e2d7da4987e51ffc13dbbbe6a3f31e65.ppt
- Количество слайдов: 27

International Conference on Sustainable Built Environment NANCO AND UNIVERSITY OF MELBOURNE JOINT RESEARCH SESSION ON NANOTECHNOLOGY AND SUSTAINABLE BUILT ENVIRONMENT 13 -14 December, 2010 at Earls Regency, Kandy, Sri Lanka

DEFLOURINATION OF DRINKING WATER USING LAYERED DOUBLE HYDROXIDES Presented by: Nadeesh Madusanka Other group members: Eranga Warsakoon Nuwan Gunawardene Hasitha Kalahe Imalka Munaweera Nilwala Kottegoda

Introduction ØAbout 1 billion people in the world, mostly in developing countries, have no access to potable water Ø 2. 6 billion people lacking access to adequate sanitation.

ØSri Lanka currently faces a number of water related problems. Ø 70 % of the Sri Lankan population satisfies their water needs from dug wells, deep wells, reservoirs and rivers.
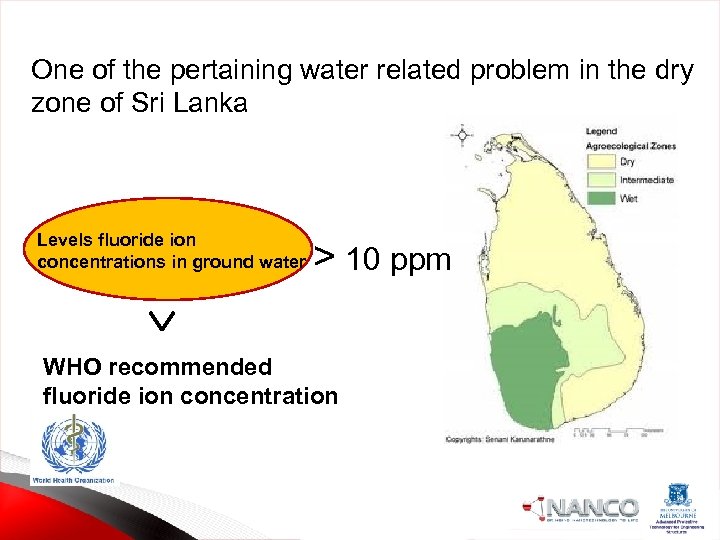
One of the pertaining water related problem in the dry zone of Sri Lanka Levels fluoride ion concentrations in ground water > 10 ppm WHO recommended fluoride ion concentration 1 ppm

Adverse effects due to the excessive exposure to fluoride in drinking-water Chronic kidney disease (CKD) Mild dental fluorosis to crippling skeletal fluorosis

Fluoride removing methods in water ØAdsorption ØPrecipitation or Adsorption ØIon-exchange ØElectro dialysis ØElectrochemical Processes

The most common approach to remove fluoride in the dry zone of Sri Lanka • Use of brick filters -low efficiency. • Combination of alum (or aluminium chloride) and lime (or sodium aluminate), together with bleaching powder, are added to high-fluoride water, stirred and left to settle. • Flocculation, sedimentation and filtration.

ØCurrently, nanotechnology has not left any field Untouched. ØThis new technology can be harnessed to provide sustainable solutions to water related problems prevailing in Sri Lanka particularly to remove fluoride ions from drinking water.

Possible Nanomaterials ØLayered Double Hydroxide ØNano Ti. O 2 ØNano Magenetite
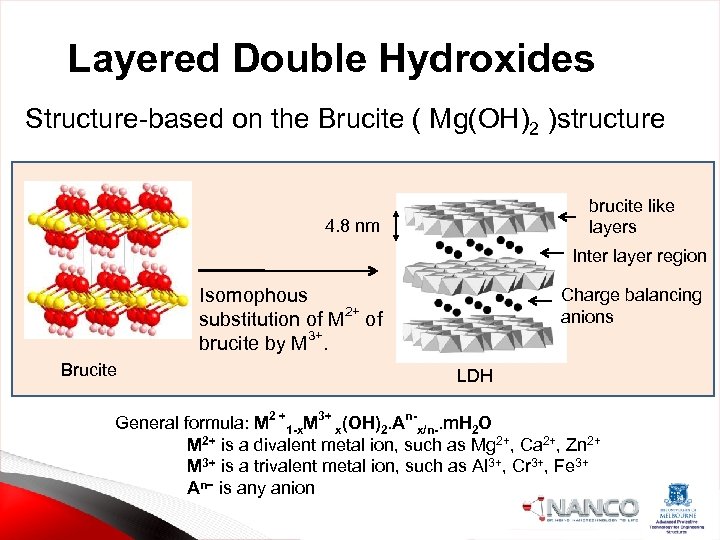
Layered Double Hydroxides Structure-based on the Brucite ( Mg(OH)2 )structure brucite like layers 4. 8 nm Inter layer region Isomophous substitution of M 2+ of 3+ brucite by M. Brucite Charge balancing anions LDH General formula: M 2 +1 -x. M 3+ x(OH)2. An-x/n-. m. H 2 O M 2+ is a divalent metal ion, such as Mg 2+, Ca 2+, Zn 2+ M 3+ is a trivalent metal ion, such as Al 3+, Cr 3+, Fe 3+ An− is any anion

Characteristics of Layered Double Hydroxides ØThermally stable. ØA range of trivalent and divalent cationic combinations are available. ØThe cationic ratio can be modified within a wide range. ØLarge anion exchange capacity. ØCan be synthesized as bulk quantities with relatively low cost and under ambient conditions.

Applications of Layered Double Hydroxides ØBiomedical applications-Drug stabilizer, Antacids, Gene and Drug Delivery ØCatalysis. Hydrogenation, Polymerizations ØPolymer nanocomposites ØIon Exchanger
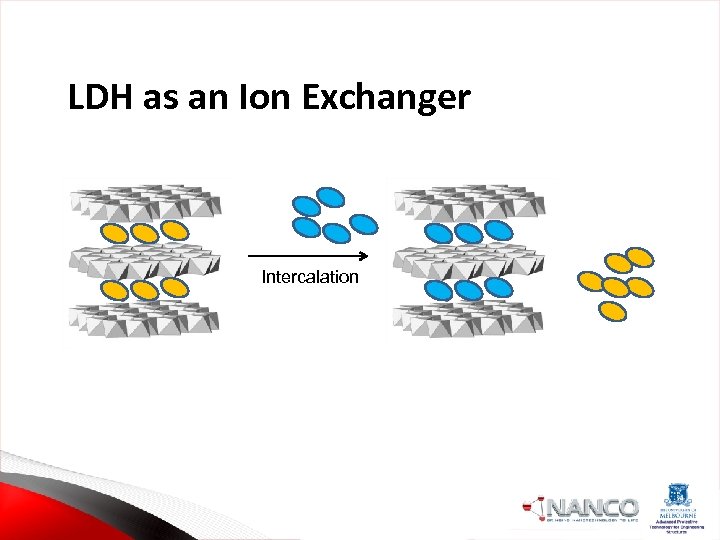
LDH as an Ion Exchanger Intercalation

Research methodology 1) Synthesis of Mg-Al-hydroxide LDH (Mg-Al-OH) Mg-Al Mixed metal solution + 50 ml 25 o. C, p. H=10 Anionic solution (Al: OH- =1: 10) 50 ml co- precipitation reaction Under N 2 atmosphere Mg. Al-OH- LDH Ref. Hibino and Jones , 2001 J. Mater. Chem 11 1321

2) Flouride removal from drinking water using Mg-Al-OH-LDH ØDifferent weights of LDH were added to water samples containing fluoride ions (8. 2 ppm , 100 ml) and kept it for 24 hours on the magnetic stirrer. ØFinal concentration of the fluoride solution was tested using an ion selective electrode. ØThe efficiency of the LDH material was compared with that of the conventional method where brick powder is used as the ion exchange medium.

3) Regeneration of the material ØExhausted LDH powder was calcined at 400 o. C for three hours. ØThen the resulting mixed oxide was characterized by PXRD and FTIR. ØThen it was exposed to 1 M Na. OH solution for 24 hours with mechanical stirring and the resulting product was characterized using PXRD and FTIR. ØThe fluoride gas coming out was allowed to absorb into 1 M calcium hydroxide solution.

4) Characterization ØScanning Electron Microscopy ØPowder X-ray Diffraction ØFourier Transform Infra Red Spectroscopy ØChemical Analysis

Results SEM Images of LDHs
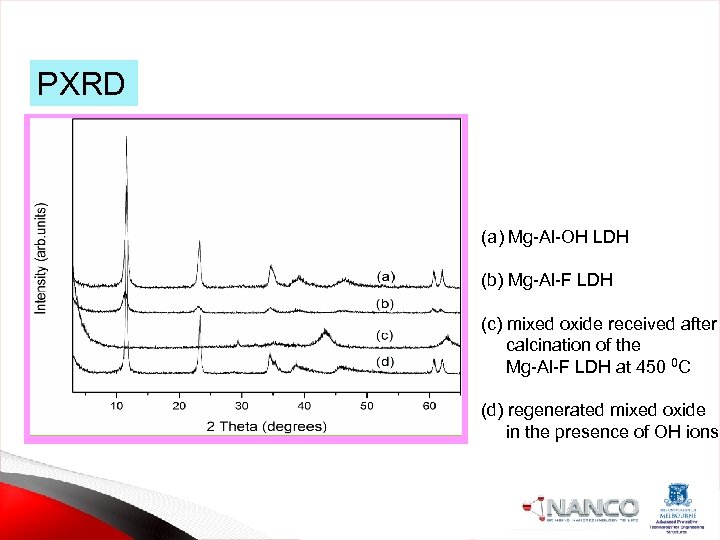
PXRD (a) Mg-Al-OH LDH (b) Mg-Al-F LDH (c) mixed oxide received after calcination of the Mg-Al-F LDH at 450 0 C (d) regenerated mixed oxide in the presence of OH ions.
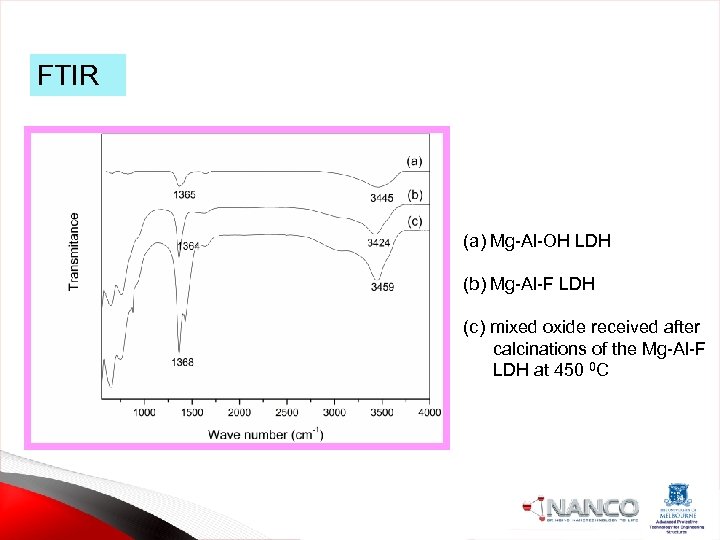
FTIR (a) Mg-Al-OH LDH (b) Mg-Al-F LDH (c) mixed oxide received after calcinations of the Mg-Al-F LDH at 450 0 C
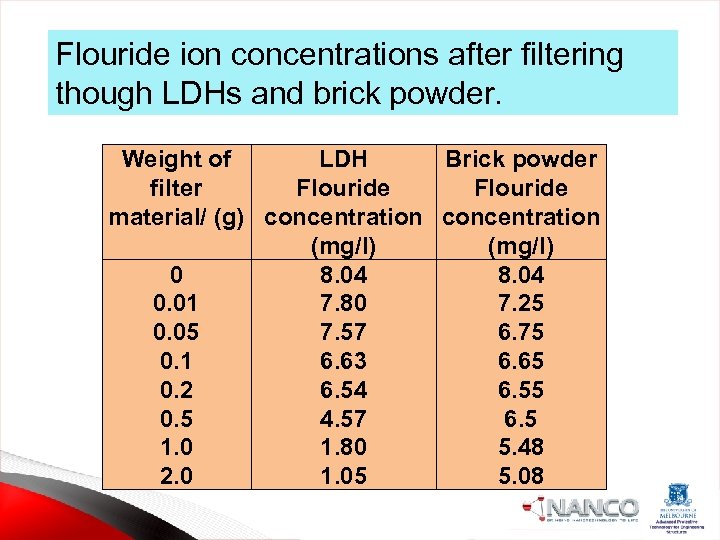
Flouride ion concentrations after filtering though LDHs and brick powder. Weight of LDH Brick powder filter Flouride material/ (g) concentration (mg/l) 0 8. 04 0. 01 7. 80 7. 25 0. 05 7. 57 6. 75 0. 1 6. 63 6. 65 0. 2 6. 54 6. 55 0. 5 4. 57 6. 5 1. 0 1. 80 5. 48 2. 0 1. 05 5. 08
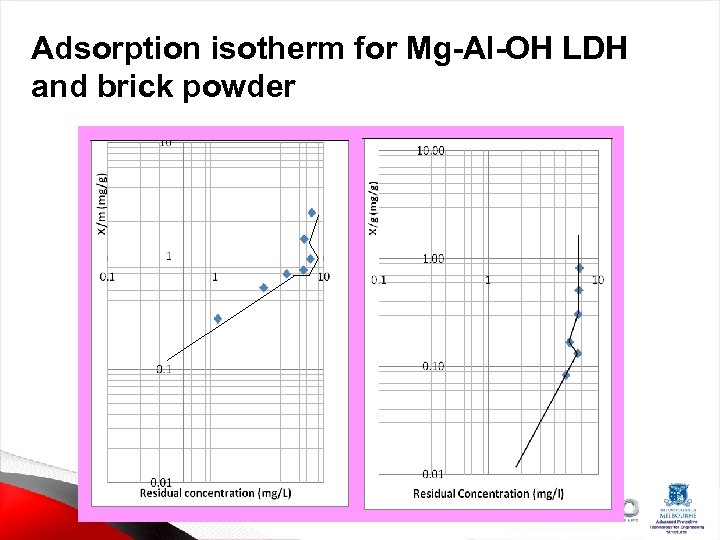
Adsorption isotherm for Mg-Al-OH LDH and brick powder
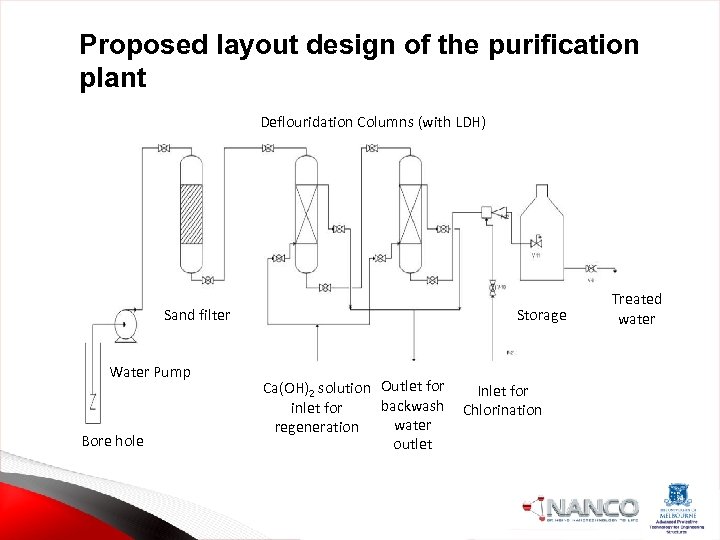
Proposed layout design of the purification plant Deflouridation Columns (with LDH) Storage Sand filter Water Pump Bore hole Ca(OH)2 solution Outlet for backwash inlet for water regeneration outlet Inlet for Chlorination Treated water

Conclusions ØLayered double hydroxides have displayed high efficiency in removing fluoride ions from drinking water compared to the basic methods currently used. ØLDHs can be used in either regional water purification units or domestic house hold filters. ØProduct is sustainable. economically and environmentally ØAn added advantage of using LDHs would be its superior capability of removing other anions such as carbonate, sulfates etc

Acknowledgement ØD. S. Senanayake College, Colombo 07. ØSri Lanka Institute of Nanotechnology

DEFLOURINATION OF DRINKING WATER USING LAYERED DOUBLE HYDROXIDES Research Group: Nadeesh Madusanka Eranga Warsakoon Nuwan Gunawardene Hasitha Kalahe Imalka Munaweera Nilwala Kottegoda
e2d7da4987e51ffc13dbbbe6a3f31e65.ppt