c64c4ae0f7a863ae10015250968707b7.ppt
- Количество слайдов: 37

Investigating the influence of the marine biosphere on climate: Oxygen isotope measurements and model simulations Becky Alexander Harvard University Department of Earth and Planetary Sciences USC May 4, 2004

Overview • Sulfate aerosols: Importance and uncertainties • D 17 O sulfate: Resolve uncertainties • GEOS-CHEM: Global 3 D model • INDOEX: INDian Ocean EXperiment • Sulfate formation in the marine boundary layer (MBL): Seasalt, biogenic DMS, Climate • Future Plans
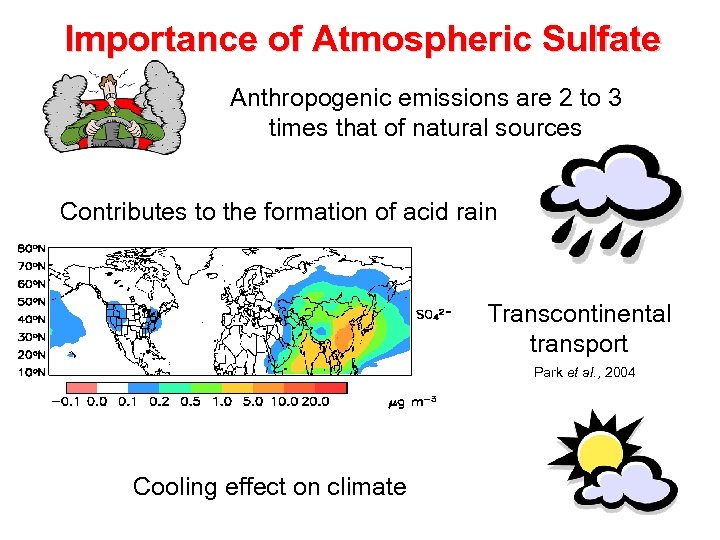
Importance of Atmospheric Sulfate Anthropogenic emissions are 2 to 3 times that of natural sources Contributes to the formation of acid rain Transcontinental transport Park et al. , 2004 Cooling effect on climate
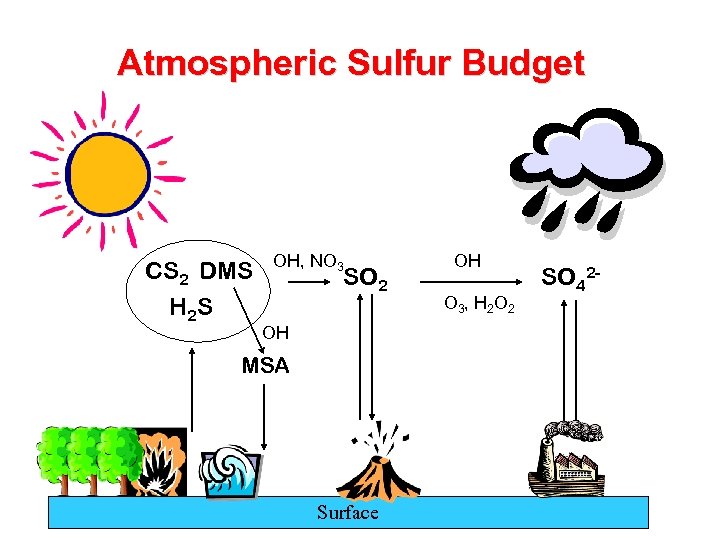
Atmospheric Sulfur Budget CS 2 DMS H 2 S OH, NO 3 SO 2 OH MSA Surface OH O 3 , H 2 O 2 SO 42 -
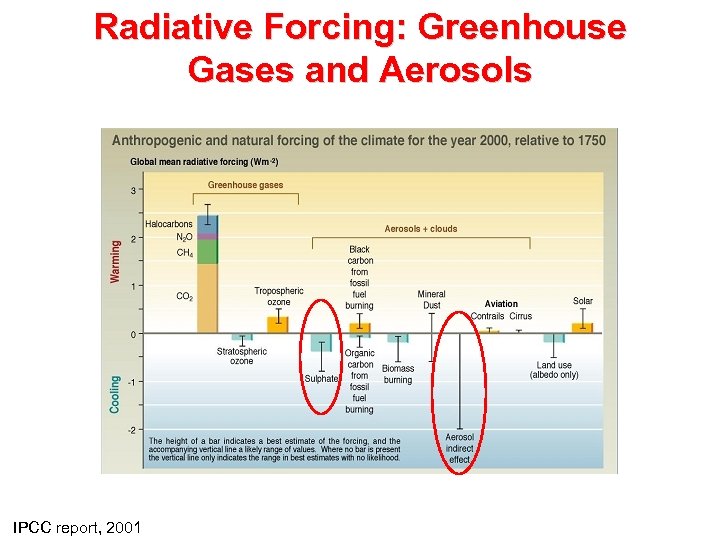
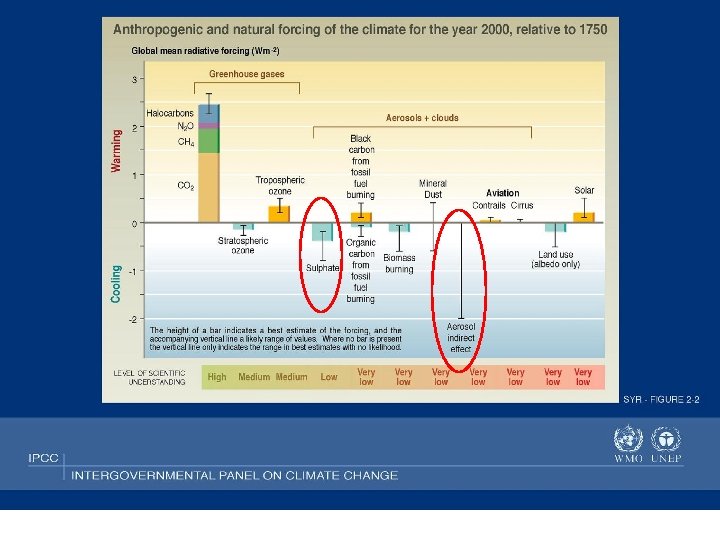
Radiative Forcing: Greenhouse Gases and Aerosols IPCC report, 2001
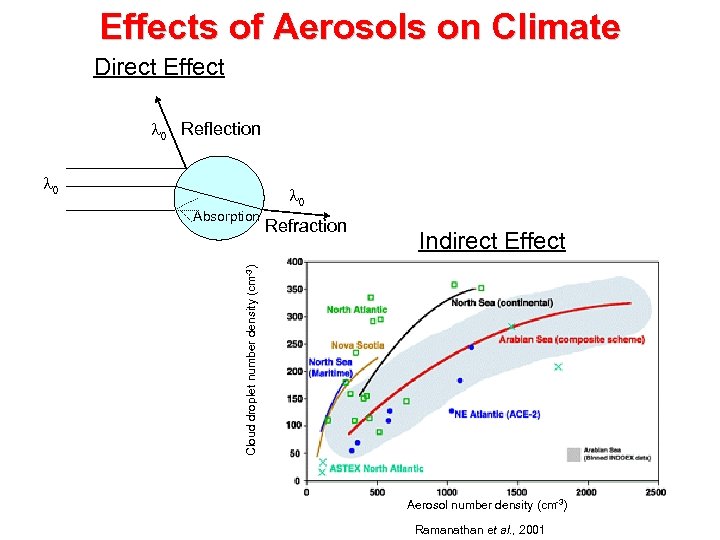
Effects of Aerosols on Climate Direct Effect l 0 Reflection l 0 Refraction Indirect Effect Cloud droplet number density (cm-3) Absorption Aerosol number density (cm-3) Ramanathan et al. , 2001

Atmospheric Aerosol Formation and Photosynthetic Rate Mt. Pinatubo volcano hn Gu et al. , 2003
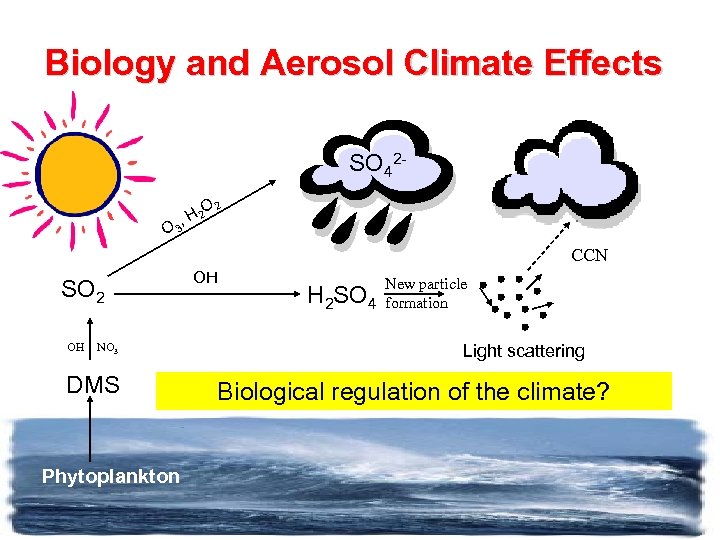
Biology and Aerosol Climate Effects SO 42 O 2 H 2 , O 3 CCN SO 2 OH NO 3 DMS Phytoplankton OH H 2 SO 4 New particle formation Light scattering Biological regulation of the climate?
![Mass-Dependent Fractionation (‰) = [(Rsample/Rstandard) – 1] 1000 18 O: R = 18 O/16 Mass-Dependent Fractionation (‰) = [(Rsample/Rstandard) – 1] 1000 18 O: R = 18 O/16](https://present5.com/presentation/c64c4ae0f7a863ae10015250968707b7/image-9.jpg)
Mass-Dependent Fractionation (‰) = [(Rsample/Rstandard) – 1] 1000 18 O: R = 18 O/16 O; 17 O: R = 17 O/16 O d 17 O/d 18 O 0. 5
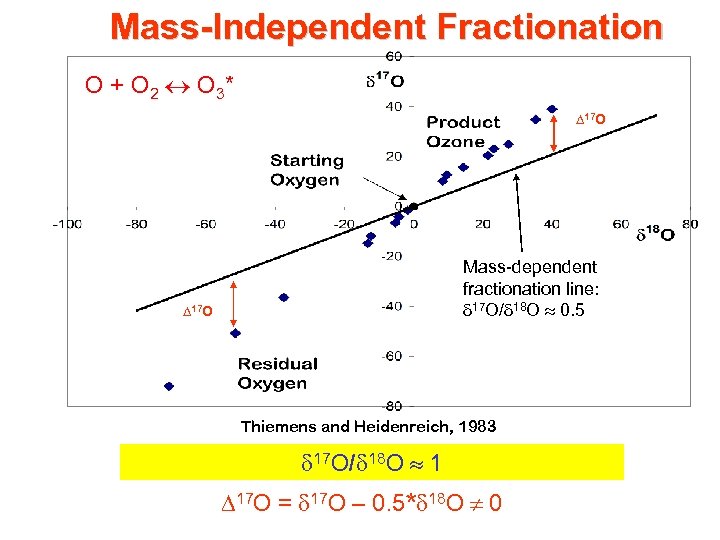
Mass-Independent Fractionation O + O 2 O 3* D 17 O Mass-dependent fractionation line: 17 O/ 18 O 0. 5 D 17 O Thiemens and Heidenreich, 1983 17 O/ 18 O 1 D 17 O = 17 O – 0. 5* 18 O 0
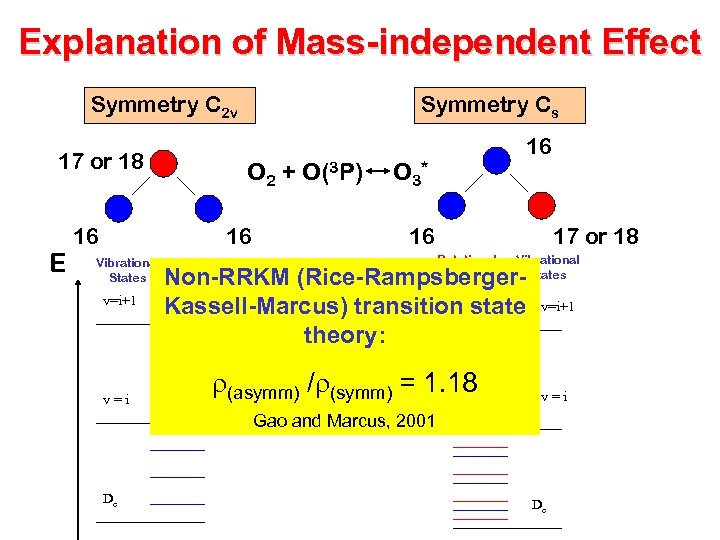
Explanation of Mass-independent Effect Symmetry C 2 v 17 or 18 E 16 Symmetry Cs O 2 + O(3 P) 16 Vibrational States v=i+1 v=i 16 O 3* 16 17 or 18 Rotational States Vibrational States Non-RRKM (Rice-Rampsberger. Kassell-Marcus) transition state theory: r(asymm) /r(symm) = 1. 18 v=i+1 v=i Gao and Marcus, 2001 De De
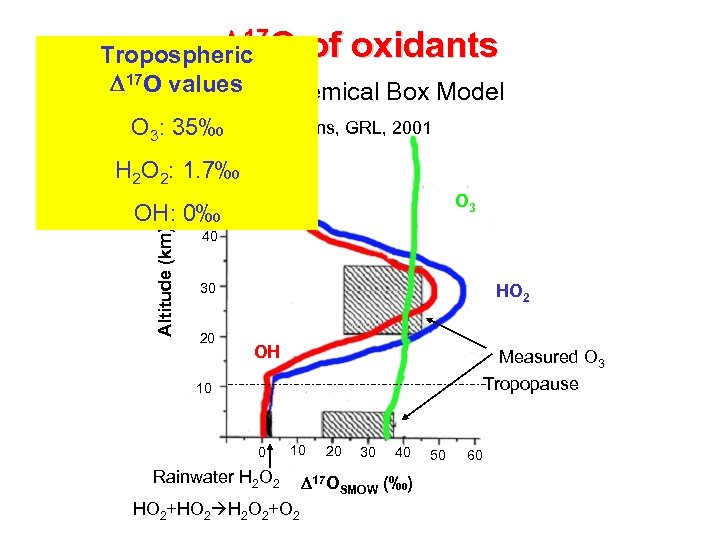
D 17 O of oxidants Tropospheric D 17 O values Photochemical Box Model O 3: 35‰ 60 Lyons, GRL, 2001 H 2 O 2: 1. 7‰ 50 O 3 Altitude (km) OH: 0‰ 40 30 20 HO 2 OH Measured O 3 Tropopause 10 0 10 Rainwater H 2 O 2 HO 2+HO 2 H 2 O 2+O 2 20 30 40 D 17 OSMOW (‰) 50 60
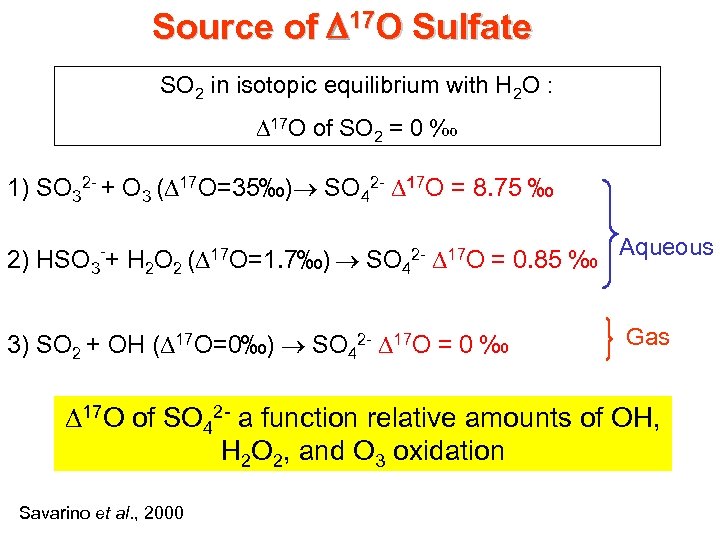
Source of D 17 O Sulfate SO 2 in isotopic equilibrium with H 2 O : D 17 O of SO 2 = 0 ‰ 1) SO 32 - + O 3 (D 17 O=35‰) SO 42 - D 17 O = 8. 75 ‰ 2) HSO 3 -+ H 2 O 2 (D 17 O=1. 7‰) SO 42 - D 17 O = 0. 85 ‰ Aqueous 3) SO 2 + OH (D 17 O=0‰) SO 42 - D 17 O = 0 ‰ Gas D 17 O of SO 42 - a function relative amounts of OH, H 2 O 2, and O 3 oxidation Savarino et al. , 2000

GEOS-CHEM http: //www-as. harvard. edu/chemistry/trop/geos/index. html • Global 3 -D model of atmospheric chemistry • 4ºx 5º horizontal resolution, 26 -30 layers in vertical • Driven by assimilated meteorology (1987 –present). • Includes aqueous and gas phase chemistry: S(IV) + OH (gas-phase) S(IV) + O 3/H 2 O 2 (in-cloud, p. H=4. 5) • Off-line sulfur chemistry (uses monthly mean OH and O 3 fields from a full chemistry, coupled aerosol simulation)

GEOS-CHEM D 17 O Sulfate Simulation Use constant, global D 17 O value for oxidants D 17 O ‰ method O 3 35 Photochemical model Lyons 2001 H 2 O 2 1. 7 Rainwater measurements Savarino and Thiemens 1999 Experimental Dubey et al. , 1997 OH SO 2 + OH 0 (gas phase) reference D 17 O=0‰ S(IV) + H 2 O 2 (in cloud, p. H=4. 5) D 17 O=0. 85‰ S(IV) + O 3 D 17 O=8. 75‰ (in cloud, p. H=4. 5)
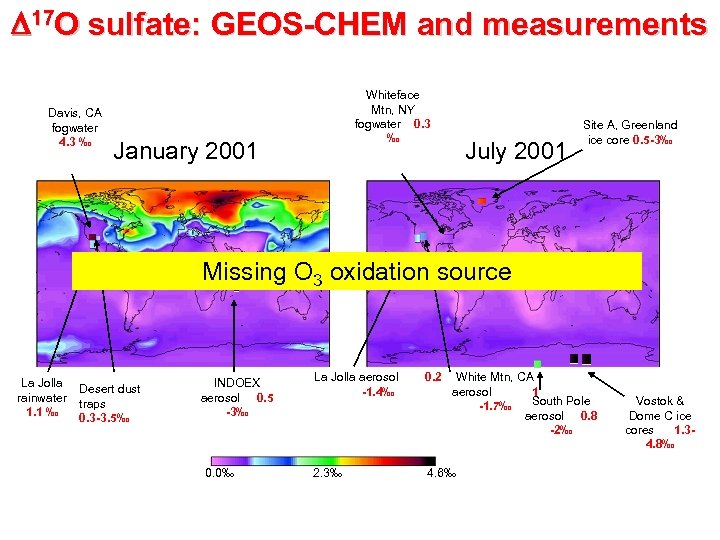
D 17 O sulfate: GEOS-CHEM and measurements Davis, CA fogwater 4. 3 ‰ Whiteface Mtn, NY fogwater 0. 3 ‰ January 2001 July 2001 Site A, Greenland ice core 0. 5 -3‰ Missing O 3 oxidation source La Jolla rainwater 1. 1 ‰ Desert dust traps 0. 3 -3. 5‰ INDOEX aerosol 0. 5 -3‰ 0. 0‰ La Jolla aerosol -1. 4‰ 0. 2 2. 3‰ 4. 6‰ White Mtn, CA aerosol 1 South Pole -1. 7‰ aerosol 0. 8 -2‰ Vostok & Dome C ice cores 1. 34. 8‰
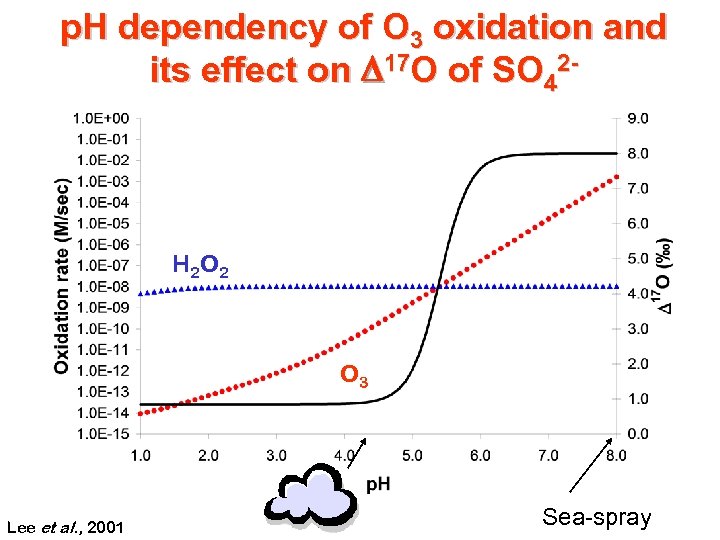
p. H dependency of O 3 oxidation and its effect on D 17 O of SO 42 - H 2 O 2 O 3 Lee et al. , 2001 O 3 Sea-spray

O 3 oxidation on sea-salt aerosols Sea-salt emissions are a function of wind speed p. H = 8, O 3 oxidation dominant Sea salt flux to atmosphere (Gong et al. , 2002): 1. 01 x 104 Tg/year 11. 1 Tg(S)/year Global DMS emissions (Seinfeld and Pandis, 1998): 15 -25 Tg(S)/year 44 -74% of SO 2 (from DMS) oxidized to sulfate by O 3 on sea-salt aerosols
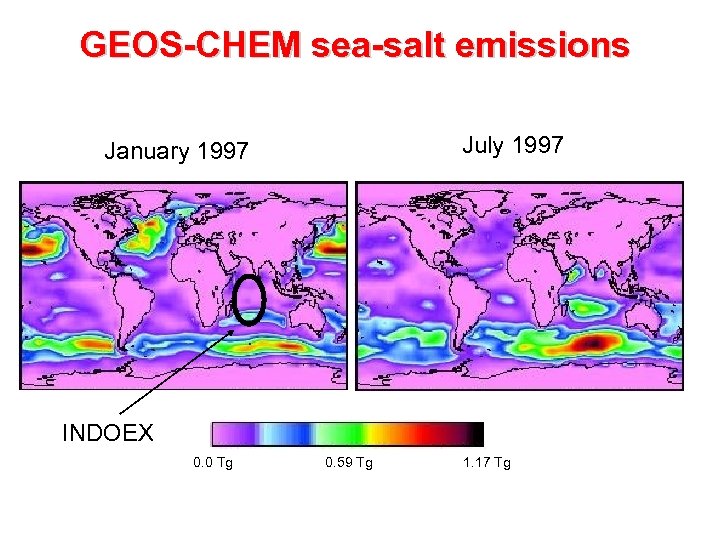
GEOS-CHEM sea-salt emissions July 1997 January 1997 INDOEX 0. 0 Tg 0. 59 Tg 1. 17 Tg
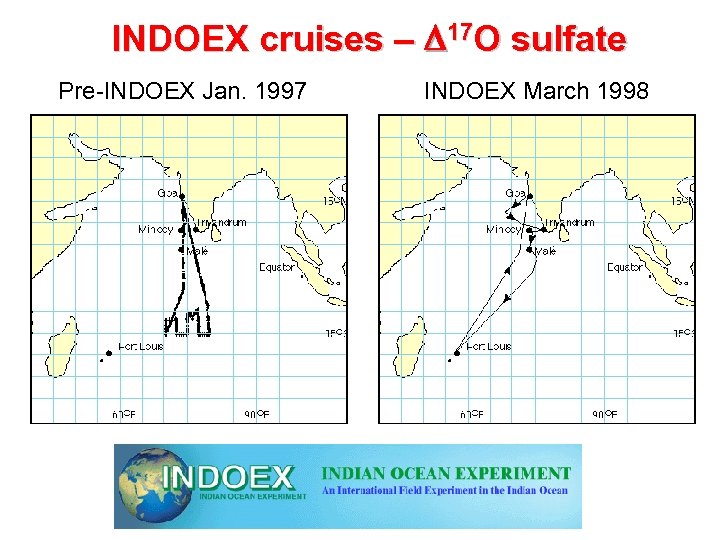
INDOEX cruises – D 17 O sulfate Pre-INDOEX Jan. 1997 INDOEX March 1998
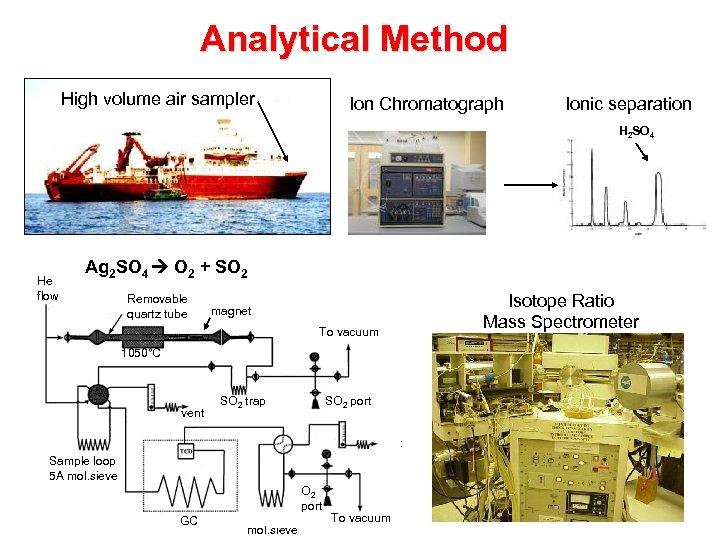
Analytical Method High volume air sampler Ion Chromatograph Ionic separation H 2 SO 4 He flow Ag 2 SO 4 O 2 + SO 2 Removable quartz tube Isotope Ratio Mass Spectrometer magnet To vacuum 1050°C vent SO 2 trap SO 2 port vent Sample loop 5 A mol. sieve GC O 2 port O 2 loop 5 A To vacuum mol. sieve
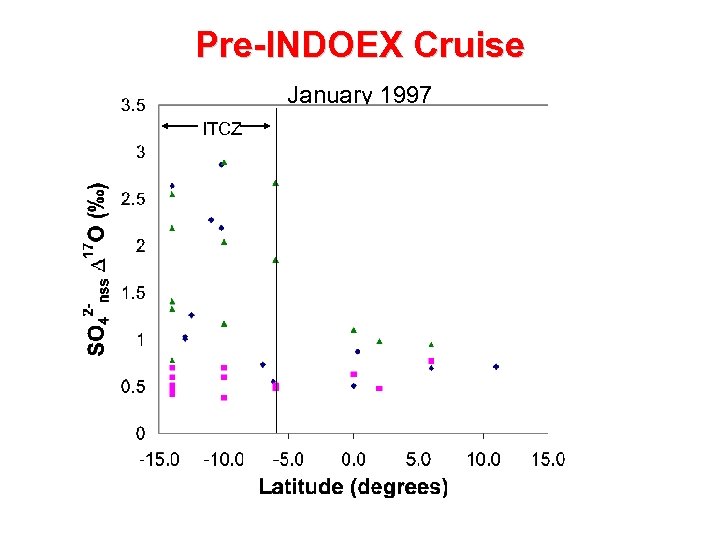
Pre-INDOEX Cruise January 1997 ITCZ

Enhanced p. H of sea-salt aerosols over sea water? Laskin et al. Science (2003): OH(g) + Cl-(interface) (HO…Cl-)interface + (HO…Cl-)interface Cl 2 + 2 OHk(OH-) k(SO 42 -)
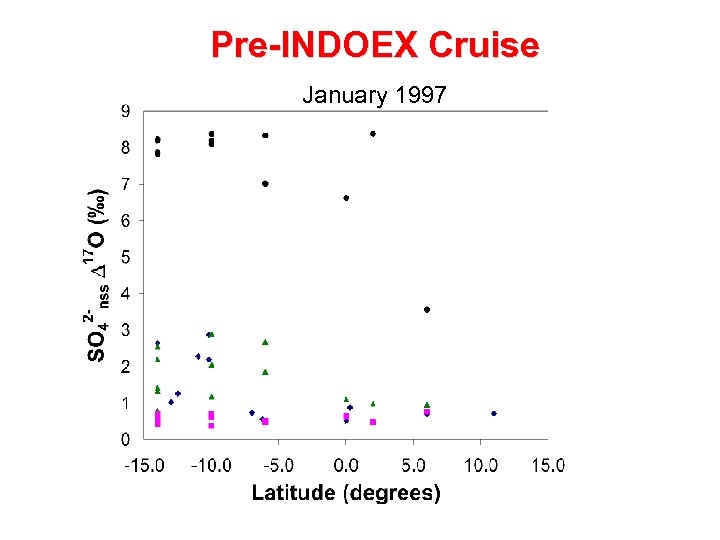
Pre-INDOEX Cruise January 1997
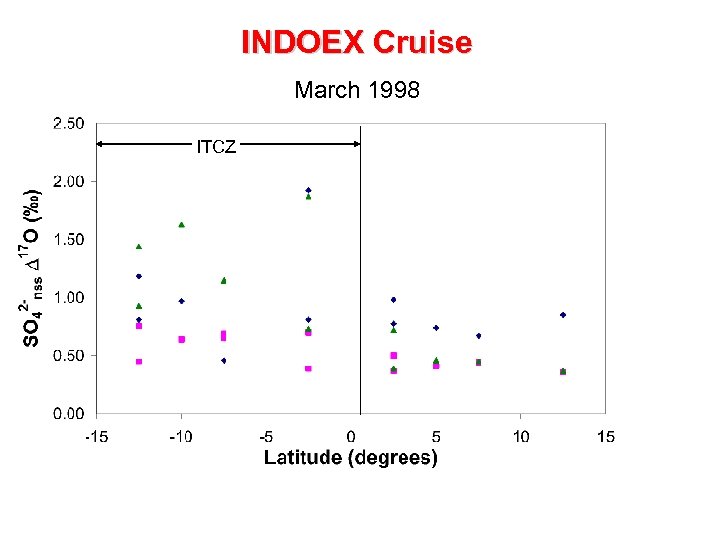
INDOEX Cruise March 1998 ITCZ
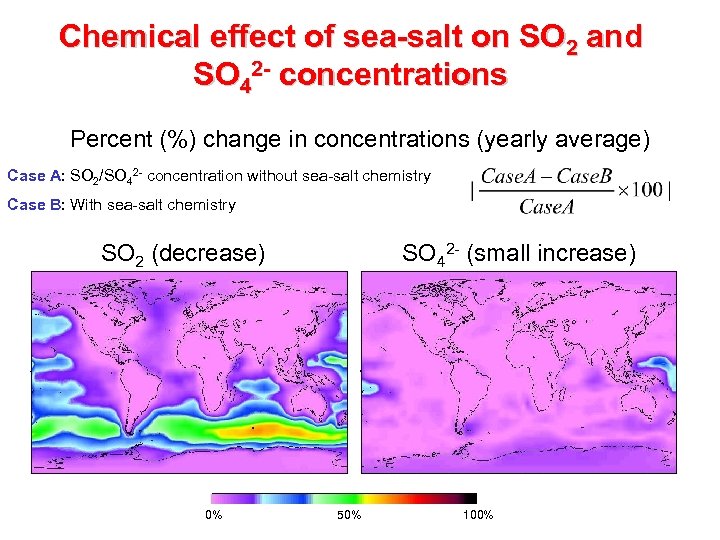
Chemical effect of sea-salt on SO 2 and SO 42 - concentrations Percent (%) change in concentrations (yearly average) Case A: SO 2/SO 42 - concentration without sea-salt chemistry Case B: With sea-salt chemistry SO 2 (decrease) 0% SO 42 - (small increase) 50% 100%
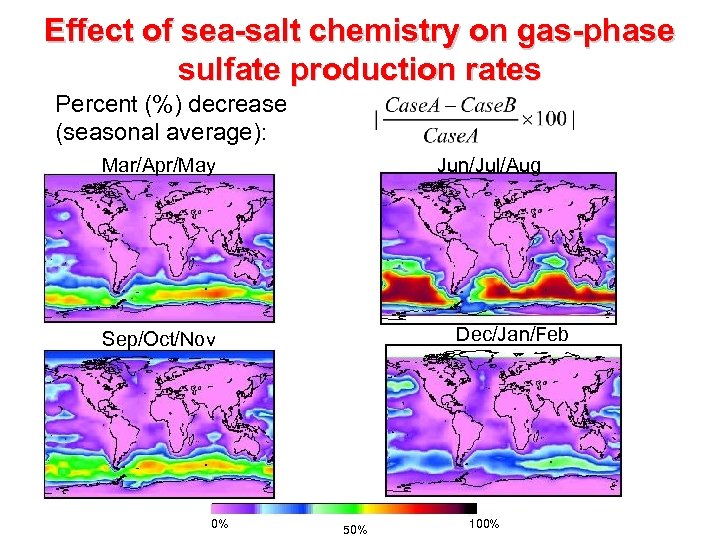
Effect of sea-salt chemistry on gas-phase sulfate production rates Percent (%) decrease (seasonal average): Mar/Apr/May Jun/Jul/Aug Dec/Jan/Feb Sep/Oct/Nov 0% 50% 100%
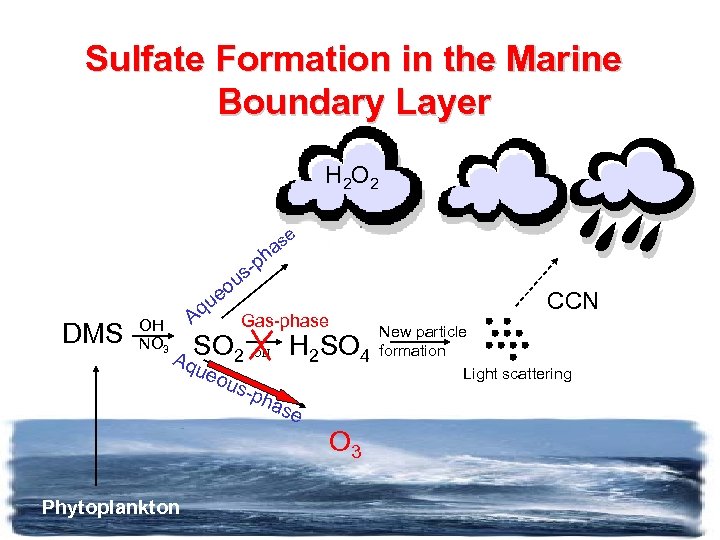
Sulfate Formation in the Marine Boundary Layer H 2 O 2 e s ha p us eo u DMS OH NO 3 Aq Gas-phase Aq. SO 2 OH H 2 SO ueo uspha se CCN New particle 4 formation Light scattering O 3 Phytoplankton
Lessons from INDOEX and GEOS-CHEM • The evolution of alkalinity depends on atmospheric acidity and has a short (< 1 hour) atmospheric lifetime. • Sulfate formation on sea-salt chemistry should be included in models estimating the radiative effects of sulfate from DMS emissions. Ship SO 2 in North Atlantic 0 3. 3 6. 6 106 kg S • The acidification of the atmosphere, particularly in the Northern Hemisphere, may have increased the effectiveness of the marine biological control on climate.
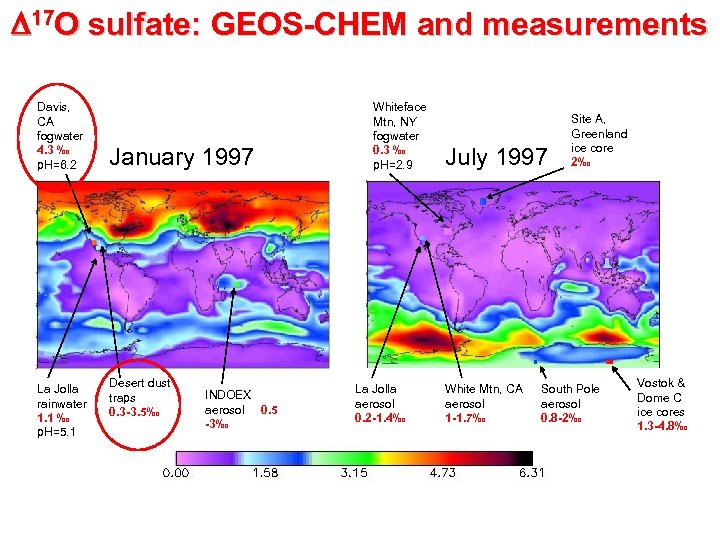
D 17 O sulfate: GEOS-CHEM and measurements Davis, CA fogwater 4. 3 ‰ p. H=6. 2 La Jolla rainwater 1. 1 ‰ p. H=5. 1 January 1997 Desert dust traps 0. 3 -3. 5‰ INDOEX aerosol 0. 5 -3‰ Whiteface Mtn, NY fogwater 0. 3 ‰ p. H=2. 9 La Jolla aerosol 0. 2 -1. 4‰ July 1997 White Mtn, CA aerosol 1 -1. 7‰ Site A, Greenland ice core 2‰ South Pole aerosol 0. 8 -2‰ Vostok & Dome C ice cores 1. 3 -4. 8‰
Future Plans – Dust
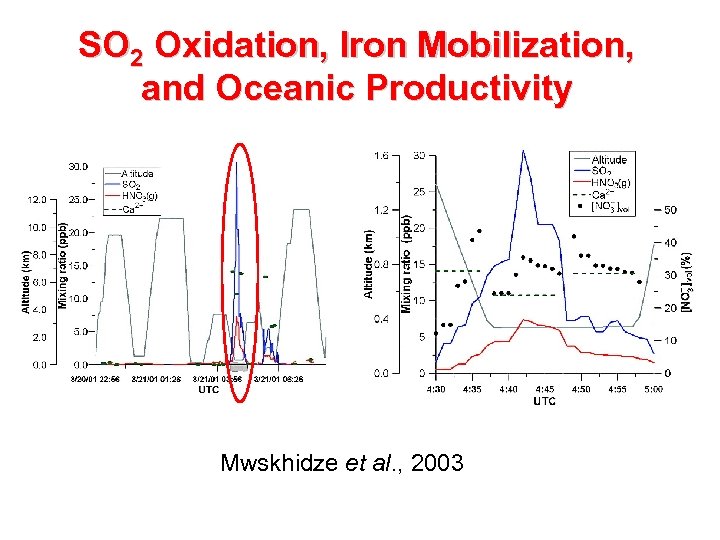
SO 2 Oxidation, Iron Mobilization, and Oceanic Productivity Mwskhidze et al. , 2003
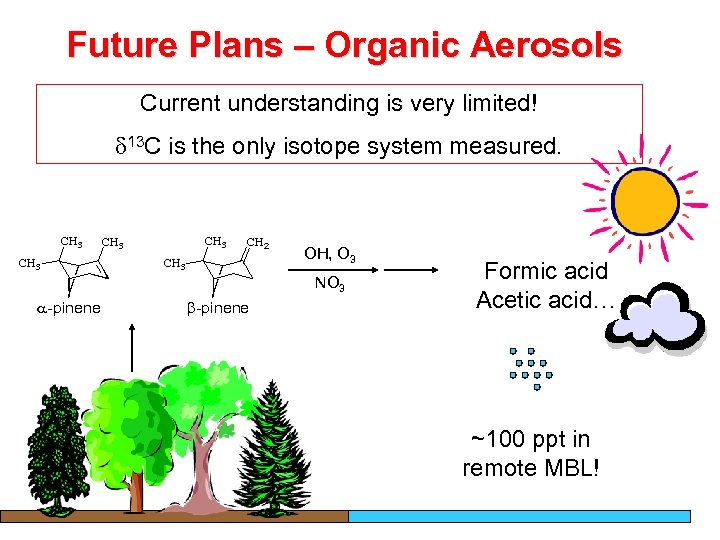
Future Plans – Organic Aerosols Current understanding is very limited! 13 C is the only isotope system measured. OH, O 3 NO 3 a-pinene b-pinene Formic acid Acetic acid… ~100 ppt in remote MBL!

Acknowledgements Mark H. Thiemens Charles Lee Daniel Jacob Dan Schrag V. Ramanathan Ann Pearson Rokjin Park Qinbin Li Joël Savarino Bob Yantosca NOAA Climate and Global Change Postdoctoral Fellowship Daly Postdoctoral Fellowship (Department of Earth and Planetary Sciences, Harvard University)

Atmospheric Aerosol Formation and Photosynthetic Rate Mt. Pinatubo volcano Aerosol Optical Depth Gu et al. , 2003
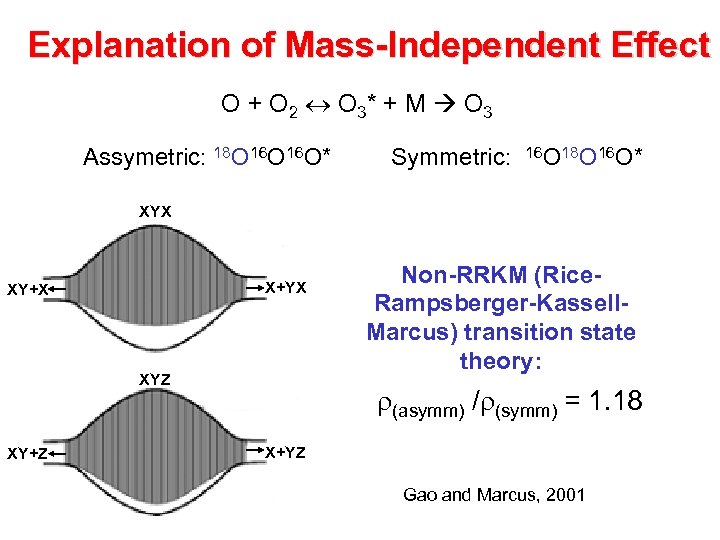
Explanation of Mass-Independent Effect O + O 2 O 3* + M O 3 Assymetric: 18 O 16 O* Symmetric: 16 O 18 O 16 O* XYX X+YX XY+X XYZ XY+Z Non-RRKM (Rice. Rampsberger-Kassell. Marcus) transition state theory: r(asymm) /r(symm) = 1. 18 X+YZ Gao and Marcus, 2001
c64c4ae0f7a863ae10015250968707b7.ppt