group theory or more correctly group representation theory. Here we will use outline some of the essential elements relevant to this course Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 86

The symmetry operations for the Schrödinger equation form a group Consider some symmetry operator, R 1, of the Hamiltonian, for example R 1 = I (inversion) for H 2 or the transposition, t 12, for any two electron system. Then If HΨ=EΨ if follows that R 1 (HΨ)=H(R 1Ψ)= E(R 1Ψ) so that R 1Ψ is an eigenfunction with the same E. The set of symmetry operators {R 1 , R 2 , … Rn} = G forms a Group. This follows since: 1). Closure: If R 1, R 2 e G (that is both are symmetry elements) then R 2 R 1 is also a symmetry element, R 2 R 1 e G (we say that the set of symmetry operations is closed). This follows since (R 2 R 1) (HΨ)= R 2 H(R 1Ψ)= R 2 E(R 1Ψ)= E(R 2 R 1Ψ)}, that is (R 2 R 1Ψ) is also an eigenstate of H with the same energy E 2. Identity. Also R 1 = e (identity) e G. Clearly eΨ is a symmetry element Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 87

The symmetry operations for the schrodinger equation form a group, continued 3. Associativity. If {R 1, R 2, R 3 } e G then (R 1 R 2)R 3 =R 1(R 2 R 3). This follows since (R 1 R 2)R 3 HΨ = (R 1 R 2)ER 3Ψ = E (R 1 R 2)R 3Ψ and R 1(R 2 R 3)HΨ = R 1 H(R 2 R 3)Ψ = R 1 E (R 2 R 3)Ψ= ER 1(R 2 R 3)Ψ 4. Inverse. If R 1 e G then the inverse, (R 1)-1 e G , where the inverse is defined as (R 1)-1 R 1=e. This follows for any finite set that is closed since, there must be some integer p, such that (R)p = e. Thus [(R)p -1]R = e = R[(R)p-1] where (R)p-1 ≡ (R )-1 1 The above four conditions are necessary and sufficient to define what the Mathematicians call a group. The theory of the properties of such a group is called Group Theory. This is a vast field, but the only part important to QM and materials is group representation theory. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 88

Group representation Theory If G is a group with n elements, consider the set of functions S={Ψ 1= R 1Ψ, Ψ 2= R 2Ψ, …Ψn= RnΨ}. From the properties of a group, any operation R e G on any Ψi e S leads to a linear combination of functions in S. This leads to a set of nxn matrices that multiple in exactly the same way as the elements of G , so the Mathematicians say that S is a basis for the group G and that these matrices form a representation of G. The mathematicians went on to show that one could derive a set of irreducible reorientations from which one can construct any possible representation. This theory was worked out ~1905 mostly in Germany but it had few real practical applications until QM. In QM these irreducible representations are important because they constitute the possible symmetries of all possible eigenfunctions of the Hamiltonian H. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 89

An example, C 2 v The point of going through these definitions is to make sure that the students understand that the use of Group Theory in QM involves very simple concepts. No need to get afraid of the complex notations and nomenclature used sometimes. For those that want to see my views on Group Theory with some simple applications. Some notes are available for this course, denoted as Volume V, chapter 9. This dates from ~1972 when I used to teach a full year course on QM and from 1976 when I included such materials in my then full year course on chemical bonding Lets consider an example, system the nonlinear H 2 A molecule, with equal bond lengths, e. g. H 2 O, CH 2, NH 2 Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 90

The symmetry operators are C 2 v And the group {e, C 2 z, sxz, syz} is denoted as C 2 z Czn denotes a rotation of 2 p/n about the z axis (C for cyclisch, German for cyclic) sxz denotes a reflection or mirror in the xz plane (s for spiegel, Ch 120 a-Goddard-L 04 2011 German for mirror) © copyright 2010 William A. Goddard III, all rights reserved 91
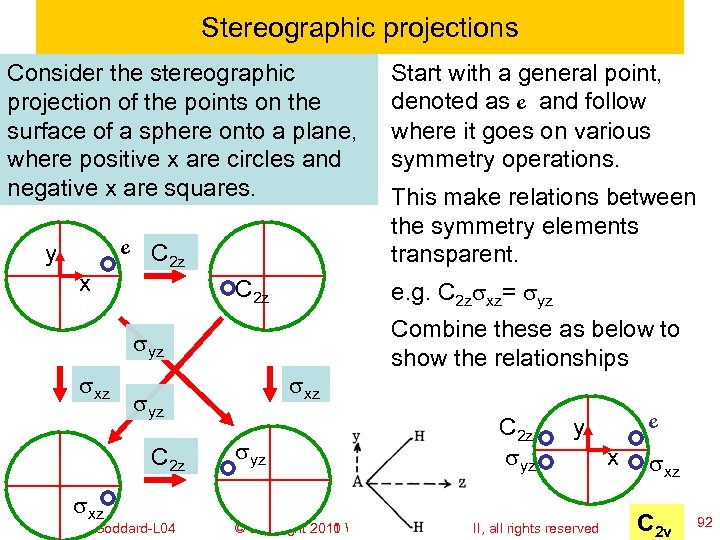
Stereographic projections Consider the stereographic projection of the points on the surface of a sphere onto a plane, where positive x are circles and negative x are squares. y x e C 2 z sxz syz Ch 120 a-Goddard-L 04 syz This make relations between the symmetry elements transparent. e. g. C 2 zsxz= syz sxz Start with a general point, denoted as e and follow where it goes on various symmetry operations. Combine these as below to show the relationships C 2 z syz e y © copyright 2010 William A. Goddard III, all rights reserved 2011 x sxz C 2 v 92
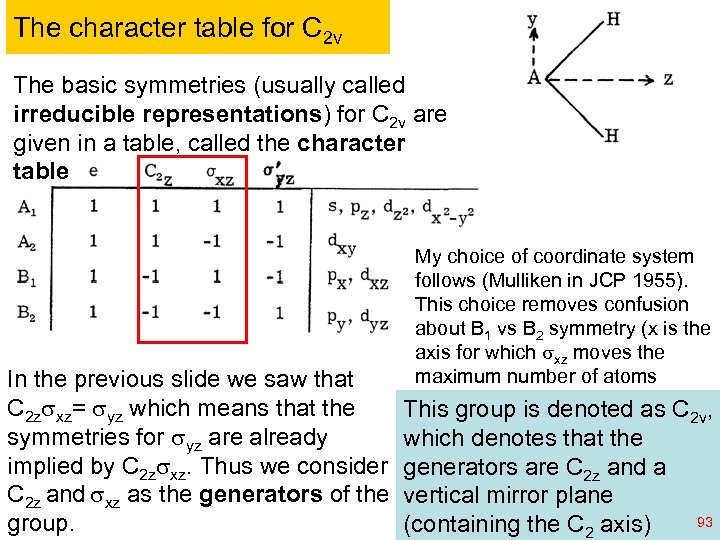
The character table for C 2 v The basic symmetries (usually called irreducible representations) for C 2 v are given in a table, called the character table My choice of coordinate system follows (Mulliken in JCP 1955). This choice removes confusion about B 1 vs B 2 symmetry (x is the axis for which sxz moves the maximum number of atoms In the previous slide we saw that C 2 zsxz= syz which means that the This group is denoted as C 2 v, symmetries for syz are already which denotes that the implied by C 2 zsxz. Thus we consider generators are C 2 z and a C 2 z and sxz as the generators of the vertical mirror plane 93 group. (containing the C 2 Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved axis) 2011

More on C 2 v Each of the symmetry elements are of order two, That is Rp = e where p=2. We saw earlier, with R = I (Inversion) and t (transposition) That with p=2 the eigenfunctions of the H must transform as symmetric (+) or antisymmetric (-) under the operation. The character table reflects this. Also since C 2 zsxz= syz , we see that the symmetry of syz is determined by those of C 2 z and sxz The general naming convention is A and B are symmetric and antisymmetric with respect to the principal rotation axis. Subscripts 1 and 2 are symmetric and antisymmetric with respect to the mirror plane generator. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 94

Applications for C 2 v Consider that an atom with a single electron in a p orbital (say B or Al) is placed at a site in a crystal with C 2 v symmetry. The character table tells us that in general, the px, py, and pz states will all have different energies. On the other hand if the symmetry were that of a square (D 4 h), px and py would be degenerate, but pz might be different, and in the symmetry of an octahedron (Oh) or tetrahedron (Td), the three p states will be degenerate. We will get to such issues later in the course but for now we will use the symmetry only to provide names for the states name 1 -e N-e a 1 a 2 b 1 b 2 Ch 120 a-Goddard-L 04 N electron wavefunction, use A 1, A 2 etc one electron orbitals: use a 1, a 2 etc © copyright 2010 William A. Goddard III, all rights reserved 2011 95
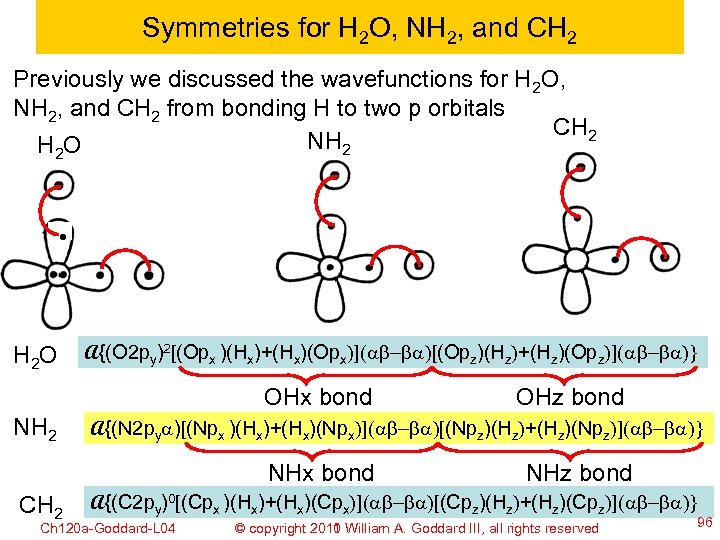
Symmetries for H 2 O, NH 2, and CH 2 Previously we discussed the wavefunctions for H 2 O, NH 2, and CH 2 from bonding H to two p orbitals CH 2 NH 2 H 2 O A{(O 2 py)2[(Opx )(Hx)+(Hx)(Opx)](ab-ba)[(Opz)(Hz)+(Hz)(Opz)](ab-ba)} OHx bond NH 2 A{(N 2 pya)[(Npx )(Hx)+(Hx)(Npx)](ab-ba)[(Npz)(Hz)+(Hz)(Npz)](ab-ba)} NHx bond CH 2 OHz bond NHz bond A{(C 2 py)0[(Cpx )(Hx)+(Hx)(Cpx)](ab-ba)[(Cpz)(Hz)+(Hz)(Cpz)](ab-ba)} Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 96
[(Cp. R)(HR)+(HR)(Cp. R)](ab-ba)} First do CH 2 singlet Ψ 1=A{(C 2 py)0[(Cp. L )(HL)+(HL)(Cp. L)](ab-ba)[(Cp. R)(HR)+(HR)(Cp. R)](ab-ba)}](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-97.jpg)
First do CH 2 singlet Ψ 1=A{(C 2 py)0[(Cp. L )(HL)+(HL)(Cp. L)](ab-ba)[(Cp. R)(HR)+(HR)(Cp. R)](ab-ba)} CHL bond CHR bond We used x and z for the bond HL y directions before but now we want new x, y, z related to the symmetry group. p. L Thus we denote bonds as L and R for left and right. Note that the C (1 s) and (2 s) pairs are invariant under all p. R operations 2 s Applying sxz to the wavefunction leads to z HR Ψ 2=A{(C 2 py)0[(Cp. R)(HR)+(HR)(Cp. R)](ab-ba)[(Cp. L)(HL)+(HL)(Cp. L)](ab-ba)} Each term involves transposing two pairs of electrons, e. g. , 1 3 and 2 4 as interchanging electrons. Since each interchange leads to a sign change we get that sxzΨ 1 = Ψ 2 = Ψ 1 Thus interchanging a copyright pair leaves Ψ invariant bond 2010 William A. Goddard III, all rights reserved Ch 120 a-Goddard-L 04 © 2011 97
[(Cp. R)(HR)+(HR)(Cp. R)](ab-ba)} CHL Finish CH 2 singlet Ψ 1=A{(C 2 py)0[(Cp. L )(HL)+(HL)(Cp. L)](ab-ba)[(Cp. R)(HR)+(HR)(Cp. R)](ab-ba)} CHL](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-98.jpg)
Finish CH 2 singlet Ψ 1=A{(C 2 py)0[(Cp. L )(HL)+(HL)(Cp. L)](ab-ba)[(Cp. R)(HR)+(HR)(Cp. R)](ab-ba)} CHL bond CHR bond y Applying C 2 z to the wavefunction also interchanges two bond pairs so that C 2 zΨ 1 = Ψ 2 = Ψ 1 A 1 or A 2 symmetry Also sxzΨ 1 = Ψ 2 = Ψ 1 A 1 or B 1 symmetry HL z p. L 2 s p. R HR Thus we conclude that the symmetry of this state of CH 2 is 1 A 1, where the superscript 1 spin singlet or S=0 Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 98
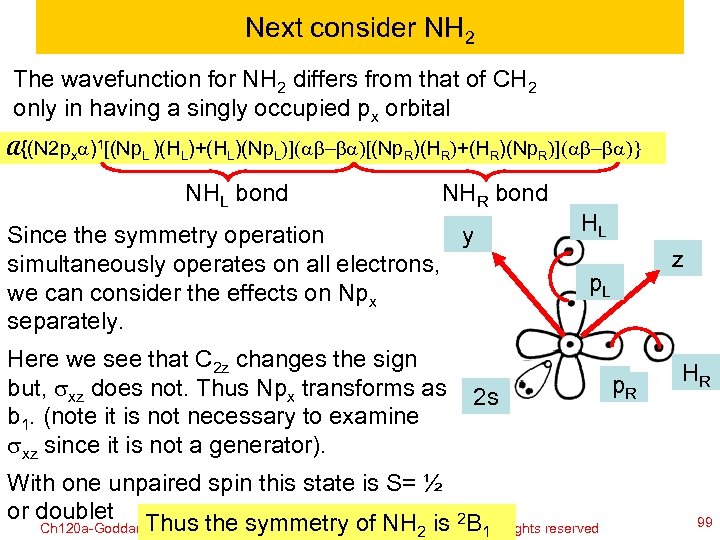
Next consider NH 2 The wavefunction for NH 2 differs from that of CH 2 only in having a singly occupied px orbital A{(N 2 pxa)1[(Np. L )(HL)+(HL)(Np. L)](ab-ba)[(Np. R)(HR)+(HR)(Np. R)](ab-ba)} NHL bond NHR bond Since the symmetry operation y simultaneously operates on all electrons, we can consider the effects on Npx separately. Here we see that C 2 z changes the sign but, sxz does not. Thus Npx transforms as b 1. (note it is not necessary to examine sxz since it is not a generator). z p. L 2 s With one unpaired spin this state is S= ½ or doublet Thus the symmetry of NH is 2 B Ch 120 a-Goddard-L 04 HL © copyright 2010 William A. Goddard III, all rights reserved 2011 2 1 p. R HR 99
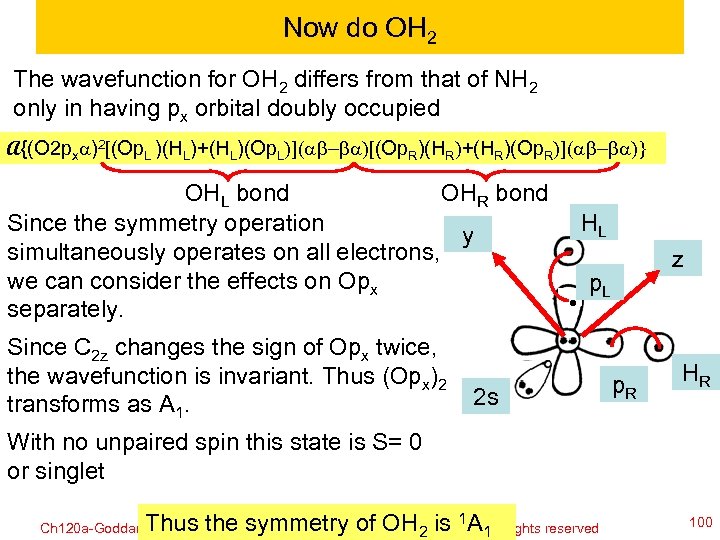
Now do OH 2 The wavefunction for OH 2 differs from that of NH 2 only in having px orbital doubly occupied A{(O 2 pxa)2[(Op. L )(HL)+(HL)(Op. L)](ab-ba)[(Op. R)(HR)+(HR)(Op. R)](ab-ba)} OHL bond OHR bond Since the symmetry operation y simultaneously operates on all electrons, we can consider the effects on Opx separately. Since C 2 z changes the sign of Opx twice, the wavefunction is invariant. Thus (Opx)2 transforms as A 1. HL z p. L 2 s p. R HR With no unpaired spin this state is S= 0 or singlet 1 Thus the©symmetry. William A. Goddard A 1 all rights reserved copyright 2010 of OH 2 is III, 2011 Ch 120 a-Goddard-L 04 100
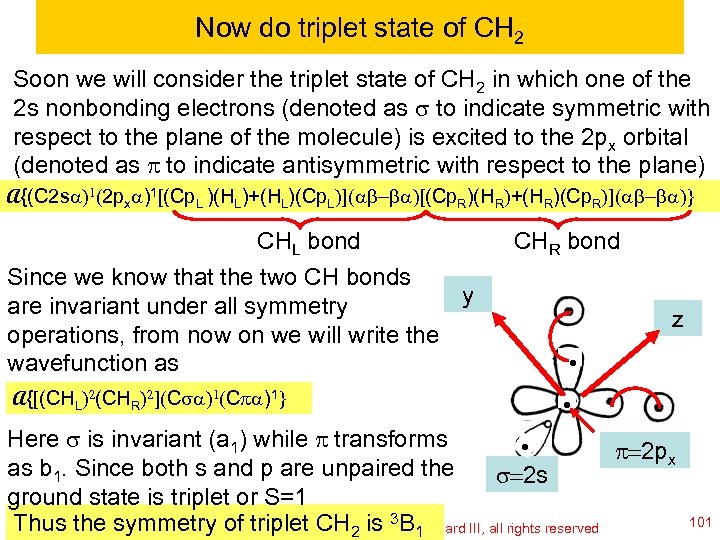
Now do triplet state of CH 2 Soon we will consider the triplet state of CH 2 in which one of the 2 s nonbonding electrons (denoted as s to indicate symmetric with respect to the plane of the molecule) is excited to the 2 px orbital (denoted as p to indicate antisymmetric with respect to the plane) A{(C 2 sa)1(2 pxa)1[(Cp. L )(HL)+(HL)(Cp. L)](ab-ba)[(Cp. R)(HR)+(HR)(Cp. R)](ab-ba)} CHL bond Since we know that the two CH bonds y are invariant under all symmetry operations, from now on we will write the wavefunction as CHR bond z A{[(CHL)2(CHR)2](Csa)1(Cpa)1} Here s is invariant (a 1) while p transforms p=2 px as b 1. Since both s and p are unpaired the s=2 s ground state is triplet or S=1 Thus the symmetry of copyright 2010 William 3 BGoddard III, all rights reserved CH Ch 120 a-Goddard-L 04 © triplet 2011 2 is A. 1 101
[(Npx )(Hx)+(Hx)(Npx)](ab-ba)[(Npz)(Hz)+(Hz)(Npz)] Second example, C 3 v, with NH 3 as the prototype A{[(Npy )(Hy)+(Hy)(Npy)](ab-ba)[(Npx )(Hx)+(Hx)(Npx)](ab-ba)[(Npz)(Hz)+(Hz)(Npz)]](https://present5.com/presentation/da94676851e08ca4c7119b0b9daf27c4/image-102.jpg)
Second example, C 3 v, with NH 3 as the prototype A{[(Npy )(Hy)+(Hy)(Npy)](ab-ba)[(Npx )(Hx)+(Hx)(Npx)](ab-ba)[(Npz)(Hz)+(Hz)(Npz)] (ab-ba)} NHb bond NHx bond We will consider a system such as NH 3, with three equal bond lengths. Here we will take the z axis as the symmetry axis and will have one H in the xz plane. NHc bond z x The other two NH bonds will be denoted as b and c. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 102

C 3 v The symmetry elements are: and the symmetry group is denoted as C 3 v. Ch 120 a-Goddard-L 04 z © copyright 2010 William A. Goddard III, all rights reserved 2011 x 103
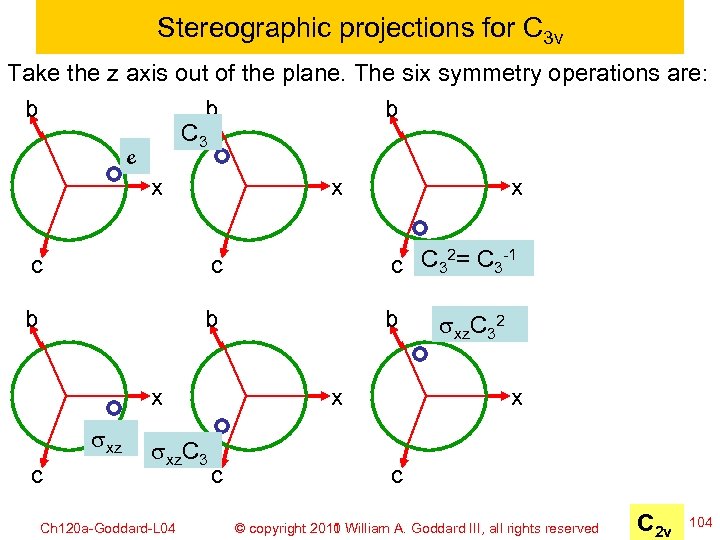
Stereographic projections for C 3 v Take the z axis out of the plane. The six symmetry operations are: b b b C 3 e x x x c c C 32= C 3 -1 c b b b x sxz c sxz. C 3 Ch 120 a-Goddard-L 04 x c sxz. C 32 x c © copyright 2010 William A. Goddard III, all rights reserved 2011 C 2 v 104
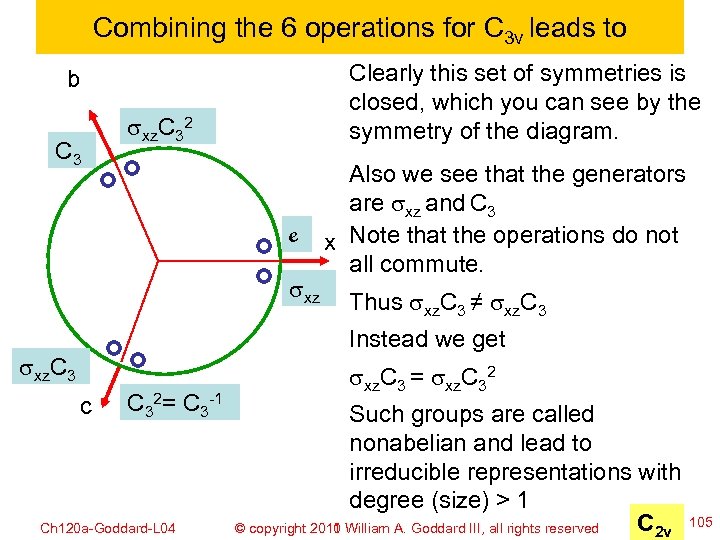
Combining the 6 operations for C 3 v leads to Clearly this set of symmetries is closed, which you can see by the symmetry of the diagram. b C 3 sxz. C 32 e sxz Also we see that the generators are sxz and C 3 x Note that the operations do not all commute. Thus sxz. C 3 ≠ sxz. C 3 Instead we get sxz. C 3 c C 32= C 3 -1 Ch 120 a-Goddard-L 04 sxz. C 3 = sxz. C 32 Such groups are called nonabelian and lead to irreducible representations with degree (size) > 1 C 2 v © copyright 2010 William A. Goddard III, all rights reserved 2011 105
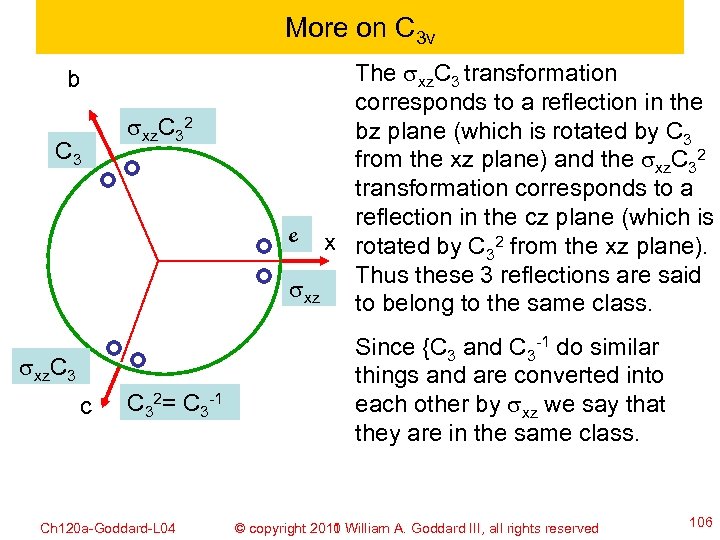
More on C 3 v b C 3 sxz. C 32 sxz. C 3 c C 32= C 3 -1 Ch 120 a-Goddard-L 04 The sxz. C 3 transformation corresponds to a reflection in the bz plane (which is rotated by C 3 from the xz plane) and the sxz. C 32 transformation corresponds to a reflection in the cz plane (which is e x rotated by C 2 from the xz plane). 3 Thus these 3 reflections are said sxz to belong to the same class. Since {C 3 and C 3 -1 do similar things and are converted into each other by sxz we say that they are in the same class. © copyright 2010 William A. Goddard III, all rights reserved 2011 106

The character table for C 3 v Here the E irreducible representation is of degree 2. This means that if φpx is an eigenfunction of the Hamiltonian, the so is φpy and they are degenerate. This set of degenerate functions would be denoted as {ex, ey} and said to belong to the E irreducible representation. The characters in this table are used to analyze the symmetries, but we will not make use of this until much later in the course. Thus an atom in a P state, say C(3 P) at a site with C 3 v symmetry, would generally split into 2 levels, {3 Px and 3 Py} of 3 E symmetry 107 and 3 Pz of 3 A 1 symmetry. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011

Application for C 3 v, NH 3 z s. LP x x We will write the wavefunction for NH 3 as A{(s. LP)2[(NHb bond)2(NHc bond)2(NHx bond)2]} Where we combined the 3 Valence bond wavefunctions in 3 pair functions and we denote what started as the 2 s pair as slp b c Consider first the effect of sxz. This leaves the NHx bond pair invariant but it interchanges the NHb and NHc bond pairs. Since the interchange two pairs of electrons the wavefunction does not change sign. Also the s. LP orbital is invariant. Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 108

Consider the effect of C 3 Next consider the C 3 symmetry operator. It does not change s. LP. It moves the NHx bond pair into the NHb pair, moves the NHb pair into the NHc pair and moves the NHc pair into the NHx pair. A cyclic permutation on three electrons can be written as (135) = (13)(35). For example. (135) φ(1)φ(3)φ(5) φ(3)φ(5)φ(1) (say this as e 1 is replaced by e 3 is replaced by e 5 is replace by 1) This is the same as (13) (35) φ(1)φ(3)φ(5) φ(1)φ(5)φ(3) φ(3)φ(5)φ(1) The point is that this is equivalent to two transpostions. Hence by the PP, the wavefunction will not change sign. Since C 3 does this cyclic permutation on 6 electrons, eg (135)(246)= (13)(35)(24)(46), we still get no sign change. Thus the wavefunction for NH 3 has 1 A 1 symmetry Ch 120 a-Goddard-L 04 © copyright 2010 William A. Goddard III, all rights reserved 2011 109
da94676851e08ca4c7119b0b9daf27c4.ppt