447c2ab3bec86f49a52c280041dede3d.ppt
- Количество слайдов: 39

INTERMOLECULAR FORCES My name is Bond James Bond A member of the Intermolecular Forces

INTERMOLECULAR FORCES Intermolecular forces are the forces that hold different molecules together. Let’s have a brief history lesson here.

INTERMOLECULAR FORCES Way way back we learned about electronic configuration. We used electronic configuration to help us draw Lewis Structures. We used Lewis Structures to help us try to understand the 3 D shape of the molecules. These concepts all help us to understand how ONE molecule is put together.

INTERMOLECULAR FORCES What we will be doing now is looking at how one molecule interacts with another molecule. The following topics are distinctly different from chemical reactions.

Analogy Time Chemical Reaction John and Mary go to the Prom. John and Mary get married that night. 12 months later John and Mary are parents. CHEMICAL REACTION Intermolecular Forces John and Mary go the Prom. John looks at Mary and winks John and Mary flirt all night. They leave separately.

INTERMOLECULAR FORCES Intermolecular forces are WEAK forces that cause molecules to have minor interactions with each other. The molecules might have slightly different properties because of intermolecular forces but are still the same basic molecule.

INTERMOLECULAR FORCES There are four intermolecular forces. 1) ionic--the strongest--between ions 2) hydrogen bonding--2 nd strongest 3) dipole--the 3 rd strongest and 4) London Dispersion Forces-the 4 th strongest (this makes it the weakest)

INTERMOLECULAR FORCES On a scale of 1 -100, ionic intermolecular forces are about a 100 hydrogen bonding forces are about a 10 dipole-dipole forces are about a 3 London dispersion forces are less than 1. For comparison, bonding Forces are about 100 or so.

INTERMOLECULAR FORCES We are not going to look at the ionic intermolecular force. We will look at the other three forces and then we will look at how these forces influence the nature of the molecules.

Hydrogen Bonding is an intermolecular force that is created when a HYDROGEN bonded to one of the three bullies (fluorine, oxygen, or nitrogen) has a weak attraction to another atom in a different molecule that is also from one of the three bullies.

Hydrogen Bonding Story… The bullies (F, O, and N) LIKE electrons They like them a lot. In a INTRAmolecular bond between a bully and a hydrogen, the electron will be very close to the bully and not very close to the hydrogen. This means hydrogen is lonely for electrons.

Hydrogen Bonding Story… The bully basically says to the hydrogen: “I will put these two electrons in my pocket and take good care of them. If you are nice I will let look at them once a week and MAYBE you can hold them if you are nice. So, hydrogen is lonely for electrons.

Hydrogen Bonding Story… Hydrogen doesn’t want to totally abandon the bully, but hydrogen may do a little serious window shopping at the local electron store. If another bully walks by, the hydrogen will come up beside this second bully and say “hey big bully, what you doing, Can I see your electrons? ”

Hydrogen Bonding We end up with a 3 dimensional arrangement of molecules loosely held together by the intermolecular bonds. Hydrogen bonding provides a weak three dimensional network to the water molecules. Each water molecule has some basic interaction with other molecules
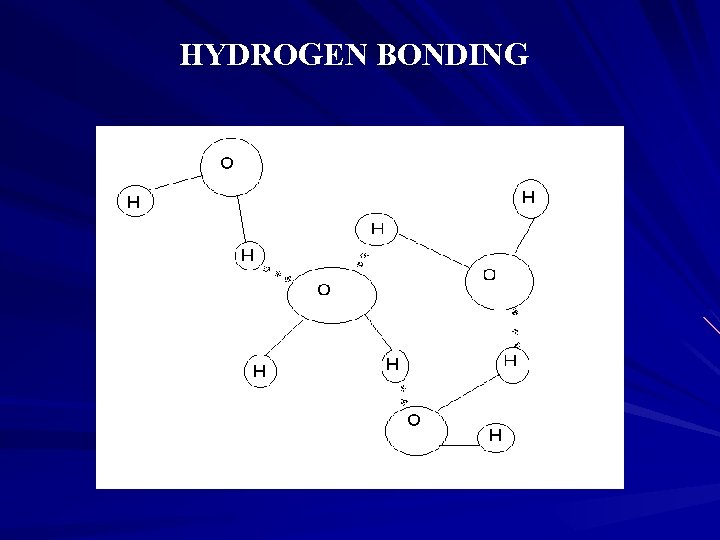
HYDROGEN BONDING

Dipole-Dipole Forces Dipole-Dipole forces are forces that result when two POLAR molecules come into contact with each other. Dipole-Dipole forces are similar to hydrogen bonding only much weaker. Dipole-Dipole forces do not involve bullies

Induced Dipole This is an instantaneous dipolethat is produced in a nonpolar molecule when the molecule’s electrons are momentarily attracted by a polar molecule. This attraction accounts for solubility of nonpolar compounds like oxygen, O 2 , in polar compounds like water

London Dispersion Forces are the result of two non-polar molecules interacting with each other. London Forces are the result of molecules bumping into each other. When they bump into each other, they become deformed. When they are deformed, they become polar. This is called an instantaneous dipole. If the molecules are going slow enough, the instantaneous dipoles created in the collision will cause the molecules to stick to each other.

London Dispersion Forces London dispersion forces are proportional to the surface area of the molecule, however we usually use molar mass as a proxy for surface area as it is more easily found and is generally adequate.
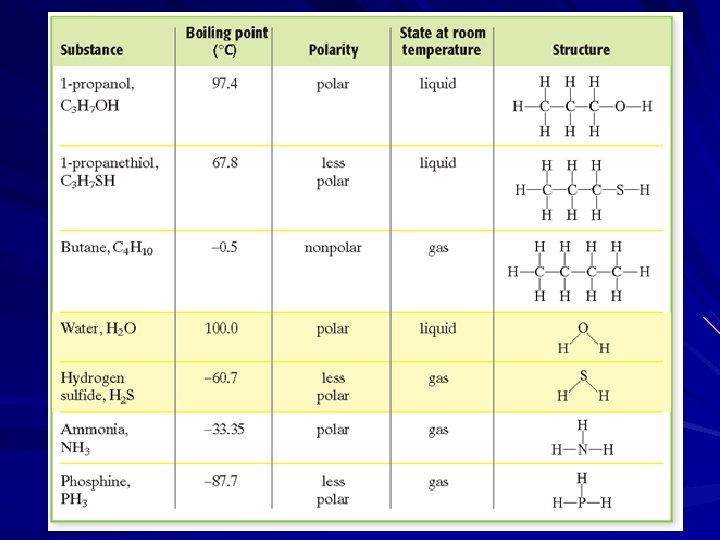

One thing to keep in mind is that we often talk about the strongest intermolecular force in a molecule. Any molecule that can exhibit hydrogen bonding also has dipole-dipole and London dispersion forces present. Any molecule that exhibits dipole-dipole forces also has London dispersion forces.

SUMMARY v Ionic Forces v Dipole-dipole Force we aren’t going to deal with them v Hydrogen Bonding H + bully (F, O, N) relatively strong two polar molecules that don’t have H bonding medium strength v London Dispersion Force 2 non-polar molecules weak

Summary In determining the type of forces present in a molecule, start with the strongest (Hbonding) and go to the weakest until you find the first one that fits. If a house is held together with chewing gum and nails, the nails will provide the majority of the strength Look for the strongest intermolecular force

Practice… H 2 O H-bonding H 2 S dipole-dipole HCl dipole-dipole PH 3 dipole-dipole CO 2 London CH 4 London Si. H 4 London

OK, so now what Let’s look at why intermolecular forces are important. Intermolecular forces affect many properties of chemicals. These properties include, boiling point, melting point, vapor pressure, and viscosity.

Properties Boiling Point where a liquid becomes a gas Melting Point where a solid becomes a liquid Vapor Pressure the pressure of a gas above the liquid of the same chemical Viscosity the resistance to flow

Boiling Point In order for a chemical to boil, the molecules must be physically given enough energy so that the molecules can get very far apart from each other (remember the Ideal Gas criteria) This takes lots of energy.

Boiling Point The stronger the intermolecular force, the more energy that it will take to pull the molecules apart. The stronger the force, the higher the boiling point for the compound. This is why water with a molecular weight of 18 g/mole has such a high boiling point compared to H 2 S which has a molecular weight of 34 g/mole.

Melting Point Remember Boiling Point trends. Melting point is the same idea. The molecules in a solid have three dimensional arrangements that must be broken before the material can melt. The stronger these 3 dimensional forces, the higher the melting point.

Viscosity is the resistance to flow. Honey is viscous--it does not flow. Soda is not viscous --it flows easily. The stronger the forces between the molecules, the more difficult it is for one molecule to sneak away from the other.

Viscosity, Trend The stronger the intermolecular force, the higher the viscosity and the slower the material will flow. The weaker the force, the lower the viscosity and the faster the material will flow.

But Wait Water has hydrogen bonding but it flows easily and honey flows slowly--why? Water forms a few attachments with each other. Not only is viscosity a function of the type of force, it is also a function of the number of physical places at which that force is exerted.

Vapor Pressure Vapor pressure is the pressure of a gas above a liquid of the same material. Vapor Pressure basically tells us how EASILY a material becomes a gas. The higher the vapor pressure, the easier it is to become a gas which means that the boiling point will probably be low.

Vapor Pressure Gasoline has a high vapor pressure. You can smell it very easily if you spill it on the ground. Steel has a very low vapor pressure. It has no noticeable odor.

Vapor Pressure, Trends The WEAKER the force, the higher the vapor pressure We are looking at how EASY it becomes a gas so this trend is backwards from the other three trends we have looked at in this section.

Solids We are not going to spend much time on the forces holding solids together. ATOMIC SOLIDS solids formed by atoms of the same element an example is solid neon or solid argon METALLIC SOLIDS made by metals, an example is Cu or Fe

Solids MOLECULAR SOLIDS solids formed by molecular compounds examples include dry ice COVALENT NETWORK SOLIDS This is a solid that has 3 dimensional bonding among non-metals. Examples include graphite and diamond

Solids IONIC SOLIDS these are solids formed by ionic compounds

Solids Ionic solids, covalent network solids and metallic solids are all strong forces and have high melting points, boiling points, and low vapor pressure. Molecular and atomic solids are weak forces and have low melting point, low boiling points and high vapor pressure.
447c2ab3bec86f49a52c280041dede3d.ppt