ba1d7b50008c17848dc5520d75c0c2c4.ppt
- Количество слайдов: 89
 JUNE 2012 REGULATORY MANAGEMENT Marie D’Amico, GRACP, CCRP Manager, RTOG Regulatory Affairs www. rtog. org 1
JUNE 2012 REGULATORY MANAGEMENT Marie D’Amico, GRACP, CCRP Manager, RTOG Regulatory Affairs www. rtog. org 1
 Disclaimer The following presentation is being given for RTOG studies only. The information represented here while current may change due to changes within RTOG internal processes or because of federal regulatory guidelines. Therefore, you may not reproduce or distribute any of the presentation without obtaining RTOG’s prior written consent. www. rtog. org 2
Disclaimer The following presentation is being given for RTOG studies only. The information represented here while current may change due to changes within RTOG internal processes or because of federal regulatory guidelines. Therefore, you may not reproduce or distribute any of the presentation without obtaining RTOG’s prior written consent. www. rtog. org 2
 JUNE 2012 Regulatory Documents & the CTSU www. rtog. org 4
JUNE 2012 Regulatory Documents & the CTSU www. rtog. org 4
 Regulatory Documents All regulatory documents for US, Canadian & Non North American sites are submitted to the CTSU for cataloguing and data entry – – Continuing Reviews Approvals of amendments IRB initial study approvals IRB approved consent forms • (translation required for Non English speaking countries/provinces) www. rtog. org 5
Regulatory Documents All regulatory documents for US, Canadian & Non North American sites are submitted to the CTSU for cataloguing and data entry – – Continuing Reviews Approvals of amendments IRB initial study approvals IRB approved consent forms • (translation required for Non English speaking countries/provinces) www. rtog. org 5
 Continuing IRB Approval • • IRB approval must continue until Finite Studies – All subjects accrued have completed treatment and follow up • Non Finite Studies – All subjects are deceased • Additional requirements for both finite & non finite studies – – No further accrual is anticipated All data forms have been submitted All queries to date have been resolved RTOG approves the site for early closure (see form) • IRB approval remains necessary, even after RTOG study closure and until RTOG termination unless all the above criteria/requirements have been met • Only RTOG can classify a study as "terminated" (Database locked) • Notification of Study Termination will be received via broadcast • A formal request is required for early closure when all criteria met www. rtog. org 6
Continuing IRB Approval • • IRB approval must continue until Finite Studies – All subjects accrued have completed treatment and follow up • Non Finite Studies – All subjects are deceased • Additional requirements for both finite & non finite studies – – No further accrual is anticipated All data forms have been submitted All queries to date have been resolved RTOG approves the site for early closure (see form) • IRB approval remains necessary, even after RTOG study closure and until RTOG termination unless all the above criteria/requirements have been met • Only RTOG can classify a study as "terminated" (Database locked) • Notification of Study Termination will be received via broadcast • A formal request is required for early closure when all criteria met www. rtog. org 6
 Form to Request Early IRB Closure http: //www. rtog. org/Research. Associates/Regulatory. Information/Early. IRBClosure. aspx www. rtog. org 7
Form to Request Early IRB Closure http: //www. rtog. org/Research. Associates/Regulatory. Information/Early. IRBClosure. aspx www. rtog. org 7
 Requests for Early IRB Closure • Request form must be submitted to and approved by RTOG • The Form has Multiple purposes – Request consideration for early closure with IRB – Request closure with IRB when no patients enrolled on study – Transfer IRB responsibilities • RTOG response with approval of the request is required prior to taking any closure action • Internal multi department review at RTOG HQ can take 7 -10 business days to complete • Sites will still be required to respond to queries even after study closure with the IRB www. rtog. org 8
Requests for Early IRB Closure • Request form must be submitted to and approved by RTOG • The Form has Multiple purposes – Request consideration for early closure with IRB – Request closure with IRB when no patients enrolled on study – Transfer IRB responsibilities • RTOG response with approval of the request is required prior to taking any closure action • Internal multi department review at RTOG HQ can take 7 -10 business days to complete • Sites will still be required to respond to queries even after study closure with the IRB www. rtog. org 8
 Continuing IRB Approval • IMPORTANT NOTE Full member institutions that have affiliates that use the Full Member IRB as their IRB of record must ensure that those affiliate/ Joint Center institutions had also never put any patients on studies they are considering closing with the IRB. If they have, then the main IRB must keep this study open for annual review. www. rtog. org 9
Continuing IRB Approval • IMPORTANT NOTE Full member institutions that have affiliates that use the Full Member IRB as their IRB of record must ensure that those affiliate/ Joint Center institutions had also never put any patients on studies they are considering closing with the IRB. If they have, then the main IRB must keep this study open for annual review. www. rtog. org 9
 Special Handling • Handling of all other study specific regulatory documentation is detailed in Sections 5. 0 or 7. 0 of the protocol • • • Study Agent Shipment Form (SASF) Sponsor required documentation Questionnaires Training Certifications RT Certifications • Non English speaking Canadian & Non North American sites are required to supply the Certified/Verified English translation and native language copy of the IC with their regulatory approvals • Other special procedures may exist for Non North American sites www. rtog. org 10
Special Handling • Handling of all other study specific regulatory documentation is detailed in Sections 5. 0 or 7. 0 of the protocol • • • Study Agent Shipment Form (SASF) Sponsor required documentation Questionnaires Training Certifications RT Certifications • Non English speaking Canadian & Non North American sites are required to supply the Certified/Verified English translation and native language copy of the IC with their regulatory approvals • Other special procedures may exist for Non North American sites www. rtog. org 10
 Correlative Components • Each participating institution must be prepared to participate in all correlative components/have a plan in place to facilitate participation and must offer every patient the opportunity to participate in these components. • ONLY THE PATIENT can decline to participate in the correlative components of a study • The majority of RTOG studies require varying degrees of specimen collection as part of eligibility, stratification or protocol treatment. • Sites are encouraged to become more organized in their efforts and coordinate tissue banking collections with other protocol required specimen collections whenever possible. • In the event tissue is collected/residing at another facility RTOG expects a good faith effort to obtain the retrospective specimens but the site will not be penalized if it is not possible. • Prospectively the site must inform the collecting entity that future specimens collected are expected to be forwarded along with the appropriate submission form for tissue banking as noted in the protocol. www. rtog. org 11
Correlative Components • Each participating institution must be prepared to participate in all correlative components/have a plan in place to facilitate participation and must offer every patient the opportunity to participate in these components. • ONLY THE PATIENT can decline to participate in the correlative components of a study • The majority of RTOG studies require varying degrees of specimen collection as part of eligibility, stratification or protocol treatment. • Sites are encouraged to become more organized in their efforts and coordinate tissue banking collections with other protocol required specimen collections whenever possible. • In the event tissue is collected/residing at another facility RTOG expects a good faith effort to obtain the retrospective specimens but the site will not be penalized if it is not possible. • Prospectively the site must inform the collecting entity that future specimens collected are expected to be forwarded along with the appropriate submission form for tissue banking as noted in the protocol. www. rtog. org 11
 CTSU Forms & Guidance Can be found on the RTOG website www. rtog. org under RA/Regulatory Info/CTSU Filing Requirements – – – IRB Certification Forms 310 Certification Form IRB Transmittal Forms Guidance for completing the forms RSS Flow sheet NCI Required Forms are also available here – FDA 1572 – CTEP Supplemental Investigator Data Form (IDF) – CTEP Financial Disclosure www. rtog. org 12
CTSU Forms & Guidance Can be found on the RTOG website www. rtog. org under RA/Regulatory Info/CTSU Filing Requirements – – – IRB Certification Forms 310 Certification Form IRB Transmittal Forms Guidance for completing the forms RSS Flow sheet NCI Required Forms are also available here – FDA 1572 – CTEP Supplemental Investigator Data Form (IDF) – CTEP Financial Disclosure www. rtog. org 12
 Proper Labeling Ensures Proper Processing • Please identify all information submitted to the CTSU with the institution’s NCI Code on the transmittal/certification form • If the submission covers more than one institution or an entire CCOP all assigned individual NCI institution codes covered under the approval must be listed on the transmittal/certification form or supplemental page www. rtog. org 13
Proper Labeling Ensures Proper Processing • Please identify all information submitted to the CTSU with the institution’s NCI Code on the transmittal/certification form • If the submission covers more than one institution or an entire CCOP all assigned individual NCI institution codes covered under the approval must be listed on the transmittal/certification form or supplemental page www. rtog. org 13
 CTSU Forms Transmittal/Certification www. rtog. org 14
CTSU Forms Transmittal/Certification www. rtog. org 14
 Resolving Regulatory Issues • All issues for U. S. , Canadian and Non North American sites with submitted regulatory documents should be handled directly through the CTSU. RTOG does not have access to copies of regulatory documents that have been submitted to the CTSU. www. rtog. org 15
Resolving Regulatory Issues • All issues for U. S. , Canadian and Non North American sites with submitted regulatory documents should be handled directly through the CTSU. RTOG does not have access to copies of regulatory documents that have been submitted to the CTSU. www. rtog. org 15
 Checking Registration Status Visit the CTSU Website: https: //www. ctsu. org/public/ • • • Requires a CTSU username & password Sites are identified by NCI Code Lists all requirements for study Lists items not in compliance Now available for Non North American sites www. rtog. org 16
Checking Registration Status Visit the CTSU Website: https: //www. ctsu. org/public/ • • • Requires a CTSU username & password Sites are identified by NCI Code Lists all requirements for study Lists items not in compliance Now available for Non North American sites www. rtog. org 16
 To obtain a CTSU password… www. rtog. org 17
To obtain a CTSU password… www. rtog. org 17
 Points of Contact for the CTSU • • • Help Desk: 1 -888 -823 -5923 Regulatory Help Desk: 866 -651 -CTSU (2878) E-mail for questions: ctsucontact@westat. com For Regulatory Submissions: • Fax Number: 215 -569 -0206 • E-mail : CTSURegulatory@ctsu. coccg. org Mailing Address: CTSU Regulatory Office Coalition of National Cancer Cooperative Groups 1818 Market Street, Suite 1100 Philadelphia, PA 19103 • Website: http: //members. ctsu. org/ www. rtog. org 18
Points of Contact for the CTSU • • • Help Desk: 1 -888 -823 -5923 Regulatory Help Desk: 866 -651 -CTSU (2878) E-mail for questions: ctsucontact@westat. com For Regulatory Submissions: • Fax Number: 215 -569 -0206 • E-mail : CTSURegulatory@ctsu. coccg. org Mailing Address: CTSU Regulatory Office Coalition of National Cancer Cooperative Groups 1818 Market Street, Suite 1100 Philadelphia, PA 19103 • Website: http: //members. ctsu. org/ www. rtog. org 18
 Record Retention According to the NCI Investigator Handbook: • 10. 3 Retention of Records FDA regulations require that all research records (including patient charts, case report forms, x-rays and scans that document response, IRB approvals, signed informed consent documents and all agent accountability records) must be kept by the investigator for at least 2 years after an NDA or BLA has been approved for that indication or the CTEP IND has been closed. CTEP will notify investigators when these events occur. This requirement is an explicit part of the FDA Form 1572, http: //ctep. cancer. gov/handbook/index. html • For NON IND Studies: The point of reference used to determine the length of time required for record retention for non IND studies would be RTOG’s termination date for the study, which is when the database is locked and no further information would be required. The recommended timeframe for record retention is 3 years past termination The institution’s (and IRB’s) guidelines would also need to be considered. www. rtog. org 19
Record Retention According to the NCI Investigator Handbook: • 10. 3 Retention of Records FDA regulations require that all research records (including patient charts, case report forms, x-rays and scans that document response, IRB approvals, signed informed consent documents and all agent accountability records) must be kept by the investigator for at least 2 years after an NDA or BLA has been approved for that indication or the CTEP IND has been closed. CTEP will notify investigators when these events occur. This requirement is an explicit part of the FDA Form 1572, http: //ctep. cancer. gov/handbook/index. html • For NON IND Studies: The point of reference used to determine the length of time required for record retention for non IND studies would be RTOG’s termination date for the study, which is when the database is locked and no further information would be required. The recommended timeframe for record retention is 3 years past termination The institution’s (and IRB’s) guidelines would also need to be considered. www. rtog. org 19
 Password Protected Documents on the RTOG Website • An RTOG user name and password may be required to gain access to some password protected documents • The username and password used for web registration will permit access to these documents • The application to obtain a username & password is now web based. Paper submissions will no linger be accepted. Complete the online application at the following link: http: //www. rtog. org/About. Us/RTOGPassword. Application. aspx • The usual turnaround time for password authorization requests is two business days www. rtog. org 20
Password Protected Documents on the RTOG Website • An RTOG user name and password may be required to gain access to some password protected documents • The username and password used for web registration will permit access to these documents • The application to obtain a username & password is now web based. Paper submissions will no linger be accepted. Complete the online application at the following link: http: //www. rtog. org/About. Us/RTOGPassword. Application. aspx • The usual turnaround time for password authorization requests is two business days www. rtog. org 20
 Informed Consent Concerns • RTOG provides an NCI and ACR IRB Approved consent template for each protocol • RTOG prefers no changes be made other than to add local context • IRB requested additions to the document for clarity are permitted • The Deletion of any information should be avoided to preserve the integrity of the document • The essential elements of the RTOG Informed Consent Template must remain intact • IRB required deletions of risks and alternative procedures will require written justification • Please refer to RTOG Guidance Document for making changes to the informed consent template (revised 5/2010) • http: //www. rtog. org/Link. Click. aspx? fileticket=d 5 UGGw 5 p. Ejc%3 d&tabid=309 www. rtog. org 21
Informed Consent Concerns • RTOG provides an NCI and ACR IRB Approved consent template for each protocol • RTOG prefers no changes be made other than to add local context • IRB requested additions to the document for clarity are permitted • The Deletion of any information should be avoided to preserve the integrity of the document • The essential elements of the RTOG Informed Consent Template must remain intact • IRB required deletions of risks and alternative procedures will require written justification • Please refer to RTOG Guidance Document for making changes to the informed consent template (revised 5/2010) • http: //www. rtog. org/Link. Click. aspx? fileticket=d 5 UGGw 5 p. Ejc%3 d&tabid=309 www. rtog. org 21
 Informed Consent Approval • It remains your IRB’s responsibility to review and approve the informed consent content and any changes made at the site level • A CERTFIED or VERIFIED English & native language translation of the IC is required for Non English speaking Canadian & Non North American sites • Please note the RTOG Regulatory Compliance Department does not review consents. All consent content will be reviewed at time of audit. • Problems and deficiencies with IRB approved consents found during the audit will be reported as appropriate. www. rtog. org 22
Informed Consent Approval • It remains your IRB’s responsibility to review and approve the informed consent content and any changes made at the site level • A CERTFIED or VERIFIED English & native language translation of the IC is required for Non English speaking Canadian & Non North American sites • Please note the RTOG Regulatory Compliance Department does not review consents. All consent content will be reviewed at time of audit. • Problems and deficiencies with IRB approved consents found during the audit will be reported as appropriate. www. rtog. org 22
 Broadcasts & Summary of Changes • Information relating to protocol updates and amendments can be found on the RTOG web site: www. rtog. org • RTOG suggest sites check the website regularly to assure they have received all broadcast information • NCI requires that cooperative groups provide a Summary of Changes which describes the changes made to a protocol, and indicates whether the changes are administrative (update) or treatment related (amendment) • On the website select Clinical Trials/Protocol Table, then the protocol number: – Select “Summary of Changes” under Protocol Documents scroll through the document to see all previous – Select “Broadcasts”, from left column, a list of protocol specific broadcasts will open. www. rtog. org 23
Broadcasts & Summary of Changes • Information relating to protocol updates and amendments can be found on the RTOG web site: www. rtog. org • RTOG suggest sites check the website regularly to assure they have received all broadcast information • NCI requires that cooperative groups provide a Summary of Changes which describes the changes made to a protocol, and indicates whether the changes are administrative (update) or treatment related (amendment) • On the website select Clinical Trials/Protocol Table, then the protocol number: – Select “Summary of Changes” under Protocol Documents scroll through the document to see all previous – Select “Broadcasts”, from left column, a list of protocol specific broadcasts will open. www. rtog. org 23
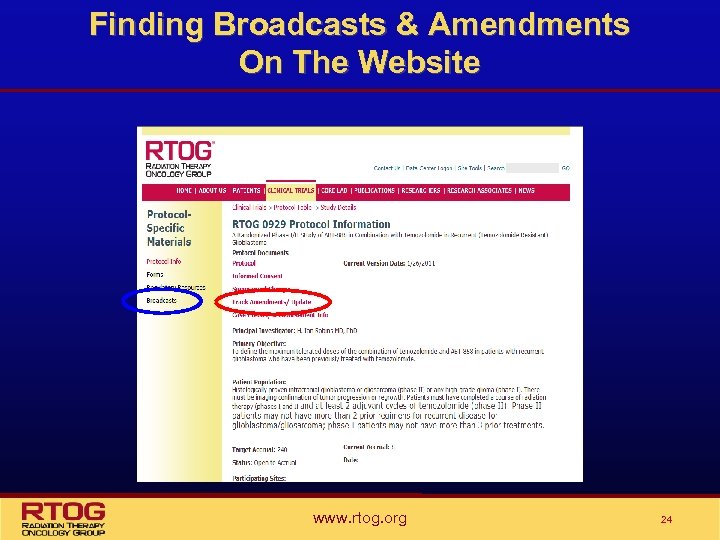 Finding Broadcasts & Amendments On The Website www. rtog. org 24
Finding Broadcasts & Amendments On The Website www. rtog. org 24
 Protocol Updates • Updates are editorial/administrative changes that RTOG is allowed to make without prior NCI approval (updates are sent to NCI for their information). • Examples of protocol updates might include: – – correction of typos changes/additions to administrative processes change in the address for data submission change in a Co-Chair’s contact information. • Per NCI, an update cannot involve patient treatment or anything that would impact patient safety. • The RTOG broadcasts indicate “IRB review of an update is not required; however, these changes must be reported to the site IRB” www. rtog. org 25
Protocol Updates • Updates are editorial/administrative changes that RTOG is allowed to make without prior NCI approval (updates are sent to NCI for their information). • Examples of protocol updates might include: – – correction of typos changes/additions to administrative processes change in the address for data submission change in a Co-Chair’s contact information. • Per NCI, an update cannot involve patient treatment or anything that would impact patient safety. • The RTOG broadcasts indicate “IRB review of an update is not required; however, these changes must be reported to the site IRB” www. rtog. org 25
 Protocol Amendments • All treatment or safety related changes to protocols are amendments, • Amendments are sent by RTOG to NCI for review and approval. • As “Per CTMB Guidelines, amendments must be reviewed and approved by local IRBs within 90 days. • For Canadian sites the broadcast of Health Canada’s approval of the amendment begins the 90 day timeframe www. rtog. org 26
Protocol Amendments • All treatment or safety related changes to protocols are amendments, • Amendments are sent by RTOG to NCI for review and approval. • As “Per CTMB Guidelines, amendments must be reviewed and approved by local IRBs within 90 days. • For Canadian sites the broadcast of Health Canada’s approval of the amendment begins the 90 day timeframe www. rtog. org 26
 JUNE 2012 Drug Procurement & Accountability www. rtog. org 27
JUNE 2012 Drug Procurement & Accountability www. rtog. org 27
 Possible Sources of Drug Supply • • Pharmaceutical Companies Third Party Distributor NCI-Pharmaceutical Management Branch (PMB) Commercially available products * More than one may apply in certain studies. www. rtog. org 28
Possible Sources of Drug Supply • • Pharmaceutical Companies Third Party Distributor NCI-Pharmaceutical Management Branch (PMB) Commercially available products * More than one may apply in certain studies. www. rtog. org 28
 Canadian Review Board (CRB) • Purpose: To identify barriers, specifically related to drug availability and differing RT standards in the various member provinces early in the development of the protocol when there is the potential to address these issues. • Initiated in April 2009 • Representation: a provincial board member from each member province was appointed by the Canadian vice chair. • Please remember to share all drug or RT related protocol concerns with your provincial board member as early as possible. www. rtog. org 29
Canadian Review Board (CRB) • Purpose: To identify barriers, specifically related to drug availability and differing RT standards in the various member provinces early in the development of the protocol when there is the potential to address these issues. • Initiated in April 2009 • Representation: a provincial board member from each member province was appointed by the Canadian vice chair. • Please remember to share all drug or RT related protocol concerns with your provincial board member as early as possible. www. rtog. org 29
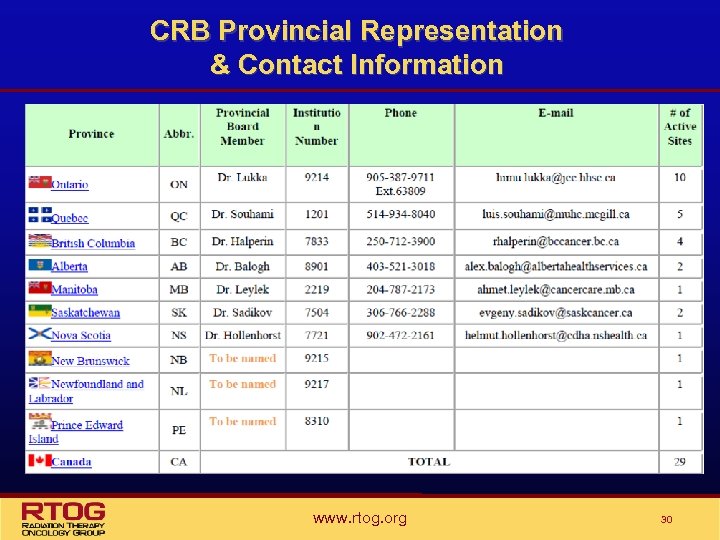 CRB Provincial Representation & Contact Information www. rtog. org 30
CRB Provincial Representation & Contact Information www. rtog. org 30
 Important Note About Non North American Sites • Non North American Institutions are advised not to submit a study for approval by their local regulatory body until their participation has been confirmed in the written notice of an approved Letter of Intent (LOI) from RTOG HQ. • All Non North American Institutions must submit an LOI to RTOG HQ indicating their interest in participating in any RTOG Study. • Formal Approval of the LOI will be received via e-mail with IMPORTANT specific instructions for moving forward • Once the LOI has been approved, documentation of completion of all applicable regulatory pre-registration requirements as noted in the protocol must be submitted directly to the CTSU before enrollment can begin. www. rtog. org 31
Important Note About Non North American Sites • Non North American Institutions are advised not to submit a study for approval by their local regulatory body until their participation has been confirmed in the written notice of an approved Letter of Intent (LOI) from RTOG HQ. • All Non North American Institutions must submit an LOI to RTOG HQ indicating their interest in participating in any RTOG Study. • Formal Approval of the LOI will be received via e-mail with IMPORTANT specific instructions for moving forward • Once the LOI has been approved, documentation of completion of all applicable regulatory pre-registration requirements as noted in the protocol must be submitted directly to the CTSU before enrollment can begin. www. rtog. org 31
 Important Note About Non North American Sites • Participation will not be possible if any part of the study specific requirements outlined in the protocol conflict with your country's Ministry of Health regulations. • It is your institution's responsibility to understand comply with all national Ministry of Health Regulations. • Translation of documents is critical. The cost of all translation would be your institution's responsibility. All regulatory documents, data and supporting documentation (when required) would need to be provided in English with certified/verified translation documentation • Your institution will be responsible for acquiring any drug noted in the protocol as commercially available. • Non North American participation in studies with a drug component will only be permitted if RTOG can procure and provide a supply of the study agent for your region www. rtog. org 32
Important Note About Non North American Sites • Participation will not be possible if any part of the study specific requirements outlined in the protocol conflict with your country's Ministry of Health regulations. • It is your institution's responsibility to understand comply with all national Ministry of Health Regulations. • Translation of documents is critical. The cost of all translation would be your institution's responsibility. All regulatory documents, data and supporting documentation (when required) would need to be provided in English with certified/verified translation documentation • Your institution will be responsible for acquiring any drug noted in the protocol as commercially available. • Non North American participation in studies with a drug component will only be permitted if RTOG can procure and provide a supply of the study agent for your region www. rtog. org 32
 International Letter of Intent (Pages 1& 2 of 3) www. rtog. org 33
International Letter of Intent (Pages 1& 2 of 3) www. rtog. org 33
 International Letter of Intent (page 3 of 3) “****Please review important information attached with response****”. • This portion of the form will be returned to the site by RTOG HQ noting approval or disapproval • A second very important document will also be provided with the approval, entitled International Letter of Intent to Participate Response, outlining expectations and requirements • This document must be retained as a checklist to assure preregistration compliance prior to patient registration/enrollment. www. rtog. org 34
International Letter of Intent (page 3 of 3) “****Please review important information attached with response****”. • This portion of the form will be returned to the site by RTOG HQ noting approval or disapproval • A second very important document will also be provided with the approval, entitled International Letter of Intent to Participate Response, outlining expectations and requirements • This document must be retained as a checklist to assure preregistration compliance prior to patient registration/enrollment. www. rtog. org 34
 Finding International Forms on the Website • International Letter of Intent (LOI) www. rtog. org 35
Finding International Forms on the Website • International Letter of Intent (LOI) www. rtog. org 35
 Pharmaceutical Company Supplied Drug Procurement • Procedure can vary by study. • Detailed instructions for obtaining drug can be found in Section 7. 0 of each protocol • First step a pre-registration requirement, as noted in Section 5. 0, submit the Study Agent Shipment Form (SASF) to CTSU • Processing of this form approves the site as eligible to receive drug – Registration/Randomization is the trigger for the initial shipment www. rtog. org 36
Pharmaceutical Company Supplied Drug Procurement • Procedure can vary by study. • Detailed instructions for obtaining drug can be found in Section 7. 0 of each protocol • First step a pre-registration requirement, as noted in Section 5. 0, submit the Study Agent Shipment Form (SASF) to CTSU • Processing of this form approves the site as eligible to receive drug – Registration/Randomization is the trigger for the initial shipment www. rtog. org 36
 SAMPLE (SASF) Study Agent Shipment Form www. rtog. org 37
SAMPLE (SASF) Study Agent Shipment Form www. rtog. org 37
 Finding RTOG SASFs On the Website Clinical Trials/Protocol Table/Select Study/Regulatory Resources www. rtog. org 38
Finding RTOG SASFs On the Website Clinical Trials/Protocol Table/Select Study/Regulatory Resources www. rtog. org 38
 Pharmaceutical Company Supplied Drug Procurement Submitting your SASF for processing as soon as the individual responsible for the drug at your institution has been identified will assist you in: – avoiding delays in the initial drug shipment – alleviating frustration and inconvenience for yourself and most importantly the patient www. rtog. org 39
Pharmaceutical Company Supplied Drug Procurement Submitting your SASF for processing as soon as the individual responsible for the drug at your institution has been identified will assist you in: – avoiding delays in the initial drug shipment – alleviating frustration and inconvenience for yourself and most importantly the patient www. rtog. org 39
 Pharmaceutical Company Supplied Drug Procurement • Realistic Treatment Start Dates – If the SASF has already been processed, drug is received much faster, generally for overnight or two day delivery. – Most distributors will not ship prior to a weekend or holiday. – Shipments are generally restricted between December 23 and January 1. – Canadian sites should take into consideration the possibility of customs delays www. rtog. org 40
Pharmaceutical Company Supplied Drug Procurement • Realistic Treatment Start Dates – If the SASF has already been processed, drug is received much faster, generally for overnight or two day delivery. – Most distributors will not ship prior to a weekend or holiday. – Shipments are generally restricted between December 23 and January 1. – Canadian sites should take into consideration the possibility of customs delays www. rtog. org 40
 NCI Drug Procurement • The Pharmaceutical Management Branch (PMB) of the NCI is responsible for supplying study agent for studies where the NCI holds the IND. • When the PMB supplies the drug for a study they are also responsible for providing the Investigator Brochure. • On-line Agent Order Processing (OAOP) https: //eappsctep. nci. nih. gov/OAOP/pages/login. jspx is now required for PMB provided drug • • • NIH Form 986/Clinical Drug Request – OBSOLETE OAOP Training Guide available All NCI Forms are available on the NCI website at http: //ctep. cancer. gov/forms/index. html • Investigational Drug Accountability Record Form (DARF) • Drug Return • Drug Transfer www. rtog. org 41
NCI Drug Procurement • The Pharmaceutical Management Branch (PMB) of the NCI is responsible for supplying study agent for studies where the NCI holds the IND. • When the PMB supplies the drug for a study they are also responsible for providing the Investigator Brochure. • On-line Agent Order Processing (OAOP) https: //eappsctep. nci. nih. gov/OAOP/pages/login. jspx is now required for PMB provided drug • • • NIH Form 986/Clinical Drug Request – OBSOLETE OAOP Training Guide available All NCI Forms are available on the NCI website at http: //ctep. cancer. gov/forms/index. html • Investigational Drug Accountability Record Form (DARF) • Drug Return • Drug Transfer www. rtog. org 41
 Canadian Drug Accountability • There is a PMB approved Canadian Drug Accountability Form, which can be used by Canadian institutions for both PMB and pharmaceutically supplied drug. • This form is available on the RTOG website with the other Canadian resources www. rtog. org 42
Canadian Drug Accountability • There is a PMB approved Canadian Drug Accountability Form, which can be used by Canadian institutions for both PMB and pharmaceutically supplied drug. • This form is available on the RTOG website with the other Canadian resources www. rtog. org 42
 NCI Drug Procurement Investigators must be registered with the NCI and have an Investigator NCI Number to receive drug from the NCI. Maintenance of this NCI Investigator Number requires the annual submission of: – FDA 1572 with CVs – signed Supplemental Investigator Data Form (IDF) – Financial Disclosure Form NCI/PMB Contact Information: Phone: (301) 496 -5725 Fax: (301) 402 -0429 www. rtog. org 43
NCI Drug Procurement Investigators must be registered with the NCI and have an Investigator NCI Number to receive drug from the NCI. Maintenance of this NCI Investigator Number requires the annual submission of: – FDA 1572 with CVs – signed Supplemental Investigator Data Form (IDF) – Financial Disclosure Form NCI/PMB Contact Information: Phone: (301) 496 -5725 Fax: (301) 402 -0429 www. rtog. org 43
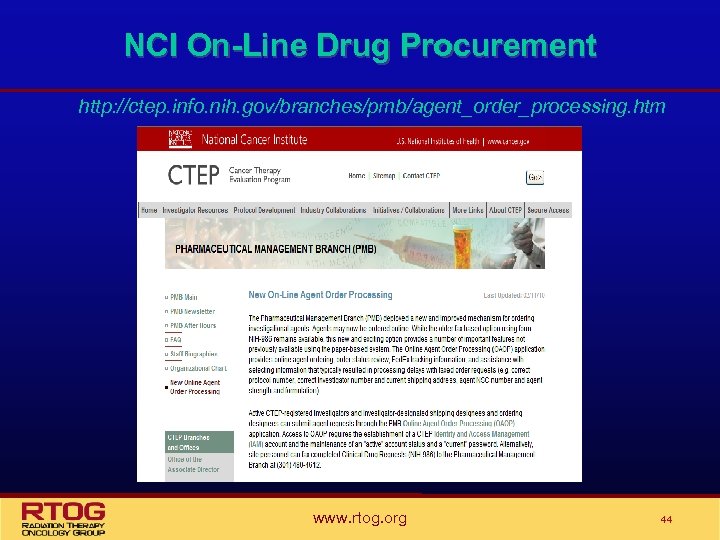 NCI On-Line Drug Procurement http: //ctep. info. nih. gov/branches/pmb/agent_order_processing. htm www. rtog. org 44
NCI On-Line Drug Procurement http: //ctep. info. nih. gov/branches/pmb/agent_order_processing. htm www. rtog. org 44
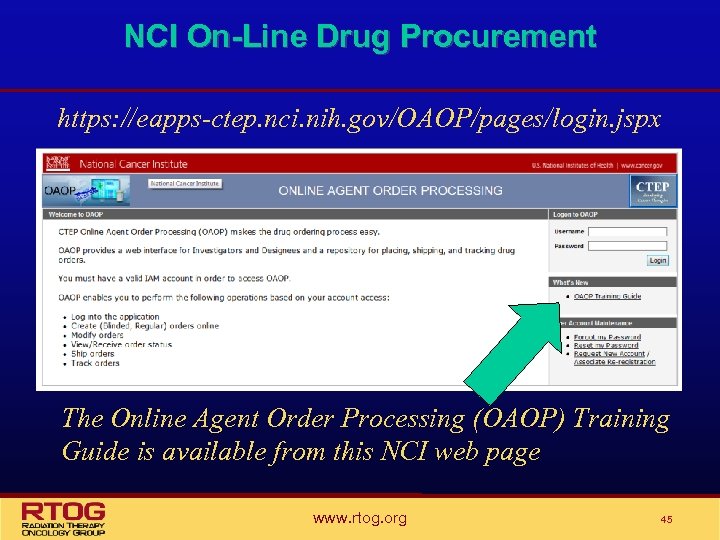 NCI On-Line Drug Procurement https: //eapps-ctep. nci. nih. gov/OAOP/pages/login. jspx The Online Agent Order Processing (OAOP) Training Guide is available from this NCI web page www. rtog. org 45
NCI On-Line Drug Procurement https: //eapps-ctep. nci. nih. gov/OAOP/pages/login. jspx The Online Agent Order Processing (OAOP) Training Guide is available from this NCI web page www. rtog. org 45
 Drug Accountability Record Forms NCI RTOG www. rtog. org 46
Drug Accountability Record Forms NCI RTOG www. rtog. org 46
 Finding RTOG Accountability Records on the Website Clinical Trials/Protocol Table/Select Study/Regulatory Resources www. rtog. org 47
Finding RTOG Accountability Records on the Website Clinical Trials/Protocol Table/Select Study/Regulatory Resources www. rtog. org 47
 Commercially Available Products • Supplied free of charge through pharmaceutical support directly from the pharmaceutical company or via a third party distributor OR • Obtained via prescription from the physician www. rtog. org 48
Commercially Available Products • Supplied free of charge through pharmaceutical support directly from the pharmaceutical company or via a third party distributor OR • Obtained via prescription from the physician www. rtog. org 48
 End of Study Drug Disposition • Can vary by sponsor/protocol • Records of accountability documentation may require submission – Receipt of Drug – Dispensation of Drug – Disposition of Drug (returned/destroyed used/unused) • Instructions usually documented in the protocol • Institutional Drug Destruction Policy or SOP – Required for site drug destruction www. rtog. org 49
End of Study Drug Disposition • Can vary by sponsor/protocol • Records of accountability documentation may require submission – Receipt of Drug – Dispensation of Drug – Disposition of Drug (returned/destroyed used/unused) • Instructions usually documented in the protocol • Institutional Drug Destruction Policy or SOP – Required for site drug destruction www. rtog. org 49
 Return/Destruction of Study Drug • When applicable, unexpired, unused, unopened, nontransferable agents remaining after a study has been permanently closed to accrual by RTOG must be returned to the distributor within 90 days of the RTOG closure broadcast. • If end of study drug destruction is noted in the protocol it must be carried out within 90 days of study closure and accountability records must be retained. • If the institution lacks a written policy or SOP pertaining to drug destruction, drug must be returned to the distributor for destruction within 90 days of study closure. www. rtog. org 50
Return/Destruction of Study Drug • When applicable, unexpired, unused, unopened, nontransferable agents remaining after a study has been permanently closed to accrual by RTOG must be returned to the distributor within 90 days of the RTOG closure broadcast. • If end of study drug destruction is noted in the protocol it must be carried out within 90 days of study closure and accountability records must be retained. • If the institution lacks a written policy or SOP pertaining to drug destruction, drug must be returned to the distributor for destruction within 90 days of study closure. www. rtog. org 50
 Blinded Studies RTOG saw a marked increase it drug errors on blinded RTOG studies particularly RTOG 0825. Please take the time to consider the importance of reviewing drug administration and safety policies with related staff in an effort to eliminate these errors. RTOG suggests a double or even triple check of the drug label and patient ID or a two person check similar to hospital drug transfusion policies. Contact RTOG HQ if you would like assistance with training your research pharmacy or staff. www. rtog. org 51
Blinded Studies RTOG saw a marked increase it drug errors on blinded RTOG studies particularly RTOG 0825. Please take the time to consider the importance of reviewing drug administration and safety policies with related staff in an effort to eliminate these errors. RTOG suggests a double or even triple check of the drug label and patient ID or a two person check similar to hospital drug transfusion policies. Contact RTOG HQ if you would like assistance with training your research pharmacy or staff. www. rtog. org 51
 Case & Point www. rtog. org 52
Case & Point www. rtog. org 52
 Blinded Studies Important Points to remember: • The number of RTOG blinded drug studies is increasing. • The order of the initials entered during registration significantly impacts our ability to quickly unblind a case in an emergency situation • Initials must be entered as First Middle Last (FML) or F-L if there is no middle • The full DOB (MM/DD/YYYY) is also required for all blinded studies to allow us to confirm we have the right patient during unblinding in the event there are cases with the same initials www. rtog. org 53
Blinded Studies Important Points to remember: • The number of RTOG blinded drug studies is increasing. • The order of the initials entered during registration significantly impacts our ability to quickly unblind a case in an emergency situation • Initials must be entered as First Middle Last (FML) or F-L if there is no middle • The full DOB (MM/DD/YYYY) is also required for all blinded studies to allow us to confirm we have the right patient during unblinding in the event there are cases with the same initials www. rtog. org 53
 Emergency Unblinding Procedures “Decisions to break the code will only be considered for life threatening events or extraordinary clinical circumstances where it can be demonstrated knowledge of the drug treatment assignment will affect clinical judgment. ” • During RTOG business hours (8: 30 AM-5 PM ET) call RTOG HQ at 215 -574 -3150 and ask to speak to a statistician. • After hours (5 PM-8: 30 AM ET) call 215 -459 -3576 • Calls received after hours or during weekends and holidays will be returned as soon as possible. Please leave your name, contact information, the first and last name of the patient who experienced the event, the study & case number and the reason for the unblinding request. • If the unblinding request pertains to RTOG 0825 and tumor progression, please contact Treena Trotman at 215 -940 -8913 during regular business hours www. rtog. org 54
Emergency Unblinding Procedures “Decisions to break the code will only be considered for life threatening events or extraordinary clinical circumstances where it can be demonstrated knowledge of the drug treatment assignment will affect clinical judgment. ” • During RTOG business hours (8: 30 AM-5 PM ET) call RTOG HQ at 215 -574 -3150 and ask to speak to a statistician. • After hours (5 PM-8: 30 AM ET) call 215 -459 -3576 • Calls received after hours or during weekends and holidays will be returned as soon as possible. Please leave your name, contact information, the first and last name of the patient who experienced the event, the study & case number and the reason for the unblinding request. • If the unblinding request pertains to RTOG 0825 and tumor progression, please contact Treena Trotman at 215 -940 -8913 during regular business hours www. rtog. org 54
 Emergency Unblinding • Requests to unblind either case in the situation of a corruption or crossover in drug administration will likely result in both cases being removed from the study • The Emergency Unblinding line is for true emergencies only • Please do not leave messages pertaining to anything but emergency unblinding • This is NOT a catch all after hours office line • Messages left for any other reason will not be forwarded or receive a response www. rtog. org 55
Emergency Unblinding • Requests to unblind either case in the situation of a corruption or crossover in drug administration will likely result in both cases being removed from the study • The Emergency Unblinding line is for true emergencies only • Please do not leave messages pertaining to anything but emergency unblinding • This is NOT a catch all after hours office line • Messages left for any other reason will not be forwarded or receive a response www. rtog. org 55
 Minimum Required Information for Emergency Unblinding Request The unblinding officer will contact you for further information. www. rtog. org 56
Minimum Required Information for Emergency Unblinding Request The unblinding officer will contact you for further information. www. rtog. org 56
 Protocol Deviations • CTEP has a policy in place related to the issuance of waivers for protocol deviations: • The policy applies to all components of CTEP-approved protocols, including eligibility criteria, treatment schedules, dose modifications, toxicity assessment, response criteria, and statistical aspects. • Link to Policy: http: //ctep. cancer. gov/protocol. Development/policies_deviation s. htm • CTEP does not issue or approve any waivers for protocol deviations www. rtog. org 57
Protocol Deviations • CTEP has a policy in place related to the issuance of waivers for protocol deviations: • The policy applies to all components of CTEP-approved protocols, including eligibility criteria, treatment schedules, dose modifications, toxicity assessment, response criteria, and statistical aspects. • Link to Policy: http: //ctep. cancer. gov/protocol. Development/policies_deviation s. htm • CTEP does not issue or approve any waivers for protocol deviations www. rtog. org 57
 NCI Deviation Policy www. rtog. org 58
NCI Deviation Policy www. rtog. org 58
 JUNE 2012 RTOG SAE Safety Reporting www. rtog. org 59
JUNE 2012 RTOG SAE Safety Reporting www. rtog. org 59
 Guidelines for AE Reporting Ø The protocol provides basic instruction regarding adverse events (AE) and Protocol Specific Special Reporting (PSSR). Ø Those serious adverse events meeting the criteria described in the protocol must be reported via the Adverse Event Expedited Reporting System (Ad. EERS) Ø Ad. EERS related training and guidance is also available from the NCI. www. rtog. org 60
Guidelines for AE Reporting Ø The protocol provides basic instruction regarding adverse events (AE) and Protocol Specific Special Reporting (PSSR). Ø Those serious adverse events meeting the criteria described in the protocol must be reported via the Adverse Event Expedited Reporting System (Ad. EERS) Ø Ad. EERS related training and guidance is also available from the NCI. www. rtog. org 60
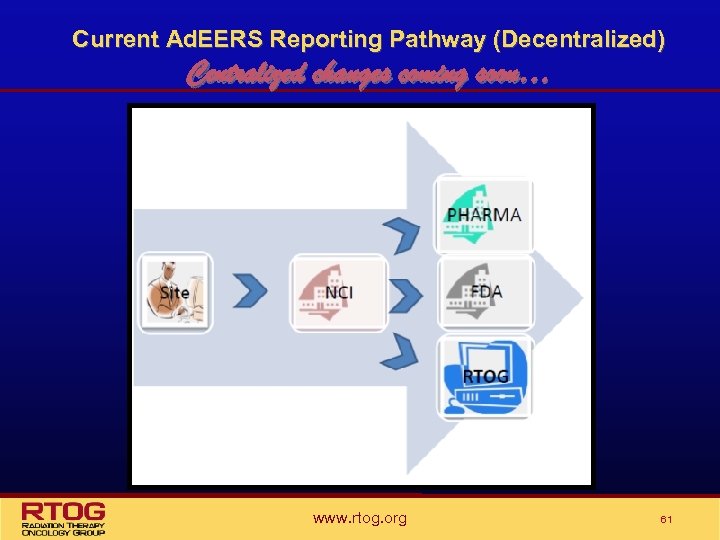 Current Ad. EERS Reporting Pathway (Decentralized) Centralized changes coming soon… www. rtog. org 61
Current Ad. EERS Reporting Pathway (Decentralized) Centralized changes coming soon… www. rtog. org 61
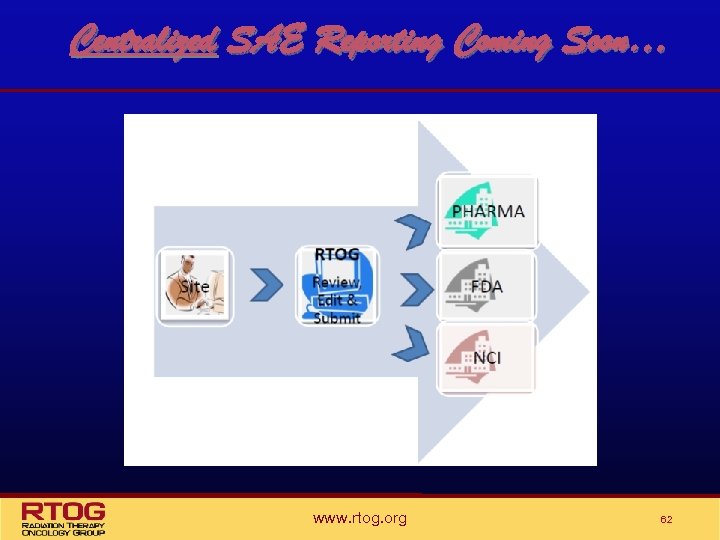 Centralized SAE Reporting Coming Soon… www. rtog. org 62
Centralized SAE Reporting Coming Soon… www. rtog. org 62
 RTOG Centralized SAE Review • An SAE Coordinator position has been created at RTOG HQ – To coordinate the timely review of submitted information, perform necessary edits for accuracy & clarity and submit report s of all UNEXPECTED and RELATED SAEs to the NCI, pharmaceutical sponsor and to the FDA as required by regulations. • Timely feedback will be required to meet the regulatory reporting timeline • The site RA should remain alert for requests from the SAE Coordinator in an effort to obtain information to ensure the clarity and accuracy of the SAE report www. rtog. org 63
RTOG Centralized SAE Review • An SAE Coordinator position has been created at RTOG HQ – To coordinate the timely review of submitted information, perform necessary edits for accuracy & clarity and submit report s of all UNEXPECTED and RELATED SAEs to the NCI, pharmaceutical sponsor and to the FDA as required by regulations. • Timely feedback will be required to meet the regulatory reporting timeline • The site RA should remain alert for requests from the SAE Coordinator in an effort to obtain information to ensure the clarity and accuracy of the SAE report www. rtog. org 63
 Centralized reporting expectations for the reporter… As the reporter you will • Receive notice of all AE/SAEs you submit. – SAEs deemed unexpected and related will be designated high priority and may require prompt feedback from you in order to meet NCI and FDA reporting requirements • to ensure clarity and accuracy of the information provided • The timeline for reporting to regulatory authorities begins with your submission Ø time is an important factor Ø 5 calendar days for fatal and life-threatening unexpected, related SAE Ø 10 calendar days for all other unexpected, related SAE www. rtog. org 64
Centralized reporting expectations for the reporter… As the reporter you will • Receive notice of all AE/SAEs you submit. – SAEs deemed unexpected and related will be designated high priority and may require prompt feedback from you in order to meet NCI and FDA reporting requirements • to ensure clarity and accuracy of the information provided • The timeline for reporting to regulatory authorities begins with your submission Ø time is an important factor Ø 5 calendar days for fatal and life-threatening unexpected, related SAE Ø 10 calendar days for all other unexpected, related SAE www. rtog. org 64
 Variations… In addition to the previously stated expectations it is important to note Foundation Studies (3500 series) will • Require the completion of an SAE Report Form in place of Ad. EERS • RTOG will transcribe the information provided to the FDA Med. Watch platform and report to regulatory authorities • Likely require additional input form the reporter and or investigator. • The site will receive a copy of the Med. Watch when submitted www. rtog. org 65
Variations… In addition to the previously stated expectations it is important to note Foundation Studies (3500 series) will • Require the completion of an SAE Report Form in place of Ad. EERS • RTOG will transcribe the information provided to the FDA Med. Watch platform and report to regulatory authorities • Likely require additional input form the reporter and or investigator. • The site will receive a copy of the Med. Watch when submitted www. rtog. org 65
 AE/SAE Coordinator Sara Mc. Cartney www. rtog. org 66
AE/SAE Coordinator Sara Mc. Cartney www. rtog. org 66
 The NEW FDA Final Rule for Expedited Reporting • The Final Rule: • 21 CFR Parts 312 and 320 [Docket No. FDA-2000 -N-0108] (formerly Docket No. 00 N-1484) • RIN 0910 -AG 13 • Investigational New Drug Safety Reporting Requirements for Human Drug and Biological Products and Safety Reporting Requirements for Bioavailability and Bioequivalence Studies in Humans • http: //frwebgate. access. gpo. gov/cgibin/getdoc. cgi? dbname=2010_register&docid=fr 29 se 10 -3. pdf • This regulation requires changes to expedited serious adverse event reporting requirements for investigators to IND sponsors such as NCI/CTEP www. rtog. org 67
The NEW FDA Final Rule for Expedited Reporting • The Final Rule: • 21 CFR Parts 312 and 320 [Docket No. FDA-2000 -N-0108] (formerly Docket No. 00 N-1484) • RIN 0910 -AG 13 • Investigational New Drug Safety Reporting Requirements for Human Drug and Biological Products and Safety Reporting Requirements for Bioavailability and Bioequivalence Studies in Humans • http: //frwebgate. access. gpo. gov/cgibin/getdoc. cgi? dbname=2010_register&docid=fr 29 se 10 -3. pdf • This regulation requires changes to expedited serious adverse event reporting requirements for investigators to IND sponsors such as NCI/CTEP www. rtog. org 67
 Pharmaceutical/Industry SAE Reporting Requirements Special considerations when reporting on pharmaceutically supported studies using the Ad. EERS system • Pharmaceutically supported studies often require additional reporting over and above that which is required by the NCI. • Complying with pharmaceutical standards will require bypassing the “reporting not required” screen in Ad. EERS and going on to complete and submit the report. www. rtog. org 68
Pharmaceutical/Industry SAE Reporting Requirements Special considerations when reporting on pharmaceutically supported studies using the Ad. EERS system • Pharmaceutically supported studies often require additional reporting over and above that which is required by the NCI. • Complying with pharmaceutical standards will require bypassing the “reporting not required” screen in Ad. EERS and going on to complete and submit the report. www. rtog. org 68
 SAE Definition A Serious Adverse Event (SAE) is defined by the Code of Federal Regulations (CFR) as: Any untoward medical occurrence that at any dose results in • death • is life threatening • requires inpatient hospitalization or prolongation of existing hospitalization • results in persistent or significant disability/incapacity • or is a congenital anomaly/birth defect. The phrase “at any dose” should be replaced with “during protocol treatment and 30 days after” for RTOG purposes to capture SAEs that occur during any part of protocol therapy including radiation therapy or surgery. Please note reporting outside the 30 day window is required when the event is determined to be possibly, probably or definitely related to any part of protocol treatment. www. rtog. org 69
SAE Definition A Serious Adverse Event (SAE) is defined by the Code of Federal Regulations (CFR) as: Any untoward medical occurrence that at any dose results in • death • is life threatening • requires inpatient hospitalization or prolongation of existing hospitalization • results in persistent or significant disability/incapacity • or is a congenital anomaly/birth defect. The phrase “at any dose” should be replaced with “during protocol treatment and 30 days after” for RTOG purposes to capture SAEs that occur during any part of protocol therapy including radiation therapy or surgery. Please note reporting outside the 30 day window is required when the event is determined to be possibly, probably or definitely related to any part of protocol treatment. www. rtog. org 69
 Important Points • Hospitalization is defined as an admission to an inpatient bed for at least 24 hours. • Grade, attribution or expectedness are not a consideration for RTOG in the reporting of SAEs, simply meeting the criteria in the SAE definition makes the event an SAE. • The new FDA Final Rule RE: Expedited Reporting now also requires all SAEs be reported unless the protocol states otherwise www. rtog. org 70
Important Points • Hospitalization is defined as an admission to an inpatient bed for at least 24 hours. • Grade, attribution or expectedness are not a consideration for RTOG in the reporting of SAEs, simply meeting the criteria in the SAE definition makes the event an SAE. • The new FDA Final Rule RE: Expedited Reporting now also requires all SAEs be reported unless the protocol states otherwise www. rtog. org 70
 Medically Significant Events • Medical and scientific judgment should be exercised in deciding whether reporting as an SAE is appropriate in other situations, such as important medical events that may not be immediately life threatening or result in death or hospitalization but may jeopardize the patient or may require intervention to prevent one of the outcomes listed in the SAE definition. www. rtog. org 71
Medically Significant Events • Medical and scientific judgment should be exercised in deciding whether reporting as an SAE is appropriate in other situations, such as important medical events that may not be immediately life threatening or result in death or hospitalization but may jeopardize the patient or may require intervention to prevent one of the outcomes listed in the SAE definition. www. rtog. org 71
 Site Initiated SAE/Safety Reporting Serious Adverse Events (SAE) • Ad. EERS is the required reporting format for SAEs on all NCI supported RTOG studies • A study specific SAE form will be used for upcoming Foundation Studies (3500 series studies) for transcription by RTOG into the FDA Med. Watch Form • All events that meet the criteria for an SAE must be reported regardless of grade or attribution • SAE/Safety Reporting is separate, and in addition to Data Management AE reporting (on the CRF) • All SAEs must be reported to your IRB www. rtog. org 72
Site Initiated SAE/Safety Reporting Serious Adverse Events (SAE) • Ad. EERS is the required reporting format for SAEs on all NCI supported RTOG studies • A study specific SAE form will be used for upcoming Foundation Studies (3500 series studies) for transcription by RTOG into the FDA Med. Watch Form • All events that meet the criteria for an SAE must be reported regardless of grade or attribution • SAE/Safety Reporting is separate, and in addition to Data Management AE reporting (on the CRF) • All SAEs must be reported to your IRB www. rtog. org 72
 SAE Serious Adverse Events (SAEs) are defined by FDA and therefore seriousness (not severity) serves as a guide for defining regulatory reporting obligations for patient/subject safety. ‘Seriousness’ is based on patient/event outcome or action criteria usually associated with events that pose a threat to a patient’s life or functioning. FDA Federal Regulations require IND sponsors to report serious adverse events via Expedited reporting. The two terms “serious” and “severe” are not interchangeable and their meanings should not be confused. www. rtog. org 73
SAE Serious Adverse Events (SAEs) are defined by FDA and therefore seriousness (not severity) serves as a guide for defining regulatory reporting obligations for patient/subject safety. ‘Seriousness’ is based on patient/event outcome or action criteria usually associated with events that pose a threat to a patient’s life or functioning. FDA Federal Regulations require IND sponsors to report serious adverse events via Expedited reporting. The two terms “serious” and “severe” are not interchangeable and their meanings should not be confused. www. rtog. org 73
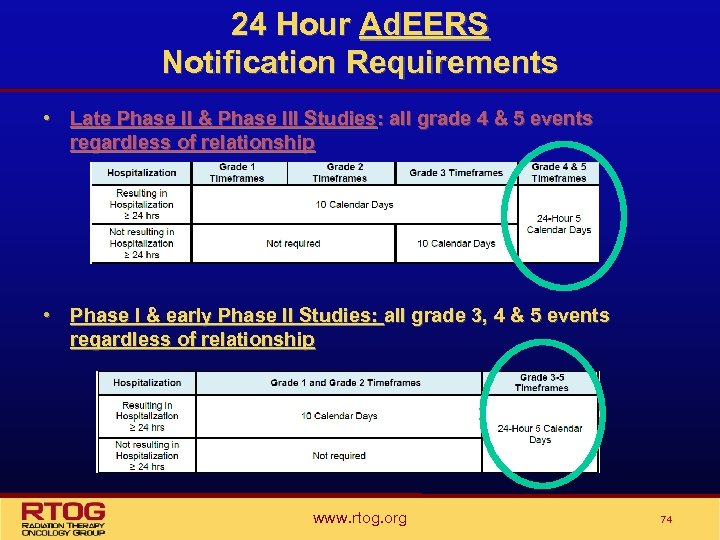 24 Hour Ad. EERS Notification Requirements • Late Phase II & Phase III Studies: all grade 4 & 5 events regardless of relationship • Phase I & early Phase II Studies: all grade 3, 4 & 5 events regardless of relationship www. rtog. org 74
24 Hour Ad. EERS Notification Requirements • Late Phase II & Phase III Studies: all grade 4 & 5 events regardless of relationship • Phase I & early Phase II Studies: all grade 3, 4 & 5 events regardless of relationship www. rtog. org 74
 24 Hour Ad. EERS Notification Requirements • The timeframe for the complete Ad. EERS Report associated with any 24 Hour Ad. EERS Notifications is 5 days. • All other SAEs that do not require the submission of a 24 Hour Ad. EERS Notification will be reported via Ad. EERS using the 10 day reporting timeline. www. rtog. org 75
24 Hour Ad. EERS Notification Requirements • The timeframe for the complete Ad. EERS Report associated with any 24 Hour Ad. EERS Notifications is 5 days. • All other SAEs that do not require the submission of a 24 Hour Ad. EERS Notification will be reported via Ad. EERS using the 10 day reporting timeline. www. rtog. org 75
 Foundation Study SAE Report Form (SAERF) 24 Hour Notification Requirements • Late Phase II & Phase III Studies: all grade 4 & 5 events regardless of relationship • Phase I & early Phase II Studies: all grade 3, 4 & 5 events regardless of relationship • The timeframe for the complete Report associated with any event requiring 24 Hour Notifications is 5 days. • All other SAEs that do not require the submission of a 24 Hour Notification will be reported using the 10 day reporting timeline. www. rtog. org 76
Foundation Study SAE Report Form (SAERF) 24 Hour Notification Requirements • Late Phase II & Phase III Studies: all grade 4 & 5 events regardless of relationship • Phase I & early Phase II Studies: all grade 3, 4 & 5 events regardless of relationship • The timeframe for the complete Report associated with any event requiring 24 Hour Notifications is 5 days. • All other SAEs that do not require the submission of a 24 Hour Notification will be reported using the 10 day reporting timeline. www. rtog. org 76
 Timelines • The time frame for submitting the required type of report will vary from FIVE to TEN CALENDAR DAYS Please refer to the protocol for specifics • The day the event is reported is counted as day one. • The reporting deadline is required to allow for RTOG and/or pharmaceutical sponsors to fulfill their external regulatory reporting obligations. www. rtog. org 77
Timelines • The time frame for submitting the required type of report will vary from FIVE to TEN CALENDAR DAYS Please refer to the protocol for specifics • The day the event is reported is counted as day one. • The reporting deadline is required to allow for RTOG and/or pharmaceutical sponsors to fulfill their external regulatory reporting obligations. www. rtog. org 77
 Proper Identification Proper identification of follow-up or amended reports, supporting source documentation and/or other information relevant to the SAE is imperative and must always include the RTOG study and case number • Please use the RTOG Case Number as the Patient ID in the Ad. EERS System * DO NOT ADD LEADING ZEROS • Please follow the specific instructions on the SAE Report Form for Foundation (3500 series) studies www. rtog. org 78
Proper Identification Proper identification of follow-up or amended reports, supporting source documentation and/or other information relevant to the SAE is imperative and must always include the RTOG study and case number • Please use the RTOG Case Number as the Patient ID in the Ad. EERS System * DO NOT ADD LEADING ZEROS • Please follow the specific instructions on the SAE Report Form for Foundation (3500 series) studies www. rtog. org 78
 What type of other documentation must be submitted with the an SAE Report? • Ad. EERS: Only documentation noted by the reporter as being provided in the additional information section of Ad. EERS need be submitted initially • Foundation (3500 series)SAE Report Form: None unless specifically requested • If the pharmaceutical company requires further documentation or clarification, RTOG will contact you via e-mail with the query/ request • The NCI may also contact you directly to request specific information or documentation www. rtog. org 79
What type of other documentation must be submitted with the an SAE Report? • Ad. EERS: Only documentation noted by the reporter as being provided in the additional information section of Ad. EERS need be submitted initially • Foundation (3500 series)SAE Report Form: None unless specifically requested • If the pharmaceutical company requires further documentation or clarification, RTOG will contact you via e-mail with the query/ request • The NCI may also contact you directly to request specific information or documentation www. rtog. org 79
 Dedicated SAE Fax Line 215 -717 -0990 All necessary supporting source documentation relating to the SAE noted as being supplied in the Additional Information Section of the Ad. EERS Report must be properly labeled with the RTOG Study and Case Number as well as the date and Ticket Number of the corresponding event and provided within the allotted timeframe or as soon as it becomes available. Information pertaining to Foundation (3500 series) Studies must also be properly labeled with the Study and Case Number as well as the date of the event www. rtog. org 80
Dedicated SAE Fax Line 215 -717 -0990 All necessary supporting source documentation relating to the SAE noted as being supplied in the Additional Information Section of the Ad. EERS Report must be properly labeled with the RTOG Study and Case Number as well as the date and Ticket Number of the corresponding event and provided within the allotted timeframe or as soon as it becomes available. Information pertaining to Foundation (3500 series) Studies must also be properly labeled with the Study and Case Number as well as the date of the event www. rtog. org 80
 May 27, 2008 www. rtog. org 81
May 27, 2008 www. rtog. org 81
 Guidance and/or Assistance with the Ad. EERS System Ø It may be helpful to view the Ad. EERS Tutorial at the following link: http: //ctep. info. nih. gov/reporting/Ad. EERS_CBT_v 3/welcome. html Ø Problems or issues with the Ad. EERS System for reporting AE/SAEs may also be referred to the following contacts: Ø For all medical and policy related issues, please contact the Ad. EERS Coordinators by email at Ad. EERSMD@tech-res. com
Guidance and/or Assistance with the Ad. EERS System Ø It may be helpful to view the Ad. EERS Tutorial at the following link: http: //ctep. info. nih. gov/reporting/Ad. EERS_CBT_v 3/welcome. html Ø Problems or issues with the Ad. EERS System for reporting AE/SAEs may also be referred to the following contacts: Ø For all medical and policy related issues, please contact the Ad. EERS Coordinators by email at Ad. EERSMD@tech-res. com
 Guidance and/or Assistance with the Foundation (3500 series) SAE Report Form Ø You can view both the Foundation SAE Report Form and Guidance for use/completion on the RTOG website www. rtog. org. Ø The Foundation SAE Report Form and Guidance are study specific and will be located under the study specific Regulatory Resources tab on the website Ø The RTOG SAE Coordinator: Sara Mc. Cartney[ smccartney@acr. org or 267 -9404 can also provide assistance with using the Foundation SAE Report Form www. rtog. org 83
Guidance and/or Assistance with the Foundation (3500 series) SAE Report Form Ø You can view both the Foundation SAE Report Form and Guidance for use/completion on the RTOG website www. rtog. org. Ø The Foundation SAE Report Form and Guidance are study specific and will be located under the study specific Regulatory Resources tab on the website Ø The RTOG SAE Coordinator: Sara Mc. Cartney[ smccartney@acr. org or 267 -9404 can also provide assistance with using the Foundation SAE Report Form www. rtog. org 83
 NCI/Pharmaceutical Initiated/External Safety Reports (ESR) • Events detailed in safety reports originating from the NCI or pharmaceutical company can reference a more global use of the drug • These safety reports are intended to inform treating physicians regarding events that have occurred in the interim that will be included in the next revised Investigator Brochure • No action other than providing these reports to your IRB is required unless indicated • Any questions regarding the overall significance of events reported in these safety reports should be directed to the NCI or the pharmaceutical company that initiated the report. www. rtog. org 84
NCI/Pharmaceutical Initiated/External Safety Reports (ESR) • Events detailed in safety reports originating from the NCI or pharmaceutical company can reference a more global use of the drug • These safety reports are intended to inform treating physicians regarding events that have occurred in the interim that will be included in the next revised Investigator Brochure • No action other than providing these reports to your IRB is required unless indicated • Any questions regarding the overall significance of events reported in these safety reports should be directed to the NCI or the pharmaceutical company that initiated the report. www. rtog. org 84
 External Safety Report (ESR) IRB Review • Local IRB policy can limit the review of ESRs – Auditors will require a copy of the written IRB policy or SOP • In the event the Local IRB elects not to review all ESRs – ESRs must still be reviewed by the PI or designee – The reviewer should indicate review by signing and dating the report – All ESRs must still be maintained as part of the study file for review during a site audit. • Sites who previously relied on the CIRB review of ESRs for Phase III studies – This review is now accomplished by way of review of the RTOG Data Monitoring Committee (DMC) reports for both Phase IIR & III studies www. rtog. org 85
External Safety Report (ESR) IRB Review • Local IRB policy can limit the review of ESRs – Auditors will require a copy of the written IRB policy or SOP • In the event the Local IRB elects not to review all ESRs – ESRs must still be reviewed by the PI or designee – The reviewer should indicate review by signing and dating the report – All ESRs must still be maintained as part of the study file for review during a site audit. • Sites who previously relied on the CIRB review of ESRs for Phase III studies – This review is now accomplished by way of review of the RTOG Data Monitoring Committee (DMC) reports for both Phase IIR & III studies www. rtog. org 85
 Finding Safety Reports on the Website • Safety Reports can be found under the study specific broadcasts on the RTOG website (Clinical Trials > Protocol Table > Study Details > Broadcasts) • Safety reports are sent via e-mail broadcast to individuals from participating institutions who are listed on the RTOG Broadcast List • If you would like to be added/removed from the RTOG broadcast list or wish to update your e-mail address, send a message to RTOG-User. Accounts@acr. org • Sites should not reply directly to any broadcast. Institutions can share a comment or ask a question by accessing the appropriate department at RTOG Headquarters on the RTOG web site at www. rtog. org then select the member information tab and click HQ Information & Telephone Listings. Sites can also call 1 -800 -227 -5463 ext. 4189, which provides prompts to reach department • Canadian research staff should be sure to request they be added specifically to the Canadian Broadcast List to assure the receipt of all broadcast information relevant to Canadian participation www. rtog. org 86
Finding Safety Reports on the Website • Safety Reports can be found under the study specific broadcasts on the RTOG website (Clinical Trials > Protocol Table > Study Details > Broadcasts) • Safety reports are sent via e-mail broadcast to individuals from participating institutions who are listed on the RTOG Broadcast List • If you would like to be added/removed from the RTOG broadcast list or wish to update your e-mail address, send a message to RTOG-User. Accounts@acr. org • Sites should not reply directly to any broadcast. Institutions can share a comment or ask a question by accessing the appropriate department at RTOG Headquarters on the RTOG web site at www. rtog. org then select the member information tab and click HQ Information & Telephone Listings. Sites can also call 1 -800 -227 -5463 ext. 4189, which provides prompts to reach department • Canadian research staff should be sure to request they be added specifically to the Canadian Broadcast List to assure the receipt of all broadcast information relevant to Canadian participation www. rtog. org 86
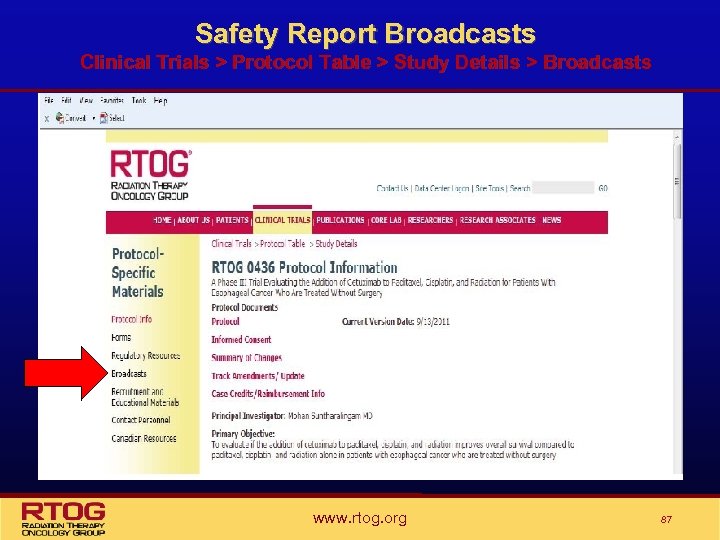 Safety Report Broadcasts Clinical Trials > Protocol Table > Study Details > Broadcasts www. rtog. org 87
Safety Report Broadcasts Clinical Trials > Protocol Table > Study Details > Broadcasts www. rtog. org 87
 On a Lighter Note… www. rtog. org 88
On a Lighter Note… www. rtog. org 88
 Regulatory Staff www. rtog. org 89
Regulatory Staff www. rtog. org 89
 www. rtog. org 90
www. rtog. org 90


