7384e2eb50d1bd95e7da2b324697cd3f.ppt
- Количество слайдов: 43

Introduction to Acids and Bases Khady Guiro NJIT C 2 PRISM Fellow

DO NOW • Brainstorm – What are common acids and bases? – What tools can be used to measure acids and bases?

Introduction • Organisms are often very sensitive to the effect of acids and bases in their environment. • They need to maintain a stable internal p. H in order to survive—even in the event of environmental changes. • Many naturally occurring biological, geological, and man-made chemicals are capable of stabilizing the environment’s p. H. • This may allow organisms to better survive in diverse environments found throughout the earth.

Introduction Cont. • Acids and bases are a way of classifying compounds based upon what happens to them when you place them in water. • An acid is a substance that yields an excess of hydrogen ions when dissolved in water. • A base is a substance that yields an excess of hydroxide ions when dissolved in water.
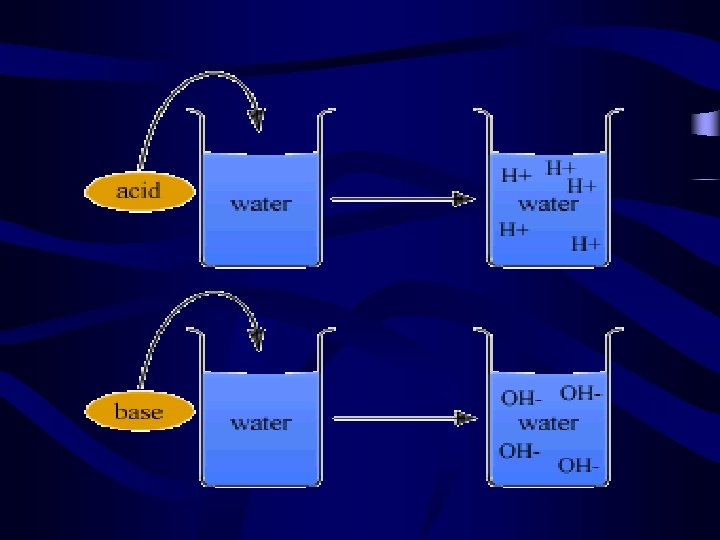

Definitions of Acids and Base s • An acid is a substance that breaks into ions in an aqueous solution. • A Base (is a substance that breaks into ions in an aqueous solution. • Note: aqueous solution is any solution where is the solvent.

ACIDS

Common Acids Citric Acid Vinegar (acetic or ethanoic acid)

Characteristics of Acids • Sour taste (for edible acids) • May burn your skin • Turn litmus paper from blue to red.

Other Examples of Acids • HCl Hydrochloric Acid • H 2 SO 4 Sulfuric Acid • CH 3 COOH Acetic Acid (Vinegar)

BASES

Common Bases Ammonia

Characteristics of Bases • Bitter taste (for edible bases only) • Can be poisonous and corrosive. • Feels slippery • Turn litmus from red to blue.

Other examples of Bases • Sodium Hydroxide Na. OH • Potassium Hydroxide. KOH • Ammonia NH 3

Three Definitions of Acids and Bases

Three Definitions of Acids and Bases • Svante Arrhenius (Swedish 1887) • Bronsted- Lowry (Danish and English 1923 independently proposed definition) • Gilbert Lewis (American 1920’s)

Arrhenius Definition of Acids and Bases • Acids produce hydrogen ions (H+) in an aqueous solution, while bases produce hydroxide ion (OH-). Acid: HCl (aq) H+ (aq) + Cl- (aq) Base: Na. OH(aq) Na+(aq) + OH-(aq)

Bronsted- Lowry Definition of Acids and Bases • An acid is a proton (H+) donor. • A base is a proton (H+) acceptor. • Example: HF + H 2 O H 3 O+ + FAcid Base H 3 O+ : hydronium ion

Lewis Definition of Acids and Bases • An acid accepts a pair of electrons. • A base donates a pair of electrons. • This is a more general definition than the previous two. • Example: : NH 3 + H+ NH 4+

Three Models of Acids and Bases Model Definition of Acid Definition of Base Arrhenius H+ producer OH- producer Bronsted-Lowry H+ donor H+ acceptor Lewis Electron-pair donor Electron-pair acceptor

Conjugate Acids and Bases • Conjugate acid is the particle formed that has received the proton. (ex: H 3 O+) • Conjugate base is the particle left from the acid once it has donated the proton.

Conjugate Acids and Bases • General expression: HA (aq) + H 2 O (l) H 3 O+ (aq) + A- (aq) Acid Base Conjugate Acid Conjugate Base

Importance of WATER • Water acts as an acid and as a base. It is amphoteric

Acids • A dilute acid has lots of water and a small amount of acid • A concentrated acid has lots of acid and not much water so must be handled carefully • A strong acid releases lots of H+ • A weak acid releases fewer H+

The extent to which a compound is acidic or basic is measured by the p. H scale.

Measuring acid strength? • To decide if something is an acid or a base we can use an indicator. • Litmus and Universal Indicator are examples of indicators. • They change colour depending on if they are in an acid or a base.

What is the p. H scale? • The p. H scale measures how acidic or basic a solution is.

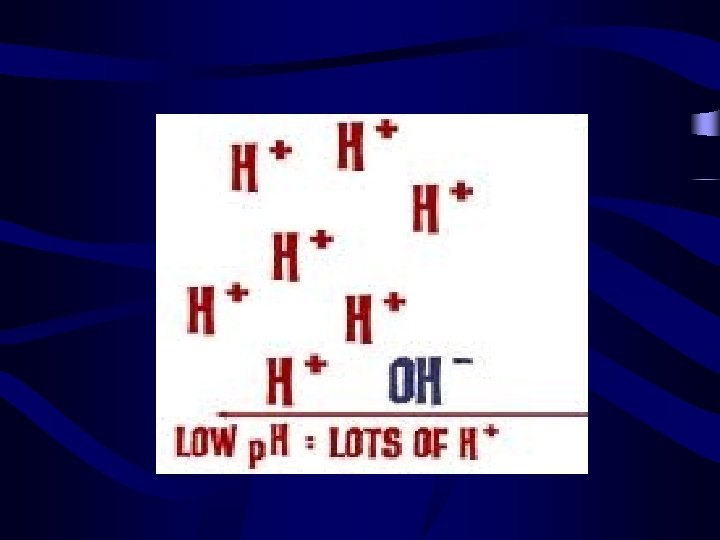
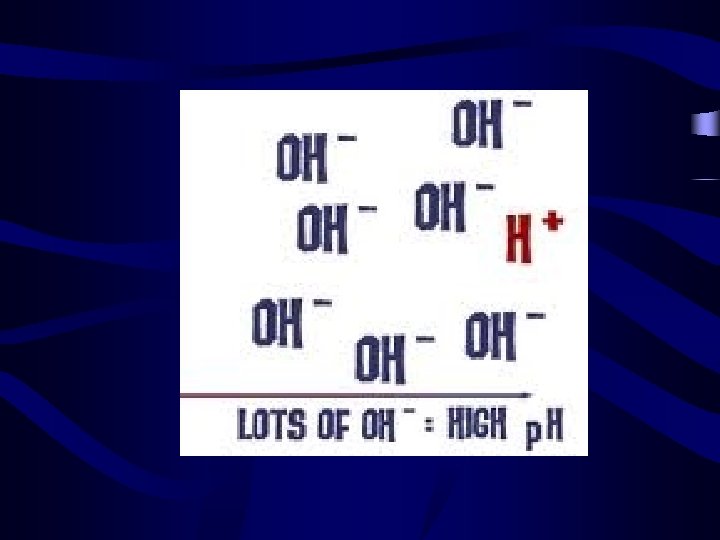
Identifying Acids and Bases • Acids have a ph from 0 -7 • Lower p. H value indicates a stronger acid. • Bases have a p. H from 7 -14 • Higher p. H value indicates a stronger base.

Did we Miss something? ? • What happens when the p. H of a substance is 7? • Ans: A p. H level of 7 indicates a Neutral Substance i. e: Water!

Why Learn about Acids & Bases? • What do you think is the p. H level of NYC tap water? • The p. H of a swimming pool must be checked periodically. Why? • Is it important for Lakes & Rivers to maintain a certain p. H?

Situations in which p. H is controlled • • • “Heartburn” Planting vegetables and flowers Fish Tanks and Ponds Blood Swimming pools

Acid Rain • Pollution in the air (sulfur dioxide, carbon dioxide, nitrogen dioxide) combines with water to form various acids. .


p. H in the Digestive System • Mouth-p. H around 7. Saliva contains amylase, an enzyme which begins to break carbohydrates into sugars. • Stomach- p. H around 2. Proteins are broken down into amino acids by the enzyme pepsin. • Small intestine-p. H around 8. Most digestion ends. Small molecules move to bloodstream toward cells that use them

Review

Acids + þ Produce H (as H 3 O+) ions in water þ Produce a negative ion (-) too þ Taste sour þ Corrode metals þ React with bases to form salts and water

Bases l. Produce OH ions in water l. Taste bitter, chalky l. Are electrolytes l. Feel soapy, slippery l. React with acids to form salts and water

p. H of Common Substances Timberlake, Chemistry 7 th Edition, page 335

Buffers • A buffer is a solution that resists changes in p. H when small amounts of acids and bases are added.

Vernier experiment

Today’s Experiment • Using Vernier Software: – Calibrate p. H sensor using Buffers – Test the p. H of distilled water, HCL, NAOH. – Test p. H of 5 Unknowns – Test p. H of common house hold items (Ketchup, coffee, tap water, Detergent)
7384e2eb50d1bd95e7da2b324697cd3f.ppt