169e8a0254dadfde723a5a44ab805087.ppt
- Количество слайдов: 24

La “trasferibilita’” dei risultati clinici tra device. Il caso dello stent medicato Roberto Violini Cardiologia Interventistica AO S. Camillo Forlanini - Roma

L’angioplastica coronarica RESTENOSI 30 -40% solo pallone 15 -20% stent < 6 % stent medicato
Lo stent
DES – Drug eluting stent

Lots of DES ever received CE-Certificate !

Transferability of data between different DES: 1. Does a CE mark mean, a stent is safe and effective ? 2. Are all DES the same ? (DES-1 vs. DES-2) 3. Can data be transferred for modifications of a specific stent ? (DES-1 a vs. DES-1 b)

Transferability of data between different DES: 1. Does a CE mark mean, a stent is safe and effective ? 2. Are all DES the same ? (DES-1 vs. DES-2) 3. Can data be transferred for modifications of a specific stent ? (DES-1 a vs. DES-1 b)
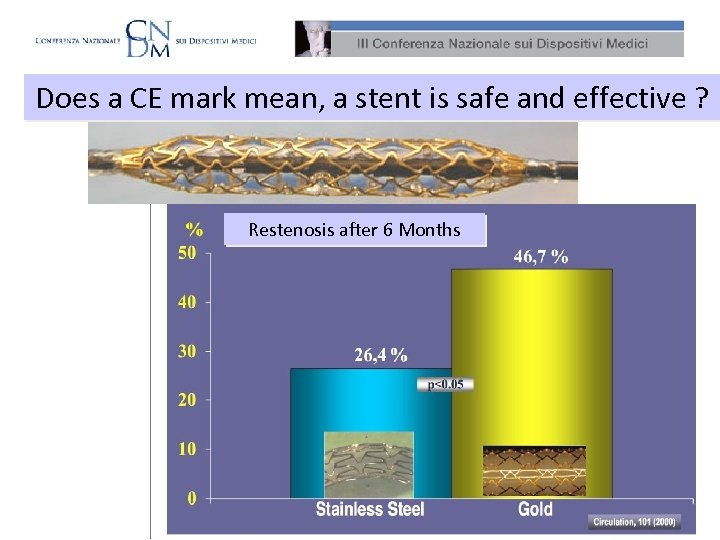
Does a CE mark mean, a stent is safe and effective ? Restenosis after 6 Months

CE-approved DES in Europe without randomized studies STENT Company Platform Carrier APOLLO COROFLEX Please In. Tek Jupiter Polysulfon B Braun Coroflex Polysulfon EUCA-TAX Eucatech ST Flex LUC-CHOPIN Balton Chopin Globamed Omega OMEGA TAXCOR . . . -el ing ut el x Genius Megaflex Euro. COR ita acl ll P ea ar Biopolymer S? U AX Paclitaxel T like Paclitaxel s ent st biodegradable Polymer no Polymer Drug Number of Randomized Studies 0 0 Paclitaxel 0

Transferability of data between different DES: 1. Does a CE mark mean, a stent is safe and effective ? NO ! 2. Are all DES the same ? (DES-1 vs. DES-2) 3. Can data be transferred for modifications of a specific stent ? (DES-1 a vs. DES-1 b)

Transferability of data between different DES: 1. Does a CE mark mean, a stent is safe and effective ? 2. Are all DES the same ? (DES-1 vs. DES-2) 3. Can data be transferred for modifications of a specific stent ? (DES-1 a vs. DES-1 b)

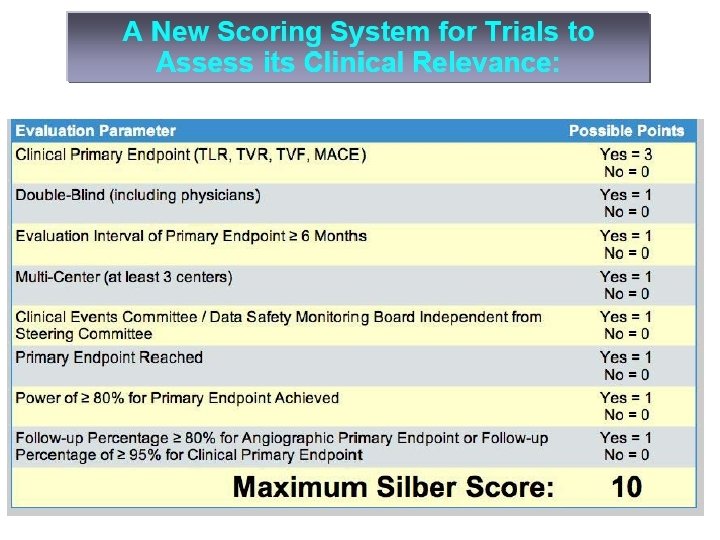
103 randomized DES Studies in 58. 356 Patients ABSOLUTE, ACTION, APPLAUSE, ASPECT, AVELLINO, BASKET, BBK, BRADES, CACTUS, CARDIA, CHINA-1, CHINA-2, CHREDIT, COMPARE, CORPAL-Bif-1, CORPAL-Bif-2, COSTAR-II, C-SIRIUS, Cypher/ Be. Stent, Cypher/Vision, DEDICATION-DES, DECODE, DELIVER-I, DESSERT, DEXAMET, DIABETES, EAGLE, ELUTES, ENDEAVOR-III, ENDEAVOR-IV, E-SIRIUS, ESTRADIOL, ETHOS-I, EUROSTAR-II, FUTURE-II, GENESIS, GENOUS-STEMI, HAAMU, HORIZONS-AMI, ISARDESIRE, ISAR-DIABETICS, ISAR-LM, ISAR-PEACE, ISAR-SMART-3, ISAR-TEST-3, ISAR-TEST-4, JUPITER-II, LABEM, LEADERS, LONG-DES-II, MISSION, NOBORI-I Phase 1, NOBORI-I Phase 2, NORDIC-Bif-I, NORDIC-Bif. II, PAINT, PASSION, PATENCY, PISA-LAD, PRISON-II, PROSIT, RAPPAC, RAVEL, RIGA-LM, RRISC, REALITY, RIBS-II, SCANDSTENT, SCORE, SCORPIUS, SESAMI, SES-SMART, SEVILLA, SIRIUS-Bif, SIRTAX, SISR, SORT-OUT-III, SOS, SPIRIT-II, SPIRIT-III, SPIRIT-IV, STEALTH-I, STRATEGY, SYNTAX, TAXI, TAXUS-II, TAXUS-IV, TAXUS -V, TAXUS-V-ISR, TAXUS-VI, TYPHOON, ZEST, ZOOMAX-II ul , mf e, ar tiv e h fec er ef w ive ES re in fect e D we ef om ES S ery e D ES v som e D som

Transferability of data between different DES: 1. Does a CE mark mean, a stent is safe and effective ? 2. Are all DES the same ? (DES-1 vs. DES-2) NO ! 3. Can data be transferred for modifications of a specific stent ? (DES-1 a vs. DES-1 b)

Transferability of data between different DES: 1. Does a CE mark mean, a stent is safe and effective ? 2. Are all DES the same ? (DES-1 vs. DES-2) 3. Can data be transferred for modifications of a specific stent ? (DES-1 a vs. DES-1 b)

Stent: Material: better deliverability, better expansion Possible Improvements for DES: Design: better deliverability, better expansion Polymer: better drug release Drug: high efficacy without prolonged DAPT Balloon: better stent deliverability, better stent expansion d, dy tu os n but m for in re ire qu s ian ic s phy the Shaft: better stent deliverability

DES Components: Stent Material Stent Design

DES – Una storia vera • La multinazionale A compra la multinazionale B • Per motivi di antitrust, A rivende alcuni brevetti alla multinazionale C • A e C concordano il comarketing per il DES X • Successivamente A realizza lo stent X 1 C realizza lo stent X 2
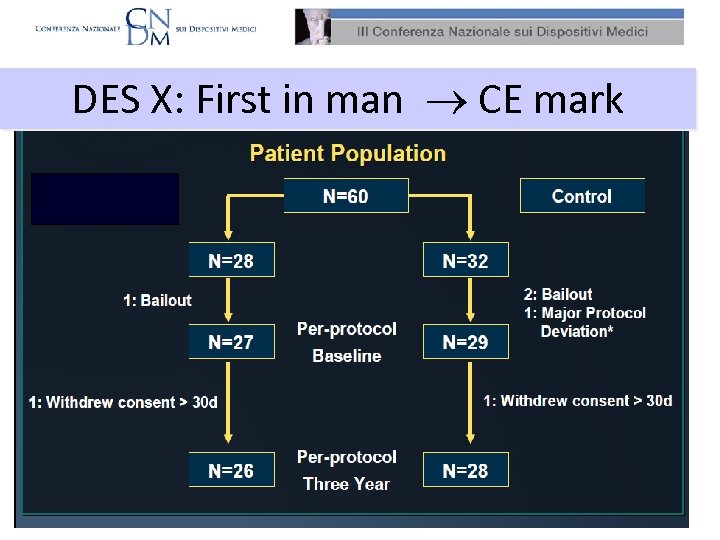
DES X: First in man CE mark
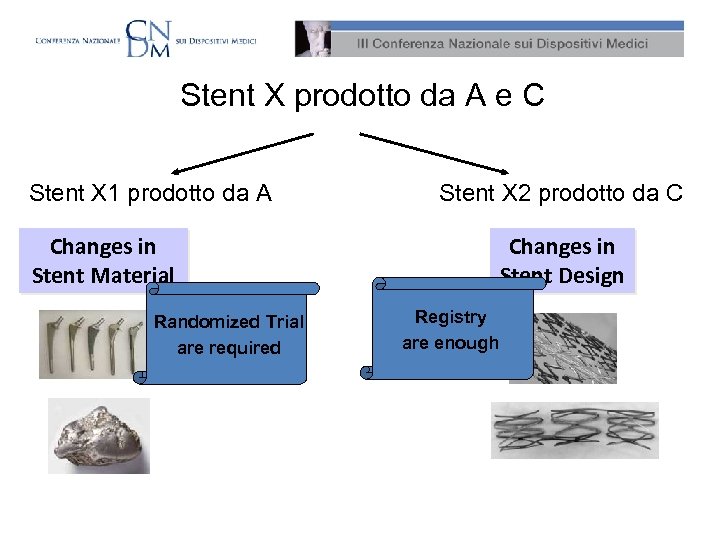
Stent X prodotto da A e C Stent X 1 prodotto da A Stent X 2 prodotto da C Changes in Stent Material Randomized Trial are required Changes in Stent Design Registry are enough

Transferability of data between different DES: 1. Does a CE mark mean, a stent is safe and effective ? 2. Are all DES the same ? (DES-1 vs. DES-2) 3. Can data be transferred for modifications of a specific stent ? (DES-1 a vs. DES-1 b) sometimes YES sometimes NO

Conclusione Al di la’ della problematica regolatoria e, per le strutture pubbliche, della normativa per gli acquisti esiste la responsabilita’ della scelta che grava sull’utilizzatore finale. Attenta valutazione dei risultati clinici e spirito critico sono gli strumenti per arrivare ad una scelta responsabile dei device.

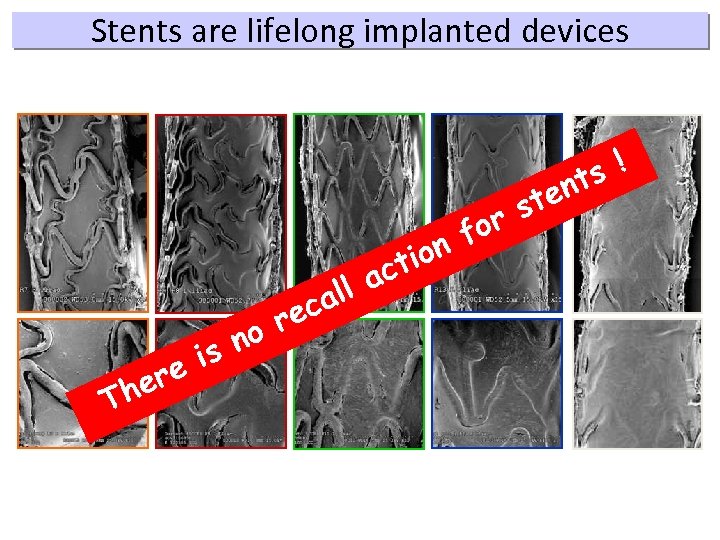
Stents are lifelong implanted devices ion ct r no is ere Th ll a ca e or f s! nt ste
169e8a0254dadfde723a5a44ab805087.ppt