Колебательная задача 2003.ppt
- Количество слайдов: 11
Колебательная задача Gaussian B 3 LYP/6 -311 G(d, p) #P B 3 LYP/6 -311 G(d, p) Opt Freq
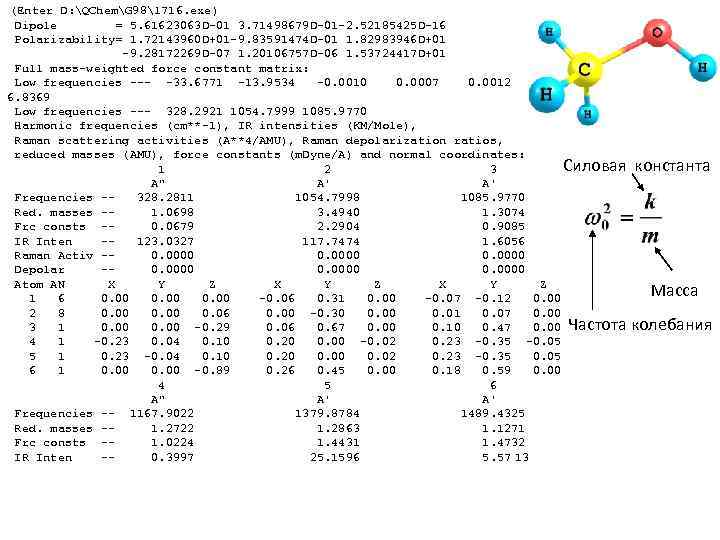
(Enter D: QChemG 98l 716. exe) Dipole = 5. 61623063 D-01 3. 71498679 D-01 -2. 52185425 D-16 Polarizability= 1. 72143960 D+01 -9. 83591474 D-01 1. 82983946 D+01 -9. 28172269 D-07 1. 20106757 D-06 1. 53724417 D+01 Full mass-weighted force constant matrix: Low frequencies --- -33. 6771 -13. 9534 -0. 0010 0. 0007 0. 0012 6. 8369 Low frequencies --- 328. 2921 1054. 7999 1085. 9770 Harmonic frequencies (cm**-1), IR intensities (KM/Mole), Raman scattering activities (A**4/AMU), Raman depolarization ratios, reduced masses (AMU), force constants (m. Dyne/A) and normal coordinates: Силовая 1 2 3 A" A' A' Frequencies -328. 2811 1054. 7998 1085. 9770 Red. masses -1. 0698 3. 4940 1. 3074 Frc consts -0. 0679 2. 2904 0. 9085 IR Inten -123. 0327 117. 7474 1. 6056 Raman Activ -0. 0000 Depolar -0. 0000 Atom AN X Y Z 1 6 0. 00 -0. 06 0. 31 0. 00 -0. 07 -0. 12 0. 00 2 8 0. 00 0. 06 0. 00 -0. 30 0. 01 0. 07 0. 00 3 1 0. 00 -0. 29 0. 06 0. 67 0. 00 0. 10 0. 47 0. 00 Частота 4 1 -0. 23 0. 04 0. 10 0. 20 0. 00 -0. 02 0. 23 -0. 35 -0. 05 5 1 0. 23 -0. 04 0. 10 0. 20 0. 02 0. 23 -0. 35 0. 05 6 1 0. 00 -0. 89 0. 26 0. 45 0. 00 0. 18 0. 59 0. 00 4 5 6 A" A' A' Frequencies -- 1167. 9022 1379. 8784 1489. 4325 Red. masses -1. 2722 1. 2863 1. 1271 Frc consts -1. 0224 1. 4431 1. 4732 IR Inten -0. 3997 25. 1596 5. 57 13 константа Масса колебания

---------- Thermochemistry ---------Temperature 298. 150 Kelvin. Pressure 1. 00000 Atm. Atom 1 has atomic number 6 and mass 12. 00000 Atom 2 has atomic number 8 and mass 15. 99491 Atom 3 has atomic number 1 and mass 1. 00783 Atom 4 has atomic number 1 and mass 1. 00783 Atom 5 has atomic number 1 and mass 1. 00783 Atom 6 has atomic number 1 and mass 1. 00783 Molecular mass: 32. 02621 amu. Principal axes and moments of inertia in atomic units: 1 2 3 EIGENVALUES -14. 08780 72. 92585 75. 56331 IA I B I c X -0. 05407 0. 99854 0. 00000 эксп-нт Y 0. 99854 0. 05407 0. 00000 Z 0. 00000 1. 00000 THIS MOLECULE IS AN ASYMMETRIC TOP. ROTATIONAL SYMMETRY NUMBER 1. ROTATIONAL TEMPERATURES (KELVIN) 6. 14812 1. 18769 1. 14624 ROTATIONAL CONSTANTS (GHZ) 128. 10671 24. 74762 23. 88383 Zero-point vibrational energy 134198. 2 (Joules/Mol) 32. 07414 (Kcal/Mol) WARNING-- EXPLICIT CONSIDERATION OF 1 DEGREES OF FREEDOM AS VIBRATIONS MAY CAUSE SIGNIFICANT ERROR VIBRATIONAL TEMPERATURES: 472. 32 1517. 61 1562. 47 1680. 34 1985. 33 (KELVIN) 2142. 95 2145. 92 2170. 02 4277. 84 4336. 95 4464. 50 5524. 24 Эксперимент - 127. 63076 24. 68418 = 1705 amu 3 A 6, 1723 23. 76536

Zero-point correction= Thermal correction to Energy= Thermal correction to Enthalpy= Thermal correction to Gibbs Free Energy= Sum of electronic and zero-point Energies= Sum of electronic and thermal Enthalpies= Sum of electronic and thermal Free Energies= TOTAL ELECTRONIC TRANSLATIONAL ROTATIONAL VIBRATION 1 TOTAL BOT TOTAL V=0 VIB (BOT) 1 VIB (V=0) 1 ELECTRONIC TRANSLATIONAL ROTATIONAL E (Thermal) KCAL/MOL 34. 155 0. 000 0. 889 32. 378 0. 712 Q 0. 889128 D-13 0. 287963 D+11 0. 395711 D-23 0. 569769 D+00 0. 128159 D+01 0. 125805 D+01 0. 100000 D+01 0. 712392 D+07 0. 315404 D+04 0. 051113 (Hartree/Particle) 0. 054430 0. 055374 0. 028374 -115. 706281 -115. 702964 -115. 702020 -115. 729021 CV CAL/MOL-KELVIN 8. 762 0. 000 2. 981 2. 801 1. 619 LOG 10(Q) -13. 051036 10. 459336 -23. 402622 -0. 244301 0. 107750 0. 099697 0. 000000 6. 852719 3. 498867 S CAL/MOL-KELVIN 56. 827 0. 000 36. 324 18. 991 1. 512 1. 269 LN(Q) -30. 051120 24. 083511 -53. 886528 -0. 562524 0. 248104 0. 229561 0. 000000 15. 778968 8. 056439 Эксп-нт: 57. 3 Эксп-нт: 8. 56
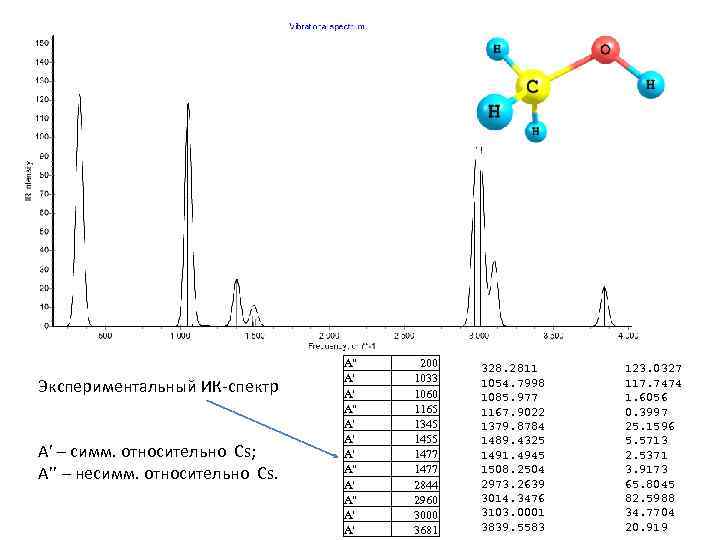
Экспериментальный ИК-спектр A’ – симм. относительно Cs; A’’ – несимм. относительно Cs. A" A' A' A' A" A' A' 200 1033 1060 1165 1345 1455 1477 2844 2960 3000 3681 328. 2811 1054. 7998 1085. 977 1167. 9022 1379. 8784 1489. 4325 1491. 4945 1508. 2504 2973. 2639 3014. 3476 3103. 0001 3839. 5583 123. 0327 117. 7474 1. 6056 0. 3997 25. 1596 5. 5713 2. 5371 3. 9173 65. 8045 82. 5988 34. 7704 20. 919
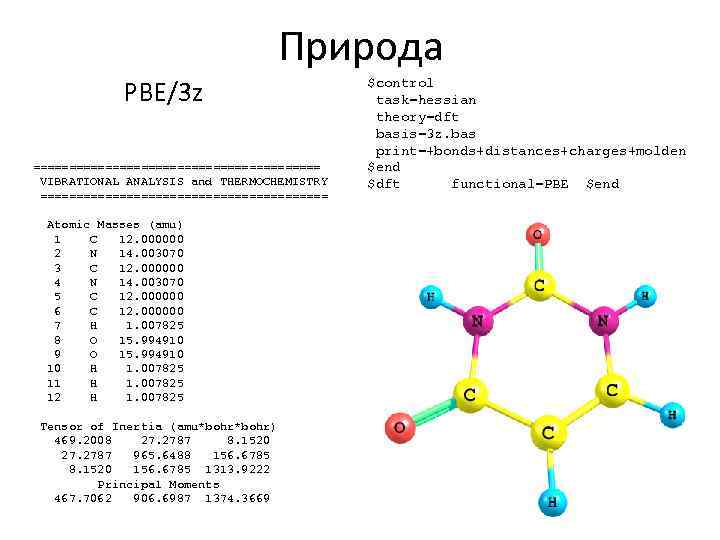
Природа PBE/3 z ==================== VIBRATIONAL ANALYSIS and THERMOCHEMISTRY ==================== Atomic Masses (amu) 1 C 12. 000000 2 N 14. 003070 3 C 12. 000000 4 N 14. 003070 5 C 12. 000000 6 C 12. 000000 7 H 1. 007825 8 O 15. 994910 9 O 15. 994910 10 H 1. 007825 11 H 1. 007825 12 H 1. 007825 Tensor of Inertia (amu*bohr) 469. 2008 27. 2787 8. 1520 27. 2787 965. 6488 156. 6785 8. 1520 156. 6785 1313. 9222 Principal Moments 467. 7062 906. 6987 1374. 3669 $control task=hessian theory=dft basis=3 z. bas print=+bonds+distances+charges+molden $end $dft functional=PBE $end
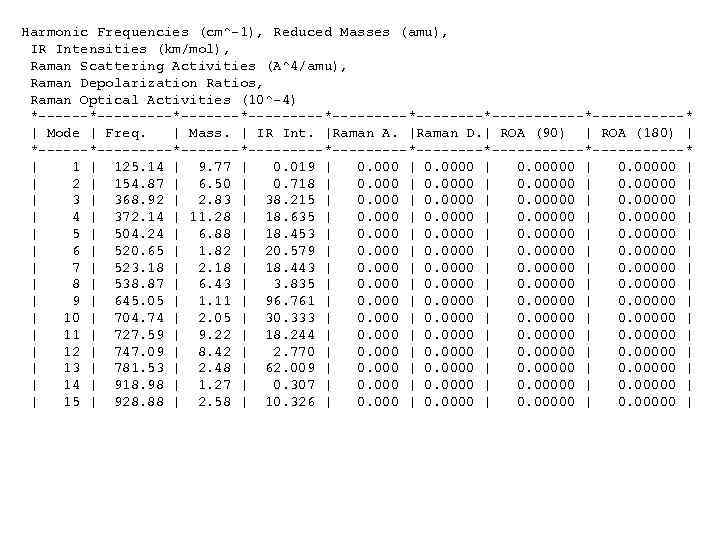
Harmonic Frequencies (cm^-1), Reduced Masses (amu), IR Intensities (km/mol), Raman Scattering Activities (A^4/amu), Raman Depolarization Ratios, Raman Optical Activities (10^-4) *---------*---------*--------*-----------* | Mode | Freq. | Mass. | IR Int. |Raman A. |Raman D. | ROA (90) | ROA (180) | *---------*---------*--------*-----------* | 125. 14 | 9. 77 | 0. 019 | 0. 00000 | | 2 | 154. 87 | 6. 50 | 0. 718 | 0. 00000 | | 368. 92 | 2. 83 | 38. 215 | 0. 00000 | | 4 | 372. 14 | 11. 28 | 18. 635 | 0. 00000 | | 504. 24 | 6. 88 | 18. 453 | 0. 00000 | | 6 | 520. 65 | 1. 82 | 20. 579 | 0. 00000 | | 7 | 523. 18 | 2. 18 | 18. 443 | 0. 00000 | | 8 | 538. 87 | 6. 43 | 3. 835 | 0. 00000 | | 9 | 645. 05 | 1. 11 | 96. 761 | 0. 00000 | | 10 | 704. 74 | 2. 05 | 30. 333 | 0. 00000 | | 11 | 727. 59 | 9. 22 | 18. 244 | 0. 00000 | | 12 | 747. 09 | 8. 42 | 2. 770 | 0. 00000 | | 13 | 781. 53 | 2. 48 | 62. 009 | 0. 00000 | | 14 | 918. 98 | 1. 27 | 0. 307 | 0. 00000 | | 15 | 928. 88 | 2. 58 | 10. 326 | 0. 00000 |
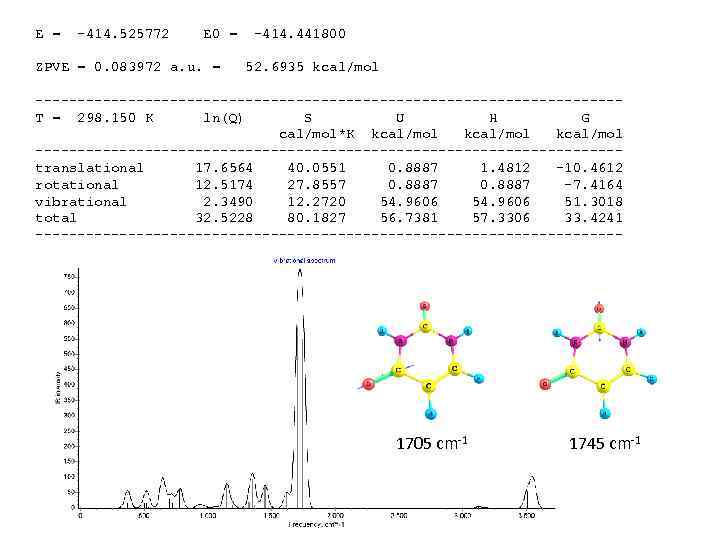
E = -414. 525772 E 0 = ZPVE = 0. 083972 a. u. = -414. 441800 52. 6935 kcal/mol -----------------------------------T = 298. 150 K ln(Q) S U H G cal/mol*K kcal/mol -----------------------------------translational 17. 6564 40. 0551 0. 8887 1. 4812 -10. 4612 rotational 12. 5174 27. 8557 0. 8887 -7. 4164 vibrational 2. 3490 12. 2720 54. 9606 51. 3018 total 32. 5228 80. 1827 56. 7381 57. 3306 33. 4241 ----------------------------------- 1705 cm-1 1745 cm-1
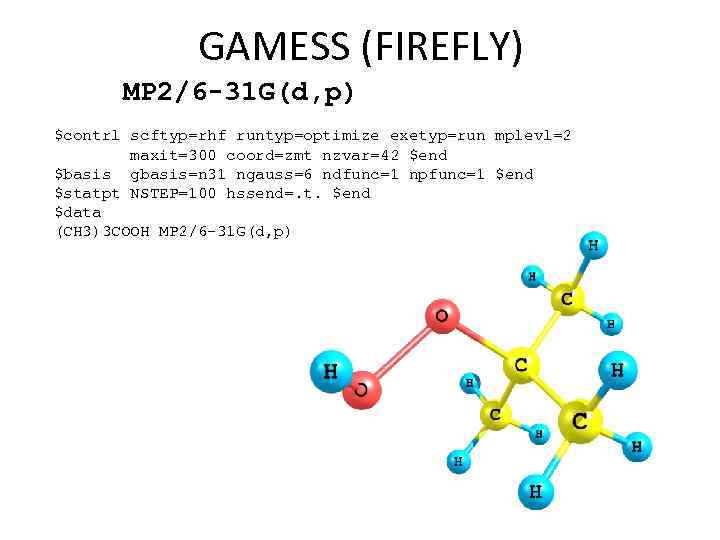
GAMESS (FIREFLY) MP 2/6 -31 G(d, p) $contrl scftyp=rhf runtyp=optimize exetyp=run mplevl=2 maxit=300 coord=zmt nzvar=42 $end $basis gbasis=n 31 ngauss=6 ndfunc=1 npfunc=1 $end $statpt NSTEP=100 hssend=. t. $end $data (CH 3)3 COOH MP 2/6 -31 G(d, p) geometry optimization
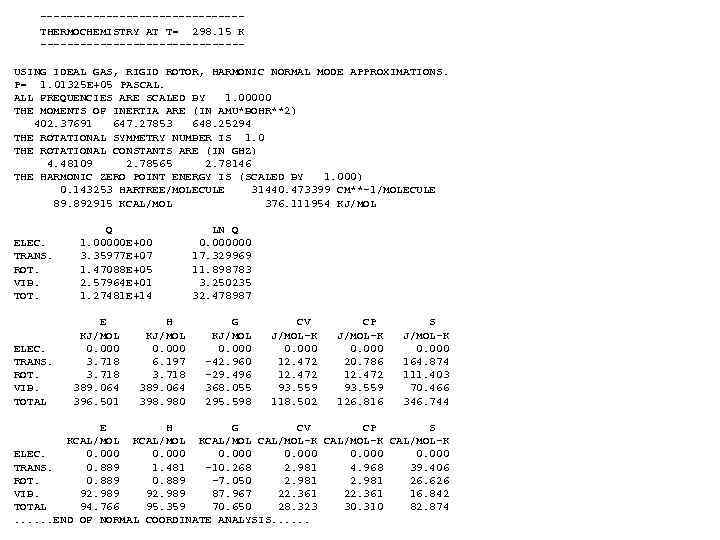
---------------THERMOCHEMISTRY AT T= 298. 15 K ---------------USING IDEAL GAS, RIGID ROTOR, HARMONIC NORMAL MODE APPROXIMATIONS. P= 1. 01325 E+05 PASCAL. ALL FREQUENCIES ARE SCALED BY 1. 00000 THE MOMENTS OF INERTIA ARE (IN AMU*BOHR**2) 402. 37691 647. 27853 648. 25294 THE ROTATIONAL SYMMETRY NUMBER IS 1. 0 THE ROTATIONAL CONSTANTS ARE (IN GHZ) 4. 48109 2. 78565 2. 78146 THE HARMONIC ZERO POINT ENERGY IS (SCALED BY 1. 000) 0. 143253 HARTREE/MOLECULE 31440. 473399 CM**-1/MOLECULE 89. 892915 KCAL/MOL 376. 111954 KJ/MOL ELEC. TRANS. ROT. VIB. TOTAL Q 1. 00000 E+00 3. 35977 E+07 1. 47088 E+05 2. 57964 E+01 1. 27481 E+14 E KJ/MOL 0. 000 3. 718 389. 064 396. 501 H KJ/MOL 0. 000 6. 197 3. 718 389. 064 398. 980 LN Q 0. 000000 17. 329969 11. 898783 3. 250235 32. 478987 G KJ/MOL 0. 000 -42. 960 -29. 496 368. 055 295. 598 CV J/MOL-K 0. 000 12. 472 93. 559 118. 502 CP J/MOL-K 0. 000 20. 786 12. 472 93. 559 126. 816 S J/MOL-K 0. 000 164. 874 111. 403 70. 466 346. 744 E H G CV CP S KCAL/MOL CAL/MOL-K ELEC. 0. 000 TRANS. 0. 889 1. 481 -10. 268 2. 981 4. 968 39. 406 ROT. 0. 889 -7. 050 2. 981 26. 626 VIB. 92. 989 87. 967 22. 361 16. 842 TOTAL 94. 766 95. 359 70. 650 28. 323 30. 310 82. 874. . . END OF NORMAL COORDINATE ANALYSIS. . .
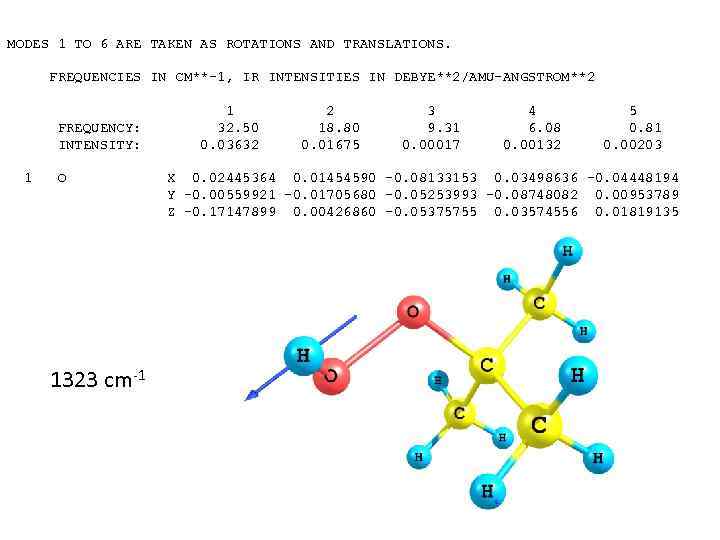
MODES 1 TO 6 ARE TAKEN AS ROTATIONS AND TRANSLATIONS. FREQUENCIES IN CM**-1, IR INTENSITIES IN DEBYE**2/AMU-ANGSTROM**2 FREQUENCY: INTENSITY: 1 O 1323 cm-1 1 32. 50 0. 03632 2 18. 80 0. 01675 3 9. 31 0. 00017 4 6. 08 0. 00132 5 0. 81 0. 00203 X 0. 02445364 0. 01454590 -0. 08133153 0. 03498636 -0. 04448194 Y -0. 00559921 -0. 01705680 -0. 05253993 -0. 08748082 0. 00953789 Z -0. 17147899 0. 00426860 -0. 05375755 0. 03574556 0. 01819135
Колебательная задача 2003.ppt