История кафедры биохимии на английском!.ppt
- Количество слайдов: 26

История кафедры биохимии 1931 -1960 – д. б. н. проф. Ильин Какуев Б. И. 1960 -1984 – д. м. н. , проф. Верболович П. А 1984 -2012 д. м. н. , проф. Плешкова С. М. 2012 по настоящее время д. б. н. , проф. Шарипов К. О.

Расположение кафедры биохимии u 1931 -1972 г – ул. Пушкина/Карла. Маркса (деревянное здание около Зеленого Базара) u 1972 -2014 – ул. Комсомольская/просп. Сейфуллина u С 2014 г. В корпусе на ул. Богенбай Батыра (3 этаж, бывшая кафедра информатики и физики



Первые годы кафедры биохимии



Кафедра наших дней

С 2012 года – 4 заведующий кафедрой биохимии, д. б. н. профессор Шарипов К. О.

Зачем студенту-медику биохимия? u 1) Ломоносов: «Без должного познания химии не может быть совершенного врача» u 2) Пререквизит фармакологии, пат. физиологии, пат. анатомии u 3) Клиническая биохимия u 4) ………. . (вопрос студентам)

Conjugated proteins The plan: 1. Glycoproteins - structure, representatives, importance 2. Chromoproteins - structure, representatives, importance

Conjugated proteins consist of apoprotein and prosthetic group (from greek word «prostheto» bound)

Apoprotein is a protein part; prosthetic group is nonprotein part of molecule

Conjugated proteins are divided into 6 main classes: phosphoproteins, chromoproteins, glycoproteins, lipoproteins, metalloproteins, nucleoproteins
Phosphoproteins – structure, chemical properties, representatives and importance Phosphproteins consist of apoprotein and residues of phosphoric acid, bound to hydroxygroups of hydroxyamino acids (Ser, Tyr, Hyp)
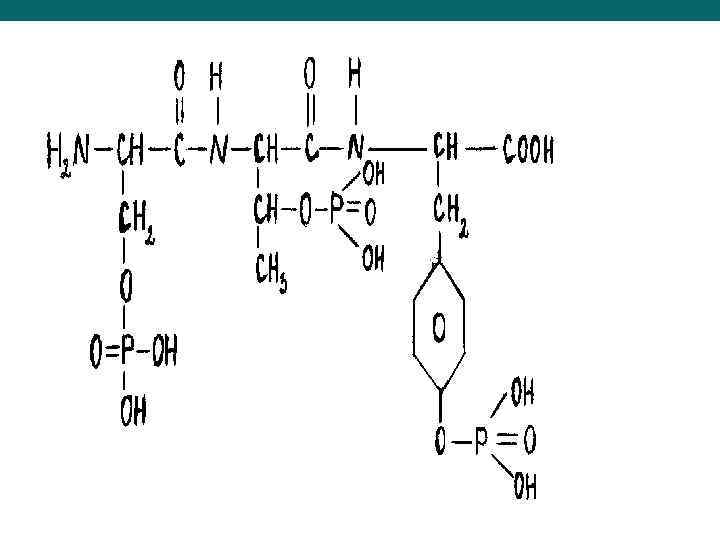

u. Phosphoproteins are full value proteins with a big molecular weight. They aren’t coagulated by the heating (i. e. thermo stabile), water insoluble, but well soluble in diluted solutions of alkalies and salts. They have an acidic character (IEP~ 4, 7) due to residues of phosphoric acid, therefore they are well precipitated by acids

The importance of Ph. Pr: 1) They serve as structural material for bones and teeth (sources of phosphorus) 2) Play an important role in growth (sources of essential amino acids)

Representatives: 1) Casein is Ph. Pr of milk (~ 1% of phosphorus); 2) Vitellin, Phosvitin are Ph. Pr of egg yolk (10% of phosphorus); 3) Ihtullin is Ph. Pr of caviar (fish eggs) (more than 10% of Phosphorus)

. Metalloproteins – structure, representatives, role MP are conjugated proteins, containing free metals as prosthetical group Representatives 1) Ferritin contains 20% of 3 valency iron and carries out the role of iron storage in the body. It is stored in liver and splen

2) Transferrin contains 0, 13% of iron and carries out the role of transport of iron in the body. Each molecule binds 2 ions of 2 valency iron

3) Ceruloplasmin transports copper in the body and possesses enzymic activity (ferrooxydase) 4) Iron-sulfur proteins occurs in the complex with flavinproteins and cytochrome b in the respiratory chain; 5) metaloenzymes, such as carboxypeptidase (Zn), carbonic anhydrase (Zn), superoxide dismutase (Cu, Zn) etc

Lipoproteins – the concept of structure and role LP consist of apoproteins and lipids (phospholipids, cholesterol and its esters, TAG). There are free and membrane-bound LP. Free LP (LP of blood serum) are composed as micelles, i. e. they have hydrophobic nucleus, containing cholesterol and TAG. Nucleus is surrounded by hydrophilic cover from proteins and phospholipids. There are LDL, HDL, VLDL, which are distinguished by various content of proteins and lipids. u Membrane-bound LP (structural LP): inside proteins, outside – lipids u

Feed back questions u 1) representatives of phosphoproteins u 2) representatives of metalloproteins u 3) what kinds of lipoproteins are called as atherogenic, and what are antiatherogenic?

References Main literature: u M. Edwin – Biochemistry in clinical correlation, 2001 u Harper’s Biochemistry – R. K. Murray, D. K. Granner, P. A. Mayes, V. W. Rodwell – Connecticut, 2012 u U. Satyanarayana – Biochemistry – Books and Allied (P/LTD, Kolkata (India), 2012 u M. N. Chatterjea, R. Shinde – Textbook of Medical Biochemistry – Jaypee Brothers, New Delhi (India), 2010 Additional literature: u Ozols J: Amino acid analysis. Methods Enzymol. 1990; 182: 587 u Biemann K: Mass spectrometry of peptides and proteins. Annu Rev Biochem 1992; 61: 977 u Benner S. A. et al: Predicting the conformation of proteins from sequences: Progress and future progress. Adv Enzyme Regul 1994; 34: 269

u. Thank you for your attention! u. See you!
История кафедры биохимии на английском!.ppt