3d6d3b9dc6f71f5046cd50d6c87fa890.ppt
- Количество слайдов: 50

Jointly sponsored by Postgraduate Institute for Medicine and Clinical Care Options, LLC 6 th International Symposium on Translational Research in Oncology October 11 -14, 2007 Dublin, Ireland This program is supported by educational grants from

6 th International Symposium on Translational Research in Oncology Dennis J. Slamon, MD, Ph. D Chief, Division of Hematology/Oncology David Geffen School of Medicine at UCLA Los Angeles, California John Crown, MD, MPH Head, Medical Oncology Research St Vincent’s Hospital Elm Park Dublin, Ireland

6 th International Symposium on Translational Research in Oncology Program Overview Now in its sixth year, this annual symposium has a firmly established reputation as a premier meeting at which the world’s leading researchers gather to present and discuss new directions in oncology research with a focus on translating the most recent laboratory developments into improved clinical outcomes for cancer patients. Under the direction of Dennis J. Slamon, MD, Ph. D, and John Crown, MD, MPH, the program includes didactic presentations and interactive discussions. Faculty are carefully selected from among the researchers at the forefront of the translational work in the topic, whether from academia, government, or industry. The program encourages networking and interaction between the attendees and the renowned faculty members. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology About These Slides § Users are encouraged to include these slides in their own presentations, but we ask that content and attribution not be changed. Users are asked to honor this intent. § These slides may not be published or posted online or used for any other commercial purpose without written permission from Clinical Care Options. § We are grateful to Fairooz Kabbinavar, MD, the Chair of the Session, who aided in the preparation of this slideset. § We are also grateful to the speakers in the session who gave us permission to use a select group of their slides from the meeting to make this slideset possible: Kari Alitalo, MD; Fairooz Kabbinavar, MD; Jean Maral, MD, Ph. D; and Donald Mc. Donald, MD, Ph. D. Disclaimer The materials published on the Clinical Care Options Web site reflect the views of the authors, not those of Clinical Care Options, LLC, the CME providers, or the companies providing educational grants. The materials may discuss uses and dosages for therapeutic products that have not been approved by the United States Food and Drug Administration. A qualified healthcare professional should be consulted before using any therapeutic product discussed. Readers should verify all information and data before treating patients or using any therapies described in these materials. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Faculty and Staff Disclosures The faculty and CCO staff reported the following financial relationships or relationships to products or devices they or their spouse/life partner have with commercial interests related to the content of this CME activity. Fairooz Kabbinavar, MD, has no significant relationships to disclose. Edward King, MA; Deborah Janssen, MS; Andy Bowser; Jim Mortimer; Robert S. Mocharnuk, MD; and Gordon Kelley have no significant relationships to disclose. The following PIM clinical content reviewers, Linda Graham, RN; Jan Hixon, RN; and Trace Hutchison, Pharm. D, hereby state that they or their spouse/life partner do not have any financial relationships or relationships to products or devices with any commercial interests related to the content of this CME activity of any amount during the past 12 months. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Goal The goal of this activity is to provide an update on major topics in translational research to biomedical researchers and clinicians involved in the field. Target Audience This activity is intended for biomedical investigators in the area of cancer research from academia and industry, as well as medical, surgical, and radiation oncologists. Learning Objectives At the conclusion of this activity, participants should be able to: § Describe the role of vascular endothelial growth factor in tumor growth § Discuss combinations of antiangiogenic and chemotherapeutic agents used in the treatment of cancer § Identify novel antiangiogenic agents used in the treatment of cancer clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Physician Continuing Medical Education Release Date § Release date: January 7, 2008; valid for credit through January 6, 2009. Accreditation § This activity has been planned and implemented in accordance with the Essential Areas and Policies of the Accreditation Council for Continuing Medical Education (ACCME) through the joint sponsorship of Postgraduate Institute for Medicine and Clinical Care Options, LLC. Postgraduate Institute for Medicine is accredited by the ACCME to provide continuing medical education for physicians. Credit Designation § Postgraduate Institute for Medicine designates this educational activity for a maximum of 1. 0 AMA PRA Category 1 Credit(s)™. Physicians should only claim credit commensurate with the extent of their participation in the activity. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Instructions for Credit § There are no fees for participating and receiving credit for this activity. Participation in this self-study activity should be completed in approximately 1 hour. To successfully complete this activity and receive credit, participants must follow these steps during the period from January 7, 2008, through January 6, 2009: 1. Register online at http: //clinicaloptions. com. 2. Read the target audience, learning objectives, and faculty disclosures. 3. Study the educational activity online or printed out. 4. Submit answers to the posttest questions and evaluation questions online. § You must receive a test score of at least 70% and respond to all evaluation questions to receive a certificate. After submitting the evaluation, you may access your online certificate by selecting the certificate link on the posttest confirmation page. Records of all CME activities completed can be found on the “My CME” page. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Disclosure of Unlabeled Use Statement § This educational activity may contain discussion of published and/or investigational uses of agents that are not indicated by the FDA. Postgraduate Institute of Medicine (PIM) and Clinical Care Options, LLC do not recommend the use of any agent outside of the labeled indications. clinicaloptions. com/oncology

Session IV: Malignant Angiogenesis and Antiangiogenic Therapies Fairooz Kabbinavar, MD Associate Professor of Medicine Division of Hematology/Oncology Department of Medicine Jonsson Comprehensive Cancer Center University of California, Los Angeles, California
6 th International Symposium on Translational Research in Oncology Angiogenesis VEGF clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Biological Role of VEGF § Regulation of angiogenesis § Endothelial cell migration, proliferation, and survival § Vascular permeability § Regulation of hemodynamics § Modulation of immune responses clinicaloptions. com/oncology
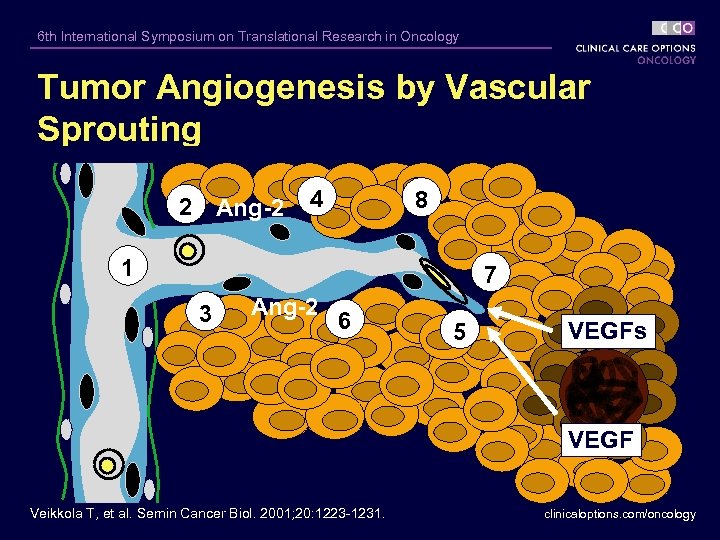
6 th International Symposium on Translational Research in Oncology Tumor Angiogenesis by Vascular Sprouting 2 Ang-2 4 8 1 7 3 Ang-2 6 5 VEGFs VEGF Veikkola T, et al. Semin Cancer Biol. 2001; 20: 1223 -1231. clinicaloptions. com/oncology
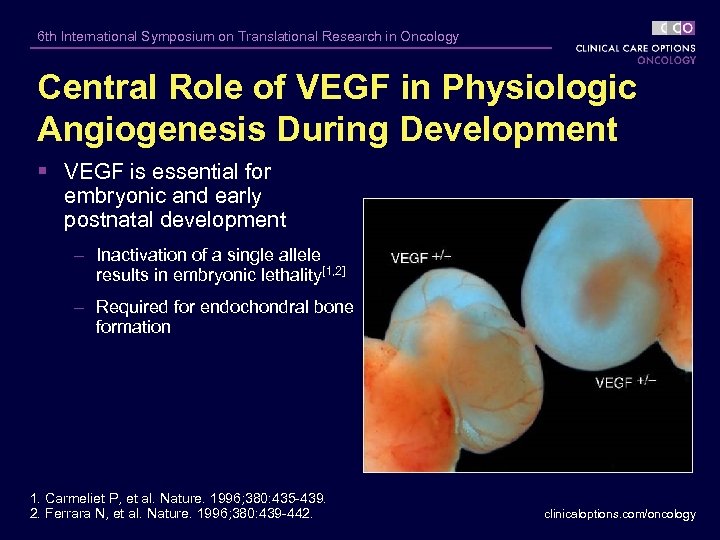
6 th International Symposium on Translational Research in Oncology Central Role of VEGF in Physiologic Angiogenesis During Development § VEGF is essential for embryonic and early postnatal development – Inactivation of a single allele results in embryonic lethality[1, 2] – Required for endochondral bone formation 1. Carmeliet P, et al. Nature. 1996; 380: 435 -439. 2. Ferrara N, et al. Nature. 1996; 380: 439 -442. clinicaloptions. com/oncology
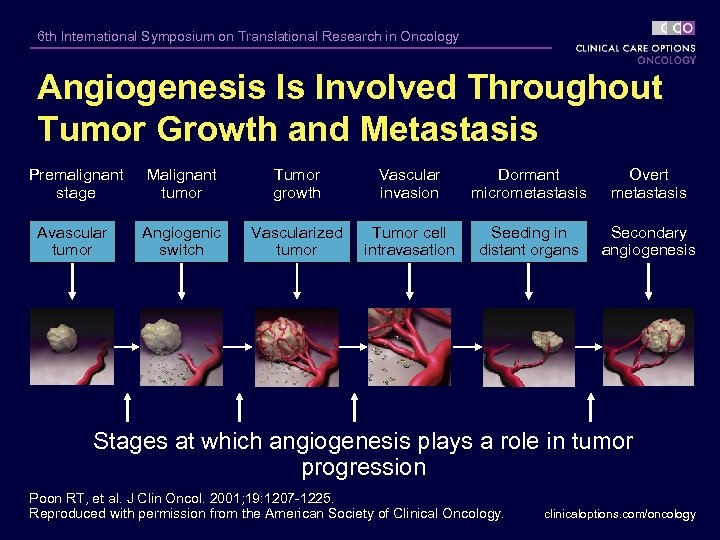
6 th International Symposium on Translational Research in Oncology Angiogenesis Is Involved Throughout Tumor Growth and Metastasis Premalignant stage Malignant tumor Tumor growth Vascular invasion Dormant micrometastasis Overt metastasis Avascular tumor Angiogenic switch Vascularized tumor Tumor cell intravasation Seeding in distant organs Secondary angiogenesis Stages at which angiogenesis plays a role in tumor progression Poon RT, et al. J Clin Oncol. 2001; 19: 1207 -1225. Reproduced with permission from the American Society of Clinical Oncology. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology VEGF Expression Is Induced by Tumor Hypoxia § VEGF m. RNA is rapidly induced in the presence of hypoxia in various cell types[1 -4] § VEGF m. RNA is highly expressed in tumor cells near areas of necrosis[4] § Hypoxia is a major inducer of VEGF gene expression[5] 1. Minchenko A, et al. Lab Invest. 1994; 71: 374 -379. 2. Shima DT, et al. Mol Med. 1995; 1: 182 -193. 3. Brogi E, et al. Circulation. 1994; 90: 649 -652. 4. Shweiki D, et al. Nature. 1992; 359: 843 -845. 5. Ferrara N, et al. Endocr Rev. 1997; 18: 4 -25. clinicaloptions. com/oncology
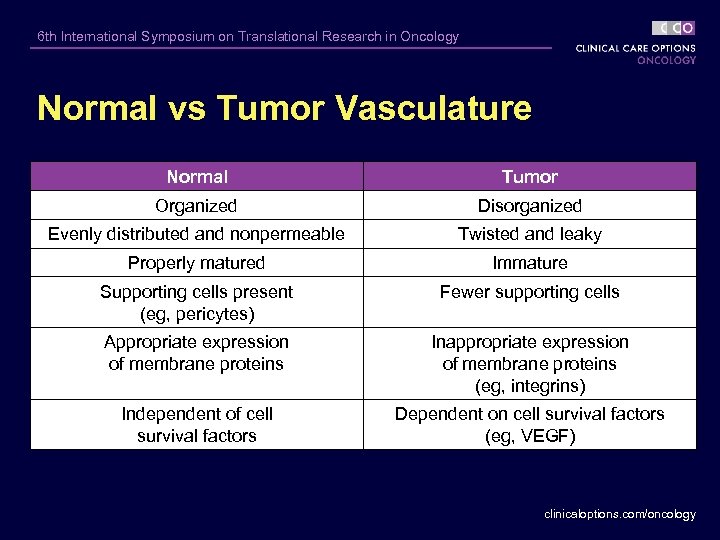
6 th International Symposium on Translational Research in Oncology Normal vs Tumor Vasculature Normal Tumor Organized Disorganized Evenly distributed and nonpermeable Twisted and leaky Properly matured Immature Supporting cells present (eg, pericytes) Fewer supporting cells Appropriate expression of membrane proteins Inappropriate expression of membrane proteins (eg, integrins) Independent of cell survival factors Dependent on cell survival factors (eg, VEGF) clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology VEGF Makes Tumor Blood Vessels Permeable and Leaky § Increased permeability induced by VEGF overexpression – Allows plasma proteins to escape to form a fibrin matrix on which activated endothelial cells migrate during angiogenesis[1] – Increases interstitial pressure, which hinders the diffusion of larger molecules (eg, chemotherapy or antitumor antibodies) into the tumor[2] 1. Pettersson A, et al. Lab Invest. 2000; 80: 99 -115. 2. Netti PA, et al. Proc Natl Acad Sci U S A. 1999; 96: 3137 -3142. clinicaloptions. com/oncology
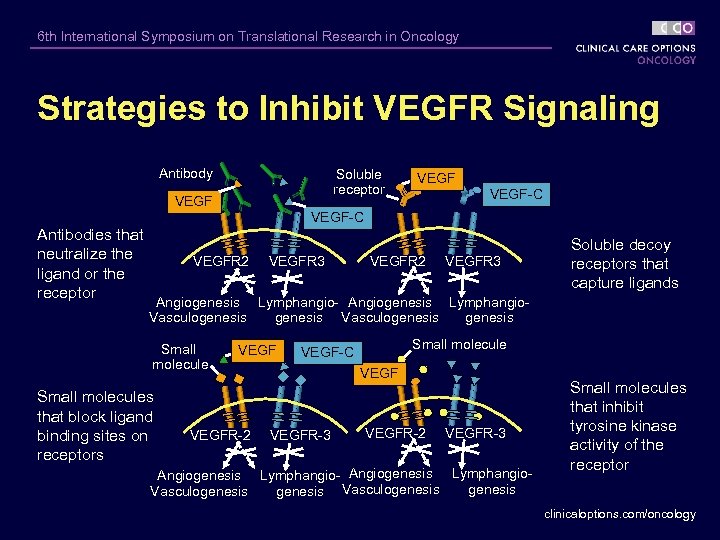
6 th International Symposium on Translational Research in Oncology Strategies to Inhibit VEGFR Signaling Antibody Soluble receptor VEGF-C Antibodies that neutralize the ligand or the receptor VEGFR 2 VEGFR 3 Soluble decoy receptors that capture ligands Angiogenesis Lymphangio- Angiogenesis Lymphangio. Vasculogenesis Small molecules that block ligand binding sites on receptors VEGFR-2 Small molecule VEGF-C VEGFR-3 VEGFR-2 VEGFR-3 Angiogenesis Lymphangio- Angiogenesis Lymphangiogenesis Vasculogenesis Small molecules that inhibit tyrosine kinase activity of the receptor clinicaloptions. com/oncology
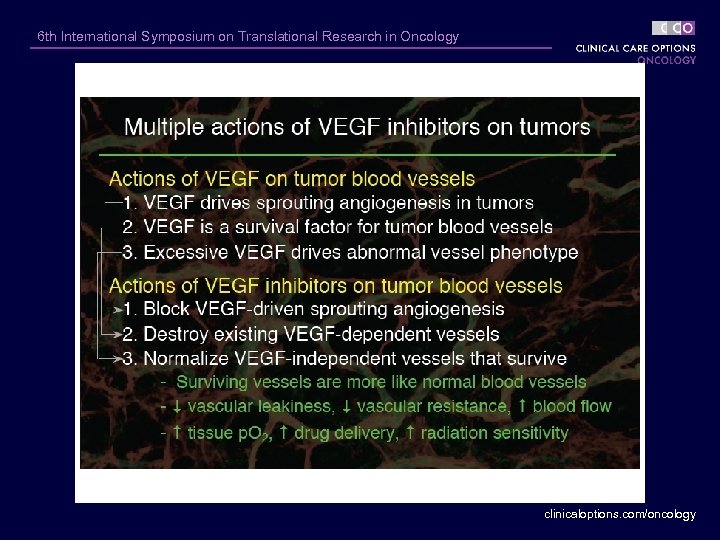
6 th International Symposium on Translational Research in Oncology clinicaloptions. com/oncology
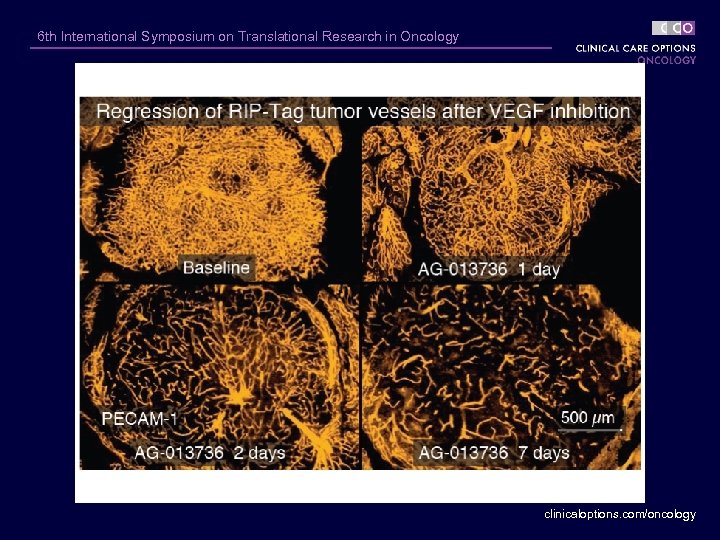
6 th International Symposium on Translational Research in Oncology clinicaloptions. com/oncology
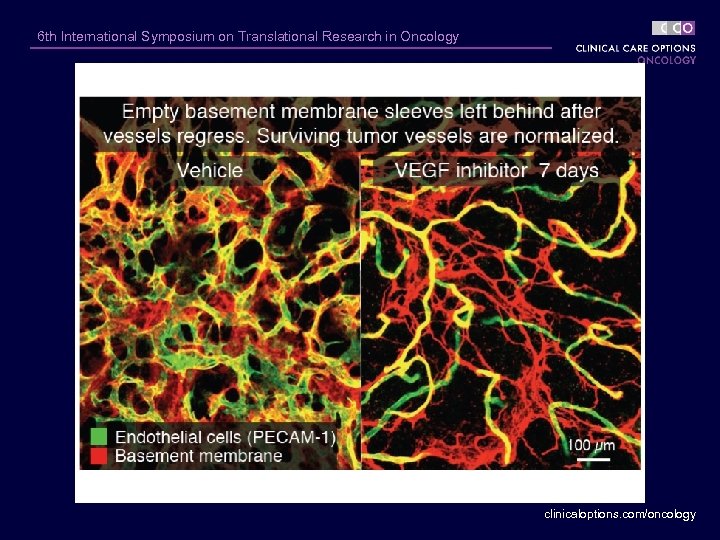
6 th International Symposium on Translational Research in Oncology clinicaloptions. com/oncology
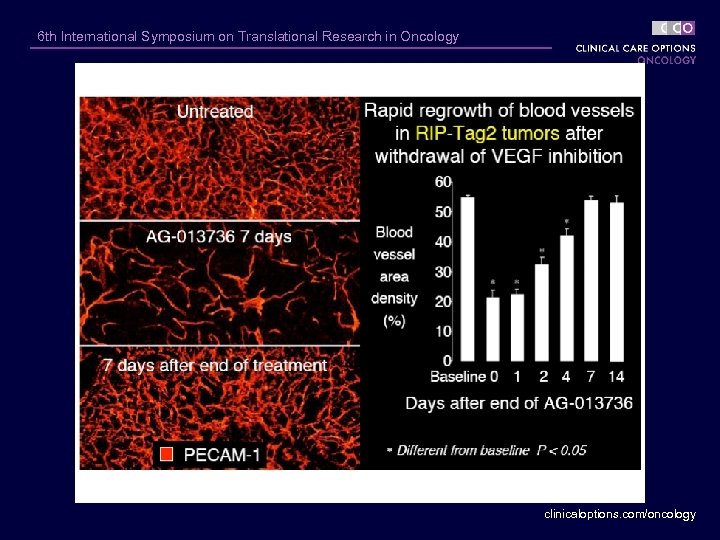
6 th International Symposium on Translational Research in Oncology clinicaloptions. com/oncology

VEGF Trap

6 th International Symposium on Translational Research in Oncology Traps Represent a New Therapeutic Class § Traps are analogous to soluble receptors like etanercept but incorporate 2 different receptor components § Discoveries of basic receptor signaling mechanisms during the 1990 s led to this new class of blockers, which target well-validated targets such as cytokines, interleukins, and growth factors Economides AN, et al. Nat Med. 2002; 9: 47 -52. Holash J, et al. Proc Natl Acad Sci U S A. 2002; 99: 11393 -11398. clinicaloptions. com/oncology
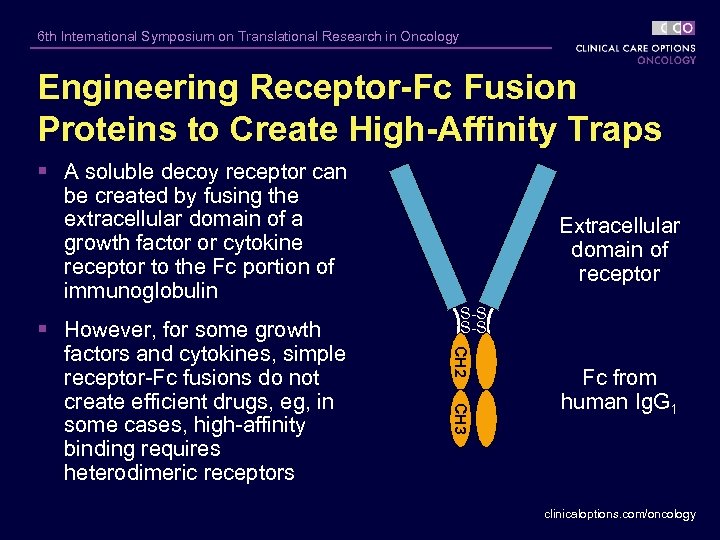
6 th International Symposium on Translational Research in Oncology Engineering Receptor-Fc Fusion Proteins to Create High-Affinity Traps § A soluble decoy receptor can be created by fusing the extracellular domain of a growth factor or cytokine receptor to the Fc portion of immunoglobulin S-S CH 2 CH 3 § However, for some growth factors and cytokines, simple receptor-Fc fusions do not create efficient drugs, eg, in some cases, high-affinity binding requires heterodimeric receptors Extracellular domain of receptor Fc from human Ig. G 1 clinicaloptions. com/oncology
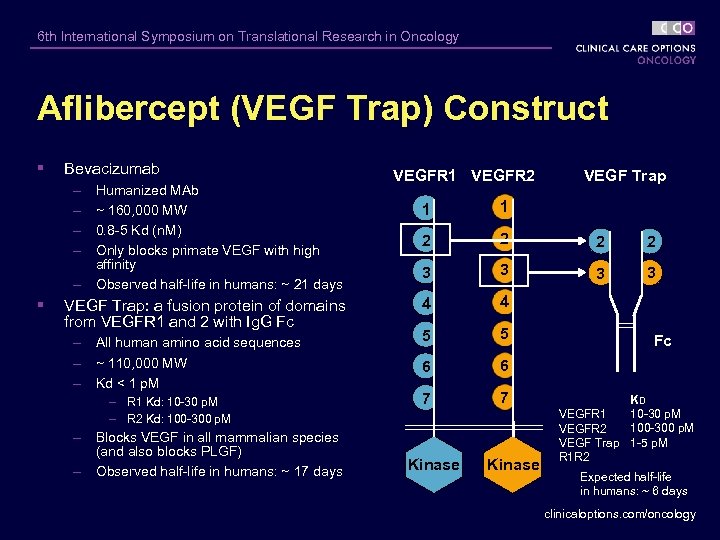
6 th International Symposium on Translational Research in Oncology Aflibercept (VEGF Trap) Construct § Bevacizumab – – – § Humanized MAb ~ 160, 000 MW 0. 8 -5 Kd (n. M) Only blocks primate VEGF with high affinity Observed half-life in humans: ~ 21 days VEGF Trap: a fusion protein of domains from VEGFR 1 and 2 with Ig. G Fc – All human amino acid sequences – ~ 110, 000 MW – Kd < 1 p. M – R 1 Kd: 10 -30 p. M – R 2 Kd: 100 -300 p. M – Blocks VEGF in all mammalian species (and also blocks PLGF) – Observed half-life in humans: ~ 17 days VEGFR 1 VEGFR 2 VEGF Trap 1 1 2 2 3 3 4 4 5 5 6 6 7 7 Kinase Fc KD 10 -30 p. M VEGFR 1 100 -300 p. M VEGFR 2 VEGF Trap 1 -5 p. M R 1 R 2 Expected half-life in humans: ~ 6 days clinicaloptions. com/oncology
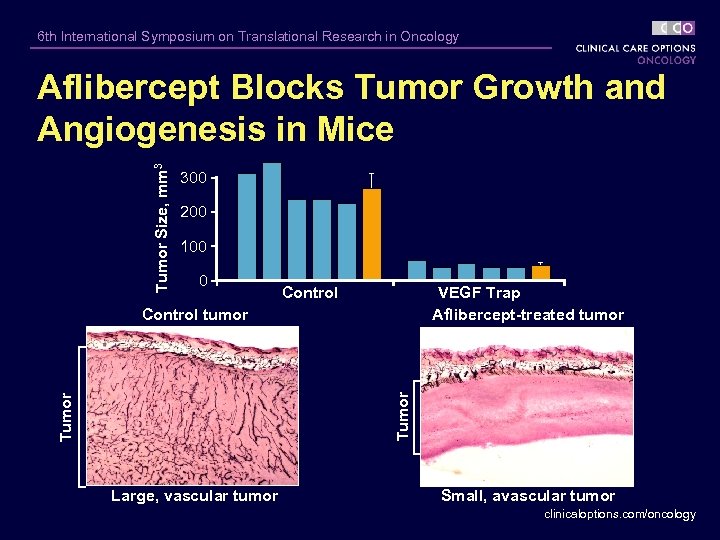
6 th International Symposium on Translational Research in Oncology Tumor Size, mm 3 Aflibercept Blocks Tumor Growth and Angiogenesis in Mice 300 200 100 0 Control VEGF Trap Aflibercept-treated tumor Tumor Control tumor Large, vascular tumor Small, avascular tumor clinicaloptions. com/oncology
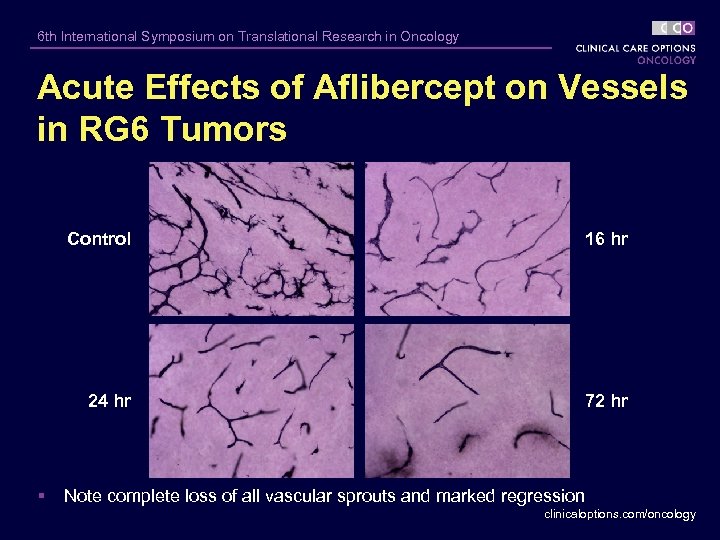
6 th International Symposium on Translational Research in Oncology Acute Effects of Aflibercept on Vessels in RG 6 Tumors Control 24 hr § 16 hr 72 hr Note complete loss of all vascular sprouts and marked regression clinicaloptions. com/oncology
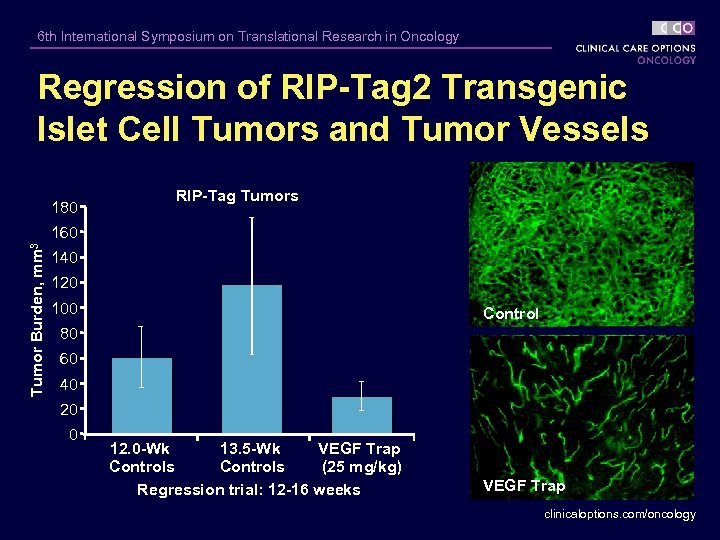
6 th International Symposium on Translational Research in Oncology Regression of RIP-Tag 2 Transgenic Islet Cell Tumors and Tumor Vessels 180 RIP-Tag Tumors Tumor Burden, mm 3 160 140 120 100 Control 80 60 40 20 0 12. 0 -Wk 13. 5 -Wk VEGF Trap Controls (25 mg/kg) Regression trial: 12 -16 weeks VEGF Trap clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Single-Agent Aflibercept: Phase I Studies § N = 53 § Dosing: aflibercept ≤ 7 mg/kg (sc/iv) § DLT § ≤ 2 patients per dose level experienced DLT up to 5 mg/kg – DLT (including hypertension) has been generally manageable and reversible § Safety profile – Hypertension – Dysphonia – Epistaxis – Proteinuria – Headache – Musculoskeletal discomfort – Fatigue clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Single-Agent Aflibercept: Phase I Studies (cont’d) § Immunogenicity: anti-VEGF Trap antibodies have not been detected § Adverse effects – Hypertension and proteinuria are class effects of VEGF/VEGFR inhibitors – Majority of reported adverse events were mild to moderate in severity – Adverse events are consistent with VEGF blockade clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Aflibercept in Cancer § Aflibercept is a novel and potent anti-VEGF angiogenesis inhibitor with unique features that may yield antiangiogenic and antitumor activity – Neutralizes all VEGF-A isoforms and Pl. GF – Binds VEGF-A 100 - to 1000 -fold more tightly than antibodies § Aflibercept well tolerated § Aflibercept has demonstrated activity as a single-agent in early clinical trials § Robust development program is progressing rapidly – Phase III studies currently under way clinicaloptions. com/oncology

Integrating Antiangiogenesis Therapy in the Management of Metastatic Cancer

6 th International Symposium on Translational Research in Oncology Bevacizumab + IFL for Metastatic CRC: Phase III AVF 2107 g Trial Hurwitz H, et al. N Engl J Med. 2004; 350: 2335 -2342. clinicaloptions. com/oncology
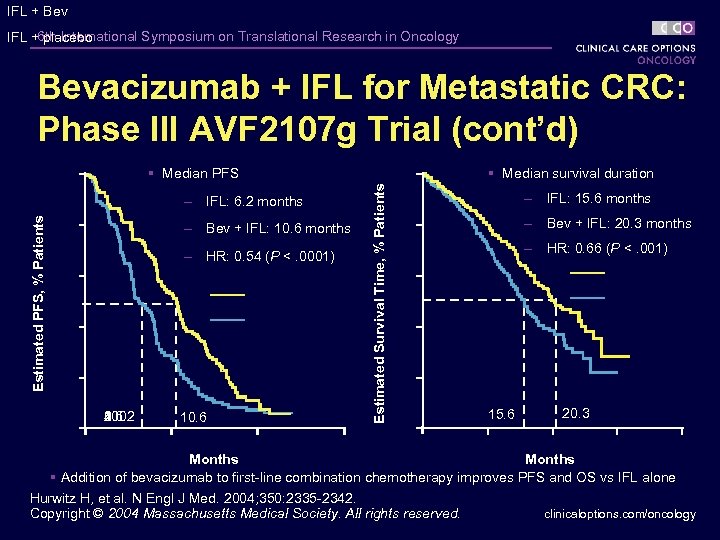
IFL + Bev IFL +6 th International Symposium on Translational Research in Oncology placebo Bevacizumab + IFL for Metastatic CRC: Phase III AVF 2107 g Trial (cont’d) Estimated PFS, % Patients – IFL: 6. 2 months – Bev + IFL: 10. 6 months – HR: 0. 54 (P <. 0001) 00 30 20 1 100 80 60 40 2 6. 2 10. 6 § Median survival duration Estimated Survival Time, % Patients § Median PFS – IFL: 15. 6 months – Bev + IFL: 20. 3 months – HR: 0. 66 (P <. 001) 15. 6 20. 3 Months § Addition of bevacizumab to first-line combination chemotherapy improves PFS and OS vs IFL alone Hurwitz H, et al. N Engl J Med. 2004; 350: 2335 -2342. clinicaloptions. com/oncology Copyright © 2004 Massachusetts Medical Society. All rights reserved.
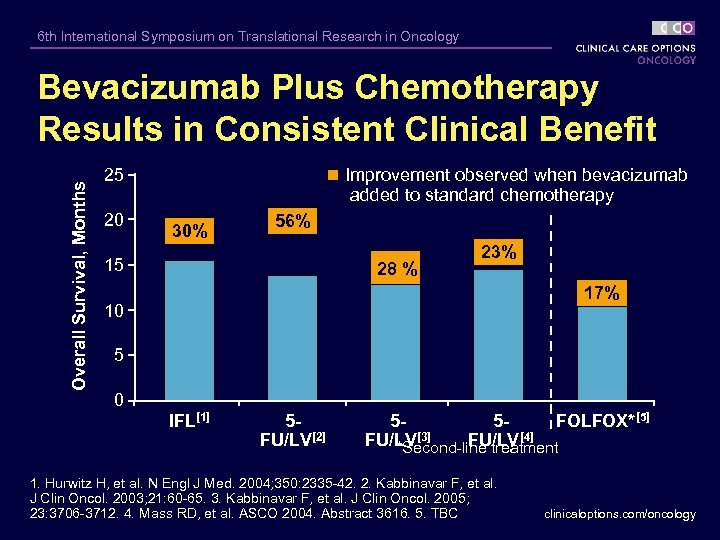
6 th International Symposium on Translational Research in Oncology Overall Survival, Months Bevacizumab Plus Chemotherapy Results in Consistent Clinical Benefit 25 20 Improvement observed when bevacizumab added to standard chemotherapy 30% 56% 15 28 % 23% 17% 10 5 0 IFL[1] 5 FU/LV[2] 55 FOLFOX*[5] FU/LV[3] FU/LV[4] *Second-line treatment 1. Hurwitz H, et al. N Engl J Med. 2004; 350: 2335 -42. 2. Kabbinavar F, et al. J Clin Oncol. 2003; 21: 60 -65. 3. Kabbinavar F, et al. J Clin Oncol. 2005; 23: 3706 -3712. 4. Mass RD, et al. ASCO 2004. Abstract 3616. 5. TBC clinicaloptions. com/oncology
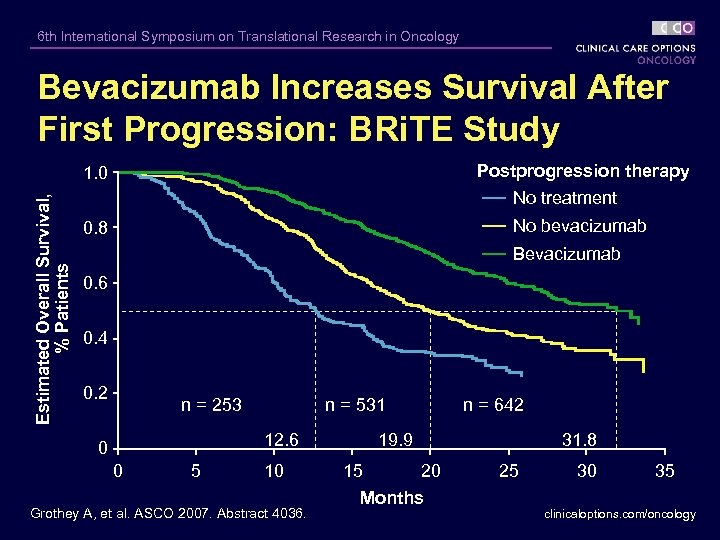
6 th International Symposium on Translational Research in Oncology Bevacizumab Increases Survival After First Progression: BRi. TE Study Postprogression therapy Estimated Overall Survival, % Patients 1. 0 No treatment No bevacizumab 0. 8 Bevacizumab 0. 6 0. 4 0. 2 n = 253 n = 531 12. 6 0 0 5 10 Grothey A, et al. ASCO 2007. Abstract 4036. n = 642 19. 9 15 31. 8 20 Months 25 30 35 clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Bevacizumab in Advanced NSCLC: Clinical Trial Evidence Trial Phase Patients Treatment Arms* PC 6 AVF 0757 g[1] II 98 Bevacizumab 7. 5 mg/kg Q 3 W + PC 6 Bevacizumab 15 mg/kg Q 3 W + PC 6 E 4599[2] III 878 PC 6 (no crossover allowed) Bevacizumab 15 mg/kg Q 3 W + PC 6 *Bevacizumab was given until disease progression or unacceptable toxicity. After disease progression, patients in the control arm of AVF 0757 g had the option to receive single-agent bevacizumab 15 mg/kg Q 3 W. 1. Johnson DH, et al. J Clin Oncol. 2004; 22: 2184 -2191. 2. Avastin [package insert]. South San Francisco, Calif: Genentech; 2006. clinicaloptions. com/oncology
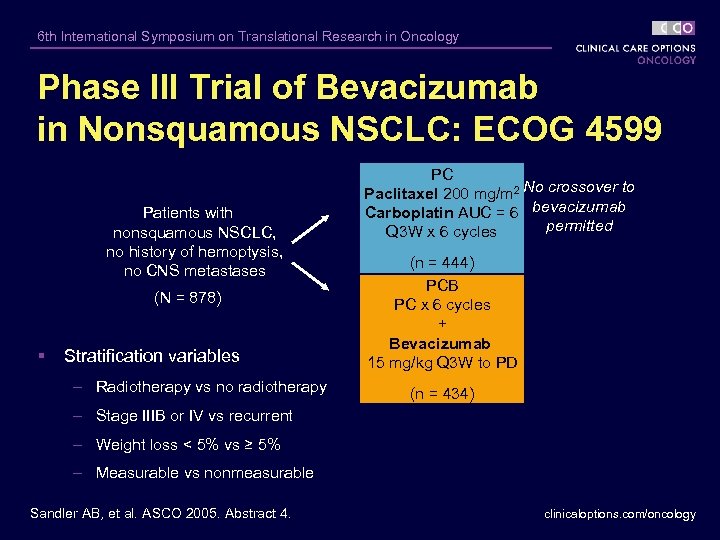
6 th International Symposium on Translational Research in Oncology Phase III Trial of Bevacizumab in Nonsquamous NSCLC: ECOG 4599 Patients with nonsquamous NSCLC, no history of hemoptysis, no CNS metastases (N = 878) § Stratification variables – Radiotherapy vs no radiotherapy PC Paclitaxel 200 mg/m 2 No crossover to Carboplatin AUC = 6 bevacizumab permitted Q 3 W x 6 cycles (n = 444) PCB PC x 6 cycles + Bevacizumab 15 mg/kg Q 3 W to PD (n = 434) – Stage IIIB or IV vs recurrent – Weight loss < 5% vs ≥ 5% – Measurable vs nonmeasurable Sandler AB, et al. ASCO 2005. Abstract 4. clinicaloptions. com/oncology
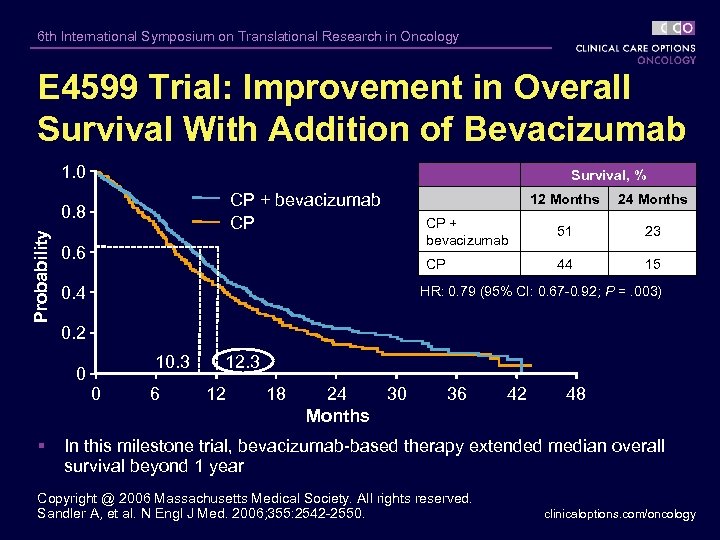
6 th International Symposium on Translational Research in Oncology E 4599 Trial: Improvement in Overall Survival With Addition of Bevacizumab 1. 0 Survival, % CP + bevacizumab CP Probability 0. 6 0. 4 CP + bevacizumab 51 23 44 15 HR: 0. 79 (95% CI: 0. 67 -0. 92; P =. 003) 0. 2 0 § 24 Months CP 0. 8 12 Months 10. 3 0 6 12. 3 12 18 24 30 Months 36 42 48 In this milestone trial, bevacizumab-based therapy extended median overall survival beyond 1 year Copyright @ 2006 Massachusetts Medical Society. All rights reserved. Sandler A, et al. N Engl J Med. 2006; 355: 2542 -2550. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Motzer RJ, et al. N Engl J Med. 2007; 356: 115 -124. Escudier B, et al. N Engl J Med. 2007; 356: 125 -134. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Sunitinib vs Interferon alfa for m. RCC: Phase III Study Patients with unresectable or m. RCC, no previous systemic treatment, and ECOG PS 0/1 (N = 750) Sunitinib, 50 mg QD 4 weeks on, 2 weeks off (n = 375) Interferon alfa 3 MU TIW, 6 MU TIW, 9 MU TIW (n = 375) § Primary endpoint: PFS § Secondary endpoints: RR, OS, safety, patient reported outcomes § Demographics – MSKCC patients with good or intermediate risk – Clear-cell carcinoma Motzer RJ, et al. N Engl J Med. 2007; 356: 115 -124. clinicaloptions. com/oncology
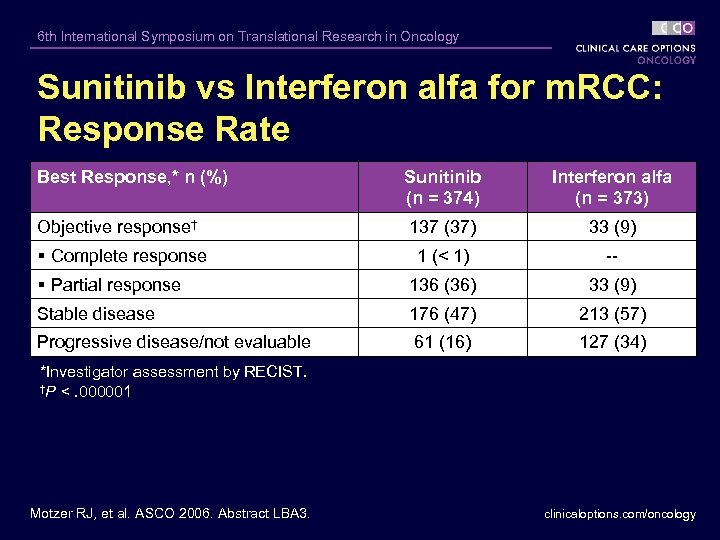
6 th International Symposium on Translational Research in Oncology Sunitinib vs Interferon alfa for m. RCC: Response Rate Best Response, * n (%) Sunitinib (n = 374) Interferon alfa (n = 373) Objective response† 137 (37) 33 (9) § Complete response 1 (< 1) -- § Partial response 136 (36) 33 (9) Stable disease 176 (47) 213 (57) Progressive disease/not evaluable 61 (16) 127 (34) *Investigator assessment by RECIST. †P <. 000001 Motzer RJ, et al. ASCO 2006. Abstract LBA 3. clinicaloptions. com/oncology
![6 th International Symposium on Translational Research in Oncology Sorafenib § Small-molecule receptor TKI[1] 6 th International Symposium on Translational Research in Oncology Sorafenib § Small-molecule receptor TKI[1]](https://present5.com/presentation/3d6d3b9dc6f71f5046cd50d6c87fa890/image-45.jpg)
6 th International Symposium on Translational Research in Oncology Sorafenib § Small-molecule receptor TKI[1] § Inhibits VEGFR 2, VEGFR 3, FLT-3, PDGFR, c-KIT, Raf kinases[1] § Dosing: 400 mg BID continuous (1 hour AC or 2 hours PC)[2] § FDA approved December 20, 2005, for advanced RCC[3] CF 3 CI O O NH O N H N CH 3 1. Wilhelm SM, et al. Cancer Res. 2004; 64: 7099 -7109. 2. Nexavar [package insert]. West Haven, Conn: Bayer Pharmaceutical Corporation; and Emeryville, Calif: Onyx Pharmaceuticals; 2005. 3. FDA. Available at: http: //www. fda. gov/bbs/topics/NEWS/2005/NEW 01282. html. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Sorafenib in Advanced Clear-Cell RCC TARGET: Phase III Study Design Patients with unresectable and/or metastatic RCC, who failed 1 previous systemic received therapy in last 8 months, and are ECOG PS 0/1 Sorafenib 400 mg BID (n = 451) (N = 903*) Placebo (n = 452) § Primary endpoints: OS and PFS § Secondary endpoints: ORR, safety, HR Qo. L § > 200 placebo patients unblinded and crossed over Demographics – MSKCC patients with good- or intermediate-risk RCC – Clear-cell carcinoma Escudier B, et al. N Engl J Med. 2007; 356: 125 -134. clinicaloptions. com/oncology
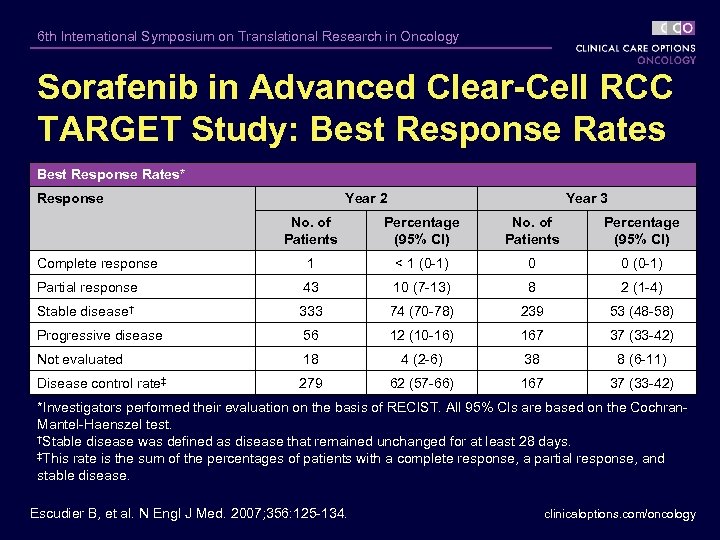
6 th International Symposium on Translational Research in Oncology Sorafenib in Advanced Clear-Cell RCC TARGET Study: Best Response Rates* Response Year 2 Year 3 No. of Patients Percentage (95% Cl) Complete response 1 < 1 (0 -1) 0 0 (0 -1) Partial response 43 10 (7 -13) 8 2 (1 -4) Stable disease† 333 74 (70 -78) 239 53 (48 -58) Progressive disease 56 12 (10 -16) 167 37 (33 -42) Not evaluated 18 4 (2 -6) 38 8 (6 -11) Disease control rate‡ 279 62 (57 -66) 167 37 (33 -42) *Investigators performed their evaluation on the basis of RECIST. All 95% Cls are based on the Cochran. Mantel-Haenszel test. †Stable disease was defined as disease that remained unchanged for at least 28 days. ‡This rate is the sum of the percentages of patients with a complete response, a partial response, and stable disease. Escudier B, et al. N Engl J Med. 2007; 356: 125 -134. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Sorafenib in Advanced Hepatocellular Carcinoma: Phase III Trial § N = 321 (143: 178) § Median survival: 10. 7 vs 7. 9 months § Overall survival: 44% improvement § Time to progresssion: 5. 5 vs 2. 8 months § Major adverse effect: diarrhea – Sorafenib: 11% – Placebo: 2% Llovet J, et al. ASCO 2007. Abstract LBA 1. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Conclusions § Inhibiting VEGF appears to be a valid strategy for treating multiple tumor types § Preclinical evidence supports single-agent efficacy and synergistic activity with chemotherapies in different tumor types § Bevacizumab, sunitinib, and sorafenib represent the validation of Folkman’s hypothesis, [1] and their use has demonstrated a survival benefit in the treatment of cancer § The large body of data from many clinical trials will confirm the value of antiangiogenic therapies in different tumor types 1. Folkman J. N Engl J Med. 1971; 285: 1182 -1186. clinicaloptions. com/oncology

6 th International Symposium on Translational Research in Oncology Now, Take the Test To receive CME credit for this activity, take the posttest: clinicaloptions. com/translational More CME-Certified Hematology/Oncology Available Online § Medical Meeting Coverage: Key data plus Expert Analysis panel discussions exploring clinical implications § Treatment Updates: Comprehensive programs covering the most important new concepts § Interactive Cases: Test your ability to manage patients clinicaloptions. com/oncology
3d6d3b9dc6f71f5046cd50d6c87fa890.ppt