a4692d7155ca11ec2b03da526e16b93e.ppt
- Количество слайдов: 17
 Key Findings XXX have statutory and other legal obligations that we are failing to adequately discharge – this trend is deteriorating RECALL: Bactroban Cream Manufacturer: Crawley Market: Spain RECALL: Havrix Paediatric Vaccine Manufacturer: Rixensart Market: Portugal RECALL: Augmentin Syrup 250 mg Manufacturer: Mayenne Market: Belgium Leaflet contains erroneous information regarding the temperature of storage. Not more than 30°C is stated rather than 25°C Erroneous information on leaflet referring to the product as Havrix intravenous, when it is given in the muscle Incorrect information on carton due to artwork error by studio RECALL: Varilrix Vaccine Manufacturer: Rixensart Market: India RECALL: Augmentin Solution for Nebuliser Manufacturer: Mayenne Market: Belgium/Luxembourg/Iceland RECALL: Ventolin Solution for Nebuliser Manufacturer: Bad Oldesloe Market: Belgium Leaflet error states “Combined Hepatitis A Hepatitis B vaccine” whereas this vaccine is for the treatment of chicken pox Recalls Low priority recall commencing 1 August 2002. Formula detail on label is for 250 mg strength Missing warning statement on PIL RECALL: Viani Diskus Manufacturer: Bad Oldesloe Market: Germany Error on PIL due to cut and paste on artwork text by marketing company in Munich. Approximately 100, 000 packs on the market and 590, 000 packs in stock We have had 7 artwork-related recalls so far in 2002 0129 -03. 2 -Update 2 v 9. ppt Page 0
Key Findings XXX have statutory and other legal obligations that we are failing to adequately discharge – this trend is deteriorating RECALL: Bactroban Cream Manufacturer: Crawley Market: Spain RECALL: Havrix Paediatric Vaccine Manufacturer: Rixensart Market: Portugal RECALL: Augmentin Syrup 250 mg Manufacturer: Mayenne Market: Belgium Leaflet contains erroneous information regarding the temperature of storage. Not more than 30°C is stated rather than 25°C Erroneous information on leaflet referring to the product as Havrix intravenous, when it is given in the muscle Incorrect information on carton due to artwork error by studio RECALL: Varilrix Vaccine Manufacturer: Rixensart Market: India RECALL: Augmentin Solution for Nebuliser Manufacturer: Mayenne Market: Belgium/Luxembourg/Iceland RECALL: Ventolin Solution for Nebuliser Manufacturer: Bad Oldesloe Market: Belgium Leaflet error states “Combined Hepatitis A Hepatitis B vaccine” whereas this vaccine is for the treatment of chicken pox Recalls Low priority recall commencing 1 August 2002. Formula detail on label is for 250 mg strength Missing warning statement on PIL RECALL: Viani Diskus Manufacturer: Bad Oldesloe Market: Germany Error on PIL due to cut and paste on artwork text by marketing company in Munich. Approximately 100, 000 packs on the market and 590, 000 packs in stock We have had 7 artwork-related recalls so far in 2002 0129 -03. 2 -Update 2 v 9. ppt Page 0
 Key Findings Our continuing failure to discharge our legal obligations presents a risk to our business security · · · The legal significance of the product information leaflet (PIL) is increasing. It is being considered as the primary patient instruction, not the prescribing information. – we have already been sued for errors on a PIL We have recalled product due to incorrect trademark referencing – the referencing was illegal and we were at risk of having those trademark rights removed Our current performance presents risks to: – our corporate reputation – our position as licensee of choice – and ultimately our business performance . . . but why is this happening? 0129 -03. 2 -Update 2 v 9. ppt Page 1
Key Findings Our continuing failure to discharge our legal obligations presents a risk to our business security · · · The legal significance of the product information leaflet (PIL) is increasing. It is being considered as the primary patient instruction, not the prescribing information. – we have already been sued for errors on a PIL We have recalled product due to incorrect trademark referencing – the referencing was illegal and we were at risk of having those trademark rights removed Our current performance presents risks to: – our corporate reputation – our position as licensee of choice – and ultimately our business performance . . . but why is this happening? 0129 -03. 2 -Update 2 v 9. ppt Page 1
 Key Findings We are experiencing numerous counterfeit issues associated with our products and packaging Product – Country • Sensodyne Toothpaste – China/Gulf States Issue Product – Country • Epivir and Combivir – Belgium/Holland Issue • Police raid in Lishui City, China, revealed large quantity of counterfeit packs, presumably for export to Middle East Investigation Findings • Counterfeits had very good copy of hologram label, introduced as a result of previous incidents in the Gulf region Corrective & Preventative Actions: • Shipments of product originally sold into Central Africa seized by Belgian police in Antwerp en route for Holland Investigation Findings • Goods being examined for signs of counterfeit packs – total shipment value over £ 25 m Corrective & Preventative Actions: • The OVERT elements of the hologram were very effectively copied – and • Charitable donations to developing countries can be diverted to Western the covert element was ignored • Holograms are much less effective than we believe • Clear differentiation of Access medicines is needed Product – Country • Zentel – China Issue markets for huge profits, and may be further dilutes with counterfeits Product – Country • Augmentin tablets – Nigeria Issue • Police raid in Yixing City, China, revealed large quantities of counterfeit Zentel, destined for Nigeria Investigation Findings • The packs were counterfeits of those made in Mayenne, France for Central Africa (different from packs sold in China) Corrective & Preventative Actions: • Suspect packs bearing genuine batch number, but incorrect batch size Investigation Findings • All components counterfeit, and tablets contain NO active material Corrective & Preventative Actions: • Anti-counterfeit features required on all packs to aid detection • Clear proof of organised international trade in counterfeit medicines • Greater diligence and international liaison is required Other sectors (e. g. FMCG) strictly control access to artwork, print templates and plates to reduce threat of counterfeiting. With approx. 250 artwork studios and over 400 print suppliers we are not able to control this supply base to prevent counterfeiting. 0129 -03. 2 -Update 2 v 9. ppt Page 2
Key Findings We are experiencing numerous counterfeit issues associated with our products and packaging Product – Country • Sensodyne Toothpaste – China/Gulf States Issue Product – Country • Epivir and Combivir – Belgium/Holland Issue • Police raid in Lishui City, China, revealed large quantity of counterfeit packs, presumably for export to Middle East Investigation Findings • Counterfeits had very good copy of hologram label, introduced as a result of previous incidents in the Gulf region Corrective & Preventative Actions: • Shipments of product originally sold into Central Africa seized by Belgian police in Antwerp en route for Holland Investigation Findings • Goods being examined for signs of counterfeit packs – total shipment value over £ 25 m Corrective & Preventative Actions: • The OVERT elements of the hologram were very effectively copied – and • Charitable donations to developing countries can be diverted to Western the covert element was ignored • Holograms are much less effective than we believe • Clear differentiation of Access medicines is needed Product – Country • Zentel – China Issue markets for huge profits, and may be further dilutes with counterfeits Product – Country • Augmentin tablets – Nigeria Issue • Police raid in Yixing City, China, revealed large quantities of counterfeit Zentel, destined for Nigeria Investigation Findings • The packs were counterfeits of those made in Mayenne, France for Central Africa (different from packs sold in China) Corrective & Preventative Actions: • Suspect packs bearing genuine batch number, but incorrect batch size Investigation Findings • All components counterfeit, and tablets contain NO active material Corrective & Preventative Actions: • Anti-counterfeit features required on all packs to aid detection • Clear proof of organised international trade in counterfeit medicines • Greater diligence and international liaison is required Other sectors (e. g. FMCG) strictly control access to artwork, print templates and plates to reduce threat of counterfeiting. With approx. 250 artwork studios and over 400 print suppliers we are not able to control this supply base to prevent counterfeiting. 0129 -03. 2 -Update 2 v 9. ppt Page 2
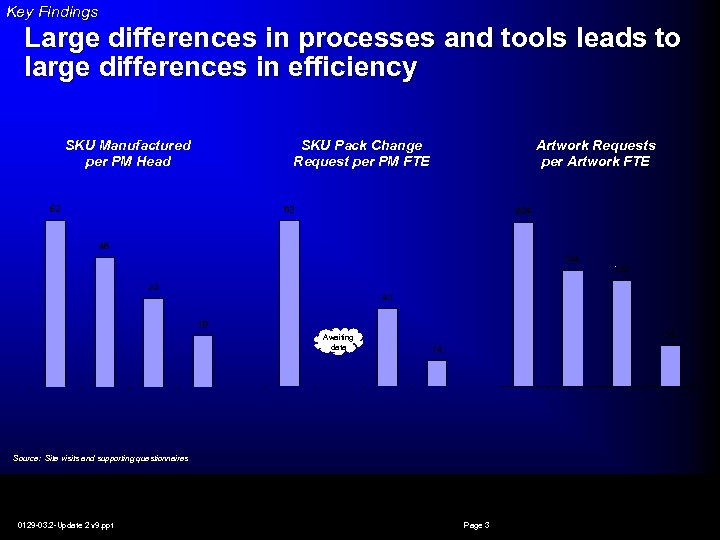 Key Findings Large differences in processes and tools leads to large differences in efficiency SKU Manufactured per PM Head SKU Pack Change Request per PM FTE Artwork Requests per Artwork FTE Awaiting data Source: Site visits and supporting questionnaires 0129 -03. 2 -Update 2 v 9. ppt Page 3
Key Findings Large differences in processes and tools leads to large differences in efficiency SKU Manufactured per PM Head SKU Pack Change Request per PM FTE Artwork Requests per Artwork FTE Awaiting data Source: Site visits and supporting questionnaires 0129 -03. 2 -Update 2 v 9. ppt Page 3
 Key Findings . . . it’s not surprising given that all sites do it differently H? H Integ’d Site 2 12 4 M M Integ’d Site 3 ~80 ~50 H H Integ’d ? Site 4 1 1 L L Silo’d GCV Validation Use of IT 31 Poor H X XXX Best in Class M? In. Good house H X No Pack Tech M? Inhouse M X “Crippled by QA” L H X Very High Cost H Outsourced (colocate) Fragmen ted & Lots of Paper In. Good house Source: Data from site visits, site questionnaires and focus interviews 0129 -03. 2 -Update 2 v 9. ppt IT Data Management 89 Artwork Execution Site 1 Efficiency Internal Org Pack Management Operations Shared Service Component Complexity Pack Tech Artwork Market Brief Preparation Languages Markets SKUs Product Complexity Service Provision Page 4
Key Findings . . . it’s not surprising given that all sites do it differently H? H Integ’d Site 2 12 4 M M Integ’d Site 3 ~80 ~50 H H Integ’d ? Site 4 1 1 L L Silo’d GCV Validation Use of IT 31 Poor H X XXX Best in Class M? In. Good house H X No Pack Tech M? Inhouse M X “Crippled by QA” L H X Very High Cost H Outsourced (colocate) Fragmen ted & Lots of Paper In. Good house Source: Data from site visits, site questionnaires and focus interviews 0129 -03. 2 -Update 2 v 9. ppt IT Data Management 89 Artwork Execution Site 1 Efficiency Internal Org Pack Management Operations Shared Service Component Complexity Pack Tech Artwork Market Brief Preparation Languages Markets SKUs Product Complexity Service Provision Page 4
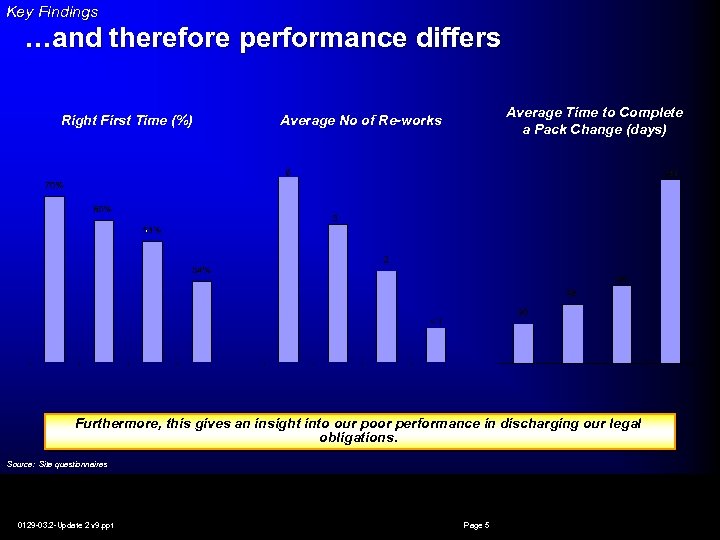 Key Findings …and therefore performance differs Right First Time (%) Average Time to Complete a Pack Change (days) Average No of Re-works Furthermore, this gives an insight into our poor performance in discharging our legal obligations. Source: Site questionnaires 0129 -03. 2 -Update 2 v 9. ppt Page 5
Key Findings …and therefore performance differs Right First Time (%) Average Time to Complete a Pack Change (days) Average No of Re-works Furthermore, this gives an insight into our poor performance in discharging our legal obligations. Source: Site questionnaires 0129 -03. 2 -Update 2 v 9. ppt Page 5
 Key Findings The development of a Pack involves many functions within XXX Legal FDA/Regulatory Medical/Product Development Sales Administration Retailer / Consumer PDD Plant Operations / QI Legal Marketing Regulatory FDA Consumer Affairs Product Development FDA / Medical Legal Regulatory / Marketing Promotions Sales / Marketing PDD Engineering/Plant Operators/QI Registered Trademark Legal Marketing FDA Product Development/ Regulatory/PDD Regulatory Legal Product Development FDA / Regulatory Registration for graphic logo Legal Marketing Corporate Identify Logo Legal Branding Marketing Construction Conformance/ Integrity PDD Engineering / Operations Legal PDD Plants/QI Primary Display Panel Marketing Category Management Can this be simplified? Source: Study by US Consumer Healthcare 0129 -03. 2 -Update 2 v 9. ppt Page 6
Key Findings The development of a Pack involves many functions within XXX Legal FDA/Regulatory Medical/Product Development Sales Administration Retailer / Consumer PDD Plant Operations / QI Legal Marketing Regulatory FDA Consumer Affairs Product Development FDA / Medical Legal Regulatory / Marketing Promotions Sales / Marketing PDD Engineering/Plant Operators/QI Registered Trademark Legal Marketing FDA Product Development/ Regulatory/PDD Regulatory Legal Product Development FDA / Regulatory Registration for graphic logo Legal Marketing Corporate Identify Logo Legal Branding Marketing Construction Conformance/ Integrity PDD Engineering / Operations Legal PDD Plants/QI Primary Display Panel Marketing Category Management Can this be simplified? Source: Study by US Consumer Healthcare 0129 -03. 2 -Update 2 v 9. ppt Page 6
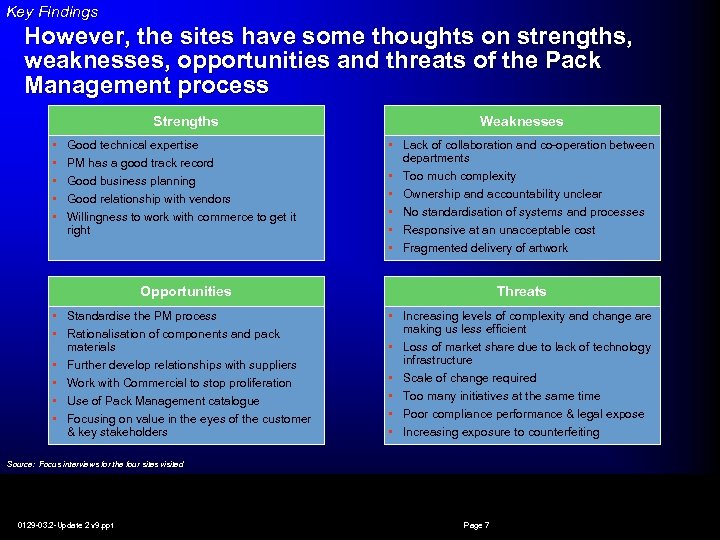 Key Findings However, the sites have some thoughts on strengths, weaknesses, opportunities and threats of the Pack Management process Strengths • • • Good technical expertise PM has a good track record Good business planning Good relationship with vendors Willingness to work with commerce to get it right Weaknesses • Lack of collaboration and co-operation between departments • Too much complexity • Ownership and accountability unclear • No standardisation of systems and processes • Responsive at an unacceptable cost • Fragmented delivery of artwork Opportunities • Standardise the PM process • Rationalisation of components and pack materials • Further develop relationships with suppliers • Work with Commercial to stop proliferation • Use of Pack Management catalogue • Focusing on value in the eyes of the customer & key stakeholders Threats • Increasing levels of complexity and change are making us less efficient • Loss of market share due to lack of technology infrastructure • Scale of change required • Too many initiatives at the same time • Poor compliance performance & legal expose • Increasing exposure to counterfeiting Source: Focus interviews for the four sites visited 0129 -03. 2 -Update 2 v 9. ppt Page 7
Key Findings However, the sites have some thoughts on strengths, weaknesses, opportunities and threats of the Pack Management process Strengths • • • Good technical expertise PM has a good track record Good business planning Good relationship with vendors Willingness to work with commerce to get it right Weaknesses • Lack of collaboration and co-operation between departments • Too much complexity • Ownership and accountability unclear • No standardisation of systems and processes • Responsive at an unacceptable cost • Fragmented delivery of artwork Opportunities • Standardise the PM process • Rationalisation of components and pack materials • Further develop relationships with suppliers • Work with Commercial to stop proliferation • Use of Pack Management catalogue • Focusing on value in the eyes of the customer & key stakeholders Threats • Increasing levels of complexity and change are making us less efficient • Loss of market share due to lack of technology infrastructure • Scale of change required • Too many initiatives at the same time • Poor compliance performance & legal expose • Increasing exposure to counterfeiting Source: Focus interviews for the four sites visited 0129 -03. 2 -Update 2 v 9. ppt Page 7
 Key Findings Verona’s relatively simple pack catalogue has saved ~7% in Pack Management labour, reduced errors, improved process management and reduced lead time • • Verona ‘Pack Catalogue Scope’ • Form based requests single electronic repository for Pack Management documents History and status Before Key Benefits • • • Manual collection of performance data Manual/verbal management of workload Manual document creation Manual pharma code allocation Manual search for incomplete brief data by several people Many forms for the same activity After • • • Automated document - no risks of confusion Pharma code automatically allocated Brief finalising automated One form for all activity Easy search and view of all documents in real time Management information automatically provided Assumptions: 2, 625 changes per year; 35 hours per week x 45 weeks per year 1 FTE; PM FTEs = 25 Source: Verona site visit It is also facilitating the standardisation of the future range 0129 -03. 2 -Update 2 v 9. ppt Page 8 30 minutes per brief before, 3 minutes after
Key Findings Verona’s relatively simple pack catalogue has saved ~7% in Pack Management labour, reduced errors, improved process management and reduced lead time • • Verona ‘Pack Catalogue Scope’ • Form based requests single electronic repository for Pack Management documents History and status Before Key Benefits • • • Manual collection of performance data Manual/verbal management of workload Manual document creation Manual pharma code allocation Manual search for incomplete brief data by several people Many forms for the same activity After • • • Automated document - no risks of confusion Pharma code automatically allocated Brief finalising automated One form for all activity Easy search and view of all documents in real time Management information automatically provided Assumptions: 2, 625 changes per year; 35 hours per week x 45 weeks per year 1 FTE; PM FTEs = 25 Source: Verona site visit It is also facilitating the standardisation of the future range 0129 -03. 2 -Update 2 v 9. ppt Page 8 30 minutes per brief before, 3 minutes after
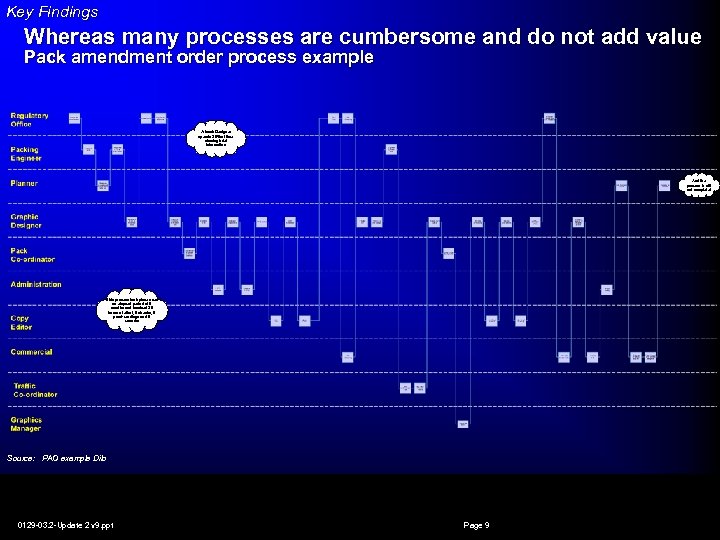 Key Findings Whereas many processes are cumbersome and do not add value Pack amendment order process example Artwork Designer spends 30% of time chasing brief information And the process is still not complete! This process took place over an elapsed period of 6 months and involved 20 hours of effort, 9 checks, 5 proof-readings and 6 reworks Source: PAO example Dilo 0129 -03. 2 -Update 2 v 9. ppt Page 9
Key Findings Whereas many processes are cumbersome and do not add value Pack amendment order process example Artwork Designer spends 30% of time chasing brief information And the process is still not complete! This process took place over an elapsed period of 6 months and involved 20 hours of effort, 9 checks, 5 proof-readings and 6 reworks Source: PAO example Dilo 0129 -03. 2 -Update 2 v 9. ppt Page 9
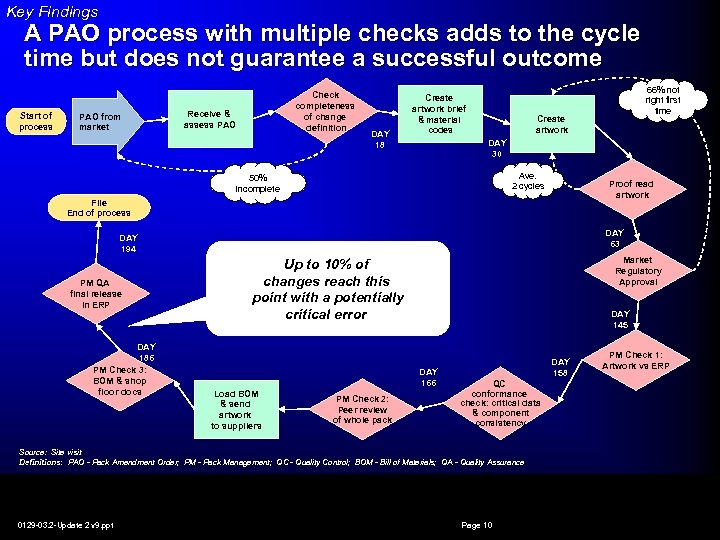 Key Findings A PAO process with multiple checks adds to the cycle time but does not guarantee a successful outcome Start of process PAO from market Check completeness of change definition Receive & assess PAO DAY 18 Create artwork brief & material codes Create artwork DAY 30 Ave. 2 cycles 50% incomplete Proof read artwork File End of process DAY 63 DAY 194 PM QA final release in ERP DAY 186 PM Check 3: BOM & shop floor docs Market Regulatory Approval Up to 10% of changes reach this point with a potentially critical error DAY 145 DAY 166 Load BOM & send artwork to suppliers PM Check 2: Peer review of whole pack DAY 158 QC conformance check: critical data & component consistency Source: Site visit Definitions: PAO - Pack Amendment Order; PM - Pack Management; QC - Quality Control; BOM - Bill of Materials; QA - Quality Assurance 0129 -03. 2 -Update 2 v 9. ppt 66% not right first time Page 10 PM Check 1: Artwork vs ERP
Key Findings A PAO process with multiple checks adds to the cycle time but does not guarantee a successful outcome Start of process PAO from market Check completeness of change definition Receive & assess PAO DAY 18 Create artwork brief & material codes Create artwork DAY 30 Ave. 2 cycles 50% incomplete Proof read artwork File End of process DAY 63 DAY 194 PM QA final release in ERP DAY 186 PM Check 3: BOM & shop floor docs Market Regulatory Approval Up to 10% of changes reach this point with a potentially critical error DAY 145 DAY 166 Load BOM & send artwork to suppliers PM Check 2: Peer review of whole pack DAY 158 QC conformance check: critical data & component consistency Source: Site visit Definitions: PAO - Pack Amendment Order; PM - Pack Management; QC - Quality Control; BOM - Bill of Materials; QA - Quality Assurance 0129 -03. 2 -Update 2 v 9. ppt 66% not right first time Page 10 PM Check 1: Artwork vs ERP
 Key Findings In many cases, the distributed/fragmented nature of pack management drives very poor quality input from the markets · ~ 200 markets · Process ~ 20, 000 SKU changes / year · The top 50 markets deal with an average of 27 factories each, in many cases having individual studios · Each studio operates a different change process – containing several Brand Managers and Regulatory contacts – as GSN changes the supply routing, markets assumptions about PM process become invalid Ý Market employee makes 1 pack change per month using a different process every time Ý Very poor quality briefs The challenge is to make it easy for commercial to get RFT. Source: CLEIT; . GSN; Site visit data 0129 -03. 2 -Update 2 v 9. ppt Page 11
Key Findings In many cases, the distributed/fragmented nature of pack management drives very poor quality input from the markets · ~ 200 markets · Process ~ 20, 000 SKU changes / year · The top 50 markets deal with an average of 27 factories each, in many cases having individual studios · Each studio operates a different change process – containing several Brand Managers and Regulatory contacts – as GSN changes the supply routing, markets assumptions about PM process become invalid Ý Market employee makes 1 pack change per month using a different process every time Ý Very poor quality briefs The challenge is to make it easy for commercial to get RFT. Source: CLEIT; . GSN; Site visit data 0129 -03. 2 -Update 2 v 9. ppt Page 11
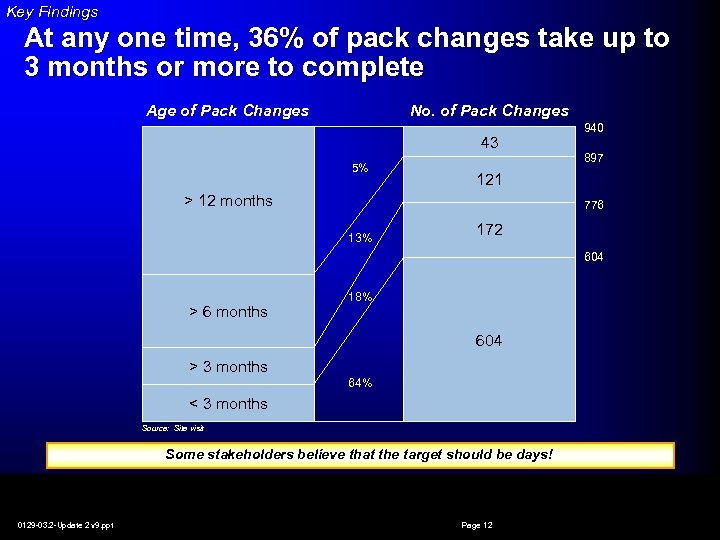 Key Findings At any one time, 36% of pack changes take up to 3 months or more to complete Age of Pack Changes No. of Pack Changes 43 5% 940 897 121 > 12 months 776 13% 172 604 > 6 months 18% 604 > 3 months 64% < 3 months Source: Site visit Some stakeholders believe that the target should be days! 0129 -03. 2 -Update 2 v 9. ppt Page 12
Key Findings At any one time, 36% of pack changes take up to 3 months or more to complete Age of Pack Changes No. of Pack Changes 43 5% 940 897 121 > 12 months 776 13% 172 604 > 6 months 18% 604 > 3 months 64% < 3 months Source: Site visit Some stakeholders believe that the target should be days! 0129 -03. 2 -Update 2 v 9. ppt Page 12
 Key Findings What are our competitors doing? Conva. Tec a Bristol-Myers Squibb company Eli Lilly · · The world's leading manufacturer of ostomy care, modern wound and skin care products have implemented an electronic system for artwork tracking and electronic approvals. They have also changed from pre-printed cartons to “generic” cartons with label printed, validated and applied on line – cost reduction – cycle time reduction – reduced component inventory – small batch sizes (>1000 packs) – virtually no packaging component waste – significantly reduced finished pack inventory Have developed an online printing and packaging system in their Glessen facility in Germany. The line handles all orders of less than 5000 packs, which supplies 70 countries with 750 SKU’s. 14 million vials per annum – delivery time of new orders cut from 17 weeks to 5 days – 70% reduction in packaging materials cost – 30% overall cost reduction – waste product/materials cut from 12% to near 0% Novo Nordisk Bristol-Myers Squibb · · BMS are implementing Printcafe’s enterprise-wide Web solution, to procure commercial print across the company allowing them to purchase and manage printing projects across multiple locations and departments · Printcafe's print supply chain solutions have been installed by more than 4, 000 customers worldwide, including 24 of the 25 largest printing companies in North America and over 50 businesses in the Fortune 1000 including AOL Time Warner, Hewlett-Packard, and General Motors. Have developed a computerised validated system for producing packaging materials for pharmaceutical products in different languages. The system is currently up & running in 39 countries. Packaging materials can now be made within 2 hours. Source: Press & Trade journals 0129 -03. 2 -Update 2 v 9. ppt Page 13
Key Findings What are our competitors doing? Conva. Tec a Bristol-Myers Squibb company Eli Lilly · · The world's leading manufacturer of ostomy care, modern wound and skin care products have implemented an electronic system for artwork tracking and electronic approvals. They have also changed from pre-printed cartons to “generic” cartons with label printed, validated and applied on line – cost reduction – cycle time reduction – reduced component inventory – small batch sizes (>1000 packs) – virtually no packaging component waste – significantly reduced finished pack inventory Have developed an online printing and packaging system in their Glessen facility in Germany. The line handles all orders of less than 5000 packs, which supplies 70 countries with 750 SKU’s. 14 million vials per annum – delivery time of new orders cut from 17 weeks to 5 days – 70% reduction in packaging materials cost – 30% overall cost reduction – waste product/materials cut from 12% to near 0% Novo Nordisk Bristol-Myers Squibb · · BMS are implementing Printcafe’s enterprise-wide Web solution, to procure commercial print across the company allowing them to purchase and manage printing projects across multiple locations and departments · Printcafe's print supply chain solutions have been installed by more than 4, 000 customers worldwide, including 24 of the 25 largest printing companies in North America and over 50 businesses in the Fortune 1000 including AOL Time Warner, Hewlett-Packard, and General Motors. Have developed a computerised validated system for producing packaging materials for pharmaceutical products in different languages. The system is currently up & running in 39 countries. Packaging materials can now be made within 2 hours. Source: Press & Trade journals 0129 -03. 2 -Update 2 v 9. ppt Page 13
 Key Findings The streams have been using a process to capture and quantify opportunities at site 0129 -03. 2 -Update 2 v 9. ppt Page 14
Key Findings The streams have been using a process to capture and quantify opportunities at site 0129 -03. 2 -Update 2 v 9. ppt Page 14
 Key Findings We have identified 23 individual opportunities Site Description Zebulon • Artwork/Component Visibility to Regulatory • Inadequate Specifications for Components • Pack change inefficiencies • Component Write Off • Colour Keys / Wet Proofs • Reduction in colour complexity • Market Packaging Artwork Concept Creation Verona • • Barnard Castle • • • Xochimilco • Pack change inefficiencies • Standardised Artwork Brief Process • Market Visibility of Impact of change requests • Timely approval of artwork • Repackaging and Scrapping of components • Consistent Range definition process • Full IT Support model for A/W Studios • Colour Reduction Cartons • Foil and Aged Stock Colour Reduction Cartons Reduce SKU Complexity Standardisation of components Providing Leaflets for SKUs supplied from Italy to the USA • MPD system replacement • Label Material Cost Savings • File Transfer • • • Colour reduction cartons Reduce SKU complexity Component cost – vial plugs Pack change inefficiencies SKU reduction Consolidation of hospital and trade packs Improve line change over Write off stock DHL hard copy transfer Reduction in USA leaflets Electronic PI Critical incidents Source: Site visits 0129 -03. 2 -Update 2 v 9. ppt Page 15
Key Findings We have identified 23 individual opportunities Site Description Zebulon • Artwork/Component Visibility to Regulatory • Inadequate Specifications for Components • Pack change inefficiencies • Component Write Off • Colour Keys / Wet Proofs • Reduction in colour complexity • Market Packaging Artwork Concept Creation Verona • • Barnard Castle • • • Xochimilco • Pack change inefficiencies • Standardised Artwork Brief Process • Market Visibility of Impact of change requests • Timely approval of artwork • Repackaging and Scrapping of components • Consistent Range definition process • Full IT Support model for A/W Studios • Colour Reduction Cartons • Foil and Aged Stock Colour Reduction Cartons Reduce SKU Complexity Standardisation of components Providing Leaflets for SKUs supplied from Italy to the USA • MPD system replacement • Label Material Cost Savings • File Transfer • • • Colour reduction cartons Reduce SKU complexity Component cost – vial plugs Pack change inefficiencies SKU reduction Consolidation of hospital and trade packs Improve line change over Write off stock DHL hard copy transfer Reduction in USA leaflets Electronic PI Critical incidents Source: Site visits 0129 -03. 2 -Update 2 v 9. ppt Page 15
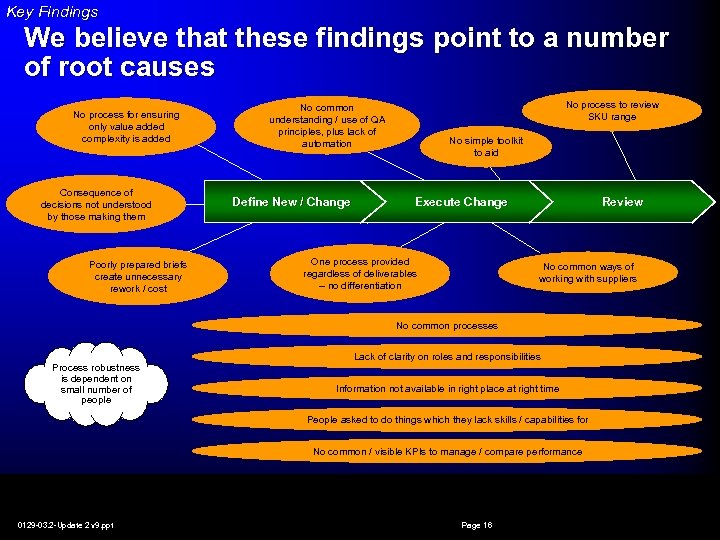 Key Findings We believe that these findings point to a number of root causes No process for ensuring only value added complexity is added Consequence of decisions not understood by those making them Poorly prepared briefs create unnecessary rework / cost No process to review SKU range No common understanding / use of QA principles, plus lack of automation Define New / Change No simple toolkit to aid Execute Change One process provided regardless of deliverables – no differentiation Review No common ways of working with suppliers No common processes Process robustness is dependent on small number of people Lack of clarity on roles and responsibilities Information not available in right place at right time People asked to do things which they lack skills / capabilities for No common / visible KPIs to manage / compare performance 0129 -03. 2 -Update 2 v 9. ppt Page 16
Key Findings We believe that these findings point to a number of root causes No process for ensuring only value added complexity is added Consequence of decisions not understood by those making them Poorly prepared briefs create unnecessary rework / cost No process to review SKU range No common understanding / use of QA principles, plus lack of automation Define New / Change No simple toolkit to aid Execute Change One process provided regardless of deliverables – no differentiation Review No common ways of working with suppliers No common processes Process robustness is dependent on small number of people Lack of clarity on roles and responsibilities Information not available in right place at right time People asked to do things which they lack skills / capabilities for No common / visible KPIs to manage / compare performance 0129 -03. 2 -Update 2 v 9. ppt Page 16


