f415d27deb154de9ed96365af172d7c1.ppt
- Количество слайдов: 38

Le. Bow College of Business Drexel University Rx Roulette: Counterfeit Pharmaceuticals in Developing Nations Dr. Kristina M. Lybecker December 6, 2003 Prepared for the conference, “Markets for Pharmaceuticals and the Health of Developing Nations, ” Toulouse, France, December 5 -6, 2003. 1

“As early as the fourth century BC people were warned about the dangers of adulterated medicines, and despite all the advances made over the years this concern has not disappeared. ” World Health Organization. “‘In the next ten years, spurious drugs will be the single biggest problem’ in public health. ” Ranjit Roychoudhury, President of the Delhi Society for the Promotion of the Use of Rational Drugs, as quoted in The Lancet. “Some health officials in Africa have stated that counterfeit medicines are a greater public health threat than AIDS or malaria. ” Harvey Bale, President of the International Federation of Pharmaceutical Manufacturers Associations (IFPMA), as quoted in The Lancet. 2

Introduction § § § Challenge to Public Health Characteristics of the Problem Facilitating Factors Available Strategies to Combat Counterfeiting Conclusions 3

Counterfeiting and Public Health Mounting Evidence § Health Consequences § § Less Visible Barrier to Access § Therapeutic Failure § Prolonged Illness & Death § Global Microbial Resistance § Loss of Confidence in the System § Squandered Resources § $43. 5 billion annually 4

Dimensions of the Problem Significant & Growing Problem § Definition § § World Health Organization § U. S. FDA 5

Counterfeiting Definition § “a counterfeit medicine is one which is deliberately and fraudulently mislabeled with respect to identity and/or source. Counterfeiting can apply to both branded and generic products and counterfeit products may include products with the correct ingredients, wrong ingredients, without active ingredients, with incorrect quantity of active ingredient or with fake packaging. ” World Health Organization (1997) 6

Dimensions of the Problem Estimates: HA! § Magnitude of the Problem: § § World Health Organization estimate: 6% – 10% § Some developing nations: more than 80% Drugs: Aspirin to Zyprexa § Nations: Global problem § Financial Impact: $43. 5 billion/year § Sophistication: varied § 7

Dimensions of the Problem Drug Quality § In two studies analyzing counterfeit medications, the results show that: § 50 – 60% : no active ingredient § 15 – 20% : wrong active ingredient § 15 – 20% : incorrect quantity of active ingredient § 5% : correct active ingredient 8

Counterfeiting Specifics & Examples 9

Dimensions of the Problem Examples § Aspirin: “Because tablet-making machines are easily obtainable, even counterfeit ‘aspirin’ tablets containing little or no acetylsalicylic acid can be profitable, especially at open-air markets such as those in African villages” Mc. Gregor 1997 10

Dimensions of the Problem Examples § HIV and Cancer Drugs: “drugs to boost the immune system of cancer and HIV patients have become a favorite of counterfeiters. In one case. . . criminals realized a $28 million profit from a shipment of 11, 000 boxes of counterfeit Epogen and Procrit, which is also often prescribed to cancer, AIDS and kidneyfailure patients” Associated Press 2003 11

Dimensions of the Problem Examples § Children’s Vaccines: “As many as 80, 000 children in Nigeria have gotten fake meningitis vaccines. India has seen bogus polio vaccines” Knox 2003 12

Dimensions of the Problem Examples § Ponstan: Authorities “interdicted millions of yellow tablets that were virtually indistinguishable from the genuine product – including the company logo. These tablets were made of boric acid, floor wax, and lead-based yellow paint used for road markings. Sacks of these ‘raw materials’ were stacked throughout the counterfeiter’s site” Christian 2001 13

Counterfeiting Operation 14

Counterfeiters GMP 15
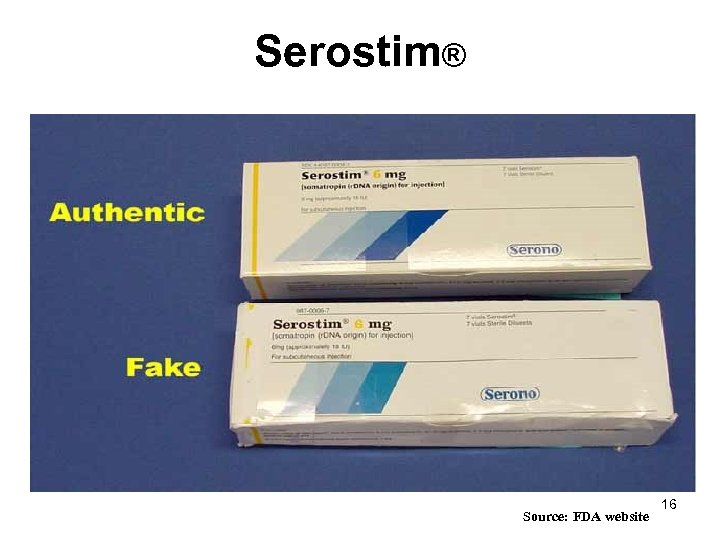
Serostim® Source: FDA website 16
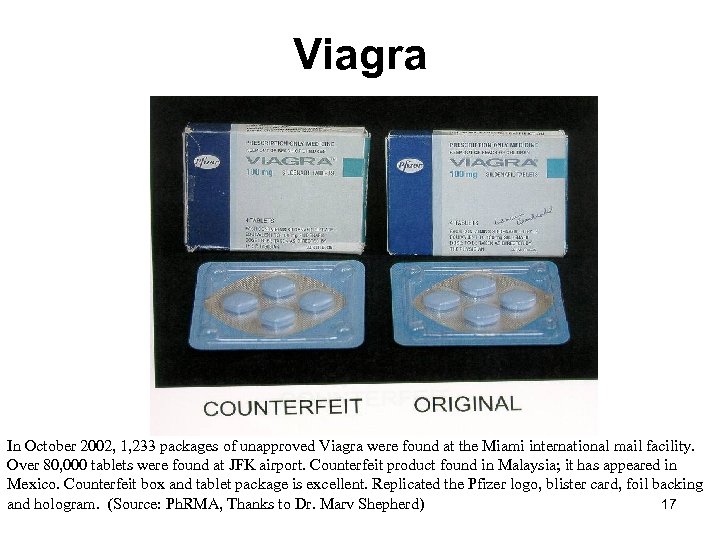
Viagra In October 2002, 1, 233 packages of unapproved Viagra were found at the Miami international mail facility. Over 80, 000 tablets were found at JFK airport. Counterfeit product found in Malaysia; it has appeared in Mexico. Counterfeit box and tablet package is excellent. Replicated the Pfizer logo, blister card, foil backing and hologram. (Source: Ph. RMA, Thanks to Dr. Marv Shepherd) 17
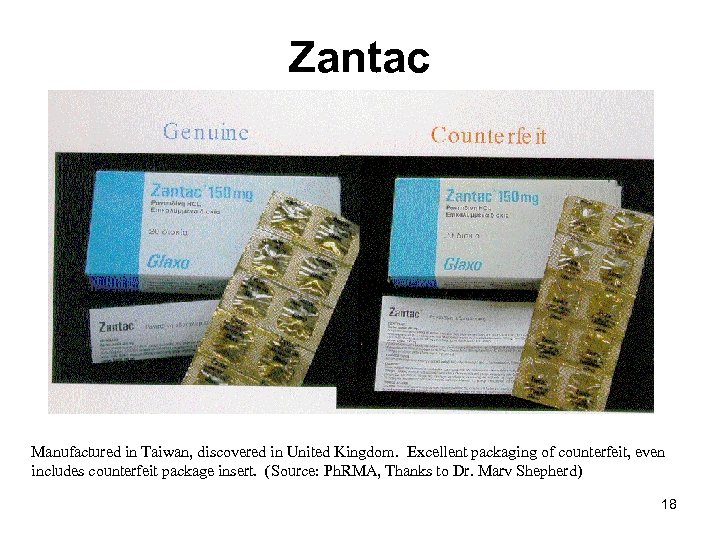
Zantac Manufactured in Taiwan, discovered in United Kingdom. Excellent packaging of counterfeit, even includes counterfeit package insert. (Source: Ph. RMA, Thanks to Dr. Marv Shepherd) 18

Deca-Durabolin Counterfeit Genuine An anemia product used to treat renal insufficiency was purchased in Mexico and illegally imported to the U. S. Packaging is very similar to original, but in testing the counterfeit product, it only contained sesame seed oil. (Source: Ph. RMA, Thanks to Dr. Marv Shepherd) 19

Ponstan is an anti-inflammatory product. This counterfeit was found in Columbia. First is the yellow powder; it consist of boric acid, floor wax, yellow highway paint. Pressed into tablets and placed in foil packs with labeling. (Source: Ph. RMA, Thanks to Dr. Marv Shepherd) 20
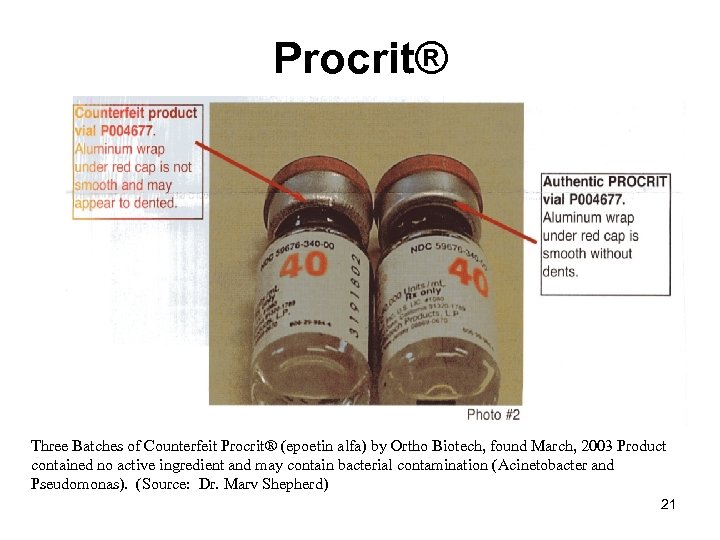
Procrit® Three Batches of Counterfeit Procrit® (epoetin alfa) by Ortho Biotech, found March, 2003 Product contained no active ingredient and may contain bacterial contamination (Acinetobacter and Pseudomonas). (Source: Dr. Marv Shepherd) 21

Prozac Photos taken from a plant in Asia. Manufactured in China, shipped to Taiwan and from there shipped around the world. Package in counterfeit Lilly blister packs. There is no assurance of quality, manufacturing processes or conditions. (Source: Dr. Marv Shepherd) 22

Manufacturing Facility for Counterfeit Panadol (Source: Ph. RMA Thanks to Dr. Marv Shepherd) 23
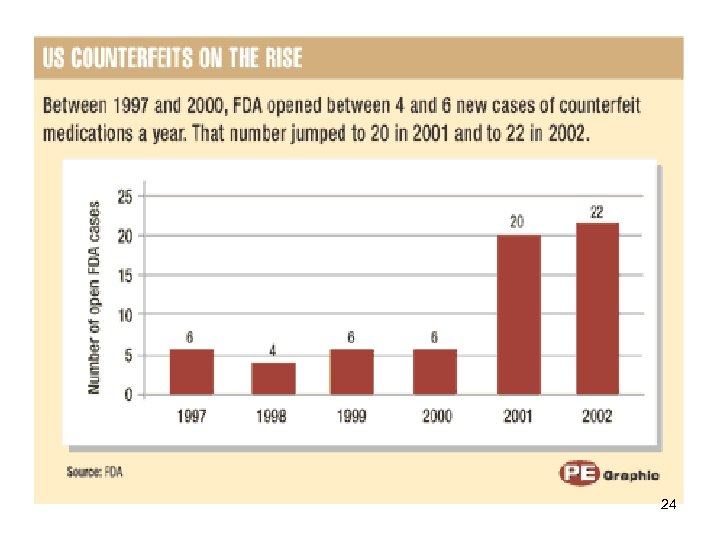
24

Dimensions of the Problem Common Threads § § § Small numbers of very costly drugs Large quantities of less costly drugs Expensive branded drugs Clear injectibles Simple tablets 25

Facilitating Factors WHO’s List Counterfeiting is facilitated where: § § § weak drug regulatory control and enforcement, scarcity and/or erratic supply of basic medicines, extended, relatively unregulated markets and distribution chains, price differentials, lack of effective intellectual property protection, due regard is not paid to quality assurance World Health Organization 1992 26

Facilitating Factors The Attraction § § § § Organized Crime Less Dangerous Less Risky Difficult Detection Naiveté of Law Enforcement Negligible punishment Enhanced IPRs Changing Technology 27

Facilitating Factors Negligible Punishment § “In Malaysia the maximum fine for manufacturing counterfeit medicine is 25, 000 ringgit ($6, 580). . . The law also allows for a maximum three-year jail sentence, but a representative of the Pharmaceutical Association of Malaysia says no one has ever done time for making fake medicine in Malaysia” Saywell & Mc. Manus 2002 28

Facilitating Factors Negligible Punishment § In Vietnam, 64 per cent of all artesunate (a key antimalarial drug in Southeast Asia) samples tested were found to be fake. Nevertheless, while prison terms of “ 20 years have been given in Vietnam for trading in fake sildenafil (Viagra). . . there have been no prosecutions of fake antimalarial traders” Newton et al. 2001 29

Facilitating Factors Enhanced IPR Protection Changing Market Conditions § Increased Intellectual Property Rights § Focus on two elements § § Access to medicines § Research on neglected diseases § Exacerbating the Problems of Counterfeiting § “First pass” at Enforcement § “Wholehearted” approach to Enforcement 30

Facilitating Factors Changing Technology Desktop Publishing § Available Equipment on e. Bay § Distinctive bottles replaced by uniform vials § Internet sourcing § 31

Strategies for Combating Counterfeiting Parallel Imports § Local Production § Enforcement Efforts § Education § 32

Strategies Parallel Imports § Disappointing Results § Israel, the Philippines and Kenya § Low-priced markets see prices increase § Evidence in EU § Variety of Packaging 33

Strategies Local Production § “DAP often did not advocate local production, particularly in countries with populations of less than 25 million. . . the value component in local-drug production was small and that quality and reliability of supplies were frequently questionable. . . DAP recommended that they start with packaging from bulk and only gradually move towards simple formulation of drugs” Lauridsen 1997 34

Strategies Local Production Self-sufficiency solution § High Fixed Costs § Supplies § § Questionable quality & reliability § Alternative: Bulk Purchase Programs 35

Strategies Enforcement § Technology § Deter counterfeiters § Facilitate authentication Raise the costs to the counterfeiter § Security features § § Overt § Covert § Forensic 36

Conclusions Critical Timing § Important Tradeoff § § Access to safe medicines vs. rewards to innovation Vulnerable Populations § Need for Change with Safeguards § 37

Thank you for your attention. 38
f415d27deb154de9ed96365af172d7c1.ppt