2b5dba95a19decce1c8a6747741a5f4d.ppt
- Количество слайдов: 34

Investing in Health Systems of the Future Budapest, Hungary 10 – 12 May 2011

European EHR Market Analysis The Euro. Rec Institute Jos Devlies, M. D.

3 Topics • The EHR-QTN project ▫ Purpose of the project ▫ Partners ▫ Main results • The European EHR Market Analysis ▫ ▫ ▫ Methodology The EHR system suppliers overview The EHR stakeholders overview Cross – domain suppliers Cross – border suppliers European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

4 The EHR-QTN Project European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

5 EHR-QTN • Thematic Network project : 238912 • “The main objective of the Thematic Network is to better prepare the health community across Europe for systematic and comparable quality assurance and certification of e-Health products, more specifically of Electronic Healthcare Record systems. ” • Three years (February 2009 – January 2012) • Budget: 789. 000€ • 28 partners in 25 countries European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011
6 Partners European Institute for Health Records Pro. Rec Austria Pro. Rec Belgium RAMIT Pro. Rec Bulgaria Hrvatsko drustvo za medicinsku informatiku Cypriotic Society for Medical Informatics České národní fórum pro e. Health, o. s MEDIQ A/S Eesti E-tervise Sihtasutus Centro Poliklinika Vilnius – Lithuania Pro. Rec Germany Foundation for Research and Technology – Hellas National Institute for Strategic Health Research - ESKI Pro. Rec Ireland - Irish Centre for Health Telematics Ltd Pro. Rec Italy CRP Henri Tudor - SANTEC Stichting Pro. Rec Nederland KITH AS Marshal's Office of the Lodz Region Administraçao Central do Sistema de Saúde, I. P. Pro. Rec Romania - Romanian Association for Electronic Registration of Medical Data Pro. Rec Serbia - Srpsko udruženje za elektronski zdravstveni karton Pro. Rec Slovakia Ustanova - Pro. Rec Slovenia Hospital Universitario de Fuenlabrada Instituto de Salud Carlos III Pro. Rec United Kingdom European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

7 Main Results of the EHR-QTN • Awareness (72 National Workshops) • Validation of the Euro. Rec Descriptive Functional Statements (+1. 700) and the Certification Procedures • Translation of the Statements in 19 languages • Euro. Rec Seal Level 1 and Level 2 definition • Quality labelling of EHR Market Products • EHR Market Overview • Roadmap towards Sustainable Pan-European Certification of EHR Systems European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

8 The European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

9 European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

10 Methodology • Data collection per country ▫ Template ▫ Web Questionnaire ▫ Completed by the EHR-QTN partner • Where feasible: strict inclusion criteria: ▫ Suppliers based on market share. Goal: > 90% of the market ▫ Other stakeholders: “decisional impact” Authorities User groups (mostly on demanding side but also possible obstructers) European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

11 Origin of the information • Not so easy to collect • Either not available as such or considered as “classified” • Possibilities: ▫ Official list of labelled applications (a few countries for a limited number of domains of application) ▫ ▫ Expertise of the partners Web searches (e. g. professional organisations) Health Ministries Health IT suppliers organisations European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

12 Result • 24 National Market Overviews • Two Deliverables ▫ Del. D 3. 1 Part I: Suppliers and Supplier Organisations: 1. 019 supplier / product ID ▫ Del. D 3. 2 Part II: Authorities and important stakeholders: 663 addresses European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

13 Suppliers and their products European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

14 EHR Market Overview: Suppliers • 849 • 25 • 101 “Commercial” EHR systems “in house” large HIS with EHR health IT service providers (health telematics, privay enhancing etc…) • 44 • Total: e. Health / Health IT representative organisations 1. 019 addresses, contact persons etc… European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011
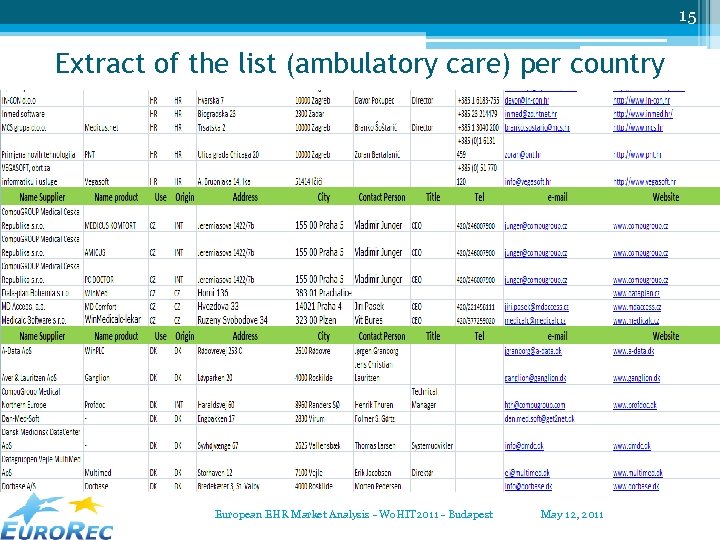
15 Extract of the list (ambulatory care) per country European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

16 Extract of Hospital Systems (Alphabetically) European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011
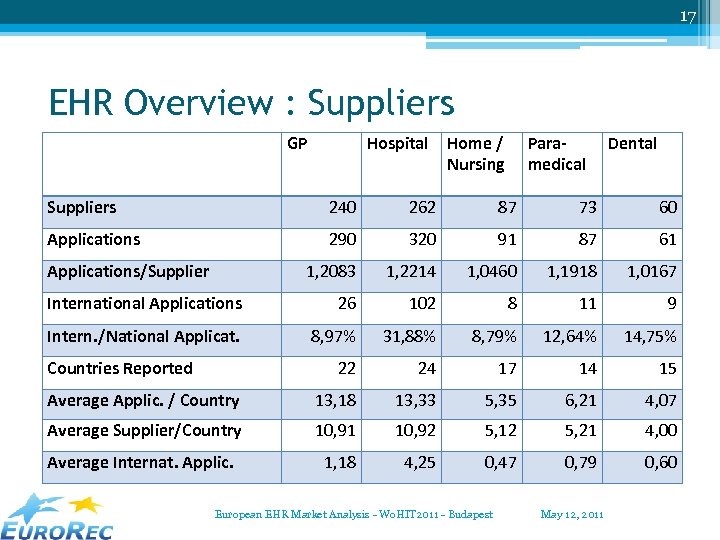
17 EHR Overview : Suppliers GP Hospital Home / Nursing Paramedical Dental Suppliers 240 262 87 73 60 Applications 290 320 91 87 61 1, 2083 1, 2214 1, 0460 1, 1918 1, 0167 International Applications 26 102 8 11 9 Intern. /National Applicat. 8, 97% 31, 88% 8, 79% 12, 64% 14, 75% 22 24 17 14 15 Average Applic. / Country 13, 18 13, 33 5, 35 6, 21 4, 07 Average Supplier/Country 10, 91 10, 92 5, 12 5, 21 4, 00 Average Internat. Applic. 1, 18 4, 25 0, 47 0, 79 0, 60 Applications/Supplier Countries Reported European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

18 Some conclusions • International applications/suppliers: ▫ rather limited ▫ most in Hospital Care and in Dental Care • Number of applications: most “medical” applications … easiest to get the information • Most suppliers support ONE application only (and in the same domain). Average 1, 21 for ambulatory systems, 1, 22 for hospital systems. European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011
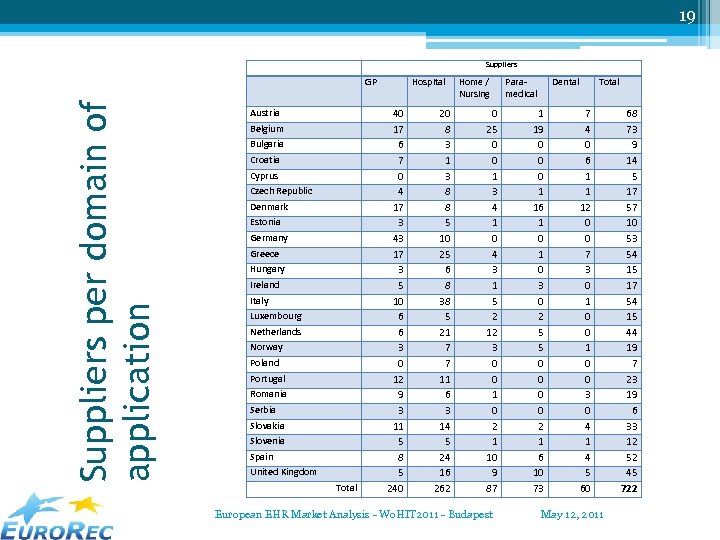
19 Suppliers per domain of application GP Austria Belgium Bulgaria Croatia Cyprus Czech Republic Denmark Estonia Germany Greece Hungary Ireland Italy Luxembourg Netherlands Norway Poland Portugal Romania Serbia Slovakia Slovenia Spain United Kingdom Total Hospital 40 17 6 7 0 4 17 3 43 17 3 5 10 6 6 3 0 12 9 3 11 5 8 5 240 20 8 3 1 3 8 8 5 10 25 6 8 38 5 21 7 7 11 6 3 14 5 24 16 262 Home / Nursing 0 25 0 0 1 3 4 1 0 4 3 1 5 2 12 3 0 0 1 0 2 1 10 9 87 European EHR Market Analysis - Wo. HIT 2011 - Budapest Paramedical Dental 1 19 0 0 0 1 16 1 0 3 0 2 5 5 0 0 2 1 6 10 73 Total 7 4 0 6 1 1 12 0 0 7 3 0 1 0 0 3 0 4 1 4 5 60 May 12, 2011 68 73 9 14 5 17 57 10 53 54 15 17 54 15 44 19 7 23 19 6 33 12 52 45 722
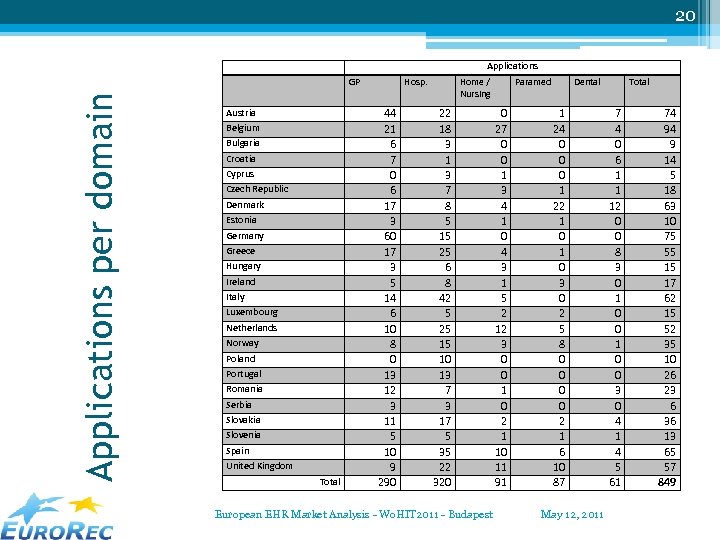
20 Applications per domain GP Austria Belgium Bulgaria Croatia Cyprus Czech Republic Denmark Estonia Germany Greece Hungary Ireland Italy Luxembourg Netherlands Norway Poland Portugal Romania Serbia Slovakia Slovenia Spain United Kingdom Total Hosp. 44 21 6 7 0 6 17 3 60 17 3 5 14 6 10 8 0 13 12 3 11 5 10 9 290 Home / Nursing 22 18 3 1 3 7 8 5 15 25 6 8 42 5 25 15 10 13 7 3 17 5 35 22 320 European EHR Market Analysis - Wo. HIT 2011 - Budapest Paramed 0 27 0 0 1 3 4 1 0 4 3 1 5 2 12 3 0 0 1 0 2 1 10 11 91 Dental 1 24 0 0 0 1 22 1 0 3 0 2 5 8 0 0 2 1 6 10 87 May 12, 2011 Total 7 4 0 6 1 1 12 0 0 8 3 0 1 0 0 3 0 4 1 4 5 61 74 94 9 14 5 18 63 10 75 55 15 17 62 15 52 35 10 26 23 6 36 13 65 57 849
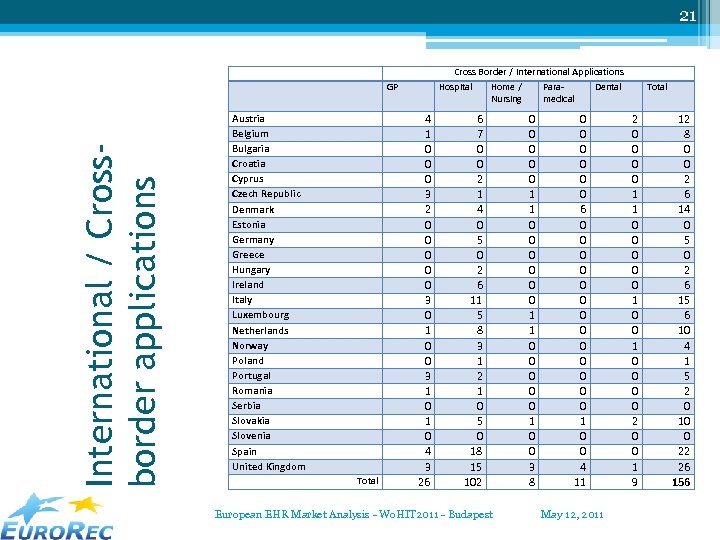
21 Cross Border / International Applications International / Crossborder applications GP Austria Belgium Bulgaria Croatia Cyprus Czech Republic Denmark Estonia Germany Greece Hungary Ireland Italy Luxembourg Netherlands Norway Poland Portugal Romania Serbia Slovakia Slovenia Spain United Kingdom Total Hospital 4 1 0 0 0 3 2 0 0 0 3 0 1 0 0 3 1 0 4 3 26 Home / Nursing 6 7 0 0 2 1 4 0 5 0 2 6 11 5 8 3 1 2 1 0 5 0 18 15 102 European EHR Market Analysis - Wo. HIT 2011 - Budapest Paramedical 0 0 0 0 0 1 1 0 0 0 1 0 0 3 8 Dental 0 0 0 6 0 0 0 0 1 0 0 4 11 May 12, 2011 Total 2 0 0 1 1 0 0 0 0 2 0 0 1 9 12 8 0 0 2 6 14 0 5 0 2 6 15 6 10 4 1 5 2 0 10 0 22 26 156

22 International suppliers / applications • International = ▫ Product used in more than one country ▫ Product owned by an entity in another country • Countries with most international systems: United Kingdom, Spain, Italy, Denmark and Austria. • Domains with most international systems: ▫ ▫ Hospital systems 31, 88% Dental systems 14, 75% Paramedical systems 12, 64% GP – Ambulatory systems 8, 97% European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

23 Cross-Border & Cross-Domain • International suppliers: ▫ 6 in Primary / Ambulatory Care ▫ 19 in Hospital Care (… hardware oriented? ) • Cross-Border applications ▫ None or nearly no applications in primary care / ambulatory care ▫ Same “product names” in several countries: same application ? ? ? (Alert, Clinicom, HP-HCIS, Impax, Lorenzo, Maxims Clinical, Orbis, PCS, Sorian, Stapro. Akord, Cerner) • Cross-Domain suppliers: 89 (at least in one country more than one target market) European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

24 Authorities, users and other stakeholders European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

25 Importance of stakeholders • Involvement of all important stakeholders is vital to the success of implementation and use of EHR systems. (EHR-Implement project) • Listed stakeholders are ▫ ▫ ▫ Health authorities Healthcare insurance organisations Public health organisations Research organisations – Academic institutes Healthcare institutes (associations) Healthcare professionals organisations European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

26 Selection criteria • Do they actually have an active role in quality assurance of EHR systems, if any in the country? • Did they manifest a particular knowledge of the domain (certification / quality labelling) in the past? • Is their approval or at least their cooperation essential in order to introduce successfully certification and quality labelling of EHR systems? • Is there a risk of obstruction not including them in the process of certification and quality labelling? • Are they representative or an important party in the particular EHR market, addressing a specified group of users? European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011
![[1] 27 Not all the regional health authorities and regional organisations are listed; [2] [1] 27 Not all the regional health authorities and regional organisations are listed; [2]](https://present5.com/presentation/2b5dba95a19decce1c8a6747741a5f4d/image-27.jpg)
[1] 27 Not all the regional health authorities and regional organisations are listed; [2] Specific for England Country Numberof Authorit. National Ministry Contact Person Departm. Identified Authorities National Ministry Austria Belgium Bulgaria Croatia Cyprus Czech Republic Denmark Estonia Germany Greece Hungary Ireland Italy Lithuania Luxembourg Netherlands Norway Poland Portugal Romania Serbia Slovakia Slovenia Spain England (UK) Other Public National Organisat. Specific Responsible Entity Regional Organis. National Public Entity 7 5 2 1 1 Y Y Y - 6 1 1 - Y Y Y - 3 - 2 Y Y Y 1 Y - 6 4 18 14 2 3 26 1 1 3 2 7 2 2 1 3 1 19 2 Y Y Y(2) Y Y Y Y Y Y Y - Y Y Y Y Y - 3 3 6 1 2 1 - Y Y Y - 2 16 7 22 5 17 Sev European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011
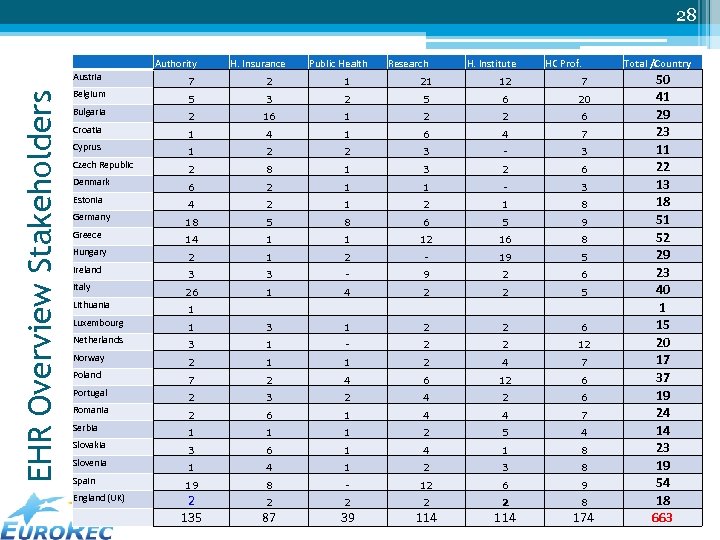
28 Authority H. Insurance Public Health Research H. Institute HC Prof. EHR Overview Stakeholders Austria 7 2 1 21 12 7 Belgium 5 3 2 5 6 20 Bulgaria 2 16 1 2 2 6 Croatia 1 4 1 6 4 7 Cyprus 1 2 2 3 - 3 Czech Republic 2 8 1 3 2 6 Denmark 6 2 1 1 - 3 Estonia 4 2 1 8 Germany 18 5 8 6 5 9 Greece 14 1 1 12 16 8 Hungary 2 1 2 - 19 5 Ireland 3 3 - 9 2 6 26 1 4 2 2 5 Italy Lithuania 1 Luxembourg 1 3 1 2 2 6 Netherlands 3 1 - 2 2 12 Norway 2 1 1 2 4 7 Poland 7 2 4 6 12 6 Portugal 2 3 2 4 2 6 Romania 2 6 1 4 4 7 Serbia 1 1 1 2 5 4 Slovakia 3 6 1 4 1 8 Slovenia 1 4 1 2 3 8 19 8 - 12 6 9 2 2 Spain England (UK) 2 135 European 87 Market Analysis - Wo. HIT 2011 - Budapest 114 EHR 39 114 8 May 12, 2011 174 Total /Country 50 41 29 23 11 22 13 18 51 52 29 23 40 1 15 20 17 37 19 24 14 23 19 54 18 663
29 (e)Health authorities European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

30 Role of Stakeholders (also regarding EHR and EHR Certification) European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

31 Market Fragmentation… • Not only a problem of the market • Internationally operating trusts ▫ Not per definition leading to product consolidation… even differentiating the same product in different countries. ▫ More interested (actually) in Market presence Making use of the outcome of EHR systems: clinical content • More consolidation within one area (country / region) between the domains than cross-border consolidation within the same domain. European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

32 And now? Maintenance is an important issue Maintenance requires resources Involve the market to maintain the database Use the database to promote quality labelling and certification • Make the data available to the market (restricted) • • European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011

33 Exploitation of the Overview • Mail to National Health Authorities ▫ Euro. Rec Letter ▫ National Partner Letter • End of April: mail to the Suppliers about ▫ Importance of quality assessment ▫ The Euro. Rec Seal European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011
34 Thank you European EHR Market Analysis - Wo. HIT 2011 - Budapest May 12, 2011
2b5dba95a19decce1c8a6747741a5f4d.ppt