f6a9156972999d082c4badf72e9ab6dc.ppt
- Количество слайдов: 25
 Intro to chemistry Video 1. 1 Metric
Intro to chemistry Video 1. 1 Metric
 Our country still uses an old system with non uniform measurements such as: fractions of an inch. . . 12 inches to a foot…. 3 feet to a yard…. 5. 5 yards to a rod. . . 320 rods to a mile. . . 43, 560 sq ft to an acre. . . But almost all other countries use the metric system, which is disadvantageous for us.
Our country still uses an old system with non uniform measurements such as: fractions of an inch. . . 12 inches to a foot…. 3 feet to a yard…. 5. 5 yards to a rod. . . 320 rods to a mile. . . 43, 560 sq ft to an acre. . . But almost all other countries use the metric system, which is disadvantageous for us.
 But we do use the metric for a few things: We buy cola in liters. . . We buy memory cards in bites… We run 10 km races. . . We swim in 25 meter pools. . . Why haven’t we switched entirely to metric?
But we do use the metric for a few things: We buy cola in liters. . . We buy memory cards in bites… We run 10 km races. . . We swim in 25 meter pools. . . Why haven’t we switched entirely to metric?
 Measuring Length in meters When measuring a person we would use meters. If we are measuring an ant, would meters still be feasible? What should we use? If we are measuring the distance from your house to the school, what should we use? Always pick a prefix with a value close to what you are measuring.
Measuring Length in meters When measuring a person we would use meters. If we are measuring an ant, would meters still be feasible? What should we use? If we are measuring the distance from your house to the school, what should we use? Always pick a prefix with a value close to what you are measuring.
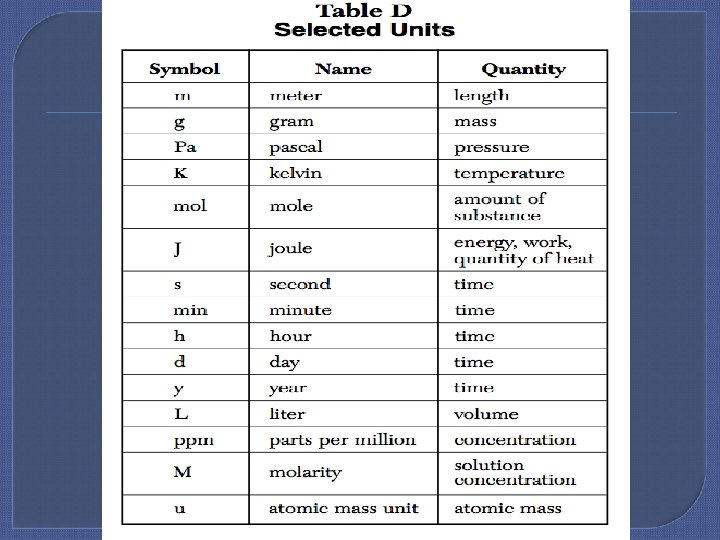
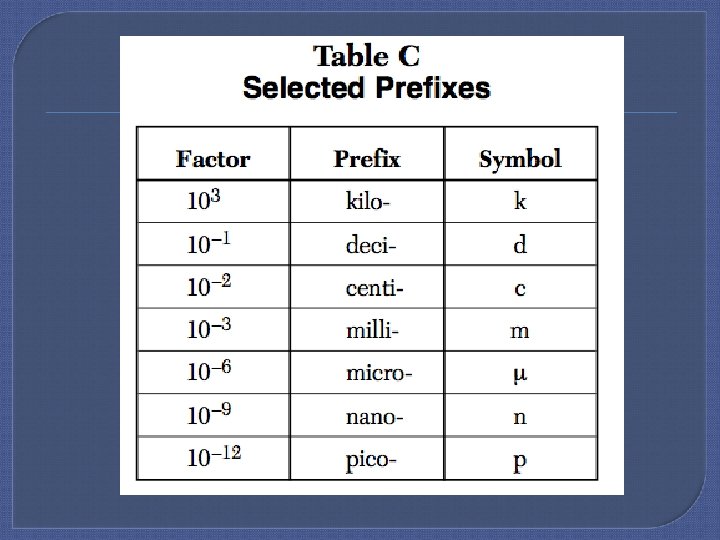
 The Metric System If a unit is getting larger (m km) the number must get smaller. [If the unit gets smaller (m cm) the number gets larger. ] Examples: • 1. 23. 5 cm = 0. 000235 km • 2. 3567 m. L = L 3. 567 • 3. 0. 0984 g = ug 98400
The Metric System If a unit is getting larger (m km) the number must get smaller. [If the unit gets smaller (m cm) the number gets larger. ] Examples: • 1. 23. 5 cm = 0. 000235 km • 2. 3567 m. L = L 3. 567 • 3. 0. 0984 g = ug 98400
 Temperature Conversions Notice that each scales is marked with BP and FP of water as well as absolute zero. The degree size of Celsius is equal to Kelvin. Therefore we adjust only for zero points: C = K – 273 K = C + 273
Temperature Conversions Notice that each scales is marked with BP and FP of water as well as absolute zero. The degree size of Celsius is equal to Kelvin. Therefore we adjust only for zero points: C = K – 273 K = C + 273
 Thinking in Celsius -10° Celsius = frigid (14° F) 0° Celsius = cold (32° F) 10° Celsius = cool (50° F) 20° Celsius = comfortably warm (68° F) 30° Celsius = hot (86° F) 40° Celsius = very hot (104° F) 50° Celsius = Phoenix Hot (120°F)
Thinking in Celsius -10° Celsius = frigid (14° F) 0° Celsius = cold (32° F) 10° Celsius = cool (50° F) 20° Celsius = comfortably warm (68° F) 30° Celsius = hot (86° F) 40° Celsius = very hot (104° F) 50° Celsius = Phoenix Hot (120°F)
 Intro to chemistry Video 1. 2 Scientific Notation
Intro to chemistry Video 1. 2 Scientific Notation
 Scientific Notation What is the purpose for using scientific notation in science?
Scientific Notation What is the purpose for using scientific notation in science?
 Scientific Notation M x 10 n M is between 1 and 10 n is the number of decimal spaces moved to make M
Scientific Notation M x 10 n M is between 1 and 10 n is the number of decimal spaces moved to make M
 Rules 1. 2. 3. 4. Find the decimal point. If it is not written, it is at the end of the number. Move the decimal point to make the number between 1 and 10 Place the number of space you moved the decimal in the n spot. If you original number was above 1, the exponent is positive. If the number was smaller that 1, the exponent is negative.
Rules 1. 2. 3. 4. Find the decimal point. If it is not written, it is at the end of the number. Move the decimal point to make the number between 1 and 10 Place the number of space you moved the decimal in the n spot. If you original number was above 1, the exponent is positive. If the number was smaller that 1, the exponent is negative.
 Scientific Notation 1020000 is equal to • 1. 02 x 106 0. 00789 is equal to • 7. 89 x 10 -3 3. 45 x 105 is equal to • 345000 1. 23 x 10 -4 is equal to • 0. 000123
Scientific Notation 1020000 is equal to • 1. 02 x 106 0. 00789 is equal to • 7. 89 x 10 -3 3. 45 x 105 is equal to • 345000 1. 23 x 10 -4 is equal to • 0. 000123
 Scientific Notation Mathematics Multiplication and Division: 1. 2. Multiply or divide the base numbers. When multiplying, add exponents. When dividing, subtract exponents. (8 x 105)(2 x 103) = 16 x 108 or 1. 6 x 109 (8 x 105)/(2 x 103) = 4 x 102
Scientific Notation Mathematics Multiplication and Division: 1. 2. Multiply or divide the base numbers. When multiplying, add exponents. When dividing, subtract exponents. (8 x 105)(2 x 103) = 16 x 108 or 1. 6 x 109 (8 x 105)/(2 x 103) = 4 x 102
 Scientific Notation Mathematics Addition and Subtraction: The exponents must be the same. Change your numbers to make this possible. 2. Add or subtract base numbers and do not change the exponent. * Remember: if the decimal move makes the base number smaller, the exponent increases. 1. 5 x 105 + 3 x 104 = 5 x 105 + 0. 3 x 105 = 5. 3 x 105
Scientific Notation Mathematics Addition and Subtraction: The exponents must be the same. Change your numbers to make this possible. 2. Add or subtract base numbers and do not change the exponent. * Remember: if the decimal move makes the base number smaller, the exponent increases. 1. 5 x 105 + 3 x 104 = 5 x 105 + 0. 3 x 105 = 5. 3 x 105
 Intro to Chemistry Video 1. 3 Significant Figures
Intro to Chemistry Video 1. 3 Significant Figures
 Precision Versus Accuracy Precision: reproducibility, repeatability Accuracy: closeness to the correct answer 1. A student obtains the following data: 2. 57 m. L 2. 59 m. L 2. 58 m. L 2. 98 m. L Compare these pieces in terms of precision and accuracy.
Precision Versus Accuracy Precision: reproducibility, repeatability Accuracy: closeness to the correct answer 1. A student obtains the following data: 2. 57 m. L 2. 59 m. L 2. 58 m. L 2. 98 m. L Compare these pieces in terms of precision and accuracy.
 Precision Versus Accuracy Describe these diagrams in terms of precision and accuracy: The first shows precision, not accuracy. The second shows accuracy, not precision.
Precision Versus Accuracy Describe these diagrams in terms of precision and accuracy: The first shows precision, not accuracy. The second shows accuracy, not precision.
 In this classroom, what is more important: Precision or Accuracy?
In this classroom, what is more important: Precision or Accuracy?
 Precision! Due to lack of precise equipment and variable climates we will most likely not end up with accurate results. Therefore, we will focus on refining our lab skills and strive for precise results.
Precision! Due to lack of precise equipment and variable climates we will most likely not end up with accurate results. Therefore, we will focus on refining our lab skills and strive for precise results.
 Significant Figures When scientists take measurements their equipment can measure with varying degrees of precision. A scientists final calculation can only be as precise as their least precise measurement. Count digits in your measured number to determine their level of precision.
Significant Figures When scientists take measurements their equipment can measure with varying degrees of precision. A scientists final calculation can only be as precise as their least precise measurement. Count digits in your measured number to determine their level of precision.
 Counting Significant Figures All natural numbers 1 -9 count 569 has 3 SF 3. 456 has 4 SF The once. number zero is tricky… • Zeros always count between natural numbers: 109 have 3 SF 50089 has 5 SF • Zeros before a decimal and natural number never count. 0. 00789 has 3 SF 001234 has 4 SF • Zeros after natural numbers only count IF there is a decimal present. 100 has 1 SF 100. has 3 SF 100. 0 has 4 SF
Counting Significant Figures All natural numbers 1 -9 count 569 has 3 SF 3. 456 has 4 SF The once. number zero is tricky… • Zeros always count between natural numbers: 109 have 3 SF 50089 has 5 SF • Zeros before a decimal and natural number never count. 0. 00789 has 3 SF 001234 has 4 SF • Zeros after natural numbers only count IF there is a decimal present. 100 has 1 SF 100. has 3 SF 100. 0 has 4 SF
 Adding/Subtracting Significant Figures Remember: you can only be as precise as your least precise measurement. Therefore, when adding and subtracting, round your answer to the least number of DECIMAL PLACES. 2. 0 + 5. 61 = 7. 6 5. 67 + 102. 111 = 107. 78 23 + 11. 10 = 34
Adding/Subtracting Significant Figures Remember: you can only be as precise as your least precise measurement. Therefore, when adding and subtracting, round your answer to the least number of DECIMAL PLACES. 2. 0 + 5. 61 = 7. 6 5. 67 + 102. 111 = 107. 78 23 + 11. 10 = 34
 Multiplying/Dividing Significant Figures Remember: you can only be as precise as your least precise measurement. Therefore, when multiplying or dividing, round your answer to the least number of significant figures. 2. 0 x 35. 1 = 70. 2 = 70. 5. 11 x 98. 654 = 504. 12194 = 504 72. 1 / 3. 123 = 23. 0867755 = 23. 1
Multiplying/Dividing Significant Figures Remember: you can only be as precise as your least precise measurement. Therefore, when multiplying or dividing, round your answer to the least number of significant figures. 2. 0 x 35. 1 = 70. 2 = 70. 5. 11 x 98. 654 = 504. 12194 = 504 72. 1 / 3. 123 = 23. 0867755 = 23. 1


